Myokines and Resistance Training: A Narrative Review
Abstract
1. Introduction
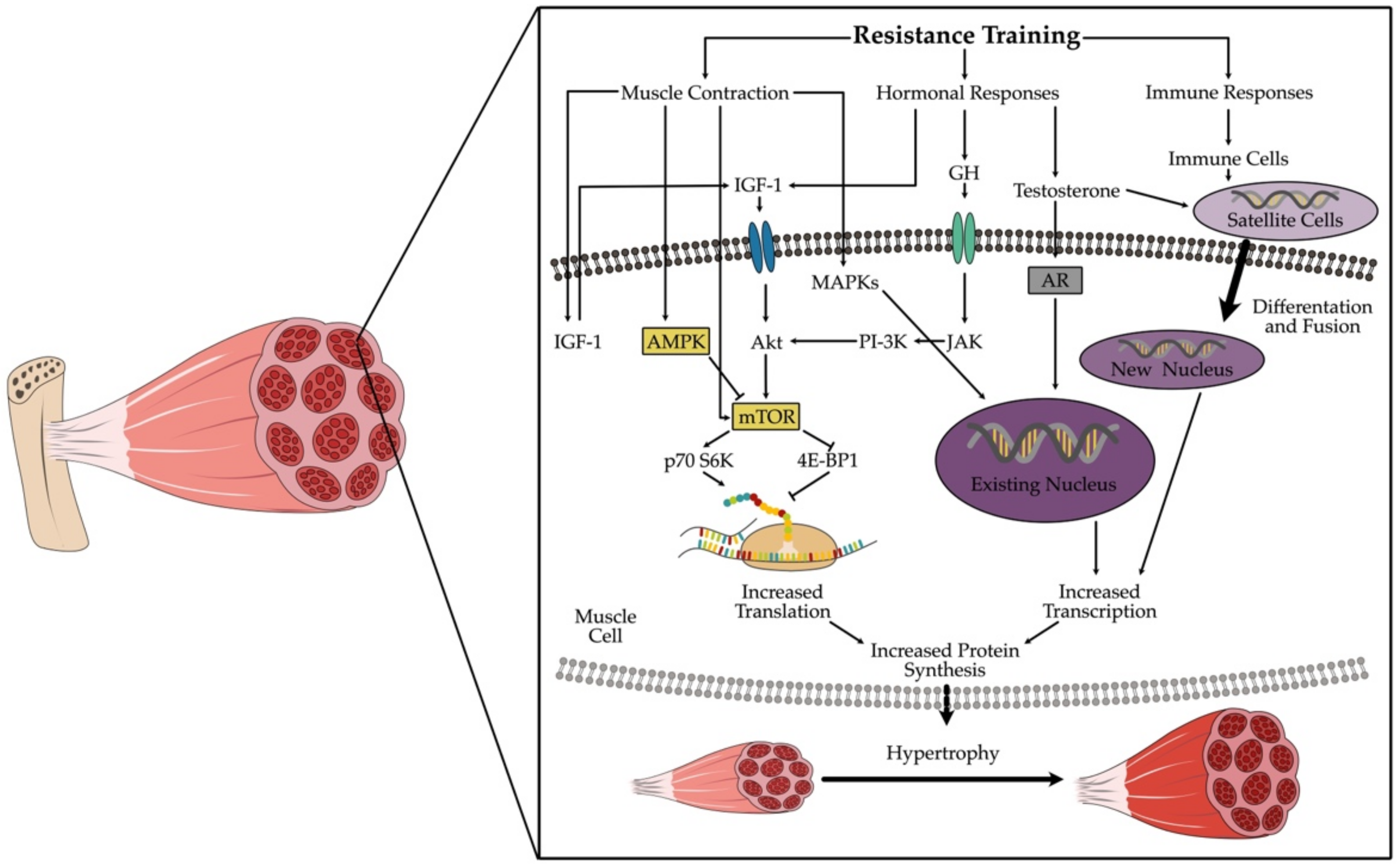
2. The Healthy Muscle Cell and Its Reaction to Resistance Training
2.1. Mechanical Tension
2.2. Muscle Damage
2.3. Metabolic Stress
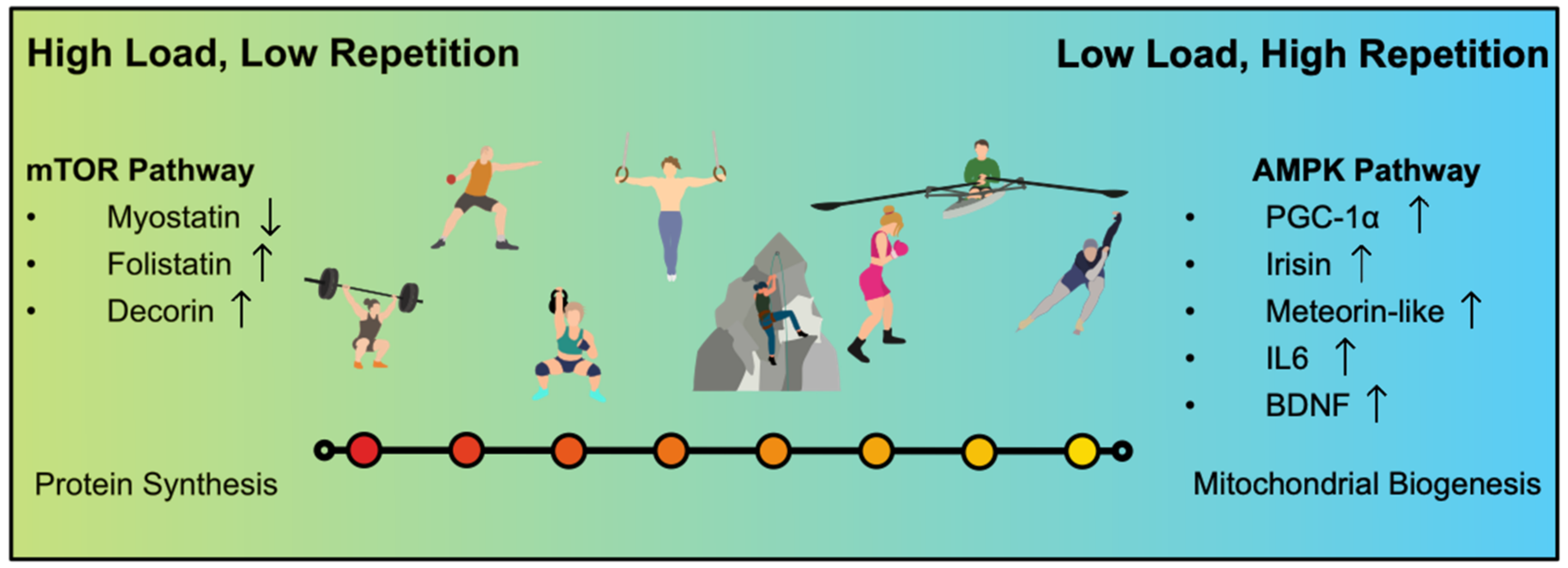
3. Different Types of Resistance Training
3.1. Hypertrophy Training
3.2. Strength Endurance Training
4. Myokines
4.1. IL-6
4.2. Myostatin Group
4.2.1. Myostatin
4.2.2. Decorin and Follistatin
4.3. BDNF
4.4. The PGC-1 Alpha Group
4.4.1. Irisin
4.4.2. Meteorin-Like
5. Myokines and Resistance Exercise
5.1. IL-6
5.2. Myostatin
5.3. Decorin
5.4. Follistatin
5.5. Irisin
5.6. BDNF
| Myokine | Study | n | Study Cohort | Training Protocol | Trial | Results |
|---|---|---|---|---|---|---|
| IL-6 | Buford et al. [145] | 24 | Physically active postmenopausal women | 3 × (10 × 80% 1 RM) | 1 session | Muscle biopsies: IL-6-mRNA ↑ |
| Della Gatta et al. [147] | 16 | Young men n = 8; elderly men n = 8 | 2 × (8–12 × 50–80% 1 RM), progressive increase | 12 weeks | Muscle biopsies: IL-6 ↑; not significant different between the groups | |
| Mendham et al. [44] | 12 | Sedentary men | 3 × (10 × 60% 1 RM) vs. 3 × (10 × 80% 1 RM) vs. Low intensity aerobic exercise (40 min) vs. Moderate intensity aerobic exercise (40 min) | 4 sessions, randomized cross-over | Serum- IL-6 ↑ in the moderate intensity groups (RE and AE) | |
| Phillips et al. [146] | 14 | Healthy men | 3 × (12 × 65% 1 RM) vs. 3 × (8 × 85% 1 RM) | 2 sessions, controlled | Serum-IL-6 ↑, with association to total volume load | |
| Quiles et al. [148] | 15 | RE-experienced men | 4–5 × (8–12 × 60–70% 1 RM) vs. 8–10 × (2–6 × 75–85% 1 RM) | 3 weekly, 6 weeks, randomized | Plasma-IL-6 ↑ in both groups; no significant effect on BDNF | |
| Tomeleri et al. [149] | 38 | Obese older women | 3 × (10–15 maximum repetitions) | 3 × weekly, 8 weeks, randomized controlled | Baseline-Serum-IL-6 ↓ | |
| Myostatin | Kazemi et al. [150] | 24 | Healthy men | 3 × (15 × 55% 1 RM) | 1 session | Plasma-Myostatin ↓ |
| Raue et al. [151] | 14 | Young women n = 8 Old women n = 6 | 3 × (10 × 70% 1 RM) | 1 session | Muscle biopsies: Myostatin mRNA ↓ | |
| Decorin | Bugera et al. [152] | 10 | Physically active young men | 4 × (7 × 80% 1 RM) vs. 4 × (15–30 × 30% 1 RM) vs. BFR 3 × (15–30 × 30% 1 RM) | 1 session | Plasma-Decorin ↑ |
| Kanzleiter et al. [153] | 10 | Young men | 3 × (8 × max weight) | Human study I, single bout | Plasma-Decorin ↑ | |
| Follistatin | Bagheri et al. [154] | 40 | Middle aged men | 3–4 × (15 > 12 > 10 > 8 × 50–80% 1 RM) Upper body vs. lower body vs. both vs. control | 3 × weekly, 8 weeks, 10% increase every 2 weeks, randomized controlled | Serum-Follistatin ↑, serum-Myostatin ↓, depending on the volume of activated muscle mass |
| Hofmann et al. [157] | 91 | Elderly women | 1–2 × 15, elastic bands vs. Training + nutritional supplements vs. Cognitive training | Progressive increase of resistance; 6 months; randomized | Serum-Follistatin ↑ only in the training group | |
| Negaresh et al. [155] | 31 | Elderly men n = 15 Young men n = 16 | 4 × (10 × 50%−85% 1 RM) | 3 × weekly, 8 weeks, 5% increase per week | Plasma-Follistatin ↑; plasma-Myostatin ↓ in both groups | |
| Irisin | Blizzard Leblanc et al. [9] | 11 | Obese youth | 4 × (12–15 × 60–65% 1 RM) | 3 × weekly, 6 weeks | Plasma-Irisin → after a single bout of RE, but ↑ after a single bout of AE; with greater ↑↑ after 6 weeks of RE |
| Ellefsen et al. [158] | 18 | Untrained women | Progressive full body heavy strength | 3 × weekly, 12 weeks | Serum-Irisin → | |
| Huh et al. [117] | 20 | Sedentary healthy men n = 14 Men with metabolic syndrome n = 6 | 3 × (8–12 × 75–80% 1 RM) vs. HIIT (4 ×4 min 90% Vo2 max) vs. Continuous moderate exercise (36 min 65% Vo2 max) | 1 session, randomized cross-over | Serum-Irisin ↑ immediately and ↓ after 1 h back to baseline; no difference between the groups | |
| Kim et al. [159] | 28 | Obese adults | 3 × (10–12 × 65–80% 1 RM) vs. Aerobic exercise (50 min 65–80% HR max) | 5 × weekly, 8 weeks, randomized controlled | Plasma-Irisin ↑ only in the RE group | |
| Norheim et al. [160] | 26 | Healthy physically inactive men n = 13 Pre-diabetic men n = 13 | Combined strength and endurance training, 2 × weekly 60 min ergometer and 2 × weekly 60 min full body strength workout | 4 × weekly, 12 weeks | Plasma-Irisin higher in the prediabetic group; acute ↑ after exercise; baseline ↓ after 12 weeks | |
| Nygaard et al. [161] | 9 | Moderately trained, healthy adults | 3 × (10–12 × max weight) vs. 6 × 5 min high intensity treadmill, Borg > 18 | 1 session, randomized cross-over | Plasma-Irisin ↑ in both groups, but remained higher in the RE group | |
| Pekkala et al. [162] | 56 | Untrained healthy men | Aerobic exercise (60 min 50% Vo2 max) vs. 5 × 10 repetitions until failure (leg press only) vs. Long term endurance exercise vs. Long term RE and endurance exercise | Single bout vs. 2 × weekly, 21 weeks | Serum-Irisin → | |
| Tibana et al. [163] | 49 | Inactive women: Obese n = 26 Non obese n = 23 | 3 × (6–12 × repetitions maximum) | 2 × weekly, 16 weeks | Plasma-Irisin ↓ in the non-obese group after intervention; no change in the obese group | |
| Tsuchiya et al. [116] | 10 | Healthy men | 3–4 × (12 ×65% 1 RM) vs. Aerobic exercise (60 min 65% Vo2 max) vs. 30 min RE + 30 min AE | 3 single bouts of exercise; randomized cross-over | Plasma-Irisin ↑, significant higher in the RE group | |
| Zhao et al. [164] | 17 | Older male adults | Class of leg muscle strength and core strength training | 2 × weekly, 12 weeks, randomized controlled | Serum-Irisin ↑ | |
| BDNF | Church et al. [101] | 20 | RE-experienced young men | 4 × (10–12 × 70% 1 RM) vs. 4 × (3–5 × 90% 1 RM) | 4 × weekly, 7 weeks | Plasma-BDNF ↑ |
| Domínguez-Sanchéz et al. [166] | 51 | Physically inactive, obese men | (12–15 × 50–70% 1 RM) vs. HIIT vs. HIIT + RE | 1 session, randomized controlled | Plasma-BDNF ↑, highest ↑↑ in the combined group | |
| Figueiredo et al. [89] | 21 | Physically active men | 1 min 100%VO2 max + 8 exercises 8–12 RM | 8 weeks, control group | Plasma-BDNF ↑ | |
| Forti et al. [167] | 65 | Healthy elderly | 2 × (10–15 × 80% 1 RM) vs. 1 × (80–100 × 20% 1 RM) vs. 1 × (60 × 20% 1 RM) + 1 × (10–20 × 40% 1 RM) | 3 × weekly, 12 weeks, randomized | Serum-BDNF ↑ in male participants, only in the 3rd group | |
| Lodo et al. [168] | 20 | Young healthy adolescents | 4 × (5 × 70% 1 RM) vs. 4 × (10 × 35% 1 RM) | 2 bouts of exercise with equated total load lifted | Serum-BDNF → | |
| Jørgensen et al. [169] | 30 | Persons with multiple sclerosis | Progressive high intensity | 2 × weekly, 24 weeks, randomized controlled | Plasma-BDNF → | |
| Marston et al. [171] | 45 | Healthy adults | 5 × (5 × 85% 1 RM) vs. 3 × (10 × 70% 1 RM) vs. | 2 × weekly, 12 weeks, randomized controlled | Serum-BDNF (↑) only in the high load group | |
