Magnetic Nanostructures and Stem Cells for Regenerative Medicine, Application in Liver Diseases
Abstract
1. Introduction
2. Bioengineering and Stem Cells
3. Stem Cells as an Alternative Treatment to Liver Diseases
4. Nanotechnology
5. Nanotechnology and TERM (Tissue Engineering and Regenerative Medicine): A Rising Treatment Field
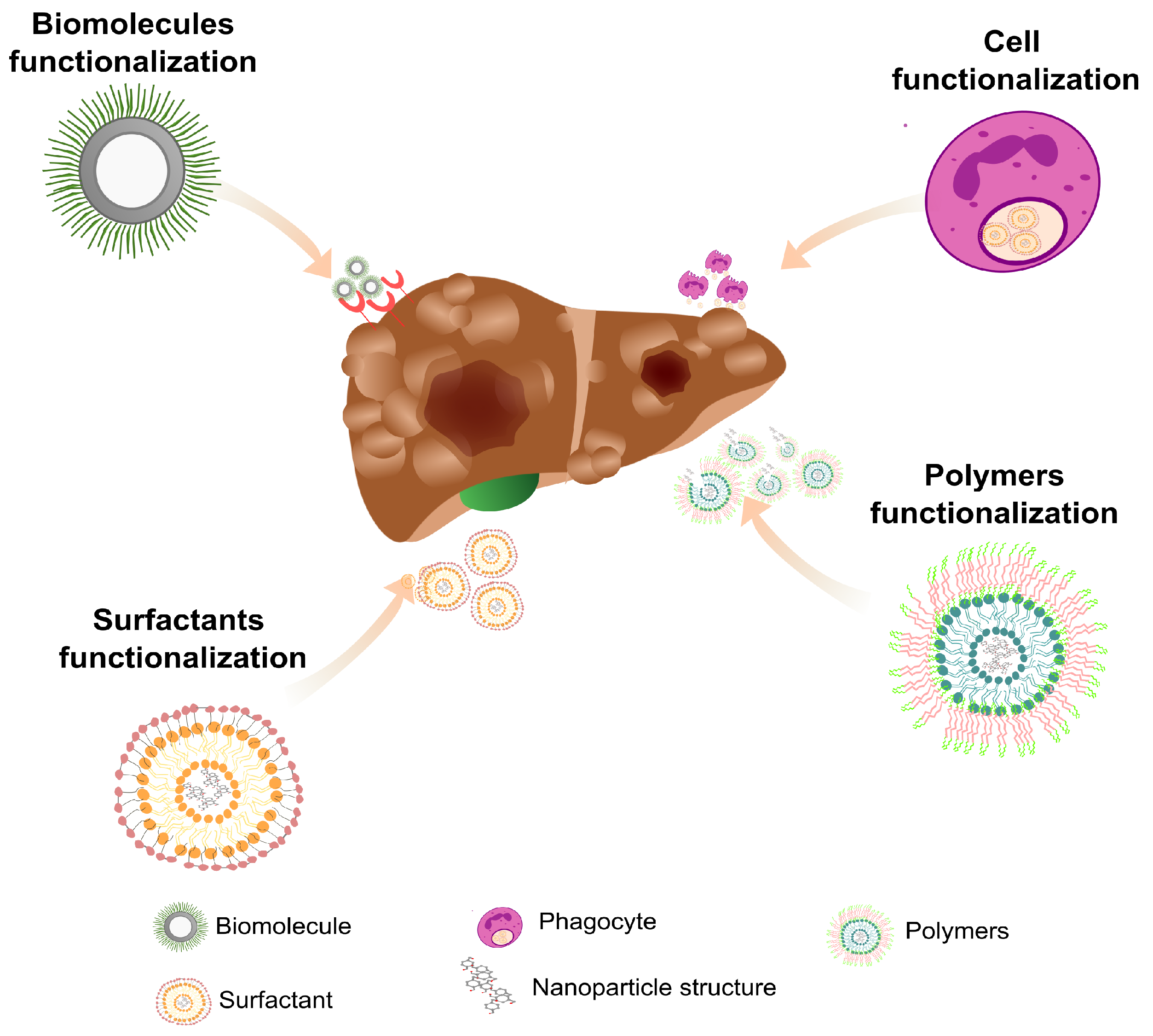
6. Mechanisms for Nanoparticles Tissue Targeting
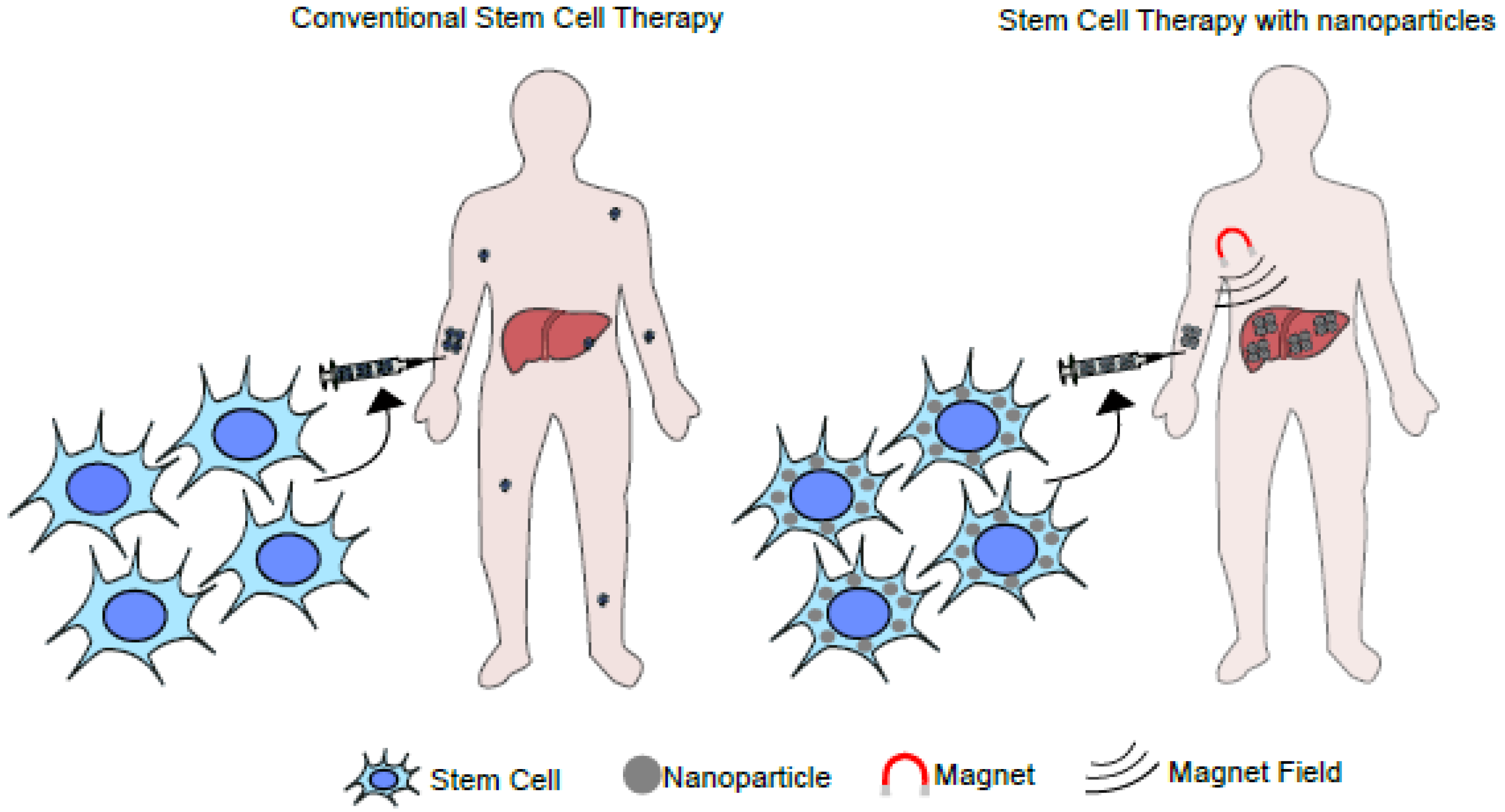
7. Applications of Stem Cells in Nanomedicine
8. Conclusions and Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, Y.; Lu, L.; Cai, X. Liver Regeneration and Cell Transplantation for End-Stage Liver Disease. Biomolecules 2021, 11, 1907. [Google Scholar] [CrossRef]
- Nicolas, C.T.; Hickey, R.D.; Chen, H.S.; Mao, S.A.; Lopera Higuita, M.; Wang, Y.; Nyberg, S.L. Concise Review: Liver Regenerative Medicine: From Hepatocyte Transplantation to Bioartificial Livers and Bioengineered Grafts. Stem Cells 2017, 35, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Lotowska, J.M.; Sobaniec-Lotowska, M.E.; Sobaniec, P. Ultrastructural Profile Combined with Immunohistochemistry of a Hepatic Progenitor Cell Line in Pediatric Autoimmune Hepatitis: New Insights into the Morphological Pattern of the Disease. Cells 2021, 10, 1899. [Google Scholar] [CrossRef]
- Tao, Y.-C.; Chen, E.-Q. Clinical Application of Stem Cell in Patients with End-Stage Liver Disease: Progress and Challenges. Ann. Transl. Med. 2020, 8, 564. [Google Scholar] [CrossRef] [PubMed]
- Doi, Y.; Sugahara, H.; Yamamoto, K.; Uji-ie, H.; Kakimoto, T.; Sakoda, H. Immune-Mediated Peripheral Neuropathy Occurring Simultaneously with Recurrent Graft-versus-Host Disease after Allogenic Hematopoietic Stem Cell Transplantation. Leuk. Res. 2012, 36, e63–e65. [Google Scholar] [CrossRef] [PubMed]
- Eggenhofer, E.; Benseler, V.; Kroemer, A.; Popp, F.C.; Geissler, E.K.; Schlitt, H.J.; Baan, C.C.; Dahlke, M.H.; Hoogduijn, M.J. Mesenchymal Stem Cells Are Short-Lived and Do Not Migrate beyond the Lungs after Intravenous Infusion. Front. Immunol. 2012, 3, 297. [Google Scholar] [CrossRef] [PubMed]
- Truong, N.H.; Nguyen, N.H.; Le, T.V.; Vu, N.B.; Huynh, N.; Nguyen, T.V.; Le, H.M.; Phan, N.K.; Pham, P.V. Comparison of the Treatment Efficiency of Bone Marrow-Derived Mesenchymal Stem Cell Transplantation via Tail and Portal Veins in CCl4-Induced Mouse Liver Fibrosis. Stem Cells Int. 2016, 2016, 5720413. [Google Scholar] [CrossRef]
- El-Ansary, M.; Abdel-Aziz, I.; Mogawer, S.; Abdel-Hamid, S.; Hammam, O.; Teaema, S.; Wahdan, M. Phase II Trial: Undifferentiated versus Differentiated Autologous Mesenchymal Stem Cells Transplantation in Egyptian Patients with HCV Induced Liver Cirrhosis. Stem Cell Rev. Rep. 2012, 8, 972–981. [Google Scholar] [CrossRef]
- Salama, H.; Zekri, A.-R.N.; Medhat, E.; Al Alim, S.A.; Ahmed, O.S.; Bahnassy, A.A.; Lotfy, M.M.; Ahmed, R.; Musa, S. Peripheral Vein Infusion of Autologous Mesenchymal Stem Cells in Egyptian HCV-Positive Patients with End-Stage Liver Disease. Stem Cell Res. Ther. 2014, 5, 70. [Google Scholar] [CrossRef]
- Deng, Q.; Cai, T.; Zhang, S.; Hu, A.; Zhang, X.; Wang, Y.; Huang, J. Autologous Peripheral Blood Stem Cell Transplantation Improves Portal Hemodynamics in Patients with Hepatitis B Virus-Related Decompensated Cirrhosis. Hepat. Mon. 2015, 15, e32498. [Google Scholar] [CrossRef]
- Mohamadnejad, M.; Namiri, M.; Bagheri, M.; Hashemi, S.M.; Ghanaati, H.; Zare Mehrjardi, N.; Kazemi Ashtiani, S.; Malekzadeh, R.; Baharvand, H. Phase 1 Human Trial of Autologous Bone Marrow-Hematopoietic Stem Cell Transplantation in Patients with Decompensated Cirrhosis. World J. Gastroenterol. 2007, 13, 3359–3363. [Google Scholar] [CrossRef] [PubMed]
- Alhaj-Suliman, S.O.; Wafa, E.I.; Salem, A.K. Engineering Nanosystems to Overcome Barriers to Cancer Diagnosis and Treatment. Adv. Drug. Deliv. Rev. 2022, 189, 114482. [Google Scholar] [CrossRef] [PubMed]
- Nagórniewicz, B.; Mardhian, D.F.; Booijink, R.; Storm, G.; Prakash, J.; Bansal, R. Engineered Relaxin as Theranostic Nanomedicine to Diagnose and Ameliorate Liver Cirrhosis. Nanomedicine 2019, 17, 106–118. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Priyadarshani, J.; Ganguly, R.; Chakraborty, S. Targeting Magnetic Nanoparticles in Physiologically Mimicking Tissue Microenvironment. ACS Appl. Mater. Interfaces 2022, 14, 31689–31701. [Google Scholar] [CrossRef]
- Choi, B.; Lee, S.-H. Nano/Micro-Assisted Regenerative Medicine. Int. J. Mol. Sci. 2018, 19, 2187. [Google Scholar] [CrossRef]
- Esfahani, S.N.; Resto Irizarry, A.M.; Xue, X.; Lee, S.B.-D.; Shao, Y.; Fu, J. Micro/Nanoengineered Technologies for Human Pluripotent Stem Cells Maintenance and Differentiation. Nano Today 2021, 41, 101310. [Google Scholar] [CrossRef]
