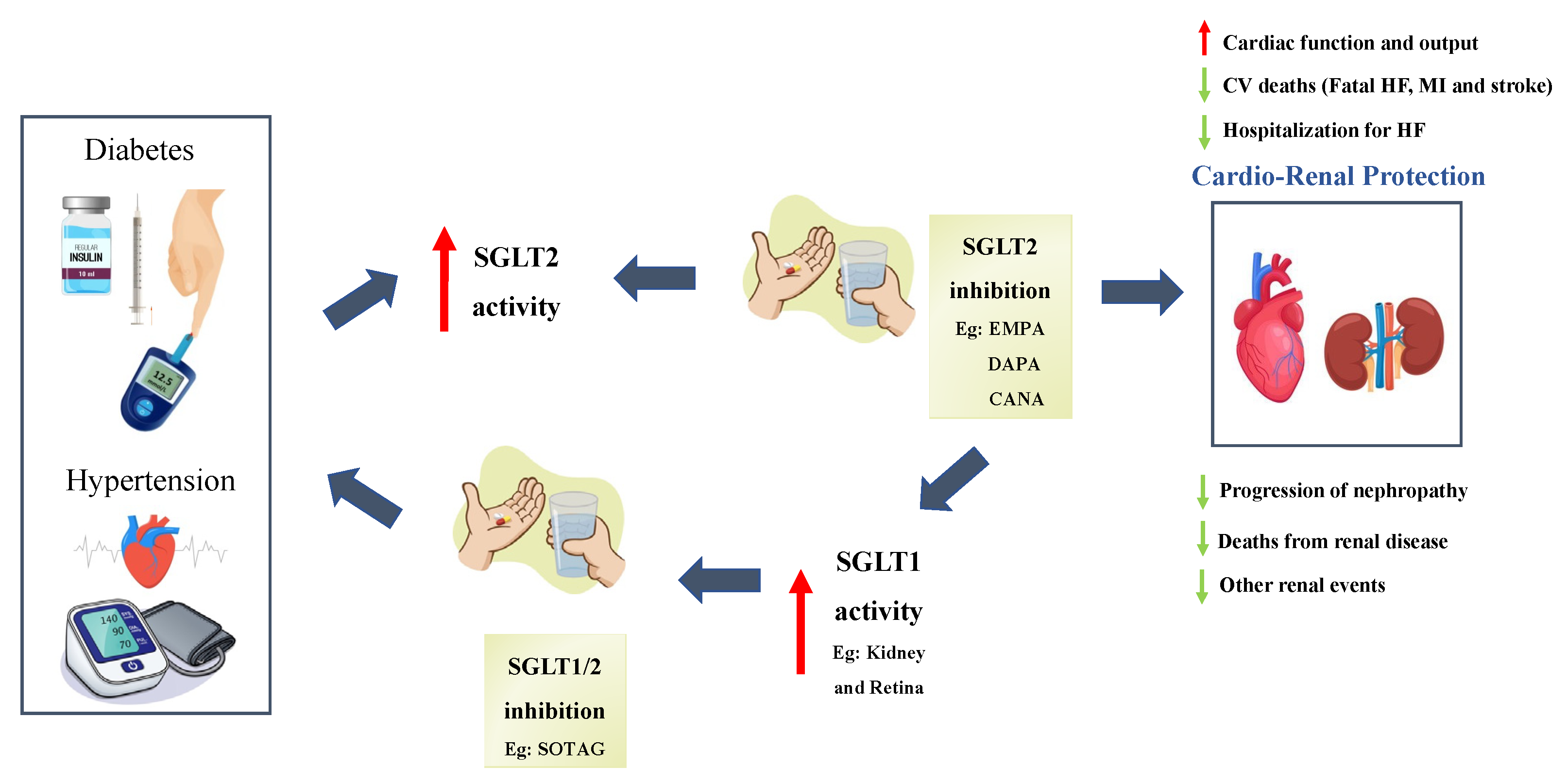
The Impact of SGLT2 Inhibitors in the Heart and Kidneys Regardless of Diabetes Status
