Resistance Is Not Futile: Widespread Convergent Evolution of Resistance to Alpha-Neurotoxic Snake Venoms in Caecilians (Amphibia: Gymnophiona)
Abstract
1. Introduction
2. Results
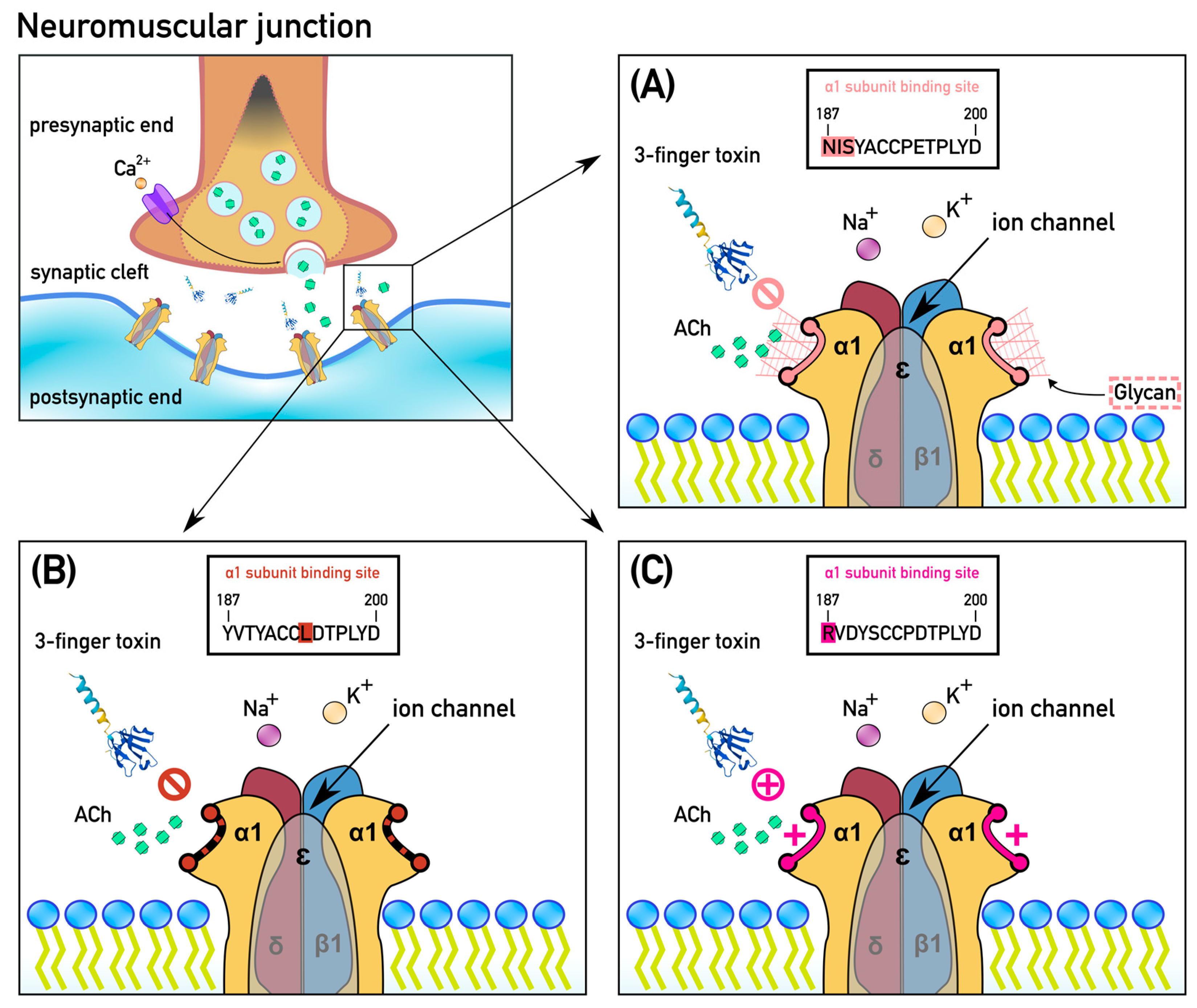
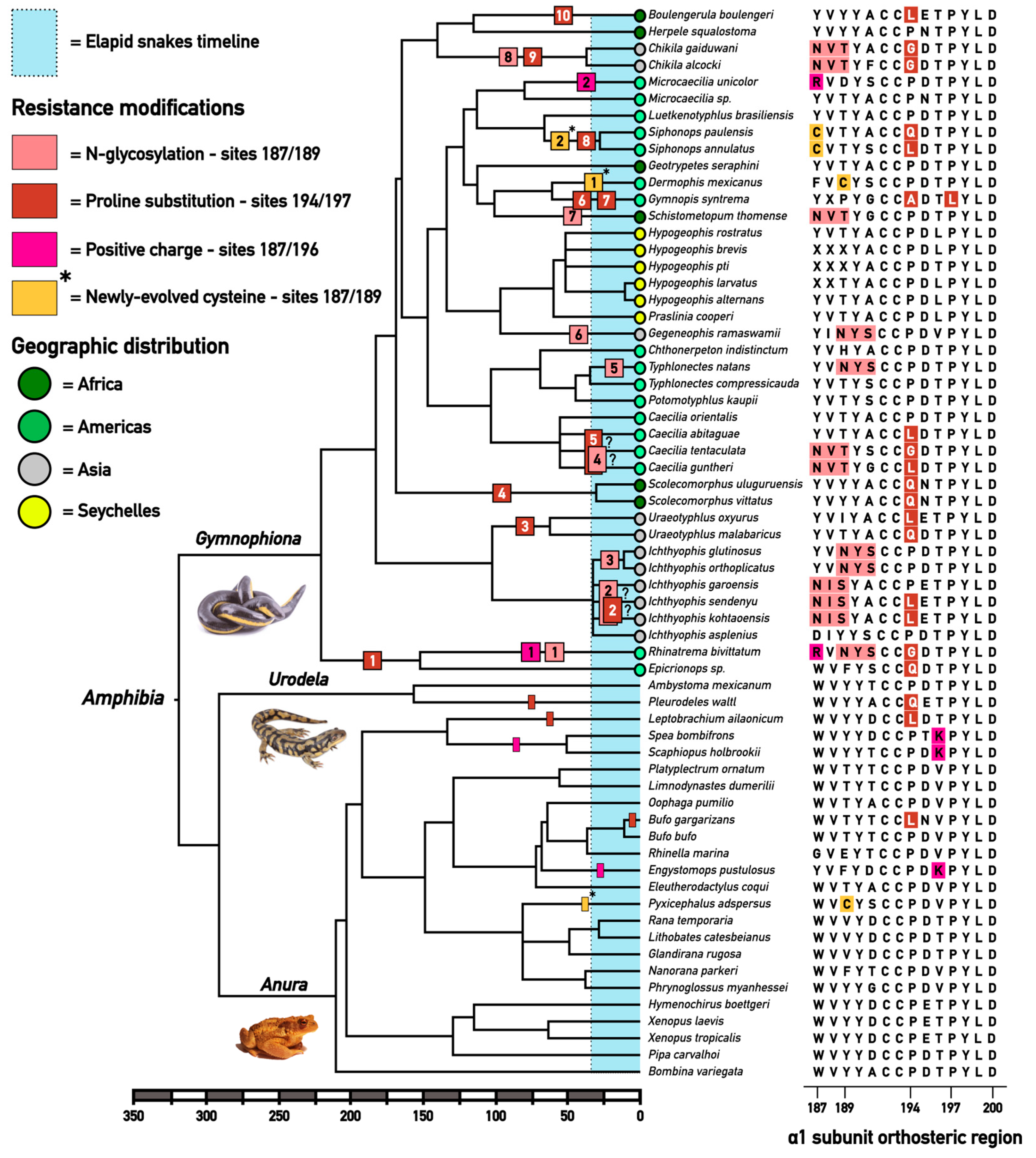
2.1. Extensive Convergent Evolution of Motifs of Resistance to Alpha-Neurotoxins
2.2. Comparative Selection Analyses Indicate Strong Adaptive Pressure to Neurotoxin-Resistance in American and Asian Caecilians
3. Discussion
4. Materials and Methods
4.1. Taxon Selection
4.2. DNA Extraction and Sequencing
4.3. Evolutionary Reconstruction of Toxin Resistance Motifs
4.4. Selection Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Toledo, R.D.; Jared, C. Cutaneous granular glands and amphibian venoms. Comp. Biochem. Physiol. Part A Physiol. 1995, 111, 1–29. [Google Scholar]
- Lüddecke, T.; Schulz, S.; Steinfartz, S.; Vences, M. A salamander’s toxic arsenal: Review of skin poison diversity and function in true salamanders, genus Salamandra. Sci. Nat. 2018, 105, 56. [Google Scholar]
- Kotipoyina, H.R.; Kong, E.L.; Warrington, S.J. Tetrodotoxin toxicity. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- O’Connell, L.A.; Course, L.I.S.L.; O’Connell, J.D.; Paulo, J.A.; Trauger, S.A.; Gygi, S.P.; Murray, A.W. Rapid toxin sequestration modifies poison frog physiology. J. Exp. Biol. 2021, 224, jeb230342. [Google Scholar]
- Jared, C.; Mailho-Fontana, P.L.; Antoniazzi, M.M.; Mendes, V.A.; Barbaro, K.C.; Rodrigues, M.T.; Brodie, E.D. Venomous Frogs Use Heads as Weapons. Curr. Biol. 2015, 25, 2166–2170. [Google Scholar] [CrossRef]
- Mauricio, B.; Mailho-Fontana, P.L.; Sato, L.A.; Barbosa, F.F.; Astray, R.M.; Kupfer, A.; Brodie, E.D.; Jared, C.; Antoniazzi, M.M. Morphology of the cutaneous poison and mucous glands in amphibians with particular emphasis on caecilians (Siphonops annulatus). Toxins 2021, 13, 779. [Google Scholar]
- Fernandez, J.D. First record of Micrurus mipartitus feeding on Oscaecilia polyzona. Rev. Latinoam. Herpetol. 2021, 4, 199–202. [Google Scholar]
- Fernandez, J.D.; Sánchez, D.G. First record of a defensive behavior in Microcaecilia nicefori (Gymnophiona: Siphonopidae) provoked by the snakebite of Micrurus dumerilii (serpentes: Elapidae). Rev. Latinoam. Herpetol. 2021, 4, 144–149. [Google Scholar]
- Fernández-Roldán, J.D.; Gómez-Sánchez, D.A. First record of feeding and defensive behaviour in Thompson’s caecilian Caecilia thompsoni from the Upper Magdalena Valley, Tolima, Colombia. Herpetol. Bull. 2022, 159, 29–31. [Google Scholar]
- Viana, P.F.; de Mello Mendes, D.M. Feeding behavior and first record of Rhinatrema bivittatum (Guérin-Méneville, 1829) as part of the diet of the ribbon coral snake, Micrurus lemniscatus (Linnaeus, 1758) in the Central Amazon region (Serpentes: Elapidae). Herpetol. Notes 2015, 8, 445–447. [Google Scholar]
- Bittencourt-Silva, G.B.; Wilkinson, M. First record of predation on the caecilian Microcaecilia unicolor (Duméril, 1863). Herpetol. Notes 2018, 11, 641–644. [Google Scholar]
- Gower, D.J.; Rasmussen, J.B.; Loader, S.P.; Wilkinson, M. The caecilian amphibian Scolecomorphus kirkii Boulenger as prey of the burrowing asp Atractaspis aterrima Günther: Trophic relationships of fossorial vertebrates. Afr. J. Ecol. 2004, 42, 83–87. [Google Scholar]
