The Endless World of Carotenoids—Structural, Chemical and Biological Aspects of Some Rare Carotenoids
Abstract
1. Introduction
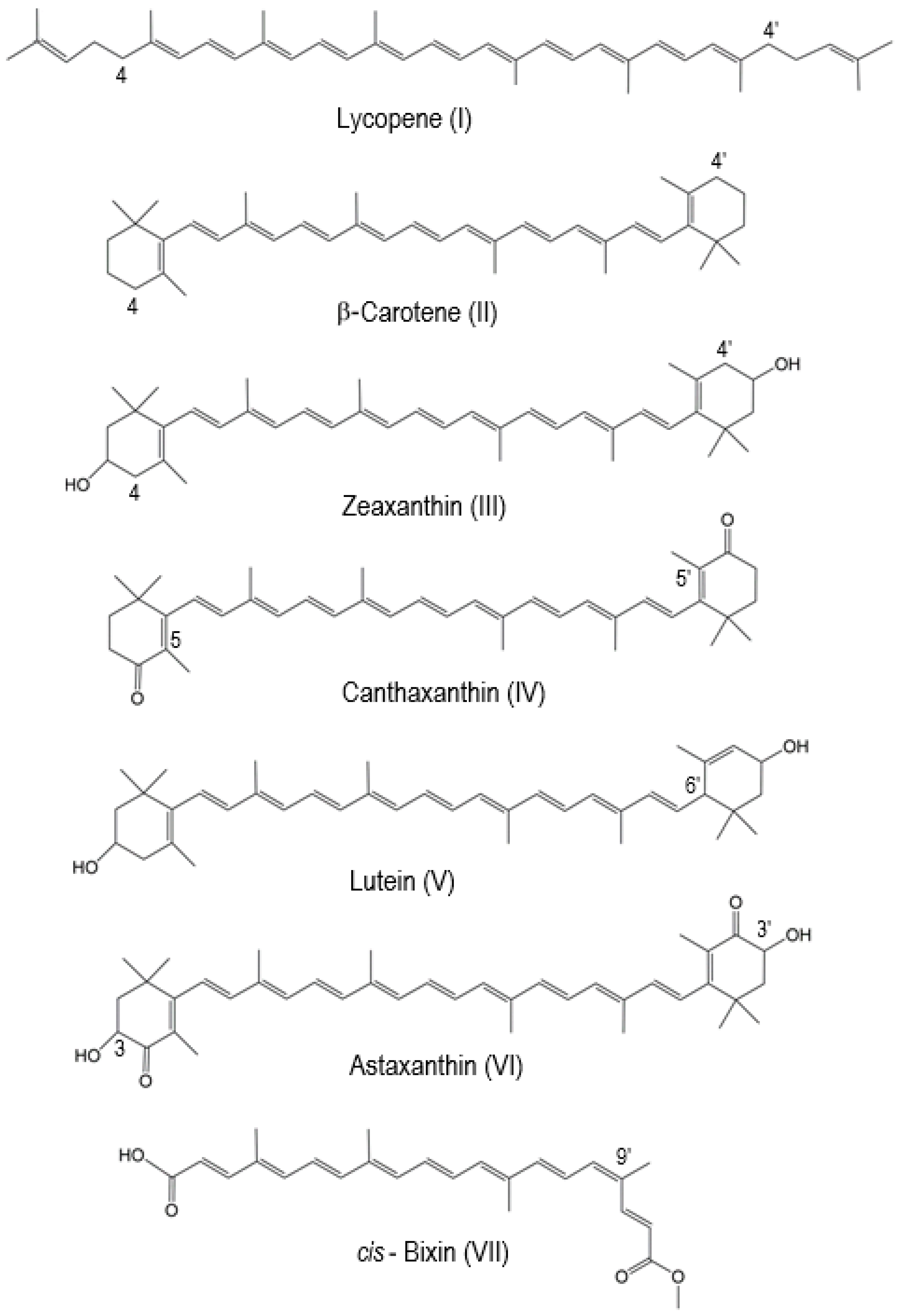
2. Studies of Electron and Proton Loss of Conventional Carotenoids by DFT and EPR
| Carotenoid | The Most Favorable Proton Loss from the Radical Cation | Reference |
|---|---|---|
| Lycopene | C4(4′) | [14] |
| β-carotene | C4(4′) | [15] |
| Zeaxanthin | C4(4′) | [15,16] |
| Canthaxanthin | C5(5′) | [15] |
| Lutein | C6′ | [15] |
| Astaxanthin | C3(C3′) | [17] |
| cis-Bixin | C9′ | [20] |
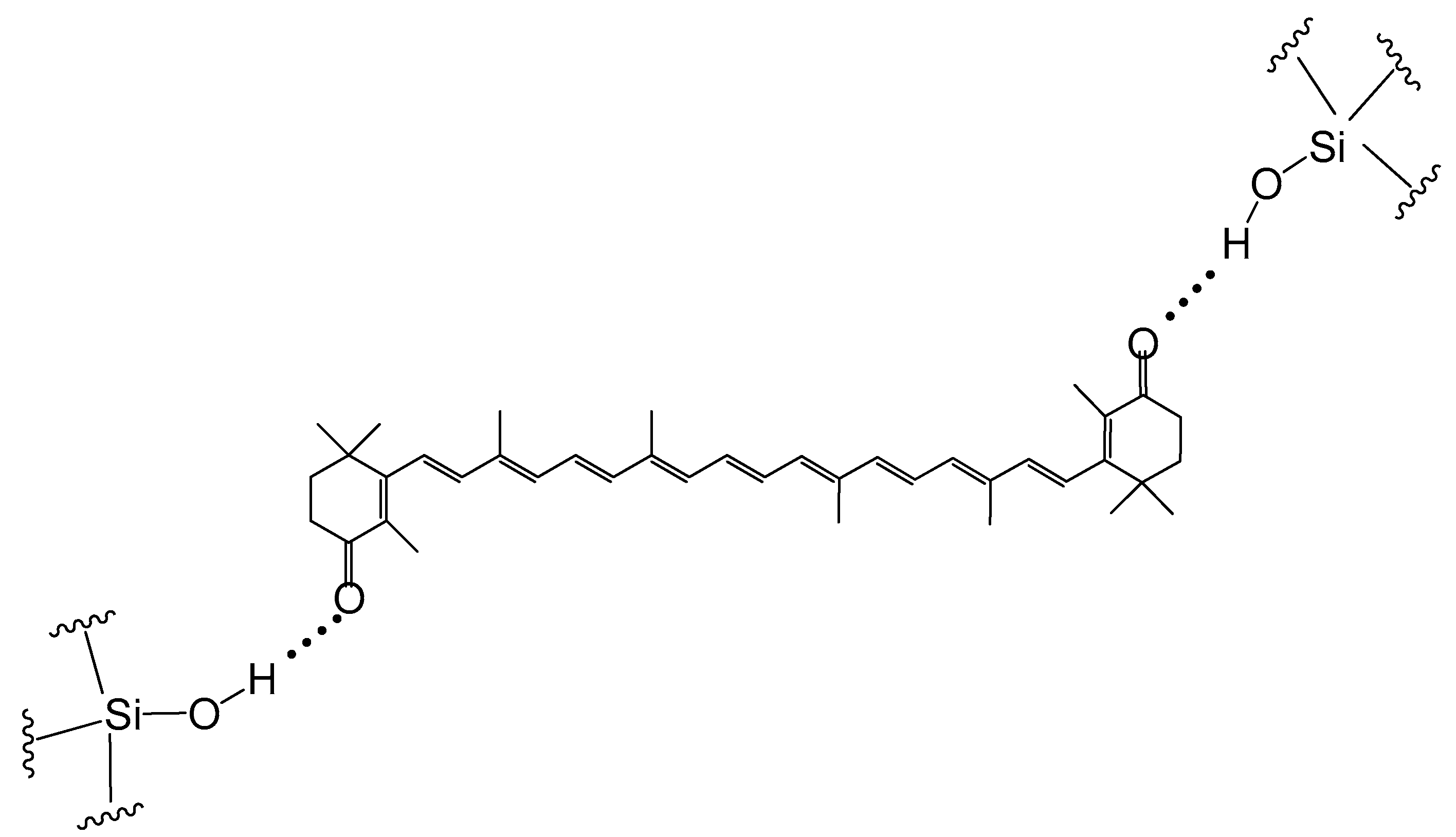
3. Improvement of Carotenoid Features by Nonpolar Environment and Absence of Hydrogen Bonding—Rare Carotenoids Discussion

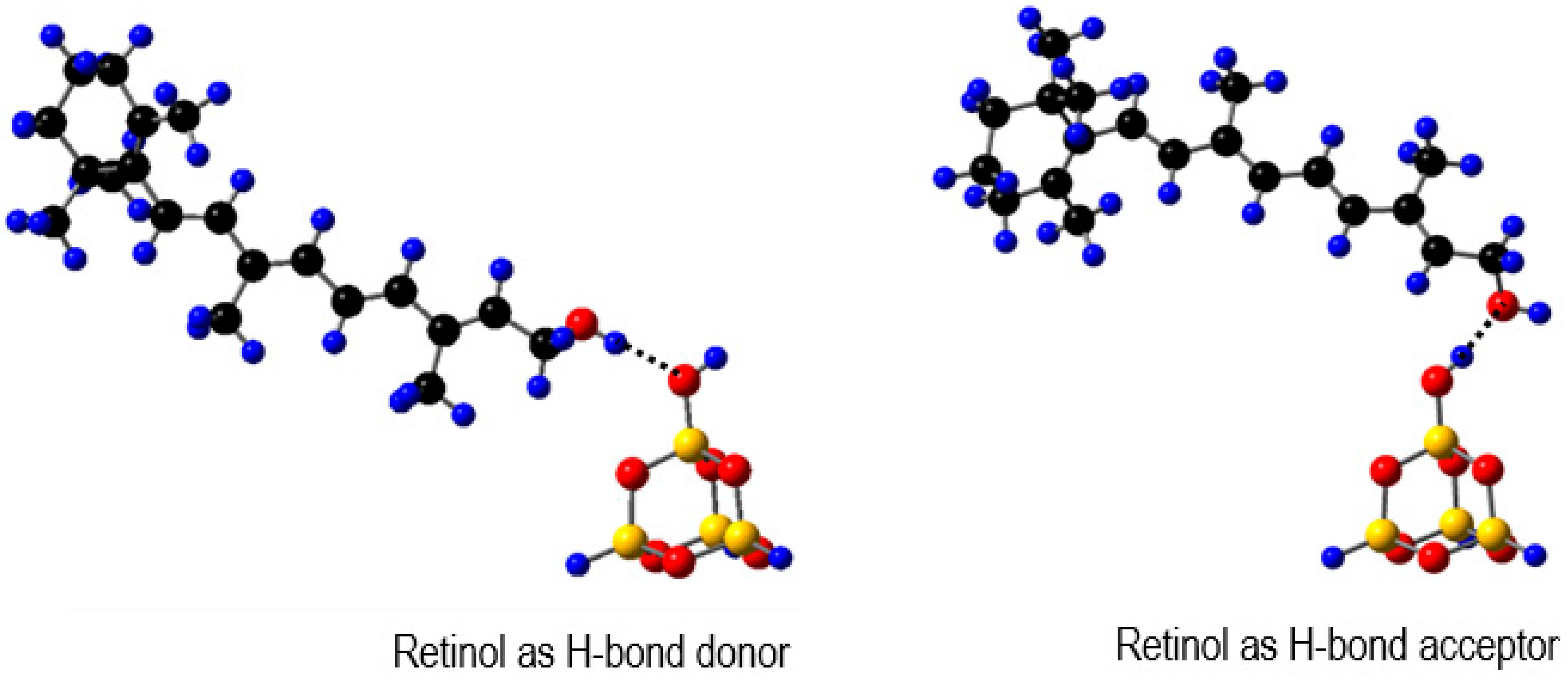
4. Improvement of Carotenoid Features by Supramolecular Delivery Systems
4.1. Biological Activities of Rare Carotenoids
4.2. Supramolecular Delivery Systems of Carotenoids
4.3. Several Examples of Using Supramolecular Delivery Systems for Rare Carotenoids
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yabuzaki, J. Carotenoids database: Structures, chemical fingerprints and distribution among organisms. Database J. Biol. Databases Curation 2017, 2017, bax004. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.Y.; Fang, J.C.; Gao, Z.H.; Zhang, C.; Xie, S.Y. Higher intake of fruits, vegetables or their fiber reduces the risk of type 2 diabetes: A meta-analysis. J. Diabetes Investig. 2016, 7, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.T.; Keum, N.; Norat, T.; Greenwood, D.C.; Riboli, E.; Vatten, L.J.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-A systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 46, 1029–1056. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Rengasamy, K.R.; Mahomoodally, F.M.; Keum, Y.-S. Protective effects of lycopene in cancer, cardiovascular, and neurodegenerative diseases: An update on epidemiological and mechanistic perspectives. Pharmacol. Res. 2020, 155, 104730. [Google Scholar] [CrossRef] [PubMed]
- Eggersdorfer, M.; Wyss, A. Carotenoids in human nutrition and health. Arch. Biochem. Biophys. 2018, 652, 18–26. [Google Scholar] [CrossRef]
- Castenmiller, J.J.M.; West, C.E. Bioavailability and bioconversion of carotenoids. Annu. Rev. Nutr. 1998, 18, 19–38. [Google Scholar] [CrossRef] [PubMed]
- dela Seña, C.; Riedl, K.M.; Narayanasamy, S.; Curley, R.W., Jr.; Schwartz, S.J.; Harrison, E.H. The human enzyme that converts dietary provitamin A carotenoids to vitamin A is a dioxygenase. J. Biol. Chem. 2014, 289, 13661–13666. [Google Scholar] [CrossRef]
- Collini, E. Carotenoids in photosynthesis: The revenge of the “accessory” pigments. Chem 2019, 5, 494–495. [Google Scholar] [CrossRef]
