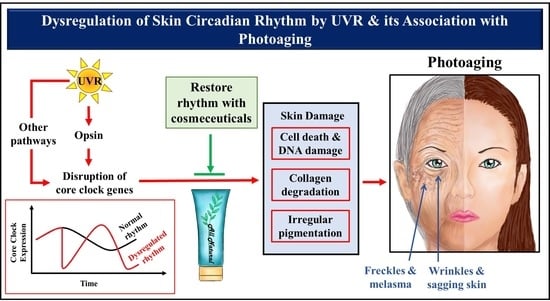
Cosmeceutical Therapy: Engaging the Repercussions of UVR Photoaging on the Skin’s Circadian Rhythm
Abstract
1. Introduction
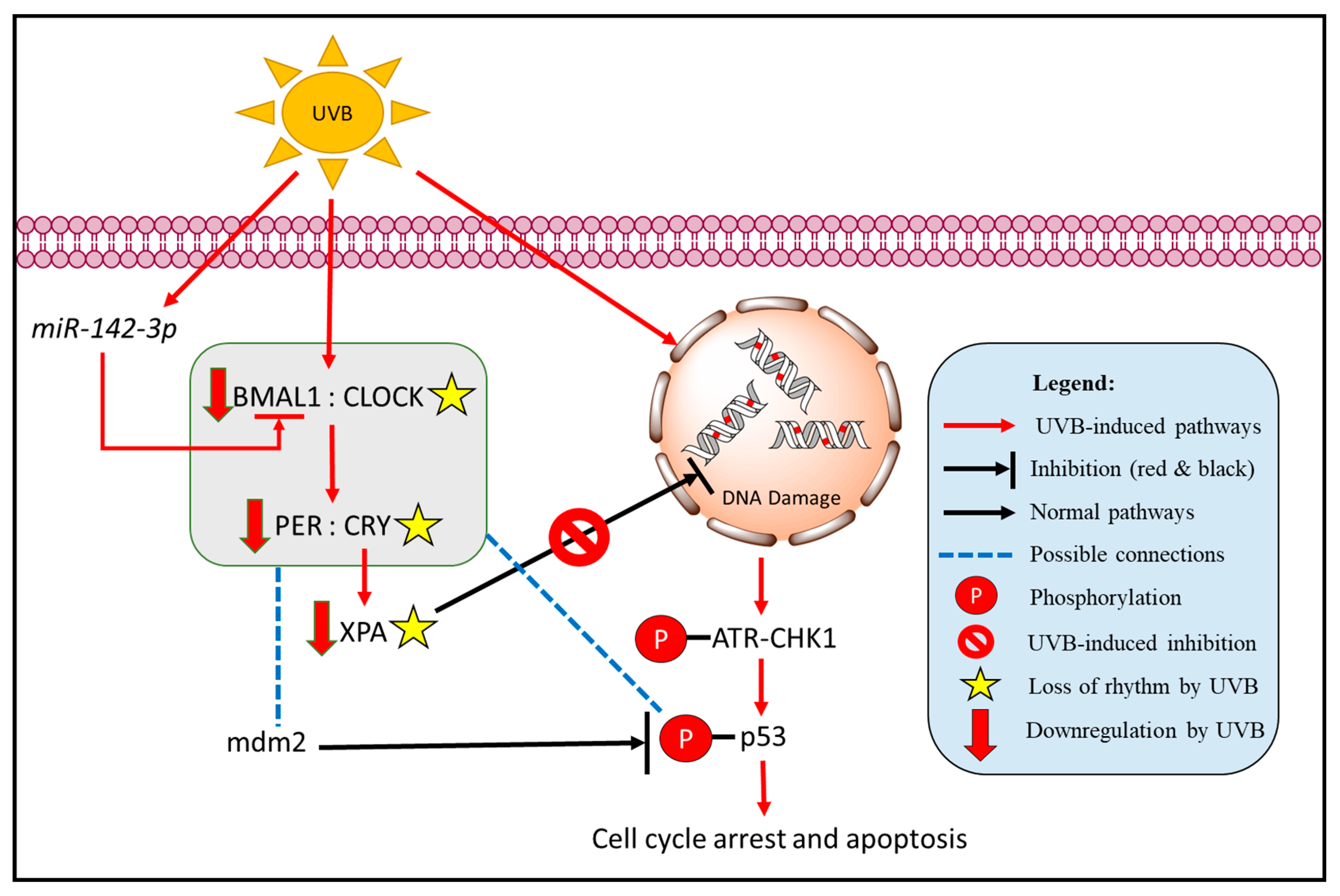
2. The Dance between Core Clock Genes and Solar Erythema
2.1. The Effect of NB-UVB BB-UVB on the Circadian Rhythm
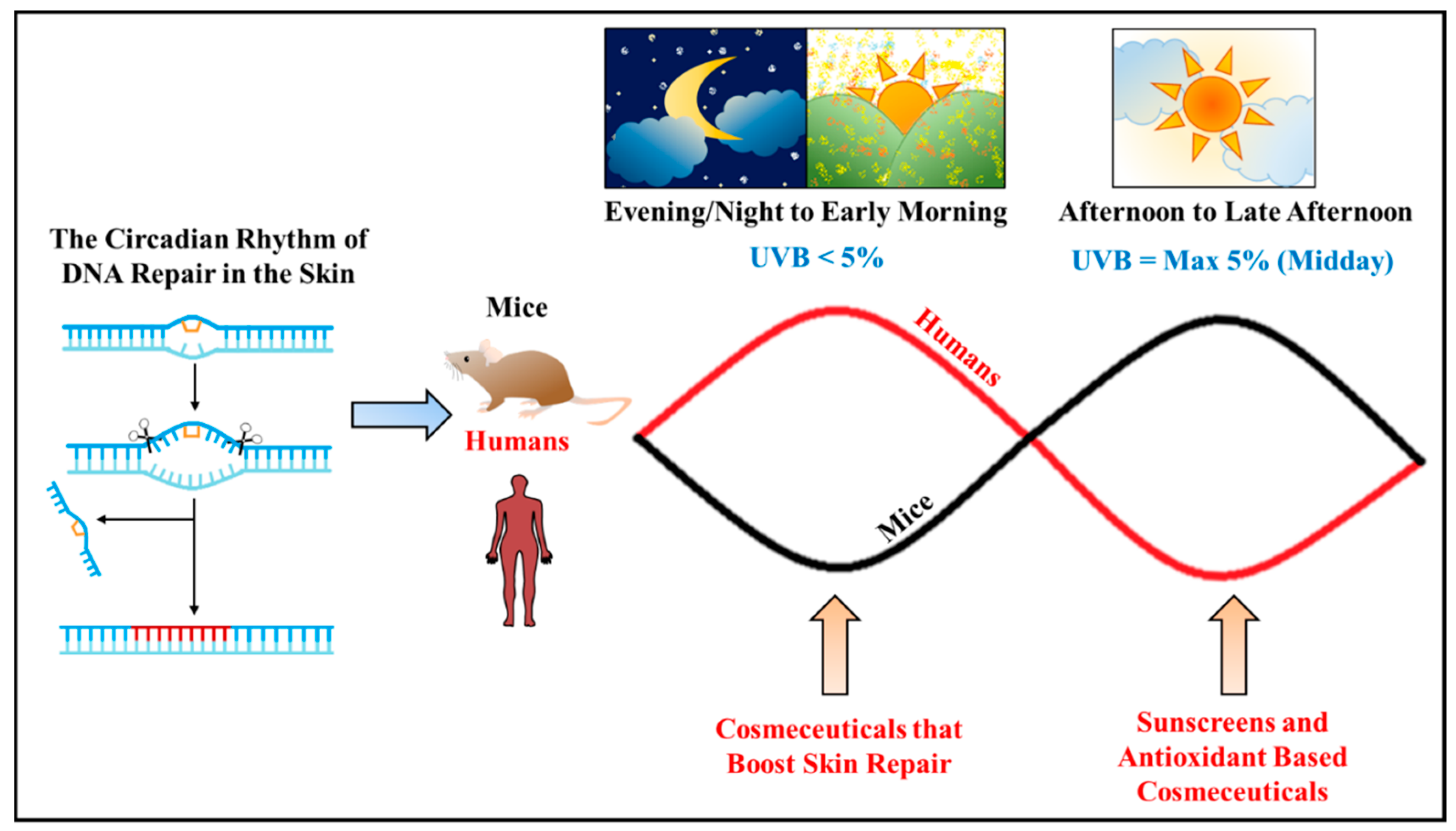
2.2. Rhythms in Animals and Humans: How Do We Maximize Our Cosmetic Product?
3. The Role of the Circadian Rhythm in Skin Pigmentation
4. Loss of Circadian Rhythm alongside Skin Elasticity and Collagen Degradation
4.1. Rhythmic Expressions of TIMP3 and AQP3
4.2. Role of OPN3 in Collagen Degradation
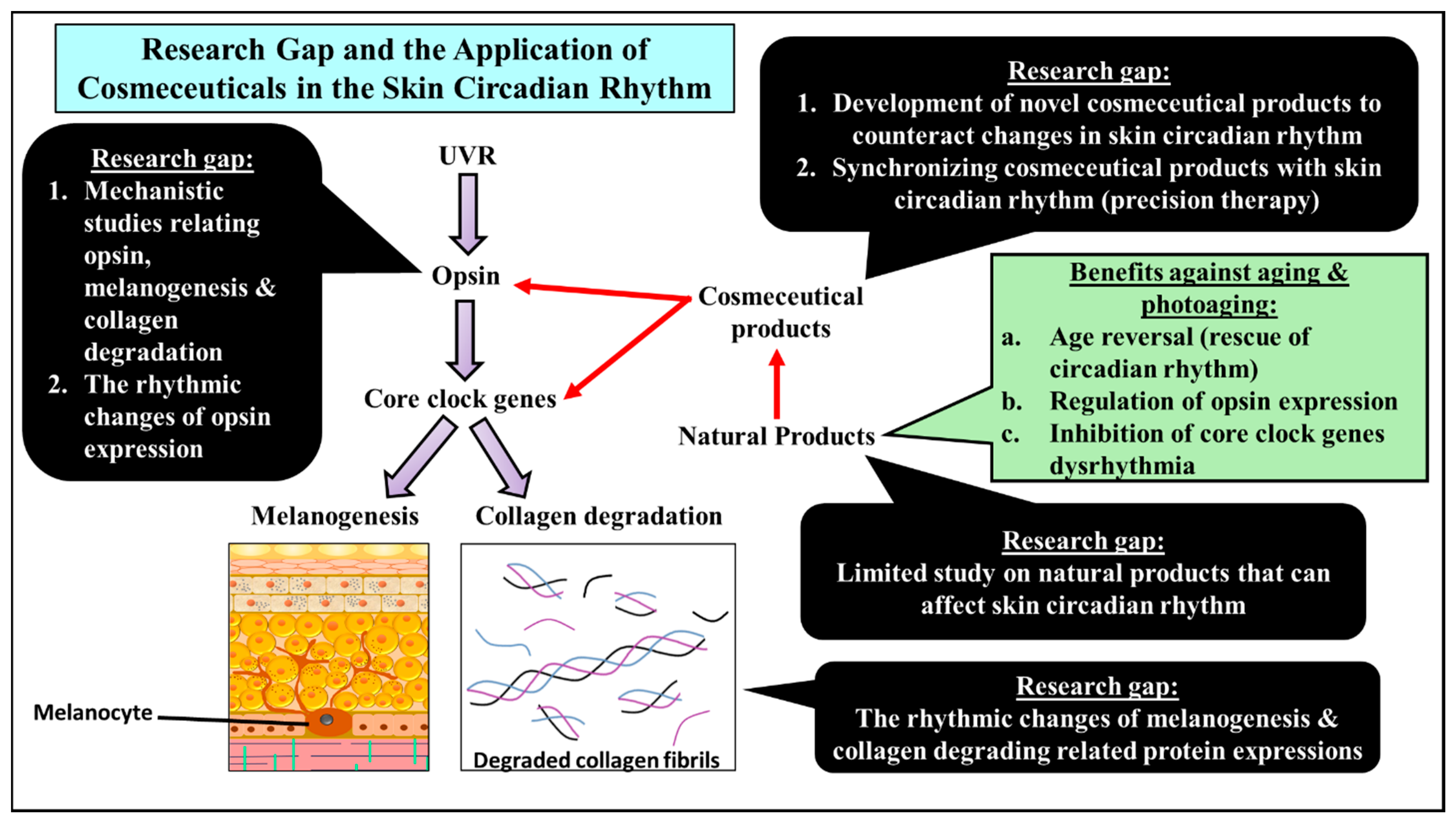
5. Limitations and Research Gap in the Study of Skin Circadian Rhythm
6. Melatonin and Natural Products as Cosmeceutical Agents
6.1. Melatonin as a Cosmeceutical Product
6.2. Potential Use of Natural Products as a Modulator of the Circadian Rhythm
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sarkar, S.; Gaddameedhi, S. UV-B-induced erythema in human skin: The circadian clock is ticking. J. Investig. Dermatol. 2018, 138, 248–251. [Google Scholar] [CrossRef] [PubMed]
- Buhr, E.D.; Takahashi, J.S. Molecular components of the mammalian circadian clock. Handb. Exp. Pharmacol. 2013, 15, 3–27. [Google Scholar] [CrossRef]
- Akashi, M.; Takumi, T. The orphan nuclear receptor RORα regulates circadian transcription of the mammalian core-clock Bmal1. Nat. Struct. Mol. Biol. 2005, 12, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Lazar, M.A. The orphan nuclear receptor Rev-erbalpha recruits the N-CoR/histone deacetylase 3 corepressor to regulate the circadian Bmal1 gene. Mol. Endocrinol. 2005, 19, 1452–1459. [Google Scholar] [CrossRef]
- Guillaumond, F.; Dardente, H.; Giguère, V.; Cermakian, N. Differential control of Bmal1 circadian transcription by REV-ERB and ROR nuclear receptors. J. Biol. Rhythm. 2005, 20, 391–403. [Google Scholar] [CrossRef]
- Manzella, N.; Bracci, M.; Strafella, E.; Staffolani, S.; Ciarapica, V.; Copertaro, A.; Rapisarda, V.; Ledda, C.; Amati, M.; Valentino, M.; et al. Circadian modulation of 8-oxoguanine DNA damage repair. Sci. Rep. 2015, 5, 13752. [Google Scholar] [CrossRef]
- Gambichler, T.; Bader, A.; Vojvodic, M.; Bechara, F.G.; Sauermann, K.; Altmeyer, P.; Hoffmann, K. Impact of UVA exposure on psychological parameters and circulating serotonin and melatonin. BMC Dermatol. 2002, 2, 1–7. [Google Scholar] [CrossRef][Green Version]
- Zittermann, A.; Gummert, J.F. Nonclassical Vitamin D Actions. Nutrients 2010, 2, 408–425. [Google Scholar] [CrossRef]
- Slominski, A.T.; Chaiprasongsuk, A.; Janjetovic, Z.; Kim, T.K.; Stefan, J.; Slominski, R.M.; Hanumanthu, V.S.; Raman, C.; Qayyum, S.; Song, Y.; et al. Photoprotective Properties of Vitamin D and Lumisterol Hydroxyderivatives. Cell Biochem. Biophys. 2020, 78, 165–180. [Google Scholar] [CrossRef]
- Bocheva, G.; Slominski, R.M.; Slominski, A.T. The Impact of Vitamin D on Skin Aging. Int. J. Mol. Sci. 2021, 22, 97. [Google Scholar] [CrossRef]
- Slominski, A.T.; Zmijewski, M.A.; Plonka, P.M.; Szaflarski, J.P.; Paus, R. How UV Light Touches the Brain and Endocrine System Through Skin, and Why. Endocrinology 2018, 159, 1992–2007. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T. Ultraviolet radiation (UVR) activates central neuro-endocrine-immune system. Photodermatol. Photoimmunol. Photomed. 2015, 31, 121–123. [Google Scholar] [CrossRef] [PubMed]
- Skobowiat, C.; Postlethwaite, A.E.; Slominski, A.T. Skin Exposure to Ultraviolet B Rapidly Activates Systemic Neuroendocrine and Immunosuppressive Responses. Photochem. Photobiol. 2017, 93, 1008–1015. [Google Scholar] [CrossRef] [PubMed]
- Seiffert, K.; Granstein, R.D. Neuropeptides and neuroendocrine hormones in ultraviolet radiation-induced immunosuppression. Methods 2002, 28, 97–103. [Google Scholar] [CrossRef]
- Moattari, C.R.; Granstein, R.D. Neuropeptides and neurohormones in immune, inflammatory and cellular responses to ultraviolet radiation. Acta Physiol. 2021, 232, e13644. [Google Scholar] [CrossRef]
- Scharffetter–Kochanek, K.; Brenneisen, P.; Wenk, J.; Herrmann, G.; Ma, W.; Kuhr, L.; Meewes, C.; Wlaschek, M. Photoaging of the skin from phenotype to mechanisms. Exp. Gerontol. 2000, 35, 307–316. [Google Scholar] [CrossRef]
- You, Y.-H.; Lee, D.-H.; Yoon, J.-H.; Nakajima, S.; Yasui, A.; Pfeifer, G.P. Cyclobutane pyrimidine dimers are responsible for the vast majority of mutations induced by UVB irradiation in mammalian cells. J. Biol. Chem. 2001, 276, 44688–44694. [Google Scholar] [CrossRef]
- Mahendra, C.K.; Abidin, S.A.Z.; Htar, T.T.; Chuah, L.H.; Khan, S.U.; Ming, L.C.; Tang, S.Y.; Pusparajah, P.; Goh, B.H. Counteracting the ramifications of UVB irradiation and photoaging with Swietenia macrophylla King seed. Molecules 2021, 26, 2000. [Google Scholar] [CrossRef]
- Fisher, G.J.; Kang, S.; Varani, J.; Bata-Csorgo, Z.; Wan, Y.; Datta, S.; Voorhees, J.J. Mechanisms of photoaging and chronological skin aging. Arch. Dermatol. 2002, 138, 1462–1470. [Google Scholar] [CrossRef]
- Sandu, C.; Dumas, M.; Malan, A.; Sambakhe, D.; Marteau, C.; Nizard, C.; Schnebert, S.; Perrier, E.; Challet, E.; Pévet, P.; et al. Human skin keratinocytes, melanocytes, and fibroblasts contain distinct circadian clock machineries. Cell. Mol. Life Sci. 2012, 69, 3329–3339. [Google Scholar] [CrossRef]
- Gaddameedhi, S.; Selby, C.P.; Kaufmann, W.K.; Smart, R.C.; Sancar, A. Control of skin cancer by the circadian rhythm. Proc. Natl. Acad. Sci. USA 2011, 108, 18790–18795. [Google Scholar] [CrossRef] [PubMed]
