Synergistic Antimicrobial Activities of Chitosan Mixtures and Chitosan–Copper Combinations
Abstract
:1. Introduction
2. Results
2.1. Structural Characterization of the Chitosan Samples
2.2. Antifungal Activity
2.3. Synergistic Antimicrobial Activity of Chitosan Oligomers and Polymers
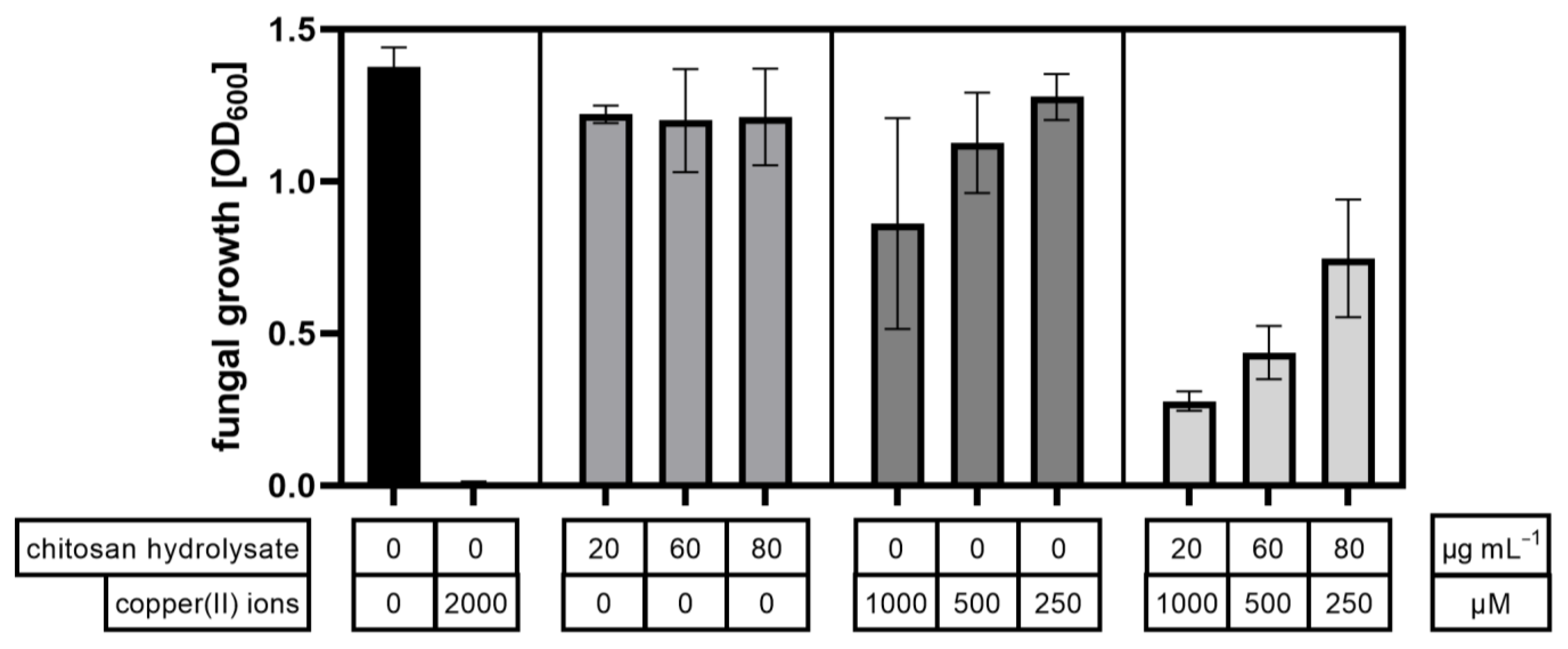
2.4. Antimicrobial Activities of Chitosan Mixtures in Combination with Copper(II) Ions
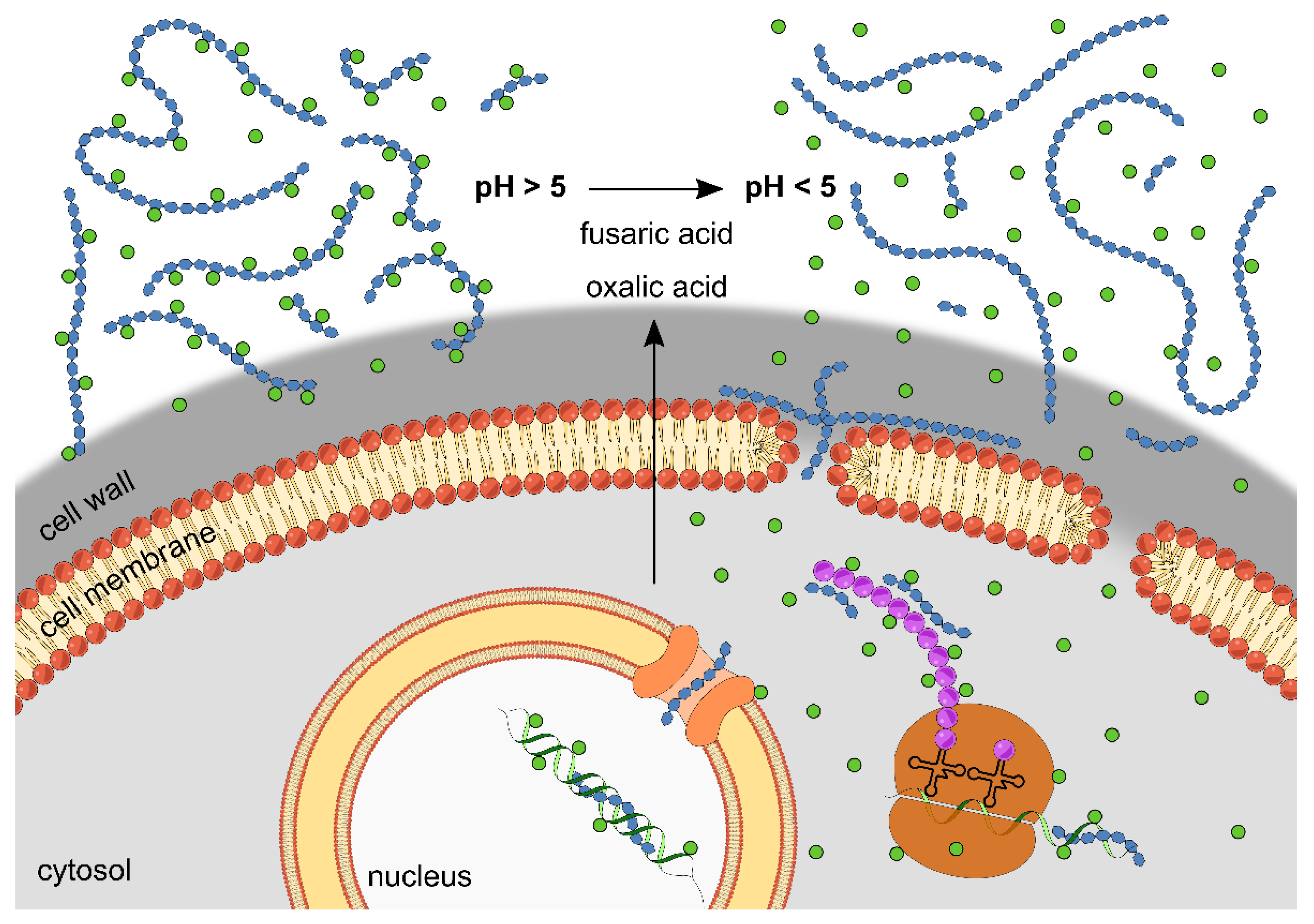
3. Discussion
4. Materials and Methods
4.1. Preparation of Chitosan Solutions
4.2. Chitosan Characterization
4.3. Chitosan Fractionation
4.4. Cultivation and Induction of Conidia Production of Fusarium graminearum
4.5. Antifungal Activity
4.6. Test for Synergistic Activity
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brunel, F.; El Gueddari, N.E.; Moerschbacher, B.M. Moerschbacher. Complexation of copper(II) with chitosan nanogels: Toward control of microbial growth. Carbohydr. Polym. 2013, 92, 1348–1356. [Google Scholar] [CrossRef] [PubMed]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- El Hadrami, A.; Adam, L.R.; El Hadrami, I.; Daayf, F. Chitosan in plant protection. Mar. Drugs 2010, 8, 968–987. [Google Scholar] [CrossRef] [PubMed]
- Orzali, L.; Corsi, B.; Forni, C.; Riccioni, L. Chitosan in agriculture: A new challenge for managing plant disease. In Biological Activities and Application of Marine Polysaccharides; InTech: London, UK, 2017. [Google Scholar] [CrossRef] [Green Version]
- Xing, K.; Zhu, X.; Peng, X.; Qin, S. Chitosan antimicrobial and eliciting properties for pest control in agriculture: A review. Agron. Sustain. Dev. 2015, 35, 569–588. [Google Scholar] [CrossRef] [Green Version]
- Malerba, M.; Cerana, R. Chitosan effects on plant systems. Int. J. Mol. Sci. 2016, 17, 996. [Google Scholar] [CrossRef]
- Sharif, R.; Mujtaba, M.; Rahman, M.U.; Shalmani, A.; Ahmad, H.; Anwar, T.; Tianchan, D.; Wang, X. The multifunctional role of chitosan in horticultural crops; A review. Molecules 2018, 23, 872. [Google Scholar] [CrossRef] [Green Version]
- Wattjes, J.; Sreekumar, S.; Richter, C.; Cord-Landwehr, S.; Singh, R.; El Gueddari, N.E.; Moerschbacher, B.M. Patterns matter part 1: Chitosan polymers with non-random patterns of acetylation. React. Funct. Polym. 2020, 151, 104583. [Google Scholar] [CrossRef]
