Poly-Alanine-ε-Caprolacton-Methacrylate as Scaffold Material with Tuneable Biomechanical Properties for Osteochondral Implants
Abstract
:1. Introduction
2. Results and Discussion
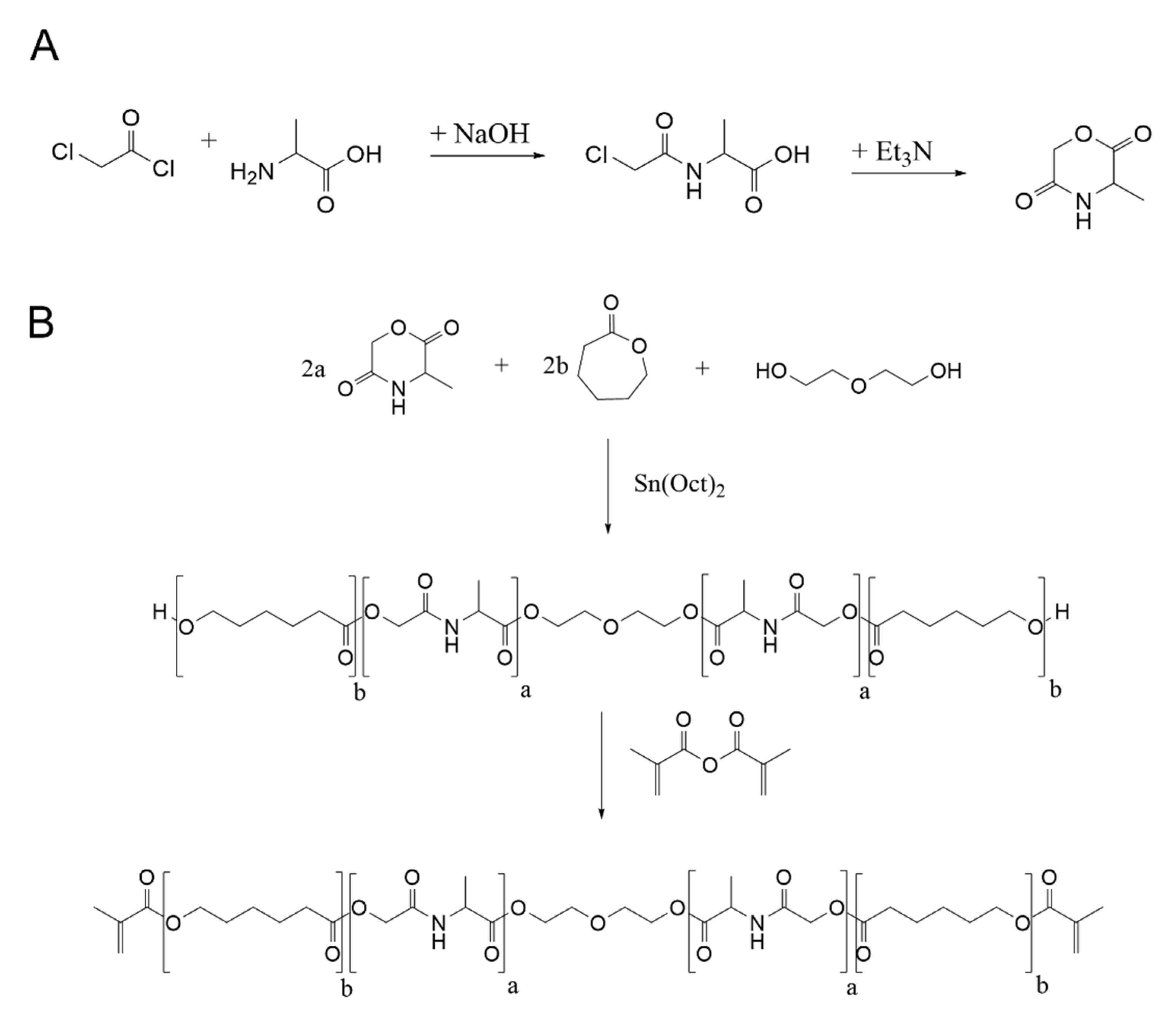
2.1. Synthesis of 2PP Processable Polymers
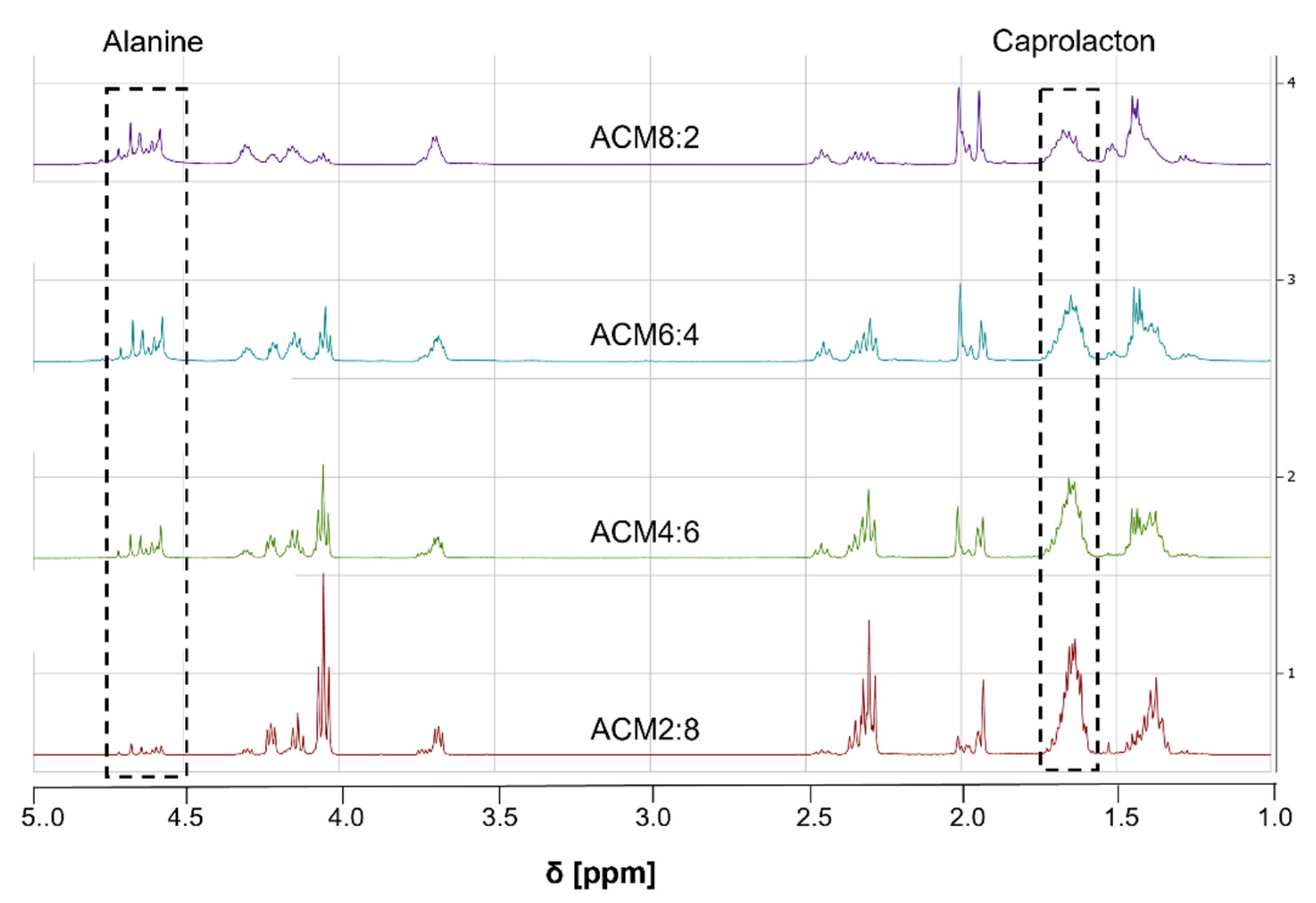
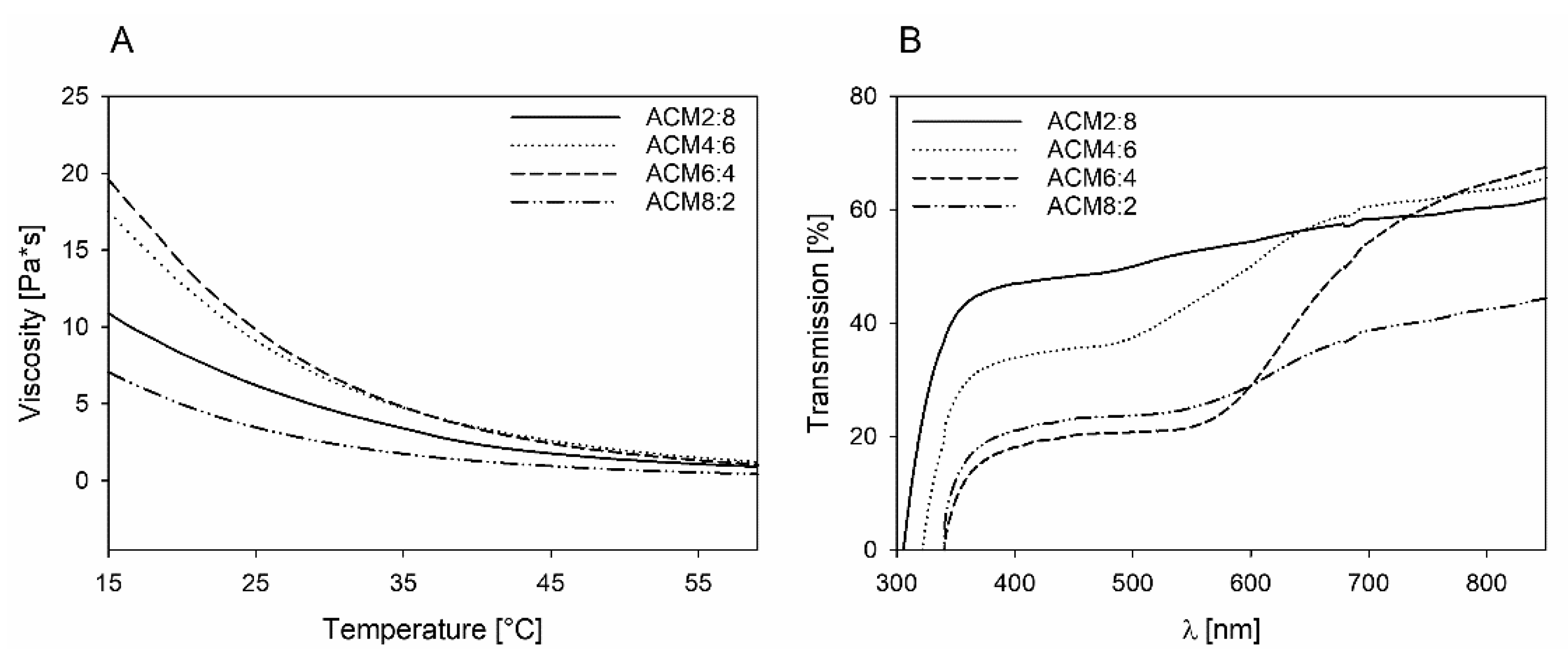
2.2. Physicochemical Properties of the Polymers
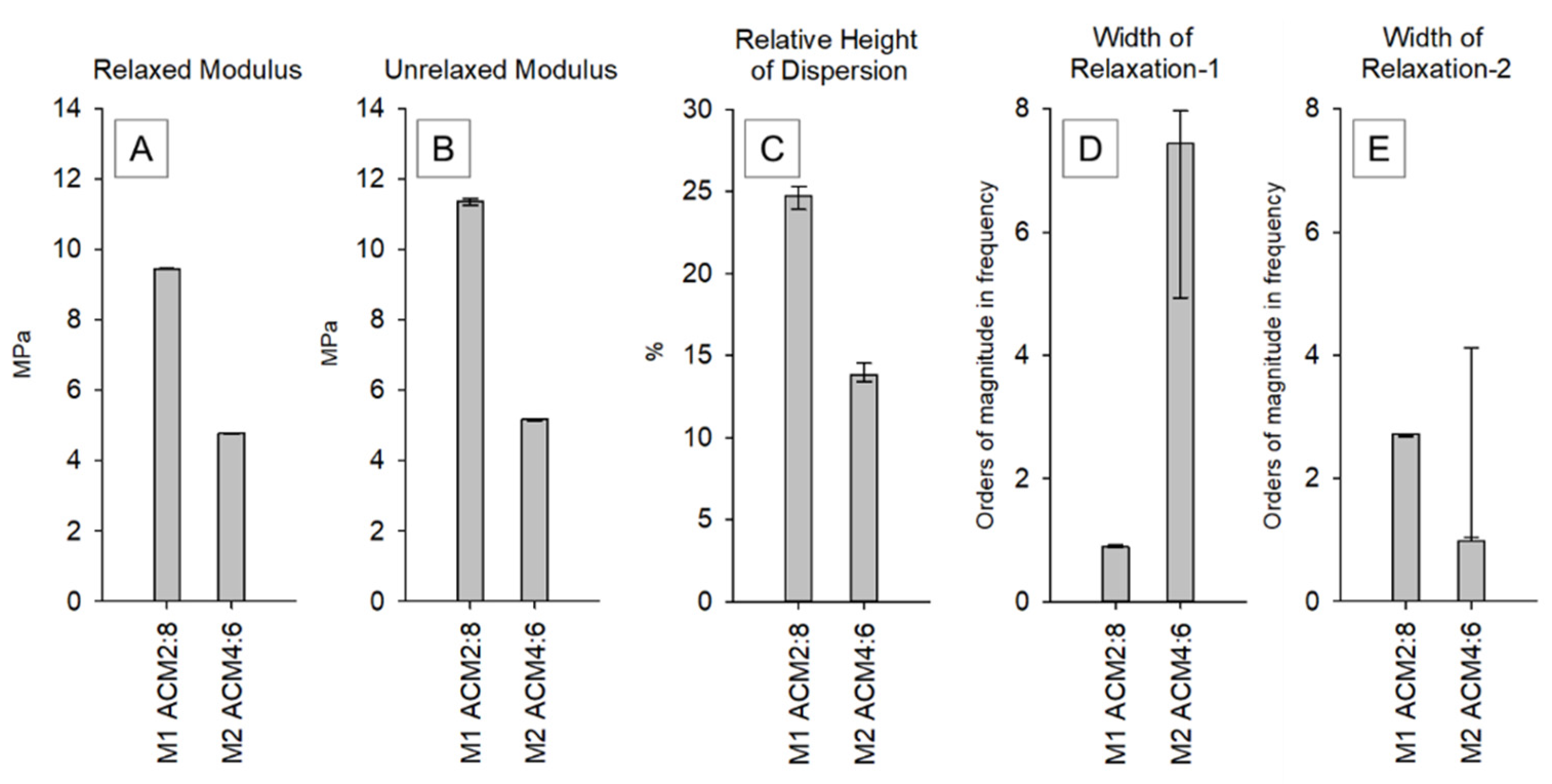
2.3. Viscoelastic Properties of ACM Copolymers
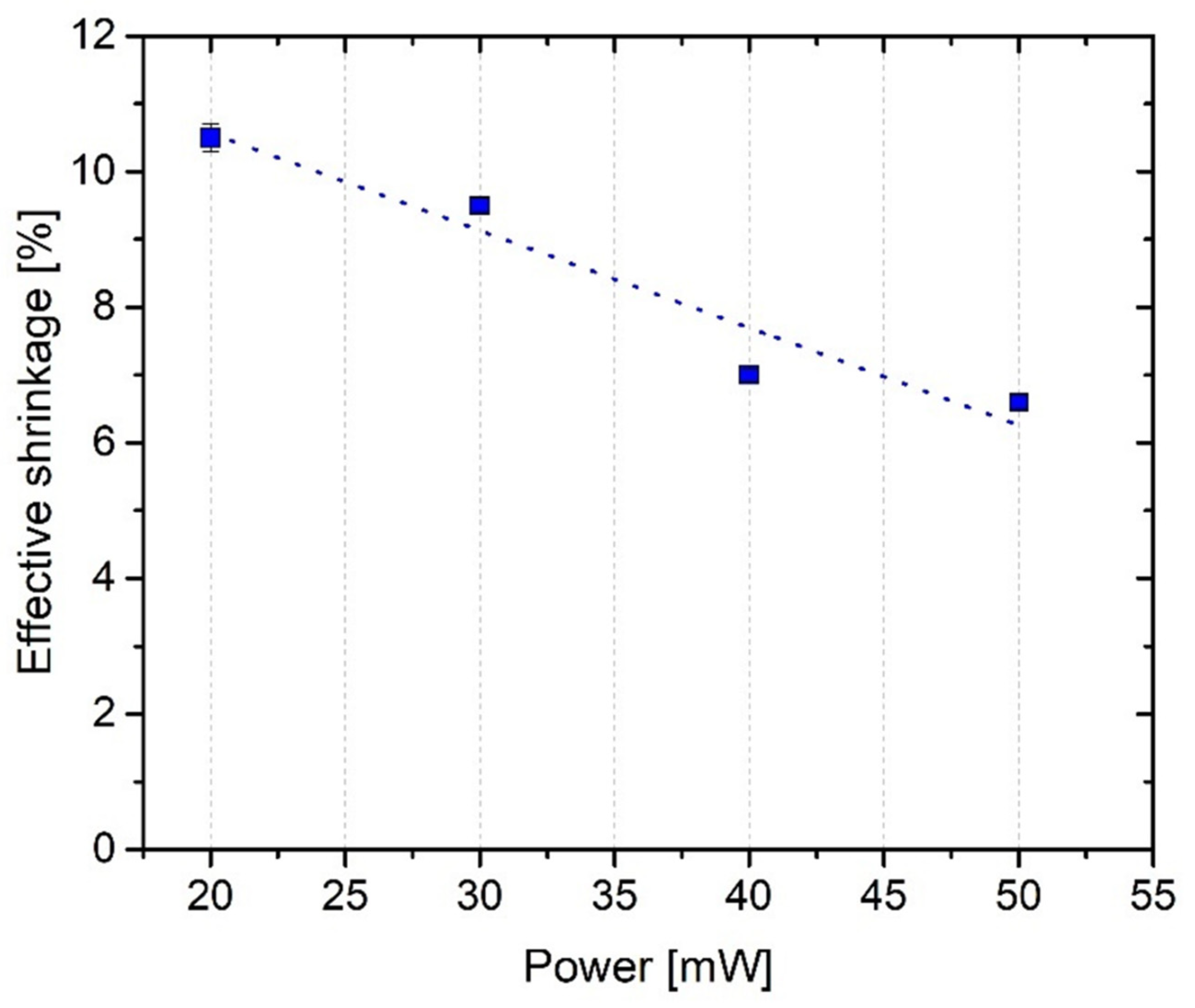
2.4. PP Structuring of ACM
2.5. Proliferation of Mesenchymal Stem Cells
3. Materials and Methods
3.1. Materials
3.2. Synthesis of 3-Methyl-morpholine-2,5-dione
3.3. Synthesis of Poly-(alanine-co-ε-caprolacton)-dimethacrylate (ACM 2:8 and ACM 4:6)
3.4. Synthesis of Poly-(alanine-co-ε-caprolacton)-dimethacrylate (ACM 6:4 and ACM 8:2)
3.5. Molecular Characterisation
3.6. Contact Angle and Swelling Behavior
3.7. Mechanical Measurement
3.8. Two-Photon Polymerization (2PP) Setup and Processing
3.9. Cultivation of Mesenchymal Stem Cells
3.9.1. Coating
3.9.2. Cell Cultivation
3.9.3. Staining
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Winkler, T.; Sass, F.A.; Duda, G.N.; Schmidt-Bleek, K. A review of biomaterials in bone defect healing, remaining shortcomings and future opportunities for bone tissue engineering: The unsolved challenge. Bone Joint Res. 2018, 7, 232–243. [Google Scholar] [CrossRef]
- Karpinski, K.; Häner, M.; Bierke, S.; Petersen, W. Matrix-induced chondrogenesis is a valid and safe cartilage repair option for small- to medium-sized cartilage defects of the knee: A systematic review. Knee Surg. Sports Traumatol. Arthrosc. 2021, 29, 4213–4222. [Google Scholar] [CrossRef] [PubMed]
