Isoflavones Mediate Dendritogenesis Mainly through Estrogen Receptor α
Abstract
1. Introduction
2. Results
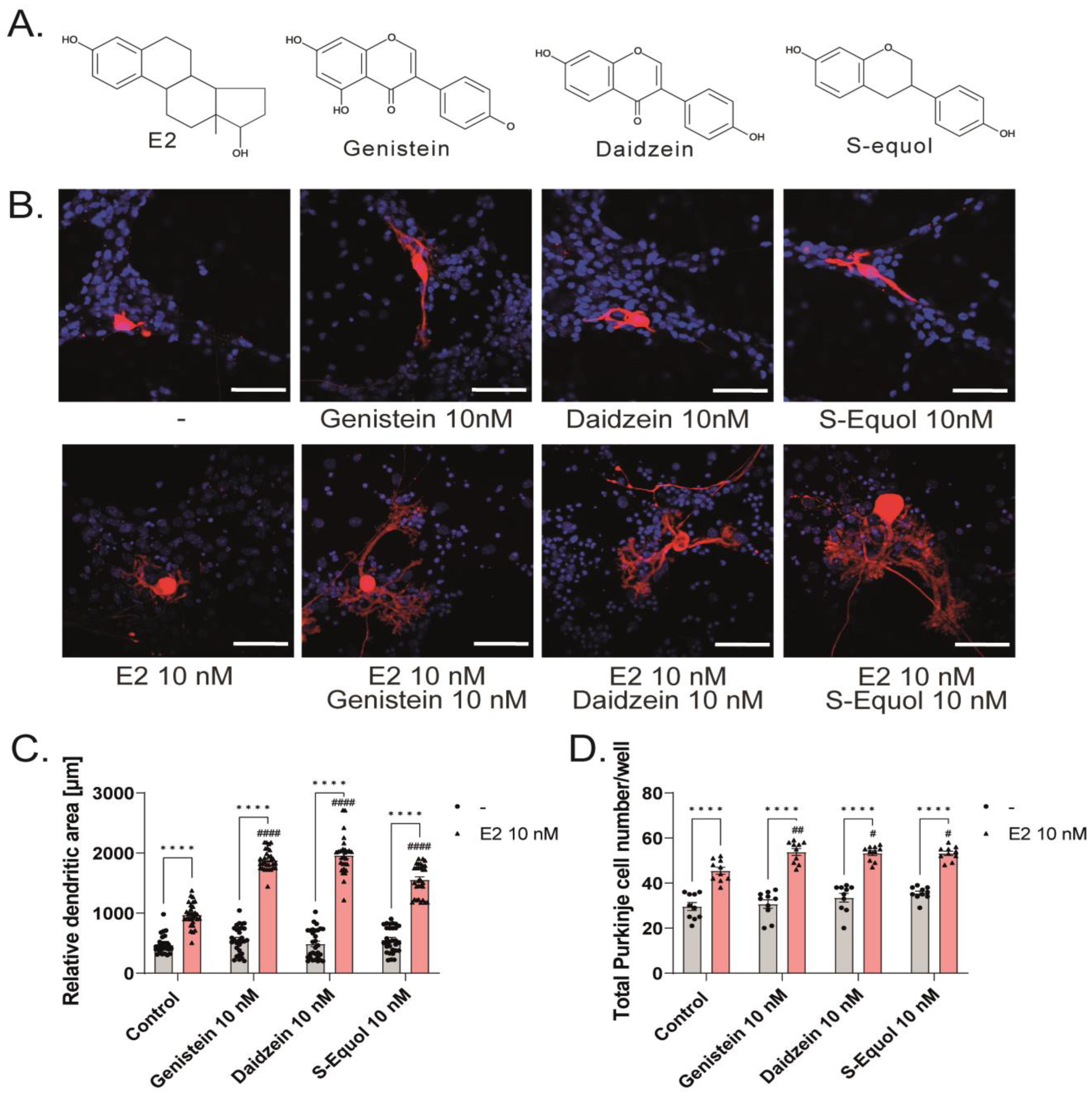
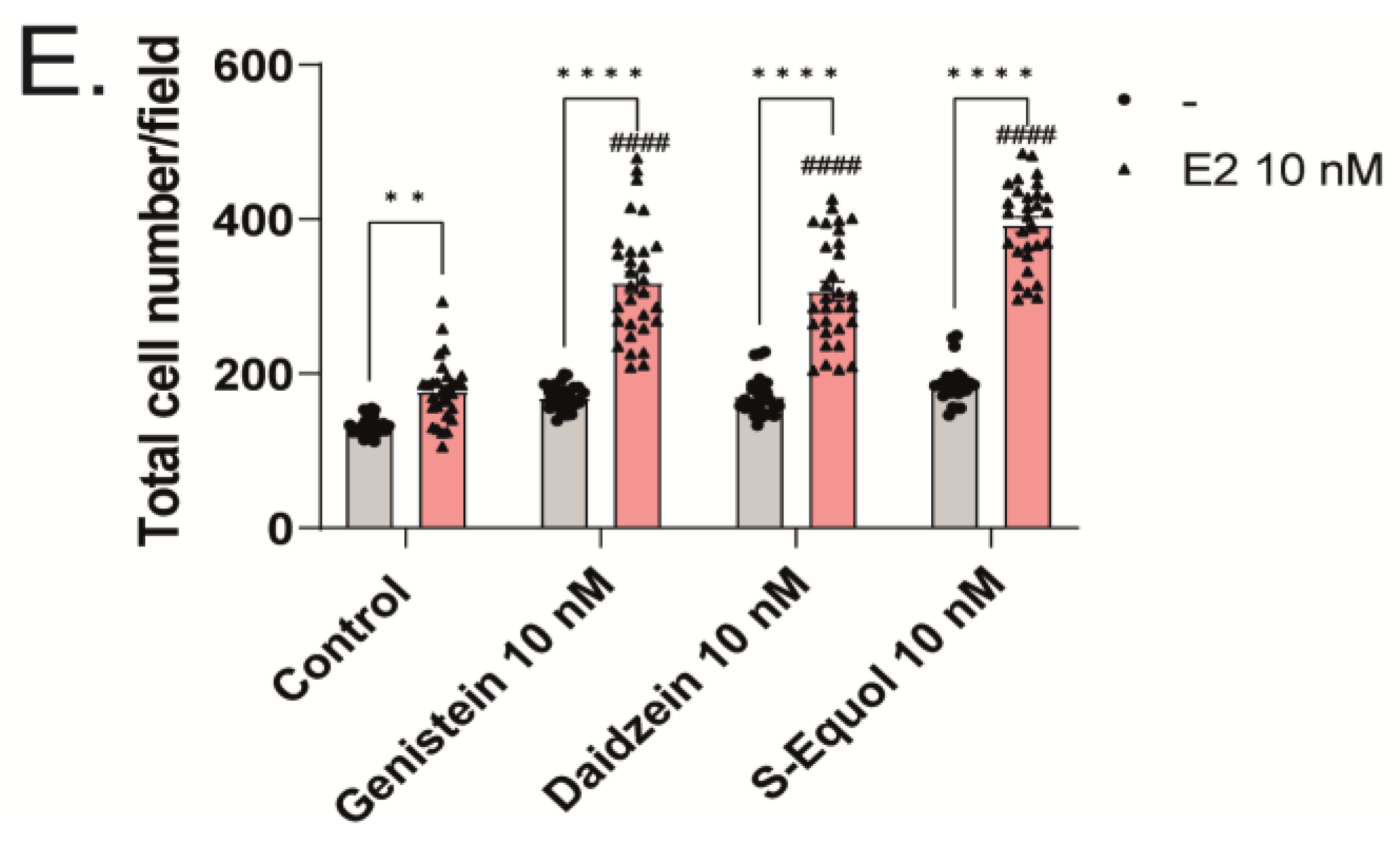
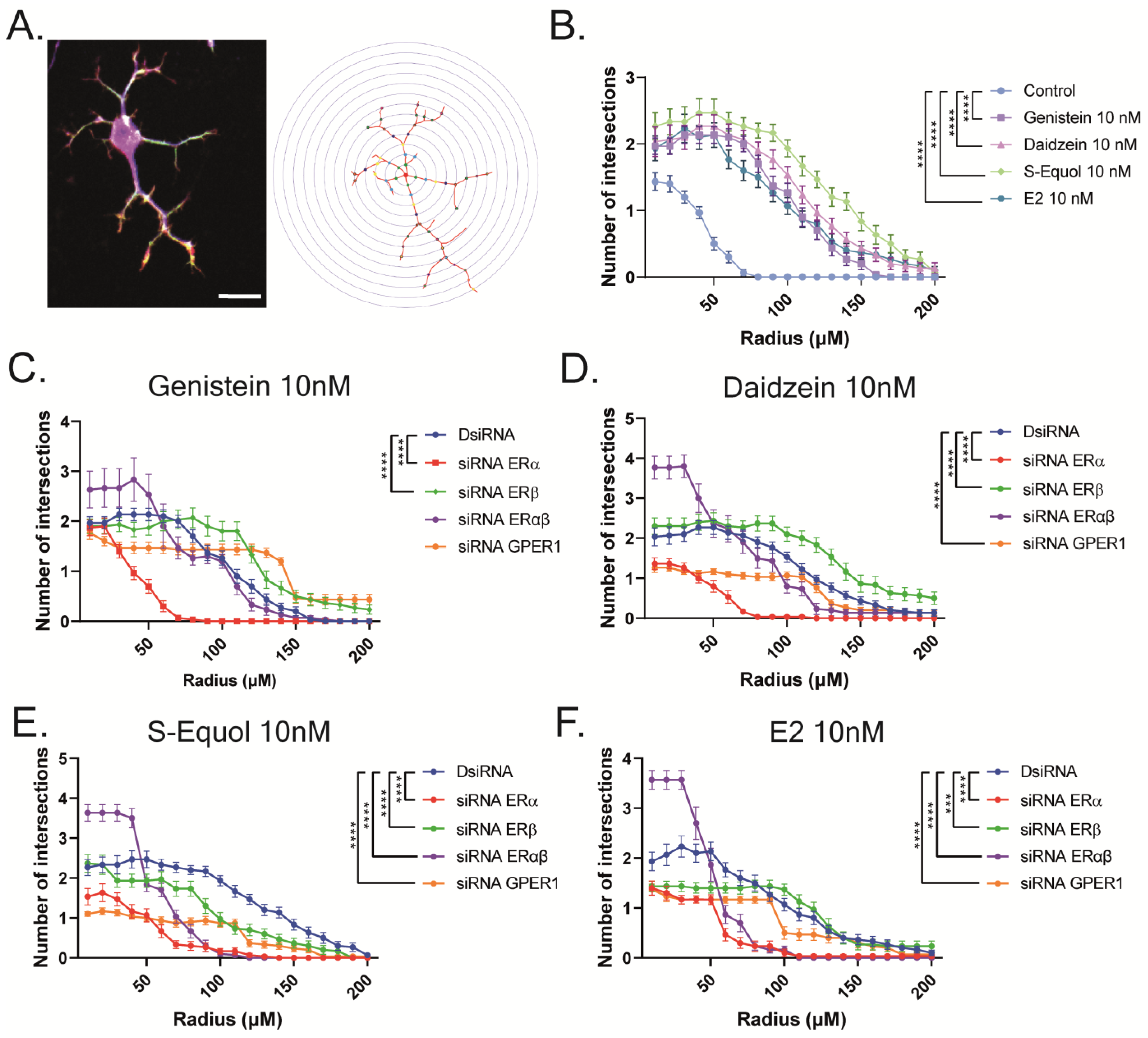
2.1. Isoflavones Enhanced Cell Number and Dendrite Arborization in Purkinje Cells in the Primary Culture of Mouse Cerebellar Cells
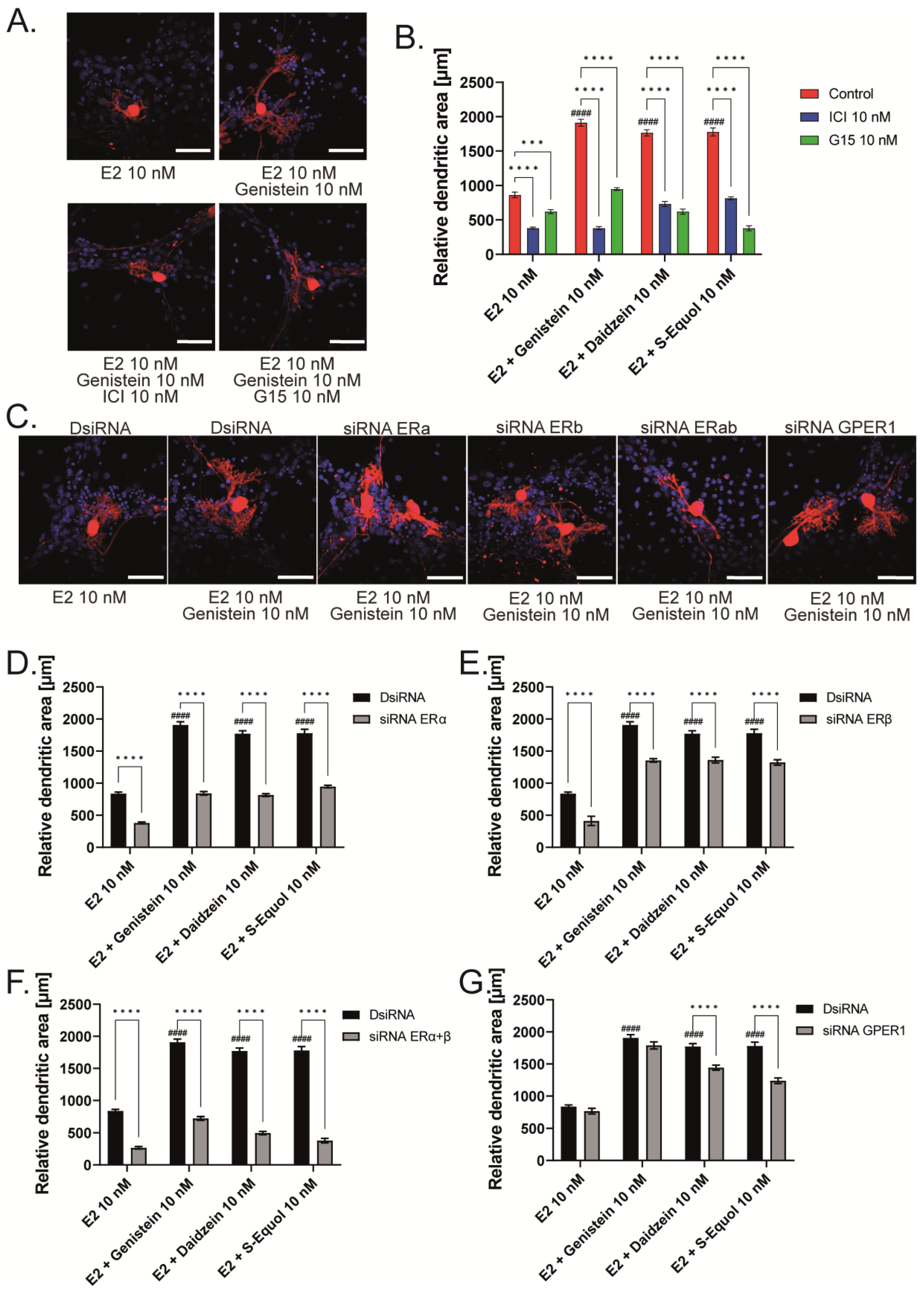
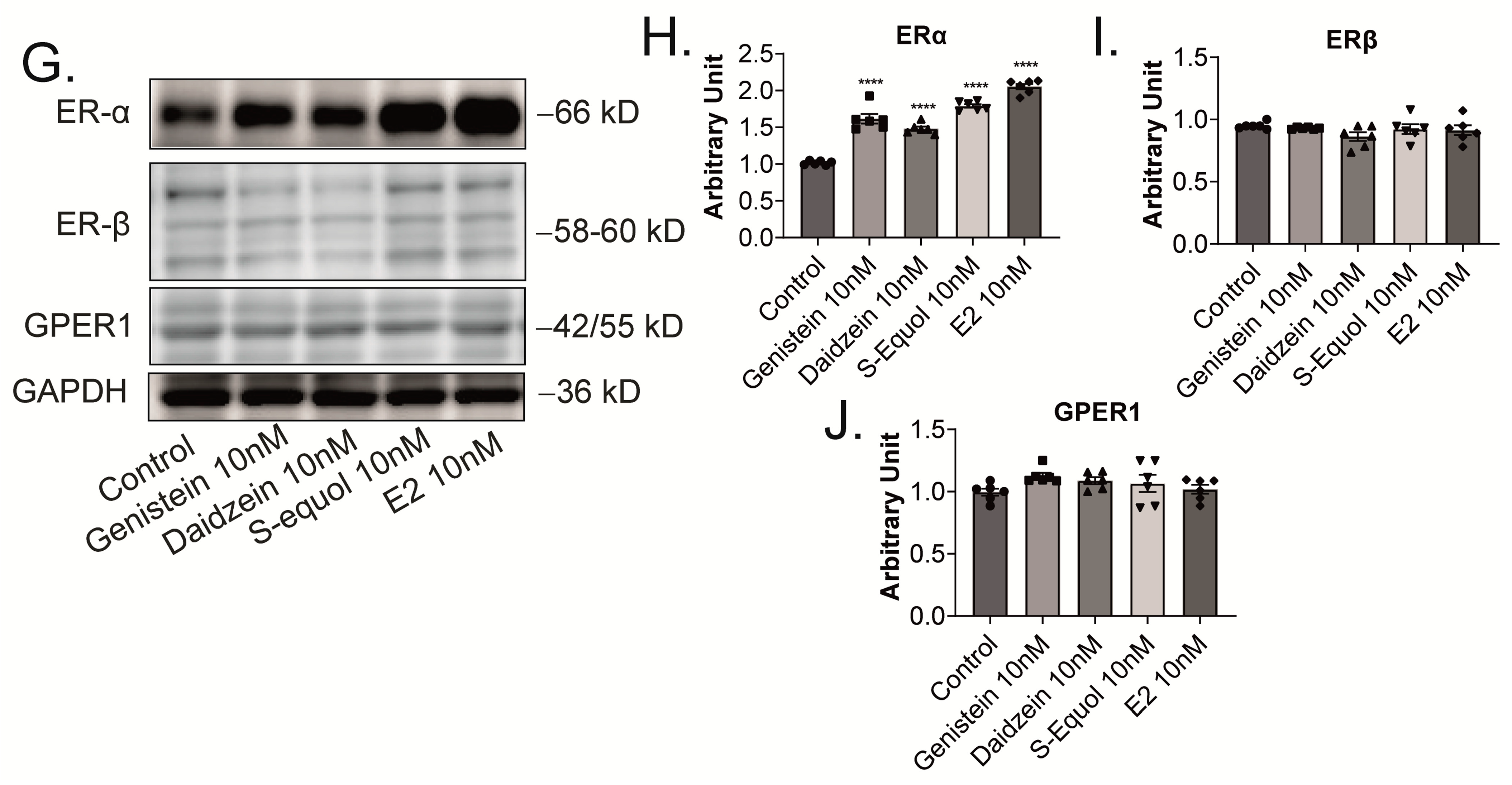
2.2. GPER1 and ER Are Required for Isoflavone-Enhanced E2-Mediated Dendrite Arborization in Purkinje Cells
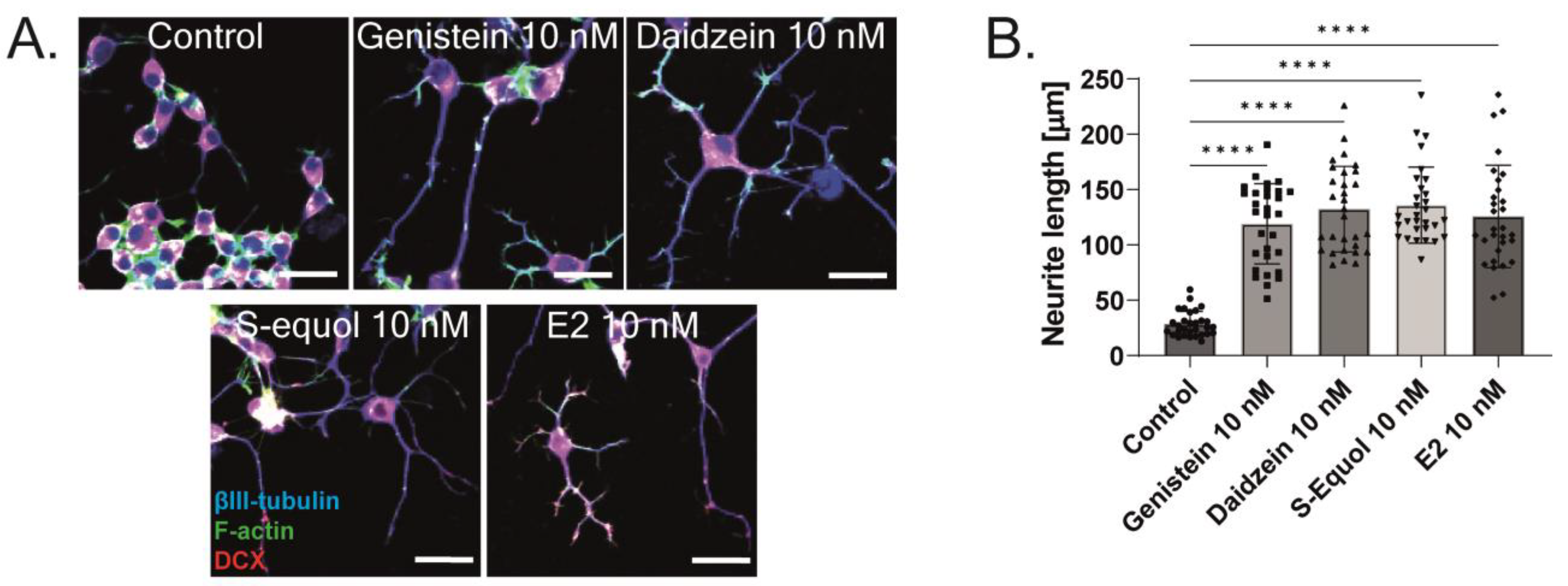
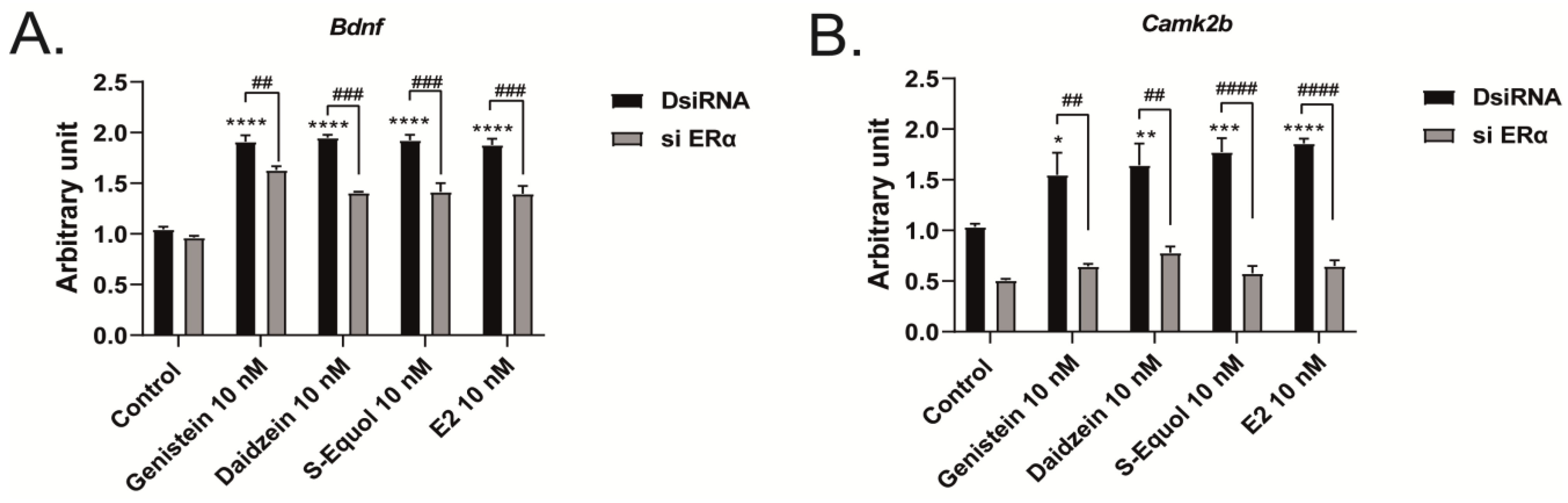
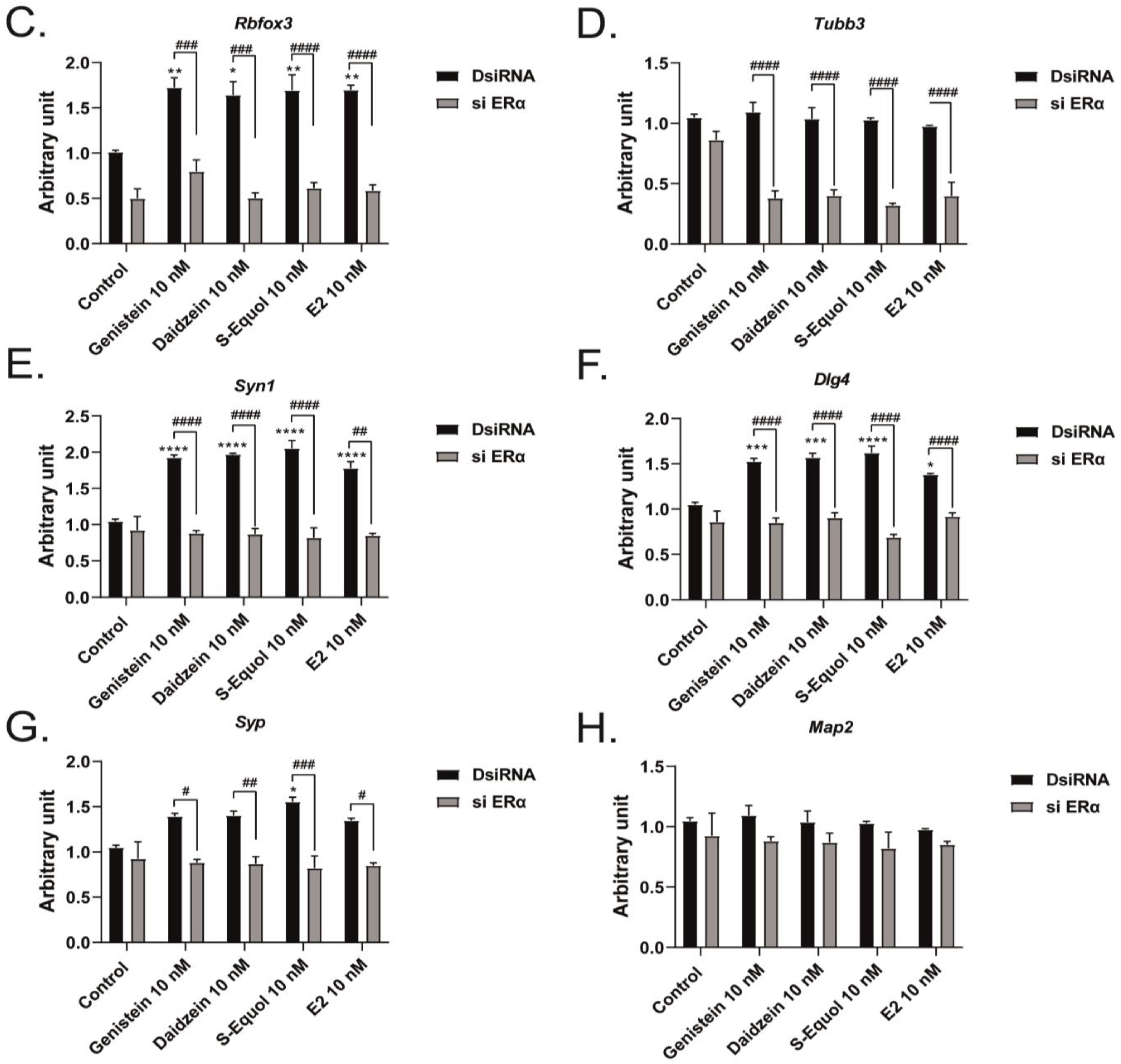
2.3. ERα Mediated Isoflavone-Induced Neurite Outgrowth in Neuro-2A Cells
2.4. Neuron–Glia Interaction Enhanced Isoflavone-Mediated Neuritogenesis
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Primary Culture of Mouse Cerebellar Cells
4.3. Primary Culture of Mouse Cerebellar Astrocytes
4.4. Mouse Neuro-2A and Co-Culture Studies
4.5. RNA Interference Assay
4.6. Western Blotting
4.7. Software and Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Miadoková, E. Isoflavonoids—An overview of their biological activities and potential health benefits. Interdiscip. Toxicol. 2009, 2, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Zaheer, K.; Humayoun Akhtar, M. An updated review of dietary isoflavones: Nutrition, processing, bioavailability and impacts on human health. Crit. Rev. Food Sci. Nutr. 2017, 57, 1280–1293. [Google Scholar] [CrossRef]
- Gorzkiewicz, J.; Bartosz, G.; Sadowska-Bartosz, I. The potential effects of phytoestrogens: The role in neuroprotection. Molecules 2021, 26, 2954. [Google Scholar] [CrossRef]
- Křížová, L.; Dadáková, K.; Kašparovská, J.; Kašparovský, T. Isoflavones. Molecules 2019, 24, 1076. [Google Scholar] [CrossRef] [PubMed]
