Targeted Delivery of Cisplatin by Gold Nanoparticles: The Influence of Nanocarrier Surface Modification Type on the Efficiency of Drug Binding Examined by CE-ICP-MS/MS
Abstract
:1. Introduction
2. Results and Discussion
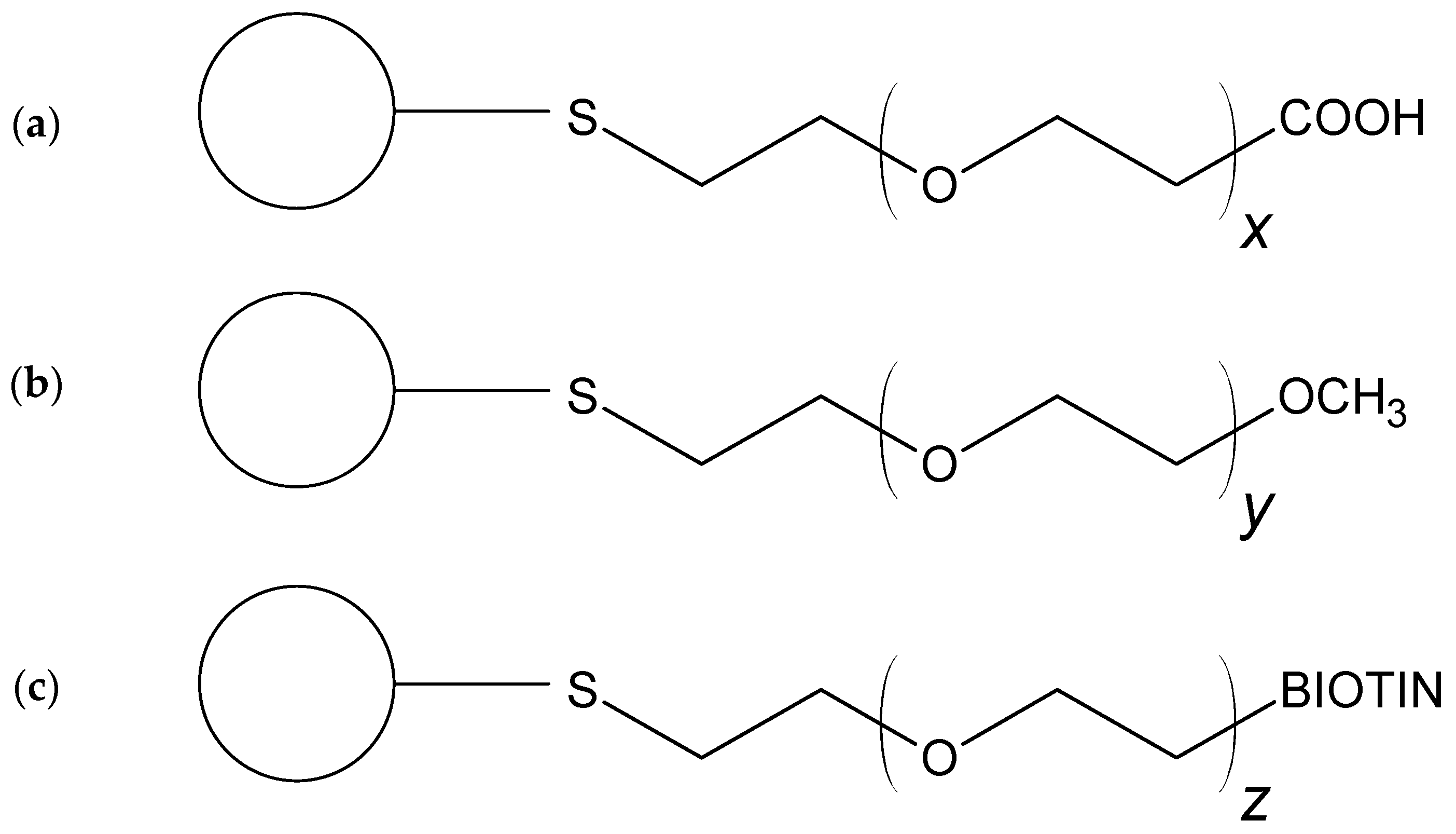
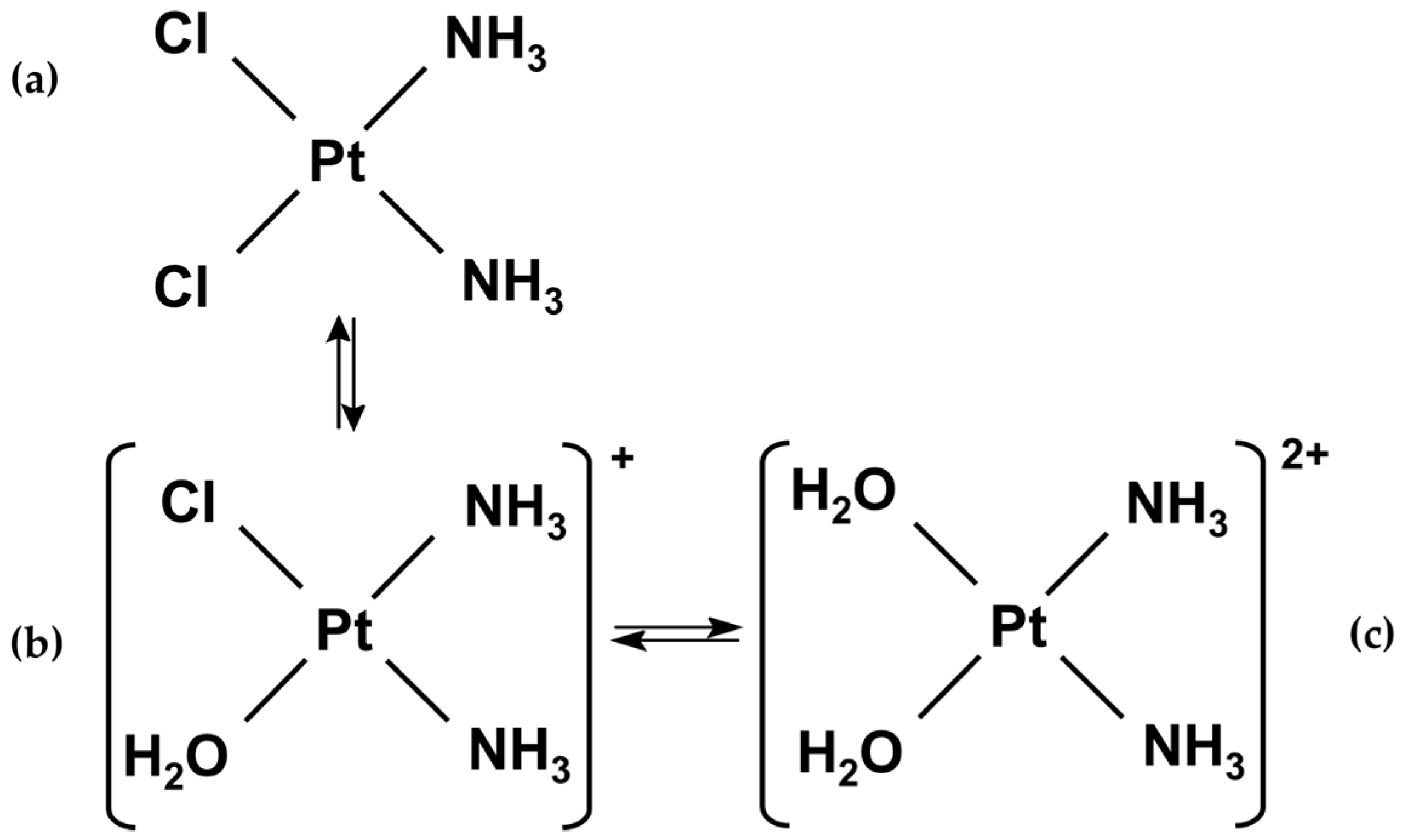
2.1. Gold Nanoparticle–Cisplatin Systems Preparation
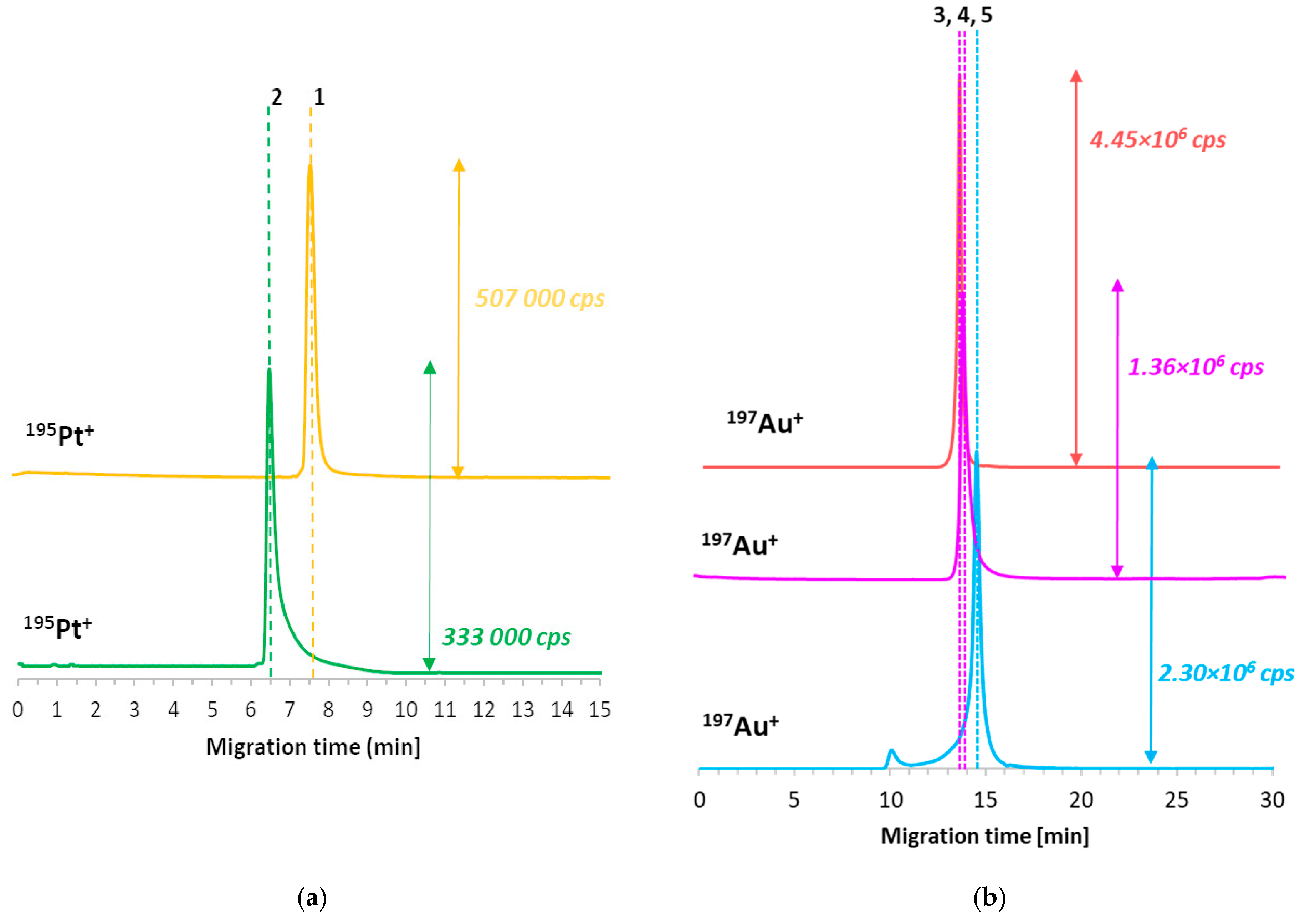