- Hashimoto, H.; Uragami, C.; Cogdell, R.J. Carotenoids and photosynthesis. In Carotenoids in Nature; Stange, C., Ed.; Subcellular Biochemistry; Springer: Cham, Switzerland, 2016; Volume 79. [Google Scholar]
- Simkin, A.J.; Kapoor, L.; Doss, C.G.P.; Hofmann, T.A.; Lawson, T.; Ramamoorthy, S. The role of photosynthesis related pigments in light harvesting, photoprotection and enhancement of photosynthetic yield in planta. Photosynth. Res. 2022, 152, 23–42. [Google Scholar] [CrossRef]
- Focsan, A.L.; Polyakov, N.E.; Kispert, L.D. Carotenoids: Importance in daily life—Insight gained from EPR and ENDOR. Appl. Magn. Reson. 2021, 52, 1093–1112. [Google Scholar] [CrossRef]
- Konovalova, T.A.; Krzystek, J.; Bratt, P.J.; van Tol, J.; Brunel, L.-C.; Kispert, L.D. 95–670 GHz EPR studies of canthaxanthin radical cation stabilized on a silica−Alumina surface. J. Phys. Chem. B 1999, 103, 5782–5786. [Google Scholar] [CrossRef]
- Focsan, A.L.; Kispert, L.D. Radicals formed from proton loss of carotenoid radical cations: A special form of carotenoid neutral radical occurring in photoprotection. J. Photochem. Photobiol. B Biol. 2017, 166, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Focsan, A.L.; Bowman, M.K.; Molnár, P.; Deli, J.; Kispert, L.D. Carotenoid radical formation: Dependence on conjugation length. J. Phys. Chem. B 2011, 115, 9495–9506. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, J.; Focsan, A.L.; Konovalova, T.A.; Molnar, P.; Deli, J.; Bowman, M.K.; Kispert, L.D. Pulsed electron nuclear double resonance studies of carotenoid oxidation in Cu(II)-substituted MCM-41 molecular sieves. J. Phys. Chem. B 2008, 112, 5449–5457. [Google Scholar] [CrossRef]
- Focsan, A.L.; Bowman, M.K.; Konovalova, T.A.; Molnár, P.; Deli, J.; Dixon, D.A.; Kispert, L.D. Pulsed EPR and DFT characterization of radicals produced by photo-oxidation of zeaxanthin and violaxanthin on silica−alumina. J. Phys. Chem. B 2008, 112, 1806–1819. [Google Scholar] [CrossRef]
- Focsan, A.L.; Bowman, M.K.; Shamshina, J.; Krzyaniak, M.D.; Magyar, A.; Polyakov, N.E.; Kispert, L.D. EPR study of the astaxanthin n-octanoic acid monoester and diester radicals on silica–alumina. J. Phys. Chem. B 2012, 116, 13200–13210. [Google Scholar] [CrossRef]
- Rivera-Madrid, R.; Aguilar-Espinosa, M.; Cardenas-Conejo, Y.; Garza-Caligaris, L.E. Carotenoid derivates in achiote (Bixa orellana) seeds: Synthesis and health promoting properties. Front. Plant Sci. 2016, 7, 1406. [Google Scholar] [CrossRef]
- Hernandez-Marin, E.; Galano, A.; Martínez, A. Cis carotenoids: Colorful molecules and free radical quenchers. J. Phys. Chem. B 2013, 117, 4050–4061. [Google Scholar] [CrossRef]
- Tay-Agbozo, S.S.; Krzyaniak, M.D.; Bowman, M.K.; Street, S.; Kispert, L.D. DFT and ENDOR study of bixin radical cations and neutral radicals on silica–alumina. J. Phys. Chem. B 2015, 119, 7170–7179. [Google Scholar] [CrossRef]