- Gois Beghini, D.; Iwao Horita, S.; Cascabulho, C.M.; Anastácio Alves, L.; Henriques-Pons, A. Induced Pluripotent Stem Cells: Hope in the Treatment of Diseases, Including Muscular Dystrophies. Int. J. Mol. Sci. 2020, 21, 5467. [Google Scholar] [CrossRef] [PubMed]
- Beghini, D.G.; Horita, S.I.; Henriques-Pons, A. Mesenchymal Stem Cells in the Treatment of COVID-19, a Promising Future. Cells 2021, 10, 2588. [Google Scholar] [CrossRef]
- Mazini, L.; Rochette, L.; Admou, B.; Amal, S.; Malka, G. Hopes and Limits of Adipose-Derived Stem Cells (ADSCs) and Mesenchymal Stem Cells (MSCs) in Wound Healing. Int. J. Mol. Sci. 2020, 21, 1306. [Google Scholar] [CrossRef]
- Henriques-Pons, A.; Beghini, D.G.; Silva, V.D.S.; Iwao Horita, S.; da Silva, F.A.B. Pulmonary Mesenchymal Stem Cells in Mild Cases of COVID-19 Are Dedicated to Proliferation; In Severe Cases, They Control Inflammation, Make Cell Dispersion, and Tissue Regeneration. Front. Immunol. 2021, 12, 780900. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of Pluripotent Stem Cells from Mouse Embryonic and Adult Fibroblast Cultures by Defined Factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Date, S.; Sato, T. Mini-Gut Organoids: Reconstitution of the Stem Cell Niche. Annu. Rev. Cell Dev. Biol. 2015, 31, 269–289. [Google Scholar] [CrossRef] [PubMed]
- Koffler, J.; Zhu, W.; Qu, X.; Platoshyn, O.; Dulin, J.N.; Brock, J.; Graham, L.; Lu, P.; Sakamoto, J.; Marsala, M.; et al. Biomimetic 3D-Printed Scaffolds for Spinal Cord Injury Repair. Nat. Med. 2019, 25, 263–269. [Google Scholar] [CrossRef]
- Zhong, Z.; Balayan, A.; Tian, J.; Xiang, Y.; Hwang, H.H.; Wu, X.; Deng, X.; Schimelman, J.; Sun, Y.; Ma, C.; et al. Bioprinting of Dual ECM Scaffolds Encapsulating Limbal Stem/Progenitor Cells in Active and Quiescent Statuses. Biofabrication 2021, 13, 044101. [Google Scholar] [CrossRef] [PubMed]
- Michalopoulos, G.K. Liver Regeneration. J. Cell. Physiol. 2007, 213, 286–300. [Google Scholar] [CrossRef] [PubMed]
- Trefts, E.; Gannon, M.; Wasserman, D.H. The Liver. Curr. Biol. CB 2017, 27, R1147–R1151. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yang, X.; Plummer, R.; Hayashi, Y.; Deng, X.-S.; Nie, Y.-Z.; Taniguchi, H. Human Pluripotent Stem Cell-Derived Hepatocyte-Like Cells and Organoids for Liver Disease and Therapy. Int. J. Mol. Sci. 2021, 22, 10471. [Google Scholar] [CrossRef]
- Reza, H.A.; Okabe, R.; Takebe, T. Organoid Transplant Approaches for the Liver. Transpl. Int. 2021, 34, 2031–2045. [Google Scholar] [CrossRef]
- Meirelles Júnior, R.F.; Salvalaggio, P.; Rezende, M.B.D.; Evangelista, A.S.; Guardia, B.D.; Matielo, C.E.L.; Neves, D.B.; Pandullo, F.L.; Felga, G.E.G.; Alves, J.A. da S.; et al. Liver Transplantation: History, Outcomes and Perspectives. Einstein 2015, 13, 149–152. [Google Scholar] [CrossRef]
- Yu, Y.; Fisher, J.E.; Lillegard, J.B.; Rodysill, B.; Amiot, B.; Nyberg, S.L. Cell Therapies for Liver Diseases. Liver Transpl. 2012, 18, 9–21. [Google Scholar] [CrossRef]
- Amato, G.; Saleh, T.; Carpino, G.; Gaudio, E.; Alvaro, D.; Cardinale, V. Cell Therapy and Bioengineering in Experimental Liver Regenerative Medicine: In Vivo Injury Models and Grafting Strategies. Curr. Transpl. Rep. 2021, 8, 76–89. [Google Scholar] [CrossRef]
- Hu, C.; Wu, Z.; Li, L. Pre-Treatments Enhance the Therapeutic Effects of Mesenchymal Stem Cells in Liver Diseases. J. Cell. Mol. Med. 2020, 24, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.H.; Kim, M.Y.; Eom, Y.W.; Baik, S.K. Mesenchymal Stem Cells for the Treatment of Liver Disease: Present and Perspectives. Gut Liver 2020, 14, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Trounson, A.; McDonald, C. Stem Cell Therapies in Clinical Trials: Progress and Challenges. Cell Stem. Cell 2015, 17, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Zhao, L.; Zhang, L.; Bao, Q.; Li, L. Mesenchymal Stem Cell-Based Cell-Free Strategies: Safe and Effective Treatments for Liver Injury. Stem Cell Res. Ther. 2020, 11, 377. [Google Scholar] [CrossRef]
- Wang, J.; Sun, M.; Liu, W.; Li, Y.; Li, M. Stem Cell-Based Therapies for Liver Diseases: An Overview and Update. Tissue Eng. Regen. Med. 2019, 16, 107–118. [Google Scholar] [CrossRef]
- Jia, Y.; Shu, X.; Yang, X.; Sun, H.; Cao, H.; Cao, H.; Zhang, K.; Xu, Q.; Li, G.; Yang, Y. Enhanced Therapeutic Effects of Umbilical Cord Mesenchymal Stem Cells after Prolonged Treatment for HBV-Related Liver Failure and Liver Cirrhosis. Stem Cell Res. Ther. 2020, 11, 277. [Google Scholar] [CrossRef]
- Wang, L.; Li, J.; Liu, H.; Li, Y.; Fu, J.; Sun, Y.; Xu, R.; Lin, H.; Wang, S.; Lv, S.; et al. A Pilot Study of Umbilical Cord-Derived Mesenchymal Stem Cell Transfusion in Patients with Primary Biliary Cirrhosis. J. Gastroenterol. Hepatol. 2013, 28, 85–92. [Google Scholar] [CrossRef]
- Li, T.-T.; Wang, Z.-R.; Yao, W.-Q.; Linghu, E.-Q.; Wang, F.-S.; Shi, L. Stem Cell Therapies for Chronic Liver Diseases: Progress and Challenges. Stem Cells Transl. Med. 2022, 11, 900–911. [Google Scholar] [CrossRef]
