Polyvalent Ionic Energetic Salts Based on 4-Amino-3-hydrazino-5-methyl-1,2,4-triazole
Abstract
:1. Introduction
2. Results and Discussion
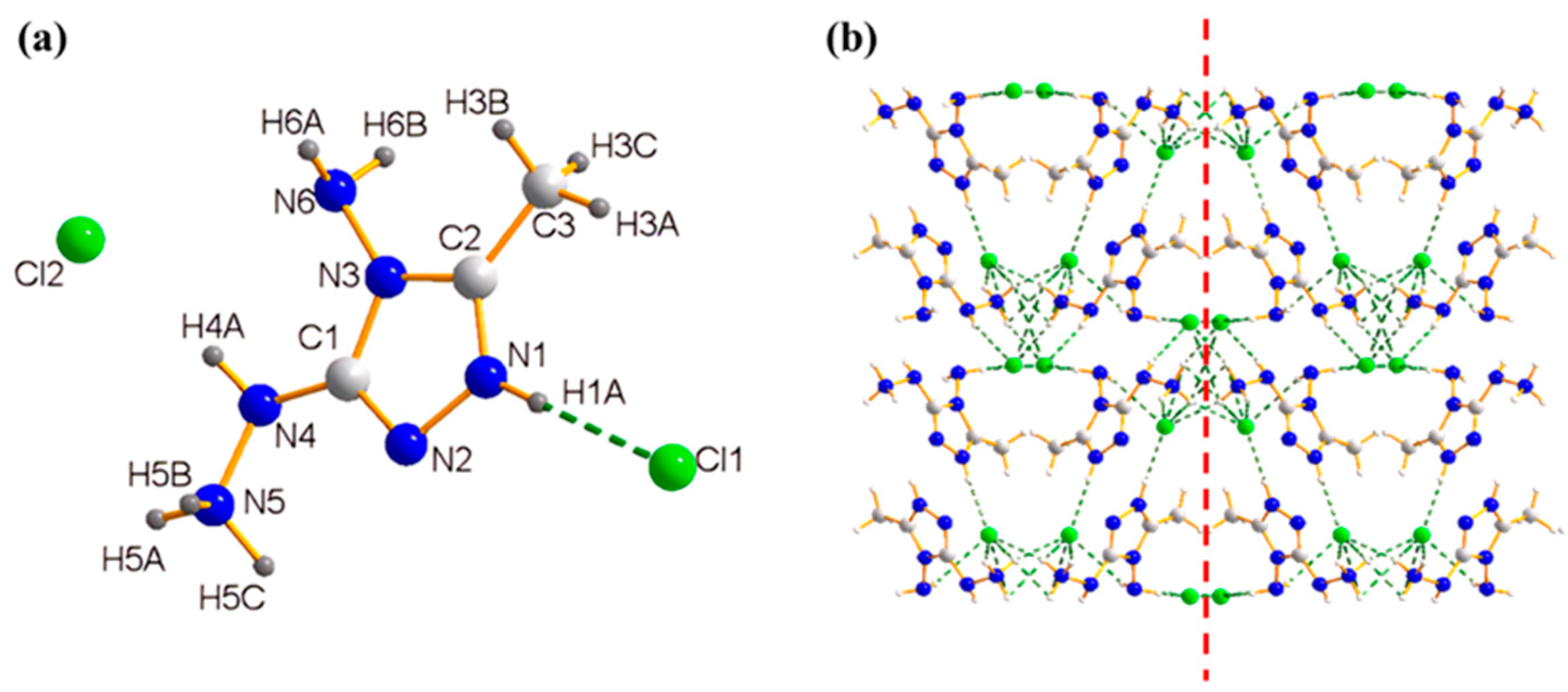
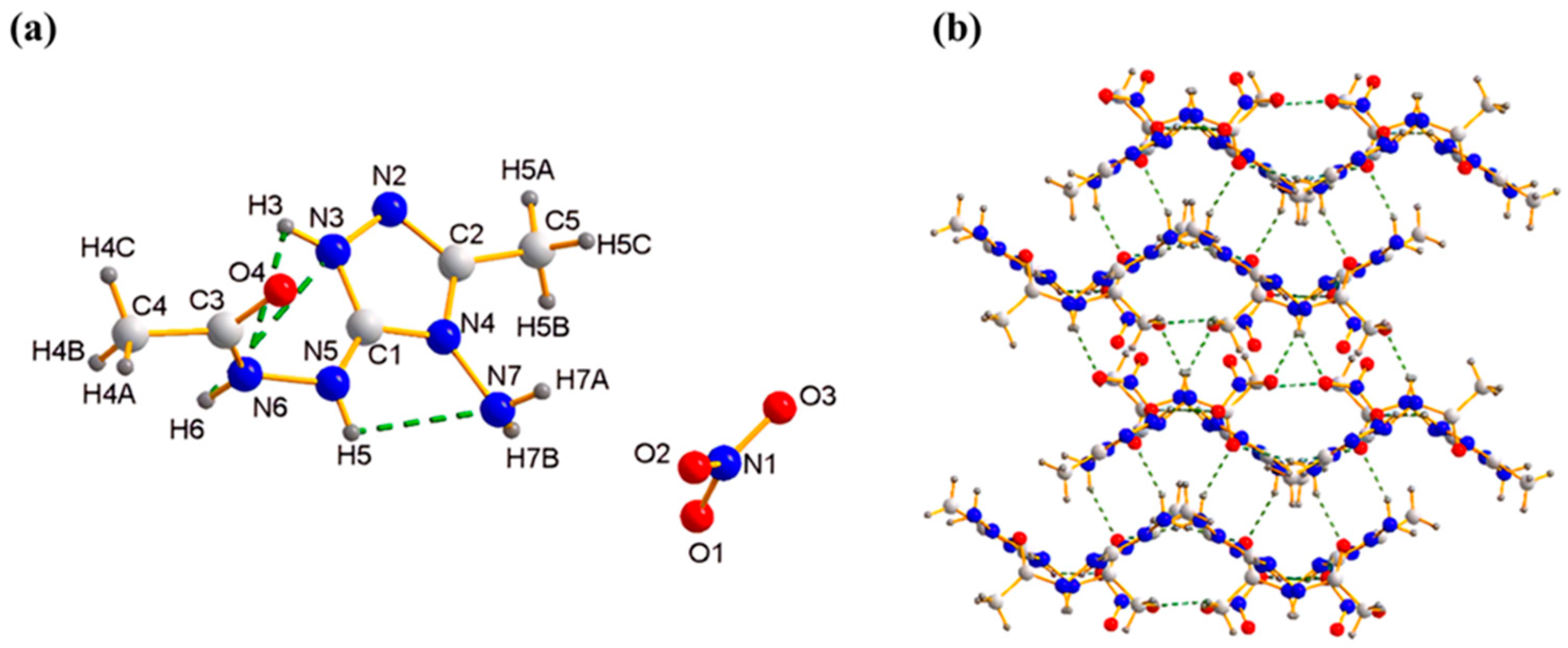
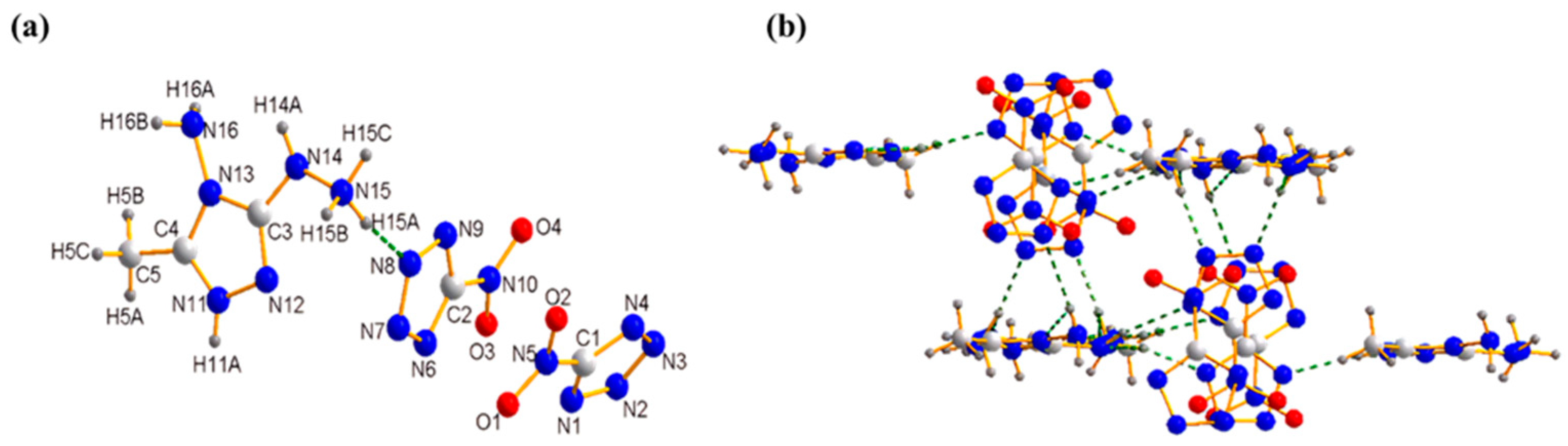
2.1. X-ray Crystallography
2.2. Physicochemical and Energetic Properties
2.3. 2D Fingerprints and Hirshfeld Surface Analysis
2.4. Noncovalent Interaction (NCI) Analysis
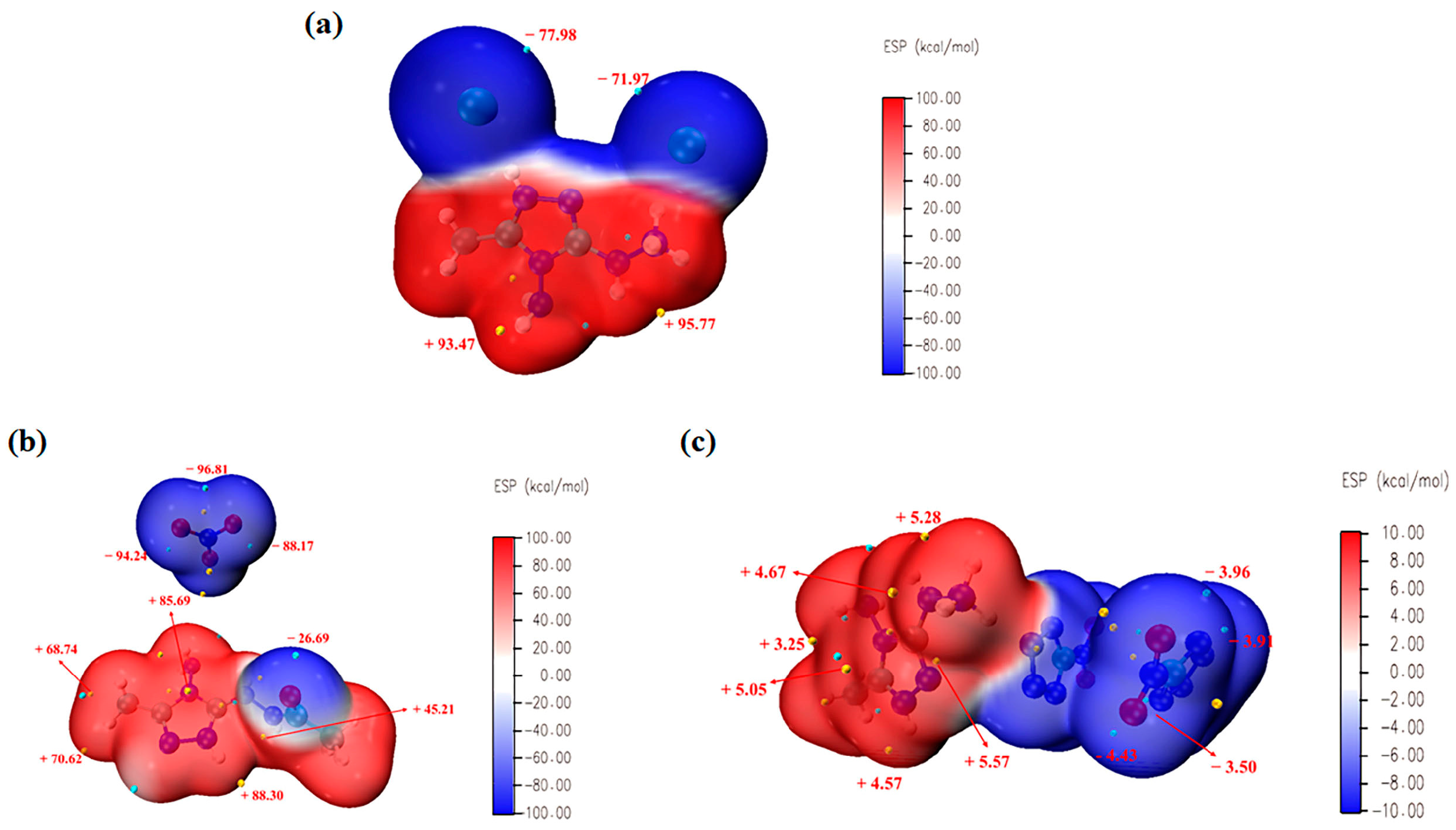
2.5. Electrostatic Potential Surface (ESP) Analysis
3. Materials and Methods
3.1. Experimental Section
3.1.1. Caution
3.1.2. Synthesis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, J.H.; Xia, M.; Jiang, N. Critical shock initiation characteristics of TNT with different charging types. Shock Waves 2023, 33, 39–49. [Google Scholar] [CrossRef]
- Sorescu, D.C.; Larentzos, J.P.; Rice, B.M.; Brennan, J.K. Toward Addressing the Challenge to Predict the Heat Capacities of RDX and HMX Energetic Materials. Prop. Explos. Pyrotech. 2022, 47, e202100338. [Google Scholar] [CrossRef]
- Jin, D.; Xu, J.; Zhang, H.; Lei, M.; Sun, J. Comparative Study of Experiments and Calculations on the Guest Molecules’ Escaping Mechanism of CL-20-Based Host–Guest Energetic Materials. J. Phys. Chem. C 2023, 127, 11641–11651. [Google Scholar] [CrossRef]
