Abscisic Acid Regulates Carbohydrate Metabolism, Redox Homeostasis and Hormonal Regulation to Enhance Cold Tolerance in Spring Barley
Abstract
1. Introduction
2. Results
2.1. Phenotypic Symptoms and Photosynthetic Electron Transport
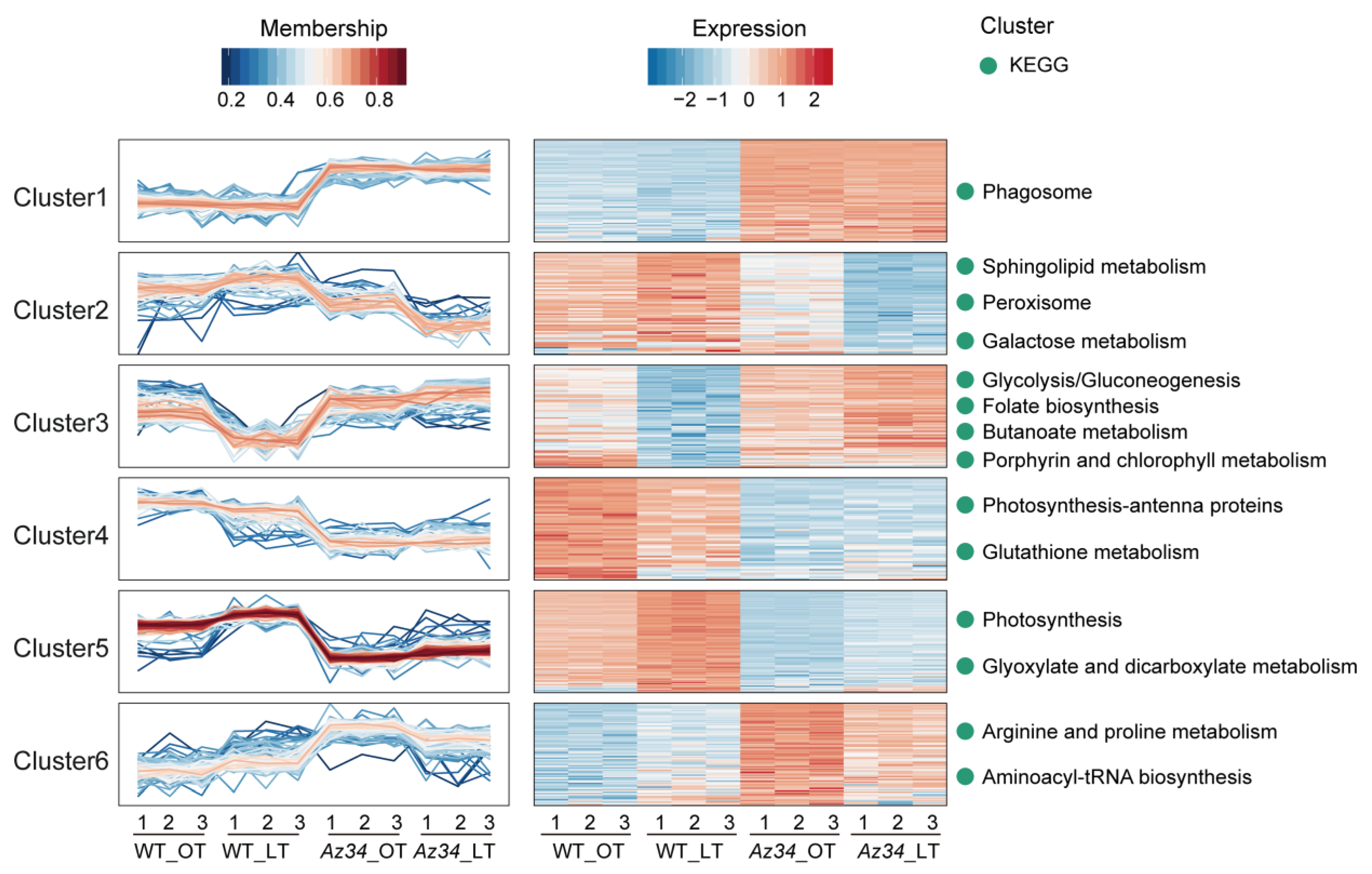
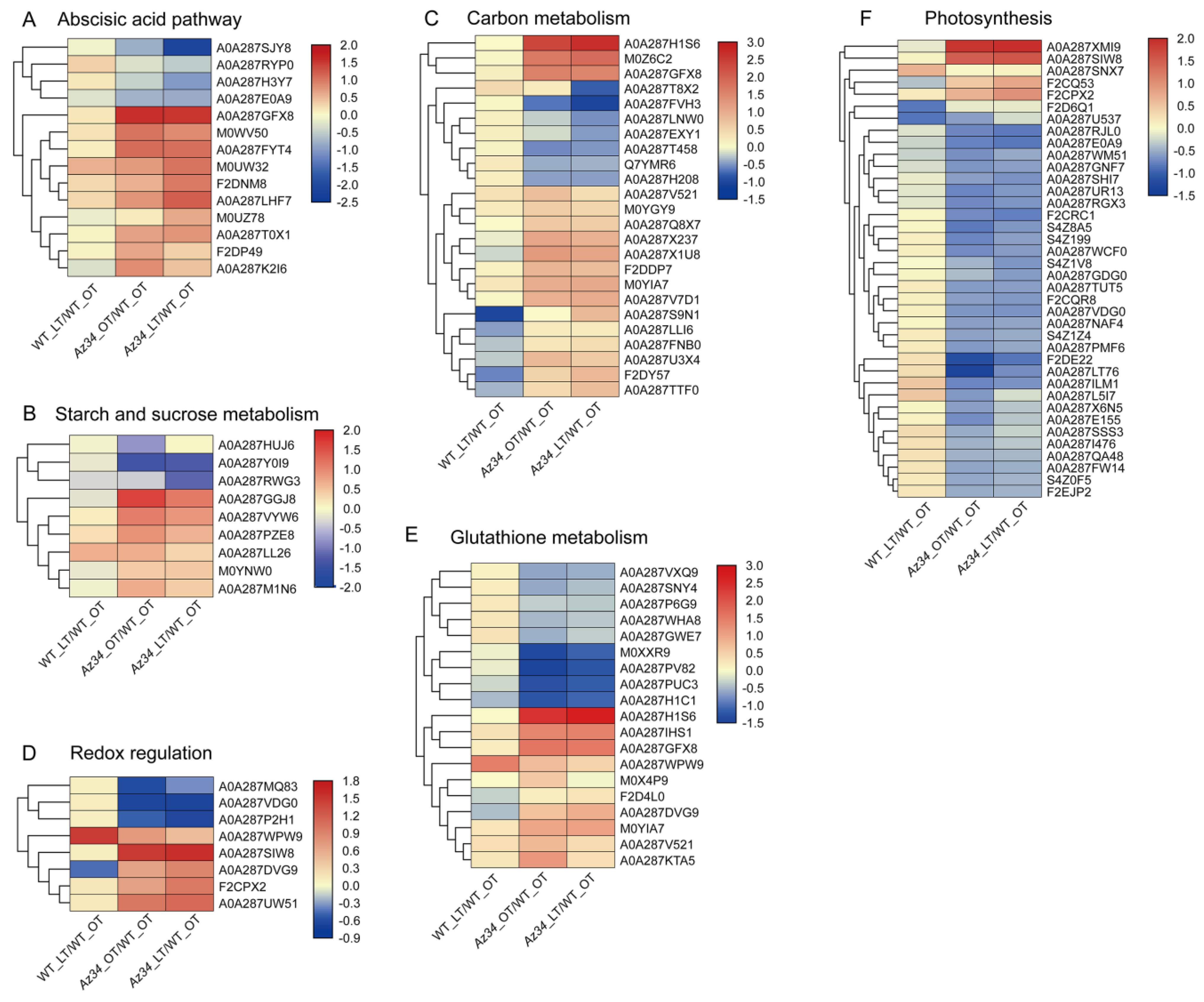
2.2. Proteomics
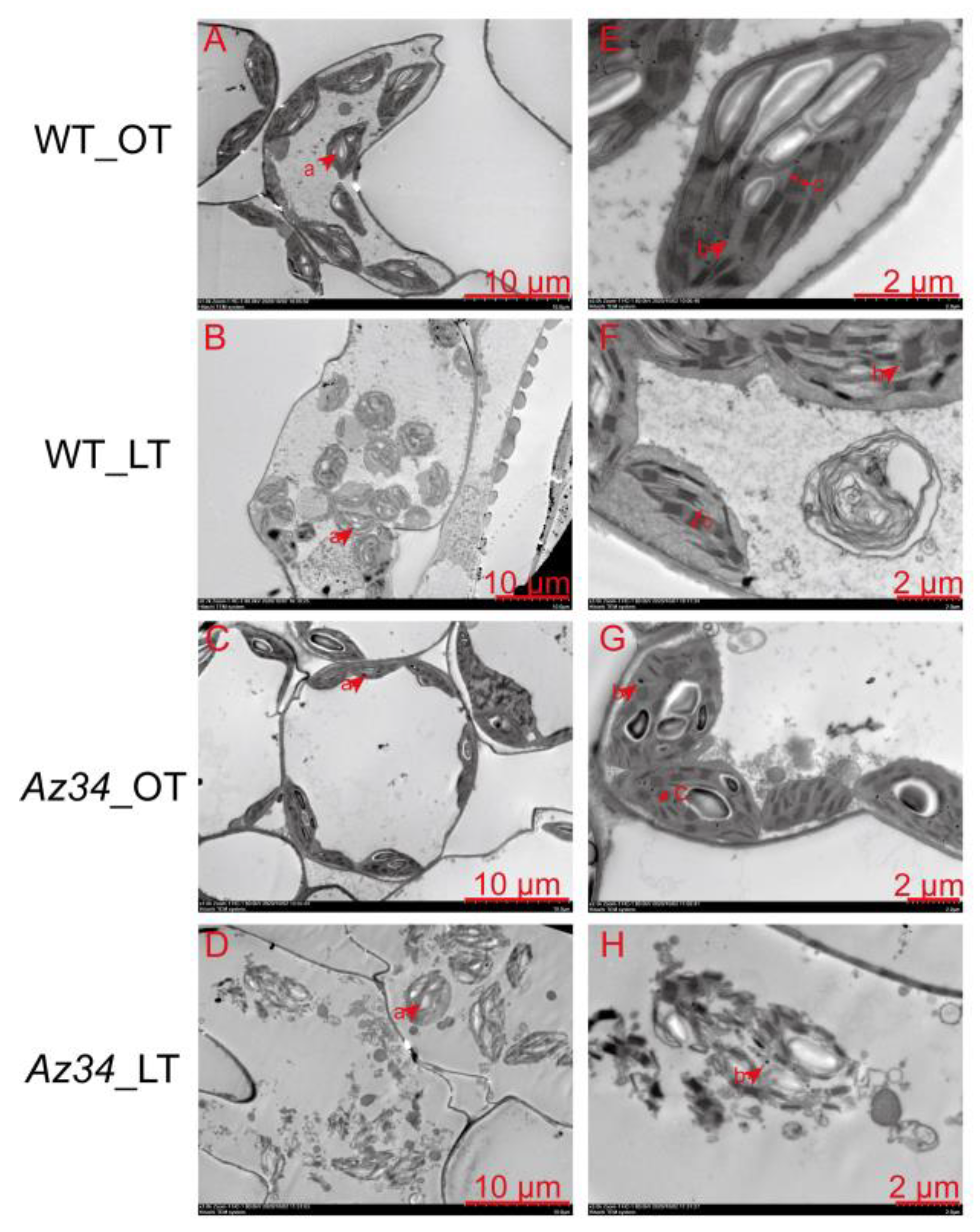
2.3. Chloroplast Ultrastructure
2.4. Carbohydrate and ROS Metabolisms
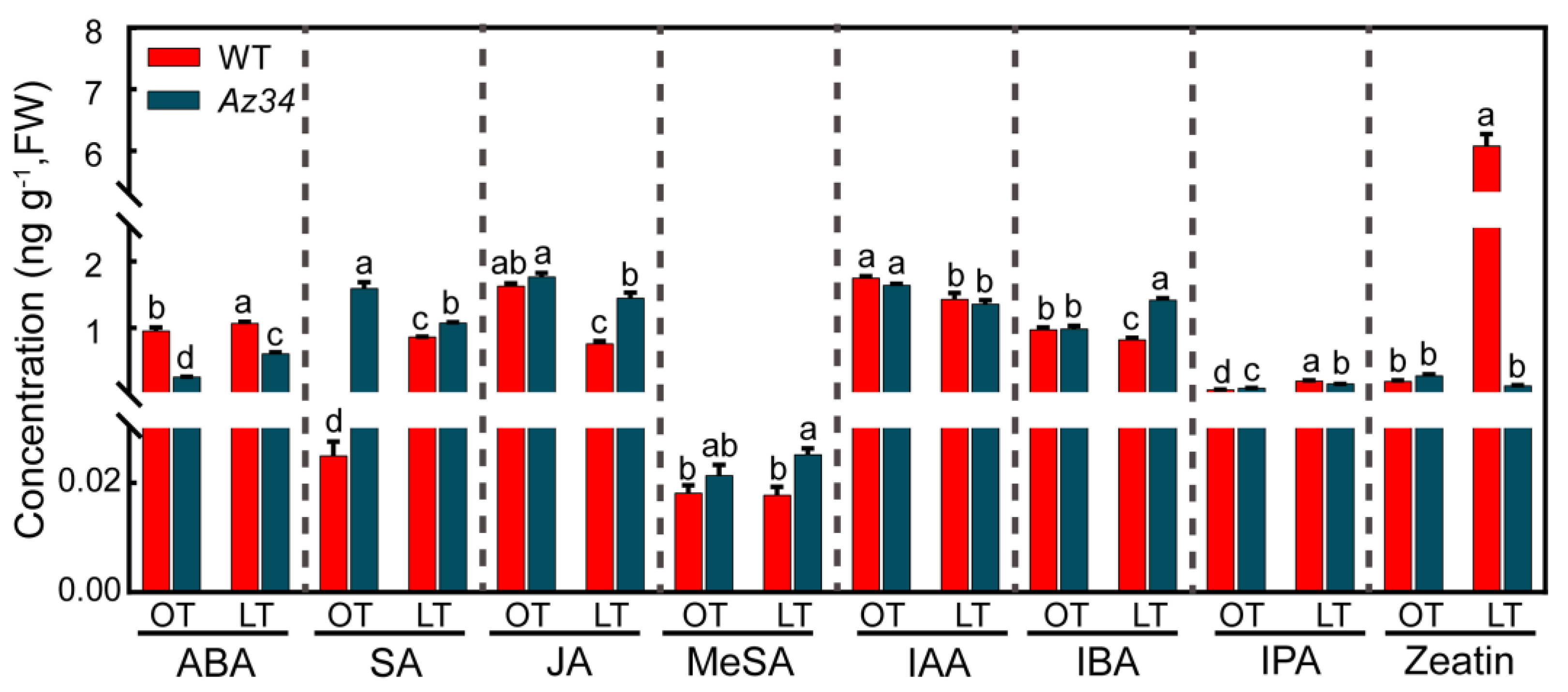
2.5. Hormonal Regulatory Network
2.6. Principal Component Analysis
3. Discussion
3.1. ABA Deficiency Increased the Sensibility of Photosynthetic System and Chloroplast Ultrastructure to Low Temperature
3.2. ABA Induced Modifications in Metabolisms of Carbohydrate and ROS Contributed to Low Temperature Tolerance
3.3. ABA Deficiency Induced Changes in Hormonal Regulatory Network Were Related to Low Temperature
4. Materials and Methods
4.1. Experimental Design
4.2. Chlorophyll a Fluorescence and Gas Exchange
4.3. Protein Extraction, Quantification and Identification by 4D-Proteomics
4.4. Database Search and Bioinformatics
4.5. Transmission Electron Microscopy for Chloroplast Ultrastructure Analysis
4.6. Determination of Hormone Concentration in Barley Leaf
4.7. Determination of Activity of Enzymes Involved in Carbohydrate and ROS Metabolisms
4.8. Quantifications of Proline and H2O2
4.9. Quantifications of Soluble Sugars and Soluble Proteins
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Asseng, S.; Foster, I.A.N.; Turner, N.C. The impact of temperature variability on wheat yields. Glob. Change Biol. 2011, 17, 997–1012. [Google Scholar] [CrossRef]
- Airaki, M.; Leterrier, M.; Mateos, R.M.; Valderrama, R.; Chaki, M.; Barroso, J.B.; Del Rio, L.A.; Palma, J.M.; Corpas, F.J. Metabolism of reactive oxygen species and reactive nitrogen species in pepper (Capsicum annuum L.) plants under low temperature stress. Plant Cell Environ. 2012, 35, 281–295. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Jiang, Y.; Han, X.; Wang, H.; Pan, J.; Yu, D. Jasmonate regulates leaf senescence and tolerance to cold stress: Crosstalk with other phytohormones. J. Exp. Bot. 2017, 68, 1361–1369. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liu, S.; Guo, J.; Liu, F.; Song, F.; Li, X. Effect of the transgenerational exposure to elevated CO2 on low temperature tolerance of winter wheat: Chloroplast ultrastructure and carbohydrate metabolism. J. Agron. Crop Sci. 2020, 206, 773–783. [Google Scholar] [CrossRef]
- Jurczyk, B.; Grzesiak, M.; Pociecha, E.; Wlazlo, M.; Rapacz, M. Diverse stomatal behaviors mediating photosynthetic acclimation to low temperatures in Hordeum vulgare. Front. Plant Sci. 2019, 9, 1963. [Google Scholar] [CrossRef]
- Marshall, C.J. Cold-adapted enzymes. Trends Biotechnol. 1997, 15, 359–364. [Google Scholar] [CrossRef]