- Hofmann, J.; Hackl, V.; Esser, H.; Meszaros, A.T.; Fodor, M.; Öfner, D.; Troppmair, J.; Schneeberger, S.; Hautz, T. Cell-Based Regeneration and Treatment of Liver Diseases. Int. J. Mol. Sci. 2021, 22, 10276. [Google Scholar] [CrossRef]
- Mansoori, G.A.; Soelaiman, T.F. Nanotechnology—An Introduction for the Standards Community. J. ASTM Int. 2005, 2, 1–22. [Google Scholar] [CrossRef]
- Jr, C.P.P.; Owens, F.J. Introduction to Nanotechnology; John Wiley & Sons: Hoboken, NJ, USA, 2003; ISBN 978-0-471-07935-4. [Google Scholar]
- Sm, M.; Ac, H.; Jc, M. Nanomedicine: Current Status and Future Prospects. FASEB J. 2005, 19, 311–330. [Google Scholar] [CrossRef]
- Chaturvedi, V.K.; Singh, A.; Singh, V.K.; Singh, M.P. Cancer Nanotechnology: A New Revolution for Cancer Diagnosis and Therapy. Curr. Drug. Metab. 2019, 20, 416–429. [Google Scholar] [CrossRef] [PubMed]
- Misra, R.; Acharya, S.; Sahoo, S.K. Cancer Nanotechnology: Application of Nanotechnology in Cancer Therapy. Drug. Discov. Today 2010, 15, 842–850. [Google Scholar] [CrossRef]
- Haeri, A.; Osouli, M.; Bayat, F.; Alavi, S.; Dadashzadeh, S. Nanomedicine Approaches for Sirolimus Delivery: A Review of Pharmaceutical Properties and Preclinical Studies. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Roy, I.; Yang, C.; Prasad, P.N. Nanochemistry and Nanomedicine for Nanoparticle-Based Diagnostics and Therapy. Chem. Rev. 2016, 116, 2826–2885. [Google Scholar] [CrossRef] [PubMed]
- Lan, J. Overview of Application of Nanomaterials in Medical Domain. Contrast Media Mol. Imaging 2022, 2022, 3507383. [Google Scholar] [CrossRef]
- Lee, H.; Park, S.Y.; Lee, S.; Choi, S.; Seo, S.; Kim, H.; Won, J.; Choi, K.; Kang, K.; Park, H.; et al. Black Phosphorus (BP) Nanodots for Potential Biomedical Applications. Small 2015, 12, 214–219. [Google Scholar] [CrossRef]
- Warner, J.H.; Hoshino, A.; Yamamoto, K.; Tilley, R.D. Water-Soluble Photoluminescent Silicon Quantum Dots. Angew. Chem. Int. Ed. 2005, 44, 4550–4554. [Google Scholar] [CrossRef]
- Yang, D.; Yang, G.; Yang, P.; Lv, R.; Gai, S.; Li, C.; He, F.; Lin, J. Assembly of Au Plasmonic Photothermal Agent and Iron Oxide Nanoparticles on Ultrathin Black Phosphorus for Targeted Photothermal and Photodynamic Cancer Therapy. Adv. Funct. Mater. 2017, 27, 1700371. [Google Scholar] [CrossRef]
- Sun, C.; Wen, L.; Zeng, J.; Wang, Y.; Sun, Q.; Deng, L.; Zhao, C.; Li, Z. One-Pot Solventless Preparation of PEGylated Black Phosphorus Nanoparticles for Photoacoustic Imaging and Photothermal Therapy of Cancer. Biomaterials 2016, 91, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Kim, D.; Wang, J.; Han, Y.; Zuidema, J.M.; Hariri, A.; Park, J.-H.; Jokerst, J.V.; Sailor, M.J. Enhanced Performance of a Molecular Photoacoustic Imaging Agent by Encapsulation in Mesoporous Silicon Nanoparticles. Adv. Mater. 2018, 30, e1800512. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, J.; Li, W.; Chen, R.; Zhang, Z.; Zhang, W.; Tang, Y.; Chen, X.; Liu, G.; Lee, C.-S. Degradable Hollow Mesoporous Silicon/Carbon Nanoparticles for Photoacoustic Imaging-Guided Highly Effective Chemo-Thermal Tumor Therapy in Vitro and in Vivo. Theranostics 2017, 7, 3007–3020. [Google Scholar] [CrossRef]
- Virlan, M.J.R.; Miricescu, D.; Radulescu, R.; Sabliov, C.M.; Totan, A.; Calenic, B.; Greabu, M. Organic Nanomaterials and Their Applications in the Treatment of Oral Diseases. Molecules 2016, 21, 207. [Google Scholar] [CrossRef] [PubMed]
- Qiu, M.; Singh, A.; Wang, D.; Qu, J.; Swihart, M.; Zhang, H.; Prasad, P.N. Biocompatible and Biodegradable Inorganic Nanostructures for Nanomedicine: Silicon and Black Phosphorus. Nano Today 2019, 25, 135–155. [Google Scholar] [CrossRef]
- Meng, H.; Chen, Z.; Xing, G.; Yuan, H.; Chen, C.; Zhao, F.; Zhang, C.; Zhao, Y. Ultrahigh Reactivity Provokes Nanotoxicity: Explanation of Oral Toxicity of Nano-Copper Particles. Toxicol. Lett. 2007, 175, 102–110. [Google Scholar] [CrossRef]
- Hirt, N.; Body-Malapel, M. Immunotoxicity and Intestinal Effects of Nano- and Microplastics: A Review of the Literature. Part. Fibre Toxicol. 2020, 17, 57. [Google Scholar] [CrossRef]
- Bostan, H.B.; Rezaee, R.; Valokala, M.G.; Tsarouhas, K.; Golokhvast, K.; Tsatsakis, A.M.; Karimi, G. Cardiotoxicity of Nano-Particles. Life Sci. 2016, 165, 91–99. [Google Scholar] [CrossRef]
- Hesler, M.; Aengenheister, L.; Ellinger, B.; Drexel, R.; Straskraba, S.; Jost, C.; Wagner, S.; Meier, F.; von Briesen, H.; Büchel, C.; et al. Multi-Endpoint Toxicological Assessment of Polystyrene Nano- and Microparticles in Different Biological Models in Vitro. Toxicol. Vitr. 2019, 61, 104610. [Google Scholar] [CrossRef]
- Gatoo, M.A.; Naseem, S.; Arfat, M.Y.; Dar, A.M.; Qasim, K.; Zubair, S. Physicochemical Properties of Nanomaterials: Implication in Associated Toxic Manifestations. Biomed. Res. Int. 2014, 2014, 498420. [Google Scholar] [CrossRef]