Abstract
:1. Introduction
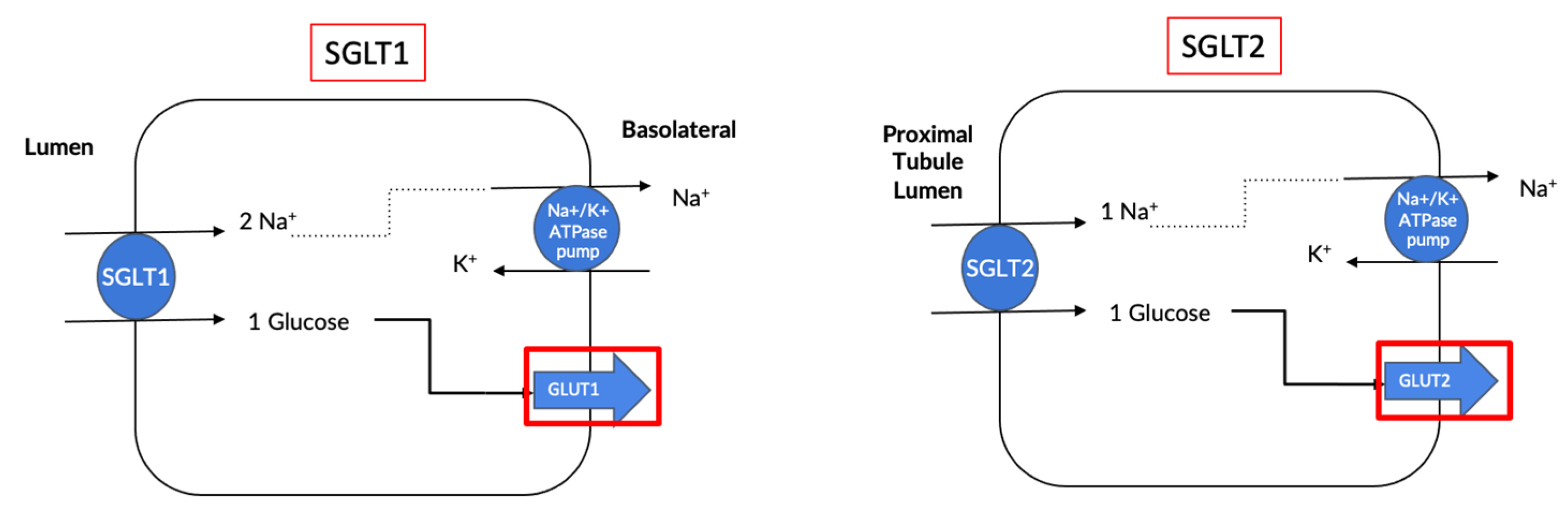
2. What Are Sodium Glucose Cotransporters?
| Location | SGLT1 | SGLT2 |
|---|---|---|
| Small Intestine | Apical membrane, K and L cells [19]. | Not expressed. |
| Eye | Retina [20]. | Retina, cornea and lens [20]. |
| Kidney | Section 3 of the proximal tubule. [21]. | Section 1 and 2 of the proximal tubules [22]. |
| Pancreas | Pancreatic alpha cells [18]. | Not expressed. |
| Liver | Biliary duct cells [19]. | Not expressed. |
| Heart | Capillaries [19]. | Not expressed. |
| Study Parameters | Key Findings |
|---|---|
| Pharmacokinetic properties | Longest plasma half-life: Canagliflozin. |
| Longest half-life in the kidney: Dapagliflozin. | |
| Highest distribution in the kidney: Ipragliflozin. | |
| Drug distribution in the kidney suggested to be dependent on chemical structure. | |
| Pharmacodynamic properties | All SGLT2i’s increased urinary glucose excretion in a dose-dependent manner. |
| Long-acting SGLT2i’s exhibited persistent action, even 18 h post dose. | |
| Close correlation between the duration of action, plasma drug concentration, drug distribution and kidney retention. | |
| Pharmacologic properties | Significant reductions in blood glucose and plasma insulin with all SGLT2i’s. |
| Significant improvement in glucose tolerance with all SGLT2i’s. | |
| Long-acting SGLT2i’s exert stronger anti-hyperglycaemic effects through persistent urine glucose excretion. | |
| Intermediate-acting SGLT2i’s may provide better glycaemic control when administered twice daily. | |
| Anti-diabetic effects | All SGLT2i’s significantly improved hyperglycaemia and hyperinsulinemia. |
| All SGLT2i’s significantly increased pancreatic insulin content by prevention of pancreatic exhaustion. | |
| Long-acting SGLT2i’s exert favourable glycaemic control over 24 h and may have slightly enhanced antidiabetic effects compared with intermediate-acting SGLT2i’s. | |
| Effects on diabetic complications | All SGLT2i’s exhibited significant improvements/trends in obesity parameters (e.g., body and visceral fat weights, lipid metabolism markers), proinflammatory cytokines and endothelial dysfunction markers. |
| All SGLT2i’s significantly decreased or showed a decreasing trend in steatohepatitis parameters (e.g., liver weight, plasma levels of liver enzymes) and renal parameters (e.g., creatinine clearance, renal tubular injury markers). | |
| Long-acting SGLT2i’s (0.3 mg/kg) demonstrated slight superiority in comparison with intermediate-acting SGLT2i’s (3 mg/kg) on several parameters (e.g., daily blood glucose control, visceral fat weight). |