2.2. CE-ICP-MS/MS Method Figures of Merit
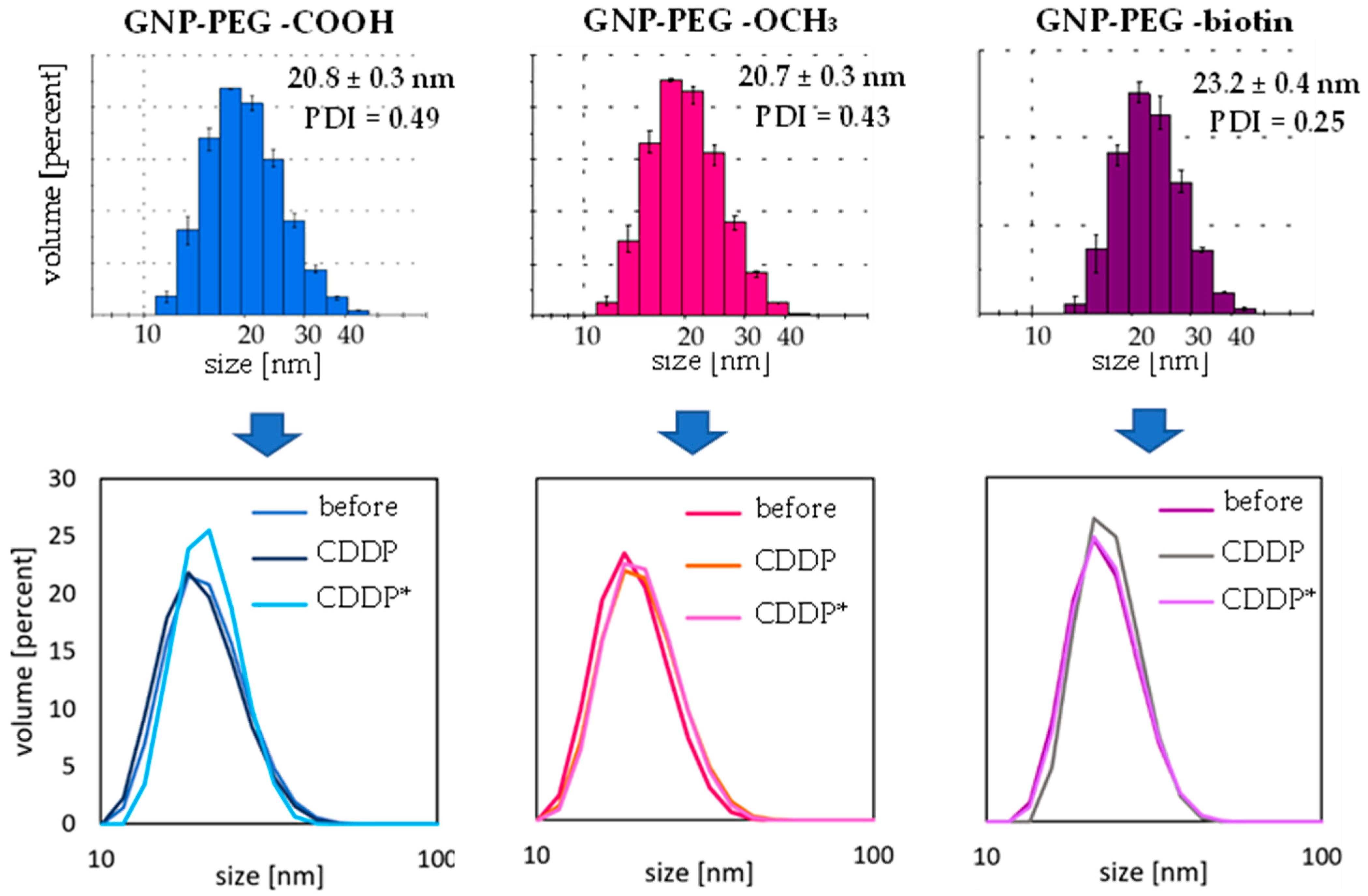
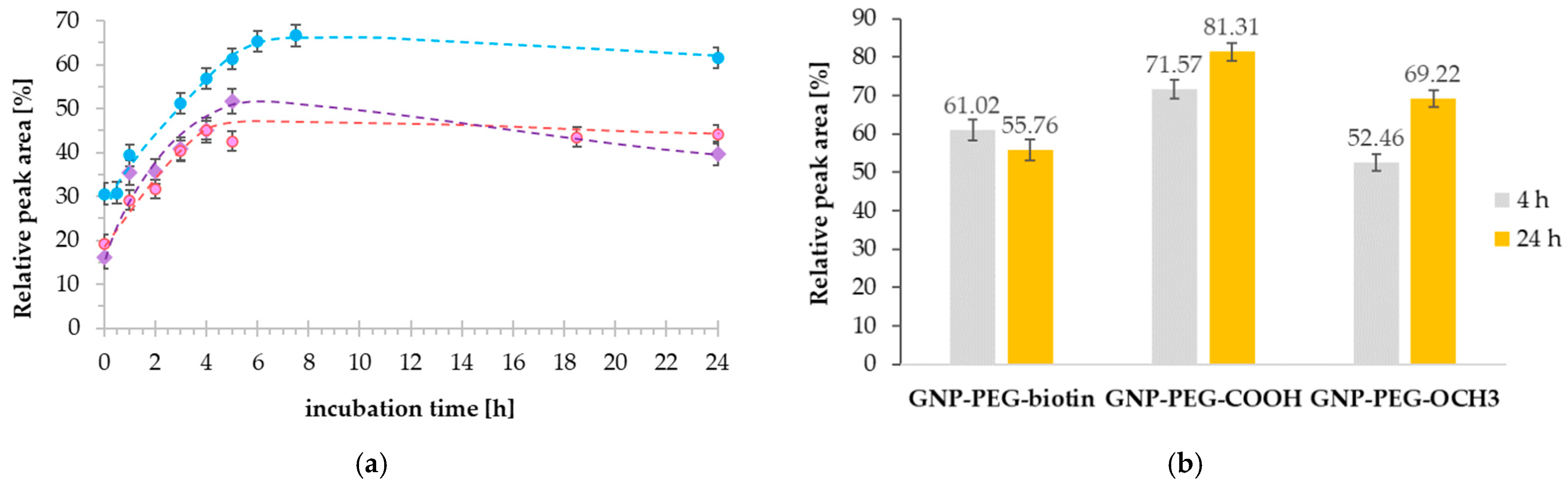
2.3. Investigation of Gold Nanoparticle–Cisplatin Systems Formation, Reaction Kinetics, and Stability
