Effects of Obesity in Old Age on the Basement Membrane of Skeletal Muscle in Mice
Abstract
1. Introduction
2. Results
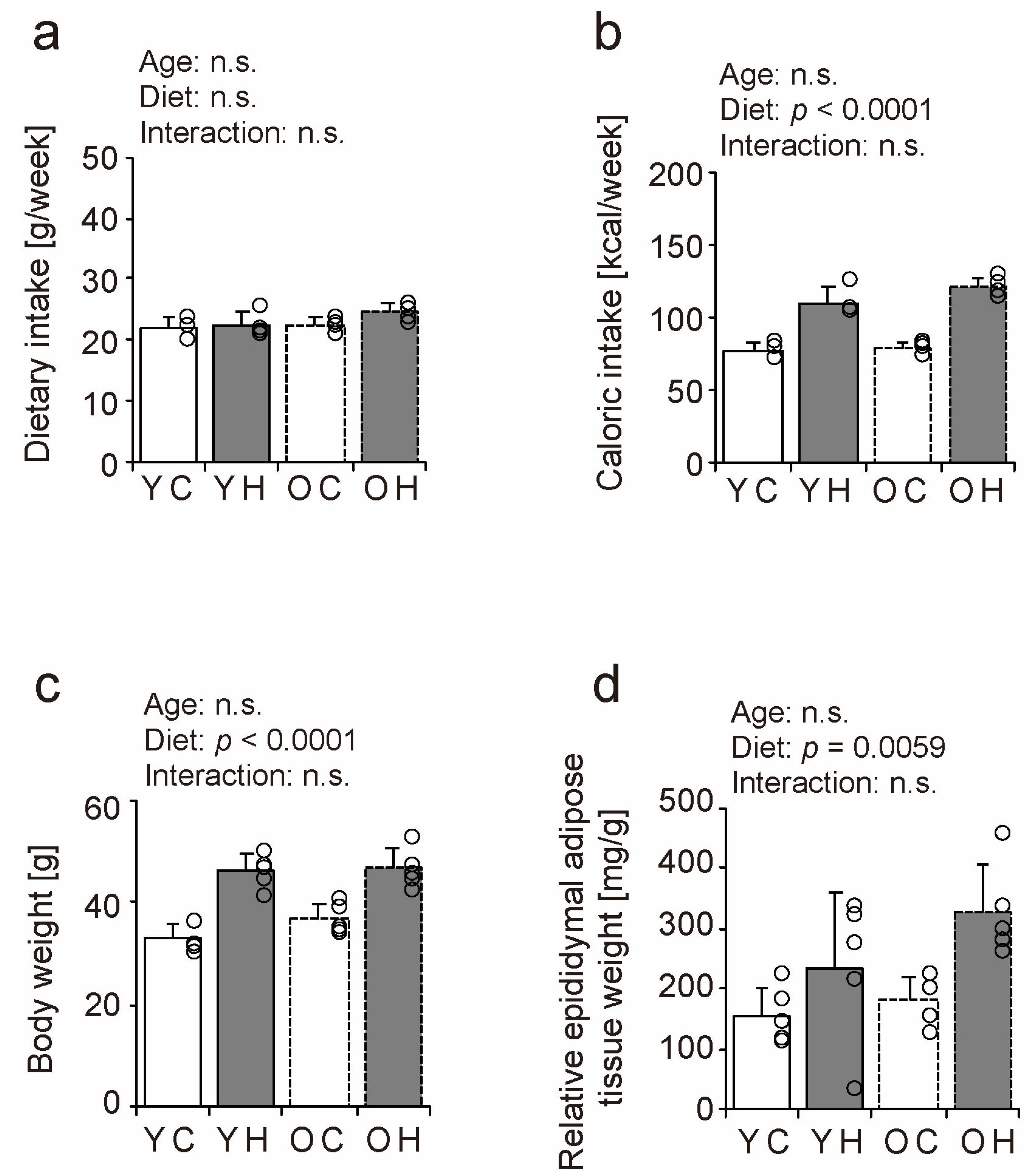
2.1. Dietary Intake, Body Weight, and Visceral Fat Content
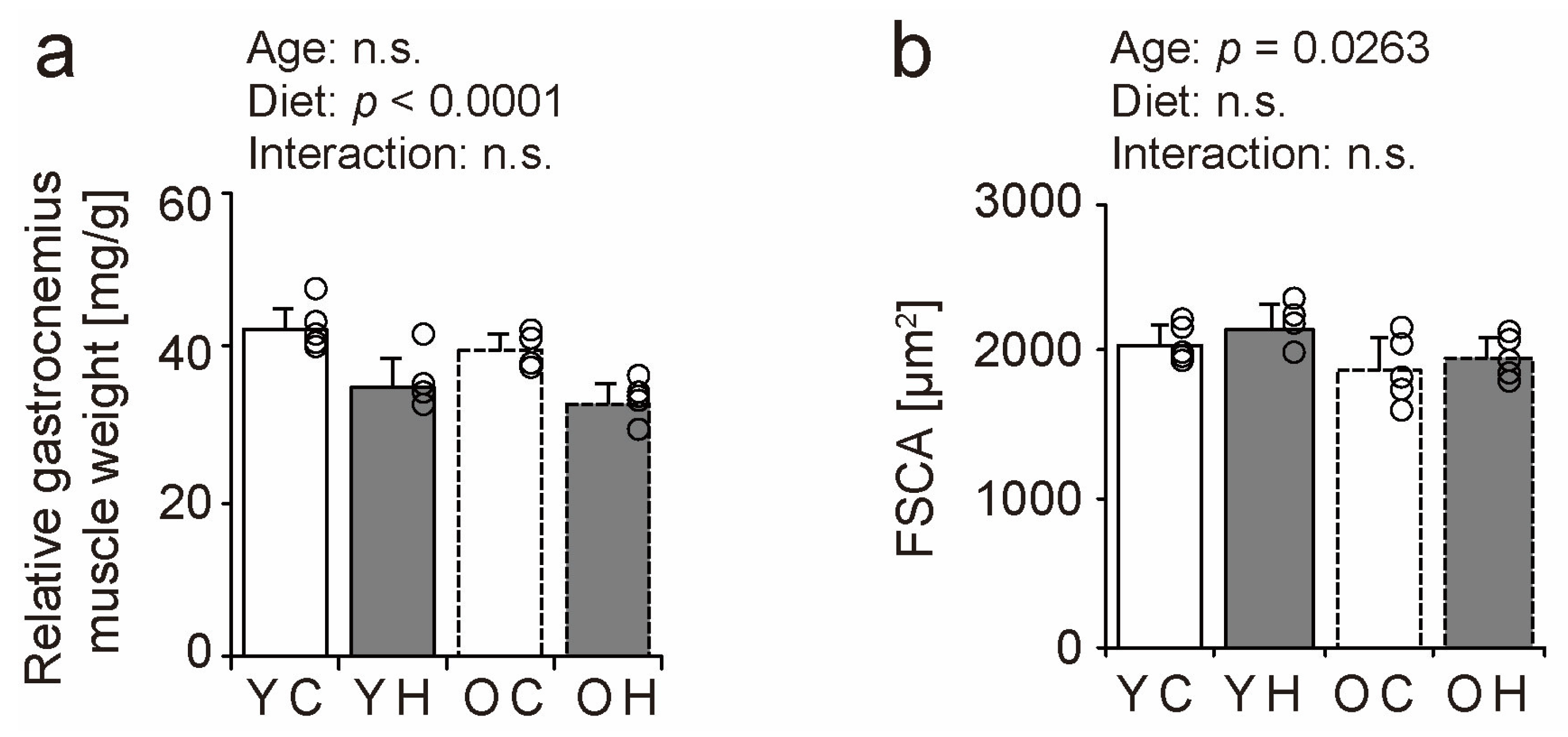
2.2. Relative Muscle Weight and Fiber Cross-Sectional Area
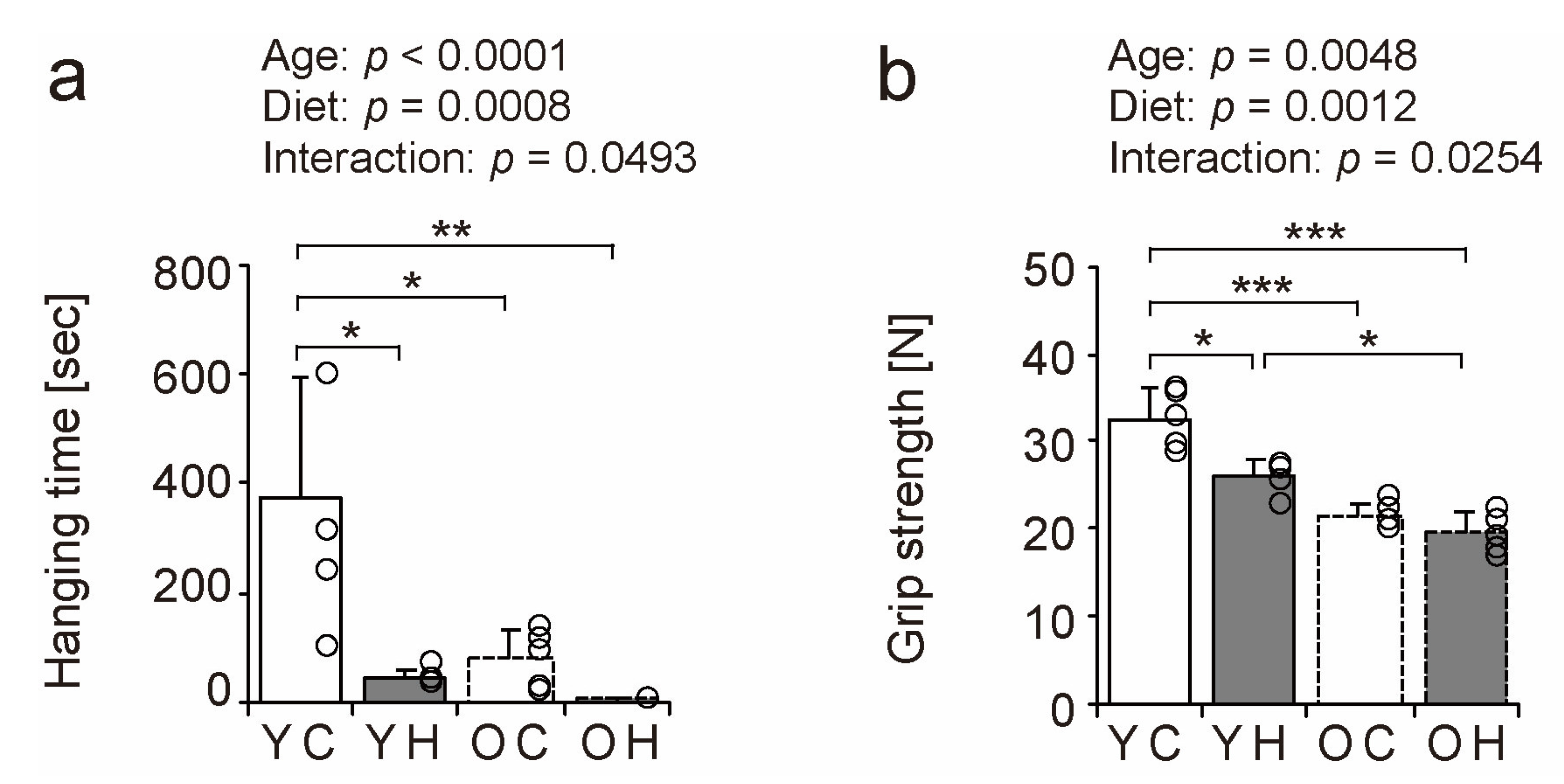
2.3. Muscle Function
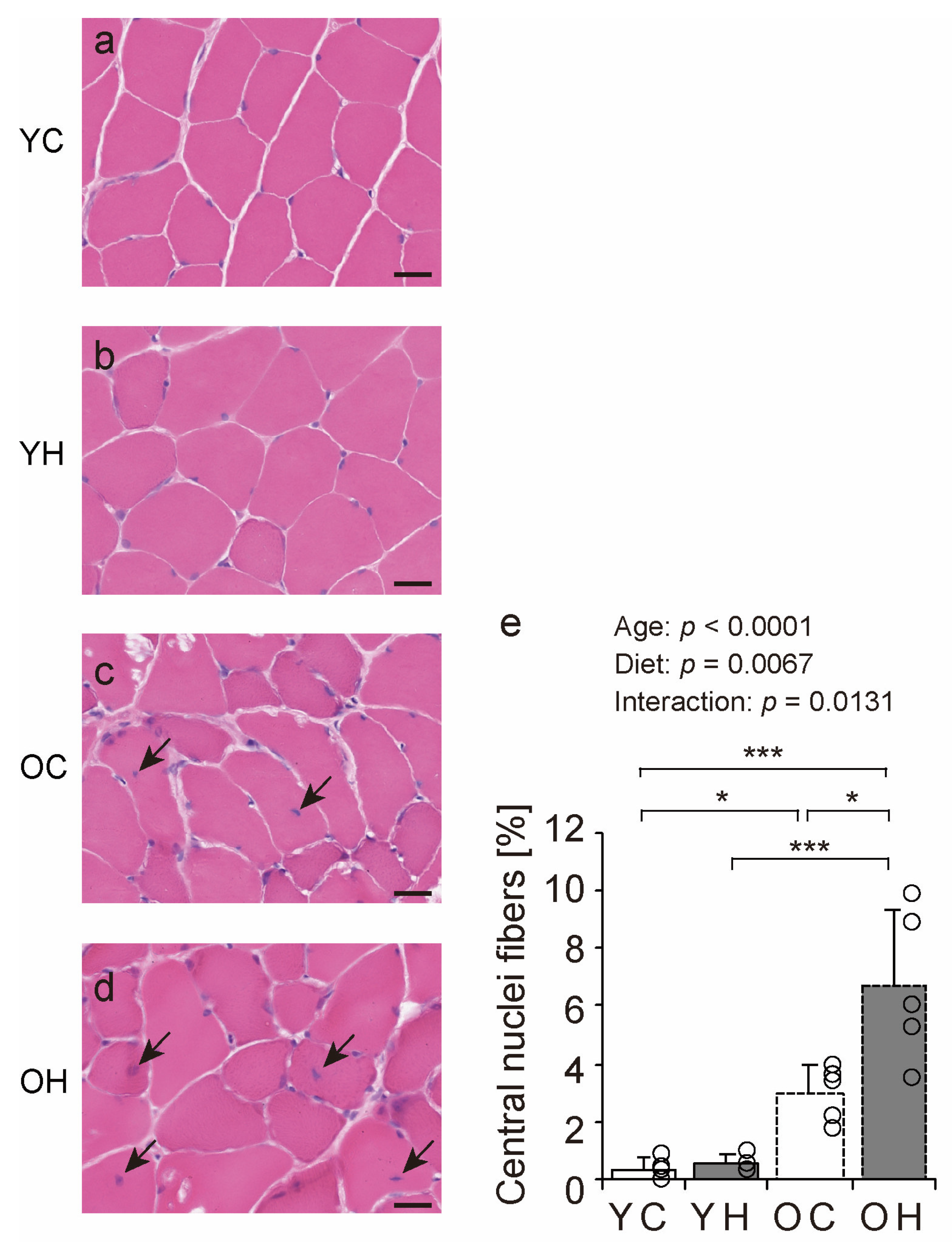
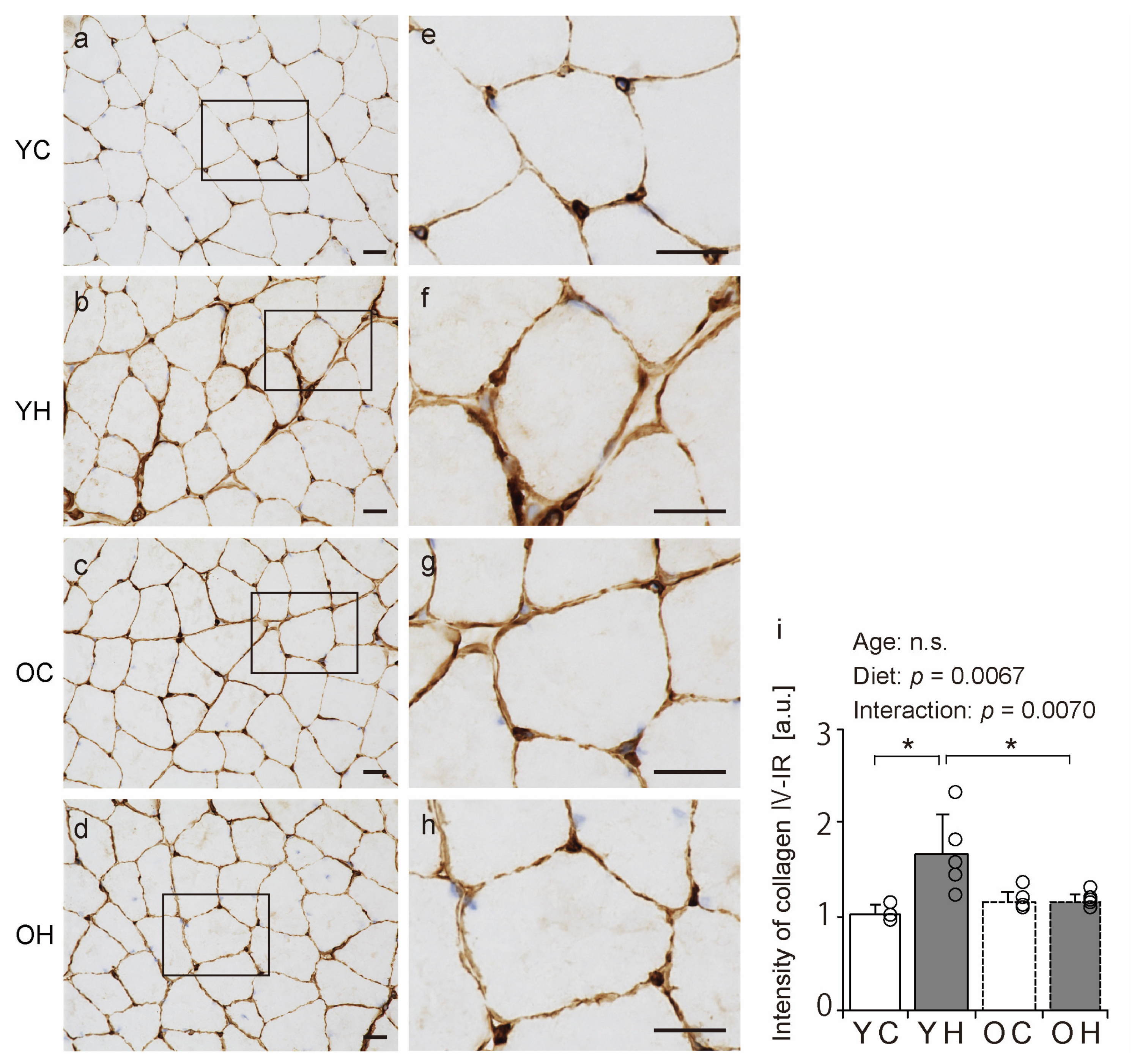
2.4. Histopathologic Findings
2.5. Collagen IV Localization
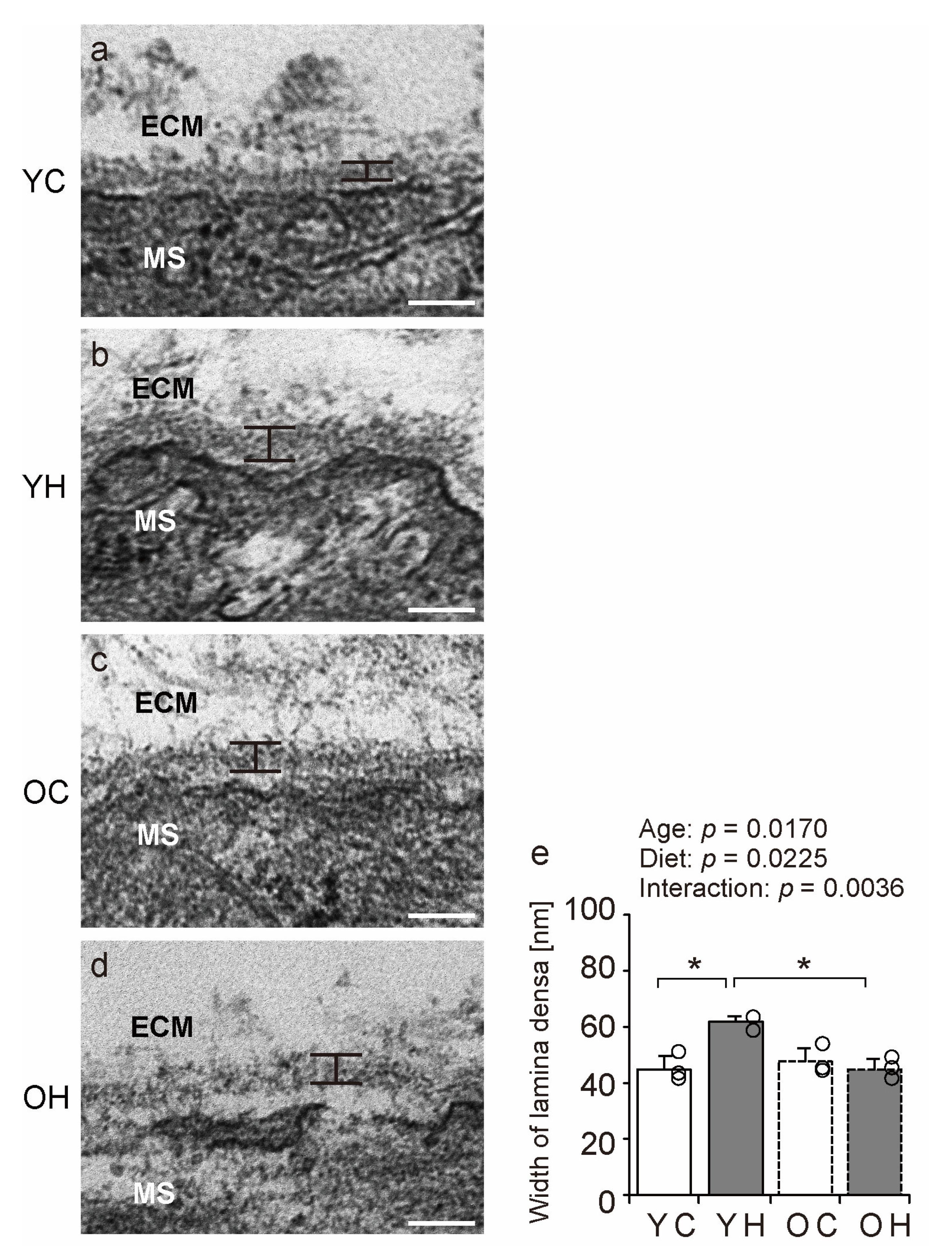
2.6. Electron Microscopy Analysis
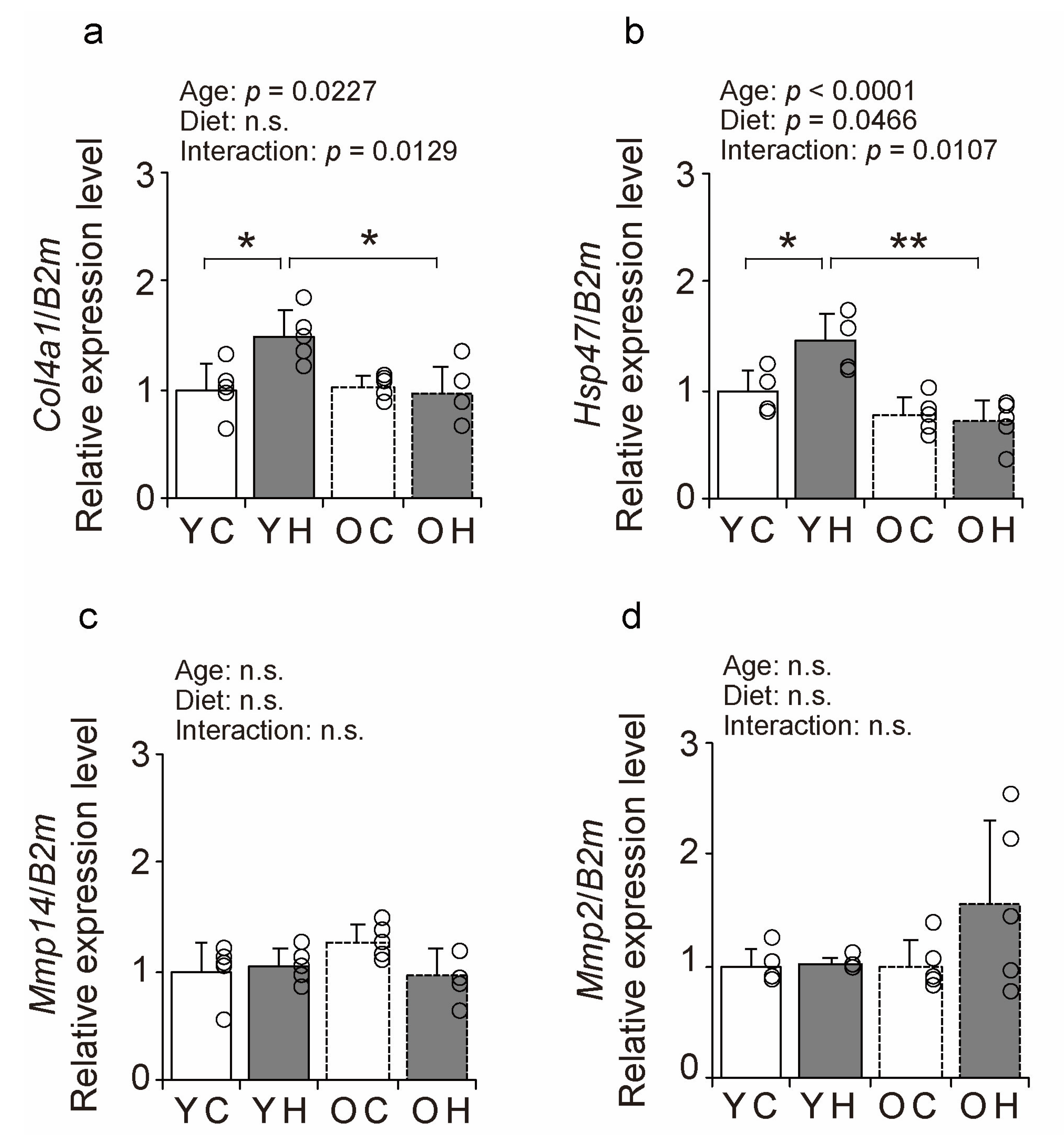
2.7. BM-Related Factors
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Diet
4.3. Group
4.4. Wire Hanging Test and Grip Strength Test
4.5. Sampling
4.6. Quantitative Polymerase Chain Reaction
4.7. Histochemical Analyses
4.8. Immunohistochemical Analysis
4.9. Electron Microscopy
4.10. Morphological Analysis
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Frontera, W.R.; Ochala, J. Skeletal Muscle: A Brief Review of Structure and Function. Calcif. Tissue Int. 2015, 96, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, C.; Weigert, C. Skeletal Muscle as an Endocrine Organ: The Role of Myokines in Exercise Adaptations. Cold Spring Harb. Perspect. Med. 2017, 7, a029793. [Google Scholar] [CrossRef] [PubMed]
- Schiaffino, S.; Dyar, K.A.; Ciciliot, S.; Blaauw, B.; Sandri, M. Mechanisms regulating skeletal muscle growth and atrophy. FEBS J. 2013, 280, 4294–4314. [Google Scholar] [CrossRef]
- Holloszy, J.O. Adaptation of skeletal muscle to endurance exercise. Med. Sci. Sports 1975, 7, 55–64. [Google Scholar] [CrossRef]