- Focsan, A.L.; Molnár, P.; Deli, J.; Kispert, L. Structure and properties of 9’-cis neoxanthin carotenoid radicals by electron paramagnetic resonance measurements and density functional theory calculations: Present in LHC II? J. Phys. Chem. 2009, 113, 6087–6096. [Google Scholar] [CrossRef]
- Gao, Y.; Xu, D.; Kispert, L.D. Hydrogen bond formation between the carotenoid canthaxanthin and the silanol group on MCM-41 surface. J. Phys. Chem. B 2015, 119, 10488–10495. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Chen, H.; Tay-Agbozo, S.; Kispert, L.D. Photo-induced electron transfer of carotenoids in mesoporous sieves (MCM-41) and surface modified MCM-41: The role of hydrogen bonds on the electron transfer. J. Photochem. Photobiol. A Chem. 2017, 341, 1–11. [Google Scholar] [CrossRef]
- Méndez-Hernández, D.D.; Tarakeshwar, P.; Gust, D.; Moore, T.A.; Moore, A.L.; Mujica, V. Simple and accurate correlation of experimental redox potentials and DFT-calculated HOMO/LUMO energies of polycyclic aromatic hydrocarbons. J. Mol. Model. 2013, 19, 2845–2848. [Google Scholar] [CrossRef]
- Gao, Y.; Focsan, A.L.; Kispert, L.D. Antioxidant activity in supramolecular carotenoid complexes favored by nonpolar environment and disfavored by hydrogen bonding. Antioxidants 2020, 9, 625. [Google Scholar] [CrossRef]
- Polyakov, N.; Leshina, T.; Salakhutdinov, N.; Konovalova, T.; Kispert, L. Antioxidant and redox properties of supramolecular complexes of carotenoids with β-glycyrrhizic acid. Free. Radic. Biol. Med. 2006, 40, 1804–1809. [Google Scholar] [CrossRef] [PubMed]
- Polyakov, N.E.; Magyar, A.; Kispert, L.D. Photochemical and optical properties of water-soluble xanthophyll antioxidants: Aggregation vs complexation. J. Phys. Chem. B 2013, 117, 10173–10182. [Google Scholar] [CrossRef]
- Focsan, A.L.; Polyakov, N.E.; Kispert, L.D. Photo protection of Haematococcus pluvialis algae by astaxanthin: Unique properties of astaxanthin deduced by EPR, optical and electrochemical studies. Antioxidants 2017, 6, 80. [Google Scholar] [CrossRef] [PubMed]
- Magyar, A.; Bowman, M.K.; Molnár, P.; Kispert, L. Neutral carotenoid radicals in photoprotection of wild-type Arabidopsis thaliana. J. Phys. Chem. B 2013, 117, 2239–2246. [Google Scholar] [CrossRef]
- Gao, Y.; Webb, S.; Kispert, L.D. Deprotonation of carotenoid radical cation and formation of a didehydrodimer. J. Phys. Chem. B 2003, 107, 13237–13240. [Google Scholar] [CrossRef]
- Gao, Y.; Focsan, A.L.; Kispert, L.D. The effect of polarity of environment on the antioxidant activity of carotenoids. Chem. Phys. Lett. 2020, 761, 138098. [Google Scholar] [CrossRef]