3. Materials and Methods
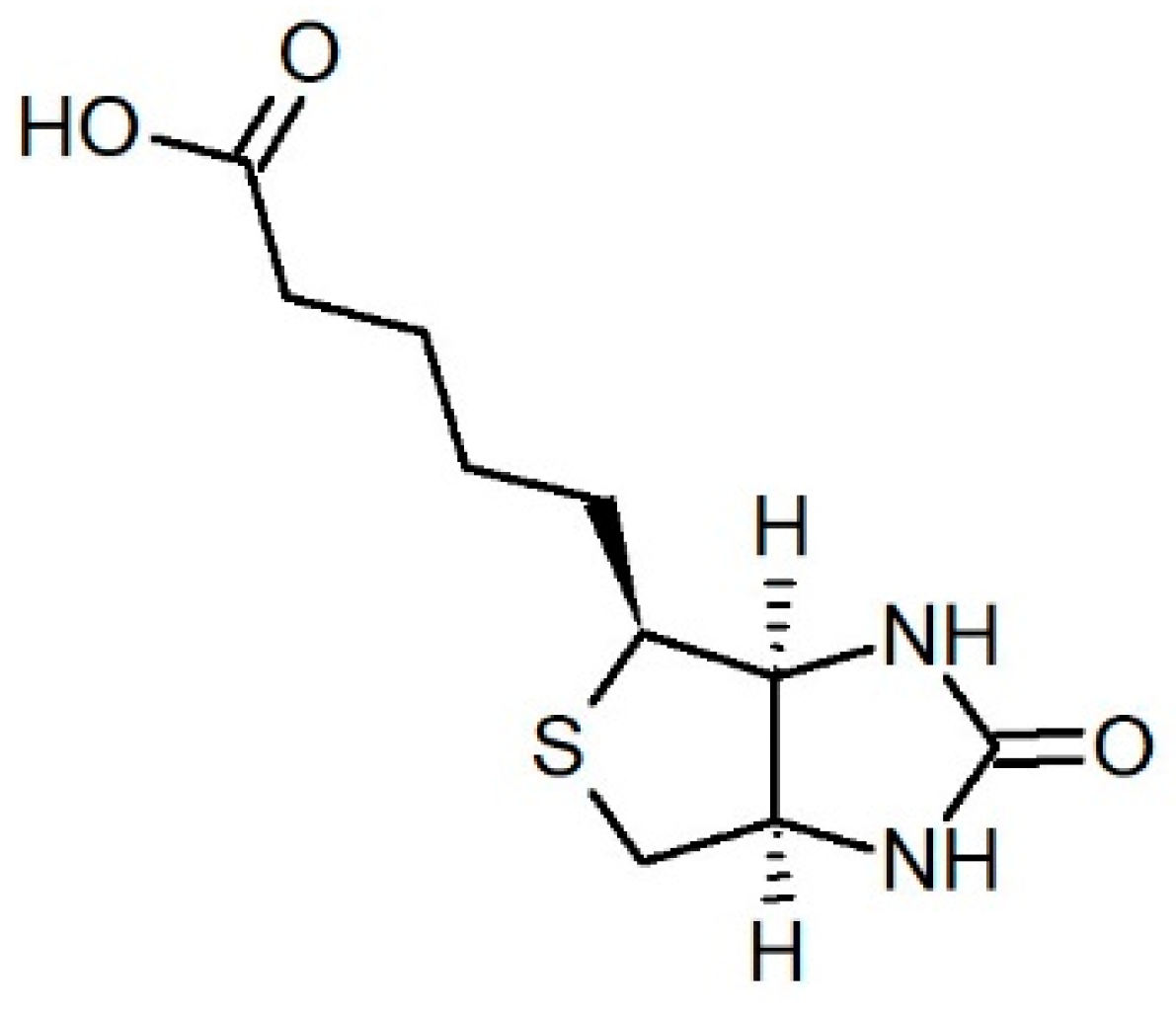
3.1. Chemicals
3.2. Nanocarrier–Drug Systems Preparation
3.3. CE-ICP-MS/MS Instrumentation
3.4. UV-Vis Measurement
3.5. DLS and ζ-Potential Measurement
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cancer. Available online: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed on 1 March 2021).
