Tauroursodeoxycholic Acid Supplementation in In Vitro Culture of Indicine Bovine Embryos: Molecular and Cellular Effects on the In Vitro Cryotolerance
Abstract
:1. Introduction
2. Results
2.1. The Effects of TUDCA during IVC of Bovine Embryos
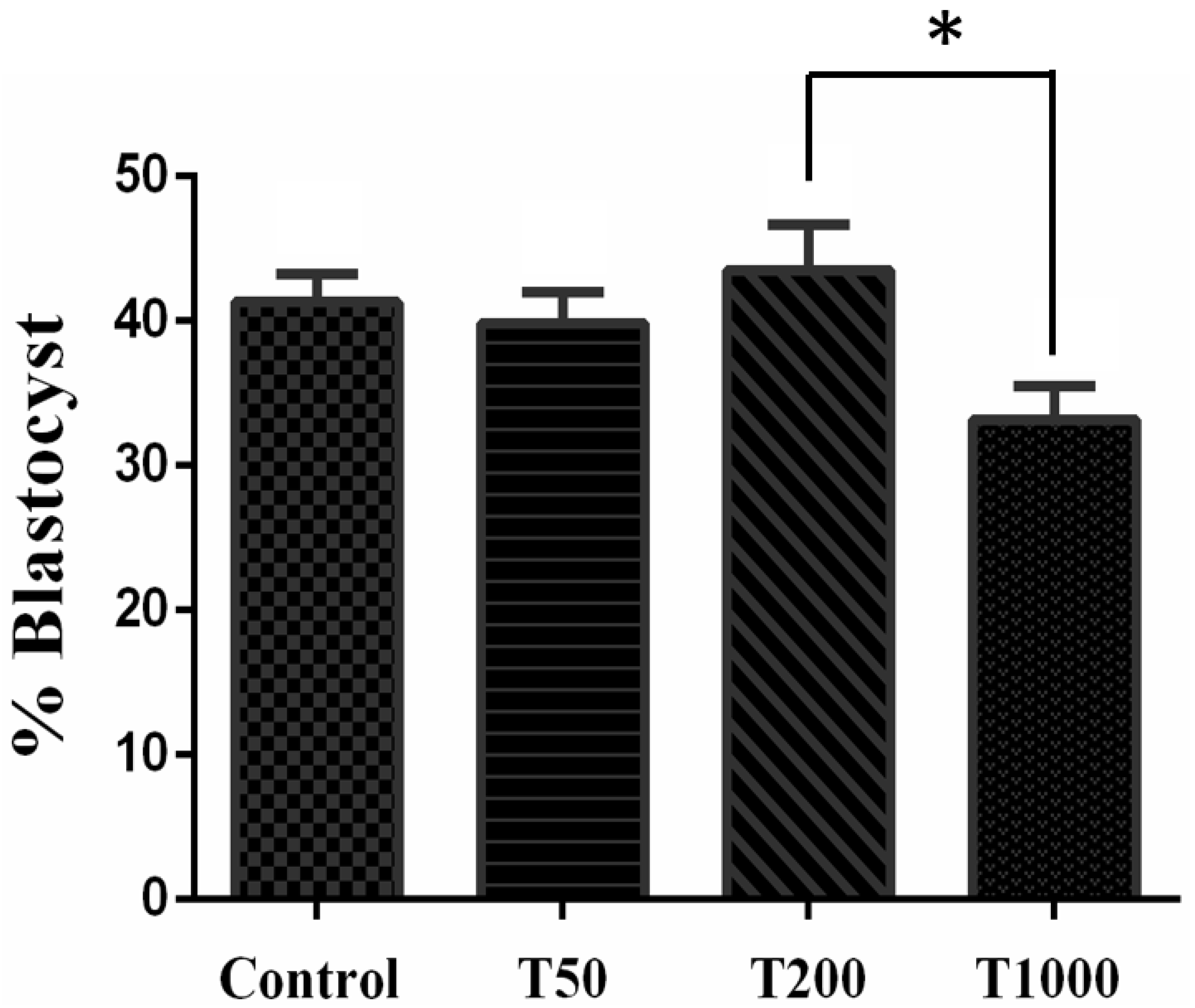
2.1.1. Developmental Competence
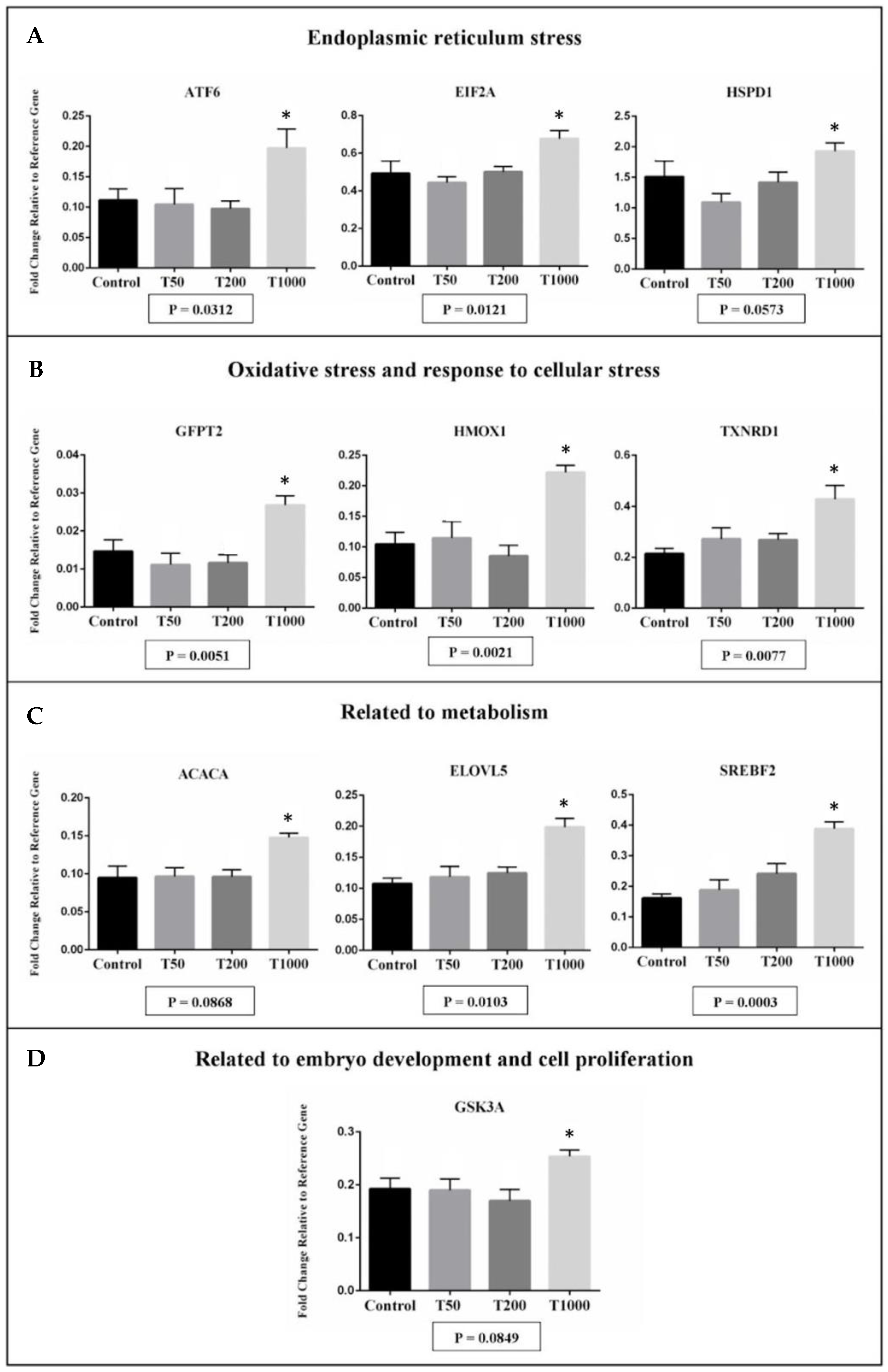
2.1.2. Gene Expression
2.2. The Effects of TUDCA during IVC on Post-Warmed Vitrified Blastocysts
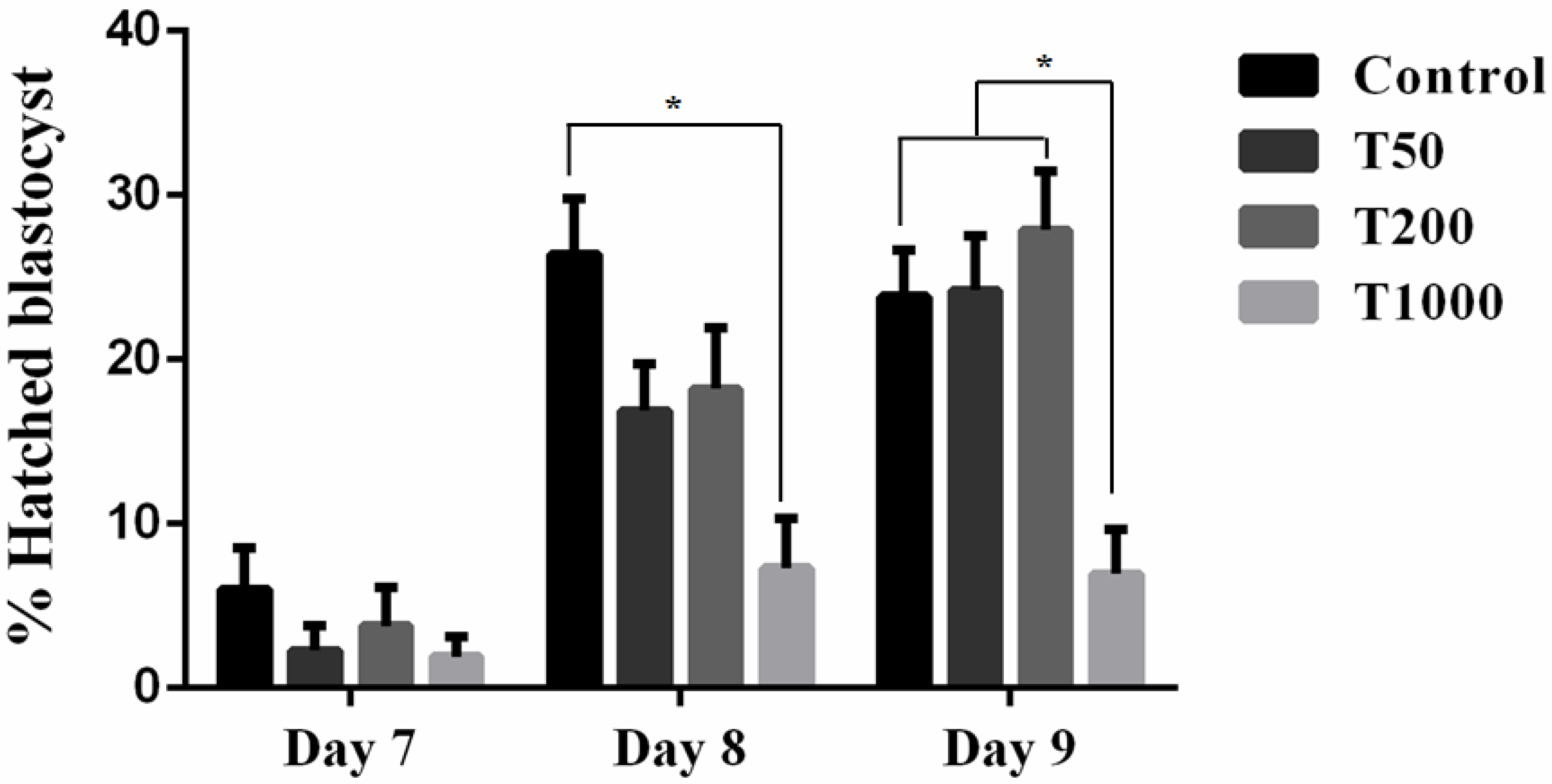
2.2.1. Developmental Competence
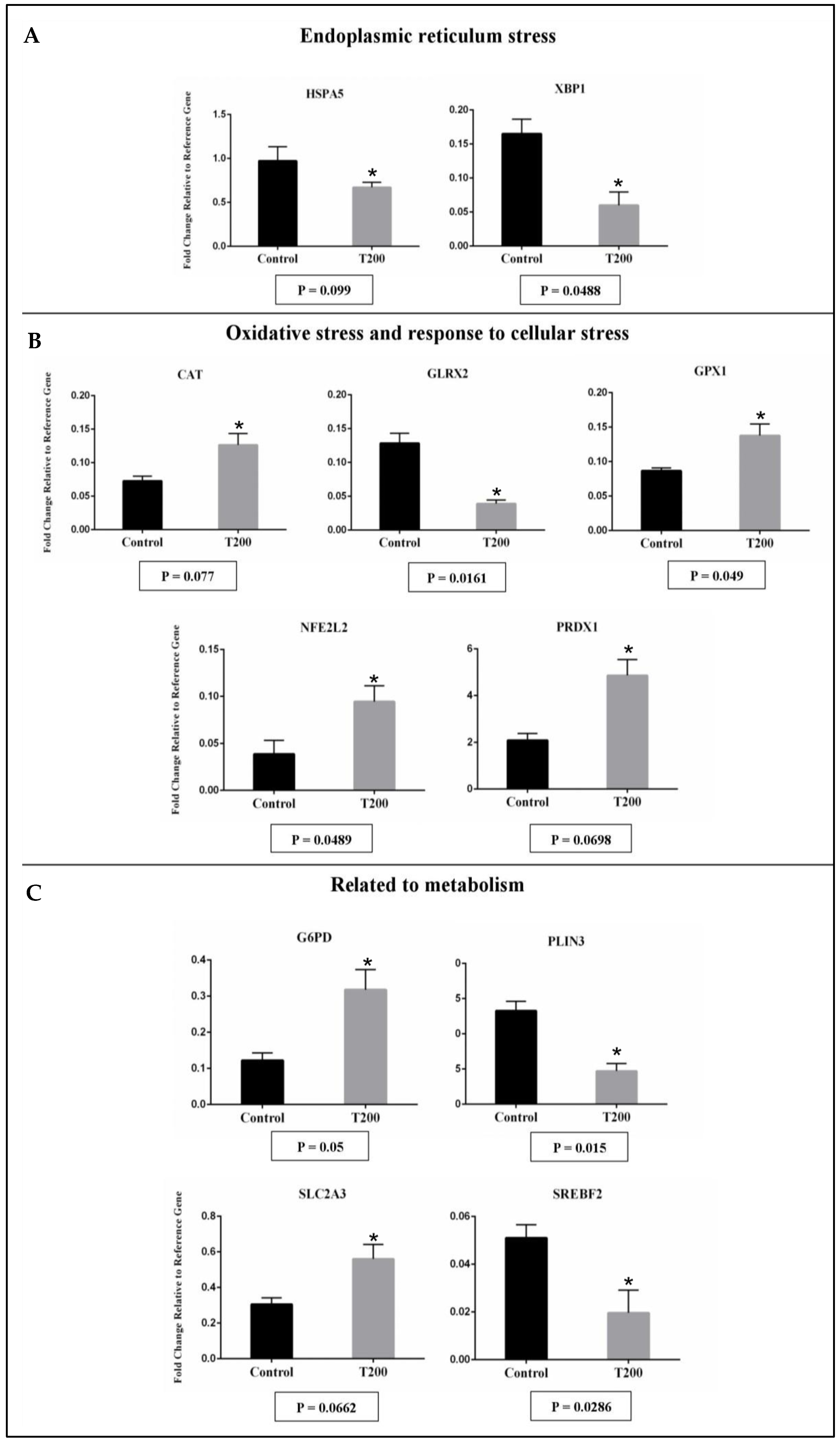
2.2.2. Gene Expression
3. Discussion
4. Materials and Methods
4.1. In Vitro Production
4.2. Chemical Treatment
4.3. Target-Transcripts Relative Quantitation: RT-qPCR
4.3.1. RNA Isolation and Reverse Transcription
4.3.2. Preamplification and Quantitative Polymerase Chain Reaction
4.4. Vitrification of Embryos
4.4.1. Embryo Freezing
4.4.2. Warming and Culture of Cryopreserved Embryos
4.5. Experimental Design
4.5.1. Experiment 1: The Effects of TUDCA during IVC on Developmental Competence and Gene Expression of Embryos
4.5.2. Experiment 2: The Effects of TUDCA during IVC on Developmental Competence and Gene Expression of Post-Warmed Vitrified Blastocysts
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chipurupalli, S.; Kannan, E.; Tergaonkar, V.; D’Andrea, R.; Robinson, N. Hypoxia Induced ER Stress Response as an Adaptive Mechanism in Cancer. Int. J. Mol. Sci. 2019, 20, 749. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, A.; Chen, A.W.; Varner, J.D. A review of the mammalian unfolded protein response. Biotechnol. Bioeng. 2011, 108, 2777–2793. [Google Scholar] [CrossRef]
- Ghemrawi, R.; Battaglia-Hsu, S.-F.; Arnold, C. Endoplasmic Reticulum Stress in Metabolic Disorders. Cells 2018, 7, 63. [Google Scholar] [CrossRef]
- Hetz, C.; Zhang, K.; Kaufman, R.J. Mechanisms, regulation and functions of the unfolded protein response. Nat. Rev. Mol. Cell Biol. 2020, 21, 421–438. [Google Scholar] [CrossRef] [PubMed]
- Iwawaki, T.; Akai, R.; Kohno, K.; Miura, M. A transgenic mouse model for monitoring endoplasmic reticulum stress. Nat. Med. 2003, 10, 98–102. [Google Scholar] [CrossRef] [PubMed]
- van Schadewijk, A.; Wout, E.F.A.V.; Stolk, J.; Hiemstra, P.S. A quantitative method for detection of spliced X-box binding protein-1 (XBP1) mRNA as a measure of endoplasmic reticulum (ER) stress. Cell Stress Chaperones 2011, 17, 275–279. [Google Scholar] [CrossRef]
