The Pathogenesis of Giant Condyloma Acuminatum (Buschke-Lowenstein Tumor): An Overview
Abstract
1. Introduction
2. Giant Condyloma Acuminatum (Buschke-Lowenstein Tumor)—A Short Description of the Disease
2.1. Classification
2.2. Clinical Presentation
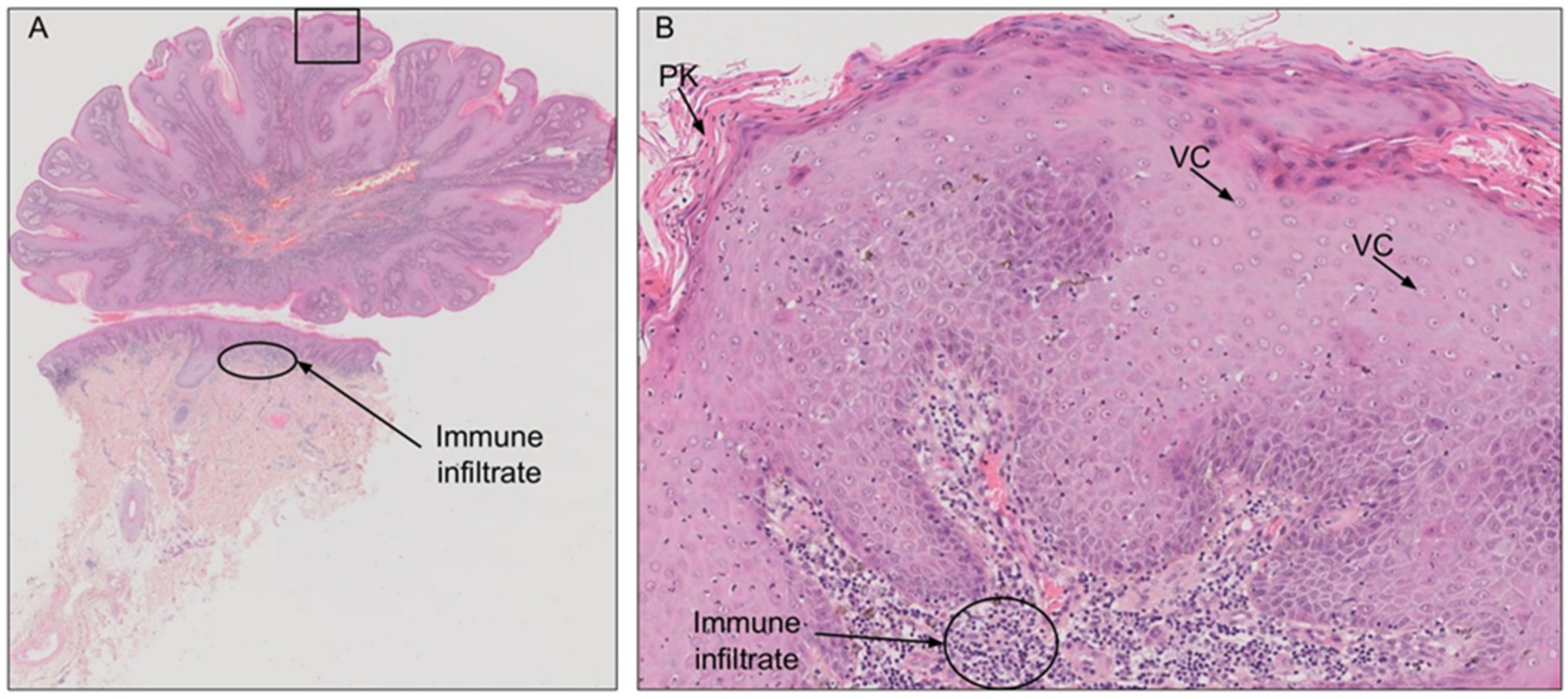
2.3. Diagnosis
2.4. Treatment
3. The Role of Low-Risk HPVs in BLT Pathogenesis
4. Possible Impact of Viral Genome Rearrangements, Mutations, and Host Genome Integration on BLT Development
5. Secondary Immunodeficiencies in the Etiology of BLT
6. Leading Genetic Causes of Susceptibility to Extensive Anogenital HPV Lesions
7. Significance of the CD28 Axis in the Development of HPV-Related Anogenital Lesions
8. Other Genes in the Pathogenesis of Anogenital HPV-Induced Lesions
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Altamura, G.; Tommasino, M.; Borzacchiello, G. Cutaneous vs. Mucosal Tropism: The Papillomavirus Paradigm Comes to an “and”. Front. Microbiol. 2020, 11, 588663. [Google Scholar] [CrossRef] [PubMed]
- McBride, A.A. Human papillomaviruses: Diversity, infection and host interactions. Nat. Rev. Microbiol. 2021, 20, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.; Campbell, C.M.P.; Waterboer, T.; Rollison, D.E.; Ingles, D.J.; Torres, B.N.; Michel, A.; Sudenga, S.L.; Pawlita, M.; Villa, L.L.; et al. Seroprevalence of cutaneous human papillomaviruses (HPVs) among men in the multinational HPV Infection in Men study. J. Gen. Virol. 2016, 97, 3291–3301. [Google Scholar] [CrossRef]
- Antonsson, A.; Green, A.C.; Mallitt, K.-A.; O’Rourke, P.K.; Pandeya, N.; Pawlita, M.; Waterboer, T.; Neale, R.E. Prevalence and stability of antibodies to 37 human papillomavirus types—A population-based longitudinal study. Virology 2010, 407, 26–32. [Google Scholar] [CrossRef]
- Tulay, P.; Serakinci, N. The role of human papillomaviruses in cancer progression. J. Cancer Metastasis Treat. 2016, 2, 201–213. [Google Scholar] [CrossRef]
- Manini, I.; Montomoli, E. Epidemiology and prevention of Human Papillomavirus. Ann Ig 2018, 30, 28–32. [Google Scholar] [CrossRef]
- de Sanjosé, S.; Bruni, L.; Alemany, L. HPV in genital cancers (at the exception of cervical cancer) and anal cancers. Presse Médicale 2014, 43, e423–e428. [Google Scholar] [CrossRef]
- Béziat, V.; Rapaport, F.; Hu, J.; Titeux, M.; des Claustres, M.B.; Bourgey, M.; Griffin, H.; Bandet, É.; Ma, C.S.; Sherkat, R.; et al. Humans with inherited T cell CD28 deficiency are susceptible to skin papillomaviruses but are otherwise healthy. Cell 2021, 184, 3812–3828.e30. [Google Scholar] [CrossRef]
- Wang, W.; Wang, C.; Xu, S.; Chen, C.; Tong, X.; Liang, Y.; Dong, X.; Lei, Y.; Zheng, X.; Yu, J.; et al. Detection of HPV-2 and identification of novel mutations by whole genome sequencing from biopsies of two patients with multiple cutaneous horns. J. Clin. Virol. 2007, 39, 34–42. [Google Scholar] [CrossRef]
- Pennycook, K.B.; McCready, T.A. Condyloma Acuminata. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK547667/ (accessed on 9 August 2021).
