Bisphenol A (BPA) Leading to Obesity and Cardiovascular Complications: A Compilation of Current In Vivo Study
Abstract
1. Introduction
Bisphenol A
2. Methods
2.1. Search Strategy
2.2. Inclusion Criteria
2.3. Exclusion Criteria
3. Results
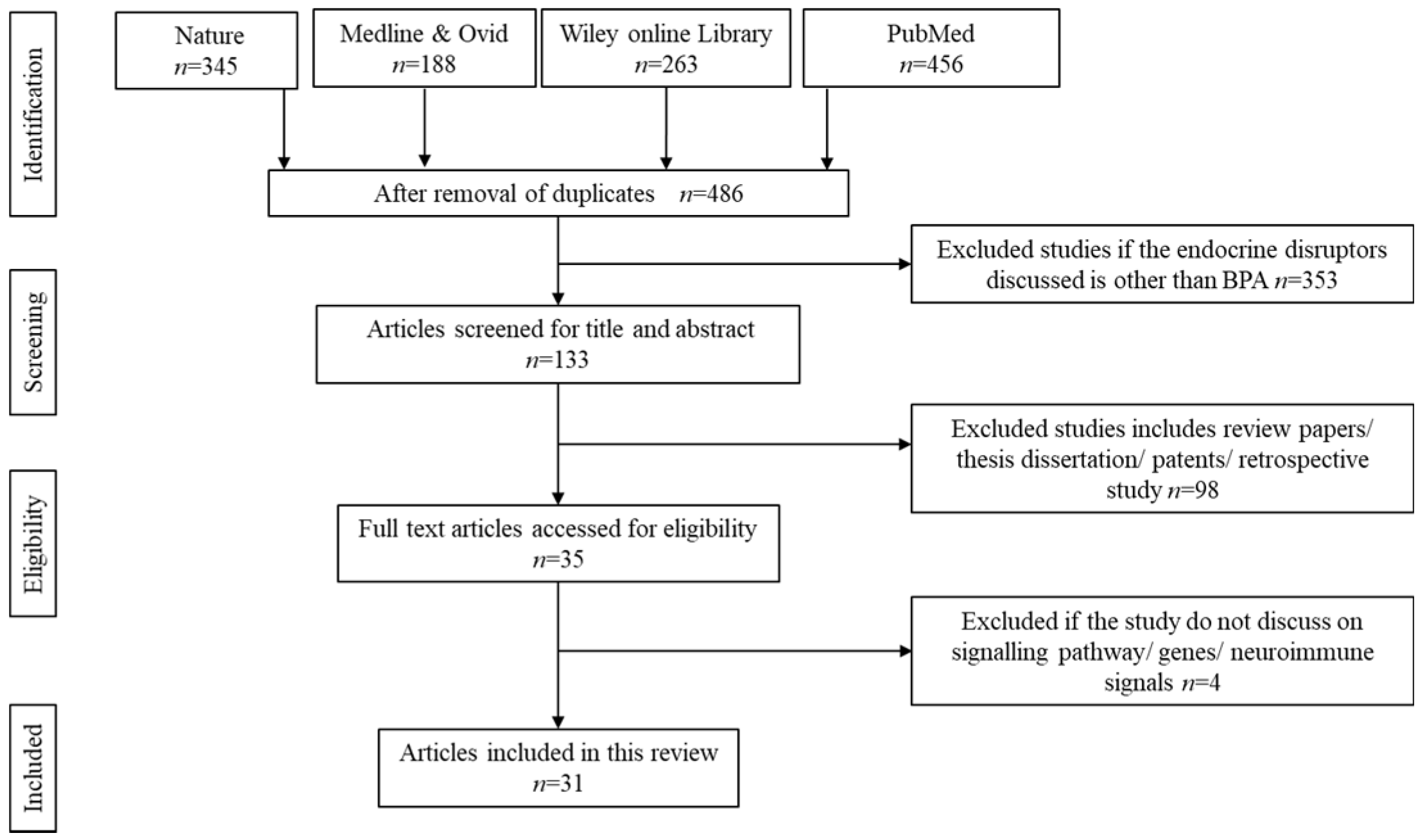
3.1. Literature Search
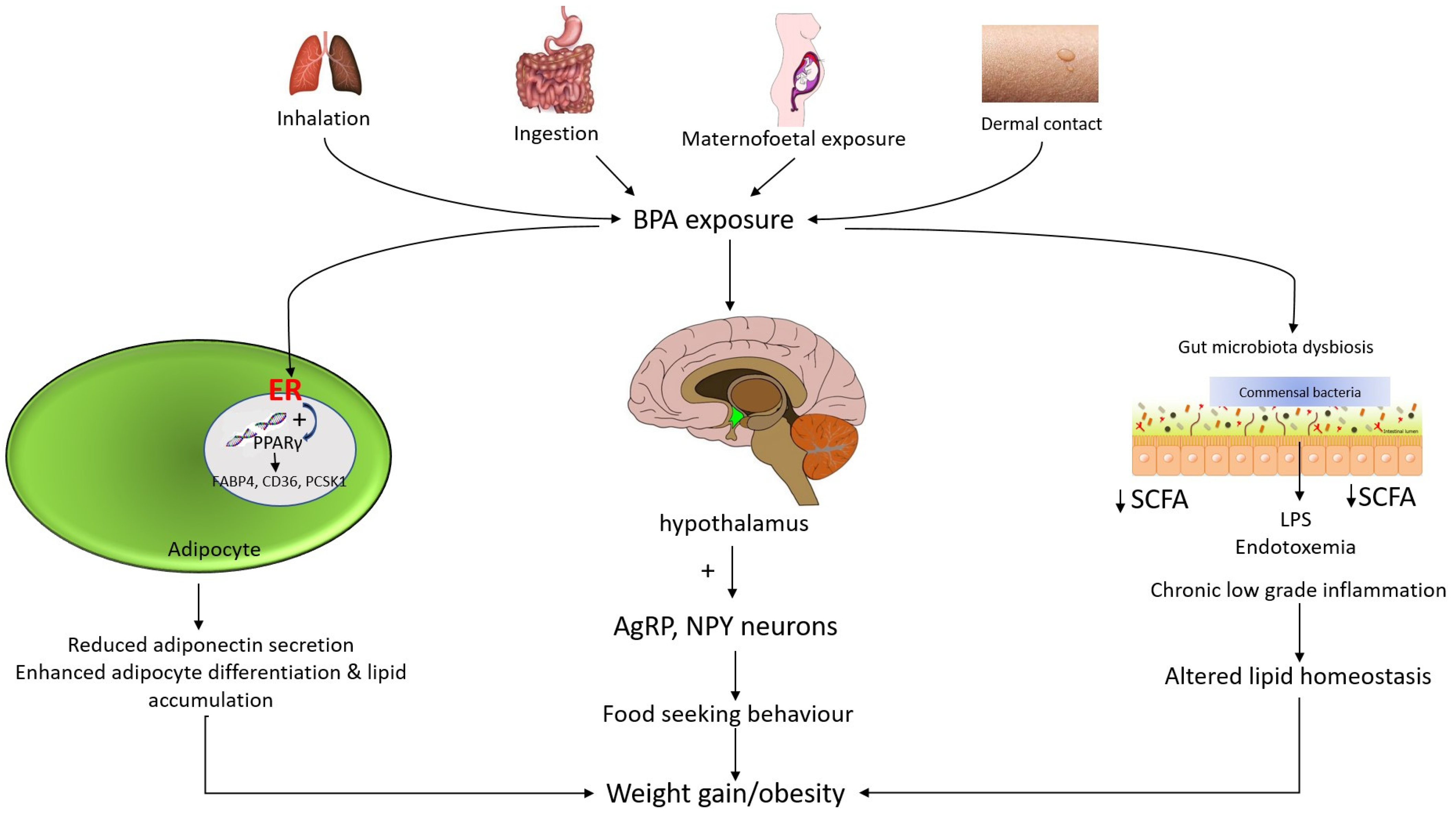
3.2. BPA and Obesity
3.3. BPA, Obesity and Neuroimmune Signals
3.4. Signaling Pathway of BPA in Obesity
3.5. BPA and Dysregulation of Gut Microbiome
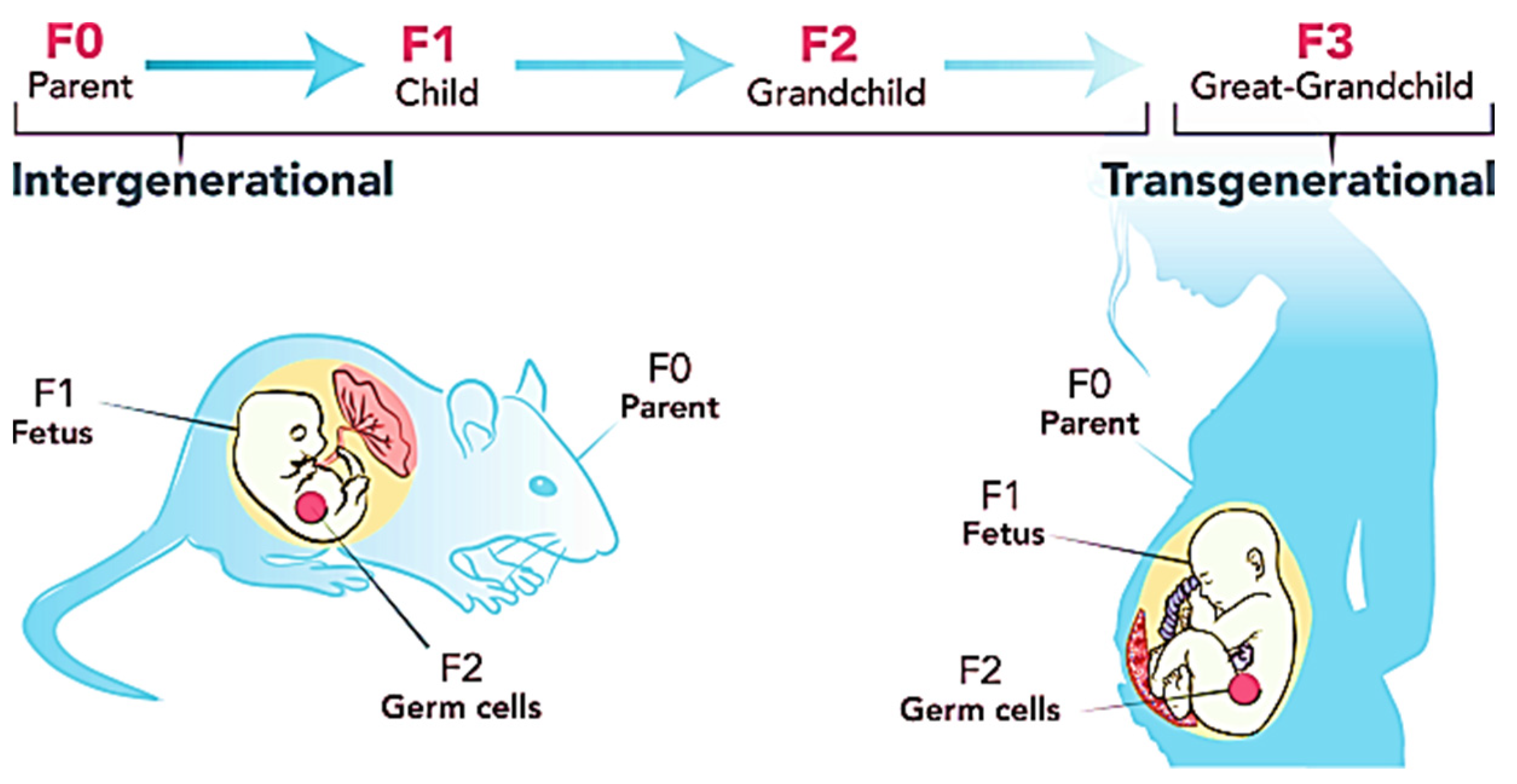
3.6. BPA and Transgenerational Effects of Obesity
3.7. BPA and Cardiovascular Complications
| Author | Subjects | Type of Exposure | Duration of Exposure | Dosage | Findings |
|---|---|---|---|---|---|
| Apaydin et al., 2019 [92] | Adult male albino rats | Oral gavage | 28 days | 130 mg/kg/bw/day |
|
| Bruno et al., 2019 [93] | 5 week old adult female balb/c mice | Drinking water | 2 weeks | 2.5, 25, and 250 µg/L |
|
| Reventun et al., 2020 [94] | 8 weeks old male wild-type CD1 mice | Drinking water | 16 weeks | 4 × 10−7 M |
|
| Bahey et al., 2019 [95] | Adult male Wistar rats | Intraperitoneal injection | 3 weeks | 1.2 mg/kg/day |
|
| Brown et al., 2019 [96] | 3rd generation homozygous TG(ERE:GFP)Casper Danio rerio zebrafish | Water in a static system | 6 h post fertilization to 5 days of post fertilization | 25 and 1000 μg/L |
|
| Friques et al., 2020 [97] | 3 weeks old male Wistar rats | Oral gavage | 60 days | 100 μg/kg/day |
|
| Amin, 2019 [98] | Adult Wistar rats | Subcutaneous injection | 6 days | 30 mg/kg/day |
|
| Makowska et al., 2021 [99] | 8 weeks old female juvenile pigs mixture of Piétrain and Duroc breed | Capsules | 28 days | 0.05 mg/kg/day |
|
| Lombó et al., 2019 [100] | 4 months old wild type Danio rerio zebrafish | Water in a static system | 24 h post fertilization | 2000 and 4000 μg/L |
