AKR1B10, One of the Triggers of Cytokine Storm in SARS-CoV2 Severe Acute Respiratory Syndrome
Abstract
:1. Introduction
2. Results
2.1. AKR1B10 Is Overexpressed in the Lung of Deceased COVID-19 Patients and Correlated with an Enrichment of Pro-Inflammatory and Cytokine Genes
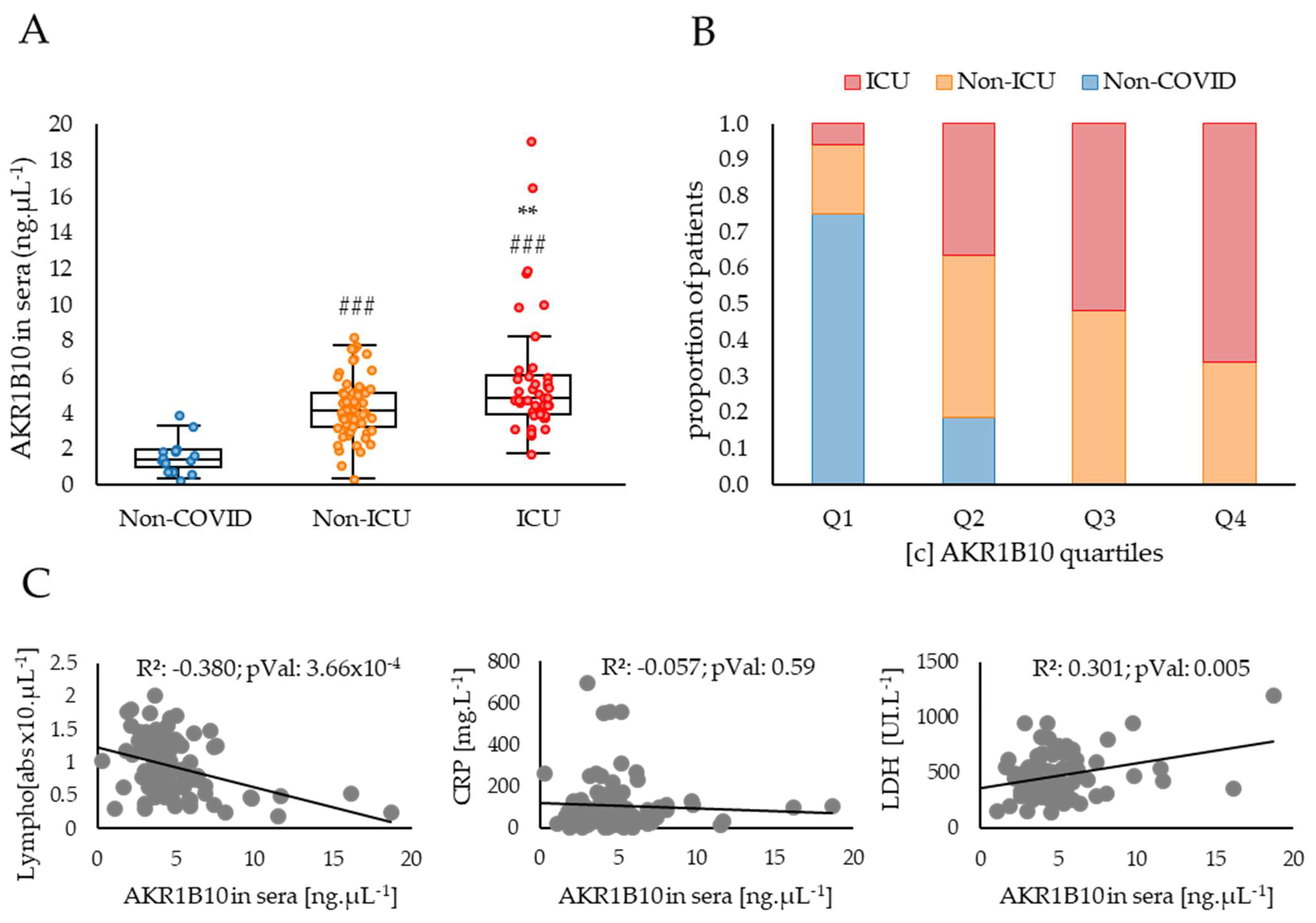
