Mucin-Phenotype and Expression of the Protein V-Set and Immunoglobulin Domain Containing 1 (VSIG1): New Insights into Gastric Carcinogenesis
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Ho, S.W.T.; Tan, P. Dissection of gastric cancer heterogeneity for precision oncology. Cancer Sci. 2019, 110, 3405–3414. [Google Scholar] [CrossRef]
- Chia, N.-Y.; Tan, P. Molecular classification of gastric cancer. Ann. Oncol. 2016, 27, 763–769. [Google Scholar] [CrossRef]
- Laurén, P. The Two Histological Main Types of Gastric Carcinoma: Diffuse and so-called Intestinal-Type Carcinoma. An Attempt at a Histo-Clinical Classification. Acta Pathol. Microbiol. Scand. 1965, 64, 31–49. [Google Scholar] [CrossRef]
- Xiao, L.-J.; Zhao, S.; Zhao, E.-H.; Zheng, X.; Gou, W.-F.; Xing, Y.-N.; Takano, Y.; Zheng, H.-C. Clinicopathological and prognostic significance of MUC-2, MUC-4 and MUC-5AC expression in japanese gastric carcinomas. Asian Pac. J. Cancer Prev. 2012, 13, 6447–6453. [Google Scholar] [CrossRef]
- Javanbakht, M.; Akhavanmoghadam, J.; Talaei, A.J.; Aghyani, M.; Mozafari, M.; Khedmat, L.; Mohebbi, M. Differential expression of two genes Oct-4 and MUC5AC associates with poor outcome in patients with gastric cancer. Clin. Exp. Pharmacol. Physiol. 2017, 44, 1099–1105. [Google Scholar] [CrossRef]
- Kim, S.M.; Kwon, C.H.; Shin, N.; Park, D.Y.; Moon, H.J.; Kim, G.H.; Jeon, T.Y. Decreased Muc5AC expression is associated with poor prognosis in gastric cancer. Int. J. Cancer 2013, 134, 114–124. [Google Scholar] [CrossRef]
- Sriamornsak, P.; Wattanakorn, N.; Takeuchi, H. Study on the mucoadhesion mechanism of pectin by atomic force microscopy and mucin-particle method. Carbohydr. Polym. 2010, 79, 54–59. [Google Scholar] [CrossRef]
- Crater, J.S.; Carrier, R.L. Barrier Properties of Gastrointestinal Mucus to Nanoparticle Transport. Macromol. Biosci. 2010, 10, 1473–1483. [Google Scholar] [CrossRef]
- Figueiras, A.; Vieira, A.; Simoes, S.; Veiga, F. Mucoadhesive Buccal Systems as a Novel Strategy for Anti-Inflammatory Drugs Administration. Anti-Inflamm. Anti-Allergy Agents Med. Chem. 2012, 10, 190–202. [Google Scholar] [CrossRef]
- Santos-Silva, F.; Fonseca, A.; Caffrey, T.; Carvalho, F.; Mesquita, P.; Reis, C.; Almeida, R.; David, L.; Hollingsworth, M.A. Thomsen-Friedenreich antigen expression in gastric carcinomas is associated with MUC1 mucin VNTR polymorphism. Glycobiology 2005, 15, 511–517. [Google Scholar] [CrossRef]
- Pinto-De-Sousa, J.; David, L.; Reis, C.; Gomes, R.; Silva, L.; Pimenta, A. Mucins MUC1, MUC2, MUC5AC and MUC6 expression in the evaluation of differentiation and clinico-biological behaviour of gastric carcinoma. Virchows Arch. 2002, 440, 304–310. [Google Scholar] [CrossRef]
- Mozgovoĭ, S.I.; Livzan, M.A.; Predvechnaia, I.K.; Kononov, A.V. Proliferative and mucin-producing activities in the foci of intestinal meta-plasia associated with chronic atrophic gastritis and gastric ulcer. Vestn. Ross. Akad. Meditsinskikh Nauk. 2010, 7, 15–18. [Google Scholar]