- Cord-Landwehr, S.; Richter, C.; Wattjes, J.; Sreekumar, S.; Singh, R.; Basa, S.; El Gueddari, N.E.; Moerschbacher, B.M. Patterns matter part 2: Chitosan oligomers with defined patterns of acetylation. React. Funct. Polym. 2020, 151, 104577. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, A. The virtuous potential of chitosan oligosaccharide for promising biomedical applications. J. Mater. Res. 2020, 35, 1123–1134. [Google Scholar] [CrossRef]
- Ahmed, K.B.M.; Khan, M.M.A.; Siddiqui, H.; Jahan, A. Chitosan and its oligosaccharides, a promising option for sustainable crop production—A review. Carbohydr. Polym. 2020, 227, 115331. [Google Scholar] [CrossRef]
- Younes, I.; Sellimi, S.; Rinaudo, M.; Jellouli, K.; Nasri, M. Influence of acetylation degree and molecular weight of homogeneous chitosans on antibacterial and antifungal activities. Int. J. Food Microbiol. 2014, 185, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Ganan, M.; Lorentzen, S.B.; Agger, J.W.; Heyward, C.A.; Bakke, O.; Knutsen, S.H.; Aam, B.B.; Eijsink, V.G.H.; Gaustad, P.; Sørlie, M. Antifungal activity of well-defined chito-oligosaccharide preparations against medically relevant yeasts. PLoS ONE 2019, 14, 1. [Google Scholar] [CrossRef] [PubMed]
- Vander, P.; Vårum, K.M.; Domard, A.; El Gueddari, N.E.; Moerschbacher, B.M. Comparison of the Ability of Partially N-Acetylated Chitosans and Chitooligosaccharides to Elicit Resistance Reactions in Wheat Leaves. Plant Physiol. 1998, 118, 1353–1359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nietzel, T.; Elsässer, M.; Ruberti, C.; Steinbeck, J.; Ugalde, J.M.; Fuchs, P.; Wagner, S.; Ostermann, L.; Moseler, A.; Lemke, P.; et al. The fluorescent protein sensor roGFP 2-Orp1 monitors in vivo H2O2 and thiol redox integration and elucidates intracellular H2O2 dynamics during elicitor-induced oxidative burst in Arabidopsis. New Phytol. 2019, 221, 1649–1664. [Google Scholar] [CrossRef] [Green Version]
- Basa, S.; Nampally, M.; Honorato, T.; Das, S.N.; Podile, A.R.; El Gueddari, N.E.; Moerschbacher, B.M. The Pattern of Acetylation Defines the Priming Activity of Chitosan Tetramers. J. Am. Chem. Soc. 2020, 142, 1975–1986. [Google Scholar] [CrossRef]
- Cord-Landwehr, S.; Niehues, A.; Wattjes, J.; Moerschbacher, B.M. New developments in the analysis of partially acetylated chitosan polymers and oligomers. In Chitin and Chitosan: Properties and Applications; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2019; pp. 81–95. [Google Scholar] [CrossRef]