2.2. AKR1B10 Levels Are Increased in the Blood of COVID-19 Patients with Severe or Critical Forms of the Disease Compared to Moderate Forms
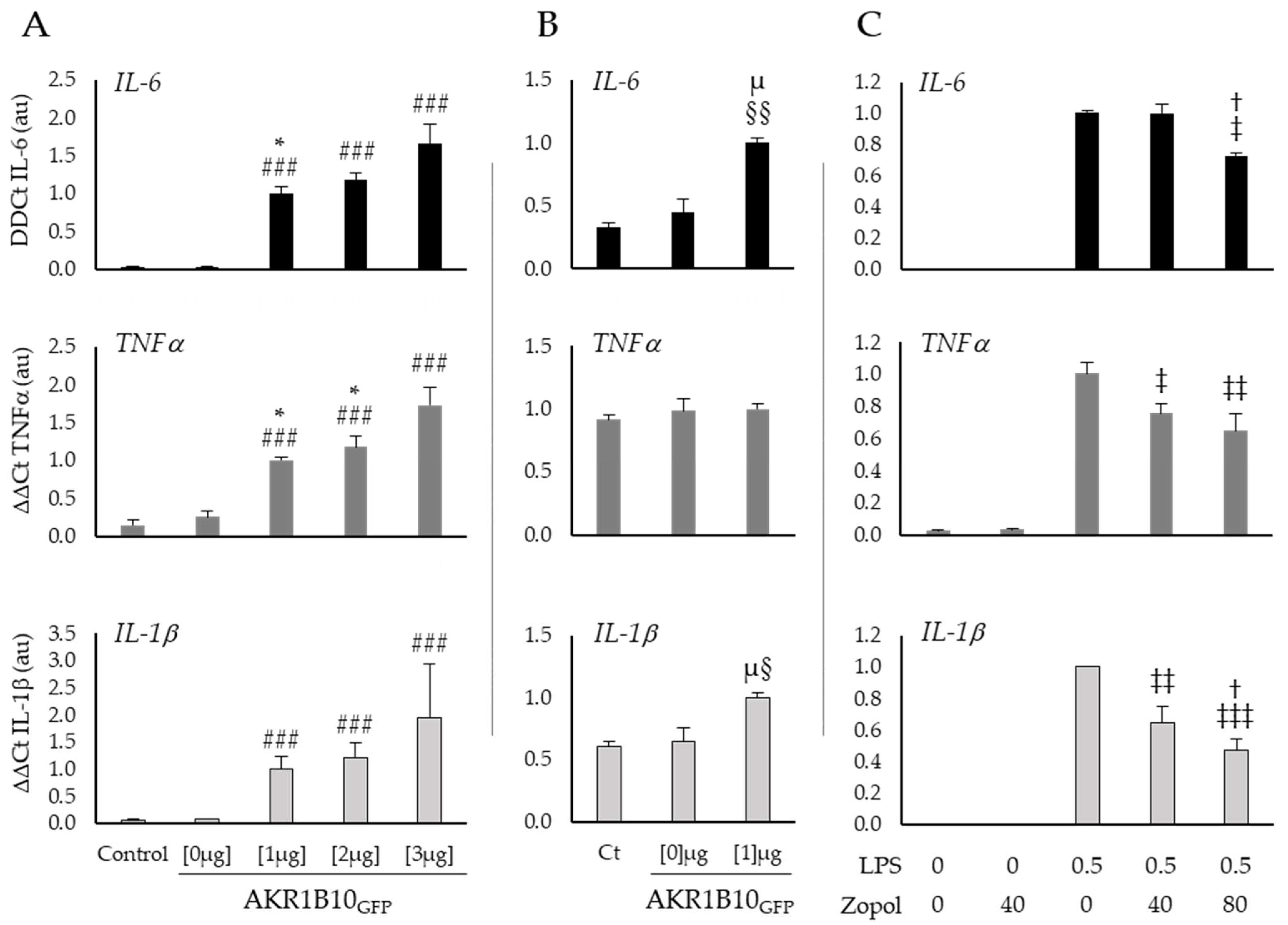
2.3. AKR1B10 Drives Cytokine Production in Cellular Models
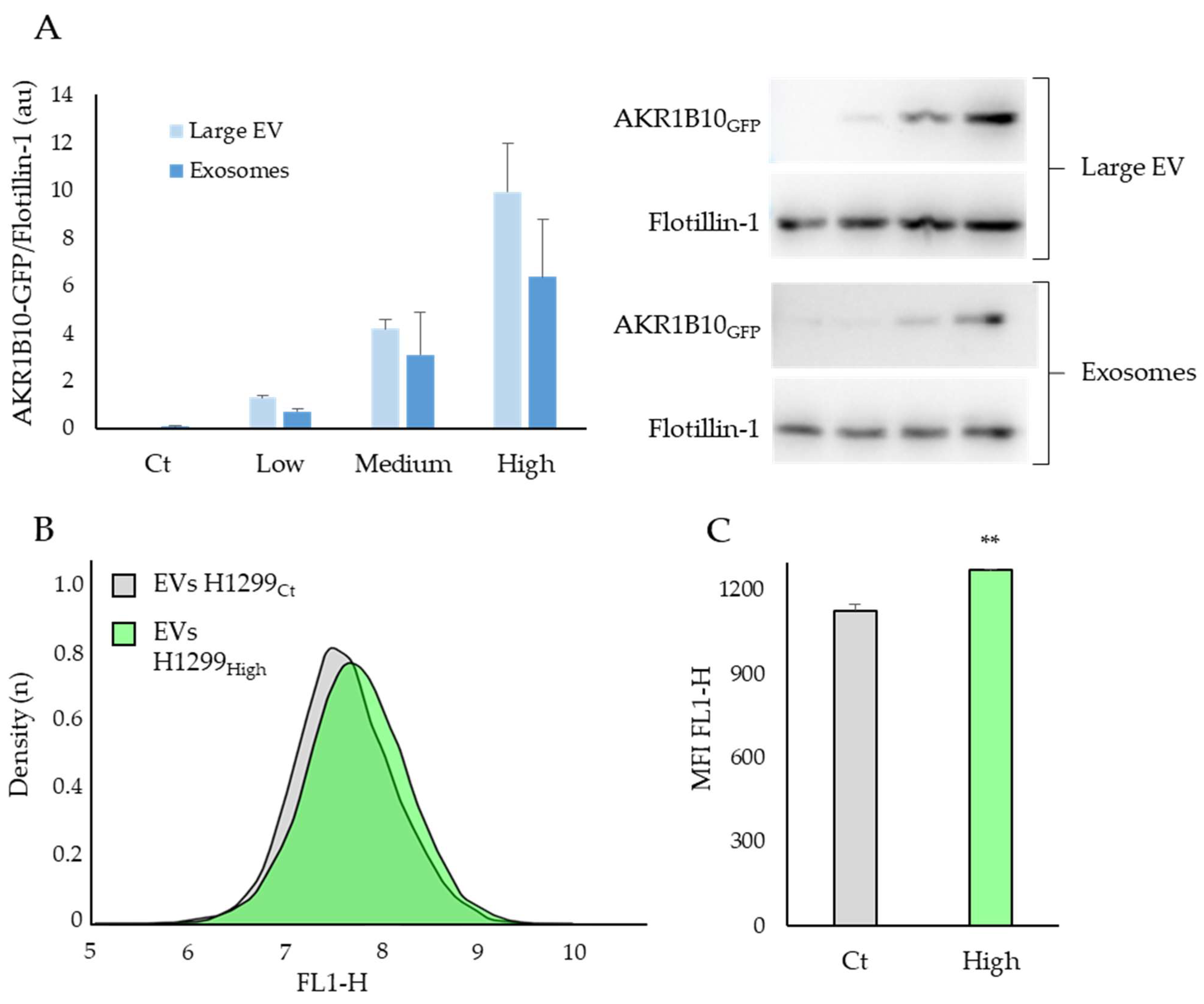
2.4. The AKR1B10 Protein Can Be Transferred between Cell Types via the Extracellular Vesicles
3. Discussion
Strength and Weakness
4. Materials and Methods
4.1. Sera
4.2. Cell Culture
4.3. RT-PCR
4.4. Western Blotting
4.5. Extracellular Vesicles (EVs) Extraction and Exposure
4.6. Transcriptomic Data Analysis
4.7. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19-11 March 2020. Available online: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 12 January 2022).