- Kocer, B.; Ulas, M.; Ustundag, Y.; Erdogan, S.; Karabeyoglu, M.; Yldrm, O.; Unal, B.; Cengiz, O.; Soran, A. A Confirmatory Report for the Close Interaction of Helicobacter pylori With Gastric Epithelial MUC5AC Expression. J. Clin. Gastroenterol. 2004, 38, 496–502. [Google Scholar] [CrossRef]
- Chen, Y.; Pan, K.; Li, S.; Xia, J.; Wang, W.; Chen, J.; Zhao, J.; Lü, L.; Wang, D.; Pan, Q.; et al. Decreased expression of V-set and immunoglobulin domain containing 1 (VSIG1) is associated with poor prognosis in primary gastric cancer. J. Surg. Oncol. 2012, 106, 286–293. [Google Scholar] [CrossRef]
- Inoue, Y.; Matsuura, S.; Yoshimura, K.; Iwashita, Y.; Kahyo, T.; Kawase, A.; Tanahashi, M.; Maeda, M.; Ogawa, H.; Inui, N.; et al. Characterization of V-set and immunoglobulin domain containing 1 exerting a tumor suppressor function in gastric, lung, and esophageal cancer cells. Cancer Sci. 2017, 108, 1701–1714. [Google Scholar] [CrossRef]
- Chandrashekar, D.S.; Bashel, B.; Balasubramanya, S.A.H.; Creighton, C.J.; Ponce-Rodriguez, I.; Chakravarthi, B.V.S.K.; Varambally, S. UALCAN: A portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia 2017, 19, 649–658. [Google Scholar] [CrossRef]
- Chang, L.; Zhou, G.; Soufan, O.; Xia, J. miRNet 2.0: Network-based visual analytics for miRNA functional analysis and systems biology. Nucleic Acids Res. 2020, 48, W244–W251. [Google Scholar] [CrossRef]
- Machlowska, J.; Baj, J.; Sitarz, M.; Maciejewski, R.; Sitarz, R. Ganstric cancer: Epidemiology, risk factors, classification, genomic characteristics, and treatment strategies. Int. J. Mol. Sci. 2020, 21, 4012. [Google Scholar] [CrossRef]
- Yusefi, A.R.; Lankarani, K.B.; Bastani, P.; Radinmanesh, M.; Kavosi, Z. Risk Factors for Gastric Cancer: A Systematic Review. Asian Pac. J. Cancer Prev. 2018, 19, 591–603. [Google Scholar] [CrossRef]
- Tatematsu, M.; Tsukamoto, T.; Inada, K. Stem cells and gastric cancer: Role of gastric and intestinal mixed intestinal metaplasia. Cancer Sci. 2003, 94, 135–141. [Google Scholar] [CrossRef]
- Kawachi, T.; Kogure, K.; Tanaka, N.; Tokunaga, A.; Sugimura, T. Studies of intestinal metaplasia in the gastric mucosa by detection of disaccharidases with “Tes-Tape”. J. Natl. Cancer Inst. 1974, 53, 19–30. [Google Scholar] [CrossRef]
- Kim, D.H.; Shin, N.; Kim, G.H.; Song, G.A.; Jeon, T.-Y.; Kim, D.-H.; Lauwers, G.Y.; Park, D.Y. Mucin Expression in Gastric Cancer: Reappraisal of Its Clinicopathologic and Prognostic Significance. Arch. Pathol. Lab. Med. 2013, 137, 1047–1053. [Google Scholar] [CrossRef]
- Saad, R.S.; Ghorab, Z.; Khalifa, M.A.; Xu, M. CDX2 as a marker for intestinal differentiation: Its utility and limitations. World J. Gastrointest. Surg. 2011, 3, 159–166. [Google Scholar] [CrossRef]
- Satala, C.-B.; Jung, I.; Kovacs, Z.; Staden, R.-I.S.-V.; Molnar, C.; Bara, T.; Patrichi, A.-I.; Gurzu, S. V-set and immunoglobulin domain containing 1 (VSIG1) as an emerging target for epithelial–mesenchymal transition of gastric cancer. Sci. Rep. 2022, 12, 16241. [Google Scholar] [CrossRef]
- VSIG1 V-Set and Immunoglobulin Domain Containing 1 [Homo Sapiens (Human)]—Gene—NCBI. Available online: https://www.ncbi.nlm.nih.gov/gene?Db=gene&Cmd=DetailsSearch&Term=340547 (accessed on 28 December 2022).