- Yuan, Y.; Cai, X.; Shen, F.; Ma, F. HPV post-infection microenvironment and cervical cancer. Cancer Lett. 2021, 497, 243–254. [Google Scholar] [CrossRef]
- Ajila, V.; Shetty, H.; Babu, S.; Shetty, V.; Hegde, S. Human Papilloma Virus Associated Squamous Cell Carcinoma of the Head and Neck. J. Sex. Transm. Dis. 2015, 2015, 91024. [Google Scholar] [CrossRef] [PubMed]
- Rantshabeng, P.S.; Moyo, S.; Moraka, N.O.; Ndlovu, A.; MacLeod, I.J.; Gaseitsiwe, S.; Kasvosve, I. Prevalence of oncogenic human papillomavirus genotypes in patients diagnosed with anogenital malignancies in Botswana. BMC Infect. Dis. 2017, 17, 731. [Google Scholar] [CrossRef]
- De Jong, S.J.; Imahorn, E.; Itin, P.; Uitto, J.; Orth, G.; Jouanguy, E.; Casanova, J.-L.; Burger, B. Epidermodysplasia Verruciformis: Inborn Errors of Immunity to Human Beta-Papillomaviruses. Front. Microbiol. 2018, 9, 1222. [Google Scholar] [CrossRef] [PubMed]
- Deau, M.C.; Favre, M.; Jablonska, S.; Rueda, L.A.; Orth, G. Genetic heterogeneity of oncogenic human papillomavirus type 5 (HPV5) and phylogeny of HPV5 variants associated with epidermodysplasia verruciformis. J. Clin. Microbiol. 1993, 31, 2918–2926. [Google Scholar] [CrossRef] [PubMed]
- Orth, G. Host Defenses against Human Papillomaviruses: Lessons from Epidermodysplasia Verruciformis. Curr. Top. Microbiol. Immunol. 2008, 321, 59–83. [Google Scholar] [CrossRef]
- Béziat, V. Human genetic dissection of papillomavirus-driven diseases: New insight into their pathogenesis. Hum. Genet. 2020, 139, 919–939. [Google Scholar] [CrossRef]
- Béziat, V.; Casanova, J.-L.; Jouanguy, E. Human genetic and immunological dissection of papillomavirus-driven diseases: New insights into their pathogenesis. Curr. Opin. Virol. 2021, 51, 9–15. [Google Scholar] [CrossRef]
- Leiding, J.W.; Holland, S.M. Warts and all: Human papillomavirus in primary immunodeficiencies. J. Allergy Clin. Immunol. 2012, 130, 1030–1048. [Google Scholar] [CrossRef]
- Ramoz, N.; Rueda, L.-A.; Bouadjar, B.; Montoya, L.-S.; Orth, G.; Favre, M. Mutations in two adjacent novel genes are associated with epidermodysplasia verruciformis. Nat. Genet. 2002, 32, 579–581. [Google Scholar] [CrossRef]
- De Jong, S.J.; Créquer, A.; Matos, I.; Hum, D.; Gunasekharan, V.K.; Lorenzo, L.; Jabot-Hanin, F.; Imahorn, E.; Arias, A.A.; Vahidnezhad, H.; et al. The human CIB1–EVER1–EVER2 complex governs keratinocyte-intrinsic immunity to β-papillomaviruses. J. Exp. Med. 2018, 215, 2289–2310. [Google Scholar] [CrossRef]
- Atschekzei, F.; Jacobs, R.; Wetzke, M.; Sogkas, G.; Schröder, C.; Ahrenstorf, G.; Dhingra, A.; Ott, H.; Baumann, U.; Schmidt, R.E. A Novel CARMIL2 Mutation Resulting in Combined Immunodeficiency Manifesting with Dermatitis, Fungal, and Viral Skin Infections as Well as Selective Antibody Deficiency. J. Clin. Immunol. 2019, 39, 274–276. [Google Scholar] [CrossRef] [PubMed]
- Buschke, A. Neisser’s Sterokopischer Atlas; Fischer: New York, NY, USA, 1896. [Google Scholar]
- Buschke, A.; Löwenstein, L. Über carcinomähnliche condylomata accuminata des Penis. Klin. Wochenschr. 1925, 7, 1726–1728. [Google Scholar] [CrossRef]
- Sabuncuoglu, M.Z.; Sabuncuoglu, A.; Celik, G.; Sozen, I.; Cetin, R. Moist Secret Mass; Buschke-Lowenste in Tumor: A Report of Three Cases. Case Rep. Clin. Med. 2014, 3, 474–478. [Google Scholar] [CrossRef][Green Version]
- Chu, Q.D.; Vezeridis, M.P.; Libbey, N.P.; Wanebo, H.J. Giant condyloma acuminatum (Buschke-Löwenstein tumor) of the anorectal and perianal regions. Analysis of 42 cases. Dis. Colon Rectum 1994, 37, 950–957. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.P. Verruciform and Condyloma-like Squamous Proliferations in the Anogenital Region. Arch. Pathol. Lab. Med. 2019, 143, 821–831. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, R.; Min, Z.; Bhanot, N. A Gigantic Anogenital Lesion: Buschke-Lowenstein Tumor. Case Rep. Dermatol. Med. 2014, 2014, 650714. [Google Scholar] [CrossRef]
- Davis, K.G.; Barton, J.S.; Orangio, G.; Bivin, W.; Krane, S. Buschke-Lowenstein Tumors: A Review and Proposed Classification System. Sex. Transm. Dis. 2021, 48, e263–e268. [Google Scholar] [CrossRef]
- Zidar, N.; Langner, C.; Odar, K.; Hošnjak, L.; Kamarádová, K.; Daum, O.; Pollheimer, M.J.; Košorok, P.; Poljak, M. Anal verrucous carcinoma is not related to infection with human papillomaviruses and should be distinguished from giant condyloma (Buschke-Löwenstein tumour). Histopathology 2017, 70, 938–945. [Google Scholar] [CrossRef]
- Haycox, C.L.; Kuypers, J.; Kriegerm, J.N. Role of human papillomavirus typing in diagnosis and clinical decision making for a giant verrucous genital lesion. Urology 1999, 53, 627–630. [Google Scholar] [CrossRef]
- Pineda-Murillo, J.; Lugo-García, J.A.; Martínez-Carrillo, G.; Torres-Aguilar, J.; Viveros-Contreras, C.; Schettino-Peredo, M.V. Buschke–Löwenstein tumor of the penis. Afr. J. Urol. 2019, 25, 9. [Google Scholar] [CrossRef]
- Kadouri, Y.; Nouini, Y. La tumeur de Buschke-Löwenstein [Buschke-Löwenstein’ tumor]. Pan Afr. Med. J. 2020, 36, 359. [Google Scholar] [CrossRef] [PubMed]
- Bastola, S.; Halalau, A.; Kc, O.; Adhikari, A. A Gigantic Anal Mass: Buschke–Löwenstein Tumor in a Patient with Controlled HIV Infection with Fatal Outcome. Case Rep. Infect. Dis. 2018, 2018, 7267213. [Google Scholar] [CrossRef]
- Diani, M.; Boneschi, V.; Ramoni, S.; Gadda, F.; Del Gobbo, A.; Cusini, M. Rapidly Invasive Buschke-Löwenstein Tumor Associated with Human Papillomavirus Types 6 and 52. Sex. Transm. Dis. 2015, 42, 547–548. [Google Scholar] [CrossRef]