3. Use of Selective SGLT2 Inhibition as an Antidiabetic Therapy
3.1. Preclinical Studies
3.1.1. Blood Pressure Reduction
3.1.2. Improved Digestive Health
3.1.3. Diabetic Retinopathy
3.1.4. Kidney Health
3.1.5. Cardiovascular Benefits
3.1.6. Improved Cognitive Function in T2D
3.2. Human Clinical Trials
3.2.1. Empagliflozin
3.2.2. Dapagliflozin
3.2.3. Canagliflozin
3.2.4. Ertugliflozin
4. Use of Selective SGLT2 Inhibition as a Nondiabetic Therapy
4.1. Preclinical Studies
4.1.1. Modulation of Sympathetic Nervous System Activity
4.1.2. Reductions in Blood Pressure
4.1.3. Inflammation Control
4.1.4. Increases in Ketone Levels
4.1.5. Improved Cardiovascular Health
4.1.6. Steatosis and Insulin Resistance
4.2. Human Clinical Trials
4.2.1. Empagliflozin
4.2.2. Dapagliflozin
4.2.3. Ipragliflozin
5. Discussion
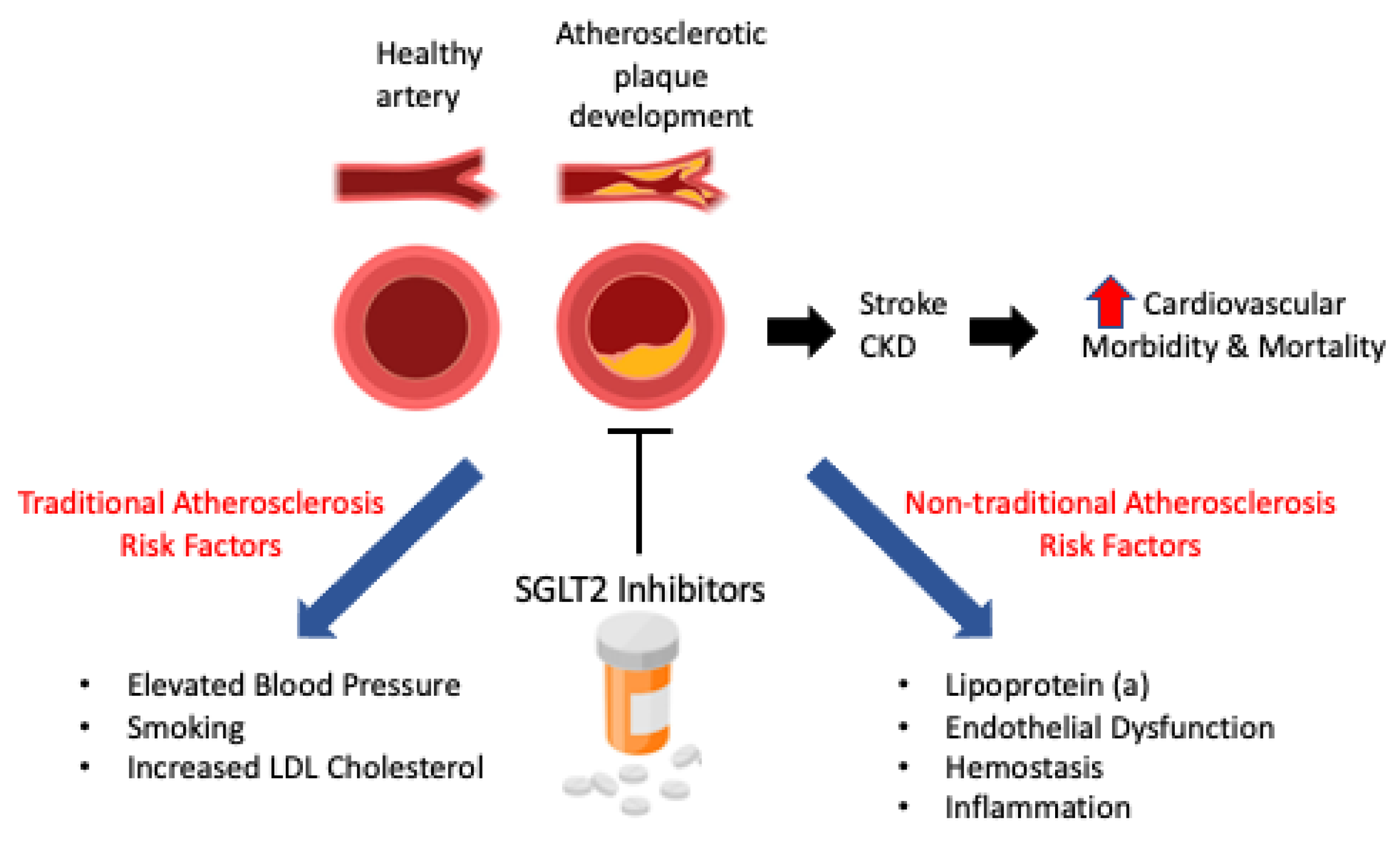
5.1. Are Dual SGLT1/2 Inhibitors More Effective Than Sole SGLT2 Inhibitors?
5.2. Interesting Avenues for SGLT2i Therapy for the Treatment of T1D
5.2.1. Animal Studies Utilizing SGLT2i’s as a Treatment for T1D
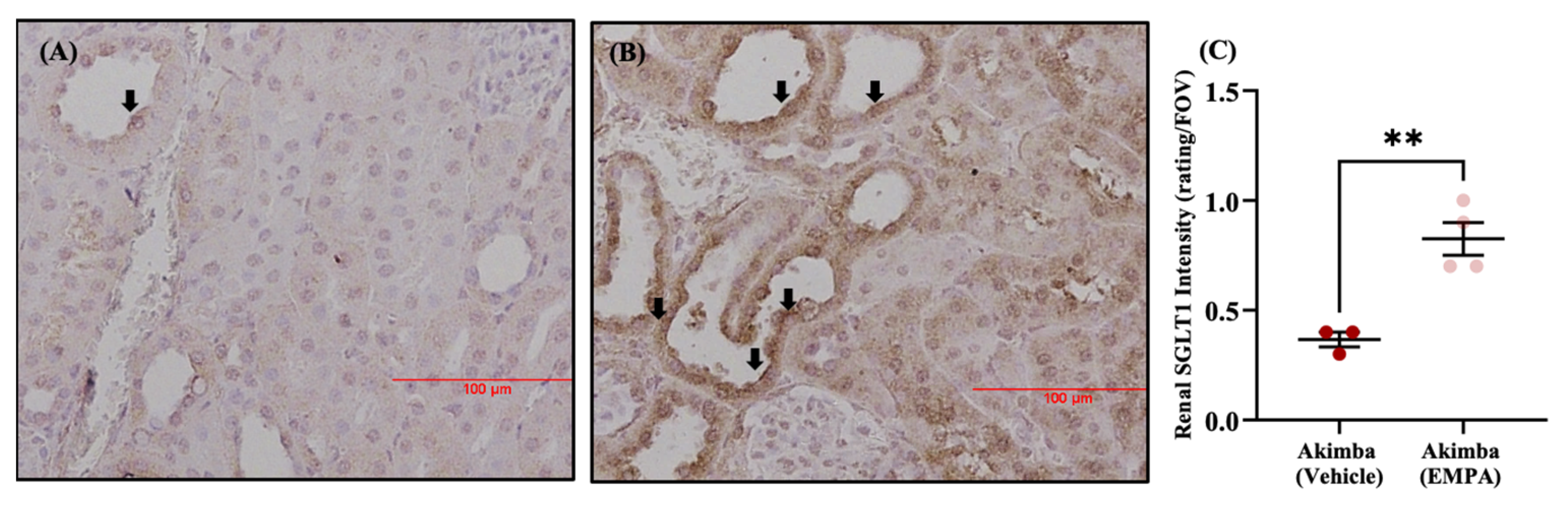
5.2.2. Human Pilot Studies/Clinical Trials Utilizing SGLT2i’s as an Add-On to Insulin for Patients with Type 1 Diabetes
5.3. Use of Dual SGLT 1/2 Inhibitors in T1D
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kovesdy, C. Epidemiology of chronic kidney disease: An update 2022. Kidney Int. Suppl. 2022, 12, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Keith, D.; Nichols, G.; Guillion, C.; Brown, J.; Smith, D. Longitudinal follow-up and outcomes among a population with chronic kidney disease in a large managed care organization. Arch. Intern. Med. 2004, 164, 659–663. [Google Scholar] [CrossRef] [PubMed]
- Aoki, J.; Nakajima, H.; Mori, M.; Sugimoto, T.; Hatori, M.; Tanimoto, S.; Amiya, E.; Hara, K. Clinical and pathologic characteristics of dilated cardiomyopathy in hemodialysis patients. Kidney Int. 2005, 67, 333–340. [Google Scholar] [CrossRef]
- Roth, G.; Mensah, G.; Johnson, C.; Addolorato, G.; Ammirati, E.; Baddour, L.; Barengo, N.; Beaton, A.; Benjamin, E.; Benziger, C.; et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef]
- Cook, C.; Cole, G.; Asaria, P.; Jabbour, R.; Francis, D. The annual global economic burden of heart failure. Int. J. Cardiol. 2014, 171, 368–376. [Google Scholar] [CrossRef] [PubMed]
- GBD 2021 Diabetes Collaborators. Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: A systematic analysis for the Global Burden of Disease Study 2021. Lancet 2023, 402, 203–234. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.; Foley, R.; Gilbertson, D.; Chen, S. United States Renal Data System public health surveillance of chronic kidney disease and end-stage renal disease. Kidney Int. Suppl. 2015, 5, 2–7. [Google Scholar] [CrossRef]
- Reutens, A. Epidemiology of diabetic kidney disease. Med. Clin. N. Am. 2013, 97, 1–18. [Google Scholar] [CrossRef]
- Hussain, S.; Chand Jamali, M.; Habib, A.; Hussain, M.; Akhtar, M.; Najmi, A. Diabetic kidney disease: An overview of prevalence, risk factors, and biomarkers. Clin. Epidemiol. Glob. Health 2021, 9, 2–6. [Google Scholar] [CrossRef]
- Pavkov, M.; Bennett, P.; Knowler, W.; Krakoff, J.; Sievers, M.; Nelson, R. Effect of youth-onset type 2 diabetes mellitus on incidence of end-stage renal disease and mortality in young and middle-aged Pima Indians. JAMA 2006, 296, 421–426. [Google Scholar] [CrossRef]
- Dasgupta, I. Cost of treating diabetic kidney disease. Indian J. Nephrol. 2014, 24, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Li, N.; Wu, Y.; Wang, M.; Yang, S.; Zheng, Y.; Deng, X.; Xiang, D.; Zhu, Y.; Xu, P.; et al. Global, Regional and National Burden of Diabetes-Related Chronic Kidney Disease from 1990 to 2019. Front. Endocrinol. 2021, 12, 672350. [Google Scholar] [CrossRef]
- Verma, D.; Firoz, A.; Garlapati, S.; Sathi, T.; Haris, M.; Dhungana, B.; Ray, B.; Shah, G.; Kc, B.; Paudel, P. Emerging treatments of Cardiorenal Syndrome: An update on Pathophysiology and Management. Cureus 2021, 13, e17240. [Google Scholar] [CrossRef] [PubMed]
- Sasso, F.; Pafundi, P.; Simeon, V.; De Nicola, L.; Chiodini, P.; Galiero, R.; Rinaldi, L.; Nevola, R.; Salvatore, T.; Sardu, C.; et al. Efficacy and durability of multifactorial intervention on mortality and MACEs: A randomized clinical trial in type-2 diabetic kidney disease. Cardiovasc. Diabetol. 2021, 20, 145. [Google Scholar] [CrossRef] [PubMed]
- Breyer, M.; Susztak, K. The next generation of therapeutics for chronic kidney disease. Nat. Rev. Drug Discov. 2016, 15, 568–588. [Google Scholar] [CrossRef]
- Sano, R.; Shinozaki, Y.; Ohta, T. Sodium-glucose cotransporters: Functional properties and pharmaceutical potential. J. Diabetes Investig. 2020, 11, 770–782. [Google Scholar] [CrossRef]
- Wright, E. The Intestinal Na+/Glucose Cotransporter. Annu. Rev. Physiol. 1993, 55, 575–589. [Google Scholar] [CrossRef] [PubMed]
