Similar Transcriptomic Responses to Early and Late Drought Stresses Produce Divergent Phenotypes in Sunflower (Helianthus annuus L.)
Abstract
1. Introduction
2. Results
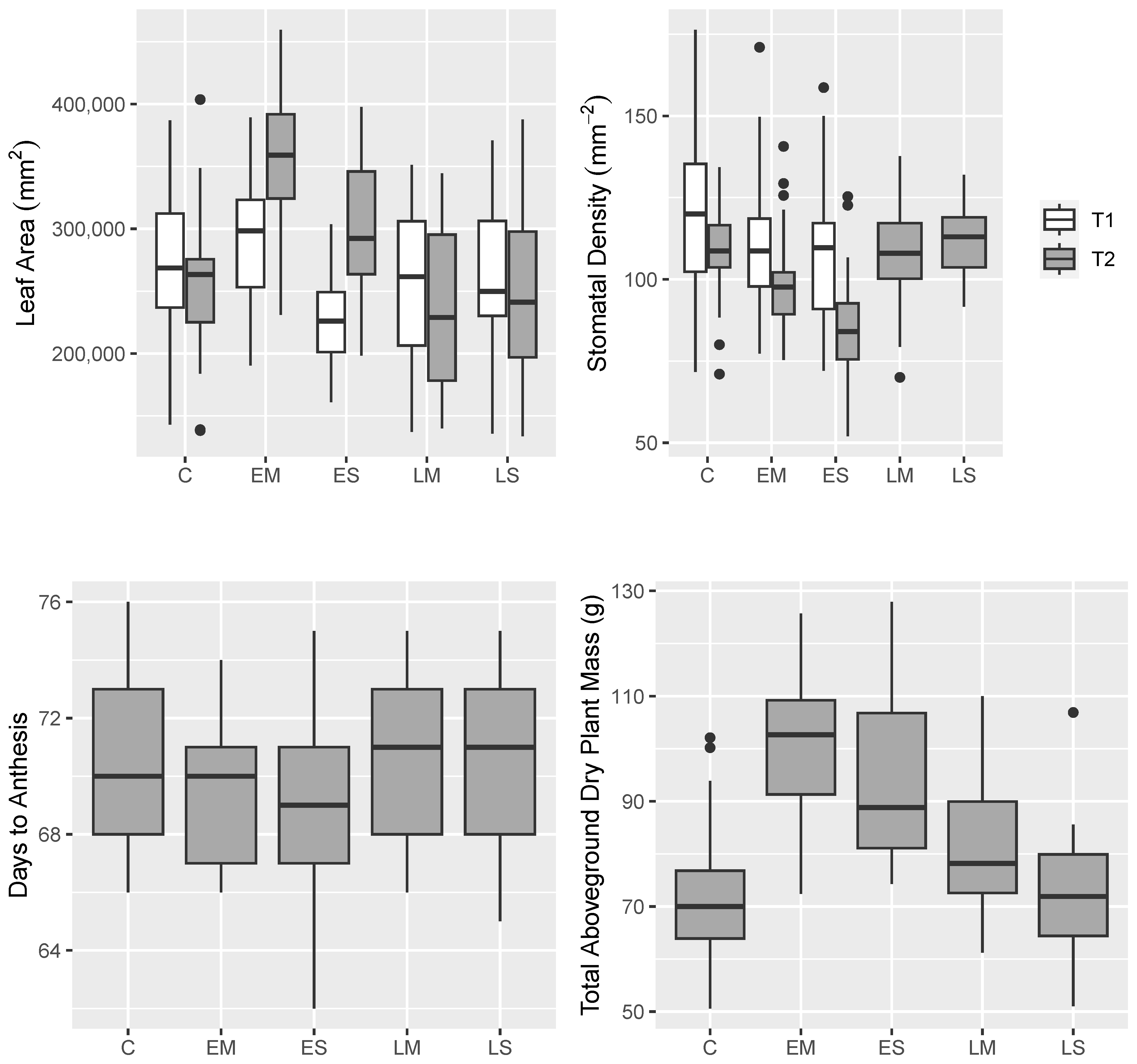
2.1. Phenotypic Analyses
2.1.1. Phenotypic Means and Variance
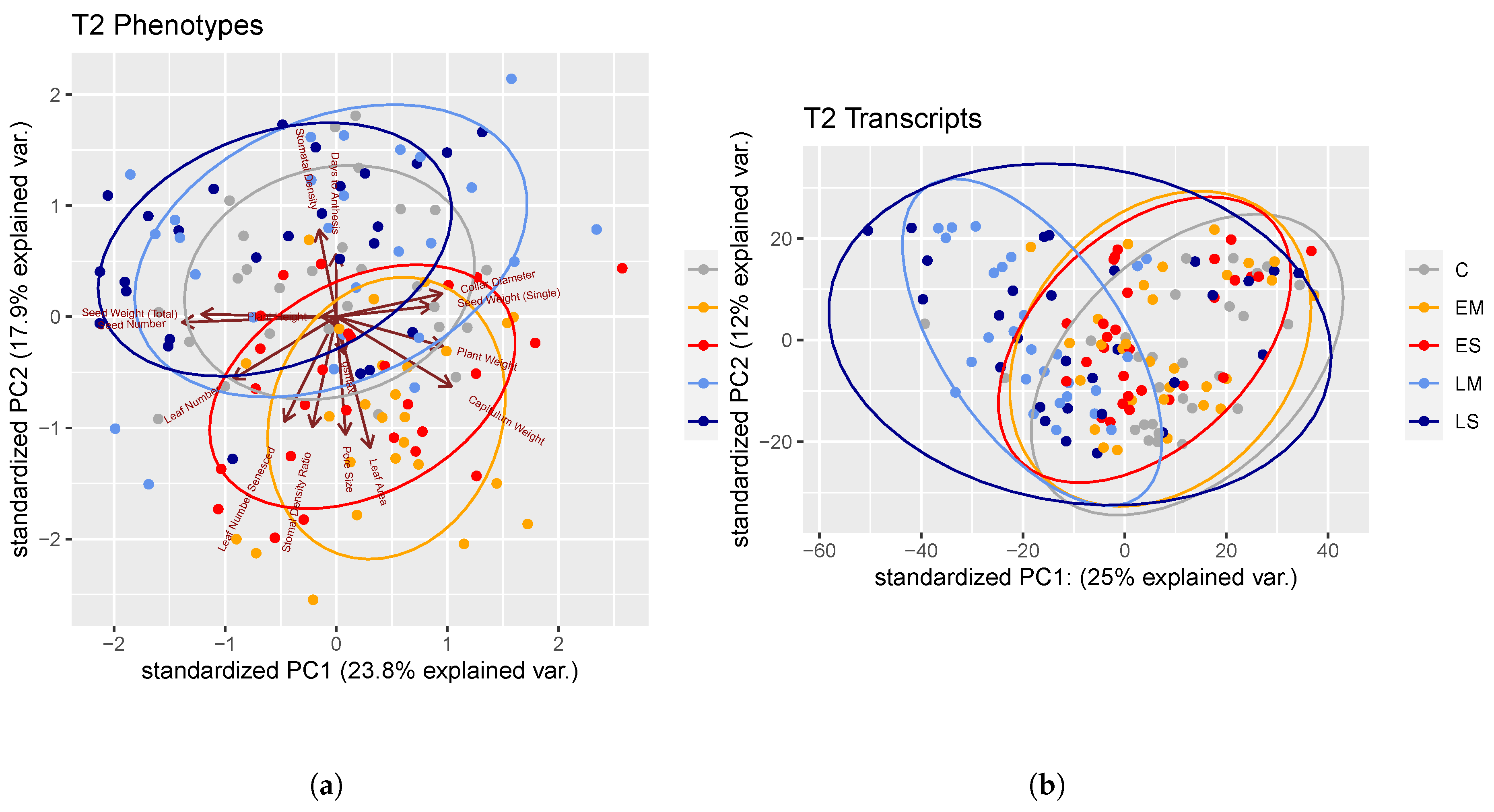
2.1.2. Trait–Trait Correlations
2.2. Transcriptomic Analyses
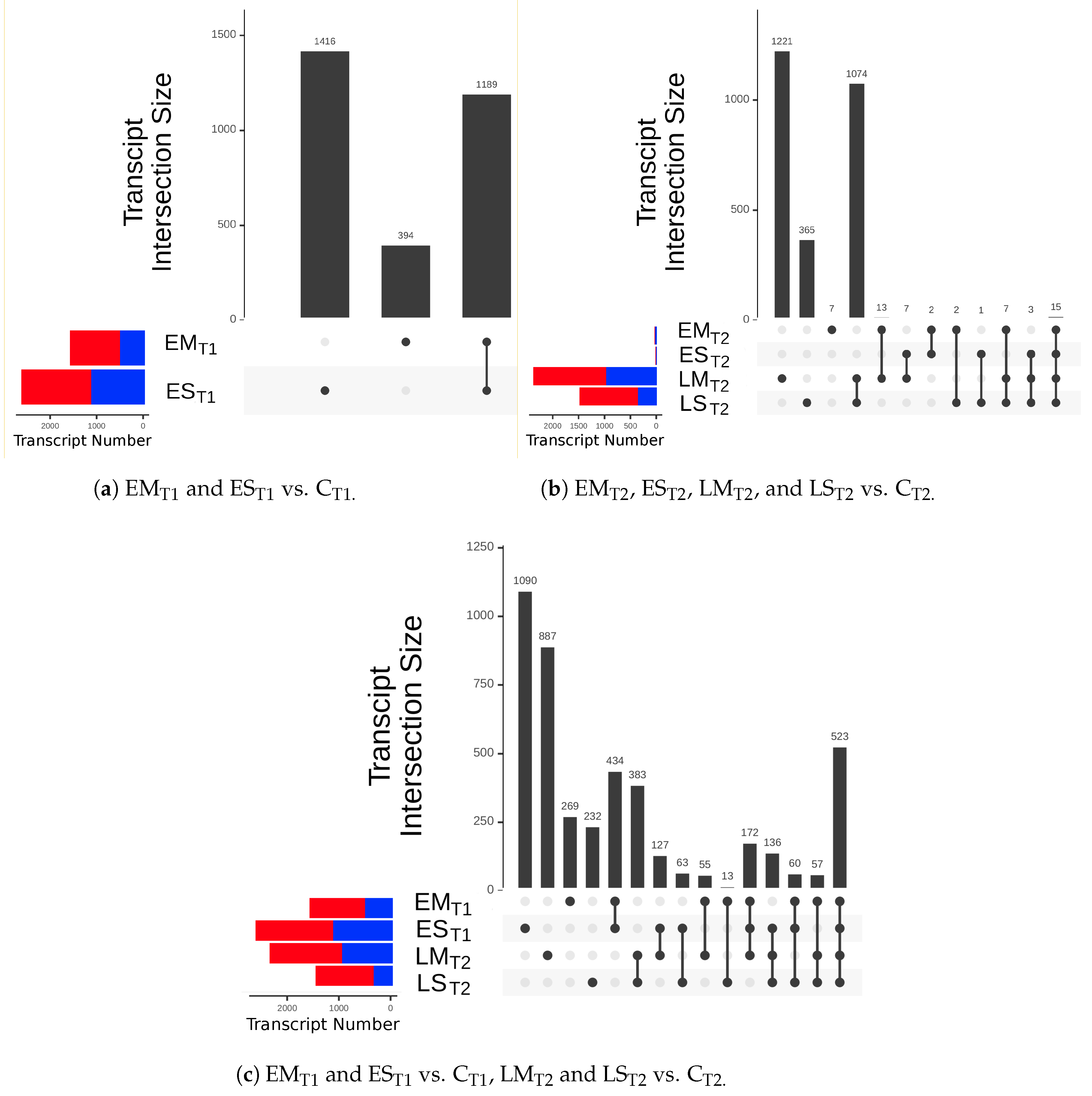
2.2.1. Differential Gene Expression
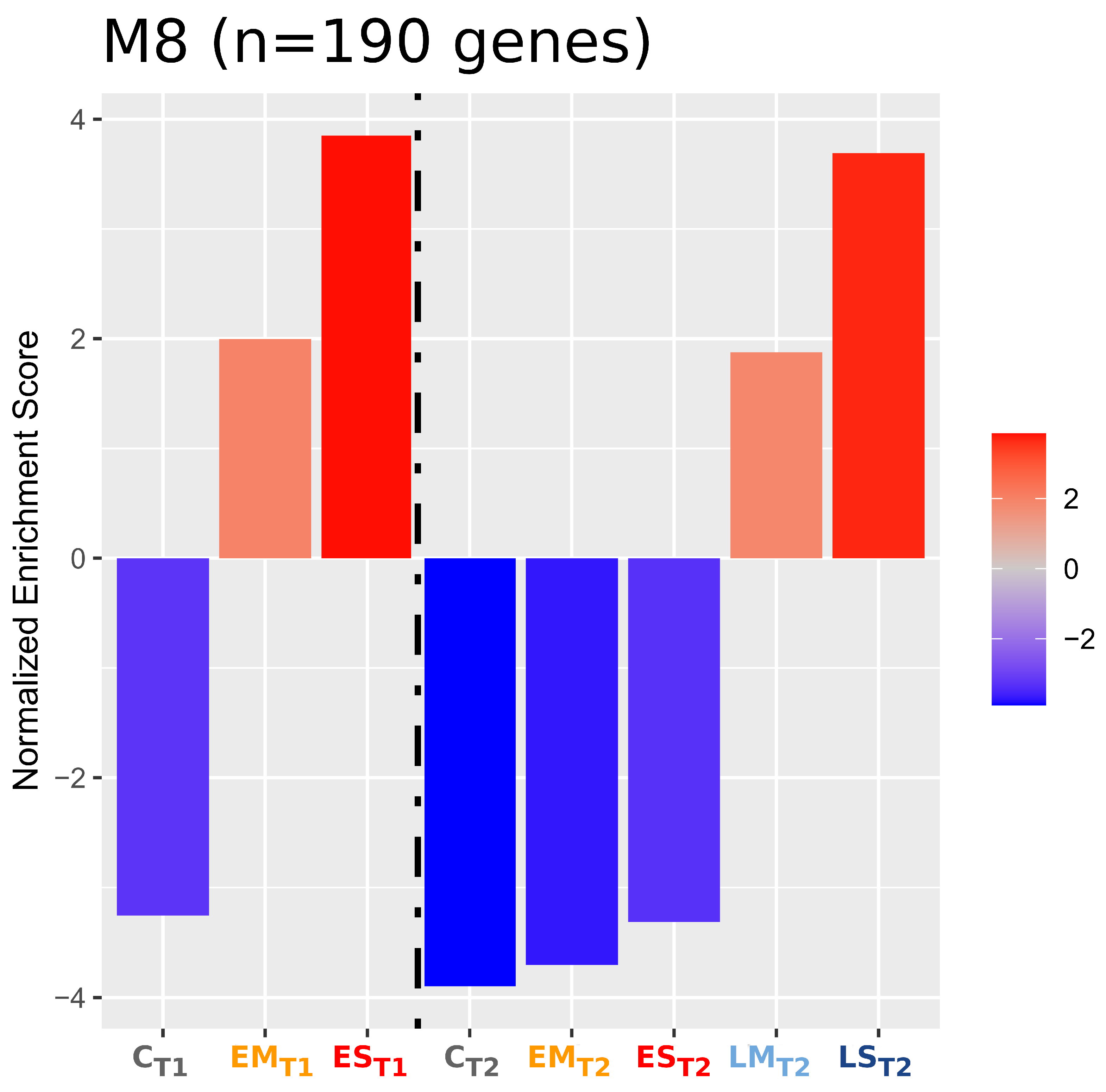
2.2.2. Gene Co-Expression Modules
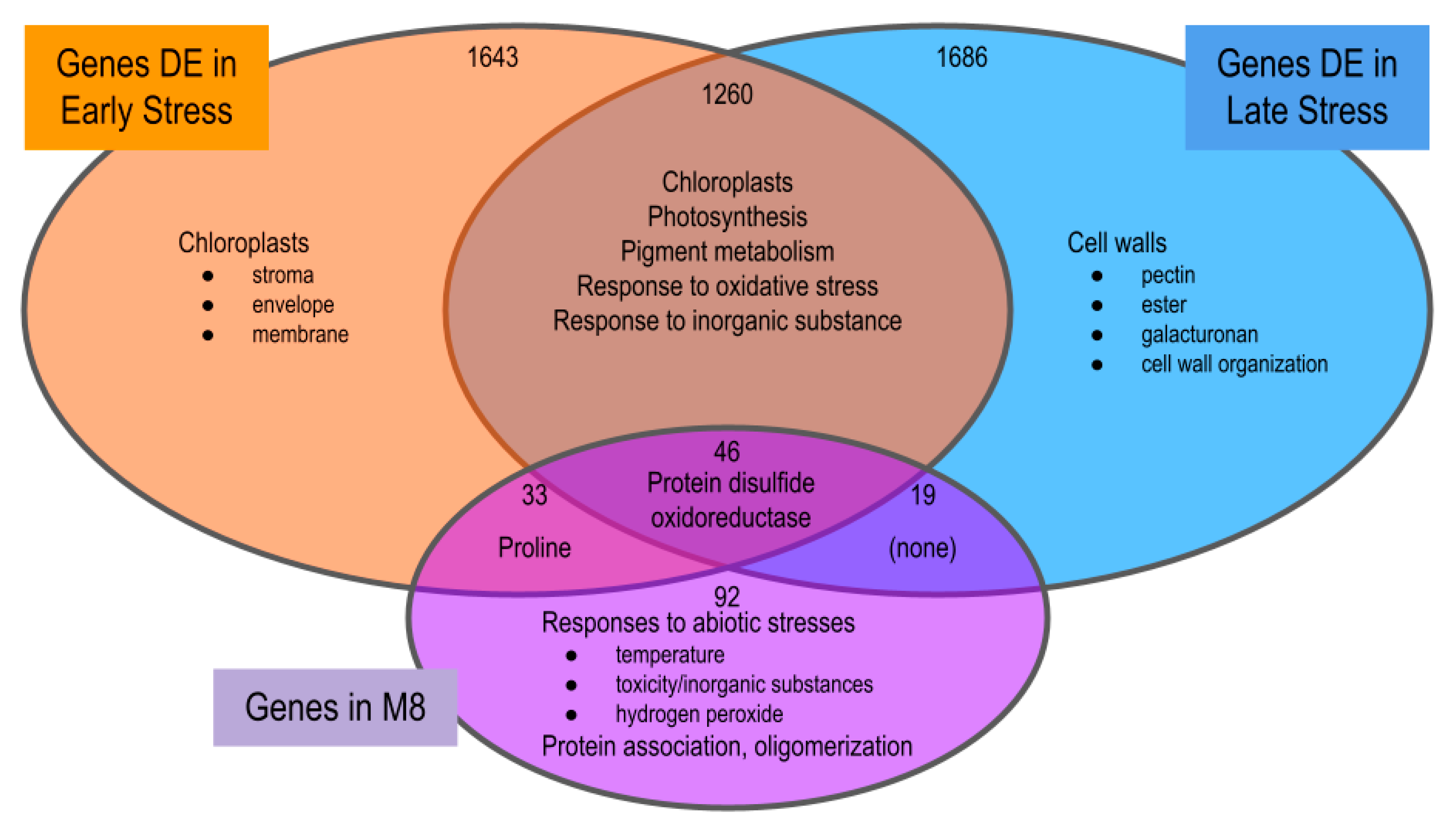
2.2.3. Overrepresentation Analysis (ORA)
3. Discussion
3.1. Similar Transcriptomic Responses to Early and Late Drought Stress Result in Divergent Phenotypic Outcomes
3.2. Divergent Phenotypic Outcomes
3.2.1. Water Use and Biomass
3.2.2. Leaf Traits
3.2.3. Photosynthesis
3.2.4. Trait–Trait Correlations
3.3. Similar Transcriptomic Responses
3.3.1. Differentially Expressed Genes
3.3.2. Co-Expression Networks
3.4. Compensatory and Overcompensatory Growth
3.5. WUE, EUW, and Drought Stress Response Strategies
4. Materials and Methods
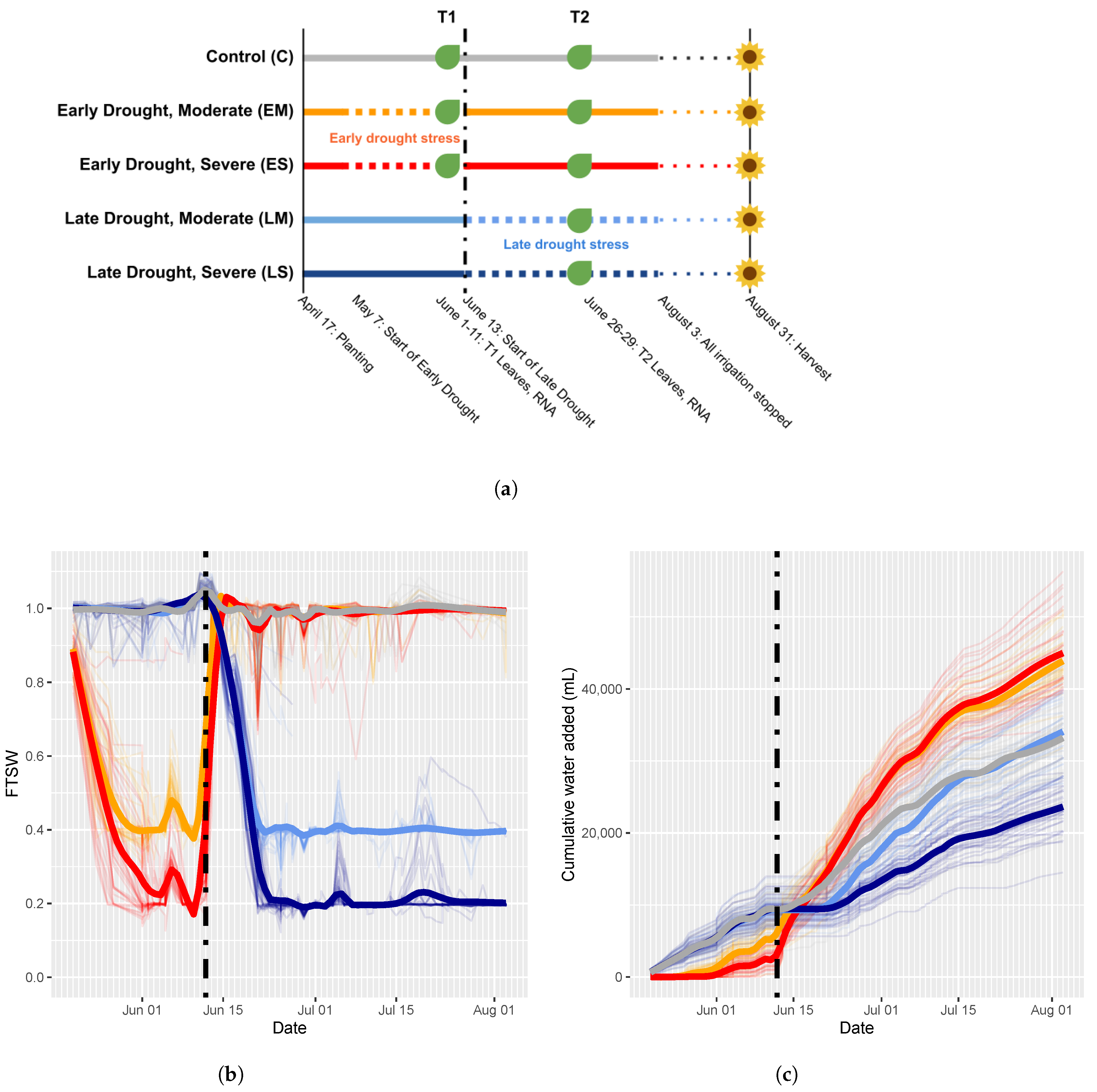
4.1. Experimental Design
4.1.1. Common Garden
4.1.2. Data Collection
4.2. RNA-Seq Pre-Processing and Transcript Quantification
4.3. Phenotypic Analyses
4.3.1. Phenotypic Means and Variance
4.3.2. Trait–Trait Correlations
4.4. Transcriptomic Analyses
4.4.1. Differential Gene Expression
4.4.2. Gene Co-Expression
4.4.3. Overrepresentation Analysis (ORA)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| C | Control |
| DE | Differentially expressed/Differential expression |
| DEG | Differentially expressed gene |
| E | Early |
| EM | Early moderate |
| ES | Early severe |
| EUW | Effective use of water |
| FTSW | Fraction of transpirable soil water |
| GAM | General additive model |
| GO | Gene ontology |
| Maximum stomatal conductance | |
| L | Late |
| LM | Late moderate |
| LS | Late severe |
| NES | Normalized enrichment score |
| ORA | Overrepresentation analysis |
| PCA | Principal component analysis |
| T1 | Timepoint 1 |
| T2 | Timepoint 2 |
| WUE | Water use efficiency |
References
- Cattivelli, L.; Rizza, F.; Badeck, F.W.; Mazzucotelli, E.; Mastrangelo, A.M.; Francia, E.; Marè, C.; Tondelli, A.; Stanca, A.M. Drought tolerance improvement in crop plants: An integrated view from breeding to genomics. Field Crop. Res. 2008, 105, 1–14. [Google Scholar] [CrossRef]
- Khan, M.I.R.; Asgher, M.; Fatma, M.; Per, T.S.; Khan, N.A. Drought stress vis a vis plant functions in the era of climate change. Clim. Chang. Environ. Sustain. 2015, 3, 13–25. [Google Scholar] [CrossRef]
- Hoerling, M.; Eischeid, J.; Perlwitz, J.; Quan, X.; Zhang, T.; Pegion, P. On the increased frequency of Mediterranean drought. J. Clim. 2012, 25, 2146–2161. [Google Scholar] [CrossRef]
- Spinoni, J.; Naumann, G.; Carrao, H.; Barbosa, P.; Vogt, J. World drought frequency, duration, and severity for 1951–2010. Int. J. Climatol. 2014, 34, 2792–2804. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, C.J. A multimodel assessment of the climate change effect on the drought severity–duration–frequency relationship. Hydrol. Process. 2013, 27, 2800–2813. [Google Scholar] [CrossRef]
- Cook, B.I.; Seager, R.; Williams, A.P.; Puma, M.J.; McDermid, S.; Kelley, M.; Nazarenko, L. Climate change amplification of natural drought variability: The historic mid-twentieth-century North American drought in a warmer world. J. Clim. 2019, 32, 5417–5436. [Google Scholar] [CrossRef]
- Levitt, J. Responses of Plants to Environmental Stresses; Academic Press: San Diego, CA, USA, 1972. [Google Scholar]