- Ariyani, W.; Miyazaki, W.; Koibuchi, N. A Novel Mechanism of S-equol Action in Neurons and Astrocytes: The Possible Involvement of GPR30/GPER1. Int. J. Mol. Sci. 2019, 20, 5178. [Google Scholar] [CrossRef] [PubMed]
- Ariyani, W.; Miyazaki, W.; Amano, I.; Hanamura, K.; Shirao, T.; Koibuchi, N. Soy Isoflavones Accelerate Glial Cell Migration via GPER-Mediated Signal Transduction Pathway. Front. Endocrinol. 2020, 11, 554941. [Google Scholar] [CrossRef]
- Prossnitz, E.R.; Maggiolini, M. Mechanisms of estrogen signaling and gene expression via GPR30. Mol. Cell. Endocrinol. 2009, 308, 32–38. [Google Scholar] [CrossRef]
- Prossnitz, E.R.; Arterburn, J.B. International Union of Basic and Clinical Pharmacology. XCVII. G Protein–Coupled Estrogen Receptor and Its Pharmacologic Modulators. Pharmacol. Rev. 2015, 67, 505–540. [Google Scholar] [CrossRef]
- Tuli, H.S.; Tuorkey, M.J.; Thakral, F.; Sak, K.; Kumar, M.; Sharma, A.K.; Sharma, U.; Jain, A.; Aggarwal, V.; Bishayee, A. Molecular mechanisms of action of genistein in cancer: Recent advances. Front. Pharmacol. 2019, 10, 1336. [Google Scholar] [CrossRef]
- Li, C.; Fan, Y.; Li, S.; Zhou, X.; Park, K.Y.; Zhao, X.; Liu, H. Antioxidant Effect of Soymilk Fermented by Lactobacillus plantarum HFY01 on D-Galactose-Induced Premature Aging Mouse Model. Front. Nutr. 2021, 8, 667643. [Google Scholar] [CrossRef]
- Ren, M.Q.; Kuhn, G.; Wegner, J.; Chen, J. Isoflavones, substances with multi-biological and clinical properties. Eur. J. Nutr. 2001, 40, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Lecomte, S.; Demay, F.; Ferri, F.; Pakdel, F. Phytochemicals Targeting Estrogen Receptors: Beneficial Rather Than Adverse Effects? Int. J. Mol. Sci. 2017, 18, 1381. [Google Scholar] [CrossRef] [PubMed]
