Protein-Coding Region Derived Small RNA in Exosomes from Influenza A Virus–Infected Cells
Abstract
1. Introduction
2. Results
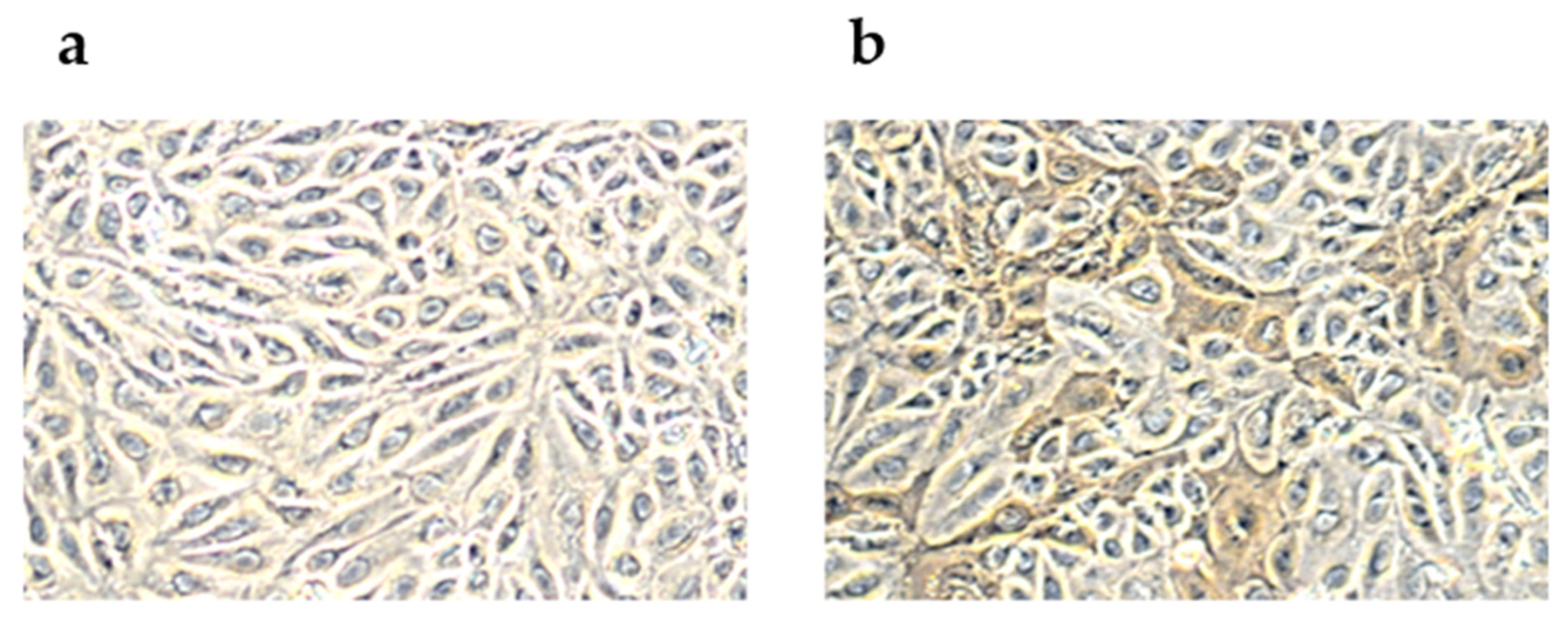
2.1. Peroxidase-Linked Assay
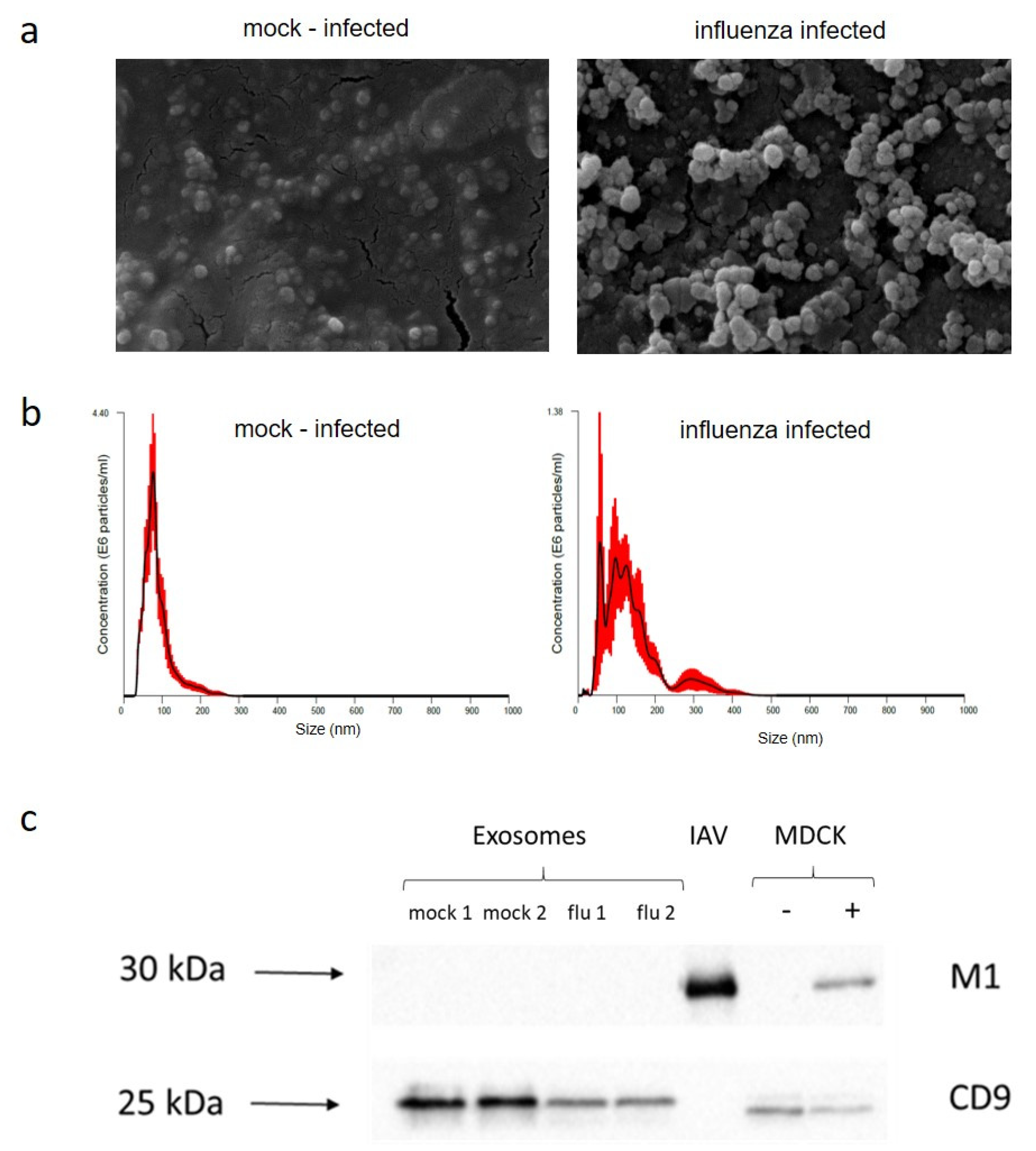
2.2. Exosome Purification
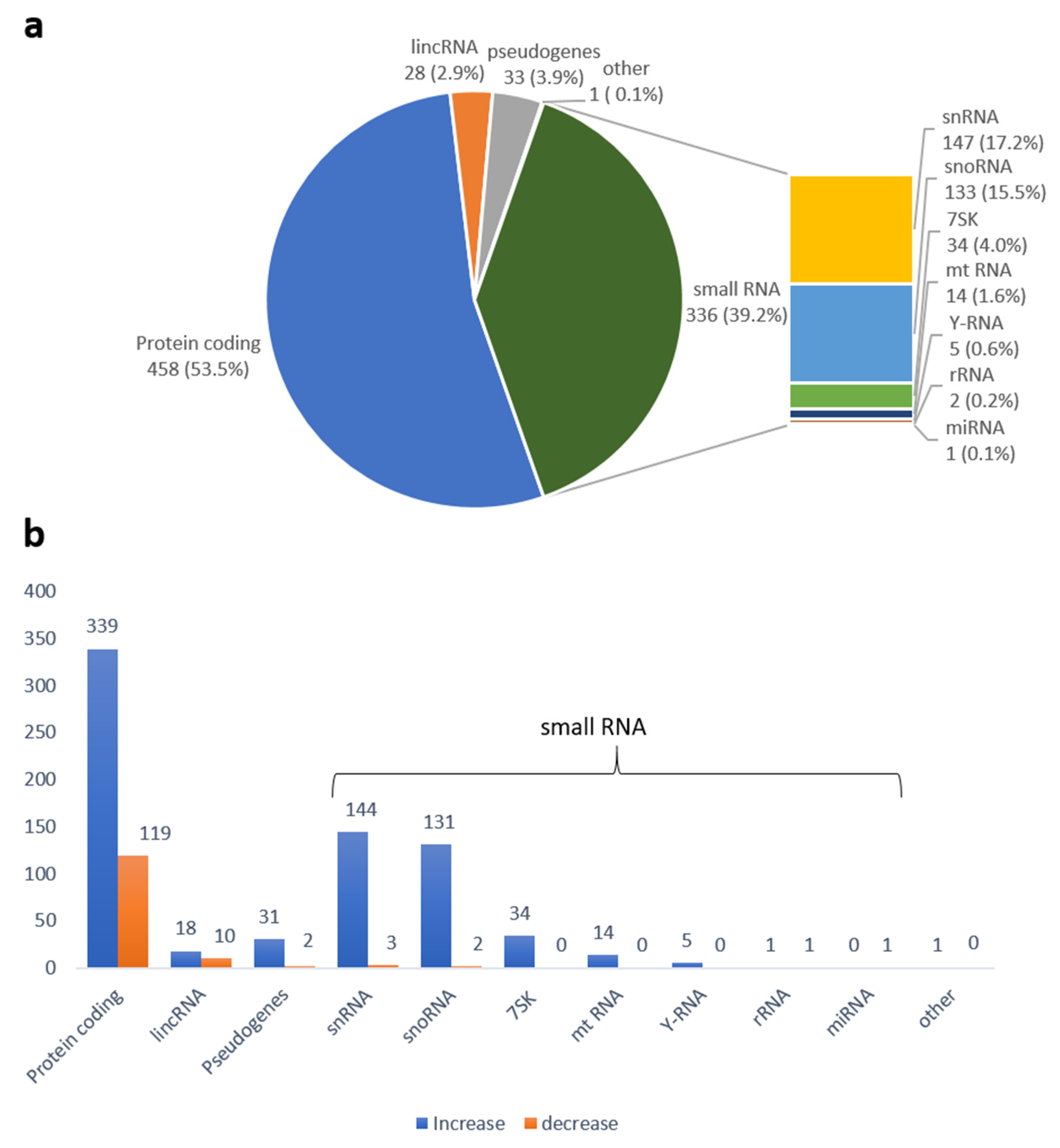
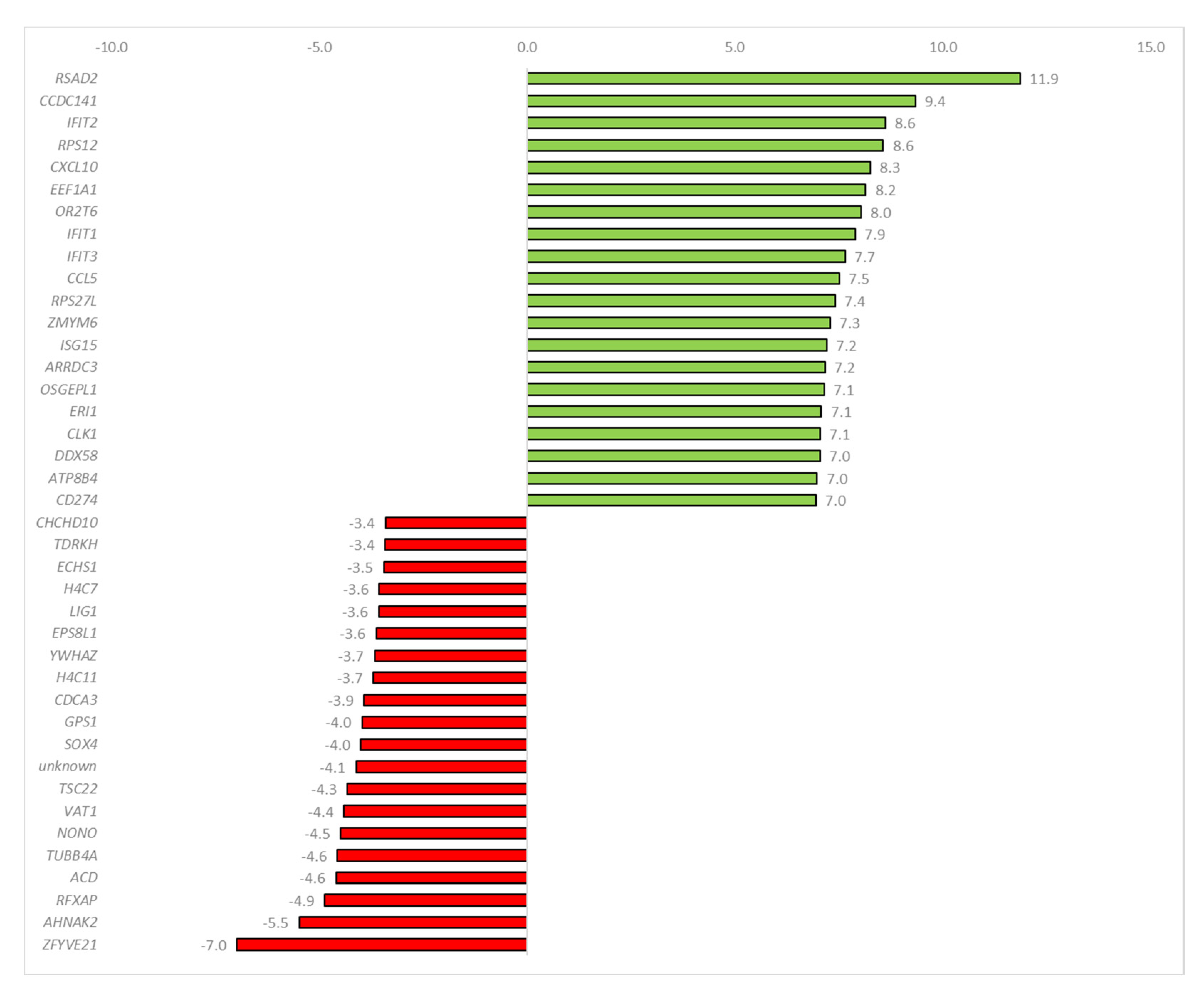
2.3. Sequencing Data Analysis
3. Discussion
4. Materials and Methods
4.1. Peroxidase-Linked Assay
4.2. Cell Culture Inoculation
4.3. Exosome Purification and RNA Extraction
4.4. Electrophoresis and Immunoblotting
4.5. Nanoparticle Tracking Analysis
4.6. Scanning Electron Microscopy
4.7. High-Throughput Sequencing and Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, J.; Guan, J.; Niu, X.; Hu, G.; Guo, S.; Li, Q.; Xie, Z.; Zhang, C.; Wang, Y. Exosomes Released from Human Induced Pluripotent Stem Cells-Derived MSCs Facilitate Cutaneous Wound Healing by Promoting Collagen Synthesis and Angiogenesis. J. Transl. Med. 2015, 13, 49. [Google Scholar] [CrossRef]
- Cui, X.; He, Z.; Liang, Z.; Chen, Z.; Wang, H.; Zhang, J. Exosomes From Adipose-Derived Mesenchymal Stem Cells Protect the Myocardium Against Ischemia/Reperfusion Injury Through Wnt/β-Catenin Signaling Pathway. J. Cardiovasc. Pharmacol. 2017, 70, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Ratajczak, J.; Miekus, K.; Kucia, M.; Zhang, J.; Reca, R.; Dvorak, P.; Ratajczak, M.Z. Embryonic Stem Cell-Derived Microvesicles Reprogram Hematopoietic Progenitors: Evidence for Horizontal Transfer of MRNA and Protein Delivery. Leukemia 2006, 20, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Men, Y.; Yelick, J.; Jin, S.; Tian, Y.; Chiang, M.S.R.; Higashimori, H.; Brown, E.; Jarvis, R.; Yang, Y. Exosome Reporter Mice Reveal the Involvement of Exosomes in Mediating Neuron to Astroglia Communication in the CNS. Nat. Commun. 2019, 10, 4136. [Google Scholar] [CrossRef] [PubMed]
- Bang, C.; Batkai, S.; Dangwal, S.; Gupta, S.K.; Foinquinos, A.; Holzmann, A.; Just, A.; Remke, J.; Zimmer, K.; Zeug, A.; et al. Cardiac Fibroblast–Derived MicroRNA Passenger Strand-Enriched Exosomes Mediate Cardiomyocyte Hypertrophy. J. Clin. Investig. 2014, 124, 2136–2146. [Google Scholar] [CrossRef] [PubMed]
- Zamani, P.; Fereydouni, N.; Butler, A.E.; Navashenaq, J.G.; Sahebkar, A. The Therapeutic and Diagnostic Role of Exosomes in Cardiovascular Diseases. Trends Cardiovasc. Med. 2019, 29, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Howitt, J.; Hill, A.F. Exosomes in the Pathology of Neurodegenerative Diseases. J. Biol. Chem. 2016, 291, 26589–26597. [Google Scholar] [CrossRef] [PubMed]
- Osaki, M.; Okada, F. Exosomes and Their Role in Cancer Progression. Yonago Acta Med. 2019, 62, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.; Liu, Y.; Liu, C.; Xiang, X.; Wang, J.; Cheng, Z.; Shah, S.V.; Zhang, S.; Zhang, L.; Zhuang, X.; et al. Immature Myeloid Cells Induced by a High-Fat Diet Contribute to Liver Inflammation. Hepatology 2009, 50, 1412–1420. [Google Scholar] [CrossRef]
- Gurung, S.; Perocheau, D.; Touramanidou, L.; Baruteau, J. The Exosome Journey: From Biogenesis to Uptake and Intracellular Signalling. Cell Commun. Signal. 2021, 19, 47. [Google Scholar] [CrossRef]
- Yoshikawa, F.S.Y.; Teixeira, F.M.E.; Sato, M.N.; Oliveira, L.M.d.S. Delivery of MicroRNAs by Extracellular Vesicles in Viral Infections: Could the News Be Packaged? Cells 2019, 8, 611. [Google Scholar] [CrossRef] [PubMed]
- Martins, S.d.T.; Alves, L.R. Extracellular Vesicles in Viral Infections: Two Sides of the Same Coin? Front. Cell. Infect. Microbiol. 2020, 10, 593170. [Google Scholar] [CrossRef] [PubMed]
- Doyle, L.; Wang, M. Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis. Cells 2019, 8, 727. [Google Scholar] [CrossRef]
- Nabhan, J.F.; Hu, R.; Oh, R.S.; Cohen, S.N.; Lu, Q. Formation and Release of Arrestin Domain-Containing Protein 1-Mediated Microvesicles (ARMMs) at Plasma Membrane by Recruitment of TSG101 Protein. Proc. Natl. Acad. Sci. USA 2012, 109, 4146–4151. [Google Scholar] [CrossRef] [PubMed]
