Emerging Role of GLP-1 Agonists in Obesity: A Comprehensive Review of Randomised Controlled Trials
Abstract
1. Introduction
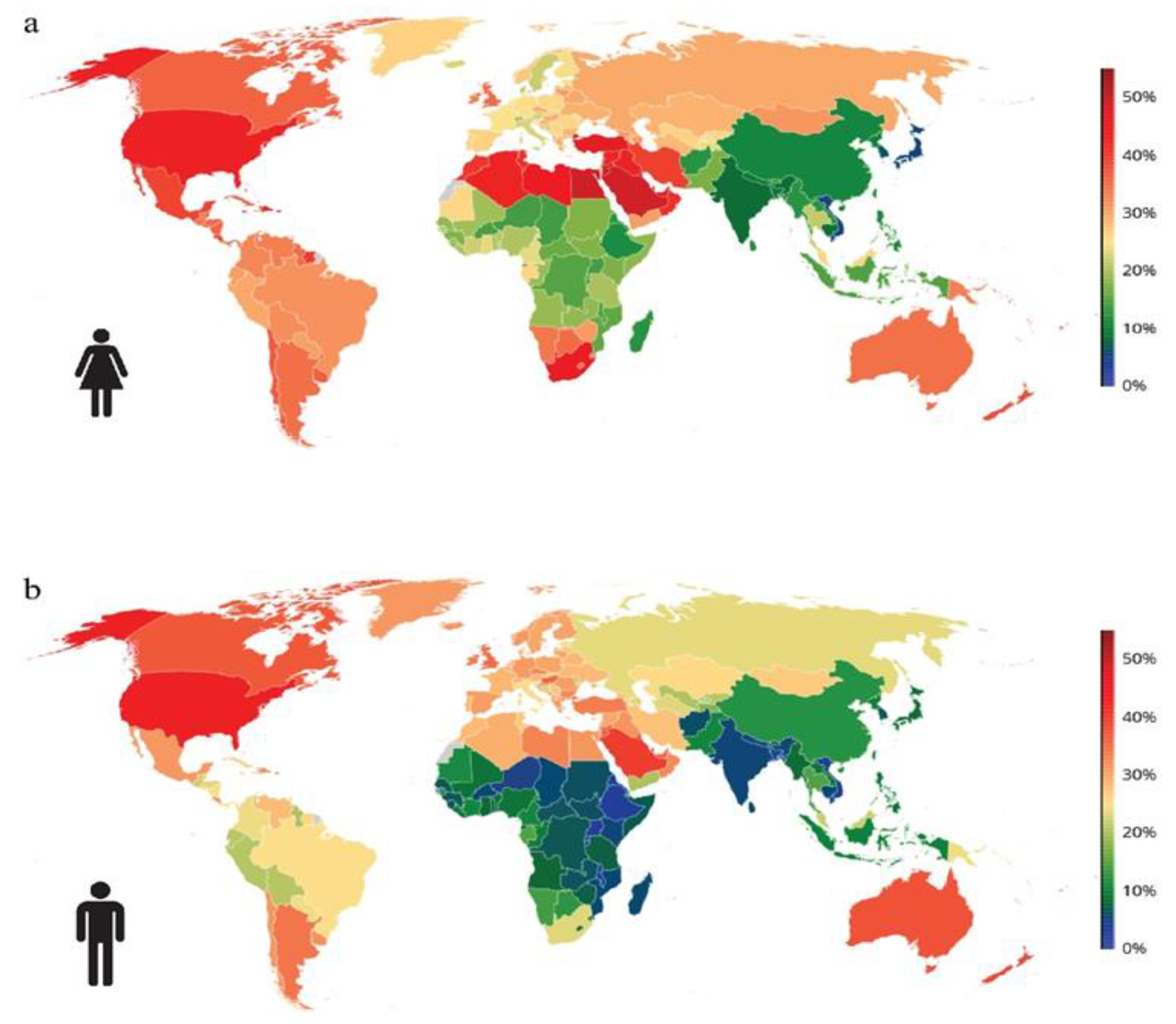
1.1. Prevalence of Obesity Globally
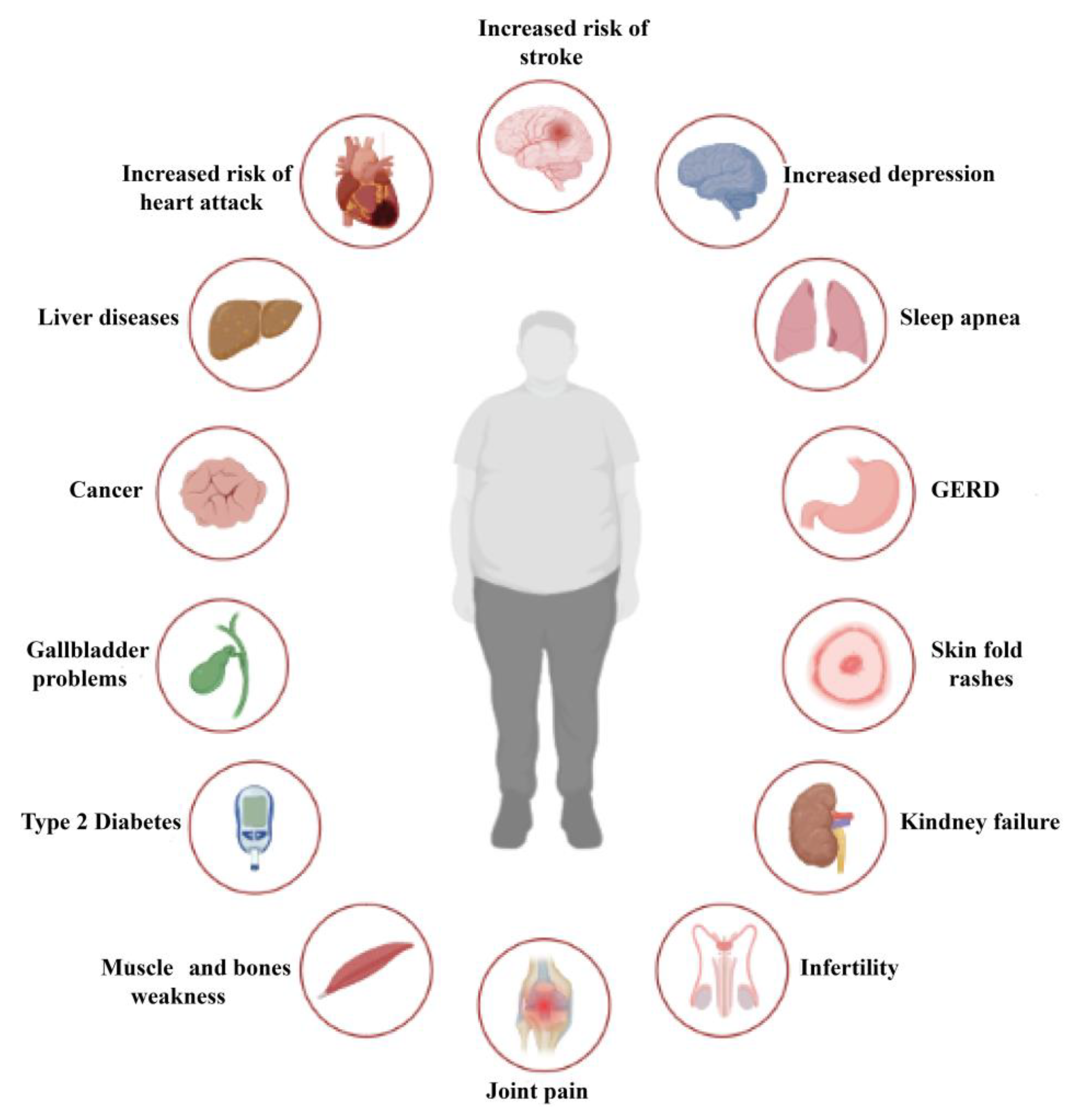
1.2. Complications of Obesity
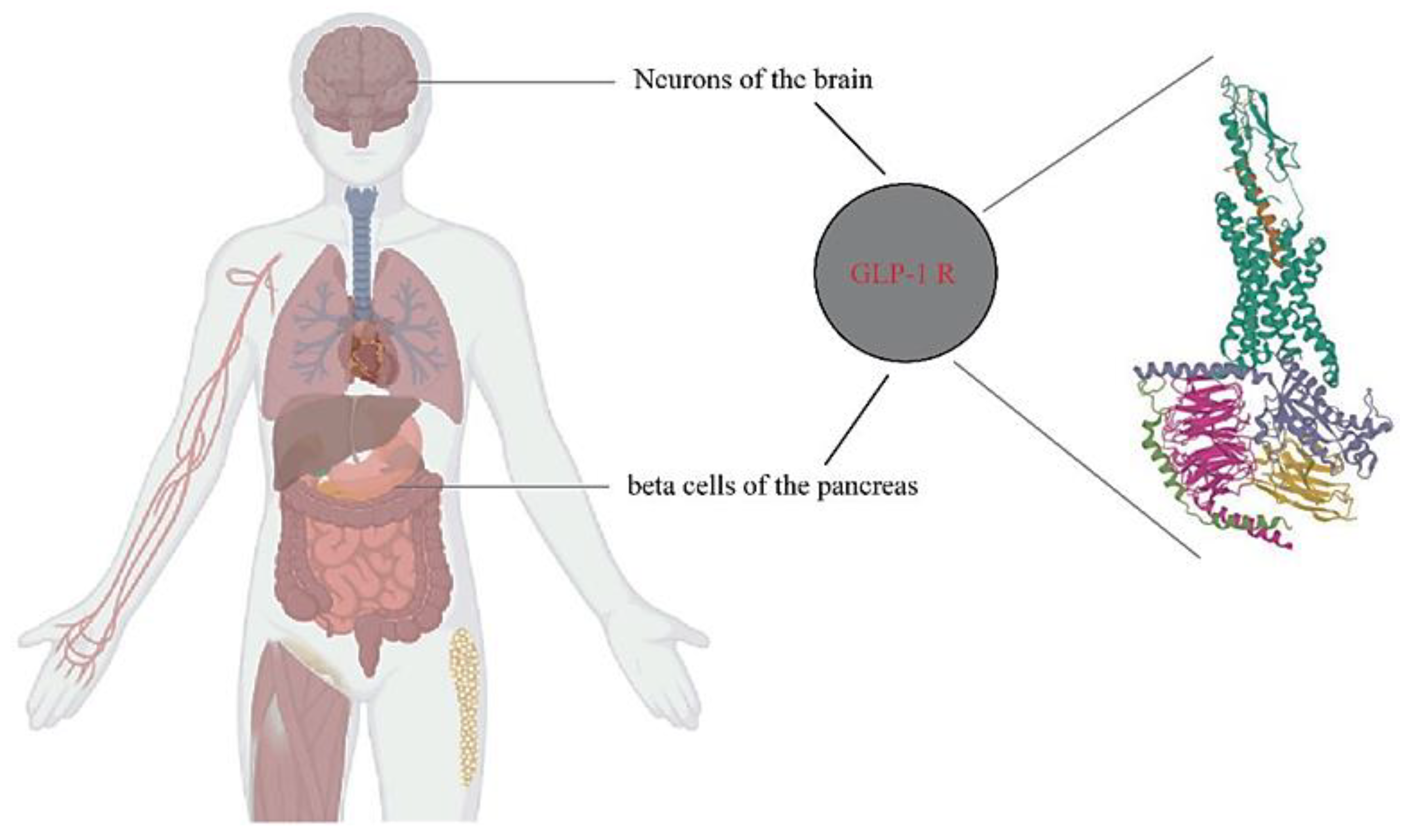
1.3. The Emergence of Glucagon-Like Peptide 1 Receptor Agonists and the First Results Obtained upon Administration of Exenatide
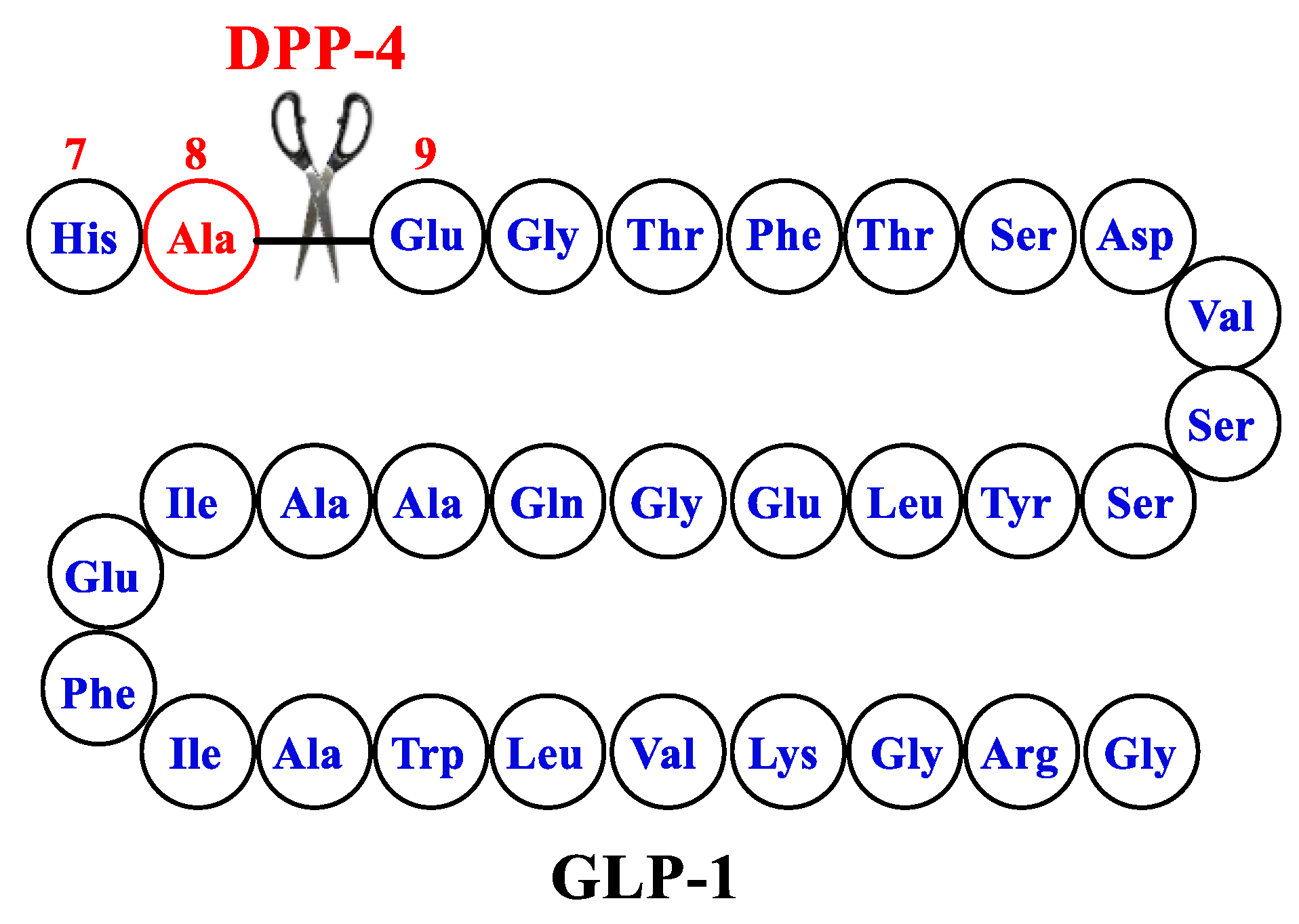
2. Design of GLP1-RA
3. Involvement of the Incretin System in Obesity
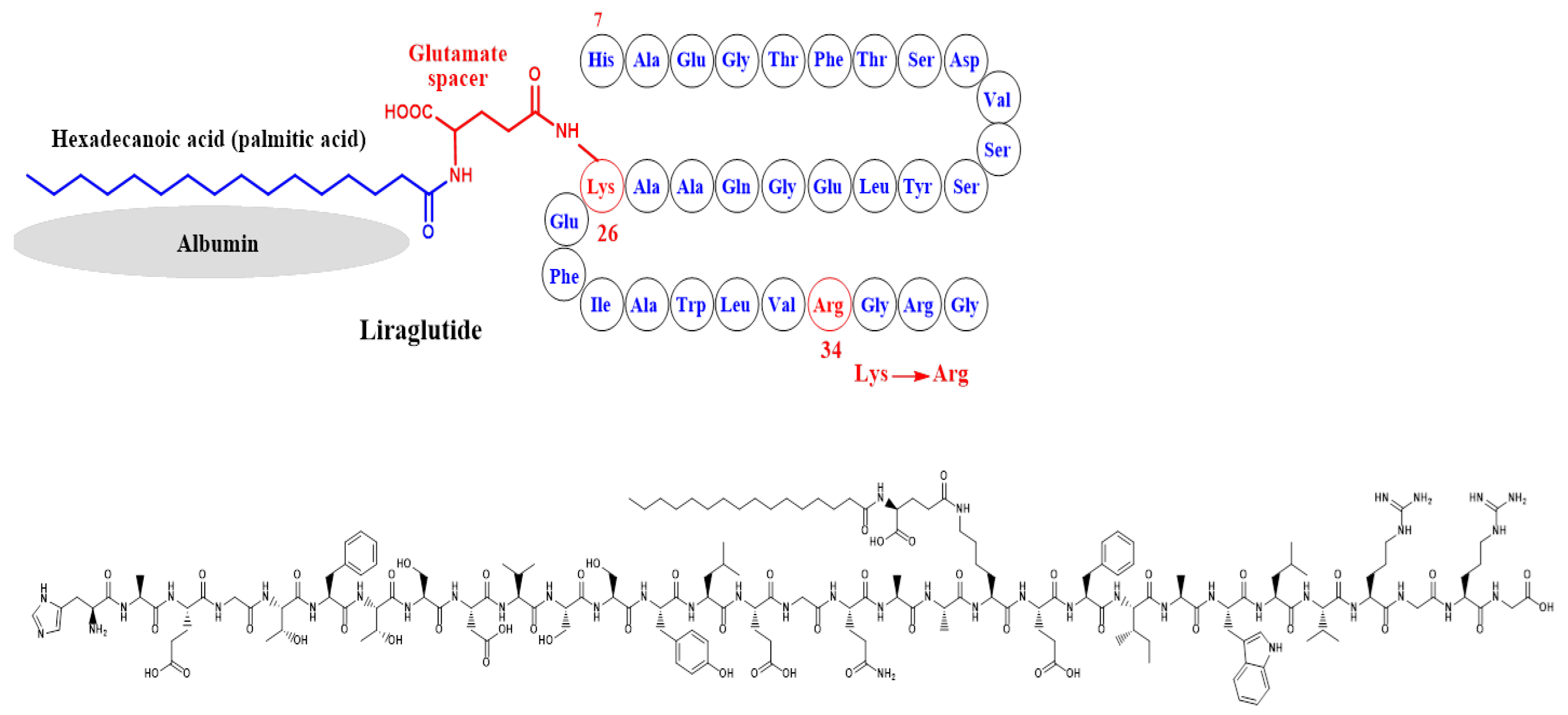
4. Liraglutide
4.1. Evidence from Clinical Trials in Diabetics and Non-Diabetics
4.2. Liraglutide at A Dose of 3 mg/Day in Non-Diabetics
4.3. Long-Term Follow-Up Studies
4.4. Average Weight Loss
4.5. Indications and Contraindications
4.5.1. Directions
