Role of Periostin Expression in Non-Small Cell Lung Cancer: Periostin Silencing Inhibits the Migration and Invasion of Lung Cancer Cells via Regulation of MMP-2 Expression
Abstract
1. Introduction
2. Results
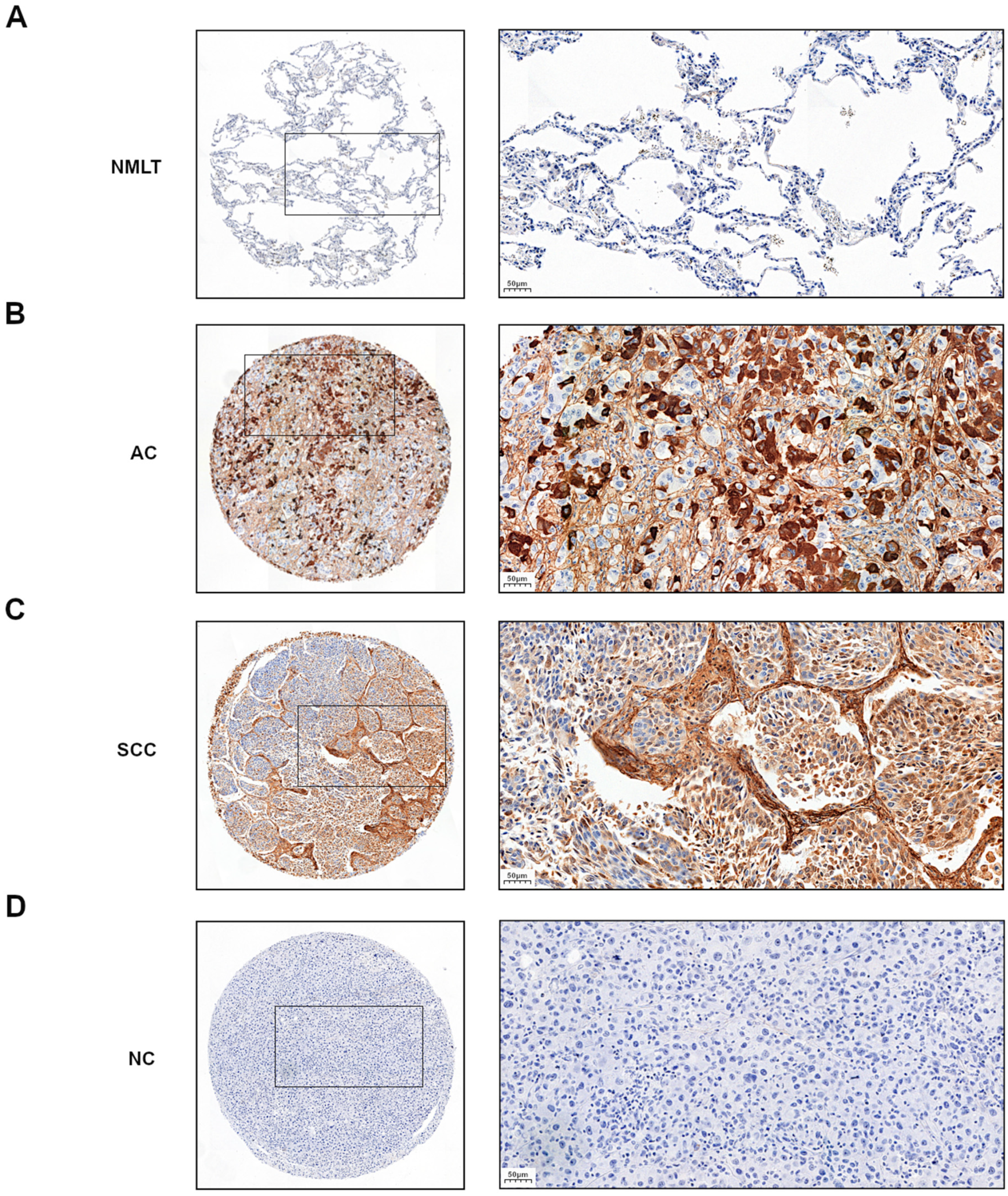
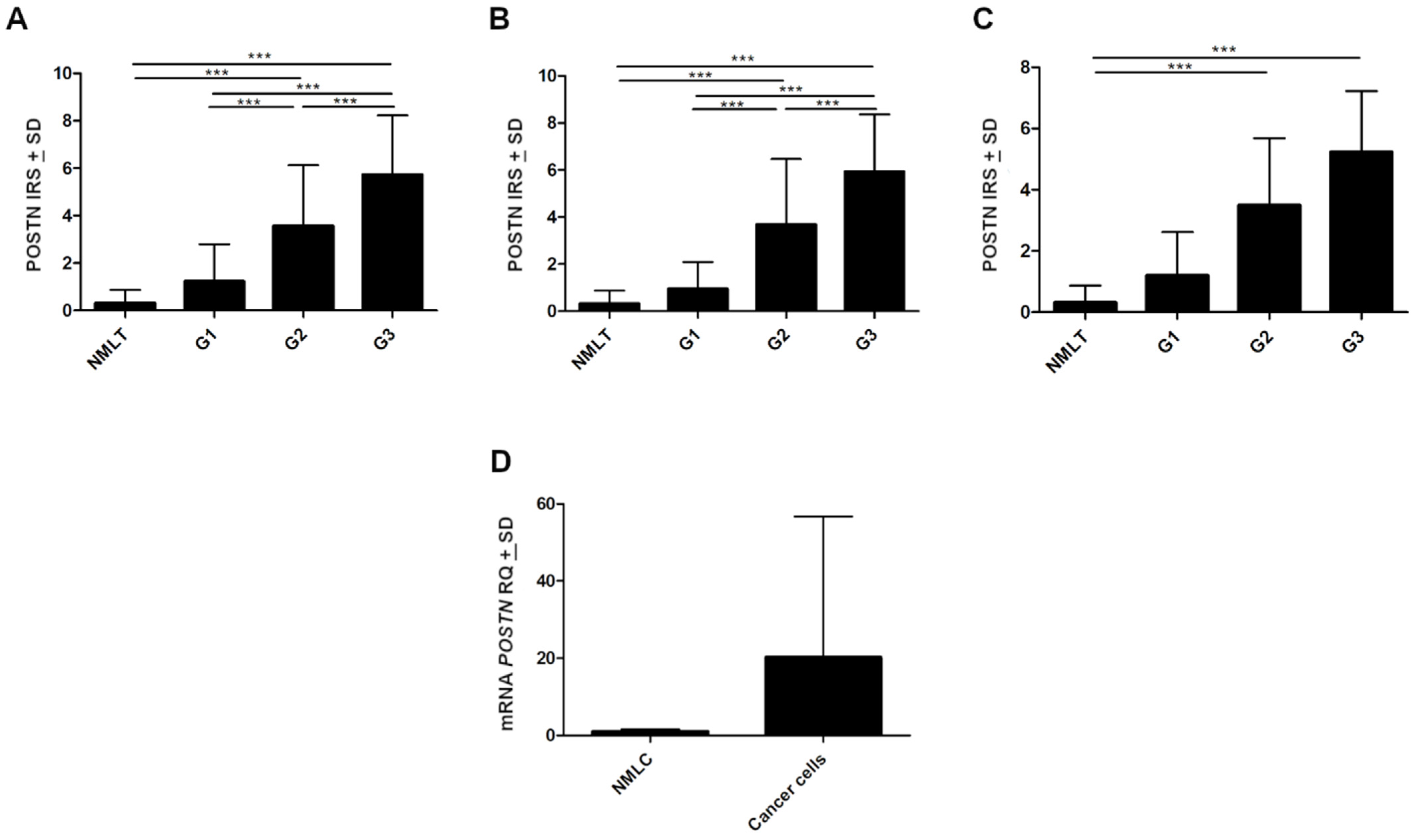
2.1. Examination of POSTN Expression Level and Its Association with Clinicopathological Parameters of Patients
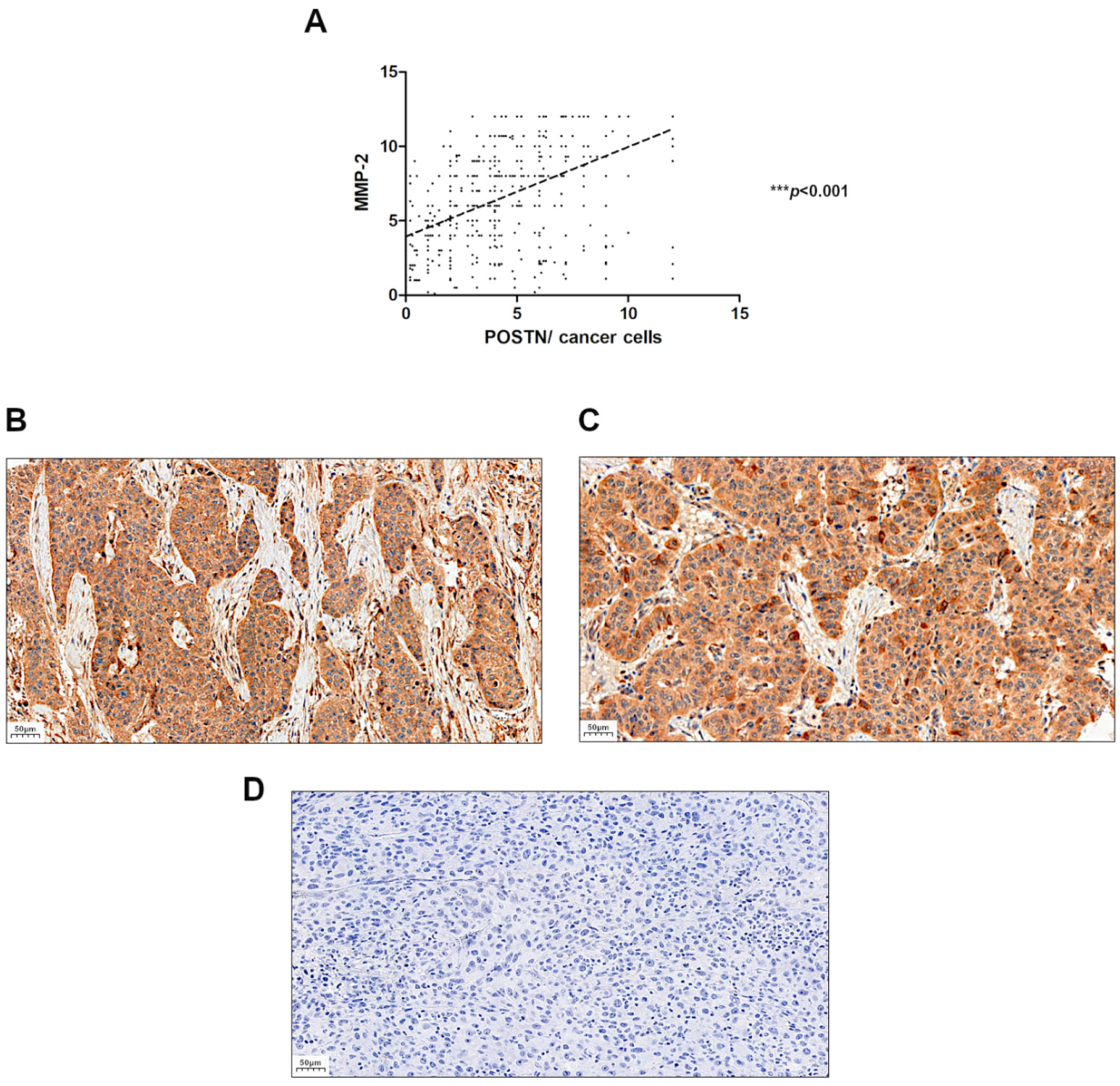
2.2. Comparison of POSTN with MMP-2 Expression Level
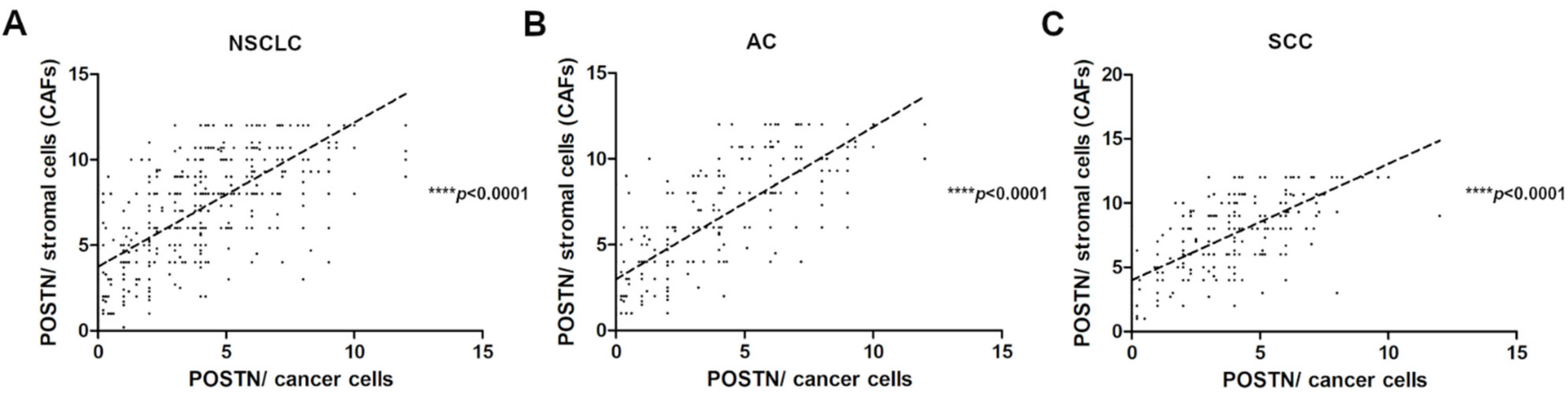
2.3. Associations between Cytoplasmic and Stromal Expression of POSTN in NSCLC
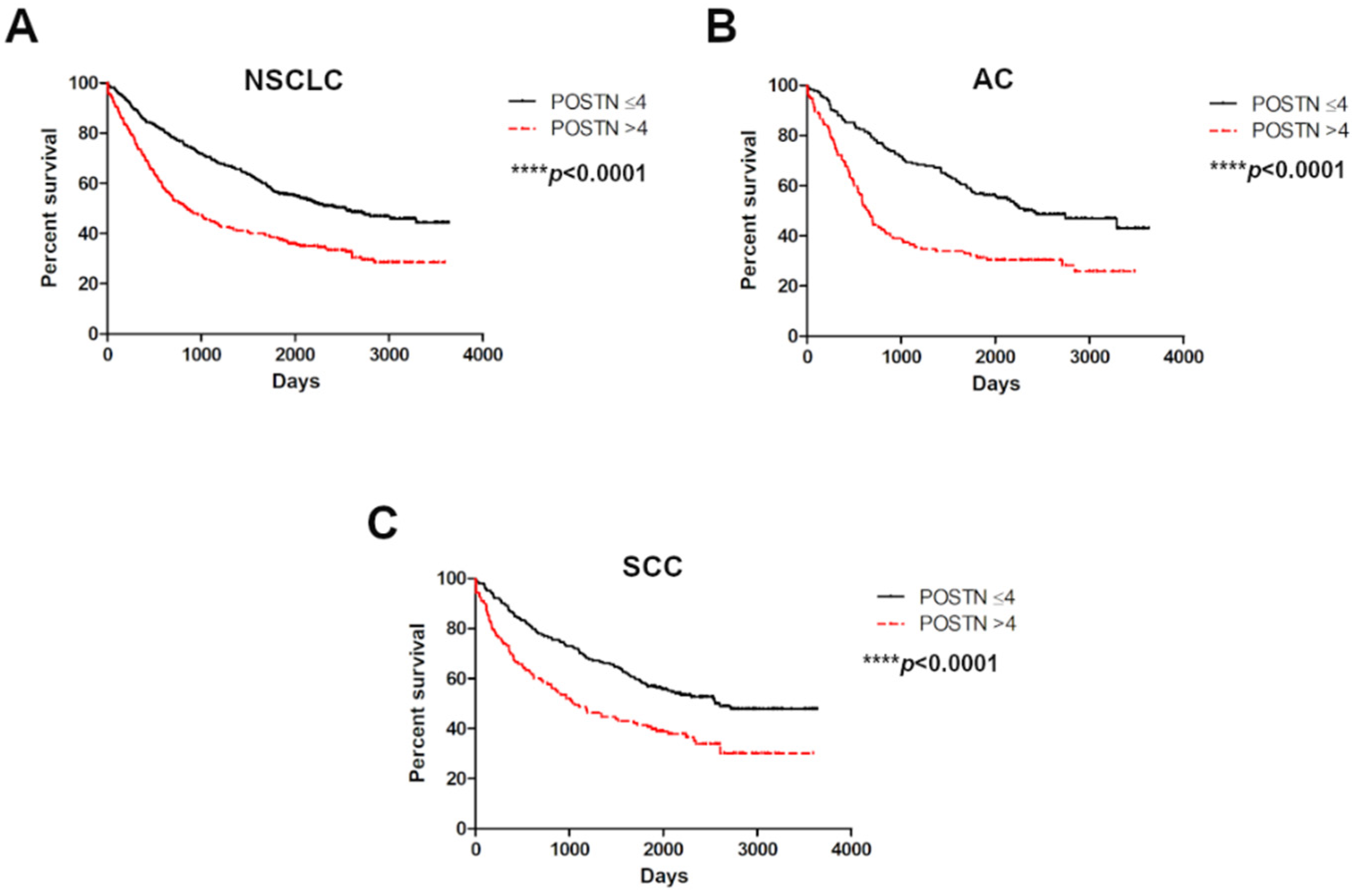
2.4. Survival Analysis
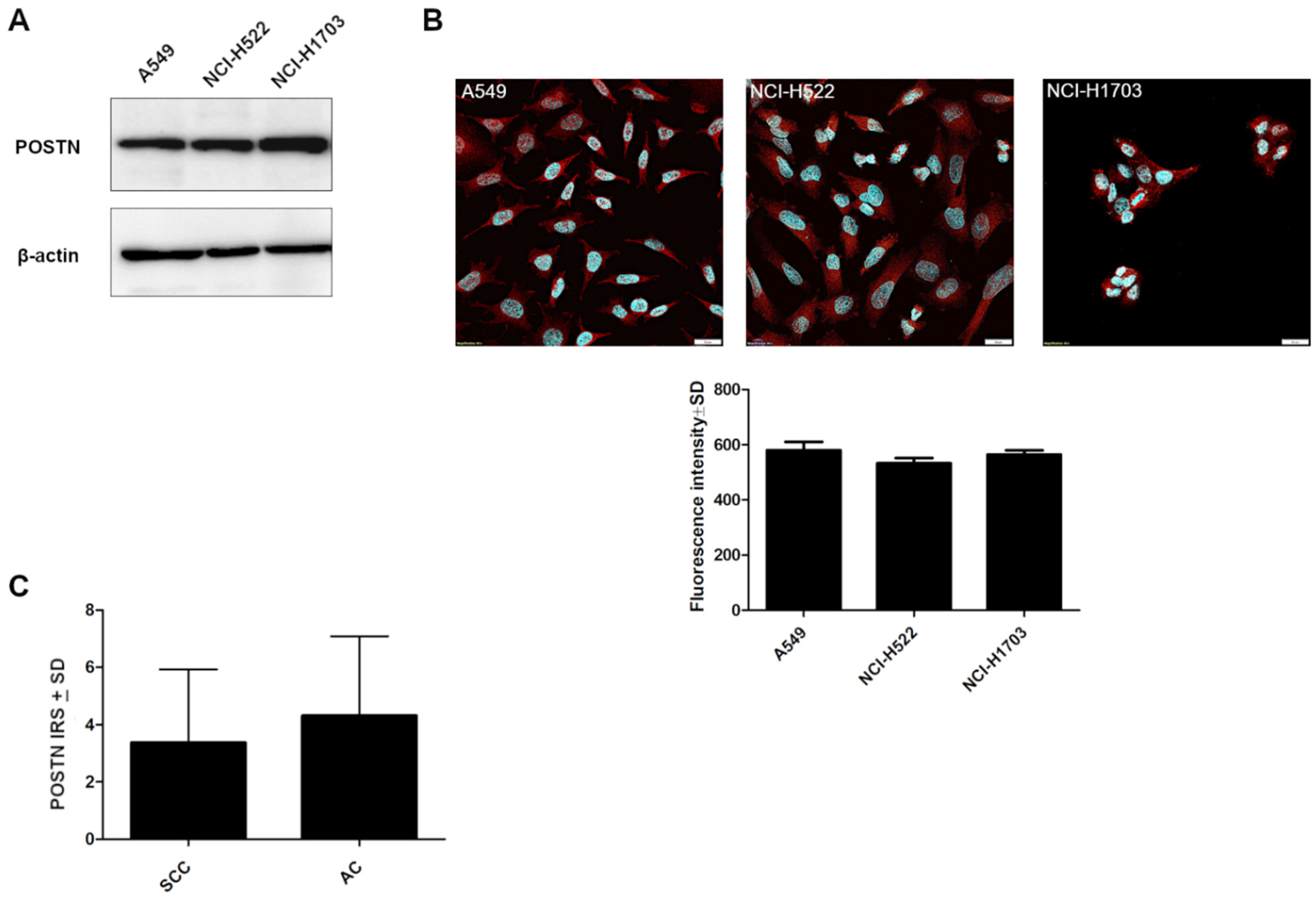
2.5. The Expression of POSTN in Lung Cancer Cell Lines
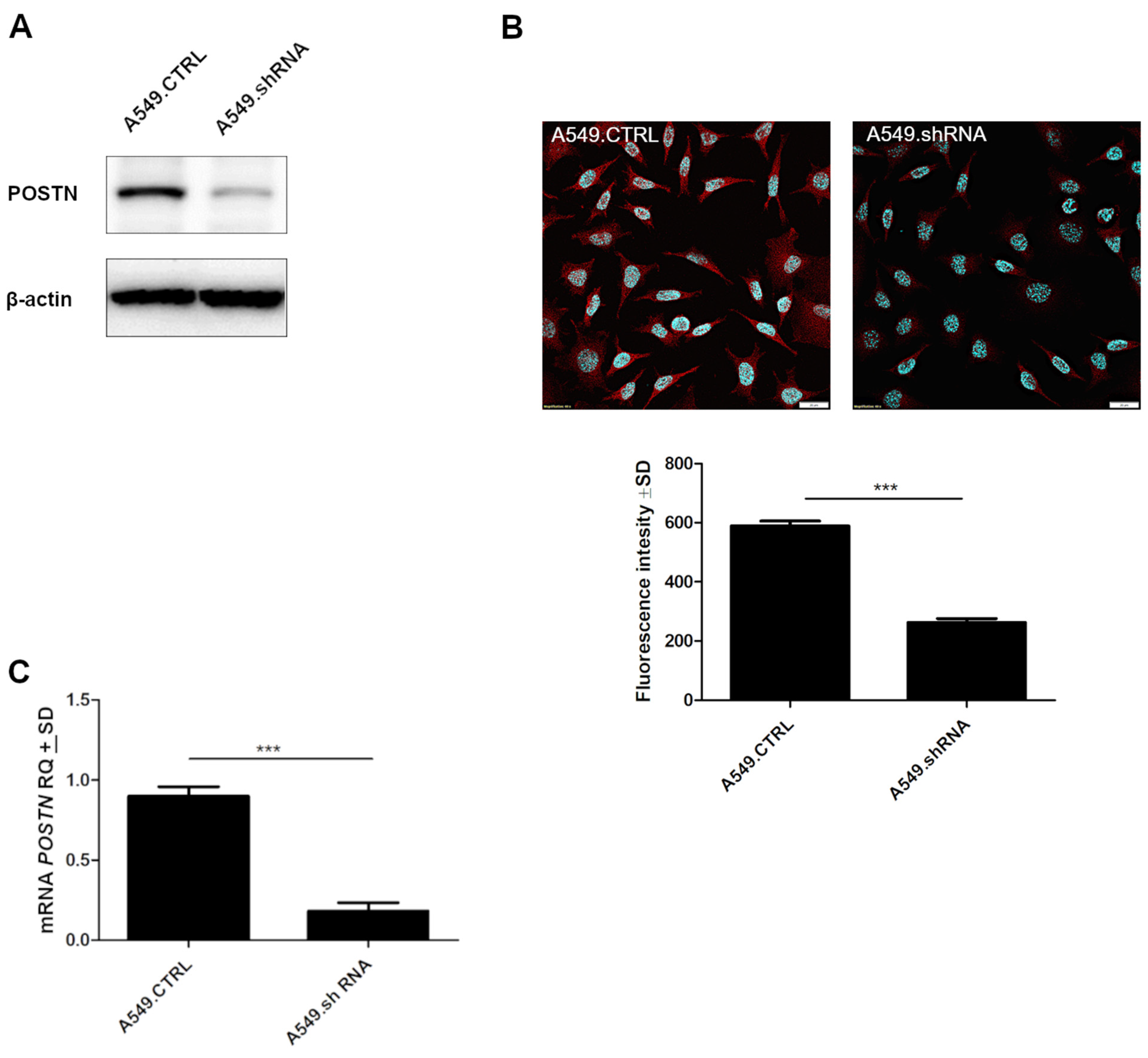
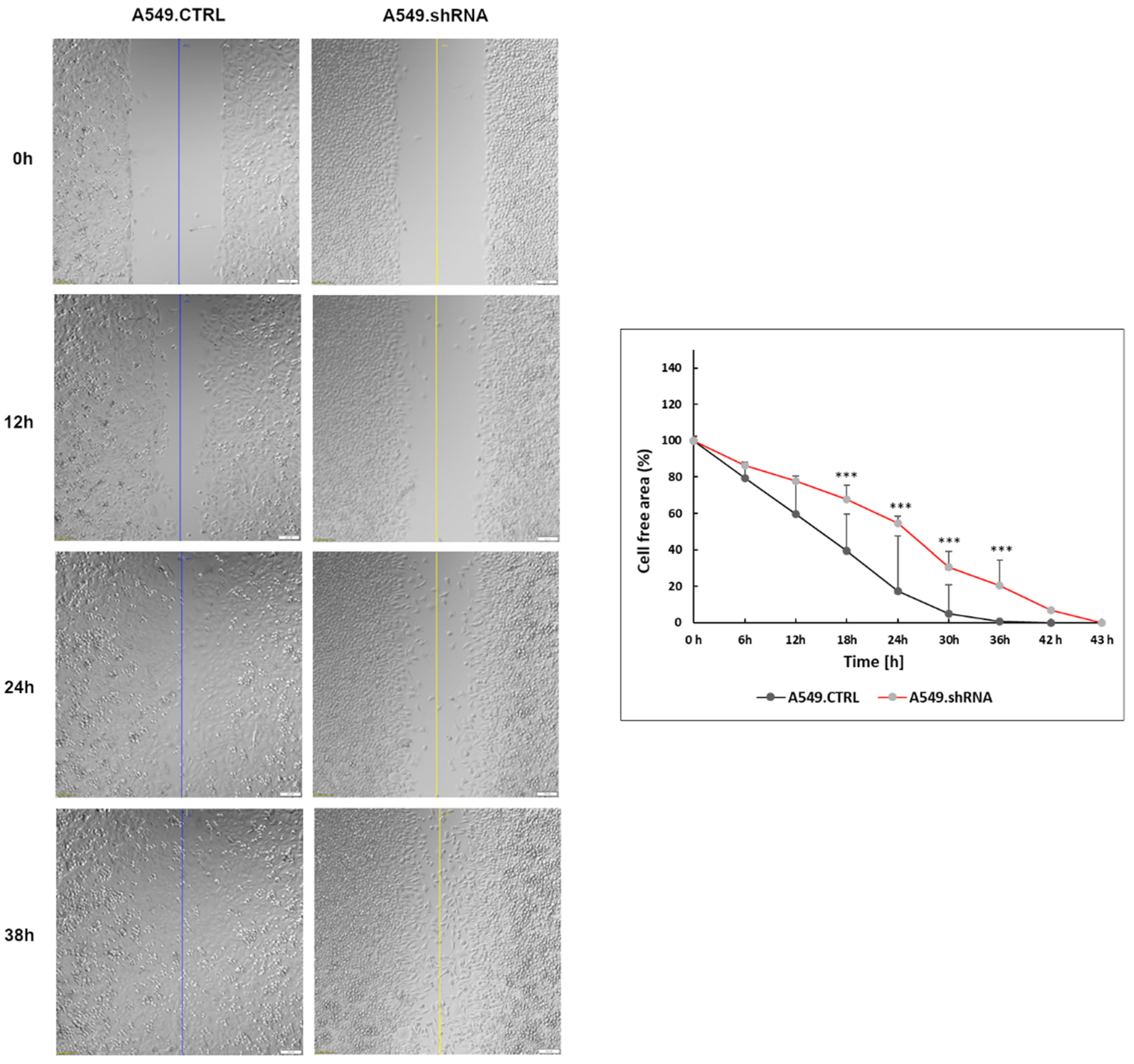
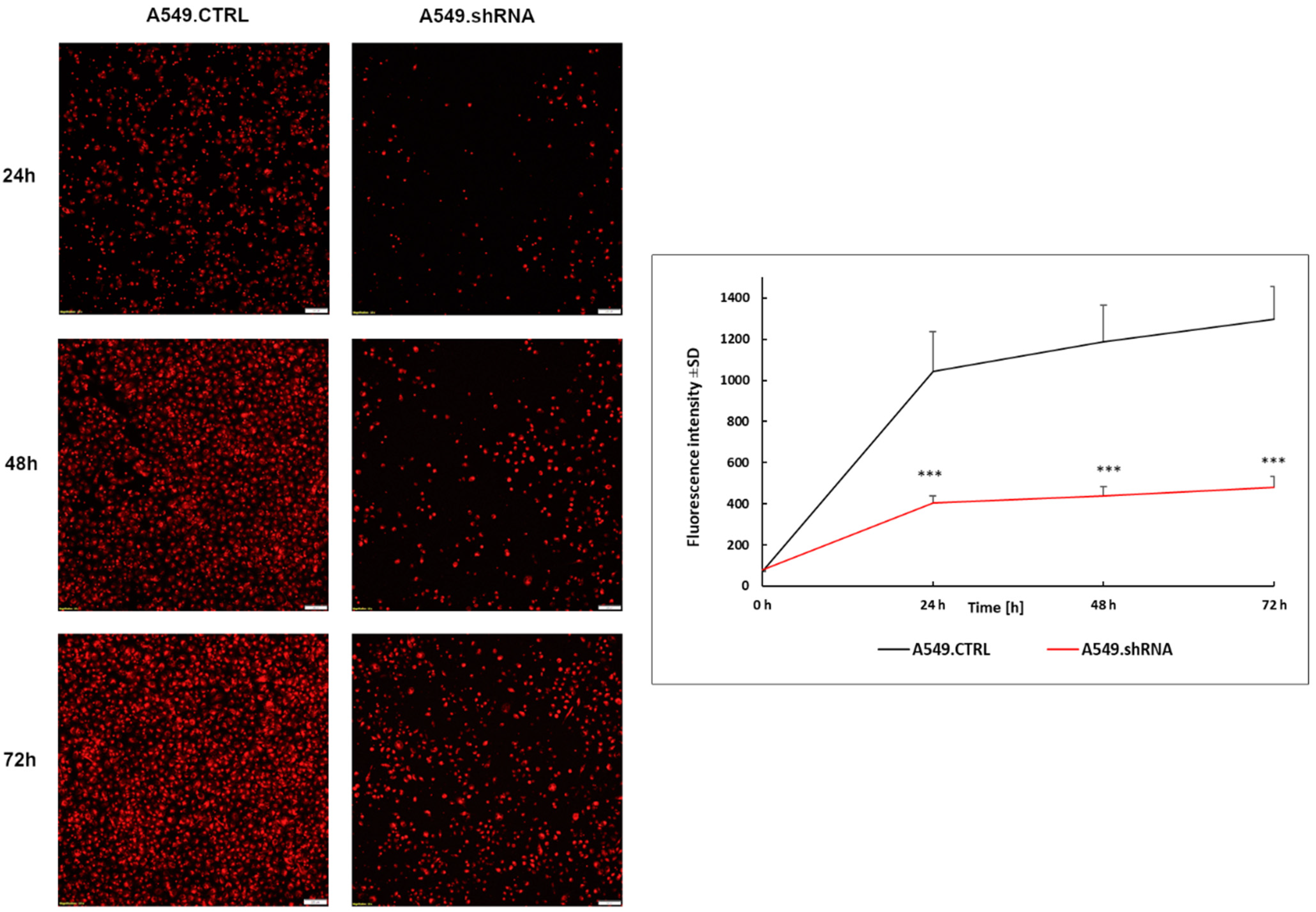
2.6. Effects of POSTN Silencing on A549 Cell Migration and Invasion In Vitro
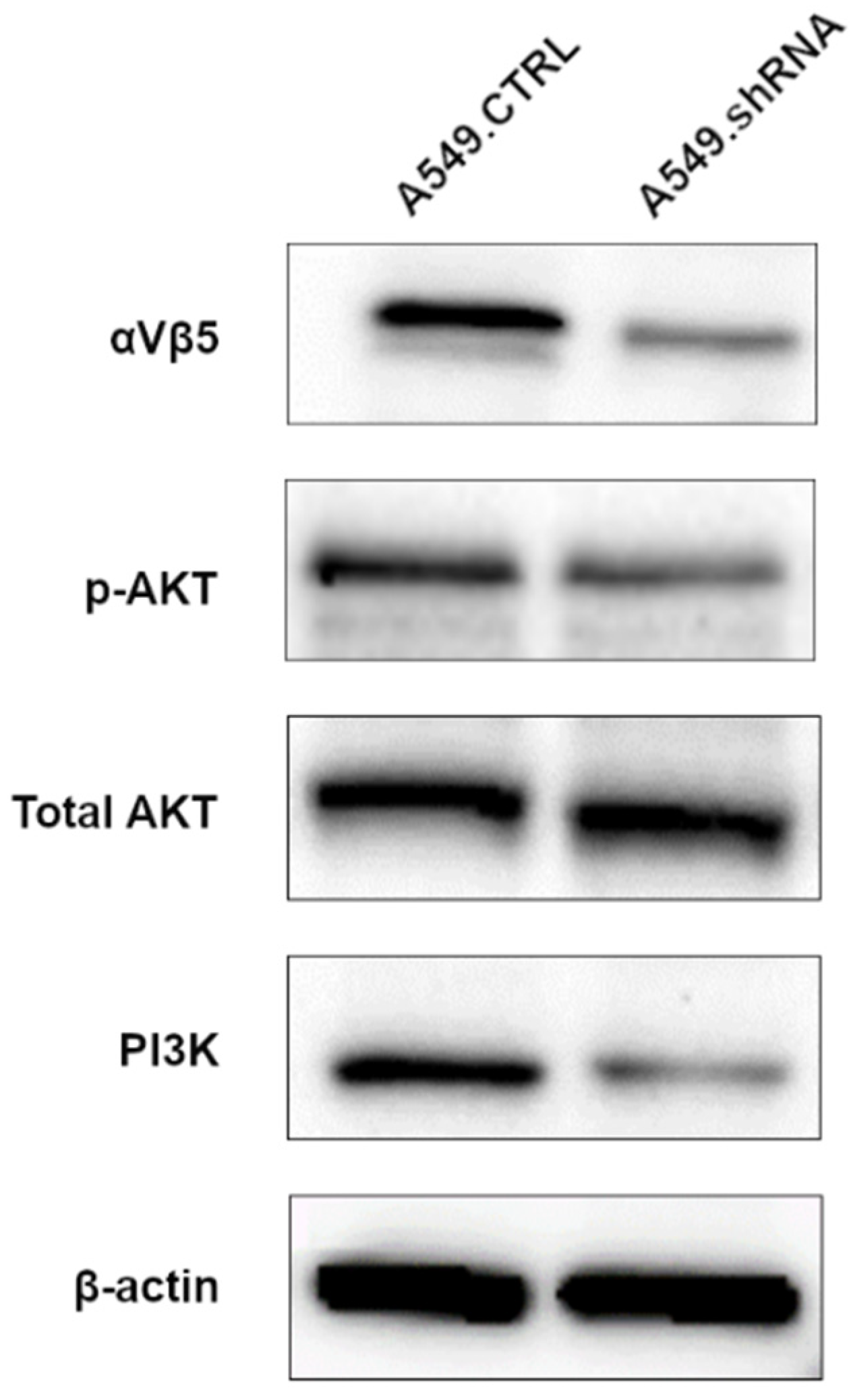
2.7. POSTN Silencing Decreases the Expression of Integrin-Signaling Pathway-Related Protein
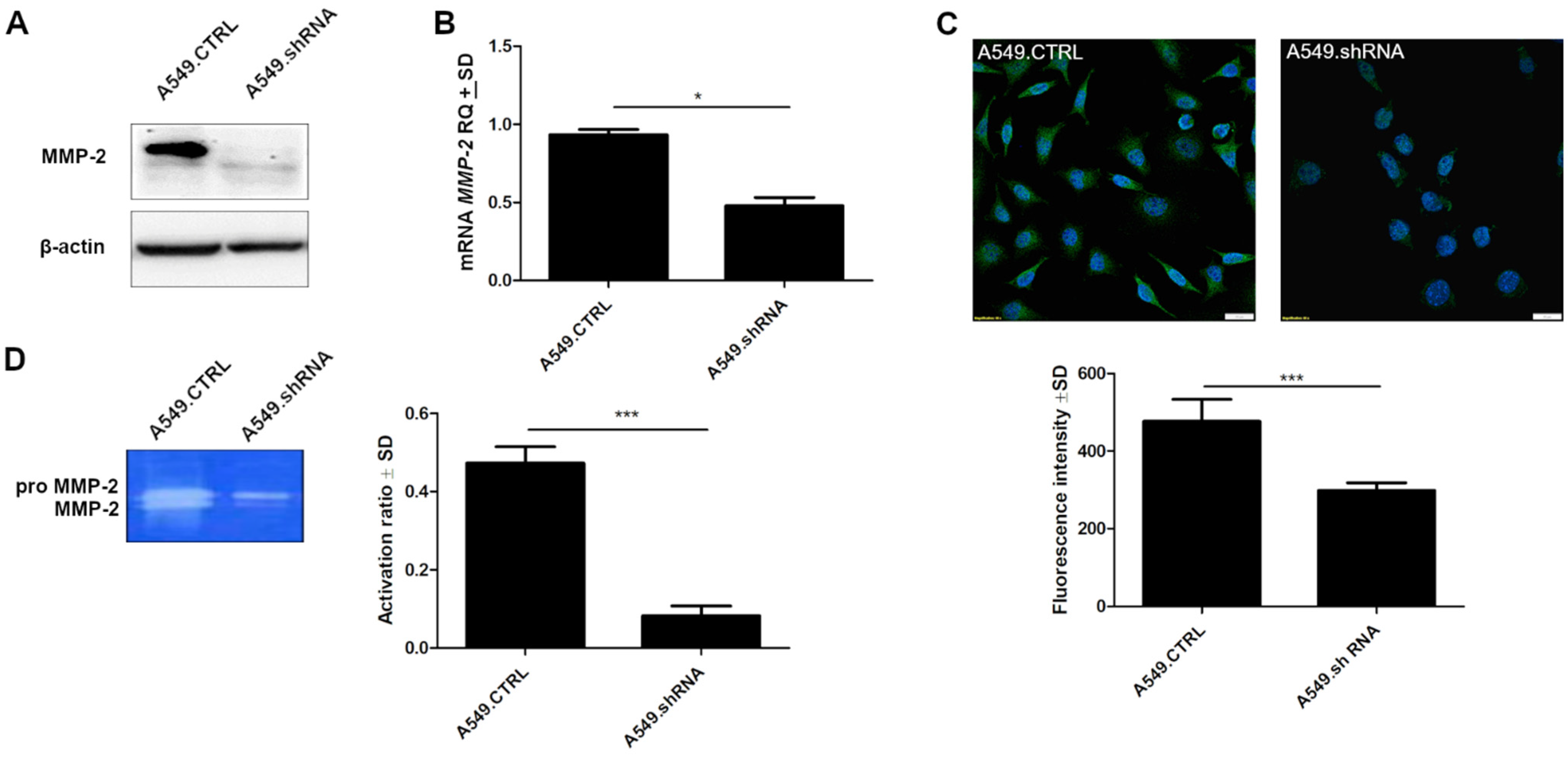
2.8. POSTN Silencing Downregulates the Protein Expression Levels of MMP-2 and MMP-2 Activity
2.9. Effects of Increased POSTN on MMP-2 Expression
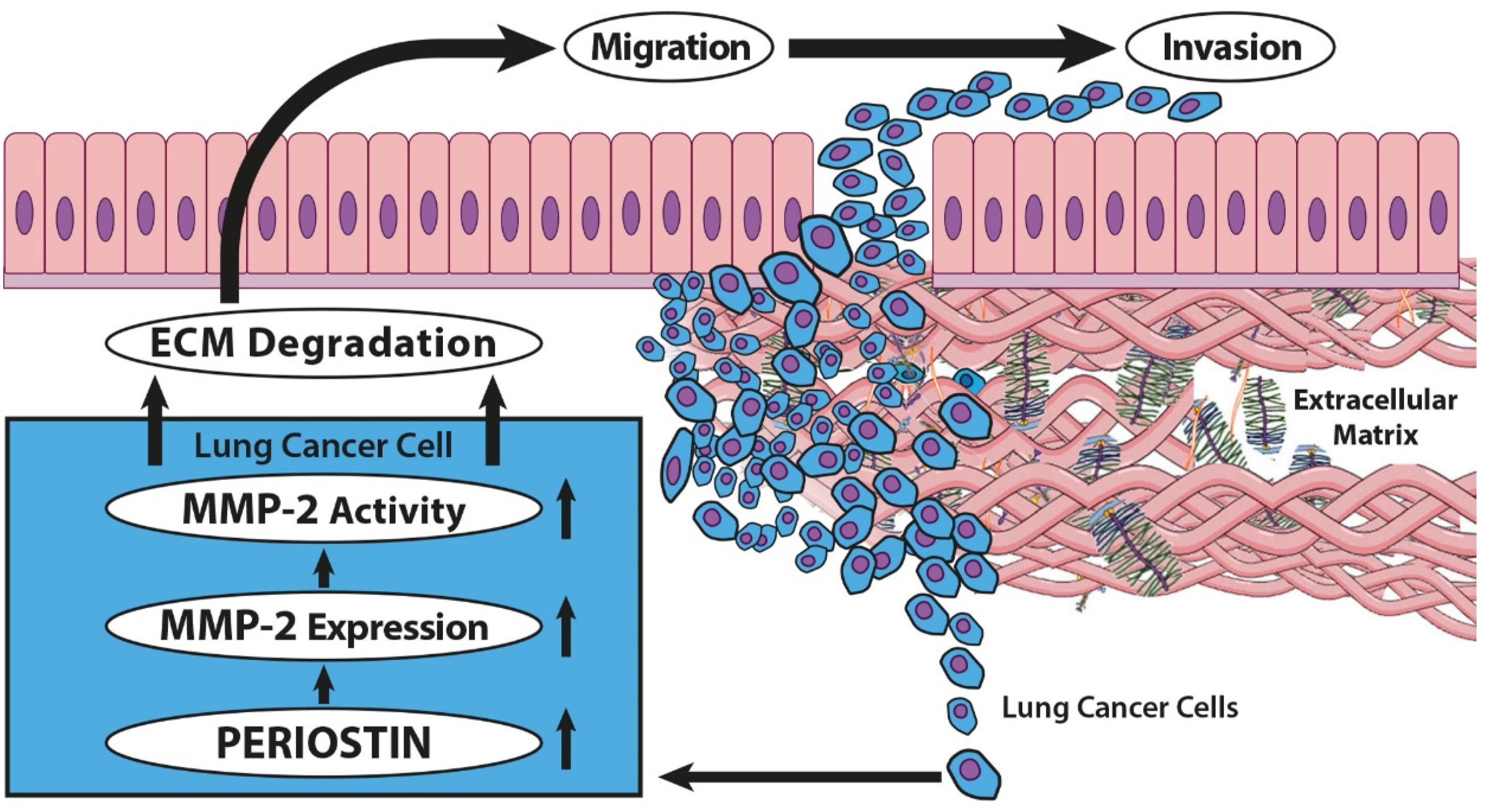
3. Discussion
4. Materials and Methods
4.1. Patient Cohort
4.2. Construction of Tissue Microarray (TMA) and Immunohistochemistry (IHC)
4.3. Laser Capture Microdissection (LCM)
4.4. Reverse Transcription–Quantitative PCR (RT–qPCR)
4.5. Cell Lines
4.6. Virus Production, Transductions and Cell Maintenance
4.7. Protein Isolation, SDS-PAGE, and Western Blotting
4.8. Wound-Healing Assay
4.9. Transwell Invasion Assay
4.10. Immunofluorescence (IF)
4.11. Gelatin Zymography
4.12. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hirsch, F.R.; Scagliotti, G.V.; Mulshine, J.L.; Kwon, R.; Curran, W.J.; Wu, Y.L.; Paz-Ares, L. Lung cancer: Current therapies and new targeted treatments. Lancet 2017, 389, 299–311. [Google Scholar] [CrossRef]
- Wadowska, K.; Bil-Lula, I.; Trembecki, Ł.; Śliwińska-Mossoń, M. Genetic markers in lung cancer diagnosis: A review. Int. J. Mol. Sci. 2020, 21, 4569. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Łaczmańska, I.; Dębicka, I.; Gil, J.; Michałowska, D.; Pawlak, I.; Sąsiadek, M.M. Personalised medicine in lung cancer. Nowotwory 2021, 71, 122–128. [Google Scholar] [CrossRef]
