Transcriptome Analysis of the Effects of Grafting Interstocks on Apple Rootstocks and Scions
Abstract
1. Introduction
2. Results
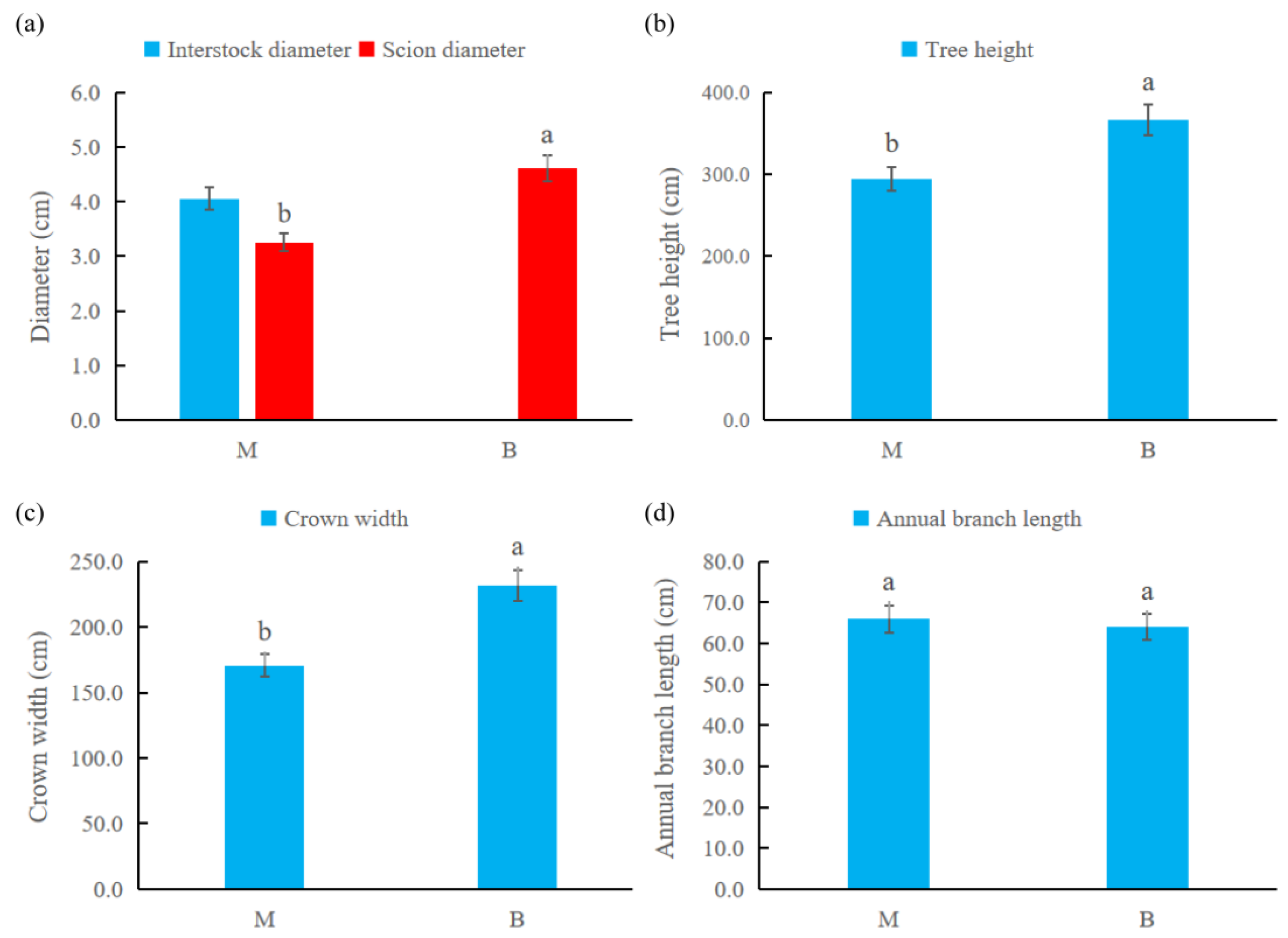
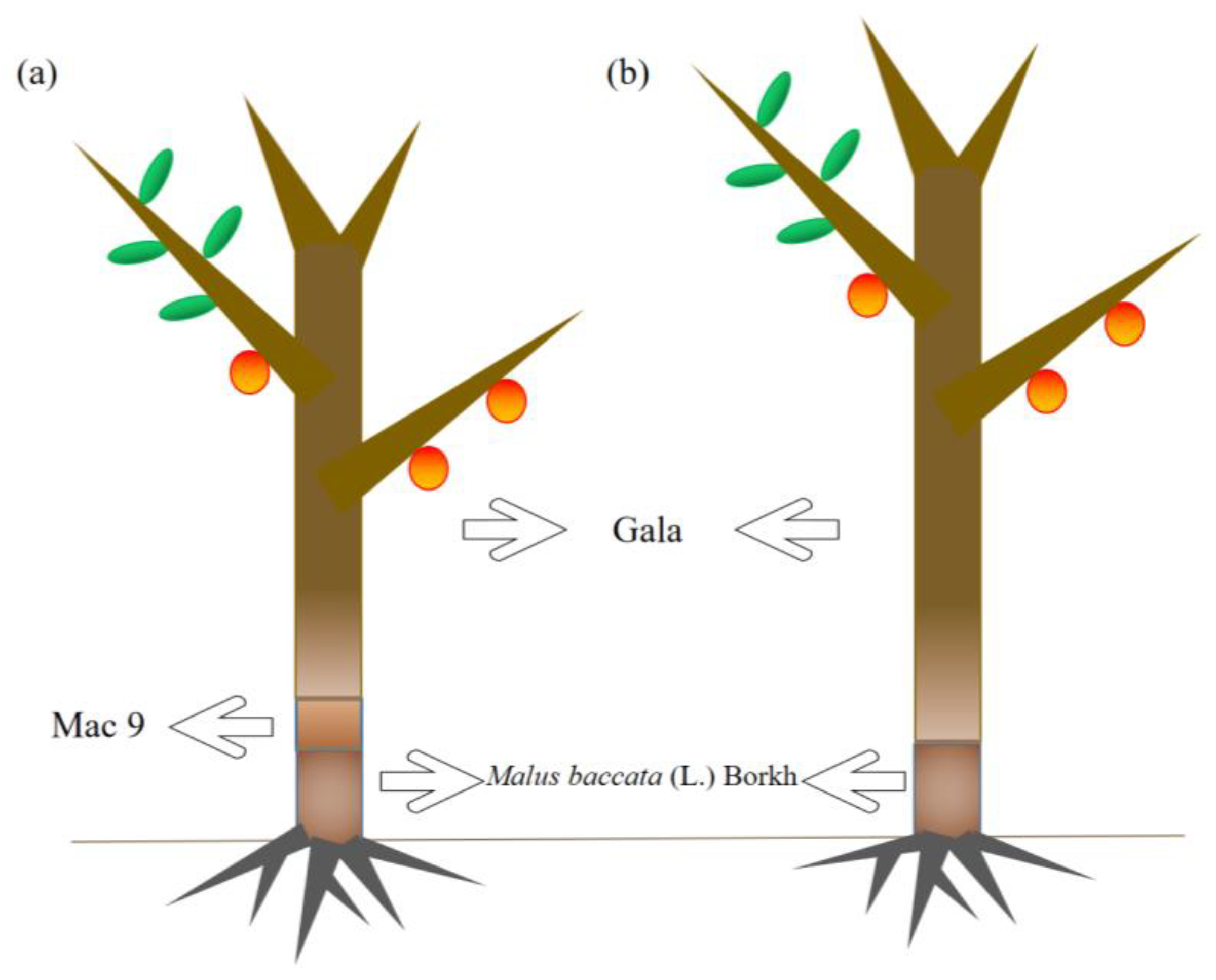
2.1. The Effect of Dwarfing Interstock on the Tree Body
2.2. Quality Control of Transcriptome Data
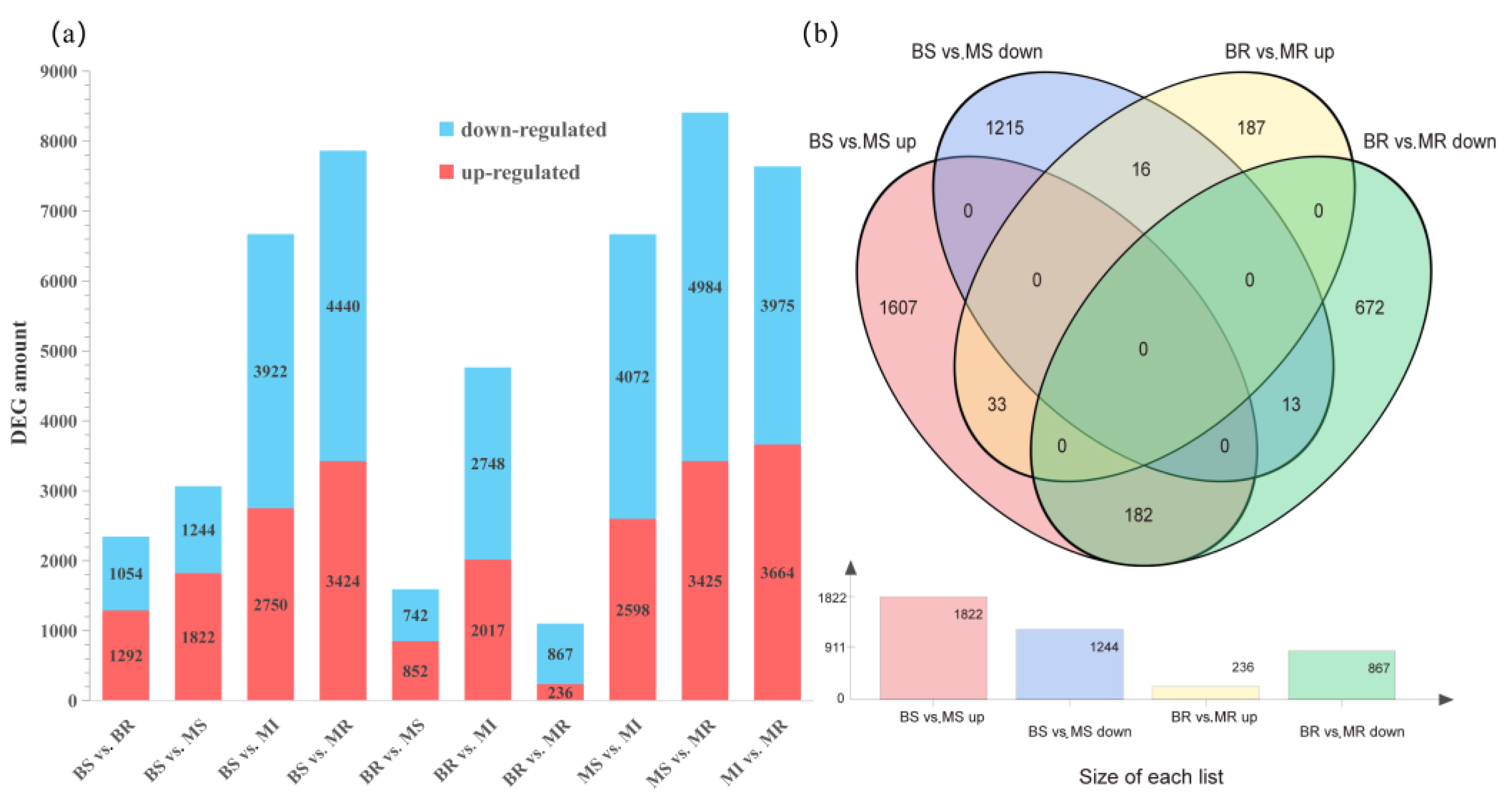
2.3. Analysis Expression Profiling of Differentially Expressed Genes (DEGs)
2.4. GO Function Analysis of DEGs
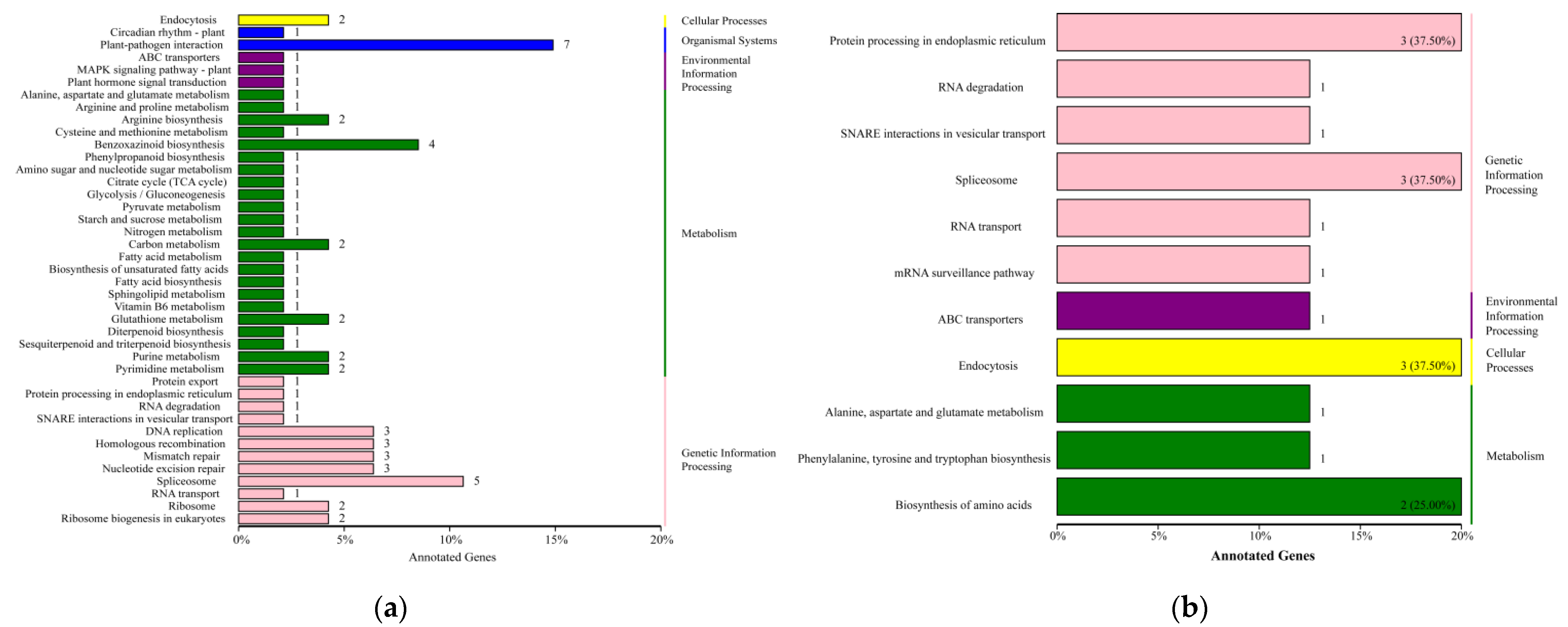
2.5. KEGG Function Analyses of DEGs
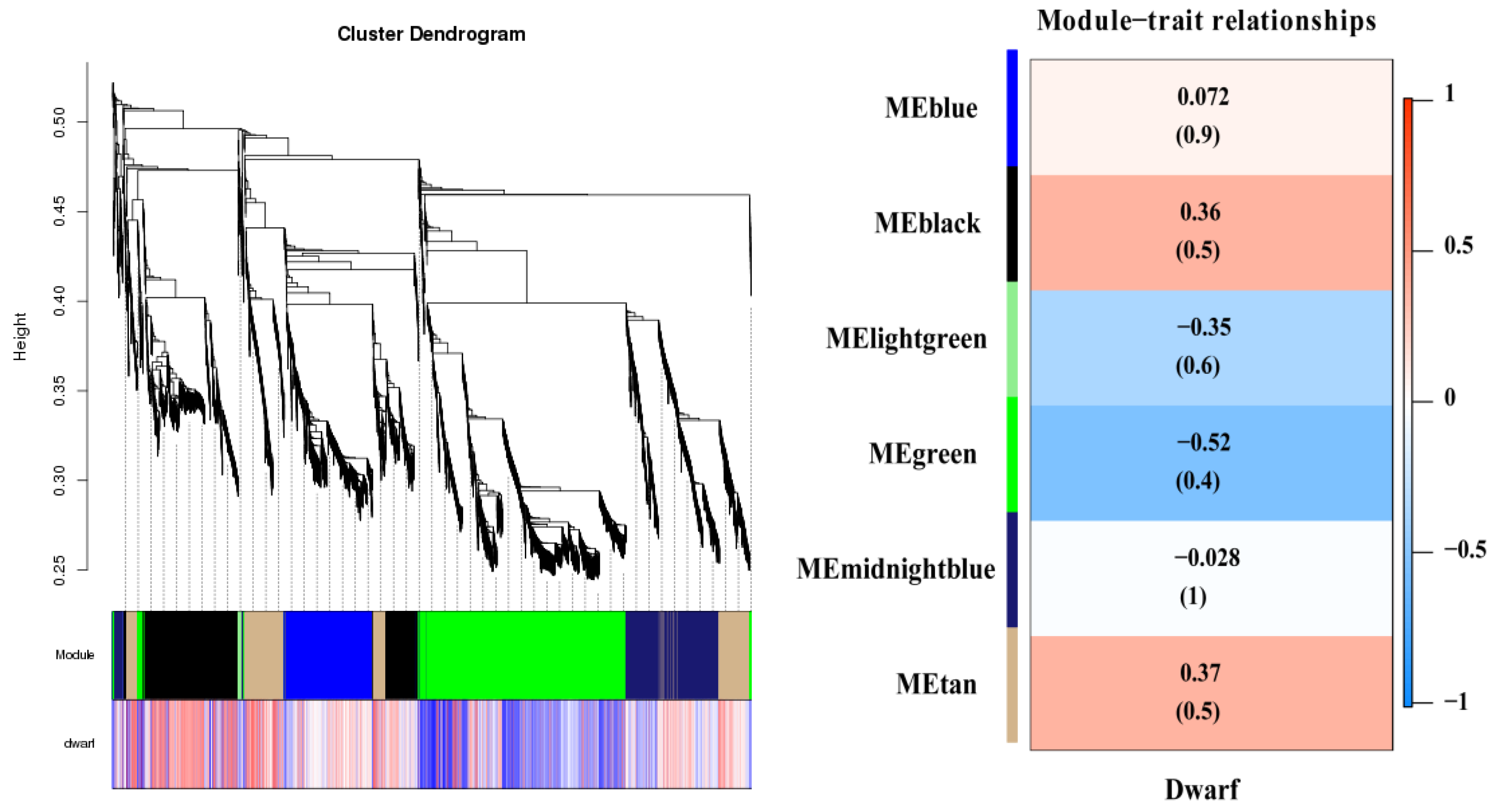
2.6. Functional Enrichment Analyses of Tissue-Specific Modules Using WGCNA
2.6.1. Analysis of Plant Hormone Metabolism Affected by Grafting Interstock
2.6.2. Analysis of DEGs Related to Photosynthesis
2.6.3. Analysis of DEGs Related to C Metabolism
2.6.4. Analysis of DEGs Related to N Metabolism
2.6.5. Analysis of DEGs Related to Secondary Metabolism
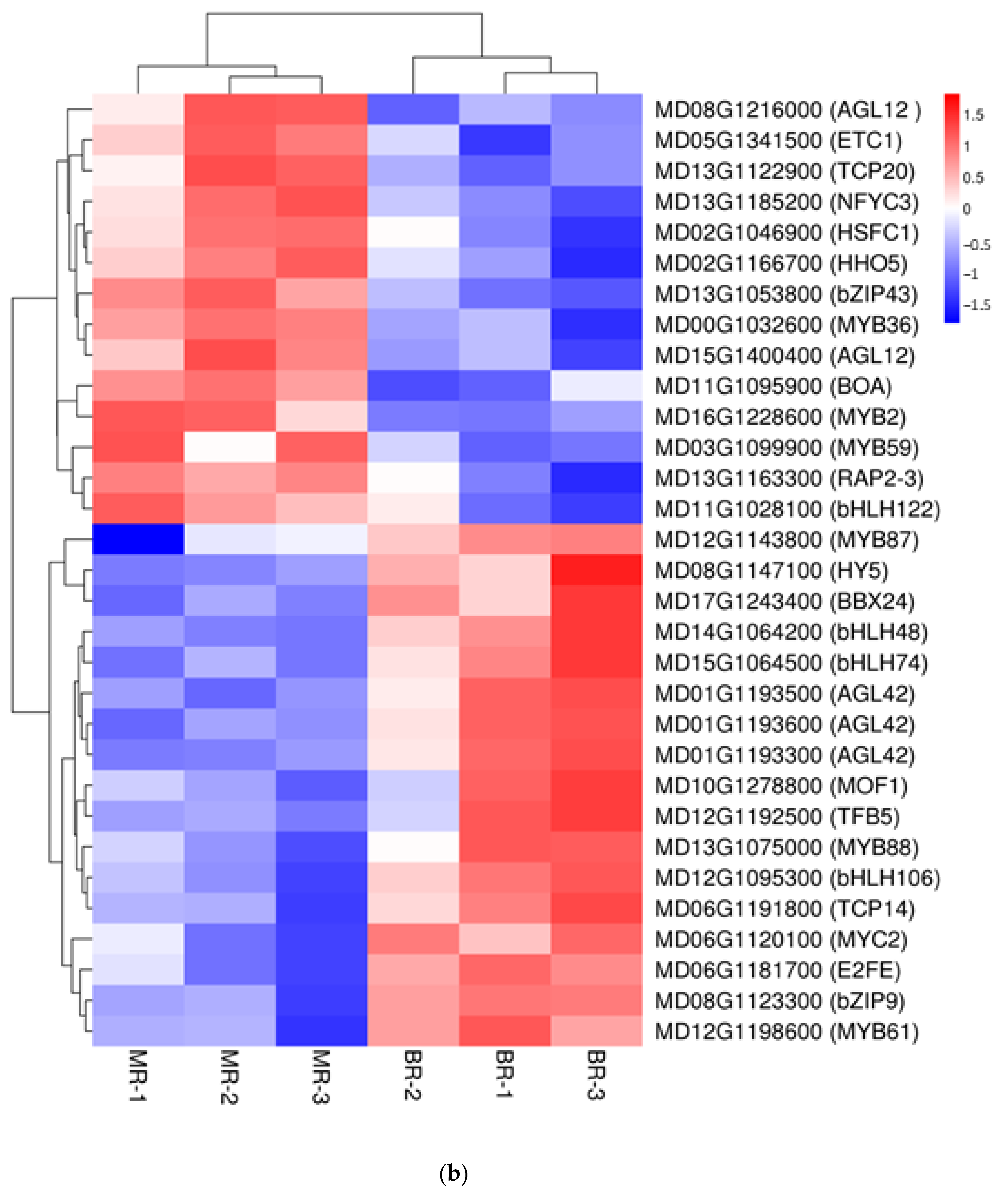
2.7. Visualization of Transcription Factors with Grafted Interstocks
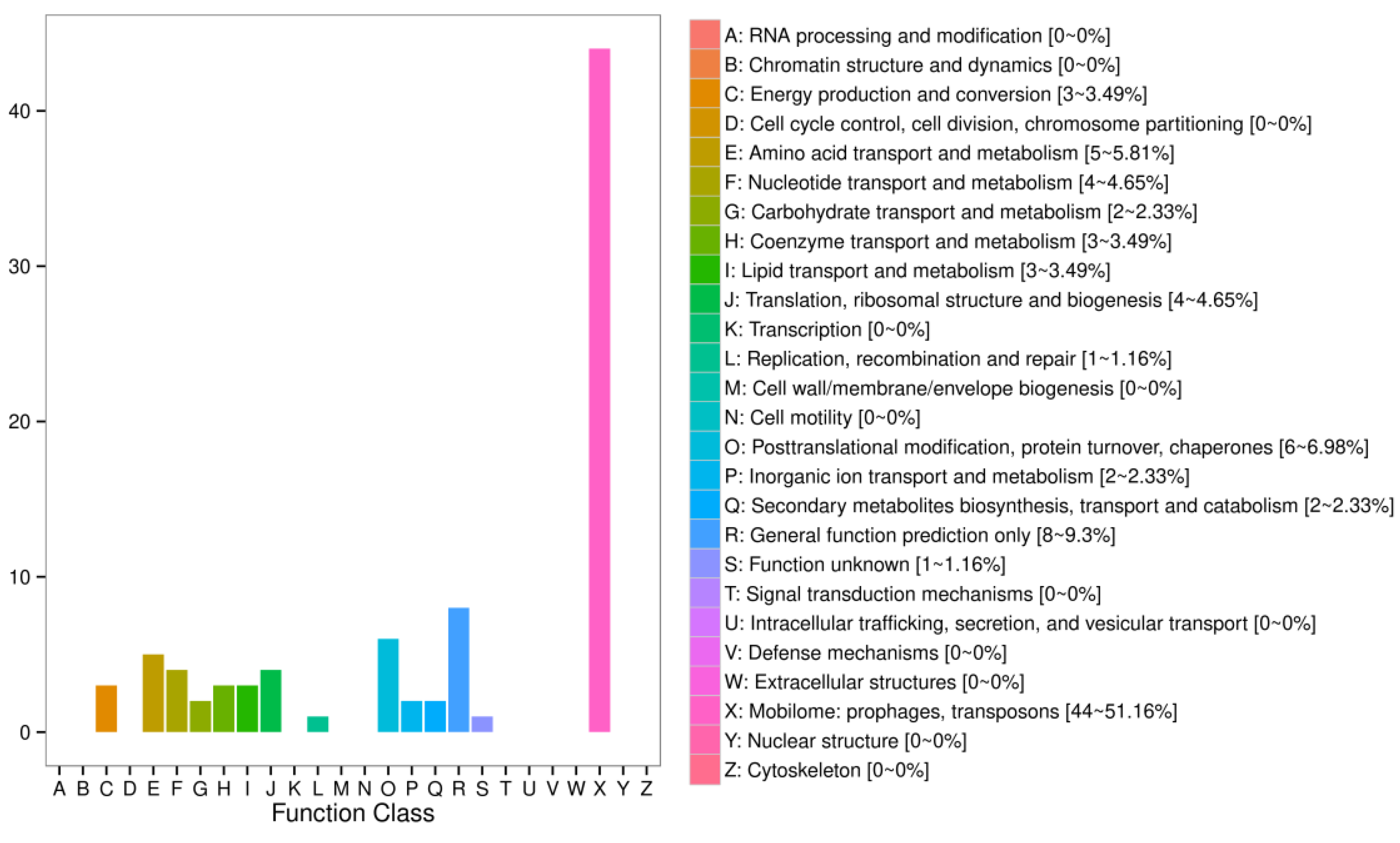
2.8. Identification of the Mobile mRNAs with Grafted Interstocks
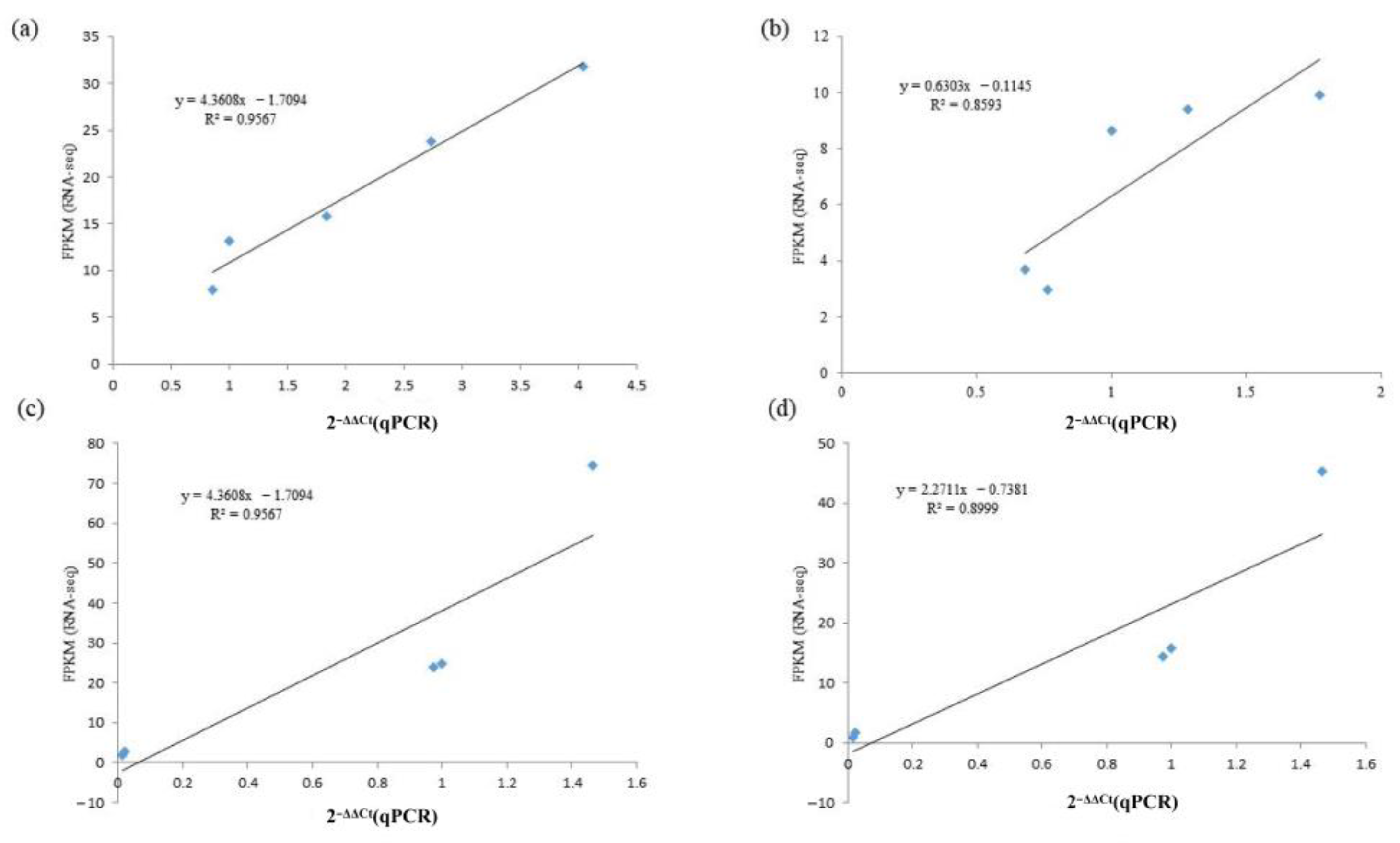
2.9. qRT-PCR Validation
3. Discussion
4. Materials and Methods
4.1. Experiments with Planting Materials and Grafting
4.2. Determination of Tree Growth
4.3. Transcriptome Sequencing Analysis
4.4. Differentially Expressed Gene (DEG) Analysis
4.5. Weighted Gene Co-Expression Network Analysis (WGCNA)
4.6. Real-Time Quantitative PCR Validation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Warschefskyf, E.J.; Klein, L.L.; Frank, M.H.; Chitwood, D.H.; Londo, J.P.; Wettberg, E.J.V.; Miller, A.J. Rootstocks: Diversity, domestication, and impacts on shoot phenotypes. Trends Plant Sci. 2015, 21, 418–437. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Yang, J.H.; Fu, X.X.; Zhang, L.; Tang, K.; Guy, K.M.; Hu, Z.Y.; Guo, S.G.; Xu, Y.; Zhang, M.F. Genome-wide identification and comparative analysis of grafting-responsive mRNA in watermelon grafted onto bottle gourd and squash rootstocks by high-throughput sequencing. Mol. Genet. Genom. 2016, 291, 621–633. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Pan, H.L.; Pan, T.F.; Tang, H.R.; Wang, X.R.; Pan, D.M. Research progress on the interaction between scion and rootstock in fruit trees. Acta Hortic. 2017, 44, 1645–1657. [Google Scholar] [CrossRef]