|
| Valokola et al., 2018 [101] | Adult male Wistar rats | Oral gavage | 4 weeks | 10, 25, and 50 mg/kg |
|
| Rameshrad et al., 2018 [102] | Male albino Wistar rats | Oral gavage | 2 months | 35 mg/kg/day |
|
| Eweda et al., 2019 [103] | Adult male Wister albino rats | Oral | 6 weeks | 30 mg/kg |
|
| Khodayar et al., 2018 [104] | 6 weeks old male Wistar rats | Oral | 30 days | 50 mg/kg |
|
| Sivashanmugam et al., 2017 [105] | 12 to 14 weeks old male albino Wistar rats | Oral | 30 days | 10,100, and 400 mg BPA/kg |
|
| Kasneci et al., 2017 [106] | C57bl/6n mice | Drinking water | 22 days | 25 ng/ml |
|
| Prudencio et al., 2021 [107] | 3 to 4 months old female Sprague Dawley rats | Not specified | Not specified | 0.0–100 µM |
|
| Oluranti et al., 2021 [108] | Wistar rats | Oral | 28 days | 25, and 50 mg/kg |
|
| Rasdi et al., 2020 [109] | 6 to 8 weeks old female Sprague Dawley rats | Drinking water | Pregnancy day 2 up to 21 days | 0.05 and 0.2 mg/ml |
|
| Gear et al., 2017 [110] | Female Sprague Dawley rats | Oral gavage | 6 months | 2.5, 25, 250, 2500, and 25,000 μg/kg/day |
|
| Vanani et al., 2020 [111] | 8 to 10 weeks old male Wistar rats | Intragastric intubation | 14 days | 250 mg/kg |
|
4. BPA Interaction with Specific Receptors in Obesity
4.1. Androgen Receptor
4.2. GPR30
4.3. Glucocorticoid Receptor
4.4. Estrogen-Related Receptor Gamma (ERRγ)
5. BPA Exposure and Adverse Perinatal, Childhood, and Adult Cardiovascular Health Outcomes
6. Biomarkers
7. Safety Concerns and Dosage
8. Conclusions
9. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aungst, J. 2014 Updated Safety Assessment of Bisphenol A (BPA) for Use in Food Contact Applications. 2014. Available online: https://www.fda.gov/media/90124/download (accessed on 20 January 2022).