- Roffi, A.; Krishnakumar, G.S.; Gostynska, N.; Kon, E.; Candrian, C.; Filardo, G. The Role of Three-Dimensional Scaffolds in Treating Long Bone Defects: Evidence from Preclinical and Clinical Literature—A Systematic Review. BioMed Res. Int. 2017, 2017, 8074178. [Google Scholar] [CrossRef] [PubMed]
- Morouco, P.; Fernandes, C.; Lattanzi, W. Challenges and Innovations in Osteochondral Regeneration: Insights from Biology and Inputs from Bioengineering toward the Optimization of Tissue Engineering Strategies. J. Funct. Biomater. 2021, 12, 17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Huang, J.; Narayan, R.J. Gradient scaffolds for osteochondral tissue engineering and regeneration. J. Mater. Chem. B 2020, 8, 8149–8170. [Google Scholar] [CrossRef]
- van Osch, G.J.; Brittberg, M.; Dennis, J.E.; Bastiaansen-Jenniskens, Y.M.; Erben, R.G.; Konttinen, Y.T.; Luyten, F.P. Cartilage repair: Past and future—Lessons for regenerative medicine. J. Cell. Mol. Med. 2009, 13, 792–810. [Google Scholar] [CrossRef] [Green Version]
- Shen, S.; Chen, M.; Guo, W.; Li, H.; Li, X.; Huang, S.; Luo, X.; Wang, Z.; Wen, Y.; Yuan, Z.; et al. Three Dimensional Printing-Based Strategies for Functional Cartilage Regeneration. Tissue Eng. Part B Rev. 2019, 25, 187–201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, H.B.; Kawata, S. Two-Photon Photopolymerization and 3D Lithographic Microfabrication, NMR, 3D Analysis, Photopolymerization. Adv. Polym. Sci. 2004, 170, 5. [Google Scholar]
- Zhou, X.; Hou, Y.H.; Lin, J. A review on the processing accuracy of two-photon polymerization. AIP Adv. 2015, 5, 030701. [Google Scholar] [CrossRef]
- Hauptmann, N.; Lian, Q.; Ludolph, J.; Rothe, H.; Hildebrand, G.; Liefeith, K. Biomimetic Designer Scaffolds Made of D,L-Lactide-varepsilon-Caprolactone Polymers by 2-Photon Polymerization. Tissue Eng. Part B Rev. 2019, 25, 167–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melissinaki, V.; Gill, A.A.; Ortega, I.; Vamvakaki, M.; Ranella, A.; Haycock, J.W.; Fotakis, C.; Farsari, M.; Claeyssens, F. Direct laser writing of 3D scaffolds for neural tissue engineering applications. Biofabrication 2011, 3, 045005. [Google Scholar] [CrossRef] [PubMed]