- Beckinghausen, J.; Sillitoe, R.V. Insights into cerebellar development and connectivity. Neurosci. Lett. 2019, 688, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Ariyani, W.; Iwasaki, T.; Miyazaki, W.; Yu, L.; Takeda, S.; Koibuchi, N. A possible novel mechanism of action of genistein and daidzein for activating thyroid hormone receptor-mediated transcription. Toxicol. Sci. 2018, 164, 417–427. [Google Scholar] [CrossRef] [PubMed]
- MacLusky, N.J.; Thomas, G.; Leranth, C. Low dietary soy isoflavonoids increase hippocampal spine synapse density in ovariectomized rats. Brain Res. 2017, 1657, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Rivera, P.; Pérez-Martín, M.; Pavón, F.J.; Serrano, A.; Crespillo, A.; Cifuentes, M.; López-Ávalos, M.D.; Grondona, J.M.; Vida, M.; Fernández-Llebrez, P.; et al. Pharmacological Administration of the Isoflavone Daidzein Enhances Cell Proliferation and Reduces High Fat Diet-Induced Apoptosis and Gliosis in the Rat Hippocampus. PLoS ONE 2013, 8, e64750. [Google Scholar] [CrossRef]
- Giri, R.; Shen, Y.; Stins, M.; Du Yan, S.; Schmidt, A.M.; Stern, D.; Kim, K.S.; Zlokovic, B.; Kalra, V.K. β-Amyloid-induced migration of monocytes across human brain endothelial cells involves RAGE and PECAM-1. Am. J. Physiol. Cell Physiol. 2000, 279, 1772–1781. [Google Scholar] [CrossRef]
- Atlante, A.; Bobba, A.; Paventi, G.; Pizzuto, R.; Passarella, S. Genistein and daidzein prevent low potassium-dependent apoptosis of cerebellar granule cells. Biochem. Pharmacol. 2010, 79, 758–767. [Google Scholar] [CrossRef]
- Patisaul, H.B. Phytoestrogen action in the adult and developing brain. J. Neuroendocrinol. 2005, 17, 57–64. [Google Scholar] [CrossRef]
- Lephart, E.D.; West, T.W.; Weber, K.S.; Rhees, R.W.; Setchell, K.D.R.; Adlercreutz, H.; Lund, T.D. Neurobehavioral effects of dietary soy phytoestrogens. Neurotoxicol. Teratol. 2002, 24, 5–16. [Google Scholar] [CrossRef]
