Toxic Effects of Glyphosate on the Nervous System: A Systematic Review
Abstract
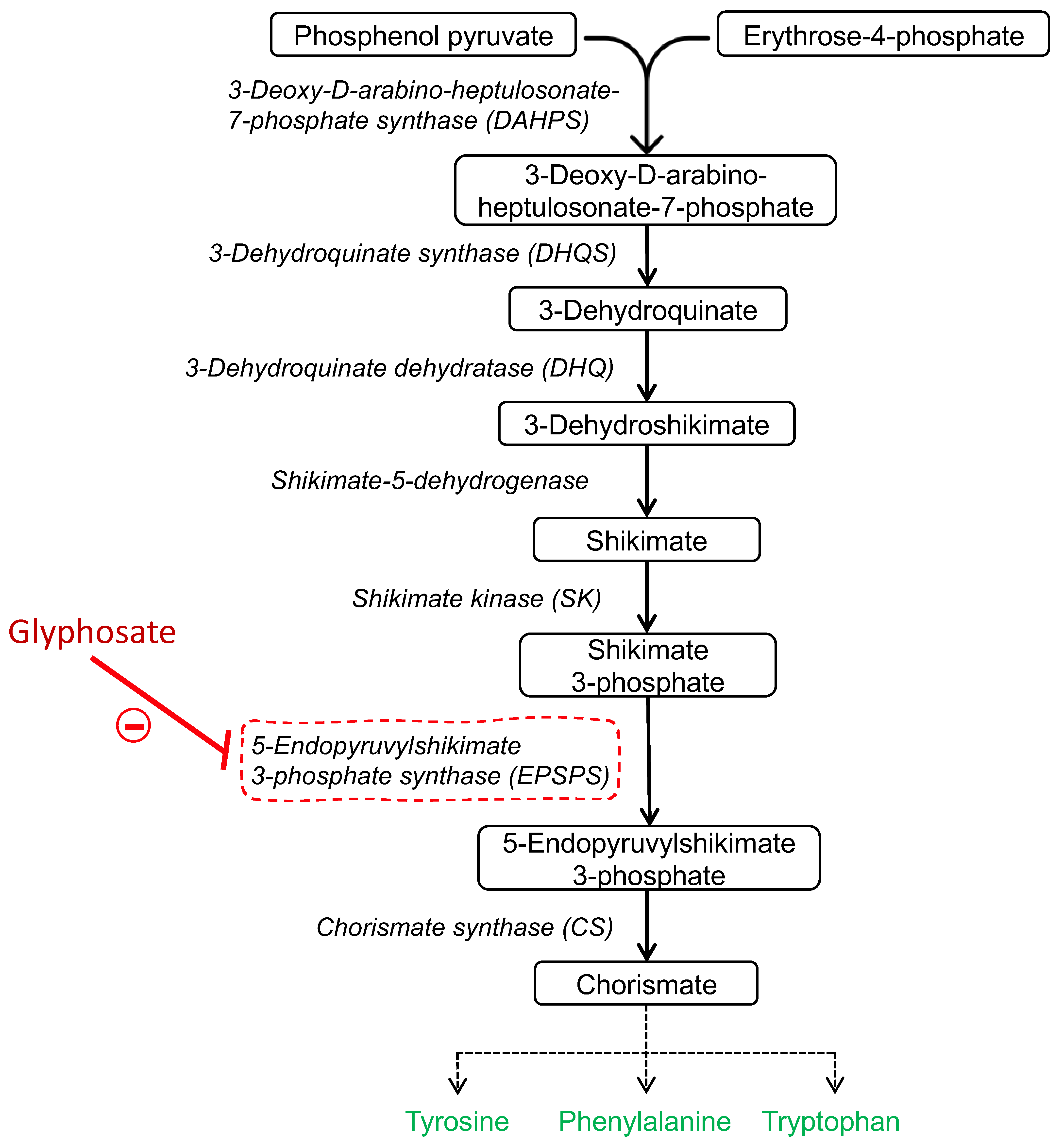
1. Introduction
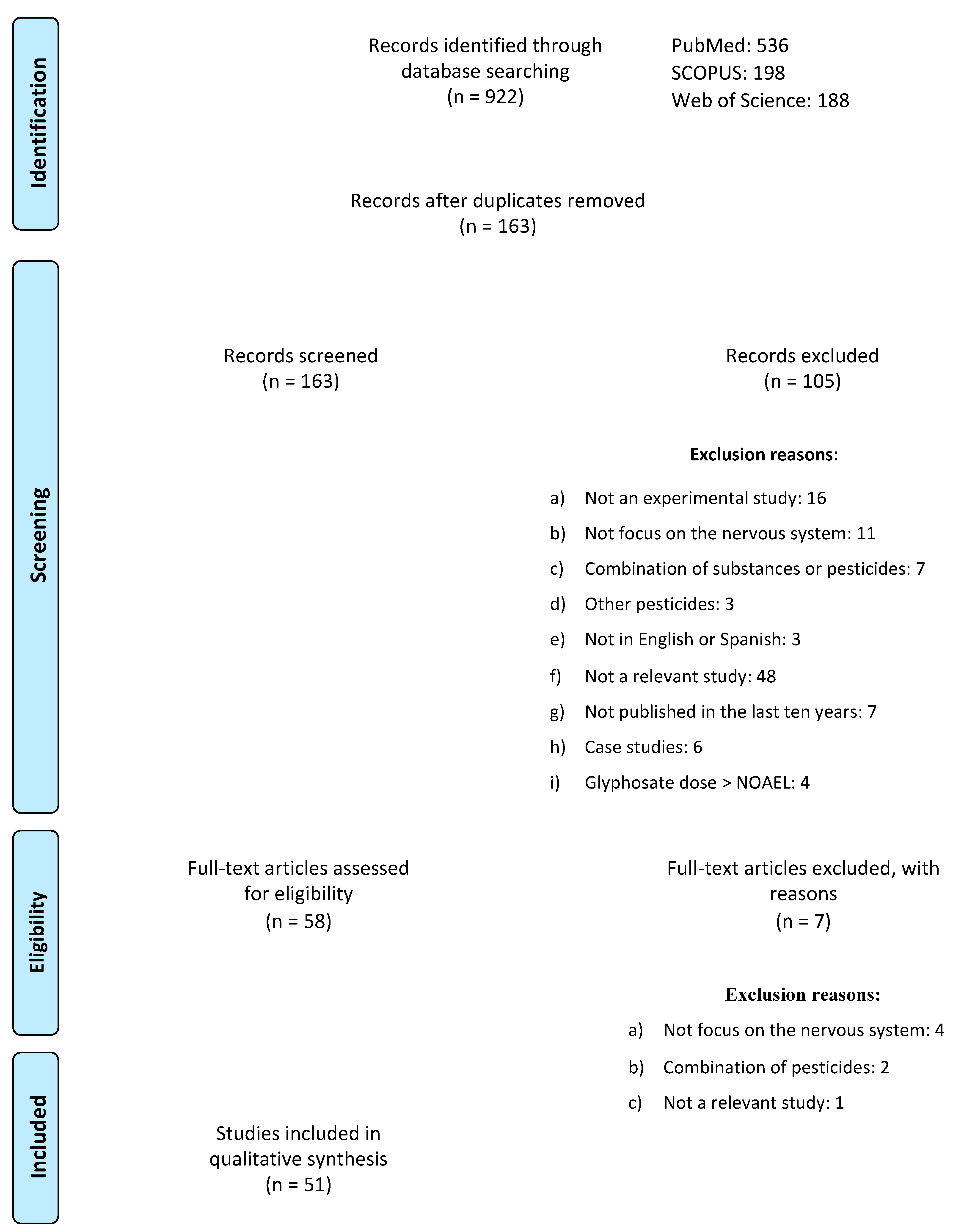
2. Methodology
Exclusion and Inclusion Criteria
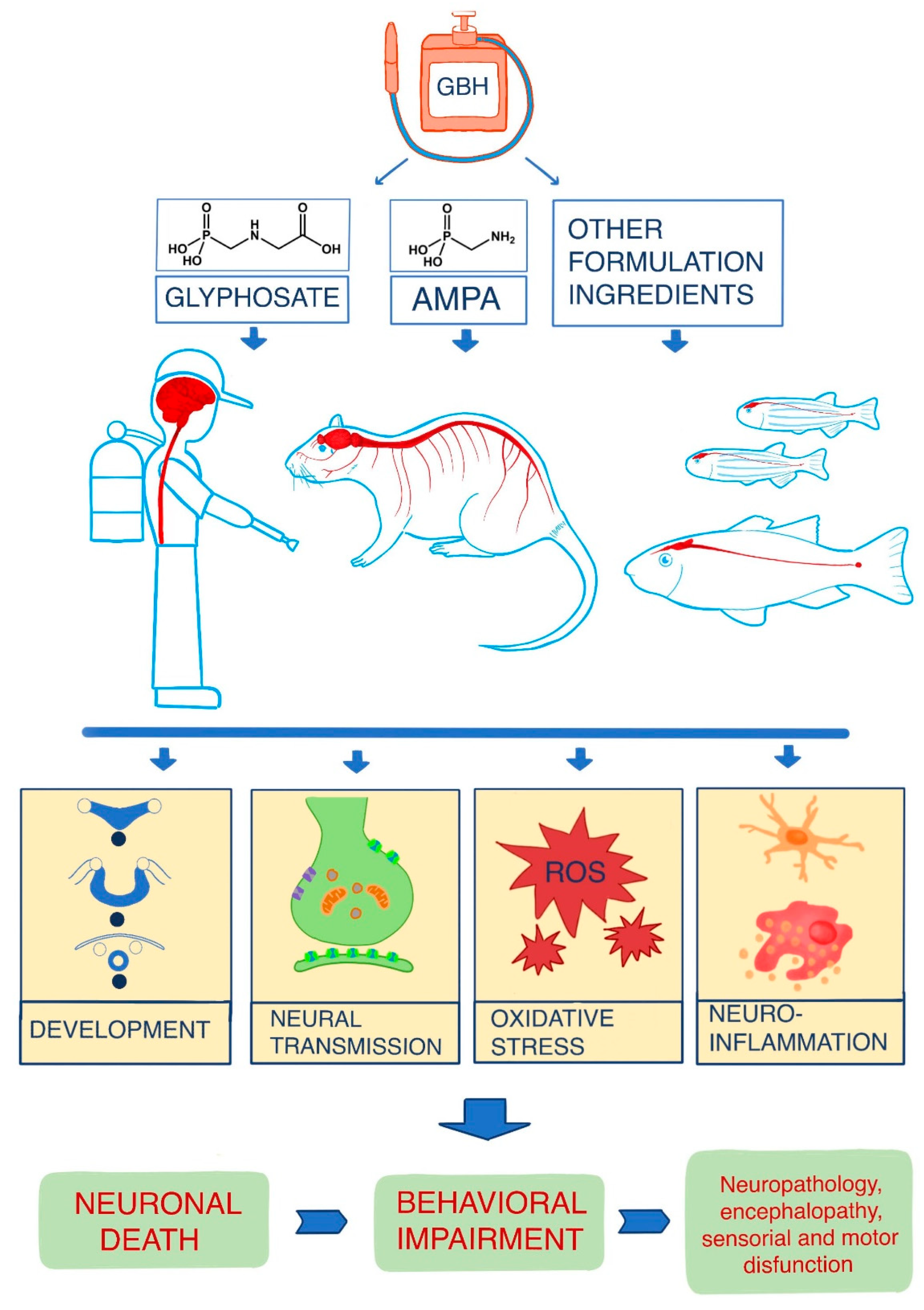
3. Results
3.1. Effects of Glyphosate in Humans
3.1.1. Descriptive and Analytical Studies
3.1.2. In Vitro Studies with Human Line Cells
3.2. Effects of Glyphosate in Rodents
3.2.1. Development of Nervous System
3.2.2. Effects on Neurotransmission
3.2.3. Effects on Behavior
3.2.4. Induction of Oxidative Stress and Inflammation
3.2.5. Induction of Apoptosis and Autophagy
3.3. Effects of Glyphosate in Fish
3.3.1. Development of Nervous System
3.3.2. Effects on Behavior
3.3.3. Effects on Neurotransmission
3.3.4. Induction of Oxidative Stress and Inflammation
3.3.5. Effects on Energy Metabolism
3.4. Effects of Glyphosate on Invertebrates
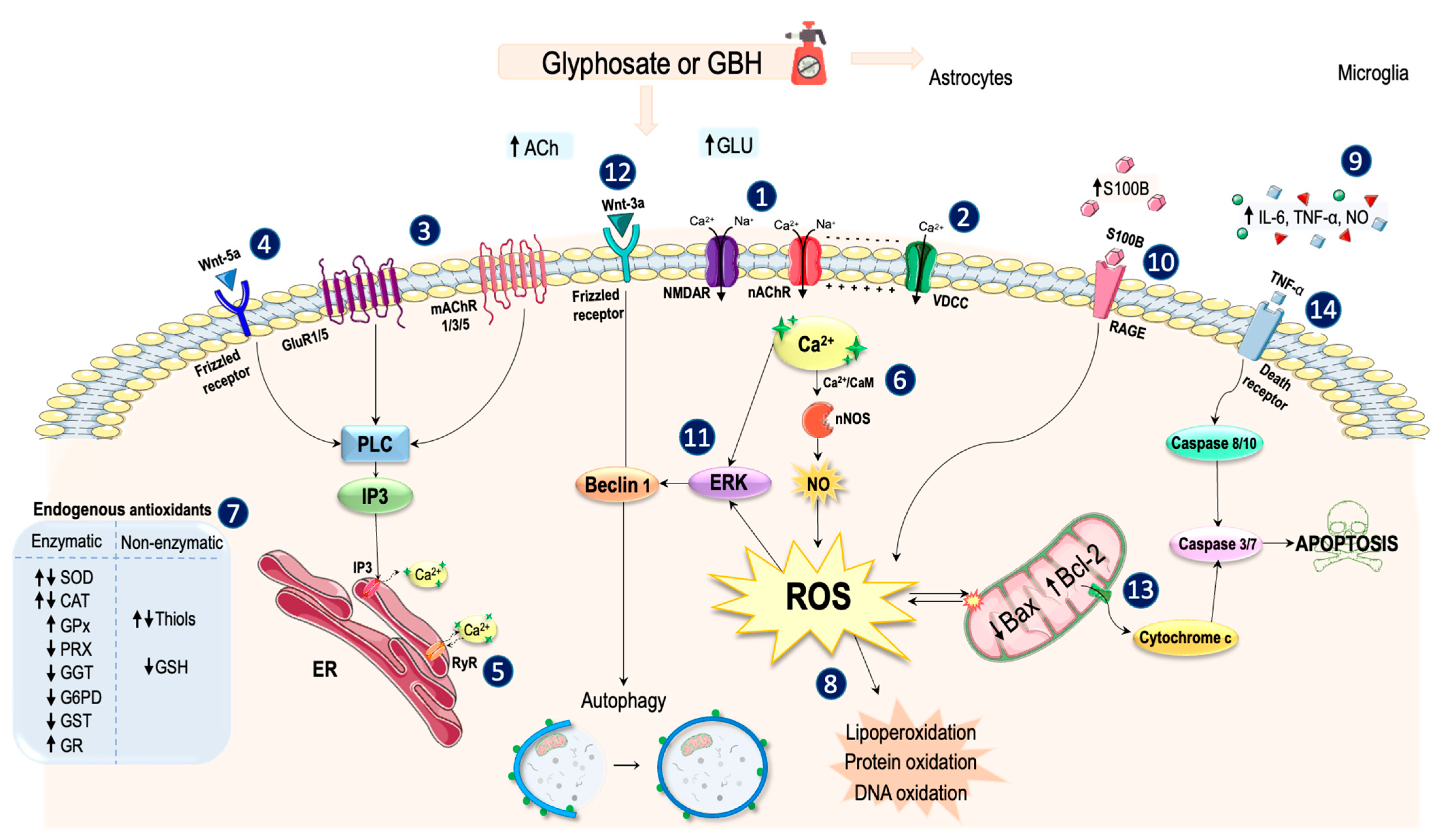
4. Discussion
4.1. Overview of the Main Mechanisms of Action of Glyphosate on the Nervous System
4.2. Relationship between Glyphosate Doses and Neurotoxic Effects in Rodents and Humans
4.3. Exposure Levels and Neurotoxic Effects in Fish
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dayan, F.E.; Barker, A.; Takano, H.; Bough, R.; Ortiz, M.; Duke, S.O. Herbicide mechanism of action and resistance. In Comprehensive Biotechnology, 3rd ed.; Moo-Young, M., Ed.; Elsevier: Amsterdam, The Netherlands, 2020; Volume 4, p. 4826. [Google Scholar]
- Duke, S.O. The history and current status of glyphosate. Pest Manag. Sci. 2018, 74, 1027–1034. [Google Scholar] [CrossRef]
- Cattani, D.; Cesconetto, P.A.; Tavares, M.K.; Parisotto, E.B.; Oliveira, P.V.; Rieg, C.E.H.; Leite, M.C.; Prediger, R.; Wendt, N.; Razzera, G.; et al. Developmental exposure to glyphosate-based herbicide and depressive-like behavior in adult offspring: Implication of glutamate excitotoxicity and oxidative stress. Toxicology 2017, 387, 67–80. [Google Scholar] [CrossRef]
- Klümper, W.; Qaim, M. A Meta-Analysis of the Impacts of Genetically Modified Crops. PLoS ONE 2014, 9, e111629. [Google Scholar] [CrossRef]
- Maggi, F.; la Cecilia, D.; Tang, F.H.; McBratney, A. The global environmental hazard of glyphosate use. Sci. Total Environ. 2020, 717, 137167. [Google Scholar] [CrossRef] [PubMed]
- Saunders, L.E.; Pezeshki, R. Glyphosate in Runoff Waters and in the Root-Zone: A Review. Toxics 2015, 3, 462–480. [Google Scholar] [CrossRef] [PubMed]
- Funke, T.; Han, H.; Healy-Fried, M.L.; Fischer, M.; Schönbrunn, E. Molecular basis for the herbicide resistance of Roundup Ready crops. Proc. Natl. Acad. Sci. USA 2006, 103, 13010–13015. [Google Scholar] [CrossRef] [PubMed]
- Meftaul, I.M.; Venkateswarlu, K.; Dharmarajan, R.; Annamalai, P.; Asaduzzaman; Parven, A.; Megharaj, M. Controversies over human health and ecological impacts of glyphosate: Is it to be banned in modern agriculture? Environ. Pollut. 2020, 263, 114372. [Google Scholar] [CrossRef]
- Van Bruggen, A.H.C.; He, M.M.; Shin, K.; Mai, V.; Jeong, K.C.; Finckh, M.R.; Morris, J.G., Jr. Environmental and health effects of the herbicide glyphosate. Sci. Total Environ. 2018, 616–617, 255–268. [Google Scholar] [CrossRef]
- Cox, C. Glyphosate (roundup). J. Pestic. Reform. 1998, 18, 3–17. [Google Scholar]
- Kier, L.D.; Kirkland, D.J. Review of genotoxicity studies of glyphosate and glyphosate-based formulations. Crit. Rev. Toxicol. 2013, 43, 283–315. [Google Scholar] [CrossRef]
- Williams, G.M.; Aardema, M.; Acquavella, J.; Berry, S.C.; Brusick, D.; Burns, M.M.; De Camargo, J.L.V.; Garabrant, D.; Greim, H.A.; Kier, L.D.; et al. A review of the carcinogenic potential of glyphosate by four independent expert panels and comparison to the IARC assessment. Crit. Rev. Toxicol. 2016, 46, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Martínez, M.-A.; Ares, I.; Rodríguez, J.-L.; Martínez, M.; Martínez-Larrañaga, M.-R.; Anadón, A. Neurotransmitter changes in rat brain regions following glyphosate exposure. Environ. Res. 2018, 161, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.M.; Kroes, R.; Munro, I.C. Safety Evaluation and Risk Assessment of the Herbicide Roundup and Its Active Ingredient, Glyphosate, for Humans. Regul. Toxicol. Pharmacol. 2000, 31, 117–165. [Google Scholar] [CrossRef] [PubMed]
- Kanissery, R.; Gairhe, B.; Kadyampakeni, D.; Batuman, O.; Alferez, F. Glyphosate: Its Environmental Persistence and Impact on Crop Health and Nutrition. Plants 2019, 8, 499. [Google Scholar] [CrossRef]
- Bento, C.P.M.; Yang, X.; Gort, G.; Xue, S.; van Dam, R.; Zomer, P.; Mol, H.G.J.; Ritsema, C.J.; Geissen, V. Persistence of glyphosate and aminomethylphosphonic acid in loess soil under different combinations of temperature, soil moisture and light/darkness. Sci. Total Environ. 2016, 572, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Bergström, L.; Börjesson, E.; Stenström, J. Laboratory and Lysimeter Studies of Glyphosate and Aminomethylphosphonic Acid in a Sand and a Clay Soil. J. Environ. Qual. 2011, 40, 98–108. [Google Scholar] [CrossRef]
- Laitinen, P.; Siimes, K.; Eronen, L.; Rämö, S.; Welling, L.; Oinonen, S.; Mattsoff, L.; Ruohonen-Lehto, M. Fate of the herbicides glyphosate, glufosinate-ammonium, phenmedipham, ethofumesate and metamitron in two Finnish arable soils. Pest Manag. Sci. 2006, 62, 473–491. [Google Scholar] [CrossRef]
- Tzanetou, E.; Karasali, H. Glyphosate Residues in Soil and Air: An Integrated Review. In Pests, Weeds and Diseases in Agricultural Crop and Animal Husbandry Production; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef]
- De Andrea, M.M.; Peres, T.B.; Luchini, L.C.; Bazarin, S.; Papini, S.; Matallo, M.B.; Savoy, V.L.T. Influence of repeated applications of glyphosate on its persistence and soil bioactivity. Pesqui. Agropecuária Bras. 2003, 38, 1329–1335. [Google Scholar] [CrossRef]
- Carretta, L.; Cardinali, A.; Onofri, A.; Masin, R.; Zanin, G. Dynamics of Glyphosate and Aminomethylphosphonic Acid in Soil Under Conventional and Conservation Tillage. Int. J. Environ. Res. 2021, 15, 1037–1055. [Google Scholar] [CrossRef]
- Leoci, R.; Ruberti, M. Glyphosate in Agriculture: Environmental Persistence and Effects on Animals. A Review. J. Agric. Environ. Int. Dev. 2020, 114, 99–122. [Google Scholar] [CrossRef]
- Battaglin, W.A.; Meyer, M.T.; Kuivila, K.M.; Dietze, J.E. Glyphosate and Its Degradation Product AMPA Occur Frequently and Widely in U.S. Soils, Surface Water, Groundwater, and Precipitation. JAWRA J. Am. Water Resour. Assoc. 2014, 50, 275–290. [Google Scholar] [CrossRef]
- Berman, M.C.; Marino, D.; Quiroga, M.V.; Zagarese, H. Occurrence and levels of glyphosate and AMPA in shallow lakes from the Pampean and Patagonian regions of Argentina. Chemosphere 2018, 200, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Mercurio, P.; Flores, F.; Mueller, J.F.; Carter, S.; Negri, A.P. Glyphosate persistence in seawater. Mar. Pollut. Bull. 2014, 85, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Review of the existing maximum residue levels for glyphosate according to Article 12 of Regulation (EC) No 396/2005. EFSA J. 2018, 16. [CrossRef]
- Edge, C.; Brown, M.; Heartz, S.; Thompson, D.; Ritter, L.; Ramadoss, M. The Persistence of Glyphosate in Vegetation One Year after Application. Forests 2021, 12, 601. [Google Scholar] [CrossRef]
- Niemann, L.; Sieke, C.; Pfeil, R.; Solecki, R. A critical review of glyphosate findings in human urine samples and comparison with the exposure of operators and consumers. J. Verbr. Lebensm. 2015, 10, 3–12. [Google Scholar] [CrossRef]
- Reddy, K.N.; Rimando, A.M.; Duke, S.O.; Nandula, V.K. Aminomethylphosphonic Acid Accumulation in Plant Species Treated with Glyphosate. J. Agric. Food Chem. 2008, 56, 2125–2130. [Google Scholar] [CrossRef]