- Kandaraki, E.; Chatzigeorgiou, A.; Livadas, S.; Palioura, E.; Economou, F.; Koutsilieris, M.; Palimeri, S.; Panidis, D.; Diamanti-Kandarakis, E. Endocrine disruptors and polycystic ovary syndrome (PCOS): Elevated serum levels of bisphenol A in women with PCOS. J. Clin. Endocrinol. Metab. 2011, 96, E480–E484. [Google Scholar] [CrossRef] [PubMed]
- Mahalingaiah, S.; Meeker, J.D.; Pearson, K.R.; Calafat, A.M.; Ye, X.; Petrozza, J.; Hauser, R. Temporal variability and predictors of urinary bisphenol A concentrations in men and women. Environ. Health Perspect. 2008, 116, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Li, M.; Chen, B.; Xu, M.; Xu, Y.; Huang, Y.; Lu, J.; Chen, Y.; Wang, W.; Li, X.; et al. Urinary Bisphenol A (BPA) Concentration Associates with Obesity and Insulin Resistance. J. Clin. Endocrinol. Metab. 2012, 97, E223–E227. [Google Scholar] [CrossRef]
- Gao, X.; Wang, H.S. Impact of Bisphenol A on the Cardiovascular System—Epidemiological and Experimental Evidence and Molecular Mechanisms. Int. J. Environ. Res. Public Health 2014, 11, 8413. [Google Scholar] [CrossRef]
- Carwile, J.L.; Luu, H.T.; Bassett, L.S.; Driscoll, D.A.; Yuan, C.; Chang, J.Y.; Ye, X.; Calafat, A.M.; Michels, K.B. Polycarbonate Bottle Use and Urinary Bisphenol A Concentrations. Environ. Health Perspect. 2009, 117, 1372. [Google Scholar] [CrossRef]
- WHO; UN and Food and Agriculture Organization of the United Nations. Toxicological and Health Aspects of Bisphenol A, Ottawa, Canada. 2010. Available online: https://apps.who.int/iris/bitstream/handle/10665/44624/97892141564274_eng.pdf;sequence=1 (accessed on 20 January 2022).
- Matuszczak, E.; Komarowska, M.D.; Debek, W.; Hermanowicz, A. The Impact of Bisphenol A on Fertility, Reproductive System, and Development: A Review of the Literature. Int. J. Endocrinol. 2019, 2019, 4068717. [Google Scholar] [CrossRef]
- Wood, L. Global Bisphenol A Market Report 2018: Analysis 2013–2017 & Forecasts 2018–2023; Transparncy Market Research: Dublin, Ireland, 2018; Available online: https://www.prnewswire.com/news-releases/global-bisphenol-a-market-report-2018-analysis-2013-2017--forecasts-2018-2023-300757673.html (accessed on 20 January 2022).
- Genuis, S.J.; Beesoon, S.; Birkholz, D.; Lobo, R.A. Human Excretion of Bisphenol A: Blood, Urine, and Sweat (BUS) Study. J. Environ. Public Health 2012, 2012, 185731. [Google Scholar] [CrossRef]
- Yang, C.; Lee, H.K.; Kong, A.P.S.; Lim, L.L.; Cai, Z.; Chung, A.C.K. Early-life exposure to endocrine disrupting chemicals associates with childhood obesity. Ann. Pediatr. Endocrinol. Metab. 2018, 23, 195. [Google Scholar] [CrossRef] [PubMed]
- Hutton, B.; Salanti, G.; Caldwell, D.M.; Chaimani, A.; Schmid, C.H.; Cameron, C.; Ioannidis, J.P.A.; Straus, S.; Thorlund, K.; Jansen, J.P.; et al. The PRISMA Extension Statement for Reporting of Systematic Reviews Incorporating Network Meta-analyses of Health Care Interventions: Checklist and Explanations. Ann. Intern. Med. 2015, 162, 777–784. [Google Scholar] [CrossRef]
- Naomi, R.; Bahari, H.; Yazid, M.D.; Othman, F.; Zakaria, Z.A.; Hussain, M.K. Potential Effects of Sweet Potato (Ipomoea batatas) in Hyperglycemia and Dyslipidemia—A Systematic Review in Diabetic Retinopathy Context. Int. J. Mol. Sci. 2021, 22, 10816. [Google Scholar] [CrossRef]
- Desai, M.; Ferrini, M.G.; Jellyman, J.K.; Han, G.; Ross, M.G. In vivo and in vitro bisphenol A exposure effects on adiposity. J. Dev. Orig. Health Dis. 2018, 9, 678–687. [Google Scholar] [CrossRef]
- Shih, M.K.; Tain, Y.L.; Chen, Y.W.; Hsu, W.H.; Yeh, Y.T.; Chang, S.K.C.; Liao, J.X.; Hou, C.Y. Resveratrol Butyrate Esters Inhibit Obesity Caused by Perinatal Exposure to Bisphenol A in Female Offspring Rats. Molecules 2021, 26, 4010. [Google Scholar] [CrossRef] [PubMed]
- Dabeer, S.; Afjal, M.A.; Ahmad, S.; Fatima, M.; Habib, H.; Parvez, S.; Raisuddin, S. Transgenerational effect of parental obesity and chronic parental bisphenol A exposure on hormonal profile and reproductive organs of preadolescent Wistar rats of F1 generation: A one-generation study. Hum. Exp. Toxicol. 2019, 39, 59–76. [Google Scholar] [CrossRef]
- Taylor, J.A.; Shioda, K.; Mitsunaga, S.; Yawata, S.; Angle, B.M.; Nagel, S.C.; vom Saal, F.S.; Shioda, T. Prenatal Exposure to Bisphenol A Disrupts Naturally Occurring Bimodal DNA Methylation at Proximal Promoter of fggy, an Obesity-Relevant Gene Encoding a Carbohydrate Kinase, in Gonadal White Adipose Tissues of CD-1 Mice. Endocrinology 2018, 159, 779–794. [Google Scholar] [CrossRef]
- Santangeli, S.; Notarstefano, V.; Maradonna, F.; Giorgini, E.; Gioacchini, G.; Forner-Piquer, I.; Habibi, H.R.; Carnevali, O. Effects of diethylene glycol dibenzoate and Bisphenol A on the lipid metabolism of Danio rerio. Sci. Total Environ. 2018, 636, 641–655. [Google Scholar] [CrossRef] [PubMed]
- Malaisé, Y.; Menard, S.; Cartier, C.; Gaultier, E.; Lasserre, F.; Lencina, C.; Harkat, C.; Geoffre, N.; Lakhal, L.; Castan, I.; et al. Gut dysbiosis and impairment of immune system homeostasis in perinatally-exposed mice to Bisphenol A precede obese phenotype development. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Pu, Y.; Gingrich, J.D.; Steibel, J.P.; Veiga-Lopez, A. Sex-Specific Modulation of Fetal Adipogenesis by Gestational Bisphenol A and Bisphenol S Exposure. Endocrinology 2017, 158, 3844–3858. [Google Scholar] [CrossRef] [PubMed]
- Stoker, C.; Andreoli, M.F.; Kass, L.; Bosquiazzo, V.L.; Rossetti, M.F.; Canesini, G.; Luque, E.H.; Ramos, J.G. Perinatal exposure to bisphenol A (BPA) impairs neuroendocrine mechanisms regulating food intake and kisspetin system in adult male rats. Evidences of metabolic disruptor hypothesis. Mol. Cell. Endocrinol. 2020, 499, 110614. [Google Scholar] [CrossRef] [PubMed]
- Neier, K.; Marchlewicz, E.M.; Bedrosian, L.D.; Dolinoy, D.C.; Harris, C. Characterization of the mouse white adipose tissue redox environment and associations with perinatal environmental exposures to bisphenol A and high-fat diets. J. Nutr. Biochem. 2019, 66, 86–97. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Yan, S.; Meng, Z.; Huang, S.; Sun, W.; Jia, M.; Teng, M.; Zhou, Z.; Zhu, W. New insights into bisphenols induced obesity in zebrafish (Danio rerio): Activation of cannabinoid receptor CB1. J. Hazard. Mater. 2021, 418, 126100. [Google Scholar] [CrossRef]
