A Recent Progress in the Leachate Pretreatment Methods Coupled with Anaerobic Digestion for Enhanced Biogas Production: Feasibility, Trends, and Techno-Economic Evaluation
Abstract
1. Introduction
2. Leachate Toxicity and Environmental Impacts
| Parameter | Unit | Type of Landfill Leachate | ||
|---|---|---|---|---|
| Young (0–5 y) | Intermediate (5–10 y) | Mature (10–20 y) | ||
| COD | mg/L | >10,000 | 4000–10,000 | <4000 |
| Period | Transition | Methane fermentation | Final maturation | |
| BOD | mg/L | 0.5–1.0 | 0.1–0.5 | <0.1 |
| pH | <6.5 | 6.5–7.5 | >7.5 | |
| Organic compounds | % | 80 VFA | 5–30 VFA+HFA | High fulvic fraction |
| NH3-N | mg/L | <400 | NA | >400 |
| Heavy Metals | mg/L | Low to medium | Low | Low |
| Biodegradability | Important | Medium | Low | |
| Stability | Unstable | Moderately stable | stable | |
| Phase | acidogenic | methanogenic | Stabilization/methanogenic | |
| Color appearance | Light yellow | Light yellow | Dark black green | |
3. Anaerobic Digestion: Viability for Leachate Treatment
Anaerobic Co-Digestion of Leachate
4. Hybrid Treatment
5. Pretreatment-Anaerobicdigestion(AD)Ofleachate
5.1. Electrochemicalpretreatment-AD
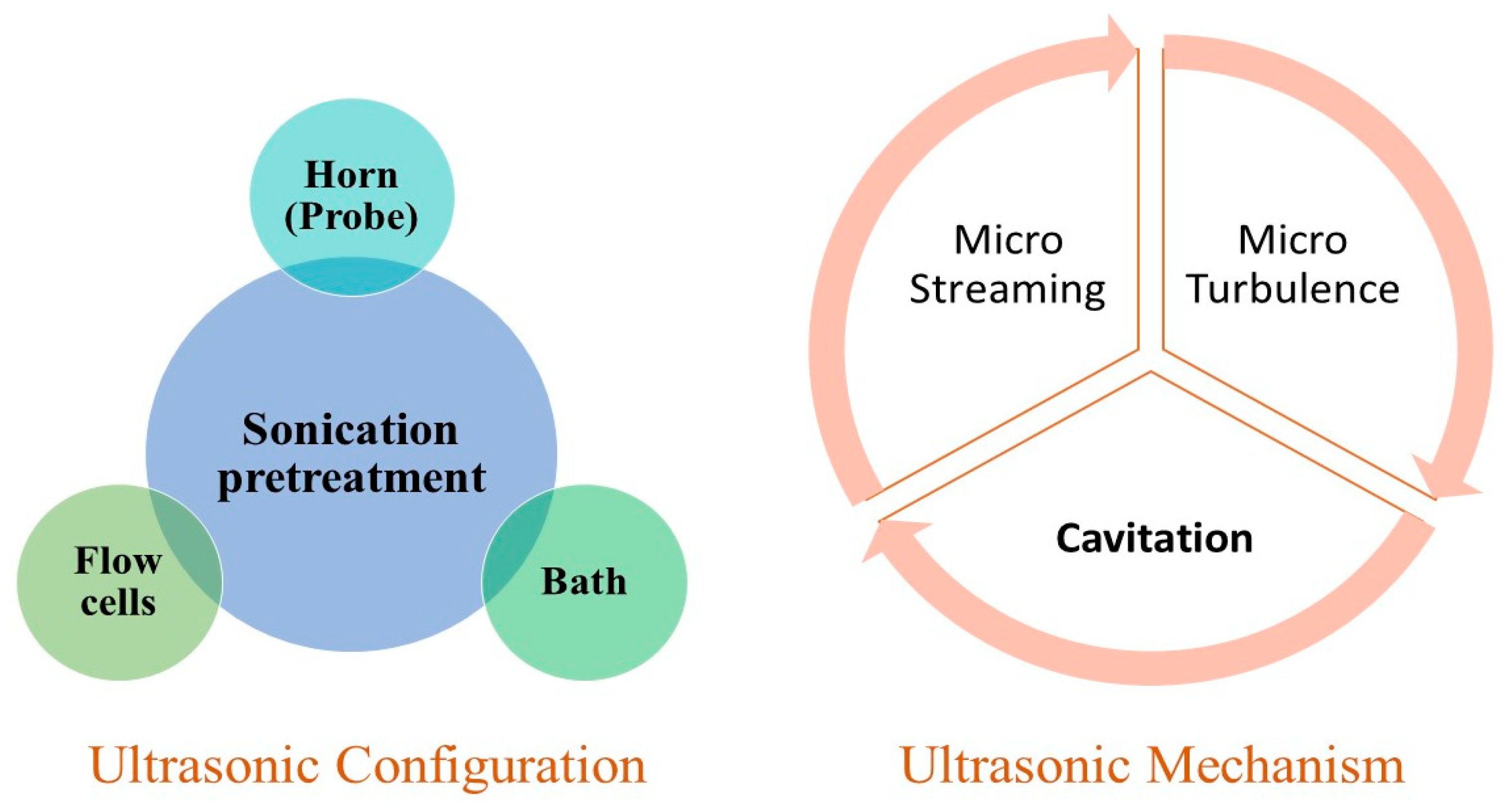
5.2. Ultrasonic Pretreatment-AD
5.3. Coagulation/Flocculationpreatreatment-AD
5.4. Airstrippingpretreatment-AD
5.5. Adsorption Filteration–AD
5.6. Alkaline Pretreatment-AD
5.7. Photocatalysispretreatment-AD
5.8. Membrane Separation-AD
6. Techno-Economic Analysis of Anaerobic Digestion and Policy Requirement
| Substrate /Leachate Type | Treatment Technology | Economic Evaluation (Cost of Process per $/m3) * | Pretreatment Applied ** | Economic Viability *** | Comments | Reference |
|---|---|---|---|---|---|---|
| LFL | MBR | 9.33 $/m−3 | − | − | Considering the commercial value of NH3, the cost of the AS/AB pretreatment technique was minimized by 47%. Furthermore, the energy cost for leachate heating can be reduced by using landfill biogas and solar energy. | [123] |
| Air-stripping/absorption (AS/AB) + MBR | 4.92 $/m−3 | + | ++ | |||
| Composting Leachat | Struvite crystallization | 8.84 $/m−3 | − | − | Compared to struvite crystallization using pure chemicals without cycling, the economic analysis revealed that SPCT could save 59.0% of the processing cost. | [124] |
| Thermal hydrolysis + Struvite pyrolysate cycling technology (SPCT) | 4.24 $/m−3 | + | ++ | |||
| Municipal solid waste leachate | MBR | 10.55 $ /m−3 | − | − | Energy usage and external carbon sources account for most MBR system operation costs. | [125] |
| steam-stripping pretreatment + MBR | 1.86 $/m−3 | + | ++ | |||
| LFL | Mechanical vapor recompression (MVR) | 6.93 $/m−3 | − | + | The cost of treatment was significantly decreased by coagulation pretreatment. | [122] |
| Coagulation + Mechanical vapor recompression | 5.03 $/m−3 | + | ++ |
7. Conclusions and Future Perspectives
- (1)
- The hydrolysis of LFL in an anaerobic reactor system was significantly assisted by optimal pretreatment before anaerobic operations.