- Cooper, W.A.; Lam, D.C.L.; O’Toole, S.A.; Minna, J.D. Molecular biology of lung cancer. J. Thorac. Dis. 2013, 5. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, H.; Pan, T.; Li, L.; Li, J.; Yang, H. STIM1 silencing inhibits the migration and invasion of A549 cells. Mol. Med. Rep. 2017, 16, 3283–3289. [Google Scholar] [CrossRef]
- Han, L.; Sheng, B.; Zeng, Q.; Yao, W.; Jiang, Q. Correlation between MMP2 expression in lung cancer tissues and clinical parameters: A retrospective clinical analysis. BMC Pulm. Med. 2020, 20, 1–9. [Google Scholar] [CrossRef]
- Hadler-Olsen, E.; Winberg, J.O.; Uhlin-Hansen, L. Matrix metalloproteinases in cancer: Their value as diagnostic and prognostic markers and therapeutic targets. Tumor Biol. 2013, 34, 2041–2051. [Google Scholar] [CrossRef]
- Kii, I.; Nishiyama, T.; Li, M.; Matsumoto, K.; Saito, M.; Amizuka, N.; Kudo, A. Incorporation of Tenascin-C into the Extracellular Matrix by Periostin Underlies an Extracellular Meshwork Architecture. J. Biol. Chem. 2010, 285, 2028–2039. [Google Scholar] [CrossRef]
- Hoersch, S.; Andrade-Navarro, M.A. Periostin shows increased evolutionary plasticity in its alternatively spliced region. BMC Evol. Biol. 2010, 10, 1–19. [Google Scholar] [CrossRef]
- González-González, L.; Alonso, J. Periostin: A matricellular protein with multiple functions in cancer development and progression. Front. Oncol. 2018, 8, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Norris, R.A.; Damon, B.; Mironov, V.; Kasyanov, V.; Ramamurthi, A.; Moreno-Rodriguez, R.; Trusk, T.; Potts, J.D.; Goodwin, R.L.; Davis, J.; et al. Periostin regulates collagen fibrillogenesis and the biomechanical properties of connective tissues. J. Cell. Biochem. 2007, 101, 695–711. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, D.W. Functional role of periostin in development and wound repair: Implications for connective tissue disease. J. Cell Commun. Signal. 2008, 2, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Ruan, K.; Bao, S.; Ouyang, G. The multifaceted role of periostin in tumorigenesis. Cell. Mol. Life Sci. 2009, 66, 2219–2230. [Google Scholar] [CrossRef]
- Zhou, Y.; Yang, W.; Ao, M.; Höti, N.; Gabrielson, E.; Chan, D.W.; Zhang, H.; Li, Q.K. Proteomic Analysis of the Air-Way Fluid in Lung Cancer. Detection of Periostin in Bronchoalveolar Lavage (BAL). Front. Oncol. 2020, 10, 1072. [Google Scholar] [CrossRef]
- Che, J.; Shen, W.Z.; Deng, Y.; Dai, Y.H.; Liao, Y.-D.; Yuan, X.L.; Zhang, P. Effects of lentivirus-mediated silencing of Periostin on tumor microenvironment and bone metastasis via the integrin-signaling pathway in lung cancer. Life Sci. 2017, 182, 10–21. [Google Scholar] [CrossRef]
