Molecular Mapping to Discover Reliable Salinity-Resilient QTLs from the Novel Landrace Akundi in Two Bi-Parental Populations Using SNP-Based Genome-Wide Analysis in Rice
Abstract
1. Introduction
2. Results
2.1. Salt Stress Reactions of the Selected F2:3 Progenies and the Parental Genotypes
2.2. Assessing Agronomic Characters under Salt Stress
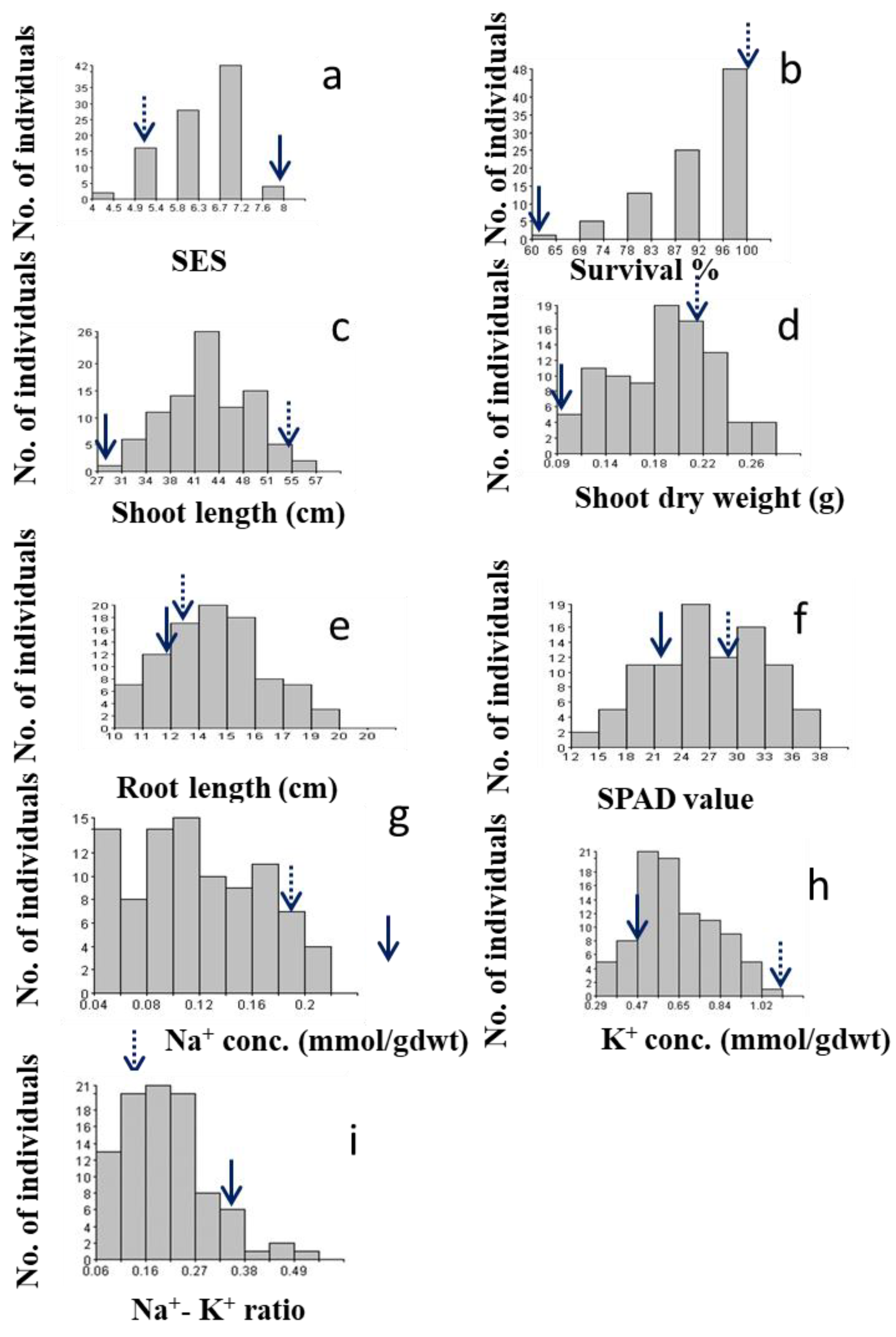
2.2.1. SES Score
2.2.2. Survival
2.2.3. Shoot Length
2.2.4. Shoot Dry Weight
2.2.5. Root Length
2.3. Characterizing Physiological Parameters under Salt Stress
2.3.1. SPAD Value
2.3.2. Na+ Concentration
2.3.3. K+ Concentration
2.3.4. Na+/K+ Ratio
2.4. Trait Correlation Analysis between Different Characters
2.5. Determining the Contribution of Component Agronomic and Physiological Traits (Independent Variables) to Overall Phenotypic Performance (SES Score: Dependent Variable) through Path Analysis
2.6. SNP Marker Polymorphism and Construction of Genetic Linkage Map
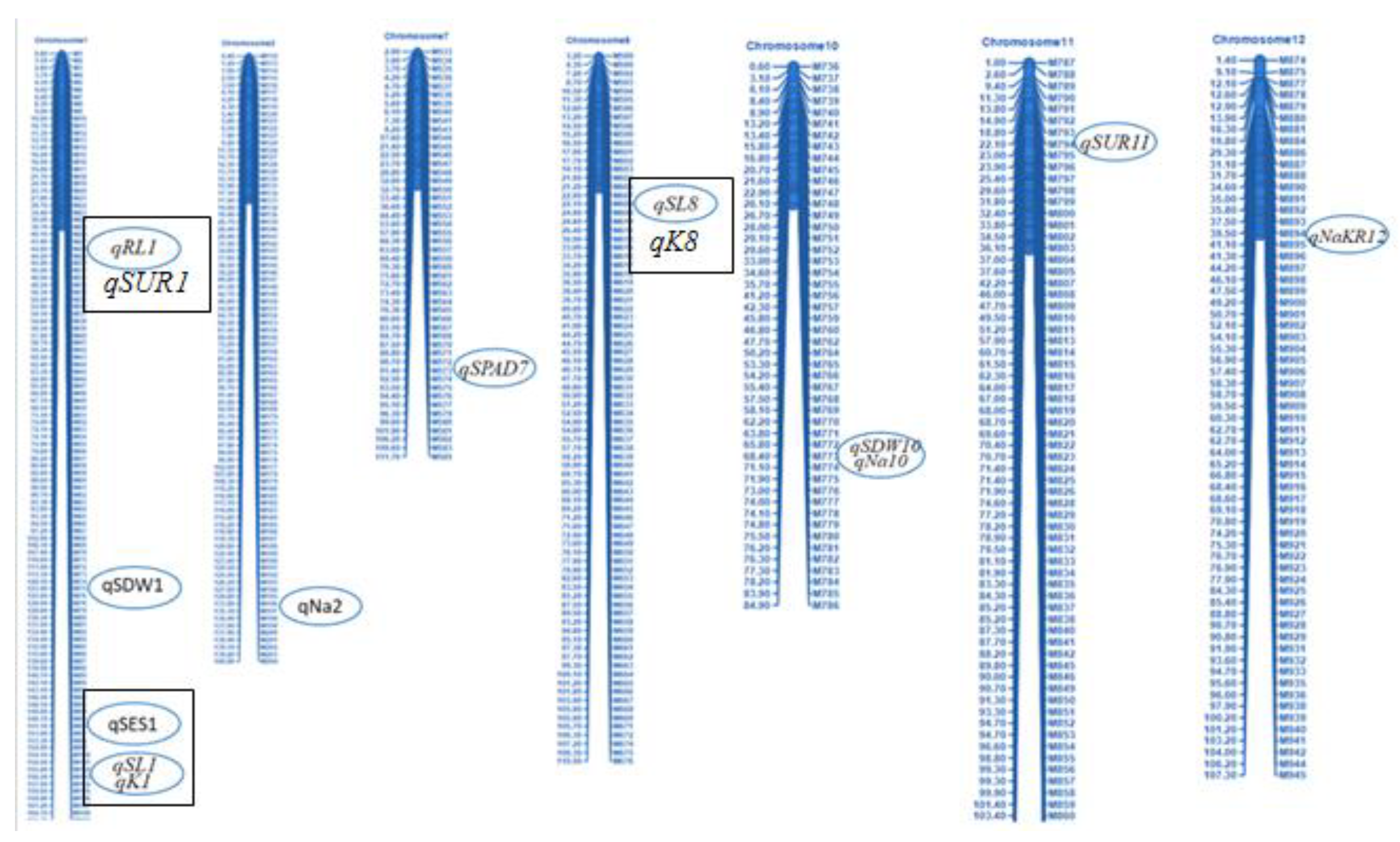
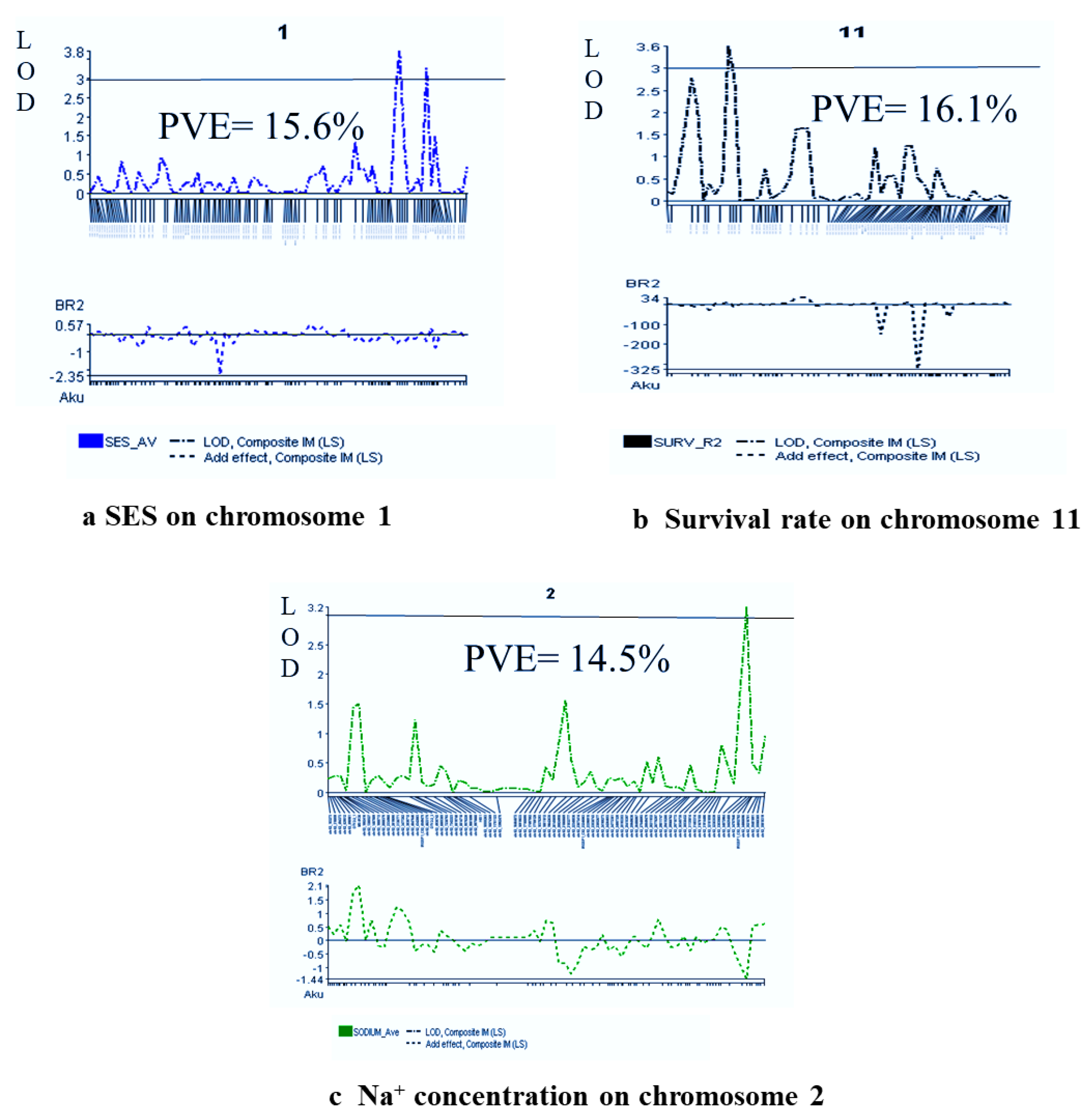
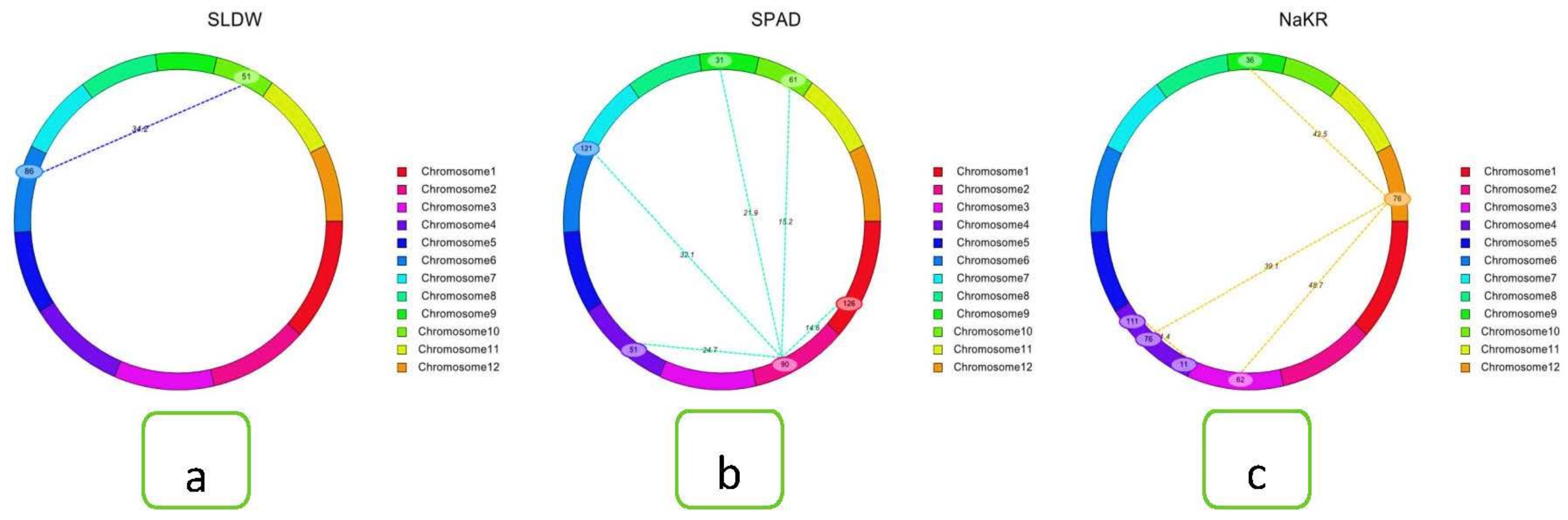
2.7. Salinity-Tolerant QTLs Controlling Agronomic Traits
2.8. QTL Regions Governing Physiological Characters
2.9. Probable Functional Genes Detection in the Different QTL Regions
2.10. Epistatic Interaction
2.11. Stable QTLs for Different Agronomic and Physiological Traits Responsible for Salinity Tolerance
3. Discussion
3.1. Assessing the Salt Stress Responses in the Selected F2:3 Progenies with Their Parents and Determining Cause and Effect Relationships via Path Analysis
3.2. Marker Segregation and Important Salt-Tolerant QTL Regions
3.3. Comparing the QTLs Revealed in the Present Study with Previously Reported QTLs
4. Materials and Methods
4.1. Parent Selection
4.2. Growing Conditions
4.3. Characterizing Agronomic Traits
4.4. Determining Physiological Traits Response
4.5. SPAD Reading
4.6. Analyzing Trait Associations (Correlation)
4.7. Path Coefficient Analysis
4.8. SNP Genotyping and Genetic Linkage Map Construction
4.9. QTL Dissection
4.10. Epistatic QTLs Identification
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yu, J.; Hu, S.; Wang, J.; Wong, G.K.S.; Li, S.; Liu, B.; Deng, Y.; Dai, L.; Zhou, Y.; Zhang, X.; et al. A draft sequence of the rice genome (Oryza sativa L. ssp. indica). Science 2002, 296, 79–92. [Google Scholar] [CrossRef] [PubMed]
