Lycopene in the Prevention of Cardiovascular Diseases
Abstract
1. Introduction
2. Mechanisms of Lycopene Action in the Cardiovascular System: Antioxidant Effect and Anti-Inflammatory Effect
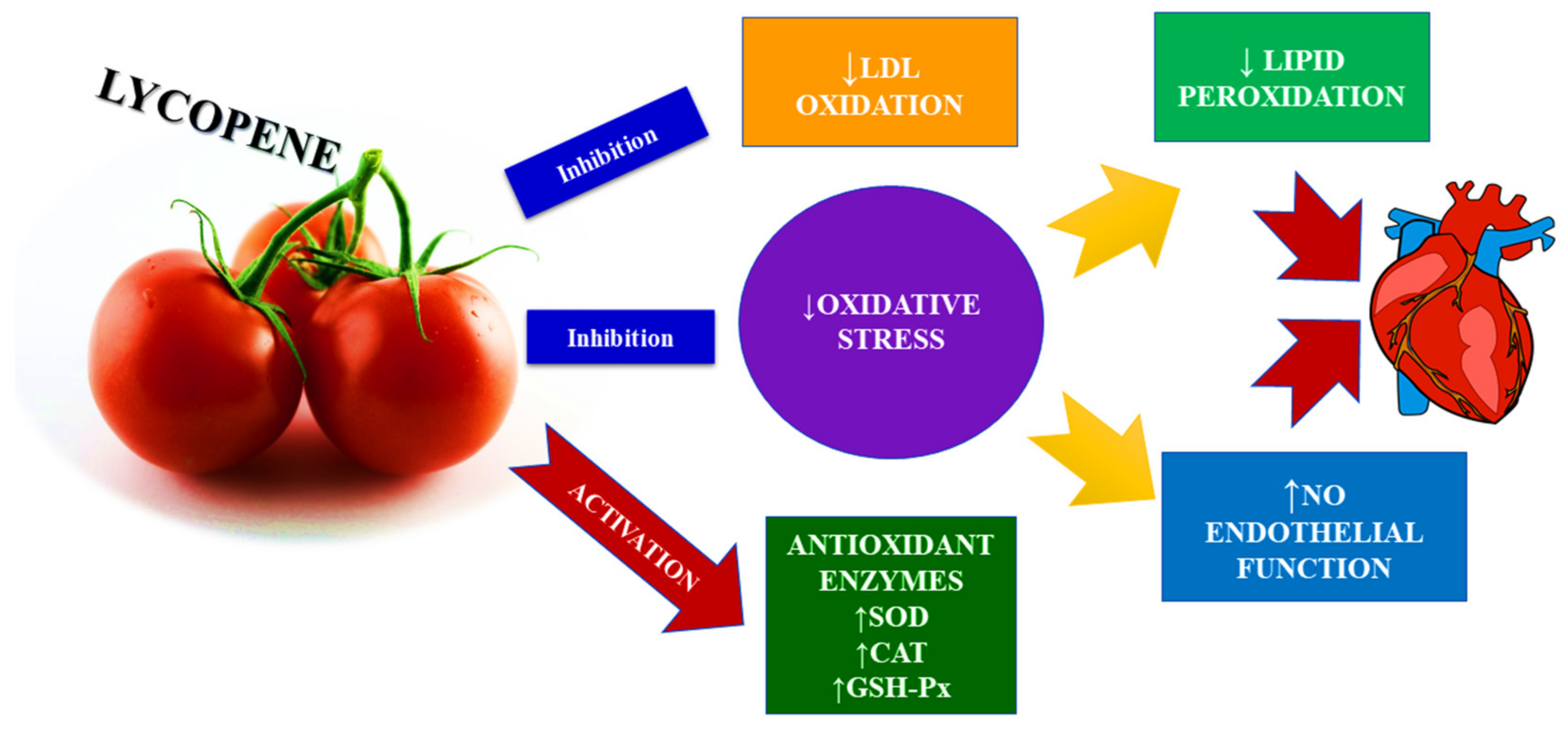
2.1. Antioxidant Effects of Lycopene
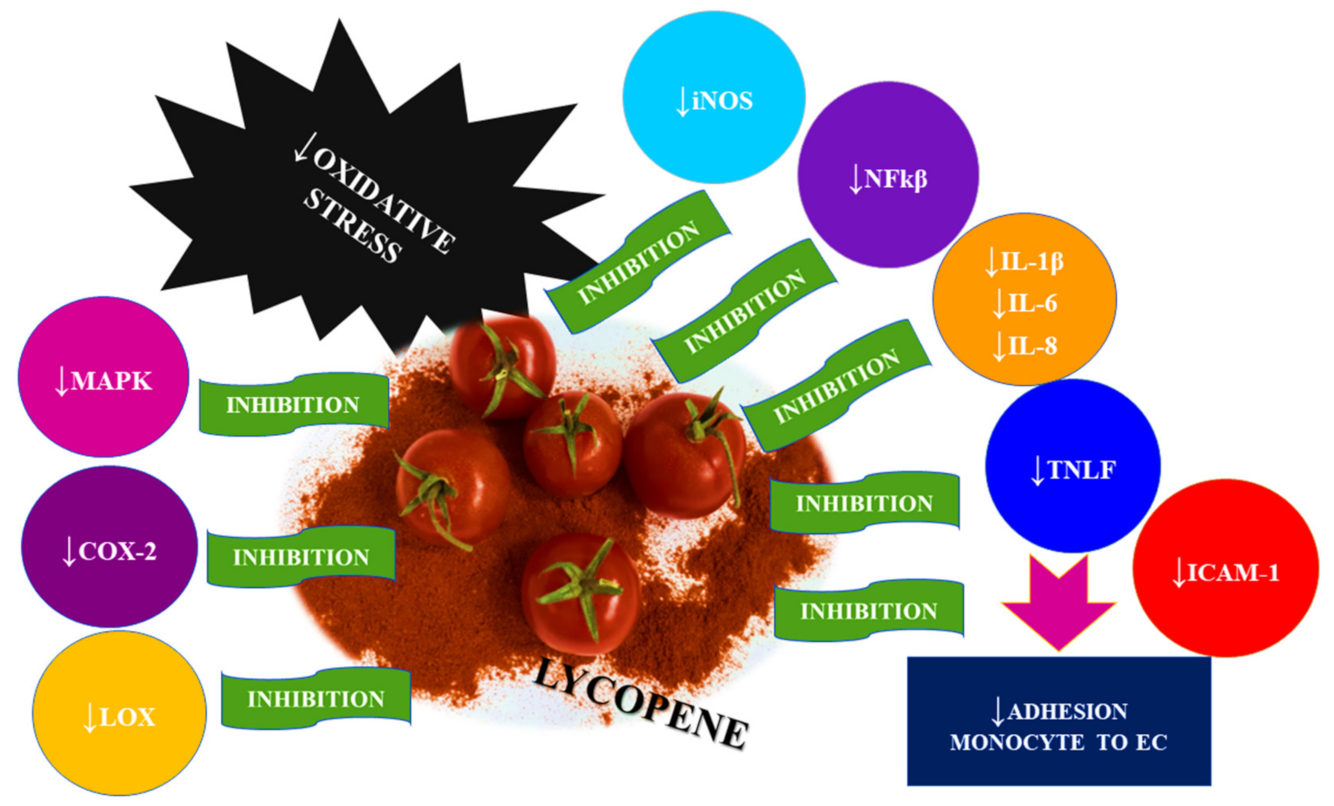
2.2. Anti-Inflammatory Mechanism of Lycopene
3. Efficacy of Lycopene in Inhibiting the Risk of Cardiovascular Diseases
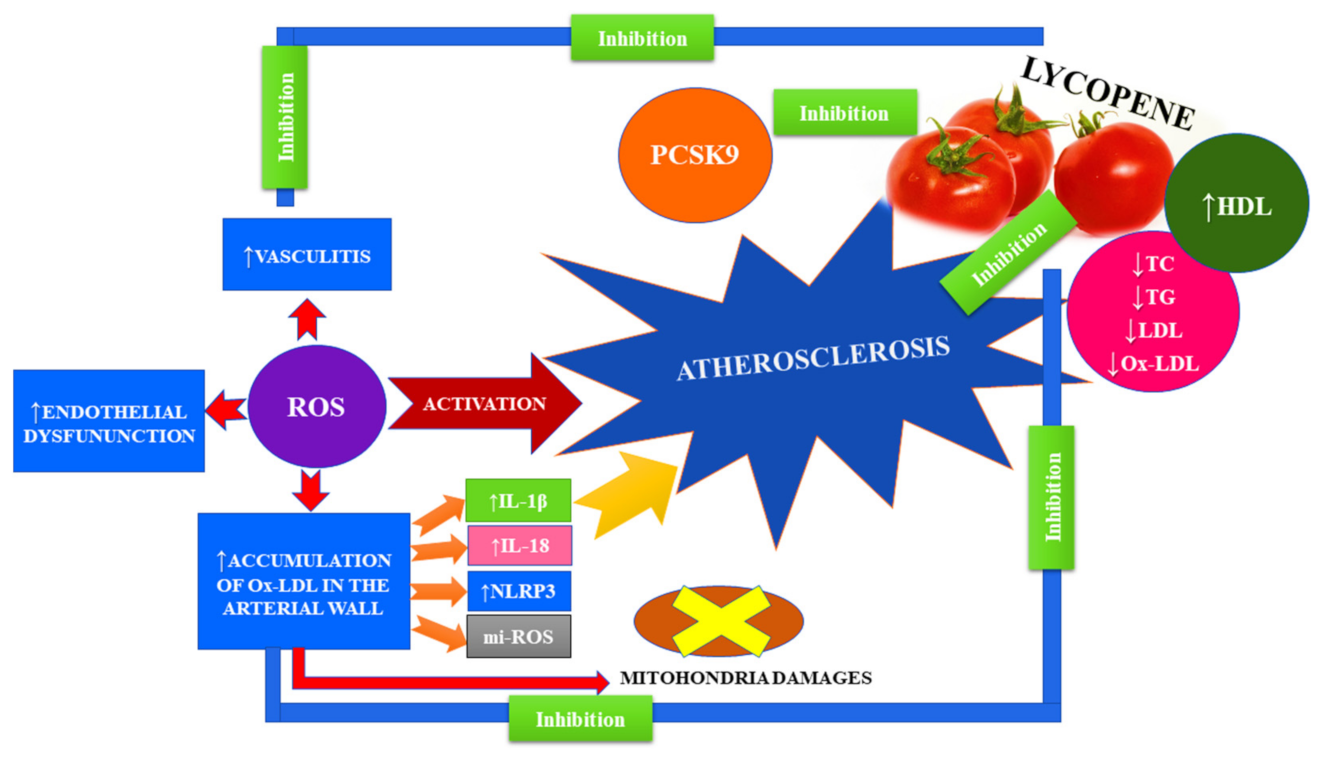
3.1. Atherosclerosis and the Action of Lycopene
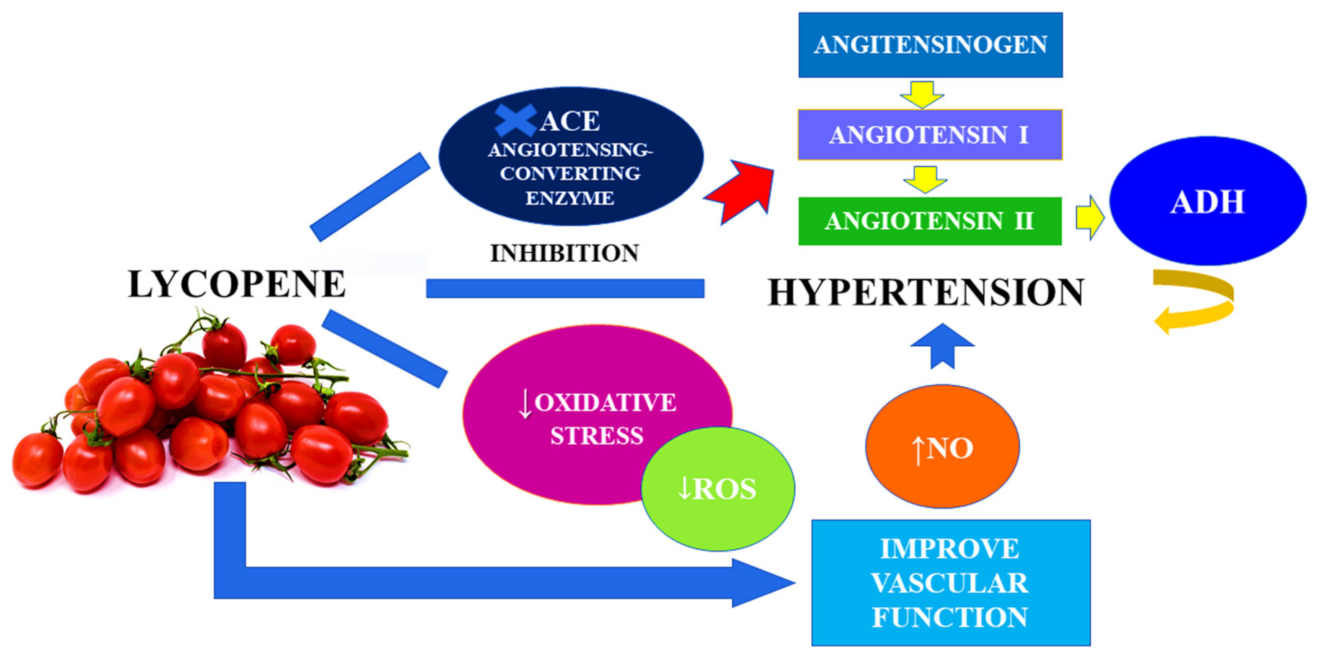
3.2. Hypertension and the Effect of Lycopene
4. Lycopene Supplementation and the Risk of Cardiovascular Diseases
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Roth, G.A.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef]
- Amini, M.; Zayeri, F.; Salehi, M. Trend analysis of cardiovascular disease mortality, incidence, and mortality-to-incidence ratio: Results from global burden of disease study 2017. BMC Public Health 2021, 21, 401. [Google Scholar] [CrossRef]
- Zhang, J.; Xu, J.H.; Qu, Q.Q.; Zhong, G.Q. Risk of Cardiovascular and Cerebrovascular Events in Polycystic Ovarian Syndrome Women: A Meta-Analysis of Cohort Studies. Front. Cardiovasc. Med. 2020, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Cardiovascular Diseases (CVDs)—World Health Organization. Definition of Cardiovascular Diseases. Available online: https://www.euro.who.int/en/health-topics/noncommunicable-diseases/cardiovascular-diseases/cardiovascular-diseases2/definition-of-cardiovascular-diseases (accessed on 19 September 2021).
- Pham, L.M.; Kim, E.C.; Ou, W.; Phung, C.D.; Nguyen, T.T.; Pham, T.T.; Poudel, K.; Gautam, M.; Nguyen, H.T.; Jeong, J.H.; et al. Targeting and clearance of senescent foamy macrophages and senescent endothelial cells by antibody-functionalized mesoporous silica nanoparticles for alleviating aorta atherosclerosis. Biomaterials 2021, 269, 120677. [Google Scholar] [CrossRef]
- Steven, S.; Frenis, K.; Oelze, M.; Kalinovic, S.; Kuntic, M.; Jimenez, M.T.B.; Vujacic-Mirski, K.; Helmstädter, J.; Kröller-Schön, S.; Münzel, T.; et al. Vascular Inflammation and Oxidative Stress: Major Triggers for Cardiovascular Disease. Oxidative Med. Cell. Longev. 2019, 2019, 1–26. [Google Scholar] [CrossRef]