- Bothe, F.; Deubel, A.K.; Hesse, E.; Lotz, B.; Groll, J.; Werner, C.; Richter, W.; Hagmann, S. Treatment of Focal Cartilage Defects in Minipigs with Zonal Chondrocyte/Mesenchymal Progenitor Cell Constructs. Int. J. Mol. Sci. 2019, 20, 653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ovsianikov, A.; Malinauskas, M.; Schlie, S.; Chichkov, B.; Gittard, S.; Narayan, R.; Lobler, M.; Sternberg, K.; Schmitz, K.P.; Haverich, A. Three-dimensional laser micro- and nano-structuring of acrylated poly(ethylene glycol) materials and evaluation of their cytoxicity for tissue engineering applications. Acta Biomater. 2011, 7, 967–974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Felfel, R.M.; Poocza, L.; Gimeno-Fabra, M.; Milde, T.; Hildebrand, G.; Ahmed, I.; Scotchford, C.; Sottile, V.; Grant, D.M.; Liefeith, K. In vitro degradation and mechanical properties of PLA-PCL copolymer unit cell scaffolds generated by two-photon polymerization. Biomed. Mater. 2016, 11, 015011. [Google Scholar] [CrossRef] [PubMed]
- Gelinsky, M. Mineralised Collagen as Biomaterial and Matrix for Bone Tissue Engineering. In Fundamentals od Tissue Engineering and Regenerative Medicine; Meyer, U., Meyer, T., Handschel, J., Wiesmann, H.P., Eds.; Springer: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Van Hoorick, J.; Gruber, P.; Markovic, M.; Tromayer, M.; Van Erps, J.; Thienpont, H.; Liska, R.; Ovsianikov, A.; Dubruel, P.; Van Vlierberghe, S. Cross-Linkable Gelatins with Superior Mechanical Properties Through Carboxylic Acid Modification: Increasing the Two-Photon Polymerization Potential. Biomacromolecules 2017, 18, 3260–3272. [Google Scholar] [CrossRef] [PubMed]