- Acquavella, J.F.; Alexander, B.H.; Mandel, J.S.; Gustin, C.; Baker, B.; Chapman, P.; Bleeke, M. Glyphosate biomonitoring for farmers and their families: Results from the Farm Family Exposure Study. Environ. Health Perspect. 2004, 112, 321–326. [Google Scholar] [CrossRef]
- Conrad, A.; Schröter-Kermani, C.; Hoppe, H.-W.; Rüther, M.; Pieper, S.; Kolossa-Gehring, M. Glyphosate in German adults—Time trend (2001 to 2015) of human exposure to a widely used herbicide. Int. J. Hyg. Environ. Health 2017, 220, 8–16. [Google Scholar] [CrossRef]
- Krüger, M.; Schledorn, P.; Schrödl, W.; Hoppe, H.W.; Lutz, W.; Shehata, A.A. Detection of Glyphosate Residues in Animals and Humans. J. Environ. Anal. Toxicol. 2014, 4, 1000210. [Google Scholar] [CrossRef]
- Krüger, M.; Schrödl, W.; Pedersen, I. Detection of Glyphosate in Malformed Piglets. J. Environ. Anal. Toxicol. 2014, 4, 1000230. [Google Scholar] [CrossRef]
- Tarazona, J.V.; Court-Marques, D.; Tiramani, M.; Reich, H.; Pfeil, R.; Istace, F.; Crivellente, F. Glyphosate toxicity and carcinogenicity: A review of the scientific basis of the European Union assessment and its differences with IARC. Arch. Toxicol. 2017, 91, 2723–2743. [Google Scholar] [CrossRef] [PubMed]
- Currie, Z.; Prosser, R.S.; Rodriguez-Gil, J.L.; Mahon, K.; Poirier, D.; Solomon, K.R. Toxicity of Cúspide 480SL® spray mixture formulation of glyphosate to aquatic organisms. Environ. Toxicol. Chem. 2015, 34, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Smeda, R.J.; Sellers, B.A.; Johnson, W.G. Influence of formulation and glyphosate salt on absorption and translocation in three annual weeds. Weed Sci. 2005, 53, 153–159. [Google Scholar] [CrossRef]
- Cattani, D.; Cavalli, V.L.D.L.O.; Rieg, C.E.H.; Domingues, J.T.; Dal-Cim, T.; Tasca, C.I.; Silva, F.R.M.B.; Zamoner, A. Mechanisms underlying the neurotoxicity induced by glyphosate-based herbicide in immature rat hippocampus: Involvement of glutamate excitotoxicity. Toxicology 2014, 320, 34–45. [Google Scholar] [CrossRef]
- Bringolf, R.B.; Cope, W.G.; Mosher, S.; Barnhart, M.C.; Shea, D. Acute and chronic toxicity of glyphosate compounds to glochidia and juveniles of lampsilis siliquoidea (unionidae). Environ. Toxicol. Chem. 2007, 26, 2094–2100. [Google Scholar] [CrossRef]
- Rodrigues, L.D.B.; de Oliveira, R.; Abe, F.R.; Brito, L.B.; Moura, D.S.; Valadares, M.C.; Grisolia, C.K.; de Oliveira, D.P.; de Oliveira, G.A.R. Ecotoxicological assessment of glyphosate-based herbicides: Effects on different organisms. Environ. Toxicol. Chem. 2016, 36, 1755–1763. [Google Scholar] [CrossRef]
- Mesnage, R.; Bernay, B.; Séralini, G.-E. Ethoxylated adjuvants of glyphosate-based herbicides are active principles of human cell toxicity. Toxicology 2013, 313, 122–128. [Google Scholar] [CrossRef]
- Nobels, I.; Spanoghe, P.; Haesaert, G.; Robbens, J.; Blust, R. Toxicity Ranking and Toxic Mode of Action Evaluation of Commonly Used Agricultural Adjuvants on the Basis of Bacterial Gene Expression Profiles. PLoS ONE 2011, 6, e24139. [Google Scholar] [CrossRef]
- Prosser, R.S.; Rodriguez-Gil, J.L.; Solomon, K.R.; Sibley, P.K.; Poirier, D.G. Effects of the herbicide surfactant MON 0818 on oviposition and viability of eggs of the ramshorn snail (Planorbella pilsbryi). Environ. Toxicol. Chem. 2016, 36, 522–531. [Google Scholar] [CrossRef]
- Bai, S.H.; Ogbourne, S. Glyphosate: Environmental contamination, toxicity and potential risks to human health via food contamination. Environ. Sci. Pollut. Res. 2016, 23, 18988–19001. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Fuhrimann, S.; Farnham, A.; Staudacher, P.; Atuhaire, A.; Manfioletti, T.; Niwagaba, C.B.; Namirembe, S.; Mugweri, J.; Winkler, M.S.; Portengen, L.; et al. Exposure to multiple pesticides and neurobehavioral outcomes among smallholder farmers in Uganda. Environ. Int. 2021, 152, 106477. [Google Scholar] [CrossRef]
- Lee, J.-W.; Choi, Y.-J.; Park, S.; Gil, H.-W.; Song, H.-Y.; Hong, S.-Y. Serum S100 protein could predict altered consciousness in glyphosate or glufosinate poisoning patients. Clin. Toxicol. 2017, 55, 357–359. [Google Scholar] [CrossRef] [PubMed]
- Von Ehrenstein, O.S.; Ling, C.; Cui, X.; Cockburn, M.; Park, A.S.; Yu, F.; Wu, J.; Ritz, B. Prenatal and infant exposure to ambient pesticides and autism spectrum disorder in children: Population based case-control study. BMJ 2019, 364, l962. [Google Scholar] [CrossRef]
- Zhang, C.; Hu, R.; Huang, J.; Huang, X.; Shi, G.; Li, Y.; Yin, Y.; Chen, Z. Health effect of agricultural pesticide use in China: Implications for the development of GM crops. Sci. Rep. 2016, 6, 34918. [Google Scholar] [CrossRef]
- Zhang, C.; Sun, Y.; Hu, R.; Huang, J.; Huang, X.; Li, Y.; Yin, Y.; Chen, Z. A comparison of the effects of agricultural pesticide uses on peripheral nerve conduction in China. Sci. Rep. 2018, 8, 9621. [Google Scholar] [CrossRef]
- Hao, Y.; Zhang, Y.; Ni, H.; Gao, J.; Yang, Y.; Xu, W.; Tao, L. Evaluation of the cytotoxic effects of glyphosate herbicides in human liver, lung, and nerve. J. Environ. Sci. Health Part B 2019, 54, 737–744. [Google Scholar] [CrossRef]
- Martinez, A.; Al-Ahmad, A.J. Effects of glyphosate and aminomethylphosphonic acid on an isogeneic model of the human blood-brain barrier. Toxicol. Lett. 2018, 304, 39–49. [Google Scholar] [CrossRef]
- Martínez, M.-A.; Rodriguez-Gutierrez, J.-L.; Torres, B.L.; Martínez, M.; Martínez-Larrañaga, M.-R.; Maximiliano, J.-E.; Anadón, A.; Ares, I. Use of human neuroblastoma SH-SY5Y cells to evaluate glyphosate-induced effects on oxidative stress, neuronal development and cell death signaling pathways. Environ. Int. 2019, 135, 105414. [Google Scholar] [CrossRef]
- Luo, J.; Chen, J.; Deng, Z.-L.; Luo, X.; Song, W.-X.; Sharff, K.A.; Tang, N.; Haydon, R.C.; Luu, H.H.; He, T.-C. Wnt signaling and human diseases: What are the therapeutic implications? Lab. Investig. 2007, 87, 97–103. [Google Scholar] [CrossRef]
- Okerlund, N.D.; Cheyette, B.N.R. Synaptic Wnt signaling—a contributor to major psychiatric disorders? J. Neurodev. Disord. 2011, 3, 162–174. [Google Scholar] [CrossRef] [PubMed]
- Kwan, V.; Unda, B.K.; Singh, K.K. Wnt signaling networks in autism spectrum disorder and intellectual disability. J. Neurodev. Disord. 2016, 8, 45. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yuan, X.-S.; Wang, Z.; Li, R. The canonical Wnt signaling pathway in autism. CNS Neurol. Disord.—Drug Targets 2014, 13, 765–770. [Google Scholar] [CrossRef] [PubMed]
- Good, P. Evidence the U.S. autism epidemic initiated by acetaminophen (Tylenol) is aggravated by oral antibiotic amoxicillin/clavulanate (Augmentin) and now exponentially by herbicide glyphosate (Roundup). Clin. Nutr. ESPEN 2018, 23, 171–183. [Google Scholar] [CrossRef] [PubMed]
- Pu, Y.; Yang, J.; Chang, L.; Qu, Y.; Wang, S.; Zhang, K.; Xiong, Z.; Zhang, J.; Tan, Y.; Wang, X.; et al. Maternal glyphosate exposure causes autism-like behaviors in offspring through increased expression of soluble epoxide hydrolase. Proc. Natl. Acad. Sci. USA 2020, 117, 11753–11759. [Google Scholar] [CrossRef]
- Swanson, N.L.; Leu, A.; Abrahamson, J.; Wallet, B. Genetically engineered crops, glyphosate and the deterioration of health in the United States of America. JOS 2014, 9, 6–37. [Google Scholar]
- Coultrap, S.J.; Vest, R.S.; Ashpole, N.M.; Hudmon, A.; Bayer, K.U. CaMKII in cerebral ischemia. Acta Pharmacol. Sin. 2011, 32, 861–872. [Google Scholar] [CrossRef]
- Zhang, X.; Connelly, J.; Levitan, E.S.; Sun, D.; Wang, J.Q. Calcium/Calmodulin–Dependent Protein Kinase II in Cerebrovascular Diseases. Transl. Stroke Res. 2021, 12, 513–529. [Google Scholar] [CrossRef]