- Koritala, B.S.C.; Porter, K.I.; Arshad, O.A.; Gajula, R.P.; Mitchell, H.D.; Arman, T.; Manjanatha, M.G.; Teeguarden, J.; Van Dongen, H.P.A.; McDermott, J.E.; et al. Night shift schedule causes circadian dysregulation of DNA repair genes and elevated DNA damage in humans. J. Pineal Res. 2021, 70, e12726. [Google Scholar] [CrossRef] [PubMed]
- Mahendra, C.K.; Tan, L.T.-H.; Yap, W.H.; Chan, C.K.; Pusparajah, P.; Goh, B.H. An optimized cosmetic screening assay for ultraviolet B (UVB) protective property of natural products. Prog. Drug Discov. Biomed. Sci. 2019, 2, a0000021. [Google Scholar] [CrossRef]
- Widel, M.; Krzywon, A.; Gajda, K.; Skonieczna, M.; Rzeszowska-Wolny, J. Induction of bystander effects by UVA, UVB, and UVC radiation in human fibroblasts and the implication of reactive oxygen species. Free Radic. Biol. Med. 2014, 68, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Nikkola, V.; Miettinen, M.E.; Karisola, P.; Grönroos, M.; Ylianttila, L.; Alenius, H.; Snellman, E.; Partonen, T. Ultraviolet B radiation modifies circadian time in epidermal skin and in subcutaneous adipose tissue. Photodermatol. Photoimmunol. Photomed. 2019, 35, 157–163. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, P.; Li, H.; Dai, J. BMAL1 and CLOCK proteins in regulating UVB-induced apoptosis and DNA damage responses in human keratinocytes. J. Cell. Physiol. 2018, 233, 9563–9574. [Google Scholar] [CrossRef]
- Kawara, S.; Mydlarski, R.; Mamelak, A.J.; Freed, I.; Wang, B.; Watanabe, H.; Shivji, G.; Tavadia, S.K.; Suzuki, H.; Bjarnason, G.A.; et al. Low-dose ultraviolet B rays alter the mRNA expression of the circadian clock genes in cultured human keratinocytes. J. Investig. Dermatol. 2002, 119, 1220–1223. [Google Scholar] [CrossRef]
- Joo, J.-H.; Hong, I.-K.; Kim, N.K.; Choi, E. Trichosanthes kirilowii extract enhances repair of UVB radiation-induced DNA damage by regulating BMAL1 and miR-142-3p in human keratinocytes. Mol. Med. Rep. 2018, 17, 877–883. [Google Scholar] [CrossRef]
- Park, S.; Kim, K.; Bae, I.H.; Lee, S.H.; Jung, J.; Lee, T.R.; Cho, E.G. TIMP3 is a CLOCK-dependent diurnal gene that inhibits the expression of UVB-induced inflammatory cytokines in human keratinocytes. FASEB J. 2018, 32, 1510–1523. [Google Scholar] [CrossRef]
- Pavlovsky, M.; Baum, S.; Shpiro, D.; Pavlovsky, L.; Pavlotsky, F. Narrow band UVB: Is it effective and safe for paediatric psoriasis and atopic dermatitis? J. Eur. Acad. Dermatol. Venereol. 2011, 25, 727–729. [Google Scholar] [CrossRef]
- Xiao, B.-H.; Wu, Y.; Sun, Y.; Chen, H.-D.; Gao, X.-H. Treatment of vitiligo with NB-UVB: A systematic review. J. Dermatolog. Treat. 2015, 26, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Pugashetti, R.; Lim, H.W.; Koo, J. Broadband UVB revisited: Is the narrowband UVB fad limiting our therapeutic options? J. Dermatolog. Treat. 2010, 21, 326–330. [Google Scholar] [CrossRef] [PubMed]
- Kunisada, M.; Kumimoto, H.; Ishizaki, K.; Sakumi, K.; Nakabeppu, Y.; Nishigori, C. Narrow-band UVB induces more carcinogenic skin tumors than broad-band UVB through the formation of cyclobutane pyrimidine dimer. J. Investig. Dermatol. 2007, 127, 2865–2871. [Google Scholar] [CrossRef] [PubMed]
- Nikkola, V.; Grönroos, M.; Huotari-Orava, R.; Kautiainen, H.; Ylianttila, L.; Karppinen, T.; Partonen, T.; Snellman, E. Circadian time effects on NB-UVB-induced erythema in human skin in vivo. J. Investig. Dermatol. 2018, 138, 464–467. [Google Scholar] [CrossRef] [PubMed]
- Papp, S.J.; Huber, A.L.; Jordan, S.D.; Kriebs, A.; Nguyen, M.; Moresco, J.J.; Yates, J.R.; Lamia, K.A. DNA damage shifts circadian clock time via Hausp-dependent Cry1 stabilization. eLife 2015, 4, e04883. [Google Scholar] [CrossRef]
- Liu, F.-T.; Newland, A.C.; Jia, L. Bax conformational change is a crucial step for PUMA-mediated apoptosis in human leukemia. Biochem. Biophys. Res. Commun. 2003, 310, 956–962. [Google Scholar] [CrossRef]
- Henseleit, U.; Zhang, J.; Wanner, R.; Haase, I.; Kolde, G.; Rosenbach, T. Role of p53 in UVB-induced apoptosis in human HaCaT keratinocytes. J. Investig. Dermatol. 1997, 109, 722–727. [Google Scholar] [CrossRef]
- Miki, T.; Matsumoto, T.; Zhao, Z.; Lee, C.C. p53 regulates Period2 expression and the circadian clock. Nat. Commun. 2013, 4, 2444. [Google Scholar] [CrossRef]
- Gaddameedhi, S.; Selby, C.P.; Kemp, M.G.; Ye, R.; Sancar, A. The circadian clock controls sunburn apoptosis and erythema in mouse skin. J. Investig. Dermatol. 2015, 135, 1119–1127. [Google Scholar] [CrossRef]
- Geyfman, M.; Kumar, V.; Liu, Q.; Ruiz, R.; Gordon, W.; Espitia, F.; Cam, E.; Millar, S.E.; Smyth, P.; Ihler, A.; et al. Brain and muscle Arnt-like protein-1 (BMAL1) controls circadian cell proliferation and susceptibility to UVB-induced DNA damage in the epidermis. Proc. Natl. Acad. Sci. USA 2012, 109, 11758–11763. [Google Scholar] [CrossRef]
- Sugitani, N.; Sivley, R.M.; Perry, K.E.; Capra, J.A.; Chazin, W.J. XPA: A key scaffold for human nucleotide excision repair. DNA Repair 2016, 44, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Ferrini, R.L.; Perlman, M.; Hill, L. American college of preventive medicine practice policy statement: Skin protection from ultraviolet light exposure. Am. J. Prev. Med. 1998, 14, 83–86. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Brenner, M.; Hearing, V.J. The Regulation of Skin Pigmentation. J. Biol. Chem. 2007, 282, 27557–27561. [Google Scholar] [CrossRef] [PubMed]
- Solano, F. Photoprotection versus photodamage: Updating an old but still unsolved controversy about melanin. Polym. Int. 2016, 65, 1276–1287. [Google Scholar] [CrossRef]