- Deschenes, M.R. Effects of aging on muscle fibre type and size. Sports Med. 2004, 34, 809–824. [Google Scholar] [CrossRef]
- Lexell, J.; Taylor, C.C.; Sjöström, M. What is the cause of the ageing atrophy? Total number, size and proportion of different fiber types studied in whole vastus lateralis muscle from 15- to 83-year-old men. J. Neurol. Sci. 1988, 84, 275–294. [Google Scholar] [CrossRef]
- Nilwik, R.; Snijders, T.; Leenders, M.; Groen, B.B.L.; van Kranenburg, J.; Verdijk, L.B.; van Loon, L.J.C. The decline in skeletal muscle mass with aging is mainly attributed to a reduction in type II muscle fiber size. Exp. Gerontol. 2013, 48, 492–498. [Google Scholar] [CrossRef]
- Kanazawa, Y.; Ikegami, K.; Sujino, M.; Koinuma, S.; Nagano, M.; Oi, Y.; Onishi, T.; Sugiyo, S.; Takeda, I.; Kaji, H.; et al. Effects of aging on basement membrane of the soleus muscle during recovery following disuse atrophy in rats. Exp. Gerontol. 2017, 98, 153–161. [Google Scholar] [CrossRef]
- Tomlinson, D.J.; Erskine, R.M.; Morse, C.I.; Winwood, K.; Onambélé-Pearson, G. The impact of obesity on skeletal muscle strength and structure through adolescence to old age. Biogerontology 2016, 17, 467–483. [Google Scholar] [CrossRef]
- Hwee, D.T.; Bodine, S.C. Age-related deficit in load-induced skeletal muscle growth. J. Gerontol. A Biol. Sci. Med. Sci. 2009, 64, 618–628. [Google Scholar] [CrossRef]
- Sanes, J.R. The basement membrane/basal lamina of skeletal muscle. J. Biol. Chem. 2003, 278, 12601–12604. [Google Scholar] [CrossRef]
- Yurchenco, P.D. Basement membranes: Cell scaffoldings and signaling platforms. Cold Spring Harb. Perspect. Biol. 2011, 3, a004911. [Google Scholar] [CrossRef]