- Du, Y.; Chen, B. Combination of drugs and carriers in drug delivery technology and its development. Drug Des. Dev. Ther. 2019, 13, 1401–1408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chauffert, B.; Mornex, F.; Bonnetain, F.; Rougier, P.; Mariette, C.; Bouché, O.; Bosset, J.F.; Aparicio, T.; Mineur, L.; Azzedine, A.; et al. Phase III trial comparing intensive induction chemoradiotherapy (60 Gy, infusional 5-FU and intermittent cisplatin) followed by maintenance gemcitabine with gemcitabine alone for locally advanced unresectable pancreatic cancer. Ann. Oncol. 2008, 19, 1592–1599. [Google Scholar] [CrossRef] [PubMed]
- Colucci, G.; Giuliani, F.; Gebbia, V.; Biglietto, M.; Rabitti, P.; Uomo, G.; Cigolari, S.; Testa, A.; Maiello, E.; Lopez, M. Gemcitabine alone or with cisplatin for the treatment of patients with locally advanced and/or metastatic pancreatic carcinoma. Cancer 2002, 94, 902–910. [Google Scholar] [CrossRef] [PubMed]
- Yafout, M.; Ousaid, A.; Khayati, Y.; El Otmani, I.S. Gold nanoparticles as a drug delivery system for standard chemotherapeutics: A new lead for targeted pharmacological cancer treatments. Sci. Afr. 2021, 11, e00685. [Google Scholar] [CrossRef]
- Zajda, J.; Wróblewska, A.; Ruzik, L.; Matczuk, M. Methodology for characterization of platinum-based drug’ s targeted delivery nanosystems. J. Control. Release 2021, 335, 178–190. [Google Scholar] [CrossRef] [PubMed]
- Daraee, H.; Eatemadi, A.; Abbasi, E.; Aval, S.F.; Kouhi, M.; Akbarzadeh, A. Application of gold nanoparticles in biomedical and drug delivery. Artif. Cells Nanomed. Biotechnol. 2016, 44, 410–422. [Google Scholar] [CrossRef]
- Bai, X.; Wang, Y.; Song, Z.; Feng, Y.; Chen, Y.; Zhang, D.; Feng, L. The basic properties of gold nanoparticles and their applications in tumor diagnosis and treatment. Int. J. Mol. Sci. 2020, 21, 2480. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Guo, Z. Targeting and delivery of platinum-based anticancer drugs. Chem. Soc. Rev. 2013, 42, 202–224. [Google Scholar] [CrossRef]
- Bhamidipati, M.; Fabris, L. Multiparametric assessment of gold nanoparticle cytotoxicity in cancerous and healthy cells: The role of size, shape, and surface chemistry. Bioconjug. Chem. 2017, 28, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Nicol, J.R.; Dixon, D.; Coulter, J.A. Gold nanoparticle surface functionalization: A necessary requirement in the development of novel nanotherapeutics. Nanomedicine 2015, 10, 1315–1326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, C.Y.-S.; Fung, S.-K.; Tong, K.-C.; Wan, P.-K.; Lok, C.-N.; Huang, Y.; Chen, T.; Che, C.-M. A multi-functional PEGylated gold(III) compound: Potent anticancer properties and self-assembly into nanostructures for drug co-delivery. Chem. Sci. 2017, 8, 1942–1953. [Google Scholar] [CrossRef] [Green Version]
- Min, Y.; Mao, C.; Xu, D.; Wang, J.; Liu, Y. Gold nanorods for platinum based prodrug delivery. Chem. Commun. 2010, 46, 8424–8426. [Google Scholar] [CrossRef] [PubMed]
- Poon, W.; Zhang, Y.-N.; Ouyang, B.; Kingston, B.R.; Wu, J.L.Y.; Wilhelm, S.; Chan, W.C.W. Elimination pathways of nanoparticles. ACS Nano 2019, 13, 5785–5798. [Google Scholar] [CrossRef] [PubMed]
- Kadkhoda, J.; Tarighatnia, A.; Barar, J.; Aghanejad, A.; Davaran, S. Recent advances and trends in nanoparticles based photothermal and photodynamic therapy. Photodiagnosis Photodyn. Ther. 2022, 37, 102697. [Google Scholar] [CrossRef] [PubMed]
- Comenge, J.; Sotelo, C.; Romero, F.; Gallego, O.; Barnadas, A.; Parada, T.G.-C.; Domínguez, F.; Puntes, V.F. Detoxifying antitumoral drugs via nanoconjugation: The case of gold nanoparticles and cisplatin. PLoS ONE 2012, 7, e47562. [Google Scholar] [CrossRef] [Green Version]