- (2)
- The pretreatment of leachates enhanced the sCOD/tCOD ratio by converting insoluble organic matter to soluble COD.
- (3)
- An anaerobic test fed with pretreatment LFL can increase biogas output and biogas or methane yield.
- (4)
- Among various pretreatment methods, it has been observed that electrochemical and photocatalysis are the most feasible and adaptive procedures due to their high capability to degrade toxic organic and inorganic pollutants. Moreover, these methods are highly cost-effective because the material can be reused and is highly stable.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Azadi, S.; Amiri, H.; Rakhshandehroo, G.R. Evaluating the ability of artificial neural network and PCA-M5P models in predicting leachate COD load in landfills. Waste Manag. 2016, 55, 220–230. [Google Scholar] [CrossRef]
- Quan, X.; Hu, R.; Chang, H.; Tang, X.; Huang, X.; Cheng, C.; Zhong, N.; Yang, L. Enhancing microalgae growth and landfill leachate treatment through ozonization. J. Clean. Prod. 2020, 248, 119182. [Google Scholar] [CrossRef]
- Xue, W.-J.; Cui, Y.-H.; Liu, Z.-Q.; Yang, S.-Q.; Li, J.-Y.; Guo, X.-L. Treatment of landfill leachate nanofiltration concentrate after ultrafiltration by electrochemically assisted heat activation of peroxydisulfate. Sep. Purif. Technol. 2020, 231, 115928. [Google Scholar] [CrossRef]
- Christensen, T.H.; Kjeldsen, P.; Bjerg, P.L.; Jensen, D.L.; Christensen, J.B.; Baun, A.; Albrechtsen, H.-J.; Heron, G. Biogeochemistry of landfill leachate plumes. Appl. Geochem. 2001, 16, 659–718. [Google Scholar] [CrossRef]
- Silva, A.L.P.; Prata, J.C.; Duarte, A.C.; Barcelò, D.; Rocha-Santos, T. An urgent call to think globally and act locally on landfill disposable plastics under and after covid-19 pandemic: Pollution prevention and technological (Bio) remediation solutions. Chem. Eng. J. 2021, 426, 131201. [Google Scholar] [CrossRef] [PubMed]
- Vanapalli, K.R.; Sharma, H.B.; Ranjan, V.P.; Samal, B.; Bhattacharya, J.; Dubey, B.K.; Goel, S. Challenges and strategies for effective plastic waste management during and post COVID-19 pandemic. Sci. Total Environ. 2021, 750, 141514. [Google Scholar] [CrossRef]
- Zolfaghari, M.; Jardak, K.; Drogui, P.; Brar, S.K.; Buelna, G.; Dubé, R. Landfill leachate treatment by sequential membrane bioreactor and electro-oxidation processes. J. Environ. Manag. 2016, 184, 318–326. [Google Scholar] [CrossRef]
- Liu, Z.; Wu, W.; Shi, P.; Guo, J.; Cheng, J. Characterization of dissolved organic matter in landfill leachate during the combined treatment process of air stripping, Fenton, SBR and coagulation. Waste Manag. 2015, 41, 111–118. [Google Scholar] [CrossRef]
- Khan, M.S.M.; Kaneesamkandi, Z. Biodegradable waste to biogas: Renewable energy option for the Kingdom of Saudi Arabia. Int. J. Innov. Appl. Stud. 2013, 4, 101–113. [Google Scholar]
- Zhou, W.; Imai, T.; Ukita, M.; Li, F.; Yuasa, A. Effect of loading rate on the granulation process and granular activity in a bench scale UASB reactor. Bioresour. Technol. 2007, 98, 1386–1392. [Google Scholar] [CrossRef]
- Demirbas, A. Biogas production from the organic fraction of municipal solid waste. Energy Sources Part A Recover. Util. Environ. Eff. 2006, 28, 1127–1134. [Google Scholar] [CrossRef]
- Hassan, M.; Wang, X.; Wang, F.; Wu, D.; Hussain, A.; Xie, B. Coupling ARB-based biological and photochemical (UV/TiO2 and UV/S2O82-) techniques to deal with sanitary landfill leachate. Waste Manag. 2017, 63, 292–298. [Google Scholar] [CrossRef]
- Eggen, T.; Moeder, M.; Arukwe, A. Municipal landfill leachates: A significant source for new and emerging pollutants. Sci. Total Environ. 2010, 408, 5147–5157. [Google Scholar] [CrossRef]
- Nika, M.C.; Ntaiou, K.; Elytis, K.; Thomaidi, V.S.; Gatidou, G.; Kalantzi, O.I.; Thomaidis, N.S.; Stasinakis, A.S. Wide-scope target analysis of emerging contaminants in landfill leachates and risk assessment using Risk Quotient methodology. J. Hazard. Mater. 2020, 394, 122493. [Google Scholar] [CrossRef] [PubMed]
- Propp, V.R.; De Silva, A.O.; Spencer, C.; Brown, S.J.; Catingan, S.D.; Smith, J.E.; Roy, J.W. Organic contaminants of emerging concern in leachate of historic municipal landfills. Environ. Pollut. 2021, 276, 116474. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.; Brame, J.; Li, Q.; Alvarez, P.J.J. Nanotechnology for a safe and sustainable water supply: Enabling integrated water treatment and reuse. Acc. Chem. Res. 2013, 46, 834–843. [Google Scholar] [CrossRef] [PubMed]
- Ferroudj, N.; Nzimoto, J.; Davidson, A.; Talbot, D.; Briot, E.; Dupuis, V.; Bée, A.; Medjram, M.S.; Abramson, S. Maghemite nanoparticles and maghemite/silica nanocomposite microspheres as magnetic Fenton catalysts for the removal of water pollutants. Appl. Catal. B Environ. 2013, 136, 9–18. [Google Scholar] [CrossRef]