- Tang, J.; Yang, H.; Cui, Y.; Cheng, G. Nitrogen-rich tricyclic-based energetic materials. Mater. Chem. Front. 2021, 5, 7108–7118. [Google Scholar] [CrossRef]
- Zhai, L.; Zhang, J.; Wu, M.; Huo, H.; Bi, F.; Wang, B. Balancing good oxygen balance and high heat of formation by incorporating of -C(NO2)2F Moiety and Tetrazole into Furoxan block. J. Mol. Struct. 2020, 1222, 128934. [Google Scholar] [CrossRef]
- Wang, Z.; Jin, D.; Du, Z.; Song, X.; Sun, S.; Zhang, Z.; Xu, J. Crystal packing driven comparable thermal expansion of a biimidazole-based energetic material. J. Therm. Anal. Calorim. 2023, 148, 4001–4014. [Google Scholar] [CrossRef]
- Tang, J.; Xiong, H.; Tang, Y.; Yang, H.; Cheng, G. The design and synthesis of new advanced energetic materials based on pyrazole–triazole backbones. Dalton Trans. 2023, 52, 3169–3175. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Shao, Y.; Lu, M. Amino-tetrazole functionalized fused triazolo-triazine and tetrazolo-triazine energetic materials. Chem. Commun. 2019, 55, 6062–6065. [Google Scholar] [CrossRef]
- Mondal, J.; Sivaramakrishna, A. Functionalized Triazines and Tetrazines: Synthesis and Applications. Top. Curr. Chem. 2022, 380, 34. [Google Scholar] [CrossRef]
- Wang, S.; Chen, X.; Chen, Y.; Nan, H.; Li, Y.; Ma, H. Synthesis, thermal behaviors, and energetic properties of asymmetrically substituted tetrazine-based energetic materials. Front. Chem. 2022, 10, 978003. [Google Scholar] [CrossRef]