- IRGSP (International Rice Genome Sequencing Project); Matsumoto, T. The map-based sequence of the rice genome. Nature 2005, 436, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.S.; Ali, M.A.; Khan, M.A.I.; Maniruzzaman, S. Prevalence and Transmission of Fusarium moniliforme: A Seed Borne Pathogen of Rice. Bangladesh Rice J. 2020, 24, 11–19. [Google Scholar] [CrossRef]
- Hossain, M.S.; Ivy, N.A.; Maniruzzaman, S.; Raihan, M.S.; Ruma, A.S.; Akter, A.; Hasan, M.R.; Kulsum, M.U.; Uddin, M.N.; Kabir, K.F. Genetic Variability of Floral and Agronomic Characteristics that Influence Outcrossing Rate Percentage of Cytoplasmic Male Sterile Rice. Asian J. Adv. Agric. Res. 2023, 22, 46–57. [Google Scholar] [CrossRef]
- Maniruzzaman, S.; Hossain, M.; Ahmed, H.U.; Hossain, M.; Jahan, G.S.; Kundu, P.K.; Haque, M.M. Development of Rice Varieties for Stress-Prone Tidal Ecosystem of Bangladesh. Middle-East J. Sci. Res. 2023, 31, 22–31. [Google Scholar]
- Xu, R.; Yang, Y.; Qin, R.; Li, H.; Qiu, C.; Li, L.; Wei, P.; Yang, J. Rapid improvement of grain weight via highly efficient CRISPR/Cas9-mediated multiplex genome editing in rice. J. Genet. Genom. 2016, 43, 529–532. [Google Scholar] [CrossRef]
- Maniruzzaman, S.; Rahman, M.A.; Hasan, M.; Rasul, M.G.; Molla, A.H.; Khatun, H.; Akter, S. Genetic Mapping to Detect Stringent QTLs Using 1k-RiCA SNP Genotyping Platform from the New Landrace Associated with Salt Tolerance at the Seedling Stage in Rice. Plants 2022, 11, 1409. [Google Scholar] [CrossRef]
- Islam, F.; Wang, J.; Farooq, M.A.; Yang, C.; Jan, M.; Mwamba, T.M.; Hannan, F.; Xu, L.; Zhou, W. Rice responses and tolerance to salt stress: Deciphering the physiological and molecular mechanisms of salinity adaptation. In Advances in Rice Research for Abiotic Stress Tolerance; Woodhead Publishing: Sawston, UK, 2019; pp. 791–819. [Google Scholar]
- Al-Tamimi, N.; Brien, C.; Oakey, H.; Berger, B.; Saade, S.; Ho, Y.S.; Schmöckel, S.M.; Tester, M.; Negrão, S. Salinity tolerance loci revealed in rice using high-throughput non-invasive phenotyping. Nat. Commun. 2016, 7, 13342. [Google Scholar] [CrossRef]
- Rasel, M.; Tahjib-Ul-Arif, M.; Hossain, M.A.; Hassan, L.; Farzana, S.; Brestic, M. Screening of salt-tolerant rice landraces by seedling stage phenotyping and dissecting biochemical determinants of tolerance mechanism. J. Plant Growth Regul. 2021, 40, 1853–1868. [Google Scholar] [CrossRef]