- Closs, G.L.; Miller, J.R. Intramolecular long-distance electron transfer in organic molecules. Science 1988, 240, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Guarr, T.; McLendon, G. Quantum mechanical effects in inorganic and bioinorganic electron transfer. Coord. Chem. Rev. 1985, 68, 1–52. [Google Scholar] [CrossRef]
- Hush, N. Distance dependence of electron transfer rates. Coord. Chem. Rev. 1985, 64, 135–157. [Google Scholar] [CrossRef]
- Marcus, R.; Sutin, N. Electron transfers in chemistry and biology. Biochim. Biophys. Acta 1985, 811, 265–322. [Google Scholar] [CrossRef]
- Gao, Y.; Lockart, M.; Kispert, L.D.; Bowman, M.K. Photo-induced charge separation in hydroxycoumarins on TiO2 and F-TiO2. Dalton Trans. 2019, 48, 10881–10891. [Google Scholar] [CrossRef]
- Gao, Y.; Lockart, M.; Kispert, L.D.; Bowman, M.K. Photoinduced charge separation in retinoic acid on TiO2: Comparison of three anchoring modes. J. Phys. Chem. C 2019, 123, 24634–24642. [Google Scholar] [CrossRef]
- Gao, Y.; Konovalova, T.A.; Lawrence, J.N.; Smitha, M.A.; Nunley, J.; Schad, R.; Kispert, L.D. Interaction of carotenoids and Cu2+ in Cu-MCM-41: Distance-dependent reversible electron transfer. J. Phys. Chem. B 2003, 107, 2459–2465. [Google Scholar] [CrossRef]
- Gao, Y.; Kispert, L.D. A DFT study of the interaction between olefins and Cu2+ on silica and MCM-41 model surfaces. Dalton Trans. 2014, 43, 6221–6228. [Google Scholar] [CrossRef]
- Polyakov, N.E.; Leshina, T.V.; Meteleva, E.S.; Dushkin, A.V.; Konovalova, T.A.; Kispert, L.D. Water soluble complexes of carotenoids with arabinogalactan. J. Phys. Chem. B 2009, 113, 275–282. [Google Scholar] [CrossRef]
- Polyakov, N.E.; Leshina, T.V.; Salakhutdinov, N.F.; Kispert, L.D. Host-guest complexes of carotenoids with β-glycyrrhizic acid. J. Phys. Chem. B 2006, 110, 6991–6998. [Google Scholar] [CrossRef]
- Polyakov, N.E.; Leshina, T.V.; Konovalova, T.A.; Hand, E.O.; Kispert, L.D. Inclusion complexes of carotenoids with cyclodextrins: 1H NMR, EPR, and optical studies. Free radic. Biol. Med. 2004, 36, 872–880. [Google Scholar] [CrossRef]
- Apanasenko, I.E.; Selyutina, O.Y.; Polyakov, N.E.; Suntsova, L.P.; Meteleva, E.S.; Dushkin, A.V.; Vachali, P.; Bernstein, P.S. Solubilization and stabilization of macular carotenoids by water soluble oligosaccharides and polysaccharides. Arch. Biochem. Biophys. 2015, 572, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Genç, Y.; Bardakci, H.; Yücel, Ç.; Karatoprak, G.; Akkol, E.K.; Barak, T.H.; Sobarzo-Sánchez, E. Oxidative stress and marine carotenoids: Application by using nanoformulations. Mar. Drugs 2020, 18, 423. [Google Scholar] [CrossRef] [PubMed]
- Polyakov, N.E.; Kispert, L.D. Water soluble biocompatible vesicles based on polysaccharides and oligosaccharides inclusion complexes for carotenoid delivery. Carbohydr. Polym. 2015, 128, 207–219. [Google Scholar] [CrossRef] [PubMed]