- Kohara, Y.; Kuwahara, R.; Kawaguchi, S.; Jojima, T.; Yamashita, K. Perinatal exposure to genistein, a soy phytoestrogen, improves spatial learning and memory but impairs passive avoidance learning and memory in offspring. Physiol. Behav. 2014, 130, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y.; Tanaka, K.; Okubo, H.; Sasaki, S.; Tokinobu, A.; Arakawa, M. Maternal consumption of soy and isoflavones during pregnancy and risk of childhood behavioural problems: The Kyushu Okinawa Maternal and Child Health Study. Int. J. Food Sci. Nutr. 2021, 72, 1118–1127. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y.; Tanaka, K.; Okubo, H.; Sasaki, S.; Furukawa, S.; Arakawa, M. Soy isoflavone intake and prevalence of depressive symptoms during pregnancy in Japan: Baseline data from the Kyushu Okinawa Maternal and Child Health Study. Eur. J. Nutr. 2018, 57, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, R.; Yamamoto, E.; Endo, Y. Morphological effects of isoflavones (daidzein and genistein) on hypothalamic oxytocin neurons in the neonatal mouse brain slice cultures. Neurosci. Lett. 2011, 505, 87–92. [Google Scholar] [CrossRef]
- Ariyani, W.; Miyazaki, W.; Amano, I.; Koibuchi, N. Involvement of integrin αvβ3 in thyroid hormone-induced dendritogenesis. Front. Endocrinol. 2022, 13, 938596. [Google Scholar] [CrossRef] [PubMed]
- Khakh, B.S.; Deneen, B. The Emerging Nature of Astrocyte Diversity. Annu. Rev. Neurosci. 2019, 42, 187–207. [Google Scholar] [CrossRef] [PubMed]
- Khakh, B.S.; Sofroniew, M.V. Diversity of astrocyte functions and phenotypes in neural circuits. Nat. Neurosci. 2015, 18, 942–952. [Google Scholar] [CrossRef]
