Double- or Triple-Tiered Protection: Prospects for the Sustainable Application of Copper-Based Antimicrobial Compounds for Another Fourteen Decades
Abstract
1. Introduction
2. Development of Copper-Based Antimicrobial Compounds
| Type | Name | Active Constituent | Advantages | Disadvantages |
|---|---|---|---|---|
| Inorganic copper fungicides | Copper sulphate | CuSO4 | Anti-microbial [8] | Phytotoxicity, Short-lasting [8] |
| Copper oxychloride | 3Cu(OH)2CuCl2 | Anti-microbial, Stable [27] | Short-lasting [39] | |
| Copper oxide | CuO | Low toxicity, Stable, Anti-microbial [8] | Low efficiency | |
| Copper hydroxide | Cu(OH)2 | Low toxicity, Stable, Anti-microbial [27] | Phytotoxicity [40] | |
| Organic copper fungicides | Oxine–copper | C18H12CuN2O2 | Low toxicity, Anti-microbial, Long-lasting [41] | Drug resistance, Environmental pollution, Phytotoxicity |
| Thiodiazole–copper | C4H4N6S4Cu | Low toxicity, Stable, Anti-microbe [42] | ||
| Copper abietate | C40H58CuO4 | Low toxicity, Stable, Anti-microbial | ||
| Copper-based nanoparticles | CuS nanoparticles | Cu and S | Slow-release, Stable, Low toxicity, High-efficiency [43] | Drug resistance, Phytotoxicity [44] |
| CuO nanoparticles | CuO | |||
| CuAlO2 nanoparticles | Cu and Al |
3. Construction of a Physical Barrier by Covering Plants with Slightly Soluble CBACs
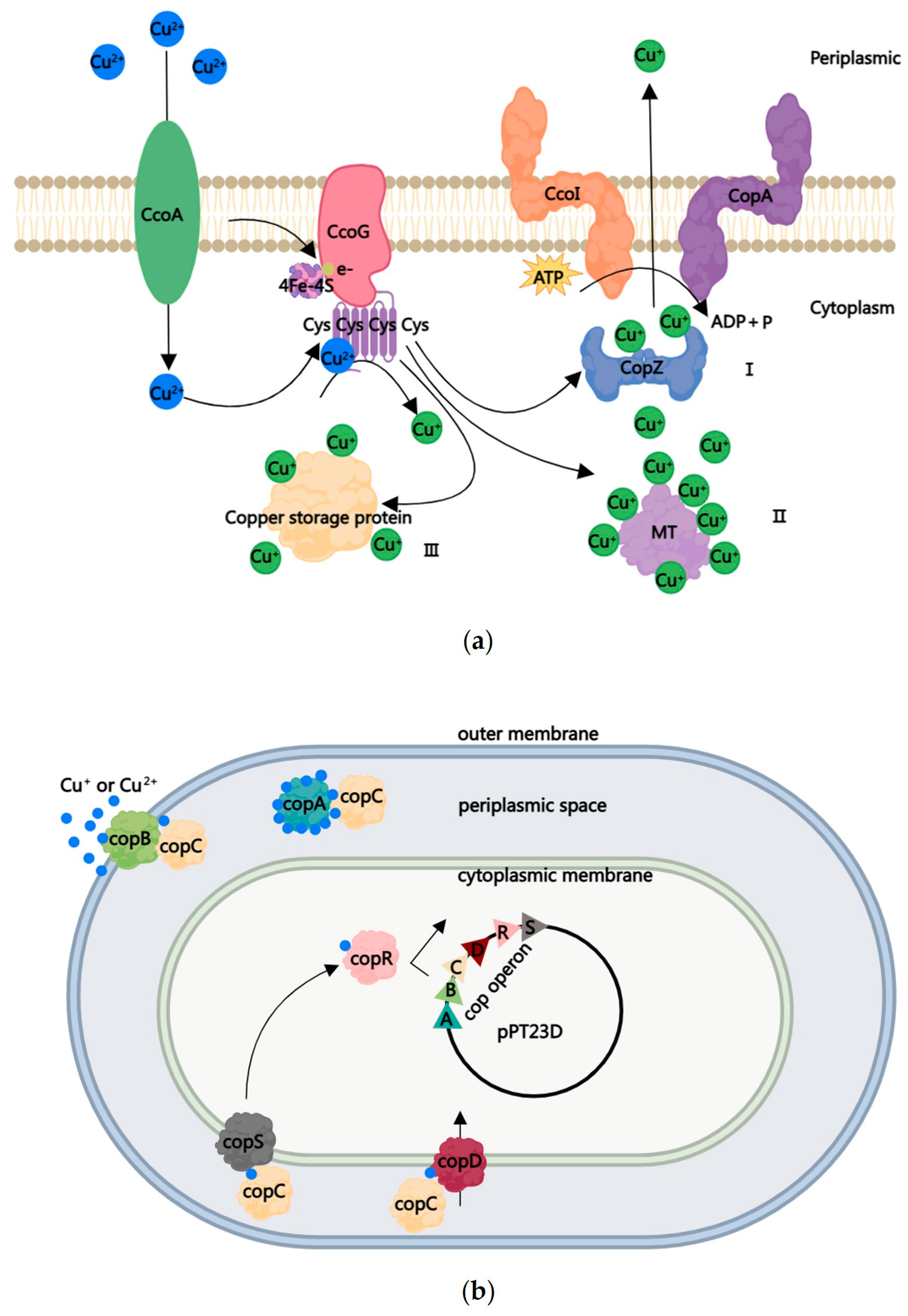
4. The Second Tier of Protection and Copper-Resistant Strains
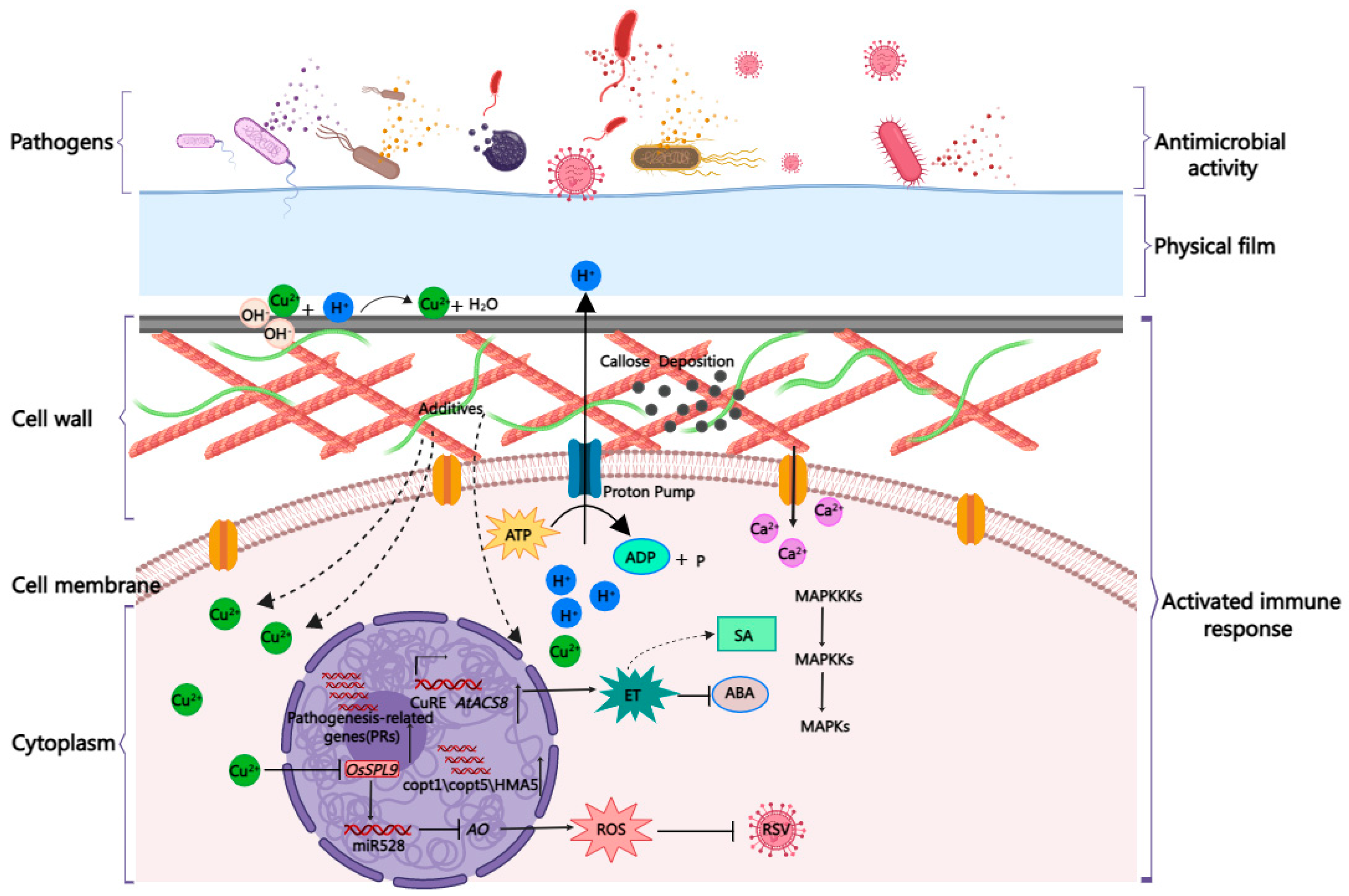
5. Enhanced Plant Resistance to Pathogens and Activation of Defense-like Responses
6. Eliciting Plant Immunity to Strengthen the Third-Tier Barrier
7. Summary and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- West, P.C.; Gerber, J.S.; Engstrom, P.M.; Mueller, N.D.; Brauman, K.A.; Carlson, K.M.; Cassidy, E.S.; Johnston, M.; MacDonald, G.K.; Ray, D.K.; et al. Leverage points for improving global food security and the environment. Science 2014, 345, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Lipper, L.; Thornton, P.; Campbell, B.M.; Baedeker, T.; Braimoh, A.; Bwalya, M.; Caron, P.; Cattaneo, A.; Garrity, D.; Henry, K.; et al. Climate-smart agriculture for food security. Nat. Clim. Change 2014, 4, 1068–1072. [Google Scholar] [CrossRef]
- Kumar, P.; Sharma, P.K. Soil salinity and food security in India. Front. Sustain. Food Syst. 2020, 4, 533781. [Google Scholar] [CrossRef]
- Lobell, D.B.; Schlenker, W.; Costa-Roberts, J. Climate trends and global crop production since 1980. Science 2011, 333, 616–620. [Google Scholar] [CrossRef] [PubMed]
- Kah, M.; Tufenkji, N.; White, J.C. Nano-enabled strategies to enhance crop nutrition and protection. Nat. Nanotechnol. 2019, 14, 532–540. [Google Scholar] [CrossRef]
- Savary, S.; Willocquet, L.; Pethybridge, S.J.; Esker, P.; McRoberts, N.; Nelson, A. The global burden of pathogens and pests on major food crops. Nat. Ecol. Evol. 2019, 3, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.J.; Jiang, F.B.; Ou, J.F. Global pesticide consumption and pollution: With China as a focus. Proc. Int. Acad. Ecol. Environ. Sci. 2011, 1, 125–144. [Google Scholar]
- Jay, R.L.; Ebrahim, O.; Franklin, B.; Jürgen, K.; Jeffrey, B.; Jones, J.A. Thirteen decades of antimicrobial copper compounds applied in agriculture. A review. Agron. Sustain. Dev. 2018, 38, 28. [Google Scholar]
- Torre, A.L.; Chiranjib, M.; Federica, C.; Valerio, B. Natural alternatives to copper and low-rate copper formulations to control grape downy mildew in organic farming. Hell. Plant Prot. J. 2012, 5, 13–21. [Google Scholar]
- Schutte, G.C.; Beeton, K.V.; Kotzé, J.M. Rind stippling on valencia oranges by copper fungicides used for control of citrus black spot in South Africa. Plant Dis. 1997, 81, 851–854. [Google Scholar] [CrossRef]
- Elkins, R.B.; Temple, T.N.; Shaffer, C.A.; Ingels, C.A.; Lindow, S.B.; Zoller, B.G.; Johnson, K.B. Evaluation of dormant-stage inoculum sanitation as a component of a fire blight management program for fresh-market bartlett pear. Plant Dis. 2015, 99, 1147–1152. [Google Scholar] [CrossRef]
- Lee, Y.A.; Milton, N.; Schroth, M.H.; Steven, E.L.; Wang, X.L.; Bill, O.; Richard, B.; Beth, L.T. Increased toxicity of iron-amended copper-containing bactericides to the walnut blight pathogen Xanthomonas campestris pv. juglandis. Phytopathology 1993, 83, 1460–1465. [Google Scholar] [CrossRef]
- Ninot, A.; Aletà, N.; Moragrega, C.; Montesinos, E. Evaluation of a reduced copper spraying program to control bacterial blight of walnut. Plant Dis. 2002, 86, 583–587. [Google Scholar] [CrossRef] [PubMed]