- Szegezdi, E.; Logue, S.E.; Gorman, A.M.; Samali, A. Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep. 2006, 7, 880–885. [Google Scholar] [CrossRef]
- Van der Kallen, C.J.; Van Greevenbroek, M.M.; Stehouwer, C.D.; Schalkwijk, C.G. Endoplasmic reticulum stress-induced apoptosis in the development of diabetes: Is there a role for adipose tissue and liver? Apoptosis 2009, 14, 1424–1434. [Google Scholar] [CrossRef]
- Zeng, F.; Schultz, R.M. RNA transcript profiling during zygotic gene activation in the preimplantation mouse embryo. Dev. Biol. 2005, 283, 40–57. [Google Scholar] [CrossRef]
- Richter, K.S. The importance of growth factors for preimplantation embryo development and in-vitro culture. Curr. Opin. Obstet. Gynecol. 2008, 20, 292–304. [Google Scholar] [CrossRef]
- Lonergan, P.; Fair, T.; Corcoran, D.; Evans, A. Effect of culture environment on gene expression and developmental characteristics in IVF-derived embryos. Theriogenology 2006, 65, 137–152. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Chaudhry, P.; Asselin, E. Bridging endometrial receptivity and implantation: Network of hormones, cytokines, and growth factors. J. Endocrinol. 2011, 210, 5–14. [Google Scholar] [CrossRef]
- Rizos, D.; Ward, F.; Duffy, P.; Boland, M.P.; Lonergan, P. Consequences of bovine oocyte maturation, fertilization or early embryo development in vitro versus in vivo: Implications for blastocyst yield and blastocyst quality. Mol. Reprod. Dev. 2002, 61, 234–248. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Lee, J.E.; Kang, J.W.; Shin, H.Y.; Bin Lee, J.; Jin, D.I. Endoplasmic Reticulum (ER) Stress and Unfolded Protein Response (UPR) in Mammalian Oocyte Maturation and Preimplantation Embryo Development. Int. J. Mol. Sci. 2019, 20, 409. [Google Scholar] [CrossRef] [PubMed]
- Latham, K.E. Stress signaling in mammalian oocytes and embryos: A basis for intervention and improvement of outcomes. Cell Tissue Res. 2016, 363, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Song, B.-S.; Yoon, S.-B.; Sim, B.-W.; Kim, Y.-H.; Cha, J.-J.; Choi, S.-A.; Jeong, K.-J.; Kim, J.-S.; Huh, J.-W.; Lee, S.-R.; et al. Valproic acid enhances early development of bovine somatic cell nuclear transfer embryos by alleviating endoplasmic reticulum stress. Reprod. Fertil. Dev. 2014, 26, 432–440. [Google Scholar] [CrossRef]