- Turner, N.C. Adaptation to water deficits: A changing perspective. Funct. Plant Biol. 1986, 13, 175–190. [Google Scholar] [CrossRef]
- Ludlow, M.M.; Muchow, R.C. A critical evaluation of traits for improving crop yields in water-limited environments. Adv. Agron. 1990, 43, 107–153. [Google Scholar]
- Liu, F.; Stützel, H. Biomass partitioning, specific leaf area, and water use efficiency of vegetable amaranth (Amaranthus spp.) in response to drought stress. Sci. Hortic. 2004, 102, 15–27. [Google Scholar] [CrossRef]
- Miyashita, K.; Tanakamaru, S.; Maitani, T.; Kimura, K. Recovery responses of photosynthesis, transpiration, and stomatal conductance in kidney bean following drought stress. Environ. Exp. Bot. 2005, 53, 205–214. [Google Scholar] [CrossRef]
- Sanders, G.J.; Arndt, S.K. Osmotic adjustment under drought conditions. In Plant Responses to Drought Stress: From Morphological to Molecular Features; Springer: Berlin/Heidelberg, Germany, 2012; pp. 199–229. [Google Scholar]
- Mashkina, E.; Usatov, A.; Skorina, M. Comparative analysis of thermotolerance of sunflower chlorophyll mutants. Russ. J. Genet 2010, 46, 178–184. [Google Scholar] [CrossRef]
- Desclaux, D.; Huynh, T.T.; Roumet, P. Identification of soybean plant characteristics that indicate the timing of drought stress. Crop. Sci. 2000, 40, 716–722. [Google Scholar] [CrossRef]
- Perrone, I.; Pagliarani, C.; Lovisolo, C.; Chitarra, W.; Roman, F.; Schubert, A. Recovery from water stress affects grape leaf petiole transcriptome. Planta 2012, 235, 1383–1396. [Google Scholar] [CrossRef]
- Tardieu, F. Any trait or trait-related allele can confer drought tolerance: Just design the right drought scenario. J. Exp. Bot. 2012, 63, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Poormohammad Kiani, S.; Grieu, P.; Maury, P.; Hewezi, T.; Gentzbittel, L.; Sarrafi, A. Genetic variability for physiological traits under drought conditions and differential expression of water stress-associated genes in sunflower (Helianthus annuus L.). Theor. Appl. Genet. 2007, 114, 193–207. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.C.; Jin, J.Q.; Ma, J.Q.; Yao, M.Z.; Ma, C.L.; Li, C.F.; Ding, Z.T.; Chen, L. Transcriptomic analysis of tea plant responding to drought stress and recovery. PLoS ONE 2016, 11, e0147306. [Google Scholar] [CrossRef]
- Hayano-Kanashiro, C.; Calderón-Vázquez, C.; Ibarra-Laclette, E.; Herrera-Estrella, L.; Simpson, J. Analysis of gene expression and physiological responses in three Mexican maize landraces under drought stress and recovery irrigation. PLoS ONE 2009, 4, e7531. [Google Scholar] [CrossRef]
- Cohen, D.; Bogeat-Triboulot, M.B.; Tisserant, E.; Balzergue, S.; Martin-Magniette, M.L.; Lelandais, G.; Ningre, N.; Renou, J.P.; Tamby, J.P.; Le Thiec, D.; et al. Comparative transcriptomics of drought responses in Populus: A meta-analysis of genome-wide expression profiling in mature leaves and root apices across two genotypes. BMC Genom. 2010, 11, 630. [Google Scholar] [CrossRef]
- Angadi, S.V.; Entz, M.H. Root system and water use patterns of different height sunflower cultivars. Agron. J. 2002, 94, 136–145. [Google Scholar] [CrossRef]
- Sadras, V.; Whitfield, D.; Connor, D. Regulation of evapotranspiration, and its partitioning between transpiration and soil evaporation by sunflower crops: A comparison between hybrids of different stature. Field Crop. Res. 1991, 28, 17–37. [Google Scholar] [CrossRef]
- Ouvrard, O.; Cellier, F.; Ferrare, K.; Tousch, D.; Lamaze, T.; Dupuis, J.M.; Casse-Delbart, F. Identification and expression of water stress-and abscisic acid-regulated genes in a drought-tolerant sunflower genotype. Plant Mol. Biol. 1996, 31, 819–829. [Google Scholar] [CrossRef] [PubMed]