- Large, E.C.; Beer, W.J. Field trials of copper fungicides for the control of potato blight; low-copper fungicides. Ann. Appl. Biol. 1946, 33, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Wimalajeewa, D.L.S.; Robert, S.; Cahill, G.H.; Hg, S.; Washbourne, J.R. Chemical control of bacterial canker (Pseudomonas syringae pv. syringae) of apricot and cherry in Victoria. Aust. J. Exp. Agric. 1991, 31, 705–708. [Google Scholar] [CrossRef]
- Furtado, I. Effect of copper fungicides on the occurrence of the pathogenic form of Colletotrichum coffeanum. Trans. Br. Mycol. Soc. 1969, 53, 325–328. [Google Scholar] [CrossRef]
- Obanor, F.O.; Walter, M.; Jones, E.E.; Jaspers, M.V. Efficacy of systemic acquired resistance inducers in olive leaf spot management. Australas. Plant Pathol. 2013, 42, 163–168. [Google Scholar] [CrossRef]
- Graham, R.D. Susceptibility to powdery mildew of wheat plants deficient in copper. Plant Soil 1980, 56, 181–185. [Google Scholar] [CrossRef]
- Karl, H.S. The influence of boron and copper deficiency upon infection by Erysiphe graminis D.C. the powdery mildew, in wheat var. kenya. Plant Soil 1967, 27, 450–452. [Google Scholar]
- Beresford, R.M.; James, T.S.; Walker, M.J.; Spink, R.R.; Marshall; White, V. Copper and slaked lime for the control of black spot and powdery mildew in apples. Fruit Crops 1995, 48, 83–88. [Google Scholar] [CrossRef]
- Mesquita, A.F.; Gonçalves, F.J.M.; Gonçalves, A.M.M. The lethal and sub-lethal effects of fluorinated and copper-based pesticides—A review. Int. J. Environ. Res. Public Health 2023, 20, 3706. [Google Scholar] [CrossRef]
- Stevens, F.L.; Hall, J.G. Diseases of economic plants. Science 1915, 33, 621–622. [Google Scholar]
- Tratman, E.E.R. Report on the Use of Metal Railroad Ties and on Preservative Processes and Metal Tie-Plates for Wooden Ties; Deptartment of Agriculture, Forestry Division: Washington, DC, USA, 1894; Bulletin 9; p. 269.
- Johnson, G.F. The early history of copper fungicides. Agric. His. 1935, 9, 67–79. [Google Scholar]
- Plant Diseases; Vermont Agricultural Experiment Station Bulletin: Burlington, VT, USA, 1892; Volume 28, p. 17.
- Mark, L. Dormant Sprays for Disease Control; Michigan State University Extension: East Lansing, MI, USA, 2008. [Google Scholar]
- Vicent, A.; Armengol, J.; García-Jiménez, J. Rain fastness and persistence of fungicides for control of alternaria brown spot of citrus. Plant Dis. 2007, 91, 393–399. [Google Scholar] [CrossRef]
- Ma, Y.; Yu, H.; Liu, W.; Qin, Y.; Xing, R.; Li, P. Integrated proteomics and metabolomics analysis reveals the antifungal mechanism of the C-coordinated O-carboxymethyl chitosan Cu (II) complex. Int. J. Biol. Macromol. 2020, 155, 1491–1509. [Google Scholar] [CrossRef] [PubMed]
- Malandrakis, A.A.; Kavroulakis, N.; Chrysikopoulos, C.V. Use of copper, silver and zinc nanoparticles against foliar and soil-borne plant pathogens. Sci. Total Environ. 2019, 670, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Hermida-Montero, L.A.; Pariona, N.; Mtz-Enriquez, A.I.; Carrión, G.; Paraguay-Delgado, F.; Rosas-Saito, G. Aqueous-phase synthesis of nanoparticles of copper/copper oxides and their antifungal effect against Fusarium oxysporum. J. Hazard. Mater. 2019, 380, 120850. [Google Scholar] [CrossRef]
- Sathiyabama, M.; Manikandan, A. Application of copper-chitosan nanoparticles stimulate growth and induce resistance in finger millet (Eleusine coracana Gaertn.) plants against blast disease. J. Agric. Food Chem. 2018, 66, 1784–1790. [Google Scholar] [CrossRef]
- Gaba, S.; Rai, A.K.; Varma, A.; Prasad, R.; Goel, A. Biocontrol potential of mycogenic copper oxide nanoparticles against Alternaria brassicae. Front. Chem. 2022, 10, 966396. [Google Scholar] [CrossRef]
- Tarakanov, R.; Shagdarova, B.; Lyalina, T.; Zhuikova, Y.; Il’ina, A.; Dzhalilov, F.; Varlamov, V. Protective properties of copper-loaded chitosan nanoparticles against soybean pathogens Pseudomonas savastanoi pv. glycinea and Curtobacterium flaccumfaciens pv. flaccumfaciens. Polymers 2023, 15, 1100. [Google Scholar]
- Sadek, M.E.; Shabana, Y.M.; Sayed-Ahmed, K.; Abou Tabl, A.H. Antifungal activities of sulfur and copper nanoparticles against cucumber postharvest diseases caused by Botrytis cinerea and Sclerotinia sclerotiorum. J. Fungi 2022, 8, 412. [Google Scholar] [CrossRef] [PubMed]
- Gomes, D.G.; Sanada, K.; Pieretti, J.C.; Shigueoka, L.H.; Sera, G.H.; Seabra, A.B.; Oliveira, H.C. Nanoencapsulation boosts the copper-induced defense responses of a susceptible Coffea arabica cultivar against Hemileia vastatrix. Antibiotics 2023, 12, 249. [Google Scholar] [CrossRef]