- Levett, P.A.; Melchels, F.P.; Schrobback, K.; Hutmacher, D.W.; Malda, J.; Klein, T.J. A biomimetic extracellular matrix for cartilage tissue engineering centered on photocurable gelatin, hyaluronic acid and chondroitin sulfate. Acta Biomater. 2014, 10, 214–223. [Google Scholar] [CrossRef] [Green Version]
- Erickson, A.E.; Sun, J.; Lan Levengood, S.K.; Swanson, S.; Chang, F.C.; Tsao, C.T.; Zhang, M. Chitosan-based composite bilayer scaffold as an in vitro osteochondral defect regeneration model. Biomed. Microdevices 2019, 21, 34. [Google Scholar] [CrossRef] [PubMed]
- Polacco, M.A.; Kahng, P.W.; Sudoko, C.K.; Gosselin, B.J. Orbital Floor Reconstruction: A Comparison of Outcomes between Absorbable and Permanent Implant Systems. Craniomaxillofac. Trauma Reconstr. 2019, 12, 193–198. [Google Scholar] [CrossRef]
- Rodrigues-Galan, A.; Franco, L.; Puiggali, J. Degradable Poly(ester amide)s for Biomedical Applications. Polymers 2011, 3, 35. [Google Scholar] [CrossRef] [Green Version]
- Elomaa, L.; Kang, Y.; Seppälä, J.V.; Yang, Y. Biodegradable Photocrosslinkable Poly(depsipeptide-co-e-caprolactone) for Tissue Engineering: Synthesis, Characterization, and In Vitro Evaluation. J. Polym. Sci. Part A Polym. Chem. 2014, 52, 3307–3315. [Google Scholar] [CrossRef]
- Ohya, Y.; Nakai, T.; Nagahama, K.; Ouchi, T. The Synthesis and Biodegradability of Poly(lactide-random-depsipeptide)-PEG-Poly(lactide-random-depsipeptide) ABA-type Triblock Copolymers. J. Bioact. Compat. Polym. 2006, 21, 557–577. [Google Scholar] [CrossRef]
- John, G.; Tsuda, S.; Morita, M. Synthesis and Modification of New Biodegradable Copolymers: Serine/Glycolic acid Based Copolymers. J. Polym. Sci. A Polym. Chem. 1997, 35, 1901–1907. [Google Scholar] [CrossRef]
- Ouchi, T.; Nozaki, T.; Ishikawa, A.; Fujimoto, I.; Ohya, Y. Synthesis and Enzymatic Hydrolysis of Lactic Acid-Depsipeptide Copolymers with Functionalized Pendant Groups. J. Polym. Sci. A Polym. Chem. 1997, 35, 377–383. [Google Scholar] [CrossRef]