- Fry, B.G.; Casewell, N.R.; Wüster, W.; Vidal, N.; Young, B.; Jackson, T.N. The structural and functional diversification of the Toxicofera reptile venom system. Toxicon 2012, 60, 434–448. [Google Scholar]
- Fry, B.G.; Scheib, H.; van der Weerd, L.; Young, B.; McNaughtan, J.; Ramjan, S.R.; Vidal, N.; Poelmann, R.E.; Norman, J.A. Evolution of an arsenal: Structural and functional diversification of the venom system in the advanced snakes (Caenophidia). Mol. Cell. Proteom. 2008, 7, 215–246. [Google Scholar]
- Fry, B. Venomous Reptiles and Their Toxins: Evolution, Pathophysiology and Biodiscovery; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Das, S.; Greenbaum, E.; Meiri, S.; Bauer, A.M.; Burbrink, F.T.; Raxworthy, C.J.; Weinell, J.L.; Brown, R.M.; Brecko, J.; Pauwels, O.S.; et al. Ultraconserved elements-based phylogenomic systematics of the snake superfamily Elapoidea, with the description of a new Afro-Asian family. Mol. Phylogenetics Evol. 2023, 180, 107700. [Google Scholar]
- Holding, M.L.; Drabeck, D.H.; Jansa, S.A.; Gibbs, H.L. Venom resistance as a model for understanding the molecular basis of complex coevolutionary adaptations. Integr. Comp. Biol. 2016, 56, 1032–1043. [Google Scholar]
- Biardi, J.E.; Ho, C.Y.L.; Marcinczyk, J.; Nambiar, K.P. Isolation and identification of a snake venom metalloproteinase inhibitor from California ground squirrel (Spermophilus beecheyi) blood sera. Toxicon 2011, 58, 486–493. [Google Scholar] [CrossRef]
- De Wit, C.A. Resistance of the prairie vole (Microtus ochrogaster) and the woodrat (Neotoma floridana), in Kansas, to venom of the Osage copperhead (Agkistrodon contortrix phaeogaster). Toxicon 1982, 20, 709–714. [Google Scholar]
- Galán, J.A.; Sánchez, E.E.; Rodríguez-Acosta, A.; Pérez, J.C. Neutralization of venoms from two Southern Pacific Rattlesnakes (Crotalus helleri) with commercial antivenoms and endothermic animal sera. Toxicon 2004, 43, 791–799. [Google Scholar]
- Gibbs, H.L.; Sanz, L.; Pérez, A.; Ochoa, A.; Hassinger, A.T.; Holding, M.L.; Calvete, J.J. The molecular basis of venom resistance in a rattlesnake-squirrel predator-prey system. Mol. Ecol. 2020, 29, 2871–2888. [Google Scholar]
- Holding, M.L.; Biardi, J.E.; Gibbs, H.L. Coevolution of venom function and venom resistance in a rattlesnake predator and its squirrel prey. Proc. R. Soc. B Biol. Sci. 2016, 283, 20152841. [Google Scholar]
- Holding, M.L.; Margres, M.J.; Rokyta, D.R.; Gibbs, H.L. Local prey community composition and genetic distance predict venom divergence among populations of the northern Pacific rattlesnake (Crotalus oreganus). J. Evol. Biol. 2018, 31, 1513–1528. [Google Scholar] [CrossRef]
- Perez, J.C.; Haws, W.C.; Hatch, C.H. Resistance of woodrats (Neotoma micropus) to Crotalus atrox venom. Toxicon 1978, 16, 198–200. [Google Scholar]
- Poran, N.S.; Coss, R.G.; Benjamini, E.L.I. Resistance of California ground squirrels (Spermophilus beecheyi) to the venom of the Northern Pacific rattlesnake (Crotalus viridis oreganus): A study of adaptive variation. Toxicon 1987, 25, 767–777. [Google Scholar]
- Pomento, A.M.; Perry, B.W.; Denton, R.D.; Gibbs, H.L.; Holding, M.L. No safety in the trees: Local and species-level adaptation of an arboreal squirrel to the venom of sympatric rattlesnakes. Toxicon 2016, 118, 149–155. [Google Scholar]
