Probing Transcriptional Crosstalk between Cryptochromes and Iron-sulfur Cluster Assembly 1 (MagR) in the Magnetoresponse of a Migratory Insect
Abstract
1. Introduction
2. Results
2.1. Transcriptional Responses of the Putative Magnetosensing Genes to Magnetic Field Intensity Changes
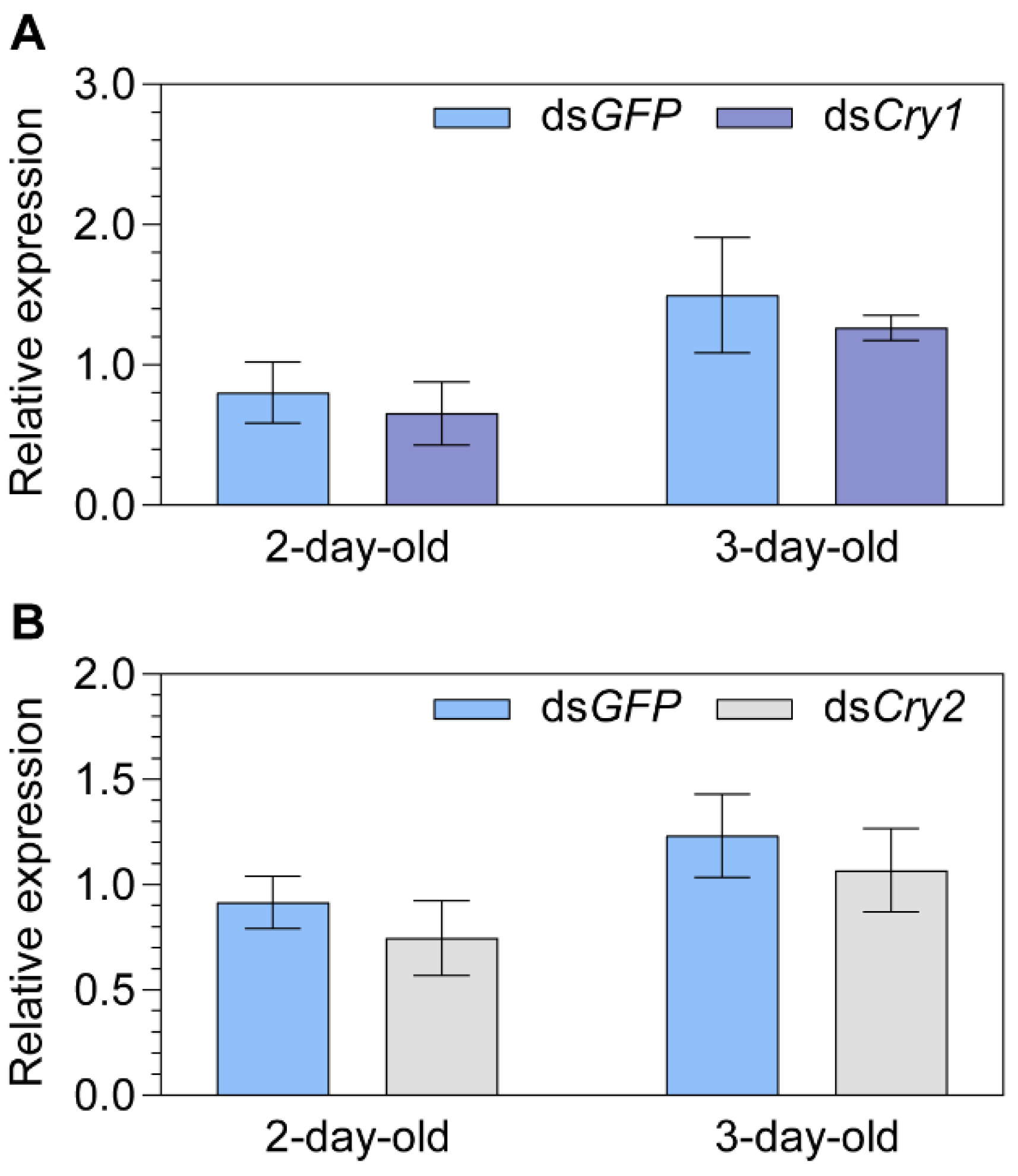
2.2. Transcript Expression of Cryptochromes after Knocking down the Putative Magnetoreceptor Iron-sulfur Cluster Assembly 1 (MagR)
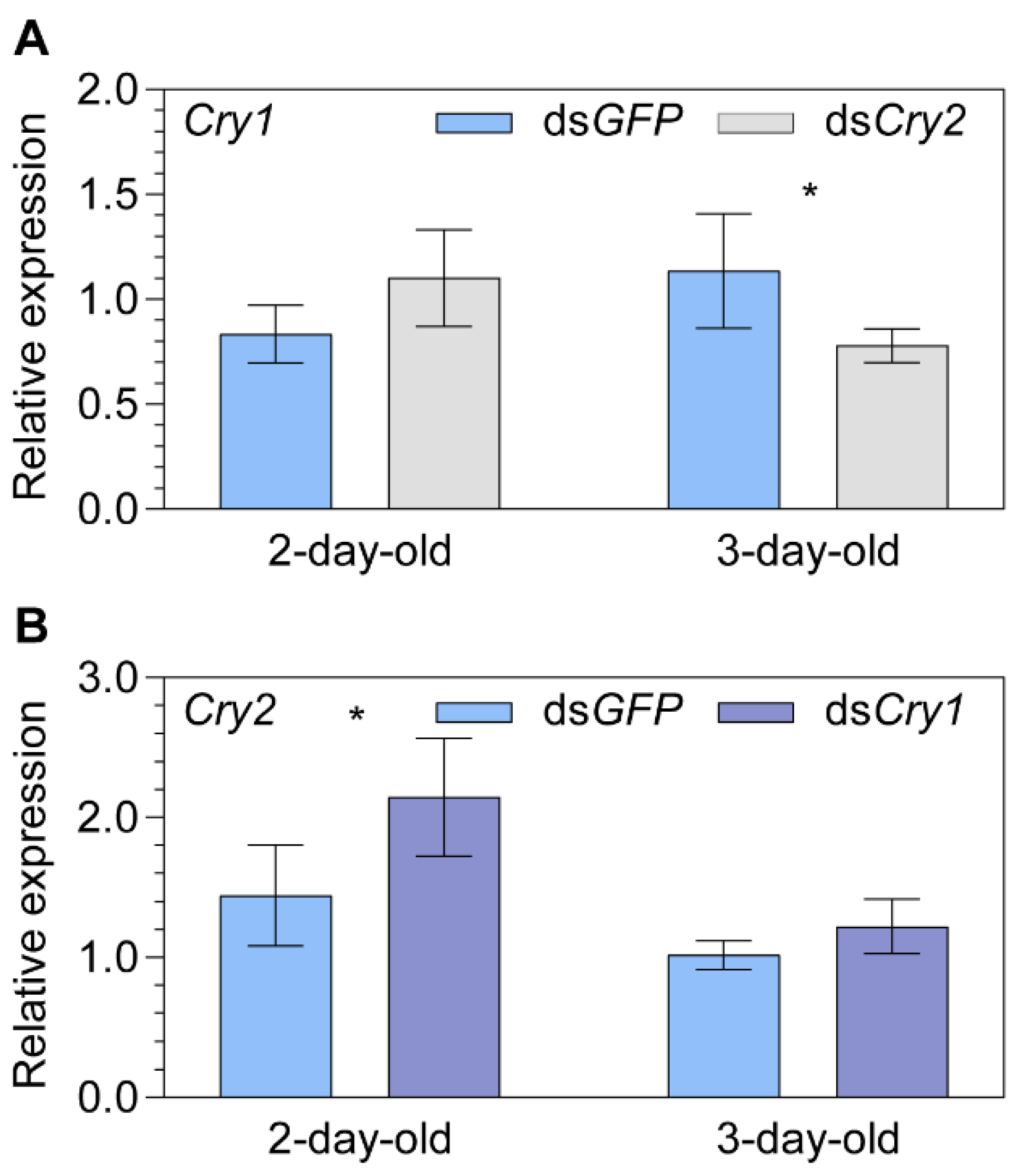
2.3. Transcript Expression of the Putative Magnetoreceptor Iron-sulfur Cluster Assembly 1 after Knocking down Cryptochrome 1 or Cryptochrome 2
2.4. Potential Transcriptional Interactions between Cryptochrome 1 and Cryptochrome 2
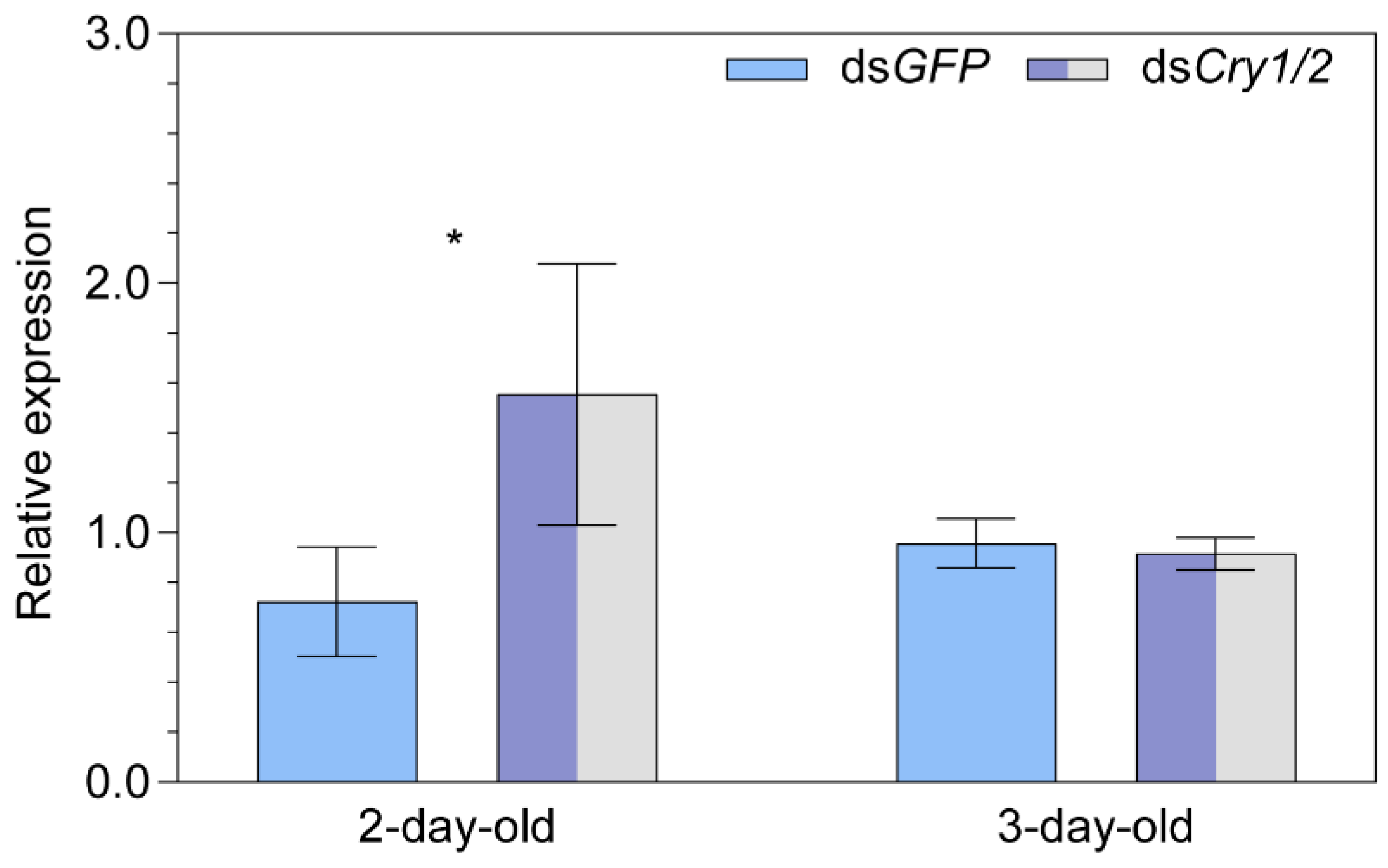
2.5. Transcript Expression of the Putative Magnetoreceptor Iron-sulfur Cluster Assembly 1 after Knocking down Both Cryptochrome 1 and Cryptochrome 2
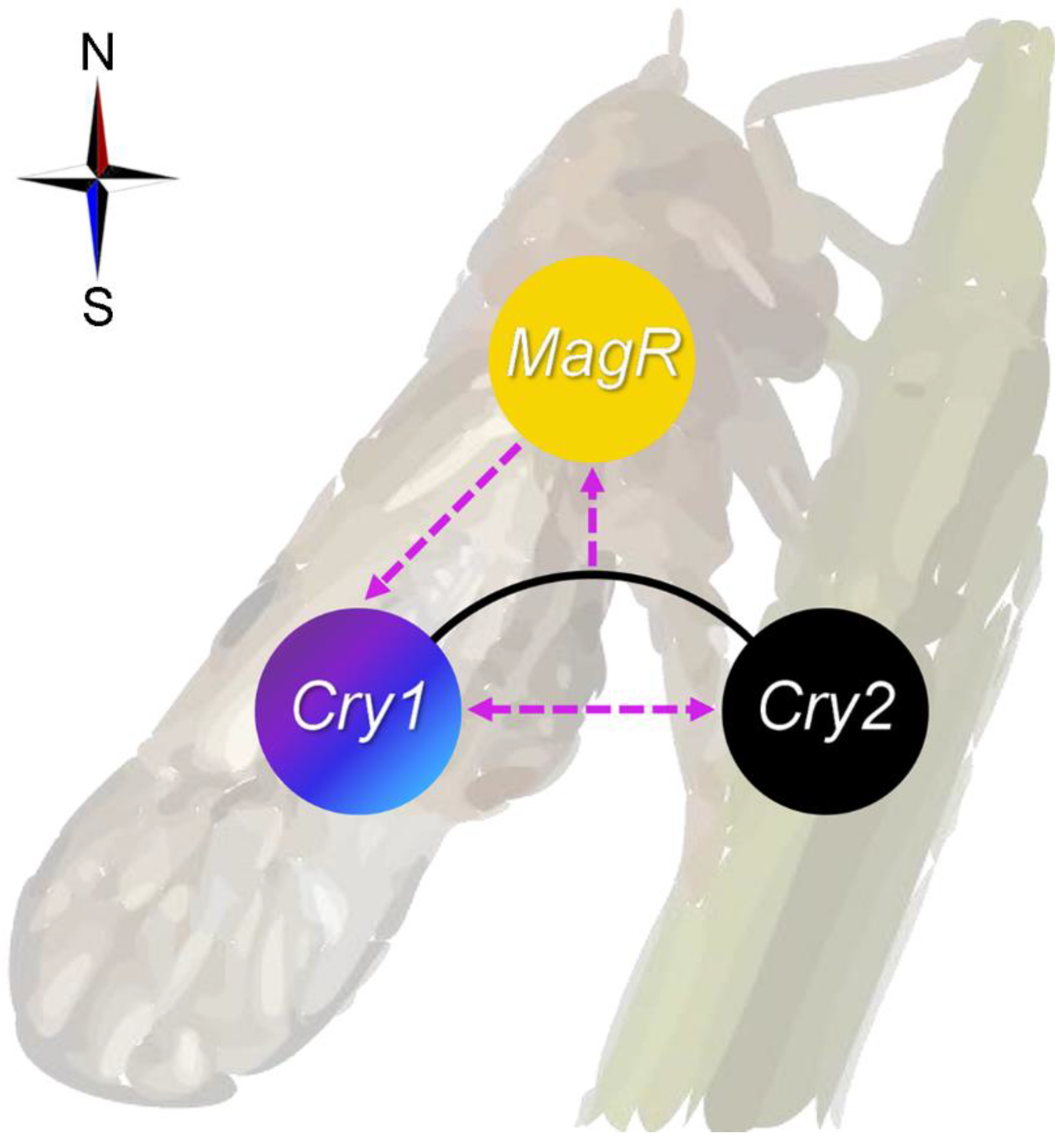
3. Discussion
4. Materials and Methods
4.1. Insects
4.2. Exposure of Insects to Magnetic Fields
4.3. RNA Isolation and cDNA Synthesis
4.4. Double-Stranded RNA Preparation
4.5. Microinjection of dsRNA
4.6. Transcript Expression Analysis
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Monteil, C.L.; Lefevre, C.T. Magnetoreception in microorganisms. Trends Microbiol. 2020, 28, 266–275. [Google Scholar] [CrossRef]
- Zhang, X. Biological Effects of Static Magnetic Fields, 2nd ed.; Springer: Singapore, 2023. [Google Scholar]
- Wang, C.X.; Hilburn, I.A.; Wu, D.A.; Mizuhara, Y.; Couste, C.P.; Abrahams, J.N.H.; Bernstein, S.E.; Matani, A.; Shimojo, S.; Kirschvink, J.L. Transduction of the geomagnetic field as evidenced from alpha-band activity in the human brain. eNeuro 2019, 6, ENEURO.0483-18.2019. [Google Scholar] [CrossRef] [PubMed]
- Mouritsen, H. Long-distance navigation and magnetoreception in migratory animals. Nature 2018, 558, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Wynn, J.; Padget, O.; Mouritsen, H.; Morford, J.; Jaggers, P.; Guilford, T. Magnetic stop signs signal a European songbird’s arrival at the breeding site after migration. Science 2022, 375, 446–449. [Google Scholar] [CrossRef] [PubMed]
- Wan, G.; Hayden, A.N.; Iiams, S.E.; Merlin, C. Cryptochrome 1 mediates light-dependent inclination magnetosensing in monarch butterflies. Nat. Commun. 2021, 12, 771. [Google Scholar] [CrossRef]