- Yang, F.; Qin, Y.; Jiang, S.; Lin, Q.; Wang, P.; Xu, Y.; Lu, M. Lithium-Promoted Formation of M-2AZTO-Li (M = N2H5+ or NH3OH+ and AZTO = Anion of 1-Hydroxytetrazole-5-hydrazide)-Type “Quaternary” Complexes with Nitrogen-Rich Characteristics: Construction of Novel Insensitive Energetic Materials. ACS Appl. Mater. Interfaces 2023, 15, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, Z.; Hsieh, Y.; Wang, B.; Huang, H.; Yang, J.; Zhang, J. A promising cation of 4-aminofurazan-3-carboxylic acid amidrazone in desensitizing energetic materials. RSC Adv. 2020, 10, 2519–2525. [Google Scholar] [CrossRef]
- Wozniak, D.R.; Zeller, M.; Byrd, E.F.C.; Piercey, D.G. 3-Methyl-1,2,3-triazolium-1N-dinitromethylylide and the strategy of zwitterionic dinitromethyl groups in energetic materials design. RSC Adv. 2021, 11, 17710–17714. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Liu, Y.; Dong, Z.; Li, H.; Ye, Z. 3-R-4-(5-Methyleneazide-1,2,4-oxadiazol-3-yl) furazan and its ionic salts as low-sensitivity and high-detonation energetic materials. New J. Chem. 2021, 45, 11380–11389. [Google Scholar] [CrossRef]
- Kang, Y.; Dong, Y.; Liu, Y.; Gao, H.; Wang, Y.; Shreeve, J.n.M. Halogen bonding (C-F···X) and its effect on creating ideal insensitive energetic materials. Chem. Eng. J. 2022, 440, 135969. [Google Scholar] [CrossRef]