- Zhang, J.Y.; Diao, Y.F.; Kim, H.R.; Jin, D.I. Inhibition of endoplasmic reticulum stress improves mouse embryo development. PLoS ONE 2012, 7, e40433. [Google Scholar] [CrossRef]
- Kim, J.-S.; Song, B.-S.; Lee, K.-S.; Kim, D.-H.; Kim, S.-U.; Choo, Y.-K.; Chang, K.-T.; Koo, D.-B. Tauroursodeoxycholic acid enhances the pre-implantation embryo development by reducing apoptosis in pigs. Reprod. Domest. Anim. 2012, 47, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.Y.; Hong, S.H.; Lee, Y.J.; Chung, S.S.; Jung, H.S.; Park, S.G.; Park, K.S. Tauroursodeoxycholate (TUDCA), chemical chaperone, enhances function of islets by reducing ER stress. Biochem. Biophys. Res. Commun. 2010, 397, 735–739. [Google Scholar] [CrossRef]
- Yoon, S.-B.; Choi, S.-A.; Sim, B.-W.; Kim, J.-S.; Mun, S.-E.; Jeong, P.-S.; Yang, H.-J.; Lee, Y.; Park, Y.-H.; Song, B.-S.; et al. Developmental competence of bovine early embryos depends on the coupled response between oxidative and endoplasmic reticulum stress. Biol. Reprod. 2014, 90, 104. [Google Scholar] [CrossRef]
- Zhang, J.Y.; Diao, Y.F.; Oqani, R.K.; Han, R.X.; Jin, D.I. Effect of endoplasmic reticulum stress on porcine oocyte maturation and parthenogenetic embryonic development in vitro. Biol. Reprod. 2012, 86, 128. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Liu, X.-J.; Li, J.-T.; Zhang, L.; Fu, Y.; Zhang, Y.-J.; Chen, R.-X.; Wei, X.-Q.; Wang, R.; Wang, Y.; et al. Endoplasmic reticulum stress inhibition is a valid therapeutic strategy in vitrifying oocytes. Cryobiology 2015, 70, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Mochizuki, M.; Miyagi, K.; Kishigami, S. Optimizing treatment of tauroursodeoxycholic acid to improve embryonic development after in vitro maturation of cumulus-free oocytes in mice. PLoS ONE 2018, 13, e0202962. [Google Scholar] [CrossRef] [PubMed]
- Khatun, H.; Wada, Y.; Konno, T.; Tatemoto, H.; Yamanaka, K.-I. Endoplasmic reticulum stress attenuation promotes bovine oocyte maturation in vitro. Reproduction 2020, 159, 361–370. [Google Scholar] [CrossRef]
- Pioltine, E.M.; Costa, C.B.; Latorraca, L.B.; Franchi, F.F.; dos Santos, P.H.; Mingoti, G.Z.; de Paula-Lopes, F.F.; Nogueira, M.F.G. Treatment of in vitro-matured bovine oocytes with tauroursodeoxycholic acid modulates the oxidative stress signaling pathway. Front. Cell Dev. Biol. 2021, 9, 623852. [Google Scholar] [CrossRef]