- Zhang, W.J.; Wang, J.Q.; Huang, Z.L.; Mi, L.; Xu, K.F.; Wu, J.J.; Fan, Y.H.; Ma, S.Y.; Jiang, D.G. Effects of low temperature at booting stage on sucrose metabolism and endogenous hormone contents in winter wheat spikelet. Front. Plant Sci. 2019, 10, 498. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhou, M.; Xu, K.; Li, J.H.; Li, S.S.; Zhang, S.H.; Yang, X.J. Integrated transcriptomics and metabolomics analyses provide insights into cold stress response in wheat. Crop J. 2019, 7, 857–866. [Google Scholar] [CrossRef]
- Ding, Y.; Shi, Y.; Yang, S. Advances and challenges in uncovering cold tolerance regulatory mechanisms in plants. N. Phytol. 2019, 222, 1690–1704. [Google Scholar] [CrossRef]
- Crosatti, C.; Rizza, F.; Badeck, F.W.; Mazzucotelli, E.; Cattivelli, L. Harden the chloroplast to protect the plant. Physiol. Plant. 2013, 147, 55–63. [Google Scholar] [CrossRef]
- Venzhik, Y.; Talanova, V.; Titov, A. The effect of abscisic acid on cold tolerance and chloroplasts ultrastructure in wheat under optimal and cold stress conditions. Acta Physiol. Plant. 2016, 38, 63. [Google Scholar] [CrossRef]
- Kawamura, Y.; Uemura, M. Mass spectrometric approach for identifying putative plasma membrane proteins of Arabidopsis leaves associated with cold acclimation. Plant J. 2003, 36, 141–154. [Google Scholar] [CrossRef]
- Goulas, E.; Schubert, M.; Kieselbach, T.; Kleczkowski, L.A.; Gardestrom, P.; Schroder, W.; Hurry, V. The chloroplast lumen and stromal proteomes of Arabidopsis thaliana show differential sensitivity to short- and long-term exposure to low temperature. Plant J. 2006, 47, 720–734. [Google Scholar] [CrossRef]
- Peleg, Z.; Blumwald, E. Hormone balance and abiotic stress tolerance in crop plants. Curr. Opin. Plant Biol. 2011, 14, 290–295. [Google Scholar] [CrossRef]
- Qin, T.; Tian, Q.Z.; Wang, G.F.; Xiong, L.M. LOWER TEMPERATURE 1 Enhances ABA responses and plant drought tolerance by modulating the stability and localization of C2-Domain ABA-related proteins in Arabidopsis. Mol. Plant 2019, 12, 1243–1258. [Google Scholar] [CrossRef]
- Soria-Garci, A.I.; Rubio, M.A.C.; Lagunas, B.; Li Pez-Gomolli, N.S.; Luji, N.M.; Di Az-Guerra, R.L.; Picorel, R.; Alfonso, M. Tissue distribution and specific contribution of Arabidopsis FAD7 and FAD8 plastid desaturases to the JA- and ABA-mediated cold stress or defense responses. Plant Cell Physiol. 2019, 60, 1025–1040. [Google Scholar] [CrossRef] [PubMed]
- Ahres, M.; Palmai, T.; Kovacs, T.; Kovacs, L.; Lacek, J.; Vankova, R.; Galiba, G.; Borbely, P. The Effect of White Light Spectrum Modifications by Excess of Blue Light on the Frost Tolerance, Lipid- and Hormone Composition of Barley in the Early Pre-Hardening Phase. Plants 2022, 12, 40. [Google Scholar] [CrossRef] [PubMed]
- Kazan, K. Diverse roles of jasmonates and ethylene in abiotic stress tolerance. Trends Plant Sci. 2015, 20, 219–229. [Google Scholar] [CrossRef]