- Moschen, S.; Di Rienzo, J.A.; Higgins, J.; Tohge, T.; Watanabe, M.; González, S.; Rivarola, M.; García-García, F.; Dopazo, J.; Hopp, H.E.; et al. Integration of transcriptomic and metabolic data reveals hub transcription factors involved in drought stress response in sunflower (Helianthus annuus L.). Plant Mol. Biol. 2017, 94, 549–564. [Google Scholar] [CrossRef] [PubMed]
- Takami, S.; Turner, N.C.; Rawson, H.M. Leaf expansion of four sunflower (Helianthus annuus L) cultivars in relation to water deficits. I. Patterns during plant development. Plant Cell Environ. 1981, 4, 399–407. [Google Scholar] [CrossRef]
- Lindhauer, M. Influence of K nutrition and drought on water relations and growth of sunflower (Helianthus annuus L.). Z. Pflanz. Bodenkd. 1985, 148, 654–669. [Google Scholar] [CrossRef]
- Manivannan, P.; Jaleel, C.A.; Sankar, B.; Kishorekumar, A.; Somasundaram, R.; Lakshmanan, G.A.; Panneerselvam, R. Growth, biochemical modifications and proline metabolism in Helianthus annuus L. as induced by drought stress. Colloids Surfaces Biointerfaces 2007, 59, 141–149. [Google Scholar] [CrossRef]
- Hussain, M.; Farooq, S.; Hasan, W.; Ul-Allah, S.; Tanveer, M.; Farooq, M.; Nawaz, A. Drought stress in sunflower: Physiological effects and its management through breeding and agronomic alternatives. Agric. Water Manag. 2018, 201, 152–166. [Google Scholar] [CrossRef]
- Comas, L.; Becker, S.; Cruz, V.M.V.; Byrne, P.F.; Dierig, D.A. Root traits contributing to plant productivity under drought. Front. Plant Sci. 2013, 4, 442. [Google Scholar] [CrossRef]
- Pekcan, V.; Evci, G.; Yilmaz, M.I.; Nalcaiyi, A.B.; Erdal, Ş.Ç.; Cicek, N.; Arslan, O.; Ekmekci, Y.; Kaya, Y. Effects of drought stress on sunflower stems and roots. In Proceedings of the International Conference on Chemical, Agricultural and Life Sciences (CALS-16), Bali, Indonesia, 4–5 February 2016; pp. 4–5. [Google Scholar]
- Blanchet, N.; Casadebaig, P.; Debaeke, P.; Duruflé, H.; Gody, L.; Gosseau, F.; Langlade, N.B.; Maury, P. Data describing the eco-physiological responses of twenty-four sunflower genotypes to water deficit. Data Brief 2018, 21, 1296–1301. [Google Scholar] [CrossRef]
- Barnhart, M.H.; Masalia, R.R.; Mosley, L.J.; Burke, J.M. Phenotypic and transcriptomic responses of cultivated sunflower seedlings (Helianthus annuus L.) to four abiotic stresses. PLoS ONE 2022, 17, e0275462. [Google Scholar] [CrossRef]
- Tran, V.H. Exploring the Physiological Basis of Salinity and Drought Tolerance in Wild and Cultivated Sunflower (Helianthus annuus). Ph.D. Thesis, University of Georgia, Athens, Georgia, 2021. [Google Scholar]
- Van der Weele, C.M.; Spollen, W.G.; Sharp, R.E.; Baskin, T.I. Growth of Arabidopsis thaliana seedlings under water deficit studied by control of water potential in nutrient-agar media. J. Exp. Bot. 2000, 51, 1555–1562. [Google Scholar] [CrossRef]
- Sharp, R.; Davies, W. Solute regulation and growth by roots and shoots of water-stressed maize plants. Planta 1979, 147, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Sponchiado, B.; White, J.W.; Castillo, J.; Jones, P. Root growth of four common bean cultivars in relation to drought tolerance in environments with contrasting soil types. Exp. Agric. 1989, 25, 249–257. [Google Scholar] [CrossRef]
- Ku, Y.S.; Au-Yeung, W.K.; Yung, Y.L.; Li, M.W.; Wen, C.Q.; Liu, X.; Lam, H.M. Drought stress and tolerance in soybean. In A Comprehensive Survey of International Soybean Research—Genetics, Physiology, Agronomy and Nitrogen Relationships; InTech: Rijeka, Croatia, 2013; pp. 209–237. [Google Scholar]
- Dalal, M.; Sahu, S.; Tiwari, S.; Rao, A.R.; Gaikwad, K. Transcriptome analysis reveals interplay between hormones, ROS metabolism and cell wall biosynthesis for drought-induced root growth in wheat. Plant Physiol. Biochem. 2018, 130, 482–492. [Google Scholar] [CrossRef]