- Rahman, M.A.; Thomson, M.J.; De Ocampo, M.; Egdane, J.A.; Salam, M.A.; Ismail, A.M. Assessing trait contribution and mapping novel QTL for salinity tolerance using the Bangladeshi rice landrace Capsule. Rice 2019, 12, 63. [Google Scholar] [CrossRef]
- Hossain, H.; Rahman, M.A.; Alam, M.S.; Singh, R.K. Mapping of quantitative trait loci associated with reproductive-stage salt tolerance in rice. J. Agron. Crop Sci. 2015, 201, 17–31. [Google Scholar] [CrossRef]
- Goto, I.; Neang, S.; Kuroki, R.; Reyes, V.P.; Doi, K.; Skoulding, N.S.; Taniguchi, M.; Yamauchi, A.; Mitsuya, S. QTL Analysis for Sodium Removal Ability in Rice Leaf Sheaths under Salinity Using an IR-44595/318 F2 Population. Front. Plant Sci. 2022, 13, 1002605. [Google Scholar] [CrossRef]
- Nayyeripasand, L.; Garoosi, G.A.; Ahmadikhah, A. Genome-wide association study (GWAS) to identify salt-tolerance QTLs carrying novel candidate genes in rice during early vegetative stage. Rice 2021, 14, 9. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Thomson, M.J.; Shah-E-Alam, M.; de Ocampo, M.; Egdane, J.; Ismail, A.M. Exploring novel genetic sources of salinity tolerance in rice through molecular and physiological characterization. Ann. Bot. 2016, 117, 1083–1097. [Google Scholar] [CrossRef] [PubMed]
- de Souza Freitas, W.E.; de Oliveira, A.B.; Mesquita, R.O.; de Carvalho, H.H.; Prisco, J.T.; Gomes-Filho, E. Sulfur-induced salinity tolerance in lettuce is due to a better P and K uptake, lower Na/K ratio and an efficient antioxidative defense system. Sci. Hortic. 2019, 257, 108764. [Google Scholar] [CrossRef]
- Singh, R.K.; Kota, S.; Flowers, T.J. Salt tolerance in rice: Seedling and reproductive stage QTL mapping come of age. Theor. Appl. Genet. 2021, 134, 3495–3533. [Google Scholar] [CrossRef]
- Mackay, T.F.; Huang, W. Charting the genotype–phenotype map: Lessons from the Drosophila melanogaster Genetic Reference Panel. Wiley Interdiscip. Rev. Dev. Biol. 2018, 7, e289. [Google Scholar] [CrossRef]
- Rahman, M.A.; Khatun, H.; Sarker, M.R.A.; Hossain, H.; Quddus, M.R.; Iftekharuddaula, K.M.; Kabir, M.S. Enhancing abiotic stress tolerance to develop climate-smart rice using holistic breeding approach. Cereal Grains 2021, 2, 91. [Google Scholar] [CrossRef]
- Kosambi, D.D. The estimation of map distance from recombination values. Annu. Eugen. 1944, 12, 172–175. [Google Scholar] [CrossRef]