| Marston et al. [170] | 16 | Untrained men n = 11 Untrained women n = 5 | 5 × 5 repetitions to-fatigue vs. 3 × 10 repetitions to fatigue | 2 bouts of exercise; cross-over | Greater serum-BDNF ↑ in the second group | |
| McKay et al. [90] | 29 | Male adolescents | 300 × maximal eccentric contractions | Single bout | Muscle-biopsy: BDNF ↑ | |
| Roh et al. [172] | 26 | Elderly, obese women | Elastic bands, intensity:10–14 RPE | 3 × weekly, 12 weeks, randomized controlled | Serum-BDNF ↑ | |
| Rojas Vega et al. [174] | 11 | Healthy adults | 3 repetitions of maximal effort isokinetic work (knee extension): 40% 1 RM vs. 110% 1 RM | 2 single bouts | Serum-BDNF (↑), but no significance | |
| Urzi et al. [173] | 20 | Elderly women | Elastic band resistance training | 12 weeks, randomized controlled | Plasma-BDNF ↑ | |
| Walsh et al. [175] Alberga et al. [177,178] | 202 | Postpubertal adolescent with obesity | HEARTY-Trial: 2–3 × (6–15 × maximum reps) vs. 20–40 min 70–85% HRmax vs. combination | 4 × weekly, 22 weeks, randomized controlled | Baseline plasma-BDNF ↑ | |
| Walsh et al. [179] | 10 | Older adults | 4 × (8–10 × 60–80% 1 RM) | 3 × weekly, 8 weeks | Serum -BDNF ↑ | |
| Wens et al. [180] | 41 | Persons with multiple sclerosis n = 22 Healthy persons n = 19 | 1 × (10 × 12–14 RPE) increased to 4 × (15 x12–14 RPE) | 5 sessions per 2 weeks, 24 weeks, randomized controlled | Baseline serum-BDNF were lower in persons with MS; Serum-BDNF ↑ after exercise in both groups | |
| Yarrow et al. [100] | 20 | Healthy young adults | 4 × (6 × 52,5% 1 RM) trad. vs. 4 × (6 × 40% 1 RM) concentric vs. 3 × (6 × 100% 1 RM) eccentric | 5 weeks | Serum BDNF ↑, with higher response at the end of intervention. No baseline change |
6. Discussion—Myokines and Resistance Training
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mero, A.A.; Hulmi, J.J.; Salmijärvi, H.; Katajavuori, M.; Haverinen, M.; Holviala, J.; Ridanpää, T.; Häkkinen, K.; Kovanen, V.; Ahtiainen, J.P.; et al. Resistance Training Induced Increase in Muscle Fiber Size in Young and Older Men. Eur. J. Appl. Physiol. 2013, 113, 641–650. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Åkerström, T.C.A.; Nielsen, A.R.; Fischer, C.P. Role of Myokines in Exercise and Metabolism. J. Appl. Physiol. 2007, 103, 1093–1098. [Google Scholar] [CrossRef] [PubMed]
- Delezie, J.; Handschin, C. Endocrine Crosstalk between Skeletal Muscle and the Brain. Front. Neurol. 2018, 9, 698. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K. Physical Activity and Muscle-Brain Crosstalk. Nat. Rev. Endocrinol. 2019, 15, 383–392. [Google Scholar] [CrossRef]
- Pedersen, B.K. Anti-Inflammatory Effects of Exercise: Role in Diabetes and Cardiovascular Disease. Eur. J. Clin. Investig. 2017, 47, 600–611. [Google Scholar] [CrossRef]
- Karstoft, K.; Pedersen, B.K. Exercise and Type 2 Diabetes: Focus on Metabolism and Inflammation. Immunol. Cell Biol. 2016, 94, 146–150. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscles, Exercise and Obesity: Skeletal Muscle as a Secretory Organ. Nat. Rev. Endocrinol. 2012, 8, 457–465. [Google Scholar] [CrossRef]
- Bay, M.L.; Pedersen, B.K. Muscle-Organ Crosstalk: Focus on Immunometabolism. Front. Physiol. 2020, 11, 567881. [Google Scholar] [CrossRef]
- Leblanc, D.R.B.; Rioux, B.V.; Pelech, C.; Moffatt, T.L.; Kimber, D.E.; Duhamel, T.A.; Dolinsky, V.W.; McGavock, J.M.; Senechal, M. Exercise-Induced Irisin Release as a Determinant of the Metabolic Response to Exercise Training in Obese Youth: The Exit Trial. Physiol. Rep. 2017, 5, e13539. [Google Scholar] [CrossRef]
- Knudsen, S.H.; Pedersen, B.K. Targeting Inflammation Through a Physical Active Lifestyle and Pharmaceuticals for the Treatment of Type 2 Diabetes. Curr. Diabetes Rep. 2015, 15, 82. [Google Scholar] [CrossRef]
- Severinsen, M.C.K.; Pedersen, B.K. Muscle–Organ Crosstalk: The Emerging Roles of Myokines. Endocr. Rev. 2020, 41, 594–609. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, B.J. The Mechanisms of Muscle Hypertrophy and Their Application to Resistance Training. J. Strength Cond. Res. 2010, 24, 2857–2872. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.M.; Hoffman, J.R.; Stout, J.R.; Fukuda, D.H.; Willoughby, D.S. Intramuscular Anabolic Signaling and Endocrine Response Following Resistance Exercise: Implications for Muscle Hypertrophy. Sports Med. 2016, 2016 46, 671–685. [Google Scholar] [CrossRef]
- Bamman, M.M.; Roberts, B.M.; Adams, G.R. Molecular Regulation of Exercise-Induced Muscle Fiber Hypertrophy. Cold Spring Harb. Perspect. Med. 2018, 8, a029751. [Google Scholar] [CrossRef] [PubMed]
- Wackerhage, H.; Ratkevicius, A. Signal Transduction Pathways That Regulate Muscle Growth. Essays Biochem. 2008, 44, 99–108. [Google Scholar] [CrossRef]
- Hornberger, T.A. Mechanotransduction and the Regulation of MTORC1 Signaling in Skeletal Muscle. Int. J. Biochem. Cell Biol. 2011, 43, 1267–1276. [Google Scholar] [CrossRef]
- Toigo, M. Trainingsrelevante Determinanten Der Molekularen Und Zellul??Ren Skelettmuskeladaptation—Teil 2: Adaptation von Querschnitt Und Fasertypusmodulen. Schweiz. Z. Sportmed. Sporttraumatol. 2006, 54, 121–132. [Google Scholar]
- Paulsen, G.; Mikkelsen, U.R.; Raastad, T.; Peake, J.M. Leucocytes, Cytokines and Satellite Cells: What Role Do They Play in Muscle Damage and Regeneration Following Eccentric Exercise? Exerc. Immunol. Rev. 2012, 18, 42–97. [Google Scholar]
- Dumont, N.A.; Wang, Y.X.; Rudnicki, M.A. Intrinsic and Extrinsic Mechanisms Regulating Satellite Cell Function. Development 2015, 142, 1572–1581. [Google Scholar] [CrossRef]
- Damas, F.; Libardi, C.A.; Ugrinowitsch, C. The Development of Skeletal Muscle Hypertrophy through Resistance Training: The Role of Muscle Damage and Muscle Protein Synthesis. Eur. J. Appl. Physiol. 2018, 118, 485–500. [Google Scholar] [CrossRef]
- Toigo, M. MuskelRevolution; Springer: Berlin/Heidelberg, Germany, 2019; ISBN 9783662547649. [Google Scholar]
- Di, W.; Lv, J.; Jiang, S.; Lu, C.; Yang, Z.; Ma, Z.; Hu, W.; Yang, Y.; Xu, B. PGC-1: The Energetic Regulator in Cardiac Metabolism. Curr. Issues Mol. Biol. 2018, 28, 29–46. [Google Scholar] [CrossRef]
- Hargreaves, M.; Spriet, L.L. Exercise Metabolism: Fuels for the Fire. Cold Spring Harb. Perspect. Med. 2018, 8, a029744. [Google Scholar] [CrossRef] [PubMed]
- Rigoulet, M.; Bouchez, C.L.; Paumard, P.; Ransac, S.; Cuvellier, S.; Duvezin-Caubet, S.; Mazat, J.P.; Devin, A. Cell Energy Metabolism: An Update. Biochim. Biophys. Acta. Bioenerg. 2020, 1861, 148276. [Google Scholar] [CrossRef] [PubMed]
- Hoppeler, H.; Baum, O.; Mueller, M.; Lurman, G. Molekulare Mechanismen Der Anpassungsfähigkeit Der Skelettmuskulatur. Schweiz. Z. Sportmed. Sporttraumatol. 2011, 59, 6–13. [Google Scholar]
- Kraemer, W.J.; Deschenes, M.R.; Fleck, S.J. Physiological Adaptations to Resistance Exercise. Implications for Athletic Conditioning. Sports Med. 1988, 6, 246–256. [Google Scholar] [CrossRef]