- Niu, J.; Zhang, S.; Liu, S.; Ma, H.; Chen, J.; Shen, Q.; Ge, C.; Zhang, X.; Pang, C.; Zhao, X. The compensation effects of physiology and yield in cotton after drought stress. J. Plant Physiol. 2018, 224, 30–48. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, J. Adaptation of rice environments. In Drought Resistance in Crops with Emphasis on Rice; Paddyfield: Manila, PA, USA, 1982; pp. 195–213. [Google Scholar]
- Sultan, S.E. Phenotypic plasticity in plants: A case study in ecological development. Evol. Dev. 2003, 5, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Chapin, F.S., III. Integrated responses of plants to stress: A centralized system of physiological responses. Bioscience 1991, 41, 29–36. [Google Scholar] [CrossRef]
- Keipp, K.; Hütsch, B.W.; Ehlers, K.; Schubert, S. Drought stress in sunflower causes inhibition of seed filling due to reduced cell-extension growth. J. Agron. Crop. Sci. 2020, 206, 517–528. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Sharp, R.E. Complexity and coordination of root growth at low water potentials: Recent advances from transcriptomic and proteomic analyses. Plant Cell Environ. 2010, 33, 590–603. [Google Scholar] [CrossRef]
- Sharp, R.; Davies, W. Regulation of growth and development of plants growing with a restricted supply of water. Plants Stress Biochem. Physiol. Ecol. Their Appl. Plant Improv. 1989, 39, 71. [Google Scholar]
- Wu, Y.; Cosgrove, D.J. Adaptation of roots to low water potentials by changes in cell wall extensibility and cell wall proteins. J. Exp. Bot. 2000, 51, 1543–1553. [Google Scholar] [CrossRef]
- Arve, L.; Torre, S.; Olsen, J.; Tanino, K. Stomatal responses to drought stress and air humidity. In Abiotic Stress in Plants-Mechanisms and Adaptations; IntechOpen: London, UK, 2011. [Google Scholar]
- Ennos, A.; Crook, M.; Grimshaw, C. A comparative study of the anchorage systems of Himalayan balsam Impatiens glandulifera and mature sunflower Helianthus annuus. J. Exp. Bot. 1993, 44, 133–146. [Google Scholar] [CrossRef]
- Condon, A.G.; Richards, R.; Rebetzke, G.; Farquhar, G. Improving intrinsic water-use efficiency and crop yield. Crop. Sci. 2002, 42, 122–131. [Google Scholar] [PubMed]
- Sakoda, K.; Taniyoshi, K.; Yamori, W.; Tanaka, Y. Drought stress reduces crop carbon gain due to delayed photosynthetic induction under fluctuating light conditions. Physiol. Plant. 2022, 174, e13603. [Google Scholar] [CrossRef] [PubMed]
- Conesa, M.À.; Muir, C.D.; Molins, A.; Galmés, J. Stomatal anatomy coordinates leaf size with Rubisco kinetics in the Balearic Limonium. AoB Plants 2020, 12, plz050. [Google Scholar] [CrossRef]
- Spence, R.; Wu, H.; Sharpe, P.; Clark, K. Water stress effects on guard cell anatomy and the mechanical advantage of the epidermal cells. Plant Cell Environ. 1986, 9, 197–202. [Google Scholar]
- Bertolino, L.T.; Caine, R.S.; Gray, J.E. Impact of stomatal density and morphology on water-use efficiency in a changing world. Front. Plant Sci. 2019, 10, 225. [Google Scholar] [CrossRef]
- Xu, Z.; Zhou, G. Responses of leaf stomatal density to water status and its relationship with photosynthesis in a grass. J. Exp. Bot. 2008, 59, 3317–3325. [Google Scholar] [CrossRef]
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S. Plant drought stress: Effects, mechanisms and management. In Sustainable Agriculture; Springer: Dordrecht, The Netherlands, 2009; pp. 153–188. [Google Scholar]
- Franks, P.J.; Doheny-Adams, T.W.; Britton-Harper, Z.J.; Gray, J.E. Increasing water-use efficiency directly through genetic manipulation of stomatal density. New Phytol. 2015, 207, 188–195. [Google Scholar] [CrossRef]