- Lin, R.; Jia, Y.; Wu, F.; Meng, Y.; Sun, Q.; Jia, L. Combined Exposure to Fructose and Bisphenol A Exacerbates Abnormal Lipid Metabolism in Liver of Developmental Male Rats. Int. J. Environ. Res. Public Health 2019, 16, 4152. [Google Scholar] [CrossRef]
- Heindel, J.J.; Newbold, R.; Schug, T.T. Endocrine disruptors and obesity. Nat. Rev. Endocrinol. 2015, 11, 653–661. [Google Scholar] [CrossRef]
- Ben-Jonathan, N.; Hugo, E.R.; Brandebourg, T.D. Effects of bisphenol A on adipokine release from human adipose tissue: Implications for the metabolic syndrome. Mol. Cell. Endocrinol. 2009, 304, 54. [Google Scholar] [CrossRef]
- Trouwborst, I.; Bowser, S.M.; Goossens, G.H.; Blaak, E.E. Ectopic Fat Accumulation in Distinct Insulin Resistant Phenotypes; Targets for Personalized Nutritional Interventions. Front. Nutr. 2018, 5, 77. [Google Scholar] [CrossRef]
- Menale, C.; Piccolo, M.T.; Cirillo, G.; Calogero, R.A.; Papparella, A.; Mita, L.; Del Giudice, E.M.; Diano, N.; Crispi, S.; Mita, D.G. Bisphenol A effects on gene expression in adipocytes from children: Association with metabolic disorders. J. Mol. Endocrinol. 2015, 54, 289–303. [Google Scholar] [CrossRef]
- Yetisgin, A.A.; Cetinel, S.; Zuvin, M.; Kosar, A.; Kutlu, O. Therapeutic nanoparticles and their targeted delivery applications. Molecules 2020, 25, 2193. [Google Scholar] [CrossRef]
- Rogero, M.M.; Calder, P.C. Obesity, inflammation, toll-like receptor 4 and fatty acids. Nutrients 2018, 10, 432. [Google Scholar] [CrossRef]
- Raetz, C.R.H.; Reynolds, C.M.; Trent, M.S.; Bishop, R.E. Lipid a modification systems in gram-negative bacteria. Annu. Rev. Biochem. 2007, 76, 295–329. [Google Scholar] [CrossRef]
- Gan, L.; Liu, Z.; Cao, W.; Zhang, Z.; Sun, C. FABP4 reversed the regulation of leptin on mitochondrial fatty acid oxidation in mice adipocytes. Sci. Rep. 2015, 5, 1–12. [Google Scholar] [CrossRef]
- Queipo-Ortuño, M.I.; Escoté, X.; Ceperuelo-Mallafré, V.; Garrido-Sanchez, L.; Miranda, M.; Clemente-Postigo, M.; Pérez-Pérez, R.; Peral, B.; Cardona, F.; Fernández-Real, J.M.; et al. FABP4 Dynamics in Obesity: Discrepancies in Adipose Tissue and Liver Expression Regarding Circulating Plasma Levels. PLoS ONE 2012, 7, e48605. [Google Scholar] [CrossRef]
- Larder, R.; Lim, C.T.; Coll, A.P. Genetic aspects of human obesity. In Handbook of Clinical Neurology; Elsevier: Cambridge, UK, 2014; Volume 124, pp. 93–106. [Google Scholar]
- Erion, K.A.; Corkey, B.E. Hyperinsulinemia: A Cause of Obesity? Curr. Obes. Rep. 2017, 6, 178. [Google Scholar] [CrossRef]
- Bonen, A.; Tandon, N.N.; Glatz, J.F.C.; Luiken, J.J.F.P.; Heigenhauser, G.J.F. The fatty acid transporter FAT/CD36 is upregulated in subcutaneous and visceral adipose tissues in human obesity and type 2 diabetes. Int. J. Obes. 2006, 30, 877–883. [Google Scholar] [CrossRef]
- Davis, R.L.; Curtis, K.S. Estrogen disruptors and neuroimmune signaling in obesity: Focus on bisphenol A. Curr. Opin. Toxicol. 2019, 19, 15–20. [Google Scholar] [CrossRef]
- Essner, R.A.; Smith, A.G.; Jamnik, A.A.; Ryba, A.R.; Trutner, Z.D.; Carter, M.E. AgRP Neurons Can Increase Food Intake during Conditions of Appetite Suppression and Inhibit Anorexigenic Parabrachial Neurons. J. Neurosci. 2017, 37, 8687. [Google Scholar] [CrossRef]
- Campos, C.A.; Bowen, A.J.; Schwartz, M.W.; Palmiter, R.D. Parabrachial CGRP Neurons Control Meal Termination. Cell Metab. 2016, 23, 811–820. [Google Scholar] [CrossRef]
- Kokot, F.; Ficek, R. Effects of Neuropeptide Y on Appetite. Miner. Electrolyte Metab. 1999, 25, 303–305. [Google Scholar] [CrossRef]
- Ahima, R.S.; Antwi, D.A. Brain regulation of appetite and satiety. Endocrinol. Metab. Clin. N. Am. 2008, 37, 811. [Google Scholar] [CrossRef]
- Yi, M.; Li, H.; Wu, Z.; Yan, J.; Liu, Q.; Ou, C.; Chen, M. A Promising Therapeutic Target for Metabolic Diseases: Neuropeptide Y Receptors in Humans. Cell. Physiol. Biochem. 2018, 45, 88–107. [Google Scholar] [CrossRef]
- Stienstra, R.; Duval, C.; Müller, M.; Kersten, S. PPARs, Obesity, and Inflammation. PPAR Res. 2007, 2007, 95974. [Google Scholar] [CrossRef]
- Franckhauser, S.; Muñ Oz, S.; Elias, I.; Ferre, T.; Bosch, F. Adipose Overexpression of Phosphoenolpyruvate Carboxykinase Leads to High Susceptibility to Diet-Induced Insulin Resistance and Obesity. Diabetes 2006, 55, 273–280. [Google Scholar] [CrossRef]
- Wang, H.; Eckel, R.H. Lipoprotein lipase: From gene to obesity. Am. J. Physiol.-Endocrinol. Metab. 2009, 297, 271–288. [Google Scholar] [CrossRef] [PubMed]
- Guan, Y.; Zhang, T.; He, J.; Jia, J.; Zhu, L.; Wang, Z. Bisphenol A disturbed the lipid metabolism mediated by sterol regulatory element binding protein 1 in rare minnow Gobiocypris rarus. Aquat. Toxicol. 2018, 207, 179–186. [Google Scholar] [CrossRef]
- Lu, M.; Shyy, J.Y.J. Sterol regulatory element-binding protein 1 is negatively modulated by PKA phosphorylation. Am. J. Physiol.-Cell Physiol. 2006, 290, 1477–1486. [Google Scholar] [CrossRef] [PubMed]
- Olofsson, L.E.; Orho-Melander, M.; William-Olsson, L.; Sjöholm, K.; Sjöströ, L.; Groop, L.; Carlsson, B.; Carlsson, L.M.S.; Olsson, B. CCAAT/Enhancer Binding Protein (C/EBP) in Adipose Tissue Regulates Genes in Lipid and Glucose Metabolism and a Genetic Variation in C/EBP Is Associated with Serum Levels of Triglycerides. J. Clin. Endocrinol. Metab. 2008, 93, 4880–4886. [Google Scholar] [CrossRef] [PubMed]
- Guo Hou, X.; Moser, S.; Sarr, M.G.; Thompson, G.B.; Que, F.G.; Jensen, M.D. Visceral and Subcutaneous Adipose Tissue Diacylglycerol Acyltransferase Activity in Humans. Obesity 2009, 17, 1129–1134. [Google Scholar] [CrossRef]
- Caporossi, L.; Papaleo, B. Exposure to Bisphenol a and Gender Differences: From Rodents to Humans Evidences and Hypothesis about the Health Effects. J. Xenobiotics 2015, 5, 5264. [Google Scholar] [CrossRef] [PubMed]
- Kaliannan, K.; Robertson, R.C.; Murphy, K.; Stanton, C.; Kang, C.; Wang, B.; Hao, L.; Bhan, A.K.; Kang, J.X. Estrogen-mediated gut microbiome alterations influence sexual dimorphism in metabolic syndrome in mice. Microbiome 2018, 6, 1–22. [Google Scholar] [CrossRef]
- Meadows, A.; Lee, J.H.; Wu, C.S.; Wei, Q.; Pradhan, G.; Yafi, M.; Lu, H.C.; Sun, Y. Deletion of G-protein-coupled receptor 55 promotes obesity by reducing physical activity. Int. J. Obes. 2015, 40, 417–424. [Google Scholar] [CrossRef]
- Sipe, J.C.; Waalen, J.; Gerber, A.; Beutler, E. Overweight and obesity associated with a missense polymorphism in fatty acid amide hydrolase (FAAH). Int. J. Obes. 2005, 29, 755–759. [Google Scholar] [CrossRef] [PubMed]