- American College of Sports Medicine. American College of Sports Medicine Position Stand. Progression Models in Resistance Training for Healthy Adults. Med. Sci. Sports Exerc. 2009, 41, 687–708. [Google Scholar] [CrossRef]
- Howe, L.P.; Read, P.; Waldron, M. Muscle Hypertrophy: A Narrative Review on Training Principles for Increasing Muscle Mass. Strength Cond. J. 2017, 39, 72–81. [Google Scholar] [CrossRef]
- Fisher, J. A Critical Commentary on the Practical Application of Resistance Training Studies. J. Trainol. 2013, 2, 10–12. [Google Scholar] [CrossRef]
- Carpinelli, R.N. Critical Commentary on the Stimulus for Muscle Hypertrophy in Experienced Trainees. Med. Sport. Pract. 2020, 21, 1–37. [Google Scholar]
- Kraemer, W.J.; Adams, K.; Cafarelli, E.; Dudley, G.A.; Dooly, C.; Feigenbaum, M.S.; Fleck, S.J.; Franklin, B.; Fry, A.C.; Hoffman, J.R.; et al. American College of Sports Medicine Position Stand. Progression Models in Resistance Training for Healthy Adults. Med. Sci. Sports Exerc. 2002, 34, 364–380. [Google Scholar] [CrossRef]
- Anderson, T.; Kearney, J.T. Effects of Three Resistance Training Programs on Muscular Strength And Absolute and Relative Endurance. Res. Q. Exerc. Sport 1982, 53, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Campos, G.; Luecke, T.; Wendeln, H.; Toma, K.; Hagerman, F.; Murray, T.; Ragg, K.; Ratamess, N.; Kraemer, W.; Staron, R. Muscular Adaptations in Response to Three Different Resistance-Training Regimens: Specificity of Repetition Maximum Training Zones. Eur. J. Appl. Physiol. 2002, 88, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, W.J.; Patton, J.F.; Gordon, S.E.; Harman, E.A.; Deschenes, M.R.; Reynolds, K.; Newton, R.U.; Triplett, N.T.; Dziados, J.E. Compatibility of High-Intensity Strength and Endurance Training on Hormonal and Skeletal Muscle Adaptations. J. Appl. Physiol. 1995, 78, 976–989. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.U.; Ghafoor, S. Myokines: Discovery Challenges and Therapeutic Impediments. J. Pak. Med. Assoc. 2019, 69, 1014–1017. [Google Scholar]
- Agarwal, M.; Singh, S.; Narayan, J.; Pandey, S.; Tiwari, S.; Sharma, P. Cardiovascular Response and Serum Interleukin-6 Level in Concentric vs. Eccentric Exercise. J. Clin. Diagn. Res. 2017, 11, CC04–CC08. [Google Scholar] [CrossRef]
- Eaton, M.; Granata, C.; Barry, J.; Safdar, A.; Bishop, D.; Little, J.P. Impact of a Single Bout of High-Intensity Interval Exercise and Short-Term Interval Training on Interleukin-6, FNDC5, and METRNL MRNA Expression in Human Skeletal Muscle. J. Sport Health Sci. 2018, 7, 191–196. [Google Scholar] [CrossRef]
- Pedersen, B.K. The Anti-Inflammatory Effect of Exercise: Its Role in Diabetes and Cardiovascular Disease Control. Essays Biochem. 2006, 42, 105–117. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscle as an Endocrine Organ: Focus on Muscle-Derived Interleukin-6. Physiol. Rev. 2008, 88, 1379–1406. [Google Scholar] [CrossRef]
- Petersen, A.M.W.; Pedersen, B.K. The Role of IL-6 in Mediating the Anti-Inflammatory Effects of Exercise. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2006, 57 (Suppl. 1), 43–51. [Google Scholar]
- Pedersen, B.K.; Steensberg, A.; Fischer, C.; Keller, C.; Keller, P.; Plomgaard, P.; Febbraio, M.; Saltin, B. Searching for the Exercise Factor: Is IL-6 a Candidate? J. Muscle Res. Cell Motil. 2003, 24, 113–119. [Google Scholar] [CrossRef]
- Ikeda, S.-I.; Tamura, Y.; Kakehi, S.; Sanada, H.; Kawamori, R.; Watada, H. Exercise-Induced Increase in IL-6 Level Enhances GLUT4 Expression and Insulin Sensitivity in Mouse Skeletal Muscle. Biochem. Biophys. Res. Commun. 2016, 473, 947–952. [Google Scholar] [CrossRef] [PubMed]
- Cornish, S.M.; Chase, J.E.; Bugera, E.M.; Giesbrecht, G.G. Systemic IL-6 and Myoglobin Response to Three Different Resistance Exercise Intensities in Older Men. J. Aging Phys. Act. 2018, 26, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Mendham, A.E.; Donges, C.E.; Liberts, E.A.; Duffield, R. Effects of Mode and Intensity on the Acute Exercise-Induced IL-6 and CRP Responses in a Sedentary, Overweight Population. Eur. J. Appl. Physiol. 2011, 111, 1035–1045. [Google Scholar] [CrossRef]
- Cullen, T.; Thomas, A.W.; Webb, R.; Hughes, M.G. The Relationship between Interleukin-6 in Saliva, Venous and Capillary Plasma, at Rest and in Response to Exercise. Cytokine 2015, 71, 397–400. [Google Scholar] [CrossRef]
- Krause, M.d.S.; de Bittencourt, P.I.H.J. Type 1 Diabetes: Can Exercise Impair the Autoimmune Event? The L-Arginine/Glutamine Coupling Hypothesis. Cell Biochem. Funct. 2008, 26, 406–433. [Google Scholar] [CrossRef]
- Gmiat, A.; Micielska, K.; Kozłowska, M.; Flis, D.J.; Smaruj, M.; Kujach, S.; Jaworska, J.; Lipińska, P.; Ziemann, E. The Impact of a Single Bout of High Intensity Circuit Training on Myokines’ Concentrations and Cognitive Functions in Women of Different Age. Physiol. Behav. 2017, 179, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Gil, A.M.; Elizondo-Montemayor, L. The Role of Exercise in the Interplay between Myokines, Hepatokines, Osteokines, Adipokines, and Modulation of Inflammation for Energy Substrate Redistribution and Fat Mass Loss: A Review. Nutrients 2020, 12, 1899. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, N.; Mohammadreza, H.A.; Abdolhosein, T.K.; Mehdi, N.; Arent, S.M. Serum Myokine Levels after Linear and Flexible Non-Linear Periodized Resistance Training in Overweight Sedentary Women. Eur. J. Sport Sci. 2021, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Furuichi, Y.; Manabe, Y.; Takagi, M.; Aoki, M.; Fujii, N.L. Evidence for Acute Contraction-Induced Myokine Secretion by C2C12 Myotubes. PLoS ONE 2018, 13, e0206146. [Google Scholar] [CrossRef]
- Mageriu, V.; Manole, E.; Bastian, A.E.; Staniceanu, F. Role of Myokines in Myositis Pathogenesis and Their Potential to Be New Therapeutic Targets in Idiopathic Inflammatory Myopathies. J. Immunol. Res. 2020, 2020, 9079083. [Google Scholar] [CrossRef]
- Weissenbach, J.; Chernajovsky, Y.; Zeevi, M.; Shulman, L.; Soreq, H.; Nir, U.; Wallach, D.; Perricaudet, M.; Tiollais, P.; Revel, M. Two Interferon MRNAs in Human Fibroblasts: In Vitro Translation and Escherichia Coli Cloning Studies. Proc. Natl. Acad. Sci. USA 1980, 77, 7152–7156. [Google Scholar] [CrossRef] [PubMed]
- Raschke, S.; Eckel, J. Adipo-Myokines: Two Sides of the Same Coin—Mediators of Inflammation and Mediators of Exercise. Mediat. Inflamm. 2013, 2013, 320724. [Google Scholar] [CrossRef] [PubMed]
- Steinbacher, P.; Eckl, P. Impact of Oxidative Stress on Exercising Skeletal Muscle. Biomolecules 2015, 5, 356–377. [Google Scholar] [CrossRef] [PubMed]
- Cornish, S.M.; Bugera, E.M.; Duhamel, T.A.; Peeler, J.D.; Anderson, J.E. A Focused Review of Myokines as a Potential Contributor to Muscle Hypertrophy from Resistance-Based Exercise. Eur. J. Appl. Physiol. 2020, 120, 941–959. [Google Scholar] [CrossRef] [PubMed]
- Proske, U.; Morgan, D.L. Muscle Damage from Eccentric Exercise: Mechanism, Mechanical Signs, Adaptation and Clinical Applications. J. Physiol. 2001, 537, 333–345. [Google Scholar] [CrossRef]
