Multiple Roles of LOXL2 in the Progression of Hepatocellular Carcinoma and Its Potential for Therapeutic Targeting
Abstract
1. Introduction
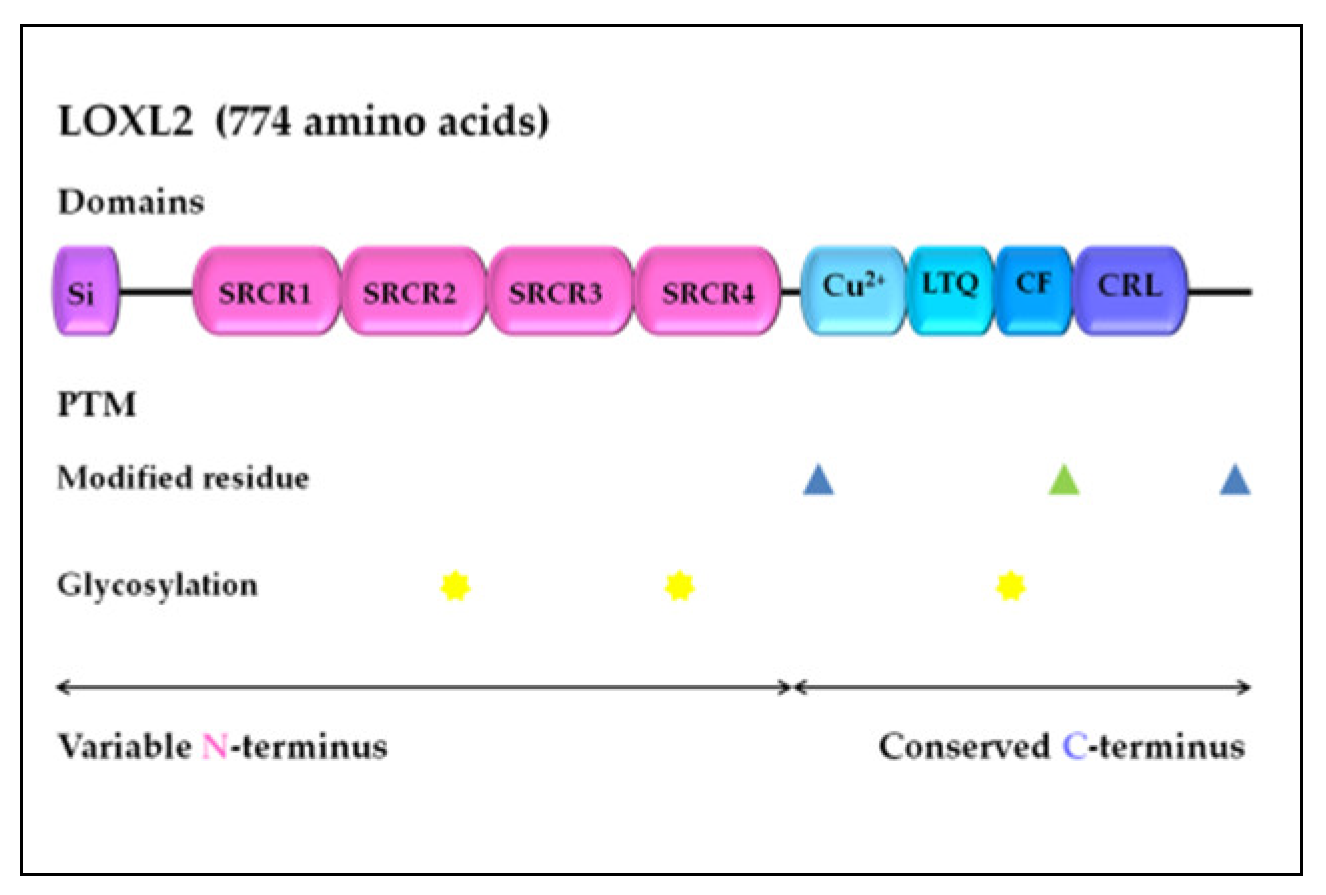
2. LOXL2 Introduction: LOX Family, Structure, and LOXL2 Function
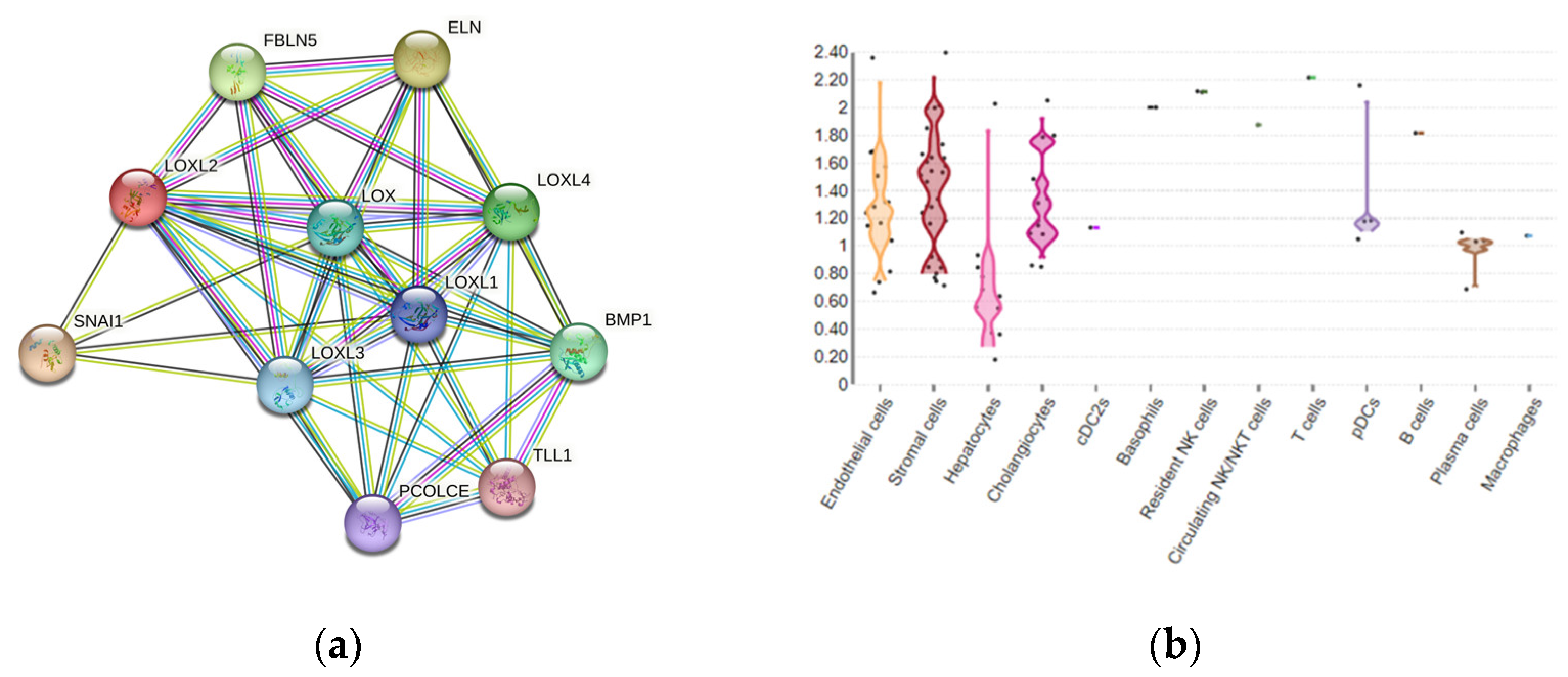
3. LOXL2 Expression in HCC and Correlation with Clinical Parameters
4. LOXL2 in the Regulation of Tumor Microenvironment and Formation of Premetastatic Niches
4.1. LOXL2 and Cancer-Associated Fibroblasts (CAFs)
4.2. LOXL2 and Tumor-Associated Macrophages (TAMs)
4.3. LOXL2 in the Formation of Premetastatic Niches (PNM)
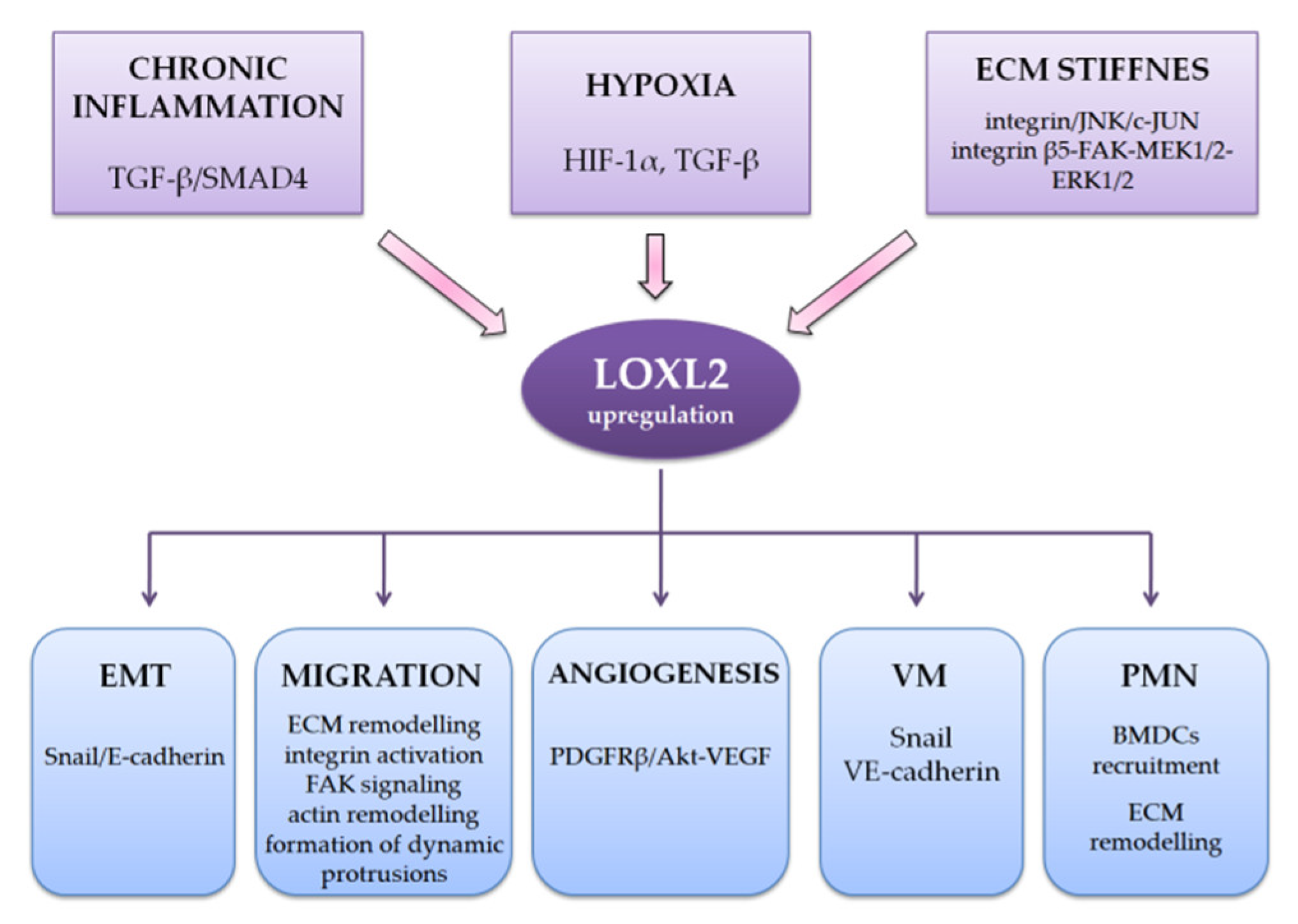
5. LOXL2 Role in Epithelial–Mesenchymal Transition
6. LOXL2 and Hypoxia, Angiogenesis, and Vasculogenic Mimicry
7. LOXL2 and micro-RNAs in HCC
8. LOXL2 as Potential Target for Treatment of HCC
| Type | Agent | Target | References |
|---|---|---|---|
| monoclonal antibody | AB0023 | LOXL2 | [46,121,122] |
| AB0024 | LOXL2 | [123,124,125] | |
| small-molecule inhibitor | BAPN | LOX/LOXL1-4 | [100,133,134] |
| LOXL2-IN-1 | LOXL2 | [122] | |
| PXS-5338 | LOXL2 | [156] | |
| PXS-5382 | LOXL2 | [156] | |
| PXS-5878 | LOXL2 | [156] | |
| PXS-5153A | LOXL2/LOXL3 | [157] | |
| (2-chloropyridin-4-yl) methenamine | LOXL2 | [158] |
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| HCC | Hepatocellular carcinoma; |
| LOXL2 | Lysyloxidase-like 2; |
| HBV | Hepatitis B virus; |
| HCV | Hepatitis C virus; |
| MASLD | Metabolic dysfunction-associated steatotic liver disease; |
| TACE | Transarterialchemoembolization; |
| VEGF | Vascular endothelial growth factor; |
| TME | Tumor microenvironment; |
| CAF | Cancer-associated fibroblasts; |
| ECM | Extracellular matrix; |
| LTQ | Lysine tyrosylquinone; |
| ELN | Elastin; |
| FBLN5 | Fibulin-5; |
| PCOLCE | Procollagen C-endopeptidase enhancer 1; |
| TLL1 | Tolloid-like protein 1; |
| BMP | Bone morphogenic protein 1; |
| CAIX | Carbon anhydrase IX; |
| HSC | Hepatic stellate cells; |
| BMDC | Bone marrow-derived cells; |
| TGF-ß | Transforming growth factor-beta; |
| HIF-1α | Hypoxia-inducible factor 1 alpha; |
| JNK-c | Jun N-terminal kinase; |
| 5FU | 5-fluorouracil; |
| FAK | Focaladhesion kinase; |
| ROCK | Rho-associated protein kinase; |
| CCL5 | Chemokine ligand 5; |
| ZEB1 | Zinc finger E-box-binding homeobox 1; |
| MEK1/2 | Mitogen-activated protein kinase 1/2; |
| ERK 1/2 | Extracellular signal-regulated kinase 1/2; |
| MMP | Matrix metalloproteinase |
| HRE | Hypoxia responsive element; |
| FBP1 | Fructose-1,6-biphosphatase protein 1; |
| PDGFRß | Platelet-derived growth factor receptor beta; |
| miR | micro-RNA; |
| BAPN | ß-aminopropionitrile. |
References
- Marrero, J.A.; Kulik, L.M.; Sirlin, C.B.; Zhu, A.X.; Finn, R.S.; Abecassis, M.M.; Roberts, L.R.; Heimbach, J.K. Diagnosis, Staging, and Management of Hepatocellular Carcinoma: 2018 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology 2018, 68, 723–750. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CACancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Yang, J.D.; Hainaut, P.; Gores, G.J.; Amadou, A.; Plymoth, A.; Roberts, L.R. A Global View of Hepatocellular Carcinoma: Trends, Risk, Prevention and Management. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 589–604. [Google Scholar] [CrossRef] [PubMed]
- Affo, S.; Yu, L.-X.; Schwabe, R.F. The Role of Cancer-Associated Fibroblasts and Fibrosis in Liver Cancer. Annu. Rev. Pathol. 2017, 12, 153–186. [Google Scholar] [CrossRef] [PubMed]
- Sangiovanni, A.; Prati, G.M.; Fasani, P.; Ronchi, G.; Romeo, R.; Manini, M.; Del Ninno, E.; Morabito, A.; Colombo, M. The Natural History of Compensated Cirrhosis Due to Hepatitis C Virus: A 17-Year Cohort Study of 214 Patients. Hepatology 2006, 43, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Dhanasekaran, R.; Bandoh, S.; Roberts, L.R. Molecular Pathogenesis of Hepatocellular Carcinoma and Impact of Therapeutic Advances. F1000Research 2016, 5, 879. [Google Scholar] [CrossRef]
- Wege, H.; Li, J.; Ittrich, H. Treatment Lines in Hepatocellular Carcinoma. Visc. Med. 2019, 35, 266–272. [Google Scholar] [CrossRef]
- Suresh, D.; Srinivas, A.N.; Prashant, A.; Harikumar, K.B.; Kumar, D.P. Therapeutic Options in Hepatocellular Carcinoma: A Comprehensive Review. Clin. Exp. Med. 2023. [Google Scholar] [CrossRef]
- Altekruse, S.F.; McGlynn, K.A.; Reichman, M.E. Hepatocellular Carcinoma Incidence, Mortality, and Survival Trends in the United States from 1975 to 2005. J. Clin. Oncol. 2009, 27, 1485–1491. [Google Scholar] [CrossRef]
- Bao, M.H.-R.; Wong, C.C.-L. Hypoxia, Metabolic Reprogramming, and Drug Resistance in Liver Cancer. Cells 2021, 10, 1715. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.-F.; De Oliveira, A.C.; Santoro, A.; Raoul, J.-L.; Forner, A.; et al. Sorafenib in Advanced Hepatocellular Carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef]
- Cheng, A.-L.; Kang, Y.-K.; Chen, Z.; Tsao, C.-J.; Qin, S.; Kim, J.S.; Luo, R.; Feng, J.; Ye, S.; Yang, T.-S.; et al. Efficacy and Safety of Sorafenib in Patients in the Asia-Pacific Region with Advanced Hepatocellular Carcinoma: A Phase III Randomised, Double-Blind, Placebo-Controlled Trial. Lancet Oncol. 2009, 10, 25–34. [Google Scholar] [CrossRef]
- Llovet, J.M.; Kelley, R.K.; Villanueva, A.; Singal, A.G.; Pikarsky, E.; Roayaie, S.; Lencioni, R.; Koike, K.; Zucman-Rossi, J.; Finn, R.S. Hepatocellular Carcinoma. Nat. Rev. Dis. Primer 2021, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Novikova, M.V.; Khromova, N.V.; Kopnin, P.B. Components of the Hepatocellular Carcinoma Microenvironment and Their Role in Tumor Progression. Biochem. Biokhimiia 2017, 82, 861–873. [Google Scholar] [CrossRef] [PubMed]