- Wollert, T.; Hurley, J.H. Molecular Mechanism of Multivesicular Body Biogenesis by ESCRT Complexes. Nature 2010, 464, 864–869. [Google Scholar] [CrossRef]
- Neal, C.S.; Michael, M.Z.; Pimlott, L.K.; Yong, T.Y.; Li, J.Y.Z.; Gleadle, J.M. Circulating MicroRNA Expression Is Reduced in Chronic Kidney Disease. Nephrol. Dial. Transplant. 2011, 26, 3794–3802. [Google Scholar] [CrossRef]
- Hornick, N.I.; Huan, J.; Doron, B.; Goloviznina, N.A.; Lapidus, J.; Chang, B.H.; Kurre, P. Serum Exosome MicroRNA as a Minimally-Invasive Early Biomarker of AML. Sci. Rep. 2015, 5, 11295. [Google Scholar] [CrossRef]
- Wang, J.; Liu, Y.; Sun, W.; Zhang, Q.; Gu, T.; Li, G. Plasma Exosomes as Novel Biomarker for the Early Diagnosis of Gastric Cancer. CBM 2018, 21, 805–812. [Google Scholar] [CrossRef]
- Bobrie, A.; Colombo, M.; Raposo, G.; Théry, C. Exosome Secretion: Molecular Mechanisms and Roles in Immune Responses. Traffic 2011, 12, 1659–1668. [Google Scholar] [CrossRef]
- Chaput, N.; Théry, C. Exosomes: Immune Properties and Potential Clinical Implementations. Semin. Immunopathol. 2011, 33, 419–440. [Google Scholar] [CrossRef]
- O’Brien, K.; Breyne, K.; Ughetto, S.; Laurent, L.C.; Breakefield, X.O. RNA Delivery by Extracellular Vesicles in Mammalian Cells and Its Applications. Nat. Rev. Mol. Cell Biol. 2020, 21, 585–606. [Google Scholar] [CrossRef] [PubMed]
- Kosaka, N.; Iguchi, H.; Yoshioka, Y.; Takeshita, F.; Matsuki, Y.; Ochiya, T. Secretory Mechanisms and Intercellular Transfer of MicroRNAs in Living Cells. J. Biol. Chem. 2010, 285, 17442–17452. [Google Scholar] [CrossRef] [PubMed]
- Nolte-’t Hoen, E.N.M.; Buermans, H.P.J.; Waasdorp, M.; Stoorvogel, W.; Wauben, M.H.M.; ’t Hoen, P.A.C. Deep Sequencing of RNA from Immune Cell-Derived Vesicles Uncovers the Selective Incorporation of Small Non-Coding RNA Biotypes with Potential Regulatory Functions. Nucleic Acids Res. 2012, 40, 9272–9285. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, F.A.; Benoit Bouvrette, L.P.; Perras, L.; Blanchet-Cohen, A.; Garnier, D.; Rak, J.; Lécuyer, É. Comparative Transcriptomic Analysis of Human and Drosophila Extracellular Vesicles. Sci. Rep. 2016, 6, 27680. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Yuan, J.; Liu, H.; Wu, C.; Huang, D.; Zhu, J.; Guo, J.; Ma, L.; Shi, H.; Zou, Y.; et al. Dendritic Cells Derived Exosomes Migration to Spleen and Induction of Inflammation Are Regulated by CCR7. Sci. Rep. 2017, 7, 42996. [Google Scholar] [CrossRef]
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-Mediated Transfer of MRNAs and MicroRNAs Is a Novel Mechanism of Genetic Exchange between Cells. Nat. Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef]
- Dong, L.; Lin, W.; Qi, P.; Xu, M.; Wu, X.; Ni, S.; Huang, D.; Weng, W.; Tan, C.; Sheng, W.; et al. Circulating Long RNAs in Serum Extracellular Vesicles: Their Characterization and Potential Application as Biomarkers for Diagnosis of Colorectal Cancer. Cancer Epidemiol. Biomark. Prev. 2016, 25, 1158–1166. [Google Scholar] [CrossRef]
- Prieto-Vila, M.; Yoshioka, Y.; Ochiya, T. Biological Functions Driven by MRNAs Carried by Extracellular Vesicles in Cancer. Front. Cell Dev. Biol. 2021, 9, 620498. [Google Scholar] [CrossRef]
- Madison, M.N.; Jones, P.H.; Okeoma, C.M. Exosomes in Human Semen Restrict HIV-1 Transmission by Vaginal Cells and Block Intravaginal Replication of LP-BM5 Murine AIDS Virus Complex. Virology 2015, 482, 189–201. [Google Scholar] [CrossRef]
- van Dongen, H.M.; Masoumi, N.; Witwer, K.W.; Pegtel, D.M. Extracellular Vesicles Exploit Viral Entry Routes for Cargo Delivery. Microbiol. Mol. Biol. Rev. 2016, 80, 369–386. [Google Scholar] [CrossRef]
- Saad, M.H.; Badierah, R.; Redwan, E.M.; El-Fakharany, E.M. A Comprehensive Insight into the Role of Exosomes in Viral Infection: Dual Faces Bearing Different Functions. Pharmaceutics 2021, 13, 1405. [Google Scholar] [CrossRef] [PubMed]