- Siciliano, A.; Limonti, C.; Curcio, G.M.; Calabrò, V. Biogas generation through anaerobic digestion of compost leachate in semi-continuous completely stirred tank reactors. Processes 2019, 7, 635. [Google Scholar] [CrossRef]
- Tatsi, A.A.; Zouboulis, A.I.; Matis, K.A.; Samaras, P. Coagulation--flocculation pretreatment of sanitary landfill leachates. Chemosphere 2003, 53, 737–744. [Google Scholar] [CrossRef]
- Vilar, V.J.P.; Capelo, S.M.S.; Silva, T.F.C.V.; Boaventura, R.A.R. Solar photo-Fenton as a pre-oxidation step for biological treatment of landfill leachate in a pilot plant with CPCs. Catal. Today 2011, 161, 228–234. [Google Scholar] [CrossRef]
- Liu, C.; Du, Y.; Zhou, B. Evaluation of estrogenic activities and mechanism of action of perfluorinated chemicals determined by vitellogenin induction in primary cultured tilapia hepatocytes. Aquat. Toxicol. 2007, 85, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Mohana, A.A.; Islam, M.M.; Rahman, M.; Pramanik, S.K.; Haque, N.; Gao, L.; Pramanik, B.K. Generation and consequence of nano/microplastics from medical waste and household plastic during the COVID-19 pandemic. Chemosphere 2022, 311, 137014. [Google Scholar] [CrossRef]
- Shi, J.; Li, X.; Zhang, S.; Sharma, E.; Sivakumar, M.; Sherchan, S.P.; Jiang, G. Enhanced decay of coronaviruses in sewers with domestic wastewater. Sci. Total Environ. 2022, 813, 151919. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Reguyal, F.; Zhuang, T. Risk assessment and investigation of landfill leachate as a source of emerging organic contaminants to the surrounding environment: A case study of the largest landfill in Jinan City, China. Environ. Sci. Pollut. Res. 2021, 28, 18368–18381. [Google Scholar] [CrossRef] [PubMed]
- Qi, C.; Huang, J.; Wang, B.; Deng, S.; Wang, Y.; Yu, G. Contaminants of emerging concern in landfill leachate in China: A review. Emerg. Contam. 2018, 4, 1–10. [Google Scholar] [CrossRef]
- Threedeach, S.; Chiemchaisri, W.; Watanabe, T.; Chiemchaisri, C.; Honda, R.; Yamamoto, K. Antibiotic resistance of Escherichia coli in leachates from municipal solid waste landfills: Comparison between semi-aerobic and anaerobic operations. Bioresour. Technol. 2012, 113, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Xiang, J.; Xie, Y.; Yu, K.; Li, J.; Wang, H.; Li, P.; Gin, K.Y.-H.; He, Y. Removal behavior and key drivers of antibiotic resistance genes in two full-scale leachate treatment plants. Water Res. 2022, 226, 119239. [Google Scholar] [CrossRef]
- Li, X.; Wang, P.; Chu, S.; Su, Y.; Wu, D.; Xie, B. The variation of antibiotic resistance genes and their links with microbial communities during full-scale food waste leachate biotreatment processes. J. Hazard. Mater. 2021, 416, 125744. [Google Scholar] [CrossRef]
- Wang, J.-Y.; An, X.-L.; Huang, F.-Y.; Su, J.-Q. Antibiotic resistome in a landfill leachate treatment plant and effluent-receiving river. Chemosphere 2020, 242, 125207. [Google Scholar] [CrossRef]
- Needham, A.D.; Smith, J.W.N.; Gallagher, E.M.G. The service life of polyethylene geomembrane barriers. Eng. Geol. 2006, 85, 82–90. [Google Scholar] [CrossRef]
- Awasthi, A.K.; Pandey, A.K.; Khan, J. Municipal solid waste leachate impact on metabolic activity of wheat (Triticum aestivum L.) seedlings. Environ. Sci. Pollut. Res. 2017, 24, 17250–17254. [Google Scholar] [CrossRef] [PubMed]
- Rapti-Caputo, D.; Vaccaro, C. Geochemical evidences of landfill leachate in groundwater. Eng. Geol. 2006, 85, 111–121. [Google Scholar] [CrossRef]
- Prechthai, T.; Parkpian, P.; Visvanathan, C. Assessment of heavy metal contamination and its mobilization from municipal solid waste open dumping site. J. Hazard. Mater. 2008, 156, 86–94. [Google Scholar] [CrossRef]
- Xiaoli, C.; Shimaoka, T.; Xianyan, C.; Qiang, G.; Youcai, Z. Characteristics and mobility of heavy metals in an MSW landfill: Implications in risk assessment and reclamation. J. Hazard. Mater. 2007, 144, 485–491. [Google Scholar] [CrossRef]
- Weiland, P. Biogas production: Current state and perspectives. Appl. Microbiol. Biotechnol. 2010, 85, 849–860. [Google Scholar] [CrossRef]
- Cuéllar, A.D.; Webber, M.E. Cow power: The energy and emissions benefits of converting manure to biogas. Environ. Res. Lett. 2008, 3, 34002. [Google Scholar] [CrossRef]
- Liu, J.; Zhong, J.; Wang, Y.; Liu, Q.; Qian, G.; Zhong, L.; Guo, R.; Zhang, P.; Xu, Z.P. Effective bio-treatment of fresh leachate from pretreated municipal solid waste in an expanded granular sludge bed bioreactor. Bioresour. Technol. 2010, 101, 1447–1452. [Google Scholar] [CrossRef]
- Mokhtarani, N.; Bayatfard, A.; Mokhtarani, B. Full scale performance of compost’s leachate treatment by biological anaerobic reactors. Waste Manag. Res. 2012, 30, 524–529. [Google Scholar] [CrossRef]
- Ebrahimi, A.; Hashemi, H.; Eslami, H.; Fallahzadeh, R.A.; Khosravi, R.; Askari, R.; Ghahramani, E. Kinetics of biogas production and chemical oxygen demand removal from compost leachate in an anaerobic migrating blanket reactor. J. Environ. Manag. 2018, 206, 707–714. [Google Scholar] [CrossRef]