- Scanlan, M.J.; Ritter, G.; Yin, B.W.T.; Williams, C.; Cohen, L.S.; Coplan, K.A.; Fortunato, S.R.; Frosina, D.; Lee, S.-Y.; Murray, A.E.; et al. Glycoprotein A34, a novel target for antibody-based cancer immunotherapy. Cancer Immun. 2006, 6, 2. [Google Scholar]
- Kim, J.H.; Kim, K.-J.; Rhee, Y.-Y.; Bae, J.M.; Cho, N.-Y.; Lee, H.S.; Kang, G.H. Gastric-type expression signature in serrated pathway–associated colorectal tumors. Hum. Pathol. 2015, 46, 643–656. [Google Scholar] [CrossRef]
- Oidovsambuu, O.; Nyamsuren, G.; Liu, S.; Göring, W.; Engel, W.; Adham, I.M. Adhesion Protein VSIG1 Is Required for the Proper Differentiation of Glandular Gastric Epithelia. PLoS ONE 2011, 6, e25908. [Google Scholar] [CrossRef]
- Gurzu, S.; Sugimura, H.; Szederjesi, J.; Szodorai, R.; Braicu, C.; Kobori, L.; Fodor, D.; Jung, I. Interaction between cadherins, vimentin, and V-set and immunoglobulin domain containing 1 in gastric-type hepatocellular carcinoma. Histochem. Cell Biol. 2021, 156, 377–390. [Google Scholar] [CrossRef]
- Kende, A.I.; Carr, N.J.; Sobin, L.H. Expression of cytokeratins 7 and 20 in carcinomas of the gastrointestinal tract. Histopathology 2003, 42, 137–140. [Google Scholar] [CrossRef]
- Gulmann, C.; Counihan, I.; Grace, A.; Patchett, S.; Leen, E.; Leader, M.; Kay, E. Cytokeratin 7/20 and mucin expression patterns in oesophageal, cardia and distal gastric adenocarcinomas. Histopathology 2003, 43, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Marín, F.; Bonet, C.; Muñoz, X.; García, N.; Pardo, M.L.; Ruiz-Liso, J.M.; Alonso, P.; Capellà, G.; Sanz-Anquela, J.M.; González, C.A.; et al. Genetic variation in MUC1, MUC2 and MUC6 genes and evolution of gastric cancer precursor lesions in a long-term follow-up in a high-risk area in Spain. Carcinogenesis 2012, 5, 1072–1080. [Google Scholar] [CrossRef] [PubMed]
