Activation of TRPV4 Induces Exocytosis and Ferroptosis in Human Melanoma Cells
Abstract
:1. Introduction
2. Results
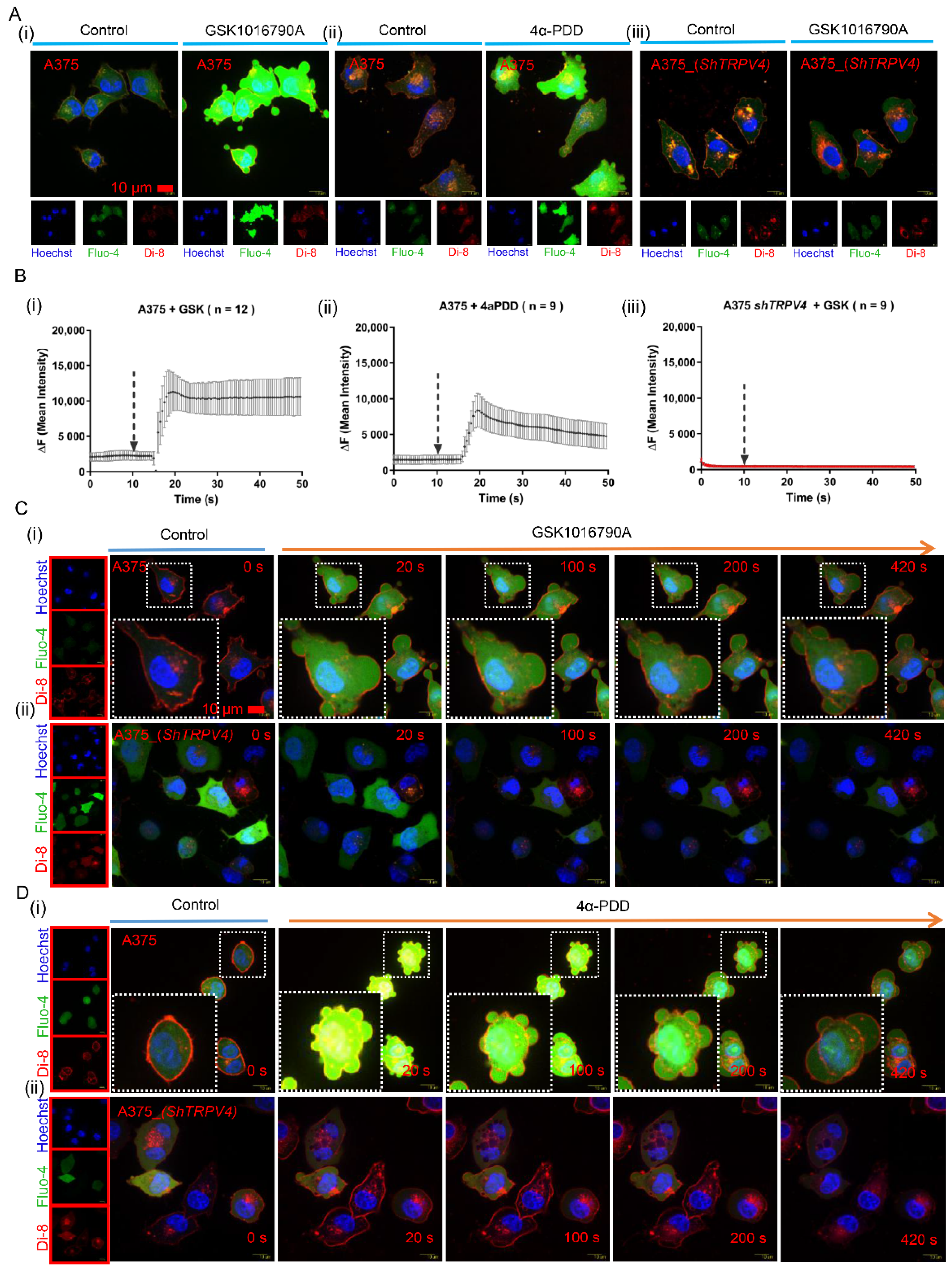
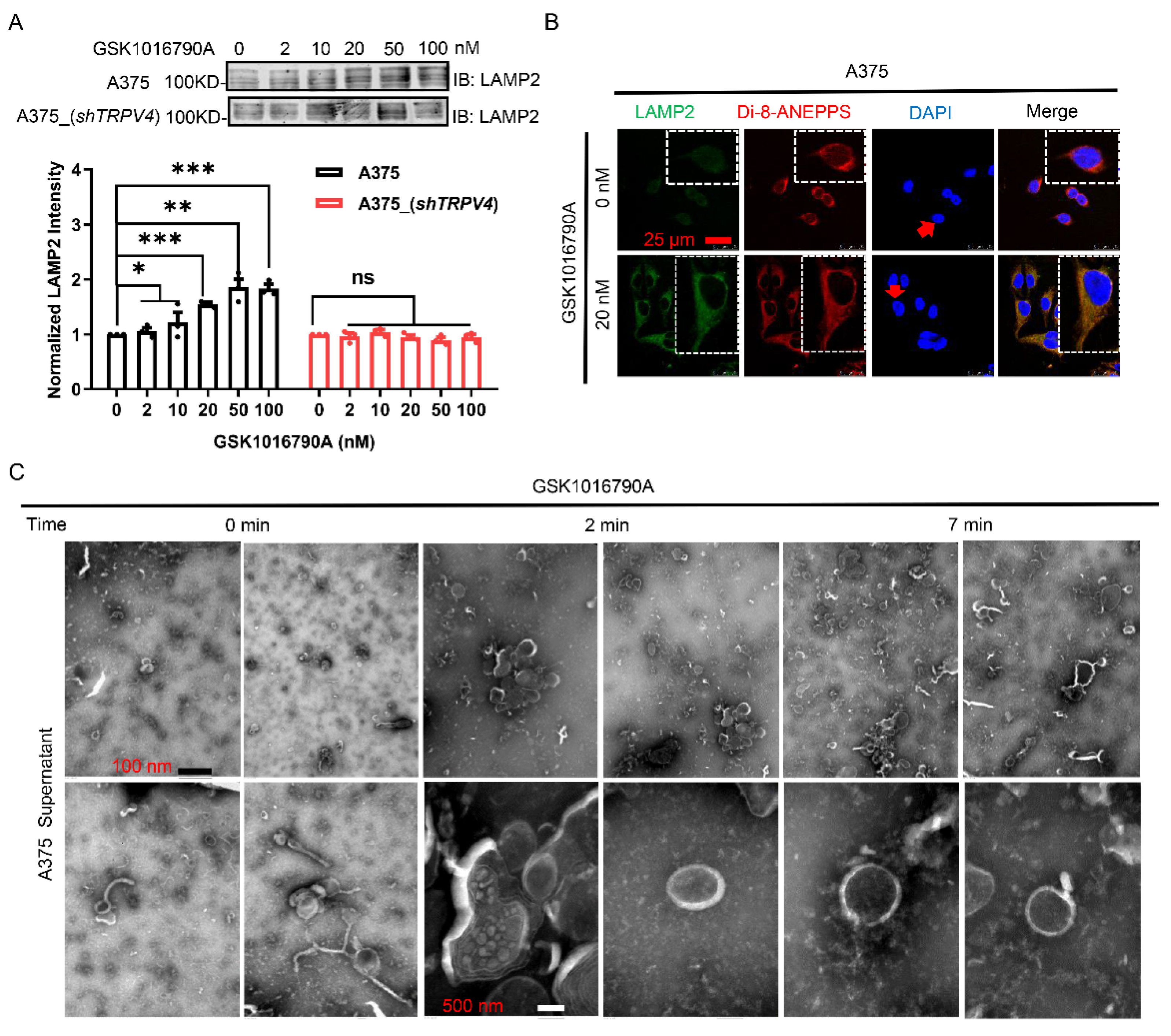
2.1. Activation of TRPV4 Induced Prominent Exocytosis in Human Melanoma A375 Cells
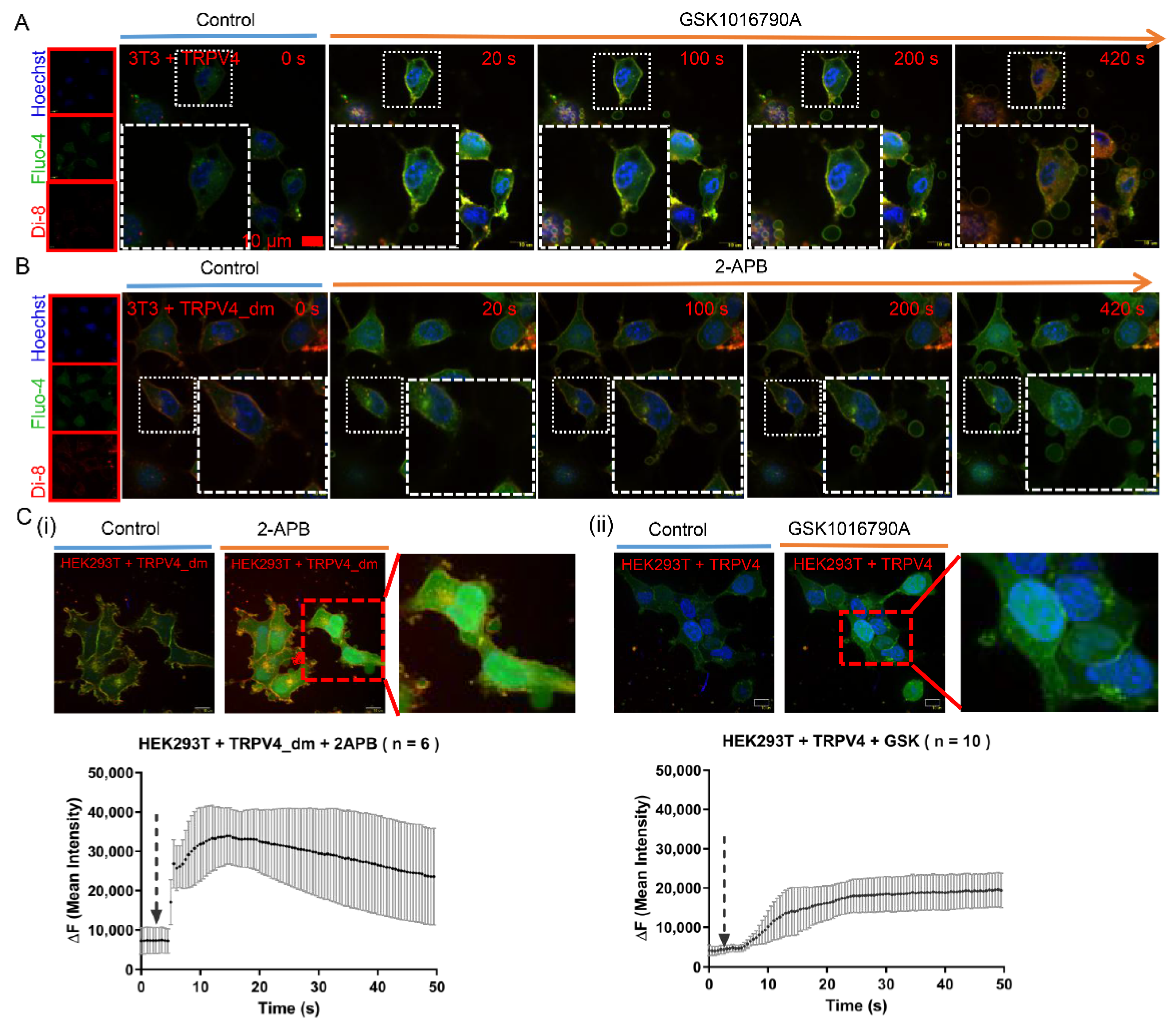
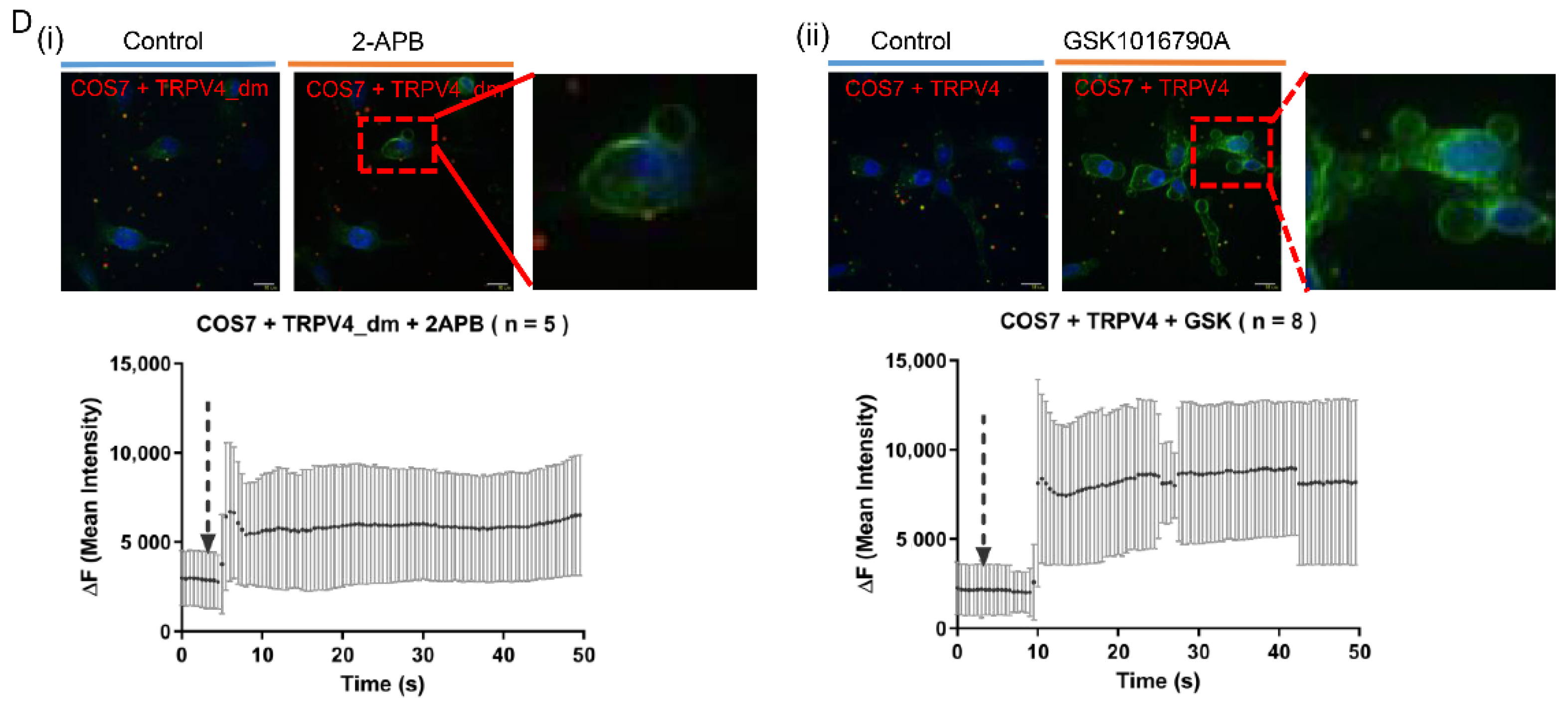
2.2. Exocytosis Is Mediated by Agonist Stimulation in Exogenous TRPV4-Expressing Cells
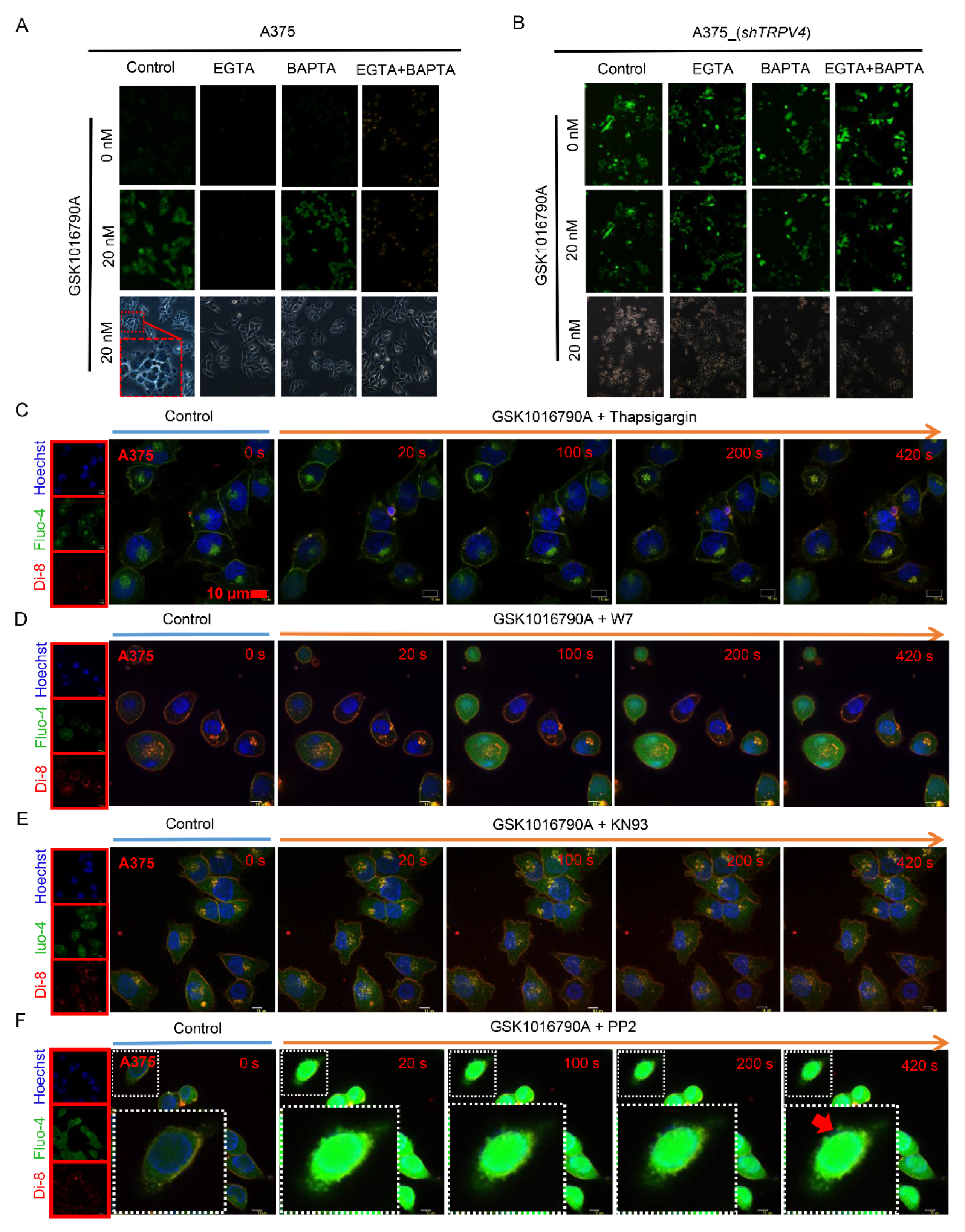
2.3. TRPV4-Mediated Exocytosis in A375 Melanoma Cells Required Ca2+/CaM/CaMKII
2.4. Accumulation of Lysosome-Associated Proteins Facilitated TRPV4-Mediated Exocytosis
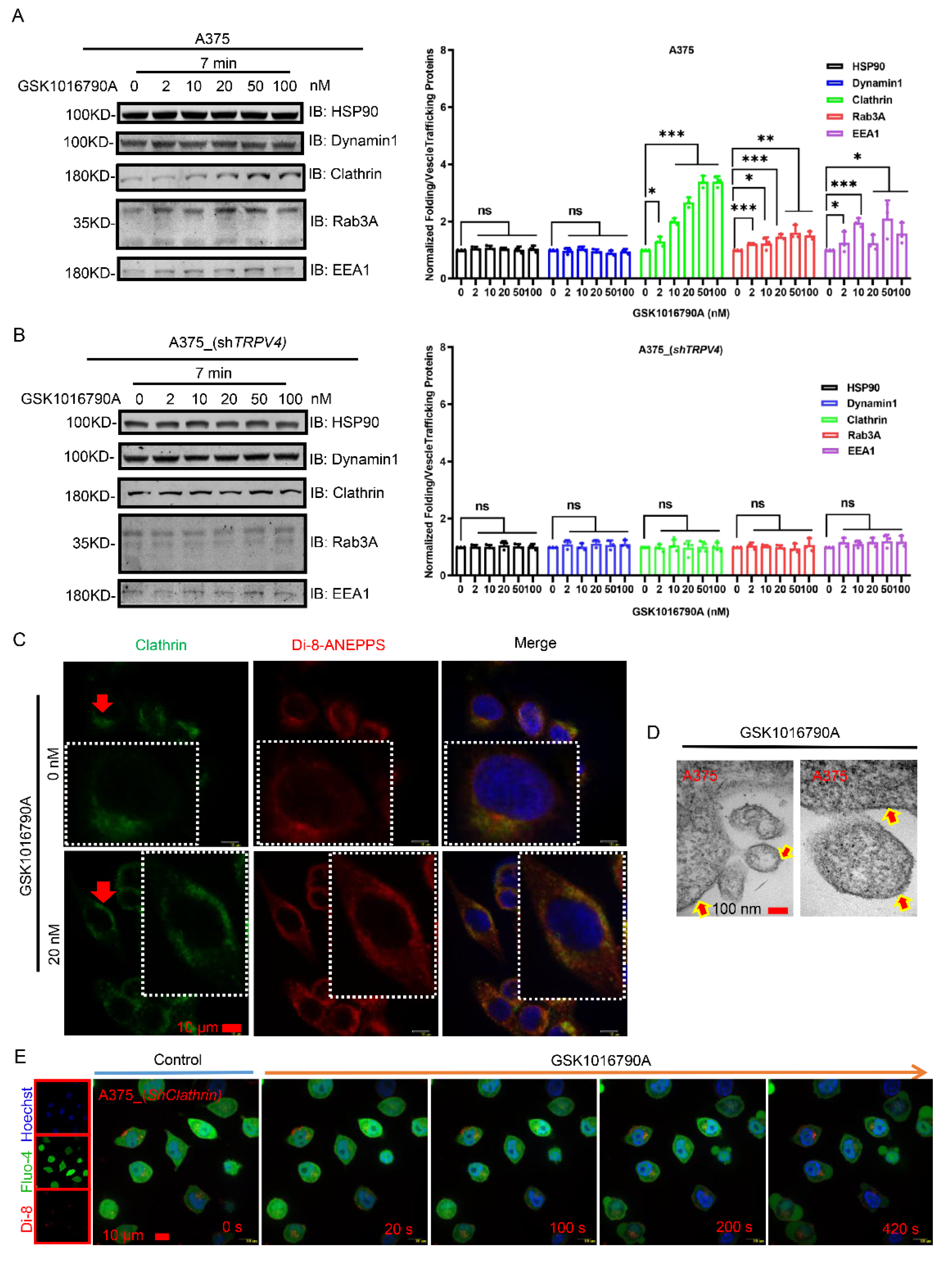
2.5. Cellular Exocytosis Response Is Aided by Recruitment of Folding and Vesicle Trafficking Proteins
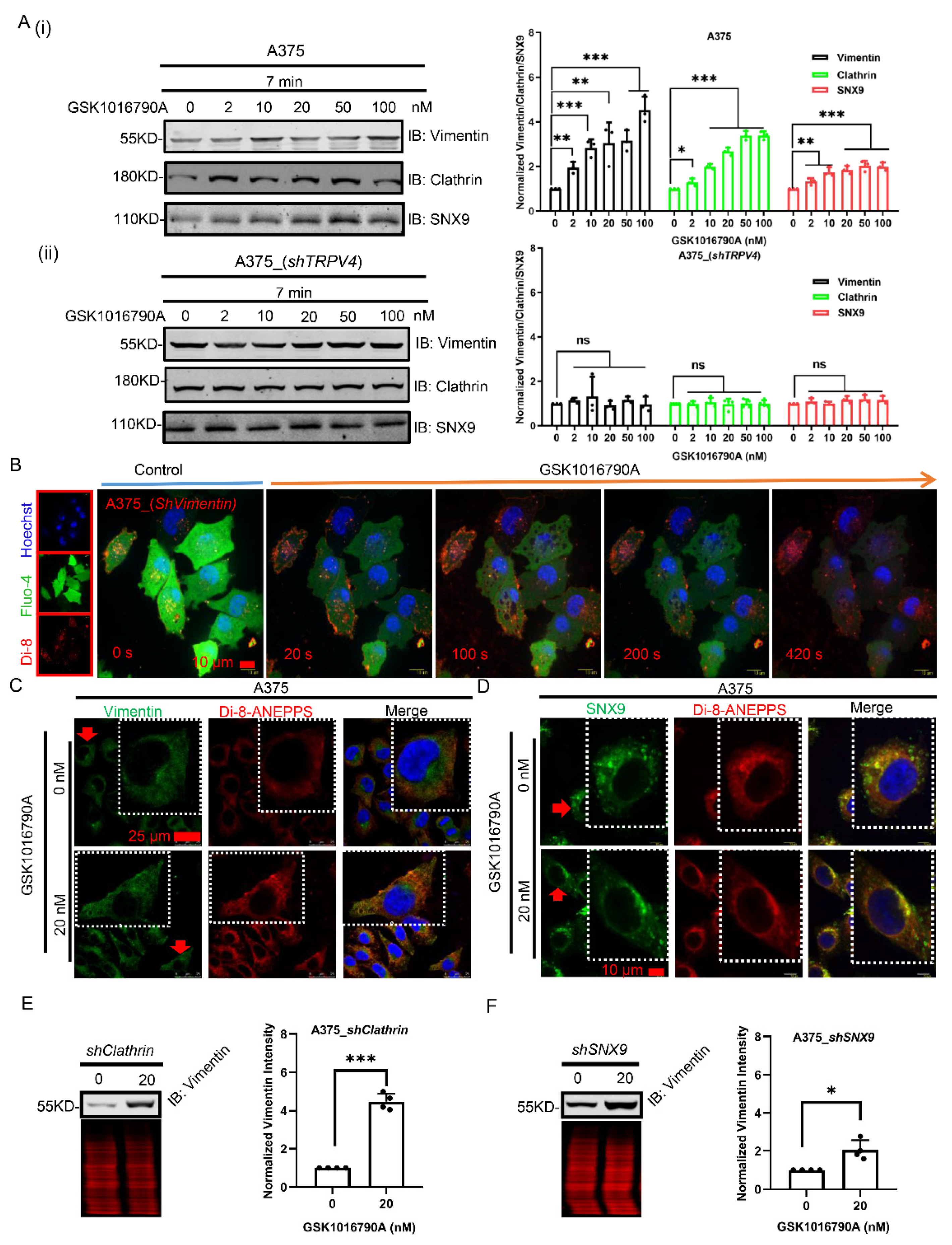
2.6. Vimentin and SNX9 Are Up-Regulated in Cellular Exocytosis after Aagonist Stimulation of TRPV4
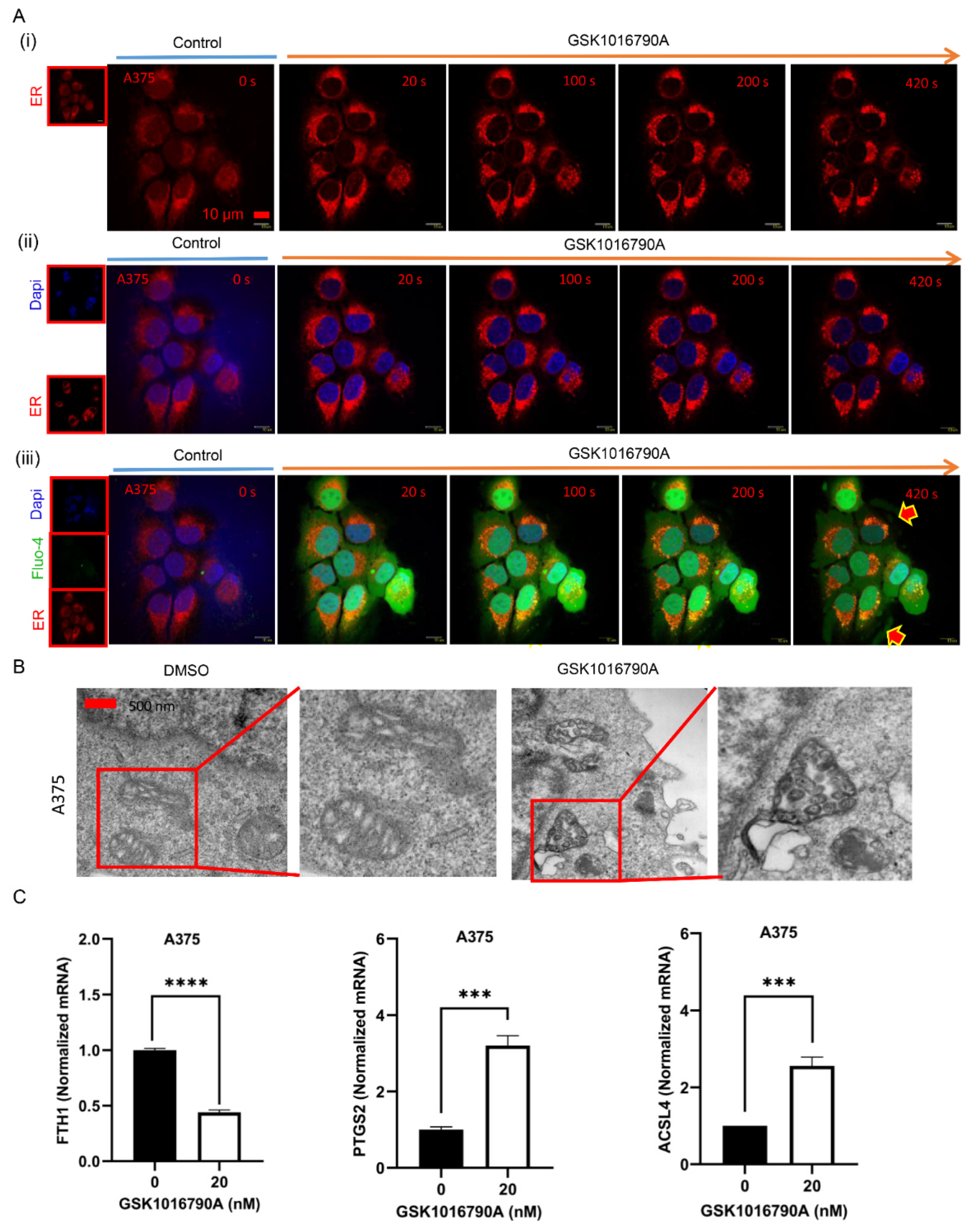
2.7. TRPV4 Mediates Exocytosis Priming from ER and May Result in Cell Ferroptosis
3. Discussion
3.1. TRPV4 Ion Channel and Calcium Signaling in Exocytosis
3.2. The Interplay among TRPV4, Folding/Trafficking Proteins, and Skeleton Proteins
3.3. TRPV4, Ferroptosis and Cellular Signalling
4. Materials and Methods
4.1. Antibodies and Chemical Regents
4.2. Cell Culture and Transfection
4.3. Western Blot
4.4. Transmission Electron Microscopy
4.5. Intracellular Calcium Measurements
4.6. Post Transcriptional Gene Silencing
4.7. Imunostaining and Imaging
4.8. Time-Lapse Microscopy
4.9. Real-Time PCR Validation
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Urry, L.A.; Cain, M.L.; Wasserman, S.A.; Minorsky, P.V.; Orr, R.B. Campbell Biology, 12th ed.; Pearson: New York, NY, USA, 2020; pp. 139–141. [Google Scholar]
- Slomiany, A.; Grabska, M.; Piotrowski, E.; Sengupta, S.; Morita, M.; Kasinathan, C.; Slomiany, B.L. Intracellular processes associated with vesicular transport from endoplasmic reticulum to Golgi and exocytosis: Ethanol-induced changes in membrane biogenesis. Arch. Biochem. Biophys. 1994, 310, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Penalva, M.A.; Moscoso-Romero, E.; Hernandez-Gonzalez, M. Tracking exocytosis of a GPI-anchored protein in Aspergillus nidulans. Traffic 2020, 21, 675–688. [Google Scholar] [CrossRef] [PubMed]