- Zhan, J.S.; Gao, K.; Chai, R.C.; Jia, X.H.; Luo, D.P.; Ge, G.; Jiang, Y.W.; Fung, Y.-W.W.; Li, L.; Yu, A.C.H. Astrocytes in Migration. Neurochem. Res. 2017, 42, 272–282. [Google Scholar] [CrossRef]
- Bohlen, C.J.; Friedman, B.A.; Dejanovic, B.; Sheng, M. Microglia in Brain Development, Homeostasis, and Neurodegeneration. Annu. Rev. Genet. 2019, 53, 263–288. [Google Scholar] [CrossRef]
- Kolb, B.; Harker, A.; Gibb, R. Principles of plasticity in the developing brain. Dev. Med. Child Neurol. 2017, 59, 1218–1223. [Google Scholar] [CrossRef]
- Menassa, D.A.; Gomez-Nicola, D. Microglial dynamics during human brain development. Front. Immunol. 2018, 9, 1014. [Google Scholar] [CrossRef]
- Baez-Jurado, E.; Rincón-Benavides, M.A.; Hidalgo-Lanussa, O.; Guio-Vega, G.; Ashraf, G.M.; Sahebkar, A.; Echeverria, V.; Garcia-Segura, L.M.; Barreto, G.E. Molecular mechanisms involved in the protective actions of Selective Estrogen Receptor Modulators in brain cells. Front. Neuroendocrinol. 2019, 52, 44–64. [Google Scholar] [CrossRef]
- D’Angelo, E. Physiology of the Cerebellum, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2018; Volume 154, ISBN 9780444639561. [Google Scholar]
- van Essen, M.J.; Nayler, S.; Becker, E.B.E.; Jacob, J. Deconstructing cerebellar development cell by cell. PLoS Genet. 2020, 16, e1008630. [Google Scholar] [CrossRef]
- Bohne, P.; Schwarz, M.K.; Herlitze, S.; Mark, M.D. A New Projection From the Deep Cerebellar Nuclei to the Hippocampus via the Ventrolateral and Laterodorsal Thalamus in Mice. Front. Neural Circuits 2019, 13, 51. [Google Scholar] [CrossRef]
- Rojas, H.; Ramos, M.; Dipolo, R. A genistein-sensitive Na+/Ca2+ exchange is responsible for the resting [Ca2+]i and most of the Ca2+ plasma membrane fluxes in stimulated rat cerebellar type 1 astrocytes. Jpn. J. Physiol. 2004, 54, 249–262. [Google Scholar] [CrossRef]