- Attjioui, M.; Gillet, D.; El Gueddari, N.E.; Moerschbacher, B.M. Synergistic antimicrobial effect of chitosan polymers and oligomers. Mol. Plant-Microbe Interact. 2021, 34, 770–778. [Google Scholar] [CrossRef]
- Rahman, H.; Shovan, L.R.; Hjeljord, L.G.; Aam, B.B.; Eijsink, V.G.H.; Sørlie, M.; Tronsmo, A. Inhibition of fungal plant pathogens by synergistic action of chito-oligosaccharides and commercially available fungicides. PLoS ONE 2014, 9, 4. [Google Scholar] [CrossRef]
- Mekahlia, S.; Bouzid, B. Chitosan-Copper (II) complex as antibacterial agent: Synthesis, characterization and coordinating bond- activity correlation study. Phys. Procedia 2009, 2, 1045–1053. [Google Scholar] [CrossRef] [Green Version]
- Hadwiger, L.A.; McBride, P.O. Low-Level Copper Plus Chitosan Applications Provide Protection Against Late Blight of Potato. Plant Health Prog. 2006, 7, 22. [Google Scholar] [CrossRef] [Green Version]
- Tantubay, S.; Mukhopadhyay, S.K.; Kalita, H.; Konar, S.; Dey, S.; Pathak, A.; Pramanik, P. Carboxymethylated chitosan-stabilized copper nanoparticles: A promise to contribute a potent antifungal and antibacterial agent. J. Nanoparticle Res. 2015, 17, 243. [Google Scholar] [CrossRef]
- Liu, W.; Qin, Y.; Liu, S.; Xing, R.; Yu, H.; Chen, X.; Li, K.; Li, P. Synthesis, characterization and antifungal efficacy of C-coordinated O-carboxymethyl chitosan Cu(II) complexes. Carbohydr. Polym. 2017, 160, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Yu, H.; Liu, W.; Qin, Y.; Xing, R.; Li, P. Integrated proteomics and metabolomics analysis reveals the antifungal mechanism of the C-coordinated O-carboxymethyl chitosan Cu(II) complex. Int. J. Biol. Macromol. 2020, 155, 1491–1509. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Gao, K.; Yu, H.; Liu, W.; Qin, Y.; Xing, R.; Liu, S.; Li, P. C-coordinated O-carboxymethyl chitosan Cu(II) complex exerts antifungal activity by disrupting the cell membrane integrity of Phytophthora capsici Leonian. Carbohydr. Polym. 2021, 261, 117821. [Google Scholar] [CrossRef] [PubMed]
- Il’Ina, A.V.; Shagdarova, B.T.; Lun’Kov, A.P.; Kulikov, S.N.; Varlamov, V.P. In vitro antifungal activity of metal complexes of a quaternized chitosan derivative with copper ions. Microbiology 2017, 86, 590–595. [Google Scholar] [CrossRef]
- Saharan, V.; Sharma, G.; Yadav, M.; Choudhary, M.K.; Sharma, S.; Pal, A.; Raliya, R.; Biswas, P. Synthesis and in vitro antifungal efficacy of Cu-chitosan nanoparticles against pathogenic fungi of tomato. Int. J. Biol. Macromol. 2015, 75, 346–353. [Google Scholar] [CrossRef]