4.5.2. Contraindications and Precautions
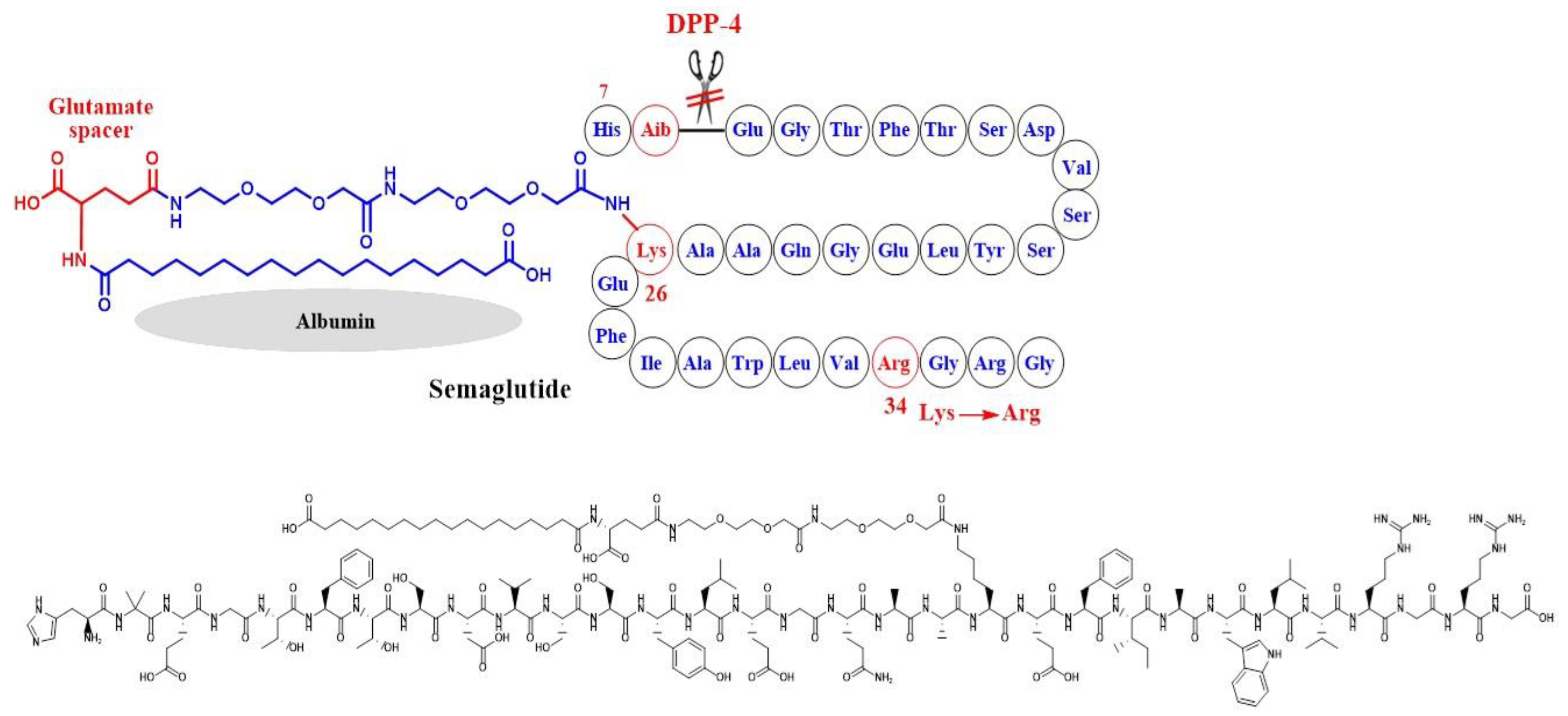
5. Semaglutide
5.1. Weight Loss Evidence from Clinical Trials in Diabetics and Non-Diabetics
5.2. Semaglutide High Doses for Non-Diabetic Obese
5.3. Long-Term Follow-Up Studies
5.4. Average Weight Loss
5.5. Indications and Contraindications
5.6. Adverse Effects
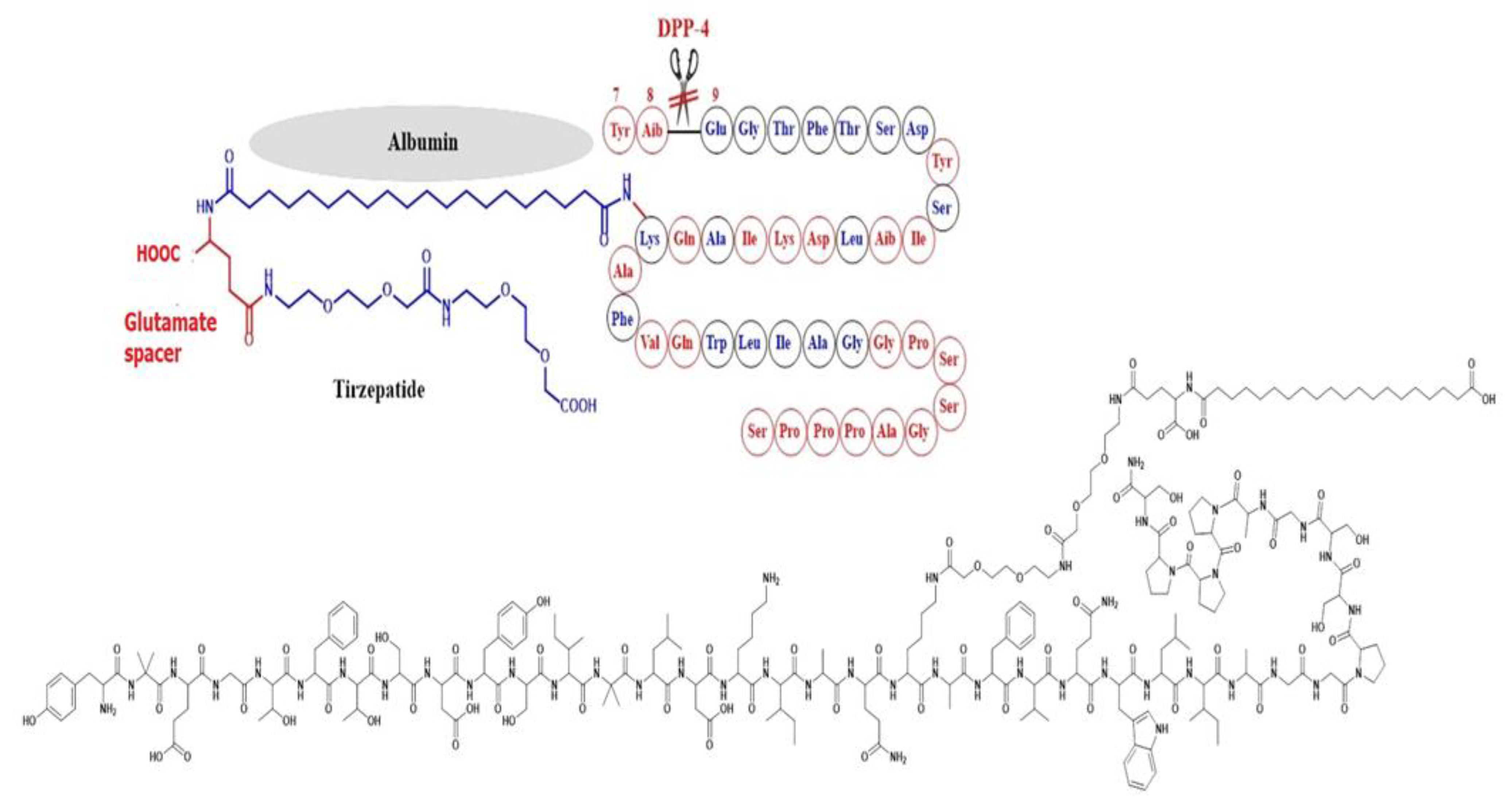
6. Tirzepatide
6.1. Studies Completed on Tirzepatide
6.2. Average Weight Loss
6.3. Indications and Contraindications
6.4. Adverse Effects
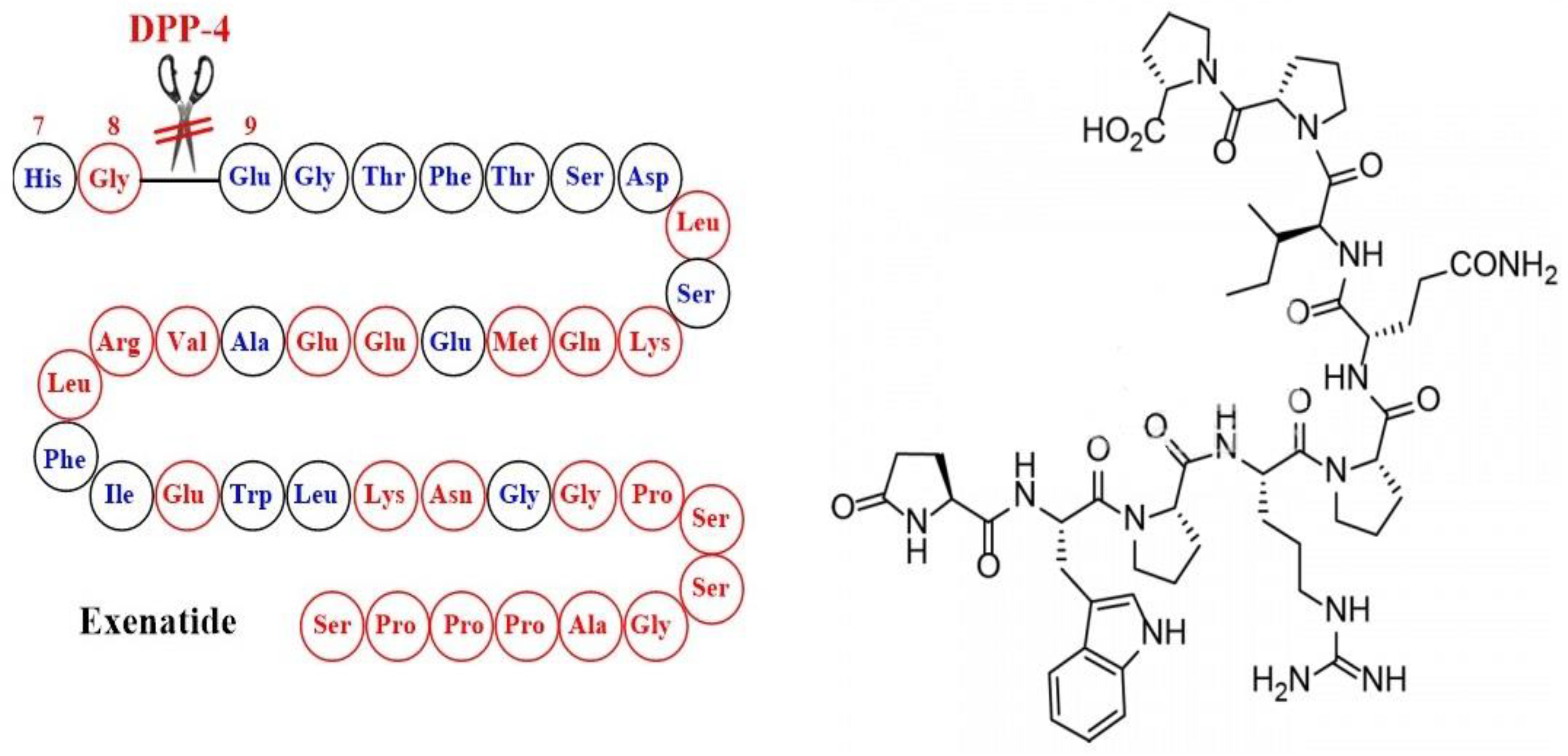
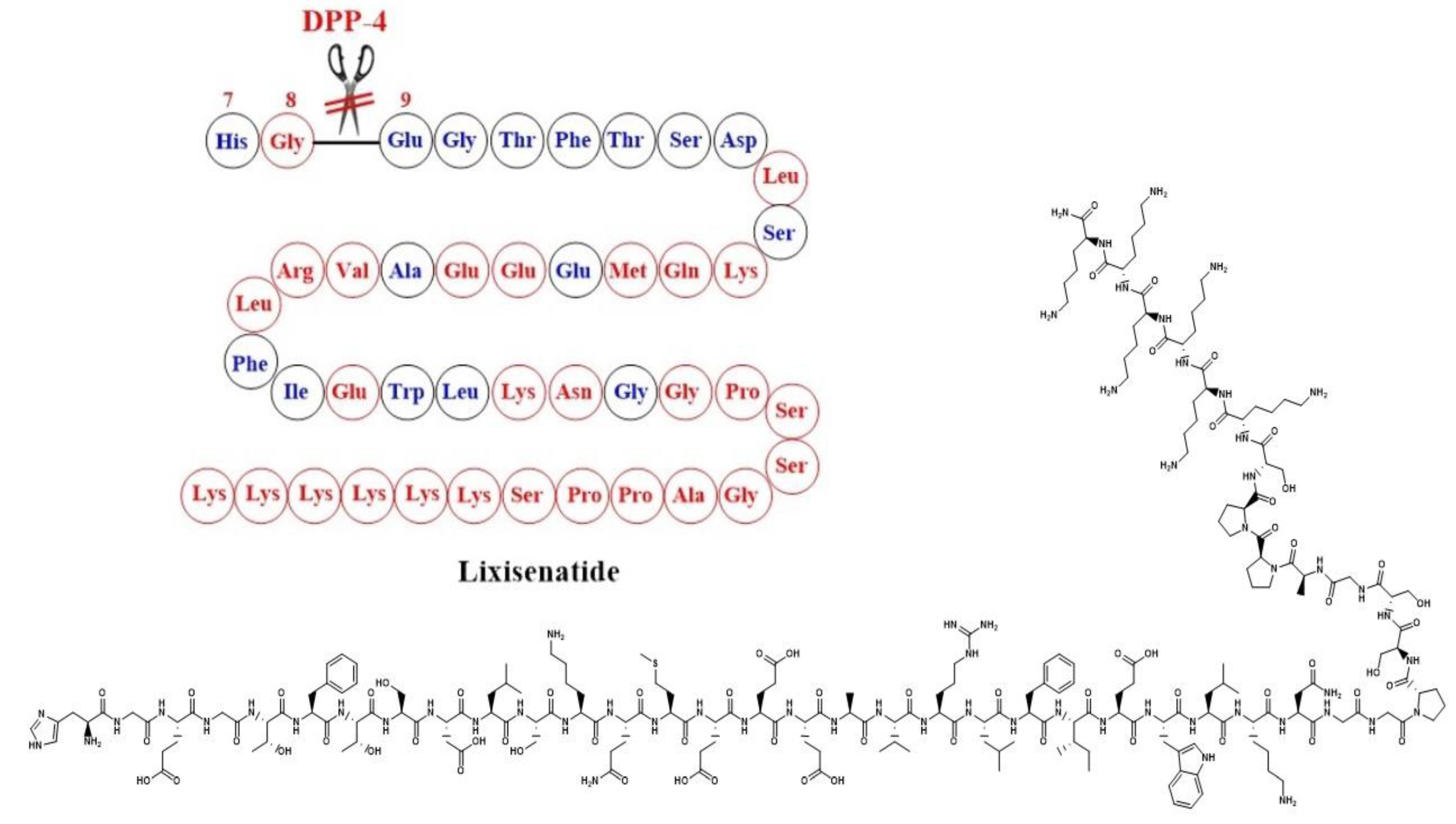
7. Lixisenatide and Exenatide-STUDIES and Evidence
7.1. Lixisenatide
7.2. Exenatide
8. Conclusions and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ritchie, H.; Roser, M. “Obesity”. Published online at OurWorldInData.org. 2017. Available online: https://ourworldindata.org/obesity (accessed on 10 January 2023).
- Lebovitz, H.E.; Banerji, M.A. Point: Visceral Adiposity Is Causally Related to Insulin Resistance. Diabetes Care 2005, 28, 2322–2325. [Google Scholar] [CrossRef]
- Apovian, C.M. Obesity: Definition, comorbidities, causes, and burden. Am. J. Manag. Care 2016, 22 (Suppl. S7), s176–s185. [Google Scholar]
- WHO European Regional Obesity Report 2022; WHO Regional Office for Europe: Copenhagen, Denmark, 2022.
- Roman, G.; Bala, C.; Creteanu, G.; Graur, M.; Morosanu, M.; Amorin, P.; Pîrcalaboiu, L.; Radulian, G.; Timar, R.; Cadariu, A.A. Obesity and Health-Related lifestyle factors in the general population in romania: A cross sectional study. Acta Endocrinol. 2015, 11, 64–71. [Google Scholar] [CrossRef]
- NCD-RisC. Homepage > NCD-RisC. Available online: https://www.ncdrisc.org/ (accessed on 15 February 2023).