- Catanese, J.J.; Kress, L.F. Opossum serum. Alpha. 1-proteinase inhibitor: Purification, linear sequence, and resistance to inactivation by rattlesnake venom metalloproteinase. Biochemistry 1993, 32, 509–515. [Google Scholar]
- Neves-Ferreira, A.G.; Perales, J.; Ovadia, M.; Moussatché, H.; Domont, G.B. Inhibitory properties of the antibothropic complex from the South American opossum (Didelphis marsupialis) serum. Toxicon 1997, 35, 849–863. [Google Scholar]
- Sánchez, E.E.; García, C.; Pérez, J.C.; De La Zerda, S.J. The detection of hemorrhagic proteins in snake venoms using monoclonal antibodies against Virginia opossum (Didelphis virginiana) serum. Toxicon 1998, 36, 1451–1459. [Google Scholar]
- Voss, R.S. Opossums (Mammalia: Didelphidae) in the diets of Neotropical pitvipers (Serpentes: Crotalinae): Evidence for alternative coevolutionary outcomes? Toxicon 2013, 66, 1–6. [Google Scholar]
- Barchan, D.; Kachalsky, S.; Neumann, D.; Vogel, Z.; Ovadia, M.; Kochva, E.; Fuchs, S. How the mongoose can fight the snake: The binding site of the mongoose acetylcholine receptor. Proc. Natl. Acad. Sci. USA 1992, 89, 7717–7721. [Google Scholar] [CrossRef]
- Rodrigues, C.F.B.; Zdenek, C.N.; Serino-Silva, C.; de Morais-Zani, K.; Grego, K.F.; Bénard-Valle, M.; Neri-Castro, E.; Alagón, A.; Tanaka-Azevedo, A.M.; Fry, B.G. BoaγPLI from boa constrictor blood is a broad-spectrum inhibitor of venom PLA2 pathophysiological actions. J. Chem. Ecol. 2021, 47, 907–914. [Google Scholar]
- Goetz, S.M.; Piccolomini, S.; Hoffman, M.; Bogan, J.; Holding, M.L.; Mendonça, M.T.; Steen, D.A. Serum-based inhibition of pitviper venom by eastern indigo snakes (Drymarchon couperi). Biol. Open 2019, 8, bio040964. [Google Scholar]
- Domont, G.B.; Perales, J.; Moussatché, H. Natural anti-snake venom proteins. Toxicon 1991, 29, 1183–1194. [Google Scholar]
- HAINS, P.G.; BROADY, K.W. Purification and inhibitory profile of phospholipase A2 inhibitors from Australian elapid sera. Biochem. J. 2000, 346, 139–146. [Google Scholar]
- Jansa, S.A.; Voss, R.S. Adaptive Evolution of the Venom-Targeted vWF Protein in Opossums that Eat Pitvipers. PLoS ONE 2011, 6, e20997. [Google Scholar] [CrossRef]
- Harris, R.J.; Fry, B.G. Electrostatic resistance to alpha-neurotoxins conferred by charge reversal mutations in nicotinic acetylcholine receptors. Proc. R. Soc. B 2021, 288, 20202703. [Google Scholar]
- Jones, L.; Harris, R.J.; Fry, B.G. Not Goanna Get Me: Mutations in the Savannah Monitor Lizard (Varanus exanthematicus) Nicotinic Acetylcholine Receptor Confer Reduced Susceptibility to Sympatric Cobra Venoms. Neurotox. Res. 2021, 39, 1116–1122. [Google Scholar] [CrossRef]
- Khan, M.A.; Dashevsky, D.; Kerkkamp, H.; Kordiš, D.; de Bakker, M.A.; Wouters, R.; van Thiel, J.; op den Brouw, B.; Vonk, F.J.; Kini, R.M. Widespread evolution of molecular resistance to snake venom α-neurotoxins in vertebrates. Toxins 2020, 12, 638. [Google Scholar]
- Harris, R.J.; Nekaris, K.A.-I.; Fry, B.G. Monkeying around with venom: An increased resistance to α-neurotoxins supports an evolutionary arms race between Afro-Asian primates and sympatric cobras. BMC Biol. 2021, 19, 253. [Google Scholar]