- Kuo, D.S.; Labelle-Dumais, C.; Gould, D.B. COL4A1 and COL4A2 mutations and disease: Insights into pathogenic mecha-nisms and potential therapeutic targets. Hum. Mol. Genet. 2012, 21, 97–110. [Google Scholar] [CrossRef]
- Snyman, C.; Niesler, C.U. MMP-14 in skelet al muscle repair. J. Muscle Res. Cell. Motil. 2015, 36, 215–225. [Google Scholar] [CrossRef]
- Monaco, S.; Sparano, V.; Gioia, M.; Sbardella, D.; Pierro, D.D.; Marini, S.; Coletta, M. Enzymatic processing of Collagen Ⅳ by MMP-2 (gelatinase A) affects neutrophil migration and it is modulated by extracatalytic domains. Protein Sci. 2006, 15, 2805–2815. [Google Scholar] [CrossRef]
- Chioran, A.; Duncan, S.; Catalano, A.; Brown, T.J.; Ringuette, M.J. Collagen Ⅳ trafficking: The inside-out and beyond story. Dev. Biol. 2017, 431, 124–133. [Google Scholar] [CrossRef]
- Labelle-Dumais, C.; Dilworth, D.J.; Harrington, E.P.; de Leau, M.; Lyons, D.; Kabaeva, Z.; Manzini, M.C.; Dobyns, W.B.; Walsh, C.A.; Michele, D.D.; et al. COL4A1 mutations cause ocular dysgenesis, neuronal localization defects, and my-opathy in mice and walker-warburg syndrome in humans. PLoS Genet. 2011, 7, e1002062. [Google Scholar] [CrossRef]
- Kanazawa, Y.; Nagano, M.; Koinuma, S.; Sugiyo, S.; Shigeyoshi, Y. Effects of Aging on Basement Membrane of Tibialis Ante-rior Muscle During Recovery Following Muscle Injury in Rats. Microscopy 2022, 71, 245–248. [Google Scholar] [CrossRef]
- Kanazawa, Y.; Nagano, M.; Koinuma, S.; Sugiyo, S.; Shigeyoshi, Y. Effects of endurance exercise on basement membrane in the soleus muscle of aged rats. Acta Histochem. Cytochem. 2021, 54, 167–175. [Google Scholar] [CrossRef]