- Mechanick, J.I.; Garber, A.J.; Handelsman, Y.; Garvey, W.T.; Beir, D.M.; Bohannon, N.J.; Bray, G.A.; Bush, M.A.; Evans, J.G.; Hurley, D.L.; et al. American Association of Clinical Endocrinologists’ Position Statement on Obesity and Obesity Medicine. Endocr. Pract. 2012, 18, 642–648. [Google Scholar] [CrossRef]
- Lindsted, K.; Tonstad, S.; Kazma, J.W. Body mass index and patterns of mortality amongst Seventh-day Adventist men. Int. J. Obes. 1991, 15, 397–406. [Google Scholar]
- Garvey, W.T. New Tools for Weight-Loss Therapy Enable a More Robust Medical Model for Obesity Treatment: Rationale for a Complications-Centric Approach. Endocr. Pract. 2013, 19, 864–874. [Google Scholar] [CrossRef]
- Garvey, W.T.; Mechanick, J.I.; Brett, E.M.; Garber, A.J.; Hurley, D.L.; Jastreboff, A.M.; Nadolsky, K.; Pessah-Pollack, R.; Plodkowski, R. American association of clinical endocrinologists and american college of endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocr. Pract. 2016, 22 (Suppl. S3), 1–203. [Google Scholar] [CrossRef]
- Mechanick, J.I.; Farkouh, M.E.; Newman, J.D.; Garvey, W.T. Cardiometabolic-based chronic disease, adiposity and dysglycemia drivers: JACC state-of-the-art review. J. Am. Coll. Cardiol. 2020, 75, 525–538. [Google Scholar] [CrossRef]
- Garvey, W.T. New Horizons. A New Paradigm for Treating to Target with Second-Generation Obesity Medications. J. Clin. Endocrinol. Metab. 2022, 107, e1339–e1347. [Google Scholar] [CrossRef]
- Sjostrom, C.D.; Lissner, L.; Wedel, H.; Sjostrom, L. Reduction in incidence of diabetes, hypertension and lipid disturbances after interventional weight loss induced by bariatric surgery: The SOS Intervention Study. Obes. Res. 1999, 7, 477–484. [Google Scholar] [CrossRef]
- Wilson, P.W.; D’Agostino, R.B.; Sullivan, L.; Parise, H.; Kannel, W.B. Overweight and obesity as determinants of cardiovascular risk: The Framingham experience. Arch. Intern. Med. 2002, 162, 1867–1872. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, E.J.; Levy, D.; Vaziri, S.M.; D’Agostino, R.B.; Belanger, A.J.; Wolf, P.A. Independent risk factors for atrial fibrillation in a population-based cohort. The Framingham Heart Study. JAMA 1994, 271, 840–844. [Google Scholar] [CrossRef]
- Colditz, G.; Willett, W.C.; Rotnitzky, A.; Manson, J.E. Weight Gain as a Risk Factor for Clinical Diabetes Mellitus in Women. Ann. Intern. Med. 1995, 122, 481–486. [Google Scholar] [CrossRef]
- Tirosh, A.; Shai, I.; Tekes-Manova, D.; Israeli, E.; Pereg, D.; Shochat, T.; Kochba, I.; Rudich, A. Normal Fasting Plasma Glucose Levels and Type 2 Diabetes in Young Men. N. Engl. J. Med. 2005, 353, 1454–1462. [Google Scholar] [CrossRef]
- Grundy, S.M.; Barnett, J.P. Metabolic and health complications of obesity. Dis. Mon. 1990, 36, 641–731. [Google Scholar]
- Grundy, S.M.; Cleeman, J.I.; Daniels, S.R.; Donato, K.A.; Eckel, R.H.; Franklin, B.A.; Gordon, D.J.; Krauss, R.M.; Savage, P.J.; Smith, S.C., Jr.; et al. Diagnosis and management of the metabolic syndrome. An American Heart Association/National Heart, Lung, and Blood Institute scientific statement. Curr. Opin. Cardiol. 2006, 21, 1–6. [Google Scholar] [CrossRef]
- Shetty, S.; Parthasarathy, S. Obesity hypoventilation syndrome. Curr. Pulmonol. Rep. 2015, 4, 42–55. [Google Scholar] [CrossRef] [PubMed]
- Canargo, C.A., Jr.; Weiss, S.T.; Zhang, S.; Willett, W.C.; Speizer, F.E. Prospective study of body mass index, weight change and risk of adult-onset asthma in women. Arch. Intern. Med. 1999, 159, 2582–2588. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, M.; Malhi, H.; Acosta, A. Gastrointestinal Complications of Obesity. Gastroenterology 2017, 152, 1656–1670. [Google Scholar] [CrossRef]
- Locke, G.R., III; Talley, N.J.; Fett, S.L.; Zinsmeister, A.R.; Melton, L.J., III. Risk factors associated with symptoms of gastroesophageal reflux. Am. J. Med. 1999, 106, 642–649. [Google Scholar] [CrossRef]
- Cavalu, S.; Popa, A.; Bratu, I.; Borodi, G.; Maghiar, A. New Evidences of Key Factors Involved in “Silent Stones” Etiopathogenesis and Trace Elements: Microscopic, Spectroscopic, and Biochemical Approach. Biol.Trace Elem. Res. 2015, 168, 311–320. [Google Scholar] [CrossRef]
- Padberg, F., Jr.; Cerveira, J.J.; Lal, B.K.; Pappas, P.J.; Varma, S.; Hobson, R.W., II. Does severe venous insufficiency have a different etiology in the morbidly obese? Is it venous? J. Vasc. Surg. 2003, 37, 79–85. [Google Scholar] [CrossRef]
- Barlow, S.E.; Dietz, W.H. Obesity Evaluation and Treatment: Expert Committee Recommendations. Pediatrics 1998, 102, e29. [Google Scholar] [CrossRef]
- McLaughlin, T.; Abbasi, F.; Kim, H.S.; Lamendola, C.; Schaaf, P.; Reaven, G. Relationship between insulin resistance, weight loss, and coronary heart disease in healthy, obese women. Metabolism 2001, 50, 759–800. [Google Scholar] [CrossRef] [PubMed]
- Calle, E.E.; Thun, M.J. Obesity and cancer. Oncogene 2004, 23, 6365–6378. [Google Scholar] [CrossRef]
- Danaei, G.; Vander Hoorn, S.; Lopez, A.D.; Murray, C.J.; Ezzati, M.; Comparative Risk Assessment Collaborating Group. Causes of cancer in the world: Comparative risk assessment of nine behavioural and environmental risk factors. Lancet 2005, 366, 1784–1793. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, C.L.; Ross, R.K.; Paganini-Hill, A.; Bernstein, L. Effect of family history, obesity and exercise on breast cancer risk among postmenopausal women. Int. J. Cancer 2003, 106, 96–102. [Google Scholar] [CrossRef]
- Vainio, H.; Bianchini, F. International Agency for Cancer handbook of cancer prevention. In Weight Control and Physical Activity; IARC: Lyon, France, 2002; Volume 6. [Google Scholar]
- The Rotterdam ESHRE/ASRM-sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil. Steril. 2004, 81, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Asfari, M.M.; Sarmini, M.T.; Baidoun, F.; Al-Khadra, Y.; Ezzaizi, Y.; Dasarathy, S.; McCullough, A. Association of non-alcoholic fatty liver disease and polycystic ovarian syndrome. BMJ Open Gastroenterol. 2020, 7, e000352. [Google Scholar] [CrossRef]
- Alorabi, M.; Cavalu, S.; Al-kuraishy, H.M.; Al-Gareeb, A.I.; Mostafa-Hedeab, G.; Negm, W.A.; Youssef, A.; El-Kadem, A.H.; Saad, H.M.; El-Saber Batiha, G. Pentoxifylline and berberine mitigate diclofenac-induced acute nephrotoxicity in male rats via modulation of inflammation and oxidative stress. Biomed. Pharmacother. 2022, 152, 113225. [Google Scholar] [CrossRef]
- Baig, M.S.; Banu, A.; Zehravi, M.; Rana, R.; Burle, S.S.; Khan, S.L.; Islam, F.; Siddiqui, F.A.; Massoud, E.E.S.; Rahman, M.H.; et al. An Overview of Diabetic Foot Ulcers and Associated Problems with Special Emphasis on Treatments with Antimicrobials. Life 2022, 12, 1054. [Google Scholar] [CrossRef]
- Sarma, S.; Sockalingam, S.; Dash, S. Obesity as a multisystem disease: Trends in obesity rates and obesity-related complications. Diabetes Obes. Metab. 2021, 23, 3–16. [Google Scholar] [CrossRef]
- Avila, C.; Holloway, A.C.; Hahn, M.K.; Morrison, K.M.; Restivo, M.; Anglin, R.; Taylor, V.H. An Overview of Links Between Obesity and Mental Health. Curr. Obes. Rep. 2015, 4, 303–310. [Google Scholar] [CrossRef]
- Lockhart, S.M.; O’Rahilly, S. When two pandemics meet: Why is obesity associated with increased COVID-19 mortality? Med 2020, 1, 33–42. [Google Scholar] [CrossRef]
- Sumithran, P.; Proietto, J. The defence of body weight: A physiological basis for weight regain after weight loss. Clin. Sci. 2013, 124, 231–241. [Google Scholar] [CrossRef]
- Leibel, R.L.; Rosenbaum, M.; Hirsch, J. Changes in Energy Expenditure Resulting from Altered Body Weight. N. Engl. J. Med. 1995, 332, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Al-Omar, M.A.; Al-Suwailem, A.K.; Al-Tamimi, A.S.; Al-Suhibani, M.S. Safety and mechanism of action of orlistat (tetrahydrolipstatin) as the first local antiobesity drug. J. Appl. Sci. Res. 2006, 2, 205–208. [Google Scholar]
- Lean, M.E.J.; Malkova, D. Altered gut and adipose tissue hormones in overweight and obese individuals: Cause or consequence? Int. J. Obes. 2016, 40, 622–632. [Google Scholar] [CrossRef]
- Rubino, D.; Abrahamsson, N.; Davies, M.; Hesse, D.; Greenway, F.L.; Jensen, C.; Lingvay, I.; Mosenzon, O.; Rosenstock, J.; Rubio, M.A.; et al. Effect of continued weekly subcutaneous semaglutide vs placebo on weight loss maintenance in adults with overweight or obesity: The STEP 4 randomized clinical trial. JAMA 2021, 325, 1414–1425. [Google Scholar] [CrossRef]
- Anderson, S.L.; Beutel, T.R.; Trujillo, J.M. Oral semaglutide in type 2 diabetes. J. Diabetes Its Complicat. 2020, 34, 107520. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, D. The structure and function of the glucagon-like peptide-1 receptor and its ligands. Br. J. Pharmacol. 2012, 166, 27–41. [Google Scholar] [CrossRef]
- Norris, S.L.; Lee, N.; Thakurta, S.; Chan, B.K.S. Exenatide efficacy and safety: A systematic review. Diabet. Med. 2009, 26, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Christensen, M.; Knop, F.K.; Holst, J.J.; Vilsboll, T. Lixisenatide, a novel GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus. IDrugs Investig. Drugs J. 2009, 12, 503–513. [Google Scholar]
- Cheang, J.Y.; Moyle, P.M. Glucagon-like peptide-1 (GLP-1)-based therapeutics: Current status and future opportunities beyond type 2 diabetes. ChemMedChem 2018, 13, 662–671. [Google Scholar] [CrossRef]
- Lau, J.; Bloch, P.; Schäffer, L.; Pettersson, I.; Spetzler, J.; Kofoed, J.; Madsen, K.; Knudsen, L.B.; McGuire, J.; Steensgaard, D.B.; et al. Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide. J. Med. Chem. 2015, 58, 7370–7380. [Google Scholar] [CrossRef]
- Wang, L. Designing a Dual GLP-1R/GIPR Agonist from Tirzepatide: Comparing Residues Between Tirzepatide, GLP-1, and GIP. Drug Des. Dev. Ther. 2022, 16, 1547–1559. [Google Scholar] [CrossRef]
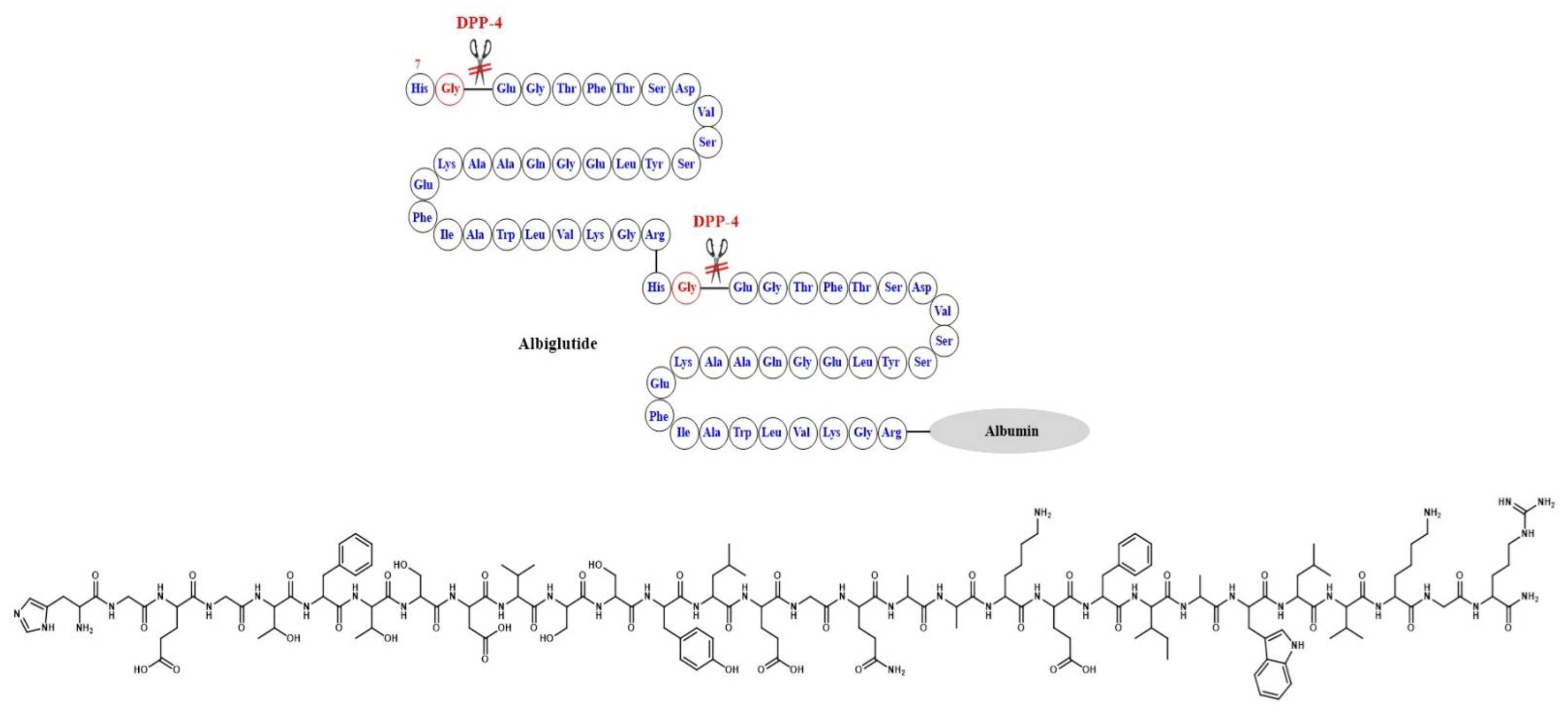
- Rendell, M.S. Albiglutide: A unique GLP-1 receptor agonist. Expert Opin. Biol. Ther. 2016, 16, 1557–1569. [Google Scholar] [CrossRef]
- Kaspar, A.A.; Reichert, J.M. Future directions for peptide therapeutics development. Drug Discov. Today 2013, 18, 807–817. [Google Scholar] [CrossRef]
- Pannacciulli, N.; Le, D.S.; Salbe, A.D.; Chen, K.; Reiman, E.M.; Tataranni, P.A.; Krakoff, J. Postprandial glucagon-like peptide-1 (GLP-1) response is positively associated with changes in neuronal activity of brain areas implicated in satiety and food intake regulation in humans. NeuroImage 2007, 35, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Crawley, J.N.; Beinfeld, M.C. Rapid development of tolerance to the behavioural actions of cholecystokinin. Nature 1983, 302, 703–706. [Google Scholar] [CrossRef]