- De Jong, A.R.; Weiss, J.C.; Brent, R.L. Condyloma Acuminata in Children. Am. J. Dis. 1982, 136, 704–706. [Google Scholar] [CrossRef]
- Nieves-Condoy, J.F.; Acuña-Pinzón, C.L.; Chavarría-Chavira, J.L.; Hinojosa-Ugarte, D.; Zúñiga-Vázquez, L.A. Giant Condyloma Acuminata (Buschke-Lowenstein Tumor): Review of an Unusual Disease and Difficult to Manage. Infect. Dis. Obstet. Gynecol. 2021, 2021, 9919446. [Google Scholar] [CrossRef] [PubMed]
- Purzycka-Bohdan, D.; Szczerkowska-Dobosz, A.; Swiatecka-Czaj, J.; Peksa, R.; Urban, M.; Szczypior, M.; Nowicki, R.J. Buschke-Löwenstein tumour associated with low-risk human papillomavirus genotypes successfully treated surgically. Postepy Dermatol. Allergol. 2019, 36, 112–114. [Google Scholar] [CrossRef]
- El Khoury, A.; Jensen, J.C.; Pacioles, T. Neoadjuvant chemotherapy and penile conservation in the management of Buschke–Lowenstein tumor, a case report. Urol. Case Rep. 2019, 29, 101111. [Google Scholar] [CrossRef] [PubMed]
- Combaud, V.; Verhaeghe, C.; El Hachem, H.; Legendre, G.; Descamps, P.; Martin, L.; Bouet, P.-E. Giant condyloma acuminatum of the vulva: Successful management with imiquimod. JAAD Case Rep. 2018, 4, 692–694. [Google Scholar] [CrossRef] [PubMed]
- Anissa, Z.; Houda, H.-G.; Wafa, K.; Olfa, M.; Rym, B.-M.; Achraf, D.; Samy, F. Successful treatment with topical imiquimod of anal Buschke-Löwenstein tumor in a child. Dermatol. Ther. 2015, 28, 363–365. [Google Scholar] [CrossRef] [PubMed]
- Dinleyici, M.; Saracoglu, N.; Eren, M.; Kiliç, Ö.; Ciftci, E.; Dinleyici, E.C.; Sag, C.; Kara, A. Giant Condyloma Acuminate Due to Human Papillomavirus Type 16 in an Infant Successfully Treated with Topical Imiquimod Therapy. Dermatol. Rep. 2015, 7, 6134. [Google Scholar] [CrossRef] [PubMed]
- Hum, M.; Chow, E.; Schuurmans, N.; Dytoc, M. Case of giant vulvar condyloma acuminata successfully treated with imiquimod 3.75% cream: A case report. SAGE Open Med. Case Rep. 2018, 6, 2050313X18802143. [Google Scholar] [CrossRef] [PubMed]
- Geusau, A.; Heinz-Peer, G.; Volc-Platzer, B.; Stingl, G.; Kirnbauer, R. Regression of Deeply Infiltrating Giant Condyloma (Buschke-Löwenstein Tumor) Following Long-term Intralesional Interferon Alfa Therapy. Arch. Dermatol. 2000, 136, 707–710. [Google Scholar] [CrossRef]
- Petrini, C.G.; Melli, P.P.D.S.; Magnani, P.S.; Rocha, L.P.; Faria, F.M.; Duarte, G.; Quintana, S.M. Giant Condyloma (Buschke-Loewenstein Tumor) in a 16-year-old Patient: Case Report. Rev. Bras. Ginecol. Obs. 2016, 38, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Grassegger, A.; Höpfl, R.; Hussl, H.; Wicke, K.; Fritsch, P. Buschke—Loewenstein tumour infiltrating pelvic organs. Br. J. Dermatol. 1994, 130, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Antony, F.C.; Ardern-Jones, M.; Evans, A.V.; Rosenbaum, T.; Russell-Jones, R. Giant condyloma of Buschke-Loewenstein in association with erythroderma. Clin. Exp. Dermatol. 2003, 28, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Bastard, P.; Michailidis, E.; Hoffmann, H.-H.; Chbihi, M.; Le Voyer, T.; Rosain, J.; Philippot, Q.; Seeleuthner, Y.; Gervais, A.; Materna, M.; et al. Auto-antibodies to type I IFNs can underlie adverse reactions to yellow fever live attenuated vaccine. J. Exp. Med. 2021, 218, e20202486. [Google Scholar] [CrossRef]
- Bustamante, J. Mendelian susceptibility to mycobacterial disease: Recent discoveries. Hum. Genet. 2020, 139, 993–1000. [Google Scholar] [CrossRef]
- Duncan, C.J.; Mohamad, S.M.; Young, D.F.; Skelton, A.J.; Leahy, T.R.; Munday, D.C.; Butler, K.M.; Morfopoulou, S.; Brown, J.R.; Hubank, M.; et al. Human IFNAR2 deficiency: Lessons for antiviral immunity. Sci. Transl. Med. 2015, 7, 307ra154. [Google Scholar] [CrossRef]
- Bowman, I.A.; Parra, A.; Arriaga, Y. Metastatic Giant Condyloma Acuminata (Buschke-Löwenstein Tumor). J. Oncol. Pract. 2016, 12, 951–953. [Google Scholar] [CrossRef]
- Lukács, A.; Máté, Z.; Farkas, N.; Mikó, A.; Tenk, J.; Hegyi, P.; Németh, B.; Czumbel, L.M.; Wuttapon, S.; Kiss, I.; et al. The quadrivalent HPV vaccine is protective against genital warts: A meta-analysis. BMC Public Health 2020, 20, 691. [Google Scholar] [CrossRef]
- Kazlouskaya, M.; Fiadorchanka, N. Regression of giant condyloma acuminata after one dose of 9-valent human papillomavirus (HPV) vaccine. Int. J. Dermatol. 2019, 58, e245–e247. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.; Smith-Matthews, S.; Ho, J. Giant condyloma of Buschke-Lowenstein in a patient with pemphigus vegetans treated with intralesional and systemic human papillomavirus vaccine. JAAD Case Rep. 2021, 20, 54–57. [Google Scholar] [CrossRef] [PubMed]
- McCutcheon, T. Anal Condyloma Acuminatum. Gastroenterol. Nurs. 2009, 32, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Danielewski, J.A.; Garland, S.M.; McCloskey, J.; Hillman, R.J.; Tabrizi, S.N. Human Papillomavirus Type 6 and 11 Genetic Variants Found in 71 Oral and Anogenital Epithelial Samples from Australia. PLoS ONE 2013, 8, e63892. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Franceschi, S.; Clifford, G.M. Human papillomavirus types from infection to cancer in the anus, according to sex and HIV status: A systematic review and meta-analysis. Lancet Infect. Dis. 2018, 18, 198–206. [Google Scholar] [CrossRef]
- Li, N.; Franceschi, S.; Howell-Jones, R.; Snijders, P.J.; Clifford, G.M. Human papillomavirus type distribution in 30,848 invasive cervical cancers worldwide: Variation by geographical region, histological type and year of publication. Int. J. Cancer 2011, 128, 927–935. [Google Scholar] [CrossRef]