- Hardman, R. A Toxicologic Review of Quantum Dots: Toxicity Depends on Physicochemical and Environmental Factors. Environ. Health Perspect. 2006, 114, 165–172. [Google Scholar] [CrossRef]
- Aggarwal, P.; Hall, J.B.; McLeland, C.B.; Dobrovolskaia, M.A.; McNeil, S.E. Nanoparticle Interaction with Plasma Proteins as It Relates to Particle Biodistribution, Biocompatibility and Therapeutic Efficacy. Adv. Drug. Deliv. Rev. 2009, 61, 428–437. [Google Scholar] [CrossRef]
- Mason, C.; Brindley, D.A.; Culme-Seymour, E.J.; Davie, N.L. Cell Therapy Industry: Billion Dollar Global Business with Unlimited Potential. Regen. Med. 2011, 6, 265–272. [Google Scholar] [CrossRef]
- Au, P.; Hursh, D.A.; Lim, A.; Moos, M.C.; Oh, S.S.; Schneider, B.S.; Witten, C.M. FDA Oversight of Cell Therapy Clinical Trials. Sci. Transl. Med. 2012, 4, 149fs31. [Google Scholar] [CrossRef]
- Alzate-Correa, D.; Lawrence, W.R.; Salazar-Puerta, A.; Higuita-Castro, N.; Gallego-Perez, D. Nanotechnology-Driven Cell-Based Therapies in Regenerative Medicine. AAPS J. 2022, 24, 43. [Google Scholar] [CrossRef]
- Bayda, S.; Adeel, M.; Tuccinardi, T.; Cordani, M.; Rizzolio, F. The History of Nanoscience and Nanotechnology: From Chemical–Physical Applications to Nanomedicine. Molecules 2020, 25, 112. [Google Scholar] [CrossRef]
- Mitchell, M.J.; Billingsley, M.M.; Haley, R.M.; Wechsler, M.E.; Peppas, N.A.; Langer, R. Engineering Precision Nanoparticles for Drug Delivery. Nat. Rev. Drug Discov. 2021, 20, 101–124. [Google Scholar] [CrossRef]
- Suh, W.H.; Suslick, K.S.; Stucky, G.D.; Suh, Y.-H. Nanotechnology, Nanotoxicology, and Neuroscience. Prog. Neurobiol. 2009, 87, 133–170. [Google Scholar] [CrossRef]
- Ranganathan, R.; Madanmohan, S.; Kesavan, A.; Baskar, G.; Krishnamoorthy, Y.R.; Santosham, R.; Ponraju, D.; Rayala, S.K.; Venkatraman, G. Nanomedicine: Towards Development of Patient-Friendly Drug-Delivery Systems for Oncological Applications. Int. J. Nanomed. 2012, 7, 1043–1060. [Google Scholar] [CrossRef]
- Huang, Y.; Gao, X.; Chen, J. Leukocyte-Derived Biomimetic Nanoparticulate Drug Delivery Systems for Cancer Therapy. Acta Pharm. Sin. B 2018, 8, 4–13. [Google Scholar] [CrossRef]
- Xuan, M.; Shao, J.; Dai, L.; He, Q.; Li, J. Macrophage Cell Membrane Camouflaged Mesoporous Silica Nanocapsules for In Vivo Cancer Therapy. Adv. Healthc. Mater. 2015, 4, 1645–1652. [Google Scholar] [CrossRef]
- Zinger, A.; Sushnitha, M.; Naoi, T.; Baudo, G.; De Rosa, E.; Chang, J.; Tasciotti, E.; Taraballi, F. Enhancing Inflammation Targeting Using Tunable Leukocyte-Based Biomimetic Nanoparticles. ACS Nano 2021, 15, 6326–6339. [Google Scholar] [CrossRef]
- Pierigè, F.; Serafini, S.; Rossi, L.; Magnani, M. Cell-Based Drug Delivery. Adv. Drug Deliv. Rev. 2008, 60, 286–295. [Google Scholar] [CrossRef]
- Terracciano, R.; Carcamo-Bahena, Y.; Royal, A.L.R.; Messina, L.; Delk, J.; Butler, E.B.; Demarchi, D.; Grattoni, A.; Wang, Z.; Cristini, V.; et al. Zonal Intratumoral Delivery of Nanoparticles Guided by Surface Functionalization. Langmuir 2022, 38, 13983–13994. [Google Scholar] [CrossRef]
- Silva, R.C. Da Avaliação da Eficiência Fotodinâmica de Fotossensibilizadores com Aplicação em Terapia Fotodinâmica. Master’s Thesis, Universidade de São Paulo, São Paulo, Brazil, 2007. [Google Scholar]
- Shi, H.; Sadler, P.J. How Promising Is Phototherapy for Cancer? Br. J. Cancer 2020, 123, 871–873. [Google Scholar] [CrossRef]
- Yi, G.; Son, J.; Yoo, J.; Park, C.; Koo, H. Emulsan-Based Nanoparticles for in Vivo Drug Delivery to Tumors. Biochem. Biophys. Res. Commun. 2019, 508, 326–331. [Google Scholar] [CrossRef]
- Schlachet, I.; Moshe Halamish, H.; Sosnik, A. Mixed Amphiphilic Polymeric Nanoparticles of Chitosan, Poly(Vinyl Alcohol) and Poly(Methyl Methacrylate) for Intranasal Drug Delivery: A Preliminary In Vivo Study. Molecules 2020, 25, 4496. [Google Scholar] [CrossRef]
- Chakraborty, I.; Aberasturi, D.J.D.; Pazos-Perez, N.; Guerrini, L.; Masood, A.; Alvarez-Puebla, R.A.; Feliu, N.; Parak, W.J. Ion-Selective Ligands: How Colloidal Nano- and Micro-Particles Can Introduce New Functionalities. Z. Für Phys. Chem. 2018, 232, 1307–1317. [Google Scholar] [CrossRef]
- Tietjen, G.T.; Bracaglia, L.G.; Saltzman, W.M.; Pober, J.S. Focus on Fundamentals: Achieving Effective Nanoparticle Targeting. Trends Mol. Med. 2018, 24, 598–606. [Google Scholar] [CrossRef]
- Jain, K.K. Nanoparticles as Targeting Ligands. Trends Biotechnol. 2006, 24, 143–145. [Google Scholar] [CrossRef]
- Ishiguro, K.; Yan, I.K.; Lewis-Tuffin, L.; Patel, T. Targeting Liver Cancer Stem Cells Using Engineered Biological Nanoparticles for the Treatment of Hepatocellular Cancer. Hepatol. Commun. 2020, 4, 298–313. [Google Scholar] [CrossRef]
- Zhao, J.; Vykoukal, J.; Abdelsalam, M.; Recio-Boiles, A.; Huang, Q.; Qiao, Y.; Singhana, B.; Wallace, M.; Avritscher, R.; Melancon, M.P. Stem Cell-Mediated Delivery of SPIO-Loaded Gold Nanoparticles for the Theranosis of Liver Injury and Hepatocellular Carcinoma. Nanotechnology 2014, 25, 405101. [Google Scholar] [CrossRef]