- Lai, X.; Wichers, H.J.; Soler-Lopez, M.; Dijkstra, B.W. Structure of human tyrosinase related protein 1 reveals a binuclear zinc active site important for melanogenesis. Angew. Chem. Int. Ed. Engl. 2017, 56, 9812–9815. [Google Scholar] [CrossRef] [PubMed]
- Haltaufderhyde, K.; Ozdeslik, R.N.; Wicks, N.L.; Najera, J.A.; Oancea, E. Opsin expression in human epidermal skin. Photochem. Photobiol. 2015, 91, 117–123. [Google Scholar] [CrossRef]
- Terakita, A. The opsins. Genome Biol. 2005, 6, 213. [Google Scholar] [CrossRef]
- Regazzetti, C.; Sormani, L.; Debayle, D.; Bernerd, F.; Tulic, M.K.; De Donatis, G.M.; Chignon-Sicard, B.; Rocchi, S.; Passeron, T. Melanocytes sense blue light and regulate pigmentation through opsin-3. J. Investig. Dermatol. 2018, 138, 171–178. [Google Scholar] [CrossRef]
- Panda, S.; Sato Trey, K.; Castrucci, A.M.; Rollag Mark, D.; DeGrip Willem, J.; Hogenesch John, B.; Provencio, I.; Kay Steve, A. Melanopsin (Opn4) requirement for normal light-induced circadian phase shifting. Science 2002, 298, 2213–2216. [Google Scholar] [CrossRef]
- Buhr, E.D.; Yue, W.W.S.; Ren, X.; Jiang, Z.; Liao, H.-W.R.; Mei, X.; Vemaraju, S.; Nguyen, M.-T.; Reed, R.R.; Lang, R.A.; et al. Neuropsin (OPN5)-mediated photoentrainment of local circadian oscillators in mammalian retina and cornea. Proc. Natl. Acad. Sci. USA 2015, 112, 13093–13098. [Google Scholar] [CrossRef]
- de Assis, L.V.M.; Moraes, M.N.; Castrucci, A.M.L. Heat shock antagonizes UVA-induced responses in murine melanocytes and melanoma cells: An unexpected interaction. Photochem. Photobiol. Sci. 2017, 16, 633–648. [Google Scholar] [CrossRef] [PubMed]
- de Assis, L.V.M.; Mendes, D.; Silva, M.M.; Kinker, G.S.; Pereira-Lima, I.; Moraes, M.N.; Menck, C.F.M.; Castrucci, A.M.d.L. Melanopsin mediates UVA-dependent modulation of proliferation, pigmentation, apoptosis, and molecular clock in normal and malignant melanocytes. Biochim. Biophys. Acta Mol. Cell Res. 2020, 1867, 118789. [Google Scholar] [CrossRef] [PubMed]
- Hardman, J.A.; Tobin, D.J.; Haslam, I.S.; Farjo, N.; Farjo, B.; Al-Nuaimi, Y.; Grimaldi, B.; Paus, R. The peripheral clock regulates human pigmentation. J. Investig. Dermatol. 2015, 135, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, S.; Porter, K.I.; Dakup, P.P.; Gajula, R.P.; Koritala, B.S.C.; Hylton, R.; Kemp, M.G.; Wakamatsu, K.; Gaddameedhi, S. Circadian clock protein BMAL1 regulates melanogenesis through MITF in melanoma cells. Pigment Cell Melanoma Res. 2021, 34, 955–965. [Google Scholar] [CrossRef] [PubMed]
- de Assis, L.V.M.; Moraes, M.N.; da Silveira Cruz-Machado, S.; Castrucci, A.M.L. The effect of white light on normal and malignant murine melanocytes: A link between opsins, clock genes, and melanogenesis. Biochim. Biophys. Acta 2016, 1863, 1119–1133. [Google Scholar] [CrossRef] [PubMed]
- de Assis, L.V.M.; Moraes, M.N.; Magalhães-Marques, K.K.; Castrucci, A.M.d.L. Melanopsin and rhodopsin mediate UVA-induced immediate pigment darkening: Unravelling the photosensitive system of the skin. Eur. J. Cell Biol. 2018, 97, 150–162. [Google Scholar] [CrossRef]
- Wang, Y.; Lan, Y.; Yang, X.; Gu, Y.; Lu, H. TGFβ2 upregulates tyrosinase activity through opsin-3 in human skin melanocytes in vitro. J. Investig. Dermatol. 2021, 141, 2679–2689. [Google Scholar] [CrossRef]
- Ozdeslik, R.N.; Olinski, L.E.; Trieu, M.M.; Oprian, D.D.; Oancea, E. Human nonvisual opsin 3 regulates pigmentation of epidermal melanocytes through functional interaction with melanocortin 1 receptor. Proc. Natl. Acad. Sci. USA 2019, 116, 11508–11517. [Google Scholar] [CrossRef]
- Lan, Y.; Zeng, W.; Dong, X.; Lu, H. Opsin 5 is a key regulator of ultraviolet radiation-induced melanogenesis in human epidermal melanocytes. Br. J. Dermatol. 2021, 185, 391–404. [Google Scholar] [CrossRef]
- Ota, W.; Nakane, Y.; Hattar, S.; Yoshimura, T. Impaired circadian photoentrainment in Opn5-null mice. iScience 2018, 6, 299–305. [Google Scholar] [CrossRef]
- Buhr, E.D.; Vemaraju, S.; Diaz, N.; Lang, R.A.; Van Gelder, R.N. Neuropsin (OPN5) mediates local light-dependent induction of circadian clock genes and circadian photoentrainment in exposed murine skin. Curr. Biol. 2019, 29, 3478–3487. [Google Scholar] [CrossRef] [PubMed]
- Feller, L.; Masilana, A.; Khammissa, R.A.G.; Altini, M.; Jadwat, Y.; Lemmer, J. Melanin: The biophysiology of oral melanocytes and physiological oral pigmentation. Head Face Med. 2014, 10, 8. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Tobin, D.J.; Shibahara, S.; Wortsman, J. Melanin pigmentation in mammalian skin and its hormonal regulation. Physiol. Rev. 2004, 84, 1155–1228. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Wortsman, J. Neuroendocrinology of the Skin. Endocr. Rev. 2000, 21, 457–487. [Google Scholar] [CrossRef] [PubMed]
- Enkhtaivan, E.; Lee, C.H. Role of Amine Neurotransmitters and Their Receptors in Skin Pigmentation: Therapeutic Implication. Int. J. Mol. Sci. 2021, 22, 8071. [Google Scholar] [CrossRef]
- Chiang, H.-M.; Lin, T.-J.; Chiu, C.-Y.; Chang, C.-W.; Hsu, K.-C.; Fan, P.-C.; Wen, K.-C. Coffea arabica extract and its constituents prevent photoaging by suppressing MMPs expression and MAP kinase pathway. Food Chem. Toxicol. 2011, 49, 309–318. [Google Scholar] [CrossRef]
- Heng, J.K.; Aw, D.C.W.; Tan, K.B. Solar elastosis in its papular form: Uncommon, mistakable. Case Rep. Dermatol. 2014, 6, 124–128. [Google Scholar] [CrossRef]