- Wróblewska, A.; Matczuk, M. First application of CE-ICP-MS for monitoring the formation of cisplatin targeting delivery systems with gold nanocarriers. Electrophoresis 2020, 41, 394–398. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, R.; Patra, C.R.; Earl, A.; Wang, S.; Katarya, A.; Lu, L.; Kizhakkedathu, J.N.; Yaszemski, M.J.; Greipp, P.R.; Mukhopadhyay, D.; et al. Attaching folic acid on gold nanoparticles using noncovalent interaction via different polyethylene glycol backbones and targeting of cancer cells. Nanomed. Nanotechnol. Biol. Med. 2007, 3, 224–238. [Google Scholar] [CrossRef]
- Tan, J.; Cho, T.J.; Tsai, D.-H.; Liu, J.; Pettibone, J.M.; You, R.; Hackley, V.A.; Zachariah, M.R. Surface modification of cisplatin-complexed gold nanoparticles and its influence on colloidal stability, drug loading, and drug release. Langmuir 2018, 34, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Wong, P.T.; Choi, S.K. Mechanisms of drug release in nanotherapeutic delivery systems. Chem. Rev. 2015, 115, 3388–3432. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-D.; Huang, L. Stealth nanoparticles: High density but sheddable PEG is a key for tumor targeting. J. Control. Release 2010, 145, 178–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahme, K.; Chen, L.; Hobbs, R.G.; Morris, M.A.; O’Driscoll, C.; Holmes, J.D. PEGylated gold nanoparticles: Polymer quantification as a function of PEG lengths and nanoparticle dimensions. RSC Adv. 2013, 3, 6085–6094. [Google Scholar] [CrossRef] [Green Version]
- Reznickova, A.; Slavikova, N.; Kolska, Z.; Kolarova, K.; Belinova, T.; Hubalek Kalbacova, M.; Cieslar, M.; Svorcik, V. PEGylated gold nanoparticles: Stability, cytotoxicity and antibacterial activity. Colloids Surf. A 2019, 560, 26–34. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, H.; Kuzmenko, I.; Mallapragada, S.; Vaknin, D. Assembling bare Au nanoparticles at positively charged templates. Sci. Rep. 2016, 6, 26462. [Google Scholar] [CrossRef] [PubMed]
- Amendola, V.; Meneghetti, M. Size evaluation of gold nanoparticles by UV-vis spectroscopy. J. Phys. Chem. C 2009, 113, 4277–4285. [Google Scholar] [CrossRef]
- Haiss, W.; Thanh, N.T.K.; Aveyard, J.; Fernig, D.G. Determination of size and concentration of gold nanoparticles from UV-Vis spectra. Anal. Chem. 2007, 79, 4215–4221. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Miao, L.; Wang, Y.; Huang, L. Unmodified drug used as a material to construct nanoparticles: Delivery of cisplatin for enhanced anticancer therapy. J. Control. Release 2014, 174, 137–142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- May, T.W.; Wiedmeyer, R.H. A table of polyatomic interferences in ICP-MS. At. Spectrosc. 1998, 19, 150–155. [Google Scholar]
- Singh, P.; Pandit, S.; Mokkapati, V.R.S.S.; Garg, A.; Ravikumar, V.; Mijakovic, I. Gold nanoparticles in diagnostics and therapeutics for human cancer. Int. J. Mol. Sci. 2018, 19, 1979. [Google Scholar] [CrossRef]
- Gong, Z.; Yang, R.; Sun, C.-Q.; Han, W.-N.; Jiang, X.-H.; Hu, S.-X.; Wang, Y. Simultaneous determination of P and S in human serum , blood plasma and whole blood by ICP-MS with collision/reaction cell technology. Int. J. Mass Spectrom. 2019, 445, 116193. [Google Scholar] [CrossRef]
- Persson, D.P.; Hansen, T.H.; Laursen, K.H.; Schjoerring, J.K.; Husted, S. Simultaneous iron, zinc, sulfur and phosphorus speciation analysis of barley grain tissues using SEC-ICP-MS and IP-ICP-MS. Metallomics 2009, 1, 418–426. [Google Scholar] [CrossRef]
- Balcaen, L.; Bolea-Fernandez, E.; Resano, M.; Vanhaecke, F. Inductively coupled plasma—Tandem mass spectrometry (ICP-MS/MS): A powerful and universal tool for the interference-free determination of (ultra)trace elements—A tutorial review. Anal. Chim. Acta 2015, 894, 7–19. [Google Scholar] [CrossRef]