- Gao, S.; Durstine, J.L.; Koh, H.J.; Carver, W.E.; Frizzell, N.; Carson, J.A. Acute Myotube Protein Synthesis Regulation by IL-6-Related Cytokines. Am. J. Physiol. Cell Physiol. 2017, 313, C487–C500. [Google Scholar] [CrossRef]
- Rose-John, S. IL-6 Trans-Signaling via the Soluble IL-6 Receptor: Importance for the pro-Inflammatory Activities of IL-6. Int. J. Biol. Sci. 2012, 8, 1237–1247. [Google Scholar] [CrossRef]
- Uciechowski, P.; Dempke, W.C.M. Interleukin-6: A Masterplayer in the Cytokine Network. Oncology 2020, 98, 131–137. [Google Scholar] [CrossRef]
- McPherron, A.C.; Lawler, A.M.; Lee, S.J. Regulation of Skeletal Muscle Mass in Mice by a New TGF-Beta Superfamily Member. Nature 1997, 387, 83–90. [Google Scholar] [CrossRef]
- Iizuka, K.; Machida, T.; Hirafuji, M. Skeletal Muscle Is an Endocrine Organ. J. Pharmacol. Sci. 2014, 125, 125–131. [Google Scholar] [CrossRef]
- Argilés, J.M.; Orpí, M.; Busquets, S.; López-Soriano, F.J. Myostatin: More than Just a Regulator of Muscle Mass. Drug Discov. Today 2012, 17, 702–709. [Google Scholar] [CrossRef] [PubMed]
- De Caestecker, M. The Transforming Growth Factor-β Superfamily of Receptors. Cytokine Growth Factor Rev. 2004, 15, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Braun, T.; Gautel, M. Transcriptional Mechanisms Regulating Skeletal Muscle Differentiation, Growth and Homeostasis. Nat. Rev. Mol. Cell Biol. 2011, 12, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Allen, D.L.; Hittel, D.S.; McPherron, A.C. Expression and Function of Myostatin in Obesity, Diabetes, and Exercise Adaptation. Med. Sci. Sports Exerc. 2011, 43, 1828–1835. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Cadavid, N.F.; Taylor, W.E.; Yarasheski, K.; Sinha-Hikim, I.; Ma, K.; Ezzat, S.; Shen, R.; Lalani, R.; Asa, S.; Mamita, M.; et al. Organization of the Human Myostatin Gene and Expression in Healthy Men and HIV-Infected Men with Muscle Wasting. Proc. Natl. Acad. Sci. USA 1998, 95, 14938–14943. [Google Scholar] [CrossRef]
- Berezin, A.E.; Berezin, A.A.; Lichtenauer, M. Myokines and Heart Failure: Challenging Role in Adverse Cardiac Remodeling, Myopathy, and Clinical Outcomes. Dis. Markers 2021, 2021, 6644631. [Google Scholar] [CrossRef]
- Hittel, D.S.; Berggren, J.R.; Shearer, J.; Boyle, K.; Houmard, J.A. Increased Secretion and Expression of Myostatin in Skeletal Muscle from Extremely Obese Women. Diabetes 2009, 58, 30–38. [Google Scholar] [CrossRef]
- Gilson, H.; Schakman, O.; Combaret, L.; Lause, P.; Grobet, L.; Attaix, D.; Ketelslegers, J.M.; Thissen, J.P. Myostatin Gene Deletion Prevents Glucocorticoid-Induced Muscle Atrophy. Endocrinology 2007, 148, 452–460. [Google Scholar] [CrossRef]
- Ma, K.; Mallidis, C.; Bhasin, S.; Mahabadi, V.; Artaza, J.; Gonzalez-Cadavid, N.; Arias, J.; Salehian, B. Glucocorticoid-Induced Skeletal Muscle Atrophy Is Associated with Upregulation of Myostatin Gene Expression. Am. J. Physiol. Endocrinol. Metab. 2003, 285, E363–E371. [Google Scholar] [CrossRef]
- Nielsen, T.L.; Vissing, J.; Krag, T.O. Antimyostatin Treatment in Health and Disease: The Story of Great Expectations and Limited Success. Cells 2021, 10, 533. [Google Scholar] [CrossRef]
- Brandan, E.; Fuentes, M.E.; Andrade, W. The Proteoglycan Decorin Is Synthesized and Secreted by Differentiated Myotubes. Eur. J. Cell Biol. 1991, 55, 209–216. [Google Scholar] [PubMed]
- Miura, T.; Kishioka, Y.; Wakamatsu, J.; Hattori, A.; Hennebry, A.; Berry, C.J.; Sharma, M.; Kambadur, R.; Nishimura, T. Decorin Binds Myostatin and Modulates Its Activity to Muscle Cells. Biochem. Biophys. Res. Commun. 2006, 340, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Kishioka, Y.; Thomas, M.; Wakamatsu, J.-I.; Hattori, A.; Sharma, M.; Kambadur, R.; Nishimura, T. Decorin Enhances the Proliferation and Differentiation of Myogenic Cells through Suppressing Myostatin Activity. J. Cell. Physiol. 2008, 215, 856–867. [Google Scholar] [CrossRef] [PubMed]
- Baghy, K.; Reszegi, A.; Tátrai, P.; Kovalszky, I. Decorin in the Tumor Microenvironment. Adv. Exp. Med. Biol. 2020, 1272, 17–38. [Google Scholar] [CrossRef]
- Tortoriello, D.V.; Sidis, Y.; Holtzman, D.A.; Holmes, W.E.; Schneyer, A.L. Human Follistatin-Related Protein: A Structural Homologue of Follistatin with Nuclear Localization. Endocrinology 2001, 142, 3426–3434. [Google Scholar] [CrossRef]
- Hemmati-Brivanlou, A.; Kelly, O.G.; Melton, D.A. Follistatin, an Antagonist of Activin, Is Expressed in the Spemann Organizer and Displays Direct Neuralizing Activity. Cell 1994, 77, 283–295. [Google Scholar] [CrossRef]
- Hill, J.J.; Qiu, Y.; Hewick, R.M.; Wolfman, N.M. Regulation of Myostatin in Vivo by Growth and Differentiation Factor-Associated Serum Protein-1: A Novel Protein with Protease Inhibitor and Follistatin Domains. Mol. Endocrinol. 2003, 17, 1144–1154. [Google Scholar] [CrossRef]
- Gilson, H.; Schakman, O.; Kalista, S.; Lause, P.; Tsuchida, K.; Thissen, J.-P. Follistatin Induces Muscle Hypertrophy through Satellite Cell Proliferation and Inhibition of Both Myostatin and Activin. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E157–E164. [Google Scholar] [CrossRef]
- Suryawan, A.; Frank, J.W.; Nguyen, H.V.; Davis, T.A. Expression of the TGF-Beta Family of Ligands Is Developmentally Regulated in Skeletal Muscle of Neonatal Rats. Pediatric Res. 2006, 59, 175–179. [Google Scholar] [CrossRef][Green Version]
- Zhu, J.; Li, Y.; Lu, A.; Gharaibeh, B.; Ma, J.; Kobayashi, T.; Quintero, A.J.; Huard, J. Follistatin Improves Skeletal Muscle Healing after Injury and Disease through an Interaction with Muscle Regeneration, Angiogenesis, and Fibrosis. Am. J. Pathol. 2011, 179, 915–930. [Google Scholar] [CrossRef]
- Fukumoto, M.; Takeuchi, T.; Koubayashi, E.; Harada, S.; Ota, K.; Kojima, Y.; Higuchi, K. Induction of Brain-derived Neurotrophic Factor in Enteric Glial Cells Stimulated by Interleukin-1β via a C-Jun N-terminal Kinase Pathway. J. Clin. Biochem. Nutr. 2020, 66, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Barde, Y.A.; Edgar, D.; Thoenen, H. Purification of a New Neurotrophic Factor from Mammalian Brain. EMBO J. 1982, 1, 549–553. [Google Scholar] [CrossRef] [PubMed]
- Camandola, S.; Mattson, M.P. Brain Metabolism in Health, Aging, and Neurodegeneration. EMBO J. 2017, 36, 1474–1492. [Google Scholar] [CrossRef] [PubMed]
- Vilar, M.; Mira, H. Regulation of Neurogenesis by Neurotrophins during Adulthood: Expected and Unexpected Roles. Front. Neurosci. 2016, 10, 26. [Google Scholar] [CrossRef]
- Lee, J.; Duan, W.; Mattson, M.P. Evidence That Brain-Derived Neurotrophic Factor Is Required for Basal Neurogenesis and Mediates, in Part, the Enhancement of Neurogenesis By Dietary Restriction in the Hippocampus of Adult Mice. J. Neurochem. 2002, 82, 1367–1375. [Google Scholar] [CrossRef]
- Chao, M.V. Neurotrophins and Their Receptors: A Convergence Point for Many Signalling Pathways. Nat. Rev. Neurosci. 2003, 4, 299–309. [Google Scholar] [CrossRef]