- Gong, L.; Zhang, Y.; Yang, Y.; Yan, Q.; Ren, J.; Luo, J.; Tiu, Y.C.; Fang, X.; Liu, B.; Lam, R.H.W.; et al. Inhibition of Lysyl Oxidase-like 2 Overcomes Adhesion-dependent Drug Resistance in the Collagen-enriched Liver Cancer Microenvironment. Hepatol. Commun. 2022, 6, 3194–3211. [Google Scholar] [CrossRef] [PubMed]
- Hernandez–Gea, V.; Toffanin, S.; Friedman, S.L.; Llovet, J.M. Role of the Microenvironment in the Pathogenesis and Treatment of Hepatocellular Carcinoma. Gastroenterology 2013, 144, 512–527. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.C.-L.; Tse, A.P.-W.; Huang, Y.-P.; Zhu, Y.-T.; Chiu, D.K.-C.; Lai, R.K.-H.; Au, S.L.-K.; Kai, A.K.-L.; Lee, J.M.-F.; Wei, L.L.; et al. Lysyl Oxidase-like 2 Is Critical to Tumor Microenvironment and Metastatic Niche Formation in Hepatocellular Carcinoma. Hepatology 2014, 60, 1645–1658. [Google Scholar] [CrossRef]
- Payne, S.L.; Hendrix, M.J.C.; Kirschmann, D.A. Paradoxical Roles for Lysyl Oxidases in Cancer—A Prospect. J. Cell. Biochem. 2007, 101, 1338–1354. [Google Scholar] [CrossRef]
- Zhan, X.; Jiao, J.; Zhang, H.; Li, C.; Zhao, J.; Liao, L.; Wu, J.; Wu, B.; Wu, Z.; Wang, S.; et al. A Three-Gene Signature from Protein-Protein Interaction Network of LOXL2- and Actin-Related Proteins for Esophageal Squamous Cell Carcinoma Prognosis. Cancer Med. 2017, 6, 1707–1719. [Google Scholar] [CrossRef]
- Moreno-Bueno, G.; Salvador, F.; Martín, A.; Floristán, A.; Cuevas, E.P.; Santos, V.; Montes, A.; Morales, S.; Castilla, M.A.; Rojo-Sebastián, A.; et al. Lysyl Oxidase-like 2 (LOXL2), a New Regulator of Cell Polarity Required for Metastatic Dissemination of Basal-like Breast Carcinomas. EMBO Mol. Med. 2011, 3, 528–544. [Google Scholar] [CrossRef]
- Park, J.S.; Lee, J.; Lee, Y.S.; Kim, J.K.; Dong, S.M.; Yoon, D.S. Emerging Role of LOXL2 in the Promotion of Pancreas Cancer Metastasis. Oncotarget 2016, 7, 42539–42552. [Google Scholar] [CrossRef]
- Torres, S.; Garcia-Palmero, I.; Herrera, M.; Bartolomé, R.A.; Peña, C.; Fernandez-Aceñero, M.J.; Padilla, G.; Peláez-García, A.; Lopez-Lucendo, M.; Rodriguez-Merlo, R.; et al. LOXL2 Is Highly Expressed in Cancer-Associated Fibroblasts and Associates to Poor Colon Cancer Survival. Clin. Cancer Res. 2015, 21, 4892–4902. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, W.; Xu, J. Prognostic Utility and Clinical Significance of Lysyl Oxidase-like 2 Protein Expression in Digestive System Cancers. J. Cell. Physiol. 2019, 234, 20713–20720. [Google Scholar] [CrossRef]
- Lin, H.-Y.; Li, C.-J.; Yang, Y.-L.; Huang, Y.-H.; Hsiau, Y.-T.; Chu, P.-Y. Roles of Lysyl Oxidase Family Members in the Tumor Microenvironment and Progression of Liver Cancer. Int. J. Mol. Sci. 2020, 21, 9751. [Google Scholar] [CrossRef] [PubMed]
- Wen, B.; Xu, L.-Y.; Li, E.-M. LOXL2 in Cancer: Regulation, Downstream Effectors and Novel Roles. Biochim. Biophys. Acta BBA-Rev. Cancer 2020, 1874, 188435. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Zhu, Y. The Function and Mechanisms of Action of LOXL2 in Cancer (Review). Int. J. Mol. Med. 2015, 36, 1200–1204. [Google Scholar] [CrossRef] [PubMed]
- Peng, T.; Deng, X.; Tian, F.; Li, Z.; Jiang, P.; Zhao, X.; Chen, G.; Chen, Y.; Zheng, P.; Li, D.; et al. The Interaction of LOXL2 with GATA6 Induces VEGFA Expression and Angiogenesis in Cholangiocarcinoma. Int. J. Oncol. 2019, 55, 657–670. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Xu, S.; Tian, Y.; Ju, A.; Hou, Q.; Liu, J.; Fu, Y.; Luo, Y. Lysyl Oxidase-Like Protein 2 Promotes Tumor Lymphangiogenesis and Lymph Node Metastasis in Breast Cancer. Neoplasia 2019, 21, 413–427. [Google Scholar] [CrossRef]
- Shao, B.; Zhao, X.; Liu, T.; Zhang, Y.; Sun, R.; Dong, X.; Liu, F.; Zhao, N.; Zhang, D.; Wu, L.; et al. LOXL2 Promotes Vasculogenic Mimicry and Tumour Aggressiveness in Hepatocellular Carcinoma. J. Cell. Mol. Med. 2019, 23, 1363–1374. [Google Scholar] [CrossRef]
- Barker, H.E.; Bird, D.; Lang, G.; Erler, J.T. Tumor-Secreted LOXL2 Activates Fibroblasts through FAK Signaling. Mol. Cancer Res. 2013, 11, 1425–1436. [Google Scholar] [CrossRef]
- Xing, X.; Wang, Y.; Zhang, X.; Gao, X.; Li, M.; Wu, S.; Zhao, Y.; Chen, J.; Gao, D.; Chen, R.; et al. Matrix Stiffness-mediated Effects on Macrophages Polarization and Their LOXL2 Expression. FEBS J. 2021, 288, 3465–3477. [Google Scholar] [CrossRef] [PubMed]
- Mäki, J.M.; Kivirikko, K.I. Cloning and Characterization of a Fourth Human Lysyl Oxidase Isoenzyme. Biochem. J. 2001, 355, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Molnar, J.; Fong, K.S.K.; He, Q.P.; Hayashi, K.; Kim, Y.; Fong, S.F.T.; Fogelgren, B.; MolnarneSzauter, K.; Mink, M.; Csiszar, K. Structural and Functional Diversity of Lysyl Oxidase and the LOX-like Proteins. Biochim. Biophys. Acta BBA-Proteins Proteom. 2003, 1647, 220–224. [Google Scholar] [CrossRef]
- Kenyon, K.; Modi, W.S.; Contente, S.; Friedman, R.M. A Novel Human CDNA with a Predicted Protein Similar to Lysyl Oxidase Maps to Chromosome 15q24-Q25. J. Biol. Chem. 1993, 268, 18435–18437. [Google Scholar] [CrossRef]
- Kim, Y.; Boyd, C.D.; Csiszar, K. A New Gene with Sequence and Structural Similarity to the Gene Encoding Human Lysyl Oxidase. J. Biol. Chem. 1995, 270, 7176–7182. [Google Scholar] [CrossRef] [PubMed]