- Zandrini, T.; Liaros, N.; Jiang, L.J.; Lu, Y.F.; Fourkas, J.T.; Osellame, R.; Baldacchini, T. Effect of the resin viscosity on the writing properties of two-photon polymerization. Opt. Mater. Express 2019, 9, 2601–2616. [Google Scholar] [CrossRef]
- Mueller, J.B.; Fischer, J.; Mayer, F.; Kadic, M.; Wegener, M. Polymerization kinetics in three-dimensional direct laser writing. Adv. Mater. 2014, 26, 6566–6571. [Google Scholar] [CrossRef] [PubMed]
- Burmeister, F.; Steenhusen, S.; Houbertz, R.; Zeitner, U.D.; Nolte, S.; Tünnermann, A. Materials and technologies for fabrication of three-dimensional microstructures with sub-100 nm feature sizes by two-photon polymerization. J. Appl. Laser Appl. 2012, 24, 042014. [Google Scholar] [CrossRef] [Green Version]
- Ovsianikov, A.; Viertl, J.; Chichkov, B.; Oubaha, M.; MacCraith, B.; Sakellari, I.; Giakoumaki, A.; Gray, D.; Vamvakaki, M.; Farsari, M.; et al. Ultra-low shrinkage hybrid photosensitive material for two-photon polymerization microfabrication. ACS Nano 2008, 2, 2257–2262. [Google Scholar] [CrossRef]
- Sugioka, K.; Xu, J.; Wu, D.; Hanada, Y.; Wang, Z.; Cheng, Y.; Midorikawa, K. Femtosecond laser 3D micromachining: A powerful tool for the fabrication of microfluidic, optofluidic, and electrofluidic devices based on glass. Lab Chip 2014, 14, 3447–3458. [Google Scholar] [CrossRef]
- Vogler, E.A. Protein adsorption in three dimensions. Biomaterials 2012, 33, 1201–1237. [Google Scholar] [CrossRef] [Green Version]
- McMahon, R.E.; Wang, L.; Skoracki, R.; Mathur, A.B. Development of nanomaterials for bone repair and regeneration. J Biomed. Mater. Res. B Appl. Biomater. 2013, 101, 387–397. [Google Scholar] [CrossRef]