- Sokolova, O.; Naumann, M. NF-κB Signaling in Gastric Cancer. Toxins 2017, 9, 119. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.-H.; Zheng, J.-B.; Qi, J.; Yang, K.; Wu, Y.-H.; Wang, K.; Wang, C.-B.; Sun, X.-J. Bile acids promote gastric intestinal metaplasia by upregulating CDX2 and MUC2 expression via the FXR/NF-κB signalling pathway. Int. J. Oncol. 2019, 54, 879–892. [Google Scholar] [CrossRef] [PubMed]
- Kolell, K.J.; Crawford, D.L. Evolution of Sp Transcription Factors. Mol. Biol. Evol. 2002, 19, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Peng, K.; Li, H.; Zhuang, R.; Wang, Y.; Li, W.; Yu, S.; Liang, L.; Xu, X.; Liu, T. SP1 upregulated FoxO3a promotes tumor progression in colorectal cancer. Oncol. Rep. 2018, 39, 2235–2242. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Chen, Q.; Wang, Y.; Zhang, N.; Meng, P.; Liu, T.; Bu, Y. Sp1 Mediates the Constitutive Expression and Repression of the PDSS2 Gene in Lung Cancer Cells. Genes 2019, 10, 977. [Google Scholar] [CrossRef]
- Liu, Y.; Du, Y.; Hu, X.; Zhao, L.; Xia, W. Up-regulation of ceRNA TINCR by SP1 contributes to tumorigenesis in breast cancer. BMC Cancer 2018, 18, 367. [Google Scholar] [CrossRef]
- Vellingiri, B.; Iyer, M.; Subramaniam, M.D.; Jayaramayya, K.; Siama, Z.; Giridharan, B.; Narayanasamy, A.; Dayem, A.A.; Cho, S.-G. Understanding the Role of the Transcription Factor Sp1 in Ovarian Cancer: From Theory to Practice. Int. J. Mol. Sci. 2020, 21, 1153. [Google Scholar] [CrossRef]
- Shi, S.; Zhang, Z. Role of Sp1 expression in gastric cancer: A meta-analysis and bioinformatics analysis. Oncol. Lett. 2019, 18, 4126–4135. [Google Scholar] [CrossRef]
- Cai, Y.; Yi, M.; Chen, D.; Liu, J.; Guleng, B.; Ren, J.; Shi, H. Trefoil factor family 2 expression inhibits gastric cancer cell growth and invasion in vitro via interactions with the transcription factor Sp3. Int. J. Mol. Med. 2016, 38, 1474–1480. [Google Scholar] [CrossRef]