- Tsiloulis, T.; Watt, M.J. Exercise and the Regulation of Adipose Tissue Metabolism, 1st ed.; Bouchard, C., Ed.; Elsevier: Victoria, Australia, 2015; Volume 135, ISBN 9780128039915. [Google Scholar]
- Jialal, I.; Kaur, H.; Devaraj, S. Toll-like receptor status in obesity and metabolic syndrome: A translational perspective. J. Clin. Endocrinol. Metab. 2014, 99, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Neuman, H.; Debelius, J.W.; Knight, R.; Koren, O. Microbial endocrinology: The interplay between the microbiota and the endocrine system. FEMS Microbiol. Rev. 2015, 39, 509–521. [Google Scholar] [CrossRef]
- Yin, Y.-N.; Yu, Q.-F.; Fu, N.; Liu, X.-W.; Lu, F.-G. Effects of four Bifidobacteria on obesity in high-fat diet induced rats. World J. Gastroenterol. 2010, 16, 3394. [Google Scholar] [CrossRef] [PubMed]
- An, H.M.; Park, S.Y.; Lee, D.K.; Kim, J.R.; Cha, M.K.; Lee, S.W.; Lim, H.T.; Kim, K.J.; Ha, N.J. Antiobesity and lipid-lowering effects of Bifidobacterium spp. in high fat diet-induced obese rats. Lipids Health Dis. 2011, 10, 116. [Google Scholar] [CrossRef] [PubMed]
- Mohajer, N.; Du, C.Y.; Checkcinco, C.; Blumberg, B. Obesogens: How They Are Identified and Molecular Mechanisms Underlying Their Action. Front. Endocrinol. 2021, 12, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Magne, F.; Gotteland, M.; Gauthier, L.; Zazueta, A.; Pesoa, S.; Navarrete, P.; Balamurugan, R. The firmicutes/bacteroidetes ratio: A relevant marker of gut dysbiosis in obese patients? Nutrients 2020, 12, 1474. [Google Scholar] [CrossRef]
- Shang, H.; Sun, J.; Chen, Y.Q. Clostridium Butyricum CGMCC0313.1 Modulates Lipid Profile, Insulin Resistance and Colon Homeostasis in Obese Mice. PLoS ONE 2016, 11, e0154373. [Google Scholar] [CrossRef]
- Rosenfeld, C.S. Gut dysbiosis in animals due to environmental chemical exposures. Front. Cell. Infect. Microbiol. 2017, 7, 396. [Google Scholar] [CrossRef] [PubMed]
- Javurek, A.B.; Spollen, W.G.; Johnson, S.A.; Bivens, N.J.; Bromert, K.H.; Givan, S.A.; Rosenfeld, C.S. Effects of exposure to bisphenol A and ethinyl estradiol on the gut microbiota of parents and their offspring in a rodent model. Gut Microbes 2016, 7, 471–485. [Google Scholar] [CrossRef]
- Wu, W.; Li, M.; Liu, A.; Wu, C.; Li, D.; Deng, Q.; Zhang, B.; Du, J.; Gao, X.; Hong, Y. Bisphenol A and the Risk of Obesity a Systematic Review With Meta-Analysis of the Epidemiological Evidence. Dose-Response 2020, 18, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yao, Y.; Li, H.; Qiao, F.; Wu, J.; Du, Z.Y.; Zhang, M. Influence of endogenous and exogenous estrogenic endocrine on intestinal microbiota in zebrafish. PLoS ONE 2016, 11, e0163895. [Google Scholar] [CrossRef]
- Lai, K.-P.; Chung, Y.-T.; Li, R.; Wan, H.-T.; Wong, C.K.C. Bisphenol A alters gut microbiome: Comparative metagenomics analysis. Environ. Pollut. 2016, 218, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Massier, L.; Chakaroun, R.; Tabei, S.; Crane, A.; Didt, K.D.; Fallmann, J.; Von Bergen, M.; Haange, S.B.; Heyne, H.; Stumvoll, M.; et al. Adipose tissue derived bacteria are associated with inflammation in obesity and type 2 diabetes. Gut 2020, 69, 1796–1806. [Google Scholar] [CrossRef] [PubMed]
- Gálvez-Ontiveros, Y.; Páez, S.; Monteagudo, C.; Rivas, A. Endocrine disruptors in food: Impact on gut microbiota and metabolic diseases. Nutrients 2020, 12, 1158. [Google Scholar] [CrossRef] [PubMed]
- Reddivari, L.; Veeramachaneni, D.N.R.; Walters, W.A.; Lozupone, C.; Palmer, J.; Hewage, M.K.K.; Bhatnagar, R.; Amir, A.; Kennett, M.J.; Knight, R.; et al. Perinatal Bisphenol A Exposure Induces Chronic Inflammation in Rabbit Offspring via Modulation of Gut Bacteria and Their Metabolites. mSystems 2017, 2, 93–110. [Google Scholar] [CrossRef] [PubMed]
- Chambers, E.S.; Preston, T.; Frost, G.; Morrison, D.J. Role of Gut Microbiota-Generated Short-Chain Fatty Acids in Metabolic and Cardiovascular Health. Curr. Nutr. Rep. 2018, 7, 206. [Google Scholar] [CrossRef] [PubMed]
- Corces, V.G.; Jung, Y.H.; Bixler, B.J.; Ruiz, D.; Wang, H.-L.V.; Linsenbaum, H.; Xiang, J.-F.; Shafik, A.M.; Jin, P. Transgenerational inheritance of BPA-induced obesity correlates with transmission of new CTCF sites in the Fto gene. bioRxiv 2020, 1, 114585. [Google Scholar] [CrossRef]
- Cimmino, I.; Fiory, F.; Perruolo, G.; Miele, C.; Beguinot, F.; Formisano, P.; Oriente, F. Potential mechanisms of bisphenol a (BPA) contributing to human disease. Int. J. Mol. Sci. 2020, 21, 5761. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zuo, X.; He, D.; Ding, S.; Xu, F.; Yang, H.; Jin, X.; Fan, Y.; Ying, L.; Tian, C.; et al. Long-term exposure to a “safe” dose of bisphenol A reduced protein acetylation in adult rat testes. Sci. Rep. 2017, 9, 40337. [Google Scholar] [CrossRef] [PubMed]
- Manikkam, M.; Tracey, R.; Guerrero-Bosagna, C.; Skinner, M.K. Plastics Derived Endocrine Disruptors (BPA, DEHP and DBP) Induce Epigenetic Transgenerational Inheritance of Obesity, Reproductive Disease and Sperm Epimutations. PLoS ONE 2013, 8, e55387. [Google Scholar] [CrossRef] [PubMed]
- Champroux, A.; Cocquet, J.; Henry-Berger, J.; Drevet, J.R.; Kocer, A. A decade of exploring the mammalian sperm epigenome: Paternal epigenetic and transgenerational inheritance. Front. Cell Dev. Biol. 2018, 6, 1–19. [Google Scholar] [CrossRef]
- Hart, R.J. Physiological aspects of female fertility: Role of the environment, modern lifestyle, and genetics. Physiol. Rev. 2016, 96, 873–909. [Google Scholar] [CrossRef]
- Church, C.; Moir, L.; McMurray, F.; Girard, C.; Banks, G.T.; Teboul, L.; Wells, S.; Brüning, J.C.; Nolan, P.M.; Ashcroft, F.M.; et al. Overexpression of Fto leads to increased food intake and results in obesity. Nat. Genet. 2010, 42, 1086–1092. [Google Scholar] [CrossRef]
- Shi, M.; Whorton, A.E.; Sekulovski, N.; Maclean, J.A.; Hayashi, K. Prenatal Exposure to Bisphenol A, E, and S Induces Transgenerational Effects on Male Reproductive Functions in Mice. Toxicol. Sci. 2019, 172, 303–315. [Google Scholar] [CrossRef]
- Alonso-Magdalena, P.; Rivera, F.J.; Guerrero-Bosagna, C. Bisphenol-A and metabolic diseases: Epigenetic, developmental and transgenerational basis. Environ. Epigenet. 2016, 2, dvw022. [Google Scholar] [CrossRef]