- Wells, M.; Robertson, S.; Lewis, F.; Dixon, M.F. Squamous carcinoma arising in a giant peri-anal condyloma associated with human papillomavirus types 6 and 11. Histopathology 1988, 12, 319–323. [Google Scholar] [CrossRef]
- Martin, J.M.; Molina, I.; Monteagudo, C.; Martí, N.; Lopez, V.; Jorda, E. Buschke-Lowenstein tumor. J. Dermatol. Case Rep. 2008, 2, 60–62. [Google Scholar] [CrossRef]
- Handisurya, A.; Rieger, A.; Bago-Horvath, Z.; Schellenbacher, C.; Bankier, A.; Salat, A.; Stingl, G.; Kirnbauer, R. Rapid progression of an anal Buschke-Lowenstein tumour into a metastasising squamous cell carcinoma in an HIV-infected patient. Sex. Transm. Infect. 2009, 85, 261–263. [Google Scholar] [CrossRef]
- Egawa, N.; Doorbar, J. The low-risk papillomaviruses. Virus Res. 2017, 231, 119–127. [Google Scholar] [CrossRef]
- Hoppe-Seyler, K.; Bossler, F.; Braun, J.A.; Herrmann, A.L.; Hoppe-Seyler, F. The HPV E6/E7 Oncogenes: Key Factors for Viral Carcinogenesis and Therapeutic Targets. Trends Microbiol. 2018, 26, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Vande Pol, S.B.; Klingelhutz, A.J. Papillomavirus E6 oncoproteins. Virology 2013, 445, 115–137. [Google Scholar] [CrossRef] [PubMed]
- Roman, A.; Munger, K. The papillomavirus E7 proteins. Virology 2013, 445, 138–168. [Google Scholar] [CrossRef]
- Mwapagha, L.M.; Tiffin, N.; Parker, M.I. Delineation of the HPV11E6 and HPV18E6 Pathways in Initiating Cellular Transformation. Front. Oncol. 2017, 7, 258. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Xiao, Y.; Zhou, S.; Wang, Y.; Wang, J. Transcriptomic Landscape of Gene Expression Profiles and Pathways in Juvenile-Onset Recurrent Respiratory Papillomatosis Tumor Tissues and Human Papillomavirus 6 and 11 E6- and E7-Overexpressing Head and Neck Squamous Cell Carcinoma Cell Lines. J. Virol. 2022, 96, e0134221. [Google Scholar] [CrossRef] [PubMed]
- Murakami, I.; Egawa, N.; Griffin, H.; Yin, W.; Kranjec, C.; Nakahara, T.; Kiyono, T.; Doorbar, J. Roles for E1-independent replication and E6-mediated p53 degradation during low-risk and high-risk human papillomavirus genome maintenance. PLoS Pathog. 2019, 15, e1007755. [Google Scholar] [CrossRef] [PubMed]
- Gage, J.R.; Meyers, C.; Wettstein, F.O. The E7 proteins of the nononcogenic human papillomavirus type 6b (HPV-6b) and of the oncogenic HPV-16 differ in retinoblastoma protein binding and other properties. J. Virol. 1990, 64, 723–730. [Google Scholar] [CrossRef]
- Pilotti, S.; Donghi, R.; D’Amato, L.; Giarola, M.; Longoni, A.; Della Torre, G.; De Palo, G.; Pierotti, M.A.; Rilke, F. HPV detection and p53 alteration in squamous cell verrucous malignancies of the lower genital tract. Diagn. Mol. Pathol. 1993, 2, 248–256. [Google Scholar] [CrossRef]
- McBride, A.A.; Warburton, A. The role of integration in oncogenic progression of HPV-associated cancers. PLoS Pathog. 2017, 13, e1006211. [Google Scholar] [CrossRef]
- Péré, H.; Vernet, R.; Pernot, S.; Pavie, J.; Robillard, N.; Puech, J.; Lameiras, S.; Lucas, M.-L.; Nicolas, A.; Badoual, C.; et al. Episomal HPV16 responsible for aggressive and deadly metastatic anal squamous cell carcinoma evidenced in peripheral blood. Sci. Rep. 2021, 11, 4633. [Google Scholar] [CrossRef]
- Cullen, A.P.; Reid, R.; Campion, M.; Lörincz, A.T. Analysis of the physical state of different human papillomavirus DNAs in intraepithelial and invasive cervical neoplasm. J. Virol. 1991, 65, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Lehn, H.; Ernst, T.-M.; Sauer, G. Transcription of Episomal Papillomavirus DNA in Human Condylomata Acuminata and Buschke-Löwenstein Tumours. J. Gen. Virol. 1984, 65, 2003–2010. [Google Scholar] [CrossRef] [PubMed]
- Boshart, M.; zur Hausen, H. Human papillomaviruses in Buschke-Löwenstein tumors: Physical state of the DNA and identification of a tandem duplication in the noncoding region of a human papillomavirus 6 subtype. J. Virol. 1986, 58, 963–966. [Google Scholar] [CrossRef] [PubMed]
- Palefsky, J.M.; Giuliano, A.R.; Goldstone, S.; Moreira, E.D., Jr.; Aranda, C.; Jessen, H.; Hillman, R.; Ferris, D.; Coutlee, F.; Stoler, M.H.; et al. HPV Vaccine against Anal HPV Infection and Anal Intraepithelial Neoplasia. N. Engl. J. Med. 2011, 365, 1576–1585. [Google Scholar] [CrossRef]
- Insinga, R.P.; Liaw, K.-L.; Johnson, L.G.; Madeleine, M.M. A Systematic Review of the Prevalence and Attribution of Human Papillomavirus Types among Cervical, Vaginal, and Vulvar Precancers and Cancers in the United States. Cancer Epidemiol. Biomark. Prev. 2008, 17, 1611–1622. [Google Scholar] [CrossRef]
- Manias, D.A.; Ostrow, R.S.; McGlennen, R.C.; Estensen, R.D.; Faras, A.J. Characterization of integrated human papillomavirus type 11 DNA in primary and metastatic tumors from a renal transplant recipient. Cancer Res. 1989, 49, 2514–2519. [Google Scholar]
- Reidy, P.M.; Dedo, H.H.; Rabah, R.; Field, J.B.; Mathog, R.H.; Gregoire, L.; Lancaster, W.D. Integration of Human Papillomavirus Type 11 in Recurrent Respiratory Papilloma-Associated Cancer. Laryngoscope 2004, 114, 1906–1909. [Google Scholar] [CrossRef]
- Bercovich, J.A.; Centeno, C.R.; Aguilar, O.G.; Grinsteinm, S.; Kahn, T. Presence and integration of human papillomavirus type 6 in a tonsillar carcinoma. J. Gen. Virol. 1991, 72, 2569–2572. [Google Scholar] [CrossRef]
- Kahn, T.; Turazza, E.; Ojeda, R.; Bercovich, A.; Stremlau, A.; Lichter, P.; Poustka, A.; Grinstein, S.; zur Hausen, H. Integration of human papillomavirus type 6a DNA in a tonsillar carcinoma: Chromosomal localization and nucleotide sequence of the genomic target region. Cancer Res. 1994, 54, 1305–1312. [Google Scholar]