- Bali, Y.A.; Ba-Mhamed, S.; Bennis, M. Behavioral and Immunohistochemical Study of the Effects of Subchronic and Chronic Exposure to Glyphosate in Mice. Front. Behav. Neurosci. 2017, 11, 146. [Google Scholar] [CrossRef]
- Bali, Y.A.; Kaikai, N.-E.; Ba-M’Hamed, S.; Bennis, M. Learning and memory impairments associated to acetylcholinesterase inhibition and oxidative stress following glyphosate based-herbicide exposure in mice. Toxicology 2019, 415, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Ait-Bali, Y.; Ba-M’Hamed, S.; Gambarotta, G.; Sassoè-Pognetto, M.; Giustetto, M.; Bennis, M. Pre- and postnatal exposure to glyphosate-based herbicide causes behavioral and cognitive impairments in adult mice: Evidence of cortical ad hippocampal dysfunction. Arch. Toxicol. 2020, 94, 1703–1723. [Google Scholar] [CrossRef] [PubMed]
- Baier, C.J.; Gallegos, C.E.; Raisman-Vozari, R.; Minetti, A. Behavioral impairments following repeated intranasal glyphosate-based herbicide administration in mice. Neurotoxicology Teratol. 2017, 64, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Cattani, D.; Struyf, N.; Steffensen, V.; Bergquist, J.; Zamoner, A.; Brittebo, E.; Andersson, M. Perinatal exposure to a glyphosate-based herbicide causes dysregulation of dynorphins and an increase of neural precursor cells in the brain of adult male rats. Toxicology 2021, 461, 152922. [Google Scholar] [CrossRef]
- Coullery, R.; Pacchioni, A.M.; Rosso, S.B. Exposure to glyphosate during pregnancy induces neurobehavioral alterations and downregulation of Wnt5a-CaMKII pathway. Reprod. Toxicol. 2020, 96, 390–398. [Google Scholar] [CrossRef]
- Dechartres, J.; Pawluski, J.L.; Gueguen, M.; Jablaoui, A.; Maguin, E.; Rhimi, M.; Charlier, T.D. Glyphosate and glyphosate-based herbicide exposure during the peripartum period affects maternal brain plasticity, maternal behaviour and microbiome. J. Neuroendocr. 2019, 31, e12731. [Google Scholar] [CrossRef]
- Gallegos, C.E.; Bartos, M.; Gumilar, F.; Raisman-Vozari, R.; Minetti, A.; Baier, C.J. Intranasal glyphosate-based herbicide administration alters the redox balance and the cholinergic system in the mouse brain. NeuroToxicology 2020, 77, 205–215. [Google Scholar] [CrossRef]
- Hernández-Plata, I.; Giordano, M.; Díaz-Muñoz, M.; Rodríguez, V.M. The herbicide glyphosate causes behavioral changes and alterations in dopaminergic markers in male Sprague-Dawley rat. NeuroToxicology 2015, 46, 79–91. [Google Scholar] [CrossRef]
- Ji, H.; Xu, L.; Wang, Z.; Fan, X.; Wu, L. Differential microRNA expression in the prefrontal cortex of mouse offspring induced by glyphosate exposure during pregnancy and lactation. Exp. Ther. Med. 2017, 15, 2457–2467. [Google Scholar] [CrossRef]
- Joaquim, A.; Spinosa, H.; Macrini, D.J.; Rodrigues, P.A.; Ricci, E.L.; Artiolli, T.S.; Moreira, N.; Suffredini, I.B.; Bernardi, M.M. Behavioral effects of acute glyphosate exposure in male and female Balb/c mice. Braz. J. Vet. Res. Anim. Sci. 2012, 49, 367–376. [Google Scholar] [CrossRef][Green Version]
- Larsen, K.E.; Lifschitz, A.L.; Lanusse, C.E.; Virkel, G.L. The herbicide glyphosate is a weak inhibitor of acetylcholinesterase in rats. Environ. Toxicol. Pharmacol. 2016, 45, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Luna, S.; Neila, L.P.; Vena, R.; Borgatello, C.; Rosso, S.B. Glyphosate exposure induces synaptic impairment in hippocampal neurons and cognitive deficits in developing rats. Arch. Toxicol. 2021, 95, 2137–2150. [Google Scholar] [CrossRef] [PubMed]
- Gui, Y.-X.; Fan, X.-N.; Wang, H.-M.; Wang, G.; Chen, S.-D. Glyphosate induced cell death through apoptotic and autophagic mechanisms. Neurotoxicol. Teratol. 2012, 34, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Masood, M.I.; Naseem, M.; Warda, S.A.; Tapia-Laliena, M.; Rehman, H.U.; Nasim, M.J.; Schäfer, K.H. Environment permissible concentrations of glyphosate in drinking water can influence the fate of neural stem cells from the subventricular zone of the postnatal mouse. Environ. Pollut. 2020, 270, 116179. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, K.N.; Cappellaro, L.G.; Ueda, C.N.; Rodrigues, L.; Remor, A.P.; Martins, R.D.P.; Latini, A.; Glaser, V. Glyphosate-based herbicide impairs energy metabolism and increases autophagy in C6 astroglioma cell line. J. Toxicol. Environ. Health Part A 2020, 83, 153–167. [Google Scholar] [CrossRef] [PubMed]
- Szepanowski, F.; Szepanowski, L.-P.; Mausberg, A.K.; Albrecht, P.; Kleinschnitz, C.; Kieseier, B.C.; Stettner, M. Differential impact of pure glyphosate and glyphosate-based herbicide in a model of peripheral nervous system myelination. Acta Neuropathol. 2018, 136, 979–982. [Google Scholar] [CrossRef]
- Szepanowski, F.; Kleinschnitz, C.; Stettner, M. Glyphosate-based herbicide: A risk factor for demyelinating conditions of the peripheral nervous system? Neural Regen. Res. 2019, 14, 2079–2080. [Google Scholar] [CrossRef]
- Arredondo, S.B.; Guerrero, F.G.; Herrera-Soto, A.; Jensen-Flores, J.; Bustamante, D.B.; Oñate-Ponce, A.; Henny, P.; Varas-Godoy, M.; Inestrosa, N.C.; Varela-Nallar, L. Wnt5a promotes differentiation and development of adult-born neurons in the hippocampus by noncanonical Wnt signaling. Stem Cells 2019, 38, 422–436. [Google Scholar] [CrossRef]
- Habtemariam, S. The brain-derived neurotrophic factor in neuronal plasticity and neuroregeneration: New pharmacological concepts for old and new drugs. Neural Regen. Res. 2018, 13, 983–984. [Google Scholar] [CrossRef]
- Murray, P.S.; Holmes, P.V. An Overview of Brain-Derived Neurotrophic Factor and Implications for Excitotoxic Vulnerability in the Hippocampus. Int. J. Pept. 2011, 2011, 1–12. [Google Scholar] [CrossRef]
- Rothermundt, M.; Peters, M.; Prehn, J.; Arolt, V. S100B in brain damage and neurodegeneration. Microsc. Res. Tech. 2003, 60, 614–632. [Google Scholar] [CrossRef] [PubMed]
- Pomper, N.; Liu, Y.; Hoye, M.L.; Dougherty, J.D.; Miller, T.M. CNS microRNA profiles: A database for cell type enriched microRNA expression across the mouse central nervous system. Sci. Rep. 2020, 10, 4921. [Google Scholar] [CrossRef] [PubMed]
- Cruz, C.D.; Cruz, F. The ERK 1 and 2 Pathway in the Nervous System: From Basic Aspects to Possible Clinical Applications in Pain and Visceral Dysfunction. Curr. Neuropharmacol. 2007, 5, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Nan, G. The extracellular signal-regulated kinase 1/2 pathway in neurological diseases: A potential therapeutic target (Review). Int. J. Mol. Med. 2017, 39, 1338–1346. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, S.; Unsicker, K. ERK and cell death: ERK1/2 in neuronal death. FEBS J. 2009, 277, 22–29. [Google Scholar] [CrossRef]
- Zhao, Y.; Luo, P.; Guo, Q.; Li, S.; Zhang, L.; Zhao, M.; Xu, H.; Yang, Y.; Poon, W.; Fei, Z. Interactions between SIRT1 and MAPK/ERK regulate neuronal apoptosis induced by traumatic brain injury in vitro and in vivo. Exp. Neurol. 2012, 237, 489–498. [Google Scholar] [CrossRef]
- Jiang, Q.; Gu, Z.; Zhang, G.; Jing, G. Diphosphorylation and involvement of extracellular signal-regulated kinases (ERK1/2) in glutamate-induced apoptotic-like death in cultured rat cortical neurons. Brain Res. 2000, 857, 71–77. [Google Scholar] [CrossRef]
- Satoh, T.; Nakatsuka, D.; Watanabe, Y.; Nagata, I.; Kikuchi, H.; Namura, S. Neuroprotection by MAPK/ERK kinase inhibition with U0126 against oxidative stress in a mouse neuronal cell line and rat primary cultured cortical neurons. Neurosci. Lett. 2000, 288, 163–166. [Google Scholar] [CrossRef]
- Schwarzer, C. 30 years of dynorphins—New insights on their functions in neuropsychiatric diseases. Pharmacol. Ther. 2009, 123, 353–370. [Google Scholar] [CrossRef]