- Hossain, S.; Mizutani, F.; Onguso, J.M.; Shereif, A.R.; Yamada, H. Effect of interstock and spiral bark ringing on the growth and yield of peach. Bulg. J. Agric. Sci. 2005, 7, 117–120. [Google Scholar] [CrossRef]
- Rozpara, E.; Grzyb, Z.S.; Glowacka, A. Effect of rootstock and interstock on growth and yield of three sweet cherry cultivars. Acta Hortic. 2011, 903, 541–545. [Google Scholar] [CrossRef]
- Cui, Z.H.; Zhang, H.; Galarneau, E.R.; Yang, Y.J.; Li, D.L.; Song, J.K.; Ma, C.H.; Zhang, S.L.; Wang, R.K. Metabolome analysis reveals important compounds related to dwarfing effect of interstock on scion in pear. Ann. Appl. Biol. 2021, 719, 108–122. [Google Scholar] [CrossRef]
- Shen, Y.Y.; Zhuang, W.B.; Tu, X.T.; Gao, Z.H.; Xiong, A.S.; Yu, X.Y.; Li, X.H.; Li, F.H.; Qu, S.C. Transcriptomic analysis of interstock induced dwarfism in Sweet Persimmon (Diospyros kaki Thunb.). Hortic. Res. 2019, 6, 17. [Google Scholar] [CrossRef] [PubMed]
- Koepke, T.; Dhingra, A. Rootstock scion somatogenetic interactions in perennial composite plants. Plant Cell Rep. 2013, 32, 1321–1337. [Google Scholar] [CrossRef]
- Guo, G.N.; Yan, Z.L.; Zhang, H.T.; Zhang, S.N.; Liu, Z.Z. Current situation of production of early and mid-season apple cultivars in china and progress in breeding for early and mid-season apple cultivars. J. Fruit Sci. 2009, 26, 871–877. [Google Scholar] [CrossRef]
- Bhat, S.A.; Rizwan, D.; Mir, S.A.; Wani, S.M.; Masoodi, F.A. Advances in apple packaging: A review. J. Food Sci. Technol. 2022, 1–13. [Google Scholar] [CrossRef]
- Li, M.J.; Zhang, Q.; Li, X.L.; Zhou, B.B.; Yang, Y.Z.; Zhou, J.; Zhang, J.K.; Wei, Q.P. Effect of three different tree shapes on growth, yield and fruit quality of ‘Fuji’ apple trees on dwarfing interstocks. Sci. Agric. Sin. 2017, 50, 3789–3796. [Google Scholar]
- Li, M.J.; Zhang, Q.; Li, X.L.; Zhou, B.B.; Yang, Y.Z.; Zhang, J.K.; Zhou, J.; Wei, Q.P. Effects of 4 dwarfing rootstocks on growth, yield and fruit quality of ‘Fuji’ sapling in apple replant orchard. Sci. Agric. Sin. 2020, 53, 2264–2271. [Google Scholar] [CrossRef]
- Robinson, T.L. Recent advances and future directions in orchard planting systems. Acta Hortic. 2004, 732, 367–381. [Google Scholar] [CrossRef]
- Wang, T.; Xiong, B.; Tan, L.P.; Yang, Y.T.; Zhang, Y.; Ma, M.M.; Xu, Y.H.; Liao, L.; Sun, G.C.; Liang, D.; et al. Effects of interstocks on growth and photosynthetic characteristics in ‘Yuanxiaochun’ Citrus seedlings. Funct. Plant Biol. 2020, 47, 977–987. [Google Scholar] [CrossRef]