- Kagan, V.E.; Mao, G.; Qu, F.; Angeli, J.P.; Doll, S.; Croix, C.S.; Dar, H.H.; Liu, B.; Tyurin, V.A.; Ritov, V.B.; et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat. Chem. Biol. 2017, 13, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.Y.; Dixon, S.J. Mechanisms of ferroptosis. Cell. Mol. Life Sci. 2016, 73, 2195–2209. [Google Scholar] [CrossRef] [Green Version]
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: An iron-dependent form of nonapoptotic cell death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef] [Green Version]
- Yagoda, N.; von Rechenberg, M.; Zaganjor, E.; Bauer, A.J.; Yang, W.S.; Fridman, D.J.; Wolpaw, A.J.; Smukste, I.; Peltier, J.M.; Boniface, J.J.; et al. RAS-RAF-MEK-dependent oxidative cell death involving voltage-dependent anion channels. Nature 2007, 447, 864–868. [Google Scholar] [CrossRef] [Green Version]
- Simoes, F.; Ousingsawat, J.; Wanitchakool, P.; Fonseca, A.; Cabrita, I.; Benedetto, R.; Schreiber, R.; Kunzelmann, K. CFTR supports cell death through ROS-dependent activation of TMEM16F (anoctamin 6). Pflug. Arch. Eur. J. Physiol. 2018, 470, 305–314. [Google Scholar] [CrossRef]
- Guo, X.W.; Zhang, H.; Huang, J.Q.; Wang, S.N.; Lu, Y.; Cheng, B.; Dong, S.H.; Wang, Y.Y.; Li, F.S.; Li, Y.W. PIEZO1 Ion Channel Mediates Ionizing Radiation-Induced Pulmonary Endothelial Cell Ferroptosis via Ca2+/Calpain/VE-Cadherin Signaling. Front. Mol. Biosci. 2021, 8, 725274. [Google Scholar] [CrossRef]
- Ousingsawat, J.; Schreiber, R.; Kunzelmann, K. TMEM16F/Anoctamin 6 in Ferroptotic Cell Death. Cancers 2019, 11, 625. [Google Scholar] [CrossRef] [Green Version]
- Tsoi, J.; Robert, L.; Paraiso, K.; Galvan, C.; Sheu, K.M.; Lay, J.; Wong, D.J.L.; Atefi, M.; Shirazi, R.; Wang, X.; et al. Multi-stage Differentiation Defines Melanoma Subtypes with Differential Vulnerability to Drug-Induced Iron-Dependent Oxidative Stress. Cancer Cell 2018, 33, 890–904. [Google Scholar] [CrossRef] [Green Version]
- Stockwell, B.R.; Friedmann Angeli, J.P.; Bayir, H.; Bush, A.I.; Conrad, M.; Dixon, S.J.; Fulda, S.; Gascon, S.; Hatzios, S.K.; Kagan, V.E.; et al. Ferroptosis: A Regulated Cell Death Nexus Linking Metabolism, Redox Biology, and Disease. Cell 2017, 171, 273–285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, J. Molecular mechanism of TRP channels. Compr. Physiol. 2013, 3, 221–242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clapham, D.E. TRP channels as cellular sensors. Nature 2003, 426, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Zheng, J. Distribution and Assembly of TRP Ion Channels. Adv. Exp. Med. Biol. 2021, 1349, 111–138. [Google Scholar] [CrossRef]