- Diniz, B.S.; Teixeira, A.L. Brain-Derived Neurotrophic Factor and Alzheimer’s Disease: Physiopathology and Beyond. Neuromol. Med. 2011, 13, 217–222. [Google Scholar] [CrossRef]
- Figueiredo, C.; Antunes, B.M.; Giacon, T.R.; Vanderlei, L.C.M.; Campos, E.Z.; Peres, F.P.; Clark, N.W.; Panissa, V.L.G.; de Lira, F.S. Influence of Acute and Chronic High-Intensity Intermittent Aerobic plus Strength Exercise on BDNF, Lipid and Autonomic Parameters. J. Sports Sci. Med. 2019, 18, 359–368. [Google Scholar]
- McKay, B.R.; Nederveen, J.P.; Fortino, S.A.; Snijders, T.; Joanisse, S.; Kumbhare, D.A.; Parise, G. Brain-Derived Neurotrophic Factor Is Associated with Human Muscle Satellite Cell Differentiation in Response to Muscle-Damaging Exercise. Appl. Physiol. Nutr. Metab. 2020, 45, 581–590. [Google Scholar] [CrossRef]
- Krabbe, K.S.; Nielsen, A.R.; Krogh-Madsen, R.; Plomgaard, P.; Rasmussen, P.; Erikstrup, C.; Fischer, C.P.; Lindegaard, B.; Petersen, A.M.W.; Taudorf, S.; et al. Brain-Derived Neurotrophic Factor (BDNF) and Type 2 Diabetes. Diabetologia 2007, 50, 431–438. [Google Scholar] [CrossRef]
- Jamshed, H.; Beyl, R.A.; Manna, D.L.D.; Yang, E.S.; Ravussin, E.; Peterson, C.M. Early Time-Restricted Feeding Improves 24-Hour Glucose Levels and Affects Markers of the Circadian Clock, Aging, and Autophagy in Humans. Nutrients 2019, 11, 1234. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Goulding, E.H.; Zang, K.; Cepoi, D.; Cone, R.D.; Jones, K.R.; Tecott, L.H.; Reichardt, L.F. Brain-Derived Neurotrophic Factor Regulates Energy Balance Downstream of Melanocortin-4 Receptor. Nat. Neurosci. 2003, 6, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, K.; Parry, D.J.; Jasmin, B.J. BDNF Rescues Myosin Heavy Chain IIB Muscle Fibers after Neonatal Nerve Injury. Am. J. Physiol. Cell Physiol. 2004, 287, C22–C29. [Google Scholar] [CrossRef] [PubMed]
- Omura, T.; Sano, M.; Omura, K.; Hasegawa, T.; Doi, M.; Sawada, T.; Nagano, A. Different Expressions of BDNF, NT3, and NT4 in Muscle and Nerve after Various Types of Peripheral Nerve Injuries. J. Peripher. Nerv. Syst. JPNS 2005, 10, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Clow, C.; Jasmin, B.J. Brain-Derived Neurotrophic Factor Regulates Satellite Cell Differentiation and Skeltal Muscle Regeneration. Mol. Biol. Cell 2010, 21, 2182–2190. [Google Scholar] [CrossRef]
- Yu, T.; Chang, Y.; Gao, X.L.; Li, H.; Zhao, P. Dynamic Expression and the Role of BDNF in Exercise-Induced Skeletal Muscle Regeneration. Int. J. Sports Med. 2017, 38, 959–966. [Google Scholar] [CrossRef]
- Matthews, V.B.; Aström, M.-B.; Chan, M.H.S.; Bruce, C.R.; Krabbe, K.S.; Prelovsek, O.; Akerström, T.; Yfanti, C.; Broholm, C.; Mortensen, O.H.; et al. Brain-Derived Neurotrophic Factor Is Produced by Skeletal Muscle Cells in Response to Contraction and Enhances Fat Oxidation via Activation of AMP-Activated Protein Kinase. Diabetologia 2009, 52, 1409–1418. [Google Scholar] [CrossRef]
- Yamanaka, M.; Tsuchida, A.; Nakagawa, T.; Nonomura, T.; Ono-Kishino, M.; Sugaru, E.; Noguchi, H.; Taiji, M. Brain-Derived Neurotrophic Factor Enhances Glucose Utilization in Peripheral Tissues of Diabetic Mice. Diabetes Obes. Metab. 2007, 9, 59–64. [Google Scholar] [CrossRef]
- Yarrow, J.F.; White, L.J.; McCoy, S.C.; Borst, S.E. Training Augments Resistance Exercise Induced Elevation of Circulating Brain Derived Neurotrophic Factor (BDNF). Neurosci. Lett. 2010, 479, 161–165. [Google Scholar] [CrossRef]
- Church, D.D.; Hoffman, J.R.; Mangine, G.T.; Jajtner, A.R.; Townsend, J.R.; Beyer, K.S.; Wang, R.; la Monica, M.B.; Fukuda, D.H.; Stout, J.R. Comparison of High-Intensity vs. High-Volume Resistance Training on the BDNF Response to Exercise. J. Appl. Physiol. 2016, 121, 123–128. [Google Scholar] [CrossRef]
- Palasz, E.; Wysocka, A.; Gasiorowska, A.; Chalimoniuk, M.; Niewiadomski, W.; Niewiadomska, G. BDNF as a Promising Therapeutic Agent in Parkinson’s Disease. Int. J. Mol. Sci. 2020, 21, 1170. [Google Scholar] [CrossRef] [PubMed]
- Linker, R.A.; Lee, D.-H.; Demir, S.; Wiese, S.; Kruse, N.; Siglienti, I.; Gerhardt, E.; Neumann, H.; Sendtner, M.; Lühder, F.; et al. Functional Role of Brain-Derived Neurotrophic Factor in Neuroprotective Autoimmunity: Therapeutic Implications in a Model of Multiple Sclerosis. Brain 2010, 133, 2248–2263. [Google Scholar] [CrossRef] [PubMed]
- Whone, A.; Luz, M.; Boca, M.; Woolley, M.; Mooney, L.; Dharia, S.; Broadfoot, J.; Cronin, D.; Schroers, C.; Barua, N.U.; et al. Randomized Trial of Intermittent Intraputamenal Glial Cell Line-Derived Neurotrophic Factor in Parkinson’s Disease. Brain 2019, 142, 512–525. [Google Scholar] [CrossRef] [PubMed]
- Denyer, R.; Douglas, M.R. Gene Therapy for Parkinson’s Disease. Parkinson’s Dis. 2012, 2012, 757305. [Google Scholar] [CrossRef]
- Géral, C.; Angelova, A.; Lesieur, S. From Molecular to Nanotechnology Strategies for Delivery of Neurotrophins: Emphasis on Brain-Derived Neurotrophic Factor (BDNF). Pharmaceutics 2013, 5, 127–167. [Google Scholar] [CrossRef]
- Boström, P.; Wu, J.; Jedrychowski, M.P.; Korde, A.; Ye, L.; Lo, J.C.; Rasbach, K.A.; Boström, E.A.; Choi, J.H.; Long, J.Z.; et al. A PGC1α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 2012, 481, 463–468. [Google Scholar] [CrossRef]
- Polyzos, S.A.; Anastasilakis, A.D.; Efstathiadou, Z.A.; Makras, P.; Perakakis, N.; Kountouras, J.; Mantzoros, C.S. Irisin in Metabolic Diseases. Endocrine 2018, 59, 260–274. [Google Scholar] [CrossRef]
- Perakakis, N.; Triantafyllou, G.A.; Fernández-Real, J.M.; Huh, J.Y.; Park, K.H.; Seufert, J.; Mantzoros, C.S. Physiology and role of irisin in glucose homeostasis. Nat. Rev. Endocrinol. 2017, 13, 324–337. [Google Scholar] [CrossRef]
- Albrecht, E.; Norheim, F.; Thiede, B.; Holen, T.; Ohashi, T.; Schering, L.; Lee, S.; Brenmoehl, J.; Thomas, S.; Drevon, C.A.; et al. Irisin—A Myth Rather than an Exercise-Inducible Myokine. Sci. Rep. 2015, 5, 8889. [Google Scholar] [CrossRef]
- Jedrychowski, M.P.; Wrann, C.D.; Paulo, J.A.; Gerber, K.K.; Szpyt, J.; Robinson, M.M.; Nair, K.S.; Gygi, S.P.; Spiegelman, B.M. Detection and Quantitation of Circulating Human Irisin by Tandem Mass Spectrometry. Cell Metab. 2015, 22, 734–740. [Google Scholar] [CrossRef]
- Hecksteden, A.; Wegmann, M.; Steffen, A.; Kraushaar, J.; Morsch, A.; Ruppenthal, S.; Kaestner, L.; Meyer, T. Irisin and Exercise Training in Humans—Results from a Randomized Controlled Training Trial. BMC Med. 2013, 11, 235. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.; Rioux, B.V.; Goulet, E.D.B.; Johanssen, N.M.; Swift, D.L.; Bouchard, D.R.; Loewen, H.; Sénéchal, M. Effect of an Acute Exercise Bout on Immediate Post-Exercise Irisin Concentration in Adults: A Meta-Analysis. Scand. J. Med. Sci. Sports 2018, 28, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Cai, X.; Sun, Z.; Schumann, U.; Zügel, M.; Steinacker, J.M. Chronic Exercise Training and Circulating Irisin in Adults: A Meta-Analysis. Sports Med. 2015, 45, 1577–1588. [Google Scholar] [CrossRef] [PubMed]