- Li, H.L.; Zhang, H.; Yu, C.; Ma, L.; Wang, Y.; Zhang, X.Z.; Han, Z.H. Possible roles of auxin and zeatin for initiating the dwarfing effect of M9 used as apple rootstock or interstock. Acta Physiol. Plant. 2012, 34, 235–244. [Google Scholar] [CrossRef]
- Shi, B.S.; Xu, J.Z.; Chen, H.J.; Ma, B.K.; Guo, R.F. The studies on water transmitting resistance with stems of different stocks and red Fuji apple grafted on interstocks. Hebei J. For. Orchard. Res. 2001, 16, 136–139. [Google Scholar]
- He, P.; Li, L.; Wang, H.; Chang, Y. Effects of five dwarfing interstocks on shoot growth, fruiting and accumulation of mineral elements in leaves of Yishui red Fuji apple. Sci. Agric. Sin. 2018, 51, 750–757. [Google Scholar] [CrossRef]
- Zhao, T.S.; Zhao, G.D.; Zhang, C.H.; Zhang, X.S.; Yang, F.Q.; Chen, D.M.; Zhao, Y.B.; You, F. Effect of dwarfing interstocks on tree growth, yields and fruit quality of ‘Miyazakifuji’ apple. J. Food Sci. 2016, 33, 1379–1387. [Google Scholar] [CrossRef]
- Palmer, J.W.; Seymour, S.; Cashmore, W.M. Early results with dwarfing rootstocks used as interstocks for apples in New Zealand. Acta Hortic. 1997, 451, 161–162. [Google Scholar] [CrossRef]
- Zhang, X.; Zhu, Z.Z.; Sun, L.L.; Li, F.L.; Wei, D.C.; Zhu, J.S.; Fan, L.D.; Zhao, Z.Y. Effects of different dwarfing interstocks on cold resistance of ’Changfu 2’ apple in Longdong area. J. Food Sci. 2020, 37, 985–996. [Google Scholar] [CrossRef]
- Michelesi, J.C.; Masseron, A. Apple rootstocks. Initial resutls of a network experimental programme. Infos 1995, 111, 37–43. [Google Scholar]
- Du, X.M.; Yang, Y.Z.; Gao, J.D.; Wang, Q.; Cai, H.C.; Li, C.Y. Advances of effect of apple rootstocks on grafted varieties. Acat Agric. Boreali-Occident. Sin. 2020, 29, 487–495. [Google Scholar] [CrossRef]
- Qiao, X.; Li, M.; Yin, H.; Li, L.T.; Wu, J.; Zhang, S.L. Advances on whole genome sequencing in fruit trees. Acta Hortic. Sin. 2014, 41, 165–177. [Google Scholar] [CrossRef]
- Gaut, B.S.; Miller, A.J.; Seymour, D.K. Living with two genomes: Grafting and its implications for plant genome-to-genome interactions, phenotypic variation, and evolution. Annu. Rev. Genet. 2019, 53, 195–215. [Google Scholar] [CrossRef] [PubMed]
- Notaguchi, M.; Higashiyama, T.; Suzuki, T. Identification of mRNAs that move over long distances using an RNA-Seq analysis of Arabidopsis/Nicotiana benthamiana heterografts. Plant Cell Physiol. 2015, 56, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, A.; Bhogale, S.; Kang, I.H.; Hannape, D.J.; Banerjee, A.K. The mRNA of a Knotted1-like transcription factor of potato is phloem mobile. Plant Mol. Biol. 2012, 79, 595–608. [Google Scholar] [CrossRef]