- Eid, A.M.; Fouda, A.; Hassan, S.E.-D.; Hamza, M.F.; Alharbi, N.K.; Elkelish, A.; Alharthi, A.; Salem, W.M. Plant-based copper oxide nanoparticles; biosynthesis, characterization, antibacterial activity, tanning wastewater treatment, and heavy metals sorption. Catalysts 2023, 13, 348. [Google Scholar] [CrossRef]
- Aien, J.; Khan, A.A.; Haq, S.; Khan, A.R.; Elmnasri, K.; Ben Ali, M.; AL-Harbi, M.S.; Alghonaim, M.I.; Alsalamah, S.A.; Qurtam, A.A.; et al. Antibacterial, antioxidant and physicochemical properties of Pipper nigram aided copper oxide nanoparticles. Crystals 2023, 13, 330. [Google Scholar] [CrossRef]
- Malandrakis, A.A.; Kavroulakis, N.; Chrysikopoulos, C.V. Synergy between Cu-NPs and fungicides against Botrytis cinerea. Sci. Total Environ. 2020, 703, 135557. [Google Scholar] [CrossRef]
- Pérez-Rodríguez, P.; Paradelo, M.; Rodríguez-Salgado, I.; Fernández-Calviño, D.; López-Periago, J.E. Modeling the influence of raindrop size on the wash-off losses of copper-based fungicides sprayed on potato (Solanum tuberosum L.) leaves. J. Environ. Sci. Health A 2013, 48, 737–746. [Google Scholar] [CrossRef] [PubMed]
- Montag, J.; Schreiber, L.; Schönherr, J. An in vitro study on the postinfection activities of copper hydroxide and copper sulfate against conidia of Venturia inaequalis. J. Agric. Food Chem. 2006, 54, 893–899. [Google Scholar] [CrossRef]
- Liu, X.; Yang, Y.; Chen, Y.; Zhang, Q.; Lu, P.; Hu, D. Dissipation, residues and risk assessment of oxine-copper and pyraclostrobin in citrus. Food Addit. Contam. 2019, 36, 1538–1550. [Google Scholar] [CrossRef]
- Tang, P.; Xiong, X.P.; Du, J.S. Control effect of 20% Longkejun (thiodiazole-copper) on crop bacterial diseases. Agric. Sci. Tech. Equip. 2008, 3, 35–36. [Google Scholar]
- Ahmad, H.; Venugopal, K.; Bhat, A.H.; Kavitha, K.; Ramanan, A.; Rajagopal, K.; Srinivasan, R.; Manikandan, E. Enhanced biosynthesis synthesis of copper oxide nanoparticles (CuO-NPs) for their antifungal activity toxicity against major phyto-pathogens of apple orchards. Pharmacol. Res. 2020, 37, 246. [Google Scholar] [CrossRef]
- Dimkpa, C.O.; McLean, J.E.; Britt, D.W.; Anderson, A.J. Nano-CuO and interaction with nano-ZnO or soil bacterium provide evidence for the interference of nanoparticles in metal nutrition of plants. Ecotoxicology 2015, 24, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Millardet, A. Traitement du Mildiou et du Rot. Agric. Prat. 1885, 49, 513–516. [Google Scholar]
- Li, J.H.; Zhang, H.Q.; Li, N.; Shu, G.P.; Gu, Y.H.; Deng, Z.N. Influences of five surface active agents on control effect of copper fungicides to citrus canker disease of bingtang sweet orange. Hunan Agric. Sci. 2013, 5, 71–73. [Google Scholar]
- Menkissoglu, O. Relationship of free ionic copper and toxicity to bacteria in solutions of organic compounds. Phytopathology 1991, 81, 1258–1263. [Google Scholar] [CrossRef]
- Menkissoglu, O.; Lindow, S.E. Chemical forms of copper on leaves in relation to the bactericidal activity of cupric hydroxide deposits on plants. Phytopathology 1991, 81, 1263–1270. [Google Scholar] [CrossRef]
- Arman, P.; Wain, R.L. The role of leaf exudates in the solution of copper from Bordeaux mixture. Ann. Appl. Biol. 1958, 46, 366–374. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Y.; Yang, D.; Jin, Q.; Wu, C.; Cui, J. Multifunctional molybdenum disulfide-copper nanocomposite that enhances the antibacterial activity, promotes rice growth and induces rice resistance. J. Hazard 2020, 394, 122551. [Google Scholar] [CrossRef]
- Aftab, M.; Butt, M.Z.; Ali, D.; Aftab, Z.H.; Tanveer, M.U.; Fayyaz, B. Investigation of antifungal response of NiO and copper-doped NiO thin films against Aspergillus niger and Macrophomina phaseolina fungi. ESPR 2022, 29, 3840–3852. [Google Scholar] [CrossRef]
- Galimberti, A.; Alyokhin, A. Lethal and sublethal effects of mineral oil on potato pests. J. Econ. Entomol. 2018, 111, 1261–1267. [Google Scholar] [CrossRef]
- Silva Junior, G.J.; Moraes, M.R.; Moreira, R.R.; Behlau, F. Tree age and cultivar-oriented use of mineral oil added to fungicide tank mixture for the control of citrus black spot in sweet orange orchards. Pest Manag. Sci. 2022, 78, 488–498. [Google Scholar] [CrossRef]
- Lu, H.L.; OuYang, G.C.; Tan, B.L.; Hou, B.H.; Meng, X.; Fang, X.R. Synergistic effect of mineral spray oil on inorganic copper fungicides to control Elsinoe fawcettii and Panonychus citri. Agrochemicals 2018, 57, 383–386. [Google Scholar]