- Ismail, A.M.; Horie, T. Genomics, physiology, and molecular breeding approaches for improving salt tolerance. Annu. Rev. Plant Biol. 2017, 68, 405–434. [Google Scholar] [CrossRef]
- Van Zelm, E.; Zhang, Y.; Testerink, C. Salt Tolerance Mechanisms of Plants. Annu. Rev. Plant Biol. 2020, 71, 403–433. [Google Scholar] [CrossRef]
- Gong, Z. Plant abiotic stress: New insights into the factors that activate and modulate plant responses. J. Integr. Plant Biol. 2021, 63, 429–430. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Ye, G.; Wang, J. A modified algorithm for the improvement of composite interval mapping. Genetics 2007, 175, 361–374. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, H.; Li, Z.; Wang, J. Interactions between markers can be caused by the dominance effect of quantitative trait loci. Genetics 2008, 180, 1177–1190. [Google Scholar] [CrossRef]
- Rahman, M.A.; Bimpong, I.K.; Bizimana, J.B.; Pascual, E.D.; Arceta, M.; Swamy, B.P.M.; Diaw, F.; Singh, R.K. Mapping QTLs using a novel source of salinity tolerance from Hasawi and their interaction with environments in rice. Rice 2017, 10, 47. [Google Scholar] [CrossRef] [PubMed]
- Nakhla, W.R.; Sun, W.; Fan, K.; Yang, K.; Zhang, C.; Yu, S. Identification of QTLs for salt tolerance at the germination and seedling stages in rice. Plants 2021, 10, 428. [Google Scholar] [CrossRef]
- Debsharma, S.K.; Syed, A.; Ali, H.; Maniruzzaman, S.; Roy, P.R.; Brestic, M.; Gaber, A.; Hossain, A. Harnessing on Genetic Variability and Diversity of Rice (Oryza sativa L.) Genotypes Based on Quantitative and Qualitative Traits for Desirable Crossing Materials. Genes 2023, 14, 10. [Google Scholar] [CrossRef] [PubMed]
- Rajasekar, R.; Jeyaprakash, P.; Manonmani, K.; Nithila, S.; Thirumurugan, T. Trait relationship and path analysis under sodicity in Nagina 22 rice mutants. Electron. J. Plant Breed. 2021, 12, 963–968. [Google Scholar]
- Thomson, M.J.; de Ocampo, M.; Egdane, J.; Rahman, M.A.; Sajise, A.G.; Adorada, D.L.; Tumimbang-Raiz, E.; Blumwald, E.; Seraj, Z.I.; Singh, R.K.; et al. Characterizing the Saltol quantitative trait locus for salinity tolerance in rice. Rice 2010, 3, 148–160. [Google Scholar] [CrossRef]
- Manohara, K.K.; Morajkar, S.; Shanbhag, Y.; Phadte, P.; Singh, N.K. Haplotype analysis of Saltol QTL region in diverse landraces, wild rice and introgression lines of rice (Oryza sativa L.). Plant Genet. Resour. 2021, 19, 289–298. [Google Scholar] [CrossRef]