- Omar, H.S.; Al Mutery, A.; Osman, N.H.; Reyad, N.E.-H.A.; Abou-Zeid, M.A. Genetic diversity, antifungal evaluation and molecular docking studies of Cu-chitosan nanoparticles as prospective stem rust inhibitor candidates among some Egyptian wheat genotypes. PLoS ONE 2021, 16, e0257959. [Google Scholar] [CrossRef]
- Mondéjar-López, M.; López-Jiménez, A.J.; Martínez, J.C.G.; Ahrazem, O.; Gómez-Gómez, L.; Niza, E. Thymoquinone-Loaded Chitosan Nanoparticles as Natural Preservative Agent in Cosmetic Products. Int. J. Mol. Sci. 2022, 23, 898. [Google Scholar] [CrossRef]
- Wei, L.; Tan, J.; Li, L.; Wang, H.; Liu, S.; Chen, J.; Weng, Y.; Liu, T. Chitosan/Alginate Hydrogel Dressing Loaded FGF/VE-Cadherin to Accelerate Full-Thickness Skin Regeneration and More Normal Skin Repairs. Int. J. Mol. Sci. 2022, 23, 1249. [Google Scholar] [CrossRef]
- Calvo, N.L.; Sreekumar, S.; Svetaz, L.A.; Lamas, M.C.; Moerschbacher, B.M.; Leonardi, D. Design and Characterization of Chitosan Nanoformulations for the Delivery of Antifungal Agents. Int. J. Mol. Sci. 2019, 20, 3686. [Google Scholar] [CrossRef] [Green Version]
- Cavallaro, G.; Micciulla, S.; Chiappisi, L.; Lazzara, G. Chitosan-based smart hybrid materials: A physico-chemical perspective. J. Mater. Chem. B 2021, 9, 594–611. [Google Scholar] [CrossRef]
- Ponts, N. Mycotoxins are a component of Fusarium graminearum stress-response system. Front. Microbiol. 2015, 6, 1234. [Google Scholar] [CrossRef] [Green Version]
- Muñoz, I.; Rodríguez, C.; Gillet, D.M.; Moerschbacher, B.M. Life cycle assessment of chitosan production in India and Europe. Int. J. Life Cycle Assess. 2018, 23, 1151–1160. [Google Scholar] [CrossRef] [Green Version]
- Kheiri, A.; Jorf, S.M.; Malihipour, A.; Saremi, H.; Nikkhah, M. Application of chitosan and chitosan nanoparticles for the control of Fusarium head blight of wheat (Fusarium graminearum) in vitro and greenhouse. Int. J. Biol. Macromol. 2016, 93, 1261–1272. [Google Scholar] [CrossRef] [PubMed]
- Francesconi, S.; Steiner, B.; Buerstmayr, H.; Lemmens, M.; Sulyok, M.; Balestra, G.M. Chitosan hydrochloride decreases fusarium graminearum growth and virulence and boosts growth, development and systemic acquired resistance in two durum wheat genotypes. Molecules 2020, 25, 4752. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Aliem, H.A.; Gibriel, A.Y.; Rasmy, N.M.H.; Sahab, A.F.; El-Nekeety, A.A.; Abdel-Wahhab, M.A. Antifungal efficacy of chitosan nanoparticles against phytopathogenic fungi and inhibition of zearalenone production by Fusarium graminearum. Comun. Sci. 2019, 10, 338–345. [Google Scholar] [CrossRef] [Green Version]