- Rübben, A.; Beaudenon, S.; Favre, M.; Schmitz, W.; Spelten, B.; Grussendorf-Conen, E.-I. Rearrangements of the upstream regulatory region of human papillomavirus type 6 can be found in both Buschke-Löwenstein tumours and in condylomata acuminata. J. Gen. Virol. 1992, 73, 3147–3153. [Google Scholar] [CrossRef]
- Albert, R.; Spelten, B.; Albrecht, J.; Grußendorf-Conen, E.-I. Demonstration of URR-duplication variants of human papillomavirus type 6 in paraffin-embedded tissue sections of one condyloma acuminatum and one buschke-lowenstein tumour. J. Pathol. 1994, 174, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Kitasato, H.; Delius, H.; zur Hausen, H.; Sorger, K.; Rösl, F.; de Villiers, E.-M. Sequence Rearrangements in the Upstream Regulatory Region of Human Papillomavirus Type 6: Are these Involved in Malignant Transition? J. Gen. Virol. 1994, 75, 1157–1162. [Google Scholar] [CrossRef] [PubMed]
- Rosen, M.; Auborn, K. Duplication of the upstream regulatory sequences increases the transformation potential of human papillomavirus type 11. Virology 1991, 185, 484–487. [Google Scholar] [CrossRef]
- Di Luca, D.; Caselli, E.; Monini, P.; Rotola, A.; Savioli, A.; Cassai, E. Episomal HPV 16 DNA isolated from a cervical carcinoma presents a partial duplication of the early region. Virus Res. 1989, 14, 49–55. [Google Scholar] [CrossRef]
- Byrne, J.C.; Tsao, M.S.; Fraser, R.S.; Howley, P.M. Human Papillomavirus-11 DNA in a Patient with Chronic Laryngotracheobronchial Papillomatosis and Metastatic Squamous-Cell Carcinoma of the Lung. N. Engl. J. Med. 1987, 317, 873–878. [Google Scholar] [CrossRef]
- DiLorenzo, T.P.; Tamsen, A.; Abramson, A.L.; Steinberg, B.M. Human papillomavirus type 6a DNA in the lung carcinoma of a patient with recurrent laryngeal papillomatosis is characterized by a partial duplication. J. Gen. Virol. 1992, 73, 423–428. [Google Scholar] [CrossRef]
- Kasher, M.S.; Roman, A. Characterization of human papillomavirus type 6b DNA isolated from an invasive squamous carcinoma of the vulva. Virology 1988, 165, 225–233. [Google Scholar] [CrossRef]
- Rando, R.F.; Groff, D.E.; Chirikjian, J.G.; Lancaster, W.D. Isolation and characterization of a novel human papillomavirus type 6 DNA from an invasive vulvar carcinoma. J. Virol. 1986, 57, 353–356. [Google Scholar] [CrossRef]
- Rando, R.F.; Lancaster, W.D.; Han, P.; Lopez, C. The noncoding region of HPV-6vc contains two distinct transcriptional enhancing elements. Virology 1986, 155, 545–556. [Google Scholar] [CrossRef]
- Farr, A.; Wang, H.; Kasher, M.S.; Roman, A. Relative enhancer activity and transforming potential of authentic human papillomavirus type 6 genomes from benign and malignant lesions. J. Gen. Virol. 1991, 72, 519–526. [Google Scholar] [CrossRef]
- Sporkert, M.; Rübben, A. Buschke-Löwenstein-Tumor [Buschke-Lowenstein tumors]. Hautarzt 2017, 68, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Wieland, U.; Kreuter, A.; Pfister, H. Human Papillomavirus and Immunosuppression. Curr. Probl. Dermatol. 2014, 45, 154–165. [Google Scholar] [CrossRef] [PubMed]
- Chin-Hong, P.V.; Reid, G.E.; The AST Infectious Diseases Community of Practice. Human papillomavirus infection in solid organ transplant recipients: Guidelines from the American Society of Transplantation Infectious Diseases Community of Practice. Clin. Transplant. 2019, 33, e13590. [Google Scholar] [CrossRef] [PubMed]
- Gormley, R.H.; Kovarik, C.L. Human papillomavirus–related genital disease in the immunocompromised host: Part I. J. Am. Acad. Dermatol. 2012, 66, 867.e1–867.e17. [Google Scholar] [CrossRef] [PubMed]
- Tschachler, E.; Bergstresser, P.R.; Stingl, G. HIV-related skin diseases. Lancet 1996, 348, 659–663. [Google Scholar] [CrossRef]
- Coleman, N.; Birley, H.D.; Renton, A.M.; Hanna, N.F.; Ryait, B.K.; Byrne, M.; Taylor-Robinson, D.; Stanley, M.A. Immunological Events in Regressing Genital Warts. Am. J. Clin. Pathol. 1994, 102, 768–774. [Google Scholar] [CrossRef]
- Fierlbeck, G.; Schiebel, U.; Müller, C. Immunohistology of Genital Warts in Different Stages of Regression after Therapy with Interferon Gamma. Dermatology 1989, 179, 191–195. [Google Scholar] [CrossRef]
- Akinboro, A.O.; Onayemi, O.; Mejiuni, A.D. Frequency, pattern, and extent of skin diseases in relation to CD4+ cell count among adults with human immunodeficiency virus infection or acquired immunodeficiency syndrome in Osogbo, southwestern Nigeria. Int. J. Dermatol. 2014, 53, 416–424. [Google Scholar] [CrossRef]
- Reusser, N.M.; Downing, C.; Guidry, J.; Tyring, S.K. HPV Carcinomas in Immunocompromised Patients. J. Clin. Med. 2015, 4, 260–281. [Google Scholar] [CrossRef]
- Dhumale, S.B. Ano-Genital Warts and HIV Status–A Clinical Study. J. Clin. Diagn. Res. 2017, 11, WC01–WC04. [Google Scholar] [CrossRef]
- De Araújo, P.S.R.; Padilha, C.E.G.; Soares, M.F. Buschke-Lowenstein tumor in a woman living with HIV/AIDS. Rev. Soc. Bras. Med. Trop. 2017, 50, 577. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, A.L.; Pursell, N.; Sisay, A. The Giant Condyloma (Buschke-Löwenstein Tumor) in the Immunocompromised Patient. Case Rep. Obstet. Gynecol. 2014, 2014, 793534. [Google Scholar] [CrossRef] [PubMed]
- Ledouble, V.; Sclafani, F.; Hendlisz, A.; Galdon, M.G.; Liberale, G. Buschke-Löwenstein tumor in a human immunodeficiency virus-positive patient: A case report and short literature review. Acta Gastroenterol. Belg. 2021, 84, 343–345. [Google Scholar] [CrossRef] [PubMed]