- Meckes, D.G. Exosomal Communication Goes Viral. J. Virol. 2015, 89, 5200–5203. [Google Scholar] [CrossRef] [PubMed]
- Cone, A.S.; York, S.B.; Meckes, D.G. Extracellular Vesicles in Epstein-Barr Virus Pathogenesis. Curr. Clin. Micro Rep. 2019, 6, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Cai, X.; Yao, J.; Guo, H.; Yin, L.; Leung, W.; Xu, C. Role of Extracellular Vesicles in Influenza Virus Infection. Front. Cell. Infect. Microbiol. 2020, 10, 366. [Google Scholar] [CrossRef]
- Crenshaw, B.J.; Gu, L.; Sims, B.; Matthews, Q.L. Exosome Biogenesis and Biological Function in Response to Viral Infections. TOVJ 2018, 12, 134–148. [Google Scholar] [CrossRef]
- Hong, Y.; Truong, A.D.; Vu, T.H.; Lee, S.; Heo, J.; Kang, S.; Lillehoj, H.S.; Hong, Y.H. Profiling and Analysis of Exosomal MiRNAs Derived from Highly Pathogenic Avian Influenza Virus H5N1-Infected White Leghorn Chickens. Poult. Sci. 2022, 101, 102123. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.; Zhou, J.; Wang, H. Host MicroRNAs and Exosomes That Modulate Influenza Virus Infection. Virus Res. 2020, 279, 197885. [Google Scholar] [CrossRef]
- Turchinovich, A.; Drapkina, O.; Tonevitsky, A. Transcriptome of Extracellular Vesicles: State-of-the-Art. Front. Immunol. 2019, 10, 202. [Google Scholar] [CrossRef]
- Cunningham, F.; Achuthan, P.; Akanni, W.; Allen, J.; Amode, M.R.; Armean, I.M.; Bennett, R.; Bhai, J.; Billis, K.; Boddu, S.; et al. Ensembl 2019. Nucleic Acids Res. 2019, 47, D745–D751. [Google Scholar] [CrossRef]
- Hutchinson, E.; Fodor, E. Purification of Influenza Virions by Haemadsorption and Ultracentrifugation. Protoc. Exch. 2014. [Google Scholar] [CrossRef]
- Zabrodskaya, Y.; Plotnikova, M.; Gavrilova, N.; Lozhkov, A.; Klotchenko, S.; Kiselev, A.; Burdakov, V.; Ramsay, E.; Purvinsh, L.; Egorova, M.; et al. Exosomes Released by Influenza-Virus-Infected Cells Carry Factors Capable of Suppressing Immune Defense Genes in Naïve Cells. Viruses 2022, 14, 2690. [Google Scholar] [CrossRef] [PubMed]
- Abramowicz, A.; Story, M.D. The Long and Short of It: The Emerging Roles of Non-Coding RNA in Small Extracellular Vesicles. Cancers 2020, 12, 1445. [Google Scholar] [CrossRef] [PubMed]
- Miranda, K.C.; Bond, D.T.; Levin, J.Z.; Adiconis, X.; Sivachenko, A.; Russ, C.; Brown, D.; Nusbaum, C.; Russo, L.M. Massively Parallel Sequencing of Human Urinary Exosome/Microvesicle RNA Reveals a Predominance of Non-Coding RNA. PLoS ONE 2014, 9, e96094. [Google Scholar] [CrossRef] [PubMed]
- Jenjaroenpun, P.; Kremenska, Y.; Nair, V.M.; Kremenskoy, M.; Joseph, B.; Kurochkin, I.V. Characterization of RNA in Exosomes Secreted by Human Breast Cancer Cell Lines Using Next-Generation Sequencing. PeerJ 2013, 1, e201. [Google Scholar] [CrossRef]
- Pérez-Boza, J.; Lion, M.; Struman, I. Exploring the RNA Landscape of Endothelial Exosomes. RNA 2018, 24, 423–435. [Google Scholar] [CrossRef]
- Batagov, A.O.; Kurochkin, I.V. Exosomes Secreted by Human Cells Transport Largely MRNA Fragments That Are Enriched in the 3′-Untranslated Regions. Biol. Direct 2013, 8, 12. [Google Scholar] [CrossRef]
- Mercer, T.R.; Dinger, M.E.; Mattick, J.S. Long Non-Coding RNAs: Insights into Functions. Nat. Rev. Genet. 2009, 10, 155–159. [Google Scholar] [CrossRef]
- Fromont-Racine, M.; Senger, B.; Saveanu, C.; Fasiolo, F. Ribosome Assembly in Eukaryotes. Gene 2003, 313, 17–42. [Google Scholar] [CrossRef]
- Xu, X.; Xiong, X.; Sun, Y. The Role of Ribosomal Proteins in the Regulation of Cell Proliferation, Tumorigenesis, and Genomic Integrity. Sci. China Life Sci. 2016, 59, 656–672. [Google Scholar] [CrossRef]
- Chen, F.W.; Ioannou, Y.A. Ribosomal Proteins in Cell Proliferation and Apoptosis. Int. Rev. Immunol. 1999, 18, 429–448. [Google Scholar] [CrossRef]
- Bhavsar, R.B.; Makley, L.N.; Tsonis, P.A. The Other Lives of Ribosomal Proteins. Hum. Genom. 2010, 4, 327. [Google Scholar] [CrossRef]