- Chen, X.; Yang, M.; Sun, F.; Liang, C.; Wei, Y.; Wang, L.; Yue, J.; Chen, B.; Li, S.; Liu, S.; et al. Expression and cellular distribution of transient receptor potential vanilloid 4 in cortical tubers of the tuberous sclerosis complex. Brain Res. 2016, 1636, 183–192. [Google Scholar] [CrossRef]
- Roberts, M.W.G.; Sui, G.; Wu, R.; Rong, W.; Wildman, S.; Montgomery, B.; Ali, A.; Langley, S.; Ruggieri, M.R., Sr.; Wu, C. TRPV4 receptor as a functional sensory molecule in bladder urothelium: Stretch-independent, tissue-specific actions and pathological implications. FASEB J. 2020, 34, 263–286. [Google Scholar] [CrossRef] [Green Version]
- Tang, B.; Wu, J.; Zhu, M.X.; Sun, X.; Liu, J.; Xie, R.; Dong, T.X.; Xiao, Y.; Carethers, J.M.; Yang, S.; et al. VPAC1 couples with TRPV4 channel to promote calcium-dependent gastric cancer progression via a novel autocrine mechanism. Oncogene 2019, 38, 3946–3961. [Google Scholar] [CrossRef]
- Yang, P.; Lu, P.; Luo, J.; Du, L.; Feng, J.; Cai, T.; Yuan, Y.; Cheng, H.; Hu, H. Transient stimulation of TRPV4-expressing keratinocytes promotes hair follicle regeneration in mice. Br. J. Pharmacol. 2020, 177, 4181–4192. [Google Scholar] [CrossRef]
- Fu, Y.; Subramanya, A.; Rozansky, D.; Cohen, D.M. WNK kinases influence TRPV4 channel function and localization. Am. J. Physiol.-Ren. Physiol. 2006, 290, F1305–F1314. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.C.; Choe, S.Y. Age-related changes in the distribution of transient receptor potential vanilloid 4 channel (TRPV4) in the central nervous system of rats. J. Mol. Histol. 2014, 45, 497–505. [Google Scholar] [CrossRef]
- Lei, L.; Cao, X.; Yang, F.; Shi, D.J.; Tang, Y.Q.; Zheng, J.; Wang, K. A TRPV4 channel C-terminal folding recognition domain critical for trafficking and function. J. Biol. Chem. 2013, 288, 10427–10439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Fu, X.; Gaiser, S.; Kottgen, M.; Kramer-Zucker, A.; Walz, G.; Wegierski, T. OS-9 regulates the transit and polyubiquitination of TRPV4 in the endoplasmic reticulum. J. Biol. Chem. 2007, 282, 36561–36570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Y.S.; Cho, S.I.; Choi, M.G.; Choi, Y.H.; Kwak, I.S.; Park, C.W.; Kim, H.O. Increased expression of three types of transient receptor potential channels (TRPA1, TRPV4 and TRPV3) in burn scars with post-burn pruritus. Acta Derm. Venereol. 