- Morra, L.; Moch, H. Periostin expression and epithelial-mesenchymal transition in cancer: A review and an update. Virchows Arch. 2011, 459, 465–475. [Google Scholar] [CrossRef]
- Murakami, D.; Takamori, S.; Kawahara, A.; Mitsuoka, M.; Kashihara, M.; Yoshiyama, K.; Matsumoto, R.; Yokoyama, S.; Fujimoto, K.; Kawaguchi, A.; et al. Periostin expression in non-small cell lung cancer: Clinical significance. Kurume Med. J. 2017, 64, 13–20. [Google Scholar] [CrossRef]
- Bao, S.; Ouyang, G.; Bai, X.; Huang, Z.; Ma, C.; Liu, M.; Shao, R.; Anderson, R.M.; Rich, J.N.; Wang, X.F. Periostin potently promotes metastatic growth of colon cancer by augmenting cell survival via the Akt/PKB pathway. Cancer Cell 2004, 5, 329–339. [Google Scholar] [CrossRef]
- Shao, R.; Bao, S.; Bai, X.; Blanchette, C.; Anderson, R.M.; Dang, T.; Gishizky, M.L.; Marks, J.R.; Wang, X.-F. Acquired Expression of Periostin by Human Breast Cancers Promotes Tumor Angiogenesis through Up-Regulation of Vascular Endothelial Growth Factor Receptor 2 Expression. Mol. Cell. Biol. 2004, 24, 3992–4003. [Google Scholar] [CrossRef]
- Kim, C.J.; Yoshioka, N.; Tambe, Y.; Kushima, R.; Okada, Y.; Inoue, H. Periostin is down-regulated in high grade human bladder cancers and suppresses in vitro cell invasiveness and in vivo metastasis of cancer cells. Int. J. Cancer 2005, 117, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Kanno, A.; Satoh, K.; Masamune, A.; Hirota, M.; Kimura, K.; Umino, J.; Hamada, S.; Satoh, A.; Egawa, S.; Motoi, F.; et al. Periostin, secreted from stromal cells, has biphasic effect on cell migration and correlates with the epithelial to mesenchymal transition of human pancreatic cancer cells. Int. J. Cancer 2008, 122, 2707–2718. [Google Scholar] [CrossRef] [PubMed]
- Ye, D.; Zhou, C.; Wang, S.; Deng, H.; Shen, Z. Tumor suppression effect of targeting periostin with siRNA in a nude mouse model of human laryngeal squamous cell carcinoma. J. Clin. Lab. Anal. 2019, 33, e22622. [Google Scholar] [CrossRef] [PubMed]
- Ratajczak-Wielgomas, K.; Kmiecik, A.; Grzegrzołka, J.; Piotrowska, A.; Gomulkiewicz, A.; Partynska, A.; Pawelczyk, K.; Nowinska, K.; Podhorska-Okolow, M.; Dziegiel, P. Prognostic significance of stromal periostin expression in non-small cell lung cancer. Int. J. Mol. Sci. 2020, 21, 7025. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, Z.; Cui, D.; Ouyang, G. The Multiaspect Functions of Periostin in Tumor Progression. In Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2019; Volume 1132, pp. 125–136. [Google Scholar]