- Wang, X.; Li, Q.; Xie, J.; Huang, M.; Cai, J.; Zhou, Q.; Dai, T.; Jiang, D. Abscisic acid and jasmonic acid are involved in drought priming-induced tolerance to drought in wheat. Crop J. 2021, 9, 120–132. [Google Scholar] [CrossRef]
- Hu, Y.; Jiang, L.; Wang, F.; Yu, D. Jasmonate regulates the inducer of CBF expression-C-repeat binding factor/DRE binding factor1 cascade and freezing tolerance in Arabidopsis. Plant Cell 2013, 25, 2907–2924. [Google Scholar] [CrossRef]
- Wang, W.; Wang, X.; Huang, M.; Cai, J.; Zhou, Q.; Dai, T.; Cao, W.; Jiang, D. Hydrogen peroxide and abscisic acid mediate salicylic acid-induced freezing tolerance in wheat. Front Plant Sci. 2018, 9, 1137. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; An, C.F.; Park, S.; Gilmour, S.J.; Wang, L.; Renna, L.; Brandizzi, F.; Grumet, R.; Thomashow, M.F. CAMTA-mediated regulation of salicylic acid immunity pathway genes in Arabidopsis exposed to low temperature and pathogen infection. Plant Cell 2017, 29, 2465–2477. [Google Scholar] [CrossRef]
- Meier, F.; Brunner, A.D.; Koch, S.; Koch, H.; Lubeck, M.; Krause, M.; Goedecke, N.; Decker, J.; Kosinski, T.; Park, M.A.; et al. Online Parallel accumulation-serial fragmentation (pasef) with a novel trapped ion mobility mass spectrometer. Mol. Cell Proteom. 2018, 17, 2534–2545. [Google Scholar] [CrossRef]
- Yobi, A.; Bagaza, C.; Batushansky, A.; Shrestha, V.; Emery, M.L.; Holden, S.; Turner-Hissong, S.; Miller, N.D.; Mawhinney, T.P.; Angelovici, R. The complex response of free and bound amino acids to water stress during the seed setting stage in Arabidopsis. Plant J. 2020, 102, 838–855. [Google Scholar] [CrossRef]
- Yu, L.; Iqbal, S.; Zhang, Y.; Zhang, G.; Ali, U.; Lu, S.; Yao, X.; Guo, L. Proteome-wide identification of S-sulphenylated cysteines in Brassica napus. Plant Cell Environ. 2021, 44, 3571–3582. [Google Scholar] [CrossRef]
- Marmiroli, N.; Terzi, V.; Odoardi Stanca, M.; Lorenzoni, C.; Stanca, A.M. Protein synthesis during cold shock in barley tissues: Comparison of two genotypes with winter and spring growth habit. Theor. Appl. Genet. 1986, 73, 190–196. [Google Scholar] [CrossRef]
- Zhu, X.; Liu, S.; Sun, L.; Song, F.; Liu, F.; Li, X. Cold tolerance of photosynthetic electron transport system is enhanced in wheat plants grown under elevated CO2. Front. Plant Sci. 2018, 9, 933. [Google Scholar] [CrossRef]
- Chen, C.; Chen, H.; Zhang, Y.; Thomas, H.R.; Frank, M.H.; He, Y.; Xia, R. TBtools: An integrative toolkit developed for interactive analyses of big biological data. Mol. Plant 2020, 13, 1194–1202. [Google Scholar] [CrossRef] [PubMed]
- Ritonga, F.N.; Chen, S. Physiological and molecular mechanism involved in cold stress tolerance in plants. Plants 2020, 9, 560. [Google Scholar] [CrossRef] [PubMed]
- Fimognari, L.; Dolker, R.; Kaselyte, G.; Jensen, C.N.G.; Akhtar, S.S.; Grosskinsky, D.K.; Roitsch, T. Simple semi-high throughput determination of activity signatures of key antioxidant enzymes for physiological phenotyping. Plant Methods 2020, 16, 42. [Google Scholar] [CrossRef]