- Xu, L.J.; Fu, J.G.; Yuan, Q.H.; Zou, G.J.; Wang, Y.Q.; Chen, J. A Bamboo Vinegar Copper Preparation and Its Application in Tobacco Disease Control and Preparation Method. Patent CN106857666A, 2 June 2020. [Google Scholar]
- Shen, Y.; Borgatta, J.; Ma, C.; Elmer, W.; Hamers, R.J.; White, J.C. Copper nanomaterial morphology and composition control foliar transfer through the cuticle and mediate resistance to root fungal disease in tomato (Solanum lycopersicum). J. Agric. Food Chem. 2020, 68, 11327–11338. [Google Scholar] [CrossRef] [PubMed]
- Ekici, S.; Yang, H.; Koch, H.G.; Daldal, F. Novel transporter required for biogenesis of cbb3-type cytochrome c oxidase in Rhodobacter capsulatus. mBio 2012, 3, e00293-11. [Google Scholar] [CrossRef]
- Ekici, S.; Turkarslan, S.; Pawlik, G.; Dancis, A.; Baliga, N.S.; Koch, H.G.; Daldal, F. Intracytoplasmic copper homeostasis controls cytochrome c oxidase production. mBio 2014, 5, e01055-13. [Google Scholar] [CrossRef]
- Khalfaoui-Hassani, B.; Wu, H.; Blaby-Haas, C.E.; Zhang, Y.; Sandri, F.; Verissimo, A.F.; Koch, H.G.; Daldal, F. Widespread distribution and functional specificity of the copper importer CcoA: Distinct Cu uptake routes for bacterial cytochrome c oxidases. mBio 2018, 9, e00065-18. [Google Scholar] [CrossRef] [PubMed]
- Chillappagari, S.; Miethke, M.; Trip, H.; Kuipers, O.P.; Marahiel, M.A. Copper acquisition is mediated by YcnJ and regulated by YcnK and CsoR in Bacillus subtilis. J. Bacteriol. 2009, 191, 2362–2370. [Google Scholar] [CrossRef] [PubMed]
- Marckmann, D.; Trasnea, P.I.; Schimpf, J.; Winterstein, C.; Andrei, A.; Schmollinger, S.; Blaby-Haas, C.E.; Friedrich, T.; Daldal, F.; Koch, H.G. The cbb3-type cytochrome oxidase assembly factor CcoG is a widely distributed cupric reductase. Proc. Natl. Acad. Sci. USA 2019, 116, 21166–21175. [Google Scholar] [CrossRef]
- Koch, H.G.; Winterstein, C.; Saribas, A.S.; Alben, J.O.; Daldal, F. Roles of the ccoGHIS gene products in the biogenesis of the cbb3-type cytochrome c oxidase. J. Mol. Biol. 2000, 297, 49–65. [Google Scholar] [CrossRef] [PubMed]
- Macomber, L.; Imlay, J.A. The iron-sulfur clusters of dehydratases are primary intracellular targets of copper toxicity. Proc. Natl. Acad. Sci. USA 2009, 106, 8344–8349. [Google Scholar] [CrossRef] [PubMed]
- Espírito Santo, C.; Lam, E.W.; Elowsky, C.G.; Quaranta, D.; Domaille, D.W.; Chang, C.J.; Grass, G. Bacterial killing by dry metallic copper surfaces. Appl. Environ. Microbiol. 2011, 77, 794–802. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C. Oxygen-toxicity, oxygen radicals, transition-metals and disease. Biochem. J. 1984, 219, 1–14. [Google Scholar] [CrossRef]
- Dupont, C.L.; Grass, G.; Rensing, C. Copper toxicity and the origin of bacterial resistance-new insights and applications. Metallomics 2011, 3, 1109–1118. [Google Scholar] [CrossRef]
- Singleton, C.; Hearnshaw, S.; Zhou, L.; Le Brun, N.E.; Hemmings, A.M. Mechanistic insights into Cu(I) cluster transfer between the chaperone CopZ and its cognate Cu(I)-transporting P-type ATPase, CopA. Biochem. J. 2009, 424, 347–356. [Google Scholar] [CrossRef]
- Meydan, S.; Klepacki, D.; Karthikeyan, S.; Margus, T.; Thomas, P.; Jones, J.E.; Khan, Y.; Briggs, J.; Dinman, J.D.; Vázquez-Laslop, N.; et al. Programmed ribosomal frameshifting generates a copper transporter and a copper chaperone from the same gene. Mol. Cell 2017, 65, 207–219. [Google Scholar] [CrossRef] [PubMed]
- Utz, M.; Andrei, A.; Milanov, M.; Trasnea, P.I.; Marckmann, D.; Daldal, F.; Koch, H.G. The Cu chaperone CopZ is required for Cu homeostasis in Rhodobacter capsulatus and influences cytochrome cbb3 oxidase assembly. Mol. Microbiol. 2019, 111, 764–783. [Google Scholar] [CrossRef]
- Kulajta, C.; Thumfart, J.O.; Haid, S.; Daldal, F.; Koch, H.G. Multi-step assembly pathway of the cbb3-type cytochrome c oxidase complex. J. Mol. Biol. 2006, 355, 989–1004. [Google Scholar] [CrossRef]
- Peters, A.; Kulajta, C.; Pawlik, G.; Daldal, F.; Koch, H.G. Stability of the cbb3-type cytochrome oxidase requires specific CcoQ-CcoP interactions. J. Bacteriol. 2008, 190, 5576–5586. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Bremner, I. Oxygen free radicals and metallothionein. Free Radic. Biol. Med. 1993, 14, 325–337. [Google Scholar] [CrossRef]
- Sutherland, D.E.; Stillman, M.J. The „magic numbers” of metallothionein. Metallomics 2011, 3, 444–463. [Google Scholar] [CrossRef]
