Evidence of a Novel Mitochondrial Signature in Systemic Sclerosis Patients with Chronic Fatigue Syndrome
Abstract
1. Introduction
2. Results
2.1. Demographic and Clinical Characteristics of Study Population
2.2. ME/CFS and FM in SSc
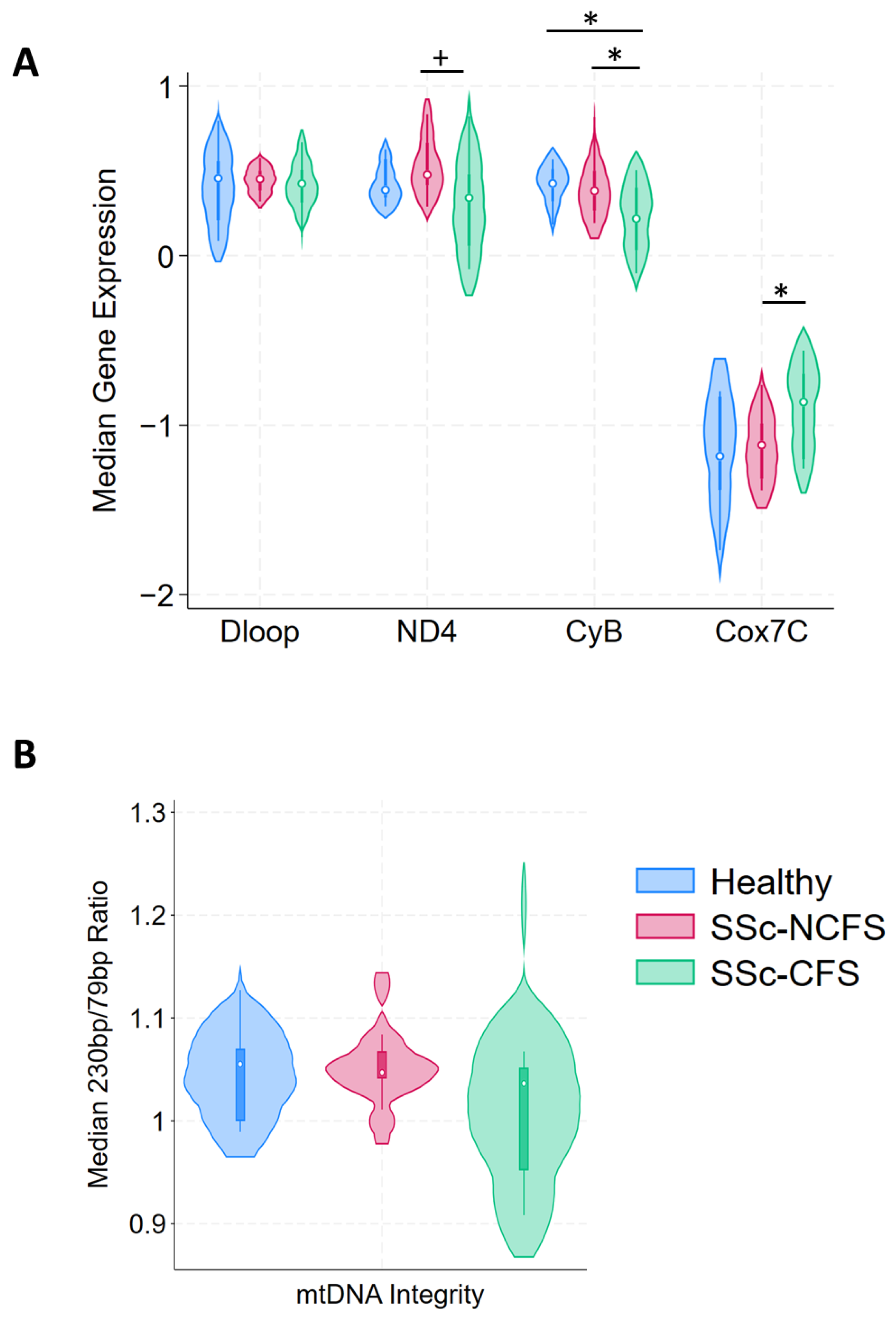
2.3. ETC Differential Expression and Cell Free Mitochondrial DNA Integrity
3. Discussion
4. Materials and Methods
4.1. Participants
4.2. Questionnaires
4.3. Mitochondrial Electron Transport Chain Expression
4.4. Cell-Free Mitochondrial DNA Analysis
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Minier, T.; Guiducci, S.; Bellando-Randone, S.; Bruni, C.; Lepri, G.; Czirják, L.; Distler, O.; A Walker, U.; Fransen, J.; Allanore, Y.; et al. Preliminary analysis of the Very Early Diagnosis of Systemic Sclerosis (VEDOSS) EUSTAR multicentre study: Evidence for puffy fingers as a pivotal sign for suspicion of systemic sclerosis. Ann. Rheum. Dis. 2014, 73, 2087–2093. [Google Scholar] [CrossRef]
- Basta, F.; Afeltra, A.; Margiotta, D.P.E. Fatigue in systemic sclerosis: A systematic review. Ann. Rheum. Dis. 2018, 36, 150–160. [Google Scholar]
- Van Eeden, C.; Osman, M.S.; Tervaert, J.W.C. Fatigue in ANCA-associated vasculitis (AAV) and systemic sclerosis (SSc): Similarities with Myalgic encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). A critical review of the literature. Expert Rev. Clin. Immunol. 2022, 18, 1049–1070. [Google Scholar] [CrossRef] [PubMed]
- Afari, N.; Buchwald, D. Chronic Fatigue Syndrome: A Review. Am. J. Psychiatry 2003, 160, 221–236. [Google Scholar] [CrossRef] [PubMed]
- Ostuni, P.; Botsios, C.; Sfriso, P.; Bertagnin, A.; Cozzi, F.; Doria, A.; Todesco, S. Prevalence and clinical features of fibromyalgia in systemic lupus erythematosus, systemic sclerosis and Sjögren’s syndrome. Minerva Medica 2002, 93, 203–209. [Google Scholar]
- Perrot, S.; Peixoto, M.; Dieude, P.; Hachulla, E.; Avouac, J.; Ottaviani, S.; Allanore, Y. Patient phenotypes in fibromyalgia comorbid with systemic sclerosis or rheumatoid arthritis: Influence of diagnostic and screening tests. Screening with the FiRST questionnaire, diagnosis with the ACR 1990 and revised ACR 2010 criteria. Ann. Rheum. Dis. 2017, 35, 35–42. [Google Scholar]
- El-Rabbat, M.S.; Mahmoud, N.K.; Gheita, T.A. Clinical significance of fibromyalgia syndrome in different rheumatic diseases: Relation to disease activity and quality of life. Reumatol. Clin. 2018, 14, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Castro-Marrero, J.; Cordero, M.D.; Sáez-Francas, N.; Jimenez-Gutierrez, C.; Aguilar-Montilla, F.J.; Aliste, L.; Alegre-Martin, J. Could Mitochondrial Dysfunction Be a Differentiating Marker Between Chronic Fatigue Syndrome and Fibromyalgia. Antioxid. Redox Signal. 2013, 19, 1855–1860. [Google Scholar] [CrossRef] [PubMed]