- Zhong, H.X.; Liu, Z.J.; Zhang, F.C.; Zhou, X.M.; Sun, X.X.; Li, Y.Y.; Liu, W.W.; Xiao, H.; Wang, N.; Lu, H.; et al. Metabolomic and transcriptomic analyses reveal the effects of self- and hetero-grafting on anthocyanin biosynthesis in grapevine. Hortic. Res. 2022, 9, 103. [Google Scholar] [CrossRef]
- Karlidağ, H.; Aslantaş, R.; Eşitken, A. Effects of interstock (M9) length grafted onto MM106 rootstock on sylleptic shoot formation, growth and yield in some apple cultivars. J. Agric. Sci 2014, 20, 331–336. [Google Scholar] [CrossRef]
- Cao, Y.H. Comparative Study on the Photosynthetic Characteristics and Physiological Effect of ‘Fuji’ Apple Saplings in Different Rootstock-Scion Combinations. Master’s Thesis, Gansu Agricultural University, Lanzhou, China, 2017. [Google Scholar]
- Li, Q.S.; Gao, Y.; Wang, K.; Sun, S.M.; Lu, X.; Li, L.W.; Feng, J.R.; Wang, D.J. Effects of different dwarfing interstocks on the total phenols and compounds of polyphenols in ‘Huahong’ apple. J. Fruit Sci. 2022, 7, 1710. [Google Scholar] [CrossRef]
- Li, G.F.; Ma, J.J.; Tan, M.; Mao, J.P.; An, N.; Sha, G.L.; Zhang, D.; Zhao, C.P.; Han, M.Y. Transcriptome analysis reveals the effects of sugar metabolism and auxin and cytokinin signaling pathways on root growth and development of grafted apple. BMC Genom. 2016, 17, 150. [Google Scholar] [CrossRef]
- Tietel, Z.; Srivastava, S.; Fait, A.; Tel-zur, N.; Carmi, N.; Raveh, E. Impact of scion/rootstock reciprocal effects on metabolomics of fruit juice and phloem sap in grafted citrus reticulata. PLoS ONE 2020, 15, e0227192. [Google Scholar] [CrossRef] [PubMed]
- Seleznyova, N.A.; Tustin, D.S.; Thorp, T.G. Apple dwarfing rootstocks and interstocks affect the type of growth units produced during the annual growth cycle: Precocious transition to flowering affects the composition and vigour of annual shoots. AOB 2008, 101, 679–687. [Google Scholar] [CrossRef] [PubMed]
- Foster, T.M.; McAtee, P.A.; Waite, C.N.; Boldingh, H.L.; McGhie, T.K. Apple dwarfing rootstocks exhibit an imbalance in carbohydrate allocation and reduced cell growth and metabolism. Hortic. Res. 2017, 4, 17009. [Google Scholar] [CrossRef] [PubMed]
- Kviklys, D.; Samuolienė, G. Relationships among the rootstock, crop load, and sugar hormone signaling of apple tree, and their effects on biennial bearing. Front. Plant Sci. 2020, 11, 1213. [Google Scholar] [CrossRef] [PubMed]