- Suga, T.; Kikuchi, O.; Kobayashi, M.; Matsui, S.; Yokota-Hashimoto, H.; Wada, E.; Kohno, D.; Sasaki, T.; Takeuchi, K.; Kakizaki, S.; et al. SGLT1 in pancreatic a cells regulate glucagon secretion in mice, possibly explaining the distinct effects of SGLT2 inhibitors on plasma glucagon levels. Mol. Metab. 2019, 19, 1–12. [Google Scholar] [CrossRef]
- Vrhovac, I.; Eror, D.; Klessen, D.; Burger, C.; Breljak, D.; Kraus, O.; Radovic, N.; Jadrijevic, S.; Aleksic, I.; Walles, T.; et al. Localizations of Na+-d-glucose cotransporters SGLT1 and SGLT2 in human kidney and of SGLT1 in human small intestine, liver, lung and heart. Pflug. Arch. 2015, 467, 1881–1898. [Google Scholar] [CrossRef]
- Herat, L.Y.; Matthews, V.; Rakoczy, E.; Carnagarin, R.; Schlaich, M. Focusing on Sodium Glucose Co-transporter-2 (SGLT2) and its potential impact in diabetic retinopathy. Int. J. Endocrinol. 2018, 2018, 9254126. [Google Scholar] [CrossRef]
- Matthews, J.; Hibbs, M.; Herat, L.; Schlaich, M.; Matthews, V. The Sympathetic Nervous System Regulates Sodium Glucose Co-Transporter 1 Expression in the Kidney. Biomedicines 2023, 11, 819. [Google Scholar] [CrossRef] [PubMed]
- Herat, L.; Magno, A.; Rudnicka, C.; Hricova, J.; Carnagarin, R.; Ward, N.; Arcambal, A.; Kiuchi, M.; Head, G.; Schlaich, M.; et al. SGLT2 Inhibitor-Induced Sympathoinhibition: A Novel Mechanism for Cardiorenal Protection. JACC Basic Transl. Sci. 2020, 5, 169–179. [Google Scholar] [CrossRef]
- Bailey, C.; Day, C.; Bellary, S. Renal Protection with SGLT2 inhibitors: Effects in Acute and Chronic Kidney Disease. Curr. Diabetes Rep. 2022, 22, 39–52. [Google Scholar] [CrossRef]
- Chao, E.; Henry, R. SGLT2 Inhibition—A novel strategy for diabetes treatment. Nat. Rev. Drug Discov. 2010, 9, 551–559. [Google Scholar] [CrossRef]
- Zinman, B.; Wanner, C.; Lachin, J.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johanssen, E.; Woerle, H.; et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N. Engl. J. Med. 2015, 373, 2117–2128. [Google Scholar] [CrossRef]
- Wiviott, S.D.; Raz, I.; Bonaca, M.; Mosenzon, O.; Kato, E.; Cahn, A.; Silverman, M.; Zelniker, T.; Kuder, J.; Murphy, S.; et al. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2019, 380, 347–357. [Google Scholar] [CrossRef]
- Neal, B.; Perkovic, V.; Mahaffey, K.; Zeeuw, D.; Fulcher, G.; Erondu, N.; Shaw, W.; Law, G.; Desai, M.; Matthews, D. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N. Engl. J. Med. 2017, 377, 644–657. [Google Scholar] [CrossRef]
- Xiang, B.; Zhao, X.; Zhou, X. Cardiovascular benefits of sodium glucose cotransporter 2 inhibitors in diabetic and nondiabetic patients. Cardiovasc. Diabetol. 2021, 20, 78. [Google Scholar] [CrossRef]
- Dutka, M.; Bobinski, R.; Ulman-Wlodarz, I.; Hajduga, M.; Bujok, J.; Pajak, C.; Cwiertnia, M. Sodium glucose cotransporter 2 inhibitors: Mechanisms of action in heart failure. Heart Fail. Rev. 2021, 26, 603–622. [Google Scholar] [CrossRef]
- Elsayed, N.; Aleppo, G.; Aroda, V.; Bannuru, R.; Brown, F.; Bruemmer, D.; Collins, B.; Hilliard, M.; Isaacs, D.; Johnson, E.; et al. Chronic Kidney Disease and Risk Management: Standards of Care in Diabetes—2023. Diabetes Care 2023, 46 (Suppl. S1), S191–S202. [Google Scholar] [CrossRef]
- Tahara, A.; Takasu, T.; Yokono, M.; Imamura, M.; Kurosaki, E. Characterization and comparison of sodium-glucose cotransporter 2 inhibitors in pharmacokinetics, pharmacodynamics, and pharmacologic effects. J. Pharmacol. Sci. 2016, 130, 159–169. [Google Scholar] [CrossRef]
- Tahara, A.; Takasu, T.; Yokono, M.; Imamura, M.; Kurosaki, E. Characterization and comparison of sodium-glucose cotransporter 2 inhibitors: Part 2. Antidiabetic effects in type 2 diabetic mice. J. Pharmacol. Sci. 2016, 131, 198–208. [Google Scholar] [CrossRef]
- Tahara, A.; Takasu, T.; Yokono, M.; Imamura, M.; Kurosaki, E. Characterization and comparison of sodium-glucose cotransporter 2 inhibitors: Part 3. Effects on diabetic complications in type 2 diabetic mice. Eur. J. Pharmacol. 2017, 809, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Vallon, V.; Verma, S. Effects of SGLT2 Inhibitors on Kidney and Cardiovascular Function. Annu. Rev. Physiol. 2021, 83, 503–528. [Google Scholar] [CrossRef] [PubMed]
- Novikov, A.; Fu, Y.; Huang, W.; Freeman, B.; Patel, R.; van Ginkel, C.; Koepsell, H.; Busslinger, M.; Onishi, A.; Nespoux, J.; et al. SGLT2 inhibition and renal urate excretion: Role of luminal glucose, GLUT9, and URAT1. Am. J. Physiol. Ren. Physiol. 2019, 316, F173–F185. [Google Scholar] [CrossRef] [PubMed]
- Majewski, C.; Bakris, G. Blood Pressure Reduction: An Added Benefit of Sodium-Glucose Cotransporter 2 Inhibitors in Patients with Type 2 Diabetes. Diabetes Care 2015, 38, 429–430. [Google Scholar] [CrossRef] [PubMed]
- Tikkanen, I.; Narko, K.; Zeller, C.; Green, A.; Salsali, A.; Broedl, U.; Woerle, H. Empagliflozin reduces blood pressure in patients with type 2 diabetes and hypertension. Diabetes Care 2015, 38, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Sjostrom, C.; Johansson, P.; Ptaszynska, A.; List, J.; Johnsson, E. Dapagliflozin lowers blood pressure in hypertensive and non-hypertensive patients with type 2 diabetes. Diabetes Vasc. Dis. Res. 2015, 12, 352–358. [Google Scholar] [CrossRef]