- Rawson, H.M.; Turner, N. Recovery from water stress in five sunflower (Helianthus annuus L.) cultivars. II. The development of leaf area. Funct. Plant Biol. 1982, 9, 449–460. [Google Scholar] [CrossRef]
- Rengel, D.; Arribat, S.; Maury, P.; Martin-Magniette, M.L.; Hourlier, T.; Laporte, M.; Vares, D.; Carrere, S.; Grieu, P.; Balzergue, S.; et al. A gene-phenotype network based on genetic variability for drought responses reveals key physiological processes in controlled and natural environments. PLoS ONE 2012, 10, e45249. [Google Scholar] [CrossRef]
- Wahid, A.; Rasul, E.; Rao, R.; Iqbal, R. Photosynthesis in leaf, stem, flower and fruit. Handb. Photosynth. 2005, 2, 479–497. [Google Scholar]
- Arslan, Ö.; Nalçaiyi, A.B.; Erdal, Ş.Ç.; Pekcan, V.; Kaya, Y.; Çiçek, N.; Ekmekçi, Y. Analysis of drought response of sunflower inbred lines by chlorophyll a fluorescence induction kinetics. Photosynthetica 2020, 58, 163–172. [Google Scholar] [CrossRef]
- Liang, C.; Wang, W.; Wang, J.; Ma, J.; Li, C.; Zhou, F.; Zhang, S.; Yu, Y.; Zhang, L.; Li, W.; et al. Identification of differentially expressed genes in sunflower (Helianthus annuus) leaves and roots under drought stress by RNA sequencing. Bot. Stud. 2017, 58, 42. [Google Scholar] [CrossRef]
- Meinzer, F.C. Stomatal control of transpiration. Trends Ecol. Evol. 1993, 8, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Rizhsky, L.; Liang, H.; Shuman, J.; Shulaev, V.; Davletova, S.; Mittler, R. When defense pathways collide. The response of Arabidopsis to a combination of drought and heat stress. Plant Physiol. 2004, 134, 1683–1696. [Google Scholar] [CrossRef] [PubMed]
- Laxa, M.; Liebthal, M.; Telman, W.; Chibani, K.; Dietz, K.J. The role of the plant antioxidant system in drought tolerance. Antioxidants 2019, 8, 94. [Google Scholar] [CrossRef] [PubMed]
- Chaves, M.; Costa, J.M.; Zarrouk, O.; Pinheiro, C.; Lopes, C.; Pereira, J.S. Controlling stomatal aperture in semi-arid regions—The dilemma of saving water or being cool? Plant Sci. 2016, 251, 54–64. [Google Scholar] [CrossRef]
- Gosseau, F.; Blanchet, N.; Varès, D.; Burger, P.; Campergue, D.; Colombet, C.; Gody, L.; Liévin, J.F.; Mangin, B.; Tison, G.; et al. Heliaphen, an outdoor high-throughput phenotyping platform for genetic studies and crop modeling. Front. Plant Sci. 2019, 9, 1908. [Google Scholar] [CrossRef]
- Rüdiger, H.; Gabius, H.J. Plant lectins: Occurrence, biochemistry, functions and applications. Glycoconj. J. 2001, 18, 589–613. [Google Scholar] [CrossRef]
- Singh, P.; Bhaglal, P.; Bhullar, S. Wheat germ agglutinin (WGA) gene expression and ABA accumulation in the developing embryos of wheat (Triticum aestivum) in response to drought. Plant Growth Regul. 2000, 30, 145–150. [Google Scholar] [CrossRef]
- Tsaneva, M.; Van Damme, E.J. 130 years of plant lectin research. Glycoconj. J. 2020, 37, 533–551. [Google Scholar] [CrossRef] [PubMed]
- Vítámvás, P.; Urban, M.O.; Škodáček, Z.; Kosová, K.; Pitelková, I.; Vítámvás, J.; Renaut, J.; Prášil, I.T. Quantitative analysis of proteome extracted from barley crowns grown under different drought conditions. Front. Plant Sci. 2015, 6, 479. [Google Scholar] [CrossRef] [PubMed]
- Alsamman, A.M.; Bousba, R.; Baum, M.; Hamwieh, A.; Fouad, N. Comprehensive analysis of the gene expression profile of wheat at the crossroads of heat, drought and combined stress. Highlights Biosci. 2021, 4, bs202104. [Google Scholar] [CrossRef]
- Kregel, K.C. Invited review: Heat shock proteins: Modifying factors in physiological stress responses and acquired thermotolerance. J. Appl. Physiol. 2002, 92, 2177–2186. [Google Scholar] [CrossRef]