- Moon, H.S.; Chamberland, J.P.; Diakopoulos, K.N.; Fiorenza, C.G.; Ziemke, F.; Schneider, B.; Mantzoros, C.S. Leptin and amylin act in an additive manner to activate overlapping signaling pathways in peripheral tissues: In vitro and ex vivo studies in humans. Diabetes Care 2011, 34, 132–138. [Google Scholar] [CrossRef]
- Drucker, D.J.; Nauck, M.A. The incretin system: Glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet 2006, 368, 1696–1705. [Google Scholar] [CrossRef]
- Nauck, M.A.; Meier, J.J. Incretin hormones: Their role in health and disease. Diabetes Obes. Metab. 2018, 20 (Suppl. S1), 5–21. [Google Scholar] [CrossRef] [PubMed]
- Bettge, K.; Kahle, M.; El Aziz, M.S.A.; Meier, J.J.; Nauck, M.A. Occurrence of nausea, vomiting and diarrhoea reported as adverse events in clinical trials studying glucagon-like peptide-1 receptor agonists: A systematic analysis of published clinical trials. Diabetes Obes. Metab. 2017, 19, 336–347. [Google Scholar] [CrossRef]
- van Bloemendaal, L.; Kulve, J.S.T.; la Fleur, S.E.; Ijzerman, R.G.; Diamant, M. Effects of glucagon-like peptide 1 on appetite and body weight: Focus on the CNS. J. Endocrinol. 2014, 221, T1–T16. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Belousoff, M.J.; Zhao, P.; Kooistra, A.J.; Truong, T.T.; Ang, S.Y.; Underwood, C.R.; Egebjerg, T.; Šenel, P.; Stewart, G.D.; et al. Differential GLP-1R Binding and Activation by Peptide and Non-peptide Agonists. Mol. Cell 2020, 80, 485–500.e7. [Google Scholar] [CrossRef]
- Kim, D.; MacConell, L.; Zhuang, D.; Kothare, P.A.; Trautmann, M.; Fineman, M.; Taylor, K. Effects of Once-Weekly Dosing of a Long-Acting Release Formulation of Exenatide on Glucose Control and Body Weight in Subjects with Type 2 Diabetes. Diabetes Care 2007, 30, 1487–1493. [Google Scholar] [CrossRef] [PubMed]
- Hedrington, M.S.; Davis, S.N. Oral semaglutide for the treatment of type 2 diabetes. Expert Opin. Pharmacother. 2019, 20, 133–141. [Google Scholar] [CrossRef]
- Nauck, M.A.; Quast, D.R.; Wefers, J.; Meier, J.J. GLP-1 receptor agonists in the treatment of type 2 diabetes—State-of-the-art. Mol. Metab. 2021, 46, 101102. [Google Scholar] [CrossRef]
- Sheahan, K.H.; Wahlberg, E.A.; Gilbert, M.P. An overview of GLP-1 agonists and recent cardiovascular outcomes trials. Postgrad. Med. J. 2020, 96, 156–161. [Google Scholar] [CrossRef]
- Zelniker, T.A.; Wiviott, S.D.; Raz, I.; Im, K.; Goodrich, E.; Bonaca, M.P.; Mosenzon, O.; Kato, E.; Cahn, A.; Furtado, R.H.M.; et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: A systematic review and meta-analysis of cardiovascular outcome trials. Lancet 2019, 393, 31–39. [Google Scholar] [CrossRef]
- Marre, M.; Shaw, J.; Brändle, M.; Bebakar, W.M.W.; Kamaruddin, N.A.; Strand, J.; Zdravkovic, M.; Le Thi, T.D.; Colagiuri, S.; LEAD-1 SU study group. Liraglutide, a once-daily human GLP-1 analogue, added to a sulphonylurea over 26 weeks produces greater improvements in glycaemic and weight control compared with adding rosiglitazone or placebo in subjects with Type 2 diabetes (LEAD-1 SU). Diabet. Med. 2009, 26, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Nauck, M.; Frid, A.; Hermansen, K.; Shah, N.S.; Tankova, T.; Mitha, I.H.; Zdravkovic, M.; Düring, M.; Matthews, D.R.; LEAD-2 Study Group. Efficacy and safety comparison of liraglutide, glimepiride, and placebo, all in combination with metformin, in type 2 diabetes: The LEAD (liraglutide effect and action in diabetes)-2 study. Diabetes Care 2009, 32, 84–90. [Google Scholar] [CrossRef]
- Garber, A.; Henry, R.; Ratner, R.; Garcia-Hernandez, P.A.; Rodriguez-Pattzi, H.; Olvera-Alvarez, I.; Hale, P.M.; Zdravkovic, M.; Bode, B.; LEAD-3 (Mono) Study Group. Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD-3 Mono): A randomised, 52-week, phase III, double-blind, parallel-treatment trial. Lancet 2009, 373, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Zinman, B.; Gerich, J.; Buse, J.B.; Lewin, A.; Schwartz, S.; Raskin, P.; Hale, P.M.; Zdravkovic, M.; Blonde, L.; LEAD-4 Study Investigators. Efficacy and Safety of the Human Glucagon-Like Peptide-1 Analog Liraglutide in Combination with Metformin and Thiazolidinedione in Patients With Type 2 Diabetes (LEAD-4 Met+TZD). Diabetes Care 2009, 32, 1224–1230. [Google Scholar] [CrossRef] [PubMed]
- Buse, J.B.; Rosenstock, J.; Sesti, G.; Schmidt, W.E.; Montanya, E.; Brett, J.H.; Zychma, M.; Blonde, L.; LEAD-6 Study Group. Liraglutide once a day versus exenatide twice a day for type 2 diabetes: A 26-week randomised, parallel-group, multinational, open-label trial (LEAD-6). Lancet 2009, 374, 39–47. [Google Scholar] [CrossRef]
- Astrup, A.; Rössner, S.; Van Gaal, L.; Rissanen, A.; Niskanen, L.; Al Hakim, M.; Madsen, J.; Rasmussen, M.F.; Lean, M.E.; NN8022-1807 Study Group. Effects of liraglutide in the treatment of obesity: A randomised, double-blind, placebo-controlled study. Lancet 2009, 374, 1606–1616. [Google Scholar] [CrossRef] [PubMed]
- Kanwal, S.; AlEmadi, E.A. Liraglutide for Weight Management, Critical Analysis of Efficacy and Side effects in Non diabetic, individuals with obesity: A Comprehensive Systematic Review. Middle East J. Fam. Med. 2021, 19, 72–83. [Google Scholar]
- Field, B.C.T.; Wren, A.M.; Cooke, D.; Bloom, S.R. Gut Hormones as Potential New Targets for Appetite Regulation and the Treatment of Obesity. Drugs 2008, 68, 147–163. [Google Scholar] [CrossRef]
- Zander, M.; Madsbad, S.; Madsen, J.L.; Holst, J.J. Effect of 6-week course of glucagon-like peptide 1 on glycaemic control, insulin sensitivity, and β-cell function in type 2 diabetes: A parallel-group study. Lancet 2002, 359, 824–830. [Google Scholar] [CrossRef]
- Garber, A.J. Incretin-based therapies in the management of type 2 diabetes: Rationale and reality in a managed care setting. Am. J. Manag. Care 2010, 16 (Suppl. S7), S187–S194. [Google Scholar] [PubMed]
- Isidro, M.L.; Cordido, F. Drug treatment of obesity: Established and emerging therapies. Mini-Rev. Med. Chem. 2009, 9, 664–673. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.J.; Kela, R.; Khunti, K. Liraglutide—Overview of the preclinical and clinical data and its role in the treatment of type 2 diabetes. Diabetes Obes. Metab. 2011, 13, 207–220. [Google Scholar] [CrossRef]
- Lean, M.E.; Carraro, R.; Finer, N.; Hartvig, H.; Lindegaard, M.L.; Rössner, S.; Van Gaal, L.; Astrup, A. Tolerability of nausea and vomiting and associations with weight loss in a randomized trial of liraglutide in obese, non-diabetic adults. Int. J. Obes. 2014, 38, 689–697. [Google Scholar] [CrossRef]
- Sullivan, S.D.; Alfonso-Cristancho, R.; Conner, C.; Hammer, M.; Blonde, L. Long-term outcomes in patients with type 2 diabetes receiving glimepiride combined with liraglutide or rosiglitazone. Cardiovasc. Diabetol. 2009, 8, 12–19. [Google Scholar] [CrossRef]
- le Roux, C.W.; Astrup, A.; Fujioka, K.; Greenway, F.; Lau, D.C.W.; Van Gaal, L.; Ortiz, R.V.; Wilding, J.P.H.; Skjøth, T.V.; Manning, L.S.; et al. 3 years of liraglutide versus placebo for type 2 diabetes risk reduction and weight management in individuals with prediabetes: A randomised, double-blind trial. Lancet 2017, 389, 1399–1409. [Google Scholar] [CrossRef]
- Russell-Jones, D.; Vaag, A.; Schmitz, O.; Sethi, B.K.; Lalic, N.; Antic, S.; Zdravkovic, M.; Ravn, G.M.; Simó, R.; Liraglutide Effect and Action in Diabetes 5 (LEAD-5) met+SU Study Group. Liraglutide vs. insulin glargine and placebo in combination with metformin and sulfonylurea therapy in type 2 diabetes mellitus (LEAD-5 met+SU): A randomised controlled trial. Diabetologia 2009, 52, 2046–2055. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.; Marso, S.P.; Neeland, I.J. Liraglutide for weight management: A critical review of the evidence. Obes. Sci. Pract. 2017, 3, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Lundgren, J.R.; Janus, C.; Jensen, S.B.; Juhl, C.R.; Olsen, L.M.; Christensen, R.M.; Svane, M.S.; Bandholm, T.; Bojsen-Møller, K.N.; Blond, M.B.; et al. Healthy Weight Loss Maintenance with Exercise, Liraglutide, or Both Combined. N. Engl. J. Med. 2021, 384, 1719–1730. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, L.V.; Flint, A.; Olsen, A.K.; Ingwersen, S.H. Liraglutide in Type 2 Diabetes Mellitus: Clinical Pharmacokinetics and Pharmacodynamics. Clin. Pharmacokinet. 2016, 55, 657–672. [Google Scholar] [CrossRef] [PubMed]
- Dejgaard, T.F.; Frandsen, C.S.; Hansen, T.S.; Almdal, T.; Urhammer, S.; Pedersen-Bjergaard, U.; Jensen, T.; Jensen, A.K.; Holst, J.J.; Tarnow, L.; et al. Efficacy and safety of liraglutide for overweight adult patients with type 1 diabetes and insufficient glycaemic control (Lira-1): A randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 2016, 4, 221–232. [Google Scholar] [CrossRef]
- Zhang, P.; Liu, Y.; Ren, Y.; Bai, J.; Zhang, G.; Cui, Y. The efficacy and safety of liraglutide in the obese, non-diabetic individuals: A systematic review and meta-analysis. Afr. Health Sci. 2019, 19, 2591–2599. [Google Scholar] [CrossRef] [PubMed]
- Siamashvili, M.; Davis, S.N. Update on the effects of GLP-1 receptor agonists for the treatment of polycystic ovary syndrome. Expert Rev. Clin. Pharmacol. 2021, 14, 1081–1089. [Google Scholar] [CrossRef]
- Mikhail, N. Cardiovascular Effects of Liraglutide. Curr. Hypertens. Rev. 2019, 15, 64–69. [Google Scholar] [CrossRef]
- Marso, S.P.; Daniels, G.H.; Brown-Frandsen, K.; Kristensen, P.; Mann, J.F.E.; Nauck, M.A.; Nissen, S.E.; Pocock, S.; Poulter, N.R.; Ravn, L.S.; et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Consoli, A.; Formoso, G. Potential side effects to GLP-1 agonists: Understanding their safety and tolerability. Expert Opin. Drug Saf. 2015, 14, 207–218. [Google Scholar] [CrossRef]
- Egan, J.M.; Chia, C.W. Incretin Therapy and Pancreatic Pathologies: Background Pathology Versus Drug-Induced Pathology in Rats. Diabetes 2014, 63, 1174–1178. [Google Scholar] [CrossRef]
- Monami, M.; Dicembrini, I.; Nardini, C.; Fiordelli, I.; Mannucci, E. Glucagon-like peptide-1 receptor agonists and pancreatitis: A meta-analysis of randomized clinical trials. Diabetes Res. Clin. Pract. 2014, 103, 269–275. [Google Scholar] [CrossRef]
- Steinberg, W.M.; Rosenstock, J.; Wadden, T.A.; Donsmark, M.; Jensen, C.B.; DeVries, J.H. Impact of Liraglutide on Amylase, Lipase, and Acute Pancreatitis in Participants With Overweight/Obesity and Normoglycemia, Prediabetes, or Type 2 Diabetes: Secondary Analyses of Pooled Data From the SCALE Clinical Development Program. Diabetes Care 2017, 40, 839–848. [Google Scholar] [CrossRef]