- Olafson, R.W.; Abel, K.; Sim, R.G. Prokaryotic metallothionein: Preliminary characterization of a blue-green alga heavy metal-binding protein. Biochem. Biophys. Res. Commun. 1979, 89, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Calvo, J.; Jung, H.; Meloni, G. Copper metallothioneins. IUBMB Life 2017, 69, 236–245. [Google Scholar] [CrossRef]
- Straw, M.L.; Chaplin, A.K.; Hough, M.A.; Paps, J.; Bavro, V.N.; Wilson, M.T.; Vijgenboom, E.; Worrall, J. A cytosolic copper storage protein provides a second level of copper tolerance in Streptomyces lividans. Metallomics 2018, 10, 180–193. [Google Scholar] [CrossRef]
- Masami, N.; Masao, G.; Katsumi, A.; Tadaaki, H. Nucleotide sequence and organization of copper resistance genes from Pseudomonas syringae pv. actinidiae. Eur. J. Plant Pathol. 2004, 110, 223–226. [Google Scholar] [CrossRef]
- Richard, D.; Tribot, N.; Boyer, M.; Terville, K.; Boyer, S.; Javegny, M.; Roux-Cuvelier, O.; Pruvost, A.; Chabirand, M.A.; Vernière, C. First report of copper-resistant Xanthomonas citri pv. citri pathotype A causing Asiatic citrus canker in Réunion, France. Plant Dis. 2017, 101, 503. [Google Scholar] [CrossRef]
- Sholberg, P.L.; Bedford, K.E.; Haag, P.; Randall, P. Survey of Erwinia amylovora isolates from British Columbia for resistance to bactericides and virulence on apple. Can. J. Plant Pathol. 2001, 23, 60–67. [Google Scholar] [CrossRef]
- Behlau, F.; Hong, J.C.; Jones, J.B.; Graham, J.H. Evidence for acquisition of copper resistance genes from different sources in citrus-associated xanthomonads. Phytopathology 2013, 103, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Basim, H.; Minsavage, G.V.; Stall, R.E.; Wang, J.F.; Shanker, S.; Jones, J.B. Characterization of a unique chromosomal copper resistance gene cluster from Xanthomonas campestris pv. vesicatoria. Appl. Environ. Microb. 2005, 71, 8284–8291. [Google Scholar] [CrossRef]
- Cooksey, D.A. Genetics of bactericide resistance in plant pathogenic bacteria. Annu. Rev. Phytopathol. 1990, 28, 201–219. [Google Scholar] [CrossRef]
- Voloudakis, A.E.; Reignier, T.M.; Cooksey, D.A. Regulation of resistance to copper in Xanthomonas axonopodis pv. vesicatoria. Appl. Environ. Microbiol. 2005, 71, 782–789. [Google Scholar] [CrossRef]
- Graham, J.H.; Gottwald, T.R.; Cubero, J.; Achor, D.S. Xanthomonas axonopodis pv. citri: Factors affecting successful eradication of citrus canker. Mol. Plant Pathol. 2004, 5, 1–15. [Google Scholar] [PubMed]
- Gottwald, T.R.; Bassanezi, R.B.; Amorim, L.; Bergamin-Filho, A. Spatial pattern analysis of citrus canker-infected plantings in são paulo, Brazil, and augmentation of infection elicited by the asian leafminer. Phytopathology 2007, 97, 674–683. [Google Scholar] [CrossRef] [PubMed]
- Berg, J.; Tom-Petersen, A.; Nybroe, O. Copper amendment of agricultural soil selects for bacterial antibiotic resistance in the field. Lett. Appl. Microbiol. 2005, 40, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Bondarczuk, K.; Piotrowska-Seget, Z. Molecular basis of active copper resistance mechanisms in Gram-negative bacteria. Cell Biol. Toxicol. 2013, 29, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.S.; Cooksey, D.A. Copper resistance in Pseudomonas syringae mediated by periplasmic and outer membrane proteins. Proc. Natl. Acad. Sci. USA 1991, 88, 8915–8919. [Google Scholar] [CrossRef] [PubMed]
- Cooksey, D.A. Copper uptake and resistance in bacteria. Mol. Microbiol. 1993, 7, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Arnesano, F.; Banci, L.; Bertini, I.; Thompsett, A.R. Solution structure of CopC: A cupredoxin-like protein involved in copper homeostasis. Structure 2002, 10, 1337–1347. [Google Scholar] [CrossRef]
- Puig, S.; Rees, E.M.; Thiele, D.J. The ABCDs of periplasmic copper trafficking. Structure 2002, 10, 1292–1295. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Koay, M.; Maher, M.J.; Xiao, Z.; Wedd, A.G. Intermolecular transfer of copper ions from the CopC protein of Pseudomonas syringae. Crystal structures of fully loaded CuI CuII forms. J. Am. Chem. Soc. 2006, 128, 5834–5850. [Google Scholar] [CrossRef]
- Brown, N.L.; Barrett, S.R.; Camakaris, J.; Lee, B.T.; Rouch, D.A. Molecular genetics and transport analysis of the copperresistance determinant (pco) from Escherichia coli plasmid pRJ1004. Mol. Microbiol. 1995, 17, 1153–1166. [Google Scholar] [CrossRef]
- Lee, S.M.; Grass, G.; Rensing, C.; Barrett, S.R.; Yates, C.J.; Stoyanov, J.V.; Brown, N.L. The Pco proteins are involved in periplasmic copper handling in Escherichia coli. Biochem. Biophys. Res. Commun. 2002, 295, 616–620. [Google Scholar] [CrossRef]