- Saito, H.; Papaconstantinou, J.; Sato, H.; Goldstein, S. Regulation of a Novel Gene Encoding a Lysyl Oxidase-Related Protein in Cellular Adhesion and Senescence. J. Biol. Chem. 1997, 272, 8157–8160. [Google Scholar] [CrossRef]
- Huang, Y. Cloning and Characterization of a Human Lysyl Oxidase-like 3 Gene (HLOXL3). Matrix Biol. 2001, 20, 153–157. [Google Scholar] [CrossRef]
- Siegel, R.C.; Pinnell, S.R.; Martin, G.R. Cross-Linking of Collagen and Elastin. Properties of Lysyl Oxidase. Biochemistry 1970, 9, 4486–4492. [Google Scholar] [CrossRef]
- Pinnell, S.R.; Martin, G.R. The Cross-Linking of Collagen and Elastin: Enzymatic Conversion of Lysine in Peptide Linkage to Alpha-Aminoadipic-Delta-Semialdehyde (Allysine) by an Extract from Bone. Proc. Natl. Acad. Sci. USA 1968, 61, 708–716. [Google Scholar] [CrossRef]
- Jourdan-Le Saux, C.; Le Saux, O.; Donlon, T.; Boyd, C.D.; Csiszar, K. The Human Lysyl Oxidase-Related Gene (LOXL2) Maps between Markers D8S280 and D8S278 on Chromosome 8p21.2–P21.3. Genomics 1998, 51, 305–307. [Google Scholar] [CrossRef]
- Csiszar, K. Lysyl Oxidases: A Novel Multifunctional Amine Oxidase Family. In Progress in Nucleic Acid Research and Molecular Biology; Elsevier: Amsterdam, The Netherlands, 2001; Volume 70, pp. 1–32. ISBN 978-0-12-540070-1. [Google Scholar]
- Lucero, H.A.; Kagan, H.M. Lysyl Oxidase: An Oxidative Enzyme and Effector of Cell Function. Cell. Mol. Life Sci. 2006, 63, 2304–2316. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Ge, G. Lysyl Oxidase, Extracellular Matrix Remodeling and Cancer Metastasis. Cancer Microenviron. 2012, 5, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Meier, A.A.; Kuczera, K.; Mure, M. A 3D–Predicted Structure of the Amine Oxidase Domain of Lysyl Oxidase–Like 2. Int. J. Mol. Sci. 2022, 23, 13385. [Google Scholar] [CrossRef] [PubMed]
- Boufraqech, M.; Zhang, L.; Nilubol, N.; Sadowski, S.M.; Kotian, S.; Quezado, M.; Kebebew, E. Lysyl Oxidase (LOX) Transcriptionally Regulates SNAI2 Expression and TIMP4 Secretion in Human Cancers. Clin. Cancer Res. 2016, 22, 4491–4504. [Google Scholar] [CrossRef]
- Hornstra, I.K.; Birge, S.; Starcher, B.; Bailey, A.J.; Mecham, R.P.; Shapiro, S.D. Lysyl Oxidase Is Required for Vascular and Diaphragmatic Development in Mice. J. Biol. Chem. 2003, 278, 14387–14393. [Google Scholar] [CrossRef]
- Dongiovanni, P.; Meroni, M.; Baselli, G.A.; Bassani, G.A.; Rametta, R.; Pietrelli, A.; Maggioni, M.; Facciotti, F.; Trunzo, V.; Badiali, S.; et al. Insulin Resistance Promotes Lysyl Oxidase Like 2 Induction and Fibrosis Accumulation in Non-Alcoholic Fatty Liver Disease. Clin. Sci. 2017, 131, 1301–1315. [Google Scholar] [CrossRef]
- Zhao, W.; Yang, A.; Chen, W.; Wang, P.; Liu, T.; Cong, M.; Xu, A.; Yan, X.; Jia, J.; You, H. Inhibition of Lysyl Oxidase-like 1 (LOXL1) Expression Arrests Liver Fibrosis Progression in Cirrhosis by Reducing Elastin Crosslinking. Biochim. Biophys. Acta BBA-Mol. Basis Dis. 2018, 1864, 1129–1137. [Google Scholar] [CrossRef]
- Ikenaga, N.; Peng, Z.-W.; Vaid, K.A.; Liu, S.B.; Yoshida, S.; Sverdlov, D.Y.; Mikels-Vigdal, A.; Smith, V.; Schuppan, D.; Popov, Y.V. Selective Targeting of Lysyl Oxidase-like 2 (LOXL2) Suppresses Hepatic Fibrosis Progression and Accelerates Its Reversal. Gut 2017, 66, 1697–1708. [Google Scholar] [CrossRef]
- Guilliams, M.; Bonnardel, J.; Haest, B.; Vanderborght, B.; Wagner, C.; Remmerie, A.; Bujko, A.; Martens, L.; Thoné, T.; Browaeys, R.; et al. Spatial Proteogenomics Reveals Distinct and Evolutionarily Conserved Hepatic Macrophage Niches. Cell 2022, 185, 379–396.e38. [Google Scholar] [CrossRef]
- Human All Liver Cells. Available online: https://www.livercellatlas.org/umap-humanAll.php (accessed on 10 June 2023).
- String. Available online: https://string-db.org/cgi/network?taskId=b4sTPzQtSCz5&sessionId=bljZKLXOKXys (accessed on 10 June 2023).
- Wang, T.-H.; Hsia, S.-M.; Shieh, T.-M. Lysyl Oxidase and the Tumor Microenvironment. Int. J. Mol. Sci. 2016, 18, 62. [Google Scholar] [CrossRef]
- Xu, L.; Go, E.P.; Finney, J.; Moon, H.; Lantz, M.; Rebecchi, K.; Desaire, H.; Mure, M. Post-Translational Modifications of Recombinant Human Lysyl Oxidase-like 2 (RhLOXL2) Secreted from Drosophila S2 Cells. J. Biol. Chem. 2013, 288, 5357–5363. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Chang, R.; Zhong, J.; Pandey, A.; Semenza, G.L. Histone Demethylase JMJD2C Is a Coactivator for Hypoxia-Inducible Factor 1 That Is Required for Breast Cancer Progression. Proc. Natl. Acad. Sci. USA 2012, 109, E3367–E3376. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhu, M.-X.; Zhang, X.-D.; Xu, X.-E.; Wu, Z.-Y.; Liao, L.-D.; Li, L.-Y.; Xie, Y.-M.; Wu, J.-Y.; Zou, H.-Y.; et al. SMYD3 Stimulates EZR and LOXL2 Transcription to Enhance Proliferation, Migration, and Invasion in Esophageal Squamous Cell Carcinoma. Hum. Pathol. 2016, 52, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Guo, T.; Sakai, A.; Ren, S.; Fukusumi, T.; Ando, M.; Sadat, S.; Saito, Y.; Califano, J.A. A Novel Splice Variant of LOXL2 Promotes Progression of Human Papillomavirus–Negative Head and Neck Squamous Cell Carcinoma. Cancer 2020, 126, 737–748. [Google Scholar] [CrossRef]
- Lv, G.-Q.; Zou, H.-Y.; Liao, L.-D.; Cao, H.-H.; Zeng, F.-M.; Wu, B.-L.; Xie, J.-J.; Fang, W.-K.; Xu, L.-Y.; Li, E.-M. Identification of a Novel Lysyl Oxidase-like 2 Alternative Splicing Isoform, LOXL2 Δe13, in Esophageal Squamous Cell Carcinoma. Biochem. Cell Biol. 2014, 92, 379–389. [Google Scholar] [CrossRef]