- Missailidis, D.; Annesley, S.J.; Allan, C.Y.; Sanislav, O.; Lidbury, B.A.; Lewis, D.P.; Fisher, P.R. An Isolated Complex V Inefficiency and Dysregulated Mitochondrial Function in Immortalized Lymphocytes from ME/CFS Patients. Int. J. Mol. Sci. 2020, 21, 1074. [Google Scholar] [CrossRef]
- Cogliati, S.; Lorenzi, I.; Rigoni, G.; Caicci, F.; Soriano, M.E. Regulation of Mitochondrial Electron Transport Chain Assembly. J. Mol. Biol. 2018, 430, 4849–4873. [Google Scholar] [CrossRef]
- Lane, R.K.; Hilsabeck, T.; Rea, S.L. The role of mitochondrial dysfunction in age-related diseases. Biochim. Biophys. Acta 2015, 1847, 1387–1400. [Google Scholar] [CrossRef] [PubMed]
- Pandit, A.; Vadnal, J.; Houston, S.; Freeman, E.; McDonough, J. Impaired regulation of electron transport chain subunit genes by nuclear respiratory factor 2 in multiple sclerosis. J. Neurol. Sci. 2009, 279, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Yu, E.P.; Reinhold, J.; Yu, H.; Starks, L.; Uryga, A.K.; Foote, K.; Finigan, A.; Figg, N.; Pung, Y.-F.; Logan, A.; et al. Mitochondrial Respiration Is Reduced in Atherosclerosis, Promoting Necrotic Core Formation and Reducing Relative Fibrous Cap Thickness. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 2322–2332. [Google Scholar] [CrossRef]
- Chistiakov, D.A.; Sobenin, I.A.; Revin, V.V.; Orekhov, A.N.; Bobryshev, Y.V. Mitochondrial Aging and Age-Related Dysfunction of Mitochondria. BioMed. Res. Int. 2014, 2014, 238463. [Google Scholar] [CrossRef] [PubMed]
- Zahn, J.M.; Sonu, R.; Vogel, H.; Crane, E.; Mazan-Mamczarz, K.; Rabkin, R.; Davis, R.W.; Becker, K.G.; Owen, A.B.; Kim, S.K. Transcriptional Profiling of Aging in Human Muscle Reveals a Common Aging Signature. PLOS Genet. 2006, 2, e115. [Google Scholar] [CrossRef]
- Dromparis, P.; Michelakis, E.D. Mitochondria in Vascular Health and Disease. Annu. Rev. Physiol. 2013, 75, 95–126. [Google Scholar] [CrossRef]
- Li, X.; Zhang, W.; Cao, Q.; Wang, Z.; Zhao, M.; Xu, L.; Zhuang, Q. Mitochondrial dysfunction in fibrotic diseases. Cell Death Discov. 2020, 6, 80. [Google Scholar] [CrossRef]
- Ma, Z.; Mulder, D.J.; Gniadecki, R.; Tervaert, J.W.C.; Osman, M. Methods of Assessing Nailfold Capillaroscopy Compared to Video Capillaroscopy in Patients with Systemic Sclerosis—A Critical Review of the Literature. Diagnostics 2023, 13, 2204. [Google Scholar] [CrossRef]
- Fallahi, P.; Ruffilli, I.; Giuggioli, D.; Colaci, M.; Ferrari, S.M.; Antonelli, A.; Ferri, C. Associations between Systemic Sclerosis and Thyroid Diseases. Front. Endocrinol. 2017, 8, 266. [Google Scholar] [CrossRef]