- de las Heras-Rubio, A.; Perucho, L.; Paciucci, R.; Vilardell, J.; LLeonart, M.E. Ribosomal Proteins as Novel Players in Tumorigenesis. Cancer Metastasis Rev. 2014, 33, 115–141. [Google Scholar] [CrossRef] [PubMed]
- Madjar, J.J.; Simonin, D.; Masse, T.; Diaz, J.J. Persistence of Ribosomal Protein Synthesis after Infection of HeLa Cells by Herpes Simplex Virus Type 1. J. Gen. Virol. 1997, 78, 435–443. [Google Scholar] [CrossRef]
- Greco, A.; Laurent, A.-M.; Madjar, J.-J. Repression of β-Actin Synthesis and Persistence of Ribosomal Protein Synthesis after Infection of HeLa Cells by Herpes Simplex Virus Type 1 Infection Are under Translational Control. Mol. Gen. Genet. 1997, 256, 320–327. [Google Scholar] [CrossRef]
- Zhai, Y.; Franco, L.M.; Atmar, R.L.; Quarles, J.M.; Arden, N.; Bucasas, K.L.; Wells, J.M.; Niño, D.; Wang, X.; Zapata, G.E.; et al. Host Transcriptional Response to Influenza and Other Acute Respiratory Viral Infections--A Prospective Cohort Study. PLoS Pathog. 2015, 11, e1004869. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.-L.; Liu, C.-D.; You, R.-I.; Ching, Y.-H.; Liang, J.; Ke, L.; Chen, Y.-L.; Chen, H.-C.; Hsu, H.-J.; Liou, J.-W.; et al. Ribosome Protein L4 Is Essential for Epstein–Barr Virus Nuclear Antigen 1 Function. Proc. Natl. Acad. Sci. USA 2016, 113, 2229–2234. [Google Scholar] [CrossRef]
- Fatima, G.; Mathan, G.; Kumar, V. The HBx Protein of Hepatitis B Virus Regulates the Expression, Intracellular Distribution and Functions of Ribosomal Protein S27a. J. Gen. Virol. 2012, 93, 706–715. [Google Scholar] [CrossRef]
- Li, S. Regulation of Ribosomal Proteins on Viral Infection. Cells 2019, 8, 508. [Google Scholar] [CrossRef]
- Fernandez-Pol, J.A.; Hamilton, P.D.; Klos, D.J. Essential Viral and Cellular Zinc and Iron Containing Metalloproteins as Targets for Novel Antiviral and Anticancer Agents: Implications for Prevention and Therapy of Viral Diseases and Cancer. Anticancer Res 2001, 21, 931–957. [Google Scholar]
- Fernandez-Pol, J.A. Conservation of Multifunctional Ribosomal Protein Metallopanstimulin-1 (RPS27) through Complex Evolution Demonstrates Its Key Role in Growth Regulation in Archaea, Eukaryotic Cells, DNA Repair, Translation and Viral Replication. Cancer Genom. Proteom. 2011, 8, 105–126. [Google Scholar]
- Karlas, A.; Machuy, N.; Shin, Y.; Pleissner, K.-P.; Artarini, A.; Heuer, D.; Becker, D.; Khalil, H.; Ogilvie, L.A.; Hess, S.; et al. Genome-Wide RNAi Screen Identifies Human Host Factors Crucial for Influenza Virus Replication. Nature 2010, 463, 818–822. [Google Scholar] [CrossRef] [PubMed]
- Cherry, S.; Doukas, T.; Armknecht, S.; Whelan, S.; Wang, H.; Sarnow, P.; Perrimon, N. Genome-Wide RNAi Screen Reveals a Specific Sensitivity of IRES-Containing RNA Viruses to Host Translation Inhibition. Genes Dev. 2005, 19, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lu, Z.; Zhang, L.; Gao, L.; Wang, N.; Gao, X.; Wang, Y.; Li, K.; Gao, Y.; Cui, H.; et al. Ribosomal Protein L4 Interacts with Viral Protein VP3 and Regulates the Replication of Infectious Bursal Disease Virus. Virus Res. 2016, 211, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Duan, X.; Fu, M.; Liu, Y.; Wang, Y.; Li, X.; Cao, H.; Zheng, S.J. The Association of Ribosomal Protein L18 (RPL18) with Infectious Bursal Disease Virus Viral Protein VP3 Enhances Viral Replication. Virus Res. 2018, 245, 69–79. [Google Scholar] [CrossRef]
- Seo, J.-Y.; Yaneva, R.; Cresswell, P. Viperin: A Multifunctional, Interferon-Inducible Protein That Regulates Virus Replication. Cell Host Microbe 2011, 10, 534–539. [Google Scholar] [CrossRef]
- Teng, T.-S.; Foo, S.-S.; Simamarta, D.; Lum, F.-M.; Teo, T.-H.; Lulla, A.; Yeo, N.K.W.; Koh, E.G.L.; Chow, A.; Leo, Y.-S.; et al. Viperin Restricts Chikungunya Virus Replication and Pathology. J. Clin. Investig. 2012, 122, 4447–4460. [Google Scholar] [CrossRef]