- Pi-Sunyer, X.; Astrup, A.; Fujioka, K.; Greenway, F.; Halpern, A.; Krempf, M.; Lau, D.C.W.; Le Roux, C.W.; Ortiz, R.V.; Jensen, C.B.; et al. A Randomized, Controlled Trial of 3.0 mg of Liraglutide in Weight Management. N. Engl. J. Med. 2015, 373, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Nexøe-Larsen, C.C.; Sørensen, P.H.; Hausner, H.; Agersnap, M.; Baekdal, M.; Brønden, A.; Gustafsson, L.N.; Sonne, D.P.; Vedtofte, L.; Vilsbøll, T.; et al. Effects of liraglutide on gallbladder emptying: A randomized, placebo-controlled trial in adults with overweight or obesity. Diabetes Obes. Metab. 2018, 20, 2557–2564. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, P.M.; Aroda, V.R.; Astrup, A.; Kushner, R.; Lau, D.C.W.; Wadden, T.A.; Brett, J.; Cancino, A.-P.; Wilding, J.P.H.; Satiety and Clinical Adiposity—Liraglutide Evidence in individuals with and without diabetes (SCALE) study groups. Neuropsychiatric safety with liraglutide 3.0 mg for weight management: Results from randomized controlled phase 2 and 3a trials. Diabetes Obes. Metab. 2017, 19, 1529–1536. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.J.; Bain, S.C.; Atkin, S.L.; Rossing, P.; Scott, D.; Shamkhalova, M.S.; Bosch-Traberg, H.; Syrén, A.; Umpierrez, G.E. Efficacy and Safety of Liraglutide Versus Placebo as Add-on to Glucose-Lowering Therapy in Patients With Type 2 Diabetes and Moderate Renal Impairment (LIRA-RENAL): A Randomized Clinical Trial. Diabetes Care 2016, 39, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Davidson, J.A.; Brett, J.; Falahati, A.; Scott, D. Mild renal impairment and the efficacy and safety of liraglutide. Endocr. Pract. 2011, 17, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Tomlinson, B.; Hu, M.; Zhang, Y.; Chan, P.; Liu, Z.-M. Effects of glucose-lowering drugs on cardiovascular outcomes in patients with type 2 diabetes. Expert Opin. Drug Metab. Toxicol. 2016, 12, 1267–1271. [Google Scholar] [CrossRef]
- Jorsal, A.; Kistorp, C.; Holmager, P.; Tougaard, R.S.; Nielsen, R.; Hänselmann, A.; Nilsson, B.; Møller, J.E.; Hjort, J.; Rasmussen, J.; et al. Effect of liraglutide, a glucagon-like peptide-1 analogue, on left ventricular function in stable chronic heart failure patients with and without diabetes (LIVE)-a multicentre, double-blind, randomised, placebo-controlled trial. Eur. J. Heart Fail. 2017, 19, 69–77. [Google Scholar] [CrossRef]
- Lin, C.-H.; Shao, L.; Zhang, Y.-M.; Tu, Y.-J.; Zhang, Y.; Tomlinson, B.; Chan, P.; Liu, Z. An evaluation of liraglutide including its efficacy and safety for the treatment of obesity. Expert Opin. Pharmacother. 2019, 21, 275–285. [Google Scholar] [CrossRef]
- Wilding, J.P.H.; Batterham, R.L.; Calanna, S.; Davies, M.; Van Gaal, L.F.; Lingvay, I.; McGowan, B.M.; Rosenstock, J.; Tran, M.T.; Wadden, T.A.; et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N. Engl. J. Med. 2021, 384, 989–1002. [Google Scholar] [CrossRef]
- Davies, M.; Færch, L.; Jeppesen, O.K.; Pakseresht, A.; Pedersen, S.D.; Perreault, L.; Rosenstock, J.; Shimomura, I.; Viljoen, A.; Wadden, T.A.; et al. Semaglutide 2·4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2): A randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial. Lancet 2021, 397, 971–984. [Google Scholar] [CrossRef] [PubMed]
- Wadden, T.A.; Bailey, T.S.; Billings, L.K.; Davies, M.; Frias, J.P.; Koroleva, A.; Lingvay, I.; O’Neil, P.M.; Rubino, D.M.; Skovgaard, D.; et al. Effect of Subcutaneous Semaglutide vs Placebo as an Adjunct to Intensive Behavioral Therapy on Body Weight in Adults With Overweight or Obesity: The STEP 3 Randomized Clinical Trial. JAMA 2021, 325, 1403–1413. [Google Scholar] [CrossRef] [PubMed]
- Garvey, W.T.; Batterham, R.L.; Bhatta, M.; Buscemi, S.; Christensen, L.N.; Frias, J.P.; Jódar, E.; Kandler, K.; Rigas, G.; Wadden, T.A.; et al. Two-year effects of semaglutide in adults with overweight or obesity: The STEP 5 trial. Nat. Med. 2022, 28, 2083–2091. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, T.; Isendahl, J.; Khalid, U.; Lee, S.Y.; Nishida, T.; Ogawa, W.; Tobe, K.; Yamauchi, T.; Lim, S.; STEP 6 investigators. Semaglutide once a week in adults with overweight or obesity, with or without type 2 diabetes in an east Asian population (STEP 6): A randomised, double-blind, double-dummy, placebo-controlled, phase 3a trial. Lancet Diabetes Endocrinol. 2022, 10, 193–206. [Google Scholar] [CrossRef]
- Rubino, D.M.; Greenway, F.L.; Khalid, U.; O’neil, P.M.; Rosenstock, J.; Sørrig, R.; Wadden, T.A.; Wizert, A.; Garvey, W.T.; STEP 8 Investigators; et al. Effect of Weekly Subcutaneous Semaglutide vs Daily Liraglutide on Body Weight in Adults with Overweight or Obesity Without Diabetes: The STEP 8 Randomized Clinical Trial. JAMA 2022, 327, 138–150. [Google Scholar] [CrossRef]
- Weghuber, D.; Barrett, T.; Barrientos-Pérez, M.; Gies, I.; Hesse, D.; Jeppesen, O.K.; Kelly, A.S.; Mastrandrea, L.D.; Sørrig, R.; Arslanian, S.; et al. Once-Weekly Semaglutide in Adolescents with Obesity. N. Engl. J. Med. 2022, 387, 2245–2257. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Hua, X.; Wang, X.; Xu, W.; Zhang, Y.; Shi, C.; Gu, M. Efficacy and safety of semaglutide on weight loss in obese or overweight patients without diabetes: A systematic review and meta-analysis of randomized controlled trials. Front. Pharmacol. 2022, 13, 935823. [Google Scholar] [CrossRef] [PubMed]
- Shaman, A.M.; Bain, S.C.; Bakris, G.L.; Buse, J.B.; Idorn, T.; Mahaffey, K.W.; Mann, J.F.; Nauck, M.A.; Rasmussen, S.; Rossing, P.; et al. Effect of the Glucagon-Like Peptide-1 Receptor Agonists Semaglutide and Liraglutide on Kidney Outcomes in Patients With Type 2 Diabetes: Pooled Analysis of SUSTAIN 6 and LEADER. Circulation 2022, 145, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Hughes, S.; Neumiller, J.J. Oral Semaglutide. Clin. Diabetes 2020, 38, 109–111. [Google Scholar] [CrossRef] [PubMed]
- Clinical Review Report: Semaglutide (Ozempic): (Novo Nordisk Canada Inc.): Indication: For the Treatment of Adult Patients with Type 2 Diabetes Mellitus to Improve Glycemic Control, in Combination with Metformin (Second-Line Treatment), and in Combination with Metformin and Sulfonylurea (Third-Line Treatment) [Internet]; Canadian Agency for Drugs and Technologies in Health: Ottawa, ON, Canada, 2019.
- Monami, M.; Nreu, B.; Scatena, A.; Cresci, B.; Andreozzi, F.; Sesti, G.; Mannucci, E. Safety issues with glucagon-like peptide-1 receptor agonists (pancreatitis, pancreatic cancer and cholelithiasis): D ata from randomized controlled trials. Diabetes Obes. Metab. 2017, 19, 1233–1241. [Google Scholar] [CrossRef] [PubMed]
- Tsapas, A.; Avgerinos, I.; Karagiannis, T.; Malandris, K.; Manolopoulos, A.; Andreadis, P.; Liakos, A.; Matthews, D.R.; Bekiari, E. Comparative Effectiveness of Glucose-Lowering Drugs for Type 2 Diabetes: A Systematic Review and Network Meta-analysis. Ann. Intern. Med. 2020, 173, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Nevola, R.; Epifani, R.; Imbriani, S.; Tortorella, G.; Aprea, C.; Galiero, R.; Rinaldi, L.; Marfella, R.; Sasso, F.C. GLP-1 Receptor Agonists in Non-Alcoholic Fatty Liver Disease: Current Evidence and Future Perspectives. Int. J. Mol. Sci. 2023, 24, 1703. [Google Scholar] [CrossRef]
- Wong, C.; Lee, M.H.; Yaow, C.Y.L.; Chin, Y.H.; Goh, X.L.; Ng, C.H.; Lim, A.Y.L.; Muthiah, M.D.; Khoo, C.M. Glucagon-Like Peptide-1 Receptor Agonists for Non-Alcoholic Fatty Liver Disease in Type 2 Diabetes: A Meta-Analysis. Front. Endocrinol. 2021, 12, 609110. [Google Scholar] [CrossRef]
- Rosenstock, J.; Wysham, C.; Frías, J.P.; Kaneko, S.; Lee, C.J.; Landó, L.F.; Mao, H.; Cui, X.; Karanikas, C.A.A.; Thieu, V.T. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): A double-blind, randomised, phase 3 trial. Lancet 2021, 398, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Frías, J.P.; Davies, M.J.; Rosenstock, J.; Pérez Manghi, F.C.; Fernández Landó, L.; Bergman, B.K.; Liu, B.; Cui, X.; Brown, K.; SURPASS-2 Investigators. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N. Engl. J. Med. 2021, 385, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Ludvik, B.; Giorgino, F.; Jódar, E.; Frias, J.P.; Landó, L.F.; Brown, K.; Bray, R.; Rodríguez, Á. Once-weekly tirzepatide versus once-daily insulin degludec as add-on to metformin with or without SGLT2 inhibitors in patients with type 2 diabetes (SURPASS-3): A randomised, open-label, parallel-group, phase 3 trial. Lancet 2021, 398, 583–598. [Google Scholar] [CrossRef] [PubMed]
- Del Prato, S.; Kahn, S.E.; Pavo, I.; Weerakkody, G.J.; Yang, Z.; Doupis, J.; Aizenberg, D.; Wynne, A.G.; Riesmeyer, J.S.; Heine, R.J.; et al. Tirzepatide versus insulin glargine in type 2 diabetes and increased cardiovascular risk (SURPASS-4): A randomised, open-label, parallel-group, multicentre, phase 3 trial. Lancet 2021, 398, 1811–1824. [Google Scholar] [CrossRef] [PubMed]
- Dahl, D.; Onishi, Y.; Norwood, P.; Huh, R.; Bray, R.; Patel, H.; Rodríguez, Á. Effect of Subcutaneous Tirzepatide vs Placebo Added to Titrated Insulin Glargine on Glycemic Control in Patients With Type 2 Diabetes: The SURPASS-5 Randomized Clinical Trial. JAMA 2022, 327, 534. [Google Scholar] [CrossRef]
- Inagaki, N.; Takeuchi, M.; Oura, T.; Imaoka, T.; Seino, Y. Efficacy and safety of tirzepatide monotherapy compared with dulaglutide in Japanese patients with type 2 diabetes (SURPASS J-mono): A double-blind, multicentre, randomised, phase 3 trial. Lancet Diabetes Endocrinol. 2022, 10, 623–633. [Google Scholar] [CrossRef]
- Kadowaki, T.; Chin, R.; Ozeki, A.; Imaoka, T.; Ogawa, Y. Safety and efficacy of tirzepatide as an add-on to single oral antihyperglycaemic medication in patients with type 2 diabetes in Japan (SURPASS J-combo): A multicentre, randomised, open-label, parallel-group, phase 3 trial. Lancet Diabetes Endocrinol. 2022, 10, 634–644. [Google Scholar] [CrossRef]
- Jastreboff, A.M.; Aronne, L.J.; Ahmad, N.N.; Wharton, S.; Connery, L.; Alves, B.; Kiyosue, A.; Zhang, S.; Liu, B.; Bunck, M.C.; et al. Tirzepatide Once Weekly for the Treatment of Obesity. N. Engl. J. Med. 2022, 387, 205–216. [Google Scholar] [CrossRef]
- Farzam, K.; Patel, P. Tirzepatide. 2022 Nov 24. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Fonseca, V.A.; Alvarado-Ruiz, R.; Raccah, D.; Boka, G.; Miossec, P.; Gerich, J.E.; EFC6018 GetGoal-Mono Study Investigators. Efficacy and Safety of the Once-Daily GLP-1 Receptor Agonist Lixisenatide in Monotherapy: A randomized, double-blind, placebo-controlled trial in patients with type 2 diabetes (GetGoal-Mono). Diabetes Care 2012, 35, 1225–1231. [Google Scholar] [CrossRef] [PubMed]