- Foroutan, T.; Kassaee, M.Z.; Salari, M.; Ahmady, F.; Molavi, F.; Moayer, F. Magnetic Fe3O4@graphene Oxide Improves the Therapeutic Effects of Embryonic Stem Cells on Acute Liver Damage. Cell Prolif. 2021, 54, e13126. [Google Scholar] [CrossRef]
- Wang, Y.-H.; Wu, D.-B.; Chen, B.; Chen, E.-Q.; Tang, H. Progress in Mesenchymal Stem Cell–Based Therapy for Acute Liver Failure. Stem Cell Res. Ther. 2018, 9, 227. [Google Scholar] [CrossRef]
- Foroutan, T.; Kabiri, F.; Motamedi, E. Silica Magnetic Graphene Oxide Improves the Effects of Stem Cell-Conditioned Medium on Acute Liver Failure. ACS Omega 2021, 6, 21194–21206. [Google Scholar] [CrossRef]
- Abd Ellah, N.H.; Abouelmagd, S.A. Surface Functionalization of Polymeric Nanoparticles for Tumor Drug Delivery: Approaches and Challenges. Expert. Opin. Drug Deliv. 2017, 14, 201–214. [Google Scholar] [CrossRef]
- Qi, J.; Yu, Z.L.; Liao, G.P.; Luo, Z.Y.; Bai, B.F. Effect of Nanoparticle Surfactants on Droplet Formation in a Flow-Focusing Microchannel. Phys. Fluids 2021, 33, 112008. [Google Scholar] [CrossRef]
- Cheng, Q.; Wei, T.; Jia, Y.; Farbiak, L.; Zhou, K.; Zhang, S.; Wei, Y.; Zhu, H.; Siegwart, D.J. Dendrimer-Based Lipid Nanoparticles Deliver Therapeutic FAH MRNA to Normalize Liver Function and Extend Survival in a Mouse Model of Hepatorenal Tyrosinemia Type I. Adv. Mater. 2018, 30, 1805308. [Google Scholar] [CrossRef]
- Petros, R.A.; DeSimone, J.M. Strategies in the Design of Nanoparticles for Therapeutic Applications. Nat. Rev. Drug Discov. 2010, 9, 615–627. [Google Scholar] [CrossRef]
- Thanh, N.T.K.; Green, L.A.W. Functionalisation of Nanoparticles for Biomedical Applications. Nano Today 2010, 5, 213–230. [Google Scholar] [CrossRef]
- Fang, J.; Nakamura, H.; Maeda, H. The EPR Effect: Unique Features of Tumor Blood Vessels for Drug Delivery, Factors Involved, and Limitations and Augmentation of the Effect. Adv. Drug Deliv. Rev. 2011, 63, 136–151. [Google Scholar] [CrossRef]
- Wu, J. The Enhanced Permeability and Retention (EPR) Effect: The Significance of the Concept and Methods to Enhance Its Application. J. Pers. Med. 2021, 11, 771. [Google Scholar] [CrossRef]
- Pérez-Herrero, E.; Fernández-Medarde, A. Advanced Targeted Therapies in Cancer: Drug Nanocarriers, the Future of Chemotherapy. Eur. J. Pharm. Biopharm. 2015, 93, 52–79. [Google Scholar] [CrossRef]
- Peer, D.; Karp, J.M.; Hong, S.; Farokhzad, O.C.; Margalit, R.; Langer, R. Nanocarriers as an Emerging Platform for Cancer Therapy. Nat. Nanotech. 2007, 2, 751–760. [Google Scholar] [CrossRef]
- Chorny, M.; Fishbein, I.; Forbes, S.; Alferiev, I. Magnetic Nanoparticles for Targeted Vascular Delivery. IUBMB Life 2011, 63, 613–620. [Google Scholar] [CrossRef]
- McBain, S.C.; Yiu, H.H.P.; Dobson, J. Magnetic Nanoparticles for Gene and Drug Delivery. Int. J. Nanomed. 2008, 3, 169–180. [Google Scholar] [CrossRef]
- Gokduman, K.; Gok, A. In Vitro Investigation of Therapeutic Potential of Bare Magnetite (Fe₃O₄) Nanoparticles (≤100 Ppm) on Hepatocellular Carcinoma Cells. J. Nanosci. Nanotechnol. 2020, 20, 1391–1400. [Google Scholar] [CrossRef]
- Liang, X.; Ding, Y.; Zhang, Y.; Tse, H.-F.; Lian, Q. Paracrine Mechanisms of Mesenchymal Stem Cell-Based Therapy: Current Status and Perspectives. Cell Transplant. 2014, 23, 1045–1059. [Google Scholar] [CrossRef]
- Bulte, J.W.M. Intracellular Endosomal Magnetic Labeling of Cells. Methods Mol. Med. 2006, 124, 419–439. [Google Scholar] [CrossRef]
- Walkey, C.D.; Chan, W.C.W. Understanding and Controlling the Interaction of Nanomaterials with Proteins in a Physiological Environment. Chem. Soc. Rev. 2012, 41, 2780–2799. [Google Scholar] [CrossRef]
- Roohi, F.; Lohrke, J.; Ide, A.; Schütz, G.; Dassler, K. Studying the Effect of Particle Size and Coating Type on the Blood Kinetics of Superparamagnetic Iron Oxide Nanoparticles. IJN 2012, 7, 4447–4458. [Google Scholar] [CrossRef]
- Moros, M.; Hernáez, B.; Garet, E.; Dias, J.T.; Sáez, B.; Grazú, V.; González-Fernández, A.; Alonso, C.; de la Fuente, J.M. Monosaccharides versus PEG-Functionalized NPs: Influence in the Cellular Uptake. ACS Nano 2012, 6, 1565–1577. [Google Scholar] [CrossRef]
- Fadeel, B.; Feliu, N.; Vogt, C.; Abdelmonem, A.M.; Parak, W.J. Bridge over Troubled Waters: Understanding the Synthetic and Biological Identities of Engineered Nanomaterials. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2013, 5, 111–129. [Google Scholar] [CrossRef]
- Chiu, C.-Y.; Chung, T.-W.; Chen, S.-Y.; Ma, Y.-H. Effects of PEGylation on Capture of Dextran-Coated Magnetic Nanoparticles in Microcirculation. Int. J. Nanomed. 2019, 14, 4767–4780. [Google Scholar] [CrossRef]