- Singh, P.K.; Sharma, S.K.; Sinha, A.; Dutta, P. Can thyroid volume predict thyroid dysfunction in patients with systemic sclerosis? A prospective cross-sectional study from a tertiary care center in North West India. Clin. Rheumatol. 2016, 35, 765–769. [Google Scholar] [CrossRef]
- Fineschi, S.; Klar, J.; Gustafsson, K.A.; Jonsson, K.; Karlsson, B.; Dahl, N. Inflammation and Interferon Signatures in Peripheral B-Lymphocytes and Sera of Individuals with Fibromyalgia. Front. Immunol. 2022, 13, 874490. [Google Scholar] [CrossRef] [PubMed]
- Powell, F.C.; Winkelmann, R.K.; Venencie-Lemarchand, F.; Spurbeck, J.L.; Schroeter, A.L. The Anticentromere Antibody: Disease Specificity and Clinical Significance. Mayo Clin. Proc. 1984, 59, 700–706. [Google Scholar] [CrossRef]
- Igusa, T.; Hummers, L.K.; Visvanathan, K.; Richardson, C.; Wigley, F.M.; Casciola-Rosen, L.; Rosen, A.; A Shah, A. Autoantibodies and scleroderma phenotype define subgroups at high-risk and low-risk for cancer. Ann. Rheum. Dis. 2018, 77, 1179–1186. [Google Scholar] [CrossRef] [PubMed]
- Caetano, J.; I Nihtyanova, S.; Harvey, J.; Denton, C.P.; Ong, V.H. Distinctive clinical phenotype of anti-centromere antibody-positive diffuse systemic sclerosis. Rheumatol. Adv. Pract. 2018, 2, rky002. [Google Scholar] [CrossRef]
- Miao, Z.; Alvarez, M.; Ko, A.; Bhagat, Y.; Rahmani, E.; Jew, B.; Heinonen, S.; Muñoz-Hernandez, L.L.; Herrera-Hernandez, M.; Aguilar-Salinas, C.; et al. The causal effect of obesity on prediabetes and insulin resistance reveals the important role of adipose tissue in insulin resistance. PLOS Genet. 2020, 16, e1009018. [Google Scholar] [CrossRef] [PubMed]
- Distler, O.; Assassi, S.; Cottin, V.; Cutolo, M.; Danoff, S.K.; Denton, C.P.; Distler, J.H.; Hoffmann-Vold, A.-M.; Johnson, S.R.; Ladner, U.M.; et al. Predictors of progression in systemic sclerosis patients with interstitial lung disease. Eur. Respir. J. 2020, 55, 1902026. [Google Scholar] [CrossRef]
- Bueno, M.; Papazoglou, A.; Valenzi, E.; Rojas, M.; Lafyatis, R.; Mora, A.L. Mitochondria, Aging, and Cellular Senescence: Implications for Scleroderma. Curr. Rheumatol. Rep. 2020, 22, 37. [Google Scholar] [CrossRef]
- Sandusky, S.B.; McGuire, L.; Smith, M.T.; Wigley, F.M.; Haythornthwaite, J.A. Fatigue: An overlooked determinant of physical function in scleroderma. Rheumatology 2009, 48, 165–169. [Google Scholar] [CrossRef]
- Tomas, C.; Brown, A.; Strassheim, V.; Elson, J.L.; Newton, J.; Manning, P. Cellular bioenergetics is impaired in patients with chronic fatigue syndrome. PLoS ONE 2017, 12, e0186802. [Google Scholar] [CrossRef]