- Rouch, D.A.; Brown, N.L. Copper-inducible transcriptional regulation at two promoters in the Escherichia coli copper resistance determinant pco. Microbiology 1997, 143, 1191–1202. [Google Scholar] [CrossRef] [PubMed]
- Huffman, D.L.; Huyett, J.; Outten, F.W.; Doan, P.E.; Finney, L.A.; Hoffman, B.M.; O’Halloran, T.V. Spectroscopy of Cu (II)-PcoC and the multicopper oxidase function of PcoA, two essential components of Escherichia coli pco copper resistance operon. Biochemistry 2002, 41, 10046–10055. [Google Scholar] [CrossRef]
- Cooksey, D.A.; Azad, H.R.; Cha, J.R.; Lim, C.K. Copper resistance gene homologs in pathogenic and saprophytic bacterial species from tomato. Appl. Environ. Microbe 1990, 56, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Monchy, S.; Benotmane, M.A.; Wattiez, R.; van Aelst, S.; Auquier, V.; Borremans, B.; Mergeay, M.; Taghavi, S.; van der Lelie, D.; Vallaeys, T. Transcriptomic and proteomic analyses of the pMOL30-encoded copper resistance in Cupriavidus metallidurans strain CH34. Microbiology 2006, 152, 1765–1776. [Google Scholar] [CrossRef] [PubMed]
- Rensing, C.; Grass, G. Escherichia coli mechanisms of copper homeostasis in a changing environment. FEMS Microbiol. Rev. 2003, 27, 197–213. [Google Scholar] [CrossRef] [PubMed]
- Richard, D.; Ravigné, V.; Rieux, A.; Facon, B.; Boyer, C.; Boyer, K.; Grygiel, P.; Javegny, S.; Terville, M.; Canteros, B.I.; et al. Adaptation of genetically monomorphic bacteria: Evolution of copper resistance through multiple horizontal gene transfers of complex and versatile mobile genetic elements. Mol. Ecol. 2017, 26, 2131–2149. [Google Scholar] [CrossRef]
- Behlau, F.; Canteros, B.I.; Minsavage, G.V.; Jones, J.B.; Graham, J.H. Molecular characterization of copper resistance genes from Xanthomonas citri subsp. citri and Xanthomonas alfalfae subsp. citrumelonis. Appl. Environ. Microb. 2011, 77, 4089–4096. [Google Scholar] [CrossRef]
- Behlau, F.; Canteros, B.I.; Jones, J.B.; Graham, J.H. Copper resistance genes from different xanthomonads and citrus epiphytic bacteria confer resistance to Xanthomonas citri subsp citri. Eur. J. Plant Pathol. 2012, 133, 949–963. [Google Scholar] [CrossRef]
- Pruvost, O.; Richard, D.; Boyer, K.; Javegny, S.; Boyer, C.; Chiroleu, F.; Grygiel, P.; Parvedy, E.; Robène, I.; Maillot-Lebon, V.; et al. Diversity and geographical structure of Xanthomonas citri pv. citri on citrus in the south west Indian ocean region. Microorganisms 2021, 9, 945. [Google Scholar]
- Robinson, J.R.; Isikhuemhen, O.S.; Anike, F.N. Fungal-metal interactions: A review of toxicity and homeostasis. J. Fungi 2021, 7, 225. [Google Scholar] [CrossRef]
- Ran, M.; Zhao, G.; Jiao, L.; Gu, Z.; Yang, K.; Wang, L.; Cao, X.; Xu, L.; Yan, J.; Yan, Y.; et al. Copper ion mediates yeast-to-hypha transition in Yarrowia lipolytica. J. Fungi 2023, 9, 249. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.; Ju, Y.; Dong, R.; Ding, X.; Li, N.; Chu, Z. The mutation of CopAB results in the sensitivity to copper and the reduction of pathogenicity for PXO99. Acta Phytopathol. Sin. 2018, 42, 176–186. (In Chinese) [Google Scholar]
- Yang, B.; Sugio, A.; White, F.F. Os8N3 is a host disease-susceptibility gene for bacterial blight of rice. Proc. Natl. Acad. Sci. USA 2006, 103, 10503–10508. [Google Scholar] [CrossRef]
- Yuan, M.; Chu, Z.; Li, X.; Xu, C.; Wang, S. The bacterial pathogen Xanthomonas oryzae overcomes rice defenses by regulating host copper redistribution. Plant Cell 2010, 22, 3164–3176. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Hou, B.; Lalonde, S.; Takanaga, H.; Hartung, M.L.; Qu, X.; Guo, W.J.; Kim, J.G.; Underwood, W.; Chaudhuri, B.; et al. Sugar transporters for intercellular exchange and nutrition of pathogens. Nature 2010, 468, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Adaskaveg, J.E.; Hine, R.B. Copper tolerance and zinc sensitivity of Mexican strains of Xanthomonas campestris pv. vesicatoria, causal agent of bacterial spot of pepper. Plant Dis. 1985, 69, 993–996. [Google Scholar]
- Andersen, G.L.; Menkissoglou, O.; Lindow, S.E. Occurrence and properties of copper-tolerant strains of Pseudomonas syringae isolated from fruit trees in California. Phytopathology 1991, 81, 648–656. [Google Scholar] [CrossRef]
- Marco, G.M.; Stall, R.E. Control of bacterial spot of pepper initiated by strains of Xanthomonas campestris pv. vesicatoria that differ in sensitivity to copper. Plant Dis. 1983, 67, 779–781. [Google Scholar]