- Khatun, H.; Ihara, Y.; Takakura, K.; Egashira, J.; Wada, Y.; Konno, T.; Tatemoto, H.; Yamanaka, K.-I. Role of endoplasmic reticulum stress on developmental competency and cryo-tolerance in bovine embryos. Theriogenology 2020, 142, 131–137. [Google Scholar] [CrossRef]
- Valente, R.S.; Marsico, T.V.; Sudano, M.J. Basic and applied features in the cryopreservation progress of bovine embryos. Anim. Reprod. Sci. 2022, 239, 106970. [Google Scholar] [CrossRef]
- Abe, H.; Yamashita, S.; Satoh, T.; Hoshi, H. Accumulation of cytoplasmic lipid droplets in bovine embryos and cryotolerance of embryos developed in different culture systems using serum-free or serum-containing media. Mol. Reprod. Dev. 2001, 61, 57–66. [Google Scholar] [CrossRef]
- Sudano, M.J.; Paschoal, D.M.; da Silva Rascado, T.; Crocomo, L.F.; Magalhães, L.C.O.; Junior, A.M.; Machado, R.; da Cruz Landim-Alvarenga, F. Crucial surviving aspects for vitrified in vitro-produced bovine embryos. Zygote 2014, 22, 124–131. [Google Scholar] [CrossRef]
- Takahashi, M.; Keicho, K.; Takahashi, H.; Ogawa, H.; Schulte, R.; Okano, A. Effect of oxidative stress on development and DNA damage in in-vitro cultured bovine embryos by comet assay. Theriogenology 2000, 54, 137–145. [Google Scholar] [CrossRef]
- Xie, Y.; Wang, F.; Puscheck, E.; Rappolee, D. Pipetting causes shear stress and elevation of phosphorylated stress-activated protein kinase/jun kinase in preimplantation embryos. Mol. Reprod. Dev. 2007, 74, 1287–1294. [Google Scholar] [CrossRef]
- Lin, T.; Zhang, J.Y.; Diao, Y.F.; Kang, J.W.; Jin, D.-I. Effects of sorbitol on porcine oocyte maturation and embryo development in vitro. Zygote 2015, 23, 297–306. [Google Scholar] [CrossRef] [PubMed]
- López-Damián, E.P.; Jiménez-Medina, J.A.; Alarcón, M.A.; Lammoglia, M.A.; Hernández, A.; Galina, C.S.; Fiordelisio, T. Cryopreservation induces higher oxidative stress levels in Bos indicus embryos compared with Bos taurus. Theriogenology 2020, 143, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Paschoal, D.M.; Sudano, M.J.; Schwarz, K.R.L.; Maziero, R.R.D.; Guastali, M.D.; Crocomo, L.F.; Magalhães, L.C.O.; Martins, J.A., Jr.; Leal, C.L.V.; Landim-Alvarenga, F.D.C. Cryotolerance and global gene-expression patterns of Bos taurus indicus and Bos taurus taurus in vitro- and in vivo-produced blastocysts. Reprod. Fertil. Dev. 2014, 26, 1129–1141. [Google Scholar] [CrossRef]