- Qu, Y.; Xiao, H.; Xiao, W.; Xiong, Z.; Hu, W.; Gao, Y.; Ru, Z.; Wang, C.; Bao, L.; Wang, K.; et al. Upregulation of MIAT Regulates LOXL2 Expression by Competitively Binding MiR-29c in Clear Cell Renal Cell Carcinoma. Cell. Physiol. Biochem. 2018, 48, 1075–1087. [Google Scholar] [CrossRef]
- Fukumoto, I.; Kikkawa, N.; Matsushita, R.; Kato, M.; Kurozumi, A.; Nishikawa, R.; Goto, Y.; Koshizuka, K.; Hanazawa, T.; Enokida, H.; et al. Tumor-Suppressive MicroRNAs (MiR-26a/b, MiR-29a/b/c and MiR-218) Concertedly Suppressed Metastasis-Promoting LOXL2 in Head and Neck Squamous Cell Carcinoma. J. Hum. Genet. 2016, 61, 109–118. [Google Scholar] [CrossRef]
- Ye, M.; Zhang, J.; Guo, T.; Pan, X. MiR-504 Inhibits Cell Proliferation and Invasion by Targeting LOXL2 in Non Small Cell Lung Cancer. Biomed. Pharmacother. 2018, 97, 1289–1295. [Google Scholar] [CrossRef]
- Reily, C.; Stewart, T.J.; Renfrow, M.B.; Novak, J. Glycosylation in Health and Disease. Nat. Rev. Nephrol. 2019, 15, 346–366. [Google Scholar] [CrossRef]
- Go, E.P.; Moon, H.-J.; Mure, M.; Desaire, H. Recombinant Human Lysyl Oxidase-like 2 Secreted from Human Embryonic Kidney Cells Displays Complex and Acidic Glycans at All Three N-Linked Glycosylation Sites. J. Proteome Res. 2018, 17, 1826–1832. [Google Scholar] [CrossRef]
- Wang, W.; Wang, X.; Yao, F.; Huang, C. Lysyl Oxidase Family Proteins: Prospective Therapeutic Targets in Cancer. Int. J. Mol. Sci. 2022, 23, 12270. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, S.; Saraiva, N.; Rijo, P.; Fernandes, A.S. LOXL2 Inhibitors and Breast Cancer Progression. Antioxidants 2021, 10, 312. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.-J.; Finney, J.; Xu, L.; Moore, D.; Welch, D.R.; Mure, M. MCF-7 Cells Expressing Nuclear Associated Lysyl Oxidase-like 2 (LOXL2) Exhibit an Epithelial-to-Mesenchymal Transition (EMT) Phenotype and Are Highly Invasive in Vitro. J. Biol. Chem. 2013, 288, 30000–30008. [Google Scholar] [CrossRef]
- Wang, M.; Zhao, X.; Zhu, D.; Liu, T.; Liang, X.; Liu, F.; Zhang, Y.; Dong, X.; Sun, B. HIF-1α Promoted Vasculogenic Mimicry Formation in Hepatocellular Carcinoma through LOXL2 up-Regulation in Hypoxic Tumor Microenvironment. J. Exp. Clin. Cancer Res. 2017, 36, 60. [Google Scholar] [CrossRef]
- Zhao, N.; Chen, C.; Guo, Y.; Liu, T.; Che, N.; Zhang, D.; Liang, X.; Zhang, Y.; Zhao, X. LOXL2 Serves as a Prognostic Biomarker for Hepatocellular Carcinoma by Mediating Immune Infiltration and Vasculogenic Mimicry. Dig. Liver Dis. 2023, 55, 661–672. [Google Scholar] [CrossRef]
- Yang, Y.-L.; Tsai, M.-C.; Chang, Y.-H.; Wang, C.-C.; Chu, P.-Y.; Lin, H.-Y.; Huang, Y.-H. MIR29A Impedes Metastatic Behaviors in Hepatocellular Carcinoma via Targeting LOX, LOXL2, and VEGFA. Int. J. Mol. Sci. 2021, 22, 6001. [Google Scholar] [CrossRef] [PubMed]
- Umezaki, N.; Nakagawa, S.; Yamashita, Y.; Kitano, Y.; Arima, K.; Miyata, T.; Hiyoshi, Y.; Okabe, H.; Nitta, H.; Hayashi, H.; et al. Lysyl Oxidase Induces Epithelial-mesenchymal Transition and Predicts Intrahepatic Metastasis of Hepatocellular Carcinoma. Cancer Sci. 2019, 110, 2033–2043. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Chung, T.; Rhee, H.; Kim, Y.-J.; Jeon, Y.; Yoo, J.E.; Noh, S.; Han, D.H.; Park, Y.N. Increased Expression of the Matrix-Modifying Enzyme Lysyl Oxidase-Like 2 in Aggressive Hepatocellular Carcinoma with Poor Prognosis. Gut Liver 2019, 13, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Zhang, Y.; Zhu, Y.; Cong, Q.; Xiang, Y.; Fu, L. The Effect of LOXL2 in Hepatocellular Carcinoma. Mol. Med. Rep. 2016, 14, 1923–1932. [Google Scholar] [CrossRef]
- Sas, Z.; Cendrowicz, E.; Weinhäuser, I.; Rygiel, T.P. Tumor Microenvironment of Hepatocellular Carcinoma: Challenges and Opportunities for New Treatment Options. Int. J. Mol. Sci. 2022, 23, 3778. [Google Scholar] [CrossRef]
- Satilmis, B.; Sahin, T.T.; Cicek, E.; Akbulut, S.; Yilmaz, S. Hepatocellular Carcinoma Tumor Microenvironment and Its Implications in Terms of Anti-Tumor Immunity: Future Perspectives for New Therapeutics. J. Gastrointest. Cancer 2021, 52, 1198–1205. [Google Scholar] [CrossRef]
- Guizhen, Z.; Guanchang, J.; Liwen, L.; Huifen, W.; Zhigang, R.; Ranran, S.; Zujiang, Y. The Tumor Microenvironment of Hepatocellular Carcinoma and Its Targeting Strategy by CAR-T Cell Immunotherapy. Front. Endocrinol. 2022, 13, 918869. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Rong, D.; Zhang, B.; Zheng, W.; Wang, X.; Chen, Z.; Tang, W. Current Perspectives on the Immunosuppressive Tumor Microenvironment in Hepatocellular Carcinoma: Challenges and Opportunities. Mol. Cancer 2019, 18, 130. [Google Scholar] [CrossRef] [PubMed]
- Reungoat, E.; Grigorov, B.; Zoulim, F.; Pécheur, E.-I. Molecular Crosstalk between the Hepatitis C Virus and the Extracellular Matrix in Liver Fibrogenesis and Early Carcinogenesis. Cancers 2021, 13, 2270. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.-X.; Ling, Y.; Wang, H.-Y. Role of Nonresolving Inflammation in Hepatocellular Carcinoma Development and Progression. Npj Precis. Oncol. 2018, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Gilkes, D.M.; Semenza, G.L.; Wirtz, D. Hypoxia and the Extracellular Matrix: Drivers of Tumour Metastasis. Nat. Rev. Cancer 2014, 14, 430–439. [Google Scholar] [CrossRef]
- Wu, S.; Zheng, Q.; Xing, X.; Dong, Y.; Wang, Y.; You, Y.; Chen, R.; Hu, C.; Chen, J.; Gao, D.; et al. Matrix Stiffness-Upregulated LOXL2 Promotes Fibronectin Production, MMP9 and CXCL12 Expression and BMDCs Recruitment to Assist Pre-Metastatic Niche Formation. J. Exp. Clin. Cancer Res. 2018, 37, 99. [Google Scholar] [CrossRef]