- Arauna, D.; Furrianca, M.; Espinosa-Parrilla, Y.; Fuentes, E.; Alarcón, M.; Palomo, I. Natural Bioactive Compounds as Protectors of Mitochondrial Dysfunction in Cardiovascular Diseases and Aging. Molecules 2019, 24, 4259. [Google Scholar] [CrossRef] [PubMed]
- Kura, B.; Parikh, M.; Slezak, J.; Pierce, G.N. The Influence of Diet on MicroRNAs that Impact Cardiovascular Disease. Molecules 2019, 24, 1509. [Google Scholar] [CrossRef] [PubMed]
- Salas-Salvadó, J.; Becerra-Tomás, N.; García-Gavilán, J.F.; Bulló, M.; Barrubés, L. Mediterranean Diet and Cardiovascular Disease Prevention: What Do We Know? Prog. Cardiovasc. Dis. 2018, 61, 62–67. [Google Scholar] [CrossRef]
- Martínez-González, M.A.; Gea, A.; Ruiz-Canela, M. The Mediterranean Diet and Cardiovascular Health. Circ. Res. 2019, 124, 779–798. [Google Scholar] [CrossRef]
- Violeta Nour, V.; Panaiteb, T.D.; Ropotab, M.; Turcub, R.; Trandafirc, I.; Corbua, A.R. Nutritional and bioactive compounds in dried tomato processing waste. CyTA J. Food. 2018, 16, 222–229. [Google Scholar] [CrossRef]
- Wolak, T.; Sharoni, Y.; Levy, J.; Linnewiel-Hermoni, K.; Stepensky, D.; Paran, E. Effect of Tomato Nutrient Complex on Blood Pressure: A Double Blind, Randomized Dose-Response Study. Nutrients 2019, 11, 950. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.M.; Koutsidis, G.; Lodge, J.K.; Ashorb, A.W.; Siervo, M.; Lara, J. Lycopene and tomato and risk of cardiovascular diseases: A systematic review and meta-analysis of epidemiological evidence. Crit. Rev. Food Sci. Nutr. 2019, 59, 141–158. [Google Scholar] [CrossRef] [PubMed]
- Przybylska, S. Lycopene—A bioactive carotenoid offering multiple health benefits: A review. Int. J. Food Sci. Technol. 2020, 55, 11–32. [Google Scholar] [CrossRef]
- Carvalho, G.C.; Sábio, R.M.; Chorilli, M. An overview of properties and analytical methods for lycopene in organic nanocarriers. Crit. Rev. Anal. Chem. 2020, 51, 1–13. [Google Scholar] [CrossRef]
- Colle, I.; Lemmens, L.; Van Buggenhout, S. Effect of thermal processing on the degradation, isomerization and bioaccessibility of lycopene in tomato pulp. J. Food Sci. 2010, 75, 735–759. [Google Scholar] [CrossRef]
- USDA National Nutrient Database for Standard Reference Legacy Release. FoodData Central. Available online: https://ndb.nal.usda.gov (accessed on 12 April 2019).