- Ren, Z.H.; Gao, J.P.; Li, L.G.; Cai, X.L.; Huang, W.; Chao, D.Y. A rice quantitative trait locus for salt tolerance encodes a sodium transporter. Nat. Genet. 2005, 37, 1141–1146. [Google Scholar] [CrossRef]
- Kaur, N.; Prashanth, K.V.H.; Bhatti, M.S.; Pati, P.K. OsSalT gene cloned from rice provides evidence of its role in salinity and drought stress tolerance. Plant Sci. 2022, 320, 111306. [Google Scholar] [CrossRef]
- Claes, B.; Dekeyser, R.; Villaroel, R.; Van den Bulcke, M.; Bauw, G.; Van Montagu, M.; Caplan, A. Characterization of rice gene showing organ specific expression in response to salt stress and drought. Plant Cell 1990, 2, 19–27. [Google Scholar] [PubMed]
- Ali, A.; Maggio, A.; Bressan, R.A.; Yun, D.-J. Role and Functional Differences of HKT1-Type Transporters in Plants under Salt Stress. Int. J. Mol. Sci. 2019, 20, 1059. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Guo, Y. Elucidating the molecular mechanisms mediating plant salt-stress responses. New Phytol. 2017, 217, 523–539. [Google Scholar] [CrossRef] [PubMed]
- Sakuraba, Y.; Bülbül, S.; Piao, W.; Choi, G.; Paek, N. Arabidopsis early flowering 3 increases salt tolerance by suppressing salt stress response pathways. Plant J. 2017, 92, 1106–1120. [Google Scholar] [CrossRef]
- Wei, H.; Wang, X.; He, Y.; Xu, H.; Wang, L. Clock component OsPRR73 positively regulates rice salt tolerance by modulating OsHKT2;1-mediated sodium homeostasis. EMBO J. 2021, 40, e105086. [Google Scholar] [CrossRef] [PubMed]
- Ayadi, M.; Brini, F.; Masmoudi, K. Overexpression of a Wheat Aquaporin Gene, TdPIP2;1, Enhances Salt and Drought Tolerance in Transgenic Durum Wheat cv. Maali. Int. J. Mol. Sci. 2019, 20, 2389. [Google Scholar] [CrossRef]
- Jahan, N.; Zhang, Y.; Lv, Y.; Song, M.; Zhao, C.; Hu, H.; Cui, Y.; Wang, Z.; Yang, S.; Zhang, A.; et al. QTL analysis for rice salinity tolerance and fine mapping of a candidate locus qSL7 for shoot length under salt stress. Plant Growth Regul. 2020, 90, 307–319. [Google Scholar] [CrossRef]
- O’connor, K.; Hayes, B.; Hardner, C.; Nock, C.; Baten, A.; Alam, M.; Henry, R.; Topp, B. Genome-wide association studies for yield component traits in a macadamia breeding population. BMC Genom. 2020, 21, 199. [Google Scholar] [CrossRef]
- BRRI. Adhunik Dhaner Chash, 23rd ed.; Bangladesh Rice Research Institute: Gazipur, Bangladesh, 2020.