- Van den Hoogen, F.; Khanna, D.; Fransen, J.; Johnson, S.R.; Baron, M.; Tyndall, A.; Matucci-Cerinic, M.; Naden, R.P.; Medsger, T.A., Jr.; Carreira, P.E.; et al. 2013 classification criteria for systemic sclerosis: An American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheum. 2013, 65, 2737–2747. [Google Scholar] [CrossRef]
- Koenig, M.; Joyal, F.; Fritzler, M.J.; Roussin, A.; Abrahamowicz, M.; Boire, G.; Goulet, J.-R.; Rich, E.; Grodzicky, T.; Raymond, Y.; et al. Autoantibodies and microvascular damage are independent predictive factors for the progression of Raynaud’s phenomenon to systemic sclerosis: A twenty-year prospective study of 586 patients, with validation of proposed criteria for early systemic sclerosis. Arthritis Rheum. 2008, 58, 3902–3912. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.; van Eeden, C.; Moazab, N.; Redmond, D.; Phan, C.; Keeling, S.; Gniadecki, R.; Tervaert, J.W.C.; Osman, M. Nailfold Capillaroscopy Abnormalities Correlate with Disease Activity in Adult Dermatomyositis. Front. Med. 2021, 8, 708432. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, F.; Clauw, D.J.; Fitzcharles, M.A.; Goldenberg, D.L.; Häuser, W.; Katz, R.L.; Mease, P.J.; Russel, A.S.; Russell, I.J.; Walitt, R. 2016 Revisions to the 2010/2011 fibromyalgia diagnostic criteria. Semin. Arthritis Rheum. 2016, 46, 319–329. [Google Scholar] [CrossRef]
- Wolfe, F.; Clauw, D.J.; Fitzcharles, M.-A.; Goldenberg, D.L.; Katz, R.S.; Mease, P.; Russell, A.S.; Russell, I.J.; Winfield, J.B.; Yunus, M.B. The American College of Rheumatology Preliminary Diagnostic Criteria for Fibromyalgia and Measurement of Symptom Severity. Arthritis Care Res. 2010, 62, 600–610. [Google Scholar] [CrossRef]
- Clayton, E.W. Beyond myalgic encephalomyelitis/chronic fatigue syndrome: An IOM report on redefining an illness. JAMA 2015, 313, 1101–1102. [Google Scholar] [CrossRef] [PubMed]
- Carruthers, B.M.; Van De Sande, M.I.; De Meirleir, K.L.; Klimas, N.G.; Broderick, G.; Mitchell, T.; Staines, D.; Powles, A.C.; Speight, N.; Vallings, R.; et al. Myalgic encephalomyelitis: International Consensus Criteria. J. Intern. Med. 2011, 270, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Van Eeden, C.; Mohazab, N.; Redmond, D.; Yacyshyn, E.; Clifford, A.; Russell, A.S.; Osman, M.S.; Tervaert, J.W.C. Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and fibromyalgia: PR3-versus MPO-ANCA-associated vasculitis, an exploratory cross-sectional study. Lancet Reg. Health Am. 2023, 20, 100460. [Google Scholar]
- Ellinger, J.; Müller, S.C.; Wernert, N.; von Ruecker, A.; Bastian, P.J. Mitochondrial DNA in serum of patients with prostate cancer: A predictor of biochemical recurrence after prostatectomy. BJU Int. 2008, 102, 628–632. [Google Scholar] [CrossRef]