- Bradlaugh, A.A.; Fedele, G.; Munro, A.L.; Hansen, C.N.; Hares, J.M.; Patel, S.; Kyriacou, C.P.; Jones, A.R.; Rosato, E.; Baines, R.A. Essential elements of radical pair magnetosensitivity in Drosophila. Nature 2023, 615, 111–116. [Google Scholar] [CrossRef]
- Xu, J.; Jarocha, L.E.; Zollitsch, T.; Konowalczyk, M.; Henbest, K.B.; Richert, S.; Golesworthy, M.J.; Schmidt, J.; Déjean, V.; Sowood, D.J.C.; et al. Magnetic sensitivity of cryptochrome 4 from a migratory songbird. Nature 2021, 594, 535–540. [Google Scholar] [CrossRef]
- Binhi, V.N.; Rubin, A.B. Theoretical concepts in magnetobiology after 40 years of research. Cells 2022, 11, 274. [Google Scholar] [CrossRef] [PubMed]
- Clites, B.L.; Pierce, J.T. Identifying cellular and molecular mechanisms for magnetosensation. Annu. Rev. Neurosci. 2017, 40, 231–250. [Google Scholar] [CrossRef]
- Hore, P.J.; Mouritsen, H. The radical-pair mechanism of magnetoreception. Annu. Rev. Biophys. 2016, 45, 299–344. [Google Scholar] [CrossRef]
- Ritz, T.; Thalau, P.; Phillips, J.B.; Wiltschko, R.; Wiltschko, W. Resonance effects indicate a radical-pair mechanism for avian magnetic compass. Nature 2004, 429, 177–180. [Google Scholar] [CrossRef]
- Kirschvink, J.L.; Walker, M.M.; Diebel, C.E. Magnetite-based magnetoreception. Curr. Opin. Neurobiol. 2001, 11, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Ritz, T.; Adem, S.; Schulten, K. A model for photoreceptor-based magnetoreception in birds. Biophys. J. 2000, 78, 707–718. [Google Scholar] [CrossRef]
- Qin, S.; Yin, H.; Yang, C.; Dou, Y.; Liu, Z.; Zhang, P.; Yu, H.; Huang, Y.; Feng, J.; Hao, J.; et al. A magnetic protein biocompass. Nat. Mater. 2016, 15, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Tong, T.; Wei, M.; Zhang, P.; Fei, F.; Zhou, X.; Guo, Z.; Zhang, J.; Xu, H.; Zhang, L.; et al. Towards magnetism in pigeon MagR: Iron- and iron-sulfur binding work indispensably and synergistically. Zool. Res. 2023, 44, 142–152. [Google Scholar] [CrossRef]
- Xie, C. Searching for unity in diversity of animal magnetoreception: From biology to quantum mechanics and back. Innovation 2022, 3, 100229. [Google Scholar] [CrossRef]
- Ozturk, N. Light-dependent reactions of animal circadian photoreceptor cryptochrome. FEBS J. 2022, 289, 6622–6639. [Google Scholar] [CrossRef] [PubMed]
- Emery, P.; Stanewsky, R.; Helfrich-Förster, C.; Emery-Le, M.; Hall, J.C.; Rosbash, M. Drosophila CRY is a deep brain circadian photoreceptor. Neuron 2000, 26, 493–504. [Google Scholar] [CrossRef]