- Ahrén, B.; Dimas, A.L.; Miossec, P.; Saubadu, S.; Aronson, R. Efficacy and Safety of Lixisenatide Once-Daily Morning or Evening Injections in Type 2 Diabetes Inadequately Controlled on Metformin (GetGoal-M). Diabetes Care 2013, 36, 2543–2550. [Google Scholar] [CrossRef] [PubMed]
- Rosenstock, J.; Raccah, D.; Korányi, L.; Maffei, L.; Boka, G.; Miossec, P.; Gerich, J.E. Efficacy and Safety of Lixisenatide Once Daily Versus Exenatide Twice Daily in Type 2 Diabetes Inadequately Controlled on Metformin: A 24-week, randomized, open-label, active-controlled study (GetGoal-X). Diabetes Care 2013, 36, 2945–2951. [Google Scholar] [CrossRef] [PubMed]
- Bolli, G.B.; Munteanu, M.; Dotsenko, S.; Niemoeller, E.; Boka, G.; Wu, Y.; Hanefeld, M. Efficacy and safety of lixisenatide once daily vs. placebo in people with Type 2 diabetes insufficiently controlled on metformin (GetGoal-F1). Diabet. Med. 2014, 31, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Rosenstock, J.; Hanefeld, M.; Shamanna, P.; Min, K.W.; Boka, G.; Miossec, P.; Zhou, T.; Muehlen-Bartmer, I.; Ratner, R.E. Beneficial effects of once-daily lixisenatide on overall and postprandial glycemic levels without significant excess of hypoglycemia in Type 2 diabetes inadequately controlled on a sulfonylurea with or without metformin (GetGoal-S). J. Diabetes Its Complicat. 2014, 28, 386–392. [Google Scholar] [CrossRef] [PubMed]
- Pinget, M.; Goldenberg, R.; Niemoeller, E.; Muehlen-Bartmer, I.; Guo, H.; Aronson, R. Efficacy and safety of lixisenatide once daily versus placebo in type 2 diabetes insufficiently controlled on pioglitazone (GetGoal-P). Diabetes Obes. Metab. 2013, 15, 1000–1007. [Google Scholar] [CrossRef] [PubMed]
- Riddle, M.C.; Aronson, R.; Home, P.; Marre, M.; Niemoeller, E.; Miossec, P.; Ping, L.; Ye, J.; Rosenstock, J. Adding Once-Daily Lixisenatide for Type 2 Diabetes Inadequately Controlled by Established Basal Insulin: A 24-week, randomized, placebo-controlled comparison (GetGoal-L). Diabetes Care 2013, 36, 2489–2496. [Google Scholar] [CrossRef]
- Riddle, M.C.; Forst, T.; Aronson, R.; Sauque-Reyna, L.; Souhami, E.; Silvestre, L.; Ping, L.; Rosenstock, J. Adding Once-Daily Lixisenatide for Type 2 Diabetes Inadequately Controlled With Newly Initiated and Continuously Titrated Basal Insulin Glargine: A 24-week, randomized, placebo-controlled study (GetGoal-Duo 1). Diabetes Care 2013, 36, 2497–2503. [Google Scholar] [CrossRef]
- Rosenstock, J.; Guerci, B.; Hanefeld, M.; Gentile, S.; Aronson, R.; Tinahones, F.J.; Roy-Duval, C.; Souhami, E.; Wardecki, M.; Ye, J.; et al. Prandial Options to Advance Basal Insulin Glargine Therapy: Testing Lixisenatide Plus Basal Insulin Versus Insulin Glulisine Either as Basal-Plus or Basal-Bolus in Type 2 Diabetes: The GetGoal Duo-2 Trial. Diabetes Care 2016, 39, 1318–1328. [Google Scholar] [CrossRef]
- Meneilly, G.S.; Roy-Duval, C.; Alawi, H.; Dailey, G.; Bellido, D.; Trescoli, C.; Hurtado, H.M.; Guo, H.; Pilorget, V.; Perfetti, R.; et al. Lixisenatide Therapy in Older Patients With Type 2 Diabetes Inadequately Controlled on Their Current Antidiabetic Treatment: The GetGoal-O Randomized Trial. Diabetes Care 2017, 40, 485–493. [Google Scholar] [CrossRef]
- Philis-Tsimikas, A.; Wysham, C.H.; Hardy, E.; Han, J.; Iqbal, N. Efficacy and tolerability of exenatide once weekly over 7 years in patients with type 2 diabetes: An open-label extension of the DURATION-1 study. J. Diabetes Its Complicat. 2019, 33, 223–230. [Google Scholar] [CrossRef]
- Wysham, C.; Bergenstal, R.; Malloy, J.; Yan, P.; Walsh, B.; Malone, J.; Taylor, K. DURATION-2: Efficacy and safety of switching from maximum daily sitagliptin or pioglitazone to once-weekly exenatide. Diabet. Med. 2011, 28, 705–714. [Google Scholar] [CrossRef]
- Blevins, T.; Pullman, J.; Malloy, J.; Yan, P.; Taylor, K.; Schulteis, C.; Trautmann, M.; Porter, L. DURATION-5: Exenatide Once Weekly Resulted in Greater Improvements in Glycemic Control Compared with Exenatide Twice Daily in Patients with Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2011, 96, 1301–1310. [Google Scholar] [CrossRef]
- Buse, J.B.; Nauck, M.; Forst, T.; Sheu, W.H.-H.; Shenouda, S.K.; Heilmann, C.R.; Hoogwerf, B.J.; Gao, A.; Boardman, M.K.; Fineman, M.; et al. Exenatide once weekly versus liraglutide once daily in patients with type 2 diabetes (DURATION-6): A randomised, open-label study. Lancet 2013, 381, 117–124. [Google Scholar] [CrossRef]
- Guja, C.; Frías, J.P.; Somogyi, A.; Jabbour, S.; Wang, H.; Hardy, E.; Rosenstock, J. Effect of exenatide QW or placebo, both added to titrated insulin glargine, in uncontrolled type 2 diabetes: The DURATION-7 randomized study. Diabetes Obes. Metab. 2018, 20, 1602–1614. [Google Scholar] [CrossRef]
- Wysham, C.H.; Rosenstock, J.; Vetter, M.L.; Wang, H.; Hardy, E.; Iqbal, N. Further improvement in glycemic control after switching from exenatide two times per day to exenatide once-weekly autoinjected suspension in patients with type 2 diabetes: 52-week results from the DURATION-NEO-1 study. BMJ Open Diabetes Res. Care 2020, 8, e000773. [Google Scholar] [CrossRef]
- Gadde, K.M.; Vetter, M.L.; Iqbal, N.; Hardy, E.; Öhman, P.; DURATION-NEO-2 Study Investigators. Efficacy and safety of autoinjected exenatide once-weekly suspension versus sitagliptin or placebo with metformin in patients with type 2 diabetes: The DURATION-NEO-2 randomized clinical study. Diabetes Obes. Metab. 2017, 19, 979–988. [Google Scholar] [CrossRef]
- Reed, J.; Bain, S.; Kanamarlapudi, V. A Review of Current Trends with Type 2 Diabetes Epidemiology, Aetiology, Pathogenesis, Treatments and Future Perspectives. Diabetes Metab. Syndr. Obes. Targets Ther. 2021, 14, 3567–3602. [Google Scholar] [CrossRef]
- Tilinca, M.C.; Tiuca, R.A.; Niculas, C.; Varga, A.; Tilea, I. Future perspectives in diabesity treatment: Semaglutide, a glucagon-like peptide 1 receptor agonist (Review). Exp. Ther. Med. 2021, 22, 1167. [Google Scholar] [CrossRef]
- Diamant, M.; Van Gaal, L.; Stranks, S.; Guerci, B.; MacConell, L.; Haber, H.; Scism-Bacon, J.; Trautmann, M. Safety and Efficacy of Once-Weekly Exenatide Compared With Insulin Glargine Titrated to Target in Patients With Type 2 Diabetes Over 84 Weeks. Diabetes Care 2012, 35, 683–689. [Google Scholar] [CrossRef]
- Russell-Jones, D.; Cuddihy, R.M.; Hanefeld, M.; Kumar, A.; González, J.G.; Chan, M.; Wolka, A.M.; Boardman, M.K.; DURATION-4 Study Group. Efficacy and Safety of Exenatide Once Weekly Versus Metformin, Pioglitazone, and Sitagliptin Used as Monotherapy in Drug-Naive Patients With Type 2 Diabetes (DURATION-4): A 26-week double-blind study. Diabetes Care 2012, 35, 252–258. [Google Scholar] [CrossRef]
| Study | Dosage/Number of Patients/Duration | Main Outcomes, Weight Reduction | Side Effects | Ref. |
|---|---|---|---|---|
| LEAD-1 | liraglutide 0.6, 1.2, or 1.8 mg/day with sulfonylurea versus placebo N = 1041, 26 weeks | −0.2 kg (baseline 83.0 kg) for 1.8 mg and +0.3 kg (baseline 80.0 kg) for 1.2 mg/day, better glycemic control | Hypoglycemia, nausea, vomiting, diarrhea | [66] |
| LEAD-2 | liraglutide 0.6, 1.2, or 1.8 mg/day with metformin as a background treatment N = 1091, 26 weeks | Body weight decreased in all groups (1.8–2.8 kg), comparable glycemic control | Minor hypoglycemia, nausea | [67] |
| LEAD-3 | liraglutide 1.2 mg/day [N = 251] or 1.8 mg/day [N = 247], versus glimepiride, 52 weeks | A1c decreased by 0.51% with glimepiride versus 0.84% with liraglutide 1.2 mg and 1.14% with liraglutide 1.8 mg | Vomiting | [68] |
| LEAD-4 | liraglutide 1.2 or 1.8 mg/day versus placebo, N = 533, 26 weeks | Dose-dependent weight loss of 1–2 kg with 1.2 and 1.8 mg liraglutide versus weight gain of about 0.6 kg in placebo | Minor hypoglycemia, early gastrointestinal adverse events | [69] |
| LEAD-5 | liraglutide 1.8 mg/day [N = 232], placebo [N = 115], and open-label insulin glargine [N = 234], all in combination with metformin (1 g/twice per day) and glimepiride (4 mg/day), 26 weeks | Average reduction of 1.8 kg in the liraglutide group versus 0.42 kg in the placebo group and a 1.6 kg increase in the glargine group. | Nausea (14%) reported in the liraglutide group | [67] |
| LEAD-6 | liraglutide 1.8 mg/day [N = 233] or exenatide 10 μg twice/day [N = 231] open-label, parallel-group, multinational study (15 countries), 26 weeks | Average weight loss 3.24 kg (liraglutide group) and 2.87 kg (exenatide group) | Minor hypoglycaemia, less frequent in the liraglutide group compared to the exenatide one | [70] |
| Study | Dosage/Number of Patients/Duration | Main Outcomes, Weight Reduction | Side Effects | Ref. |
|---|---|---|---|---|
| STEP-1 | Semaglutide 2.4 mg/week versus placebo N = 1961, 68 weeks | 14.9% reduction of body weight versus 2.4% in placebo | Nausea and diarrhea | [102] |
| STEP-2 | Semaglutide 2.4 mg, or 1.0 mg once/week versus placebo N = 1210, 68 weeks | 9.64%, 6.99%, and 3.42% average body weight reduction with semaglutide 2.4 mg, 1.0 mg, and placebo, respectively. | Mild to moderate gastrointestinal adverse events more frequent with semaglutide 2.4 mg than with placebo | [103,115] |
| STEP 3 | Semaglutide 2.4 mg once/week N = 611, 68 weeks | 16.0% average weight reduction versus 5.7% with placebo. The co-primary endpoint of at least a 5% reduction in body weight was met by 86.6% versus 47.6%. | Gastrointestinal adverse events more frequent with semaglutide vs. placebo | [104] |
| STEP-4 | Semaglutide 2.4 mg/week for the first 20 weeks, followed by random semaglutide or placebo for the remaining 48 weeks. N = 902, 68 weeks | A total weight loss of 5.0% | Gastrointestinal events in 49.1% of participants in semaglutide group vs. 26.1% in placebo | [43] |
| STEP-5 | Semaglutide 2.4 mg versus placebo N = 304, 104 weeks | Decreasing weight until week 60, maintained through week 104; average placebo-corrected weight loss of 12.6 % | Gastrointestinal disorders, nausea, diarrhea, vomiting, and constipation more frequent in semaglutide group | [105] |
| STEP-6 | Semaglutide 2.4 or 1.7 mg/week versus placebo in Asian people N = 401, 20 weeks | Body weight reduction 13.2%, 9.6%and 2.1%, respectively, for 2.4 mg, 1.7 mg, and placebo | Mild to moderate gastrointestinal disorders predominantly in semaglutide 2.4 group | [106,108] |