- Kanazawa, Y.; Nagano, M.; Koinuma, S.; Sugiyo, S.; Shigeyoshi, Y. Effects of aging on basement membrane-related gene ex-pression of the skeletal muscle in rats. Biomed. Res. 2021, 42, 115–119. [Google Scholar] [CrossRef]
- Deaver, J.W.; Schrems, E.R.; Brown, L.A.; Haynie, W.A.; Perry Jr, R.A.; Rosa-Caldwell, M.E.; Tedrowe, M.A.; Greene, N.P.; Washington, T.A. The effect of diet-induced obesity on extracellular matrix remodeling during skeletal muscle regeneration. Sports Med. Health Sci. 2006, 3, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.S.J.; Anderson, J.E.; Joya, J.E.; Head, S.I.; Pather, N.; Kee, A.J.; Gunning, P.W.; Hardeman, E.C. Aged skeletal muscle re-tains the ability to fully regenerate functional architecture. Bioarchitecture 2013, 2, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Jungbluth, H.; Treves, S.; Zorzato, F.; Sarkozy, A.; Ochala, J.; Sewry, C.; Phadke, R.; Gautel, M.; Muntoni, F. Congenital myo-pathies: Disorders of excitation–contraction coupling and muscle contraction. Nat. Rev. Neurol. 2018, 14, 151–167. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Sakiyama, K.; Kitamura, K.; Yamamoto, Y.; Takagi, T.; Sekiya, S.; Watanabe, G.; Taniguchi, S.; Ogawa, Y.; Ishizuka, S.; et al. Development and Regeneration of Muscle, Tendon, and Myotendinous Junctions in Striated Skeletal Muscle. Int. J. Mol. Sci. 2022, 23, 3006. [Google Scholar] [CrossRef]
- Rooney, J.E.; Welser, J.V.; Dechert, M.A.; Flintoff-Dye, N.L.; Kaufman, S.J.; Burkin, D.J. Severe muscular dystrophy in mice that lack dystrophin and alpha 7 integrin. J. Cell. Sci. 2006, 119, 2185–2195. [Google Scholar] [CrossRef]