- Jiang, W.; Jin, Z.; Zhou, F.; Cui, J.; Wang, L.; Wang, L. High co-expression of Sp1 and HER-2 is correlated with poor prognosis of gastric cancer patients. Surg. Oncol. 2015, 24, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Gurzu, S.; Kadar, Z.; Sugimura, H.; Orlowska, J.; Bara, T.; Szederjesi, J.; Jung, I. Maspin-related Orchestration of Aggressiveness of Gastric Cancer. Appl. Immunohistochem. Mol. Morphol. 2016, 24, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Nagtegaal, I.D.; Odze, R.D.; Klimstra, D.; Paradis, V.; Rugge, M.; Schirmacher, P.; Washington, K.M.; Carneiro, F.; Cree, I.A.; the WHO Classification of Tumours Editorial Board. The 2019 WHO classification of tumours of the digestive system. Histopathology 2020, 76, 182–188. [Google Scholar] [CrossRef]
- Gurzu, S.; Sugimura, H.; Orlowska, J.; Szederjesi, J.; Szentirmay, Z.; Bara, T.; Fetyko, A.; Jung, I. Proposal of a Dukes-Mac-Like Staging System for Gastric Cancer. J. Investig. Med. 2017, 65, 316–322. [Google Scholar] [CrossRef] [PubMed]
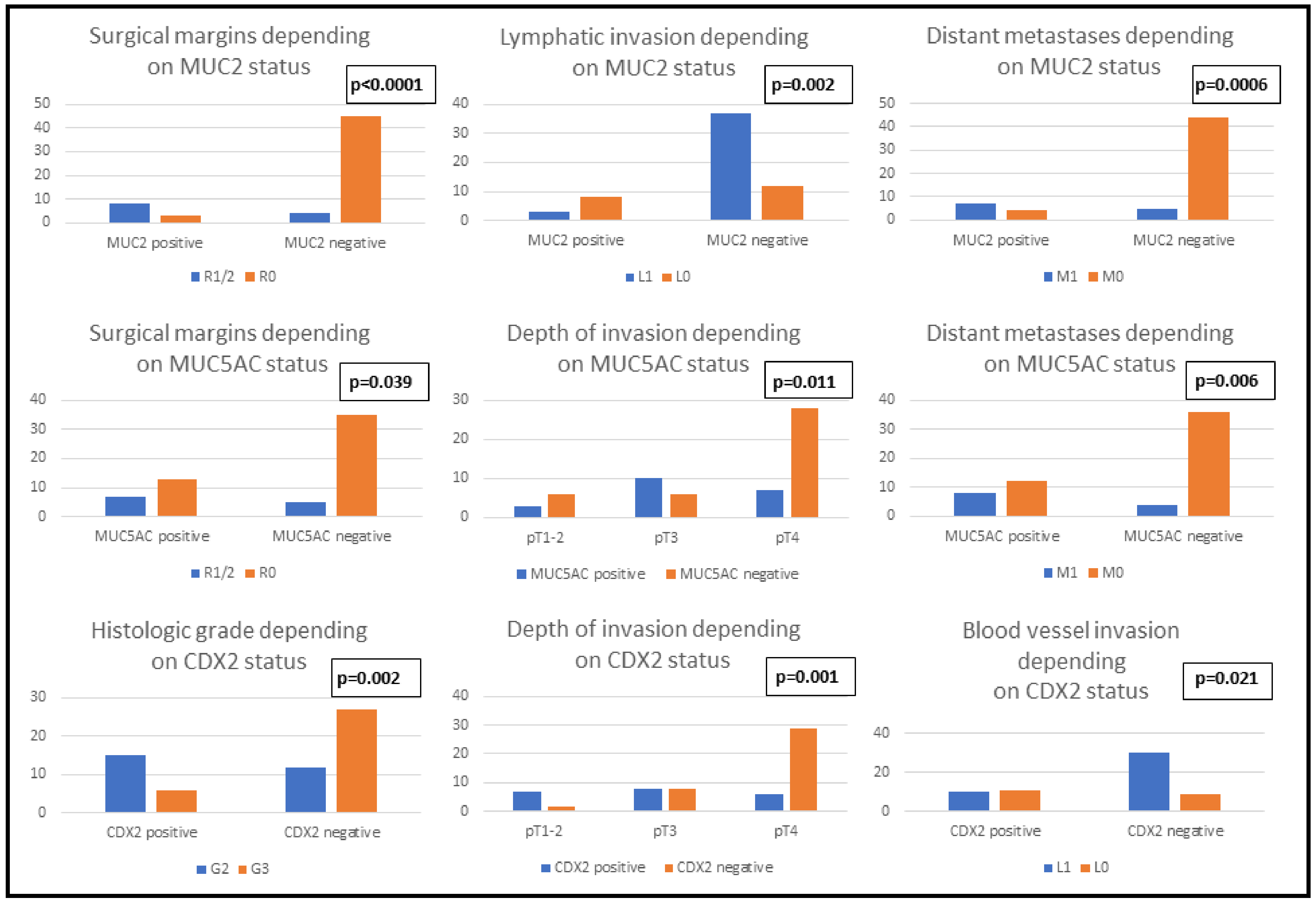
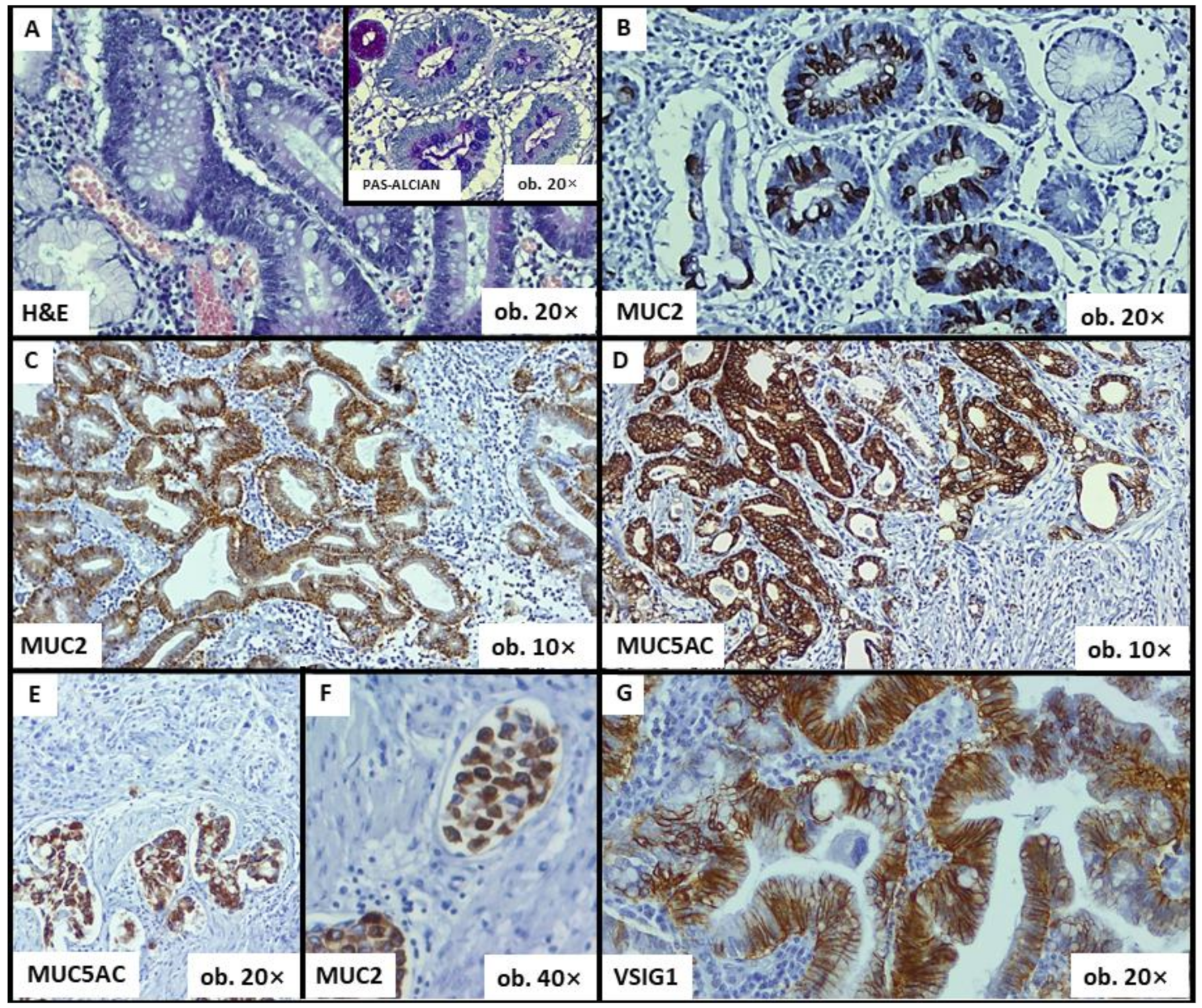
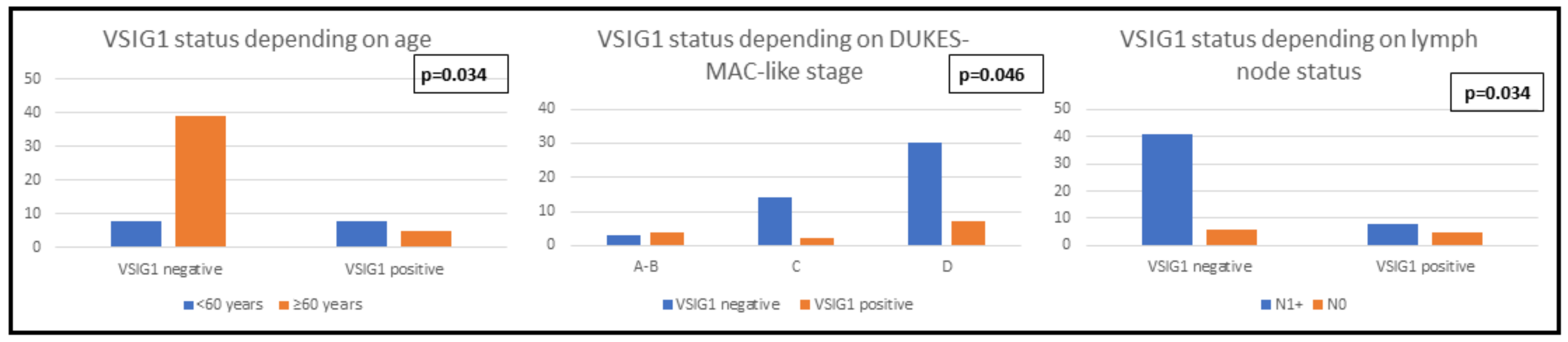
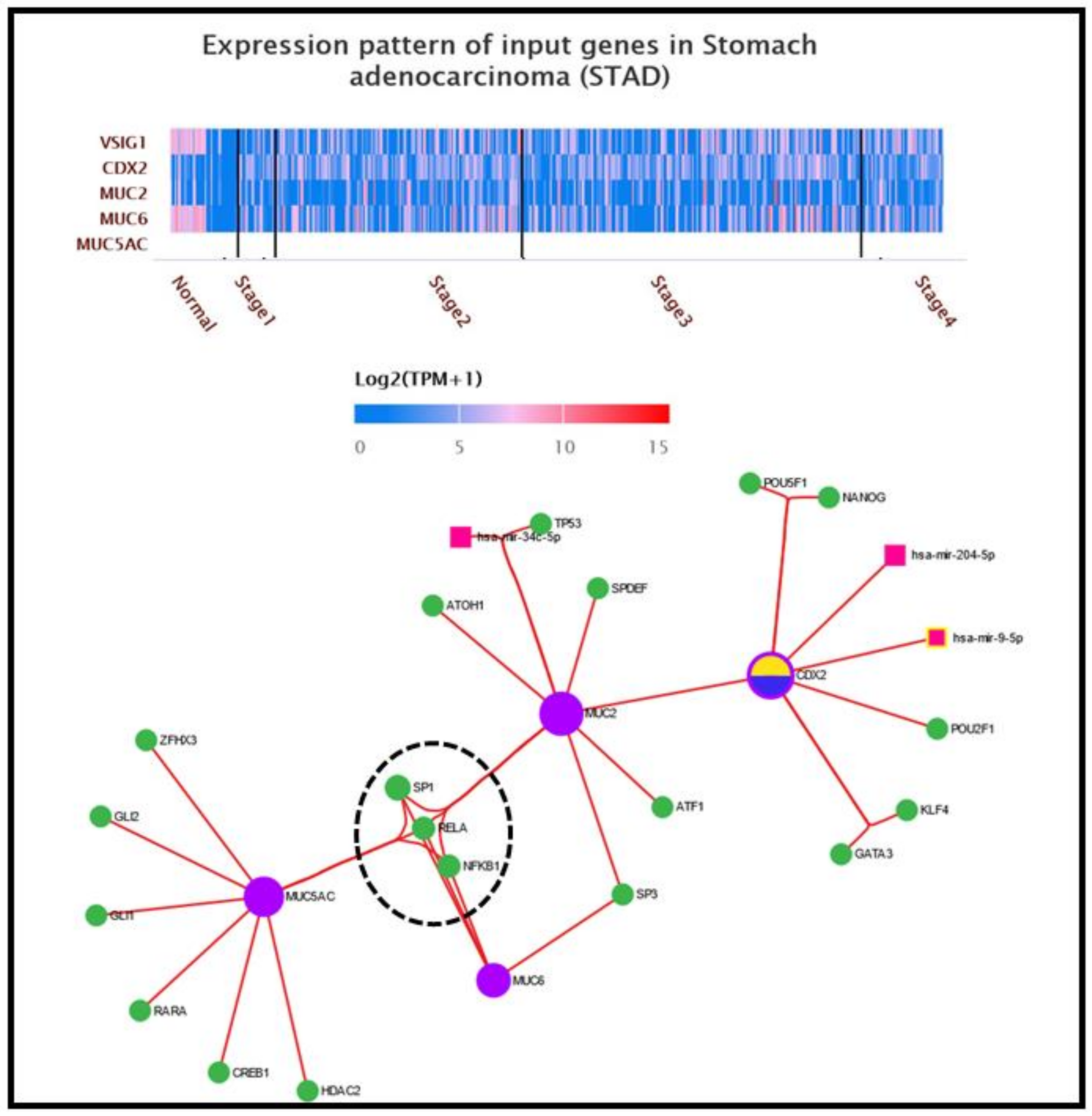

| Parameter | Intestinal Metaplasia (n = 16) | Without Metaplasia (n = 44) | p-Value | |
|---|---|---|---|---|
| Sex | Male | 31.25% (5) | 95.45% (42) | <0.0001 |
| Female | 68.75% (11) | 4.55% (2) | ||
| Age (years) | <60 | 62.5% (10) | 13.63% (6) | 0.001 |
| ≥60 | 37.5% (6) | 86.37% (38) | ||
| Histologic Grade | G2 | 43.75% (7) | 45.45% (20) | 0.906 |
| G3 | 56.25% (9) | 54.55% (24) | ||
| Tumor Stage | pT1-2 | 18.75% (3) | 13.63% (6) | 0.727 |
| pT3 | 31.25% (5) | 25% (11) | ||
| pT4 | 50% (8) | 61.37% (27) | ||
| Dukes-MAC-like Stage | A-B | 12.5% (2) | 11.36% (5) | 0.865 |
| C | 31.25 (5) | 25% (11) | ||
| D | 56.25% (9) | 63.64% (28) | ||
| Surgical Margins | Positive (R1/R2) | 18.75% (3) | 20.45% (9) | 0.883 |
| Negative (R0) | 81.25% (13) | 79.55% (35) | ||
| Lymphatic Invasion | Present (L1) | 62.5% (10) | 68.18% (30) | 0.679 |
| Absent (L0) | 37.5% (6) | 31.82% (14) | ||
| Blood Vessel Invasion | Present (V1) | 43.75% (7) | 54.55% (24) | 0.459 |
| Absent (V0) | 56.25% (9) | 45.45% (20) | ||