- Tartof, S.Y.; Slezak, J.M.; Fischer, H.; Hong, V.; Ackerson, B.K.; Ranasinghe, O.N.; Frankland, T.B.; Ogun, O.A.; Zamparo, J.M.; Gray, S.; et al. Effectiveness of MRNA BNT162b2 COVID-19 Vaccine up to 6 Months in a Large Integrated Health System in the USA: A Retrospective Cohort Study. Lancet 2021, 398, 1407–1416. [Google Scholar] [CrossRef]
- Agrawal, U.; Katikireddi, S.V.; McCowan, C.; Mulholland, R.H.; Azcoaga-Lorenzo, A.; Amele, S.; Fagbamigbe, A.F.; Vasileiou, E.; Grange, Z.; Shi, T.; et al. COVID-19 Hospital Admissions and Deaths after BNT162b2 and ChAdOx1 NCoV-19 Vaccinations in 2·57 Million People in Scotland (EAVE II): A Prospective Cohort Study. Lancet Respir. Med. 2021, 9, 1439–1449. [Google Scholar] [CrossRef]
- Lam, C.Y.K.; Zhang, Q.; Yin, B.; Huang, Y.; Wang, H.; Yang, M.; Wong, S.H.D. Recent Advances in Two-Dimensional Transition Metal Dichalcogenide Nanocomposites Biosensors for Virus Detection before and during COVID-19 Outbreak. J. Compos. Sci. 2021, 5, 190. [Google Scholar] [CrossRef]
- Yin, B.; Ho, W.K.H.; Zhang, Q.; Li, C.; Huang, Y.; Yan, J.; Yang, H.; Hao, J.; Wong, S.H.D.; Yang, M. Magnetic-Responsive Surface-Enhanced Raman Scattering Platform with Tunable Hot Spot for Ultrasensitive Virus Nucleic Acid Detection. ACS Appl. Mater. Interfaces 2022, 14, 4714–4724. [Google Scholar] [CrossRef]
- Mizrahi, B.; Shilo, S.; Rossman, H.; Kalkstein, N.; Marcus, K.; Barer, Y.; Keshet, A.; Shamir-Stein, N.; Shalev, V.; Zohar, A.E.; et al. Longitudinal Symptom Dynamics of COVID-19 Infection. Nat. Commun. 2020, 11, 6208. [Google Scholar] [CrossRef]
- Hu, B.; Huang, S.; Yin, L. The Cytokine Storm and COVID-19. J. Med. Virol. 2021, 93, 250–256. [Google Scholar] [CrossRef]
- Karki, R.; Sharma, B.R.; Tuladhar, S.; Williams, E.P.; Zalduondo, L.; Samir, P.; Zheng, M.; Sundaram, B.; Banoth, B.; Malireddi, R.K.S.; et al. Synergism of TNF-α and IFN-γ Triggers Inflammatory Cell Death, Tissue Damage, and Mortality in SARS-CoV-2 Infection and Cytokine Shock Syndromes. Cell 2021, 184, 149–168.e17. [Google Scholar] [CrossRef]
- Rezagholizadeh, A.; Khiali, S.; Sarbakhsh, P.; Entezari-Maleki, T. Remdesivir for Treatment of COVID-19; an Updated Systematic Review and Meta-Analysis. Eur. J. Pharmacol. 2021, 897, 173926. [Google Scholar] [CrossRef]
- Deng, J.; Zhou, F.; Ali, S.; Heybati, K.; Hou, W.; Huang, E.; Wong, C.Y. Efficacy and Safety of Ivermectin for the Treatment of COVID-19: A Systematic Review and Meta-Analysis. QJM Int. J. Med. 2021, 114, 721–732. [Google Scholar] [CrossRef]