- Baron, D.; Amaro, A.C.E.; Pina, A.; Ferreira, G. An overview of grafting re-establishment in woody fruit species. Sci. Hortic. 2019, 243, 84–91. [Google Scholar] [CrossRef]
- Albacete, A.; Martínez-Andújar, C.; Martínez-Pérez, A.; Thompson, A.J.; Dodd, I.C.; Perez-Alfovea, F. Unravelling rootstock × scion interactions to improve food security. J. Exp. Bot. 2015, 66, 2211–2226. [Google Scholar] [CrossRef]
- Olszewski, N.; Sun, T.P.; Gubler, F. Gibberellin signaling: Biosynthesis, catabolism, and response pathways. Plant Cell 2002, 14, S61–S80. [Google Scholar] [CrossRef]
- Zhou, Y.; Underhill, S.J.R. Expression of gibberellin metabolism genes and signalling components in dwarf phenotype of breadfruit (Artocarpus altilis) plants growing on marang (Artocarpus odoratissimus) rootstocks. Plants 2020, 9, 634. [Google Scholar] [CrossRef]
- Wang, Z.H. Bioinformatics of GA20ox, GA2ox, GA3ox Genes and Functional of GA20ox1 Analyses in Cucumis sativus. Master’s Thesis, Shandong University, Jinan, China, 2014. [Google Scholar]
- El-Sharkawy, I.; Kayal, W.E.; Prasath, D.; Bouzayen, M.; Svircev, A.M.; Jayasankar, S.; Fernández, H. Identification and genetic characterization of a gibberellin 2-oxidase gene that controls tree stature and reproductive growth in plum. J. Exp. Bot. 2012, 63, 1225–1239. [Google Scholar] [CrossRef]
- Liu, X.Y.; Li, J.; Liu, M.M.; Yao, Q.; Chen, J.Z. Transcriptome profiling to understand the effect of citrus rootstocks on the growth of ‘Shatangju’ mandarin. PLoS ONE 2017, 12, e0169897. [Google Scholar] [CrossRef]
- Van, H.B.; Woolley, D.J.; Warrington, I.J.; Tustin, D.S. Initial alteration of scion architecture by dwarfing apple rootstocks may involve shoot-root-shoot signalling by auxin, gibberellin, and cytokinin. J. Hortic. Sci. Biotechnol. 2010, 85, 59–65. [Google Scholar] [CrossRef]
- Gan, Z.Y. Study on the Regulatory Effects of Apple Auxin Transporter PIN on Dwarfing of M9 Interstem and Pollen Development. Ph.D. Thesis, China Agriculture University, Beijing, China, 2018. [Google Scholar]
- An, C. Cloning and Functional Analysis of Auxin Transfusion Gene LAX3 from ‘Guizimai No.1’. Master’s Thesis, Guizhou University, Guiyang, China, 2022. [Google Scholar] [CrossRef]
- Feng, Y. Research on the Mechanisms of IPT5b Gene Expression and Dwarfing Regulation in Root of M9 Rootstock. Ph.D. Thesis, China Agriculture University, Beijing, China, 2017. [Google Scholar]