- Focsan, A.L.; Polyakov, N.E.; Kispert, L.D. Supramolecular carotenoid complexes of enhanced solubility and stability—The way of bioavailability improvement. Molecules 2019, 24, 3947. [Google Scholar] [CrossRef] [PubMed]
- Soukoulis, C.; Bohn, T. A comprehensive overview on the micro- and nano-technological encapsulation advances for enhancing the chemical stability and bioavailability of carotenoids. Crit. Rev. Food Sci. Nutr. 2018, 58, 1–36. [Google Scholar] [CrossRef] [PubMed]
- Terao, J.; Minami, Y.; Bando, N. Singlet molecular oxygen-quenching activity of carotenoids: Relevance to protection of the skin from photoaging. J. Clin. Biochem. Nutr. 2011, 48, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Polyakov, N.E.; Leshina, T.V.; Konovalova, T.A.; Kispert, L.D. Carotenoids as scavengers of free radicals in a Fenton reaction: Antioxidants or pro-oxidants? Free. Radic. Biol. Med. 2001, 31, 398–404. [Google Scholar] [CrossRef]
- El-Agamey, A.; Lowe, G.M.; McGarvey, D.J.; Mortensen, A.; Phillip, D.M.; Truscott, T.; Young, A.J. Carotenoid radical chemistry and antioxidant/pro-oxidant properties. Arch. Biochem. Biophys. 2004, 430, 37–48. [Google Scholar] [CrossRef]
- Young, A.J.; Lowe, G.L. Carotenoids—Antioxidant properties. Antioxidants 2018, 7, 28. [Google Scholar] [CrossRef]
- Brotosudarmo, T.H.P.; Limantara, L.; Setiyono, E. Structures of astaxanthin and their consequences for therapeutic application. Int. J. Food Sci. 2020, 2020, 2156582. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, P.; Noda, K.; Manabe, Y.; Ohkubo, T.; Tanaka, Y.; Maoka, T.; Sugawara, T.; Hirata, T. Siphonaxanthin, a marine carotenoid from green algae, effectively induces apoptosis in human leukemia (HL-60) cells. Biochim. Biophys. Acta—Gen. Subj. 2011, 1810, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, T.; Ganesan, P.; Li, Z.; Manabe, Y.; Hirata, T. Siphonaxanthin, a green algal carotenoid, as a novel functional compound. Mar. Drugs 2014, 12, 3660–3668. [Google Scholar] [CrossRef] [PubMed]
- Manabe, Y.; Hirata, T.; Sugawara, T. Suppressive effects of carotenoids on the antigen-induced degranulation in RBL-2H3 rat basophilic leukemia cells. J. Oleo Sci. 2014, 63, 291–294. [Google Scholar] [CrossRef]
- Shindo, K.; Misawa, N. New and rare carotenoids isolated from marine bacteria and their antioxidant activities. Mar. Drugs 2014, 12, 1690–1698. [Google Scholar] [CrossRef]
- Shindo, K.; Kikuta, K.; Suzuki, A.; Katsuta, A.; Kasai, H.; Yasumoto-Hirose, M.; Matsuo, Y.; Misawa, N.; Takaichi, S. Rare carotenoids, (3R)-saproxanthin and (3R,2′S)-myxol, isolated from novel marine bacteria (Flavobacteriaceae) and their antioxidative activities. Appl. Microbiol. Biotechnol. 2007, 74, 1350–1357. [Google Scholar] [CrossRef]