- Rachman, A.; Hasan, N. Giant Condyloma Acuminata in Indonesian Females with SLE under Immunosuppressant and Steroid Therapy. Case Rep. Immunol. 2016, 2016, 4710979. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, N.; Waller, S.; Stasik, C.J.; Skiknem, B.S.; Ganguly, S. Giant anal condylomatosis after allogeneic bone marrow transplantation: A rare complication of human papilloma virus infection. Transpl. Infect. Dis. 2008, 10, 56–58. [Google Scholar] [CrossRef]
- Das, B.B.; Anton, K.; Knox, L.; Jarin, J.; Sue, P.K. Successful treatment of giant condyloma in a pediatric heart transplant recipient with topical cidofovir. Transpl. Infect. Dis. 2018, 20, e12989. [Google Scholar] [CrossRef]
- Wester, N.E.; Hutten, E.M.; Krikke, C.; Pol, R.A. Intra-Abdominal Localisation of a Buschke-Lowenstein Tumour: Case Presentation and Review of the Literature. Case Rep. Transplant. 2013, 2013, 187682. [Google Scholar] [CrossRef]
- Indinnimeo, M.; Impagnatiello, A.; D’Ettorre, G.; Bernardi, G.; Moschella, C.M.; Gozzo, P.; Ciardi, A.; Bangrazi, C.; De Felice, F.; Musio, D.; et al. Buschke-Löwenstein tumor with squamous cell carcinoma treated with chemo-radiation therapy and local surgical excision: Report of three cases. World J. Surg. Oncol. 2013, 11, 231. [Google Scholar] [CrossRef]
- Grodner, C.; Henn, A.; Lelièvre, J.-D.; Gallien, S. Successful improvement of Buschke-Löwenstein tumour in an HIV-infected patient with antiretroviral therapy alone. BMJ Case Rep. 2016, 2016, bcr2016217753. [Google Scholar] [CrossRef]
- Wang, Y.; Ma, C.S.; Ling, Y.; Bousfiha, A.; Camcioglu, Y.; Jacquot, S.; Payne, K.; Crestani, E.; Roncagalli, R.; Belkadi, A.; et al. Dual T cell– and B cell–intrinsic deficiency in humans with biallelic RLTPR mutations. J. Exp. Med. 2016, 213, 2413–2435. [Google Scholar] [CrossRef]
- Sorte, H.S.; Osnes, L.T.; Fevang, B.; Aukrust, P.; Erichsen, H.C.; Backe, P.H.; Abrahamsen, T.G.; Kittang, O.B.; Øverland, T.; Jhangiani, S.; et al. A potential founder variant inCARMIL2/RLTPRin three Norwegian families with warts, molluscum contagiosum, and T-cell dysfunction. Mol. Genet. Genom. Med. 2016, 4, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Meuris, F.; Carthagena, L.; Jaracz-Ros, A.; Gaudin, F.; Cutolo, P.; Deback, C.; Xue, Y.; Thierry, F.; Doorbar, J.; Bachelerie, F. The CXCL12/CXCR4 Signaling Pathway: A New Susceptibility Factor in Human Papillomavirus Pathogenesis. PLoS Pathog. 2016, 12, e1006039. [Google Scholar] [CrossRef] [PubMed]
- Handisurya, A.; Schellenbacher, C.; Reininger, B.; Koszik, F.; Vyhnanek, P.; Heitger, A.; Kirnbauer, R.; Förster-Waldl, E. A quadrivalent HPV vaccine induces humoral and cellular immune responses in WHIM immunodeficiency syndrome. Vaccine 2010, 28, 4837–4841. [Google Scholar] [CrossRef]
- McDermott, D.H.; Murphy, P.M. WHIM syndrome: Immunopathogenesis, treatment and cure strategies. Immunol. Rev. 2019, 287, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Woodbine, L.; Grigoriadou, S.; Goodarzi, A.A.; Riballo, E.; Tape, C.; Oliver, A.W.; Van Zelm, M.C.; Buckland, M.S.; Davies, E.G.; Pearl, L.H.; et al. An Artemis polymorphic variant reduces Artemis activity and confers cellular radiosensitivity. DNA Repair 2010, 9, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- Volk, T.; Pannicke, U.; Reisli, I.; Bulashevska, A.; Ritter, J.; Björkman, A.; Schäffer, A.A.; Fliegauf, M.; Sayar, E.H.; Salzer, U.; et al. DCLRE1C(ARTEMIS) mutations causing phenotypes ranging from atypical severe combined immunodeficiency to mere antibody deficiency. Hum. Mol. Genet. 2015, 24, 7361–7372. [Google Scholar] [CrossRef]
- Chu, E.Y.; Freeman, A.F.; Jing, H.; Cowen, E.W.; Davis, J.; Su, H.C.; Holland, S.M.; Turner, M.L. Cutaneous Manifestations of DOCK8 Deficiency Syndrome. Arch. Dermatol. 2012, 148, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Al-Zahrani, D.; Raddadi, A.; Massaad, M.; Keles, S.; Jabara, H.H.; Chatila, T.A.; Geha, R. Successful interferon-alpha 2b therapy for unremitting warts in a patient with DOCK8 deficiency. Clin. Immunol. 2014, 153, 104–108. [Google Scholar] [CrossRef]
- Venegas-Montoya, E.; Staines-Boone, A.T.; Sánchez-Sánchez, L.M.; García-Campos, J.A.; Córdova-Gurrola, R.A.; Salazar-Galvez, Y.; Múzquiz-Zermeño, D.; González-Serrano, M.E.; Reyes, S.O.L. Case Report: DOCK8 Deficiency without Hyper-IgE in a Child with a Large Deletion. Front. Pediatr. 2021, 9, 635322. [Google Scholar] [CrossRef]
- Hsu, A.P.; McReynolds, L.J.; Holland, S.M. GATA2 deficiency. Curr. Opin. Allergy Clin. Immunol. 2015, 15, 104–109. [Google Scholar] [CrossRef]
- Spinner, M.A.; Sanchez, L.A.; Hsu, A.P.; Shaw, P.A.; Zerbe, C.S.; Calvo, K.R.; Arthur, D.C.; Gu, W.; Gould, C.M.; Brewer, C.C.; et al. GATA2 deficiency: A protean disorder of hematopoiesis, lymphatics, and immunity. Blood 2014, 123, 809–821. [Google Scholar] [CrossRef] [PubMed]
- Kuriyama, Y.; Hattori, M.; Mitsui, T.; Nakano, H.; Oikawa, D.; Tokunaga, F.; Ishikawa, O.; Shimizu, A. Generalized verrucosis caused by various human papillomaviruses in a patient with GATA2 deficiency. J. Dermatol. 2018, 45, e108–e109. [Google Scholar] [CrossRef] [PubMed]
- Cole, K.; Avila, D.; Parta, M.; Schuver, B.; Holland, S.; Shah, N.; Hickstein, D. GATA2 Deficiency: Early Identification for Improved Clinical Outcomes. Clin. J. Oncol. Nurs. 2019, 23, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Roussel, L.; Landekic, M.; Golizeh, M.; Gavino, C.; Zhong, M.-C.; Chen, J.; Faubert, D.; Blanchet-Cohen, A.; Dansereau, L.; Parent, M.-A.; et al. Loss of human ICOSL results in combined immunodeficiency. J. Exp. Med. 2018, 215, 3151–3164. [Google Scholar] [CrossRef]