- Jammer, A.; Gasperl, A.; Luschin-Ebengreuth, N.; Heyneke, E.; Chu, H.; Cantero-Navarro, E.; Grosskinsky, D.K.; Albacete, A.A.; Stabentheiner, E.; Franzaring, J.; et al. Simple and robust determination of the activity signature of key carbohydrate metabolism enzymes for physiological phenotyping in model and crop plants. J. Exp. Bot. 2015, 66, 5531–5542. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, D.; Hu, J.Y.; Fernandez, M.G.S. Genetic architecture of photosynthesis in Sorghum bicolor under non-stress and cold stress conditions. J. Exp. Bot. 2017, 68, 4545–4557. [Google Scholar] [CrossRef] [PubMed]
- Kosova, K.; Prasil, I.T.; Vitamvas, P.; Dobrev, P.; Motyka, V.; Flokova, K.; Novak, O.; Tureckova, V.; Rolcik, J.; Pesek, B.; et al. Complex phytohormone responses during the cold acclimation of two wheat cultivars differing in cold tolerance, winter Samanta and spring Sandra. J. Plant Physiol. 2012, 169, 567–576. [Google Scholar] [CrossRef]
- Zhang, J.; Yuan, H.; Yang, Y.; Fish, T.; Lyi, S.M.; Thannhauser, T.W.; Zhang, L.; Li, L. Plastid ribosomal protein S5 is involved in photosynthesis, plant development, and cold stress tolerance in Arabidopsis. J. Exp. Bot. 2016, 67, 2731–2744. [Google Scholar] [CrossRef] [PubMed]
- Huner, N.P.; Oquist, G.; Hurry, V.M.; Krol, M.; Falk, S.; Griffith, M. Photosynthesis, photoinhibition and low temperature acclimation in cold tolerant plants. Photosynth. Res. 1993, 37, 19–39. [Google Scholar] [CrossRef] [PubMed]
- Murchie, E.H.; Lawson, T. Chlorophyll fluorescence analysis: A guide to good practice and understanding some new applications. J. Exp. Bot. 2013, 64, 3983–3998. [Google Scholar] [CrossRef] [PubMed]
- Rizza, F.; Pagani, D.; Stanca, A.M.; Cattivelli, L. Use of chlorophyll fluorescence to evaluate the cold acclimation and freezing tolerance of winter and spring oats. Plant Breed. 2001, 120, 389–396. [Google Scholar] [CrossRef]
- Akar, T.; Francia, E.; Tondelli, A.; Rizza, F.; Stanca, A.M.; Pecchioni, N. Marker-assisted characterization of frost tolerance in barley (Hordeum vulgare L.). Plant Breed. 2009, 128, 381–386. [Google Scholar] [CrossRef]
- Bussotti, F.; Desotgiu, R.; Cascio, C.; Pollastrini, M.; Gravano, E.; Gerosa, G.; Marzuoli, R.; Nali, C.; Lorenzini, G.; Salvatori, E.; et al. Ozone stress in woody plants assessed with chlorophyll a fluorescence. A critical reassessment of existing data. Environ. Exp. Bot. 2011, 73, 19–30. [Google Scholar] [CrossRef]
- Cascio, C.; Schaub, M.; Novak, K.; Desotgiu, R.; Bussotti, F.; Strasser, R.J. Foliar responses to ozone of Fagus sylvatica L. seedlings grown in shaded and in full sunlight conditions. Environ. Exp. Bot. 2010, 68, 188–197. [Google Scholar] [CrossRef]