- Hamidi, M.; Safarzadeh Kozani, P.; Pierre, G.; Michaud, P.; Delattre, C. Marine bacteria versus microalgae: Who is the best for biotechnological production of bioactive compounds with antioxidant properties and other biological applications? Mar. Drugs 2020, 18, 28. [Google Scholar] [CrossRef]
- Ji, H.-F. Insight into the strong antioxidant activity of deinoxanthin, a unique carotenoid in Deinococcus radiodurans. Int. J. Mol. Sci. 2010, 11, 4506–4510. [Google Scholar] [CrossRef]
- Choi, Y.-J.; Hur, J.-M.; Lim, S.; Jo, M.; Kim, D.H.; Choi, J.-I. Induction of apoptosis by deinoxanthin in human cancer cells. Anticancer. Res. 2014, 34, 1829–1835. [Google Scholar]
- Shin, J.; Song, M.-H.; Oh, J.-W.; Keum, Y.-S.; Saini, R.K. Pro-oxidant actions of carotenoids in triggering apoptosis of cancer cells: A review of emerging evidence. Antioxidants 2020, 9, 532. [Google Scholar] [CrossRef]
- Richter, T.K.S.; Hughes, C.C.; Moore, B.S. Sioxanthin, a novel glycosylated carotenoid, reveals an unusual subclustered biosynthetic pathway. Environ. Microbiol. 2015, 17, 2158–2171. [Google Scholar] [CrossRef] [PubMed]
- Singla, R.K.; Bhat, G.V. Crocin: An overview. Indo Glob. J. Pharm. Sci. 2011, 1, 281–286. [Google Scholar] [CrossRef]
- Chen, S.-S.; Gu, Y.; Lu, F.; Qian, D.-P.; Dong, T.-T.; Ding, Z.-H.; Zhao, S.; Yu, Z.-H. Antiangiogenic effect of crocin on breast cancer cell MDA-MB-231. J. Thorac. Dis. 2019, 11, 4464–4473. [Google Scholar] [CrossRef] [PubMed]
- Osawa, A.; Ishii, Y.; Sasamura, N.; Morita, M.; Kasai, H.; Maoka, T.; Shindo, K. Characterization and antioxidative activities of rare C-50 carotenoids-sarcinaxanthin, sarcinaxanthinmonoglucoside, and sarcinaxanthindiglucoside-obtained from Micrococcus yunnanensis. J. Oleo Sci. 2010, 59, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Berg, C.J.; Hsu, C.-C.; Merrill, B.A.; Tauber, M.J. Characterization of carotenoid aggregates by steady-state optical spectroscopy. J. Phys. Chem. B 2012, 116, 10617–10630. [Google Scholar] [CrossRef]
- Sujak, A.; Okulski, W.; Gruszecki, W.I. Organisation of xanthophyll pigments lutein and zeaxanthin in lipid membranes formed with dipalmitoylphosphatidylcholine. Biochim. Biophys. Acta—Biomembr. 2000, 1509, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, R.G.R.; Coutinho, A.J.; Pinheiro, M.; Neves, A.R. Nanoparticles for targeted brain drug delivery: What do we know? Int. J. Mol. Sci. 2021, 22, 11654. [Google Scholar] [CrossRef]
- Martínez-Álvarez, O.; Calvo, M.M.; Gómez-Estaca, J. Recent advances in astaxanthin micro/nanoencapsulation to improve its stability and functionality as a food ingredient. Mar. Drugs 2020, 18, 406. [Google Scholar] [CrossRef]
- Shibata, S. A Drug over the millennia: Pharmacognosy, chemistry, and pharmacology of licorice. Yakugaku Zasshi 2000, 120, 849–862. [Google Scholar] [CrossRef]