- Martins-Filho, P.R.; Ferreira, L.C.; Heimfarth, L.; Araújo, A.A.d.S.; Quintans-Júnior, L.J. Efficacy and Safety of Hydroxychloroquine as Pre-and Post-Exposure Prophylaxis and Treatment of COVID-19: A Systematic Review and Meta-Analysis of Blinded, Placebo-Controlled, Randomized Clinical Trials. Lancet Reg. Health Am. 2021, 2, 62. [Google Scholar] [CrossRef]
- Shah, M.; Woo, H.G. Molecular Perspectives of SARS-CoV-2: Pathology, Immune Evasion, and Therapeutic Interventions. Mol. Cells 2021, 44, 408–421. [Google Scholar] [CrossRef]
- Cano, E.J.; Fonseca Fuentes, X.; Corsini Campioli, C.; O’Horo, J.C.; Abu Saleh, O.; Odeyemi, Y.; Yadav, H.; Temesgen, Z. Impact of Corticosteroids in Coronavirus Disease 2019 Outcomes. Chest 2021, 159, 1019–1040. [Google Scholar] [CrossRef]
- Barski, O.A.; Tipparaju, S.M.; Bhatnagar, A. The Aldo-Keto Reductase Superfamily and Its Role in Drug Metabolism and Detoxification. Drug Metab. Rev. 2008, 40, 553–624. [Google Scholar] [CrossRef] [Green Version]
- Ramana, K.V. ALDOSE REDUCTASE: New Insights for an Old Enzyme. Biomol. Concepts 2011, 2, 103–114. [Google Scholar] [CrossRef] [Green Version]
- Endo, S.; Matsunaga, T.; Nishinaka, T. The Role of AKR1B10 in Physiology and Pathophysiology. Metabolites 2021, 11, 332. [Google Scholar] [CrossRef]
- Steiling, K.; Lenburg, M.E.; Spira, A. Airway Gene Expression in Chronic Obstructive Pulmonary Disease. Proc. Am. Thorac. Soc. 2009, 6, 697–700. [Google Scholar] [CrossRef] [Green Version]
- Ramana, K.V.; Bhatnagar, A.; Srivastava, S.K. Inhibition of Aldose Reductase Attenuates TNF-Alpha-Induced Expression of Adhesion Molecules in Endothelial Cells. FASEB J. 2004, 18, 1209–1218. [Google Scholar] [CrossRef]
- Yadav, U.; Mishra, R.; Aguilera-Aguirre, L.; Sur, S.; Bolodgh, I.; Ramana, K.V.; Srivatsava, S.K. Prevention of Allergic Rhinitis by Aldose Reductase Inhibition in a Murine Model. Inflamm. Allergy Drug Targets 2013, 12, 178–186. [Google Scholar] [CrossRef]
- Pladzyk, A.; Reddy, A.B.M.; Yadav, U.C.S.; Tammali, R.; Ramana, K.V.; Srivastava, S.K. Inhibition of Aldose Reductase Prevents Lipopolysaccharide-Induced Inflammatory Response in Human Lens Epithelial Cells. Inv. Ophthalmol. Vis. Sci. 2006, 47, 5395–5403. [Google Scholar] [CrossRef] [Green Version]
- Srivastava, S.K.; Yadav, U.C.S.; Reddy, A.B.; Saxena, A.; Tammali, R.; Mohammad, S.; Ansari, N.H.; Bhatnagar, A.; Petrash, M.J.; Srivastava, S.; et al. Aldose Reductase Inhibition Suppresses Oxidative Stress-Induced Inflammatory Disorders. Chem. Biol. Interact. 2011, 191, 330–338. [Google Scholar] [CrossRef] [Green Version]
- Yadav, U.C.S.; Ramana, K.V.; Srivastava, S.K. Aldose Reductase Inhibition Suppresses Airway Inflammation. Chem. Biol. Interact. 2011, 191, 339–345. [Google Scholar] [CrossRef] [Green Version]