| STEP-7 | Semaglutide 2.4 mg or placebo N = 375, 44 weeks | Not yet published | Not yet published | |
| STEP-8 | Semaglutide 2.4 mg vs. liraglutide1.8 mg/day. N = 338, 68 weeks | Significantly greater bodyweight reduction; 15.8% with semaglutide, compared to 6.4% with liraglutide | Gastrointestinal adverse events in 84.1% participants in semaglutide group and 82.7% in liraglutide | [107] |
| Study | Dosage/Number of Patients/Duration | Main Outcomes, Weight Reduction | Side Effects | Ref. |
|---|---|---|---|---|
| SURPASS-1 | Tirzepatide 5, 10, and 15 mg/week. N = 705, 40 weeks | Dose-dependent bodyweight loss ranging from 7 to 9.5 kg | Transient gastrointestinal events, nausea, diarrhea, vomiting | [117] |
| SURPASS-2 | Tirzepatide weekly doses (5, 10, and 15 mg) versus weekly injections of semaglutide 1.0 mg. N = 1879, 40 weeks | Greater reduction in body weight with tirzepatide than with semaglutide (−1.9 kg, −3.6 kg, and −5.5 kg, respectively) | Gastrointestinal events mild to moderate in both tirzepatide and semaglutide groups (nausea, diarrhea, vomiting) | [103] |
| SURPASS-3 | Tirzepatide weekly (5, 10, and 15 mg) with daily insulin degludec in people with poorly controlled blood glucose despite stable treatment with metformin, with or without SGLT2 N = 1444, 52 weeks | Average loss of 7.5, 10.7, and 12.9 kg versus average weight gain of 2.3 kg in the degludec group | Mild to moderate gastrointestinal events | [119] |
| SURPASS-4 | Tirzepatide 5 mg, 10 mg, or 15 mg/week or glargine 100 U/mL to reach fasting blood glucose < 100 mg/dLs N = 3045, 48 weeks | 74–88% of people taking tirzepatide achieved HbA1c below 7.0% without weight gain or severe hypoglycemia, versus13% in glargine group | Nausea, diarrhea, decreased appetite, and vomiting more frequent with tirzepatide than with glargine | [120] |
| SURPASS-5 | Tirzepatide 5, 10, or 15 mg/week in people taking insulin glargine for T2DM, with or without metformin. N = 475, 40 weeks | Average reduced body weight by 6.2, 8.2, and 10.9 kg, respectively | Gastrointestinal events, decreased appetite in 7–14% participants in tirzepatide group compared to 1.7% in placebo group, potentially contributing to weight loss | [121] |
| SURPASS-J-mono | Tirzepatide 5, 10, or 15 mg/week versus dulaglutide 0.75 mg/week in Japanese people with type 2 diabetes taking no other glucose-lowering medications during the study. N = 821, 96 weeks | Dose-dependent reduction of body weight in tirzepatide group (5.8 kg, 8.5 kg, and 10.7 kg, respectively), versus 0.5 kg reduction in dulaglutide group | Gastrointestinal events | [122] |
| SURPASS J-combo | Tirzepatide (5, 10, or 15 mg/week) in addition to non-incretin-based antidiabetic medications N = 484, 52 weeks | Dose-dependent reduction in body weight with tirzepatide compared with dulaglutide | Nausea, constipation, and nasopharyngitis | [123] |
| SURMOUNT-1 | Tirzepatide 5, 10, or 15 mg/week in obese people without diabetes N = 2539, 72 weeks | 15.0%, 19.5%, and 20.9%, respectively, body weight reduction in tirzepatide group, compared with just 3.1% in placebo | common gastrointestinal adverse events with tirzepatide | [124] |
| Study | Dosage/Number of Patients/Duration | Main Outcomes, Weight Reduction | Side Effects | Ref. |
|---|---|---|---|---|
| Get-Goal Mono | Lixisenatide 20 μg/day in medication-naïve people N = 361, 12 weeks | 2 kg reduction regardless of treatment allocation. | Nausea | [130] |
| Get-Goal-M | Lixisenatide 20 μg once daily, as add-on therapy in patients with T2DM insufficiently controlled with metformin alone. N = 680, 24 weeks | Mean body weight decreased to a similar extent in all groups. | Nausea and vomiting more frequently in Lixisenatide group | [131] |
| Get-Goal-X | Lixisenatide 20 µg daily versus exenatide 10µg twice daily in T2DM inadequately controlled with metformin N = 1243, 24 weeks | 25.1% of Lixisenatide patients and 31.4% of exenatide patients had ≥5% weight loss from baseline to week 24 | Gastrointestinal symptoms, treatment discontinuation for 6.3% in the Lixisenatide group and 7.6% in exenatide group | [132] |
| Get Goal F1 | (1) Lixisenatide one-step dose increase (10 μg once daily for two weeks, then 20 μg once daily; N= 161); (2) Lixisenatide two-step dose increase (10 μg once daily for one week, 15 μg once daily for one week, then 20 μg once daily; N = 161); (3) matching placebo one-step dose increase (N = 82); (4) matching placebo two-step dose increase (N = 80). N = 484, 24 weeks | Weight reduction between 2 kg and –2.7 kg in Lixisenatide group vs. 1.6 kg in placebo | Nausea and vomiting reported most frequently | [133] |
| Get Goal -S | Lixisenatide 20µg/day versus placebo inT2DM patients inadequately controlled with sulfonylurea ± metformin N = 1438, 24 weeks | ≥5% weight loss from baseline to week 24 for 14.4% in Lixisenatide patients and 7.2% in placebo patients. Significant reduction in HbA1c at week 24 versus placebo in Lixisenatide group | Nausea in Lixisenatide group, mainly in the first month of treatment | [134] |
| Get-Goal-P | Prandial Lixisenatide 20 µg/day versus placebo in T2DM patients insufficiently controlled by pioglitazone ± metformin. N = 484, 24 weeks | Average 0.2 kg reduction in body weight versus 0.2 kg increase in placebo group | Gastrointestinal disorders in Lixisenatide group | [135] |
| Get-Goal-L | Adding Lixisenatide (20µg/day) to established basal insulin therapy alone or together with metformin in people with T2DM and elevated glycated hemoglobin (HbA1c). N = 495, 24 weeks | Body weight decreased by 1.8 kg with Lixisenatide and 0.5 kg with placebo between randomization and week 24 | Hypoglycemia and nausea were increased compared with placebo, but no excess of serious adverse events | [136] |
| Get-Goal Duo 1 | Lixisenatide (20 µg/day) in patients with HbA1c still elevated after initiation of insulin glargine | Statistically significant bodyweight increase by an average of 0.3 and 1.2 kg in Lixisenatide and placebo groups, respectively | Increase in the frequency of gastrointestinal side effects and modestly increased rates of hypoglycemia | [137] |
| Study | Dosage/Patients/Duration | Main Outcomes, Weight Reduction | Side Effects | Ref. |
|---|---|---|---|---|
| Duration-1 | Exenatide 2 mg/week, against the pre-existing 10 µg/twice per day version N = 295, 30 weeks | No increased risk of hypoglycaemia and similar reductions in body weight | Nausea reported in both treatments, but more often for 10 µg/twice per day formulation | [136] |
| Duration-2 | Exenatide (2 mg once/week) versus maximum approved doses sitagliptin, thiazolidinedione, or pioglitazone, in patients treated with metformin | Average 2.3 kg weight loss in exenatide group, 0.8 kg reduction in sitagliptin group, and 2.8 kg weight gain with pioglitazone | Nausea and diarrhea in exenatide and sitagliptin groups | [137] |
| Duration-3 | Exenatide (2 mg once/week) versus insulin glargine titrated to glucose targets N = 456, 84 weeks | Average 2.6 kg decrease in bodyweight with exenatide, compared with a 1.4 kg increase with glargine, accompanied by improved glycemic control | No evidence | [145] |
| Duration-4 | Exenatide once weekly (EQW) compared with metformin, pioglitazone, and sitagliptin (SITA) N = 820, 26 weeks | 2.0 kg decrease with exenatide versus 0.8 kg reduction with sitagliptin and 1.5 kg increase with pioglitazone | Exenatide once weekly induced nausea and diarrhea | [146] |
| Duration-5 | Exenatide (2 mg once/week)) versus exenatide twice daily (5 µg during 4 weeks followed by 10 µg during 20 weeks) in order to improve glycemic control, body weight, and safety. N = 252, 24 weeks | Similar reductions in mean body weight from baseline to wk 24 observed in both groups (−2.3 ± 0.4 kg and −1.4 ± 0.4 kg) | In both groups, the majority of nausea was transient and mild to moderate in intensity, while the incidence decreased over time | [138] |
| Duration-6 | Exenatide once weekly (2 mg) versus liraglutide (1.8 mg) once daily in patients with T2DM. N = 911, 26 weeks | Better body weight reductions in liraglutide group (average 2.68–3.57 kg) | Nausea predominantly in exenatide group; diarrhea and vomiting more frequently in the liraglutide group and with decreasing incidence over time in both groups | [144] |
| Duration-7 | Exenatide 2 mg once weekly or placebo in patients with T2DM inadequately controlled despite titrated insulin glargine ± metformin. N = 461, 28 weeks | Body weight reduction average of 1.5 kg with exenatide versus placebo. | Gastrointestinal and injection-site adverse events more frequent with exenatide + IG than with placebo + IG | [140] |
| Duration Neo-1 | Exenatide 2 mg once/week, self-injectable Miglyol suspension (QWS-AI) versus exenatide 10 µg twice daily (BID), N = 375, 28 weeks | Significant body weight was reduced in both groups | Gastrointestinal adverse events were reported in 22.7% of patients within exenatide QWS-AI group and 35.6% in exenatide BID group | [141,142] |
| Duration-Neo-2 | Exenatide 2 mg once-weekly Miglyol suspension for autoinjection (QWS-AI) versus sitagliptin (100 mg once/day oraly) or placebo. N = 364, 28 weeks | Average 1.12 kg and, respectively, 1.19 kg decrease of bodyweight in exenatide and sitagliptin groups versus 0.15 kg increase in the placebo | Gastrointestinal events and injection-site reactions | [142] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Popoviciu, M.-S.; Păduraru, L.; Yahya, G.; Metwally, K.; Cavalu, S. Emerging Role of GLP-1 Agonists in Obesity: A Comprehensive Review of Randomised Controlled Trials. Int. J. Mol. Sci. 2023, 24, 10449. https://doi.org/10.3390/ijms241310449
Popoviciu M-S, Păduraru L, Yahya G, Metwally K, Cavalu S. Emerging Role of GLP-1 Agonists in Obesity: A Comprehensive Review of Randomised Controlled Trials. International Journal of Molecular Sciences. 2023; 24(13):10449. https://doi.org/10.3390/ijms241310449
Chicago/Turabian StylePopoviciu, Mihaela-Simona, Lorena Păduraru, Galal Yahya, Kamel Metwally, and Simona Cavalu. 2023. "Emerging Role of GLP-1 Agonists in Obesity: A Comprehensive Review of Randomised Controlled Trials" International Journal of Molecular Sciences 24, no. 13: 10449. https://doi.org/10.3390/ijms241310449
APA StylePopoviciu, M.-S., Păduraru, L., Yahya, G., Metwally, K., & Cavalu, S. (2023). Emerging Role of GLP-1 Agonists in Obesity: A Comprehensive Review of Randomised Controlled Trials. International Journal of Molecular Sciences, 24(13), 10449. https://doi.org/10.3390/ijms241310449