- Zachetti, V.G.L.; Cendoya, E.; Nichea, M.J.; Chulze, S.N.; Ramirez, M.L. Preliminary Study on the Use of Chitosan as an Eco-Friendly Alternative to Control Fusarium Growth and Mycotoxin Production on Maize and Wheat. Pathogens 2019, 8, 29. [Google Scholar] [CrossRef] [Green Version]
- Mejdoub-Trabelsi, B.; Touihri, S.; Ammar, N.; Riahi, A.; Daami-Remadi, M. Effect of chitosan for the control of potato diseases caused by Fusarium species. J. Phytopathol. 2020, 168, 18–27. [Google Scholar] [CrossRef]
- Hernández-Lauzardo, A.N.; Bautista-Baños, S.; Velázquez-Del Valle, M.; Méndez-Montealvo, M.G.; Sánchez-Rivera, M.; Bello-Pérez, L.A. Antifungal effects of chitosan with different molecular weights on in vitro development of Rhizopus stolonifer (Ehrenb.:Fr.) Vuill. Carbohydr. Polym. 2008, 73, 541–547. [Google Scholar] [CrossRef]
- Naqvi, S.; Moerschbacher, B.M. The cell factory approach toward biotechnological production of high-value chitosan oligomers and their derivatives: An update. Crit. Rev. Biotechnol. 2017, 37, 11–25. [Google Scholar] [CrossRef]
- Rahman, H.; Hjeljord, L.G.; Aam, B.B.; Sørlie, M.; Tronsmo, A. Antifungal effect of chito-oligosaccharides with different degrees of polymerization. Eur. J. Plant Pathol. 2015, 141, 147–158. [Google Scholar] [CrossRef]
- Kauss, H.; Jeblick, W.; Domard, A. The degrees of polymerization and N-acetylation of chitosan determine its ability to elicit callose formation in suspension cells and protoplasts of Catharanthus roseus. Planta 1989, 178, 385–392. [Google Scholar] [CrossRef] [PubMed]
- El Gueddari, N.E.; Kolkenbrock, S.; Schaaf, A.; Chilukoti, A.; Brunel, F.; Gorzelanny, C.; Fehser, S.; Chachra, S.; Bernard, F.; Nampally, M. Chitin and chitosan modifying enzymes: Versatile novel tools for the analysis of structure-function relationships of partially acetylated chitosans. Adv. Chitin Sci. 2014, 14, 40–47. [Google Scholar]