- Shaw, N.; Yang, B.; Millward, A.; Demaine, A.; Hodgkinson, A. AKR1B10 Is Induced by Hyperglycaemia and Lipopolysaccharide in Patients with Diabetic Nephropathy. Cell Stress Chaperones 2014, 19, 281–287. [Google Scholar] [CrossRef] [Green Version]
- Pettinelli, P.; Arendt, B.M.; Teterina, A.; McGilvray, I.; Comelli, E.M.; Fung, S.K.; Fischer, S.E.; Allard, J.P. Altered Hepatic Genes Related to Retinol Metabolism and Plasma Retinol in Patients with Non-Alcoholic Fatty Liver Disease. PLoS ONE 2018, 13, e0205747. [Google Scholar] [CrossRef]
- Kanno, M.; Kawaguchi, K.; Honda, M.; Horii, R.; Takatori, H.; Shimakami, T.; Kitamura, K.; Arai, K.; Yamashita, T.; Sakai, Y.; et al. Serum Aldo-Keto Reductase Family 1 Member B10 Predicts Advanced Liver Fibrosis and Fatal Complications of Nonalcoholic Steatohepatitis. J. Gastroenterol. 2019, 54, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Portillo-Sanchez, P.; Bril, F.; Maximos, M.; Lomonaco, R.; Biernacki, D.; Orsak, B.; Subbarayan, S.; Webb, A.; Hecht, J.; Cusi, K. High Prevalence of Nonalcoholic Fatty Liver Disease in Patients with Type 2 Diabetes Mellitus and Normal Plasma Aminotransferase Levels. J. Clin. Endocrinol. Metab. 2015, 100, 2231–2238. [Google Scholar] [CrossRef]
- Mitra, S.; De, A.; Chowdhury, A. Epidemiology of Non-Alcoholic and Alcoholic Fatty Liver Diseases. Transl. Gastroenterol. Hepatol. 2020, 5, 16. [Google Scholar] [CrossRef]
- Daamen, A.R.; Bachali, P.; Owen, K.A.; Kingsmore, K.M.; Hubbard, E.L.; Labonte, A.C.; Robl, R.; Shrotri, S.; Grammer, A.C.; Lipsky, P.E. Comprehensive Transcriptomic Analysis of COVID-19 Blood, Lung, and Airway. Sci. Rep. 2021, 11, 7052. [Google Scholar] [CrossRef]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene Set Enrichment Analysis: A Knowledge-Based Approach for Interpreting Genome-Wide Expression Profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Song, J.; Du, X.; Zhou, Y.; Li, Y.; Li, R.; Lyu, L.; He, Y.; Hao, J.; Ben, J.; et al. AKR1B10 (Aldo-Keto Reductase Family 1 B10) Promotes Brain Metastasis of Lung Cancer Cells in a Multi-Organ Microfluidic Chip Model. Acta Biomater. 2019, 91, 195–208. [Google Scholar] [CrossRef]
- Ye, X.; Li, C.; Zu, X.; Lin, M.; Liu, Q.; Liu, J.; Xu, G.; Chen, Z.; Xu, Y.; Liu, L.; et al. A Large-Scale Multicenter Study Validates Aldo-Keto Reductase Family 1 Member B10 as a Prevalent Serum Marker for Detection of Hepatocellular Carcinoma. Hepatology 2019, 69, 2489–2501. [Google Scholar] [CrossRef] [Green Version]