- Albillos, A.; Carbone, E.; Gandia, L.; Garcia, A.G.; Polio, A. Opioid Inhibition of Ca 2+ Channel Subtypes in Bovine Chromaffin Cells: Selectivity of Action and Voltage-dependence. Eur. J. Neurosci. 1996, 8, 1561–1570. [Google Scholar] [CrossRef]
- Rittase, W.B.; Dong, Y.; Barksdale, D.; Galdzicki, Z.; Bausch, S.B. Dynorphin up-regulation in the dentate granule cell mossy fiber pathway following chronic inhibition of GluN2B-containing NMDAR is associated with increased CREB (Ser 133) phosphorylation, but is independent of BDNF/TrkB signaling pathways. Mol. Cell. Neurosci. 2014, 60, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Wagner, J.J.; Terman, G.W.; Chavkin, C. Endogenous dynorphins inhibit excitatory neurotransmission and block LTP induction in the hippocampus. Nat. 1993, 363, 451–454. [Google Scholar] [CrossRef] [PubMed]
- Sgroi, S.; Tonini, R. Opioidergic Modulation of Striatal Circuits, Implications in Parkinson’s Disease and Levodopa Induced Dyskinesia. Front. Neurol. 2018, 9, 524. [Google Scholar] [CrossRef] [PubMed]
- Bridges, R.S. Long-term alterations in neural and endocrine processes induced by motherhood in mammals. Horm. Behav. 2015, 77, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Kohl, J.; Autry, A.E.; Dulac, C. The neurobiology of parenting: A neural circuit perspective. BioEssays 2016, 39, e201600159-11. [Google Scholar] [CrossRef]
- Pawluski, J.L.; Lambert, K.G.; Kinsley, C.H. Neuroplasticity in the maternal hippocampus: Relation to cognition and effects of repeated stress. Horm. Behav. 2016, 77, 86–97. [Google Scholar] [CrossRef]
- Lau, A.; Tymianski, M. Glutamate receptors, neurotoxicity and neurodegeneration. Pflügers Arch.-Eur. J. Physiol. 2010, 460, 525–542. [Google Scholar] [CrossRef]
- Moussawi, K.; Riegel, A.; Nair, S.; Kalivas, P.W. Extracellular Glutamate: Functional Compartments Operate in Different Concentration Ranges. Front. Syst. Neurosci. 2011, 5, 94. [Google Scholar] [CrossRef]
- Simões, A.P.; Silva, C.S.G.; Marques, J.; Pochmann, D.; Porciúncula, L.O.; Ferreira, S.; Oses, J.P.; Beleza, R.O.; Real, J.I.; Köfalvi, A.; et al. Glutamate-induced and NMDA receptor-mediated neurodegeneration entails P2Y1 receptor activation. Cell Death Dis. 2018, 9, 297. [Google Scholar] [CrossRef]
- Maurer, S.V.; Williams, C.L. The Cholinergic System Modulates Memory and Hippocampal Plasticity via Its Interactions with Non-Neuronal Cells. Front. Immunol. 2017, 8, 1489. [Google Scholar] [CrossRef]
- Picciotto, M.R.; Higley, M.J.; Mineur, Y.S. Acetylcholine as a Neuromodulator: Cholinergic Signaling Shapes Nervous System Function and Behavior. Neuron 2012, 76, 116–129. [Google Scholar] [CrossRef] [PubMed]
- Jaffard, R.; Galey, D.; Micheau, J.; Durkin, T. The cholinergic septo-hippocampal pathway, learning and memory. In Brain Plasticity, Learning, and Memory; Will, B.E., Schmitt, P., Dalrymple-Alford, J.C., Eds.; Springer: Boston, MA, USA, 1985; pp. 167–181. [Google Scholar]
- Nicoll, R.A. The septo-hippocampal projection: A model cholinergic pathway. Trends Neurosci. 1985, 8, 533–536. [Google Scholar] [CrossRef]
- Bahena-Trujillo, R.; Flores, G.; Arias-Montaño, J.A. Dopamina: Síntesis, liberación y receptores en el Sistema Nervioso Central. Rev. Biomed. 2000, 11, 39–60. [Google Scholar] [CrossRef]
- Gudi, V.; Gai, L.; Herder, V.; Tejedor, L.S.; Kipp, M.; Amor, S.; Sühs, K.-W.; Hansmann, F.; Beineke, A.; Baumgärtner, W.; et al. Synaptophysin Is a Reliable Marker for Axonal Damage. J. Neuropathol. Exp. Neurol. 2017, 76, 109–125. [Google Scholar] [CrossRef]
- Salim, S. Oxidative Stress and the Central Nervous System. J. Pharmacol. Exp. Ther. 2017, 360, 201–205. [Google Scholar] [CrossRef]
- Wang, X.; Michaelis, E.K. Selective neuronal vulnerability to oxidative stress in the brain. Front. Aging Neurosci. 2010, 2, 12. [Google Scholar] [CrossRef]
- Baba, S.P.; Bhatnagar, A. Role of thiols in oxidative stress. Curr. Opin. Toxicol. 2018, 7, 133–139. [Google Scholar] [CrossRef]
- Harris, C.; Hansen, J.M. Oxidative stress, thiols, and redox profiles. In Developmental Toxicology; Harris, C., Hansen, J., Eds.; Humana Press: Totowa, NJ, USA, 2012; pp. 325–346. [Google Scholar]
- Grant, R.S.; Guest, J.A. Effects of dietary derived antioxidants on the central nervous system. Int. J. Nutr. Pharmacol. Neurol. Dis. 2012, 2, 185. [Google Scholar] [CrossRef]
- Peng, L. Mice Brain Tissue Injury Induced by Diisononyl Phthalate Exposure and the Protective Application of Vitamin, E.J. Biochem. Mol. Toxicol. 2015, 29, 311–320. [Google Scholar] [CrossRef]
- Kwon, D.H.; Cha, H.-J.; Lee, H.; Hong, S.-H.; Park, C.; Park, S.-H.; Kim, G.-Y.; Kim, S.; Kim, H.-S.; Hwang, H.-J.; et al. Protective Effect of Glutathione against Oxidative Stress-induced Cytotoxicity in RAW 264.7 Macrophages through Activating the Nuclear Factor Erythroid 2-Related Factor-2/Heme Oxygenase-1 Pathway. Antioxidants 2019, 8, 82. [Google Scholar] [CrossRef]
- Tan, B.L.; Norhaizan, M.E.; Liew, W.-P.-P.; Rahman, H.S. Antioxidant and Oxidative Stress: A Mutual Interplay in Age-Related Diseases. Front. Pharmacol. 2018, 9, 1162. [Google Scholar] [CrossRef]
- Kumar, S.; Trivedi, P.K. Glutathione S-Transferases: Role in Combating Abiotic Stresses Including Arsenic Detoxification in Plants. Front. Plant Sci. 2018, 9, 751. [Google Scholar] [CrossRef] [PubMed]
- Astiz, M.; de Alaniz, M.J.; Marra, C.A. Effect of pesticides on cell survival in liver and brain rat tissues. Ecotoxicol. Environ. Saf. 2009, 72, 2025–2032. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Zhou, J. Neuroinflammation in the central nervous system: Symphony of glial cells. Glia 2018, 67, 1017–1035. [Google Scholar] [CrossRef]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget 2018, 9, 7204–7218. [Google Scholar] [CrossRef] [PubMed]
- Glass, C.K.; Saijo, K.; Winner, B.; Marchetto, M.C.; Gage, F.H. Mechanisms Underlying Inflammation in Neurodegeneration. Cell 2010, 140, 918–934. [Google Scholar] [CrossRef]
- Yuste, J.E.; Tarragon, E.; Campuzano, C.M.; Cros, E.T. Implications of glial nitric oxide in neurodegenerative diseases. Front. Cell. Neurosci. 2015, 9, 322. [Google Scholar] [CrossRef] [PubMed]
- Saraste, A.; Pulkki, K. Morphologic and biochemical hallmarks of apoptosis. Cardiovasc. Res. 2000, 45, 528–537. [Google Scholar] [CrossRef]
- Badadani, M. Autophagy Mechanism, Regulation, Functions, and Disorders. ISRN Cell Biol. 2012, 2012, 927064. [Google Scholar] [CrossRef]
- Edlich, F. BCL-2 proteins and apoptosis: Recent insights and unknowns. Biochem. Biophys. Res. Commun. 2018, 500, 26–34. [Google Scholar] [CrossRef]
- Mizushima, N. Autophagy: Process and function. Genes Dev. 2007, 21, 2861–2873. [Google Scholar] [CrossRef] [PubMed]
- Stavoe, A.; Holzbaur, E.L.F. Autophagy in Neurons. Annu. Rev. Cell Dev. Biol. 2019, 35, 477–500. [Google Scholar] [CrossRef] [PubMed]
- Babin, P.J.; Goizet, C.; Raldúa, D. Zebrafish models of human motor neuron diseases: Advantages and limitations. Prog. Neurobiol. 2014, 118, 36–58. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, J.A.; Könemann, S.; Krümpelmann, L.; Županič, A.; Berg, C.V. Approaches to Test the Neurotoxicity of Environmental Contaminants in the Zebrafish Model: From Behavior to Molecular Mechanisms. Environ. Toxicol. Chem. 2020, 40, 989–1006. [Google Scholar] [CrossRef]
- Flinn, L.; Bretaud, S.; Lo, C.; Ingham, P.W.; Bandmann, O. Zebrafish as a new animal model for movement disorders. J. Neurochem. 2008, 106, 1991–1997. [Google Scholar] [CrossRef]
- Faria, M.; Bedrossiantz, J.; Ramírez, J.R.R.; Mayol, M.; García, G.H.; Bellot, M.; Prats, E.; Garcia-Reyero, N.; Gómez-Canela, C.; Gómez-Oliván, L.M.; et al. Glyphosate targets fish monoaminergic systems leading to oxidative stress and anxiety. Environ. Int. 2020, 146, 106253. [Google Scholar] [CrossRef] [PubMed]