- Soares, G.A.; Faria, J.V.C.; Pinto, L.A.; Prospero, A.G.; Pereira, G.M.; Stoppa, E.G.; Buranello, L.P.; Bakuzis, A.F.; Baffa, O.; Miranda, J.R.A. Long-Term Clearance and Biodistribution of Magnetic Nanoparticles Assessed by AC Biosusceptometry. Materials 2022, 15, 2121. [Google Scholar] [CrossRef]
- Battaglia, A.M.; Chirillo, R.; Aversa, I.; Sacco, A.; Costanzo, F.; Biamonte, F. Ferroptosis and Cancer: Mitochondria Meet the “Iron Maiden” Cell Death. Cells 2020, 9, 1505. [Google Scholar] [CrossRef]
- Li, J.; Cao, F.; Yin, H.-L.; Huang, Z.-J.; Lin, Z.-T.; Mao, N.; Sun, B.; Wang, G. Ferroptosis: Past, Present and Future. Cell Death Dis. 2020, 11, 88. [Google Scholar] [CrossRef]
- He, J.; Yang, X.; Men, B.; Wang, D. Interfacial Mechanisms of Heterogeneous Fenton Reactions Catalyzed by Iron-Based Materials: A Review. J. Environ. Sci. 2016, 39, 97–109. [Google Scholar] [CrossRef]
- Jiang, X.; Stockwell, B.R.; Conrad, M. Ferroptosis: Mechanisms, Biology and Role in Disease. Nat. Rev. Mol. Cell Biol. 2021, 22, 266–282. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, Y.; Zhang, R.; Wang, F.; Wang, T.; Jiao, Y. The Role of Erastin in Ferroptosis and Its Prospects in Cancer Therapy. Onco Targets Ther. 2020, 13, 5429–5441. [Google Scholar] [CrossRef]
- Sui, X.; Zhang, R.; Liu, S.; Duan, T.; Zhai, L.; Zhang, M.; Han, X.; Xiang, Y.; Huang, X.; Lin, H.; et al. RSL3 Drives Ferroptosis Through GPX4 Inactivation and ROS Production in Colorectal Cancer. Front. Pharmacol. 2018, 9, 1371. [Google Scholar] [CrossRef]
- Jiang, Q.; Wang, K.; Zhang, X.; Ouyang, B.; Liu, H.; Pang, Z.; Yang, W. Platelet Membrane-Camouflaged Magnetic Nanoparticles for Ferroptosis-Enhanced Cancer Immunotherapy. Small 2020, 16, e2001704. [Google Scholar] [CrossRef]
- Chen, M.; Li, J.; Shu, G.; Shen, L.; Qiao, E.; Zhang, N.; Fang, S.; Chen, X.; Zhao, Z.; Tu, J.; et al. Homogenous Multifunctional Microspheres Induce Ferroptosis to Promote the Anti-Hepatocarcinoma Effect of Chemoembolization. J. Nanobiotechnol. 2022, 20, 179. [Google Scholar] [CrossRef]
- Chi, H.; Zhu, G.; Yin, Y.; Diao, H.; Liu, Z.; Sun, S.; Guo, Z.; Xu, W.; Xu, J.; Cui, C.; et al. Dual-Responsive Multifunctional “Core-Shell” Magnetic Nanoparticles Promoting Fenton Reaction for Tumor Ferroptosis Therapy. Int. J. Pharm. 2022, 622, 121898. [Google Scholar] [CrossRef]
- Zhou, Z.; Yang, L.; Gao, J.; Chen, X. Structure-Relaxivity Relationships of Magnetic Nanoparticles for Magnetic Resonance Imaging. Adv. Mater. 2019, 31, e1804567. [Google Scholar] [CrossRef]
- Ramos Rubio, E.; Llado Garriga, L. Usefulness of pre-surgical biopsy in selecting patients with hepatocellular carcinoma for liver transplant. Cir. Esp. 2010, 87, 133–138. [Google Scholar] [CrossRef]
- Zhao, H.Y.; Liu, S.; He, J.; Pan, C.C.; Li, H.; Zhou, Z.Y.; Ding, Y.; Huo, D.; Hu, Y. Synthesis and Application of Strawberry-like Fe3O4-Au Nanoparticles as CT-MR Dual-Modality Contrast Agents in Accurate Detection of the Progressive Liver Disease. Biomaterials 2015, 51, 194–207. [Google Scholar] [CrossRef]
- Maeng, J.H.; Lee, D.-H.; Jung, K.H.; Bae, Y.-H.; Park, I.-S.; Jeong, S.; Jeon, Y.-S.; Shim, C.-K.; Kim, W.; Kim, J.; et al. Multifunctional Doxorubicin Loaded Superparamagnetic Iron Oxide Nanoparticles for Chemotherapy and Magnetic Resonance Imaging in Liver Cancer. Biomaterials 2010, 31, 4995–5006. [Google Scholar] [CrossRef]
- Mohammadi, M.; Pourseyed Aghaei, F. Magnetite Fe3O4 Surface as an Effective Drug Delivery System for Cancer Treatment Drugs: Density Functional Theory Study. J. Biomol. Struct. Dyn. 2021, 39, 2798–2805. [Google Scholar] [CrossRef]
- Kawashita, M.; Tanaka, M.; Kokubo, T.; Inoue, Y.; Yao, T.; Hamada, S.; Shinjo, T. Preparation of Ferrimagnetic Magnetite Microspheres for in Situ Hyperthermic Treatment of Cancer. Biomaterials 2005, 26, 2231–2238. [Google Scholar] [CrossRef]
- Parchur, A.K.; Ansari, A.A.; Singh, B.P.; Hasan, T.N.; Syed, N.A.; Rai, S.B.; Ningthoujam, R.S. Enhanced Luminescence of CaMoO₄:Eu by Core@shell Formation and Its Hyperthermia Study after Hybrid Formation with Fe₃O₄: Cytotoxicity Assessment on Human Liver Cancer Cells and Mesenchymal Stem Cells. Integr. Biol. 2014, 6, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wei, X.; Tao, F.; Deng, C.; Lv, C.; Chen, C.; Cheng, Y. The Potential Application of Nanomaterials for Ferroptosis-Based Cancer Therapy. Biomed. Mater. 2021, 16, 042013. [Google Scholar] [CrossRef] [PubMed]
- Stanger, B.Z. Cellular Homeostasis and Repair in the Mammalian Liver. Annu. Rev. Physiol. 2015, 77, 179–200. [Google Scholar] [CrossRef] [PubMed]
- Mazzeo, A.; Santos, E.J.C. Nanotechnology and Multipotent Adult Progenitor Cells in Reparative Medicine: Therapeutic Perspectives. Einstein 2018, 16, eRB4587. [Google Scholar] [CrossRef]
- Kargozar, S.; Mozafari, M.; Hashemian, S.J.; Brouki Milan, P.; Hamzehlou, S.; Soleimani, M.; Joghataei, M.T.; Gholipourmalekabadi, M.; Korourian, A.; Mousavizadeh, K.; et al. Osteogenic Potential of Stem Cells-Seeded Bioactive Nanocomposite Scaffolds: A Comparative Study between Human Mesenchymal Stem Cells Derived from Bone, Umbilical Cord Wharton’s Jelly, and Adipose Tissue. J. Biomed. Mater. Res. Part B Appl. Biomater. 2018, 106, 61–72. [Google Scholar] [CrossRef]