- Luo, D.; Bu, Y.; Ma, J.; Rajput, S.; He, Y.; Cai, G.; Liao, D.-F.; Cao, D. Heat Shock Protein 90-α Mediates Aldo-Keto Reductase 1B10 (AKR1B10) Protein Secretion through Secretory Lysosomes. J. Biol. Chem. 2013, 288, 36733–36740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin, B.; Ni, J.; Witherel, C.E.; Yang, M.; Burdick, J.A.; Wen, C.; Wong, S.H.D. Harnessing Tissue-Derived Extracellular Vesicles for Osteoarthritis Theranostics. Theranostics 2022, 12, 207–231. [Google Scholar] [CrossRef] [PubMed]
- Fathi, N.; Rezaei, N. Lymphopenia in COVID-19: Therapeutic Opportunities. Cell Biol. Int. 2020, 44, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Xie, X.; Tu, Z.; Fu, J.; Xu, D.; Zhou, Y. The Signal Pathways and Treatment of Cytokine Storm in COVID-19. Sig. Transduct. Target Ther. 2021, 6, 1–20. [Google Scholar] [CrossRef]
- Karki, R.; Kanneganti, T.-D. The “Cytokine Storm”: Molecular Mechanisms and Therapeutic Prospects. Trends Immunol. 2021, 42, 681–705. [Google Scholar] [CrossRef]
- Rutledge, H.R.; Jiang, W.; Yang, J.; Warg, L.A.; Schwartz, D.A.; Pisetsky, D.S.; Yang, I.V. Gene Expression Profiles of RAW264.7 Macrophages Stimulated with Preparations of LPS Differing in Isolation and Purity. Innate Immun. 2012, 18, 80–88. [Google Scholar] [CrossRef]
- Wang, W.; Ye, L.; Ye, L.; Li, B.; Gao, B.; Zeng, Y.; Kong, L.; Fang, X.; Zheng, H.; Wu, Z.; et al. Up-Regulation of IL-6 and TNF-α Induced by SARS-Coronavirus Spike Protein in Murine Macrophages via NF-ΚB Pathway. Virus Res. 2007, 128, 1–8. [Google Scholar] [CrossRef]
- Liu, T.; Guo, Y.; Zhao, J.; He, S.; Bai, Y.; Wang, N.; Lin, Y.; Liu, Q.; Xu, X. Systems Pharmacology and Verification of ShenFuHuang Formula in Zebrafish Model Reveal Multi-Scale Treatment Strategy for Septic Syndrome in COVID-19. Front. Pharmacol. 2020, 11, 1464. [Google Scholar] [CrossRef]
- Nahmias, Y.; Ehrlich, A.; Ioannidis, K.; Nasar, M.; Alkian, I.A.; Hofree, M.; Tikva, S.S.; Rainy, N.; Houri, I.; Cicero, A.; et al. Metabolic Regulation of SARS-CoV-2 Infection; Research Square: San Francisco, CA, USA, 2021. [Google Scholar] [CrossRef]
- Verma, M.; Martin, H.-J.; Haq, W.; Connor, T.R.O.; Maser, E.; Balendiran, G.K. Inhibiting Wild-Type and C299S Mutant AKR1B10; a Homologue of Aldose Reductase Upregulated in Cancers. Eur. J. Pharmacol. 2008, 584, 213–221. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Livshits, M.A.; Khomyakova, E.; Evtushenko, E.G.; Lazarev, V.N.; Kulemin, N.A.; Semina, S.E.; Generozov, E.V.; Govorun, V.M. Isolation of Exosomes by Differential Centrifugation: Theoretical Analysis of a Commonly Used Protocol. Sci. Rep. 2015, 5, 17319. [Google Scholar] [CrossRef]

| Non-ICU | ICU | Adjusted p-Value | |||||
|---|---|---|---|---|---|---|---|
| Mean | ± | SEM | Mean | ± | SEM | ||
| Sex (F/M) | 26/35 | 18/25 | |||||
| Survival (% [Surv-deceased]) | 98.4% [60/1] | 37.2% [16/27] | <0.001 | ||||
| Nb. of comorbidities: | |||||||