- Kim, Y.; Wong, A.C.Y.; Power, J.M.; Tadros, S.F.; Klugmann, M.; Moorhouse, A.J.; Bertrand, P.P.; Housley, G.D. Alternative splicing of the TRPC3 ion channel calmodulin/ IP 3 receptor-binding domain in the hindbrain Enhances cation flux. J. Neurosci. 2012, 32, 11414–11423. [Google Scholar] [CrossRef]
- Tierney, A.L.; Nelson, C.A. Brain Development and the Role of Experience in the Early Years. Zero Three 2009, 30, 9–13. [Google Scholar]
- Marshall, J.J.; Mason, J.O. Mouse vs man: Organoid models of brain development & disease. Brain Res. 2019, 1724, 146427. [Google Scholar] [CrossRef] [PubMed]
- Mccarthy, M.M. Estradiol and the developing brain. Physiol. Rev. 2009, 88, 91–124. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, J.F.; Wright, C.L.; McCarthy, M.M. A Critical Period in Purkinje Cell Development Is Mediated by Local Estradiol Synthesis, Disrupted by Inflammation, and Has Enduring Consequences Only for Males. J. Neurosci. 2016, 36, 10039–10049. [Google Scholar] [CrossRef] [PubMed]
- Occhiuto, F.; Zangla, G.; Samperi, S.; Palumbo, D.R.; Pino, A.; De Pasquale, R.; Circosta, C. The phytoestrogenic isoflavones from Trifolium pratense L. (Red clover) protects human cortical neurons from glutamate toxicity. Phytomedicine 2008, 15, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.H.; Liao, C.C.; Chen, Y.; Syu, J.P.; Jeng, C.J.; Wang, S.M. Daidzein induces neuritogenesis in DRG neuronal cultures. J. Biomed. Sci. 2012, 19, 80. [Google Scholar] [CrossRef]
- Liu, E.Y.L.; Xu, M.L.; Jin, Y.; Wu, Q.; Dong, T.T.X.; Tsim, K.W.K. Genistein, a phytoestrogen in soybean, induces the expression of acetylcholinesterase via G protein-coupled receptor 30 in PC12 cells. Front. Mol. Neurosci. 2018, 11, 59. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, K.I.; Niisato, N.; Marunaka, Y. Genistein enhances the NGF-induced neurite outgrowth. Biomed. Res. 2011, 32, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chen, S.; Ma, G.; Ye, M.; Lu, G. Genistein protects dopaminergic neurons by inhibiting microglial activation. Neuroreport 2005, 16, 267–270. [Google Scholar] [CrossRef]