- Zhang, W.; Yang, Y.; Pang, S.-P.; Li, Y.-C.; Sun, C. Regulation of stability and density of energetic materials via isomerism. Phys. Chem. Chem. Phys. 2023, 25, 20168–20172. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Gong, X.; Mei, H.; Li, T.; Zhang, J.; Gozin, M. Design of Zero Oxygen Balance Energetic Materials on the Basis of Diels–Alder Chemistry. J. Org. Chem. 2018, 83, 14698–14702. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, A.T.; Lee, R.J.; Watkins, S.; Brinkman, J.; Stephens, J.C.; Dickens, J.C.; Neuber, A.A. Apparatus for skidding sensitivity testing of energetic materials. Rev. Sci. Instrum. 2021, 92, 45101. [Google Scholar] [CrossRef]
- Suceska, M. EXPLO5—Computer Program for Calculation of Detonation Parameters. In Proceedings of the 32nd International Annual Conference of ICT, Karlsruhe, Germany, 3–6 July 2001. [Google Scholar]
- Frisch, M.J.; Trucks, G.; Schlegel, H.B.; Scuseria, G.E.; Robb; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A. Gaussian 09W; revision A. 02; Gaussian Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Spackman, M.A.; Jayatilaka, D. Hirshfeld surface analysis. CrystEngComm 2009, 11, 19–32. [Google Scholar] [CrossRef]