| 0 (n (%)) | 39 (63.9%) | 15 (34.9%) | <0.01 | ||||
| 1 (n (%)) | 12 (19.7%) | 16 (37.2%) | <0.05 | ||||
| >=2 (n (%)) | 10 (16.4%) | 12 (27.9%) | 0.16 | ||||
| Nature of comorbidities: | |||||||
| Diabetes (n (%)) | 9 (15%) | 13 (30%) | 0.06 | ||||
| HT (n (%)) | 16 (26%) | 15 (35%) | 0.35 | ||||
| COPD (n (%)) | 2 (3%) | 4 (9%) | 0.20 | ||||
| Renal disease (n (%)) | 1 (2%) | 2 (5%) | 0.37 | ||||
| Cancer (n (%)) | 4 (7%) | 7 (16%) | 0.11 | ||||
| Age (year) | 66.1 | ± | 8.51 | 71.6 | ± | 10.68 | 0.31 |
| BMI (kg/m²) | 26.4 | ± | 3.66 | 26.8 | ± | 4.12 | 0.31 |
| PaO2 (mmHg) | 59.9 | ± | 7.84 | 53.3 | ± | 8.21 | <0.01 |
| CRP (mg/L) | 92.3 | ± | 10.58 | 138.1 | ± | 20.98 | 0.09 |
| CT-Scan (%) | 33.3 | ± | 4.68 | 55.3 | ± | 8.22 | <0.01 |
| Lymphocytes (abs. x10/µL) | 1.57 | ± | 0.17 | 0.86 | ± | 0.13 | <0.001 |
| Fibrinogen (mg/dL) | 570.4 | ± | 73.54 | 588.5 | ± | 87.59 | 0.99 |
| D.Dimer (ng/mL) | 2192 | ± | 286.02 | 3860 | ± | 560.52 | 0.31 |
| Creatinine (mg/dL) | 1.41 | ± | 0.18 | 1.38 | ± | 0.21 | 0.64 |
| Ferritin (ng/mL) | 546.0 | ± | 55.65 | 917.1 | ± | 152.55 | <0.05 |
| LDH (UI/I) | 421.9 | ± | 54.75 | 528.6 | ± | 79.23 | 0.06 |
| Procalcitonin (ng/mL) | 0.32 | ± | 0.04 | 1.01 | ± | 0.63 | 0.31 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chabert, C.; Vitte, A.-L.; Iuso, D.; Chuffart, F.; Trocme, C.; Buisson, M.; Poignard, P.; Lardinois, B.; Debois, R.; Rousseaux, S.; et al. AKR1B10, One of the Triggers of Cytokine Storm in SARS-CoV2 Severe Acute Respiratory Syndrome. Int. J. Mol. Sci. 2022, 23, 1911. https://doi.org/10.3390/ijms23031911
Chabert C, Vitte A-L, Iuso D, Chuffart F, Trocme C, Buisson M, Poignard P, Lardinois B, Debois R, Rousseaux S, et al. AKR1B10, One of the Triggers of Cytokine Storm in SARS-CoV2 Severe Acute Respiratory Syndrome. International Journal of Molecular Sciences. 2022; 23(3):1911. https://doi.org/10.3390/ijms23031911
Chicago/Turabian StyleChabert, Clovis, Anne-Laure Vitte, Domenico Iuso, Florent Chuffart, Candice Trocme, Marlyse Buisson, Pascal Poignard, Benjamin Lardinois, Régis Debois, Sophie Rousseaux, and et al. 2022. "AKR1B10, One of the Triggers of Cytokine Storm in SARS-CoV2 Severe Acute Respiratory Syndrome" International Journal of Molecular Sciences 23, no. 3: 1911. https://doi.org/10.3390/ijms23031911
APA StyleChabert, C., Vitte, A.-L., Iuso, D., Chuffart, F., Trocme, C., Buisson, M., Poignard, P., Lardinois, B., Debois, R., Rousseaux, S., Pepin, J.-L., Martinot, J.-B., & Khochbin, S. (2022). AKR1B10, One of the Triggers of Cytokine Storm in SARS-CoV2 Severe Acute Respiratory Syndrome. International Journal of Molecular Sciences, 23(3), 1911. https://doi.org/10.3390/ijms23031911