- Górecka, D.; Wawrzyniak, A.; Jędrusek-Golińska, A.; Dziedzic, K.; Hamułka, J.; Kowalczewski, P.Ł.; Walkowiak, J. Lycopene in tomatoes and tomato products. Open Chem. 2020, 18, 752–756. [Google Scholar] [CrossRef]
- Maiani, G.; Periago Castón, M.J.; Catasta, G.; Toti, E.; Cambrodón, I.G.; Bysted, A.; Granado-Lorencio, F.; Olmedilla-Alonso, B.; Knuthsen, P.; Valoti, M. Carotenoids: Actual knowledge on food sources, intakes, stability and bioavailability and their protective role in humans. Mol. Nutr. Food Res. 2009, 53, S194–S218. [Google Scholar] [CrossRef]
- Burton-Freeman, B.; Sesso, H.D. Whole food versus supplement: Comparing the clinical evidence of tomato intake and lycopene supplementation on cardiovascular risk factors. Adv. Nutr. 2014, 5, 457–485. [Google Scholar] [CrossRef]
- Petyaev, I.M. Lycopene Deficiency in Ageing and Cardiovascular Disease. Oxidat. Med. Cell. Longev. 2016, 3, 1–6. [Google Scholar] [CrossRef]
- Story, E.N.; Kopec, R.E.; Schwartz, S.J.; Harris, G.K. An uptade on the health effects of tomato lycopene. Annu. Rev. Food Sci. Technol. 2010, 1, 189–210. [Google Scholar] [CrossRef]
- Puah, B.P.; Jalil, J.; Attiq, A.; Kamisah, Y. New insights into molecular mechanism behind anti-cancer activities of lycopene. Molecules 2021, 26, 3888. [Google Scholar] [CrossRef] [PubMed]
- Thies, F.; Mills, L.M.; Moir, S.; Masson, L.F. Cardiovascular benefits of lycopene: Fantasy or reality? Proc. Nutr. Soc. 2017, 76, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Lammi, C.; Arnoldi, A. Food-derived antioxidants and COVID-19. J. Food Biochem. 2021, 45, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Galanakis, C.M. The food systems in the era of the coronavirus (COVID-19) pandemic crisis. Foods 2020, 9, 523. [Google Scholar] [CrossRef]
- Galanakis, C.M. Functionality of food components and emerging technologies. Foods 2021, 10, 128. [Google Scholar] [CrossRef]
- Kong, K.W.; Khoo, H.E.; Prasad, K.N.; Ismall, A.; Tan, C.P.; Rajab, N.F. Revealing the power of the natural red pigment lycopene. Molecules 2010, 15, 959–987. [Google Scholar] [CrossRef]
- Wang, X.D. Lycopene metabolism and its biological significance. Am. J. Clin. Nutr. 2010, 96, 1214–1222. [Google Scholar] [CrossRef]
- Honda, M.; Maeda, H.; Fukaya, T.; Goto, M. Effects of Z-isomerization on the bioavailability and functionality of carotenoids: A review. In Progress in Carotenoid Research; Zempka, E.Q., Lopes, E.J., de Rosso, V.V., Eds.; Intech Open: London, UK, 2018; pp. 139–159. [Google Scholar]
- Saini, R.K.A.; Bekhit, A.E.D.; Roohinejad, S.; Rengasamy, K.R.R.; Keum, Y.S. Chemical stability of lycopene in processed products: A review of the effects of processing methods and modern preservation strategies. J. Agric. Food Chem. 2020, 68, 712–726. [Google Scholar] [CrossRef]
- Carvalho, G.C.; Camargo, B.A.F.; de Thayanne, J.; de Araújo, J.T.C.; Chorilli, M. Lycopene: From tomato to its nutraceutical use and its association with nanotechnology. Trends Food Sci. Technol. 2021, 118, 447–458. [Google Scholar] [CrossRef]
- Dubois-Deruy, E.; Victoriane Peugnet, V.; Turkieh, A.; Pinet, F. Oxidative Stress in Cardiovascular Diseases. Antioxidants 2020, 9, 864. [Google Scholar] [CrossRef]
- Senoner, T.; Dichtl, W. Oxidative Stress in Cardiovascular Diseases: Still a Therapeutic Target? Nutrients 2019, 11, 2090. [Google Scholar] [CrossRef]
- Maoka, T. Carotenoids as natural functional pigments. J. Nat. Med. 2020, 74, 1–16. [Google Scholar] [CrossRef] [PubMed]
- ChemSpider Search and Share Chemistry. Search ChemSpider. Available online: http://www.chemspider.com/ (accessed on 25 November 2021).
- Arballo, J.; Amengual, J.; Erdman, J.W., Jr. Lycopene: A Critical Review of Digestion, Absorption, Metabolism, and Excretion. Antioxidants 2021, 10, 342. [Google Scholar] [CrossRef] [PubMed]
- Caseiro, M.; Ascenso, A.; Costa, A.; Creagh-Flynn, J.; Melissa Johnson, M.; Simões, S. Lycopene in human health. LWT Food Sci. Technol. 2020, 127, 109323. [Google Scholar] [CrossRef]
- Young, A.J.; Lowe, G.M. Antioxidant and prooxidant properties of carotenoids. Arch. Biochem. Biophys. 2001, 385, 20–27. [Google Scholar] [CrossRef]
- Joshi, B.; Kar, S.K.; Yadav, P.K.; Yadav, S.; Shrestha, L.; Bera, T.K. Therapeutic and medicinal uses of lycopene: A systematic review. Int. J. Res. Med Sci. 2020, 8, 1195–1201. [Google Scholar] [CrossRef]
- Krinsky, N.I.; Johnson, E.J. Carotenoid actions and their relation to health and disease. Mol. Asp. Med. 2005, 26, 459–516. [Google Scholar] [CrossRef] [PubMed]
- Khan, U.M.; Sevindik, M.; Zarrabi, A.; Nami, M.; Ozdemir, B.; Kaplan, D.N.; Selamoglu, Z.; Hasan, M.; Kumar, M.; Alshehri, M.M.; et al. Lycopene: Food Sources, Biological Activities, and Human Health Benefits. Oxid. Med. Cell. Longev. 2021, 2021, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bin-Jumah, M.N.; Nadeem, M.S.; Gilani, S.J.; Mubeen, B.; Ullah, I.; Alzarea, S.I.; Ghoneim, M.; Alshehri, S.; Al-Abbasi, F.A.; Kazmi, I. Review Lycopene: A Natural Arsenal in the War against Oxidative Stress and Cardiovascular Diseases. Antioxidants 2022, 11, 232. [Google Scholar] [CrossRef]
- Zeng, J.; Zhao, J.; Dong, B.; Cai, X.; Jiang, J.; Xue, R.; Yao, F.; Dong, Y.; Liu, C. Lycopene protects against pressure overload-induced cardiac hypertrophy by attenuating oxidative stress. J. Nutr. Biochem. 2019, 66, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Hu, H.; Chen, B.; Yue, R.; Zhou, Z.; Liu, Y.; Zhang, S.; Xu, L.; Wang, H.; Yu, Z. Lycopene Protects against Hypoxia/Reoxygenation Injury by Alleviating ER Stress Induced Apoptosis in Neonatal Mouse Cardiomyocytes. PLoS ONE 2012, 10, 1–16. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Zhou, W.; Xiong, C.; Tan, G.; Chen, M. Lycopene attenuates inflammation and apoptosis in post-myocardial infarction remodeling by inhibiting the nuclear factor kappa-κB signaling pathway. Mol. Med. Rep. 2015, 11, 374–378. [Google Scholar] [CrossRef] [PubMed]
- Mozos, I.; Stoian, D.; Caraba, A.; Malainer, C.; Horbanczuk, J.O.; Atanasov, A.G. Lycopene and Vascular Health. Front. Pharmacol. 2018, 9, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Omidyan, R.; Abedini, F.; Shahrokh, L.; Azimi, G. Excited State Deactivation Mechanism in Protonated Uracil: New Insights from Theoretical Studies. J. Phys. Chem. A. 2020, 124, 5089–5097. [Google Scholar] [CrossRef]