- Wang, C.; Huang, W.; Zhou, Y.; He, L.; He, Z.; Chen, Z.; He, X.; Tian, S.; Liao, J.; Lu, B.; et al. 3D printing of bone tissue engineering scaffolds. Bioact. Mater. 2020, 5, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Xiao, Y. Biomaterials Regulating Bone Hematoma for Osteogenesis. Adv. Healthc. Mater. 2020, 9, e2000726. [Google Scholar] [CrossRef]
- Samadian, H.; Farzamfar, S.; Vaez, A.; Ehterami, A.; Bit, A.; Alam, M.; Goodarzi, A.; Darya, G.; Salehi, M. A tailored polylactic acid/polycaprolactone biodegradable and bioactive 3D porous scaffold containing gelatin nanofibers and Taurine for bone regeneration. Sci. Rep. 2020, 10, 13366. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Feng, B.; Zhang, W.; Yan, C.; Yao, Q.; Shao, C.; Yu, F.; Li, F.; Fu, Y. Electrospun gelatin/PCL and collagen/PCL scaffolds for modulating responses of bone marrow endothelial progenitor cells. Exp. Ther. Med. 2018, 17, 3717–3726. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ho-Shui-Ling, A.; Bolander, J.; Rustom, L.E.; Johnson, A.W.; Luyten, F.P.; Picart, C. Bone regeneration strategies: Engineered scaffolds, bioactive molecules and stem cells current stage and future perspectives. Biomaterials 2018, 180, 143–162. [Google Scholar] [CrossRef] [PubMed]
- Rothe, H.; Rost, J.; Kramer, F.; Alkhatib, Y.; Petzold-Welcke, K.; Klemm, D.; Fischer, D.; Liefeith, K. Bacterial nanocellulose: Reinforcement of compressive strength using an adapted Mobile Matrix Reservoir Technology and suitable post-modification strategies. J. Mech. Behav. Biomed. Mater. 2022, 125, 104978. [Google Scholar] [CrossRef]
- Kampleitner, C.; Changi, K.; Felfel, R.M.; Scotchford, C.A.; Sottile, V.; Kluger, R.; Hoffmann, O.; Grant, D.M.; Epstein, M.M. Preclinical biological and physicochemical evaluation of two-photon engineered 3D biomimetic copolymer scaffolds for bone healing. Biomater. Sci. 2020, 8, 1683–1694. [Google Scholar] [CrossRef]