- Pittayapruek, P.; Meephansan, J.; Prapapan, O.; Komine, M.; Ohtsuki, M. Role of matrix metalloproteinases in photoaging and photocarcinogenesis. Int. J. Mol. Sci. 2016, 17, 868. [Google Scholar] [CrossRef]
- Park, S.; Lee, E.-S.; Park, N.-H.; Hwang, K.; Cho, E.-G. Circadian expression of TIMP3 is disrupted by UVB irradiation and recovered by green tea extracts. Int. J. Mol. Sci. 2019, 20, 862. [Google Scholar] [CrossRef]
- Yeom, M.; Lee, H.; Shin, S.; Park, D.; Jung, E. PER, a circadian clock component, mediates the suppression of MMP-1 expression in HaCaT keratinocytes by cAMP. Molecules 2018, 23, 745. [Google Scholar] [CrossRef]
- Matsunaga, N.; Itcho, K.; Hamamura, K.; Ikeda, E.; Ikeyama, H.; Furuichi, Y.; Watanabe, M.; Koyanagi, S.; Ohdo, S. 24-hour rhythm of aquaporin-3 function in the epidermis Is regulated by molecular clocks. J. Investig. Dermatol. 2014, 134, 1636–1644. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Hara, M.; Sougrat, R.; Verbavatz, J.M.; Verkman, A.S. Impaired stratum corneum hydration in mice lacking epidermal water channel aquaporin-3. J. Biol. Chem. 2002, 277, 17147–17153. [Google Scholar] [CrossRef] [PubMed]
- Hara, M.; Ma, T.; Verkman, A.S. Selectively reduced glycerol in skin of aquaporin-3-deficient mice may account for impaired skin hydration, elasticity, and barrier recovery. J. Biol. Chem. 2002, 277, 46616–46621. [Google Scholar] [CrossRef] [PubMed]
- Hara-Chikuma, M.; Verkman, A.S. Aquaporin-3 facilitates epidermal cell migration and proliferation during wound healing. J. Mol. Med. 2008, 86, 221–231. [Google Scholar] [CrossRef]
- Vincenti, M.P. The matrix metalloproteinase (MMP) and tissue inhibitor of metalloproteinase (TIMP) genes. In Matrix Metalloproteinase Protocols; Clark, I.M., Ed.; Humana Press: Totowa, NJ, USA, 2001; pp. 121–148. [Google Scholar]
- Kassiri, Z.; Oudit, G.Y.; Kandalam, V.; Awad, A.; Wang, X.; Ziou, X.; Maeda, N.; Herzenberg, A.M.; Scholey, J.W. Loss of TIMP3 enhances interstitial nephritis and fibrosis. J. Am. Soc. Nephrol. 2009, 20, 1223–1235. [Google Scholar] [CrossRef]
- Kandalam, V.; Basu, R.; Abraham, T.; Wang, X.; Awad, A.; Wang, W.; Lopaschuk, G.D.; Maeda, N.; Oudit, G.Y.; Kassiri, Z. Early activation of matrix metalloproteinases underlies the exacerbated systolic and diastolic dysfunction in mice lacking TIMP3 following myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 2010, 299, H1012–H1023. [Google Scholar] [CrossRef]
- Stöhr, R.; Cavalera, M.; Menini, S.; Mavilio, M.; Casagrande, V.; Rossi, C.; Urbani, A.; Cardellini, M.; Pugliese, G.; Menghini, R.; et al. Loss of TIMP3 exacerbates atherosclerosis in ApoE null mice. Atherosclerosis 2014, 235, 438–443. [Google Scholar] [CrossRef]
- Black, R.A. TIMP3 checks inflammation. Nat. Genet. 2004, 36, 934–935. [Google Scholar] [CrossRef]
- Sharma, M.R.; Mitrani, R.; Werth, V.P. Effect of TNFα blockade on UVB-induced inflammatory cell migration and collagen loss in mice. J. Photochem. Photobiol. B. 2020, 213, 112072. [Google Scholar] [CrossRef]
- Lan, Y.; Wang, Y.; Lu, H. Opsin 3 is a key regulator of ultraviolet A-induced photoageing in human dermal fibroblast cells. Br. J. Dermatol. 2020, 182, 1228–1244. [Google Scholar] [CrossRef]
- Vemaraju, S.; Nayak, G.; Buhr, E.D.; Odaka, Y.; Zhang, K.X.; Mocko, J.A.; Smith, A.N.; Upton, B.A.; Zhan, J.J.; Borra, V.J.; et al. An adipocyte light-Opsin 3 pathway regulates the circadian clock and energy balance. bioRxiv 2018, 368373. [Google Scholar] [CrossRef]
- Wang, H.; van Spyk, E.; Liu, Q.; Geyfman, M.; Salmans, M.; Kumar, V.; Ihler, A.; Li, N.; Takahashi, J.S.; Andersen, B. Time-restricted feeding shifts the skin circadian clock and alters UVB-induced DNA damage. Cell Rep. 2017, 20, 1061–1072. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Charareh, P.; Lei, X.; Zhong, J.L. Autophagy: Multiple mechanisms to protect skin from ultraviolet radiation-driven photoaging. Oxid. Med. Cell. Longev. 2019, 2019, 8135985. [Google Scholar] [CrossRef] [PubMed]
- Pernodet, N.; Dong, K.; Pelle, E. Autophagy in human skin fibroblasts: Comparison between young and aged cells and evaluation of its cellular rhythm and response to Ultraviolet A radiation. J. Cosmet. Sci. 2016, 67, 13–20. [Google Scholar] [PubMed]
- Spörl, F.; Korge, S.; Jürchott, K.; Wunderskirchner, M.; Schellenberg, K.; Heins, S.; Specht, A.; Stoll, C.; Klemz, R.; Maier, B.; et al. Krüppel-like factor 9 is a circadian transcription factor in human epidermis that controls proliferation of keratinocytes. Proc. Natl. Acad. Sci. USA 2012, 109, 10903–10908. [Google Scholar] [CrossRef]
- Dong, K.; Pelle, E.; Yarosh, D.B.; Pernodet, N. Sirtuin 4 identification in normal human epidermal keratinocytes and its relation to sirtuin 3 and energy metabolism under normal conditions and UVB-induced stress. Exp. Dermatol. 2012, 21, 231–233. [Google Scholar] [CrossRef]
- Avitabile, D.; Ranieri, D.; Nicolussi, A.; D’Inzeo, S.; Capriotti, A.L.; Genovese, L.; Proietti, S.; Cucina, A.; Coppa, A.; Samperi, R.; et al. Peroxiredoxin 2 nuclear levels are regulated by circadian clock synchronization in human keratinocytes. Int. J. Biochem. Cell Biol. 2014, 53, 24–34. [Google Scholar] [CrossRef]
- Bai, X.-Y.; Li, S.; Wang, M.; Li, X.; Yang, Y.; Xu, Z.; Li, B.; Li, Y.; Xia, K.; Chen, H.; et al. Krüppel-like factor 9 down-regulates matrix metalloproteinase 9 transcription and suppresses human breast cancer invasion. Cancer Lett. 2018, 412, 224–235. [Google Scholar] [CrossRef]
- McAlpine, C.S.; Swirski, F.K. Circadian influence on metabolism and inflammation in atherosclerosis. Circ. Res. 2016, 119, 131–141. [Google Scholar] [CrossRef]