- Zhu, H.; Yuan, Q.; Briscoe, A.D.; Froy, O.; Casselman, A.; Reppert, S.M. The two CRYs of the butterfly. Curr. Biol. 2005, 15, R953–R954. [Google Scholar] [CrossRef]
- Zoltowski, B.D.; Chelliah, Y.; Wickramaratne, A.; Jarocha, L.; Karki, N.; Xu, W.; Mouritsen, H.; Hore, P.J.; Hibbs, R.E.; Green, C.B.; et al. Chemical and structural analysis of a photoactive vertebrate cryptochrome from pigeon. Proc. Natl. Acad. Sci. USA 2019, 116, 19449–19457. [Google Scholar] [CrossRef]
- Yuan, Q.; Metterville, D.; Briscoe, A.D.; Reppert, S.M. Insect cryptochromes: Gene duplication and loss define diverse ways to construct insect circadian clocks. Mol. Biol. Evol. 2007, 24, 948–955. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Markert, M.J.; Groves, S.C.; Hardin, P.E.; Merlin, C. Vertebrate-like CRYPTOCHROME 2 from monarch regulates circadian transcription via independent repression of CLOCK and BMAL1 activity. Proc. Natl. Acad. Sci. USA 2017, 114, E7516–E7525. [Google Scholar] [CrossRef]
- Netusil, R.; Tomanova, K.; Chodakova, L.; Chvalova, D.; Dolezel, D.; Ritz, T.; Vacha, M. Cryptochrome-dependent magnetoreception in a heteropteran insect continues even after 24 h in darkness. J. Exp. Biol. 2021, 224, jeb243000. [Google Scholar] [CrossRef]
- Gao, Y.; Wen, P.; Carde, R.T.; Xu, H.; Huang, Q. In addition to cryptochrome 2, magnetic particles with olfactory co-receptor are important for magnetic orientation in termites. Commun. Biol. 2021, 4, 1121. [Google Scholar] [CrossRef] [PubMed]
- Tong, D.; Zhang, L.; Wu, N.; Xie, D.; Fang, G.; Coates, B.S.; Sappington, T.W.; Liu, Y.; Cheng, Y.; Xia, J.; et al. The oriental armyworm genome yields insights into the long-distance migration of noctuid moths. Cell Rep. 2022, 41, 111843. [Google Scholar] [CrossRef]
- Merlin, C. Insect magnetoreception: A Cry for mechanistic insights. J. Comp. Physiol. A-Neuroethol. Sens. Neural Behav. Physiol. 2023. [Google Scholar] [CrossRef]
- Fontecave, M. Iron-sulfur clusters: Ever-expanding roles. Nat. Chem. Biol. 2006, 2, 171–174. [Google Scholar] [CrossRef]
- Johnson, D.C.; Dean, D.R.; Smith, A.D.; Johnson, M.K. Structure, function, and formation of biological iron-sulfur clusters. Annu. Rev. Biochem. 2005, 74, 247–281. [Google Scholar] [CrossRef]
- Kiley, P.J.; Beinert, H. The role of Fe-S proteins in sensing and regulation in bacteria. Curr. Opin. Microbiol. 2003, 6, 181–185. [Google Scholar] [CrossRef]