- Colaianni, G.; Cinti, S.; Colucci, S.; Grano, M. Irisin and Musculoskeletal Health. Ann. N. Y. Acad. Sci. 2017, 1402, 5–9. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Ando, D.; Takamatsu, K.; Goto, K. Resistance Exercise Induces a Greater Irisin Response than Endurance Exercise. Metabolism 2015, 64, 1042–1050. [Google Scholar] [CrossRef]
- Huh, J.Y.; Siopi, A.; Mougios, V.; Park, K.H.; Mantzoros, C.S. Irisin in Response to Exercise in Humans with and without Metabolic Syndrome. J. Clin. Endocrinol. Metab. 2015, 100, E453–E457. [Google Scholar] [CrossRef]
- Mahgoub, M.O.; D’Souza, C.; al Darmaki, R.S.M.H.; Baniyas, M.M.Y.H.; Adeghate, E. An Update on the Role of Irisin in the Regulation of Endocrine and Metabolic Functions. Peptides 2018, 104, 15–23. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, Y.T.; Zhang, S.; Dubielecka, P.M.; Du, J.; Yano, N.; Chin, Y.E.; Zhuang, S.; Qin, G.; Zhao, T.C. Irisin Plays a Pivotal Role to Protect the Heart against Ischemia and Reperfusion Injury. J. Cell. Physiol. 2017, 232, 3775–3785. [Google Scholar] [CrossRef]
- Zhang, Y.; Song, H.; Zhang, Y.; Wu, F.; Mu, Q.; Jiang, M.; Wang, F.; Zhang, W.; Li, L.; Shao, L.; et al. Irisin Inhibits Atherosclerosis by Promoting Endothelial Proliferation Through MicroRNA126-5p. J. Am. Heart Assoc. 2016, 5, e004031. [Google Scholar] [CrossRef]
- Kim, H.; Wrann, C.D.; Jedrychowski, M.; Vidoni, S.; Kitase, Y.; Nagano, K.; Zhou, C.; Chou, J.; Parkman, V.-J.A.; Novick, S.J.; et al. Irisin Mediates Effects on Bone and Fat via AV Integrin Receptors. Cell 2018, 175, 1756–1768.e17. [Google Scholar] [CrossRef]
- Briganti, S.I.; Gaspa, G.; Tabacco, G.; Naciu, A.M.; Cesareo, R.; Manfrini, S.; Palermo, A. Irisin as a Regulator of Bone and Glucose Metabolism. Minerva Endocrinol. 2018, 43, 489–500. [Google Scholar] [CrossRef] [PubMed]
- Kim, O.; Song, J. The Role of Irisin in Alzheimer’s Disease. J. Clin. Med. 2018, 7, 407. [Google Scholar] [CrossRef] [PubMed]
- Maalouf, G.E.; el Khoury, D. Exercise-Induced Irisin, the Fat Browning Myokine, as a Potential Anticancer Agent. J. Obes. 2019, 2019, 6561726. [Google Scholar] [CrossRef] [PubMed]
- Gannon, N.P.; Vaughan, R.A.; Garcia-Smith, R.; Bisoffi, M.; Trujillo, K.A. Effects of the Exercise-Inducible Myokine Irisin on Malignant and Non-Malignant Breast Epithelial Cell Behavior in Vitro. Int. J. Cancer 2015, 136, E197–E202. [Google Scholar] [CrossRef] [PubMed]
- Provatopoulou, X.; Georgiou, G.P.; Kalogera, E.; Kalles, V.; Matiatou, M.A.; Papapanagiotou, I.; Sagkriotis, A.; Zografos, G.C.; Gounaris, A. Serum Irisin Levels Are Lower in Patients with Breast Cancer: Association with Disease Diagnosis and Tumor Characteristics. BMC Cancer 2015, 15, 898. [Google Scholar] [CrossRef]
- Shao, L.; Li, H.; Chen, J.; Song, H.; Zhang, Y.; Wu, F.; Wang, W.; Zhang, W.; Wang, F.; Li, H.; et al. Irisin Suppresses the Migration, Proliferation, and Invasion of Lung Cancer Cells via Inhibition of Epithelial-to-Mesenchymal Transition. Biochem. Biophys. Res. Commun. 2017, 485, 598–605. [Google Scholar] [CrossRef]
- Kong, G.; Jiang, Y.; Sun, X.; Cao, Z.; Zhang, G.; Zhao, Z.; Zhao, Y.; Yu, Q.; Cheng, G. Irisin Reverses the IL-6 Induced Epithelial-Mesenchymal Transition in Osteosarcoma Cell Migration and Invasion through the STAT3/Snail Signaling Pathway. Oncol. Rep. 2017, 38, 2647–2656. [Google Scholar] [CrossRef]
- Zhu, H.; Liu, M.; Zhang, N.; Pan, H.; Lin, G.; Li, N.; Wang, L.; Yang, H.; Yan, K.; Gong, F. Serum and Adipose Tissue MRNA Levels of ATF3 and FNDC5/Irisin in Colorectal Cancer Patients With or Without Obesity. Front. Physiol. 2018, 9, 1125. [Google Scholar] [CrossRef]
- Liu, J.; Song, N.; Huang, Y.; Chen, Y. Irisin Inhibits Pancreatic Cancer Cell Growth via the AMPK-MTOR Pathway. Sci. Rep. 2018, 8, 15247. [Google Scholar] [CrossRef]
- Gaggini, M.; Cabiati, M.; del Turco, S.; Navarra, T.; de Simone, P.; Filipponi, F.; Del Ry, S.; Gastaldelli, A.; Basta, G. Increased FNDC5/Irisin Expression in Human Hepatocellular Carcinoma. Peptides 2017, 88, 62–66. [Google Scholar] [CrossRef]
- Shi, G.; Tang, N.; Qiu, J.; Zhang, D.; Huang, F.; Cheng, Y.; Ding, K.; Li, W.; Zhang, P.; Tan, X. Irisin Stimulates Cell Proliferation and Invasion by Targeting the PI3K/AKT Pathway in Human Hepatocellular Carcinoma. Biochem. Biophys. Res. Commun. 2017, 493, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Li, D.-J.; Huang, F.; Lu, W.-J.; Jiang, G.-J.; Deng, Y.-P.; Shen, F.-M. Metformin Promotes Irisin Release from Murine Skeletal Muscle Independently of AMP-Activated Protein Kinase Activation. Acta Physiol. 2015, 213, 711–721. [Google Scholar] [CrossRef] [PubMed]
- Haemmerle, G.; Moustafa, T.; Woelkart, G.; Büttner, S.; Schmidt, A.; van de Weijer, T.; Hesselink, M.; Jaeger, D.; Kienesberger, P.C.; Zierler, K.; et al. ATGL-Mediated Fat Catabolism Regulates Cardiac Mitochondrial Function via PPAR-α and PGC-1. Nat. Med. 2011, 17, 1076–1085. [Google Scholar] [CrossRef] [PubMed]
- Dillon, L.M.; Rebelo, A.P.; Moraes, C.T. The Role of PGC-1 Coactivators in Aging Skeletal Muscle and Heart. IUBMB Life 2012, 64, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Puigserver, P.; Wu, Z.; Park, C.W.; Graves, R.; Wright, M.; Spiegelman, B.M. A Cold-Inducible Coactivator of Nuclear Receptors Linked to Adaptive Thermogenesis. Cell 1998, 92, 829–839. [Google Scholar] [CrossRef]
- Shen, L.; Wang, H.; Ye, P. Effects of pioglitazone on myocardial peroxisome proliferator-activated receptor gamma co-activator lα expression in rats with myocardial ischemia/reperfusion injury. Nan Fang Yi Ke Da Xue Xue Bao 2014, 34, 197–200. [Google Scholar]
- Sun, C.-K.; Chang, L.-T.; Sheu, J.-J.; Wang, C.-Y.; Youssef, A.A.; Wu, C.-J.; Chua, S.; Yip, H.-K. Losartan Preserves Integrity of Cardiac Gap Junctions and PGC-1 Alpha Gene Expression and Prevents Cellular Apoptosis in Remote Area of Left Ventricular Myocardium Following Acute Myocardial Infarction. Int. Heart J. 2007, 48, 533–546. [Google Scholar] [CrossRef]
- Martin, A.R.; Chung, S.; Koehler, K. Is Exercise a Match for Cold Exposure? Common Molecular Framework for Adipose Tissue Browning. Int. J. Sports Med. 2020, 41, 427–442. [Google Scholar] [CrossRef]
- Rao, R.R.; Long, J.Z.; White, J.P.; Svensson, K.J.; Lou, J.; Lokurkar, I.; Jedrychowski, M.P.; Ruas, J.L.; Wrann, C.D.; Lo, J.C.; et al. Meteorin-like Is a Hormone That Regulates Immune-Adipose Interactions to Increase Beige Fat Thermogenesis. Cell 2014, 157, 1279–1291. [Google Scholar] [CrossRef]
- Ruas, J.L.; White, J.P.; Rao, R.R.; Kleiner, S.; Brannan, K.T.; Harrison, B.C.; Greene, N.P.; Wu, J.; Estall, J.L.; Irving, B.A.; et al. A PGC-1α Isoform Induced by Resistance Training Regulates Skeletal Muscle Hypertrophy. Cell 2012, 151, 1319–1331. [Google Scholar] [CrossRef]
- Miao, Z.-W.; Hu, W.-J.; Li, Z.-Y.; Miao, C.-Y. Involvement of the Secreted Protein Metrnl in Human Diseases. Acta Pharmacol. Sin. 2020, 41, 1525–1530. [Google Scholar] [CrossRef]