2015, 95, 20–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shukla, A.K.; Kim, J.; Ahn, S.; Xiao, K.; Shenoy, S.K.; Liedtke, W.; Lefkowitz, R.J. Arresting a transient receptor potential (TRP) channel: Beta-arrestin 1 mediates ubiquitination and functional down-regulation of TRPV4. J. Biol. Chem. 2010, 285, 30115–30125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wegierski, T.; Hill, K.; Schaefer, M.; Walz, G. The HECT ubiquitin ligase AIP4 regulates the cell surface expression of select TRP channels. EMBO J. 2006, 25, 5659–5669. [Google Scholar] [CrossRef] [Green Version]
- Cuajungco, M.P.; Grimm, C.; Oshima, K.; D’Hoedt, D.; Nilius, B.; Mensenkamp, A.R.; Bindels, R.J.; Plomann, M.; Heller, S. PACSINs bind to the TRPV4 cation channel. PACSIN 3 modulates the subcellular localization of TRPV4. J. Biol. Chem. 2006, 281, 18753–18762. [Google Scholar] [CrossRef] [Green Version]
- D’Hoedt, D.; Owsianik, G.; Prenen, J.; Cuajungco, M.P.; Grimm, C.; Heller, S.; Voets, T.; Nilius, B. Stimulus-specific modulation of the cation channel TRPV4 by PACSIN 3. J. Biol. Chem. 2008, 283, 6272–6280. [Google Scholar] [CrossRef] [Green Version]
- Zheng, J.; Liu, F.; Du, S.; Li, M.; Wu, T.; Tan, X.; Cheng, W. Mechanism for Regulation of Melanoma Cell Death via Activation of Thermo-TRPV4 and TRPV2. J. Oncol. 2019, 2019, 7362875. [Google Scholar] [CrossRef]
- Hu, H.; Grandl, J.; Bandell, M.; Petrus, M.; Patapoutian, A. Two amino acid residues determine 2-APB sensitivity of the ion channels TRPV3 and TRPV4. Proc. Natl. Acad. Sci. USA 2009, 106, 1626–1631. [Google Scholar] [CrossRef] [Green Version]
- Wu, X.S.; McNeil, B.D.; Xu, J.; Fan, J.; Xue, L.; Melicoff, E.; Adachi, R.; Bai, L.; Wu, L.G. Ca2+ and calmodulin initiate all forms of endocytosis during depolarization at a nerve terminal. Nat. Neurosci. 2009, 12, 1003–1010. [Google Scholar] [CrossRef]