- Imran, M.; Ghorat, F.; Ul-Haq, I.; Ur-Rehman, H.; Aslam, F.; Heydari, M.; Shariati, M.A.; Okuskhanova, E.; Yessimbekov, Z.; Thiruvengadam, M.; et al. Lycopene as a Natural Antioxidant Used to Prevent Human Health Disorders. Antioxidants 2020, 9, 706. [Google Scholar] [CrossRef]
- Pereira, B.L.B.; Reis, P.P.; Severino, F.E.; Felix, T.F.; Braz, M.G.; Nogueira, F.R.; Silva, R.A.C.; Cardoso, A.C.; Lourenco, M.A.M.; Figueiredo, A.M.; et al. Tomato (Lycopersicon esculentum) or lycopene supplementation attenuates ventricular remodeling after myocardial infarction through different mechanistic pathways. J. Nutr. Biochem. 2017, 46, 117–124. [Google Scholar] [CrossRef]
- Sun, X.; Jia, H.; Xu, Q.; Zhao, C.; Xu, C. Lycopene alleviates H2O2-induced oxidative stress, inflammation and apoptosis in bovine mammary epithelial cells via the NFE2L2 signaling pathway. Food Funct. 2019, 10, 6276–6285. [Google Scholar] [CrossRef]
- Li, N.; Wub, X.; Zhuang, W.; Xia, L.; Chen, Y.; Wuc, C.h.; Road, Z.; Due, L.; Zhao, R.; Yi, M.; et al. Tomato and lycopene and multiple health outcomes: Umbrella view. Food Chem. 2021, 343, 128396. [Google Scholar] [CrossRef]
- Nakamura, M.; Sadoshima, J. Mechanisms of physiological and pathological cardiac hypertrophy. Nat. Rev. Cardiol. 2018, 15, 387–407. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, J.; Long, Z.; Wang, C.; Wang, L.; Sun, P.; Li, P.; Wang, T. Hydrogen (H2) inhibits isoproterenol-induced cardiac hypertrophy via antioxidative pathways. Molec. Med. Rep. 2017, 16, 8253–8258. [Google Scholar] [CrossRef]
- Kim, T.Y.; Terentyeva, R.; Roder, K.H.; Li, W.; Liu, M.; Greener, I.; Hamilton, S.; Polina, I.; Murphy, K.R.; Clements, R.T.; et al. SK channel enhancers attenuate Ca2+-dependent arrhythmia in hypertrophic hearts by regulating Mito-ROS-dependent oxidation and activity of RyR. Cardiovasc. Res. 2017, 113, 343–345. [Google Scholar]
- Sangeetha, K.N.; Lakshmi, B.S.; Niranjali-Devaraj, S. Dexamethasone promotes hypertrophy of H9C2 cardiomyocytes through calcineurin B pathway, independent of NFAT activation. Mol. Cell. Biochem. 2016, 411, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Chao, H.H.; Sung, L.C.; Chen, C.H.; Liu, J.C.; Chen, J.J.; Cheng, T.H. Lycopene inhibits urotensin-II-induced cardiomyocyte hypertrophy in neonatal rat cardiomyocytes. Evid. Based Complement. Alternat. Med. 2014, 2014, 724670. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.Y.; Hartig, N.; Kaufman, K.; Hooshmand, S.; Figueroa, A.; Kern, M. Watermelon consumption improves inflammation and antioxidant capacity in rats fed an atherogenic diet. Nutr. Res. 2015, 5, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Sorriento, D.; Laccarino, G. Inflammation and Cardiovascular Diseases: The Most Recent Findings. Int. J. Mol. Sci. 2019, 20, 3879. [Google Scholar] [CrossRef] [PubMed]
- Teague, H.; Mehta, N.N. The link between inflammatory disorders and coronary heart disease: A look at recent studies and novel drugs in development. Curr. Atheroscler. Rep. 2016, 18, 2–5. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Z.; Shao, J.; Lin, L.; Jiang, M.; Wang, L.; Lu, X.; Zhang, H.; Chen, Y.; Zhang, R. Immune and inflammation in acute coronary syndrome: Molecular mechanisms and therapeutic implications. J. Immunol. Res. 2020, 2020, 1–11. [Google Scholar] [CrossRef]
- Hung, C.F.; Huang, T.F.; Chen, B.H.; Shieh, J.M.; Wu, P.H.; Wu, W.B. Lycopene inhibits TNF-alpha-induced endothelial ICAM-1 expression and monocyte-endothelial adhesion. Eur. J. Pharmacol. 2008, 586, 275–282. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Xia, P.; Jin, H.; Zhang, Y.; Chen, B.; Xu, Z. Lycopene ameliorates transplant arteriosclerosis in vascular allograft transplantation by regulating the NO/cGMP pathways and Pho-associated kinases expression. Oxid. Med. Cell Longev. 2016, 2016, 3128280. [Google Scholar] [CrossRef]
- Li, X.N.; Lin, J.; Xia, J.; Qin, L.; Zhu, S.Y.; Li, J.L. Lycopene mitigates atrazine-induced cardiac inflammation via blocking the NF-κB pathway and NO production. J. Funct. Food. 2017, 29, 208–216. [Google Scholar] [CrossRef]
- Wang, X.; Lv, H.; Gu, Y.; Wang, X.; Cao, H.; Tang, Y.; Chen, H.; Huang, C. Protective effect of lycopene on cardiac function and myocardial fibrosis after acute myocardial infarction in rats via the modulation of p38 and MMP-9. J. Mol. Histol. 2014, 45, 113–120. [Google Scholar] [CrossRef]
- Ferreira-Santos, P.; Aparicio, R.; Carrón, R.; Sevilla, M.; Monroy-Ruiz, J.; Montero, M.J. Lycopene-supplemented diet ameliorates cardiovascular remodeling and oxidative stress in rats with hypertension induced by Angiotensin II. J. Funct. Food. 2018, 47, 279–287. [Google Scholar] [CrossRef]
- Yang, P.M.; Huang-Zhi Chen, H.Z.; Huang, Y.T.; Chia-Wen Hsieh, C.W.; Wung, B.S. Lycopene inhibits NF-kB activation and adhesion molecule expression through NrF2-mediated heme oxygenase-1 in endothelial cells. Int. J. Molec. Med. 2017, 39, 1533–1540. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.V.N.; Elango, P.; Asmathulla, S.; Kaviman, S. A Systematic Review on Lycopene and Its Beneficial Effects. Biomed. Pharmcol. J. 2017, 10, 2113–2120. [Google Scholar]
- Sung, L.C.; Chao, H.H.; Chen, C.H.; Tsai, J.C.; Liu, J.C.; Hong, H.J.; Cheng, T.H.; Chen, J.J. Lycopene inhibits cyclic strain-induced endothelin-1 expression through the suppression of reactive oxygen species generation and induction of heme oxygenase-1 in human umbilical vein endothelial cells. Clin. Exp. Pharmacol. Physiol. 2015, 42, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Sahin, K.; Orhan, C.; Tuzcu, M.; Sahin, N.; Ali, S.; Bahcecioglu, I.H.; Guler, O.; Ozercan, I.; Ilhan, N.; Kucuk, O. Orally administered lycopene attenuates diethylnitrosamine-induced hepatocarcinogenesis in rats by modulating Nrf-2/HO-1 and Akt/mTOR pathways. Nutr. Cancer 2014, 66, 590–598. [Google Scholar] [CrossRef] [PubMed]
- Tierney, A.C.; Rumble, C.E.; Billings, L.M.; George, E.S. Effect of Dietary and Supplemental Lycopene on Cardiovascular Risk Factors: A Systematic Review and Meta-Analysis. Adv. Nutr. 2020, 11, 1453–1488. [Google Scholar] [CrossRef]
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das, S.R.; et al. Heart disease and stroke statistics-2019 update: A report from the American Heart Association. Circulation 2019, 139, e56–e528. [Google Scholar] [CrossRef]