- Yang, Y.; Guo, Y. Unraveling salt stress signaling in plants. J. Integr. Plant Biol. 2018, 60, 796–804. [Google Scholar] [CrossRef]
- Yoshida, S.; Forno, D.A.; Cock, J.H. Laboratory Manual for Physiological Studies of Rice, 3rd ed.; International Rice Research Institute: Manila, Philippines, 1976. [Google Scholar]
- Zhang, M.; Cao, Y.; Wang, Z.; Wang, Z.-Q.; Shi, J.; Liang, X.; Song, W.; Chen, Q.; Lai, J.; Jiang, C. A retrotransposon in an HKT1 family sodium transporter causes variation of leaf Na+ exclusion and salt tolerance in maize. New Phytol. 2017, 217, 1161–1176. [Google Scholar] [CrossRef]
- Covington, M.F.; Maloof, J.N.; Straume, M.; Kay, S.A.; Harmer, S.L. Global transcriptome analysis reveals circadian regulation of key pathways in plant growth and development. Genome Biol. 2008, 9, R130. [Google Scholar] [CrossRef]
- IRRI. Standard Evaluation System for Rice (SES), 5th ed.; International Rice Research Institute: Los Banos, Philippines, 2014; p. 57. [Google Scholar]
- Flowers, T.J.; Yeo, A.R. Variability in the resistance of sodium chloride salinity within rice (Oryza sativa L.) varieties. New Phytol. 1981, 88, 363–373. [Google Scholar] [CrossRef]
- RStudio Team. RStudio: Integrated Development for R; RStudio PBC: Boston, MA, USA, 2020; Available online: http://www.rstudio.com/ (accessed on 1 May 2021).
- Wright, S. Coefficients of inbreeding and relationship. Am. Nat. 1922, 56, 330–338. [Google Scholar] [CrossRef]
- Dewey, D.R.; Lu, K. A correlation and path-coefficient analysis of components of crested wheatgrass seed production. Agron. J. 1959, 51, 515–518. [Google Scholar] [CrossRef]
- Arbelaez, J.D.; Dwiyanti, M.S.; Tandayu, E. 1k-RiCA (1K-Rice Custom Amplicon) a novel genotyping amplicon-based SNP assay for genetics and breeding applications in rice. Rice 2019, 12, 55. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Presting, G.; Barbazuk, W.B.; Goicoechea, J.L.; Blackmon, B.; Fang, G.; Kim, H.; Frisch, D.; Yu, Y.; Sun, S.; et al. An integrated physical and genetic map of the Rice genome. Plant Cell 2002, 14, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Joehanes, R.; Nelson, J.C. QGene 4.0, an extensible Java QTL-analysis platform. Bioinformatics 2008, 24, 2788–2789. [Google Scholar] [CrossRef]
- Churchill, G.A.; Doerge, R.W. Empirical threshold values for quantitative trait mapping. Genetics 1994, 138, 963–971. [Google Scholar] [CrossRef]
- Li, H.; Ribaut, J.-M.; Li, Z.; Wang, J. Inclusive composite interval mapping (ICIM) for digenic epistasis of quantitative traits in biparental populations. Theor. Appl. Genet. 2008, 116, 243–260. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Li, H.; Zhang, L.; Wang, J. QTL IciMapping: Integrated software for genetic linkage map construction and quantitative trait locus mapping in biparental populations. Crop J. 2015, 3, 269–283. [Google Scholar] [CrossRef]