- Matthews, V.; Elliot, R.; Rudnicka, C.; Hricova, J.; Herat, L.; Schlaich, M. Role of the sympathetic nervous system in regulation of the sodium glucose cotransporter 2. J. Hypertens. 2017, 35, 2059–2068. [Google Scholar] [CrossRef]
- Wu, J.; Chen, Y.; Yang, H.; Gu, L.; Ni, Z.; Mou, S.; Shen, J.; Che, X. Sodium glucose co-transporter 2 (SGLT2) inhibition via dapagliflozin improves diabetic kidney disease (DKD) over time associated with increasing effect on the gut microbiota in db/db mice. Front. Endocrinol. 2023, 14, 1026040. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Battson, M.; Jarrell, D.; Hou, S.; Ecton, K.; Weir, T.; Gentile, C. SGLT2 inhibition via dapagliflozin improves generalized vascular dysfunction and alters the gut microbiota in type 2 diabetic mice. Cardiovasc. Diabetol. 2018, 17, 62. [Google Scholar] [CrossRef] [PubMed]
- Herat, L.; Ward, N.; Magno, A.; Rakoczy, E.; Kiuchi, M.; Schlaich, M.; Matthews, V. Sodium glucose co-transporter 2 inhibition reduces succinate levels in diabetic mice. World J. Gastroenterol. 2020, 26, 3225–3235. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Yang, Y.; Xu, G. Empagliflozin ameliorates type 2 diabetes mellitus-related diabetic nephrophathy via altering the gut microbiota. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2022, 1867, 159234. [Google Scholar] [CrossRef]
- Hata, S.; Okamura, T.; Kobayashi, A.; Bamba, R.; Miyoshi, T.; Nakajima, H.; Kitagawa, N.; Hashimoto, Y.; Majima, S.; Senmaru, T.; et al. Gut Microbiota Changes by an SGLT2 Inhibitor, Luseogliflozin, Alters Metabolites Compared with Those in a Low Carbohydrate Diet in db/db Mice. Nutrients 2022, 14, 3531. [Google Scholar] [CrossRef]
- Gong, Q.; Zhang, R.; Wei, F.; Fang, J.; Zhang, J.; Sun, J.; Sun, Q.; Wang, H. SGLT2 inhibitor-empagliflozin treatment ameliorates diabetic retinopathy manifestations and exerts protective effects associated with augmenting branched chain amino acids catabolism and transportation in db/db mice. Biomed. Pharmacother. 2022, 152, 113222. [Google Scholar] [CrossRef] [PubMed]
- Luo, Q.; Leley, S.; Bello, E.; Dhami, H.; Mathew, D.; Bhatwadekar, A. Dapagliflozin protects neural and vascular dysfunciton of the retinas in diabetes. BMJ Open Diabetes Res. Care 2022, 10, e002801. [Google Scholar] [CrossRef]
- Tang, L.; Wu, Y.; Tian, M.; Sjostrom, C.; Johansson, U.; Peng, X.; Smith, D.; Huang, Y. Dapagliflozin slows the progression of the renal and liver fibrosis associated with type 2 diabetes. Am. J. Physiol. Endocrinol. Metab. 2017, 313, E563–E576. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.; Chung, S.; Kim, S.; Lee, E.; Yoo, Y.; Kim, J.; Ahn, Y.; Kim, E.; Moon, S.; Kim, M.; et al. Effect of Sodium-Glucose Co-Transporter 2 Inhibitor, Dapagliflozin, on Renal Renin-Angiotensin System in an Animal Model of Type 2 Diabetes. PLoS ONE 2016, 11, e0165703. [Google Scholar] [CrossRef] [PubMed]
- Kojima, N.; Williams, J.; Takahashi, T.; Miyata, N.; Roman, R. Effects of a new SGLT2 inhibitor, Luseogliflozin, on diabetic nephropathy in T2D rats. J. Pharmacol. Exp. Ther. 2013, 345, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.; Koibuchi, N.; Hasegawa, Y.; Sueta, D.; Toyama, K.; Uekawa, K.; Ma, M.; Nakagawa, T.; Husaka, H.; Kim-Mitsuyama, S. Glycemic control with empagliflozin, a novel selective SGLT2 inhibitor, ameliorates cardiovascular injury and cognitive dysfunction in obese and type 2 diabetic mice. Cardiovasc. Diabetol. 2014, 13, 148. [Google Scholar] [CrossRef] [PubMed]
- Wanner, C.; Inzucchi, S.; Lachin, J.; Fitchett, D.; Von Eynatten, M.; Mattheus, M.; Johansen, E.; Woerle, H.; Broedl, U.; Zinman, B. Empagliflozin and Progression of Kidney Disease in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Perkovic, V.; Jardine, M.; Neal, B.; Bompoint, S.; Heerspink, H.; Charytan, D.; Edwards, R.; Agarwal, R.; Bakris, G.; Bull, S.; et al. Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy. N. Engl. J. Med. 2019, 380, 2295–2306. [Google Scholar] [CrossRef] [PubMed]
- Cannon, C.; Pratley, R.; Dagogo-Jack, S.; Mancuso, J.; Huyck, S.; Masiukiewicz, U.; Charbonnel, B.; Frederich, R.; Gallo, S.; Cosentino, F.; et al. Cardiovascular Outcomes with Ertugliflozin in Type 2 Diabetes. N. Engl. J. Med. 2020, 383, 1425–1435. [Google Scholar] [CrossRef]
- McMurray, J.; Solomon, S.; Inzucchi, S.; Kober, L.; Kosiborod, M.; Martinez, F.; Ponikowski, P.; Sabatine, M.; Anand, I.; Belohlavek, J.; et al. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction. N. Engl. J. Med. 2019, 381, 1995–2008. [Google Scholar] [CrossRef]
- Cherney, D.; Dekkers, C.; Barbour, S.; Cattran, D.; Abdul Gafor, A.H.; Greasley, P.; Laverman, G.; Kun Lim, S.; Di Tanna, G.; Reich, H.; et al. Effects of the SGLT2 inhibitor dapagliflozin on proteinuria in non-diabetic patients with chronic kidney disease (DIAMOND): A randomized, double-blind, crossover trial. Lancet Diabetes Endocrinol. 2020, 8, 582–593. [Google Scholar] [CrossRef] [PubMed]