- Roy, D.; Azais, A.; Benkaraache, S.; Drogui, P.; Tyagi, R.D. Composting leachate: Characterization, treatment, and future perspectives. Rev. Environ. Sci. Bio/Technol. 2018, 17, 323–349. [Google Scholar] [CrossRef]
- Lei, Y.; Wei, L.; Liu, T.; Xiao, Y.; Dang, Y.; Sun, D.; Holmes, D.E. Magnetite enhances anaerobic digestion and methanogenesis of fresh leachate from a municipal solid waste incineration plant. Chem. Eng. J. 2018, 348, 992–999. [Google Scholar] [CrossRef]
- Pasalari, H.; Esrafili, A.; Rezaee, A.; Gholami, M.; Farzadkia, M. Electrochemical oxidation pretreatment for enhanced methane potential from landfill leachate in anaerobic co-digestion process: Performance, Gompertz model, and energy assessment. Chem. Eng. J. 2021, 422, 130046. [Google Scholar] [CrossRef]
- Nayono, S.E.; Winter, J.; Gallert, C. Anaerobic digestion of pressed off leachate from the organic fraction of municipal solid waste. Waste Manag. 2010, 30, 1828–1833. [Google Scholar] [CrossRef] [PubMed]
- Bohdziewicz, J.; Neczaj, E.; Kwarciak, A. Landfill leachate treatment by means of anaerobic membrane bioreactor. Desalination 2008, 221, 559–565. [Google Scholar] [CrossRef]
- Shin, H.-S.; Han, S.K.; Song, Y.C.; Lee, C.Y. Performance of UASB reactor treating leachate from acidogenic fermenter in the two-phase anaerobic digestion of food waste. Water Res. 2001, 35, 3441–3447. [Google Scholar] [CrossRef]
- Liao, X.; Zhu, S.; Zhong, D.; Zhu, J.; Liao, L. Anaerobic co-digestion of food waste and landfill leachate in single-phase batch reactors. Waste Manag. 2014, 34, 2278–2284. [Google Scholar] [CrossRef]
- Ding, L.; Chen, Y.; Xu, Y.; Hu, B. Improving treatment capacity and process stability via a two-stage anaerobic digestion of food waste combining solid-state acidogenesis and leachate methanogenesis/recirculation. J. Clean. Prod. 2021, 279, 123644. [Google Scholar] [CrossRef]
- Feng, S.; Hou, S.; Huang, X.; Fang, Z.; Tong, Y.; Yang, H. Insights into the microbial community structure of anaerobic digestion of municipal solid waste landfill leachate for methane production by adaptive thermophilic granular sludge. Electron. J. Biotechnol. 2019, 39, 98–106. [Google Scholar] [CrossRef]
- Begum, S.; Anupoju, G.R.; Sridhar, S.; Bhargava, S.K.; Jegatheesan, V.; Eshtiaghi, N. Evaluation of single and two stage anaerobic digestion of landfill leachate: Effect of pH and initial organic loading rate on volatile fatty acid (VFA) and biogas production. Bioresour. Technol. 2018, 251, 364–373. [Google Scholar] [CrossRef]
- de Castro, T.M.; Arantes, E.J.; de Mendonça Costa, M.S.S.; Gotardo, J.T.; Passig, F.H.; de Carvalho, K.Q.; Gomes, S.D. Anaerobic co-digestion of industrial waste landfill leachate and glycerin in a continuous anaerobic bioreactor with a fixed-structured bed (ABFSB): Effects of volumetric organic loading rate and alkaline supplementation. Renew. Energy 2021, 164, 1436–1446. [Google Scholar] [CrossRef]
- Saha, S.; Hussain, A.; Lee, J.; Lee, E.; Lee, H.-S. An integrated leachate bed reactor--anaerobic membrane bioreactor system (LBR-AnMBR) for food waste stabilization and biogas recovery. Chemosphere 2023, 311, 137054. [Google Scholar]
- Gao, M.; Yang, J.; Liu, Y.; Zhang, J.; Li, J.; Liu, Y.; Wu, B.; Gu, L. Deep insights into the anaerobic co-digestion of waste activated sludge with concentrated leachate under different salinity stresses. Sci. Total Environ. 2022, 838, 155922. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Lv, Y.; Cheng, H.; Zou, L.; Li, Y.-Y.; Liu, J. High-efficiency anaerobic co-digestion of food waste and mature leachate using expanded granular sludge blanket reactor. Bioresour. Technol. 2022, 362, 127847. [Google Scholar]
- Yusof, A.; Suja, F.; Rahman, R.A.; Basri, N.E.A.; Abdullah, N.A.; Abujazar, M.S.S. Kinetics evaluation of a pilot scale anaerobic biofilm digester treating leachate from a municipal solid waste transfer station. J. Water Process Eng. 2022, 50, 103202. [Google Scholar] [CrossRef]
- Nabi, M.; Gao, D.; Liang, H.; Cheng, L.; Yang, W.; Li, Y. Landfill leachate treatment by graphite engineered anaerobic membrane bioreactor: Performance enhancement and membrane fouling mitigation. Environ. Res. 2022, 214, 114010. [Google Scholar] [PubMed]
- Franca, L.S.; Ornelas-Ferreira, B.; Pereira, C.P.; Bassin, J.P. Performance of a percolation reactor integrated to solid-state anaerobic garage-type digesters with leachate recirculation for organic fraction of municipal solid waste treatment. Bioresour. Technol. Rep. 2022, 20, 101215. [Google Scholar] [CrossRef]
- Fazzino, F.; Folino, A.; Mauriello, F.; Pedullà, A.; Calabrò, P.S. Biofuel production from fruit and vegetable market waste and mature landfill leachate by an active filter-anaerobic digestion integrated system. Energy Convers. Manag. X 2021, 12, 100130. [Google Scholar] [CrossRef]