- Chistiakov, D.A.; Shkurat, T.P.; Melnichenko, A.A.; Grechko, A.V.; Orekhov, A.N. The role of mitochondrial dysfunction in cardiovascular disease: A brief review. Ann. Med. 2018, 50, 121–127. [Google Scholar] [CrossRef]
- Yu, E.P.K.; Reinhold, J.; Yu, H.; Starks, L.; Uryga, A.K.; Foote, K.; Finigan, A.; Figg, N.; Pung, Y.P.; Logan, A.; et al. Mitochondrial respiration is reduced in atherosclerosis, promoting necrotic core formation and reducing relative fibrous cap thickness. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 2322–2332. [Google Scholar] [CrossRef]
- Huang, D.; Gao, W.; Zhong, X.; Ge, J. NLRP3 activation in endothelia promotes development of diabetes-associated atherosclerosis. Aging 2020, 12, 18181–18191. [Google Scholar] [CrossRef] [PubMed]
- Markin, A.M.; Khotina, W.A.; Zabudskaya, X.G.; Bogatyreva, A.; Starodubova, A.V.; Ivanova, E.; Nikiforov, N.G.; Orekhov, A.N. Disturbance of mitochondrial dynamics and mitochondrial therapies in atherosclerosis. Life 2021, 11, 165. [Google Scholar] [CrossRef] [PubMed]
- Suárez-Rivero, J.M.; Pastor-Maldonado, C.J.; Povea-Cabello, S.; Álvarez-Córdoba, M.; Villalón-García, I.; Talaverón-Rey, M.; Suárez-Carrillo, A.; Munuera-Cabeza, M.; Sánchez-Alcázaret, J.A. From mitochondria to atherosclerosis: The inflammation path. Biomed. 2021, 9, 258. [Google Scholar] [CrossRef]
- Li, D.; Shengjie Yang, S.; Xing, Y.; Pan, L.; Zhao, R.; Zhao, Y.; Liu, L.; Wu, M. Novel Insights and Current Evidence for Mechanisms of Atherosclerosis: Mitochondrial Dynamics as a Potential Therapeutic Target. Front. Cell Dev. Biol. 2021, 9, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, C.A.; Baker, E.J.; Miles, E.A.; Calder, P.C. Eighteen-carbon trans fatty acids and inflammation in the context of atherosclerosis. Prog. Lipid Res. 2019, 76, 101009. [Google Scholar] [CrossRef]
- Dubuc, G.; Chamberland, A.; Wassef, H.; Davignon, J.; Seidah, N.G.; Bernier, L.; Prat, A. Statins uregulate PCSK9 the gene encoding the proprotein convertase neural apoptosis-regulated convertase-1 implicated in familid hypercholesterolemia. Arteroscler. Thromb. Vasc. Biol. 2004, 24, 1454–1459. [Google Scholar] [CrossRef]
- Brown, M.S.; Goldstein, J.L. Cholesterol feedback: From Schoenheimer’s bottle to Scap’s MELADL. J. Lipid. Res. 2009, 50, s15–s27. [Google Scholar] [CrossRef]
- Reiner, Z. Resistance and intolerance to statins. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 1057–1066. [Google Scholar] [CrossRef]
- Forrester, J.S.; Bairey-Merz, C.N.; Kaul, S. The aggressive low density lipoprotein lowering controversy. J. Am. Coll. Cardiol. 2000, 36, 1419–1425. [Google Scholar] [CrossRef][Green Version]
- Horton, J.D.; Cohen, J.C.; Hobbs, H.H. Molecular biology of PCSK9: Its role in LDL metabolism. Trends Biochem. Sci. 2007, 32, 71–77. [Google Scholar] [CrossRef]
- Alvi, S.S.; Ansari, I.A.; Khan, I.; Iqbal, J.; Khan, M.S. Potential role of lycopene in targeting proprotein convertase subtilisin/kexin type-9 to combat hypercholesterolemia. Free Radic. Biol. Med. 2017, 108, 394–403. [Google Scholar] [CrossRef]
- Alvi, S.S.; Ansari, I.A.; Khan, M.S. Pleiotropic role of lycopene in protecting various risk factors mediated atherosclerosis. Ann. Phytomed. 2015, 4, 54–60. [Google Scholar]
- Alvi, S.S.; Iqbal, D.; Ahmad, S.; Khan, M.S. Molecular rationale delineating the role of lycopene as a potent HMG-CoA reductase inhibitor: In vitro and in silico study. Nat. Product. Res. 2016, 30, 2111–2114. [Google Scholar] [CrossRef] [PubMed]
- McEneny, J.; Wade, L.; Young, I.S.; Masson, L.; Duthie, G.; McGinty, A.; McMaster, C.; Thies, F. Lycopene intervention reduces inflammation and improves HDL functionality in moderately overweight middle-aged individuals. J. Nutr. Biochem. 2013, 24, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.F.; Chang, Y.Y.; Huang, H.C.; Wu, Y.C.; Yang, M.D.; Chao, P.M. Tomato juice supplementation in young women reduces inflammatory adipokine levels independently of body fat reduction. Nutrients 2015, 31, 691–696. [Google Scholar] [CrossRef] [PubMed]
- Poznyak, A.; Grechko, A.V.; Poggio, P.; Myasoedova, V.A.; Alfieri, V.; Orekhov, A.N. The Diabetes Mellitus–Atherosclerosis Connection: The Role of Lipid and Glucose Metabolism and Chronic Inflammation. Int. J. Mol. Sci. 2020, 21, 1835. [Google Scholar] [CrossRef]
- Chiva-Blanch, G.; Jiménez, C.; Pinyol, M.; Herreras, Z.; Catalán, M.; Martínez-Huélamo, M.; Lamuela-Raventos, R.M.; Sala-Vila, A.; Cofán, M.; Gilabert, R.; et al. 5-cis-, Trans- and Total Lycopene Plasma Concentrations Inversely Relate to Atherosclerotic Plaque Burden in Newly Diagnosed Type 2 Diabetes Subjects. Nutrients 2020, 12, 1696. [Google Scholar] [CrossRef]
- Nishimura, M.; Tominaga, N.; Ishikawa-Takano, Y.; Maeda-Yamamoto, M.; Nishihira, J. Effect of 12-Week Daily Intake of the High-Lycopene Tomato (Solanum Lycopersicum), a Variety Named “PR-7”, on Lipid Metabolism: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Study. Nutrients 2019, 11, 1177. [Google Scholar] [CrossRef]
- Kumar, R.; Salwe, K.J.; Kumarappan, M. Evaluation of Antioxidant, Hypolipidemic, and Antiatherogenic Property of Lycopene and Astaxanthin in Atherosclerosis-induced Rats. Pharmacogn. Res. 2017, 9, 161–167. [Google Scholar]
- Palozza, P.; Parrone, N.; Simone, R.E.; Catalano, A. Lycopene in atherosclerosis prevention: An integrated scheme of the potential mechanisms of action from cell culture studies. Arch. Biochem. Biophys. 2010, 504, 26–33. [Google Scholar] [CrossRef]
- Slivnick, J.A.; Brent, C.; Lampert, D.O. Hypertension and Heart Failure. Heart Fail. Clin. 2019, 15, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Al Ghorani, H.; Götzinger, F.; Böhm, M.; Mahfoud, F. Arterial hypertension Clinical trials update. Nutr. Metab. Cardiovasc. Dis. 2021, 32, 21–31. [Google Scholar] [CrossRef]
- Whelton, P.K.; Carey, R.M.; Aronow, W.S.; Casey, D.E., Jr.; Collins, K.J.; Dennison Himmelfarb, C.; DePalma, S.M.; Gidding, S.; Jamerson, K.A.; Jones, D.W.; et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: Executive summary: A report of the American College of Cardiology/American Heart Association Task Force 2on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2018, 71, 2199–2269. [Google Scholar] [PubMed]
- Tackling, G.; Borhade, M.B. Hypertensive Heart Disease; StatPearls Publishing: Treasure Island, FL, USA, 2019. Available online: www.ncbi.nlm.nih.gov›NBK539800 (accessed on 10 November 2021).