- Zivcak, M.; Brestic, M.; Kalaji, H.M.; Govindjee. Photosynthetic responses of sun- and shade-grown barley leaves to high light: Is the lower PSII connectivity in shade leaves associated with protection against excess of light? Photosynth. Res. 2014, 119, 339–354. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.E.; Thomas, D.J.; Tucker, D.E.; Herbert, S.K. The effects of photooxidative stress on photosystem I measured in vivo in Chlamydomonas. Plant Cell Environ. 1997, 20, 1451–1461. [Google Scholar] [CrossRef]
- Sonoike, K. The different roles of chilling temperatures in the photoinhibition of photosystem I and photosystem II. J. Photochem. Photobiol. B Biol. 1999, 48, 136–141. [Google Scholar] [CrossRef]
- Shao, R.X.; Xin, L.F.; Zheng, H.F.; Li, L.L.; Ran, W.L.; Mao, J.; Yang, Q.H. Changes in chloroplast ultrastructure in leaves of drought-stressed maize inbred lines. Photosynthetica 2016, 54, 74–80. [Google Scholar] [CrossRef]
- Liu, L.; Li, S.; Guo, J.; Li, N.; Jiang, M.; Li, X. Low temperature tolerance is depressed in wild-type and abscisic acid-deficient mutant barley grown in Cd-contaminated soil. J. Hazard. Mater. 2022, 430, 128489. [Google Scholar] [CrossRef] [PubMed]
- Ahres, M.; Palmai, T.; Gierczik, K.; Dobrev, P.; Vankova, R.; Galiba, G. The Impact of Far-Red Light Supplementation on Hormonal Responses to Cold Acclimation in Barley. Biomolecules 2021, 11, 450. [Google Scholar] [CrossRef] [PubMed]
- Kempa, S.; Krasensky, J.; Dal Santo, S.; Kopka, J.; Jonak, C. A central role of abscisic acid in stress-regulated carbohydrate metabolism. PLoS ONE 2008, 3, e3935. [Google Scholar] [CrossRef]
- Liu, L.; Cang, J.; Yu, J.; Wang, X.; Huang, R.; Wang, J.; Lu, B. Effects of exogenous abscisic acid on carbohydrate metabolism and the expression levels of correlative key enzymes in winter wheat under low temperature. Biosci. Biotechnol. Biochem. 2013, 77, 516–525. [Google Scholar] [CrossRef]
- Pan, X.; Guan, L.; Lei, K.; Li, J.; Zhang, X. Transcriptional and physiological data revealed cold tolerance in a photo-thermo sensitive genic male sterile line Yu17S. BMC Plant Biol. 2022, 22, 44. [Google Scholar] [CrossRef]
- Roitsch, T.; Gonzalez, M.C. Function and regulation of plant invertases: Sweet sensations. Trends Plant Sci. 2004, 9, 606–613. [Google Scholar] [CrossRef]
- Farci, D.; Collu, G.; Kirkpatrick, J.; Esposito, F.; Piano, D. RhVI1 is a membrane-anchored vacuolar invertase highly expressed in Rosa hybrida L. petals. J. Exp. Bot. 2016, 67, 3303–3312. [Google Scholar] [CrossRef]
- Perez, L.; Alves, R.; Perez-Fons, L.; Albacete, A.; Farre, G.; Soto, E.; Vilaprinyo, E.; Martinez-Andujar, C.; Basallo, O.; Fraser, P.D.; et al. Multilevel interactions between native and ectopic isoprenoid pathways affect global metabolism in rice. Transgenic Res. 2022, 31, 249–268. [Google Scholar] [CrossRef]
- Snider, J.L.; Pilon, C.; Hu, W.; Wang, H.M.; Tishchenko, V.; Slaton, W.; Chastain, D.; Parkash, V. Net photosynthesis acclimates to low growth temperature in cotton seedlings by shifting temperature thresholds for photosynthetic component processes and respiration. Environ. Exp. Bot. 2022, 196, 104816. [Google Scholar] [CrossRef]