- Sengupta, S.; Tang, S.Y.; Devine, J.C.; Anderson, S.T.; Nayak, S.; Zhang, S.L.; Valenzuela, A.; Fisher, D.G.; Grant, G.R.; López, C.B.; et al. Circadian control of lung inflammation in influenza infection. Nat. Commun. 2019, 10, 4107. [Google Scholar] [CrossRef]
- Hunt, K.A.; Fate, J.; Dodds, B. Cultural and social influences on the perception of beauty: A case analysis of the cosmetics industry. J. Bus. Case Stud. 2011, 7, 1–10. [Google Scholar] [CrossRef]
- Amberg, N.; Fogarassy, C. Green consumer behavior in the cosmetics market. Resources 2019, 8, 137. [Google Scholar] [CrossRef]
- Mahendra, C.K.; Tan, L.T.; Mahendra, C.K.; Ser, H.-L.; Pusparajah, P.; Htar, T.T.; Chuah, L.-H.; Yap, W.H.; Tang, S.Y.; Ming, L.C.; et al. The Potential of sky fruit as an anti-aging and wound healing cosmeceutical agent. Cosmetics 2021, 8, 79. [Google Scholar] [CrossRef]
- Milani, M.; Sparavigna, A. Antiaging efficacy of melatonin-based day and night creams: A randomized, split-face, assessor-blinded proof-of-concept trial. Clin. Cosmet. Investig. Dermatol. 2018, 11, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Marchena, A.M.; Franco, L.; Romero, A.M.; Barriga, C.; Rodríguez, A.B. Lycopene and melatonin: Antioxidant compounds in cosmetic formulations. Skin Pharmacol. Physiol. 2020, 33, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Marto, J.; Ascenso, A.; Gonçalves, L.M.; Gouveia, L.F.; Manteigas, P.; Pinto, P.; Oliveira, E.; Almeida, A.J.; Ribeiro, H.M. Melatonin-based pickering emulsion for skin’s photoprotection. Drug Deliv. 2016, 23, 1594–1607. [Google Scholar] [CrossRef]
- Granger, C.; Brown, A.; Aladren, S.; Narda, M. Night cream containing melatonin, carnosine and Helichrysum italicum extract helps reduce skin reactivity and signs of photodamage: Ex vivo and clinical studies. Dermatol. Ther. 2020, 10, 1315–1329. [Google Scholar] [CrossRef]
- Puviani, M.; Eisendle, K.; Milani, M. Anti-wrinkle efficacy of a melatonin-based eye contour balm: An antera 3D prospective 2-month pilot trial. J. Clin. Exp. Dermatol. Res. 2020, 11, 534. [Google Scholar] [CrossRef]
- Ndiaye, M.A.; Nihal, M.; Wood, G.S.; Ahmad, N. Skin, reactive oxygen species, and circadian clocks. Antioxid. Redox Signal. 2013, 20, 2982–2996. [Google Scholar] [CrossRef]
- Slominski, A.; Tobin, D.J.; Zmijewski, M.A.; Wortsman, J.; Paus, R. Melatonin in the skin: Synthesis, metabolism and functions. Trends Endocrinol. Metab. 2008, 19, 17–24. [Google Scholar] [CrossRef]
- Juhnevica-Radenkova, K.; Moreno, D.A.; Ikase, L.; Drudze, I.; Radenkovs, V. Naturally occurring melatonin: Sources and possible ways of its biosynthesis. Compr. Rev. Food Sci. Food Saf. 2020, 19, 4008–4030. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.; Goyarts, E.; Rella, A.; Pelle, E.; Wong, Y.H.; Pernodet, N. Age associated decrease of MT-1 melatonin receptor in human dermal skin fibroblasts impairs protection against UV-induced DNA damage. Int. J. Mol. Sci. 2020, 21, 326. [Google Scholar] [CrossRef] [PubMed]
- Fischer, T.W.; Slominski, A.; Zmijewski, M.A.; Reiter, R.J.; Paus, R. Melatonin as a major skin protectant: From free radical scavenging to DNA damage repair. Exp. Dermatol. 2008, 17, 713–730. [Google Scholar] [CrossRef] [PubMed]
- Fischer, T.W.; Kleszczyński, K.; Hardkop, L.H.; Kruse, N.; Zillikens, D. Melatonin enhances antioxidative enzyme gene expression (CAT, GPx, SOD), prevents their UVR-induced depletion, and protects against the formation of DNA damage (8-hydroxy-2’-deoxyguanosine) in ex vivo human skin. J. Pineal Res. 2013, 54, 303–312. [Google Scholar] [CrossRef]
- Scheuer, C.; Pommergaard, H.C.; Rosenberg, J.; Gögenur, I. Dose dependent sun protective effect of topical melatonin: A randomized, placebo-controlled, double-blind study. J. Dermatol. Sci. 2016, 84, 178–185. [Google Scholar] [CrossRef]
- Rusanova, I.; Martínez-Ruiz, L.; Florido, J.; Rodríguez-Santana, C.; Guerra-Librero, A.; Acuña-Castroviejo, D.; Escames, G. Protective effects of melatonin on the skin: Future perspectives. Int. J. Mol. Sci. 2019, 20, 4948. [Google Scholar] [CrossRef]
- Kim, T.-K.; Kleszczyński, K.; Janjetovic, Z.; Sweatman, T.; Lin, Z.; Li, W.; Reiter, R.J.; Fischer, T.W.; Slominski, A.T. Metabolism of melatonin and biological activity of intermediates of melatoninergic pathway in human skin cells. FASEB J. 2013, 27, 2742–2755. [Google Scholar] [CrossRef]
- Slominski, A.T.; Semak, I.; Fischer, T.W.; Kim, T.-K.; Kleszczyński, K.; Hardeland, R.; Reiter, R.J. Metabolism of melatonin in the skin: Why is it important? Exp. Dermatol. 2017, 26, 563–568. [Google Scholar] [CrossRef]
- Tan, D.X.; Hardeland, R.; Manchester, L.C.; Galano, A.; Reiter, R.J. Cyclic-3-hydroxymelatonin (C3HOM), a potent antioxidant, scavenges free radicals and suppresses oxidative reactions. Curr. Med. Chem. 2014, 21, 1557–1565. [Google Scholar] [CrossRef]
- Favero, G.; Franceschetti, L.; Bonomini, F.; Rodella, L.F.; Rezzani, R. Melatonin as an anti-inflammatory agent modulating inflammasome activation. Int. J. Endocrinol. 2017, 2017, 1835195. [Google Scholar] [CrossRef]
- Slominski, A.T.; Zmijewski, M.A.; Semak, I.; Kim, T.K.; Janjetovic, Z.; Slominski, R.M.; Zmijewski, J.W. Melatonin, mitochondria, and the skin. Cell Mol. Life Sci. 2017, 74, 3913–3925. [Google Scholar] [CrossRef] [PubMed]
- Bocheva, G.; Slominski, R.M.; Janjetovic, Z.; Kim, T.-K.; Böhm, M.; Steinbrink, K.; Reiter, R.J.; Kleszczyński, K.; Slominski, A.T. Protective role of melatonin and its metabolites in skin aging. Int. J. Mol. Sci. 2022, 23, 1238. [Google Scholar] [CrossRef] [PubMed]