- Kawai, M.; Kishi, M.; Hamersley, M.R.; Nagao, N.; Hermana, J.; Toda, T. Biodegradability and methane productivity during anaerobic co-digestion of refractory leachate. Int. Biodeterior. Biodegrad. 2012, 72, 46–51. [Google Scholar] [CrossRef]
- Zhu, T.; Su, Z.; Lai, W.; Zhang, Y.; Liu, Y. Insights into the fate and removal of antibiotics and antibiotic resistance genes using biological wastewater treatment technology. Sci. Total Environ. 2021, 776, 145906. [Google Scholar] [CrossRef]
- Ahmad, I.; Chelliapan, S.; Othman, N.; Mohammad, R.; Kamaruddin, S.A. A review of municipal solid waste (MSW) landfill management and treatment of leachate. Int. J. Civ. Eng. Technol 2018, 9, 336–348. [Google Scholar]
- Liu, P.; Zhang, H.; Feng, Y.; Shen, C.; Yang, F. Integrating electrochemical oxidation into forward osmosis process for removal of trace antibiotics in wastewater. J. Hazard. Mater. 2015, 296, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Peng, L.; Su, H. The effect of a buffer function on the semi-continuous anaerobic digestion. Bioresour. Technol. 2013, 139, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Liaoa, X.; Zengb, F.; Akhilc, G.; Zhuc, J.; Liaoc, L. Experimental study on the mechanisms involved in the synergetic effect of anaerobic co-digestion of mature leachate with food waste. Desalin. Water Treat. 2021, 222, 137–144. [Google Scholar] [CrossRef]
- Luo, J.; Qian, G.; Liu, J.; Xu, Z.P. Anaerobic methanogenesis of fresh leachate from municipal solid waste: A brief review on current progress. Renew. Sustain. Energy Rev. 2015, 49, 21–28. [Google Scholar] [CrossRef]
- Montusiewicz, A.; Lebiocka, M. Co-digestion of intermediate landfill leachate and sewage sludge as a method of leachate utilization. Bioresour. Technol. 2011, 102, 2563–2571. [Google Scholar] [CrossRef]
- Takeda, P.Y.; Gotardo, J.T.; Gomes, S.D. Anaerobic co-digestion of leachate and glycerol for renewable energy generation. Environ. Technol. 2022, 43, 1118–1128. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, T.; Erdirencelebi, D.; Berktay, A. Effect of COD/SO ratio on anaerobic treatment of landfill leachate during the start-up period. Environ. Technol. 2012, 33, 313–320. [Google Scholar] [CrossRef]
- Luste, S.; Luostarinen, S.; Sillanpää, M. Effect of pre-treatments on hydrolysis and methane production potentials of by-products from meat-processing industry. J. Hazard. Mater. 2009, 164, 247–255. [Google Scholar] [CrossRef]
- Dougan, I.; Sanin, F.D. Alkaline solubilization and microwave irradiation as a combined sludge disintegration and minimization method. Water Res. 2009, 43, 2139–2148. [Google Scholar] [CrossRef]
- Song, K.; Ren, X.; Zhang, Q.; Xu, L.; Liu, D. Electrochemical treatment for leachate membrane retentate: Performance comparison of electrochemical oxidation and electro-coagulation technology. Chemosphere 2022, 303, 134986. [Google Scholar]
- Wu, Z.; Qiao, W.; Liu, Y.; Yao, J.; Gu, C.; Zheng, X.; Dong, R. Contribution of chemical precipitation to the membrane fouling in a high-solids type anaerobic membrane bioreactor treating OFMSW leachate. J. Memb. Sci. 2022, 647, 120298. [Google Scholar] [CrossRef]
- Guo, Z.; Zhang, Y.; Jia, H.; Guo, J.; Meng, X.; Wang, J. Electrochemical methods for landfill leachate treatment: A review on electrocoagulation and electrooxidation. Sci. Total Environ. 2022, 806, 150529. [Google Scholar] [CrossRef] [PubMed]
- Nomura, Y.; Fukahori, S.; Fujiwara, T. Removal of 1, 4-dioxane from landfill leachate by a rotating advanced oxidation contactor equipped with activated carbon/TiO2 composite sheets. J. Hazard. Mater. 2020, 383, 121005. [Google Scholar] [CrossRef] [PubMed]
- Teng, C.; Zhou, K.; Yang, W.; Zhang, X.; Peng, C.; Chen, W. Unveiling the degradation of membrane concentrated landfill leachate during enhanced photocatalysis using spectroscopic approaches. J. Water Process Eng. 2021, 43, 102220. [Google Scholar] [CrossRef]
- Li, R.; Li, L.; Zhang, Z.; Chen, G.; Tang, Y. Limiting factors of heavy metals removal during anaerobic biological pretreatment of municipal solid waste landfill leachate. J. Hazard. Mater. 2021, 416, 126081. [Google Scholar] [CrossRef]
- Robinson, T. Removal of toxic metals during biological treatment of landfill leachates. Waste Manag. 2017, 63, 299–309. [Google Scholar] [CrossRef]
- Lei, X.; Sugiura, N.; Feng, C.; Maekawa, T. Pretreatment of anaerobic digestion effluent with ammonia stripping and biogas purification. J. Hazard. Mater. 2007, 145, 391–397. [Google Scholar] [CrossRef]
- Fernandes, A.; Pacheco, M.J.; Cir’iaco, L.; Lopes, A. Review on the electrochemical processes for the treatment of sanitary landfill leachates: Present and future. Appl. Catal. B Environ. 2015, 176, 183–200. [Google Scholar] [CrossRef]
- Ouhadi, V.R.; Yong, R.N.; Sedighi, M. Influence of heavy metal contaminants at variable pH regimes on rheological behaviour of bentonite. Appl. Clay Sci. 2006, 32, 217–231. [Google Scholar] [CrossRef]