- Kazandjian, T.D.; Petras, D.; Robinson, S.D.; van Thiel, J.; Greene, H.W.; Arbuckle, K.; Barlow, A.; Carter, D.A.; Wouters, R.M.; Whiteley, G. Convergent evolution of pain-inducing defensive venom components in spitting cobras. Science 2021, 371, 386–390. [Google Scholar]
- Takacs, Z.; Wilhelmsen, K.C.; Sorota, S. Snake α-neurotoxin binding site on the Egyptian cobra (Naja haje) nicotinic acetylcholine receptor is conserved. Mol. Biol. Evol. 2001, 18, 1800–1809. [Google Scholar]
- Youngman, N.J.; Llinas, J.; Fry, B.G. Evidence for resistance to coagulotoxic effects of Australian elapid snake venoms by sympatric prey (blue tongue skinks) but not by predators (monitor lizards). Toxins 2021, 13, 590. [Google Scholar]
- Fry, B.G.; Roelants, K.; Champagne, D.E.; Scheib, H.; Tyndall, J.D.; King, G.F.; Nevalainen, T.J.; Norman, J.A.; Lewis, R.J.; Norton, R.S. The toxicogenomic multiverse: Convergent recruitment of proteins into animal venoms. Annu. Rev. Genom. Hum. Genet. 2009, 10, 483–511. [Google Scholar]
- Utkin, Y.; Sunagar, K.; Jackson, T.N.; Reeks, T.; Fry, B.G. Three-finger toxins (3FTXs). In Venomous reptiles and Their Toxins: Evolution, Pathophysiology and Biodiscovery; Fry, B.G., Ed.; Oxford University Press: Oxford, UK, 2015; pp. 215–227. [Google Scholar]
- Mellquist, J.L.; Kasturi, L.; Spitalnik, S.L.; Shakin-Eshleman, S.H. The amino acid following an asn-X-Ser/Thr sequon is an important determinant of N-linked core glycosylation efficiency. Biochemistry 1998, 37, 6833–6837. [Google Scholar]
- Shakin-Eshleman, S.H.; Spitalnik, S.L.; Kasturi, L. The Amino Acid at the X Position of an Asn-X-Ser Sequon Is an Important Determinant of N-Linked Core-glycosylation Efficiency. J. Biol. Chem. 1996, 271, 6363–6366. [Google Scholar]
- Kasturi, L.; Eshleman, J.R.; Wunner, W.H.; Shakin-Eshleman, S.H. The Hydroxy Amino Acid in an Asn-X-Ser/Thr Sequon Can Influence N-Linked Core Glycosylation Efficiency and the Level of Expression of a Cell Surface Glycoprotein. J. Biol. Chem. 1995, 270, 14756–14761. [Google Scholar] [CrossRef]
- Ohtsubo, K.; Marth, J.D. Glycosylation in cellular mechanisms of health and disease. Cell 2006, 126, 855–867. [Google Scholar]
- Gavel, Y.; von Heijne, G. Sequence differences between glycosylated and non-glycosylated Asn-X-Thr/Ser acceptor sites: Implications for protein engineering. Protein Eng. Des. Sel. 1990, 3, 433–442. [Google Scholar]
- Barchan, D.; Ovadia, M.; Kochva, E.; Fuchs, S. The Binding Site of the Nicotinic Acetylcholine Receptor in Animal Species Resistant to alpha-Bungarotoxin. Biochemistry 1995, 34, 9172–9176. [Google Scholar] [CrossRef]
- Kachalsky, S.G.; Jensen, B.S.; Barchan, D.; Fuchs, S. Two subsites in the binding domain of the acetylcholine receptor: An aromatic subsite and a proline subsite. Proc. Natl. Acad. Sci. USA 1995, 92, 10801–10805. [Google Scholar] [CrossRef]
- Chandrasekara, U.; Harris, R.J.; Fry, B.G. The Target Selects the Toxin: Specific Amino Acids in Snake-Prey Nicotinic Acetylcholine Receptors That Are Selectively Bound by King Cobra Venoms. Toxins 2022, 14, 528. [Google Scholar]
- Drabeck, D.H.; Dean, A.M.; Jansa, S.A. Why the honey badger don’t care: Convergent evolution of venom-targeted nicotinic acetylcholine receptors in mammals that survive venomous snake bites. Toxicon 2015, 99, 68–72. [Google Scholar]