- Van der Hoek, K.H.; Eyre, N.S.; Shue, B.; Khantisitthiporn, O.; Glab-Ampi, K.; Carr, J.M.; Gartner, M.J.; Jolly, L.A.; Thomas, P.Q.; Adikusuma, F.; et al. Viperin Is an Important Host Restriction Factor in Control of Zika Virus Infection. Sci. Rep. 2017, 7, 4475. [Google Scholar] [CrossRef]
- Hinson, E.R.; Cresswell, P. The Antiviral Protein, Viperin, Localizes to Lipid Droplets via Its N-Terminal Amphipathic α-Helix. Proc. Natl. Acad. Sci. USA 2009, 106, 20452–20457. [Google Scholar] [CrossRef]
- Gizzi, A.S.; Grove, T.L.; Arnold, J.J.; Jose, J.; Jangra, R.K.; Garforth, S.J.; Du, Q.; Cahill, S.M.; Dulyaninova, N.G.; Love, J.D.; et al. A Naturally Occurring Antiviral Ribonucleotide Encoded by the Human Genome. Nature 2018, 558, 610–614. [Google Scholar] [CrossRef]
- Chemudupati, M.; Kenney, A.D.; Bonifati, S.; Zani, A.; McMichael, T.M.; Wu, L.; Yount, J.S. From APOBEC to ZAP: Diverse Mechanisms Used by Cellular Restriction Factors to Inhibit Virus Infections. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2019, 1866, 382–394. [Google Scholar] [CrossRef]
- Ashley, C.; Abendroth, A.; McSharry, B.; Slobedman, B. Interferon-Independent Upregulation of Interferon-Stimulated Genes during Human Cytomegalovirus Infection Is Dependent on IRF3 Expression. Viruses 2019, 11, 246. [Google Scholar] [CrossRef]
- Horibata, S.; Teramoto, T.; Vijayarangan, N.; Kuhn, S.; Padmanabhan, R.; Vasudevan, S.; Gottesman, M.; Padmanabhan, R. Host Gene Expression Modulated by Zika Virus Infection of Human-293 Cells. Virology 2021, 552, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Dissanayake, T.K.; Schäuble, S.; Mirhakkak, M.H.; Wu, W.-L.; Ng, A.C.-K.; Yip, C.C.Y.; López, A.G.; Wolf, T.; Yeung, M.-L.; Chan, K.-H.; et al. Comparative Transcriptomic Analysis of Rhinovirus and Influenza Virus Infection. Front. Microbiol. 2020, 11, 1580. [Google Scholar] [CrossRef] [PubMed]
- Fensterl, V.; Sen, G.C. Interferon-Induced Ifit Proteins: Their Role in Viral Pathogenesis. J. Virol. 2015, 89, 2462–2468. [Google Scholar] [CrossRef]
- Imaizumi, T.; Hashimoto, S.; Sato, R.; Umetsu, H.; Aizawa, T.; Watanabe, S.; Kawaguchi, S.; Matsumiya, T.; Seya, K.; Ding, J.; et al. IFIT Proteins Are Involved in CXCL10 Expression in Human Glomerular Endothelial Cells Treated with a Toll-Like Receptor 3 Agonist. Kidney Blood Press. Res. 2021, 46, 74–83. [Google Scholar] [CrossRef]
- Xie, Y.; Liu, K.; Luo, J.; Liu, S.; Zheng, H.; Cao, L.; Li, X. Identification of DDX58 and CXCL10 as Potential Biomarkers in Acute Respiratory Distress Syndrome. DNA Cell Biol. 2019, 38, 1444–1451. [Google Scholar] [CrossRef] [PubMed]
- Haller, O.; Staeheli, P.; Schwemmle, M.; Kochs, G. Mx GTPases: Dynamin-like Antiviral Machines of Innate Immunity. Trends Microbiol. 2015, 23, 154–163. [Google Scholar] [CrossRef]
- Verhelst, J.; Parthoens, E.; Schepens, B.; Fiers, W.; Saelens, X. Interferon-Inducible Protein Mx1 Inhibits Influenza Virus by Interfering with Functional Viral Ribonucleoprotein Complex Assembly. J. Virol. 2012, 86, 13445–13455. [Google Scholar] [CrossRef]
- Perng, Y.-C.; Lenschow, D.J. ISG15 in Antiviral Immunity and Beyond. Nat. Rev. Microbiol. 2018, 16, 423–439. [Google Scholar] [CrossRef]
- Villarroya-Beltri, C.; Guerra, S.; Sánchez-Madrid, F. ISGylation—A Key to Lock the Cell Gates for Preventing the Spread of Threats. J. Cell Sci. 2017, 130, jcs.205468. [Google Scholar] [CrossRef]
- Radoshevich, L.; Impens, F.; Ribet, D.; Quereda, J.J.; Nam Tham, T.; Nahori, M.-A.; Bierne, H.; Dussurget, O.; Pizarro-Cerdá, J.; Knobeloch, K.-P.; et al. ISG15 Counteracts Listeria Monocytogenes Infection. eLife 2015, 4, e06848. [Google Scholar] [CrossRef] [PubMed]
- Ganz, T.; Nemeth, E. Regulation of Iron Acquisition and Iron Distribution in Mammals. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2006, 1763, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.; Moreira, A.; Mesquita, G.; Gomes, M. Modulation of Iron Metabolism in Response to Infection: Twists for All Tastes. Pharmaceuticals 2018, 11, 84. [Google Scholar] [CrossRef]