- Sirés, I.; Brillas, E.; Oturan, M.A.; Rodrigo, M.A.; Panizza, M. Electrochemical advanced oxidation processes: Today and tomorrow. A review. Environ. Sci. Pollut. Res. 2014, 21, 8336–8367. [Google Scholar] [CrossRef]
- Urtiaga, A.; Gómez-Lav’in, S.; Soriano, A. Electrochemical treatment of municipal landfill leachates and implications for poly-and perfluoroalkyl substances (PFAS) removal. J. Environ. Chem. Eng. 2022, 10, 107900. [Google Scholar] [CrossRef]
- Xiong, C.; Li, G.; Zhang, Z.; Xia, Z.; Li, J.; Ye, H. Technique for advanced electrochemical oxidation treatment of nanofiltration concentrate of landfill leachate. Wuhan Univ. J. Nat. Sci. 2014, 19, 355–360. [Google Scholar] [CrossRef]
- Appels, L.; Dewil, R.; Baeyens, J.; Degrève, J. Ultrasonically enhanced anaerobic digestion of waste activated sludge. Int. J. Sustain. Eng. 2008, 1, 94–104. [Google Scholar] [CrossRef]
- Oz, N.A.; Yarimtepe, C.C. Ultrasound assisted biogas production from landfill leachate. Waste Manag. 2014, 34, 1165–1170. [Google Scholar] [CrossRef]
- Nazimudheen, G.; Roy, K.; Sivasankar, T.; Moholkar, V.S. Mechanistic investigations in ultrasonic pretreatment and anaerobic digestion of landfill leachates. J. Environ. Chem. Eng. 2018, 6, 1690–1701. [Google Scholar] [CrossRef]
- Naddeo, V.; Belgiorno, V.; Napoli, R.M.A. Behaviour of natural organic mater during ultrasonic irradiation. Desalination 2007, 210, 175–182. [Google Scholar] [CrossRef]
- Zhang, P.; Zhang, G.; Wang, W. Ultrasonic treatment of biological sludge: Floc disintegration, cell lysis and inactivation. Bioresour. Technol. 2007, 98, 207–210. [Google Scholar] [CrossRef]
- Shah, Y.T.; Pandit, A.B.; Moholkar, V.S. Cavitation Reaction Engineering; Springer Science & Business Media: Cham, Switzerland, 1999. [Google Scholar]
- Neczaj, E.; Kacprzak, M. Ultrasound as a pre-oxidation for biological landfill leachate treatment. Water Sci. Technol. 2007, 55, 175–179. [Google Scholar] [CrossRef]
- Reddy, C.V.; Rao, D.S.; Kalamdhad, A.S. Combined treatment of high-strength fresh leachate from municipal solid waste landfill using coagulation-flocculation and fixed bed upflow anaerobic filter. J. Water Process Eng. 2022, 46, 102554. [Google Scholar] [CrossRef]
- Smaoui, Y.; Mlaik, N.; Bouzid, J.; Sayadi, S. Improvement of anaerobic digestion of landfill leachate by using coagulation-flocculation, Fenton’s oxidation and air stripping pretreatments. Environ. Prog. Sustain. Energy 2018, 37, 1041–1049. [Google Scholar] [CrossRef]
- Castrillón, L.; Fernández-Nava, Y.; Ulmanu, M.; Anger, I.; Marañón, E. Physico-chemical and biological treatment of MSW landfill leachate. Waste Manag. 2010, 30, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Zhang, L.; Xu, Y.; Liang, C.; Kong, H.; Shi, X.; Peng, Y. Advanced nitrogen removal using bio-refractory organics as carbon source for biological treatment of landfill leachate. Sep. Purif. Technol. 2016, 170, 306–313. [Google Scholar] [CrossRef]
- Smaoui, Y.; Bouzid, J.; Sayadi, S. Combination of air stripping and biological processes for landfill leachate treatment. Environ. Eng. Res. 2020, 25, 80–87. [Google Scholar] [CrossRef]
- Abood, A.R.; Bao, J.; Abudi, Z.N.; Zheng, D.; Gao, C. Pretreatment of nonbiodegradable landfill leachate by air stripping coupled with agitation as ammonia stripping and coagulation--flocculation processes. Clean Technol. Environ. Policy 2013, 15, 1069–1076. [Google Scholar] [CrossRef]
- Hedayati, M.S.; Abida, O.; Li, L.Y. Adsorption of polycyclic aromatic hydrocarbons by surfactant-modified clinoptilolites for landfill leachate treatment. Waste Manag. 2021, 131, 503–512. [Google Scholar] [CrossRef]
- Azreen, I.; Zahrim, A.Y. Overview of biologically digested leachate treatment using adsorption. In Anaerobic Digestion Processes; Springer: Berlin/Heidelberg, Germany, 2018; pp. 123–148. [Google Scholar]
- Mojiri, A.; Zhou, J.L.; Ratnaweera, H.; Ohashi, A.; Ozaki, N.; Kindaichi, T.; Asakura, H. Treatment of landfill leachate with different techniques: An overview. J. Water Reuse Desalin. 2021, 11, 66–96. [Google Scholar] [CrossRef]
- Cui, Y.-R.; Guo, Y.; Wu, Q. Influence of biological activated carbon dosage on landfill leachate treatment. Huan Jing ke Xue= Huanjing Kexue 2014, 35, 3206–3211. [Google Scholar]
- Hedayati, M.S.; Li, L.Y. Removal of polycyclic aromatic hydrocarbons from aqueous media using modified clinoptilolite. J. Environ. Manag. 2020, 273, 111113. [Google Scholar] [CrossRef]
- Luo, Y.; Li, R.; Sun, X.; Liu, X.; Li, D. The roles of phosphorus species formed in activated biochar from rice husk in the treatment of landfill leachate. Bioresour. Technol. 2019, 288, 121533. [Google Scholar] [CrossRef]
- Lin, Y.; Wang, D.; Wu, S.; Wang, C. Alkali pretreatment enhances biogas production in the anaerobic digestion of pulp and paper sludge. J. Hazard. Mater. 2009, 170, 366–373. [Google Scholar] [CrossRef]