- Hong, L.; Sun, H.; Lv, X.; Yang, D.; Zhang, J.; Shi, Y. Expression of periostin in the serum of NSCLC and its function on proliferation and migration of human lung adenocarcinoma cell line (A549) in vitro. Mol. Biol. Rep. 2010, 37, 2285–2293. [Google Scholar] [CrossRef]
- Ratajczak-Wielgomas, K.; Grzegrzolka, J.; Piotrowska, A.; Gomulkiewicz, A.; Witkiewicz, W.; Dziegiel, P. Periostin expression in cancer-associated fibroblasts of invasive ductal breast carcinoma. Oncol. Rep. 2016, 36, 2745–2754. [Google Scholar] [CrossRef]
- Baril, P.; Gangeswaran, R.; Mahon, P.C.; Caulee, K.; Kocher, H.M.; Harada, T.; Zhu, M.; Kalthoff, H.; Crnogorac-Jurcevic, T.; Lemoine, N.R. Periostin promotes invasiveness and resistance of pancreatic cancer cells to hypoxia-induced cell death: Role of the β4 integrin and the PI3k pathway. Oncogene 2007, 26, 2082–2094. [Google Scholar] [CrossRef]
- Gillan, L.; Matei, D.; Fishman, D.A.; Gerbin, C.S.; Karlan, B.Y.; Chang, D.D. Periostin Secreted by Epithelial Ovarian Carcinoma Is a Ligand for V 3 and V 5 Integrins and Promotes Cell Motility. Cancer Res. 2002, 62, 5358–5364. [Google Scholar]
- Yoshioka, N.; Fuji, S.; Shimakage, M.; Kodama, K.; Hakura, A.; Yutsudo, M.; Inoue, H.; Nojima, H. Suppression of anchorage-independent growth of human cancer cell lines by the TRIF52/periostin/OSF-2 gene. Exp. Cell Res. 2002, 279, 91–99. [Google Scholar] [CrossRef]
- Sun, C.; Zhao, X.; Xu, K.; Gong, J.; Liu, W.; Ding, W.; Gou, Y.; Xia, G.; Ding, Q. Periostin: A promising target of therapeutical intervention for prostate cancer. J. Transl. Med. 2011, 9, 1–10. [Google Scholar] [CrossRef]
- Blanco-Prieto, S.; Barcia-Castro, L.; Páez de la Cadena, M.; Rodríguez-Berrocal, F.J.; Vázquez-Iglesias, L.; Botana-Rial, M.I.; Fernández-Villar, A.; De Chiara, L. Relevance of matrix metalloproteases in non-small cell lung cancer diagnosis. BMC Cancer 2017, 17, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chuanyu, S.; Yuqing, Z.; Chong, X.; Guowei, X.; Xiaojun, Z. Periostin promotes migration and invasion of renal cell carcinoma through the integrin/focal adhesion kinase/c-Jun N-terminal kinase pathway. Tumor Biol. 2017, 39, 1010428317694549. [Google Scholar] [CrossRef]
- Egeblad, M.; Werb, Z. New functions for the matrix metalloproteinases in cancer progression. Nat. Rev. Cancer 2002, 2, 161–174. [Google Scholar] [CrossRef] [PubMed]