- Lellupitiyage Don, S.S.; Robertson, K.L.; Lin, H.-H.; Labriola, C.; Harrington, M.E.; Taylor, S.R.; Farkas, M.E. Nobiletin affects circadian rhythms and oncogenic characteristics in a cell-dependent manner. PLoS ONE 2020, 15, e0236315. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, Y.; Kawano, Y.; Yamazaki, Y.; Onishi, Y. Shikonin shortens the circadian period: Possible involvement of Top2 inhibition. Biochem. Biophys. Res. Commun. 2014, 443, 339–343. [Google Scholar] [CrossRef]
- Yang, J.-T.; Li, Z.-L.; Wu, J.-Y.; Lu, F.-J.; Chen, C.-H. An oxidative stress mechanism of shikonin in human glioma cells. PLoS ONE 2014, 9, e94180. [Google Scholar] [CrossRef]
- Lu, L.; Qin, A.; Huang, H.; Zhou, P.; Zhang, C.; Liu, N.; Li, S.; Wen, G.; Zhang, C.; Dong, W.; et al. Shikonin extracted from medicinal Chinese herbs exerts anti-inflammatory effect via proteasome inhibition. Eur. J. Pharmacol. 2011, 658, 242–247. [Google Scholar] [CrossRef]
- Yang, H.; Zhou, P.; Huang, H.; Chen, D.; Ma, N.; Cui, Q.C.; Shen, S.; Dong, W.; Zhang, X.; Lian, W.; et al. Shikonin exerts antitumor activity via proteasome inhibition and cell death induction in vitro and in vivo. Int. J. Cancer 2009, 124, 2450–2459. [Google Scholar] [CrossRef]
- Yan, Y.; Tan, F.; Miao, H.; Wang, H.; Cao, Y. Effect of shikonin against Candida albicans biofilms. Front. Microbiol. 2019, 10, 1085. [Google Scholar] [CrossRef]
- Yin, S.-Y.; Peng, A.-P.; Huang, L.-T.; Wang, Y.-T.; Lan, C.-W.; Yang, N.-S. The phytochemical shikonin stimulates epithelial-mesenchymal transition (EMT) in skin wound healing. Evid. Based Complement Alternat. Med. 2013, 2013, 262796. [Google Scholar] [CrossRef]
- Huang, X.-Y.; Fu, H.-L.; Tang, H.-Q.; Yin, Z.-Q.; Zhang, W.; Shu, G.; Yin, L.-Z.; Zhao, L.; Yan, X.-R.; Lin, J.-C. Optimization extraction of shikonin using ultrasound-assisted response surface methodology and antibacterial studies. Evid. Based Complement Alternat. Med. 2020, 2020, 1208617. [Google Scholar] [CrossRef]
- Lin, N.; Sato, T.; Takayama, Y.; Mimaki, Y.; Sashida, Y.; Yano, M.; Ito, A. Novel anti-inflammatory actions of nobiletin, a citrus polymethoxy flavonoid, on human synovial fibroblasts and mouse macrophages. Biochem. Pharmacol. 2003, 65, 2065–2071. [Google Scholar] [CrossRef]
- Murakami, A.; Nakamura, Y.; Torikai, K.; Tanaka, T.; Koshiba, T.; Koshimizu, K.; Kuwahara, S.; Takahashi, Y.; Ogawa, K.; Yano, M.; et al. Inhibitory effect of citrus nobiletin on phorbol ester-induced skin inflammation, oxidative stress, and tumor promotion in mice. Cancer Res. 2000, 60, 5059–5066. [Google Scholar] [PubMed]
- He, B.; Nohara, K.; Park, N.; Park, Y.-S.; Guillory, B.; Zhao, Z.; Garcia, J.M.; Koike, N.; Lee, C.C.; Takahashi, J.S.; et al. The small molecule nobiletin targets the molecular oscillator to enhance circadian rhythms and protect against metabolic syndrome. Cell Met. 2016, 23, 610–621. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Zhu, X.; Pan, S.; Fang, Y.; Jiang, F.; Phillips, G.O.; Xu, X. Antimicrobial activity of nobiletin and tangeretin against Pseudomonas. Food Chem. 2012, 132, 1883–1890. [Google Scholar] [CrossRef]
- Kim, H.J.; Yonezawa, T.; Teruya, T.; Woo, J.-T.; Cha, B.-Y. Nobiletin, a polymethoxy flavonoid, reduced endothelin-1 plus SCF-induced pigmentation in human melanocytes. Photochem. Photobiol. 2014, 91, 379–386. [Google Scholar] [CrossRef]
- Kim, J.-J.; Korm, S.; Kim, W.-S.; Kim, O.-S.; Lee, J.-S.; Min, H.-G.; Chin, Y.-W.; Cha, H.-J. Nobiletin suppresses MMP-9 expression through modulation of p38 MAPK activity in human dermal fibrobalsts. Biol. Pharm. Bull. 2014, 37, 158–163. [Google Scholar] [CrossRef]
- Zhou, Z.; Lin, Y.; Gao, L.; Yang, Z.; Wang, S.; Wu, B. Circadian pharmacological effects of berberine on chronic colitis in mice: Role of the clock component Rev-erbα. Biochem. Pharmacol. 2020, 172, 113773. [Google Scholar] [CrossRef]
- Song, Y.C.; Lee, Y.; Kim, H.M.; Hyun, M.Y.; Lim, Y.Y.; Song, K.Y.; Kim, B.J. Berberine regulates melanin synthesis by activating PI3K/AKT, ERK and GSK3β in B16F10 melanoma cells. Int. J. Mol. Med. 2015, 35, 1011–1016. [Google Scholar] [CrossRef]
- Kim, S.; Chung, J.H. Berberine prevents UV-induced MMP-1 and reduction of type I procollagen expression in human dermal fibroblasts. Phytomedicine 2008, 15, 749–753. [Google Scholar] [CrossRef]
- Wang, X.; Yao, X.; Zhu, Z.a.; Tang, T.; Dai, K.; Sadovskaya, I.; Flahaut, S.; Jabbouri, S. Effect of berberine on Staphylococcus epidermidis biofilm formation. Int. J. Antimicrob. Agents 2009, 34, 60–66. [Google Scholar] [CrossRef]
- Kim, H.-S.; Kim, M.-J.; Kim, E.J.; Yang, Y.; Lee, M.-S.; Lim, J.-S. Berberine-induced AMPK activation inhibits the metastatic potential of melanoma cells via reduction of ERK activity and COX-2 protein expression. Biochem. Pharmacol. 2012, 83, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Li, D.X.; Zhang, J.; Zhang, Y.; Zhao, P.W.; Yang, L.M. Inhibitory effect of berberine on human skin squamous cell carcinoma A431 cells. Genet. Mol. Res. 2015, 14, 10553–10568. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Zhou, C.; Xu, H.; Zhang, T.; Wu, B. Chronopharmacological targeting of Rev-erbα by puerarin alleviates hyperhomocysteinemia in mice. Biomed. Pharmacother. 2020, 125, 109936. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Mei, E.; Hu, M.; Zhou, C.; Li, X.; Cai, L.; Li, Z. Effect of puerarin on melanogenesis in human melanocytes and vitiligo mouse models and the underlying mechanism. Phytother. Res. 2019, 33, 205–213. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Ahn, S.-H.; Choi, M.-J.; Yang, I.-J.; Shin, H.-M. Puerarin improves dexamethasone-impaired wound healing in vitro and in vivo by enhancing keratinocyte proliferation and migration. Appl. Sci. 2021, 11, 9343. [Google Scholar] [CrossRef]