- Haussling, V.; Deninger, S.; Vidoni, L.; Rinderknecht, H.; Ruoss, M.; Arnscheidt, C.; Athanasopulu, K.; Kemkemer, R.; Nussler, A.K.; Ehnert, S. Impact of Four Protein Additives in Cryogels on Osteogenic Differentiation of Adipose-Derived Mesenchymal Stem Cells. Bioengineering 2019, 6, 67. [Google Scholar] [CrossRef] [Green Version]
- Hong, J.K.; Cooke, S.L.; Whittington, A.R.; Roman, M. Bioactive Cellulose Nanocrystal-Poly(epsilon-Caprolactone) Nanocomposites for Bone Tissue Engineering Applications. Front. Bioeng. Biotechnol. 2021, 9, 605924. [Google Scholar] [CrossRef]
- Portan, D.V.; Ntoulias, C.; Mantzouranis, G.; Fortis, A.P.; Deligianni, D.D.; Polyzos, D.; Kostopoulos, V. Gradient 3D Printed PLA Scaffolds on Biomedical Titanium: Mechanical Evaluation and Biocompatibility. Polymers 2021, 13, 682. [Google Scholar] [CrossRef]
- Krupp, A.; Dupuis, Y.; Kohlhaas, S.; Lechner, J.; Winkler, M.; Houbertz, R.; Hildebrand, G.; Hauptmann, N.; Rost, J.; Liefeith, K.; et al. Bone cartiladge transplats fabricated by TPA based on biocompatible polymers. SPIE Photonics West 2021, 2, 116760B. [Google Scholar]
- Carlotti, M.; Mattoli, V. Functional Materials for Two-Photon Polymerization in Microfabrication. Small 2019, 15, e1902687. [Google Scholar] [CrossRef] [PubMed]





| Polymer | Alanin (a) | Caprolactone (b) | Molar Mass [g/mol] |
|---|---|---|---|
| ACM 2:8 | 1.3 | 7.2 | 1183 |
| ACM4:6 | 2.9 | 5.6 | 1208 |
| ACM6:4 | 4.3 | 3.9 | 1188 |
| ACM8:2 | 5.4 | 2.2 | 1161 |
| Material Platform | Application | Methodology | Mechanical Property Range Investigated | Ref. |
|---|---|---|---|---|
| Poly-Lactide/ Caprolactone Copolymer | bone | compression; 0.5 mmxmin−1; 20% strain | 180–5500 kPa compressive strength | [38] |
| PolyHEMA Composite | bone | compression; 5 mmxmin−1; 10% strain | 60–190 kPa compressive strength | [39] |
| Cellulose/ Poly-Capro- lactone Composite | bone | tension; 10 mmxmin−1; strain not specified | 10–18 MPa tensile strength | [40] |