- Horzmann, K.A.; Freeman, J.L. Zebrafish Get Connected: Investigating Neurotransmission Targets and Alterations in Chemical Toxicity. Toxics 2016, 4, 19. [Google Scholar] [CrossRef]
- Bernal-Rey, D.L.; Cantera, C.G.; Afonso, M.D.S.; Menéndez-Helman, R.J. Seasonal variations in the dose-response relationship of acetylcholinesterase activity in freshwater fish exposed to chlorpyrifos and glyphosate. Ecotoxicol. Environ. Saf. 2019, 187, 109673. [Google Scholar] [CrossRef]
- Braz-Mota, S.; Sadauskas-Henrique, H.; Duarte, R.; Val, A.; Almeida-Val, V.M. Roundup® exposure promotes gills and liver impairments, DNA damage and inhibition of brain cholinergic activity in the Amazon teleost fish Colossoma macropomum. Chemosphere 2015, 135, 53–60. [Google Scholar] [CrossRef]
- Bridi, D.; Altenhofen, S.; Gonzalez, J.B.; Reolon, G.K.; Bonan, C.D. Glyphosate and Roundup® alter morphology and behavior in zebrafish. Toxicology 2017, 392, 32–39. [Google Scholar] [CrossRef]
- Díaz-Martín, R.D.; Valencia-Hernández, J.D.; Betancourt-Lozano, M.; Yáñez-Rivera, B. Changes in microtubule stability in zebrafish (Danio rerio) embryos after glyphosate exposure. Heliyon 2021, 7, e06027. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, J.M.D.S.; Lima, V.D.S.; De Moura, F.R.; Marisco, P.D.C.; Sinhorin, A.P.; Sinhorin, V.D.G. Acute toxicity and effects of Roundup Original® on pintado da Amazônia. Environ. Sci. Pollut. Res. 2018, 25, 25383–25389. [Google Scholar] [CrossRef] [PubMed]
- Forner-Piquer, I.; Faucherre, A.; Byram, J.; Blaquiere, M.; de Bock, F.; Gamet-Payrastre, L.; Ellero-Simatos, S.; Audinat, E.; Jopling, C.; Marchi, N. Differential impact of dose-range glyphosate on locomotor behavior, neuronal activity, glio-cerebrovascular structures, and transcript regulations in zebrafish larvae. Chemosphere 2020, 267, 128986. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Kurobe, T.; Ramírez-Duarte, W.F.; Bolotaolo, M.B.; Lam, C.H.; Pandey, P.K.; Hung, T.-C.; Stillway, M.E.; Zweig, L.; Caudill, J.; et al. Sub-lethal effects of herbicides penoxsulam, imazamox, fluridone and glyphosate on Delta Smelt (Hypomesus transpacificus). Aquat. Toxicol. 2018, 197, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.K.; Shah, N.; Gul, A.; Sahar, N.U.; Ismail, A.; Muhammad, M.; Aziz, F.; Farooq, M.; Adnan, M.; Rizwan, M. Comparative Study of Toxicological Impinge of Glyphosate and Atrazine (Herbicide) on Stress Biomarkers; Blood Biochemical and Hematological Parameters of the Freshwater Common Carp (Cyprinus carpio). Pol. J. Environ. Stud. 2016, 25, 1995–2001. [Google Scholar] [CrossRef]
- Lanzarin, G.A.B.; Félix, L.M.; Santos, D.; Venâncio, C.A.S.; Monteiro, S.M. Dose-dependent effects of a glyphosate commercial formulation–Roundup® UltraMax-on the early zebrafish embryogenesis. Chemosphere 2019, 223, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Lanzarin, G.A.; Venâncio, C.; Monteiro, S.M.; Félix, L.M. Behavioural toxicity of environmental relevant concentrations of a glyphosate commercial formulation-RoundUp® UltraMax-During zebrafish embryogenesis. Chemosphere 2020, 253, 126636. [Google Scholar] [CrossRef]
- Li, M.-H.; Xu, H.-D.; Liu, Y.; Chen, T.; Jiang, L.; Fu, Y.-H.; Wang, J.-S. Multi-tissue metabolic responses of goldfish (Carassius auratus) exposed to glyphosate-based herbicide. Toxicol. Res. 2016, 5, 1039–1052. [Google Scholar] [CrossRef]
- Li, M.; Ruan, L.-Y.; Zhou, J.-W.; Fu, Y.-H.; Jiang, L.; Zhao, H.; Wang, J.-S. Metabolic profiling of goldfish (Carassius auratis) after long-term glyphosate-based herbicide exposure. Aquat. Toxicol. 2017, 188, 159–169. [Google Scholar] [CrossRef]
- Lopes, F.M.; Caldas, S.S.; Primel, E.G.; da Rosa, C.E. Glyphosate Adversely Affects Danio rerio Males: Acetylcholinesterase Modulation and Oxidative Stress. Zebrafish 2017, 14, 97–105. [Google Scholar] [CrossRef]
- Menéndez-Helman, R.J.; Ferreyroa, G.V.; Afonso, M.D.S.; Salibián, A. Glyphosate as an Acetylcholinesterase Inhibitor in Cnesterodon decemmaculatus. Bull. Environ. Contam. Toxicol. 2011, 88, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Menéndez-Helman, R.J.; Miranda, L.A.; Afonso, M.D.S.; Salibián, A. Subcellular energy balance of Odontesthes bonariensis exposed to a glyphosate-based herbicide. Ecotoxicol. Environ. Saf. 2015, 114, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.G.; Jaramillo, M.L.; Remor, A.P.; Latini, A.; Davico, C.E.; da Silva, M.L.; Müller, Y.M.; Ammar, D.; Nazari, E.M. Low-concentration exposure to glyphosate-based herbicide modulates the complexes of the mitochondrial respiratory chain and induces mitochondrial hyperpolarization in the Danio rerio brain. Chemosphere 2018, 209, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Roy, N.M.; Carneiro, B.; Ochs, J. Glyphosate induces neurotoxicity in zebrafish. Environ. Toxicol. Pharmacol. 2016, 42, 45–54. [Google Scholar] [CrossRef]
- Sánchez, J.A.A.; Barros, D.M.; Bistoni, M.D.L.A.; Ballesteros, M.L.; Roggio, M.A.; Martins, C.D.G.M. Glyphosate-based herbicides affect behavioural patterns of the livebearer Jenynsia multidentata. Environ. Sci. Pollut. Res. 2021, 28, 29958–29970. [Google Scholar] [CrossRef]
- Sobjak, T.M.; Romão, S.; Nascimento, C.Z.D.; dos Santos, A.F.P.; Vogel, L.; Guimarães, A.T.B. Assessment of the oxidative and neurotoxic effects of glyphosate pesticide on the larvae of Rhamdia quelen fish. Chemosphere 2017, 182, 267–275. [Google Scholar] [CrossRef]
- Zhang, S.; Xu, J.; Kuang, X.; Li, S.; Li, X.; Chen, D.; Zhao, X.; Feng, X. Biological impacts of glyphosate on morphology, embryo biomechanics and larval behavior in zebrafish ( Danio rerio ). Chemosphere 2017, 181, 270–280. [Google Scholar] [CrossRef]
- Carhart-Harris, R.L.; Nutt, D.J. Serotonin and brain function: A tale of two receptors. J. Psychopharmacol. 2017, 31, 1091–1120. [Google Scholar] [CrossRef]
- Strużyńska, L.; Sulkowski, G. Relationships between glutamine, glutamate, and GABA in nerve endings under Pb-toxicity conditions. J. Inorg. Biochem. 2004, 98, 951–958. [Google Scholar] [CrossRef]
- Zorova, L.D.; Popkov, V.A.; Plotnikov, E.Y.; Silachev, D.N.; Pevzner, I.B.; Jankauskas, S.S.; Babenko, V.A.; Zorov, S.D.; Balakireva, A.V.; Juhaszova, M.; et al. Mitochondrial membrane potential. Anal. Biochem. 2018, 552, 50–59. [Google Scholar] [CrossRef]
- Tayebati, S.; Marucci, G.; Santinelli, C.; Buccioni, M.; Amenta, F. Choline-Containing Phospholipids: Structure-Activity Relationships Versus Therapeutic Applications. Curr. Med. Chem. 2015, 22, 4328–4340. [Google Scholar] [CrossRef] [PubMed]
- Zahr, N.M.; Mayer, D.; Rohlfing, T.; Sullivan, E.V.; Pfefferbaum, A. Imaging Neuroinflammation? A Perspective from MR Spectroscopy. Brain Pathol. 2014, 24, 654–664. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Ni, Y.; Jin, Y.; Fu, Z. Pesticides-induced energy metabolic disorders. Sci. Total Environ. 2020, 729, 139033. [Google Scholar] [CrossRef] [PubMed]
- Dienel, G.A. Brain Glucose Metabolism: Integration of Energetics with Function. Physiol. Rev. 2019, 99, 949–1045. [Google Scholar] [CrossRef]
- Holtzman, D. Brain Creatine Kinases and Phosphocreatine: An Update. Dev. Neurosci. 1996, 18, 522–523. [Google Scholar] [CrossRef]
- Cunnane, S.C.; Courchesne-Loyer, A.; Vandenberghe, C.; St-Pierre, V.; Fortier, M.; Hennebelle, M.; Croteau, E.; Bocti, C.; Fulop, T.; Castellano, C.-A. Can Ketones Help Rescue Brain Fuel Supply in Later Life? Implications for Cognitive Health during Aging and the Treatment of Alzheimer’s Disease. Front. Mol. Neurosci. 2016, 9, 53. [Google Scholar] [CrossRef]
- Tovar-Franco, J.A.; Ortiz, M.J.S.; Morales, L.; Espíndola, C. Regulación de la utilización del aspartato por los astrocitos durante la prelactancia. Univ. Sci. 2009, 14, 151–163. [Google Scholar] [CrossRef]