- Cord-Landwehr, S.; Moerschbacher, B.M. Deciphering the ChitoCode: Fungal Chitins and Chitosans as Functional Biopolymers. Fungal Biol. Biotechnol. 2021, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Dos Passos Braga, S.; Lundgren, G.A.; Macedo, S.A.; Tavares, J.F.; dos Santos Vieira, W.A.; Câmara, M.P.S.; de Souza, E.L. Application of coatings formed by chitosan and Mentha essential oils to control anthracnose caused by Colletotrichum gloesporioides and C. brevisporum in papaya (Carica papaya L.) fruit. Int. J. Biol. Macromol. 2019, 139, 631–639. [Google Scholar] [CrossRef]
- De Oliveira, K.Á.R.; Berger, L.R.R.; de Araújo, S.A.; Câmara, M.P.S.; de Souza, E.L. Synergistic mixtures of chitosan and Mentha piperita L. essential oil to inhibit Colletotrichum species and anthracnose development in mango cultivar Tommy Atkins. Food Microbiol. 2017, 66, 96–103. [Google Scholar] [CrossRef]
- Muslim, S.; AL Kadmy, I.M.; Ali, A.N.M.; Salman, B.K.; Ahmad, M.; Khazaal, S.S.; Hussein, N.H.; Muslim, S.N. Chitosan extracted from Aspergillus flavus shows synergistic effect, eases quorum sensing mediated virulence factors and biofilm against nosocomial pathogen Pseudomonas aeruginosa. Int. J. Biol. Macromol. 2018, 107, 52–58. [Google Scholar] [CrossRef]
- Jaime, M.D.; Lopez-Llorca, L.V.; Conesa, A.; Lee, A.Y.; Proctor, M.; Heisler, L.E.; Gebbia, M.; Giaever, G.; Westwood, J.T.; Nislow, C. Identification of yeast genes that confer resistance to chitosan oligosaccharide (COS) using chemogenomics. BMC Genom. 2012, 13, 1. [Google Scholar] [CrossRef] [Green Version]
- Lo, W.-H.; Deng, F.-S.; Chang, C.-J.; Lin, C.-H. Synergistic Antifungal Activity of Chitosan with Fluconazole against Candida albicans, Candida tropicalis, and Fluconazole-Resistant Strains. Molecules 2020, 25, 5114. [Google Scholar] [CrossRef]
- Le, V.T.; Bach, L.G.; Pham, T.T.; Le, N.T.T.; Ngoc, U.T.P.; Tran, D.-H.N.; Nguyen, D.H. Synthesis and antifungal activity of chitosan-silver nanocomposite synergize fungicide against Phytophthora capsici. J. Macromol. Sci. Part A 2019, 56, 522–528. [Google Scholar] [CrossRef]
- Sun, G.; Yang, Q.; Zhang, A.; Guo, J.; Liu, X.; Wang, Y.; Ma, Q. Synergistic effect of the combined bio-fungicides ε-poly-l-lysine and chitooligosaccharide in controlling grey mould (Botrytis cinerea) in tomatoes. Int. J. Food Microbiol. 2018, 276, 46–53. [Google Scholar] [CrossRef]
- Ngoc, U.T.P.; Nguyen, D.H. Synergistic antifungal effect of fungicide and chitosan-silver nanoparticles on Neoscytalidium dimidiatum. Green Process. Synth. 2018, 7, 132–138. [Google Scholar] [CrossRef]