- Lee, J.-H.; Jeon, Y.-D.; Lee, Y.-M.; Kim, D.-K. The suppressive effect of puerarin on atopic dermatitis-like skin lesions through regulation of inflammatory mediators in vitro and in vivo. Biochem. Biophys. Res. Commun. 2018, 498, 707–714. [Google Scholar] [CrossRef]
- Yu, Z.; Li, W. Induction of apoptosis by puerarin in colon cancer HT-29 cells. Cancer Lett. 2006, 238, 53–60. [Google Scholar] [CrossRef]
- Izumikawa, M.; Ukai, H.; Takagi, M.; Ueda, H.R.; Shin-ya, K. JBIR-26, a novel natural compound from Streptomyces sp. AK-AH76, regulates mammalian circadian clock. J. Antibiot. 2008, 61, 756–758. [Google Scholar] [CrossRef][Green Version]
- Shin, S.Y.; Lee, D.H.; Gil, H.-N.; Kim, B.S.; Choe, J.-S.; Kim, J.-B.; Lee, Y.H.; Lim, Y. Agerarin, identified from Ageratum houstonianum, stimulates circadian CLOCK-mediated aquaporin-3 gene expression in HaCaT keratinocytes. Sci. Rep. 2017, 7, 11175. [Google Scholar] [CrossRef]
- Shin, S.Y.; Gil, H.-N.; Choi, J.H.; Lim, Y.; Lee, Y.H. Agerarin inhibits α-MSH–induced TYR gene transcription via STAT3 suppression independent of CREB-MITF pathway. J. Dermatol. Sci. 2018, 91, 107–110. [Google Scholar] [CrossRef]
- Ahn, S.S.; Lee, Y.H.; Yeo, H.; Lee, Y.; Min, D.S.; Lim, Y.; Shin, S.Y. Effect of 6,7-dimethoxy-2,2-dimethyl-2H-chromene (agerarin) on the recovery of filaggrin expression through targeting of Janus kinases in the inflammatory skin. J. Food Drug Anal. 2020, 28, 449–460. [Google Scholar] [CrossRef]
- Pastorino, G.; Marchetti, C.F.; Borghesi, B.; Cornara, L.; Ribulla, S.; Burlando, B. Biological activities of the legume crops Melilotus officinalis and Lespedeza capitata for skin care and pharmaceutical applications. Ind. Crops Prod. 2017, 96, 158–164. [Google Scholar] [CrossRef]
- Oh, H.; Mun, Y.J.; Im, S.J.; Lee, S.Y.; Song, H.J.; Lee, H.S.; Woo, W.H. Cucurbitacins from Trichosanthes kirilowii as the inhibitory components on tyrosinase activity and melanin synthesis of B16/F10 melanoma cells. Planta Med. 2002, 68, 832–833. [Google Scholar] [CrossRef] [PubMed]
- Lei, X.; Li, N.; Bai, Z.; Di, J.; Zhang, H.; Dong, P.; Zhang, P. Chemical constituent from the peel of Trichosanthes kirilowii Maxim and their NF-κB inhibitory activity. Nat. Prod. Res. 2020, 35, 5132–5137. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Seo, H.S.; Choi, H.-S.; Cho, S.-G.; Kim, Y.K.; Hong, E.H.; Shin, Y.C.; Ko, S.-G. Trichosanthes kirilowii ethanol extract and Cucurbitacin D inhibit cell growth and induce apoptosis through inhibition of STAT3 activity in breast cancer cells. Evid. Based Complement Alternat. Med. 2013, 2013, 975350. [Google Scholar] [CrossRef] [PubMed]
- Portillo, M.; Mataix, M.; Alonso-Juarranz, M.; Lorrio, S.; Villalba, M.; Rodríguez-Luna, A.; González, S. The aqueous extract of Polypodium leucotomos (Fernblock®) regulates opsin 3 and prevents photooxidation of melanin precursors on skin cells exposed to blue light emitted from digital devices. Antioxidants 2021, 10, 400. [Google Scholar] [CrossRef]
- Salvador, G.; Silvia, R.L.; Pablo, D.; Angeles, J. Comparison of several hydrophilic extracts of Polypodium leucotomos reveals different antioxidant moieties and photoprotective effects in vitro. J. Med. Plant Res. 2018, 12, 336–345. [Google Scholar] [CrossRef]
- Zamarrón, A.; Lorrio, S.; González, S.; Juarranz, Á. Fernblock prevents dermal cell damage induced by visible and infrared A radiation. Int. J. Mol. Sci. 2018, 19, 2250. [Google Scholar] [CrossRef]
- Park, D.W.; Jeon, H.; So, R.; Kang, S.C. Centella asiatica extract prevents visual impairment by promoting the production of rhodopsin in the retina. Nutr. Res. Pract. 2020, 14, 203–217. [Google Scholar] [CrossRef]
- Kwon, K.J.; Bae, S.; Kim, K.; An, I.S.; Ahn, K.J.; An, S.; Cha, H.J. Asiaticoside, a component of Centella asiatica, inhibits melanogenesis in B16F10 mouse melanoma. Mol. Med. Rep. 2014, 10, 503–507. [Google Scholar] [CrossRef]
- Seo, S.B.; Kim, Y.M. Improving cosmetic activity by optimizing Centella asiatica extraction process. Nat. Prod. Commun. 2019, 14, 1–4. [Google Scholar] [CrossRef]
- Shen, X.; Guo, M.; Yu, H.; Liu, D.; Lu, Z.; Lu, Y. Propionibacterium acnes related anti-inflammation and skin hydration activities of madecassoside, a pentacyclic triterpene saponin from Centella asiatica. Biosci. Biotechnol. Biochem. 2019, 83, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Kwon, M.C.; Choi, W.Y.; Seo, Y.C.; Kim, J.S.; Yoon, C.S.; Lim, H.W.; Kim, H.S.; Ahn, J.h.; Lee, H.Y. Enhancement of the skin-protective activities of Centella asiatica L. Urban by a nano-encapsulation process. J. Biotechnol. 2012, 157, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Saranya Babu Jayaprakash, C.; Nagarajan, N. Studies on the bioactive compounds and antimicrobial activities of medicinal plant Centella asiatica (Linn). J. Med. Plants Stud. 2016, 4, 181–185. [Google Scholar]


| Type of UVR | Dose | Cell Type | Expression of Core Clock Gene and Protein after UVR | References |
|---|---|---|---|---|
| NB-UVB (311 nm) | 688 J/cm2 | Epidermal/dermal (Primary cell culture obtained from skin biopsies of skin phototypes II and III after irradiation) | ↓ CRY2 mRNA (24 h after exposure) | [25] |
| BB-UVB (280–320 nm) | 5 mJ/cm2 | HaCaT (Immortalized normal human keratinocyte cell line) | ↓ BMAL1 mRNA (immediate effect from 0 to 8 h); ↑ BMAL1 mRNA (24 h after exposure); Marginal changes in BMAL1 protein after exposure | [26] |