- Moffett, J.R.; Arun, P.; Ariyannur, P.S.; Namboodiri, A.M. N-Acetylaspartate reductions in brain injury: Impact on post-injury neuroenergetics, lipid synthesis, and protein acetylation. Front. Neuroenergetics 2013, 5, 11. [Google Scholar] [CrossRef]
- Domise, M.; Sauvé, F.; Didier, S.; Caillerez, R.; Bégard, S.; Carrier, S.; Colin, M.; Marinangeli, C.; Buée, L.; Vingtdeux, V. Neuronal AMP-activated protein kinase hyper-activation induces synaptic loss by an autophagy-mediated process. Cell Death Dis. 2019, 10, 221. [Google Scholar] [CrossRef]
- McVey, K.A.; Snapp, I.B.; Johnson, M.B.; Negga, R.; Pressley, A.S.; Fitsanakis, V.A. Exposure of C. elegans eggs to a glyphosate-containing herbicide leads to abnormal neuronal morphology. Neurotoxicol. Teratol. 2016, 55, 23–31. [Google Scholar] [CrossRef]
- Burchfield, S.L.; Bailey, D.C.; Todt, C.E.; Denney, R.D.; Negga, R.; Fitsanakis, V.A. Acute exposure to a glyphosate-containing herbicide formulation inhibits Complex II and increases hydrogen peroxide in the model organism Caenorhabditis elegans. Environ. Toxicol. Pharmacol. 2018, 66, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Fortes, C.; Mastroeni, S.; Segatto, M.M.; Hohmann, C.; Miligi, L.; Bakos, L.; Bonamigo, R. Occupational Exposure to Pesticides With Occupational Sun Exposure Increases the Risk for Cutaneous Melanoma. J. Occup. Environ. Med. 2016, 58, 370–375. [Google Scholar] [CrossRef] [PubMed]
- Jayasumana, C.; Gunatilake, S.; Senanayake, P. Glyphosate, Hard Water and Nephrotoxic Metals: Are They the Culprits Behind the Epidemic of Chronic Kidney Disease of Unknown Etiology in Sri Lanka? Int. J. Environ. Res. Public Health 2014, 11, 2125–2147. [Google Scholar] [CrossRef] [PubMed]
- Kamel, F.; Hoppin, J.A. Association of Pesticide Exposure with Neurologic Dysfunction and Disease. Environ. Health Perspect. 2004, 112, 950–958. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.J.; Alavanja, M.C.; Hoppin, J.; Rusiecki, J.A.; Kamel, F.; Blair, A.; Sandler, D.P. Mortality among Pesticide Applicators Exposed to Chlorpyrifos in the Agricultural Health Study. Environ. Health Perspect. 2007, 115, 528–534. [Google Scholar] [CrossRef]
- Le Couteur, D.; McLean, A.; Taylor, M.; Woodham, B.; Board, P. Pesticides and Parkinson’s disease. Biomed. Pharmacother. 1999, 53, 122–130. [Google Scholar] [CrossRef]
- Gasnier, C.; Dumont, C.; Benachour, N.; Clair, E.; Chagnon, M.-C.; Séralini, G.-E. Glyphosate-based herbicides are toxic and endocrine disruptors in human cell lines. Toxicology 2009, 262, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Koller, V.J.; Fürhacker, M.; Nersesyan, A.; Mišík, M.; Eisenbauer, M.; Knasmueller, S. Cytotoxic and DNA-damaging properties of glyphosate and Roundup in human-derived buccal epithelial cells. Arch. Toxicol. 2012, 86, 805–813. [Google Scholar] [CrossRef]
- Prasad, S.; Srivastava, S.; Singh, M.; Shukla, Y. Clastogenic Effects of Glyphosate in Bone Marrow Cells of Swiss Albino Mice. J. Toxicol. 2009, 2009, 308985. [Google Scholar] [CrossRef]
- Lajmanovich, R.C.; Sandoval, M.T.; Peltzer, P.M. Induction of Mortality and Malformation in Scinax nasicus Tadpoles Exposed to Glyphosate Formulations. Bull. Environ. Contam. Toxicol. 2003, 70, 612–618. [Google Scholar] [CrossRef]
- Barbosa, E.R.; da Costa, M.D.L.; Bacheschi, L.A.; Scaff, M.; Leite, C.C. Parkinsonism after glycine-derivate exposure. Mov. Disord. 2001, 16, 565–568. [Google Scholar] [CrossRef] [PubMed]
- Menkes, D.; Temple, W.; Edwards, I. Intentional Self-Poisoning with Glyphosate-Containing Herbicides. Hum. Exp. Toxicol. 1991, 10, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Mose, T.; Kjaerstad, M.B.; Mathiesen, L.; Nielsen, J.B.; Edelfors, S.; Knudsen, L.E. Placental Passage of Benzoic acid, Caffeine, and Glyphosate in an Ex Vivo Human Perfusion System. J. Toxicol. Environ. Health Part A 2008, 71, 984–991. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, M.S.; Rytting, E.; Mose, T.; Knudsen, L.E. Modeling placental transport: Correlation of in vitro BeWo cell permeability and ex vivo human placental perfusion. Toxicol. Vitr. 2009, 23, 1380–1386. [Google Scholar] [CrossRef]
- Wooltorton, J.; Pidoplichko, V.I.; Broide, R.S.; Dani, J.A. Differential Desensitization and Distribution of Nicotinic Acetylcholine Receptor Subtypes in Midbrain Dopamine Areas. J. Neurosci. 2003, 23, 3176–3185. [Google Scholar] [CrossRef]
- Balali-Mood, M.; Saber, H. Recent Advances in the Treatment of Organophosphorous Poisonings. Iran. J. Med Sci. 2012, 37, 74–91. [Google Scholar]
- Scheffel, C.; Niessen, K.V.; Rappenglück, S.; Wanner, K.; Thiermann, H.; Worek, F.; Seeger, T. Counteracting desensitization of human α7-nicotinic acetylcholine receptors with bispyridinium compounds as an approach against organophosphorus poisoning. Toxicol. Lett. 2018, 293, 149–156. [Google Scholar] [CrossRef]
- Wojda, U.; Salinska, E.; Kuznicki, J. Calcium ions in neuronal degeneration. IUBMB Life 2008, 60, 575–590. [Google Scholar] [CrossRef]
- De, A. Wnt/Ca2+ signaling pathway: A brief overview. Acta Biochim. Biophys. Sin. 2011, 43, 745–756. [Google Scholar] [CrossRef]
- McMurry, J.L.; Chrestensen, C.A.; Scott, I.M.; Lee, E.W.; Rahn, A.M.; Johansen, A.M.; Forsberg, B.J.; Harris, K.D.; Salerno, J.C. Rate, affinity and calcium dependence of nitric oxide synthase isoform binding to the primary physiological regulator calmodulin. FEBS J. 2011, 278, 4943–4954. [Google Scholar] [CrossRef]
- Donato, R.; Sorci, G.; Riuzzi, F.; Arcuri, C.; Bianchi, R.; Brozzi, F.; Tubaro, C.; Giambanco, I. S100B’s double life: Intracellular regulator and extracellular signal. Biochim. Biophys. Acta 2009, 1793, 1008–1022. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, R.; Giambanco, I.; Donato, R. S100B/RAGE-dependent activation of microglia via NF-κB and AP-1: Co-regulation of COX-2 expression by S100B, IL-1β and TNF-α. Neurobiol. Aging 2010, 31, 665–677. [Google Scholar] [CrossRef]
- Petrova, T.V.; Hu, J.; Van Eldik, L.J. Modulation of glial activation by astrocyte-derived protein S100B: Differential responses of astrocyte and microglial cultures. Brain Res. 2000, 853, 74–80. [Google Scholar] [CrossRef]
- Sun, F.; Xu, X.; Wang, X.; Zhang, B. Regulation of autophagy by Ca2+. Tumor Biol. 2016, 37, 15467–15476. [Google Scholar] [CrossRef] [PubMed]
- Ríos, J.A.; Godoy, J.A.; Inestrosa, N.C. Wnt3a ligand facilitates autophagy in hippocampal neurons by modulating a novel GSK-3β-AMPK axis. Cell Commun. Signal. 2018, 16, 15. [Google Scholar] [CrossRef] [PubMed]
- Lorzadeh, S.; Kohan, L.; Ghavami, S.; Azarpira, N. Autophagy and the Wnt signaling pathway: A focus on Wnt/β-catenin signaling. Biochim. Biophys. Acta 2020, 1868, 118926. [Google Scholar] [CrossRef]
- Hollville, E.; Romero, S.E.; Deshmukh, M. Apoptotic cell death regulation in neurons. FEBS J. 2019, 286, 3276–3298. [Google Scholar] [CrossRef]
- Fricker, M.; Tolkovsky, A.M.; Borutaite, V.; Coleman, M.; Brown, G.C. Neuronal Cell Death. Physiol. Rev. Am. Physiol. Soc. 2018, 98, 813–880. [Google Scholar] [CrossRef]
- World Health Organization. Pesticide Residues in Food-2004: Evaluations 2004: Part II-Toxicological (Vol. 178). World Health Organization. 2006. Available online: http://apps.who.int/iris/bitstream/handle/10665/43624/9241665203_eng.pdf;jsessionid=98409D614439EBC6C58392A3809839C4?sequence=1 (accessed on 30 July 2021).
- Solomon, K.R. Glyphosate in the general population and in applicators: A critical review of studies on exposures. Crit. Rev. Toxicol. 2016, 46, 21–27. [Google Scholar] [CrossRef]
- Paumgartten, F.J.R. To be or not to be a carcinogen; delving into the glyphosate classification controversy. Braz. J. Pharm. Sci. 2019, 55, 2175-97902019000118217. [Google Scholar] [CrossRef]