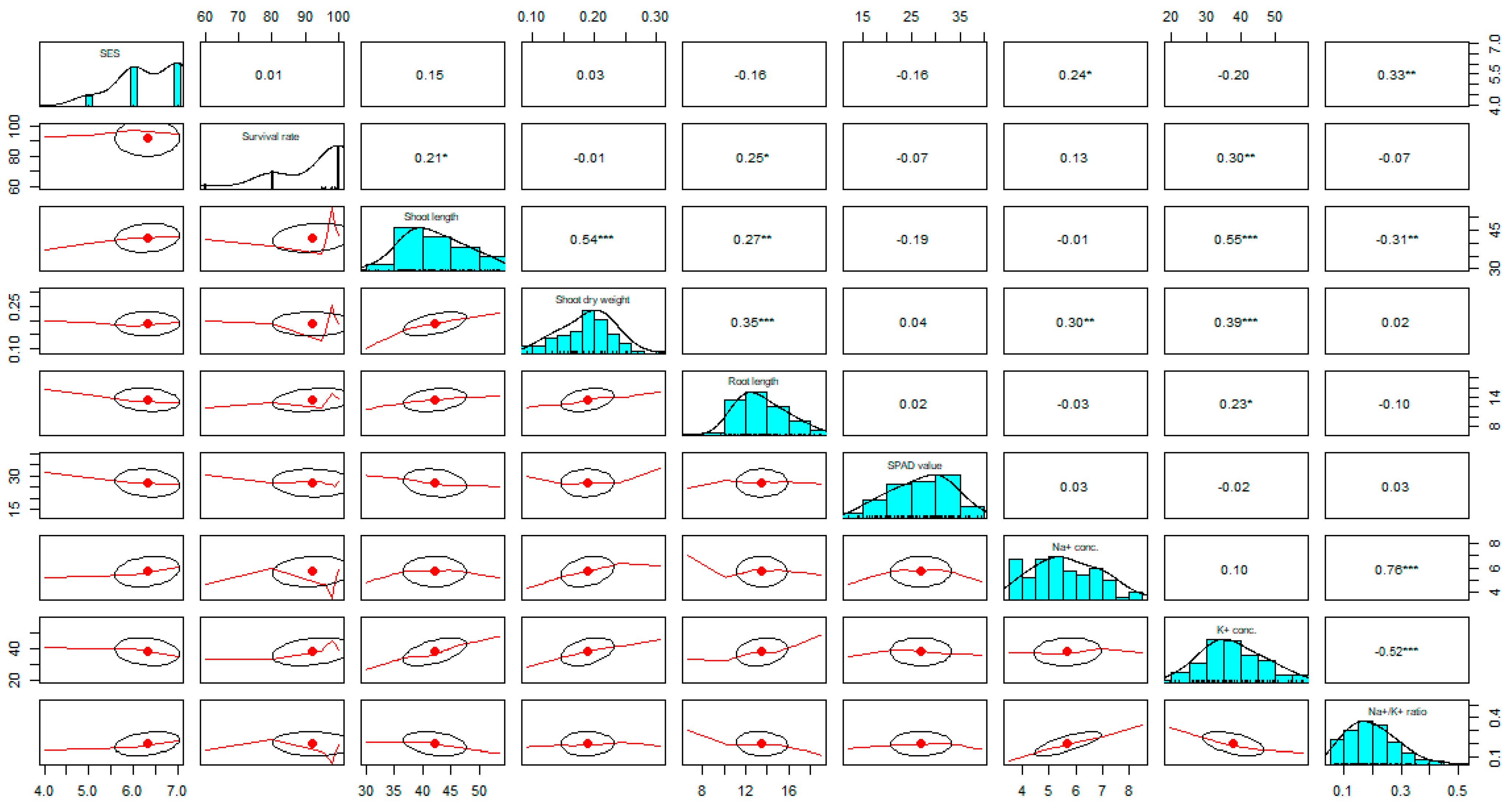

| Variable Name | Correlation | % Survival | Shoot | Root Length | SPAD Value | Na+ Concentration | K+ Concentration | Na+/K+ Ratio | Total Effect | |
|---|---|---|---|---|---|---|---|---|---|---|
| Length | Dry Weight | |||||||||
| % Survival | 0.008 | 0.035 | 0.093 | 0.001 | −0.042 | 0.006 | 0.021 | −0.096 | −0.011 | 0.008 |
| Shoot length | 0.147 | 0.007 | 0.440 | −0.046 | −0.044 | 0.016 | −0.001 | −0.173 | −0.052 | 0.147 |
| Shoot dry weight | 0.018 | 0.001 | 0.235 | −0.086 | −0.058 | −0.004 | 0.049 | −0.121 | 0.003 | 0.018 |
| Root length | −0.161 | 0.009 | 0.118 | −0.030 | −0.164 | −0.001 | −0.004 | −0.072 | −0.017 | −0.161 |
| SPAD value | −0.162 | −0.003 | −0.083 | −0.004 | −0.003 | −0.086 | 0.005 | 0.005 | 0.006 | −0.162 |
| Na+ conc. | 0.239 | 0.004 | −0.002 | −0.025 | 0.004 | −0.003 | 0.164 | −0.031 | 0.127 | 0.239 |
| K+ conc. | −0.201 | 0.011 | 0.243 | −0.033 | −0.038 | 0.001 | 0.016 | −0.314 | −0.087 | −0.201 |
| Na+/K+ ratio | 0.328 | −0.002 | −0.137 | −0.001 | 0.016 | −0.003 | 0.125 | 0.163 | 0.168 | 0.328 |
| Characters | QTL Identified | Chr. | QTL Peak Marker | QTL Position (cM) | Additive Effect | LOD | PVE (%) | QTL Detection Method | Favorable Allele Contributing Parent | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| QGene | ICIM | QGene | ICIM | QGene | ICIM | |||||||
| SES | qSES1 | 1 | chr01_38632196 | 151.8 | 150.8 | −0.61 | 3.4 | 3.0 | 15.6 | 2.59 | IM, CIM | Akundi |
| % Survival | qSUR11 | 11 | chr11_5615885 | 21 | 21 | 13.73 | 3.5 | 4.0 | 16.1 | 0.85 | IM, CIM | BR28 |
| Shoot length | qSL1 | 1 | QSES1-2_2 | 156 | - | 4.86 | 7.3 | - | 30.7 | - | IM, CIM | BR28 |
| qSL8 | 8 | GM4_4 | 22 | - | 4.29 | 3.2 | - | 16.3 | - | IM, CIM | BR28 | |
| Shoot dry weight | qSDW1 | 1 | chr01_231396842 | 123.4 | 137.8 | −0.25 | 3.6 | 6.0 | 16.4 | 13.44 | IM, CIM | Akundi |
| qSDW10 | 10 | chr10_17397576 | 68.6 | 64.6 | −0.2 | 3.6 | 5.0 | 17.1 | 17.31 | IM, CIM | Akundi | |
| Root length | qRL1 | 1 | chr01_1045259 | 42 | - | 1.93 | 3.5 | - | 26 | - | SMA | BR28 |
| SPAD value | qSPAD7 | 7 | AG3_1 | 91.4 | - | −2.32 | 3.0 | - | 12.1 | - | SMR | Akundi |
| Na+ conc. | qNa2 | 2 | chr02_34072964 | 134.4 | - | −1.43 | 3.1 | - | 14.5 | - | IM, CIM | Akundi |
| qNa10 | 10 | chr10_17397576 | 68.8 | 68.6 | −0.74 | 3.3 | 5.0 | 15.5 | 21.20 | IM, CIM | Akundi | |
| K+ conc. | qK1 | 1 | QSES1-2_2 | 156.5 | 156.8 | 0.102 | 4.28 | 4.0 | 19.3 | 18.75 | SMR | BR28 |
| Na+/K+ ratio | qNaKR12 | 12 | chr12_10051752 | 39.4 | - | −0.04 | 3.0 | - | 12.1 | - | CIM | Akundi |