- Spackman, P.R.; Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer: A program for Hirshfeld surface analysis, visualization and quantitative analysis of molecular crystals. J. Appl. Crystallogr. 2021, 54, 1006–1011. [Google Scholar] [CrossRef]
- Johnson, E.R.; Keinan, S.; Mori-Sánchez, P.; Contreras-García, J.; Cohen, A.J.; Yang, W. Revealing Noncovalent Interactions. J. Am. Chem. Soc. 2010, 132, 6498–6506. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Chen, F. Multiwfn: A multifunctional wavefunction analyzer. J. Comput. Chem. 2012, 33, 580–592. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. Model 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Zhang, J.; Lu, T. Efficient evaluation of electrostatic potential with computerized optimized code. Phys. Chem. Chem. Phys. 2021, 23, 20323–20328. [Google Scholar] [CrossRef] [PubMed]



| Salt | Td a | Tsd b | dc c/dm d | OB e | P h | D i | IS j | |
|---|---|---|---|---|---|---|---|---|
| 3 | 213.7 | - | 1.445/1.435 | −64 | 246.79/1.23 | 17.0 | 6600 | >40 |
| 5 | 145.7 | 232.9 | 1.521/1.513 | −44.61 | 374.59/1.61 | 25.1 | 7705 | 31.6 |
| 6 | 163.8 | 302.6 | 1.69/1.679 | −12.6 | −280.5/−1.10 | 25.6 | 8170 | 16.3 |
| 7 | 126.8 | 266.5 | 2.01/1.989 | 0.0 | −291.1/−0.88 | 34.2 | 8528 | 28.9 |
| 8 | 164.6 | 231.1 | 1.78/1.791 | 0.0 | 177.4/0.52 | 33.9 | 8887 | 10.7 |
| 9 | 173.4 | 233.2 | 1.69/1.621 | −26.8 | 697.8/1.95 | 23.6 | 8031 | 7.1 |