- Cariati, F.; Carbone, L.; Conforti, A.; Bagnulo, F.; Peluso, S.R.; Carotenuto, C.; Buonfantino, C.; Alviggi, E.; Alviggi, C.; Strina, I. Bisphenol A-Induced Epigenetic Changes and Its Effects on the Male Reproductive System. Front. Endocrinol. 2020, 11, 453. [Google Scholar] [CrossRef]
- Chang, H.; Wang, D.; Xia, W.; Pan, X.; Huo, W.; Xu, S.; Li, Y. Epigenetic disruption and glucose homeostasis changes following low-dose maternal bisphenol A exposure. Toxicol. Res. 2016, 5, 1400–1409. [Google Scholar] [CrossRef]
- Breton, C.V.; Landon, R.; Kahn, L.G.; Enlow, M.B.; Peterson, A.K.; Bastain, T.; Braun, J.; Comstock, S.S.; Duarte, C.S.; Hipwell, A.; et al. Exploring the evidence for epigenetic regulation of environmental influences on child health across generations. Commun. Biol. 2021, 4, 769. [Google Scholar] [CrossRef]
- Ma, Y.; Xia, W.; Wang, D.Q.; Wan, Y.J.; Xu, B.; Chen, X.; Li, Y.Y.; Xu, S.Q. Hepatic DNA methylation modifications in early development of rats resulting from perinatal BPA exposure contribute to insulin resistance in adulthood. Diabetologia 2013, 56, 2059–2067. [Google Scholar] [CrossRef]
- Junge, K.M.; Leppert, B.; Jahreis, S.; Wissenbach, D.K.; Feltens, R.; Grützmann, K.; Thürmann, L.; Bauer, T.; Ishaque, N.; Schick, M.; et al. MEST mediates the impact of prenatal bisphenol A exposure on long-term body weight development. Clin. Epigenet. 2018, 10, 1–12. [Google Scholar] [CrossRef]
- Tan, C.M.J.; Green, P.; Tapoulal, N.; Lewandowski, A.J.; Leeson, P.; Herring, N. The Role of Neuropeptide Y in Cardiovascular Health and Disease. Front. Physiol. 2018, 9, 1281. [Google Scholar] [CrossRef]
- Lomax, A.E.G.; Sharkey, K.A.; Giles, W.R. Neuropeptide Y Modulates L-Type Ca2+ Current during Heart Development. Circ. Res. 2003, 93, 891–892. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, C.Y.; Chen, W.C.; Shi, Y.C.; Wang, C.M.; Lin, S.; He, H.F. Regulation of neuropeptide Y in body microenvironments and its potential application in therapies: A review. Cell Biosci. 2021, 11, 151. [Google Scholar] [CrossRef]
- Robich, M.P.; Matyal, R.; Chu, L.M.; Feng, J.; Xu, S.H.; Laham, R.J.; Hess, P.E.; Bianchi, C.; Sellke, F.W. Effects of Neuropeptide Y on collateral development in a swine model of chronic myocardial ischemia. J. Mol. Cell. Cardiol. 2010, 49, 1030. [Google Scholar] [CrossRef]
- Sheriff, S.; Balasubramaniam, A. Inhibitory and stimulatory effects of neuropeptide Y(17-36) on rat cardiac adenylate cyclase activity. Structure-function studies. J. Biol. Chem. 1992, 267, 4680–4685. [Google Scholar] [CrossRef]
- Singh, R.M.; Cummings, E.; Pantos, C.; Singh, J. Protein kinase C and cardiac dysfunction: A review. Heart Fail. Rev. 2017, 22, 843–859. [Google Scholar] [CrossRef]
- Tallam, L.S.; Kuo, J.J.; Da Silva, A.A.; Hall, J.E. Cardiovascular, Renal, and Metabolic Responses to Chronic Central Administration of Agouti-Related Peptide. Hypertension 2004, 44, 853–858. [Google Scholar] [CrossRef]
- Apaydin, F.G.; Aslanturk, A.; Uzunhisarcikli, M.; Bas, H.; Kalender, S.; Kalender, Y. Histopathological and biochemical studies on the effect of curcumin and taurine against bisphenol A toxicity in male rats. Environ. Sci. Pollut. Res. 2019, 26, 12302–12310. [Google Scholar] [CrossRef]
- Bruno, K.A.; Mathews, J.E.; Yang, A.L.; Frisancho, J.A.; Scott, A.J.; Greyner, H.D.; Molina, F.A.; Greenaway, M.S.; Cooper, G.M.; Bucek, A.; et al. BPA Alters Estrogen Receptor Expression in the Heart After Viral Infection Activating Cardiac Mast Cells and T Cells Leading to Perimyocarditis and Fibrosis. Front. Endocrinol. 2019, 10, 598. [Google Scholar] [CrossRef]
- Reventun, P.; Sanchez-Esteban, S.; Cook, A.; Cuadrado, I.; Roza, C.; Moreno-Gomez-Toledano, R.; Muñoz, C.; Zaragoza, C.; Bosch, R.J.; Saura, M. Bisphenol A induces coronary endothelial cell necroptosis by activating RIP3/CamKII dependent pathway. Sci. Rep. 2020, 10, 1–17. [Google Scholar] [CrossRef]
- Bahey, N.G.; Elaziz, H.O.A.; Gadalla, K.K.E. Potential Toxic Effect of Bisphenol A on the Cardiac Muscle of Adult Rat and the Possible Protective Effect of Omega-3: A Histological and Immunohistochemical Study. J. Microsc. Ultrastruct. 2019, 7, 8. [Google Scholar] [CrossRef]
- Brown, A.R.; Green, J.M.; Moreman, J.; Gunnarsson, L.M.; Mourabit, S.; Ball, J.; Winter, M.J.; Trznadel, M.; Correia, A.; Hacker, C.; et al. Cardiovascular Effects and Molecular Mechanisms of Bisphenol A and Its Metabolite MBP in Zebrafish. Environ. Sci. Technol. 2019, 53, 463–474. [Google Scholar] [CrossRef]
- Friques, A.G.F.; Santos, F.D.N.; Angeli, D.B.; Silva, F.A.C.; Dias, A.T.; Aires, R.; Leal, M.A.S.; Nogueira, B.V.; Amorim, F.G.; Campagnaro, B.P.; et al. Bisphenol A contamination in infant rats: Molecular, structural, and physiological cardiovascular changes and the protective role of kefir. J. Nutr. Biochem. 2020, 75, 108254. [Google Scholar] [CrossRef]
- Amin, D.M. Role of copeptin as a novel biomarker of bisphenol A toxic effects on cardiac tissues: Biochemical, histological, immunohistological, and genotoxic study. Environ. Sci. Pollut. Res. 2019, 26, 36037–36047. [Google Scholar] [CrossRef]
- Makowska, K.; Gonkowski, S.; Licata, P.; Crupi, R.; Gugliandolo, E. Changes Caused by Low Doses of Bisphenol A (BPA) in the Neuro-Chemistry of Nerves Located in the Porcine Heart. Animals 2021, 11, 780. [Google Scholar] [CrossRef]
- Lombó, M.; González-Rojo, S.; Fernández-Díez, C.; Herráez, M.P. Cardiogenesis impairment promoted by bisphenol A exposure is successfully counteracted by epigallocatechin gallate. Environ. Pollut. 2019, 246, 1008–1019. [Google Scholar] [CrossRef]
- Valokola, M.G.; Karimi, G.; Razavi, B.M.; Kianfar, M.; Jafarian, A.H.; Jaafari, M.R.; Imenshahidi, M. The protective activity of nanomicelle curcumin in bisphenol A-induced cardiotoxicity following subacute exposure in rats. Environ. Toxicol. 2019, 34, 319–329. [Google Scholar] [CrossRef]
- Rameshrad, M.; Imenshahidi, M.; Razavi, B.M.; Iranshahi, M.; Hosseinzadeh, H. Bisphenol A vascular toxicity: Protective effect of Vitis vinifera (grape) seed extract and resveratrol. Phyther. Res. 2018, 32, 2396–2407. [Google Scholar] [CrossRef]
- Eweda, S.M.; Newairy, A.S.A.; Abdou, H.M.; Gaber, A.S. Bisphenol A-induced oxidative damage in the hepatic and cardiac tissues of rats: The modulatory role of sesame lignans. Exp. Ther. Med. 2020, 19, 33–44. [Google Scholar] [CrossRef]