| Lymph Node Metastases | Present (N1+) | 87.5% (14) | 79.55% (35) | 0.481 |
| Absent (N0) | 12.5% (2) | 20.45% (9) | ||
| Distant Metastases | Present (M1) | 25% (4) | 18.18% (8) | 0.559 |
| Absent (M0) | 75% (12) | 81.82% (36) | ||
| Parameter | MUC2 Positive (n = 11) | MUC2 Negative (n = 49) | p-Value | MUC5AC Positive (n = 20) | MUC5AC Negative (n = 40) | p-Value | CDX2 Positive (n = 21) | CDX2 Negative (n = 39) | p-Value | |
|---|---|---|---|---|---|---|---|---|---|---|
| Sex | Male | 27.27% (3) | 89.8% (44) | <0.0001 | 70% (14) | 82.5% (33) | 0.267 | 47.62% (10) | 94.88% (37) | 0.0002 |
| Female | 72.73% (8) | 10.2% (5) | 30% (6) | 17.5% (7) | 52.38% (11) | 5.12% (2) | ||||
| Age (years) | <60 | 36.36% (4) | 24.5% (12) | 0.42 | 40% (8) | 20% (8) | 0.098 | 33.33% (7) | 23.07% (9) | 0.391 |
| ≥60 | 63.64% (7) | 75.5% (37) | 60% (12) | 80% (32) | 66.67% (14) | 76.93% (30) | ||||
| Histologic Grade | G2 | 45.45% (5) | 44.9% (22) | 0.973 | 35% (7) | 50% (20) | 0.27 | 71.42% (15) | 30.76% (12) | 0.002 |
| G3 | 54.55% (6) | 55.1% (27) | 65% (13) | 50% (20) | 28.58% (6) | 69.26% (27) | ||||
| Tumor Stage | pT1-2 | 18.18% (2) | 14.28% (7) | 0.771 | 15% (3) | 15% (6) | 0.011 | 33.33% (7) | 5.12% (2) | 0.001 |
| pT3 | 18.18% (2) | 28.56% (14) | 50% (10) | 15% (6) | 38.09% (8) | 20.51% (8) | ||||
| pT4 | 63.64% (7) | 57.16% (28) | 35% (7) | 70% (28) | 28.58% (6) | 74.37% (29) | ||||
| Dukes-MAC-like Stage | A-B | 27.27% (3) | 8.16% (4) | 0.183 | 20% (4) | 7.5% (3) | 0.14 | 14.28% (3) | 10.24% (4) | 0.018 |
| C | 27.27% (3) | 26.54% (13) | 35% (7) | 22.5% (9) | 4.76% (1) | 38.46% (15) | ||||
| D | 45.45% (5) | 65.3% (32) | 45% (9) | 70% (28) | 80.96% (17) | 51.3% (20) | ||||
| Surgical Margins | R1/2 | 72.73% (8) | 8.16% (4) | <0.0001 | 35% (7) | 12.5% (5) | 0.039 | 52.38% (11) | 2.56% (1) | <0.0001 |
| R0 | 27.27% (3) | 91.84% (45) | 65% (13) | 87.5% (35) | 47.62% (10) | 97.44% (38) | ||||