- Prabhakaran, M.P.; Venugopal, J.R.; Ramakrishna, S. Mesenchymal Stem Cell Differentiation to Neuronal Cells on Electrospun Nanofibrous Substrates for Nerve Tissue Engineering. Biomaterials 2009, 30, 4996–5003. [Google Scholar] [CrossRef]
- Jia, Y.; Zhang, P.; Sun, Y.; Kang, Q.; Xu, J.; Zhang, C.; Chai, Y. Regeneration of Large Bone Defects Using Mesoporous Silica Coated Magnetic Nanoparticles during Distraction Osteogenesis. Nanomed. Nanotechnol. Biol. Med. 2019, 21, 102040. [Google Scholar] [CrossRef]
- Landázuri, N.; Tong, S.; Suo, J.; Joseph, G.; Weiss, D.; Sutcliffe, D.J.; Giddens, D.P.; Bao, G.; Taylor, W.R. Magnetic Targeting of Human Mesenchymal Stem Cells with Internalized Superparamagnetic Iron Oxide Nanoparticles. Small 2013, 9, 4017–4026. [Google Scholar] [CrossRef]
- Vandergriff, A.C.; Hensley, T.M.; Henry, E.T.; Shen, D.; Anthony, S.; Zhang, J.; Cheng, K. Magnetic Targeting of Cardiosphere-Derived Stem Cells with Ferumoxytol Nanoparticles for Treating Rats with Myocardial Infarction. Biomaterials 2014, 35, 8528–8539. [Google Scholar] [CrossRef]
- Zhou, B.; Shan, H.; Li, D.; Jiang, Z.-B.; Qian, J.-S.; Zhu, K.-S.; Huang, M.-S.; Meng, X.-C. MR Tracking of Magnetically Labeled Mesenchymal Stem Cells in Rats with Liver Fibrosis. Magn. Reson. Imaging 2010, 28, 394–399. [Google Scholar] [CrossRef]
- Cai, J.; Zhang, X.; Wang, X.; Li, C.; Liu, G. In Vivo MR Imaging of Magnetically Labeled Mesenchymal Stem Cells Transplanted into Rat Liver through Hepatic Arterial Injection. Contrast Media Mol. Imaging 2008, 3, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhao, J.; Jiang, J.; Chen, F.; Fang, X. Doxorubicin Delivered Using Nanoparticles Camouflaged with Mesenchymal Stem Cell Membranes to Treat Colon Cancer. Int. J. Nanomed. 2020, 15, 2873–2884. [Google Scholar] [CrossRef]
- Wu, H.-H.; Zhou, Y.; Tabata, Y.; Gao, J.-Q. Mesenchymal Stem Cell-Based Drug Delivery Strategy: From Cells to Biomimetic. J. Control. Release 2019, 294, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Thanuja, M.Y.; Anupama, C.; Ranganath, S.H. Bioengineered Cellular and Cell Membrane-Derived Vehicles for Actively Targeted Drug Delivery: So near and yet so Far. Adv. Drug Deliv. Rev. 2018, 132, 57–80. [Google Scholar] [CrossRef] [PubMed]

| Identifier at ClinicalTrials.gov | Liver Condition | Stem Cell Type * | Study Phase | Enrolment/ Estimated Enrolment | Status | Administration Route and Cell Dose |
|---|---|---|---|---|---|---|
| NCT03109236 | Cirrhosis | Autologous EPC CD133+ from BM | Phase 3 | 66 participants | Recruiting | 5–10 × 106 CD133 cells through the transhepatic route into the portal venous circulation. |
| NCT05331872 | Cirrhosis | UC-MSCs | Phase 1 | 20 participants | Recruiting | Route not informed. Cell dosage not informed. |
| NCT05227846 | Cirrhosis | UC-MSCs | Phase 1 | 9 participants | Recruiting | Cell dosage not informed. |
| NCT03945487 | Cirrhosis | UC-MSCs | Phase 2 | 200 participants | Recruiting | Intravenous administration of 1.0 × 106 cell/kg three times at three-week intervals. |
| NCT05121870 | Cirrhosis | UC-MSCs | Phase 2 | 240 participants | Recruiting | Intravenous administration of three doses (6.0 × 107 cells per event) at weeks 0, 4, and 8. |
| NCT03626090 | Cirrhosis | Autologous BM-MSCs | Phase 1/2 | 20 participants | Recruiting | A single dose of 0.5 to 1 × 106 cells/kg via peripheral venous access. |
| NCT03254758 | Cirrhosis | AD-MSCs | Phase 1/2 | 27 participants | Recruiting | Intravenous infusion; for phase 1, the cell dose escalated from low to mid and high; for phase 2, the recommended amount of cells was administered once a week for four weeks in the same route and time as in phase 1. Cell dosage not informed. |
| NCT05155657 | Alcoholic cirrhosis | UC-MSCs | Phase 1 | 36 participants | Recruiting | 0.5 × 106 cells/kg, 1.0 × 106 cells/kg, or 2.0 × 106 cells/kg via intravenous infusion. |
| NCT04689152 | Alcoholic cirrhosis | Autologous BM MSC | Phase 3 | 200 participants | Recruiting | 7 × 107 cells via the hepatic artery. |
| NCT03826433 | Cirrhosis due to hepatitis B | UC-MSCs | Phase 1 | 20 participants | Recruiting | 6 × 107 cells via peripheral intravenous injection. |
| NCT05507762 | Cirrhosis due to hepatitis B (compensation stage) | UC-MSCs | Phase 1/2 | 20 participants | Recruiting | 1 × 106/kg/time per injection via intravenous infusion in the elbow. |
| NCT05106972 | Cirrhosis due to hepatitis B | UC-MSCs | NT | 30 participants | Recruiting | 1 × 108 cells/dose via intravenous infusion. |
| NCT00655707 | Liver disease | Autologous expanded CD34+ HCSs | Phase 1/2 | 5 participants | Completed | 1 × 109,1 × 1010, 2 × 1010, or 5 × 1010 cells via either the hepatic artery or the portal vein. |