- Aksu, D.A.; Agca, C.; Aksu, S.; Bagis, H.; Akkoc, T.; Caputcu, A.T.; Arat, S.; Taskin, A.C.; Kizil, S.H.; Karasahin, T.; et al. Gene expression profiles of vitrified in vitro- and in vivo-derived bovine blastocysts. Mol. Reprod. Dev. 2012, 79, 613–625. [Google Scholar] [CrossRef] [PubMed]
- Hua, Y.; Kandadi, M.R.M.; Zhu, M.; Ren, J.; Sreejayan, N.M. Tauroursodeoxycholic acid attenuates lipid accumulation in endoplasmic reticulum–stressed macrophages. J. Cardiovasc. Pharmacol. 2010, 55, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Cha, B.-H.; Kim, J.-S.; Ahn, J.C.; Kim, H.-C.; Kim, B.-S.; Han, D.K.; Park, S.G.; Lee, S.-H. The role of tauroursodeoxycholic acid on adipogenesis of human adipose-derived stem cells by modulation of ER stress. Biomaterials 2014, 35, 2851–2858. [Google Scholar] [CrossRef]
- Lee, K.; Tirasophon, W.; Shen, X.; Michalak, M.; Prywes, R.; Okada, T.; Yoshida, H.; Mori, K.; Kaufman, R.J. IRE-1-mediated unconventional mRNA splicing and S2P-mediated ATF6 cleavage merges to regulate XBP1 in signaling the unfolded protein response. Genes Dev. 2002, 16, 452–466. [Google Scholar] [CrossRef]
- Hosoi, T.; Ogawa, K.; Ozawa, K. Homocysteine induces X-box-binding protein 1 splicing in the mice brain. Neurochem. Int. 2010, 56, 216–220. [Google Scholar] [CrossRef]
- Wild, A.C.; Moinova, H.R.; Mulcahy, R.T. Regulation of gamma-glutamyl-cysteine synthetase subunit gene expression by the transcription factor Nrf2. J. Biol. Chem. 1999, 274, 33627–33636. [Google Scholar] [CrossRef]
- Tanaka, Y.; Aleksunes, L.M.; Yeager, R.L.; Gyamfi, M.A.; Esterly, N.; Guo, G.L.; Klaassen, C.D. NF-E2-related factor 2 inhibits lipid accumulation and oxidative stress in mice fed a high-fat diet. J. Pharmacol. Exp. Ther. 2008, 325, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Moreira, S.; Fonseca, I.; Nunes, M.J.; Rosa, A.; Lemos, L.; Rodrigues, E.; Carvalho, A.N.; Outeiro, T.F.; Rodrigues, C.M.P.; Gama, M.J.; et al. Nrf2 activation by tauroursodeoxycholic acid in experimental models of Parkinson’s disease. Exp. Neurol. 2017, 295, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.-C.; Chen, T.-L.; Wu, Y.-H.; Cheng, K.-P.; Lin, Y.-H.; Cheng, M.-L.; Ho, H.-Y.; Lo, S.J.; Chiu, D.T.-Y. Glucose 6-phosphate dehydrogenase deficiency enhances germ cell apoptosis and causes defective embryogenesis in Caenorhabditis elegans. Cell Death Dis. 2013, 4, e616. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.H.; Lee, Y.H.; Shih, H.Y.; Chen, S.H.; Cheng, Y.C.; Tsun-Yee Chiu, D. Glucose-6-phosphate dehydrogenase is indispensable in embryonic development by modulation of epithelial-mesenchymal transition via the NOX/Smad3/miR-200b axis. Cell Death Dis. 2018, 9, 10. [Google Scholar] [CrossRef]