- Mettert, E.L.; Kiley, P.J. How is Fe-S cluster formation regulated? Annu. Rev. Microbiol. 2015, 69, 505–526. [Google Scholar] [CrossRef] [PubMed]
- Rouault, T.A. Mammalian iron-sulphur proteins: Novel insights into biogenesis and function. Nat. Rev. Mol. Cell Biol. 2015, 16, 45–55. [Google Scholar] [CrossRef]
- Ollagnier-de-Choudens, S.; Mattioli, T.; Takahashi, Y.; Fontecave, M. Iron-sulfur cluster assembly: Characterization of IscA and evidence for a specific and functional complex with ferredoxin. J. Biol. Chem. 2001, 276, 22604–22607. [Google Scholar] [CrossRef] [PubMed]
- Mandilaras, K.; Missirlis, F. Genes for iron metabolism influence circadian rhythms in Drosophila melanogaster. Metallomics 2012, 4, 928–936. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Xu, S.; Chen, X.; Wang, C.; Yang, P.; Qin, S.; Zhao, C.; Fei, F.; Zhao, X.; Tan, P.H.; et al. Modulation of MagR magnetic properties via iron-sulfur cluster binding. Sci. Rep. 2021, 11, 23941. [Google Scholar] [CrossRef] [PubMed]
- Mannino, G.; Casacci, L.P.; Bianco Dolino, G.; Badolato, G.; Maffei, M.E.; Barbero, F. The geomagnetic field (GMF) is necessary for black garden ant (Lasius niger L.) foraging and modulates orientation potentially through aminergic regulation and MagR expression. Int. J. Mol. Sci. 2023, 24, 4387. [Google Scholar] [CrossRef]
- Yang, P.; Cai, T.; Zhang, L.; Yu, D.; Guo, Z.; Zhang, Y.; Li, G.; Zhang, X.; Xie, C. A rationally designed building block of the putative magnetoreceptor MagR. Bioelectromagnetics 2022, 43, 317–326. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, Y.; Wu, J.; Wang, W.; Li, Y.; Wan, G.; Chen, F.; Sword, G.A.; Pan, W. Molecular characterization, spatial-temporal expression and magnetic response patterns of iron-sulfur cluster assembly1 (IscA1) in the rice planthopper, Nilaparvata lugens. Insect Sci. 2019, 26, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zeng, L.; Wan, G.; Jiang, X.; Chen, F.; Pan, W. Interaction between cryptochrome and iron-sulfur cluster assembly 1 protein and their roles in the development and reproduction of the brown planthopper, Nilaparvata lugens. Chin. J. Appl. Entomol. 2021, 58, 74–82. [Google Scholar]
- Zhang, Y.; Pan, W. Removal or component reversal of local geomagnetic field affects foraging orientation preference in migratory insect brown planthopper Nilaparvata lugens. PeerJ 2021, 9, e12351. [Google Scholar] [CrossRef]