- Wu, W.; Xu, J.; Wu, X.S.; Wu, L.G. Activity-dependent acceleration of endocytosis at a central synapse. J. Neurosci. Off. J. Soc. Neurosci. 2005, 25, 11676–11683. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Wu, X.S.; Xu, J.; McNeil, B.D.; Pang, Z.P.; Yang, W.; Bai, L.; Qadri, S.; Molkentin, J.D.; Yue, D.T.; et al. The role of calcium/calmodulin-activated calcineurin in rapid and slow endocytosis at central synapses. J. Neurosci. Off. J. Soc. Neurosci. 2010, 30, 11838–11847. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, S.; Yamada, K.M. Localized Lysosome Exocytosis Helps Breach Tissue Barriers. Dev. Cell 2017, 43, 377–378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vijayan, K.; Cestari, I.; Mast, F.D.; Glennon, E.K.K.; McDermott, S.M.; Kain, H.S.; Brokaw, A.M.; Aitchison, J.D.; Stuart, K.; Kaushansky, A. Plasmodium Secretion Induces Hepatocyte Lysosome Exocytosis and Promotes Parasite Entry. iScience 2019, 21, 603–611. [Google Scholar] [CrossRef] [Green Version]
- Padamsey, Z.; McGuinness, L.; Bardo, S.J.; Reinhart, M.; Tong, R.; Hedegaard, A.; Hart, M.L.; Emptage, N.J. Activity-Dependent Exocytosis of Lysosomes Regulates the Structural Plasticity of Dendritic Spines. Neuron 2017, 93, 132–146. [Google Scholar] [CrossRef] [Green Version]
- Liu, B.; Cao, W.; Li, J.; Liu, J. Lysosomal exocytosis of ATP is coupled to P2Y2 receptor in marginal cells in the stria vascular in neonatal rats. Cell Calcium 2018, 76, 62–71. [Google Scholar] [CrossRef]
- Buratta, S.; Tancini, B.; Sagini, K.; Delo, F.; Chiaradia, E.; Urbanelli, L.; Emiliani, C. Lysosomal Exocytosis, Exosome Release and Secretory Autophagy: The Autophagic- and Endo-Lysosomal Systems Go Extracellular. Int. J. Mol. Sci. 2020, 21, 2576. [Google Scholar] [CrossRef] [Green Version]
- Huynh, K.K.; Eskelinen, E.L.; Scott, C.C.; Malevanets, A.; Saftig, P.; Grinstein, S. LAMP proteins are required for fusion of lysosomes with phagosomes. EMBO J. 2007, 26, 313–324. [Google Scholar] [CrossRef]
- Manoni, M.; Tribioli, C.; Lazzari, B.; DeBellis, G.; Patrosso, C.; Pergolizzi, R.; Pellegrini, M.; Maestrini, E.; Rivella, S.; Vezzoni, P.; et al. The nucleotide sequence of a CpG island demonstrates the presence of the first exon of the gene encoding the human lysosomal membrane protein lamp2 and assigns the gene to Xq24. Genomics 1991, 9, 551–554. [Google Scholar] [CrossRef]
- Kannan, K.; Divers, S.G.; Lurie, A.A.; Chervenak, R.; Fukuda, M.; Holcombe, R.F. Cell surface expression of lysosome-associated membrane protein-2 (lamp2) and CD63 as markers of in vivo platelet activation in malignancy. Eur. J. Haematol. 1995, 55, 145–151. [Google Scholar] [CrossRef]