- van de Vijver, E.; Maddalena, A.; Sanal, Ö.; Holland, S.M.; Uzel, G.; Madkaikar, M.; de Boer, M.; van Leeuwen, K.; Köker, M.Y.; Parvaneh, N.; et al. Hematologically important mutations: Leukocyte adhesion deficiency (first update). Blood Cells Mol. Dis. 2012, 48, 53–61. [Google Scholar] [CrossRef]
- Ravell, J.C.; Matsuda-Lennikov, M.; Chauvin, S.D.; Zou, J.; Biancalana, M.; Deeb, S.J.; Price, S.; Su, H.C.; Notarangelo, G.; Jiang, P.; et al. Defective glycosylation and multisystem abnormalities characterize the primary immunodeficiency XMEN disease. J. Clin. Investig. 2020, 130, 507–522. [Google Scholar] [CrossRef]
- Li, A.L.; Walsh, S.; McKay, D.R. Surgical management of a giant condyloma of Buschke-Löwenstein in a patient with Netherton syndrome using the pedicled anterolateral thigh flap—A case report. J. Plast. Reconstr. Aesthetic Surg. 2011, 64, 1533–1536. [Google Scholar] [CrossRef]
- Ashton, R.; Moledina, J.; Sivakumar, B.; Mellerio, J.E.; Martinez, A.E. Considerations in surgical management of a Buschke-Lowenstein tumor in Netherton syndrome: A case report. Pediatr. Dermatol. 2017, 34, e328–e330. [Google Scholar] [CrossRef]
- Mehta, H.; Paz, J.C.; Sadikot, R.T. Wiskott–Aldrich syndrome with bronchiectasis. Respir. Med. CME 2008, 1, 54–58. [Google Scholar] [CrossRef][Green Version]
- Kawai, T.; Malech, H.L. WHIM syndrome: Congenital immune deficiency disease. Curr. Opin. Hematol. 2009, 16, 20–26. [Google Scholar] [CrossRef]
- Donadieu, J.; Lamant, M.; Fieschi, C.; De Fontbrune, F.S.; Caye, A.; Ouachee, M.; Beaupain, B.; Bustamante, J.; Poirel, H.A.; Isidor, B.; et al. Natural history of GATA2 deficiency in a survey of 79 French and Belgian patients. Haematologica 2018, 103, 1278–1287. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Dove, C.G.; Hor, J.L.; Murdock, H.M.; Strauss-Albee, D.M.; Garcia, J.A.; Mandl, J.N.; Grodick, R.A.; Jing, H.; Chandler-Brown, D.B.; et al. DOCK8 regulates lymphocyte shape integrity for skin antiviral immunity. J. Exp. Med. 2014, 211, 2549–2566. [Google Scholar] [CrossRef] [PubMed]
- Harada, Y.; Tanaka, Y.; Terasawa, M.; Pieczyk, M.; Habiro, K.; Katakai, T.; Hanawa-Suetsugu, K.; Kukimoto-Niino, M.; Nishizaki, T.; Shirouzu, M.; et al. DOCK8 is a Cdc42 activator critical for interstitial dendritic cell migration during immune responses. Blood 2012, 119, 4451–4461. [Google Scholar] [CrossRef] [PubMed]
- Dorjbal, B.; Stinson, J.R.; Ma, C.A.; Weinreich, M.A.; Miraghazadeh, B.; Hartberger, J.M.; Frey-Jakobs, S.; Weidinger, S.; Moebus, L.; Franke, A.; et al. Hypomorphic caspase activation and recruitment domain 11 (CARD11) mutations associated with diverse immunologic phenotypes with or without atopic disease. J. Allergy Clin. Immunol. 2019, 143, 1482–1495. [Google Scholar] [CrossRef]
- Chinn, I.K.; Sanders, R.P.; Stray-Pedersen, A.; Coban-Akdemir, Z.H.; Kim, V.H.-D.; Dadi, H.; Roifman, C.M.; Quigg, T.; Lupski, J.R.; Orange, J.S.; et al. Novel Combined Immune Deficiency and Radiation Sensitivity Blended Phenotype in an Adult with Biallelic Variations in ZAP70 and RNF168. Front. Immunol. 2017, 8, 576. [Google Scholar] [CrossRef]
- Schepp, J.; Chou, J.; Skrabl-Baumgartner, A.; Arkwright, P.D.; Engelhardt, K.R.; Hambleton, S.; Morio, T.; Röther, E.; Warnatz, K.; Geha, R.S.; et al. 14 Years after Discovery: Clinical Follow-up on 15 Patients with Inducible Co-Stimulator Deficiency. Front. Immunol. 2017, 8, 964. [Google Scholar] [CrossRef]
- Insinga, R.P.; Dasbach, E.J.; Myers, E.R. The Health and Economic Burden of Genital Warts in a Set of Private Health Plans in the United States. Clin. Infect. Dis. 2003, 36, 1397–1403. [Google Scholar] [CrossRef]
- Lipke, M.M. An Armamentarium of Wart Treatments. Clin. Med. Res. 2006, 4, 273–293. [Google Scholar] [CrossRef]
| Primary Immunodeficiency | Gene Mutated | Inheritance | Phenotype | References |
|---|---|---|---|---|
| CARMIL2 deficiency | CARMIL2 (RLTPR) | AR | Common warts, recurrent condylomas, broad susceptibility to infection, immune dysregulation, EBV-driven smooth muscle tumors | [17,22,112,113] |
| WHIM syndrome | CXCR4 | AD | Common warts, condyloma acuminata, hypogammaglobulinemia (low IgG and IgA, normal IgM), infections, myelokathexis | [114,115,116] |
| DCLRE1C deficiency | DCLRE1C | AR (hypomorphic) | Extensive HPV-related anogenital lesions, atypical EV, low numbers of B cells, hypogammaglobulinemia | [117,118] |
| DOCK8 deficiency | DOCK8 | AR | Common warts, condyloma acuminata, atypical EV, other viral cutaneous infections (VZV, HSV, molluscum contagiosum), eczema, food allergy, asthma, allergic rhinitis, bacterial pneumonia, candidiasis, abscesses, cancer, thrombocytosis, eosinophilia, lymphopenia | [119,120,121] |
| GATA2 deficiency [DCML, MDS, MonoMAC syndrome, WILD syndrome, Emberger syndrome] | GATA2 | AD | Common warts, condyloma acuminata, VZV, HSV, fungal infections, lymphedema, myelodysplasia, leukemia, panniculitis, cancer, low B-cell levels | [19,122,123,124,125] |
| ICOSL deficiency | ICOSLG | AR | Common warts, extensive condyloma acuminata, orolabial HSV infections, angular cheilitis, mouth ulcers, hypogammaglobulinemia, neutropenia, lymphopenia | [126] |
| LAD-1 | ITGB2 | AR | Extensive common warts and condyloma acuminata, frequent systemic, skin, and soft tissue infections, inflammatory bowel disease, impaired wound healing, gingivitis, periodontitis | [19,127] |