- Mills, K.T.; Bundy, J.D.; Kelly, T.N.; Reed, J.E.; Kearney, P.M.; Reynolds, K.; Chen, J.; Hee, J. Global disparities of hypertension prevalence and control. A Systematic Analysis of Population-Based Studies From 90 Countries. Circulation 2016, 134, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Mills, K.T.; Stefanescu, A.; He, J. The global epidemiology of hypertension. Nat. Rev. Nephrol. 2020, 16, 223–237. [Google Scholar] [CrossRef] [PubMed]
- Tedla, Y.G.; Bautista, L.E. Drug side effect symptoms and adherence to antihypertensive medication. Am. J. Hypertens. 2015, 29, 772–779. [Google Scholar] [CrossRef]
- Roura, L.C.; Arulkumaran, S.S. Facing the noncommunicable disease (NCD) global epidemic—The battle of prevention starts in utero—The FIGO challenge. Best Pract. Res. Clin. Obstet. Gynaecol. 2015, 29, 5–14. [Google Scholar] [CrossRef]
- Emami, M.R.; Safabakhsh, M.; Alizadeh, S.; Asbaghi, O.; Khosroshahi, M.Z. Effect of vitamin E supplementation on blood pressure: A systematic review and meta-analysis. J. Hum. Hypertens. 2019, 33, 499–507. [Google Scholar] [CrossRef]
- Khan, N.I.; Noori, S.; Mahboob, T. Efficacy of lycopene on modulation of renal antioxidant enzymes, ACE and ACE gene expression in hyperlipidaemic rats. J. Renin. Angiotensin Aldosterone Syst. 2016, 17, 1470320316664611. [Google Scholar] [CrossRef]
- Han, G.M.; Liu, P. Higher serum lycopene is associated with reduced prevalence of hypertension in overweight or obese adults. Eur. J. Integr. Med. 2017, 13, 34–40. [Google Scholar] [CrossRef]
- Kim, J.Y.; Paik, J.K.; Kim, O.Y.; Park, H.W.; Lee, J.H.; Jang, Y.; Lee, J.H. Effects of lycopene supplementation on oxidative stress and markers of endothelial function in healthy men. Atherosclerosis. 2011, 215, 189–195. [Google Scholar] [CrossRef]
- Paran, E.; Novack, V.; Engelhard, Y.N.; Hazan-Halevy, I. The effects of natural antioxidants from tomato extract in treated but uncontrolled hypertensive patients. Cardiovasc. Drugs Ther. 2009, 23, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Ried, K.; Frank, O.R.; Stocks, N.P. Dark chocolate or tomato extract for prehypertension: A randomized controlled trial. BMC Complement. Altern. Med. 2009, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hozawa, A.; Jacobs, D.R., Jr.; Steffes, M.W.; Gross, M.D.; Steffen, L.M.; Lee, D.H. Circulating carotenoid concentrations and incident hypertension: The coronary artery risk development in young adults (CARDIA) study. J. Hypertens. 2009, 27, 105. [Google Scholar] [CrossRef] [PubMed]
- Itsiopoulos, C.; Brazionis, L.; Kaimakamis, M.; Cameron, M.; Best, J.D.; O’Dea, K.; Rowley, K. Can the Mediterranean diet lower HbA1c in type 2 diabetes? Results from a randomized cross-over study. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 740–747. [Google Scholar] [CrossRef] [PubMed]
- Paterson, E.; Gordon, M.H.; Niwat, C.; George, T.W.; Parr, L.; Waroonphan, S.; Lovegrove, J.A. Supplementation with fruit and vegetable soups and beverages increases plasma carotenoid concentrations but does not alter markers of oxidative stress or cardiovascular risk factors. J. Nutr. 2006, 136, 2849–2855. [Google Scholar] [CrossRef] [PubMed]
- Gajendragadkar, P.R.; Hubsch, A.; Maki-Petaja, K.M.; Serg, M.; Wilkinson, I.B.; Cheriyan, J. Effects of oral lycopene supplementation on vascular function in patients with cardiovascular disease and healthy volunteers: A randomised controlled trial. PLoS ONE 2014, 9, e99070. [Google Scholar] [CrossRef]
- Belovic, M.M.; Girones-Vilaplana, A.; Moreno, D.A.; Milovanovic, I.L.; Novakovic, A.R.; Karaman, M.A.; Ilic, N.M. Tomato (Solanum lycopersicum L.) processing main product (Juice) and by-product (Pomace) bioactivity potential measured as antioxidant activity and angiotensin-converting enzyme inhibition. J. Food Process. Preserv. 2016, 40, 1229–1237. [Google Scholar] [CrossRef]
- Mozos, I.; Luca, C.T. Crosstalk between oxidative and nitrosative stress and arterial stiffness. Curr. Vasc. Pharmacol. 2017, 15, 446–456. [Google Scholar] [CrossRef]
- Yiannikouris, F.; Gupte, M.; Putnam, K.; Cassis, L. Adipokines and blood pressure control. Curr. Opin. Nephrol. Hypertens. 2010, 19, 195–200. [Google Scholar] [CrossRef]
- Kelishadi, M.R.; Asbaghi, O.; Nazarian, B.; Naeini, F.; Kaviani, M.; Moradi, S.; Askari, G.; Nourian, M.; Ashtary˗Larky, D. Lycopene Supplementation and Blood Pressure: Systematic review and meta-analyses of randomized trials. J. Herb. Med. 2022, 31, 100521. [Google Scholar] [CrossRef]
- Li, X.; Xu, J. Lycopene supplement and blood pressure: An updated meta-analysis of intervention trials. Nutrients 2013, 5, 3696–3712. [Google Scholar] [CrossRef] [PubMed]
- Honda, M.; Kageyama, H.; Hibino, T.; Ichihashi, K.; Takada, W.; Goto, M. Synergistic Effects of Food Ingredients and Vegetable Oils on Thermal Isomerization of Lycopene. J. Oleo. Sci. 2020, 69, 1529–1540. [Google Scholar] [CrossRef] [PubMed]
- Nemli, E.; Ozakdogan, S.; Tomas, M.; McClements, D.J.; Capanoglu, E. Increasing the Bioaccessibility of Antioxidants in Tomato Pomace Using Excipient Emulsions. Food Biophys. 2021, 16, 355–364. [Google Scholar] [CrossRef]
- Petyaev, I.M.; Dovgalevsky, P.Y.; Klochkov, V.A.; Chalyk, N.E.; Pristensky, D.V.; Chernyshova, M.P.; Udumyan, R.; Kocharyan, T.; Kyle, N.H.; Lozbiakova, M.V.; et al. Effect of lycopene supplementation on cardiovascular parameters and markers of inflammation and oxidation in patients with coronary vascular disease. Food Sci. Nutr. 2018, 6, 1770–1777. [Google Scholar] [CrossRef] [PubMed]
- Muller, L.; Caris-Veyrat, C.; Lowe, G.; Bohm, V. Lycopene and its antioxidant role in the prevention of cardiovascular diseases-A critical review. Crit. Rev. Food Sci. Nutr. 2016, 56, 1868–1879. [Google Scholar] [CrossRef]
- Desmarchelier, C.; Borel, P. Overview of carotenoid bioavailability determinants: From dietary factors to host genetic variations. Trends Food Sci. Technol. 2017, 69, 270–280. [Google Scholar] [CrossRef]
- Bohn, T.; Desmarchelier, C.; Dragsted, L.O.; Nielsen, C.S.; Stahl, W.; Ruhl, R.; Keijer, J.; Borel, P. Host-related factors explaining interindividual variability of carotenoid bioavailability and tissue concentrations in humans. Mol. Nutr. Food Res. 2017, 61, 1–37. [Google Scholar] [CrossRef]
- Shao, A.; Hathcock, J.N. Risk assessment for the carotenoids lutein and lycopene. Regul. Toxicol. Pharmacol. 2006, 45, 289–298. [Google Scholar] [CrossRef]
- Colle, I.J.P.; Lemmens, L.; Buggenhout, S.V.; Loey, A.M.V.; Hendrickx, M.E. Modeling Lycopene Degradation and Isomerization in the Presence of Lipids. Food Bioprocess. Technol. 2013, 6, 909–918. [Google Scholar] [CrossRef]
- Friedman, M. Anticarcinogenic, Cardioprotective, and Other Health Benefits of Tomato Compounds Lycopene, α-Tomatine, and Tomatidine in Pure Form and in Fresh and Processed Tomatoes. J. Agric. Food Chem. 2013, 61, 9534–9550. [Google Scholar] [CrossRef] [PubMed]
- Biddle, M.J.; Lennie, T.A.; Bricker, G.V.; Kopec, R.E.; Schwartz, S.J.; Moser, D.K. Lycopene Dietary Intervention: A pilot study in patients with heart failure. J. Card. Nurs. 2015, 30, 205–212. [Google Scholar] [CrossRef]
- Costa-Rodrigues, J.; Pinho, O.; Monteirob, P.R.R. Can lycopene be considered an effective protection against cardiovascular disease? Food Chem. 2018, 245, 1148–1153. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.M.; Koutsidis, G.; Lodge, J.K.; Ashor, A.; Siervo, M.; Lara, J. Tomato and lycopene supplementation and cardiovascular risk factors: A systematic reviewand meta-analysis. Atherosclerosis 2017, 257, 100–108. [Google Scholar] [CrossRef]