- Huang, C.H.; Vallad, G.E.; Zhang, S.; Wen, A.; Balogh, B.; Figueiredo, J.F.L.; Behlau, F.; Jones, J.B.; Momol, M.T.; Olson, S.M. Effect of application frequency and reduced rates of acibenzolar-s-methyl on the field efficacy of induced resistance against bacterial spot on tomato. Plant Dis. 2012, 96, 221–227. [Google Scholar] [CrossRef]
- Jones, J.B.; Jones, J.P. The effect of bactericides, tank mixing time and spray schedule on bacterial leaf spot of tomato. Proc. Fla. State Hort. Soc. 1985, 244, 247–298. [Google Scholar]
- Liao, Y.Y.; Strayer-Scherer, A.L.; White, J.; Mukherjee, A.; De La Torre-Roche, R.; Ritchie, L.; Colee, J.; Vallad, G.E.; Freeman, J.H.; Jones, J.B.; et al. Nano-Magnesium Oxide: A novel bactericide against copper-tolerant Xanthomonas perforans causing tomato bacterial spot. Phytopathology 2019, 109, 52–62. [Google Scholar] [CrossRef]
- Carvalho, R.; Duman, K.; Jones, J.B.; Paret, M.L. Bactericidal activity of copper-zinc hybrid nanoparticles on copper-tolerant Xanthomonas perforans. Sci. Rep. 2019, 9, 20124. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, L.A.; Kimura, O.; Castilho, A.M.C.; Castilho, K.S.C.; Ribeiro, R.L.D.; Akiba, F.; Carmo, M.G.F. Effect of copper formulations on resident Xanthomonas campestris pv. vesicatoria populations on sweet pepper leaf surfaces. Hortic. Bras. 2003, 21, 44–50. [Google Scholar]
- Strayer-Scherer, A.; Liao, Y.Y.; Young, M.; Ritchie, L.; Vallad, G.E.; Santra, S.; Freeman, J.H.; Clark, D.; Jones, J.B.; Paret, M.L. Advanced copper composites against copper-tolerant Xanthomonas perforans and tomato bacterial spot. Phytopathology 2018, 108, 196–205. [Google Scholar] [CrossRef]
- Jonak, C.; Nakagami, H.; Hirt, H. Heavy metal stress. Activation of distinct mitogen-activated protein kinase pathways by copper and cadmium. Plant Physiol. 2004, 136, 3276–3283. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.Y.; Lee, K.T.; Chi, W.C.; Hirt, H.; Chang, C.C.; Huang, H.J. Possible involvement of MAP kinase pathways in acquired metal-tolerance induced by heat in plants. Planta 2008, 228, 499–509. [Google Scholar] [CrossRef]
- Yeh, C.M.; Chien, P.S.; Huang, H.J. Distinct signalling pathways for induction of MAP kinase activities by cadmium and copper in rice roots. J. Exp. Bot. 2007, 58, 659–671. [Google Scholar] [CrossRef]
- Hung, W.C.; Huang, D.D.; Yeh, C.M.; Huang, H.J. Reactive oxygen species, calcium and serine/threonine phosphatase are required for copper-induced map kinase gene osmapk2, expression in rice. Plant Growth Regul. 2005, 45, 233–241. [Google Scholar] [CrossRef]
- Dietz, K.J.; Baier, M.; Krämer, U. Free radicals and reactive oxygen species as mediators of heavy metal toxicity in plants. In Heavy Metal Stress in Plants; Springer: Berlin/Heidelberg, Germany, 1999; pp. 73–97. [Google Scholar]
- Stoiber, T.L.; Shafer, M.M.; Armstrong, D.E. Induction of reactive oxygen species in chlamydomonas reinhardtii in response to contrasting trace metal exposures. Environ. Toxicol. 2013, 28, 516–523. [Google Scholar] [CrossRef]
- Devireddy, A.R.; Zandalinas, S.I.; Fichman, Y.; Mittler, R. Integration of reactive oxygen species and hormone signaling during abiotic stress. Plant J. 2021, 105, 459–476. [Google Scholar] [CrossRef]
- Cheng, R.; Li, G.; Fan, L.; Jiang, J.; Zhao, Y. Therapeutic iminoboronate-based polymersomes with a Cu(ii)-mediated Fenton reaction-enhanced ROS-response. Chem. Commun. 2020, 56, 12246–12249. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Miller, G.; Morales, J.; Shulaev, V.; Torres, M.A.; Mittler, R. Respiratory burst oxidases: The engines of ROS signaling. Curr. Opin. Plant Biol. 2011, 14, 691–699. [Google Scholar] [CrossRef] [PubMed]
- Iturbe-Ormaetxe, I.; Escuredo, P.R.; Arrese-Igor, C.; Becana, M. Oxidative damage in pea plants exposed to water deficit or paraquat. Plant Physiol. 1998, 116, 173–181. [Google Scholar] [CrossRef]
- Raklami, A.; Oufdou, K.; Tahiri, A.I.; Mateos-Naranjo, E.; Navarro-Torre, S.; Rodríguez-Llorente, I.D.; Meddich, A.; Redondo-Gómez, S.; Pajuelo, E. Safe cultivation of Medicago sativa in metal-polluted soils from semi-arid regions assisted by heat- and metallo-resistant PGPR. Microorganisms 2019, 7, 212. [Google Scholar] [CrossRef]
- Bianka, S. The role of ethylene and ROS in salinity, heavy metal, and flooding responses in rice. Front. Plant Sci. 2014, 5, 685. [Google Scholar]
- Wan, Z.; Erlin, G.; Muhammad, S.; Yujing, W.; Honglei, W.; Xinhui, N.; Longfu, Z. Ghumc1, a blue copper-binding protein, regulates lignin synthesis and cotton immune response. Biochem. Biophys. Res. Commun. 2018, 504, 75–81. [Google Scholar]