- Chen, H.Q.; Wang, X.J.; Jin, Z.Y.; Xu, X.M.; Zhao, J.W.; Xie, Z.J. Protective effect of isoflavones from Trifolium pratense on dopaminergic neurons. Neurosci. Res. 2008, 62, 123–130. [Google Scholar] [CrossRef]
- Subedi, L.; Ji, E.; Shin, D.; Jin, J.; Yeo, J.H.; Kim, S.Y. Equol, a Dietary Daidzein Gut Metabolite Attenuates Microglial Activation and Potentiates Neuroprotection In Vitro. Nutrients 2017, 9, 207. [Google Scholar] [CrossRef]
- Watanabe, S.; Yamaguchi, M.; Sobue, T.; Takahashi, T.; Miura, T.; Arai, Y.; Mazur, W.; Wähälä, K.; Adlercreutz, H. Pharmacokinetics of soybean isoflavones in plasma, urine and feces of men after ingestion of 60 g baked soybean powder (kinako). J. Nutr. 1998, 128, 1710–1715. [Google Scholar] [CrossRef]
- Setchell, K.D.R.; Clerici, C. Equol: Pharmacokinetics and Biological Actions. J. Nutr. 2010, 140, 1363S–1368S. [Google Scholar] [CrossRef]
- Hüser, S.; Guth, S.; Joost, H.G.; Soukup, S.T.; Köhrle, J.; Kreienbrock, L.; Diel, P.; Lachenmeier, D.W.; Eisenbrand, G.; Vollmer, G.; et al. Effects of isoflavones on breast tissue and the thyroid hormone system in humans: A comprehensive safety evaluation. Arch. Toxicol. 2018, 92, 2703–2748. [Google Scholar] [CrossRef]
- Ahn-jarvis, J.H.; Clinton, S.K.; Grainger, E.M.; Riedl, K.M.; Schwartz, S.J.; Lee, M.T.; Cruz-cano, R.; Young, G.S.; Gregory, B. Isoflavone pharmacokinetics and metabolism after consumption of a standardized soy and soy-almond bread in men with asymptomatic prostate cancer. Cancer Prev. Res. 2015, 8, 1045–1054. [Google Scholar] [CrossRef] [PubMed]