- Ushach, I.; Burkhardt, A.M.; Martinez, C.; Hevezi, P.A.; Gerber, P.A.; Buhren, B.A.; Schrumpf, H.; Valle-Rios, R.; Vazquez, M.I.; Homey, B.; et al. METEORIN-LIKE Is a Cytokine Associated with Barrier Tissues and Alternatively Activated Macrophages. Clin. Immunol. 2015, 156, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Ushach, I.; Arrevillaga-Boni, G.; Heller, G.N.; Pone, E.; Hernandez-Ruiz, M.; Catalan-Dibene, J.; Hevezi, P.; Zlotnik, A. Meteorin-like/Meteorin-β Is a Novel Immunoregulatory Cytokine Associated with Inflammation. J. Immunol. 2018, 201, 3669–3676. [Google Scholar] [CrossRef] [PubMed]
- Buford, T.W.; Cooke, M.B.; Willoughby, D.S. Resistance Exercise-Induced Changes of Inflammatory Gene Expression within Human Skeletal Muscle. Eur. J. Appl. Physiol. 2009, 107, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M.D.; Mitchell, J.B.; Currie-Elolf, L.M.; Yellott, R.C.; Hubing, K.A. Influence of Commonly Employed Resistance Exercise Protocols on Circulating IL-6 and Indices of Insulin Sensitivity. J. Strength Cond. Res. 2010, 24, 1091–1101. [Google Scholar] [CrossRef] [PubMed]
- Della Gatta, P.A.; Garnham, A.P.; Peake, J.M.; Cameron-Smith, D. Effect of Exercise Training on Skeletal Muscle Cytokine Expression in the Elderly. Brain Behav. Immun. 2014, 39, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Quiles, J.M.; Klemp, A.; Dolan, C.; Maharaj, A.; Huang, C.-J.; Khamoui, A.V.; Trexler, E.T.; Whitehurst, M.; Zourdos, M.C. Impact of Resistance Training Program Configuration on the Circulating Brain-Derived Neurotrophic Factor Response. Appl. Physiol. Nutr. Metab. 2020, 45, 667–674. [Google Scholar] [CrossRef]
- Tomeleri, C.M.; Ribeiro, A.S.; Souza, M.F.; Schiavoni, D.; Schoenfeld, B.J.; Venturini, D.; Barbosa, D.S.; Landucci, K.; Sardinha, L.B.; Cyrino, E.S. Resistance Training Improves Inflammatory Level, Lipid and Glycemic Profiles in Obese Older Women: A Randomized Controlled Trial. Exp. Gerontol. 2016, 84, 80–87. [Google Scholar] [CrossRef]
- Kazemi, F. The Correlation of Resistance Exercise-Induced Myostatin with Insulin Resistance and Plasma Cytokines in Healthy Young Men. J. Endocrinol. Investig. 2016, 39, 383–388. [Google Scholar] [CrossRef]
- Raue, U.; Slivka, D.; Jemiolo, B.; Hollon, C.; Trappe, S. Myogenic Gene Expression at Rest and after a Bout of Resistance Exercise in Young (18–30 Yr) and Old (80–89 Yr) Women. J. Appl. Physiol. 2006, 101, 53–59. [Google Scholar] [CrossRef]
- Bugera, E.M.; Duhamel, T.A.; Peeler, J.D.; Cornish, S.M. The Systemic Myokine Response of Decorin, Interleukin-6 (IL-6) and Interleukin-15 (IL-15) to an Acute Bout of Blood Flow Restricted Exercise. Eur. J. Appl. Physiol. 2018, 118, 2679–2686. [Google Scholar] [CrossRef] [PubMed]
- Kanzleiter, T.; Rath, M.; Görgens, S.W.; Jensen, J.; Tangen, D.S.; Kolnes, A.J.; Kolnes, K.J.; Lee, S.; Eckel, J.; Schürmann, A.; et al. The Myokine Decorin Is Regulated by Contraction and Involved in Muscle Hypertrophy. Biochem. Biophys. Res. Commun. 2014, 450, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, R.; Rashidlamir, A.; Motevalli, M.S.; Elliott, B.T.; Mehrabani, J.; Wong, A. Effects of Upper-Body, Lower-Body, or Combined Resistance Training on the Ratio of Follistatin and Myostatin in Middle-Aged Men. Eur. J. Appl. Physiol. 2019, 119, 1921–1931. [Google Scholar] [CrossRef] [PubMed]
- Negaresh, R.; Ranjbar, R.; Habibi, A.; Mokhtarzade, M.; Fokin, A.; Gharibvand, M.M. The Effect of Resistance Training on Quadriceps Muscle Volume and Some Growth Factors in Elderly and Young Men. Adv. Gerontol. Uspekhi Gerontol. 2017, 30, 880–887. [Google Scholar]
- Jang, K.S.; Kang, S.; Woo, S.H.; Bae, J.Y.; Shin, K.O. Effects of Combined Open Kinetic Chain and Closed Kinetic Chain Training Using Pulley Exercise Machines on Muscle Strength and Angiogenesis Factors. J. Phys. Ther. Sci. 2016, 28, 960–966. [Google Scholar] [CrossRef][Green Version]
- Hofmann, M.; Schober-Halper, B.; Oesen, S.; Franzke, B.; Tschan, H.; Bachl, N.; Strasser, E.M.; Quittan, M.; Wagner, K.H.; Wessner, B. Effects of Elastic Band Resistance Training and Nutritional Supplementation on Muscle Quality and Circulating Muscle Growth and Degradation Factors of Institutionalized Elderly Women: The Vienna Active Ageing Study (VAAS). Eur. J. Appl. Physiol. 2016, 116, 885–897. [Google Scholar] [CrossRef]
- Ellefsen, S.; Vikmoen, O.; Slettaløkken, G.; Whist, J.E.; Nygaard, H.; Hollan, I.; Rauk, I.; Vegge, G.; Strand, T.A.; Raastad, T.; et al. Irisin and FNDC5: Effects of 12-Week Strength Training, and Relations to Muscle Phenotype and Body Mass Composition in Untrained Women. Eur. J. Appl. Physiol. 2014, 114, 1875–1888. [Google Scholar] [CrossRef]
- Kim, H.J.; Lee, H.J.; So, B.; Son, J.S.; Yoon, D.; Song, W. Effect of Aerobic Training and Resistance Training on Circulating Irisin Level and Their Association with Change of Body Composition in Overweight/Obese Adults: A Pilot Study. Physiol. Res. 2016, 65, 271–279. [Google Scholar] [CrossRef]
- Norheim, F.; Langleite, T.M.; Hjorth, M.; Holen, T.; Kielland, A.; Stadheim, H.K.; Gulseth, H.L.; Birkeland, K.I.; Jensen, J.; Drevon, C.A. The Effects of Acute and Chronic Exercise on PGC-1α, Irisin and Browning of Subcutaneous Adipose Tissue in Humans. FEBS J. 2014, 281, 739–749. [Google Scholar] [CrossRef]
- Nygaard, H.; Slettaløkken, G.; Vegge, G.; Hollan, I.; Whist, J.E.; Strand, T.; Rønnestad, B.R.; Ellefsen, S. Irisin in Blood Increases Transiently after Single Sessions of Intense Endurance Exercise and Heavy Strength Training. PLoS ONE 2015, 10, e0121367. [Google Scholar] [CrossRef]
- Pekkala, S.; Wiklund, P.K.; Hulmi, J.J.; Ahtiainen, J.P.; Horttanainen, M.; Pöllänen, E.; Mäkelä, K.A.; Kainulainen, H.; Häkkinen, K.; Nyman, K.; et al. Are Skeletal Muscle FNDC5 Gene Expression and Irisin Release Regulated by Exercise and Related to Health? J. Physiol. 2013, 591, 5393–5400. [Google Scholar] [CrossRef] [PubMed]
- Tibana, R.A.; Nascimento, D.d.C.; de Souza, N.M.F.; de Souza, V.C.; Neto, I.V.d.S.; Voltarelli, F.A.; Pereira, G.B.; Navalta, J.W.; Prestes, J. Irisin Levels Are Not Associated to Resistance Training-Induced Alterations in Body Mass Composition in Older Untrained Women with and without Obesity. J. Nutr. Health Aging 2017, 21, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Su, Z.; Qu, C.; Dong, Y. Effects of 12 Weeks Resistance Training on Serum Irisin in Older Male Adults. Front. Physiol. 2017, 8, 171. [Google Scholar] [CrossRef] [PubMed]
- So, B.; Kim, H.-J.; Kim, J.; Song, W. Exercise-Induced Myokines in Health and Metabolic Diseases. Integr. Med. Res. 2014, 3, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Sanchéz, M.A.; Bustos-Cruz, R.H.; Velasco-Orjuela, G.P.; Quintero, A.P.; Tordecilla-Sanders, A.; Correa-Bautista, J.E.; Triana-Reina, H.R.; García-Hermoso, A.; González-Ruíz, K.; Peña-Guzmán, C.A.; et al. Acute Effects of High Intensity, Resistance, or Combined Protocol on the Increase of Level of Neurotrophic Factors in Physically Inactive Overweight Adults: The BrainFit Study. Front. Physiol. 2018, 9, 741. [Google Scholar] [CrossRef]