- Carlsson, M.; Lagerkvist, A.; Morgan-Sagastume, F. The effects of substrate pre-treatment on anaerobic digestion systems: A review. Waste Manag. 2012, 32, 1634–1650. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Park, C.; Kim, T.-H.; Lee, M.; Kim, S.; Kim, S.-W.; Lee, J. Effects of various pretreatments for enhanced anaerobic digestion with waste activated sludge. J. Biosci. Bioeng. 2003, 95, 271–275. [Google Scholar] [CrossRef]
- Ariunbaatar, J.; Panico, A.; Esposito, G.; Pirozzi, F.; Lens, P.N.L. Pretreatment methods to enhance anaerobic digestion of organic solid waste. Appl. Energy 2014, 123, 143–156. [Google Scholar] [CrossRef]
- us Saqib, N.; Adnan, R.; Shah, I. Modifications of pure and Ag doped TiO2 by pre-sulphated and calcination temperature treatments. Res. Chem. Intermed. 2017, 43, 6571–6588. [Google Scholar]
- Cai, F.-F.; Yang, Z.-H.; Huang, J.; Zeng, G.-M.; Wang, L.; Yang, J. Application of cetyltrimethylammonium bromide bentonite--titanium dioxide photocatalysis technology for pretreatment of aging leachate. J. Hazard. Mater. 2014, 275, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Sun, X.; Shan, H.; Zhang, H.; Xi, B. Degradation of landfill leachate using UV-TiO2 photocatalysis combination with aged waste reactors. Processes 2021, 9, 946. [Google Scholar] [CrossRef]
- Yasmin, C.; Lobna, E.; Mouna, M.; Kais, D.; Mariam, K.; Rached, S.; Abdelwaheb, C.; Ismail, T. New trend of Jebel Chakir landfill leachate pre-treatment by photocatalytic TiO2/Ag nanocomposite prior to fermentation using Candida tropicalis strain. Int. Biodeterior. Biodegrad. 2020, 146, 104829. [Google Scholar] [CrossRef]
- Mishra, S.; Sundaram, B. Efficacy of nanoparticles as photocatalyst in leachate treatment. Nanotechnol. Environ. Eng. 2022, 7, 1–20. [Google Scholar] [CrossRef]
- Chen, W.; Gu, Z.; Ran, G.; Li, Q. Application of membrane separation technology in the treatment of leachate in China: A review. Waste Manag. 2021, 121, 127–140. [Google Scholar] [CrossRef]
- Judd, S. The MBR Book: Principles and Applications of Membrane Bioreactors for Water and Wastewater Treatment; Elsevier: Amsterdam, The Netherlands, 2010. [Google Scholar]
- Hashisho, J.; El-Fadel, M. Membrane bioreactor technology for leachate treatment at solid waste landfills. Rev. Environ. Sci. Bio/Technol. 2016, 15, 441–463. [Google Scholar] [CrossRef]
- Sutzkover-Gutman, I.; Hasson, D.; Semiat, R. Humic substances fouling in ultrafiltration processes. Desalination 2010, 261, 218–231. [Google Scholar] [CrossRef]
- Saleem, M.; Spagni, A.; Alibardi, L.; Bertucco, A.; Lavagnolo, M.C. Assessment of dynamic membrane filtration for biological treatment of old landfill leachate. J. Environ. Manag. 2018, 213, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Talalaj, I.A. Mineral and organic compounds in leachate from landfill with concentrate recirculation. Environ. Sci. Pollut. Res. 2015, 22, 2622–2633. [Google Scholar] [CrossRef]
- Long, Y.; Xu, J.; Shen, D.; Du, Y.; Feng, H. Effective removal of contaminants in landfill leachate membrane concentrates by coagulation. Chemosphere 2017, 167, 512–519. [Google Scholar] [CrossRef]
- Navaratnasamy, M.; Edeogu, I.; Papworth, L. Economic Feasibility of Anaerobic Digesters; Alberta Agriculture and Rural Development: Edmonton, AB, Canada, 2008. [Google Scholar]
- Segeren, A.; Luijt, J. Segmenting the market for agricultural land in the Netherlands. Tijdschr. Econ. Soc. Geogr. 2002, 93, 570–575. [Google Scholar] [CrossRef]
- Gebrezgabher, S.A.; Meuwissen, M.P.M.; Prins, B.A.M.; Lansink, A.G.J.M.O. Economic analysis of anaerobic digestion—A case of Green power biogas plant in The Netherlands. NJAS Wagening. J. Life Sci. 2010, 57, 109–115. [Google Scholar] [CrossRef]
- Van Rooijen, S.N.M.; Van Wees, M.T. Green electricity policies in the Netherlands: An analysis of policy decisions. Energy Policy 2006, 34, 60–71. [Google Scholar] [CrossRef]
- Ye, Z.-L.; Hong, Y.; Pan, S.; Huang, Z.; Chen, S.; Wang, W. Full-scale treatment of landfill leachate by using the mechanical vapor recompression combined with coagulation pretreatment. Waste Manag. 2017, 66, 88–96. [Google Scholar] [CrossRef]
- Brasil, Y.L.; Silva, A.F.R.; Gomes, R.F.; Amaral, M.C.S. Technical and economic evaluation of the integration of membrane bioreactor and air-stripping/absorption processes in the treatment of landfill leachate. Waste Manag. 2021, 134, 110–119. [Google Scholar] [CrossRef]
- Zhang, T.; Wu, X.; Li, H.; Tsang, D.C.W.; Li, G.; Ren, H. Struvite pyrolysate cycling technology assisted by thermal hydrolysis pretreatment to recover ammonium nitrogen from composting leachate. J. Clean. Prod. 2020, 242, 118442. [Google Scholar] [CrossRef]
- Xiong, J.; Zheng, Z.; Yang, X.; Dai, X.; Zhou, T.; He, J.; Luo, X. Recovery of NH3-N from mature leachate via negative pressure steam-stripping pretreatment and its benefits on MBR systems: A pilot scale study. J. Clean. Prod. 2018, 203, 918–925. [Google Scholar] [CrossRef]