- Vitt, L.J.; Caldwell, J.P. Herpetology: An introductory Biology of Amphibians and Reptiles; Academic Press: Cambridge, MA, USA, 2013. [Google Scholar]
- Kamei, R.G.; Mauro, D.S.; Gower, D.J.; Van Bocxlaer, I.; Sherratt, E.; Thomas, A.; Babu, S.; Bossuyt, F.; Wilkinson, M.; Biju, S.D. Discovery of a new family of amphibians from northeast India with ancient links to Africa. Proc. R. Soc. B Biol. Sci. 2012, 279, 2396–2401. [Google Scholar]
- Bhardwaj, V.K.; Kapoor, R. A report on ophiophagy observed in Naja oxiana (Eichwald, 1831) from Himachal Pradesh, India. Hamadryad 2022, 39, 75–76. [Google Scholar]
- Yadav, V.; Shinde, A. A Case of Cannibalism in Common Krait Bungarus caeruleus (Schneider, 1801) (Reptilia: Serpentes: Elapidae). J. Bombay Nat. Hist. Soc. 2021. [Google Scholar] [CrossRef]
- Tan, C.H.; Oh, A.M.F.; Wong, K.Y.; Liew, J.L.; Tan, N.H.; Tan, K.Y. On characterizing the Red-headed Krait (Bungarus flaviceps) venom: Decomplexation proteomics, immunoreactivity and toxicity cross-neutralization by hetero-specific antivenoms. Comp. Biochem. Physiol. Part D Genom. Proteom. 2022, 43, 101006. [Google Scholar]
- Solanki, J.; Khare, R. Cannibalism in the Common Krait (Bungarus caeruleus) and the Indian Spectacled Cobra (Naja naja). Reptiles Amphib. 2022, 29, 470–471. [Google Scholar]
- Zhang, P.; Wake, M.H. A mitogenomic perspective on the phylogeny and biogeography of living caecilians (Amphibia: Gymnophiona). Mol. Phylogenetics Evol. 2009, 53, 479–491. [Google Scholar]
- Gower, D.J.; Wilkinson, M. Caecilians (Gymnophiona). In The Timetree of Life; Hedges, S.B., Kumar, K., Eds.; OUP: Oxford, UK, 2009; pp. 369–372. [Google Scholar]
- Wilkinson, M.; Sheps, J.A.; Oommen, O.V.; Cohen, B.L. Phylogenetic relationships of Indian caecilians (Amphibia: Gymnophiona) inferred from mitochondrial rRNA gene sequences. Mol. Phylogenetics Evol. 2002, 23, 401–407. [Google Scholar]
- San Mauro, D.; Gower, D.J.; Müller, H.; Loader, S.P.; Zardoya, R.; Nussbaum, R.A.; Wilkinson, M. Life-history evolution and mitogenomic phylogeny of caecilian amphibians. Mol. Phylogenetics Evol. 2014, 73, 177–189. [Google Scholar]
- Wilkinson, M.; San Mauro, D.; Sherratt, E.; Gower, D.J. A nine-family classification of caecilians (Amphibia: Gymnophiona). Zootaxa 2011, 2874, 41–64. [Google Scholar]
- Dubois, A.; Frétey, T. Herpetological higher taxa nomina. 6. Gymnophiona Rafinesque, 1814. Bionomina 2021, 21, 39–72. [Google Scholar]
- Benson, D.A.; Cavanaugh, M.; Clark, K.; Karsch-Mizrachi, I.; Lipman, D.J.; Ostell, J.; Sayers, E.W. GenBank. Nucleic Acids Res. 2017, 45, D37–D42. [Google Scholar] [CrossRef]
- Kumar, S.; Suleski, M.; Craig, J.M.; Kasprowicz, A.E.; Sanderford, M.; Li, M.; Stecher, G.; Hedges, S.B. TimeTree 5: An expanded resource for species divergence times. Mol. Biol. Evol. 2022, 39, msac174. [Google Scholar]
- Maddison, W.P.; Maddison, D.R. Mesquite: A Modular System for Evolutionary Analysis. Version 3.80. 2023. Available online: http://www.mesquiteproject.org (accessed on 30 July 2022).
- Xu, B.; Yang, Z. PAMLX: A graphical user interface for PAML. Mol. Biol. Evol. 2013, 30, 2723–2724. [Google Scholar]