| Poly-Lactide Composite | bone | microtensile testing; parameter not specified | 1.8–6.5 MPa tensile strength | [41] |
| Poly-Alanine-ε-Caprolacton- Methacrylate Copolymer | bone cartilage | stress relaxation after 20% initial compressive strain | 230–400 kPa complex modulus | this study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hauptmann, N.; Ludolph, J.; Rothe, H.; Rost, J.; Krupp, A.; Lechner, J.; Kohlhaas, S.; Winkler, M.; Stender, B.; Hildebrand, G.; et al. Poly-Alanine-ε-Caprolacton-Methacrylate as Scaffold Material with Tuneable Biomechanical Properties for Osteochondral Implants. Int. J. Mol. Sci. 2022, 23, 3115. https://doi.org/10.3390/ijms23063115
Hauptmann N, Ludolph J, Rothe H, Rost J, Krupp A, Lechner J, Kohlhaas S, Winkler M, Stender B, Hildebrand G, et al. Poly-Alanine-ε-Caprolacton-Methacrylate as Scaffold Material with Tuneable Biomechanical Properties for Osteochondral Implants. International Journal of Molecular Sciences. 2022; 23(6):3115. https://doi.org/10.3390/ijms23063115
Chicago/Turabian StyleHauptmann, Nicole, Johanna Ludolph, Holger Rothe, Jürgen Rost, Alexander Krupp, Jörg Lechner, Svenja Kohlhaas, Manuela Winkler, Benedikt Stender, Gerhard Hildebrand, and et al. 2022. "Poly-Alanine-ε-Caprolacton-Methacrylate as Scaffold Material with Tuneable Biomechanical Properties for Osteochondral Implants" International Journal of Molecular Sciences 23, no. 6: 3115. https://doi.org/10.3390/ijms23063115
APA StyleHauptmann, N., Ludolph, J., Rothe, H., Rost, J., Krupp, A., Lechner, J., Kohlhaas, S., Winkler, M., Stender, B., Hildebrand, G., & Liefeith, K. (2022). Poly-Alanine-ε-Caprolacton-Methacrylate as Scaffold Material with Tuneable Biomechanical Properties for Osteochondral Implants. International Journal of Molecular Sciences, 23(6), 3115. https://doi.org/10.3390/ijms23063115