- Eveland, A.L.; Jackson, D.P. Sugars, signalling, and plant development. J. Exp. Bot. 2012, 63, 3367–3377. [Google Scholar] [CrossRef]
- Kelly, G.; Moshelion, M.; David-Schwartz, R.; Halperin, O.; Wallach, R.; Attia, Z.; Belausov, E.; Granot, D. Hexokinase mediates stomatal closure. Plant J. 2013, 75, 977–988. [Google Scholar] [CrossRef] [PubMed]
- Sobczyk, E.A.; Kacperskapalacz, A. Adenine-nucleotide changes during cold-acclimation of winter rape plants. Plant Physiol. 1978, 62, 875–878. [Google Scholar] [CrossRef]
- Sturm, A.; Tang, G.Q. The sucrose-cleaving enzymes of plants are crucial for development, growth and carbon partitioning. Trends Plant Sci. 1999, 4, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; He, Y.; Zhu, M.; Ahmad, A.; Xu, S.; He, Z.; Jiang, S.; Huang, J.; Li, Z.; Liu, S.; et al. ipa1 improves rice drought tolerance at seedling stage mainly through activating abscisic acid pathway. Plant Cell Rep. 2022, 41, 221–232. [Google Scholar] [CrossRef]
- Kleczkowski, L.A. Glucose activation and metabolism through UDP-glucose pyrophosphorylase in plants. Phytochemistry 1994, 37, 1507–1515. [Google Scholar] [CrossRef]
- Xing, C.H.; Liu, Y.; Zhao, L.Y.; Zhang, S.L.; Huang, X.S. A novel MYB transcription factor regulates ascorbic acid synthesis and affects cold tolerance. Plant Cell Environ. 2019, 42, 832–845. [Google Scholar] [CrossRef]
- Li, X.; Tan, D.X.; Jiang, D.; Liu, F. Melatonin enhances cold tolerance in drought-primed wild-type and abscisic acid-deficient mutant barley. J. Pineal Res. 2016, 61, 328–339. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Cang, J.; Lu, Q.; Fan, B.; Xu, Q.; Li, W.; Wang, X. ABA enhanced cold tolerance of wheat ‘dn1’ via increasing ROS scavenging system. Plant Signal. Behav. 2020, 15, 1780403. [Google Scholar] [CrossRef]
- Asada, K. The water-water cycle in chloroplasts: Scavenging of active oxygens and dissipation of excess photons. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 601–639. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.Y.; Hsu, Y.T.; Tsai, Y.C.; Kao, C.H. Expression of ASCORBATE PEROXIDASE 8 in roots of rice (Oryza sativa L.) seedlings in response to NaCl. J. Exp. Bot. 2007, 58, 3273–3283. [Google Scholar] [CrossRef]
- Ding, H.; Wang, B.; Han, Y.; Li, S. The pivotal function of dehydroascorbate reductase in glutathione homeostasis in plants. J Exp. Bot. 2020, 71, 3405–3416. [Google Scholar] [CrossRef] [PubMed]
- Elakhdar, A.; Slaski, J.J.; Kubo, T.; Hamwieh, A.; Hernandez Ramirez, G.; Beattie, A.D.; Capo-Chichi, L.J.A. Genome-wide association analysis provides insights into the genetic basis of photosynthetic responses to low-temperature stress in spring barley. Front. Plant Sci. 2023, 14, 1159016. [Google Scholar] [CrossRef]
- Chen, Q.; Hu, T.; Li, X.; Song, C.P.; Zhu, J.K.; Chen, L.; Zhao, Y. Phosphorylation of SWEET sucrose transporters regulates plant root:shoot ratio under drought. Nat. Plants 2022, 8, 68–77. [Google Scholar] [CrossRef]