- Zaher, H.; Murphy, R.W.; Arredondo, J.C.; Graboski, R.; Machado-Filho, P.R.; Mahlow, K.; Montingelli, G.G.; Quadros, A.B.; Orlov, N.L.; Wilkinson, M.; et al. Large-scale molecular phylogeny, morphology, divergence-time estimation, and the fossil record of advanced caenophidian snakes (Squamata: Serpentes). PLoS ONE 2019, 14, e0216148. [Google Scholar]
- Gower, D.J.; Kupfer, A.; Oommen, O.V.; Himstedt, W.; Nussbaum, R.A.; Loader, S.P.; Presswell, B.; Müller, H.; Krishna, S.B.; Boistel, R.; et al. A molecular phylogeny of ichthyophiid caecilians (Amphibia: Gymnophiona: Ichthyophiidae): Out of India or out of South East Asia? Proc. R. Soc. London Ser. B Biol. Sci. 2002, 269, 1563–1569. [Google Scholar]
- Briggs, J.C. The biogeographic and tectonic history of India. J. Biogeogr. 2003, 30, 381–388. [Google Scholar]
- McLoughlin, S. The breakup history of Gondwana and its impact on pre-Cenozoic floristic provincialism. Aust. J. Bot. 2001, 49, 271–300. [Google Scholar]
- Chatterjee, S.; Goswami, A.; Scotese, C.R. The longest voyage: Tectonic, magmatic, and paleoclimatic evolution of the Indian plate during its northward flight from Gondwana to Asia. Gondwana Res. 2013, 23, 238–267. [Google Scholar]
- Aitchison, J.C.; Ali, J.R.; Davis, A.M. When and where did India and Asia collide? J. Geophys. Res. Solid Earth 2007, 112. [Google Scholar] [CrossRef]
- Guo, P.; Liu, Q.; Xu, Y.; Jiang, K.; Hou, M.; Ding, L.; Pyron, R.A.; Burbrink, F.T. Out of Asia: Natricine snakes support the Cenozoic Beringian dispersal hypothesis. Mol. Phylogenetics Evol. 2012, 63, 825–833. [Google Scholar]
- Terrat, Y.; Sunagar, K.; Fry, B.G.; Jackson, T.N.; Scheib, H.; Fourmy, R.; Verdenaud, M.; Blanchet, G.; Antunes, A.; Ducancel, F. Atractaspis aterrima toxins: The first insight into the molecular evolution of venom in side-stabbers. Toxins 2013, 5, 1948–1964. [Google Scholar]
- Oulion, B.; Dobson, J.S.; Zdenek, C.N.; Arbuckle, K.; Lister, C.; Coimbra, F.C.; Op den Brouw, B.; Debono, J.; Rogalski, A.; Violette, A.; et al. Factor X activating Atractaspis snake venoms and the relative coagulotoxicity neutralising efficacy of African antivenoms. Toxicol. Lett. 2018, 288, 119–128. [Google Scholar]
- Nussbaum, R.A.; Pfrender, M.E. Revision of the African caecilian genus Schistometopum Parker (Amphibia: Gymnophiona: Caeciliidae); Miscellaneous Publications Museum of Zoology University of Michigan: Ann Arbor, MI, USA, 1998. [Google Scholar]
- Ceriaco, L.M.; Marques, M.P.; Schmitz, A.; Bauer, A.M. The “cobra-preta” of São Tomé island, gulf of Guinea, is a new species of Naja Laurenti, 1768 (Squamata: Elapidae). Zootaxa 2017, 4324, 121–141. [Google Scholar]
- Sanmartíin, I.; Ronquist, F. Southern hemisphere biogeography inferred by event-based models: Plant versus animal patterns. Syst. Biol. 2004, 53, 216–243. [Google Scholar]
- Modahl, C.M.; Mukherjee, A.K.; Mackessy, S.P. An analysis of venom ontogeny and prey-specific toxicity in the Monocled Cobra (Naja kaouthia). Toxicon 2016, 119, 8–20. [Google Scholar]
- Wang, K.; Lyu, Z.; Wang, J.; Qi, S.; Che, J. The updated checklist and zoogeographic division of the reptilian fauna of Yunnan Province, China. Biodivers. Sci. 2022, 30, 21326. [Google Scholar]
- Yang, D.; Lu, S. Leptobrachium ailaonicum. The IUCN Red List of Threatened Species. 2004. Available online: https://dx.doi.org/10.2305/IUCN.UK.2004.RLTS.T57624A11665512.en (accessed on 15 May 2023).