- Barnett, N.M.; Naylor, A. Amino acid and protein metabolism in Bermuda grass during water stress. Plant Physiol. 1966, 41, 1222–1230. [Google Scholar] [CrossRef]
- Sánchez, F.J.; Manzanares, M.; de Andres, E.F.; Tenorio, J.L.; Ayerbe, L. Turgor maintenance, osmotic adjustment and soluble sugar and proline accumulation in 49 pea cultivars in response to water stress. Field Crop. Res. 1998, 59, 225–235. [Google Scholar] [CrossRef]
- Sofo, A.; Dichio, B.; Xiloyannis, C.; Masia, A. Lipoxygenase activity and proline accumulation in leaves and roots of olive trees in response to drought stress. Physiol. Plant. 2004, 121, 58–65. [Google Scholar] [CrossRef]
- Wang, L.; Zu, W.; Dong, S.; Liu, L.; Xu, Y.; Li, X. Effects of drought stresses and times on compensation effect after re-watering in soybean. Trans. Chin. Soc. Agric. Eng. 2015, 31, 150–156. [Google Scholar]
- Cechin, I.; Rossi, S.; Oliveira, V.; Fumis, T.D.F. Photosynthetic responses and proline content of mature and young leaves of sunflower plants under water deficit. Photosynthetica 2006, 44, 143–146. [Google Scholar] [CrossRef]
- Riemer, J.; Bulleid, N.; Herrmann, J.M. Disulfide formation in the ER and mitochondria: Two solutions to a common process. Science 2009, 324, 1284–1287. [Google Scholar] [CrossRef]
- Baena-González, E.; Aro, E.M. Biogenesis, assembly and turnover of photosystem II units. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2002, 357, 1451–1460. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, X.; Li, R.; Yuan, L.; Dai, Y.; Wang, X. Identification and functional analysis of a protein disulfide isomerase (AtPDI1) in Arabidopsis thaliana. Front. Plant Sci. 2018, 9, 913. [Google Scholar] [CrossRef] [PubMed]
- Iraki, N.M.; Singh, N.; Bressan, R.A.; Carpita, N.C. Cell walls of tobacco cells and changes in composition associated with reduced growth upon adaptation to water and saline stress. Plant Physiol. 1989, 91, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Wormit, A.; Usadel, B. The multifaceted role of pectin methylesterase inhibitors (PMEIs). Int. J. Mol. Sci. 2018, 19, 2878. [Google Scholar] [CrossRef]
- Dong, S.; Jiang, Y.; Dong, Y.; Wang, L.; Wang, W.; Ma, Z.; Yan, C.; Ma, C.; Liu, L. A study on soybean responses to drought stress and rehydration. Saudi J. Biol. Sci. 2019, 26, 2006–2017. [Google Scholar] [CrossRef]
- Poveda, K.; Díaz, M.F.; Ramirez, A. Can overcompensation increase crop production? Ecology 2018, 99, 270–280. [Google Scholar] [CrossRef]
- Hoogenboom, G.; Peterson, C.M.; Huck, M. Shoot growth rate of soybean as affected by drought stress. Agron. J. 1987, 79, 598–607. [Google Scholar] [CrossRef]
- Liu, Y.J.; Ren, J.H.; Yin, L.N.; Deng, X.P.; Ke, Q.B.; Wang, S.W. Compensation effect of re-watering after different drought stresses on source-sink metabolism during tuber expansion period of potato. Ying Yong Sheng Tai Xue Bao = J. Appl. Ecol. 2019, 30, 3777–3786. [Google Scholar]
- Xu, Z.; Zhou, G.; Shimizu, H. Plant responses to drought and rewatering. Plant Signal. Behav. 2010, 5, 649–654. [Google Scholar] [CrossRef]
- Dulloo, A.G. Human pattern of food intake and fuel-partitioning during weight recovery after starvation: A theory of autoregulation of body composition. Proc. Nutr. Soc. 1997, 56, 25–40. [Google Scholar] [CrossRef]
- Hayward, R.S.; Noltie, D.B.; Wang, N. Use of compensatory growth to double hybrid sunfish growth rates. Trans. Am. Fish. Soc. 1997, 126, 316–322. [Google Scholar] [CrossRef]
- Meckel, L.; Egli, D.; Phillips, R.; Radcliffe, D.; Leggett, J. Effect of Moisture Stress on Seed Growth in Soybeans 1. Agron. J. 1984, 76, 647–650. [Google Scholar] [CrossRef]