| MAGT-1 deficiency XMEN syndrome | MAGT-1 | XLR | Cutaneous warts, perineal condylomas, EBV infections, infections of the ear and nose, viral infections of the skin, cancers | [17,128] |
| Netherton syndrome | SPINK5 | AR | Common warts, giant condyloma acuminata, ichthyosis, eczema, bamboo hair, asthma, food allergy, high IgE levels | [129,130] |
| WAS | WAS | XLR | Common warts, condyloma acuminata, thrombocytopenia, infections, eczema, cancers, autoimmune manifestations | [17,131] |
| Gene (Official Symbol) | Gene (Official Full Name) | Function of Encoded Protein |
|---|---|---|
| CARD11 | caspase activation and recruitment domain 11 | a member of the membrane-associated guanylate kinase (MAGUK) family; plays a key role in adaptive immune response by transducing the activation of NF-kappa-B downstream of T-cell receptor and B-cell receptor engagement |
| CARMIL2 | capping protein regulator and myosin 1 linker 2 | a member of CARMIL family of proteins; involved in the CD28 cosignaling of T cells, and in cytoskeletal organization and cell migration |
| CIB1 | calcium and integrin binding 1 | regulator of diverse cellular processes including migration, adhesion, proliferation, and cell death/survival. CIB1 deficiency is associated with epidermodysplasia verruciformis |
| CXCR4 | C-X-C chemokine receptor type 4 | a receptor of the CXCL12 chemokine; is involved in multiple signaling pathways that orchestrate cell migration, hematopoiesis and cell homing, and retention in the bone marrow |
| DCLRE1C | DNA cross-link repair 1C | a nuclear protein; regulation of the cell cycle in response to DNA damage, and TCR and BCR recombination |
| DOCK8 | dedicator of cytokinesis 8 | a member of the DOCK180 family of guanine nucleotide exchange factors; critical role in cell migration and survival of several types of immune system cells |
| GATA2 | GATA-binding protein 2 | a member of the GATA family of zinc-finger transcription factors; plays a critical role in maintaining the pool of early hematopoietic cells |
| ICOS | inducible T cell costimulator | protein belonging to the CD28 and CTLA-4 cell-surface receptor family; T cell co-activating receptor, involved in T cell immune responses |
| ICOSLG | inducible T cell costimulator ligand | ligand of ICOS, involved in T cell immune responses |
| IFNAR1 | interferon alpha and beta receptor subunit 1 | forms one of the two chains of a receptor for IFN-α and IFN-β; involved in immune response; functions as an antiviral factor |
| IFNAR2 | interferon alpha and beta receptor subunit 2 | forms one of the two chains of a receptor for IFN-α and IFN-β; involved in immune response; functions as an antiviral factor |
| IFNGR1 | interferon gamma receptor 1 | the ligand-binding chain (alpha) of the gamma interferon receptor; non-redundant roles against intra-cellular pathogens (in particular mycobacteria) |
| IFNGR2 | interferon gamma receptor 2 | the non-ligand-binding beta chain of the gamma interferon receptor. non-redundant roles against intra-cellular pathogens (in particular mycobacteria) |
| ITGB2 | integrin subunit beta 2 | an integrin beta chain; participate in cell adhesion as well as cell-surface mediated signaling |
| MAGT1 | magnesium transporter 1 | a ubiquitously expressed magnesium cation transporter protein; crucial for the glycosylation and cell-surface expression of major immune receptors, including CD28 |
| SPINK5 | serine peptidase inhibitor Kazal type 5 | lympho-epithelial Kazal-type related inhibitor (LEKT1); plays a role in skin and hair morphogenesis, as well as anti-inflammatory and antimicrobial protection of mucous epithelia |
| TMC6 (EVER1) | transmembrane channel like 6 | integral membrane protein located in the endoplasmic reticulum; predicted to form transmembrane channels; TMC6 deficiency is associated with epidermodysplasia verruciformis |
| TMC8 (EVER2) | transmembrane channel like 8 | integral membrane protein located in the endoplasmic reticulum; predicted to form transmembrane channels; TMC8 deficiency is associated with epidermodysplasia verruciformis |
| ZAP70 | zeta chain of T cell receptor associated protein kinase 70 | an enzyme belonging to the protein tyrosine kinase family; plays a role in T-cell development and lymphocyte activation; essential for thymocyte development |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Purzycka-Bohdan, D.; Nowicki, R.J.; Herms, F.; Casanova, J.-L.; Fouéré, S.; Béziat, V. The Pathogenesis of Giant Condyloma Acuminatum (Buschke-Lowenstein Tumor): An Overview. Int. J. Mol. Sci. 2022, 23, 4547. https://doi.org/10.3390/ijms23094547
Purzycka-Bohdan D, Nowicki RJ, Herms F, Casanova J-L, Fouéré S, Béziat V. The Pathogenesis of Giant Condyloma Acuminatum (Buschke-Lowenstein Tumor): An Overview. International Journal of Molecular Sciences. 2022; 23(9):4547. https://doi.org/10.3390/ijms23094547
Chicago/Turabian StylePurzycka-Bohdan, Dorota, Roman J. Nowicki, Florian Herms, Jean-Laurent Casanova, Sébastien Fouéré, and Vivien Béziat. 2022. "The Pathogenesis of Giant Condyloma Acuminatum (Buschke-Lowenstein Tumor): An Overview" International Journal of Molecular Sciences 23, no. 9: 4547. https://doi.org/10.3390/ijms23094547
APA StylePurzycka-Bohdan, D., Nowicki, R. J., Herms, F., Casanova, J.-L., Fouéré, S., & Béziat, V. (2022). The Pathogenesis of Giant Condyloma Acuminatum (Buschke-Lowenstein Tumor): An Overview. International Journal of Molecular Sciences, 23(9), 4547. https://doi.org/10.3390/ijms23094547