- Khodayar, M.J.; Kalantari, H.; Mahdavinia, M.; Khorsandi, L.; Alboghobeish, S.; Samimi, A.; Alizadeh, S.; Zeidooni, L. Protective effect of naringin against BPA-induced cardiotoxicity through prevention of oxidative stress in male Wistar rats. Drug Chem. Toxicol. 2018, 43, 85–95. [Google Scholar] [CrossRef]
- Sivashanmugam, P.; Mullainadhan, V.; Karundevi, B. Dose-dependent effect of Bisphenol-A on insulin signaling molecules in cardiac muscle of adult male rat. Chem. Biol. Interact. 2017, 266, 10–16. [Google Scholar] [CrossRef]
- Kasneci, A.; Lee, J.S.; Yun, T.J.; Shang, J.; Lampen, S.; Gomolin, T.; Cheong, C.C.; Chalifour, L.E. From the Cover: Lifelong Exposure of C57bl/6n Male Mice to Bisphenol A or Bisphenol S Reduces Recovery from a Myocardial Infarction. Toxicol. Sci. 2017, 159, 189–202. [Google Scholar] [CrossRef]
- Prudencio, T.M.; Swift, L.M.; Guerrelli, D.; Cooper, B.; Reilly, M.; Ciccarelli, N.; Sheng, J.; Jaimes, R.; Posnack, N.G. Bisphenol S and Bisphenol F Are Less Disruptive to Cardiac Electrophysiology, as Compared With Bisphenol A. Toxicol. Sci. 2021, 183, 214–226. [Google Scholar] [CrossRef]
- Oluranti, O.I.; Alabi, B.A.; Michael, O.S.; Ojo, A.O.; Fatokun, B.P. Rutin prevents cardiac oxidative stress and inflammation induced by bisphenol A and dibutyl phthalate exposure via NRF-2/NF-κB pathway. Life Sci. 2021, 284, 119878. [Google Scholar] [CrossRef]
- Rasdi, Z.; Kamaludin, R.; Ab Rahim, S.; Syed, A.; Fuad, S.B.; Othman, M.H.D.; Siran, R.; Nor, N.S.M.; Hasani, N.A.H.; Kadir, S.H.S.A. The impacts of intrauterine Bisphenol A exposure on pregnancy and expression of miRNAs related to heart development and diseases in animal model. Sci. Rep. 2020, 10, 1–13. [Google Scholar] [CrossRef]
- Gear, R.; Kendziorski, J.A.; Belcher, S.M. Effects of bisphenol A on incidence and severity of cardiac lesions in the NCTR-Sprague-Dawley rat: A CLARITY-BPA study. Toxicol. Lett. 2017, 275, 123–135. [Google Scholar] [CrossRef]
- Vanani, A.R.; Mahdavinia, M.; Shirani, M.; Alizadeh, S.; Dehghani, M.A. Protective effects of quercetin against oxidative stress induced by bisphenol-A in rat cardiac mitochondria. Environ. Sci. Pollut. Res. 2020, 27, 15093–15102. [Google Scholar] [CrossRef]
- Delfosse, V.; Grimaldi, M.; Pons, J.L.; Boulahtouf, A.; Le Maire, A.; Cavailles, V.; Labesse, G.; Bourguet, W.; Balaguer, P. Structural and mechanistic insights into bisphenols action provide guidelines for risk assessment and discovery of bisphenol A substitutes. Proc. Natl. Acad. Sci. USA. 2012, 109, 14930–14935. [Google Scholar] [CrossRef]
- Chen, Q.; Wang, X.; Tan, H.; Shi, W.; Zhang, X.; Wei, S.; Giesy, J.P.; Yu, H. Molecular Initiating Events of Bisphenols on Androgen Receptor-Mediated Pathways Provide Guidelines for in Silico Screening and Design of Substitute Compounds. Environ. Sci. Technol. Lett. 2019, 6, 205–210. [Google Scholar] [CrossRef]
- Pasquali, R. Obesity and androgens: Facts and perspectives. Fertil. Steril. 2006, 85, 1319–1340. [Google Scholar] [CrossRef]
- Kelly, D.M.; Jones, T.H. Testosterone and obesity. Obes. Rev. 2015, 16, 581–606. [Google Scholar] [CrossRef]
- Thomas, P.; Dong, J. Binding and activation of the seven-transmembrane estrogen receptor GPR30 by environmental estrogens: A potential novel mechanism of endocrine disruption. J. Steroid Biochem. Mol. Biol. 2006, 102, 175–179. [Google Scholar] [CrossRef]
- Wang, A.; Luo, J.; Moore, W.; Alkhalidy, H.; Wu, L.; Zhang, J.; Zhen, W.; Wang, Y.; Clegg, D.J.; Xu, B.; et al. GPR30 regulates diet-induced adiposity in female mice and adipogenesis in vitro. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef]
- Cimini, F.A.; Barchetta, I.; Porzia, A.; Mainiero, F.; Costantino, C.; Bertoccini, L.; Ceccarelli, V.; Morini, S.; Baroni, M.G.; Lenzi, A.; et al. Circulating IL-8 levels are increased in patients with type 2 diabetes and associated with worse inflammatory and cardiometabolic profile. Acta Diabetol. 2017, 54, 961–967. [Google Scholar] [CrossRef]
- Cimmino, I.; Oriente, F.; D’esposito, V.; Liguoro, D.; Liguoro, P.; Ambrosio, M.R.; Cabaro, S.; D’andrea, F.; Beguinot, F.; Formisano, P.; et al. Low-dose bisphenol-a regulates inflammatory cytokines through GPR30 in mammary adipose cells. J. Mol. Endocrinol. 2019, 63, 273–283. [Google Scholar] [CrossRef]
- Boucher, J.G.; Boudreau, A.; Atlas, E. Bisphenol A induces differentiation of human preadipocytes in the absence of glucocorticoid and is inhibited by an estrogen-receptor antagonist. Nutr. Diabetes 2013, 4, e102. [Google Scholar] [CrossRef]
- Tohmé, M.; Prud’Homme, S.M.; Boulahtouf, A.; Samarut, E.; Brunet, F.; Bernard, L.; Bourguet, W.; Gibert, Y.; Balaguer, P.; Laudet, V. Estrogen-related receptor γ is an in vivo receptor of bisphenol A. FASEB J. 2014, 28, 3124–3133. [Google Scholar] [CrossRef]
- Beale, E.G.; Harvey, B.J.; Forest, C. PCK1 and PCK2 as candidate diabetes and obesity genes. Cell Biochem. Biophys. 2007, 48, 89–95. [Google Scholar] [CrossRef]
- Zulkifli, S.; Rahman, A.A.; Kadir, S.H.S.A.; Nor, N.S.M. Bisphenol A and its effects on the systemic organs of children. Eur. J. Pediatr. 2021, 180, 3111–3127. [Google Scholar] [CrossRef]
- Melzer, D.; Rice, N.E.; Lewis, C.; Henley, W.E.; Galloway, T.S. Association of Urinary Bisphenol A Concentration with Heart Disease: Evidence from NHANES 2003/06. PLoS ONE 2010, 5, e8673. [Google Scholar] [CrossRef]
- Shankar, A.; Teppala, S. Urinary bisphenol A and hypertension in a multiethnic sample of US adults. J. Environ. Public Health 2012, 2012, 481641. [Google Scholar] [CrossRef]
- Bae, S.; Kim, J.H.; Lim, Y.H.; Park, H.Y.; Hong, Y.C. Associations of bisphenol A exposure with heart rate variability and blood pressure. Hypertensionaha 2012, 60, 786–793. [Google Scholar] [CrossRef]
- Kataria, A.; Levine, D.; Wertenteil, S.; Vento, S.; Xue, J.; Rajendiran, K.; Kannan, K.; Thurman, J.M.; Morrison, D.; Brody, R.; et al. Exposure to bisphenols and phthalates and association with oxidant stress, insulin resistance, and endothelial dysfunction in children. Pediatr. Res. 2017, 81, 857–864. [Google Scholar] [CrossRef]
- Kubo, T.; Maezawa, N.; Osada, M.; Katsumura, S.; Funae, Y.; Imaoka, S. Bisphenol A, an environmental endocrine-disrupting chemical, inhibits hypoxic response via degradation of hypoxia-inducible factor 1alpha (HIF-1alpha): Structural requirement of bisphenol A for degradation of HIF-1alpha. Biochem. Biophys. Res. Commun. 2004, 318, 1006–1011. [Google Scholar] [CrossRef]