- Desclaux, D.; Roumet, P. Impact of drought stress on the phenology of two soybean (Glycine max L. Merr) cultivars. Field Crop. Res. 1996, 46, 61–70. [Google Scholar] [CrossRef]
- Samarah, N.H. Effects of drought stress on growth and yield of barley. Agron. Sustain. Dev. 2005, 25, 145–149. [Google Scholar] [CrossRef]
- Estrada-Campuzano, G.; Miralles, D.J.; Slafer, G.A. Genotypic variability and response to water stress of pre-and post-anthesis phases in triticale. Eur. J. Agron. 2008, 28, 171–177. [Google Scholar] [CrossRef]
- Rahman, S.M.A.; Ellis, R.H. Seed quality in rice is most sensitive to drought and high temperature in early seed development. Seed Sci. Res. 2019, 29, 238–249. [Google Scholar] [CrossRef]
- Sinclair, T.; Bennett, J.; Muchow, R. Relative sensitivity of grain yield and biomass accumulation to drought in field-grown maize. Crop. Sci. 1990, 30, 690–693. [Google Scholar] [CrossRef]
- Kasu-Bandi, B.T.; Kidinda, L.K.; Kasendue, G.N.; Longanza, L.B.; Emery, K.L.; Lubobo, A.K. Correlations between Growth and Yield Parameters of Soybean (Glycine max (L.) Merr.) under the Influence of Bradyrhizobium japonicum in Kipushi (The Democratic Republic of Congo). Am. J. Agric. Biol. Sci. 2019, 14, 86–94. [Google Scholar] [CrossRef]
- Rawson, H.M.; Turner, N. Recovery from water stress in five sunflower (Helianthus annuus L.) cultivars. I. Effects of the timing of water application on leaf area and seed production. Funct. Plant Biol. 1982, 9, 437–448. [Google Scholar] [CrossRef]
- Blum, A. Effective use of water (EUW) and not water-use efficiency (WUE) is the target of crop yield improvement under drought stress. Field Crop. Res. 2009, 112, 119–123. [Google Scholar] [CrossRef]
- Blum, A. Drought resistance, water-use efficiency, and yield potential—Are they compatible, dissonant, or mutually exclusive? Aust. J. Agric. Res. 2005, 56, 1159–1168. [Google Scholar] [CrossRef]
- Rawson, H.; Munns, R. Leaf expansion in sunflower as influenced by salinity and short-term changes in carbon fixation. Plant Cell Environ. 1984, 7, 207–213. [Google Scholar]
- Connor, D.; Sadras, V. Physiology of yield expression in sunflower. Field Crop. Res. 1992, 30, 333–389. [Google Scholar] [CrossRef]
- Palacios-de la Rosa, G. Variedades e híbridos de maíz “latentes” y tolerantes a sequía ya las heladas. Rev. Méx. Agríc. 1959, 107, 38–39. [Google Scholar]
- Srinivasan, G.; Bänziger, M.; Edmeades, G.; Lothrop, J.; Torres, J. Identification of drought tolerance in elite tropical highland maize germplasm. In Drought-and Low N-Tolerant Maize; CIMMYT: El Batan, Mexico, 1996; p. 309. [Google Scholar]
- Mandel, J.R.; Nambeesan, S.; Bowers, J.E.; Marek, L.F.; Ebert, D.; Rieseberg, L.H.; Knapp, S.J.; Burke, J.M. Association mapping and the genomic consequences of selection in sunflower. PLoS Genet. 2013, 9, e1003378. [Google Scholar] [CrossRef]
- Casadebaig, P.; Debaeke, P.; Lecoeur, J. Thresholds for leaf expansion and transpiration response to soil water deficit in a range of sunflower genotypes. Eur. J. Agron. 2008, 28, 646–654. [Google Scholar] [CrossRef]
- Earley, A.M.; Temme, A.A.; Cotter, C.R.; Burke, J.M. Genomic regions associate with major axes of variation driven by gas exchange and leaf construction traits in cultivated sunflower (Helianthus annuus L.). Plant J. 2022, 111, 1425–1438. [Google Scholar] [CrossRef]
- Dow, G.J.; Bergmann, D.C.; Berry, J.A. An integrated model of stomatal development and leaf physiology. New Phytol. 2014, 201, 1218–1226. [Google Scholar] [CrossRef]
- Parlange, J.Y.; Waggoner, P.E. Stomatal dimensions and resistance to diffusion. Plant Physiol. 1970, 46, 337–342. [Google Scholar] [CrossRef]