- Machado, R.C.; Amaral, C.D.B.; Schiavo, D.; Nóbrega, J.A.; Nogueira, A.R.A. Complex samples and spectral interferences in ICP-MS: Evaluation of tandem mass spectrometry for interference-free determination of cadmium, tin and platinum group elements. Microchem. J. 2017, 130, 271–275. [Google Scholar] [CrossRef]


| CE System | |
| Capillary | Fused silica capillary, i.d. 75 µm, o.d. 375 µm, 70 cm length |
| BGE * | Phosphate buffer, 10 mM, pH 7.4 |
| Temperature | 37 °C |
| Voltage | +17 kV |
| Current | 20–40 µA |
| Sample injection | Hydrodynamic, 30 mbar, 5 s |
| ICP-MS/MS System | |
| RF power | 1570 W |
| Sample depth | 8.2 mm |
| Plasma gas | 15.0 L min−1 |
| Nebulizer gas flow | 1.10 L min−1 |
| Sheath liquid | 10 ng mL−1 72Ge in 1 mM PB |
| Sheath liquid flow | 10 µL min−1 |
| Cell gas (O2) flow | 0.51 mL min−1 |
| Monitored mases | 72Ge+, 197Au+, 195Pt+, 32S16O+ |
| Analyte | RSD (%) (n = 3) | LOD (mol L−1) | LOQ (mol L−1) | |||
|---|---|---|---|---|---|---|
| Migration Time | Peak Area | 195Pt | 197Au | 195Pt | 197Au | |
| CDDP | 5.1 | 7.0 | 2.16 × 10−8 | – | 6.47 × 10−8 | – |
| CDDP* | 2.7 | 7.5 | 3.87 × 10−8 | – | 1.16 × 10−8 | – |
| GNP-PEG-biotin | 0.8 | 2.8 | – | 2.29 × 10−11 | – | 6.88 × 10−11 |
| GNP-PEG-OCH3 | 8.9 | 4.7 | – | 8.59 × 10−12 | – | 22.58 × 10−11 |
| GNP-PEG-COOH | 0.7 | 8.7 | – | 7.17 × 10−12 | – | 2.15 × 10−11 |
| GNPs Functionalization Type (Terminal Moiety); Reagents Ratio ** | The Estimated Density of PEG-Moieties on the GNPs Surface | The Estimated Number of PEG-Moieties on the Surface of Each GNP [-] | The Highest CDDP* Concentration in GNCSs [µM] | Efficiency of Moieties Saturation with CDDP* Molecules [%] |
|---|---|---|---|---|
| Methoxyl-; 1:400 | 4 per 1 nm2 | ~1256 | 1.77 | 4.55 |
| Carboxyl-; 1:800 | 1 per 1 nm2 | ~314 | 2.12 | 21.70 |
| Biotin; 1:400 | 0.5 per 1 nm2 | ~157 | 1.91 | 39.23 |
| GNPs Functionalization (Terminal Moiety); Reagents Ratio ** | before [mV] | CDDP [mV] | CDDP* [mV] |
|---|---|---|---|
| Methoxyl-; 1:400 | −24.6 ±1.0 | −24.1 ±1.0 | −24.3 ±1.3 |
| Carboxyl-; 1:800 | −21.4 ±1.0 | −23.1 ±1.8 | −27.1 ±0.8 |
| Biotin; 1:400 | −19.9 ±1.2 | −19.4 ±0.7 | −22.7 ±0.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wróblewska, A.M.; Milewska, A.; Drozd, M.; Matczuk, M. Targeted Delivery of Cisplatin by Gold Nanoparticles: The Influence of Nanocarrier Surface Modification Type on the Efficiency of Drug Binding Examined by CE-ICP-MS/MS. Int. J. Mol. Sci. 2022, 23, 2324. https://doi.org/10.3390/ijms23042324
Wróblewska AM, Milewska A, Drozd M, Matczuk M. Targeted Delivery of Cisplatin by Gold Nanoparticles: The Influence of Nanocarrier Surface Modification Type on the Efficiency of Drug Binding Examined by CE-ICP-MS/MS. International Journal of Molecular Sciences. 2022; 23(4):2324. https://doi.org/10.3390/ijms23042324
Chicago/Turabian StyleWróblewska, Anna M., Aleksandra Milewska, Marcin Drozd, and Magdalena Matczuk. 2022. "Targeted Delivery of Cisplatin by Gold Nanoparticles: The Influence of Nanocarrier Surface Modification Type on the Efficiency of Drug Binding Examined by CE-ICP-MS/MS" International Journal of Molecular Sciences 23, no. 4: 2324. https://doi.org/10.3390/ijms23042324
APA StyleWróblewska, A. M., Milewska, A., Drozd, M., & Matczuk, M. (2022). Targeted Delivery of Cisplatin by Gold Nanoparticles: The Influence of Nanocarrier Surface Modification Type on the Efficiency of Drug Binding Examined by CE-ICP-MS/MS. International Journal of Molecular Sciences, 23(4), 2324. https://doi.org/10.3390/ijms23042324