- Williams, A.M.; Ladva, C.N.; Leon, J.S.; Lopman, B.A.; Tangpricha, V.; Whitehead, R.D.; Armitage, A.E.; Wray, K.; Morovat, A.; Pasricha, S.-R.; et al. Changes in Micronutrient and Inflammation Serum Biomarker Concentrations after a Norovirus Human Challenge. Am. J. Clin. Nutr. 2019, 110, 1456–1464. [Google Scholar] [CrossRef]
- Zhu, Y.; Tong, L.; Nie, K.; Wiwatanaratanabutr, I.; Sun, P.; Li, Q.; Yu, X.; Wu, P.; Wu, T.; Yu, C.; et al. Host Serum Iron Modulates Dengue Virus Acquisition by Mosquitoes. Nat. Microbiol. 2019, 4, 2405–2415. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.-H.; Wang, J.-Y.; Liu, P.-Y.; Sun, J.; Wang, X.-M.; Wu, R.-H.; He, X.-T.; Tu, Z.-K.; Wang, C.-G.; Xu, H.-Q.; et al. Iron Metabolism Disorders in Patients with Hepatitis B-Related Liver Diseases. WJCC 2018, 6, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Duchemin, J.-B.; Paradkar, P.N. Iron Availability Affects West Nile Virus Infection in Its Mosquito Vector. Virol. J. 2017, 14, 103. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Read, S.A.; Shackel, N.A.; Hebbard, L.; George, J.; Ahlenstiel, G. The Role of Micronutrients in the Infection and Subsequent Response to Hepatitis C Virus. Cells 2019, 8, 603. [Google Scholar] [CrossRef]
- Schnell, S.A.; Ohtsuka, H.; Kakinuma, S.; Yoshikawa, Y.; Watanabe, K.; Orino, K. Iron and Ferritin Levels in the Serum and Milk of Bovine Leukemia Virus-Infected Dairy Cows. Front. Vet. Sci. 2015, 2, 12. [Google Scholar] [CrossRef]
- McDermid, J.M.; Jaye, A.; Schim van der Loeff, M.F.; Todd, J.; Bates, C.; Austin, S.; Jeffries, D.; Awasana, A.A.; Whittle, H.C.; Prentice, A.M. Elevated Iron Status Strongly Predicts Mortality in West African Adults With HIV Infection. JAIDS J. Acquir. Immune Defic. Syndr. 2007, 46, 498–507. [Google Scholar] [CrossRef]
- Moreira, A.C.; Silva, T.; Mesquita, G.; Gomes, A.C.; Bento, C.M.; Neves, J.V.; Rodrigues, D.F.; Rodrigues, P.N.; Almeida, A.A.; Santambrogio, P.; et al. H-Ferritin Produced by Myeloid Cells Is Released to the Circulation and Plays a Major Role in Liver Iron Distribution during Infection. Int. J. Mol. Sci. 2021, 23, 269. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wu, X.; Wu, X.; Liu, J.; Yan, Y.; Wang, F.; Li, L.; Zhou, J.; Liao, M. Iron Status Is Linked to Disease Severity after Avian Influenza Virus H7N9 Infection. Asia Pac. J. Clin. Nutr. 2020, 29, 593–602. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Xu, K.; Keiner, B.; Zhou, J.; Czudai, V.; Li, T.; Chen, Z.; Liu, J.; Klenk, H.-D.; Shu, Y.L.; et al. Influenza A Virus Replication Induces Cell Cycle Arrest in G0/G1 Phase. J. Virol. 2010, 84, 12832–12840. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Batra, J.; Stuchlik, O.; Reed, M.S.; Pohl, J.; Chow, V.T.K.; Sambhara, S.; Lal, S.K. Influenza A Virus Nucleoprotein Activates the JNK Stress-Signaling Pathway for Viral Replication by Sequestering Host Filamin A Protein. Front. Microbiol. 2020, 11, 581867. [Google Scholar] [CrossRef]
- Allam, R.; Kumar, S.V.R.; Darisipudi, M.N.; Anders, H.-J. Extracellular Histones in Tissue Injury and Inflammation. J. Mol. Med. 2014, 92, 465–472. [Google Scholar] [CrossRef]
- Hoeksema, M.; van Eijk, M.; Haagsman, H.P.; Hartshorn, K.L. Histones as Mediators of Host Defense, Inflammation and Thrombosis. Future Microbiol. 2016, 11, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Marazzi, I.; Ho, J.S.Y.; Kim, J.; Manicassamy, B.; Dewell, S.; Albrecht, R.A.; Seibert, C.W.; Schaefer, U.; Jeffrey, K.L.; Prinjha, R.K.; et al. Suppression of the Antiviral Response by an Influenza Histone Mimic. Nature 2012, 483, 428–433. [Google Scholar] [CrossRef]
- Whitfield, M.L.; Zheng, L.-X.; Baldwin, A.; Ohta, T.; Hurt, M.M.; Marzluff, W.F. Stem-Loop Binding Protein, the Protein That Binds the 3′ End of Histone MRNA, Is Cell Cycle Regulated by Both Translational and Posttranslational Mechanisms. Mol. Cell. Biol. 2000, 20, 4188–4198. [Google Scholar] [CrossRef]