- Parmagnani, A.S.; D’Alessandro, S.; Maffei, M.E. Iron-sulfur complex assembly: Potential players of magnetic induction in plants. Plant Sci. 2022, 325, 111483. [Google Scholar] [CrossRef]
- Cao, Y.; Yan, P. Role of atomic spin-mechanical coupling in the problem of a magnetic biocompass. Phys. Rev. E 2018, 97, 042409. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Hu, W.; Cai, Y.; Zhao, N. Magnetic noise enabled biocompass. Phys. Rev. Lett. 2020, 124, 128101. [Google Scholar] [CrossRef]
- Zhao, X.; Chen, H.; Lu, L.; Li, Y. A mechanism of compass-free migratory navigation. J. Phys. D Appl. Phys. 2022, 55, 245004. [Google Scholar] [CrossRef]
- Parmagnani, A.S.; Betterle, N.; Mannino, G.; D’Alessandro, S.; Nocito, F.F.; Ljumovic, K.; Vigani, G.; Ballottari, M.; Maffei, M.E. The geomagnetic field (GMF) is required for lima bean photosynthesis and reactive oxygen species production. Int. J. Mol. Sci. 2023, 24, 2896. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Q.; Sun, J.; Ge, Y.; Xue, L.; Mao, H.; Zhou, L.; Zhao, J. Bionic magnetic sensor based on the MagR/Cry4 Complex-Configured graphene transistor with an integrated on-chip gate. ACS Sens. 2023, 8, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Xue, L.; Hu, T.; Guo, Z.; Yang, C.; Wang, Z.; Qin, S.; Yang, P.; Xie, C.; Xu, J.; Li, N.; et al. A novel biomimetic magnetosensor based on magneto-optically involved conformational variation of MagR/Cry4 Complex. Adv. Electron. Mater. 2020, 6, 1901168. [Google Scholar] [CrossRef]
- Chang, H.; Guo, J.; Fu, X.; Hou, Y.; Wu, K. Orientation behavior and regulatory gene expression profiles in migratory Agrotis ipsilon (Lepidoptera: Noctuidae). J. Insect Behav. 2019, 32, 59–67. [Google Scholar] [CrossRef]
- Jin, M.; Liu, B.; Zheng, W.; Liu, C.; Liu, Z.; He, Y.; Li, X.; Wu, C.; Wang, P.; Liu, K.; et al. Chromosome-level genome of black cutworm provides novel insights into polyphagy and seasonal migration in insects. BMC Biol. 2023, 21, 2. [Google Scholar] [CrossRef]
- Wu, H.; Scholten, A.; Einwich, A.; Mouritsen, H.; Koch, K.W. Protein-protein interaction of the putative magnetoreceptor cryptochrome 4 expressed in the avian retina. Sci. Rep. 2020, 10, 7364. [Google Scholar] [CrossRef]
- Xiong, X.; Liu, Y.; Zhang, J.; Wang, S.; Li, L.; Gao, M. Mutational analysis of MpPhy reveals magnetoreception and photosensitivity involvement in secondary metabolites biosynthesis in Monascus purpureus. J. Photochem. Photobiol. B-Biol. 2021, 217, 112164. [Google Scholar] [CrossRef]