- Hasan, K.; Ara, I.; Alam Mondal, M.S.; Kabir, Y. Phytochemistry, pharmacological activity, and potential health benefits of Glycyrrhiza glabra. Heliyon 2021, 7, e07240. [Google Scholar] [CrossRef]
- Ageeva, A.A.; Kruppa, A.I.; Magin, I.M.; Babenko, S.V.; Leshina, T.V.; Polyakov, N.E. New aspects of the antioxidant activity of glycyrrhizin revealed by the CIDNP technique. Antioxidants 2022, 11, 1591. [Google Scholar] [CrossRef]
- Su, X.; Wu, L.; Hu, M.; Dong, W.; Xu, M.; Zhang, P. Glycyrrhizic acid: A promising carrier material for anticancer therapy. Biomed. Pharmacother. 2017, 95, 670–678. [Google Scholar] [CrossRef]
- Chrzanowski, J.; Chrzanowska, A.; Graboń, W. Glycyrrhizin: An old weapon against a novel coronavirus. Phytother. Res. 2021, 35, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Aqib, A.I.; Atta, K.; Muneer, A.; Arslan, M.; Shafeeq, M.; Rahim, K. Saponin and its derivatives (glycyrrhizin) and SARS-CoV-2. In Application of Natural Products in SARS-CoV-2; Niaz, K., Ed.; Academic Press: Cambridge, MA, USA, 2023; pp. 25–46. [Google Scholar]
- Selyutina, O.; Polyakov, N. Glycyrrhizic acid as a multifunctional drug carrier—From physicochemical properties to biomedical applications: A modern insight on the ancient drug. Int. J. Pharm. 2019, 559, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Selyutina, O.Y.; Shelepova, E.A.; Paramonova, E.D.; Kichigina, L.A.; Khalikov, S.S.; Polyakov, N.E. Glycyrrhizin-induced changes in phospholipid dynamics studied by 1H NMR and MD simulation. Arch. Biochem. Biophys. 2020, 686, 108368. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Shelepova, E.; Evseenko, V.; Dushkin, A.; Medvedev, N.; Polyakov, N. Mechanism of the enhancing effect of glycyrrhizin on nifedipine penetration through a lipid membrane. J. Mol. Liq. 2021, 344, 117759. [Google Scholar] [CrossRef] [PubMed]
- Dushkin, A.V.; Tolstikova, T.G.; Khvostov, M.V.; Tolstikov, G.A. Complexes of polysaccharides and glycyrrhizic acid with drug molecules-mechanochemical synthesis and pharmacological activity. In The Complex World of Polysaccharides; Karunaratne, D.N., Ed.; IntechOpen: Rijeka, Croatia, 2012; pp. 573–602. [Google Scholar]
- Stancanelli, R.; Løjkner, L.D.; Larsen, K.L.; Guardo, M.; Cannavà, C.; Tommasini, S.; Ventura, C.A.; Calabrò, M.L.; Micali, N.; Villari, V.; et al. Structural and spectroscopic features of lutein/butanoyl-β-cyclodextrin nanoassemblies. J. Pharm. Biomed. 2012, 71, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Kispert, L.D. Reaction of carotenoids and ferric chloride: Equilibria, isomerization, and products. J. Phys. Chem. B 2003, 107, 5333–5338. [Google Scholar] [CrossRef]
- Li, B.; Vachali, P.P.; Shen, Z.; Gorusupudi, A.; Nelson, K.; Besch, B.M.; Bartschi, A.; Longo, S.; Mattinson, T.; Shihab, S.; et al. Retinal accumulation of zeaxanthin, lutein, and β-carotene in mice deficient in carotenoid cleavage enzymes. Exp. Eye Res. 2017, 159, 123–131. [Google Scholar] [CrossRef]