- Franks, P.J.; Farquhar, G.D. The effect of exogenous abscisic acid on stomatal development, stomatal mechanics, and leaf gas exchange in Tradescantia virginiana. Plant Physiol. 2001, 125, 935–942. [Google Scholar] [CrossRef]
- Wingett, S.W.; Andrews, S. FastQ Screen: A tool for multi-genome mapping and quality control. F1000Research 2018, 7, 1338. [Google Scholar] [CrossRef] [PubMed]
- Ewels, P.; Magnusson, M.; Lundin, S.; Käller, M. MultiQC: Summarize analysis results for multiple tools and samples in a single report. Bioinformatics 2016, 32, 3047–3048. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef] [PubMed]
- Bates, D.M. lme4: Mixed-Effects Modeling with R; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Dixon, P. VEGAN, a package of R functions for community ecology. J. Veg. Sci. 2003, 14, 927–930. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Russo, P.S.; Ferreira, G.R.; Cardozo, L.E.; Bürger, M.C.; Arias-Carrasco, R.; Maruyama, S.R.; Hirata, T.D.; Lima, D.S.; Passos, F.M.; Fukutani, K.F.; et al. CEMiTool: A Bioconductor package for performing comprehensive modular co-expression analyses. BMC Bioinform. 2018, 19, 56. [Google Scholar] [CrossRef]
- Langfelder, P.; Zhang, B.; Horvath, S. Defining clusters from a hierarchical cluster tree: The Dynamic Tree Cut package for R. Bioinformatics 2008, 24, 719–720. [Google Scholar] [CrossRef]
- Korotkevich, G.; Sukhov, V.; Budin, N.; Shpak, B.; Artyomov, M.N.; Sergushichev, A. Fast gene set enrichment analysis. BioRxiv 2021. [Google Scholar]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B Methodol. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Dong, J.; Horvath, S. Understanding network concepts in modules. BMC Syst. Biol. 2007, 1, 24. [Google Scholar] [CrossRef] [PubMed]
- Adepeju, M.; Langton, S.; Bannister, J. Akmedoids R package for generating directionally-homogeneous clusters of longitudinal data sets. J. Open Source Softw. 2020, 5, 2379. [Google Scholar] [CrossRef]
- Yu, G.; Wang, L.G.; Han, Y.; He, Q.Y. clusterProfiler: An R package for comparing biological themes among gene clusters. Omics J. Integr. Biol. 2012, 16, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Yu, G. R Package, Version 2019, Enrichplot: Visualization of Functional Enrichment Result. Available online: https://yulab-smu.top/biomedical-knowledge-mining-book/ (accessed on 20 May 2023).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janzen, G.M.; Dittmar, E.L.; Langlade, N.B.; Blanchet, N.; Donovan, L.A.; Temme, A.A.; Burke, J.M. Similar Transcriptomic Responses to Early and Late Drought Stresses Produce Divergent Phenotypes in Sunflower (Helianthus annuus L.). Int. J. Mol. Sci. 2023, 24, 9351. https://doi.org/10.3390/ijms24119351
Janzen GM, Dittmar EL, Langlade NB, Blanchet N, Donovan LA, Temme AA, Burke JM. Similar Transcriptomic Responses to Early and Late Drought Stresses Produce Divergent Phenotypes in Sunflower (Helianthus annuus L.). International Journal of Molecular Sciences. 2023; 24(11):9351. https://doi.org/10.3390/ijms24119351
Chicago/Turabian StyleJanzen, Garrett M., Emily L. Dittmar, Nicolas B. Langlade, Nicolas Blanchet, Lisa A. Donovan, Andries A. Temme, and John M. Burke. 2023. "Similar Transcriptomic Responses to Early and Late Drought Stresses Produce Divergent Phenotypes in Sunflower (Helianthus annuus L.)" International Journal of Molecular Sciences 24, no. 11: 9351. https://doi.org/10.3390/ijms24119351
APA StyleJanzen, G. M., Dittmar, E. L., Langlade, N. B., Blanchet, N., Donovan, L. A., Temme, A. A., & Burke, J. M. (2023). Similar Transcriptomic Responses to Early and Late Drought Stresses Produce Divergent Phenotypes in Sunflower (Helianthus annuus L.). International Journal of Molecular Sciences, 24(11), 9351. https://doi.org/10.3390/ijms24119351