- MaCaulay, S.L.; Stoichevska, V.; Grusovin, J.; Gough, K.H.; Castelli, L.A.; Ward, C.W. Insulin stimulates movement of sorting nexin 9 between cellular compartments: A putative role mediating cell surface receptor expression and insulin action. Biochem. J. 2003, 376, 123–134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soulet, F.; Yarar, D.; Leonard, M.; Schmid, S.L. SNX9 regulates dynamin assembly and is required for efficient clathrin-mediated endocytosis. Mol. Biol. Cell 2005, 16, 2058–2067. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miao, Y.; Li, G.; Zhang, X.; Xu, H.; Abraham, S.N. A TRP Channel Senses Lysosome Neutralization by Pathogens to Trigger Their Expulsion. Cell 2015, 161, 1306–1319. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bohme, I.; Schonherr, R.; Eberle, J.; Bosserhoff, A.K. Membrane Transporters and Channels in Melanoma. Rev. Physiol. Biochem. Pharmacol. 2021, 181, 269–374. [Google Scholar] [CrossRef]
- Ferrera, L.; Barbieri, R.; Picco, C.; Zuccolini, P.; Remigante, A.; Bertelli, S.; Fumagalli, M.R.; Zifarelli, G.; La Porta, C.A.M.; Gavazzo, P.; et al. TRPM2 Oxidation Activates Two Distinct Potassium Channels in Melanoma Cells through Intracellular Calcium Increase. Int. J. Mol. Sci. 2021, 22, 8359. [Google Scholar] [CrossRef]
- Orlando, M.; Schmitz, D.; Rosenmund, C.; Herman, M.A. Calcium-Independent Exo-endocytosis Coupling at Small Central Synapses. Cell Rep. 2019, 29, 3767–3774. [Google Scholar] [CrossRef] [Green Version]
- Ma, H.; Macdougall, L.J.; GonzalezRodriguez, A.; Schroeder, M.E.; Batan, D.; Weiss, R.M.; Anseth, K.S. Calcium Signaling Regulates Valvular Interstitial Cell Alignment and Myofibroblast Activation in Fast-Relaxing Boronate Hydrogels. Macromol. Biosci. 2020, 20, e2000268. [Google Scholar] [CrossRef]
- Clapham, D.E. Calcium signaling. Cell 2007, 131, 1047–1058. [Google Scholar] [CrossRef] [Green Version]
- Woolums, B.M.; McCray, B.A.; Sung, H.; Tabuchi, M.; Sullivan, J.M.; Ruppell, K.T.; Yang, Y.; Mamah, C.; Aisenberg, W.H.; Saavedra-Rivera, P.C.; et al. TRPV4 disrupts mitochondrial transport and causes axonal degeneration via a CaMKII-dependent elevation of intracellular Ca2+. Nat. Commun. 2020, 11, 2679. [Google Scholar] [CrossRef]