- Kawagashira, Y.; Koike, H.; Kawabata, K.; Takahashi, M.; Ohyama, K.; Hashimoto, R.; Iijima, M.; Katsuno, M.; Sobue, G. Vasculitic Neuropathy Following Exposure to a Glyphosate-based Herbicide. Intern. Med. 2017, 56, 1431–1434. [Google Scholar] [CrossRef]
- Ramwell, C.T.; Heather, A.I.J.; Shepherd, A.J. Herbicide loss following application to a roadside. Pest Manag. Sci. 2002, 58, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Langiano, V.D.C.; Martinez, C. Toxicity and effects of a glyphosate-based herbicide on the Neotropical fish Prochilodus lineatus. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2008, 147, 222–231. [Google Scholar] [CrossRef] [PubMed]
- Contardo-Jara, V.; Klingelmann, E.; Wiegand, C. Bioaccumulation of glyphosate and its formulation Roundup Ultra in Lumbriculus variegatus and its effects on biotransformation and antioxidant enzymes. Environ. Pollut. 2008, 157, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Dey, S.; Samanta, P.; Pal, S.; Mukherjee, A.K.; Kole, D.; Ghosh, A.R. Integrative assessment of biomarker responses in teleostean fishes exposed to glyphosate-based herbicide (Excel Mera 71). Emerg. Contam. 2016, 2, 191–203. [Google Scholar] [CrossRef]
- El-Sheshtawy, S.M.; Nada, M.M.; Elhafeez, M.S.A.; Samak, D.H. Protective Effect of Supplementation with Powdered Mulberry Leaves on Glyphosate-Induced Toxicity in Catfish (Clarias gariepinus). Adv. Anim. Vet.-Sci. 2021, 9, 1718–1731. [Google Scholar] [CrossRef]
- Rossi, A.S.; Fantón, N.; Michlig, M.P.; Repetti, M.R.; Cazenave, J. Fish inhabiting rice fields: Bioaccumulation, oxidative stress and neurotoxic effects after pesticides application. Ecol. Indic. 2020, 113, 106186. [Google Scholar] [CrossRef]
- Fiorino, E.; Sehonova, P.; Plhalova, L.; Blahova, J.; Svobodova, Z.; Faggio, C. Effects of glyphosate on early life stages: Comparison between Cyprinus carpio and Danio rerio. Environ. Sci. Pollut. Res. 2018, 25, 8542–8549. [Google Scholar] [CrossRef]
- Benachour, N.; Séralini, G.-E. Glyphosate Formulations Induce Apoptosis and Necrosis in Human Umbilical, Embryonic, and Placental Cells. Chem. Res. Toxicol. 2009, 22, 97–105. [Google Scholar] [CrossRef]
- Mesnage, R.; Ibragim, M.; Mandrioli, D.; Falcioni, L.; Tibaldi, E.; Belpoggi, F.; Brandsma, I.; Bourne, E.; Savage, E.; Mein, C.A.; et al. Comparative Toxicogenomics of Glyphosate and Roundup Herbicides by Mammalian Stem Cell-Based Genotoxicity Assays and Molecular Profiling in Sprague-Dawley Rats. Toxicol. Sci. 2021, 186, 83–101. [Google Scholar] [CrossRef]
- Tsui, M.T.; Chu, L.M. Aquatic toxicity of glyphosate-based formulations: Comparison between different organisms and the effects of environmental factors. Chemosphere 2003, 52, 1189–1197. [Google Scholar] [CrossRef]
- El-Shenawy, N.S. Oxidative stress responses of rats exposed to Roundup and its active ingredient glyphosate. Environ. Toxicol. Pharmacol. 2009, 28, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Peixoto, F. Comparative effects of the Roundup and glyphosate on mitochondrial oxidative phosphorylation. Chemosphere 2005, 61, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Richard, S.; Moslemi, S.; Sipahutar, H.; Benachour, N.; Seralini, G.-E. Differential Effects of Glyphosate and Roundup on Human Placental Cells and Aromatase. Environ. Health Perspect. 2005, 113, 716–720. [Google Scholar] [CrossRef] [PubMed]
| Type of Study | Toxic Agent | Exposure Mode/Objetives | Results | Reference |
|---|---|---|---|---|
| Transversal study | GBH | Occupational exposure |
| [45] |
| Prospective cohort study | GBH | Not specified |
| [46] |
| Population-based case-control study | GBH | Occupational exposure |
| [47] |
| Cohort study | GBH | Occupational exposure |
| [48] |
| Cohort study | GBH | Occupational exposure |
| [49] |
| In vitro SH-SY5Y cell line | GLY alone or mixed with other formulants: 5.33 to 3.200 μg/mL for 24 h | Investigate whether GBH toxicity is related to formulants |
| [50] |
| In vitro IMR90-c4 iPSCs line | GLY, AMPA: 0.1 to 1000 μM for 24 or 48 h | Investigate the effect of GLY on the BBB in vitro and compare it with that of AMPA and glycine |
| [51] |
| In vitro SH-SY5Y cell line | GLY, AMPA: 0.1 to 20 mM for 48 h | Investigate the effects of GLY and AMPA on oxidative stress, neurodevelopment, and cell death. |
| [52] |
| Species | Dose and Exposition | Time Exposition | Objectives | Results | Reference |
|---|---|---|---|---|---|
| Swiss mice | Roundup®: 250 or 500 mg/kg/day orally | Subchronic exposition: 6 weeks Chronic exposition: 12 weeks | Assess the effects of acute or repeated GBH exposure on the developing brain of young and adult mice | Chronic/subchronic exposure:
| [62] |
| Swiss mice | Roundup®: 250 or 500 mg/kg/day orally | Subchronic exposition: 6 weeks Chronic exposition: 12 weeks | Evaluate the effects of GBH on learning and memory functions, AChE activity, and oxidation/antioxidation homeostasis | Chronic/subchronic exposure:
| [63] |
| Swiss mice | Roundup®: 250 or 500 mg/kg/day orally | From GD0 to PND21 | Evaluate the behavioral (PND5-PND25) and biochemical (PND60) effects of gestational and lactational exposure to GBH on offspring |
| [64] |
| CF-1 mice | Glifloglex®: 50 mg/kg/day intranasally | Three days a week for four weeks | Assess the neurobehavioral effects of repeated intranasal administration of a GBH |
| [65] |
| Wistar rats | Roundup®: 70 mg/kg/day orally | Chronic exposition: from GD5 to PND15. Acute exposition: 30 min in vitro | Determine the neurotoxic effects of GBH on the hippocampal function of immature rats after chronic exposure (pregnancy and lactation) and after acute in vitro exposition. | Acute in vitro exposition:
| [37] |
| Wistar rats | Roundup®: 1% in drinking water (0.38% GLY) | Subchronic exposition: from GD5 to PND21. Chronic exposition: from GD5 to PND60. | Investigate the effects of subchronic exposure to GBH on neurochemical and behavioral parameters in immature and adult offspring |
| [3] |
| Wistar rats | Roundup®: 70 mg/kg/day orally | Subchronic exposition: from GD5 to PND15. | Investigate possible biochemical and cell-persistent effects in the brain of adult rats following perinatal exposure to GBH |
| [66] |
| Wistar rats | GLY: 24 or 35 mg/kg intraperitoneally | Dams received injections every 48 h from GD8 to GD20, totaling seven injections over two weeks | Evaluate the neurobehavioral effects of GLY in neonate rats after gestational exposure |
| [67] |
| Sprague-Dawley rats | GLY, Roundup®: 5 mg/kg/day orally | From GD10 to PND22 | Compare the potential effects of a low dose of GLY and GBH on maternal behavior and maternal neuroplasticity, focusing on the hippocampus and cingulate gyrus |
| [68] |
| CF-1 mice | Glifloglex®: 50 mg/kg/day intranasally | Four weeks (three injections per week) | Elucidate the mechanisms by which the intranasal administration of a GBH exerts its neuropathological effects |
| [69] |
| Sprague-Dawley rats | GLY: 50, 100, or 150 mg/kg intraperitoneally | Two weeks (three injections per week) | Assess the integrity of the nigrostriatal and mesolimbic dopaminergic systems and their relationship with spontaneous locomotor activity after repeated or acute exposure to GLY |
| [70] |
| ICR mice | Roundup®: 50 mg/kg/day orally | From GD14 to PND7 | Assess the miRNA expression patterns in the PFC of mouse offspring after exposure to GBH during pregnancy and lactation |
| [71] |
| Balb/c mice | Roundup®: 25, 50 or 100 mg/kg orally | Acute exposure | Investigate the behavioral effects induced by acute exposure to a GBH in increasing doses |
| [72] |
| Wistar rats | GBH: 2.5, 5, 10, 20 or 40 mM | Single dose | Assess the inhibitory potency of a GBH on AChE activity in rat tissues |
| [73] |