- Labelle-Dumais, C.; Schuitema, V.; Hayashi, G.; Hoff, K.; Gong, W.; Dao, D.Q.; Ullian, E.M.; Oishi, P.; Margeta, M.; Gould, D.B. COL4A1 Mutations Cause Neuromuscular Disease with Tissue-Specific Mechanistic Heterogeneity. Am. J. Hum. Genet. 2019, 5, 847–860. [Google Scholar] [CrossRef]
- Kanazawa, Y.; Takahashi, T.; Higuchi, T.; Miyachi, R.; Nagano, M.; Koinuma, S.; Shigeyoshi, Y. Effects of stretching on the basement membrane structure in the soleus muscle of Wistar rats. Med. Mol. Morphol. 2022, 56, 11–19. [Google Scholar] [CrossRef]
- Urciuolo, A.; Quarta, M.; Morbidoni, V.; Gattazzo, F.; Molon, S.; Grumati, P.; Montemurro, F.; Tedesco, F.S.; Blaauw, B.; Cossu, G.; et al. Collagen Ⅵ regulates satellite cell self-renewal and muscle regeneration. Nat. Commun. 2013, 4, 1964. [Google Scholar] [CrossRef]
- Reggio, S.; Rouault, C.; Poitou, C.; Bichet, J.C.; Prifti, E.; Bouillot, J.L.; Rizkalla, S.; Lacasa, D.; Tordjman, J.; Clément, K. In-creased Basement Membrane Components in Adipose Tissue During Obesity: Links With TGFβ and Metabolic Phenotypes. J. Clin. Endocrinol. Metab. 2016, 6, 2578–2587. [Google Scholar] [CrossRef]
- Song, B.; Zhang, H.; Zhang, S. Toll-like receptor 2 mediates deposition of collagen I in adipose tissue of high fat diet-induced obese mice. Mol. Med. Rep. 2018, 4, 5958–5963. [Google Scholar] [CrossRef]
- Varani, J.; Dame, M.K.; Rittie, L.; Fligiel, S.E.; Kang, S.; Fisher, G.J.; Voorhees, J.J. Decreased collagen production in chrono-logically aged skin: Roles of age-dependent alteration in fibroblast function and defective mechanical stimulation. Am. J. Pathol. 2006, 168, 1861–1868. [Google Scholar] [CrossRef]