- Chang, H.; Guo, J.; Fu, X.; Shen, X.; Hou, Y.; Wu, K. Molecular characterization and expression profiles of IscA1 gene in a long-distance migrant, Agrotis segetum. J. Asia Pac. Econ. 2018, 21, 1299–1306. [Google Scholar] [CrossRef]
- Gegear, R.J.; Casselman, A.; Waddell, S.; Reppert, S.M. Cryptochrome mediates light-dependent magnetosensitivity in Drosophila. Nature 2008, 454, 1014–1018. [Google Scholar] [CrossRef]
- Gegear, R.J.; Foley, L.E.; Casselman, A.; Reppert, S.M. Animal cryptochromes mediate magnetoreception by an unconventional photochemical mechanism. Nature 2010, 463, 804–807. [Google Scholar] [CrossRef]
- Oh, I.T.; Kwon, H.J.; Kim, S.C.; Kim, H.J.; Lohmann, K.J.; Chae, K.S. Behavioral evidence for geomagnetic imprinting and transgenerational inheritance in fruit flies. Proc. Natl. Acad. Sci. USA 2019, 117, 1216–1222. [Google Scholar] [CrossRef] [PubMed]
- Fedele, G.; Green, E.W.; Rosato, E.; Kyriacou, C.P. An electromagnetic field disrupts negative geotaxis in Drosophila via a CRY-dependent pathway. Nat. Commun. 2014, 5, 4391. [Google Scholar] [CrossRef]
- Fedele, G.; Edwards, M.D.; Bhutani, S.; Hares, J.M.; Murbach, M.; Green, E.W.; Dissel, S.; Hastings, M.H.; Rosato, E.; Kyriacou, C.P. Genetic analysis of circadian responses to low frequency electromagnetic fields in Drosophila melanogaster. PLoS Genet. 2014, 10, e1004804. [Google Scholar] [CrossRef] [PubMed]
- Yoshii, T.; Ahmad, M.; Helfrich-Forster, C. Cryptochrome mediates light-dependent magnetosensitivity of Drosophila’s circadian clock. PLoS Biol. 2009, 7, e1000086. [Google Scholar] [CrossRef]
- Menz, M.H.M.; Reynolds, D.R.; Gao, B.; Hu, G.; Chapman, J.W.; Wotton, K.R. Mechanisms and consequences of partial migration in insects. Front. Ecol. Evol. 2019, 7, 403. [Google Scholar] [CrossRef]
- Wan, G.; Jiang, S.; Zhao, Z.; Xu, J.; Tao, X.; Sword, G.A.; Gao, Y.; Pan, W.; Chen, F. Bio-effects of near-zero magnetic fields on the growth, development and reproduction of small brown planthopper, Laodelphax striatellus and brown planthopper, Nilaparvata lugens. J. Insect Physiol. 2014, 68, 7–15. [Google Scholar] [CrossRef]
- Wan, G.; Liu, R.; Li, C.; He, J.; Pan, W.; Sword, G.A.; Hu, G.; Chen, F. Change in geomagnetic field intensity alters migration-associated traits in a migratory insect. Biol. Lett. 2020, 16, 20190940. [Google Scholar] [CrossRef]