- Lo, C.-M.; Wang, H.-B.; Dembo, M.; Wang, Y. Cell Movement Is Guided by the Rigidity of the Substrate. Biophys. J. 2000, 79, 144–152. [Google Scholar] [CrossRef]
- Liburkin-Dan, T.; Toledano, S.; Neufeld, G. Lysyl Oxidase Family Enzymes and Their Role in Tumor Progression. Int. J. Mol. Sci. 2022, 23, 6249. [Google Scholar] [CrossRef]
- Schrader, J.; Gordon-Walker, T.T.; Aucott, R.L.; Van Deemter, M.; Quaas, A.; Walsh, S.; Benten, D.; Forbes, S.J.; Wells, R.G.; Iredale, J.P. Matrix Stiffness Modulates Proliferation, Chemotherapeutic Response, and Dormancy in Hepatocellular Carcinoma Cells. Hepatology 2011, 53, 1192–1205. [Google Scholar] [CrossRef]
- Tachi, Y.; Hirai, T.; Kojima, Y.; Ishizu, Y.; Honda, T.; Kuzuya, T.; Hayashi, K.; Ishigami, M.; Goto, H. Liver Stiffness Measurement Predicts Hepatocellular Carcinoma Development in Patients Treated with Direct-Acting Antivirals: Liver Stiffness and Cancer Development. JGH Open 2017, 1, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Wen, Q.; Shang, J.; Mise, S.R.L.; Bai, L. Effects of periostin on hepatocellular carcinoma cells invasion, metastasis and prognosis. Chin. J. Hepatol. 2019, 27, 766–771. [Google Scholar] [CrossRef]
- Jang, S.Y.; Park, S.Y.; Lee, H.W.; Choi, Y.-K.; Park, K.-G.; Yoon, G.S.; Tak, W.Y.; Kweon, Y.O.; Hur, K.; Lee, W.K. The Combination of Periostin Overexpression and Microvascular Invasion Is Related to a Poor Prognosis for Hepatocellular Carcinoma. Gut Liver 2016, 10, 948–954. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Wang, W.; Jia, W.-D.; Sun, Q.-K.; Li, J.-S.; Ma, J.-L.; Liu, W.-B.; Zhou, H.-C.; Ge, Y.-S.; Yu, J.-H.; et al. High-Level Expression of Periostin Is Closely Related to Metastatic Potential and Poor Prognosis of Hepatocellular Carcinoma. Med. Oncol. 2013, 30, 385. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Wang, W.; Jia, W.D.; Sun, Q.K.; Huang, M.; Zhou, H.C.; Xia, H.H.; Liu, W.B.; Chen, H.; Sun, S.N.; et al. High Preoparative Levels of Serum Periostin Are Associated with Poor Prognosis in Patients with Hepatocellular Carcinoma after Hepatectomy. Eur. J. Surg. Oncol. EJSO 2013, 39, 1129–1135. [Google Scholar] [CrossRef]
- Maruhashi, T.; Kii, I.; Saito, M.; Kudo, A. Interaction between Periostin and BMP-1 Promotes Proteolytic Activation of Lysyl Oxidase. J. Biol. Chem. 2010, 285, 13294–13303. [Google Scholar] [CrossRef]
- Kumar, P.; Smith, T.; Raeman, R.; Chopyk, D.M.; Brink, H.; Liu, Y.; Sulchek, T.; Anania, F.A. Periostin Promotes Liver Fibrogenesis by Activating Lysyl Oxidase in Hepatic Stellate Cells. J. Biol. Chem. 2018, 293, 12781–12792. [Google Scholar] [CrossRef] [PubMed]
- Vallet, S.D.; Berthollier, C.; Salza, R.; Muller, L.; Ricard-Blum, S. The Interactome of Cancer-Related Lysyl Oxidase and Lysyl Oxidase-Like Proteins. Cancers 2020, 13, 71. [Google Scholar] [CrossRef]
- Wu, J.; Jiang, J.; Chen, B.; Wang, K.; Tang, Y.; Liang, X. Plasticity of Cancer Cell Invasion: Patterns and Mechanisms. Transl. Oncol. 2021, 14, 100899. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Condeelis, J. Regulation of the Actin Cytoskeleton in Cancer Cell Migration and Invasion. Biochim. Biophys. Acta BBA-Mol. Cell Res. 2007, 1773, 642–652. [Google Scholar] [CrossRef]
- Ezzoukhry, Z.; Henriet, E.; Piquet, L.; Boyé, K.; Bioulac-Sage, P.; Balabaud, C.; Couchy, G.; Zucman-Rossi, J.; Moreau, V.; Saltel, F. TGF-Β1 Promotes Linear Invadosome Formation in Hepatocellular Carcinoma Cells, through DDR1 up-Regulation and Collagen I Cross-Linking. Eur. J. Cell Biol. 2016, 95, 503–512. [Google Scholar] [CrossRef]
- Biffi, G.; Tuveson, D.A. Diversity and Biology of Cancer-Associated Fibroblasts. Physiol. Rev. 2021, 101, 147–176. [Google Scholar] [CrossRef]
- Peng, H.; Zhu, E.; Zhang, Y. Advances of Cancer-Associated Fibroblasts in Liver Cancer. Biomark. Res. 2022, 10, 59. [Google Scholar] [CrossRef] [PubMed]
- Kubo, N.; Araki, K.; Kuwano, H.; Shirabe, K. Cancer-Associated Fibroblasts in Hepatocellular Carcinoma. World J. Gastroenterol. 2016, 22, 6841. [Google Scholar] [CrossRef] [PubMed]
- Brenner, D.A.; Waterboer, T.; Choi, S.K.; Lindquist, J.N.; Stefanovic, B.; Burchardt, E.; Yamauchi, M.; Gillan, A.; Rippe, R.A. New Aspects of Hepatic Fibrosis. J. Hepatol. 2000, 32, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Török, N.J. Recent Advances in the Pathogenesis and Diagnosis of Liver Fibrosis. J. Gastroenterol. 2008, 43, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Tacke, F.; Luedde, T.; Trautwein, C. Inflammatory Pathways in Liver Homeostasis and Liver Injury. Clin. Rev. Allergy Immunol. 2009, 36, 4–12. [Google Scholar] [CrossRef]
- Lin, Z.-Y.; Chuang, Y.-H.; Chuang, W.-L. Cancer-Associated Fibroblasts up-Regulate CCL2, CCL26, IL6 and LOXL2 Genes Related to Promotion of Cancer Progression in Hepatocellular Carcinoma Cells. Biomed. Pharmacother. 2012, 66, 525–529. [Google Scholar] [CrossRef]
- Xu, H.; Zhao, J.; Li, J.; Zhu, Z.; Cui, Z.; Liu, R.; Lu, R.; Yao, Z.; Xu, Q. Cancer Associated Fibroblast–Derived CCL5 Promotes Hepatocellular Carcinoma Metastasis through Activating HIF1α/ZEB1 Axis. Cell Death Dis. 2022, 13, 478. [Google Scholar] [CrossRef]
- Song, G.; Shi, Y.; Zhang, M.; Goswami, S.; Afridi, S.; Meng, L.; Ma, J.; Chen, Y.; Lin, Y.; Zhang, J.; et al. Global Immune Characterization of HBV/HCV-Related Hepatocellular Carcinoma Identifies Macrophage and T-Cell Subsets Associated with Disease Progression. Cell Discov. 2020, 6, 90. [Google Scholar] [CrossRef]