- Mak, K.M.; Mei, R. Basement membrane type IV collagen and laminin: An overview of their biology and value as fibrosis biomarkers of liver disease. Anat. Rec. 2017, 300, 1371–1390. [Google Scholar] [CrossRef]
- Davis, W.; Mahale, S.; Carranza, A.; Cox, B.; Hayes, K.; Jimenez, D.; Ding, Y. Exercise pre-conditioning ameliorates blood–brain barrier dysfunction in stroke by enhancing basal lamina. Neurol. Res. 2007, 29, 382–387. [Google Scholar] [CrossRef]
- Mattiello-Sverzut, A.C.; Petersen, S.G.; Kjaer, M.; Mackey, A.L. Morphological adaptation of muscle collagen and receptor of advanced glycation end product (RAGE) in osteoarthritis patients with 12 weeks of resistance training: Influence of an-ti-inflammatory or glucosamine treatment. Rheumatol. Int. 2013, 33, 2215–2224. [Google Scholar] [CrossRef]
- Gumpenberger, M.; Wessner, B.; Graf, A.; Narici, M.V.; Fink, C.; Braun, S.; Hoser, C.; Blazevich, A.J.; Csapo, R. Remodeling the Skeletal Muscle Extracellular Matrix in Older Age-Effects of Acute Exercise Stimuli on Gene Expression. Int. J. Mol. Sci. 2020, 21, 7089. [Google Scholar] [CrossRef]
- Kessler, D.; Dethlefsen, S.; Haase, I.; Plomann, M.; Hirche, F.; Krieg, T.; Eckes, B. Fibroblasts in Mechanically Stressed Collagen Lattices Assume a “Synthetic” Phenotype. J. Biol. Chem. 2001, 276, 36575–36585. [Google Scholar] [CrossRef]
- Furth, J.J. The steady-state levels of type I collagen mRNA are reduced in senes- cent fibroblasts. J. Gerontol. 1991, 46, B122–B124. [Google Scholar] [CrossRef]
- Speakman, J.R. Use of high-fat diets to study rodent obesity as a model of human obesity. Int. J. Obes. 2019, 43, 1491–1492. [Google Scholar] [CrossRef]