- Gilchrist, C.L.; Leddy, H.A.; Kaye, L.; Case, N.D.; Rothenberg, K.E.; Little, D.; Liedtke, W.; Hoffman, B.D.; Guilak, F. TRPV4-mediated calcium signaling in mesenchymal stem cells regulates aligned collagen matrix formation and vinculin tension. Proc. Natl. Acad. Sci. USA 2019, 116, 1992–1997. [Google Scholar] [CrossRef] [Green Version]
- Donate-Macian, P.; Jungfleisch, J.; Perez-Vilaro, G.; Rubio-Moscardo, F.; Peralvarez-Marin, A.; Diez, J.; Valverde, M.A. The TRPV4 channel links calcium influx to DDX3X activity and viral infectivity. Nat. Commun. 2018, 9, 2307. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Lu, K.; Yang, S.; Wu, Y.; Liao, J.; Lu, Y.; Wu, Q.; Zhao, N.; Dong, Q.; Chen, L.; et al. Activation of transient receptor potential vanilloid 4 exacerbates myocardial ischemia-reperfusion injury via JNK-CaMKII phosphorylation pathway in isolated mice hearts. Cell Calcium 2021, 100, 102483. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Kleiner, S.; Wu, J.; Sah, R.; Gupta, R.K.; Banks, A.S.; Cohen, P.; Khandekar, M.J.; Bostrom, P.; Mepani, R.J.; et al. TRPV4 is a regulator of adipose oxidative metabolism, inflammation, and energy homeostasis. Cell 2012, 151, 96–110. [Google Scholar] [CrossRef] [Green Version]
- Scheraga, R.G.; Abraham, S.; Grove, L.M.; Southern, B.D.; Crish, J.F.; Perelas, A.; McDonald, C.; Asosingh, K.; Hasday, J.D.; Olman, M.A. TRPV4 Protects the Lung from Bacterial Pneumonia via MAPK Molecular Pathway Switching. J. Immunol. 2020, 204, 1310–1321. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Li, X.; Zhang, X.; Kang, R.; Tang, D. Identification of ACSL4 as a biomarker and contributor of ferroptosis. Biochem. Biophys. Res. Commun. 2016, 478, 1338–1343. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.S.; SriRamaratnam, R.; Welsch, M.E.; Shimada, K.; Skouta, R.; Viswanathan, V.S.; Cheah, J.H.; Clemons, P.A.; Shamji, A.F.; Clish, C.B.; et al. Regulation of ferroptotic cancer cell death by GPX4. Cell 2014, 156, 317–331. [Google Scholar] [CrossRef] [Green Version]
- Earley, S.; Pauyo, T.; Drapp, R.; Tavares, M.J.; Liedtke, W.; Brayden, J.E. TRPV4-dependent dilation of peripheral resistance arteries influences arterial pressure. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H1096–H1102. [Google Scholar] [CrossRef] [Green Version]