- Zheng, H.; Peng, X.; Yang, S.; Li, X.; Huang, M.; Wei, S.; Zhang, S.; He, G.; Liu, J.; Fan, Q.; et al. Targeting Tumor-Associated Macrophages in Hepatocellular Carcinoma: Biology, Strategy, and Immunotherapy. Cell Death Discov. 2023, 9, 65. [Google Scholar] [CrossRef] [PubMed]
- Klepfish, M.; Gross, T.; Vugman, M.; Afratis, N.A.; Havusha-Laufer, S.; Brazowski, E.; Solomonov, I.; Varol, C.; Sagi, I. LOXL2 Inhibition Paves the Way for Macrophage-Mediated Collagen Degradation in Liver Fibrosis. Front. Immunol. 2020, 11, 480. [Google Scholar] [CrossRef] [PubMed]
- Peinado, H.; Zhang, H.; Matei, I.R.; Costa-Silva, B.; Hoshino, A.; Rodrigues, G.; Psaila, B.; Kaplan, R.N.; Bromberg, J.F.; Kang, Y.; et al. Pre-Metastatic Niches: Organ-Specific Homes for Metastases. Nat. Rev. Cancer 2017, 17, 302–317. [Google Scholar] [CrossRef]
- Ghanem, I.; Riveiro, M.E.; Paradis, V.; Faivre, S.; de Parga, P.M.V.; Raymond, E. Insights on the CXCL12-CXCR4 Axis in Hepatocellular Carcinoma Carcinogenesis. Am. J. Transl. Res. 2014, 6, 340–352. [Google Scholar] [PubMed]
- Park, H.J.; Gusarova, G.; Wang, Z.; Carr, J.R.; Li, J.; Kim, K.; Qiu, J.; Park, Y.; Williamson, P.R.; Hay, N.; et al. Deregulation of FoxM1b Leads to Tumour Metastasis. EMBO Mol. Med. 2011, 3, 21–34. [Google Scholar] [CrossRef]
- Wu, S.; Xing, X.; Wang, Y.; Zhang, X.; Li, M.; Wang, M.; Wang, Z.; Chen, J.; Gao, D.; Zhao, Y.; et al. The Pathological Significance of LOXL2 in Pre-Metastatic Niche Formation of HCC and Its Related Molecular Mechanism. Eur. J. Cancer 2021, 147, 63–73. [Google Scholar] [CrossRef]
- Polyak, K.; Weinberg, R.A. Transitions between Epithelial and Mesenchymal States: Acquisition of Malignant and Stem Cell Traits. Nat. Rev. Cancer 2009, 9, 265–273. [Google Scholar] [CrossRef]
- Thiery, J.P.; Acloque, H.; Huang, R.Y.J.; Nieto, M.A. Epithelial-Mesenchymal Transitions in Development and Disease. Cell 2009, 139, 871–890. [Google Scholar] [CrossRef]
- Mashita, N.; Yamada, S.; Nakayama, G.; Tanaka, C.; Iwata, N.; Kanda, M.; Kobayashi, D.; Fujii, T.; Sugimoto, H.; Koike, M.; et al. Epithelial to Mesenchymal Transition Might Be Induced via CD44 Isoform Switching in Colorectal Cancer: EMT and CD44 in Colorectal Cancer. J. Surg. Oncol. 2014, 110, 745–751. [Google Scholar] [CrossRef]
- Yamada, S.; Fuchs, B.C.; Fujii, T.; Shimoyama, Y.; Sugimoto, H.; Nomoto, S.; Takeda, S.; Tanabe, K.K.; Kodera, Y.; Nakao, A. Epithelial-to-Mesenchymal Transition Predicts Prognosis of Pancreatic Cancer. Surgery 2013, 154, 946–954. [Google Scholar] [CrossRef]
- Murai, T.; Yamada, S.; Fuchs, B.C.; Fujii, T.; Nakayama, G.; Sugimoto, H.; Koike, M.; Fujiwara, M.; Tanabe, K.K.; Kodera, Y. Epithelial-to-Mesenchymal Transition Predicts Prognosis in Clinical Gastric Cancer: EMT in Clinical Gastric Cancer. J. Surg. Oncol. 2014, 109, 684–689. [Google Scholar] [CrossRef] [PubMed]
- Christofori, G.; Semb, H. The Role of the Cell-Adhesion Molecule E-Cadherin as a Tumour-Suppressor Gene. Trends Biochem. Sci. 1999, 24, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Peinado, H.; Del Carmen Iglesias-de La Cruz, M.; Olmeda, D.; Csiszar, K.; Fong, K.S.K.; Vega, S.; Nieto, M.A.; Cano, A.; Portillo, F. A Molecular Role for Lysyl Oxidase-like 2 Enzyme in Snail Regulation and Tumor Progression. EMBO J. 2005, 24, 3446–3458. [Google Scholar] [CrossRef] [PubMed]
- Cuevas, E.P.; Moreno-Bueno, G.; Canesin, G.; Santos, V.; Portillo, F.; Cano, A. LOXL2 Catalytically Inactive Mutants Mediate Epithelial-to-Mesenchymal Transition. Biol. Open 2014, 3, 129–137. [Google Scholar] [CrossRef]
- Ninomiya, G.; Yamada, S.; Hayashi, M.; Takeda, S.; Suenaga, M.; Takami, H.; Kanda, M.; Iwata, N.; Niwa, Y.; Tanaka, C.; et al. Significance of Lysyl Oxidase-like2 Gene Expression on the Epithelial-mesenchymal Status of Hepatocellular Carcinoma. Oncol. Rep. 2018, 39, 2664–2672. [Google Scholar] [CrossRef]
- Postovit, L.-M.; Abbott, D.E.; Payne, S.L.; Wheaton, W.W.; Margaryan, N.V.; Sullivan, R.; Jansen, M.K.; Csiszar, K.; Hendrix, M.J.C.; Kirschmann, D.A. Hypoxia/Reoxygenation: A Dynamic Regulator of Lysyl Oxidase-Facilitated Breast Cancer Migration. J. Cell. Biochem. 2008, 103, 1369–1378. [Google Scholar] [CrossRef]
- Schietke, R.; Warnecke, C.; Wacker, I.; Schödel, J.; Mole, D.R.; Campean, V.; Amann, K.; Goppelt-Struebe, M.; Behrens, J.; Eckardt, K.-U.; et al. The Lysyl Oxidases LOX and LOXL2 Are Necessary and Sufficient to Repress E-Cadherin in Hypoxia. J. Biol. Chem. 2010, 285, 6658–6669. [Google Scholar] [CrossRef]
- Tse, A.P.-W.; Sze, K.M.-F.; Shea, Q.T.-K.; Chiu, E.Y.-T.; Tsang, F.H.-C.; Chiu, D.K.-C.; Zhang, M.S.; Lee, D.; Xu, I.M.-J.; Chan, C.Y.-K.; et al. Hepatitis Transactivator Protein X Promotes Extracellular Matrix Modification through HIF/LOX Pathway in Liver Cancer. Oncogenesis 2018, 7, 44. [Google Scholar] [CrossRef]
- Fan, Z.; Zheng, W.; Li, H.; Wu, W.; Liu, X.; Sun, Z.; Hu, H.; Du, L.; Jia, Q.; Liu, Q. LOXL2 Upregulates Hypoxia-inducible Factor-1α Signaling through Snail-FBP1 Axis in Hepatocellular Carcinoma Cells. Oncol. Rep. 2020, 43, 1641–1649. [Google Scholar] [CrossRef]
- Pang, R.W.C.; Joh, J.W.; Johnson, P.J.; Monden, M.; Pawlik, T.M.; Poon, R.T.P. Biology of Hepatocellular Carcinoma. Ann. Surg. Oncol. 2008, 15, 962–971. [Google Scholar] [CrossRef]
- Fang, J.; Zhou, H.; Zhang, C.; Shang, L.; Zhang, L.; Xu, J.; Zheng, L.; Yuan, Y.; Guo, R.; Jia, W.; et al. A Novel Vascular Pattern Promotes Metastasis of Hepatocellular Carcinoma in an Epithelial–Mesenchymal Transition–Independent Manner. Hepatology 2015, 62, 452–465. [Google Scholar] [CrossRef] [PubMed]