| Trait | Population-1 | Favorable Allele Contributing Parent | Trait | Population-2 | Favorable Allele Contributing Parent | Common and Stable QTLs in Different Genetic Background | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| QTL Detected in Pop. 1 | Chr. No. | QTL Position (cM) | QTL Detected in Pop. 2 | Chr. No. | QTL Position (cM) | |||||
| SES | qSES1 | 1 | 151.8 | Akundi | SES | qSES1 | 1 | 151.8 | Akundi | qSES1 |
| Shoot length | qSL1 | 1 | 156 | BR28 | Shoot length | qSL1 | 1 | 156 | BR49 | qSL1 |
| Root length | qRL1 | 1 | 42 | BR28 | Root length | qRL1 | 1 | 48.8 | Akundi | qRL1 |
| Root length | qRL1 | 1 | 42 | BR28 | Survival (%) | qSUR1 | 1 | 50 | BR49 | qSUR1 |
| Shoot length | qSL8 | 8 | 22 | BR28 | K+ Conc. | qK8 | 8 | 18.8 | Akundi | qSL8 |
| Shoot length | qSL8 | 8 | 22 | BR28 | K+ Conc. | qK8 | 8 | 18.8 | Akundi | qK8 |
| K+ conc. | qK1 | 1 | 156.5 | BR28 | Shoot length | qSL1 | 1 | 156 | BR49 | qK1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maniruzzaman, S.; Rahman, M.A.; Hasan, M.; Rasul, M.G.; Molla, A.H.; Khatun, H.; Iftekharuddaula, K.M.; Kabir, M.S.; Akter, S. Molecular Mapping to Discover Reliable Salinity-Resilient QTLs from the Novel Landrace Akundi in Two Bi-Parental Populations Using SNP-Based Genome-Wide Analysis in Rice. Int. J. Mol. Sci. 2023, 24, 11141. https://doi.org/10.3390/ijms241311141
Maniruzzaman S, Rahman MA, Hasan M, Rasul MG, Molla AH, Khatun H, Iftekharuddaula KM, Kabir MS, Akter S. Molecular Mapping to Discover Reliable Salinity-Resilient QTLs from the Novel Landrace Akundi in Two Bi-Parental Populations Using SNP-Based Genome-Wide Analysis in Rice. International Journal of Molecular Sciences. 2023; 24(13):11141. https://doi.org/10.3390/ijms241311141
Chicago/Turabian StyleManiruzzaman, Sheikh, M. Akhlasur Rahman, Mehfuz Hasan, Mohammad Golam Rasul, Abul Hossain Molla, Hasina Khatun, K. M. Iftekharuddaula, Md. Shahjahan Kabir, and Salma Akter. 2023. "Molecular Mapping to Discover Reliable Salinity-Resilient QTLs from the Novel Landrace Akundi in Two Bi-Parental Populations Using SNP-Based Genome-Wide Analysis in Rice" International Journal of Molecular Sciences 24, no. 13: 11141. https://doi.org/10.3390/ijms241311141
APA StyleManiruzzaman, S., Rahman, M. A., Hasan, M., Rasul, M. G., Molla, A. H., Khatun, H., Iftekharuddaula, K. M., Kabir, M. S., & Akter, S. (2023). Molecular Mapping to Discover Reliable Salinity-Resilient QTLs from the Novel Landrace Akundi in Two Bi-Parental Populations Using SNP-Based Genome-Wide Analysis in Rice. International Journal of Molecular Sciences, 24(13), 11141. https://doi.org/10.3390/ijms241311141