| Wistar rats | GLY: 35 or 70 mg/kg subcutaneous injection | From PND7 to PND27 | Evaluate the effects of glyphosate on hippocampal synapses and cognitive functioning |
| [74] |
| Wistar rats | GLY: 35, 75, 150 or 800 mg/kg/day orally | Six days | Determine the effects of GLY on the levels of DA, NE, and 5-HT and their metabolites, as well as the turnover in striatum, hippocampus, PFC, hypothalamus, and midbrain. |
| [13] |
| Cellular Line | Dose and Time of Exposure | Objectives | Results | Reference |
|---|---|---|---|---|
| PC12 cells | GLY: 0, 5, 10, 20, or 40 mM for 12, 24, 48, or 72 h | Investigate the neurotoxicity of GLY in differentiated rat PC12 cells and explore the role of apoptosis and autophagy pathways in toxicity |
| [75] |
| Hippocampal pyramidal cells | GLY: 0.5 or 1 mg/mL for five or ten days | Examine the effects of glyphosate on synapse formation and maturation in the hippocampus |
| [74] |
| NSC | GLY: 0.1, 700, 7000, or 36,000 μg/L for 24 h | Understand the effects of two maximum permissible concentrations of GLY on the basic processes of neurogenesis in NSCs of the postnatal mouse subventricular zone. |
| [76] |
| Astroglioma (C6) | GLY: concentrations from 0 to 160 mM for 24 h | Determine the activity of enzymes related to energy metabolism, as well as parameters of oxidative stress, mitochondrial mass, nuclear area, and autophagy in astrocytes treated with GBH |
| [77] |
| Embryonic DRG and pure Schwann cells | GLY, Roundup®: 0.0005% and 0.005% for ten days (DRG) or 72 h (Schwann cells) | Investigate the effects of pure GLY and GBH in murine embryonic DRG cultures |
| [78] |
| Embryonic DRG and pure Schwann cells | GLY, Roundup®: doses not specified for ten days (DRG) or 72 h (Schwann cells) | Study and compare the effects of pure GLY and GBH in murine embryonic DRG explant cultures |
| [79] |
| Species | Dose and Time Exposure | Objectives | Results | Reference |
|---|---|---|---|---|
| Cnesterodon decemmaculatus (Ten-spotted livebearer) | GLY: 1 or 10 mg/L for 96 h | Assess the effect of seasonal variability on AChE activity in fish exposed to chlorpyrifos and GLY |
| [132] |
| Colossoma macropomum (Blackfin pacu) | Roundup®: 10 or 15 mg/L for 96 h | Investigate the effects of GBH on gill morphology and function, hematological parameters, biotransformation enzymes, the antioxidant system in the gills and liver, as well as on both neurological and erythrocytic DNA damage |
| [133] |
| Danio rerio (Zebrafish) | Roundup®, GLY: 0.01, 0.065, or 0.5 mg/L for 96 h | Evaluate the effects of GLY and GBH on morphological and behavioral parameters in larvae and adult zebrafish | GLY and GBH caused:
| [134] |
| Danio rerio (Zebrafish) | GLY: 5, 10, or 50 μg/mL for 96 h | Identify a possible mechanism of toxicity for GLY related to changes in microtubule stability, which could alter the distribution and dynamics of cytoskeletal components |
| [135] |
| Pintado da Amazônia | Roundup®: 0.37, 0.75, 2.25, 4.5, 7.5, 11.25, 15, 22.5, or 30 mg/L for 24, 48, 72, or 96 h | Evaluate the lethal concentration of the GLY and the oxidative stress parameters in tests with sublethal concentrations |
| [136] |
| Danio rerio (Zebrafish) | GLY: 0.3 or 3 μg/L for 2 weeks | Analyze the neurotoxicity of GLY in adult zebrafish after exposure through water to environmentally relevant concentrations |
| [130] |
| Danio rerio (Zebrafish) | GLY: 0.05 to 10.000 μg/L for a period of 1.5 to 120 h after fertilization | Explore the effects of the use of different concentrations of GLY on anatomy and behavior of fish | High concentrations of GLY (≥1000 μg/L) caused:
| [137] |
| Hypomesus transpacificus (Delta smelt) | Roundup®: 0.064, 0.64, 6.4, 64, or 640 mg/L for 6 h | Compare the sublethal toxicity of four herbicides (penoxsulam, imazamox, fluridone, and GBH). |
| [138] |
| Cyprinus carpio (European carp) | GLY: 0.02, 0.05, 0.07, or 0.1 mg/L for 24, 48, 72, or 96 h | Analyze the effect of GLY and atrazine on the hematological and biochemical parameters of blood and on behavioral aspects |
| [139] |
| Danio rerio (Zebrafish) | Roundup®: 2, 5, or 8.5 μg/mL for 72 h | Investigate the lethal and sublethal developmental effects, neurotoxic potential, and oxidative stress responses after GBH exposure | High concentrations of GBH caused:
| [140] |
| Danio rerio (Zebrafish) | Roundup®: 1, 2, or 5 μg/mL for 72 h | Assess GBH effects at environmentally relevant concentrations through a set of behavioral patterns |
| [141] |
| Carassius auratus (Goldenfish) | Nongteshi®: 0.22, 0.44, or 0.88 mmol/L for 96 h | Investigate the toxic effects of GBH exposure using a metabolomic approach supplemented with histological inspection and hematological evaluation |
| [142] |
| Carassius auratus | Nongteshi®: 0.2 mmol/L for 90 days | Assess GBH toxicity after prolonged exposure | Results in the brain:
| [143] |
| Danio rerio (Zebrafish) | GLY: 5 or 10 mg/L for 24 and 96 h | Evaluate oxidative stress parameters, as well as the activity and expression of AChE |
| [144] |
| Cnesterodon decemmaculatus | GLY: 1, 17.5, or 35 mg/L for 96 h | Assess the toxic effect of acute exposure to sublethal GLY concentrations on AChE activity in different parts of the body |
| [145] |
| Odontesthes bonariensis (Argentinian silverside) | GBH: 1 or 10 mg/L for 15 days | Determine the basal levels of adenylates, phosphagens, and the AEC index in the brain, muscle, and liver, as well as the impact of exposure to sublethal GBH on the subcellular energy balance |
| [146] |
| Danio rerio (Zebrafish) | GBH, GLY: 0.065, 1, 10, 160, 1.6 × 103, 4 × 103, or 8 × 103 mg/L for 3 h (in vitro) or for 7 days (in vivo) | Investigate the neurotoxic effects of GBH by focusing on acute toxicity, activity, and transcription levels of mitochondrial respiratory chain complexes, mitochondrial membrane potential, reactive species formation, and behavioral repertoire | In vivo exposure to GBH (7 days) caused:
| [147] |
| Danio rerio (Zebrafish) | Roundup®, GLY: 50 μg/mL for 24 h | Investigate the neurotoxic effects of GBH and GLY exposure on the developing brain | Both GBH and GLY caused:
| [148] |
| Jenynsia multidentate (Onesided livebearer) | Roundup® (Original, Transorb or WG): 0.5 mg/L for 96 h | Evaluate and compare the effects of three GBH formulations on behavior patterns | Roundup WG® was the most harmful formulation and negatively affected:
| [149] |
| Rhamdia quelen (Silver catfish) | GLY: 6.5 mg/L for 12, 24, 48, or 72 h | Investigate the effects of GLY on the antioxidant system, as well as the neurotoxic effects on eggs and larvae |
| [150] |
| Danio rerio (Zebrafish) | GLY: 0.01, 0.1, 0.5, 1, 5, 10, 100, 200, 400, or 600 mg/L from 3 hpf until 96 hpf | Assess the developmental, morphological, and genetic effects of GLY in zebrafish embryos |
| [151] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costas-Ferreira, C.; Durán, R.; Faro, L.R.F. Toxic Effects of Glyphosate on the Nervous System: A Systematic Review. Int. J. Mol. Sci. 2022, 23, 4605. https://doi.org/10.3390/ijms23094605
Costas-Ferreira C, Durán R, Faro LRF. Toxic Effects of Glyphosate on the Nervous System: A Systematic Review. International Journal of Molecular Sciences. 2022; 23(9):4605. https://doi.org/10.3390/ijms23094605
Chicago/Turabian StyleCostas-Ferreira, Carmen, Rafael Durán, and Lilian R. F. Faro. 2022. "Toxic Effects of Glyphosate on the Nervous System: A Systematic Review" International Journal of Molecular Sciences 23, no. 9: 4605. https://doi.org/10.3390/ijms23094605
APA StyleCostas-Ferreira, C., Durán, R., & Faro, L. R. F. (2022). Toxic Effects of Glyphosate on the Nervous System: A Systematic Review. International Journal of Molecular Sciences, 23(9), 4605. https://doi.org/10.3390/ijms23094605