- Yang, J.H.; Huang, X.Y.; Ye, J.F.; Yang, S.P.; Zhang, X.C.; Chan, B.P.L. A report on the herpetofauna of Tengchong Section of Gaoligongshan National Nature Reserve, China. J. Threat. Taxa 2019, 11, 14434–14451. [Google Scholar]
- Harris, R.J.; Zdenek, C.N.; Harrich, D.; Frank, N.; Fry, B.G. An Appetite for Destruction: Detecting Prey-Selective Binding of alpha-Neurotoxins in the Venom of Afro-Asian Elapids. Toxins 2020, 12, E205. [Google Scholar]
- Artimo, P.; Jonnalagedda, M.; Arnold, K.; Baratin, D.; Csardi, G.; De Castro, E.; Duvaud, S.; Flegel, V.; Fortier, A.; Gasteiger, E.; et al. ExPASy: SIB bioinformatics resource portal. Nucleic Acids Res. 2012, 40, W597–W603. [Google Scholar]
- Katoh, K.; Asimenos, G.; Toh, H. Multiple alignment of DNA sequences with MAFFT. In Bioinformatics for DNA Sequence Analysis; Springer: Berlin/Heidelberg, Germany, 2009; pp. 39–64. [Google Scholar]
- Johnson, M.; Zaretskaya, I.; Raytselis, Y.; Merezhuk, Y.; McGinnis, S.; Madden, T.L. NCBI BLAST: A better web interface. Nucleic Acids Res. 2008, 36 (Suppl. 2), W5–W9. [Google Scholar]
- Smith, J.J.; Putta, S.; Walker, J.A.; Kump, D.K.; Samuels, A.K.; Monaghan, J.R.; Weisrock, D.W.; Staben, C.; Voss, S.R. Sal-Site: Integrating new and existing ambystomatid salamander research and informational resources. BMC Genom. 2005, 6, 181. [Google Scholar]
- Matsunami, M.; Suzuki, M.; Haramoto, Y.; Fukui, A.; Inoue, T.; Yamaguchi, K.; Uchiyama, I.; Mori, K.; Tashiro, K.; Ito, Y.; et al. A comprehensive reference transcriptome resource for the Iberian ribbed newt Pleurodeles waltl, an emerging model for developmental and regeneration biology. DNA Res. 2019, 26, 217–229. [Google Scholar]
| ω Values | |||
|---|---|---|---|
| Selection Model | Clade/Branch Set | Orthosteric (Toxin-Binding Site) | Flanking Regions |
| One-ratio model (M0) | non-caecilian amphibians (24 spp.) | 0.0852 | 0.0205 |
| One-ratio model (M0) | caecilians (40 spp.) | 0.6019 | 0.1907 |
| Branch model (M2) with two branch sets | Caecilian branches in the absence of elapids | 0.2168 | 0.0104 |
| Caecilian branches in the presence of elapids | 0.9699 | 0.452 | |
| Branch model (M2) with five branch sets | Caecilian branches older than elapid radiation (Early Oligocene) | 0.2411 | 0.0119 |
| African caecilians overlapping with elapid radiation | 0.1703 | 0.2322 | |
| American caecilians overlapping with elapid radiation | 1.7789 | 0.4039 | |
| Asian Caecilians overlapping with elapid radiation | 2.6121 | 0.7442 | |
| Seychelles caecilians (in the absence of elapids) | 0.0001 | N.A. | |
| ω Values | |||
|---|---|---|---|
| Selection Model | Clade/Branch Set | chrna2 | chrna4 |
| One-ratio model (M0) | three caecilian species + five non-caecilian species (eight spp.) | 0.0001 | 0.0015 |
| Branch model (M2) | Caecilians (three spp.) | 0.0001 | 0.0037 |
| Non-caecilian amphibians (five spp.) | 0.0001 | 0.0012 | |
| Gymnophiona | ||||
|---|---|---|---|---|
| Family | Species | Serial Number | Source | Locality |
| Caeciliidae | Caecilia abitaguae | MZUTI 4030 | MZUTI | Ecuador |
| Caeciliidae | Caecilia guntheri | MZUTI 3039 | MZUTI | Ecuador |
| Caeciliidae | Caecilia orientalis | MZUTI 1371 | MZUTI | Ecuador |
| Caeciliidae | Caecilia tentaculata | MZUTI 3919 | MZUTI | Ecuador |
| Chikilidae | Chikila alcocki | BNHS 6210 (RGK 0804) | BNHS | India |