- Purcell, S.H.; Moley, K.H. Glucose transporters in gametes and preimplantation embryos. Trends Endocrinol. Metab. 2009, 20, 483–489. [Google Scholar] [CrossRef]
- Ortega, M.S.; Kelleher, A.M.; O’Neil, E.; Benne, J.; Cecil, R.; Spencer, T.E. NANOG is required to form the epiblast and maintain pluripotency in the bovine embryo. Mol. Reprod. Dev. 2020, 87, 152–160. [Google Scholar] [CrossRef]
- Song, B.-S.; Kim, J.-S.; Yoon, S.-B.; Lee, K.-S.; Koo, D.-B.; Lee, D.-S.; Choo, Y.-K.; Huh, J.-W.; Lee, S.-R.; Kim, S.-U.; et al. Inactivated Sendai-virus-mediated fusion improves early development of cloned bovine embryos by avoiding endoplasmic-reticulum-stress-associated apoptosis. Reprod. Fertil. Dev. 2011, 23, 826–836. [Google Scholar] [CrossRef]
- Sharma, A.; Agrawal, H.; Mullani, N.; Sandhu, A.; Singh, M.K.; Chauhan, M.S.; Singla, S.K.; Palta, P.; Manik, R.S. Supplementation of tauroursodeoxycholic acid during IVC did not enhance in vitro development and quality of buffalo IVF embryos but combated endoplasmic reticulum stress. Theriogenology 2015, 84, 200–207. [Google Scholar] [CrossRef]
- Lin, T.; Lee, J.E.; Oqani, R.K.; Kim, S.Y.; Cho, E.S.; Jeong, Y.D.; Baek, J.J.; Jin, D.I. Tauroursodeoxycholic acid improves pre-implantation development of porcine SCNT embryo by endoplasmic reticulum stress inhibition. Reprod. Biol. 2016, 16, 269–278. [Google Scholar] [CrossRef]
- Wang, J.; Lee, J.; Liem, D.; Ping, P. HSPA5 Gene encoding Hsp70 chaperone BiP in the endoplasmic reticulum. Gene 2017, 618, 14–23. [Google Scholar] [CrossRef]
- Campanella, C.; Pace, A.; Caruso Bavisotto, C.; Marzullo, P.; Marino Gammazza, A.; Buscemi, S.; Palumbo Piccionello, A. Heat shock proteins in Alzheimer’s disease: Role and targeting. Int. J. Mol. Sci. 2018, 19, 2603. [Google Scholar] [CrossRef] [PubMed]
- Harding, H.P.; Zhang, Y.; Bertolotti, A.; Zeng, H.; Ron, D. Perk is essential for translational regulation and cell survival during the unfolded protein response. Mol. Cell 2000, 5, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Landau, G.; Kodali, V.K.; Malhotra, J.D.; Kaufman, R.J. Detection of oxidative damage in response to protein misfolding in the endoplasmic reticulum. Methods Enzymol. 2013, 526, 231–250. [Google Scholar] [CrossRef] [PubMed]