- Wan, G.; Jiang, S.; Zhang, M.; Zhao, J.; Zhang, Y.; Pan, W.; Sword, G.A.; Chen, F. Geomagnetic field absence reduces adult body weight of a migratory insect by disrupting feeding behavior and appetite regulation. Insect Sci. 2020, 28, 251–260. [Google Scholar] [CrossRef]
- Zhang, Y.; Wan, G.; Wang, W.; Li, Y.; Yu, Y.; Zhang, Y.; Chen, F.; Pan, W. Enhancement of the geomagnetic field reduces the phototaxis of rice brown planthopper Nilaparvata lugens associated with frataxin down-regulation. Insect Sci. 2020, 27, 1043–1052. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zeng, L.; Wei, Y.; Zhang, M.; Pan, W.; Sword, G.A.; Yang, F.; Chen, F.; Wan, G. Reliable reference genes for gene expression analyses under the hypomagnetic field in a migratory insect. Front. Physiol. 2022, 13, 954228. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Wan, G.; Xu, J.; Li, X.; Liu, Y.; Qi, L.; Chen, F. Evidence for the presence of biogenic magnetic particles in the nocturnal migratory brown planthopper, Nilaparvata lugens. Sci. Rep. 2016, 6, 18771. [Google Scholar] [CrossRef]
- Xu, J.; Wan, G.; Hu, D.; He, J.; Chen, F.; Wang, X.; Hua, H.; Pan, W. Molecular characterization, tissue and developmental expression profiles of cryptochrome genes in wing dimorphic brown planthoppers, Nilaparvata lugens. Insect Sci. 2016, 23, 805–818. [Google Scholar] [CrossRef] [PubMed]
- Bazalova, O.; Kvicalova, M.; Valkova, T.; Slaby, P.; Bartos, P.; Netusil, R.; Tomanova, K.; Braeunig, P.; Lee, H.J.; Sauman, I.; et al. Cryptochrome 2 mediates directional magnetoreception in cockroaches. Proc. Natl. Acad. Sci. USA 2016, 113, 1660–1665. [Google Scholar] [CrossRef]
- Cheng, X.; WU, J.; Ma, F. Brown Planthopper: Occurrence and Control; China Agricultural Press: Beijing, China, 2003. [Google Scholar]
- Zheng, D.; Hu, G.; Yang, F.; Du, X.; Yang, H.; Zhang, G.; Qi, G.; Liang, Z.; Zhang, X.; Cheng, X.; et al. Ovarian development status and population characteristics of Sogatella furcifera (Horváth) and Nilaparvata lugens (Stål): Implications for pest forecasting. J. Appl. Entomol. 2014, 138, 67–77. [Google Scholar] [CrossRef]
- Wiltschko, R.; Niessner, C.; Wiltschko, W. The magnetic compass of birds: The role of cryptochrome. Front. Physiol. 2021, 12, 667000. [Google Scholar] [CrossRef]