- Forti, L.N.; van Roie, E.; Njemini, R.; Coudyzer, W.; Beyer, I.; Delecluse, C.; Bautmans, I. Dose-and Gender-Specific Effects of Resistance Training on Circulating Levels of Brain Derived Neurotrophic Factor (BDNF) in Community-Dwelling Older Adults. Exp. Gerontol. 2015, 70, 144–149. [Google Scholar] [CrossRef]
- Lodo, L.; Moreira, A.; Bacurau, R.F.P.; Capitani, C.D.; Barbosa, W.P.; Massa, M.; Schoenfeld, B.J.; Aoki, M.S. Resistance Exercise Intensity Does Not Influence Neurotrophic Factors Response in Equated Volume Schemes. J. Hum. Kinet. 2020, 74, 227–236. [Google Scholar] [CrossRef]
- Jørgensen, M.L.K.; Kjølhede, T.; Dalgas, U.; Hvid, L.G. Plasma Brain-Derived Neurotrophic Factor (BDNF) and Sphingosine-1-Phosphat (S1P) Are NOT the Main Mediators of Neuroprotection Induced by Resistance Training in Persons with Multiple Sclerosis—A Randomized Controlled Trial. Mult. Scler. Relat. Disord. 2019, 31, 106–111. [Google Scholar] [CrossRef]
- Marston, K.J.; Newton, M.J.; Brown, B.M.; Rainey-Smith, S.R.; Bird, S.; Martins, R.N.; Peiffer, J.J. Intense Resistance Exercise Increases Peripheral Brain-Derived Neurotrophic Factor. J. Sci. Med. Sport 2017, 20, 899–903. [Google Scholar] [CrossRef]
- Marston, K.J.; Brown, B.M.; Rainey-Smith, S.R.; Bird, S.; Wijaya, L.; Teo, S.Y.M.; Laws, S.M.; Martins, R.N.; Peiffer, J.J. Twelve Weeks of Resistance Training Does Not Influence Peripheral Levels of Neurotrophic Growth Factors or Homocysteine in Healthy Adults: A Randomized-Controlled Trial. Eur. J. Appl. Physiol. 2019, 119, 2167–2176. [Google Scholar] [CrossRef]
- Roh, H.; Cho, S.; So, W. A Cross-Sectional Study Evaluating the Effects of Resistance Exercise on Inflammation and Neurotrophic Factors in Elderly Women with Obesity. J. Clin. Med. 2020, 9, 842. [Google Scholar] [CrossRef] [PubMed]
- Urzi, F.; Marusic, U.; Ličen, S.; Buzan, E. Effects of Elastic Resistance Training on Functional Performance and Myokines in Older Women—A Randomized Controlled Trial. J. Am. Med. Dir. Assoc. 2019, 20, 830–834.e2. [Google Scholar] [CrossRef]
- Vega, S.R.; Knicker, A.; Hollmann, W.; Bloch, W.; Strüder, H.K. Effect of Resistance Exercise on Serum Levels of Growth Factors in Humans. Horm. Metab. Res. Horm.—Stoffwechs. Horm. Metab. 2010, 42, 982–986. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.J.; D’Angiulli, A.; Cameron, J.D.; Sigal, R.J.; Kenny, G.P.; Holcik, M.; Doucette, S.; Alberga, A.S.; Prud’homme, D.; Hadjiyannakis, S.; et al. Changes in the Brain-Derived Neurotrophic Factor Are Associated with Improvements in Diabetes Risk Factors after Exercise Training in Adolescents with Obesity: The Hearty Randomized Controlled Trial. Neural Plast. 2018, 2018, 7169583. [Google Scholar] [CrossRef] [PubMed]
- Goldfield, G.S.; Kenny, G.P.; Prud’homme, D.; Holcik, M.; Alberga, A.S.; Fahnestock, M.; Cameron, J.D.; Doucette, S.; Hadjiyannakis, S.; Tulloch, H.; et al. Effects of Aerobic Training, Resistance Training, or Both on Brain-Derived Neurotrophic Factor in Adolescents with Obesity: The Hearty Randomized Controlled Trial. Physiol. Behav. 2018, 191, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Alberga, A.S.; Goldfield, G.S.; Kenny, G.P.; Hadjiyannakis, S.; Phillips, P.; Prud’homme, D.; Tulloch, H.; Gougeon, R.; Wells, G.A.; Sigal, R.J. Healthy Eating, Aerobic and Resistance Training in Youth (HEARTY): Study Rationale, Design and Methods. Contemp. Clin. Trials 2012, 33, 839–847. [Google Scholar] [CrossRef]
- Alberga, A.S.; Prud’homme, D.; Kenny, G.P.; Goldfield, G.S.; Hadjiyannakis, S.; Gougeon, R.; Phillips, P.; Malcolm, J.; Wells, G.; Doucette, S.; et al. Effects of Aerobic and Resistance Training on Abdominal Fat, Apolipoproteins and High-Sensitivity C-Reactive Protein in Adolescents with Obesity: The HEARTY Randomized Clinical Trial. Int. J. Obes. 2015, 39, 1494–1500. [Google Scholar] [CrossRef]
- Walsh, J.J.; Scribbans, T.D.; Bentley, R.F.; Kellawan, J.M.; Gurd, B.; Tschakovsky, M.E. Neurotrophic Growth Factor Responses to Lower Body Resistance Training in Older Adults. Appl. Physiol. Nutr. Metab. 2016, 41, 315–323. [Google Scholar] [CrossRef]
- Wens, I.; Keytsman, C.; Deckx, N.; Cools, N.; Dalgas, U.; Eijnde, B.O. Brain Derived Neurotrophic Factor in Multiple Sclerosis: Effect of 24 Weeks Endurance and Resistance Training. Eur. J. Neurol. 2016, 23, 1028–1035. [Google Scholar] [CrossRef]
- Thompson, W.R.; Sallis, R.; Joy, E.; Jaworski, C.A.; Stuhr, R.M.; Trilk, J.L. Exercise Is Medicine. Am. J. Lifestyle Med. 2020, 14, 511–523. [Google Scholar] [CrossRef]
- Senesi, P.; Luzi, L.; Terruzzi, I. Adipokines, Myokines, and Cardiokines: The Role of Nutritional Interventions. Int. J. Mol. Sci. 2020, 21, 8372. [Google Scholar] [CrossRef]
- Duan, Y.; Li, F.; Li, Y.; Tang, Y.; Kong, X.; Feng, Z.; Anthony, T.G.; Watford, M.; Hou, Y.; Wu, G.; et al. The Role of Leucine and Its Metabolites in Protein and Energy Metabolism. Amino Acids 2016, 48, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D.C.; Lila, M.A.; Gillitt, N.D. Immunometabolism: A Multi-Omics Approach to Interpreting the Influence of Exercise and Diet on the Immune System. Annu. Rev. Food Sci. Technol. 2019, 10, 341–363. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D.C.; Pence, B.D. Exercise Immunology: Future Directions. J. Sport Health Sci. 2020, 9, 432–445. [Google Scholar] [CrossRef]
- Spiering, B.A.; Kraemer, W.J.; Anderson, J.M.; Armstrong, L.E.; Nindl, B.C.; Volek, J.S.; Maresh, C.M. Resistance Exercise Biology: Manipulation of Resistance Exercise Programme Variables Determines the Responses of Cellular and Molecular Signalling Pathways. Sports Med. 2008, 38, 527–540. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zunner, B.E.M.; Wachsmuth, N.B.; Eckstein, M.L.; Scherl, L.; Schierbauer, J.R.; Haupt, S.; Stumpf, C.; Reusch, L.; Moser, O. Myokines and Resistance Training: A Narrative Review. Int. J. Mol. Sci. 2022, 23, 3501. https://doi.org/10.3390/ijms23073501
Zunner BEM, Wachsmuth NB, Eckstein ML, Scherl L, Schierbauer JR, Haupt S, Stumpf C, Reusch L, Moser O. Myokines and Resistance Training: A Narrative Review. International Journal of Molecular Sciences. 2022; 23(7):3501. https://doi.org/10.3390/ijms23073501
Chicago/Turabian StyleZunner, Beate E. M., Nadine B. Wachsmuth, Max L. Eckstein, Lukas Scherl, Janis R. Schierbauer, Sandra Haupt, Christian Stumpf, Laura Reusch, and Othmar Moser. 2022. "Myokines and Resistance Training: A Narrative Review" International Journal of Molecular Sciences 23, no. 7: 3501. https://doi.org/10.3390/ijms23073501
APA StyleZunner, B. E. M., Wachsmuth, N. B., Eckstein, M. L., Scherl, L., Schierbauer, J. R., Haupt, S., Stumpf, C., Reusch, L., & Moser, O. (2022). Myokines and Resistance Training: A Narrative Review. International Journal of Molecular Sciences, 23(7), 3501. https://doi.org/10.3390/ijms23073501