- Ratajczak-Wielgomas, K.; Grzegrzolka, J.; Piotrowska, A.; Matkowski, R.; Wojnar, A.; Rys, J.; Ugorski, M.; Dziegiel, P. Expression of periostin in breast cancer cells. Int. J. Oncol. 2017, 51, 1300–1310. [Google Scholar] [CrossRef]
- Liu, A.Y.; Zheng, H.; Ouyang, G. Periostin, a multifunctional matricellular protein in inflammatory and tumor microenvironments. Matrix Biol. 2014, 37, 150–156. [Google Scholar] [CrossRef]
- Kudo, Y.; Siriwardena, B.S.M.S.; Hatano, H.; Ogawa, I.; Takata, T. Periostin: Novel diagnostic and therapeutic target for cancer. Histol. Histopathol. 2007, 22, 1167–1174. [Google Scholar] [CrossRef]
- Orecchia, P.; Conte, R.; Balza, E.; Castellani, P.; Borsi, L.; Zardi, L.; Mingari, M.C.; Carnemolla, B. Identification of a novel cell binding site of periostin involved in tumour growth. Eur. J. Cancer 2011, 47, 2221–2229. [Google Scholar] [CrossRef]
- Soltermann, A.; Tischler, V.; Arbogast, S.; Braun, J.; Probst-Hensch, N.; Weder, W.; Moch, H.; Kristiansen, G. Prognostic Significance of Epithelial-Mesenchymal and Mesenchymal-Epithelial Transition Protein Expression in Non-Small Cell Lung Cancer. Clin. Cancer Res. 2008, 14, 7430–7437. [Google Scholar] [CrossRef]
- Zhu, M.; Fejzo, M.S.; Anderson, L.; Dering, J.; Ginther, C.; Ramos, L.; Gasson, J.C.; Karlan, B.Y.; Slamon, D.J. Periostin promotes ovarian cancer angiogenesis and metastasis. Gynecol. Oncol. 2010, 119, 337–344. [Google Scholar] [CrossRef]
- Morra, L.; Rechsteiner, M.; Casagrande, S.; Duc Luu, V.; Santimaria, R.; Diener, P.A.; Sulser, T.; Kristiansen, G.; Schraml, P.; Moch, H.; et al. Relevance of periostin splice variants in renal cell carcinoma. Am. J. Pathol. 2011, 179, 1513–1521. [Google Scholar] [CrossRef]
- Ben, Q.W.; Jin, X.L.; Jun, L.; Cai, X.; Yuan, F.; Yuan, Y.Z. Periostin, a matrix specific protein, is associated with proliferation and invasion of pancreatic cancer. Oncol. Rep. 2011, 25, 709–716. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Riener, M.-O.; Fritzsche, F.R.; Soll, C.; Pestalozzi, B.C.; Probst-Hensch, N.; Clavien, P.-A.; Jochum, W.; Soltermann, A.; Moch, H.; Kristiansen, G. Expression of the extracellular matrix protein periostin in liver tumours and bile duct carcinomas. Histopathology 2010, 56, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Sun, Q.K.; He, Y.F.; Ma, D.C.; Xie, M.R.; Ji, C.S.; Hu, B. Overexpression of periostin is significantly correlated to the tumor angiogenesis and poor prognosis in patients with esophageal squamous cell carcinoma. Int. J. Clin. Exp. Pathol. 2014, 7, 593–601. [Google Scholar] [PubMed]