- Misra, R.; Mangi, S.; Joshi, S.; Mittal, S.; Gupta, S.K.; Pandey, R.M. LycoRed as an alternative to hormone replacement therapy in lowering serum lipids and oxidative stress markers: A randomized controlled clinical trial. J. Obstet. Gynaecol. 2006, 32, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Burton-Freeman, B.; Talbot, J.; Park, E.; Krishnankutty, S.; Edirisinghe, I. Protective activity of processed tomato products on postprandial oxidation and inflammation: A clinical trial in healthy weight men and women. Mol. Nutr. Food Res. 2012, 56, 622–631. [Google Scholar] [CrossRef] [PubMed]
- Xaplanteris, P.; Vlachopoulos, C.; Pietri, P.; Terentes-Printzios, D.; Kardara, D.; Alexopoulos, N.; Aznaouridis, K.; Miliou, A.; Stefanadis, C. Tomato paste supplementation improves endothelial dynamics and reduces plasma total oxidative status in healthy subjects. Nutr. Res. 2012, 32, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Abete, I.; Perez-Cornago, A.; Navas-Carretero, S.; Bondia-Pons, I.; Zulet, M.A.; Martinez, J.A. A regular lycopene enriched tomato sauce consumption influences antioxidant status of healthy young-subjects: A crossover study. J. Funct. Foods 2013, 5, 28–35. [Google Scholar] [CrossRef]
- Tsitsimpikou, C.; Tsarouhas, K.; Kioukia-Fougia, N.; Skondra, C.; Fragkiadaki, P.; Papalexis, P.; Stamatopoulos, P.; Kaplanis, I.; Hayes, A.W.; Tsatsakis, A.; et al. Dietary supplementation with tomato-juice in patients with metabolic syndrome: A suggestion to alleviate detrimental clinical factors. Food. Chem. Toxicol. 2014, 9, e13. [Google Scholar] [CrossRef]
- Ghavipour, M.; Sotoudeh, G.; Ghorbani, M. Tomato juice consumption improves blood antioxidative biomarkers in overweight and obese females. Clin. Nutr. 2015, 34, 805–809. [Google Scholar]
- Deplanque, X.; Muscente-Paque, D.; Chappuis, E. Proprietary tomato extract improves metabolic response to high-fat meal in healthy normal weight subjects. Food Nutr. Res. 2016, 60, 1–8. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Valderas-Martinez, P.; Chiva-Blanch, G.; Casas, R.; Arranz, S.; Martínez-Huélamo, M.; Urpi-Sarda, M.; Torrado, X.; Corella, D.; Lamuela-Raventós, R.M.; Estruch, R. Tomato sauce enriched with olive oil exerts greater effects on cardiovascular disease risk factors than raw tomato and tomato sauce: A randomized trial. Nutrients 2016, 8, 170. [Google Scholar] [CrossRef] [PubMed]
- Colmán Martínez, M.; Martinez-Huelamo, M.; Valderas-Martinez, P.; Arranz-Martinez, S.; Almanza-Aguilera, E.; Corella, D.; Estruch, R.; Lamuela-Raventos, R.M. trans-Lycopene from tomato juice attenuates inflammatory biomarkers in human plasma samples: An intervention trial. Mol. Nutr. Food Res. 2017, 61, 1600993. [Google Scholar] [CrossRef] [PubMed]
- Alvi, S.S.; Ansari, I.A.; Ahmad, M.K.; Iqbal, J.; Khan, M.S. Lycopene amends LPS induced oxidative stress and hypertriglyceridemia via modulating PCSK-9 expression and Apo-C III mediated lipoprotein lipase activity. Biomed. Pharmacother. 2017, 96, 1082–1093. [Google Scholar] [CrossRef] [PubMed]
- Ojha, S.; Goyal, S.; Sharma, C.; Arora, S.; Kumari, S.; Arya, D.S. Cardioprotective effect of lycopene against isoproterenol-induced myocardial infarction in rats. Hum. Exp. Toxicol. 2013, 32, 492–503. [Google Scholar] [CrossRef]
- Trumbo, P.R. Are there adverse effects of lycopene exposure? J. Nutr. 2005, 135, 2060S–2061S. [Google Scholar] [CrossRef] [PubMed]
- Jonker, D.; Kuper, C.F.; Fraile, N.; Estrella, A.; Rodríguez Otero, C. Ninety-day oral toxicity study of lycopene from Blakeslea trispora in rats. Regul. Toxicol. Pharmacol. 2003, 37, 396–406. [Google Scholar] [CrossRef]
- Eliassen, A.H.; Hendrickson, S.J.; Brinton, L.A.; Buring, J.E.; Campos, H.; Dai, Q.; Dorgan, J.F.; Franke, A.A.; Gao, Y.T.; Goodman, M.T. Circulating carotenoids and risk of breast cancer: Pooled analysis of eight prospective studies. J. Natl. Cancer Inst. 2012, 104, 1905–1916. [Google Scholar] [CrossRef]
- Aguilar, F.; Autrup, H.; Barlow, S.; Castle, L.; Crebelli, R.; Dekant, W.; Engel, K.H.; Gontard, N.; Gott, D.; Grilli, S. Use of lycopene as a food colour scientific opinion of the panel on food additives, flavorings, processing aids and materials in contact with food. Eur. Food Saf. Auth. J. 2008, 674, 1–66. [Google Scholar]
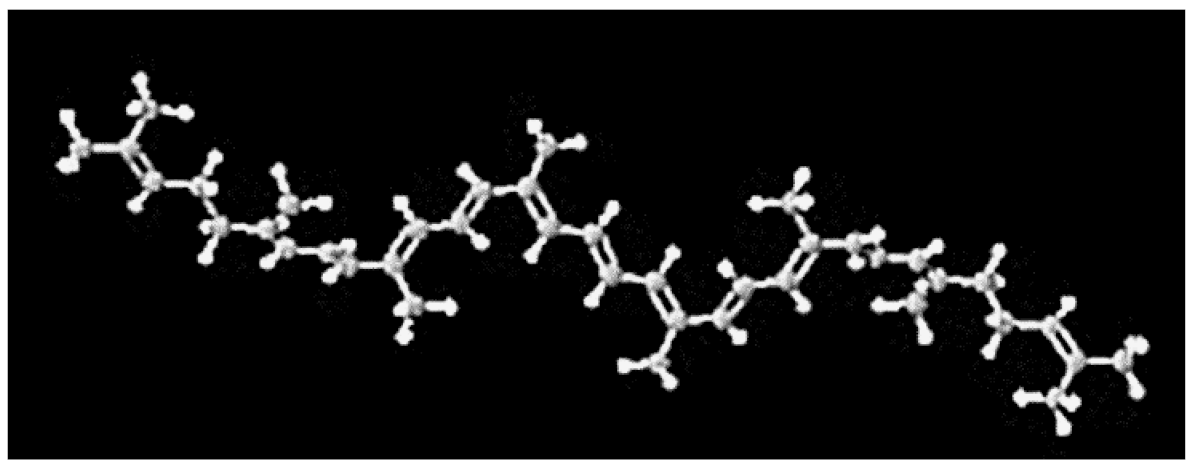
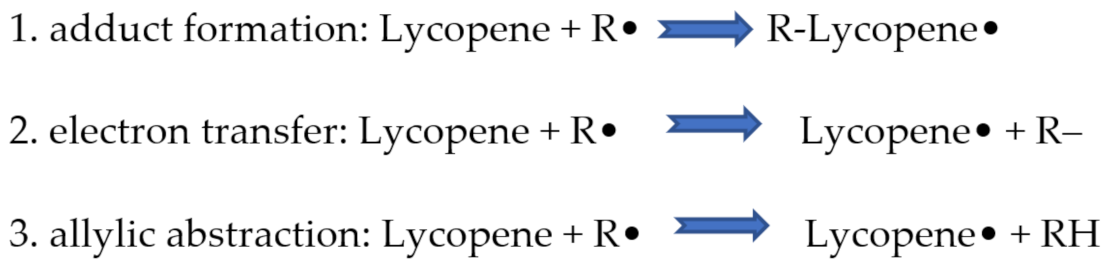
| Source | Lycopene Contents (mg/100 g) |
|---|---|
| Fresh tomato | 5.6 |
| Tomato juice | 9.04 |
| Ketchup | 16.6 |
| Tomato sauces | 23.8 |
| Tomato concentrate | 54.0 |
| Tomato puree | 21.7 |
| Tomato sun-dried | 45.9 |
| Study Authors/Country | Area of Interest | Study Design | Age and Health Condition | Dose and Treatment Period of Lycopene | Main Findings | Reference |
|---|---|---|---|---|---|---|
| Misra et al., 2006 (India) | Lipid profile | Parallel, RCT | 41 healthy postmenopausal women at the age of 46 | Two capsules of lycopene (LycoRed) 2 mg per day (n = 20 women), for 6 months; Control (n = 21) hormone replacement therapy (HRT) estradiol valerate 2 mg and norethisterone acetate 1 mg | LycoRed showed beneficial effects on serum lipids and markers of oxidative stress that were comparable to HRT. LycoRed: ↓TC(24.2%); ↓LDL(14.9%); ↑HDL (26.1%), ↓MDA, ↑GSH | [130] |
| Paran et al., 2009 (Israel) | Blood pressure | Crossover, RCT | 50 hypertensive patients with hypertension between 46 and 66 years old; SBP 140–159 mmHg DBP 90–99 mmHg | Encapsulated tomato extract (Lyc-O-Mato®) 250 mg, containing 15 mg per day (n = 26 men; n = 24 women) for 6 weeks; Control (26 men; n = 24 women) placebo capsule with soya oil and normal diet for 6 weeks | Tomato extract containing 15 mg of lycopene favorable reduction of SBP from 145.8 to 132.2 mmHg and 140.4 to 128.7 mmHg and DBP 82.1 to 77.9 mmHg. Serum lycopene content increased from 0.30 μmol/L | [107] |
| Kim et al., 2011 (Korea) | Endothelial function | Parallel, RCT, double-blind | 126 healthy men aged 22–57 years | Lycopene in the form (Lyc-O-Mato®) 6 mg per day (n = 41 men); 15 mg per day (n = 37 men) for 8 weeks; Control placebo capsule with soya oil and normal diet for 8 weeks | Supplementation with lycopene in the amount of 15 mg/day for 8 weeks in the group of tested men had a positive effect on endothelial function. Increased the RH-PAT index by 23%. Decreased oxidative DNA damage and increased plasma SOD activity. Moreover, it lowered SBP and the level of: hs-CRP, sICAM-1 and sVCAM-1. | [106] |
| Burton-Freeman et al., 2012 (USA) | Oxidative stress | RCT, Crossover | 25 healthy patients age 27 ± 8 years | Randomly selected patients received (n = 12 men and n = 13 women): I: 85 g tomato paste/day, II: a diet without the participation of tomatoes (control) by 360 min | Lycopene significantly attenuated high-fat meal ↓LDL oxidation and ↓interleukin-6 a proinflammatory cytokine and a proinflammatory cytokine and inflammation marker | [131] |
| Xaplanteris et al., 2012 (Greece) | Endothelial function | RCT, Crossover | 19 healthy patients age 39 ± 13 years | Patients (n = 8 men, n = 11 women) received: I: 70 g of tomato paste containing 33.3 mg of lycopene in their diet, II: diet without tomato paste: control for two weeks every day | Tomato paste supplementation increased FMD compared with the control period. It improves the functions of the endothelium. Moreover, it lowers plasma lipid peroxides (TOS) | [132] |