- Zhou, Y.; Jiang, S.; Jiao, Y.; Wang, H. Synergistic effects of nanochitin on inhibition of tobacco root rot disease. Int. J. Biol. Macromol. 2017, 99, 205–212. [Google Scholar] [CrossRef]
- Husak, V. Copper and Copper-Containing Pesticides: Metabolism, Toxicity and Oxidative Stress. J. Vasyl Stefanyk Precarpathian Natl. Univ. 2015, 2, 38–50. [Google Scholar] [CrossRef]
- Hu, C.; Li, G.; Wang, Y.; Li, F.; Guo, G.; Hu, H. The effect of pH on the bonding of Cu 2+ and chitosan-montmorillonite composite. Int. J. Biol. Macromol. 2017, 103, 751–757. [Google Scholar] [CrossRef]
- Chiessi, E.; Paradossi, G.; Venanzi, M.; Pispisa, B. Copper complexes immobilized to chitosan. J. Inorg. Biochem. 1992, 46, 109–118. [Google Scholar] [CrossRef]
- Basumallick, S.; Rajasekaran, P.; Tetard, L.; Santra, S. Hydrothermally derived water-dispersible mixed valence copper-chitosan nanocomposite as exceptionally potent antimicrobial agent. J. Nanoparticle Res. 2014, 16, 2675. [Google Scholar] [CrossRef]
- Mallick, S.; Sanpui, P.; Ghosh, S.S.; Chattopadhyay, A.; Paul, A. Synthesis, characterization and enhanced bactericidal action of a chitosan supported core-shell copper-silver nanoparticle composite. RSC Adv. 2015, 5, 12268–12276. [Google Scholar] [CrossRef]
- Raut, A.; Yadav, H.; Gnanamani, A.; Pushpavanam, S.; Pawar, S. Synthesis and characterization of chitosan-TiO2:Cu nanocomposite and their enhanced antimicrobial activity with visible light. Colloids Surf. B Biointerfaces 2016, 148, 566–575. [Google Scholar] [CrossRef]
- Lamichhane, J.R.; Osdaghi, E.; Behlau, F.; Köhl, J.; Jones, J.B.; Aubertot, J.-N. Thirteen decades of antimicrobial copper compounds applied in agriculture. A review. Agron. Sustain. Dev. 2018, 38, 28. [Google Scholar] [CrossRef] [Green Version]
- Schatz, C.; Viton, C.; Delair, T.; Pichot, C.; Domard, A. Typical Physicochemical Behaviors of Chitosan in Aqueous Solution. Biomacromolecules 2003, 4, 641–648. [Google Scholar] [CrossRef]
- Hirai, A.; Odani, H.; Nakajima, A. Determination of degree of deacetylation of chitosan by 1H NMR spectroscopy. Polym. Bull. 1991, 26, 87–94. [Google Scholar] [CrossRef]
- Kasaai, M.R. Determination of the degree of N-acetylation for chitin and chitosan by various NMR spectroscopy techniques: A review. Carbohydr. Polym. 2010, 79, 801–810. [Google Scholar] [CrossRef]
- Pontecorvo, G.; Roper, J.; Chemmons, L.; Macdonald, K.; Bufton, A. The Genetics of Aspergillus nidulans. Adv. Genet. 1953, 5, 141–238. [Google Scholar] [CrossRef]