- Yucheng, L.; Yalin, L.; Zhifeng, R. Mini review on photocatalysis of titanium dioxide nanoparticles and their solar applications. Nano Energy 2013, 2, 1031–1045. [Google Scholar]
- Yang, X.; Guo, S.; Wang, L.; Guan, S.; Zhou, S.; Lu, J. Function toggle of tumor microenvironment responsive nanoagent for highly efficient free radical stress enhanced chemodynamic therapy. Nano Res. 2022, 15, 8228–8236. [Google Scholar] [CrossRef]
- Esposito, E.; Drechsler, M.; Huang, N.; Pavoni, G.; Cortesi, R.; Santonocito, D.; Puglia, C. Ethosomes and organogels for cutaneous administration of crocin. Biomed. Microdevices 2016, 18, 108. [Google Scholar] [CrossRef] [PubMed]
- Muthiah, M.; Park, I.-K.; Cho, C.-S. Surface modification of iron oxide nanoparticles by biocompatible polymers for tissue imaging and targeting. Biotechnol. Adv. 2013, 31, 1224–1236. [Google Scholar] [CrossRef] [PubMed]
- El-Kharrag, R. Development of a Therapeutic Model of Early Liver Cancer Using Crocin-Coated Magnetite Nanoparticles. Ph.D. Dissertation, United Arab Emirates University, Al Ain, Abu Dhabi, 2015. [Google Scholar]
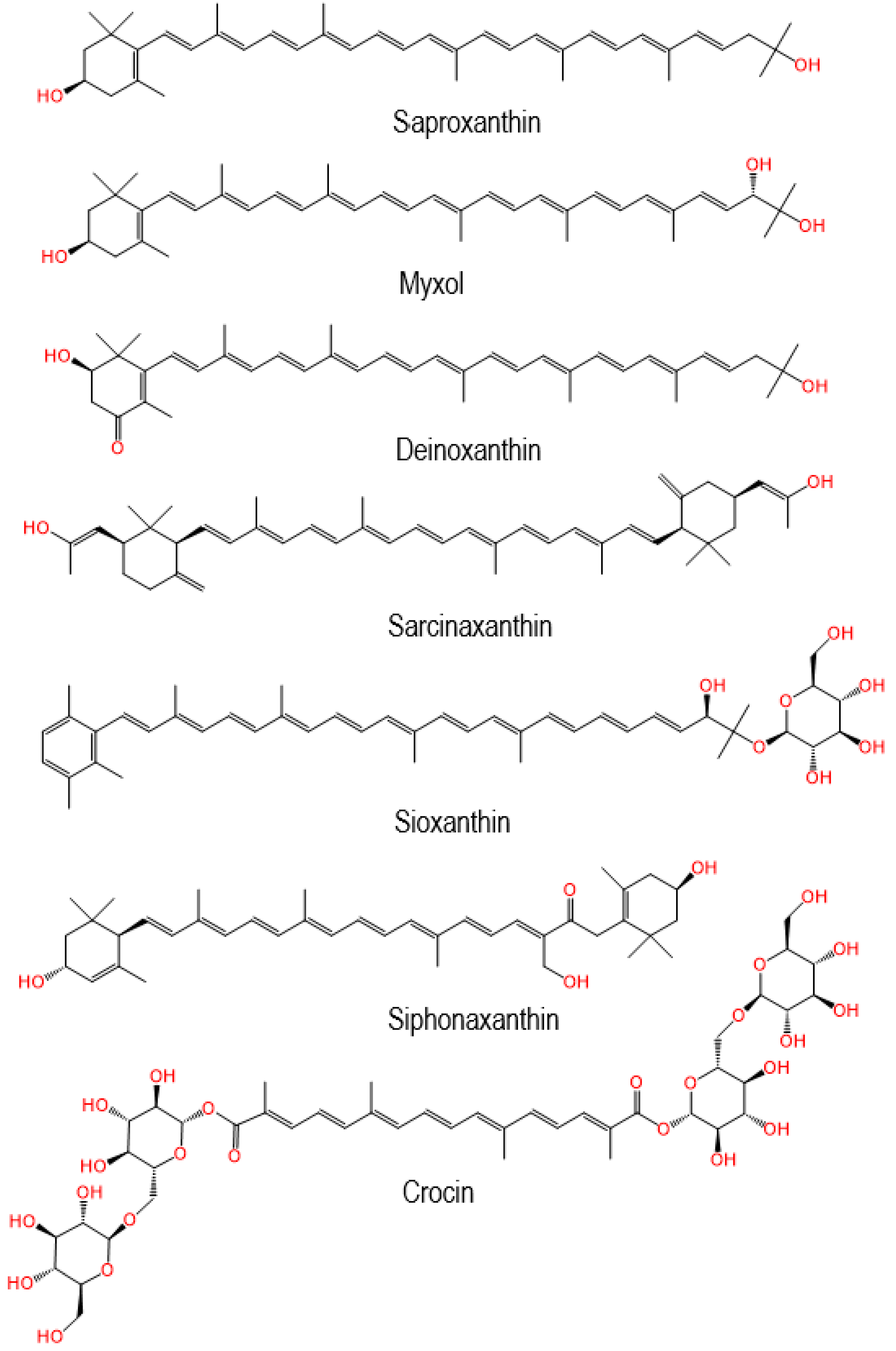

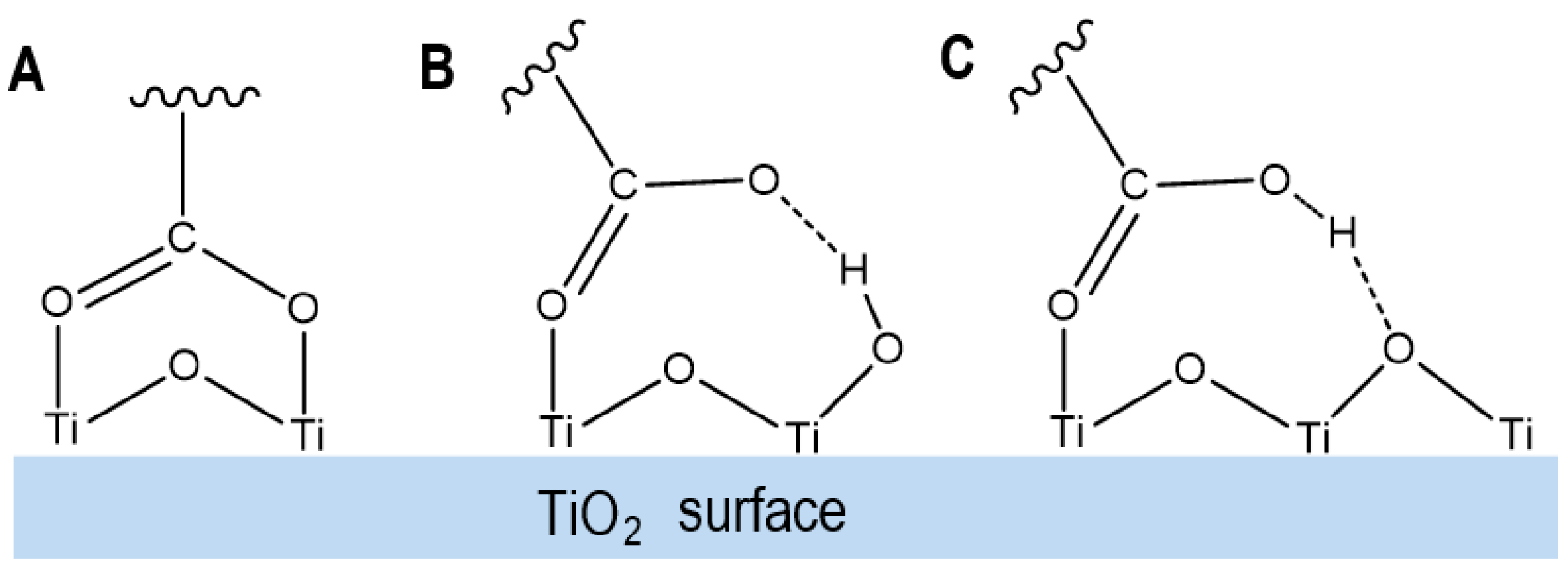
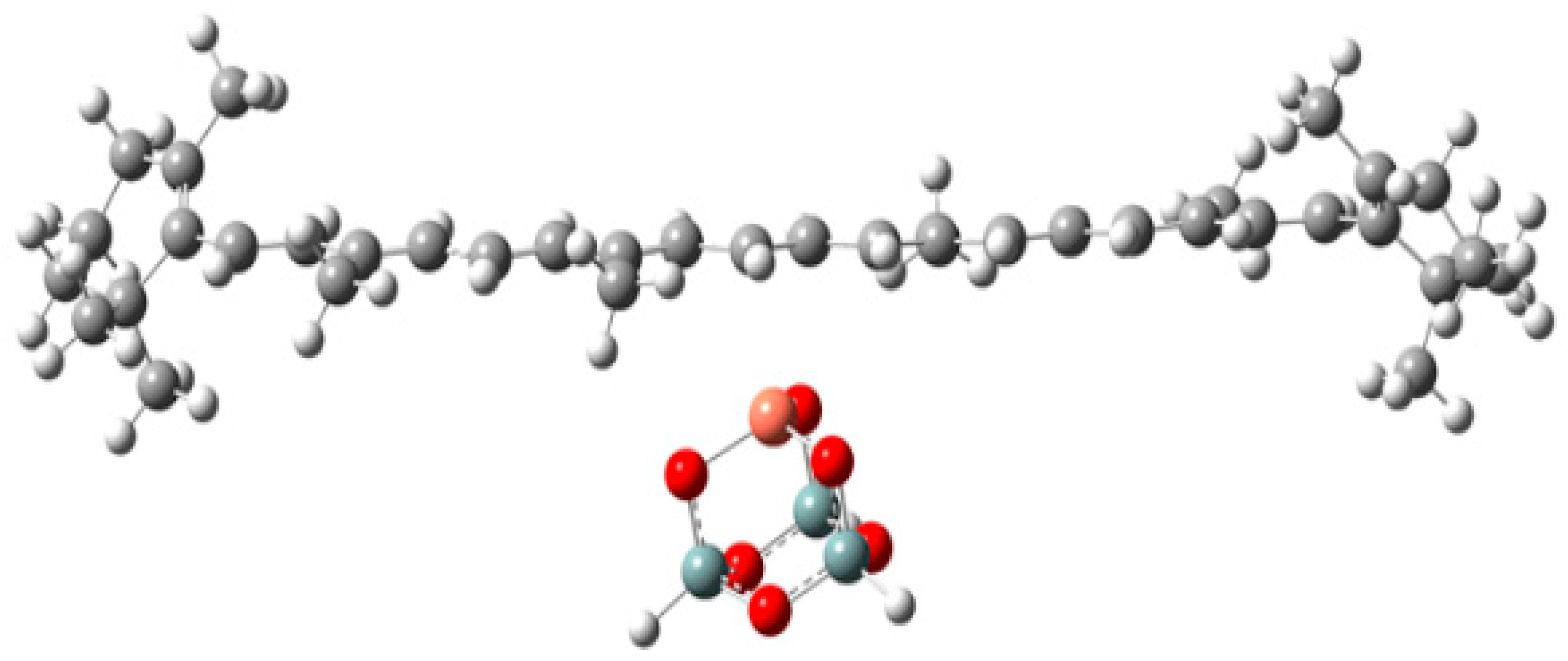
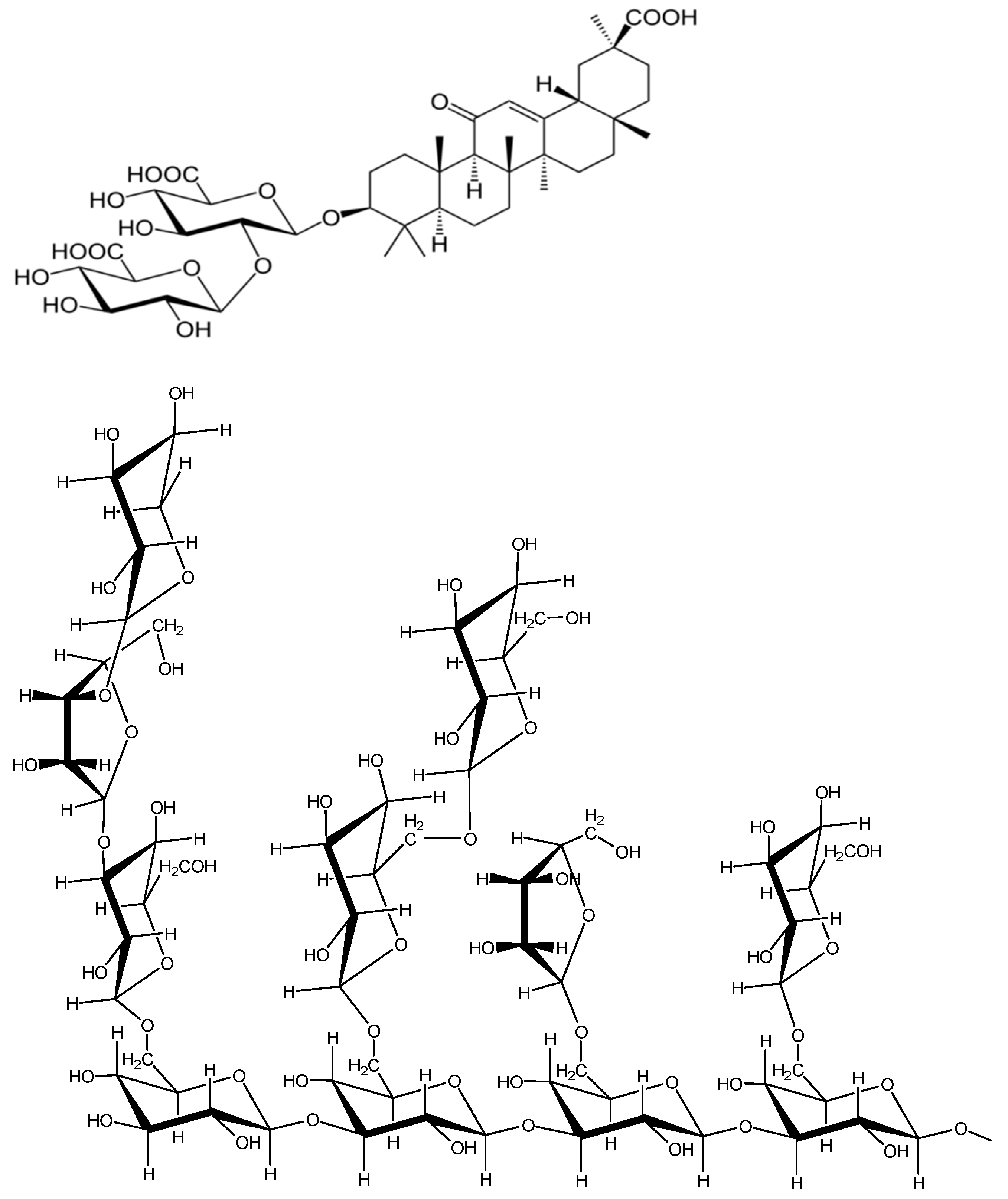
| Rare Carotenoid | Biological Role | Reference |
|---|---|---|
| Siphonaxanthin |
| [53] [54,55] |
| Saproxanthin |
| [56,57] |
| Myxol |
| [56,57] [58] |
| Sioxanthin |
| [56,62] |
| Deinoxanthin |
| [59] [60] |
| Crocin |
| [63] [64] |
| Sarcinaxantin |
| [65] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polyakov, N.E.; Focsan, A.L.; Gao, Y.; Kispert, L.D. The Endless World of Carotenoids—Structural, Chemical and Biological Aspects of Some Rare Carotenoids. Int. J. Mol. Sci. 2023, 24, 9885. https://doi.org/10.3390/ijms24129885
Polyakov NE, Focsan AL, Gao Y, Kispert LD. The Endless World of Carotenoids—Structural, Chemical and Biological Aspects of Some Rare Carotenoids. International Journal of Molecular Sciences. 2023; 24(12):9885. https://doi.org/10.3390/ijms24129885
Chicago/Turabian StylePolyakov, Nikolay E., A. Ligia Focsan, Yunlong Gao, and Lowell D. Kispert. 2023. "The Endless World of Carotenoids—Structural, Chemical and Biological Aspects of Some Rare Carotenoids" International Journal of Molecular Sciences 24, no. 12: 9885. https://doi.org/10.3390/ijms24129885
APA StylePolyakov, N. E., Focsan, A. L., Gao, Y., & Kispert, L. D. (2023). The Endless World of Carotenoids—Structural, Chemical and Biological Aspects of Some Rare Carotenoids. International Journal of Molecular Sciences, 24(12), 9885. https://doi.org/10.3390/ijms24129885