- van der Vlies, D.; Makkinje, M.; Jansens, A.; Braakman, I.; Verkleij, A.J.; Wirtz, K.W.A.; Post, J.A.; Kim, J.; Yun, M.; Kim, E.-O.; et al. Oxidation of er resident proteins upon oxidative stress: Effects of altering cellular redox/antioxidant status and implications for protein maturation. Antioxid. Redox Signal. 2003, 5, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Hausburg, M.A.; DeKrey, G.K.; Salmen, J.J.; Palic, M.R.; Gardiner, C.S. Effects of paraquat on development of preimplantation embryos in vivo and in vitro. Reprod. Toxicol. 2005, 20, 239–246. [Google Scholar] [CrossRef]
- Kammoun, H.L.; Chabanon, H.; Hainault, I.; Luquet, S.; Magnan, C.; Koike, T.; Ferré, P.; Foufelle, F. GRP78 expression inhibits insulin and ER stress–induced SREBP-1c activation and reduces hepatic steatosis in mice. J. Clin. Investig. 2009, 119, 1201–1215. [Google Scholar] [CrossRef]
- Zhang, K.; Wang, S.; Malhotra, J.; Hassler, J.R.; Back, S.H.; Wang, G.; Chang, L.; Xu, W.; Miao, H.; Leonardi, R.; et al. The unfolded protein response transducer IRE1α prevents ER stress-induced hepatic steatosis. EMBO J. 2011, 30, 1357–1375. [Google Scholar] [CrossRef]
- Lauressergues, E.; Bert, E.; Duriez, P.; Hum, D.; Majd, Z.; Staels, B.; Cussac, D. Does endoplasmic reticulum stress participate in APD-induced hepatic metabolic dysregulation? Neuropharmacology 2012, 62, 784–796. [Google Scholar] [CrossRef]
- Han, J.; Kaufman, R.J. The role of ER stress in lipid metabolism and lipotoxicity. J. Lipid. Res. 2016, 57, 1329–1338. [Google Scholar] [CrossRef]
- Harris, D.; Ben Huang, B.; Oback, B. Inhibition of MAP2K and GSK3 signaling promotes bovine blastocyst development and epiblast-associated expression of pluripotency factors 1. Biol. Reprod. 2013, 88, 74. [Google Scholar] [CrossRef]
- Botigelli, R.C.; Razza, E.M.; Pioltine, E.M.; Fontes, P.K.; Schwarz, K.R.L.; Leal, C.L.V.; Nogueira, M.F.G. Supplementing in vitro embryo production media by NPPC and sildenafil affect the cytoplasmic lipid content and gene expression of bovine cumulus-oocyte complexes and embryos. Reprod. Biol. 2018, 18, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Seneda, M.M.; Esper, C.R.; Garcia, J.M.; de Oliveira, J.A.; Vantini, R. Relationship between follicle size and ultrasound-guided transvaginal oocyte recovery. Anim. Reprod. Sci. 2001, 67, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Vajta, G.; Holm, P.; Greve, T.; Callesen, H. The submarine incubation system, a new tool for in vitro embryo culture: A technique report. Theriogenology 1997, 48, 1379–1385. [Google Scholar] [CrossRef]