- Merlin, C.; Iiams, S.E.; Lugena, A.B. Monarch butterfly migration moving into the genetic era. Trends Genet. 2020, 36, 689–701. [Google Scholar] [CrossRef]
- Filiba, O.; Borin, V.A.; Schapiro, I. The involvement of triplet states in the isomerization of retinaloids. Phys. Chem. Chem. Phys. 2022, 24, 26223–26231. [Google Scholar] [CrossRef]
- Bartos, P.; Netusil, R.; Slaby, P.; Dolezel, D.; Ritz, T.; Vacha, M. Weak radiofrequency fields affect the insect circadian clock. J. R. Soc. Interface 2019, 16, 20190285. [Google Scholar] [CrossRef] [PubMed]
- Wan, G.; Yuan, R.; Wang, W.; Fu, K.; Zhao, J.; Jiang, S.; Pan, W.; Sword, G.A.; Chen, F. Reduced geomagnetic field may affect positive phototaxis and flight capacity of a migratory rice planthopper. Anim. Behav. 2016, 121, 107–116. [Google Scholar] [CrossRef]
- Chen, T.; Jiao, Q.; Ye, C.; Wu, J.; Zheng, Y.; Sun, C.; Hao, P.; Yu, X. A novel cuticular protein-like Cpr21L is essential for nymph survival and male fecundity in the brown planthopper. Int. J. Mol. Sci. 2023, 24, 2163. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Zhang, Y.; Zhao, J.; He, J.; Xuanyuan, Z.; Pan, W.; Sword, G.A.; Chen, F.; Wan, G. Probing Transcriptional Crosstalk between Cryptochromes and Iron-sulfur Cluster Assembly 1 (MagR) in the Magnetoresponse of a Migratory Insect. Int. J. Mol. Sci. 2023, 24, 11101. https://doi.org/10.3390/ijms241311101
Zhang Y, Zhang Y, Zhao J, He J, Xuanyuan Z, Pan W, Sword GA, Chen F, Wan G. Probing Transcriptional Crosstalk between Cryptochromes and Iron-sulfur Cluster Assembly 1 (MagR) in the Magnetoresponse of a Migratory Insect. International Journal of Molecular Sciences. 2023; 24(13):11101. https://doi.org/10.3390/ijms241311101
Chicago/Turabian StyleZhang, Yuning, Ying Zhang, Jingyu Zhao, Jinglan He, Zongjin Xuanyuan, Weidong Pan, Gregory A. Sword, Fajun Chen, and Guijun Wan. 2023. "Probing Transcriptional Crosstalk between Cryptochromes and Iron-sulfur Cluster Assembly 1 (MagR) in the Magnetoresponse of a Migratory Insect" International Journal of Molecular Sciences 24, no. 13: 11101. https://doi.org/10.3390/ijms241311101
APA StyleZhang, Y., Zhang, Y., Zhao, J., He, J., Xuanyuan, Z., Pan, W., Sword, G. A., Chen, F., & Wan, G. (2023). Probing Transcriptional Crosstalk between Cryptochromes and Iron-sulfur Cluster Assembly 1 (MagR) in the Magnetoresponse of a Migratory Insect. International Journal of Molecular Sciences, 24(13), 11101. https://doi.org/10.3390/ijms241311101