- Xu, W.; Meng, Y.; Wise, R.P. Mla- and Rom1-mediated control of microRNA398 and chloroplast copper/zinc superoxide dismutase regulates cell death in response to the barley powdery mildew fungus. New Phytol. 2014, 201, 1396–1412. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, B.; Wu, T.; Ding, Y.; Ding, X.; Chu, Z. Copper ion elicits defense response in Arabidopsis thaliana by activating salicylate- and ethylene-dependent signaling pathways. Mol. Plant 2015, 8, 1550–1553. [Google Scholar] [CrossRef]
- Liu, H.; Xue, X.; Yu, Y.; Xu, M.; Lu, C.; Meng, X.; Zhang, B.; Ding, X.; Chu, Z. Copper ions suppress abscisic acid biosynthesis to enhance defence against Phytophthora infestans in potato. Mol. Plant Pathol. 2020, 21, 636–651. [Google Scholar] [CrossRef]
- Chi, Y.; Wang, C.; Wang, M.; Wan, D.; Huang, F.; Jiang, Z.; Crawford, B.M.; Vo-Dinh, T.; Yuan, F.; Wu, F.; et al. Flg22-induced Ca2+ increases undergo desensitization and resensitization. Plant Cell Environ. 2021, 44, 3563–3575. [Google Scholar] [CrossRef] [PubMed]
- Clay, N.K.; Adio, A.M.; Denoux, C.; Jander, G.; Ausubel, F.M. Glucosinolate metabolites required for an Arabidopsis innate immune response. Science 2009, 323, 95–101. [Google Scholar] [CrossRef]
- Zhang, B.; Liu, H.; Ding, X.; Qiu, J.; Zhang, M.; Chu, Z. Arabidopsis thaliana ACS8 plays a crucial role in the early biosynthesis of ethylene elicited by Cu2+ ions. J. Cell Sci. 2018, 131, jcs202424. [Google Scholar] [PubMed]
- Yu, Y.; Xu, M.; Ding, X.; Chu, Z.; Liu, H. Activating the MYB51 and MYB122 to upregulate the transcription of glucosinolates biosynthesis genes by copper ions in Arabidopsis. Plant Physiol. Biochem. 2021, 162, 496–505. [Google Scholar] [CrossRef] [PubMed]
- Chai, L.; Dong, K.; Liu, S.; Zhang, Z.; Zhang, X.; Tong, X.; Zhu, F.; Zou, J.; Wang, X. A putative nuclear copper chaperone promoters plant immunity in Arabidopsis. J. Exp. Bot. 2020, 71, 6684–6696. [Google Scholar] [CrossRef]
- Yao, S.; Kang, J.; Guo, G.; Yang, Z.; Huang, Y.; Lan, Y.; Zhou, T.; Wang, L.; Wei, C.; Xu, Z.; et al. The key micronutrient copper orchestrates broad-spectrum virus resistance in rice. Sci. Adv. 2022, 8, eabm0660. [Google Scholar] [CrossRef]
- Kamel, S.M.; Elgobashy, S.F.; Omara, R.I.; Derbalah, A.S.; Abdelfatah, M.; El-Shaer, A.; Al-Askar, A.A.; Abdelkhalek, A.; Abd-Elsalam, K.A.; Essa, T.; et al. Antifungal activity of copper oxide nanoparticles against root rot disease in cucumber. J. Fungi 2022, 8, 911. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.L.; Lopes, C.A. Pseudomonas syringae pv. tomato resistant to copper in copper-sprayed tomato fields. Fitopatol. Bras. 1995, 20, 85–89. [Google Scholar]
- Pernezny, K.; Kudela, V.; Kokoskova, B.; Hladka, I. Bacterial disease of tomato in the Czech and Slovak republics and lack of streptomycin resistance among copper-tolerant bacterial strains. Crop Prot. 1995, 14, 267–270. [Google Scholar] [CrossRef]
- Lombardo, M.F.; Panebianco, S.; Azzaro, A.; Catara, V.; Cirvilleri, G. Assessing copper-alternative products for the control of pre- and postharvest citrus Anthracnose. Plants 2023, 12, 904. [Google Scholar] [CrossRef]
- Xu, M.; Zhang, Q.; Lin, X.; Shang, Y.; Cui, X.; Guo, L.; Huang, Y.; Wu, M.; Song, K. Potential effects of metal oxides on agricultural production of rice: A mini review. Plants 2023, 12, 778. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, Y.; Liu, H.; Xia, H.; Chu, Z. Double- or Triple-Tiered Protection: Prospects for the Sustainable Application of Copper-Based Antimicrobial Compounds for Another Fourteen Decades. Int. J. Mol. Sci. 2023, 24, 10893. https://doi.org/10.3390/ijms241310893
Yu Y, Liu H, Xia H, Chu Z. Double- or Triple-Tiered Protection: Prospects for the Sustainable Application of Copper-Based Antimicrobial Compounds for Another Fourteen Decades. International Journal of Molecular Sciences. 2023; 24(13):10893. https://doi.org/10.3390/ijms241310893
Chicago/Turabian StyleYu, Yue, Haifeng Liu, Haoran Xia, and Zhaohui Chu. 2023. "Double- or Triple-Tiered Protection: Prospects for the Sustainable Application of Copper-Based Antimicrobial Compounds for Another Fourteen Decades" International Journal of Molecular Sciences 24, no. 13: 10893. https://doi.org/10.3390/ijms241310893
APA StyleYu, Y., Liu, H., Xia, H., & Chu, Z. (2023). Double- or Triple-Tiered Protection: Prospects for the Sustainable Application of Copper-Based Antimicrobial Compounds for Another Fourteen Decades. International Journal of Molecular Sciences, 24(13), 10893. https://doi.org/10.3390/ijms241310893