| Lymphatic Invasion | L1 | 27.27% (3) | 75.5% (37) | 0.002 | 50% (10) | 75% (30) | 0.052 | 61.91% (13) | 69.24% (27) | 0.565 |
| L0 | 72.73% (8) | 24.5% (12) | 50% (10) | 25% (10) | 38.09% (8) | 30.76% (12) | ||||
| Blood Vessel Invasion | V1 | 45.45% (5) | 53.06% (26) | 0.648 | 55% (11) | 50% (20) | 0.714 | 47.62% (10) | 76.92% (30) | 0.021 |
| V0 | 54.55% (6) | 46.94% (23) | 45% (9) | 50% (20) | 52.38% (11) | 23.08% (9) | ||||
| Lymph Node Metastases | N1+ | 90.9% (10) | 79.6% (39) | 0.38 | 85% (17) | 80% (32) | 0.637 | 71.42% (15) | 81.17% (34) | 0.132 |
| N0 | 9.1% (1) | 20.4% (10) | 15% (3) | 20% (8) | 28.58% (6) | 12.83% (5) | ||||
| Distant Metastases | M1 | 63.64% (7) | 10.2% (5) | 0.0006 | 40% (8) | 10% (4) | 0.006 | 28.58% (6) | 15.38% (6) | 0.223 |
| M0 | 36.36% (4) | 89.8% (44) | 60% (12) | 90% (36) | 71.42% (15) | 84.62% (33) | ||||
| Parameter | VSIG1 Negative (n = 47) | VSIG1 Positive (n = 13) | p-Values | |
|---|---|---|---|---|
| Sex | Male | 78.72% (37) | 76.92% (10) | 0.754 |
| Female | 21.28% (10) | 23.08% (3) | ||
| Age (years) | <60 | 17.02% (8) | 61.53% (8) | 0.034 |
| ≥60 | 82.98% (39) | 38.47% (5) | ||
| Histologic Grade | G2 | 44.68% (21) | 46.15% (6) | 0.924 |
| G3 | 55.32% (26) | 53.85% (7) | ||
| Tumor Stage | pT1-2 | 10.63% (5) | 30.76% (4) | 0.165 |
| pT3 | 29.78% (14) | 15.38% (2) | ||
| pT4 | 59.59% (28) | 53.85% (7) | ||
| Dukes-MAC-like Stage | A-B | 6.38% (3) | 30.76% (4) | 0.046 |
| C | 29.78% (14) | 15.38% (2) | ||
| D | 63.82% (30) | 53.85% (7) | ||
| Surgical Margins | R1/2 | 19.14% (9) | 23.08% (3) | 0.754 |
| R0 | 80.86% (38) | 76.92% (10) | ||
| Lymphatic Invasion | L1 | 65.95% (31) | 69.24% (9) | 0.824 |
| L0 | 34.05% (16) | 30.76% (4) | ||
| Blood Vessel Invasion | V1 | 48.93% (23) | 61.53% (8) | 0.421 |
| V0 | 51.07% (24) | 38.47% (5) | ||
| Lymph Node Metastases | N1+ | 87.23% (41) | 61.53% (8) | 0.034 |
| N0 | 12.76% (6) | 38.47% (5) | ||
| Distant Metastases | M1 | 19.14% (9) | 23.08% (3) | 0.754 |
| M0 | 80.86% (38) | 76.92% (10) | ||