- Packer, M.; Anker, S.; Butler, J.; Filippatos, G.; Pocock, S.; Carson, P.; Januzzi, J.; Subodh, V.; Tsutsui, H.; Brueckmann, M.; et al. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N. Engl. J. Med. 2020, 383, 1413–1424. [Google Scholar] [CrossRef]
- Heerspink, H.; Stefansson, B.; Correa-Rotter, R.; Chertow, G.; Greene, T.; Hou, F.; Mann, J.; McMurray, J.; Lindberg, M.; Rossing, P.; et al. Dapagliflozin in Patients with Chronic Kidney Disease. N. Engl. J. Med. 2020, 383, 1436–1446. [Google Scholar] [CrossRef] [PubMed]
- Packer, M.; Butler, J.; Zannad, F.; Filippatos, G.; Ferreira, J.; Pocock, S.; Carson, P.; Anand, I.; Doehner, W.; Haass, M.; et al. Effect of Empagliflozin on Worsening Heart Failure Events in Patients With Heart Failure and Preserved Ejection Fraction: EMPEROR-Preserved Trial. Circulation 2021, 144, 1284–1294. [Google Scholar] [CrossRef] [PubMed]
- Lewinski, D.; Kolesnik, E.; Tripolt, N.; Pferschy, P.; Benedikt, M.; Wallner, M.; Alber, H.; Berger, R.; Lichtnauer, M.; Saely, C.; et al. Empagliflozin in acute myocardial infarction: The EMMY trial. Eur. Heart J. 2022, 43, 4421–4432. [Google Scholar] [CrossRef] [PubMed]
- Herrington, W.; Staplin, N.; Wanner, C.; Green, J.; Hauske, S.; Emberson, J.; Preiss, D.; Judge, P.; Mayne, K.; Ng, S.; et al. Empagliflozin in Patients with Chronic Kidney Disease. N. Engl. J. Med. 2023, 388, 117–127. [Google Scholar] [CrossRef]
- Solomon, S.; McMurray, J.; Claggett, B.; Boer, R.; DeMets, D.; Hernandez, A.; Inzucchi, S.; Kosiborod, M.; Lam, C.; Martinez, F.; et al. Dapagliflozin in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N. Engl. J. Med. 2022, 387, 1089–1098. [Google Scholar] [CrossRef] [PubMed]
- Herat, L.; Matthews, J.; Azzam, O.; Schlaich, M.; Matthews, V. Targeting features of the metabolic syndrome through sympatholytic effects of SGLT inhibition. Curr. Hypertens. Rep. 2022, 24, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wu, M.; Xu, B.; Kang, L. Empagliflozin Alleviates Atherosclerosis Progression by Inhibiting Inflammation and Sympathetic Activity in a Normoglycemic Mouse Model. J. Inflamm. Res. 2021, 14, 2277–2287. [Google Scholar] [CrossRef] [PubMed]
- Matthews, J.; Herat, L.; Magno, A.; Gorman, S.; Schlaich, M.; Matthews, V. SGLT-2 Inhibitor-Induced Sympathoexcitation in White Adipose Tissue: A Novel Mechanism of Beiging. Biomedicines 2020, 8, 514. [Google Scholar] [CrossRef]
- Machado, S.; Pasquarelli-do-Nascimento, G.; Santos da Silva, D.; Farias, G.; de Oliveira Santos, I.; Baptista, L.; Magalhaes, K. Browning of the white adipose tissue regulation: New insights into nutritional and metabolic relevance in health and diseases. Nutr. Metab. 2022, 19, 61. [Google Scholar] [CrossRef] [PubMed]
- Leng, W.; Ouyang, X.; Lei, X.; Wu, M.; Chen, L.; Wu, Q.; Deng, W.; Liang, Z. The SGLT-2 Inhibitor Dapagliflozin Has a Therapeutic Effect on Atherosclerosis in Diabetic ApoE−/− Mice. Mediat. Inflamm. 2016, 2016, 6305735. [Google Scholar] [CrossRef] [PubMed]
- Papazafiropoulou, A.; Georgopoulos, M.; Katsilambros, N. Ketone bodies and the heart. Arch. Med. Sci. Atheroscler. Dis. 2021, 6, e209–e214. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Hu, X.; Xu, C.; Lu, C.; Cao, R.; Xie, Y.; Yang, J. Ketogenic diet alleviates renal fibrosis in mice by enhancing fatty acid oxidation through the free fatty acid receptor 3 pathway. Front. Nutr. 2023, 10, 1127845. [Google Scholar] [CrossRef] [PubMed]
- Pietschner, R.; Kolwelter, J.; Bosch, A.; Striepe, K.; Jung, S.; Kannenkeril, D.; Ott, C.; Schiffer, M.; Achenbach, S.; Schmieder, R. Effect of empagliflozin on ketone bodies in patients with chronic heart failure. Cardiovasc. Diabetol. 2021, 20, 219. [Google Scholar] [CrossRef]
- Park, S.; Farooq, M.; Gaertner, S.; Bruckert, C.; Qureshi, A.; Lee, H.; Benrahla, D.; Pollet, B.; Stephan, D.; Ohlmann, P.; et al. Empaglifozin improved systolic blood pressure, endothelial dysfunction and heart remodeling in the metabolic syndrome ZSF1 rat. Cardiovasc. Diabetol. 2020, 19, 19. [Google Scholar] [CrossRef]
- Tomita, I.; Kume, S.; Sugahara, S.; Osawa, N.; Yamahara, K.; Yasuda-Yamahara, M.; Takeda, N.; Chin-Kanasaki, M.; Kaneko, T.; Mayoux, E.; et al. SGLT2 inhibition mediates protection from diabetic kidney disease by promoting ketone body-induced mTORC1 inhibition. Cell Metab. 2020, 32, 404–419.e6. [Google Scholar] [CrossRef] [PubMed]
- Khunti, K.; Ruan, Y.; Davies, J.; Field, B.; Harris, S.; Kosiborod, M.; Nagi, D.; Narendran, P.; Patel, D.; Ryder, R.; et al. Association between SGLT2 Inhibitor Treatment and Diabetic Ketoacidosis and Mortality in People with Type 2 Diabetes Admitted to Hospital with COVID-19. Diabetes Care 2022, 45, 2838–2843. [Google Scholar] [CrossRef] [PubMed]