| 10 | 183.2 | 234.8 | 1.73/1.712 | −16.4 | 676.4/1.73 | 28.6 | 8524 | 6.1 |
| 11 | 173.9 | - | 1.65/1.652 | −43.4 | 354.4/1.37 | 22.6 | 8039 | >40 |
| TNT | 295.0 | - | 1.65 | −24.7 | −67/−0.295 | 19.5 | 6881 | 15 |
| RDX | 230 | - | 1.82 | −21.6 | 83.8/0.38 | 35.2 | 8977 | 7.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, Q.; Zhang, M.; Zhang, L.; Zhu, M.; Qin, K.; Wang, P. Polyvalent Ionic Energetic Salts Based on 4-Amino-3-hydrazino-5-methyl-1,2,4-triazole. Int. J. Mol. Sci. 2023, 24, 13136. https://doi.org/10.3390/ijms241713136
Lin Q, Zhang M, Zhang L, Zhu M, Qin K, Wang P. Polyvalent Ionic Energetic Salts Based on 4-Amino-3-hydrazino-5-methyl-1,2,4-triazole. International Journal of Molecular Sciences. 2023; 24(17):13136. https://doi.org/10.3390/ijms241713136
Chicago/Turabian StyleLin, Qiuhan, Mingmin Zhang, Linan Zhang, Mimi Zhu, Kaiyi Qin, and Pengcheng Wang. 2023. "Polyvalent Ionic Energetic Salts Based on 4-Amino-3-hydrazino-5-methyl-1,2,4-triazole" International Journal of Molecular Sciences 24, no. 17: 13136. https://doi.org/10.3390/ijms241713136
APA StyleLin, Q., Zhang, M., Zhang, L., Zhu, M., Qin, K., & Wang, P. (2023). Polyvalent Ionic Energetic Salts Based on 4-Amino-3-hydrazino-5-methyl-1,2,4-triazole. International Journal of Molecular Sciences, 24(17), 13136. https://doi.org/10.3390/ijms241713136