- Fang, C.; Manes, T.D.; Liu, L.; Liu, K.; Qin, L.; Li, G.; Tobiasova, Z.; Kirkiles-Smith, N.C.; Patel, M.; Merola, J.; et al. ZFYVE21 Is a Complement-Induced Rab5 Effector That Activates Non-Canonical NF-ΚB via Phosphoinosotide Remodeling of Endosomes. Nat. Commun. 2019, 10, 2247. [Google Scholar] [CrossRef]
- Banus, S.; Pennings, J.; Vandebriel, R.; Wester, P.; Breit, T.; Mooi, F.; Hoebee, B.; Kimman, T. Lung Response to Bordetella Pertussis Infection in Mice Identified by Gene-Expression Profiling. Immunogenetics 2007, 59, 555–564. [Google Scholar] [CrossRef]
- Wang, D.-W.; Zheng, H.-Z.; Cha, N.; Zhang, X.-J.; Zheng, M.; Chen, M.-M.; Tian, L.-X. Down-Regulation of AHNAK2 Inhibits Cell Proliferation, Migration and Invasion Through Inactivating the MAPK Pathway in Lung Adenocarcinoma. Technol. Cancer Res. Treat. 2020, 19, 153303382095700. [Google Scholar] [CrossRef] [PubMed]
- Pink, R.C.; Wicks, K.; Caley, D.P.; Punch, E.K.; Jacobs, L.; Francisco Carter, D.R. Pseudogenes: Pseudo-Functional or Key Regulators in Health and Disease? RNA 2011, 17, 792–798. [Google Scholar] [CrossRef]
- Milligan, M.J.; Lipovich, L. Pseudogene-Derived LncRNAs: Emerging Regulators of Gene Expression. Front. Genet. 2015, 5, 476. [Google Scholar] [CrossRef] [PubMed]
- Cheetham, S.W.; Faulkner, G.J.; Dinger, M.E. Overcoming Challenges and Dogmas to Understand the Functions of Pseudogenes. Nat. Rev. Genet. 2020, 21, 191–201. [Google Scholar] [CrossRef]
- Ogawa, Y.; Tsujimoto, M.; Yanoshita, R. Next-Generation Sequencing of Protein-Coding and Long Non-Protein-Coding RNAs in Two Types of Exosomes Derived from Human Whole Saliva. Biol. Pharm. Bull. 2016, 39, 1496–1507. [Google Scholar] [CrossRef]
- Van den Broeke, A.; Van Poucke, M.; Van Zeveren, A.; Peelman, L.J. Ribosomal Protein SA and Its Pseudogenes In Ruminants: An Extremely Conserved Gene Family. Czech J. Anim. Sci. 2013, 58, 79–90. [Google Scholar] [CrossRef]
- Cristiano, L. The Pseudogenes of Eukaryotic Translation Elongation Factors (EEFs): Role in Cancer and Other Human Diseases. Genes Dis. 2022, 9, 941–958. [Google Scholar] [CrossRef] [PubMed]
- Shi, N.; Zhang, S.; Guo, Y.; Yu, X.; Zhao, W.; Zhang, M.; Guan, Z.; Duan, M. CircRNA_0050463 Promotes Influenza A Virus Replication by Sponging MiR-33b-5p to Regulate EEF1A1. Vet. Microbiol. 2021, 254, 108995. [Google Scholar] [CrossRef] [PubMed]
- O’Grady, T.; Njock, M.-S.; Lion, M.; Bruyr, J.; Mariavelle, E.; Galvan, B.; Boeckx, A.; Struman, I.; Dequiedt, F. Sorting and Packaging of RNA into Extracellular Vesicles Shape Intracellular Transcript Levels. BMC Biol. 2022, 20, 72. [Google Scholar] [CrossRef] [PubMed]
- Guduric-Fuchs, J.; O’Connor, A.; Camp, B.; O’Neill, C.L.; Medina, R.J.; Simpson, D.A. Selective Extracellular Vesicle-Mediated Export of an Overlapping Set of MicroRNAs from Multiple Cell Types. BMC Genom. 2012, 13, 357. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Yuan, T.; Tschannen, M.; Sun, Z.; Jacob, H.; Du, M.; Liang, M.; Dittmar, R.L.; Liu, Y.; Liang, M.; et al. Characterization of Human Plasma-Derived Exosomal RNAs by Deep Sequencing. BMC Genom. 2013, 14, 319. [Google Scholar] [CrossRef] [PubMed]
- Rozek, W.; Kwasnik, M.; Żmudziński, J.F. Detection of Antibodies against Equine Influenza Virus by Cell Based Enzyme-Linked Immunosorbent Assay. Bull. Vet. Inst. Pulawy 2011, 55, 569–574. [Google Scholar]
- Théry, C.; Amigorena, S.; Raposo, G.; Clayton, A. Isolation and Characterization of Exosomes from Cell Culture Supernatants and Biological Fluids. Curr. Protoc. Cell Biol. 2006, 30, 3.22.1–3.22.29. [Google Scholar] [CrossRef] [PubMed]
- World Organisation for Animal Health. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. 2022. Available online: https://www.woah.org/en/what-we-do/standards/codes-and-manuals/terrestrial-manual-online-access/ (accessed on 16 November 2022).