- Zheng, X.D.; Zhao, Y.; Shan, D.Q.; Shi, K.; Wang, L.; Li, Q.T.; Wang, N.A.; Zhou, J.Z.; Yao, J.Z.; Xue, Y.; et al. MdWRKY9 overexpression confers intensive dwarfing in the M26 rootstock of apple by directly inhibiting brassinosteroid synthetase MdDWF4 expression. New Phytol. 2018, 217, 1086–1098. [Google Scholar] [CrossRef] [PubMed]
- Raines, C.A.; Paul, M.J. Products of leaf primary carbon metabolism modulate the developmental programme determining plant morpholodgy. J. Exp. Bot. 2006, 57, 1857–1862. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.W. Omics-Based Study on the Metabolism of C/N and Biosynthesis of Main Quality Related Components in Tea Plants Affected by Nitrogen. Ph.D. Thesis, Chinese Academy of Agricultural Sciences, Beijing, China, 2016. [Google Scholar]
- Izumi, M.; Tsunoda, H.; Suzuki, Y.; Makino, A.; Ishida, H. RBCS1A and RBCS3B, two major members within the arabidopsis rbcs multigene family, function to yield sufficient rubisco content for leaf photosynthetic capacity. J. Exp. Bot. 2012, 63, 2159–2170. [Google Scholar] [CrossRef]
- Ruan, C.J.; Shao, H.B.; Silva, J.A.T.D. A critical review on the improvement of photosynthetic carbon assimilation in C3 plants using genetic engineering. Crit. Rev. Biotechnol. 2012, 32, 1–21. [Google Scholar] [CrossRef]
- Zhou, Y.M.; Hayat, F.; Yao, J.J.; Tian, X.; Wang, Y.X.; Zhang, X.Z.; Li, W.; Wu, T.N.; Han, Z.H.; Xu, X.F.; et al. Size-controlling interstocks affect growth vigour by downregulating photosynthesis in eight-year-old ‘Red Fuji’ apple trees. Eur. J. Hortic. Sci. 2021, 86, 146–155. [Google Scholar] [CrossRef]
- Yuan, C.Z.; Han, X.J.; Li, Y.Q.; Mo, Y.Y.; Guo, F.Y.; Li, W.G.; Gai, Y.P.; Yi, X.L. Genetic transformation and biological function analysis of the Sedoheptulose-1, 7-bisphosphatase gene from mulberry. Sci. Seric. 2013, 39, 413–419. [Google Scholar] [CrossRef]
- Alvarez, C.E.; Bovdilova, A.; Hoppner, A.; Wolff, C.C.; Saigo, M.; Trajtenberg, F.; Zhang, T.; Buschiazzo, A.; Nagel-Steger, L.; Drincovich, M.F.; et al. Molecular adaptations of NADP-malic enzyme for its function in C4 photosynthesis in grasses. Nat. Plants 2019, 5, 755–765. [Google Scholar] [CrossRef]
- Geng, Y.Q.; Dong, X.C.; Zhang, C.M. Recent progress of sugar transporter in horticultural crops. Acta Hortic. Sin. 2021, 48, 676–688. [Google Scholar] [CrossRef]
- Xin, W.; Xin, L. Research progress on mechanism of nitrogen metabolism involved in plant stress resistance. Guihaia 2020, 40, 583–591. [Google Scholar] [CrossRef]
- Aguirre, P.B.; Al-Hinai, Y.K.; Roper, T.R.; Krueger, A.R. Apple tree rootstock and fertilizer application timing affect nitrogen uptake. HortScience 2001, 36, 1202–1205. [Google Scholar] [CrossRef]