- Wang, H.; Wang, Y.; Jiang, C. Stromal protein periostin identified as a progression associated and prognostic biomarker in glioma via inducing an invasive and proliferative phenotype. Int. J. Oncol. 2013, 42, 1716–1724. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shih, Y.W.; Chien, S.T.; Chen, P.S.; Lee, J.H.; Wu, S.H.; Yin, L. Te α-Mangostin Suppresses Phorbol 12-myristate 13-acetate-induced MMP-2/MMP-9 Expressions via αvβ3 Integrin/FAK/ERK and NF-κB Signaling Pathway in Human Lung Adenocarcinoma A549 Cells. Cell Biochem. Biophys. 2010, 58, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Yasue, A.; Fujihara, S.; Tanaka, E. PERIOSTIN regulates MMP-2 expression via the αvβ3 integrin/ERK pathway in human periodontal ligament cells. Arch. Oral Biol. 2012, 57, 52–59. [Google Scholar] [CrossRef]
- Yan, W.; Shao, R. Transduction of a mesenchyme-specific gene periostin into 293T cells induces cell invasive activity through epithelial-mesenchymal transformation. J. Biol. Chem. 2006, 281, 19700–19709. [Google Scholar] [CrossRef]
- Burgess, J.K.; Jonker, M.R.; Berg, M.; Hacken, N.T.H.; Meyer, K.B.; van den Berge, M.; Nawijn, M.C.; Heijink, I.H. Early View Original article Periostin: Contributor to abnormal airway epithelial function in asthma? Eur. Respir. J. 2021, 57, 2001286. [Google Scholar] [CrossRef]
- Ouanouki, A.; Lamy, S.; Annabi, B. Periostin, a signal transduction intermediate in TGF-β-induced EMT in U-87MG human glioblastoma cells, and its inhibition by anthocyanidins. Oncotarget 2018, 9, 22023–22037. [Google Scholar] [CrossRef]
- Travis, W.D.; Brambilla, E.; Nicholson, A.G.; Yatabe, Y.; Austin, J.H.M.; Beasley, M.B.; Chirieac, L.R.; Dacic, S.; Duhig, E.; Flieder, D.B.; et al. The 2015 World Health Organization Classification of Lung Tumors: Impact of Genetic, Clinical and Radiologic Advances since the 2004 Classification. J. Thorac. Oncol. 2015, 10, 1243–1260. [Google Scholar] [CrossRef]
- Detterbeck, F.C.; Boffa, D.J.; Kim, A.W.; Tanoue, L.T. The Eighth Edition Lung Cancer Stage Classification. Chest 2017, 151, 193–203. [Google Scholar] [CrossRef]