- Montazeri, P.; Fossati, S.; Warembourg, C.; Casas, M.; Clemente, D.B.P.; Garcia-Esteban, R.; Nawrot, T.S.; Vrijheid, M. Prenatal exposure to phthalates and phenols and preclinical vascular health during early adolescence. Int. J. Hyg. Environ. Health 2021, 240, 113909. [Google Scholar] [CrossRef]
- Bujalance-reyes, F.; Molina-López, A.M.; Ayala-soldado, N.; Lora-benitez, A.; Mora-Medina, R.; Moyano-Salvago, R. Analysis of Indirect Biomarkers of Effect after Exposure to Low Doses of Bisphenol A in a Study of Successive Generations of Mice. Animals 2022, 12, 300. [Google Scholar] [CrossRef]
- Pérez-Bermejo, M.; Mas-Pérez, I.; Murillo-Llorente, M.T. The Role of the Bisphenol A in Diabetes and Obesity. Biomedicines 2021, 9, 666. [Google Scholar] [CrossRef]
- Wang, M.; Rang, O.; Liu, F.; Xia, W.; Li, Y.; Zhang, Y.; Lu, S.; Xu, S. A systematic review of metabolomics biomarkers for Bisphenol A exposure. Metabolomics 2018, 14, 1–19. [Google Scholar] [CrossRef]
- Cahill, A.G.; MacOnes, G.A.; Smyser, C.D.; López, J.D.; Inder, T.E.; Mathur, A.M. Umbilical Artery Lactate Correlates with Brain Lactate in Term Infants. Am. J. Perinatol. 2017, 34, 535–540. [Google Scholar] [CrossRef]
- Cohn, P.; Fan, A.M. Bisphenol A Biomarkers and Biomonitoring, 2nd ed.; Gupta, R.C., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; ISBN 9780128146552. [Google Scholar]
- Mustieles, V.; D’Cruz, S.C.; Couderq, S.; Rodríguez-Carrillo, A.; Fini, J.B.; Hofer, T.; Steffensen, I.L.; Dirven, H.; Barouki, R.; Olea, N.; et al. Bisphenol A and its analogues: A comprehensive review to identify and prioritize effect biomarkers for human biomonitoring. Environ. Int. 2020, 144, 105811. [Google Scholar] [CrossRef]
- Steffensen, I.L.; Dirven, H.; Couderq, S.; David, A.; D’cruz, S.C.; Fernández, M.F.; Mustieles, V.; Rodríguez-Carillo, A.; Hofer, T. Bisphenols and oxidative stress biomarkers— associations found in human studies, evaluation of methods used, and strengths and weaknesses of the biomarkers. Int. J. Environ. Res. Public Health 2020, 17, 3609. [Google Scholar] [CrossRef]
- Raja, G.L.; Lite, C.; Subhashree, K.D.; Santosh, W.; Barathi, S. Prenatal bisphenol-A exposure altered exploratory and anxiety-like behaviour and induced non-monotonic, sex-specific changes in the cortical expression of CYP19A1, BDNF and intracellular signaling proteins in F1 rats. Food Chem. Toxicol. 2020, 142, 111442. [Google Scholar] [CrossRef]
- Veiga-Lopez, A.; Pennathur, S.; Kannan, K.; Patisaul, H.B.; Dolinoy, D.C.; Zeng, L.; Padmanabhan, V. Impact of gestational bisphenol A on oxidative stress and free fatty acids: Human association and interspecies animal testing studies. Endocrinology 2015, 156, 911–922. [Google Scholar] [CrossRef]
- Moon, M.K. Concern about the Safety of Bisphenol A Substitutes. Diabetes Metab. J. 2019, 43, 48. [Google Scholar] [CrossRef]
- Petre, A. What Is BPA and Why Is It Bad for You? Healthline. 2018. Available online: https://www.healthline.com/nutrition/what-is-bpa#bottom-line (accessed on 20 January 2022).
- EPA. Bisphenol A (BPA) Action Plan Summary; EPA: Washington, DC, USA, 2013.
- Bolognesi, C.; Castle, L.; Cravedi, J.-P.; Engel, K.-H.; Fowler, P.; Franz, R.; Grob, K.; Gürtler, R.; Husøy, T.; Mennes, W.; et al. Scientific Opinion on the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs. EFSA J. 2015, 13, 3978. [Google Scholar] [CrossRef]
| Author | Subjects | Type of Exposure | Duration of Exposure | Dosage | Findings |
|---|---|---|---|---|---|
| Desai et al., 2018 [14] | 12 weeks old female Sprague–Dawley rats | Oral (purified drinking water containing BPA) | Before 2 weeks of mating up to weaning stage | 5 mg/L/day |
|
| Shih et al., 2021 [15] | 15 weeks old female Sprague–Dawley rats | Oral gavage | 6th day after pregnancy up to 36 days | 50 µg/kg/day |
|
| Dabeer et al., 2019 [16] | 3 to 4 weeks old male and female Wistar rats | BPA in drinking water | 180 days before mating and up to postnatal day (PND) 35 | 10 ppm and 10 mg/L/day |
|
| Taylor et al., 2018 [17] | 3 month old nulliparous female CD-1 mice | Oral gavage | Pregnancy confirmation up to PND 22 | 5 or 500 mg/kg/bw/day |
|
| Santangeli et al., 2018 [18] | Wild type adult female Danio rerio | Water in a static system | 21 days | 5 μg/L, 10 μg/L, and 20 μg/L |
|
| Malaisé et al., 2017 [19] | 8 week old female and male C3H/HeN mice | oral | PND 170 | 50 µg/kg/day |
|
| Pu et al., 2017 [20] | Primiparous female sheep | Subcutaneous injection | 147 days | 0.5 mg/kg/day |
|
| Stoker et al., 2019 [21] | 90 days old female Wistar rats | BPA in drinking water | Gestation day 9 to weaning | 50 µg/kg/day |
|
| Neier et al., 2019 [22] | 12 to 27 weeks old C57BL/6J female mice | Chow diet | Gestation day 9 to PND 21 | 50 μg/kg/day |
|
| Tian et al., 2021 [23] | 5 months old wild type adult male Danio rerio | Water in a static system | 28 days | 20, 100, and 500 μg/L/day |
|
| Lin et al., 2019 [24] | 3 weeks old male Wistar rats | Drinking water | 8 weeks | 1 µg/mL/day |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naomi, R.; Yazid, M.D.; Bahari, H.; Keong, Y.Y.; Rajandram, R.; Embong, H.; Teoh, S.H.; Halim, S.; Othman, F. Bisphenol A (BPA) Leading to Obesity and Cardiovascular Complications: A Compilation of Current In Vivo Study. Int. J. Mol. Sci. 2022, 23, 2969. https://doi.org/10.3390/ijms23062969
Naomi R, Yazid MD, Bahari H, Keong YY, Rajandram R, Embong H, Teoh SH, Halim S, Othman F. Bisphenol A (BPA) Leading to Obesity and Cardiovascular Complications: A Compilation of Current In Vivo Study. International Journal of Molecular Sciences. 2022; 23(6):2969. https://doi.org/10.3390/ijms23062969
Chicago/Turabian StyleNaomi, Ruth, Muhammad Dain Yazid, Hasnah Bahari, Yong Yoke Keong, Retnagowri Rajandram, Hashim Embong, Soo Huat Teoh, Shariff Halim, and Fezah Othman. 2022. "Bisphenol A (BPA) Leading to Obesity and Cardiovascular Complications: A Compilation of Current In Vivo Study" International Journal of Molecular Sciences 23, no. 6: 2969. https://doi.org/10.3390/ijms23062969
APA StyleNaomi, R., Yazid, M. D., Bahari, H., Keong, Y. Y., Rajandram, R., Embong, H., Teoh, S. H., Halim, S., & Othman, F. (2022). Bisphenol A (BPA) Leading to Obesity and Cardiovascular Complications: A Compilation of Current In Vivo Study. International Journal of Molecular Sciences, 23(6), 2969. https://doi.org/10.3390/ijms23062969