- Kerbel, R.S. Tumor Angiogenesis. N. Engl. J. Med. 2008, 358, 2039–2049. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.-M.; Bird, D.; Welti, J.C.; Gourlaouen, M.; Lang, G.; Murray, G.I.; Reynolds, A.R.; Cox, T.R.; Erler, J.T. Lysyl Oxidase Plays a Critical Role in Endothelial Cell Stimulation to Drive Tumor Angiogenesis. Cancer Res. 2013, 73, 583–594. [Google Scholar] [CrossRef] [PubMed]
- Barry-Hamilton, V.; Spangler, R.; Marshall, D.; McCauley, S.; Rodriguez, H.M.; Oyasu, M.; Mikels, A.; Vaysberg, M.; Ghermazien, H.; Wai, C.; et al. Allosteric Inhibition of Lysyl Oxidase–like-2 Impedes the Development of a Pathologic Microenvironment. Nat. Med. 2010, 16, 1009–1017. [Google Scholar] [CrossRef]
- Zhou, H.; Huang, H.; Shi, J.; Zhao, Y.; Dong, Q.; Jia, H.; Liu, Y.; Ye, Q.; Sun, H.; Zhu, X.; et al. Prognostic Value of Interleukin 2 and Interleukin 15 in Peritumoral Hepatic Tissues for Patients with Hepatitis B-Related Hepatocellular Carcinoma after Curative Resection. Gut 2010, 59, 1699–1708. [Google Scholar] [CrossRef]
- Ribatti, D.; Annese, T.; Ruggieri, S.; Tamma, R.; Crivellato, E. Limitations of Anti-Angiogenic Treatment of Tumors. Transl. Oncol. 2019, 12, 981–986. [Google Scholar] [CrossRef]
- Zhu, X.-D.; Sun, H.-C. Emerging Agents and Regimens for Hepatocellular Carcinoma. J. Hematol. Oncol. 2019, 12, 110. [Google Scholar] [CrossRef]
- Bergers, G.; Hanahan, D. Modes of Resistance to Anti-Angiogenic Therapy. Nat. Rev. Cancer 2008, 8, 592–603. [Google Scholar] [CrossRef]
- Zheng, N.; Zhang, S.; Wu, W.; Zhang, N.; Wang, J. Regulatory Mechanisms and Therapeutic Targeting of Vasculogenic Mimicry in Hepatocellular Carcinoma. Pharmacol. Res. 2021, 166, 105507. [Google Scholar] [CrossRef]
- Sun, B.; Zhang, D.; Zhao, N.; Zhao, X. Epithelial-to-Endothelial Transition and Cancer Stem Cells: Two Cornerstones of Vasculogenic Mimicry in Malignant Tumors. Oncotarget 2017, 8, 30502–30510. [Google Scholar] [CrossRef]
- Sun, T.; Sun, B.; Zhao, X.; Zhao, N.; Dong, X.; Che, N.; Yao, Z.; Ma, Y.; Gu, Q.; Zong, W.; et al. Promotion of Tumor Cell Metastasis and Vasculogenic Mimicry by Way of Transcription Coactivation by Bcl-2 and Twist1: A Study of Hepatocellular Carcinoma. Hepatology 2011, 54, 1690–1706. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Gu, H.; Liu, L.; Zhang, X.-L.; Cheng, Q.-L.; Zhu, Y. Inhibitory Effects of LOXL2 Knockdown on Cellular Functions of Liver Cancer Stem Cells. Transl. Cancer Res. 2022, 11, 2013–2025. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-H.; Lian, W.-S.; Wang, F.-S.; Wang, P.-W.; Lin, H.-Y.; Tsai, M.-C.; Yang, Y.-L. MiR-29a Curbs Hepatocellular Carcinoma Incidence via Targeting of HIF-1α and ANGPT2. Int. J. Mol. Sci. 2022, 23, 1636. [Google Scholar] [CrossRef] [PubMed]
- Sequera, C.; Grattarola, M.; Holczbauer, A.; Dono, R.; Pizzimenti, S.; Barrera, G.; Wangensteen, K.J.; Maina, F. MYC and MET Cooperatively Drive Hepatocellular Carcinoma with Distinct Molecular Traits and Vulnerabilities. Cell Death Dis. 2022, 13, 994. [Google Scholar] [CrossRef]
- Dey, S.; Kwon, J.J.; Liu, S.; Hodge, G.A.; Taleb, S.; Zimmers, T.A.; Wan, J.; Kota, J. MiR-29a Is Repressed by MYC in Pancreatic Cancer and Its Restoration Drives Tumor-Suppressive Effects via Downregulation of LOXL2. Mol. Cancer Res. MCR 2020, 18, 311–323. [Google Scholar] [CrossRef]
- Wang, X.; Wu, S.; Yang, Y.; Zhao, J. LncRNA CARMN Affects Hepatocellular Carcinoma Prognosis by Regulating the MiR-192-5p/LOXL2 Axis. Oxidative Med. Cell. Longev. 2022, 2022, 9277360. [Google Scholar] [CrossRef]
- Zhu, Y.; Zheng, B.; Wang, H.; Chen, L. New Knowledge of the Mechanisms of Sorafenib Resistance in Liver Cancer. Acta Pharmacol. Sin. 2017, 38, 614–622. [Google Scholar] [CrossRef]
- Hajdú, I.; Kardos, J.; Major, B.; Fabó, G.; Lőrincz, Z.; Cseh, S.; Dormán, G. Inhibition of the LOX Enzyme Family Members with Old and New Ligands. Selectivity Analysis Revisited. Bioorg. Med. Chem. Lett. 2018, 28, 3113–3118. [Google Scholar] [CrossRef]
- Rodríguez, C.; Rodríguez-Sinovas, A.; Martínez-González, J. Lysyl Oxidase as a Potential Therapeutic Target. Drug News Perspect. 2008, 21, 218–224. [Google Scholar] [CrossRef]
- Rodriguez, H.M.; Vaysberg, M.; Mikels, A.; McCauley, S.; Velayo, A.C.; Garcia, C.; Smith, V. Modulation of Lysyl Oxidase-like 2 Enzymatic Activity by an Allosteric Antibody Inhibitor. J. Biol. Chem. 2010, 285, 20964–20974. [Google Scholar] [CrossRef]
- Verstovsek, S.; Savona, M.R.; Mesa, R.A.; Dong, H.; Maltzman, J.D.; Sharma, S.; Silverman, J.; Oh, S.T.; Gotlib, J. A Phase 2 Study of Simtuzumab in Patients with Primary, Post-Polycythaemia Vera or Post-Essential Thrombocythaemia Myelofibrosis. Br. J. Haematol. 2017, 176, 939–949. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.A.; Abdelmalek, M.F.; Caldwell, S.; Shiffman, M.L.; Diehl, A.M.; Ghalib, R.; Lawitz, E.J.; Rockey, D.C.; Schall, R.A.; Jia, C.; et al. Simtuzumab Is Ineffective for Patients with Bridging Fibrosis or Compensated Cirrhosis Caused by Nonalcoholic Steatohepatitis. Gastroenterology 2018, 155, 1140–1153. [Google Scholar] [CrossRef]
- Hecht, J.R.; Benson, A.B.; Vyushkov, D.; Yang, Y.; Bendell, J.; Verma, U. A Phase II, Randomized, Double-Blind, Placebo-Controlled Study of Simtuzumab in Combination with FOLFIRI for the Second-Line Treatment of Metastatic KRAS Mutant Colorectal Adenocarcinoma. Oncologist 2017, 22, 243-e23. [Google Scholar] [CrossRef] [PubMed]
- Pharos. Available online: https://pharos.nih.gov/targets/LOXL2 (accessed on 10 June 2023).