- Cappellini, R.A.; Peterson, J.L. Macroconidium Formation in Submerged Cultures by a Non-Sporulating Strain of Gibberella zeae. Mycologia 1965, 57, 962. [Google Scholar] [CrossRef]
- Oliveira, E.N.; El Gueddari, N.E.; Moerschbacher, B.M.; Peter, M.G.; Franco, T.T. Growth of Phytopathogenic Fungi in the Presence of Partially Acetylated Chitooligosaccharides. Mycopathologia 2008, 166, 163–174. [Google Scholar] [CrossRef] [Green Version]
- Cohen, Y.; Levy, Y. Joint action of fungicides in Mixtures: Theory and practice. Phytoparasitica 1990, 18, 159–169. [Google Scholar] [CrossRef]
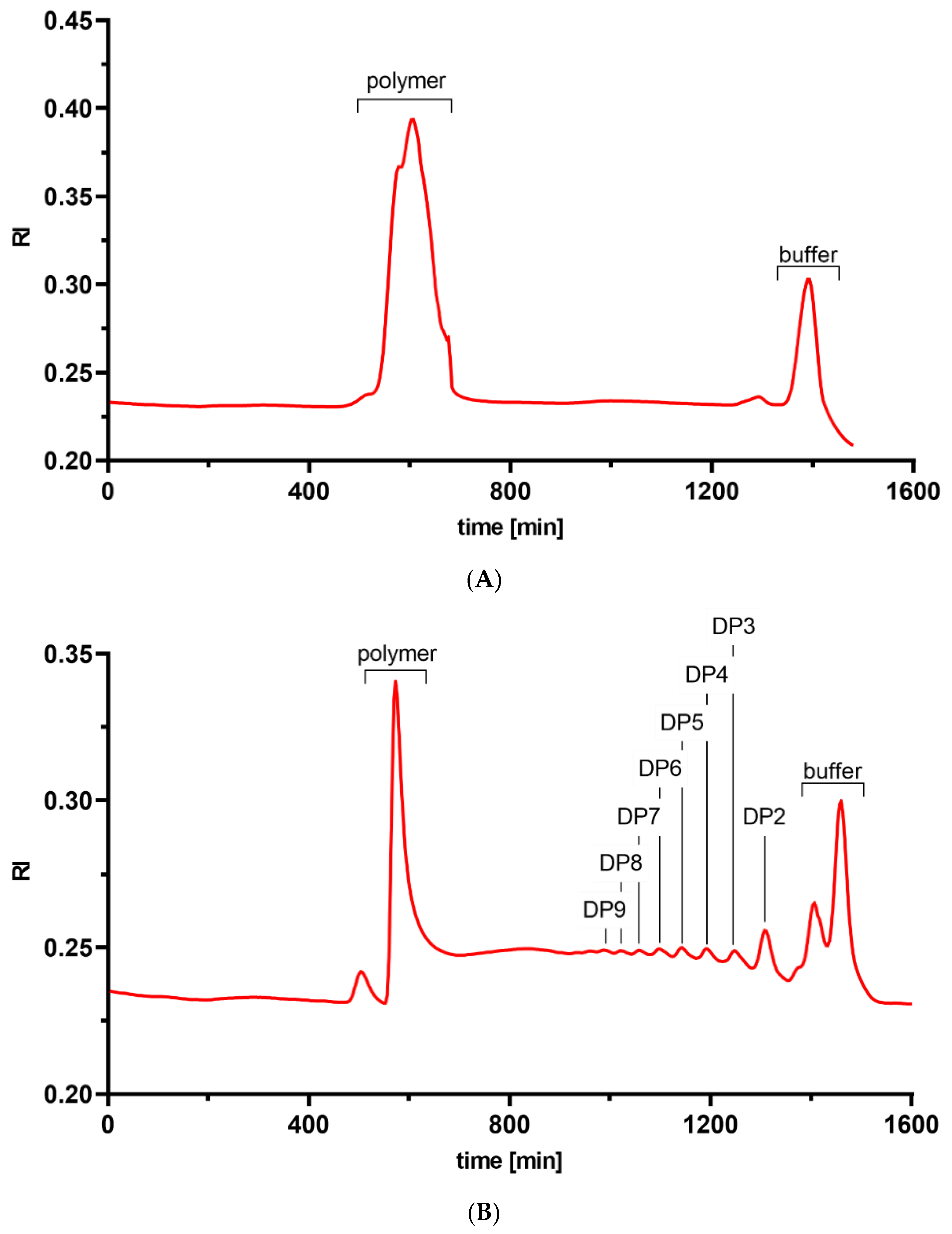
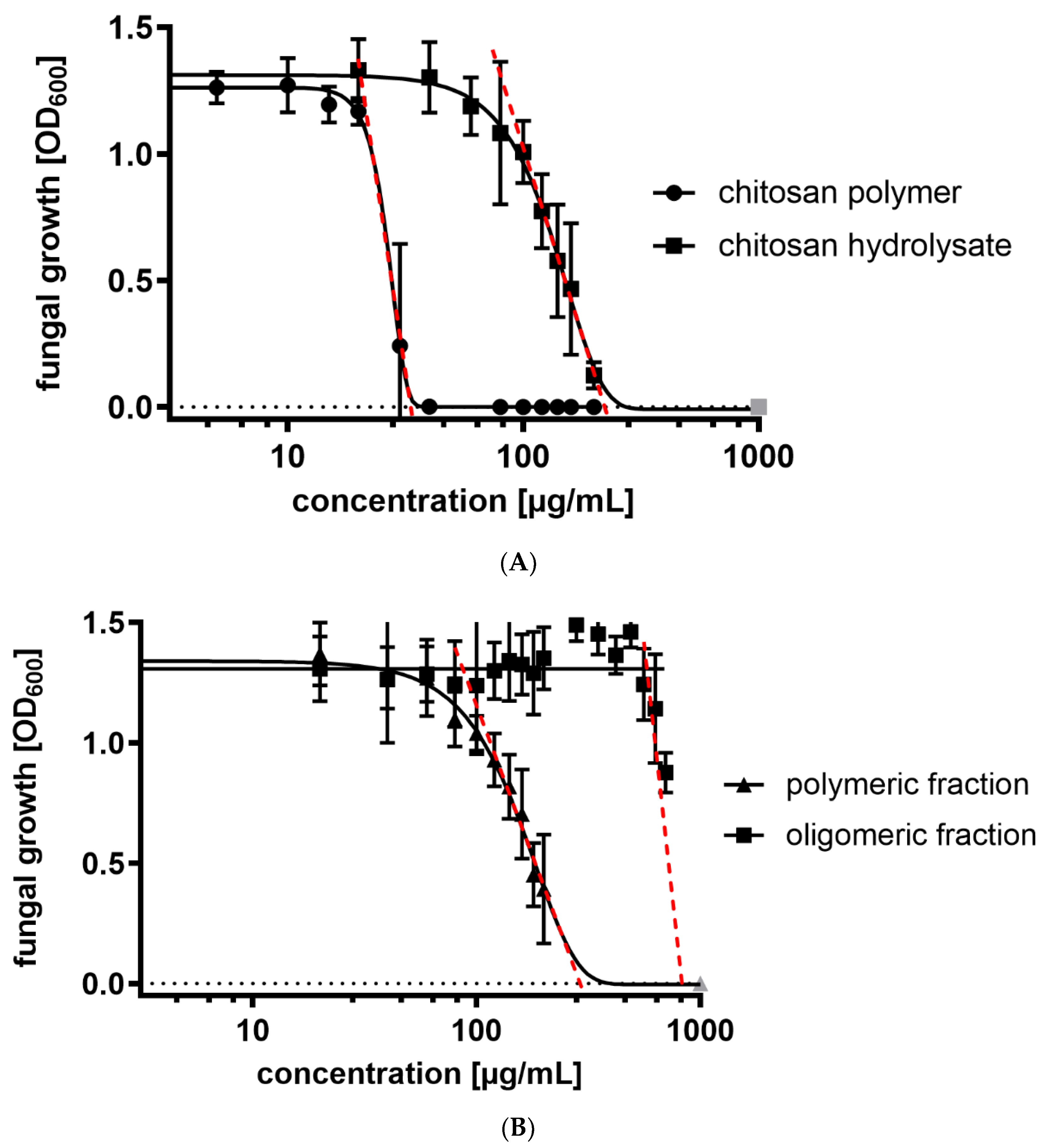
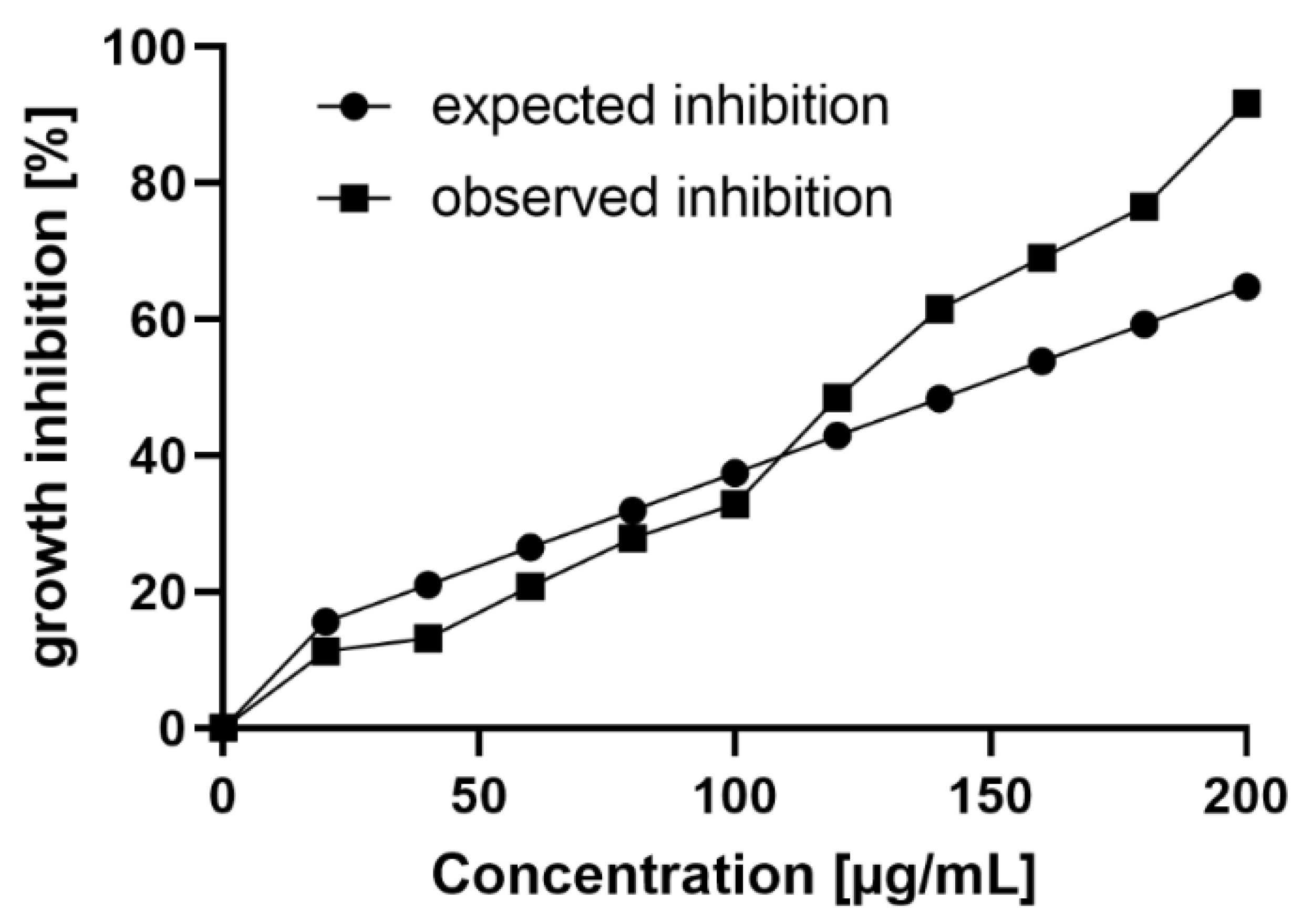
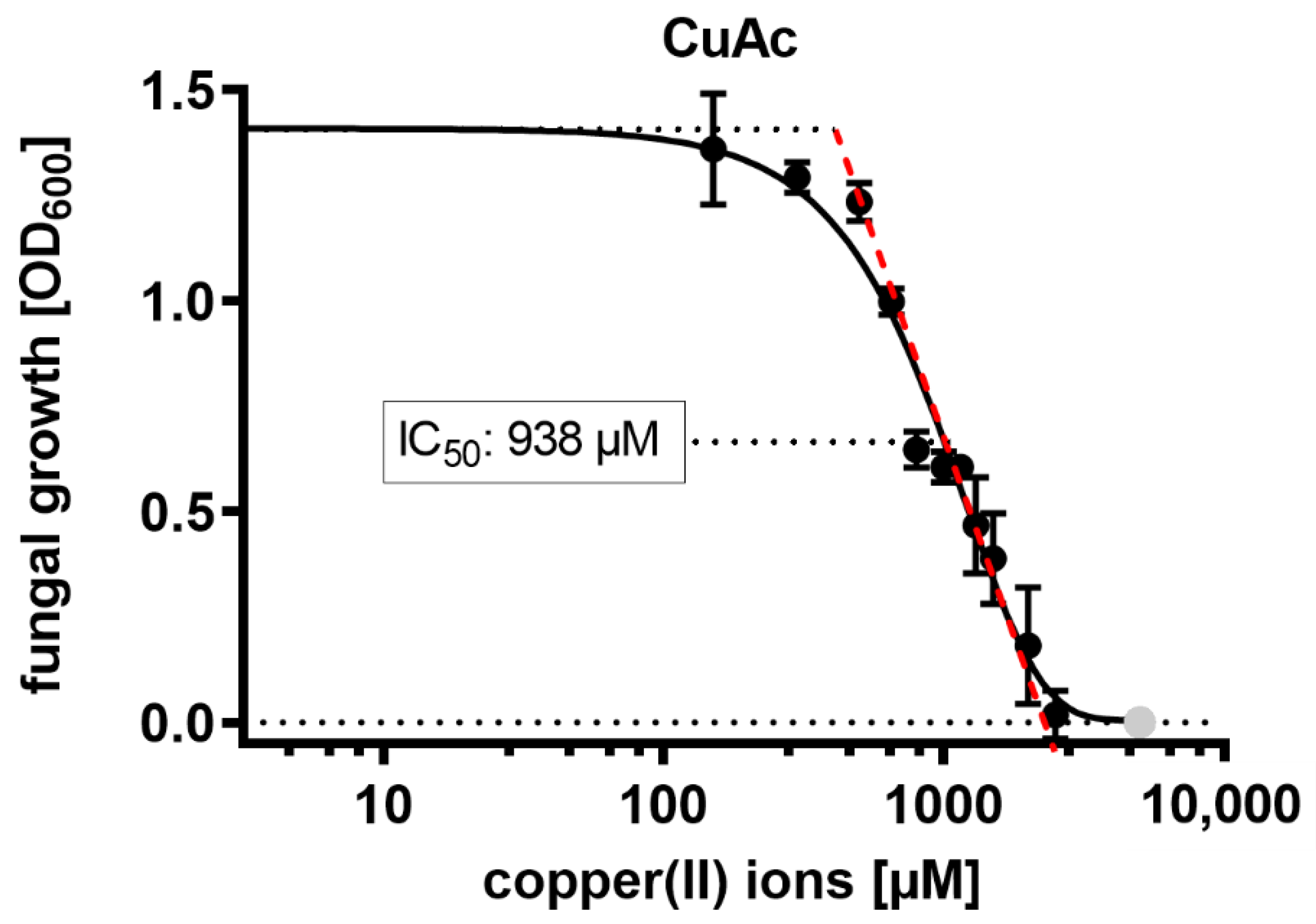
| Chitosan | FA | w.a. Mw (kDa) | w.a. DP | Đ |
|---|---|---|---|---|
| chitosan polymer | 0.2 | 58.7 | 347 | 2.2 |
| chitosan hydrolysate a | 0.2 | n/a | n/a | n/a |
| polymeric fraction b | 0.2 | 43.3 | 255 | 1.1 |
| oligomeric fraction b | 0.2 c | n/a | 2-15 | n/a |
| Compound | IC50 (µg mL−1) |
|---|---|
| chitosan polymer | 26 |
| chitosan hydrolysate | 132 |
| polymeric fraction | 155 |
| oligomeric fraction | 739 |
| Combination | Chitosan Hydrolysate µg/mL | Copper(II) Ions µM | Cobs % | Cexp % | Synergy Factor Cobs/Cexp |
|---|---|---|---|---|---|
| 1 | 20 | 1000 | 79.8 | 47.3 | 1.7 |
| 2 | 60 | 500 | 68.2 | 33.5 | 2.0 |
| 3 | 80 | 250 | 45.7 | 18.1 | 2.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lemke, P.; Jünemann, L.; Moerschbacher, B.M. Synergistic Antimicrobial Activities of Chitosan Mixtures and Chitosan–Copper Combinations. Int. J. Mol. Sci. 2022, 23, 3345. https://doi.org/10.3390/ijms23063345
Lemke P, Jünemann L, Moerschbacher BM. Synergistic Antimicrobial Activities of Chitosan Mixtures and Chitosan–Copper Combinations. International Journal of Molecular Sciences. 2022; 23(6):3345. https://doi.org/10.3390/ijms23063345
Chicago/Turabian StyleLemke, Philipp, Lena Jünemann, and Bruno M. Moerschbacher. 2022. "Synergistic Antimicrobial Activities of Chitosan Mixtures and Chitosan–Copper Combinations" International Journal of Molecular Sciences 23, no. 6: 3345. https://doi.org/10.3390/ijms23063345
APA StyleLemke, P., Jünemann, L., & Moerschbacher, B. M. (2022). Synergistic Antimicrobial Activities of Chitosan Mixtures and Chitosan–Copper Combinations. International Journal of Molecular Sciences, 23(6), 3345. https://doi.org/10.3390/ijms23063345