- Kimura, I.; Inoue, D.; Maeda, T.; Hara, T.; Ichimura, A.; Miyauchi, S.; Kobayashi, M.; Hirasawa, A.; Tsujimoto, G. Short-chain fatty acids and ketones directly regulate sympathetic nervous system via G protein-coupled receptor 41 (GPR41). Proc. Natl. Acad. Sci. USA 2011, 108, 8030–8035. [Google Scholar] [CrossRef] [PubMed]
- Kolanowski, J.; Young, J.; Landsberg, L. Stimulatory influence of D(-)3-hydroxybutyrate feeding on sympathetic nervous system activity in the rat. Metabolism 1994, 43, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Yang, N.; Luo, W.; Qian, J.; Zhu, W.; Ye, S.; Yuan, C.; Xu, D.; Liang, G.; Huang, W.; et al. Direct cardio-protection of Dapagliflozin against obesity-related cardiomyopathy via NHE1/MAPK signaling. Acta Pharmacol. Sin. 2022, 43, 2624–2635. [Google Scholar] [CrossRef]
- Xu, L.; Nagata, N.; Nagashimada, M.; Zhuge, F.; Ni, Y.; Chen, G.; Mayoux, E.; Kaneko, S.; Ota, T. SGLT2 Inhibition by Empagliflozin Promotes Fat Utilization and Browning and Attenuates Inflammation and Insulin Resistance by Polarizing M2 Macrophages in Diet-induced Obese Mice. EbioMedicine 2017, 20, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Radlinger, B.; Ress, C.; Folie, S.; Salzmann, K.; Lechuga, A.; Weiss, B.; Salvenmoser, W.; Graber, M.; Hirsch, J.; Holfeld, J.; et al. Empagliflozin protects mice against diet-induced obesity, insulin resistance and hepatic steatosis. Diabetologia 2023, 66, 754–767. [Google Scholar] [CrossRef]
- Ji, W.; Zhao, M.; Wang, M.; Yan, W.; Liu, Y.; Ren, S.; Lu, J.; Wang, B.; Chen, L. Effects of canagliflozin on weight loss in high-fat-diet-induced obese mice. PLoS ONE 2017, 12, e0179960. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.; Cherney, D. Clinical Implications of an Acute Dip in eGFR after SGLT2 Inhibitor Initiation. Clin. J. Am. Soc. Nephrol. 2021, 16, 1278–1280. [Google Scholar] [CrossRef] [PubMed]
- Kiuchi, S.; Hisatake, S.; Kabuki, T.; Fujii, T.; Oka, T.; Dobashi, S.; Hashimoto, H.; Ikeda, T. Long-term use of ipragliflozin improved cardiac sympathetic nerve activity in a patient with heart failure: A case report. Drug Discov. Ther. 2018, 12, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Young, S.; Ryan, L.; Mullins, T.; Flint, M.; Steane, S.; Walton, S.; Bielefeldt-Ohmann, H.; Carter, D.; Reichelt, M.; Gallo, L. Sotagliflozin, a dual SGLT1/2 Inhibitor, improves cardiac outcomes in a mouse model of early heart failure without diabetes. Front. Physiol. 2021, 12, 738594. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, D.; Szarek, M.; Steg, P.; Cannon, C.; Leiter, L.; McGuire, D.; Lewis, J.; Riddle, M.; Voors, A.; Metra, M.; et al. Sotagliflozin in Patients with Diabetes and Recent Worsening Heart Failure. N. Engl. J. Med. 2021, 384, 117–128. [Google Scholar] [CrossRef]
- Bhatt, D.; Szarek, M.; Pitt, B.; Cannon, C.; Leiter, L.; McGuire, D.; Lewis, J.; Riddle, M.; Inzucchi, S.; Kosiborod, M.; et al. Sotagliflozin in Patients with Diabetes and Chronic Kidney Disease. N. Engl. J. Med. 2021, 384, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Perez, M.; Rodriguez-Capitan, J.; Requena-Ibanez, J.; Santos-Gallego, C.; Zafar, M.; Escolar, G.; Mancini, D.; Mitter, S.; Lam, D.; Contreras, J.; et al. Rationale and Design of the SOTA-P-CARDIA Trial (ATRU-V): Sotagliflozin in HFpEF Patients Without Diabetes. Cardiovasc. Drugs Ther. online ahead of print. 2023. [Google Scholar] [CrossRef] [PubMed]
- Herat, L.; Matthews, J.; Ong, W.; Rakoczy, E.; Schlaich, M.; Matthews, V. Determining the Role of SGLT2 Inhibition with Dapagliflozin in the Development of Diabetic Retinopathy. Front. Biosci. 2022, 27, 320. [Google Scholar] [CrossRef] [PubMed]
- Herat, L.; Matthews, J.; Rakoczy, E.; Schlaich, M.; Matthews, V. Comparing and Contrasting the Effects of the SGLT Inhibitors Canagliflozin and Empagliflozin on the Progression of Retinopathy. Front. Biosci. 2023, 28, 83. [Google Scholar] [CrossRef] [PubMed]
- Matthews, J.; Herat, L.; Rooney, J.; Rakoczy, E.; Schlaich, M.; Matthews, V. Determining the Role of SGLT2 inhibition with Empagliflozin in the development of diabetic retinopathy. Biosci. Rep. 2022, 42, BSR20212209. [Google Scholar] [CrossRef] [PubMed]
- Matthews, J.; Schlaich, M.; Rakoczy, E.; Matthews, V.; Herat, L. The Effect of SGLT2 Inhibition on Diabetic Kidney Disease in a Model of Diabetic Retinopathy. Biomedicines 2022, 10, 522. [Google Scholar] [CrossRef] [PubMed]
- Kesherwani, V.; Shahshahan, H.; Mishra, P. Cardiac transcriptome profiling of diabetic Akita mice using microarray and next generation sequencing. PLoS ONE 2017, 12, e0182828. [Google Scholar] [CrossRef] [PubMed]
- Herat, L.; Matthews, J.; Hibbs, M.; Rakoczy, E.; Schlaich, M.; Matthews, V. SGLT1/2 inhibition improves glycemic control and multi-organ protection in type 1 diabetes. iScience 2023, 26, 107260. [Google Scholar] [CrossRef]
- Henry, R.; Thakkar, P.; Tong, C.; Polidori, D.; Alba, M. Efficacy and Safety of Canagliflozin, a Sodium-Glucose Cotransporter 2 Inhibitor, as Add-on to Insulin in Patients with Type 1 Diabetes. Diabetes Care 2015, 38, 2258–2265. [Google Scholar] [CrossRef] [PubMed]