- Sampath Narayanan, A.; Siegel, R.C.; Martin, G.R. On the Inhibition of Lysyl Oxidase by β-Aminopropionitrile. Biochem. Biophys. Res. Commun. 1972, 46, 745–751. [Google Scholar] [CrossRef]
- Shi, L.; Zhang, N.; Liu, H.; Zhao, L.; Liu, J.; Wan, J.; Wu, W.; Lei, H.; Liu, R.; Han, M. Lysyl Oxidase Inhibition via β-Aminoproprionitrile Hampers Human Umbilical Vein Endothelial Cell Angiogenesis and Migration Invitro. Mol. Med. Rep. 2018, 17, 5029–5036. [Google Scholar] [CrossRef] [PubMed]
- Kirschmann, D.A.; Seftor, E.A.; Fong, S.F.T.; Nieva, D.R.C.; Sullivan, C.M.; Edwards, E.M.; Sommer, P.; Csiszar, K.; Hendrix, M.J.C. A Molecular Role for Lysyl Oxidase in Breast Cancer Invasion. Cancer Res. 2002, 62, 4478–4483. [Google Scholar] [PubMed]
- Abourbih, D.A.; Di Cesare, S.; Orellana, M.E.; Antecka, E.; Martins, C.; Petruccelli, L.A.; Burnier, M.N. Lysyl Oxidase Expression and Inhibition in Uveal Melanoma. Melanoma Res. 2010, 20, 97–106. [Google Scholar] [CrossRef]
- Bondareva, A.; Downey, C.M.; Ayres, F.; Liu, W.; Boyd, S.K.; Hallgrimsson, B.; Jirik, F.R. The Lysyl Oxidase Inhibitor, β-Aminopropionitrile, Diminishes the Metastatic Colonization Potential of Circulating Breast Cancer Cells. PLoS ONE 2009, 4, e5620. [Google Scholar] [CrossRef]
- Yang, X.; Li, S.; Li, W.; Chen, J.; Xiao, X.; Wang, Y.; Yan, G.; Chen, L. Inactivation of Lysyl Oxidase by β-Aminopropionitrile Inhibits Hypoxia-Induced Invasion and Migration of Cervical Cancer Cells. Oncol. Rep. 2013, 29, 541–548. [Google Scholar] [CrossRef]
- Li, Q.; Zhu, C.-C.; Ni, B.; Zhang, Z.-Z.; Jiang, S.-H.; Hu, L.-P.; Wang, X.; Zhang, X.-X.; Huang, P.-Q.; Yang, Q.; et al. Lysyl Oxidase Promotes Liver Metastasis of Gastric Cancer via Facilitating the Reciprocal Interactions between Tumor Cells and Cancer Associated Fibroblasts. eBioMedicine 2019, 49, 157–171. [Google Scholar] [CrossRef]
- Liu, S.B.; Ikenaga, N.; Peng, Z.; Sverdlov, D.Y.; Greenstein, A.; Smith, V.; Schuppan, D.; Popov, Y. Lysyl Oxidase Activity Contributes to Collagen Stabilization during Liver Fibrosis Progression and Limits Spontaneous Fibrosis Reversal in Mice. FASEB J. 2016, 30, 1599–1609. [Google Scholar] [CrossRef] [PubMed]
- Findlay, A.; Turner, C.; Schilter, H.; Deodhar, M.; Zhou, W.; Perryman, L.; Foot, J.; Zahoor, A.; Yao, Y.; Hamilton, R.; et al. An Activity-Based Bioprobe Differentiates a Novel Small Molecule Inhibitor from a LOXL2 Antibody and Provides Renewed Promise for Anti-Fibrotic Therapeutic Strategies. Clin. Transl. Med. 2021, 11, e572. [Google Scholar] [CrossRef] [PubMed]
- Schilter, H.; Findlay, A.D.; Perryman, L.; Yow, T.T.; Moses, J.; Zahoor, A.; Turner, C.I.; Deodhar, M.; Foot, J.S.; Zhou, W.; et al. The Lysyl Oxidase like 2/3 Enzymatic Inhibitor, PXS-5153A, Reduces Crosslinks and Ameliorates Fibrosis. J. Cell. Mol. Med. 2019, 23, 1759–1770. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, J.H.; Rowbottom, M.W.; Lonergan, D.; Darlington, J.; Prodanovich, P.; King, C.D.; Evans, J.F.; Bain, G. Small Molecule Lysyl Oxidase-like 2 (LOXL2) Inhibitors: The Identification of an Inhibitor Selective for LOXL2 over LOX. ACS Med. Chem. Lett. 2017, 8, 423–427. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radić, J.; Kožik, B.; Nikolić, I.; Kolarov-Bjelobrk, I.; Vasiljević, T.; Vranjković, B.; Despotović, S. Multiple Roles of LOXL2 in the Progression of Hepatocellular Carcinoma and Its Potential for Therapeutic Targeting. Int. J. Mol. Sci. 2023, 24, 11745. https://doi.org/10.3390/ijms241411745
Radić J, Kožik B, Nikolić I, Kolarov-Bjelobrk I, Vasiljević T, Vranjković B, Despotović S. Multiple Roles of LOXL2 in the Progression of Hepatocellular Carcinoma and Its Potential for Therapeutic Targeting. International Journal of Molecular Sciences. 2023; 24(14):11745. https://doi.org/10.3390/ijms241411745
Chicago/Turabian StyleRadić, Jelena, Bojana Kožik, Ivan Nikolić, Ivana Kolarov-Bjelobrk, Tijana Vasiljević, Bojana Vranjković, and Sanja Despotović. 2023. "Multiple Roles of LOXL2 in the Progression of Hepatocellular Carcinoma and Its Potential for Therapeutic Targeting" International Journal of Molecular Sciences 24, no. 14: 11745. https://doi.org/10.3390/ijms241411745
APA StyleRadić, J., Kožik, B., Nikolić, I., Kolarov-Bjelobrk, I., Vasiljević, T., Vranjković, B., & Despotović, S. (2023). Multiple Roles of LOXL2 in the Progression of Hepatocellular Carcinoma and Its Potential for Therapeutic Targeting. International Journal of Molecular Sciences, 24(14), 11745. https://doi.org/10.3390/ijms241411745