- Hosoda, K.; Furuta, T.; Yokokawa, A.; Ogura, K.; Hiratsuka, A.; Ishii, K. Plasma profiling of intact isoflavone metabolites by high-performance liquid chromatography and mass spectrometric identification of flavone glycosides daidzin and genistin in human plasma after administration of kinako. Drug Metab. Dispos. 2008, 36, 1485–1495. [Google Scholar] [CrossRef] [PubMed]
- Ariyani, W.; Iwasaki, T.; Miyazaki, W.; Khongorzul, E.; Nakajima, T.; Kameo, S.; Koyama, H.; Tsushima, Y.; Koibuchi, N. Effects of gadolinium-based contrast agents on thyroid hormone receptor action and thyroid hormone-induced cerebellar purkinje cell morphogenesis. Front. Endocrinol. 2016, 7, 115. [Google Scholar] [CrossRef] [PubMed]
- Ariyani, W.; Miyazaki, W.; Tsushima, Y.; Koibuchi, N. Gadolinium-based contrast agent accelerates the migration of astrocyte via integrin αvβ3 signaling pathway. Sci. Rep. 2022, 12, 5850. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ariyani, W.; Amano, I.; Koibuchi, N. Isoflavones Mediate Dendritogenesis Mainly through Estrogen Receptor α. Int. J. Mol. Sci. 2023, 24, 9011. https://doi.org/10.3390/ijms24109011
Ariyani W, Amano I, Koibuchi N. Isoflavones Mediate Dendritogenesis Mainly through Estrogen Receptor α. International Journal of Molecular Sciences. 2023; 24(10):9011. https://doi.org/10.3390/ijms24109011
Chicago/Turabian StyleAriyani, Winda, Izuki Amano, and Noriyuki Koibuchi. 2023. "Isoflavones Mediate Dendritogenesis Mainly through Estrogen Receptor α" International Journal of Molecular Sciences 24, no. 10: 9011. https://doi.org/10.3390/ijms24109011
APA StyleAriyani, W., Amano, I., & Koibuchi, N. (2023). Isoflavones Mediate Dendritogenesis Mainly through Estrogen Receptor α. International Journal of Molecular Sciences, 24(10), 9011. https://doi.org/10.3390/ijms24109011