- Berger, A.; Boscari, A.; Araújo, N.H.; Maucourt, M.; Brouquisse, R. Plant nitrate reductases regulate nitric oxide production and nitrogen-fixing metabolism during the medicago truncatula-sinorhizobium meliloti symbiosis. Front. Plant Sci. 2020, 11, 1313. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, J.X.; Terce-Laforgue, T.; Armengaud, P.; Clement, G.; Renou, J.P.; Pelletier, S.; Catterou, M.; Azzopardi, M.; Gibon, Y.; Lea, P.J.; et al. Characterization of a NADH-dependent glutamate dehydrogenase mutant of arabidopsis demonstrates the key role of this enzyme in root carbon and nitrogen metabolism. Plant Cell 2012, 24, 4044–4065. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.; Shahzad, S.M.; Imtiaz, M.; Rizwan, M.S.; Arif, M.S.; Kausar, R. Nitrogen nutrition and adaptation of glycophytes to saline environment: A review. Arch. Agron. Soil Sci. 2018, 64, 1181–1206. [Google Scholar] [CrossRef]
- Wertheim, S.J.; Wagenmakers, P.S.; Bootsmaj, H.; Groot, M.J. Orchard systems for apple and pear: Conditions for success. Acta Hortic. 2001, 557, 209–227. [Google Scholar] [CrossRef]
- Shao, L.Q. Techno-Economic Evaluation on High-Density Dwarfing Cultivation Pattern. Ph.D. Thesis, Northwest A&F University, Xianyang, China, 2015. [Google Scholar]
- Wang, D.J.; Wang, K.; Gao, Y.; Liu, L.J.; Gong, X.; Zhao, J.R. Comprehensive evaluation of the relationship between dwarfing interstocks and growth power and functional ingredients in the ‘Honeycrisp’ apple. J. Food Sci. 2016, 33, 686–693. [Google Scholar] [CrossRef]
- Tao, R.; Zhang, T.H.; Wu, D.; Fan, M.M.; Sun, L.L.; Liu, Z.Z.; Gao, H. Effects of different dwarfing interstocks on fruit quality of Gala apple. J. Gansu Agric. Univ. 2021, 56, 2–84. [Google Scholar] [CrossRef]
- Lin, G.Y.; Zhou, P.N.; Yin, M.J.; Liu, L.C.; Dai, S.L.; Liu, C.C.; Wu, Q.N. Cloning and bioinformatics analysis of 1-deoxy-D-xylulose 5-phosphate DXS gene from Schizonepeta tenuifolia. Chin. Tradit. Herb. Drugs 2021, 52, 527–537. [Google Scholar] [CrossRef]
- Xie, D.Y.; Jackson, L.A.; Cooper, J.D.; Ferreira, D.; Paiva, N.L. Molecular and biochemical analysis of two cDNA clones encoding dihydroflavonol-4-reductase from Medicago truncatula. Plant Physiol. 2004, 134, 979–994. [Google Scholar] [CrossRef]
- Wang, Q.; Yang, T.Z.; Gao, J.D.; Cai, H.C.; Li, C.Y.; Du, X.M.; Wang, S.T.; Gong, G.H. Effects of different rootstocks on tree growth and fruit quality of Malus pumila ‘Nagafu No.2’. Chin. Agric. Sci. Bull. 2017, 33, 16–21. [Google Scholar]