- Remmele, W. Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue. Pathologe 1987, 8, 138–140. [Google Scholar] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Ratajczak-Wielgomas, K.; Gosk, J.; Rabczyński, J.; Augoff, K.; Podhorska-Okołów, M.; Gamian, A.; Rutowski, R. Expression of MMP-2, TIMP-2, TGF-β1, and Decorin in Dupuytren’s Contracture. Connect. Tissue Res. 2012, 53, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Dean, D.; Hornicek, F.J.; Chen, Z.; Duan, Z. The role of extracelluar matrix in osteosarcoma progression and metastasis. J. Exp. Clin. Cancer Res. 2020, 39, 1–11. [Google Scholar] [CrossRef] [PubMed]


| (A) | ||||||||
| Overall Survival (OS) | ||||||||
| Clinicopathological Parameters | NSCLC | |||||||
| Univariate Analysis | Multivariate Analysis | |||||||
| p-Value | HR (95% HR CI) | p-Value | HR (95% HR CI) | |||||
| Age (<62 vs. >62) | 0.0099 | 1.2915 (1.0635–1.5684) | 0.0011 | 1.3867 (1.1389–1.6885) | ||||
| POSTN cancer cells (low vs. high) | <0.0001 | 1.8496 (1.5203–2.2502) | <0.0001 | 1.5401 (1.2595–1.8831) | ||||
| Ki-67 (median) | 0.5862 | 0.9456 (0.7729–1.1567) | - | - | ||||
| TTF-1 | 0.7634 | 0.9908 (0.9333–1.052) | - | - | ||||
| p63 | 0.0938 | 0.9517 (0.898–1.0084) | - | - | ||||
| Smoking history | 0.2842 | 1.01 (0.9918–1.0286) | - | - | ||||
| Stage (I–II vs. III–IV) | <0.0001 | 2.1399 (1.7488–2.6185) | 0.1108 | 1.2661 (0.9474–1.692) | ||||
| Grade (G1 vs. G2-G3) | 0.7886 | 1.0539 (0.718–1.5471) | - | - | ||||
| Tumour size (T1-T2 vs. T3-T4) | <0.0001 | 1.9758 (1.6174–2.4136) | 0.0001 | 1.6143 (1.2715–2.0495) | ||||
| Lymph nodes involvement (N0 vs. N+) | <0.0001 | 1.8288 (1.5009–2.2284) | 0.0004 | 1.5767 (1.223–2.0327) | ||||
| pM | 0.6951 | 1.3087 (0.3408–5.0262) | - | - | ||||
| (B) | ||||||||
| Overall Survival (OS) | ||||||||
| Clinicopathological Parameters | SCC | AC | ||||||
| Univariate analysis | Multivariate analysis | Univariate analysis | Multivariate analysis | |||||
| p-Value | HR (95% HR CI) | p-Value | HR (95% HR CI) | p-Value | HR (95% HR CI) | p-Value | HR (95% HR CI) | |
| Age (<62 vs. >62) | 0.0463 | 1.3453 (1.0049–1.801) | 0.0279 | 1.3912 (1.0365–1.8673) | 0.1792 | 1.2257 (0.9108–1.6493) | - | - |
| POSTN cancer cells (low vs. high) | 0.0001 | 1.827 (1.3593–2.4556) | 0.0045 | 1.5597 (1.1476–2.1196) | <0.0001 | 2.0868 (1.548–2.8132) | 0.0025 | 1.6236 (1.1858–2.223) |
| Ki-67 (median) | 0.5593 | 0.915 (0.679–1.233) | - | - | 0.7894 | 0.9599 (0.7109–1.2962) | - | - |
| TTF-1 | 0.2344 | 1.0831 (0.9496–1.2353) | - | - | 0.0469 | 0.8994 (0.8102–0.9986) | 0.2284 | 0.9345 (0.8369–1.0434) |
| p63 | 0.0675 | 0.8935 (0.792–1.0081) | - | - | 0.7377 | 0.978 (0.8589–1.1138) | - | - |
| Smoking history | 0.3124 | 1.01 (0.9907–1.0298) | - | - | 0.0662 | 1.4783 (0.9742–2.2431) | - | - |