- Fiorio Pla, A.; Ong, H.L.; Cheng, K.T.; Brossa, A.; Bussolati, B.; Lockwich, T.; Paria, B.; Munaron, L.; Ambudkar, I.S. TRPV4 mediates tumor-derived endothelial cell migration via arachidonic acid-activated actin remodeling. Oncogene 2012, 31, 200–212. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.S.; Shin, D.H.; Nam, J.H.; Park, K.S.; Zhang, Y.H.; Kim, W.K.; Kim, S.J. Functional Expression of TRPV4 Cation Channels in Human Mast Cell Line (HMC-1). Korean J. Physiol. Pharmacol. 2010, 14, 419–425. [Google Scholar] [CrossRef] [Green Version]
- Shibasaki, K. TRPV4 ion channel as important cell sensors. J. Anesth. 2016, 30, 1014–1019. [Google Scholar] [CrossRef]

| Gene | Sense Strand | Entrez Gene ID |
|---|---|---|
| TRPV4 | 5′-ACCAAGTTTGTTACCAAGA-3′ | 59341 |
| Clathrin | 5′-GTGCTCTATTTATATAGAA-3′ | 1213 |
| Vimentin | 5′-ACACTCAGTGCAGCAATAT-3′ | 7431 |
| SNX9 | 5′-CTAACACCTACTAACACTA-3′ | 51429 |
| Gene | Forward Primer | Reverse Primer | NM-Number |
|---|---|---|---|
| FTH1 | AGAACTACCACCAGGACTCAGA | CAAAGCCCACATCATCGCGG | NM-002032.3 |
| ACSL4 | TTTTGCGAGCTTTCCGAGTG | AGCCGACAATAAAGTACGCC | NM-022977.3 |
| PTGS2 | CTGATGATTGCCCGACTCCC | CGCAGTTTACGCTGTCTAGC | NM-000963.4 |
| GPX4 | TGGACGAGGGGAGGAGC | TCGATGTCCTTGGCGGAAAA | NM-002085.5 |
| NOX1 | TAAAGGCTCACAGACCCTGC | GGCCAATGTTGACCCAAGGA | NM-007052.5 |
| β-actin | TGGCATCCACGAAACTACCTT | TCGTCATACTCCTGCTTGCTG | NM-001101.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, M.; Zheng, J.; Wu, T.; He, Y.; Guo, J.; Xu, J.; Gao, C.; Qu, S.; Zhang, Q.; Zhao, J.; et al. Activation of TRPV4 Induces Exocytosis and Ferroptosis in Human Melanoma Cells. Int. J. Mol. Sci. 2022, 23, 4146. https://doi.org/10.3390/ijms23084146
Li M, Zheng J, Wu T, He Y, Guo J, Xu J, Gao C, Qu S, Zhang Q, Zhao J, et al. Activation of TRPV4 Induces Exocytosis and Ferroptosis in Human Melanoma Cells. International Journal of Molecular Sciences. 2022; 23(8):4146. https://doi.org/10.3390/ijms23084146
Chicago/Turabian StyleLi, Mei, Jiaojiao Zheng, Tian Wu, Yulin He, Jing Guo, Jiao Xu, Chuanzhou Gao, Shuxian Qu, Qianyi Zhang, Jiayu Zhao, and et al. 2022. "Activation of TRPV4 Induces Exocytosis and Ferroptosis in Human Melanoma Cells" International Journal of Molecular Sciences 23, no. 8: 4146. https://doi.org/10.3390/ijms23084146
APA StyleLi, M., Zheng, J., Wu, T., He, Y., Guo, J., Xu, J., Gao, C., Qu, S., Zhang, Q., Zhao, J., & Cheng, W. (2022). Activation of TRPV4 Induces Exocytosis and Ferroptosis in Human Melanoma Cells. International Journal of Molecular Sciences, 23(8), 4146. https://doi.org/10.3390/ijms23084146