| Chikilidae | Chikila gaiduwani | BNHS 6211 (RGK 0102) | BNHS | India |
| Dermophiidae | Dermophis mexicanus | RAN 31534 | UMMZ | Guatemala |
| Dermophiidae | Geotrypetes seraphini | XM_033946945.1 | GenBank | Cameroon |
| Dermophiidae | Gymnopis syntrema | RAN 31499 | UMMZ | Guatemala |
| Dermophiidae | Schistometopum thomense | RAN 31503 | UMMZ | São Tomé |
| Grandisoniidae | Gegeneophis ramaswamii | UK MW331 | NHM | India |
| Grandisoniidae | Hypogeophis alternans | RAN 31465 | UMMZ | Seychelles |
| Grandisoniidae | Hypogeophis brevis | SM 691 | NHM | Seychelles |
| Grandisoniidae | Hypogeophis cooperi | RAN 31305 | UMMZ | Seychelles |
| Grandisoniidae | Hypogeophis larvatus | SM 297 | NHM | Seychelles |
| Grandisoniidae | Hypogeophis pti | SM 295 | NHM | Seychelles |
| Grandisoniidae | Hypogeophis rostratus | RAN 31441 | UMMZ | Seychelles |
| Ichthyophiidae | Ichthyophis garoensis | BNHS 6208 (RGK 0157) | BNHS | India |
| Ichthyophiidae | Ichthyophis orthoplicatus | DNM MW1723 | / | Sri Lanka |
| Ichthyophiidae | Ichthyophis sendenyu | BNHS 6209 (RGK 0809) | BNHS | India |
| Ichthyophiidae | Uraeotyphlus malabaricus | UK MW1711 | NHM | India |
| Ichthyophiidae | Uraeotyphlus oxyurus | UK MW212 | NHM | India |
| Siphonopidae | Microcaecilia sp. | NHM MW995 | NHM | Guyana |
| Siphonopidae | Siphonops annulatus | RAN 31968 | UMMZ | Ecuador |
| Siphonopidae | Siphonops paulensis | CHUNB 39114 | CHUNB | Brazil |
| Typhlonectidae | Chthonerpeton indistinctum | MCP MW15 | MCP | Brazil |
| Typhlonectidae | Potomotyphlus kaupii | UMFS 11777 | UMMZ | Peru |
| Typhlonectidae | Typhlonectes compressicauda | UMFS 11776 | UMMZ | Peru |
| Typhlonectidae | Typhlonectes natans | BMNH 2000.218. | NHM | Colombia |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mancuso, M.; Zaman, S.; Maddock, S.T.; Kamei, R.G.; Salazar-Valenzuela, D.; Wilkinson, M.; Roelants, K.; Fry, B.G. Resistance Is Not Futile: Widespread Convergent Evolution of Resistance to Alpha-Neurotoxic Snake Venoms in Caecilians (Amphibia: Gymnophiona). Int. J. Mol. Sci. 2023, 24, 11353. https://doi.org/10.3390/ijms241411353
Mancuso M, Zaman S, Maddock ST, Kamei RG, Salazar-Valenzuela D, Wilkinson M, Roelants K, Fry BG. Resistance Is Not Futile: Widespread Convergent Evolution of Resistance to Alpha-Neurotoxic Snake Venoms in Caecilians (Amphibia: Gymnophiona). International Journal of Molecular Sciences. 2023; 24(14):11353. https://doi.org/10.3390/ijms241411353
Chicago/Turabian StyleMancuso, Marco, Shabnam Zaman, Simon T. Maddock, Rachunliu G. Kamei, David Salazar-Valenzuela, Mark Wilkinson, Kim Roelants, and Bryan G. Fry. 2023. "Resistance Is Not Futile: Widespread Convergent Evolution of Resistance to Alpha-Neurotoxic Snake Venoms in Caecilians (Amphibia: Gymnophiona)" International Journal of Molecular Sciences 24, no. 14: 11353. https://doi.org/10.3390/ijms241411353
APA StyleMancuso, M., Zaman, S., Maddock, S. T., Kamei, R. G., Salazar-Valenzuela, D., Wilkinson, M., Roelants, K., & Fry, B. G. (2023). Resistance Is Not Futile: Widespread Convergent Evolution of Resistance to Alpha-Neurotoxic Snake Venoms in Caecilians (Amphibia: Gymnophiona). International Journal of Molecular Sciences, 24(14), 11353. https://doi.org/10.3390/ijms241411353