- Spencer, M.; Unal, R.; Zhu, B.; Rasouli, N.; McGehee, R.E., Jr.; Peterson, C.A.; Kern, P.A. Adipose Tissue Extracellular Matrix and Vascular Abnormalities in Obesity and Insulin Resistance. J. Clin. Endocrinol. Metab. 2011, 96, 1990–1998. [Google Scholar] [CrossRef]
- Kovanen, V.; Suominen, H.; Risteli, J.; Risteli, L. Type IV collagen and laminin in slow and fast skeletal muscle in rats--effects of age and life-time endurance training. Coll. Relat. Res. 1988, 8, 145–153. [Google Scholar] [CrossRef]
- Wood, L.K.; Kayupov, E.; Gumucio, J.P.; Mendias, C.L.; Claflin, D.R.; Brooks, S.V. Intrinsic stiffness of extracellular matrix increases with age in skeletal muscles of mice. J. Appl. Physiol. 1985 2014, 117, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Lofaro, F.D.; Cisterna, B.; Lacavalla, M.A.; Boschi, F.; Malatesta, M.; Quaglino, D.; Zancanaro, C.; Boraldi, F. Age-Related Changes in the Matrisome of the Mouse Skeletal Muscle. Int. J. Mol. Sci. 2021, 22, 10564. [Google Scholar] [CrossRef] [PubMed]
- Koskinen, S.O.A.; Ahtikoski, A.M.; Komulainen, J.; Hesselink, M.K.C.; Drost, M.R.; Takala, T.E.S. Short-term effects of forced eccentric contractions on collagen synthesis and degradation in rat skeletal muscle. Pflugers Arch. 2002, 444, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.M.; Vykoukal, J.; Lechler, P.; Zeitler, K.; Gehmert, S.; Schreml, S.; Alt, E.; Bogdahn, U.; Prantl, L. Noninvasive in vivo assessment of muscle impairment in the mdx mouse model—A comparison of two common wire hanging methods with two different results. J. Neurosci. Methods 2012, 203, 292–297. [Google Scholar] [CrossRef]