| Abete et al., 2013 (Spain) | Oxidative stress markers | RCT, Double-blind, Crossover | 30 healthy patients aged 39 ± 6 years | Randomly selected patients received (n = 9 men, n = 21 women): I: tomato sauces 160 g/day containing (27.2 mg lycopene/day), II: commercial tomato sauce 160 g/day with a reduced content of lycopene (12.3 mg lycopene/day) for 10 weeks | The consumption of tomato sauce with a higher concentration of lycopene (27 mg/day) caused a decrease in oxidized LDL-cholesterol levels | [133] |
| Grajendragadkar et al., 2014 (UK) | Vascular function | Parallel, RCT, double-blind | 72 patients, including 36 with cardiovascular disease and 36 healthy volunteers aged 30–80 years | Lycopene 7 mg per day patients healthy (n = 23 men, n = 1 women) for 2 months; Control (n = 10 men, n = 2 women) placebo capsule CVD patients (n = 15 men, n = 9 women): 7 mg per day lycopene, Control (n = 10 men, n = 2 women) placebo capsule by 8 weeks | Lycopene supplementation in CVD patients improved endothelium-dependent vasodilatation (EDV) by 53%. EDV values were close to the baseline values of healthy patients, indicating an improvement in endothelial function by lycopene. It also caused a slight reduction in SB pressure in patients with CVD by 2.9 mmHg | [112] |
| Tsitsimpikou et al., 2014 (Greece) | Metabolic syndrome Endothelial function Lipid profile | Parallel, RCT | 27 patients with metabolic syndrome aged 53 ± 10 years | Randomly selected patients received: I: tomato juice 100 mL 4 times a week over a period of two months (n = 13 men, n = 2 women), II: water: control group (n = 11 men, n = 1 women) | Tomato juice: ↓LDL, ↑HDL. In addition, it lowered the markers of inflammation TNF-α and IL-6. Endothelial function and insulin resistance improved also improved as a result of consuming tomato juice | [134] |
| Ghavipour et al., 2015 (Iran) | Oxidative stress | RCT | 64 overweight and obese female patients (BMI ¼ 25 kg/m2 or higher) aged between 20 and 30 years | Female students received: I: tomato juice 330 mL/day containing (37.0 mg/day lycopene) (n = 32), II: control: water (n = 28) for 20 days | Tomato juice consumption significantly: ↑TAC, ↑erythrocyte SOD, ↑CAT and ↑GPx of plasma and ↓MDA of serum compared with the control group after 20 days | [135] |
| Deplanque et al., 2016 (France) | Lipid profile | Parallel, RTC | 145 healthy patients aged 17–70 years | Patients divided into two groups, the first (75 patients) taking CRTE capsules containing 15 mg/day lycopene and the second (70 patients) taking placebo capsules without lycopene for 2 weeks | Supplementation of CRTE (tomato extract containing 15 mg of lycopene) for 2 weeks increased the plasma level of lycopene and improved the response of oxidized LDL to a high-fat meal in healthy, normal-weight patients. It also had a positive effect on blood glucose, insulin and TG levels | [136] |
| Valderas-Martinez et al., 2016 (Spain) | Atherosclerosis | RCT, Crossover | 40 healthy patients aged 28 ± 11 years | Randomly selected patients received (n = 19 men, n = 21 women): I: Raw Tomato (RT): 7 g of tomato/kg of body weight, II: tomato sauce (TS): 3.5 g of tomato sauce/kg of body weight; III: tomato sauce with olive oil (TSOO): 3.5 g of tomato sauce with refined olive oil/kg of BW; IV: control: 0.25 g of sugar dissolved in water/kg of BW for 14 weeks | The three groups of products used: RT, TS and TSOO among the examined patients resulted in: ↓TC, ↓LDL, ↑HDL↓. In the assessment of inflammatory markers, the products (TR, TS, TSOO) caused: ↓MCP-1. Whereas, TR and TOOO: ↓IL18 and TOOO: ↓IL6, ↓VCAM-1 | [137] |
| Colmán Martínez et al., 2017 (Spain) | Inflammatory biomarkers Atherosclerosis | RCT, Crossover | 28 patients (men) at high risk of cardiovascular disease age 69 ± 3 years | The patients randomly drank in the studies: I: 200 mL (low dose) tomato juice, II: 400 mL (high dose) tomato juice, III: control: water for 4 weeks every day | The use of alternating doses of juices resulted in: ↓ICAM-1, ↓VCAM-1, ↓IL8. A reduction was observed after the consumption of tomato juice (low dose): chemokine CXCL10, CRP, and IFN. However, these changes were not noticed after consuming the juice at a higher dose | [138] |
| Petyaev et al. 2018 (Russia) | Lipid profile | RCT | 142 patients with coronary artery disease aged 45 to 73 years | Patients divided into two groups. One group took lacto-lycopene (Nestle Inc.) (n = 68), the other group took microencapsulated GA lycopene (Lycotec Ltd.) (n = 74 patients) at 7 mg/day (1 capsule) for 4 weeks | The Ga lycopene (lycosome) supplement caused an increase in serum lycopene concentration in patients compared to lacto-lycopene. At the end of the study, it also lowered the oxidized LDL levels by five times. Such an effect was not observed in patients treated with lacto-lycopene. Ga lycopen also caused an increase in tissue oxygenation and flow-mediated dilation by the end of the observational period | [120] |
| Wolak et al., 2019 (Israel) | Blood pressure | Parallel, RCT | 61 hypertensive patients aged 35–60 years | Patients divided into 5 groups: I receiving the tomato nutrient complex, (TNC) containing 5 mg of lycopene (n = 12 patients), II receiving TNC containing 15 mg of lycopene (n = 12 patients), III receiving TNC containing 30 mg of lycopene (n = 13 patients), IV taking synthetic lycopene in the amount of 15 mg (n = 12 patients), V taking a placebo—lycopene-free capsules (soybean oil) (n = 12 patients) administered once daily for 8 weeks | Supplementation with TNC containing lycopene 15 (from 137.4 mmHg to 127.2 mmHg) and 30 mg (from 136.4 mmHg to 130 mmHg) caused a decrease in SBP. On the other hand, TNC (lycopene 5 mg) and synthetic lycopene 15 mg had no effect on the decrease of SBP. In the case of diastolic blood pressure (DBP), a TNC containing 15 mg of lycopene had a beneficial effect on its reduction (from 83.8 to 78.6) | [12] |
| Tested Animal | Lycopene Dose | Effective Operation | Reference |
|---|---|---|---|
| Male Sprague-Dawley (SD) rats injected intraperitoneally with LPS at a dose of 1000 ng/kg body weight/rat to induce infection and inflammation triggered hypertriglyceridemia | (10 mg/kg body weight/day) lycopene was dissolved in corn oil and administered to rats by intra-gastric intubation |
| [139] |
| Male Wistar rats injected with angiotensin II (Ang II) (0.3 mg/kg/day) | Lycopene was incorporated into drinking water (the Redivivo® product) 10 mg/kg/day |
| [66] |
| Male Kunming mice ATR (atrazine)-induced cardiac inflammation (50 mg/kg, 200 mg/kg) | 5 mg/kg body weight/day |
| [64] |
| Male BALA/c mice (18–25 g; 10–12 weeks) Left anterior descending coronary artery ligated | 10 mg/kg body weight/day |
| [45] |
| Male Wistar albino rats Isoproterenol (85 mg/kg) has been used to induce myocardial infarction in 29 and 30 day (two days) | (0.5, 1.0 and 1.5 mg/kg body weight/day) for 30 days |
| [140] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Przybylska, S.; Tokarczyk, G. Lycopene in the Prevention of Cardiovascular Diseases. Int. J. Mol. Sci. 2022, 23, 1957. https://doi.org/10.3390/ijms23041957
Przybylska S, Tokarczyk G. Lycopene in the Prevention of Cardiovascular Diseases. International Journal of Molecular Sciences. 2022; 23(4):1957. https://doi.org/10.3390/ijms23041957
Chicago/Turabian StylePrzybylska, Sylwia, and Grzegorz Tokarczyk. 2022. "Lycopene in the Prevention of Cardiovascular Diseases" International Journal of Molecular Sciences 23, no. 4: 1957. https://doi.org/10.3390/ijms23041957
APA StylePrzybylska, S., & Tokarczyk, G. (2022). Lycopene in the Prevention of Cardiovascular Diseases. International Journal of Molecular Sciences, 23(4), 1957. https://doi.org/10.3390/ijms23041957