- Dard, N.; Celton, J.M.; Linsmith, G.; Becker, C.; Choisne, N.; Schijlen, E.; van de Geest, H.; Bianco, L.; Micheletti, D.; Velasco, R.; et al. High-quality de novo assembly of the apple genome and methylome dynamics of early fruit development. Nat. Genet. 2017, 49, 1099–1106. [Google Scholar] [CrossRef]
- Chen, T.; Liu, Y.X.; Huang, L.Q. ImageGP: An easy-to-use data visualization web server for scientific researchers. iMeta 2022, 1, e5. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−∆∆CT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Wang, D.J.; Gao, Y.; Sun, S.M.; Li, L.W.; Wang, K. Expression profiles and characteristics of apple lncRNAs in roots, phloem, leaves, flowers, and fruit. Int. J. Mol. Sci. 2022, 23, 5931. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Chen, X.; Zhang, S.; Zhu, J.; Tang, B.; Wang, A.; Dong, L.; Zhang, Z.; Yu, C.; Sun, Y.; et al. The genome sequence archive family: Toward explosive data growth and diverse data types. Genom. Proteom. Bioinform. 2021, 19, 578–583. [Google Scholar] [CrossRef] [PubMed]
- CNCB-NGDC Members and Partners. Database resources of the national genomics data center, China National Center for Bioinformation in 2022. Nucleic Acids Res. 2022, 50, D27–D38. [Google Scholar] [CrossRef] [PubMed]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Q.; Gao, Y.; Wang, K.; Feng, J.; Sun, S.; Lu, X.; Liu, Z.; Zhao, D.; Li, L.; Wang, D. Transcriptome Analysis of the Effects of Grafting Interstocks on Apple Rootstocks and Scions. Int. J. Mol. Sci. 2023, 24, 807. https://doi.org/10.3390/ijms24010807
Li Q, Gao Y, Wang K, Feng J, Sun S, Lu X, Liu Z, Zhao D, Li L, Wang D. Transcriptome Analysis of the Effects of Grafting Interstocks on Apple Rootstocks and Scions. International Journal of Molecular Sciences. 2023; 24(1):807. https://doi.org/10.3390/ijms24010807
Chicago/Turabian StyleLi, Qingshan, Yuan Gao, Kun Wang, Jianrong Feng, Simiao Sun, Xiang Lu, Zhao Liu, Deying Zhao, Lianwen Li, and Dajiang Wang. 2023. "Transcriptome Analysis of the Effects of Grafting Interstocks on Apple Rootstocks and Scions" International Journal of Molecular Sciences 24, no. 1: 807. https://doi.org/10.3390/ijms24010807
APA StyleLi, Q., Gao, Y., Wang, K., Feng, J., Sun, S., Lu, X., Liu, Z., Zhao, D., Li, L., & Wang, D. (2023). Transcriptome Analysis of the Effects of Grafting Interstocks on Apple Rootstocks and Scions. International Journal of Molecular Sciences, 24(1), 807. https://doi.org/10.3390/ijms24010807