- Dandona, P.; Mathieu, C.; Phillip, M.; Hansen, L.; Griffen, S.; Tschope, D.; Thoren, F.; Xu, J.; Langkilde, A. Efficacy and safety of dapagliflozin in patients with inadequately controlled type 1 diabetes (DEPICT-1): 24 week results from a multicentre, double-blind, phase 3, randomised controlled trial. Lancet 2017, 5, 864–876. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, C.; Rudofsky, G.; Phillip, M.; Araki, E.; Lind, M.; Arya, N.; Thoren, F.; Scheerer, M.; Iqbal, N.; Dandona, P. Long-term efficacy and safety of dapagliflozin in patients with inadequately controlled type 1 diabetes (the DEPICT-2 study): 52-week results from a randomized controlled trial. Diabetes Obes. Metab. 2020, 22, 1516–1526. [Google Scholar] [CrossRef] [PubMed]
- Rosenstock, J.; Marquard, J.; Laffel, L.; Neubacher, D.; Kaspers, S.; Cherney, D.; Zinman, B.; Skyler, J.; George, J.; Soleymanlou, N.; et al. Empagliflozin as Adjunctive to Insulin Therapy in Type 1 Diabetes: The EASE Trials. Diabetes Care 2018, 41, 2560–2569. [Google Scholar] [CrossRef] [PubMed]
- Paik, J.; Blair, H. Dapagliflozin: A Review in Type 1 Diabetes. Drugs 2019, 79, 1877–1884. [Google Scholar] [CrossRef] [PubMed]
- Zinman, B.; Lachin, J.; Inzucchi, S. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N. Engl. J. Med. 2016, 374, 1094. [Google Scholar] [CrossRef] [PubMed]
- Cefalo, C.; Cinti, F.; Moffa, S.; Impronta, F.; Sorice, G.; Mezza, T.; Pontecorvi, A.; Giaccari, A. Sotagliflozin, the first dual SGLT inhibitor: Current outlook and perspectives. Cardiovasc. Diabetol. 2019, 18, 20. [Google Scholar] [CrossRef] [PubMed]

| Clinical Trials in Patients with Diabetes Only | |||||
| Trial/Year | SGLT2 Inhibitor | Patient Cohort | Patient # | Outcome | Ref # |
| 2015 EMPA-REG | Empagliflozin | T2D—High Risk of Cardiovascular Events | 7064 | ↓ Mortality & Hospitalisation due to HF ↓ Risk of Clinically Relevant Renal Events | [25,51] |
| 2017 CANVAS | Canagliflozin | T2D—High Risk of CVD | 10,143 | ↓ Major Adverse Cardiovascular Events ↓ Albuminuria Levels ↓ Renal Replacement Therapy/Death | [27] |
| 2018 DELCARE-TIMI | Dapagliflozin | T2D—High Risk of Atherosclerotic CVD | 17,190 | ↓ Cardiovascular Death or Hospitalisation for HF. | [26] |
| 2018 CREDENCE | Canagliflozin | T2D and Kidney Disease | 4401 | ↓ Risk of Kidney Failure/Cardiovascular Events | [52] |
| 2019 VERTIS | Ertugliflozin | T2D and CVD | 8246 | ↓ First/Total Hospitalization ↓ Risk of death from HF/CVD | [53] |
| Clinical Trials in Patients Irrespective of Diabetic Status | |||||
| Trial/Year | SGLT2 Inhibitor | Patient Cohort | Patient # | Outcome | Ref # |
| 2019 DAPA-HF | Dapagliflozin | Class II, III or IV Heart Failure & EF < 40% | 4744 | ↓ Worsening Heart Failure/Death from CV Events | [54] |
| 2019 DIAMOND | Dapagliflozin | Non Diabetic Patients with CKD | 50 | Induced an acute and reversible decline in mGFR levels. | [55] |
| 2020 EMPEROR-REDUCED | Empagliflozin | Class II, III or IV Heart Failure & EF < 40% | 3730 | ↓ Mortality & Hospitalisation due to HF | [56] |
| 2020 DAPA-CKD | Dapagliflozin | High Risk of Kidney & CDV Outcomes | 4304 | ↓ Risk of a declining GFR level ↓ End Stage Renal Disease/Death from Renal Causes | [57] |
| 2021 EMPEROR-PRESERVED | Empagliflozin | Class II, III or IV Heart Failure & EF > 40% | 5988 | ↓ Cardiovascular Death and Hospitalisation for HF. | [58] |
| 2022 EMMY | Empagliflozin | Recent acute myocardial infarction | 476 | ↓ Risk of a declining GFR level ↓ ES Renal Disease/Death from Renal Causes | [59] |
| 2022 EMPA-KIDNEY | Empagliflozin | Patients with CKD | 6609 | ↓ Progression of Kidney Disease ↓ Death from Cardiovascular Causes | [60] |
| 2022 DELIVER | Dapagliflozin | Heart Failure, Left Ventricular EF > 40% | 6263 | ↓ Worsening Heart Failure/Death from Cardiovascular Events | [61] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matthews, J.; Herat, L.; Schlaich, M.P.; Matthews, V. The Impact of SGLT2 Inhibitors in the Heart and Kidneys Regardless of Diabetes Status. Int. J. Mol. Sci. 2023, 24, 14243. https://doi.org/10.3390/ijms241814243
Matthews J, Herat L, Schlaich MP, Matthews V. The Impact of SGLT2 Inhibitors in the Heart and Kidneys Regardless of Diabetes Status. International Journal of Molecular Sciences. 2023; 24(18):14243. https://doi.org/10.3390/ijms241814243
Chicago/Turabian StyleMatthews, Jennifer, Lakshini Herat, Markus P. Schlaich, and Vance Matthews. 2023. "The Impact of SGLT2 Inhibitors in the Heart and Kidneys Regardless of Diabetes Status" International Journal of Molecular Sciences 24, no. 18: 14243. https://doi.org/10.3390/ijms241814243
APA StyleMatthews, J., Herat, L., Schlaich, M. P., & Matthews, V. (2023). The Impact of SGLT2 Inhibitors in the Heart and Kidneys Regardless of Diabetes Status. International Journal of Molecular Sciences, 24(18), 14243. https://doi.org/10.3390/ijms241814243