- Park, H.J.; Park, J.Y.; Kim, J.W.; Yang, S.G.; Jung, J.M.; Kim, M.J.; Park, J.J.; Koo, D.B. Regulation of the Endoplasmic Reticulum Stress by BIP/GRP78 is involved in Meiotic Maturation of Porcine Oocytes In Vitro. Dev. Reprod. 2017, 21, 407–415. [Google Scholar] [CrossRef] [PubMed]
| Treatment | Vitrified Embryos | Re-Exp. Rate | Re-Exp. Rate | Re-Exp. Rate | Hatch. Rate | Hatch. Rate | Hatch. Rate |
|---|---|---|---|---|---|---|---|
| (D7/D8) | 12 h (%) | 24 h (%) | 48 h (%) | 12 h (%) | 24 h (%) | 48 h (%) | |
| Control | 88 | 55.54 ± 5.62 | 67.19 ± 4.81 | 67.19 ± 4.81 | 3.23 ± 1.33 | 11.71 ± 3.28 b | 26.67 ± 4.72 b |
| T200 | 117 | 61.88 ± 3.63 | 68.22 ± 5.16 | 79.00 ± 4.51 | 7.61 ± 2.44 | 20.50 ± 3.15 a | 45.87 ± 4.62 a |
| p-value | - | 0.4254 | 0.8885 | 0.1112 | 0.1552 | 0.09 | 0.0423 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pioltine, E.M.; Costa, C.B.; Franchi, F.F.; dos Santos, P.H.; Nogueira, M.F.G. Tauroursodeoxycholic Acid Supplementation in In Vitro Culture of Indicine Bovine Embryos: Molecular and Cellular Effects on the In Vitro Cryotolerance. Int. J. Mol. Sci. 2023, 24, 14060. https://doi.org/10.3390/ijms241814060
Pioltine EM, Costa CB, Franchi FF, dos Santos PH, Nogueira MFG. Tauroursodeoxycholic Acid Supplementation in In Vitro Culture of Indicine Bovine Embryos: Molecular and Cellular Effects on the In Vitro Cryotolerance. International Journal of Molecular Sciences. 2023; 24(18):14060. https://doi.org/10.3390/ijms241814060
Chicago/Turabian StylePioltine, Elisa Mariano, Camila Bortoliero Costa, Fernanda Fagali Franchi, Priscila Helena dos Santos, and Marcelo Fábio Gouveia Nogueira. 2023. "Tauroursodeoxycholic Acid Supplementation in In Vitro Culture of Indicine Bovine Embryos: Molecular and Cellular Effects on the In Vitro Cryotolerance" International Journal of Molecular Sciences 24, no. 18: 14060. https://doi.org/10.3390/ijms241814060
APA StylePioltine, E. M., Costa, C. B., Franchi, F. F., dos Santos, P. H., & Nogueira, M. F. G. (2023). Tauroursodeoxycholic Acid Supplementation in In Vitro Culture of Indicine Bovine Embryos: Molecular and Cellular Effects on the In Vitro Cryotolerance. International Journal of Molecular Sciences, 24(18), 14060. https://doi.org/10.3390/ijms241814060