- Meng, X.; Schwarzenbach, H.; Yang, Y.; Müller, V.; Li, N.; Tian, D.; Shen, Y.; Gong, Z. Circulating Mitochondrial DNA is Linked to Progression and Prognosis of Epithelial Ovarian Cancer. Transl. Oncol. 2019, 12, 1213–1220. [Google Scholar] [CrossRef]
- Bedree, H.; Sunnquist, M.; Jason, L.A. The DePaul Symptom Questionnaire-2: A validation study. Fatigue Biomed. Heal. Behav. 2019, 7, 166–179. [Google Scholar] [CrossRef]
- Smets, E.M.A.; Garssen, B.; Bonke, B.; De Haes, J.C.J.M. The multidimensional fatigue inventory (MFI) psychometric qualities of an instrument to assess fatigue. J. Psychosom. Res. 1995, 39, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Yellen, S.B.; Cella, D.F.; Webster, K.; Blendowski, C.; Kaplan, E. Measuring fatigue and other anemia-related symptoms with the Functional Assessment of Cancer Therapy (FACT) measurement system. J. Pain Symptom Manag. 1997, 13, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Ware, J.E., Jr.; Sherbourne, C.D. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med. Care 1992, 30, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, C. International experiences with the Hospital Anxiety and Depression Scale-a review of validation data and clinical results. J. Psychosom. Res. 1997, 42, 17–41. [Google Scholar] [CrossRef]
- Buysse, D.J.; Reynolds, C.F., III; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Broadbent, D.E.; Cooper, P.F.; FitzGerald, P.; Parkes, K.R. The Cognitive Failures Questionnaire (CFQ) and its correlates. Br. J. Clin. Psychol. 1982, 21, 1–16. [Google Scholar] [CrossRef]

| SSc-CFS (n = 12) | SSc-NCFS(n = 12) | p | Healthy (n = 10) | p | |
|---|---|---|---|---|---|
| Continuous variables | Median (IQR) | Median (IQR) | Median (IQR) | ||
| Age | 47.5 (46; 57) | 53 (35.5; 63) | 0.920 | 46 (36; 51) | 0.389 |
| BMI | 30.4 (25.3; 36.0) | 27.8 (22.9; 32.9) | 0.178 | 22.5 (21.4; 34.2) | 0.095 |
| CRP (mg/L) | 3.5 (2.3; 6.5) | 2.7 (0.9; 7.9) | 0.579 | 0.6 (0.5; 4.2) | 0.146 |
| TSH (mU/L) | 2.63 (1.8; 3.4) | 1.54 (0.83; 2.15) | 0.026 | 1.29 (0.81; 1.47) | 0.009 |
| Lung function | |||||
| DLCO SB (ml/(min × mmHg)) | 17.2 (14.2; 22.7) | 19.5 (16.8; 21.5) | 0.265 | - | - |
| FEV (L) | 2.55 (2.0; 2.8) | 2.56 (2.1; 3.1) | 0.580 | - | - |
| FVC (L) | 3.18 (2.6; 3.8) | 3.32 (3.0; 3.8) | 0.580 | - | - |
| mRSS | 0 (0; 1.5) | 0 (0; 0) | 0.931 | - | - |
| SF-36 PCS | 31.25 (23.7; 44.0) | 84.6 (80.3; 91.2) | <0.001 | 96.8 (93.7; 97.5) | <0.001 |
| SF-36 MCS | 61.25 (35.7; 70.8) | 88.5 (70.6; 92.5) | 0.001 | 90.1 (88.2; 92 | <0.001 |
| Cognitive Failure | 42.5 (18.5; 51) | 22 (16; 31) | 0.061 | 26.5 (12; 38) | 0.185 |