| NCT00420134 | Liver failure /cirrhosis | Autologous MSCs | Phase 1 Phase 2 | 30 participants | Completed | The cells were administered via the portal vein. Cell dosage not informed. |
| NCT00147043 | Cirrhosis | Autologous adult stem cells | Not Applicable | 5 participants | Completed | The cells were administered via the hepatic artery or portal vein. Cell dosage not informed. |
| NCT04243681 | Cirrhosis | Autologous CD34+ HSCs and MSCs | Phase 4 | 5 participants | Completed | The cells were administered via the hepatic artery. Cell dosage not informed. |
| NCT00713934 | Cirrhosis | Autologous BM-MNCs and enriched CD133+ HSCs | Phase 1/2 | 7 participants | Completed | The cells were administered via the portal vein. Cell dosage not informed. |
| NCT02297867 | Cirrhosis | ADSCs | Phase 1 | 6 participants | Completed | One milliliter of cell suspension via intrahepatic injection. |
| NCT03632148 | Cirrhosis | MSCs | Not applicable | 9 participants | Completed | Route not informed. Cell dosage not informed. |
| NCT01342250 | Cirrhosis | UC-MSCs | Phase 1 Phase 2 | 20 participants | Completed | The cells were administered at low, medium, or high doses. Route not informed. Cell dosage not informed. |
| NCT01333228 | Cirrhosis | Autologous BM-derived endothelial progenitor cells | Phase 1/2 | 14 participants | Completed | The cells were administered via the hepatic artery. Cell dosage not informed. |
| NCT01013194 | Cirrhosis | Human fetal liver cell | Phase 1/2 | 25 participants | Completed | 5 or 10 × 108 cells via the splenic artery infusion |
| NCT01454336 | Liver cirrhosis /fibrosis | Autologous MSCs | Phase 1 | 3 participants | Completed | The cells were administered via the portal vein. Cell dosage not informed. |
| NCT01220492 | Cirrhosis | UC-MSCs | Phase 1/2 | 266 participants | Completed | The cells were administered once a week for four weeks at a dose of 0.5 × 106 /kg body and intravenously for eight weeks. |
| NCT01120925 | Cirrhosis | BM-MNCs and enriched CD133+ HSCs | Phase 1/2 | 30 participants | Completed | BM-MNC were administered at a dose of 2–3 × 109 cells and CD133 at a dose of 5–15 × 106 cells, both via the portal vein. |
| NCT03963921 | NASH—non-alcoholic steatohepatitis | Liver-derived MSCs | Phase 1/2 | 23 participants | Completed | Route not informed. Cell dosage not informed. |
| NCT01591200 | Alcoholic liver cirrhosis | Allogeneic BM-MSCs | Phase 2 | 40 participants | Completed | The cells were administered at a low, medium, or high dose via the hepatic artery. Cell dosage not informed. |
| NCT01875081 | Alcoholic cirrhosis | Autologous BM-MSCs | Phase 2 | 72 participants | Completed | 5 × 107 cells via the hepatic artery. |
| NCT01378182 | Wilson’s cirrhosis | Allogeneic BM-MSCs | Not applicable | 10 participants | Completed | 1 × 106 cells/kg in total, with 1/2 of the dose in the peripheral vein and 1/2 of the dose in the right hepatic artery. |
| NCT01062750 | Cirrhosis | Autologous adipose tissue-derived stromal cells | Not applicable | 4 participants | Completed | The cells were administered via the hepatic artery. Cell dosage not informed. |
| NCT00956891 | Liver failure due to hepatitis B | Autologous BM-MSCs | Not applicable | 158 participants | Completed | The cells were administered via the hepatic artery. Cell dosage not informed. |
| NCT05517317 | Liver cirrhosis due to biliary atresia | Autologous BM-MNCs | Phase 1 | 12 participants | Completed | The cells were administered via the hepatic artery. Cell dosage not informed. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barreto da Silva, T.; Dias, E.A.; Cardoso, L.M.d.F.; Gama, J.F.G.; Alves, L.A.; Henriques-Pons, A. Magnetic Nanostructures and Stem Cells for Regenerative Medicine, Application in Liver Diseases. Int. J. Mol. Sci. 2023, 24, 9293. https://doi.org/10.3390/ijms24119293
Barreto da Silva T, Dias EA, Cardoso LMdF, Gama JFG, Alves LA, Henriques-Pons A. Magnetic Nanostructures and Stem Cells for Regenerative Medicine, Application in Liver Diseases. International Journal of Molecular Sciences. 2023; 24(11):9293. https://doi.org/10.3390/ijms24119293
Chicago/Turabian StyleBarreto da Silva, Tatiane, Evellyn Araújo Dias, Liana Monteiro da Fonseca Cardoso, Jaciara Fernanda Gomes Gama, Luiz Anastácio Alves, and Andrea Henriques-Pons. 2023. "Magnetic Nanostructures and Stem Cells for Regenerative Medicine, Application in Liver Diseases" International Journal of Molecular Sciences 24, no. 11: 9293. https://doi.org/10.3390/ijms24119293
APA StyleBarreto da Silva, T., Dias, E. A., Cardoso, L. M. d. F., Gama, J. F. G., Alves, L. A., & Henriques-Pons, A. (2023). Magnetic Nanostructures and Stem Cells for Regenerative Medicine, Application in Liver Diseases. International Journal of Molecular Sciences, 24(11), 9293. https://doi.org/10.3390/ijms24119293