- Sharma, M.; Laxmi, A. Jasmonates: Emerging players in controlling temperature stress tolerance. Front. Plant Sci. 2015, 6, 1129. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Guo, Z.; Li, H.; Wang, M.; Onac, E.; Zhou, J.; Xia, X.; Shi, K.; Yu, J.; Zhou, Y. Phytochrome A and B function antagonistically to regulate cold tolerance via abscisic acid-dependent jasmonate signaling. Plant Physiol. 2016, 170, 459–471. [Google Scholar] [CrossRef]
- Grosskinsky, D.K.; Albacete, A.; Jammer, A.; Krbez, P.; van der Graaff, E.; Pfeifhofer, H.; Roitsch, T. A rapid phytohormone and phytoalexin screening method for physiological phenotyping. Mol. Plant 2014, 7, 1053–1056. [Google Scholar] [CrossRef]
- Garcia-Andrade, J.; Gonzalez, B.; Gonzalez-Guzman, M.; Rodriguez, P.L.; Vera, P. The role of ABA in plant immunity is mediated through the PYR1 receptor. Int. J. Mol. Sci. 2020, 21, 5852. [Google Scholar] [CrossRef] [PubMed]
- Walker-Simmons, M.; Kudrna, D.A.; Warner, R.L. Reduced accumulation of ABA during water stress in a molybdenum cofactor mutant of barley. Plant Physiol. 1989, 90, 728–733. [Google Scholar] [CrossRef]
- International Barley Genome Sequencing, C.; Mayer, K.F.; Waugh, R.; Brown, J.W.; Schulman, A.; Langridge, P.; Platzer, M.; Fincher, G.B.; Muehlbauer, G.J.; Sato, K.; et al. A physical, genetic and functional sequence assembly of the barley genome. Nature 2012, 491, 711–716. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Fales, F. The assimilation and degradation of carbohydrates by yeast cells. J. Biol. Chem. 1951, 193, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Shi, G. Dong, X. Techniques of Plant Physiological Experiment; China Agriculture Science and Technology Press: Beijing, China, 2002; pp. 84–85. (In Chinese) [Google Scholar]
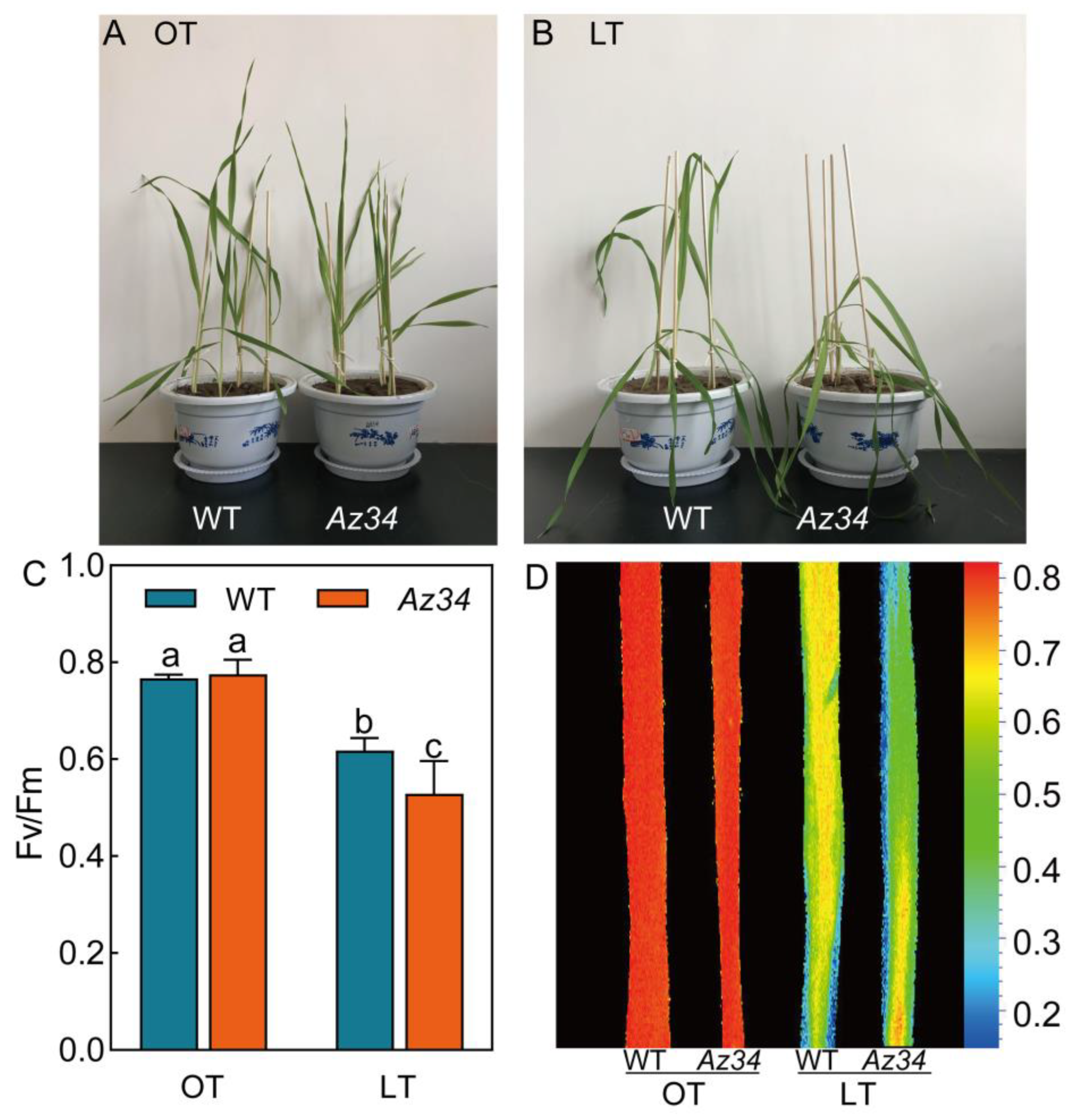



| Treatment | An (µmol CO2 m−2 s−1) | gs (mmol m−2 s−1) | φPo | ψEo | φEo | φRo | |
|---|---|---|---|---|---|---|---|
| OT | WT | 27.80 ± 0.66 a | 271.90 ± 7.86 a | 0.77 ± 0.01 a | 0.57 ± 0.02 ab | 0.44 ± 0.02 ab | 0.19 ± 0.01 bc |
| Az34 | 27.70 ± 0.60 a | 264.67 ± 4.66 a | 0.78 ± 0.03 a | 0.65 ± 0.08 a | 0.51 ± 0.09 a | 0.26 ± 0.06 ab | |
| LT | WT | 17.50 ± 0.35 c | 205.83 ± 8.90 b | 0.53 ± 0.07 b | 0.60 ± 0.09 b | 0.32 ± 0.09 b | 0.29 ± 0.04 a |
| Az34 | 14.63 ± 0.77 b | 234.90 ± 4.74 c | 0.62 ± 0.03 c | 0.51 ± 0.05 ab | 0.32 ± 0.05 b | 0.14 ± 0.04 c |
| Factor | G6PDH | AGPase | UGPase | HXK | Ald | PGI | PGM | PFK | vacInv | cwInv | cytInv | Susy | FK |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| FG | ns | * | * | ns | ** | * | ** | ns | *** | ** | ns | ns | ** |
| LT | ** | ns | * | * | ** | ns | ** | *** | *** | ns | *** | ns | ns |
| FG × LT | ns | * | * | * | ns | ns | *** | ns | *** | ** | *** | ns | ns |
| Factor | APX | CAT | cwPOX | GR | GST | POX | DHAR | MDHAR | SOD |