| MFI Score | 67.5 (60; 73.5.) | 39 (31; 43) | <0.001 | 28.5 (26; 34) | <0.001 |
| FACIT Score | 24 (20.2; 29.5) | 48 (44; 49.8) | <0.001 | 50 (50; 52) | <0.001 |
| Categorical variables | Yes (%) | Yes (%) | Yes (%) | ||
| Gender (female) | 11/12 (91.6) | 10/12 (83.3) | 0.537 | 1/10 (10.0) | 0.892 |
| Autoantibodies | |||||
| ANA | 10/12 (83.3) | 11/12 (91.6) | 0.537 | - | - |
| SSc-specific autoantibodies | 9/12 (75.0) | 10/12 (83.3) | 0.615 | - | - |
| ACA | 2/12 (16.6) | 6/12 (50.0) | 0.083 | - | - |
| Medications | |||||
| Immunomodulators | 3/12 (25.0) | 2/12 (16.6) | |||
| Vasodilators | 3/12 (25.0) | 5/12 (41.6) | |||
| Immunomodulators and vasodilators | 5/12 (41.6) | 3/12 (25.0) | |||
| No immunomodulators or vasodilators | 1/12 (8.33) | 2/12 (16.6) | 0.675 | ||
| ILD | 2/12 (16.6) | 2/12 (16.6) | 1.000 | - | - |
| Digital ulcers | 2/12 (16.6) | 4/12 (33.3) | 0.346 | - | - |
| Arthralgia | 7/12 (58.3) | 2/12 (16.6) | 0.035 | - | - |
| GERD | 5/12 (41.6) | 2/12 (16.6) | 0.178 | - | - |
| Sleep disturbances | 8/11 (72.7) | 5/12 (41.6) | 0.133 | 2/10 (20.0) | 0.016 |
| Fibromyalgia | 0/12 (0.0) | 8/12 (66.6) | 0.001 | 0/10 (0.0) | 0.001 |
| HADS depression | 0/12 (0.0) | 2/12 (16.6) | 0.140 | 0/10 (0.0) | 0.176 |
| HADS anxiety | 0/12 (0.0) | 4/12 (33.3) | 0.028 | 0/10 (0.0) | 0.044 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
van Eeden, C.; Redmond, D.; Mohazab, N.; Larché, M.J.; Mason, A.L.; Cohen Tervaert, J.W.; Osman, M.S. Evidence of a Novel Mitochondrial Signature in Systemic Sclerosis Patients with Chronic Fatigue Syndrome. Int. J. Mol. Sci. 2023, 24, 12057. https://doi.org/10.3390/ijms241512057
van Eeden C, Redmond D, Mohazab N, Larché MJ, Mason AL, Cohen Tervaert JW, Osman MS. Evidence of a Novel Mitochondrial Signature in Systemic Sclerosis Patients with Chronic Fatigue Syndrome. International Journal of Molecular Sciences. 2023; 24(15):12057. https://doi.org/10.3390/ijms241512057
Chicago/Turabian Stylevan Eeden, Charmaine, Desiree Redmond, Naima Mohazab, Maggie J. Larché, Andrew L. Mason, Jan Willem Cohen Tervaert, and Mohammed S. Osman. 2023. "Evidence of a Novel Mitochondrial Signature in Systemic Sclerosis Patients with Chronic Fatigue Syndrome" International Journal of Molecular Sciences 24, no. 15: 12057. https://doi.org/10.3390/ijms241512057
APA Stylevan Eeden, C., Redmond, D., Mohazab, N., Larché, M. J., Mason, A. L., Cohen Tervaert, J. W., & Osman, M. S. (2023). Evidence of a Novel Mitochondrial Signature in Systemic Sclerosis Patients with Chronic Fatigue Syndrome. International Journal of Molecular Sciences, 24(15), 12057. https://doi.org/10.3390/ijms241512057