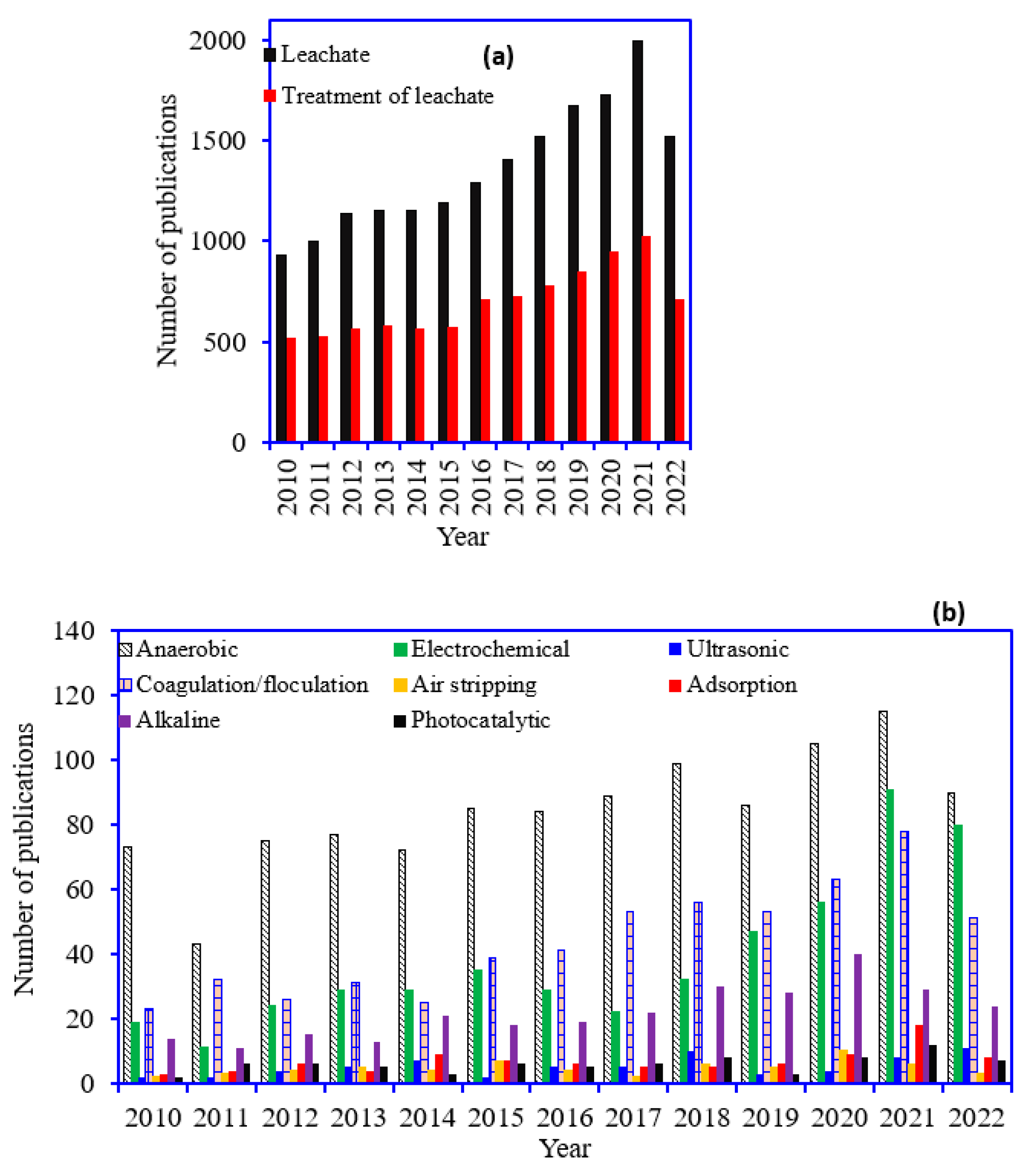





| Leachate Type | Leachate Age * | Anaerobic Reactor | Reactor Conditions | Degradation Efficiency (Organic Matter% Removal) | Methane/Biogas Yield (m3/L) | Ref. |
|---|---|---|---|---|---|---|
| Municipal Solid Waste leachate | Fresh leachate | Up-flow Anaerobic Sludge Blanket (UASB) | OLR:18.2 kgCOD/m3/d HRT: 50 d | 89 | 0.0048 | [41] |
| LFL | Fresh leachate | Batch Reactor (BR) | Temp: 37 °C HRT: 24 d | 53 | 0.292 | [42] |
| Pressed off leachate | NR | BR | OLD: 27.7 kg COD/m3/d Temp: 37 °C | 60 | 7.1 | [43] |
| Landfill leachate | Mature leachate | Anaerobic Submerged Membrane Bioreactor | OLR: 2.5 kg COD/m3/d HRT: 2 d | 90 | NR ** | [44] |
| Leachate with food waste | Fresh Leachate | UASB | OLR: 15.8 g COD/l/d. | 96 | 0.055 | [45] |
| Raw Leachate With food waste | Middle aged leachate | BR | OLR: 41.8 g VS/L. HRT 35 d | NR | 0.466 | [46] |
| Food waste leachate | NR | Bench-scale Two-Stage System (BSTSS) | Temp: 37 °C HRT: 99 d | 94.3 | 0.393 | [47] |
| Municipal Solid Waste leachate + Sludge | Fresh leachate | Stirring Batch Reactor (SBR) | Temp: 55 °C HRT: 90 d pH: 6.4–7.4 | 81.8 | 0.1173 | [48] |
| LFL | Fresh leachate | Two-stage Anaerobic Sequential System | HRT: 21 d pH: 5–11 | 81 | 0.320 | [49] |
| LFL | Fresh leachate | Anaerobic bioreactor (fixed-structured bed) | Temp: 30 °C HRT: 24 d OLR: 7.1 gCOD/L/d | NR | 0.30 | [50] |
| NR | leachate bed reactor—anaerobic membrane bioreactor system (LBR-AnMBR) | Temp: 21–22 °C Leachate recirculation rate: 4.4–13.2 L/h | 86 | 0.31 | [51] | |
| LFL + Activated sludge | Middle aged | Continuous stirred tank reactors | Temp: 37 °C, OLR: 1.3 g VS/L/day Operation: 180 d | 91.3 | 0.105 | [52] |
| Food waste + Mature leachate | Mature leachate | Expanded granular sludge blanket reactor | Temp: 35 °C OLR: 23.6 g COD/L/d | 80 | 5.87 (L/L/d) | [53] |
| MSW leachate | NR | Anaerobic biofilm digester (ABD) | OLR: 1.27 g COD/ L/d HRT: 30 d | 95 | 17 (m3/d) | [54] |
| Landfill leachate | NR | anaerobic membrane bioreactor | Temp: 35 °C Operation: 185 d Working vol: 6.3 l | 78 | 436 (ml/L approx.) | [55] |
| OFMSW + Leachate | Fresh leachate | Solid-state anaerobic garage-type digesters | HRT: 72 d OLR: 0.5 kg VS/m/d | 56% | 0.06 m3 CH4.kg VS | [56] |
| + Landfill leachate | Mature | Semi-continuous anaerobic reactors | OLR: 1.0 gVS/L/d HRT: 20 d | NR | 0.260 NL/ gVS added. | [57] |
| LFL + Synthetic wastewater | Mature | UASB | Temp: 37 °C HRT: 1 d OLR: 2 gCOD/L/day−1 | 93 | 0.035 L g-COD−1 | [58] |
| Substrate | Treatment Process/System | Target Pollutant | Pollutant Removal Efficiency | References |
|---|---|---|---|---|
| Leachate membrane retentate | Electrochemical oxidation |
|
| [70] |
| OFMSW leachate | Anaerobic membrane bioreactors |
|
| [71] |
| Mixed landfill leachate | Full scale leachate treatment plant (biological + UF-NF-RO membrane) |
|
| [72] |
| MSW leachate | Biological and photochemical processes |
|
| [15] |
| Landfill leachate | Photocatalysis |
|
| [73] |
| Membrane concentrated landfill leachate | Photocatalysis |
|
| [74] |
| Landfill leachate | Submerged anaerobic biofilm reactor |
|
| [75] |
| Landfill leachate | Ultrafiltration- biological treatment |
|
| [76] |
| Landfill leachate | anaerobic sequencing batch reactor coupled with sequencing batch reactor (ASBR-SBR) |
|
| [62] |
| Leachate (anaerobic digestion effluent) | Ammonia stripping pretreatment-Anaerobic digestion |
|
| [77] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anjum, M.; Anees, M.; Qadeer, S.; Khalid, A.; Kumar, R.; Barakat, M.A. A Recent Progress in the Leachate Pretreatment Methods Coupled with Anaerobic Digestion for Enhanced Biogas Production: Feasibility, Trends, and Techno-Economic Evaluation. Int. J. Mol. Sci. 2023, 24, 763. https://doi.org/10.3390/ijms24010763
Anjum M, Anees M, Qadeer S, Khalid A, Kumar R, Barakat MA. A Recent Progress in the Leachate Pretreatment Methods Coupled with Anaerobic Digestion for Enhanced Biogas Production: Feasibility, Trends, and Techno-Economic Evaluation. International Journal of Molecular Sciences. 2023; 24(1):763. https://doi.org/10.3390/ijms24010763
Chicago/Turabian StyleAnjum, Muzammil, Mobeena Anees, Samia Qadeer, Azeem Khalid, Rajeev Kumar, and Mohamed. A. Barakat. 2023. "A Recent Progress in the Leachate Pretreatment Methods Coupled with Anaerobic Digestion for Enhanced Biogas Production: Feasibility, Trends, and Techno-Economic Evaluation" International Journal of Molecular Sciences 24, no. 1: 763. https://doi.org/10.3390/ijms24010763
APA StyleAnjum, M., Anees, M., Qadeer, S., Khalid, A., Kumar, R., & Barakat, M. A. (2023). A Recent Progress in the Leachate Pretreatment Methods Coupled with Anaerobic Digestion for Enhanced Biogas Production: Feasibility, Trends, and Techno-Economic Evaluation. International Journal of Molecular Sciences, 24(1), 763. https://doi.org/10.3390/ijms24010763