| ↓ CLOCK mRNA (immediate effect from 0 to 8 h); CLOCK mRNA return to basal level (24 h after exposure); Marginal changes in CLOCK protein after exposure | ||||
| HKC (primary human keratinocyte cells) | ↓ BMAL1 mRNA (immediate effect from 0 to 8 h); ↑ BMAL1 (24 h after exposure) | |||
| ↓ CLOCK mRNA (immediate effect from 0 to 8 h); ↑ CLOCK mRNA (24 h after exposure); | ||||
| 10 mJ/cm2 | Normal human keratinocyte(Primary cell culture from neonatal foreskin) | ↓ BMAL1 mRNA (immediate effect from 0 to 20 h) | [27] | |
| ↓ PER1 mRNA (from 0 to 12 h) | ||||
| ↓ CLOCK mRNA (from 0 to 24 h) | ||||
| 12.5 mJ/cm2 | HaCaT | ↓ BMAL1 mRNA and protein (at 24 h) | [28] | |
| 20 mJ/cm2 | NHEK (Normal neonatal human epidermal keratinocytes) | ↓ CLOCK mRNA and protein (Immediate effect after exposure) | [29] | |
| ↓ BMAL1 mRNA and protein (Immediate effect after exposure) |
| Natural Product | Testing Model | Effect on Opsins or Core Clock Expression | Reported Bioproperties Potentially Beneficial in Cosmeceutical Development | References |
|---|---|---|---|---|
| Shikonin (isolated from Lithospermum erythrorhizon and Arnebia euchroma) | NIH3T3-derived stable cells transfected with BMAL1 promoter (NIH3T3 cells: immortalized embryonic fibroblast) | Shortens BMAL1 transcriptional oscillation | Anti-cancer, anti-tumour, anti-inflammation, wound healing, anti-bacterial, anti-fungal | [115,116,117,118,119,120,121] |
| Nobiletin (polymethoxylated flavone from the skin of citrus fruits) | U2OS cells (bone osteosarcoma) MCF7 cells (breast adenocarcinoma) MDA-MB-231 cells (breast adenocarcinoma) | Subtle circadian alterations in U2OS and MCF7 Enhancement of circadian oscillation in MDA-MB-231 (rescue of rhythmicity) | Anti-inflammation, anti-tumour, antioxidant, anti-microbial, anti-pigmentation, anti-collagen degradation | [114,122,123,124,125,126,127] |
| CLOCKΔ19/+-immortalized mice fibroblast cells Hepa1-6 cells (murine hepatoma) U2OS cells | Enhancement of amplitude and lengthen period of the PER2::LucSV reporter rhythm Modification of the expression of other core clock genes and proteins Direct binding to RORα and γ, inducing BMAL1 promoter-driven luciferase reporter expression | |||
| Berberine (Isolated from Coptidis rhizoma) | Bone marrow-derived macrophages from C57BL/6 mice HEK293 cells | Antagonization of Rev-erbα activity ↓ BMAL1 expression | Anti-inflammation, anti-pigmentation, anti-collagen degradation, anti-microbial, anti-cancer | [128,129,130,131,132,133] |
| Puerarin (Isolated from Puerariae radix) | Hepa-1c1c7 cellsHEK293 cells | Antagonization of Rev-erbα activity ↑ BMAL1 expression | Wound healing, anti-inflammation, anti-cancer Promotes melanogenesis * | [134,135,136,137,138] |
| JBIR-26 (derived from Streptomyces sp. AK-AH76) | NIH3T3 cells transfected with PER2 promoter | Lengthening of PER2 transcriptional oscillation | Not available | [139] |
| Agerarin(Isolated from Ageratum houstonianum) | HaCaT cells | ↑ CLOCK mRNA expression | Increases aquaporin 3 expression (skin moisturizing), anti-pigmentation, increases filaggrin involved in maintaining skin barrier | [140,141,142] |
| Lepedeza capitata extract | HaCaT cells | ↑ PER2 activity Lengthening of period of Per2 promoter activity | Wound healing, anti-collagen degradation | [70,143] |
| Trichosanthes kirilowii ethanolic extract | HaCaT cells | ↓ miR-142-3p which inhibits BMAL1 expression | Anti-pigmentation, protection against UVB-induced DNA damage, anti-inflammation, anti-cancer | [28,144,145,146] |
| Fernblock® (hydrophilic extract from Polypodium leucotomos leaves) | B16-F10 cells (mouse melanocyte) | ↓ OPN3 mRNA expression | Anti-pigmentation, Antioxidant, protection against UVR-induced DNA damage, anti-collagen degradation | [147,148,149] |
| Centella asiatica ethanolic extract | ARPE-19 cells (human retinal pigmented epithelium) | ↑ OPN2 protein ↑ Opsin protein | Anti-pigmentation, anti-inflammation, increase skin hydration, anti-collagen degradation, anti-microbial, anti-fungal | [150,151,152,153,154,155] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahendra, C.K.; Ser, H.-L.; Pusparajah, P.; Htar, T.T.; Chuah, L.-H.; Yap, W.H.; Tang, Y.-Q.; Zengin, G.; Tang, S.Y.; Lee, W.L.; et al. Cosmeceutical Therapy: Engaging the Repercussions of UVR Photoaging on the Skin’s Circadian Rhythm. Int. J. Mol. Sci. 2022, 23, 2884. https://doi.org/10.3390/ijms23052884
Mahendra CK, Ser H-L, Pusparajah P, Htar TT, Chuah L-H, Yap WH, Tang Y-Q, Zengin G, Tang SY, Lee WL, et al. Cosmeceutical Therapy: Engaging the Repercussions of UVR Photoaging on the Skin’s Circadian Rhythm. International Journal of Molecular Sciences. 2022; 23(5):2884. https://doi.org/10.3390/ijms23052884
Chicago/Turabian StyleMahendra, Camille Keisha, Hooi-Leng Ser, Priyia Pusparajah, Thet Thet Htar, Lay-Hong Chuah, Wei Hsum Yap, Yin-Quan Tang, Gokhan Zengin, Siah Ying Tang, Wai Leng Lee, and et al. 2022. "Cosmeceutical Therapy: Engaging the Repercussions of UVR Photoaging on the Skin’s Circadian Rhythm" International Journal of Molecular Sciences 23, no. 5: 2884. https://doi.org/10.3390/ijms23052884
APA StyleMahendra, C. K., Ser, H.-L., Pusparajah, P., Htar, T. T., Chuah, L.-H., Yap, W. H., Tang, Y.-Q., Zengin, G., Tang, S. Y., Lee, W. L., Liew, K. B., Ming, L. C., & Goh, B. H. (2022). Cosmeceutical Therapy: Engaging the Repercussions of UVR Photoaging on the Skin’s Circadian Rhythm. International Journal of Molecular Sciences, 23(5), 2884. https://doi.org/10.3390/ijms23052884