- Rozek, W.; Kwasnik, M.; Debski, J.; Zmudzinski, J.F. Mass Spectrometry Identification of Granins and Other Proteins Secreted by Neuroblastoma Cells. Tumor Biol. 2013, 34, 1773–1781. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A Flexible Trimmer for Illumina Sequence Data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Martin, M. Cutadapt Removes Adapter Sequences from High-Throughput Sequencing Reads. EMBnet J. 2011, 17, 10. [Google Scholar] [CrossRef]
- Liao, Y.; Smyth, G.K.; Shi, W. The R Package Rsubread Is Easier, Faster, Cheaper and Better for Alignment and Quantification of RNA Sequencing Reads. Nucleic Acids Res. 2019, 47, e47. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated Estimation of Fold Change and Dispersion for RNA-Seq Data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]

| Sample | Total Filtered Reads | Canis lupus familiaris | Influenza A/equi/Kentucky 81 | ||
|---|---|---|---|---|---|
| Reads | % of Total | Reads | % of Total | ||
| mock 1 | 51,779,137 | 43,271,948 | 83.57% | 150 | 0.00% |
| mock 2 | 41,938,351 | 36,352,398 | 86.68% | 2639 | 0.01% |
| flu 1 | 46,630,234 | 37,460,944 | 80.34% | 632,383 | 1.36% |
| flu 2 | 81,467,933 | 69,411,940 | 85.20% | 930,116 | 1.14% |
| Biotype | Mock 1 | % | Mock 2 | % | Flu 1 | % | Flu 2 | % |
|---|---|---|---|---|---|---|---|---|
| Protein-coding | 6045 | 85.38 | 5809 | 85.38 | 6315 | 84.55 | 6350 | 84.63 |
| Pseudogene | 141 | 1.99 | 127 | 1.87 | 153 | 2.05 | 153 | 2.04 |
| lincRNA | 412 | 5.82 | 410 | 6.03 | 429 | 5.74 | 430 | 5.73 |
| small RNA | 482 | 6.81 | 458 | 6.73 | 571 | 7.64 | 569 | 7.58 |
| snRNA | 141 | 1.99 | 121 | 1.78 | 181 | 2.42 | 181 | 2.41 |
| snoRNA | 216 | 3.05 | 225 | 3.31 | 246 | 3.29 | 246 | 3.28 |
| miRNA | 14 | 0.20 | 15 | 0.22 | 15 | 0.20 | 15 | 0.20 |
| rRNA | 69 | 0.97 | 68 | 1.00 | 70 | 0.94 | 69 | 0.92 |
| mtRNA | 1 | 0.01 | 0 | 0.00 | 1 | 0.01 | 1 | 0.01 |
| 7SK RNA | 28 | 0.40 | 17 | 0.25 | 44 | 0.59 | 43 | 0.57 |
| Y-RNA | 8 | 0.11 | 7 | 0.10 | 9 | 0.12 | 9 | 0.12 |
| Other a | 5 | 0.07 | 5 | 0.07 | 6 | 0.08 | 6 | 0.08 |
| Total | 7080 | 100.00 | 6804 | 100.00 | 7469 | 100.00 | 7503 | 100.00 |
| Parental Gene Name | Pseudogene | Protein-Coding Transcript | |||
|---|---|---|---|---|---|
| Ensembl Id | log2FC | Type | Ensembl Id | log2FC | |
| LMF2 | ENSCAFG00000031971 | 7.036 | U a | ENSCAFG00000031984 | −2.208 |
| RPL23A | ENSCAFG00000024043 | 6.881 | U | ENSCAFG00000015394 | 4.870 |
| RPS19 | ENSCAFG00000000177 | 6.813 | U | ENSCAFG00000028485 | 3.205 |
| RPL12 | ENSCAFG00000003544 | 6.785 | U | ENSCAFG00000020136 | 5.258 |
| EEF1A1 | ENSCAFG00000013990 | 6.762 | U | ENSCAFG00000009708 | 8.151 |
| EEF1A1 | ENSCAFG00000017221 | 3.509 | U | ||
| EEF1A1 | ENSCAFG00000002517 | 3.081 | U | ||
| EEF1A1 | ENSCAFG00000002748 | 2.978 | U | ||
| EEF1A1 | ENSCAFG00000031557 | 4.839 | U | ||
| FTH1 | ENSCAFG00000001606 | 6.071 | P b | ENSCAFG00000015901 | 3.723 |
| RPS6 | ENSCAFG00000017909 | 5.896 | U | ENSCAFG00000008776 | 4.971 |
| RPS6 | ENSCAFG00000014664 | 5.016 | U | ||
| RPL21 | ENSCAFG00000029619 | 5.644 | U | ENSCAFG00000015987 | 6.408 |
| RPL32 | ENSCAFG00000007159 | 5.549 | U | ENSCAFG00000004198 | 5.967 |
| RPL32 | ENSCAFG00000008590 | 4.968 | U | ||
| RPL32 | ENSCAFG00000018104 | 3.346 | U | ||
| RPSA | ENSCAFG00000016319 | 5.543 | P | ENSCAFG00000005101 | 4.353 |
| RPSA | ENSCAFG00000006064 | 4.191 | U | ||
| RPSA | ENSCAFG00000017317 | 5.249 | P | ||
| RPL15 | ENSCAFG00000025328 | 5.342 | U | ENSCAFG00000005764 | 2.109 |
| RPLP0P | ENSCAFG00000009876 | 5.128 | P | ENSCAFG00000010227 | 4.484 |
| RPL31 | ENSCAFG00000017238 | 4.322 | U | ENSCAFG00000030034 | 4.237 |
| RPL19 | ENSCAFG00000004857 | 4.007 | U | - c | - |
| RPL19 | ENSCAFG00000002237 | 3.371 | U | - | - |
| RPL4 | ENSCAFG00000018632 | 3.822 | U | - | - |
| RPS2 | ENSCAFG00000013905 | 3.580 | U | - | - |
| SNRPG | ENSCAFG00000003361 | 3.346 | P | ENSCAFG00000032595 | 6.411 |
| RPL10A | ENSCAFG00000030323 | 3.211 | U | - | - |
| HSPA8 | ENSCAFG00000012507 | 2.885 | U | ENSCAFG00000011666 | 2.231 |
| RPS26 | ENSCAFG00000011389 | 2.227 | U | - | - |
| UBA52 | ENSCAFG00000018652 | 2.107 | P | - | - |
| PSMA4 | ENSCAFG00000016728 | −2.689 | U | - | - |
| CFl1 | ENSCAFG00000010275 | −2.385 | U | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwasnik, M.; Socha, W.; Czech, B.; Wasiak, M.; Rola, J.; Rozek, W. Protein-Coding Region Derived Small RNA in Exosomes from Influenza A Virus–Infected Cells. Int. J. Mol. Sci. 2023, 24, 867. https://doi.org/10.3390/ijms24010867
Kwasnik M, Socha W, Czech B, Wasiak M, Rola J, Rozek W. Protein-Coding Region Derived Small RNA in Exosomes from Influenza A Virus–Infected Cells. International Journal of Molecular Sciences. 2023; 24(1):867. https://doi.org/10.3390/ijms24010867
Chicago/Turabian StyleKwasnik, Malgorzata, Wojciech Socha, Bartosz Czech, Magdalena Wasiak, Jerzy Rola, and Wojciech Rozek. 2023. "Protein-Coding Region Derived Small RNA in Exosomes from Influenza A Virus–Infected Cells" International Journal of Molecular Sciences 24, no. 1: 867. https://doi.org/10.3390/ijms24010867
APA StyleKwasnik, M., Socha, W., Czech, B., Wasiak, M., Rola, J., & Rozek, W. (2023). Protein-Coding Region Derived Small RNA in Exosomes from Influenza A Virus–Infected Cells. International Journal of Molecular Sciences, 24(1), 867. https://doi.org/10.3390/ijms24010867