|---|---|---|---|---|---|---|---|---|---|
| FG | ** | ns | ns | ns | ns | ns | ns | *** | *** |
| LT | ** | ns | ns | ns | ns | ns | * | ns | *** |
| FG × LT | ns | ns | ns | ns | ns | ns | ns | ns | *** |
| Factor | ABA | SA | JA | MeSA | IAA | IBA | IPA | Zeatin |
|---|---|---|---|---|---|---|---|---|
| FG | *** | *** | *** | ** | ns | *** | ns | *** |
| LT | *** | ** | *** | ns | *** | ** | *** | *** |
| FG × LT | ** | *** | ** | ns | ns | *** | *** | *** |
| Fluorescence Parameters | Illustrations |
|---|---|
| Fo ≌ F20 μs | Minimal fluorescence, when all PSII RCs are open |
| Fv =Ft − Fo | Variable fluorescence at time t |
| Fm = FP | Maximal recorded fluorescence intensity, at the peak P of OJIP |
| φPo = TRo/ABS = 1 − Fo/FM = Fv/Fm | Maximum quantum yield for PS II primary photochemistry att = 0 |
| ψEo = ETO/TRO | Probability that an electron moves further than QA |
| φEo = ETO/ABS = (1 − FO/FM) × (1 − VJ) | Quantum yield for PS II electron transport at t = 0 (ET) |
| φRo = REO/ABS | Quantum yield for reduction of end electronacceptors at the PSI acceptor side (RE) |
| Compounds | Ion Model | Parental Ion (M/Z) | Daughter Ion (M/Z) | Fragmentor Voltage (V) | Collision Energy (V) |
|---|---|---|---|---|---|
| ABA | Negative | 263.1 | 153.0/204.2 | −60 | −14/−27 |
| IAA | Positive | 176.2 | 129.8/102.9 | 65 | 12/42 |
| IBA | Negative | 202 | 116.0/158.0 | −80 | −20/−18 |
| IPA | Positive | 336.2 | 204.2/136.3 | 59 | 28/32 |
| JA | Negative | 209.2 | 58.9 | −54 | −16 |
| MeSA | Negative | 153.0 | 121.0/93.1 | −131 | −24/−39 |
| SA | Negative | 137 | 92.6/65 | −50 | −20/−39 |
| Zeatin | Positive | 220.4 | 136.0/147.9 | 92 | 22/16 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, J.; Beemster, G.T.S.; Liu, F.; Wang, Z.; Li, X. Abscisic Acid Regulates Carbohydrate Metabolism, Redox Homeostasis and Hormonal Regulation to Enhance Cold Tolerance in Spring Barley. Int. J. Mol. Sci. 2023, 24, 11348. https://doi.org/10.3390/ijms241411348
Guo J, Beemster GTS, Liu F, Wang Z, Li X. Abscisic Acid Regulates Carbohydrate Metabolism, Redox Homeostasis and Hormonal Regulation to Enhance Cold Tolerance in Spring Barley. International Journal of Molecular Sciences. 2023; 24(14):11348. https://doi.org/10.3390/ijms241411348
Chicago/Turabian StyleGuo, Junhong, Gerrit T. S. Beemster, Fulai Liu, Zongming Wang, and Xiangnan Li. 2023. "Abscisic Acid Regulates Carbohydrate Metabolism, Redox Homeostasis and Hormonal Regulation to Enhance Cold Tolerance in Spring Barley" International Journal of Molecular Sciences 24, no. 14: 11348. https://doi.org/10.3390/ijms241411348
APA StyleGuo, J., Beemster, G. T. S., Liu, F., Wang, Z., & Li, X. (2023). Abscisic Acid Regulates Carbohydrate Metabolism, Redox Homeostasis and Hormonal Regulation to Enhance Cold Tolerance in Spring Barley. International Journal of Molecular Sciences, 24(14), 11348. https://doi.org/10.3390/ijms241411348