- Crowe, A.R.; Wei, Y. Semi-quantitative Determination of Protein Expression Using Immunohistochemistry Staining and Analysis: An Integrated Protocol. Bio. Protoc. 2019, 9, 3465. [Google Scholar] [CrossRef]
- Schindelin, J.; Carreras, I.A.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biologi-cal-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kanazawa, Y.; Ikeda-Matsuo, Y.; Sato, H.; Nagano, M.; Koinuma, S.; Takahashi, T.; Suzuki, H.; Miyachi, R.; Shigeyoshi, Y. Effects of Obesity in Old Age on the Basement Membrane of Skeletal Muscle in Mice. Int. J. Mol. Sci. 2023, 24, 9209. https://doi.org/10.3390/ijms24119209
Kanazawa Y, Ikeda-Matsuo Y, Sato H, Nagano M, Koinuma S, Takahashi T, Suzuki H, Miyachi R, Shigeyoshi Y. Effects of Obesity in Old Age on the Basement Membrane of Skeletal Muscle in Mice. International Journal of Molecular Sciences. 2023; 24(11):9209. https://doi.org/10.3390/ijms24119209
Chicago/Turabian StyleKanazawa, Yuji, Yuri Ikeda-Matsuo, Hiaki Sato, Mamoru Nagano, Satoshi Koinuma, Tatsuo Takahashi, Hirokazu Suzuki, Ryo Miyachi, and Yasufumi Shigeyoshi. 2023. "Effects of Obesity in Old Age on the Basement Membrane of Skeletal Muscle in Mice" International Journal of Molecular Sciences 24, no. 11: 9209. https://doi.org/10.3390/ijms24119209
APA StyleKanazawa, Y., Ikeda-Matsuo, Y., Sato, H., Nagano, M., Koinuma, S., Takahashi, T., Suzuki, H., Miyachi, R., & Shigeyoshi, Y. (2023). Effects of Obesity in Old Age on the Basement Membrane of Skeletal Muscle in Mice. International Journal of Molecular Sciences, 24(11), 9209. https://doi.org/10.3390/ijms24119209