| MUC2 | Negative | 85.1% (40) | 69.24% (9) | 0.368 |
| Positive | 14.9% (7) | 30.76% (4) | ||
| MUC5AC | Negative | 61.7% (29) | 84.62% (11) | 0.049 |
| Positive | 38.3% (18) | 15.38% (2) | ||
| CDX2 | Negative | 76.6% (36) | 23.08% (3) | 0.405 |
| Positive | 23.4% (11) | 76.92% (10) | ||
| Immunohistochemical Marker | With Intestinal Metaplasia (n = 16) | Without Intestinal Metaplasia (n = 44) | p-Values | |
|---|---|---|---|---|
| VSIG1 | Negative | 62.5% (10) | 84.1% (37) | 0.046 |
| Positive | 37.5% (6) | 15.9% (7) | ||
| MUC2 | Negative | 62.5% (10) | 88.63% (39) | 0.02 |
| Positive | 37.5% (6) | 11.37% (5) | ||
| CDX2 | Negative | 25% (4) | 79.55% (35) | 0.0009 |
| Positive | 75% (12) | 20.45% (9) | ||
| MUC5AC | Negative | 68.75% (11) | 65.9% (29) | 0.836 |
| Positive | 31.25% (5) | 34.1% (15) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Satala, C.-B.; Jung, I.; Gurzu, S. Mucin-Phenotype and Expression of the Protein V-Set and Immunoglobulin Domain Containing 1 (VSIG1): New Insights into Gastric Carcinogenesis. Int. J. Mol. Sci. 2023, 24, 8697. https://doi.org/10.3390/ijms24108697
Satala C-B, Jung I, Gurzu S. Mucin-Phenotype and Expression of the Protein V-Set and Immunoglobulin Domain Containing 1 (VSIG1): New Insights into Gastric Carcinogenesis. International Journal of Molecular Sciences. 2023; 24(10):8697. https://doi.org/10.3390/ijms24108697
Chicago/Turabian StyleSatala, Catalin-Bogdan, Ioan Jung, and Simona Gurzu. 2023. "Mucin-Phenotype and Expression of the Protein V-Set and Immunoglobulin Domain Containing 1 (VSIG1): New Insights into Gastric Carcinogenesis" International Journal of Molecular Sciences 24, no. 10: 8697. https://doi.org/10.3390/ijms24108697
APA StyleSatala, C.-B., Jung, I., & Gurzu, S. (2023). Mucin-Phenotype and Expression of the Protein V-Set and Immunoglobulin Domain Containing 1 (VSIG1): New Insights into Gastric Carcinogenesis. International Journal of Molecular Sciences, 24(10), 8697. https://doi.org/10.3390/ijms24108697