| Stage (I vs. II–IV) | 0.0001 | 1.9025 (1.394–2.5965) | 0.3043 | 1.2635 (0.8087–1.9741) | <0.0001 | 2.3709 (1.751–3.2103) | 0.3528 | 1.2374 (0.7896–1.9392) |
| Grade (G1 vs. G2-G3) | 0.2417 | 0.6367 (0.299–1.3557) | - | - | 0.2646 | 1.3118 (0.8144–2.113) | - | - |
| Tumour size (T1-T2 vs. T3-T4) | <0.0001 | 1.8701 (1.3894–2.517) | 0.0221 | 1.5217 (1.0623–2.1798) | <0.0001 | 2.0099 (1.4645–2.7583) | 0.0362 | 1.4849 (1.0258–2.1496) |
| Lymph nodes involvement (N0 vs. N+) | 0.0039 | 1.5464 (1.1504–2.0788) | 0.1079 | 1.3633 (0.9343–1.9892) | <0.0001 | 2.3189 (1.7176–3.1306) | 0.0026 | 1.8544 (1.2414–2.7701) |
| pM | - | - | - | - | 0.7036 | 1.3302 (0.3058–5.7855) | - | - |
| Characteristics | NSCLC | AC | SCC | LCC | ||||
|---|---|---|---|---|---|---|---|---|
| N = 715 | % | N = 298 | % | N = 370 | % | N = 47 | % | |
| Age | ||||||||
| ≤62 | 36 | 5.03% | 21 | 7.05% | 10 | 2.70% | 26 | 55.32% |
| >62 | 679 | 94.97% | 277 | 92.95% | 360 | 97.30% | 21 | 44.68% |
| Tumor size | ||||||||
| pT1 | 184 | 25.73% | 74 | 24.83% | 93 | 25.14% | 14 | 29.79% |
| pT2 | 313 | 43.78% | 145 | 48.66% | 155 | 41.89% | 18 | 38.30% |
| pT3 | 149 | 20.84% | 52 | 17.45% | 85 | 22.97% | 9 | 19.15% |
| pT4 | 69 | 9.65% | 27 | 9.06% | 37 | 10.00% | 6 | 12.77% |
| Tumor grade | ||||||||
| G1 | 52 | 7.27% | 37 | 12.42% | 12 | 3.24% | 2 | 4.26% |
| G2 | 565 | 79.02% | 205 | 68.79% | 301 | 81.35% | 39 | 82.98% |
| G3 | 98 | 13.71% | 56 | 18.79% | 57 | 15.41% | 6 | 12.77% |
| Lymph node involvement | ||||||||
| pN0 | 471 | 65.87% | 193 | 64.77% | 240 | 64.86% | 34 | 72.34% |
| pN1 | 131 | 18.32% | 43 | 14.43% | 84 | 22.70% | 4 | 8.51% |
| pN2 | 113 | 15.80% | 62 | 20.81% | 46 | 12.43% | 9 | 19.15% |
| Stage | ||||||||
| I | 262 | 36.64% | 114 | 38.26% | 131 | 35.41% | 15 | 31.91% |
| II | 249 | 34.83% | 88 | 29.53% | 140 | 37.84% | 16 | 34.04% |
| III | 198 | 27.69% | 91 | 30.54% | 98 | 26.49% | 15 | 31.91% |
| IV | 6 | 0.84% | 5 | 1.68% | 1 | 0.27% | 1 | 2.13% |
| Smoking status | ||||||||
| Neg. | 104 | 14.55% | 52 | 17.45% | 38 | 10.27% | 4 | 8.51% |
| Pos. | 611 | 85.45% | 246 | 82.55% | 332 | 89.73% | 43 | 91.49% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ratajczak-Wielgomas, K.; Kmiecik, A.; Dziegiel, P. Role of Periostin Expression in Non-Small Cell Lung Cancer: Periostin Silencing Inhibits the Migration and Invasion of Lung Cancer Cells via Regulation of MMP-2 Expression. Int. J. Mol. Sci. 2022, 23, 1240. https://doi.org/10.3390/ijms23031240
Ratajczak-Wielgomas K, Kmiecik A, Dziegiel P. Role of Periostin Expression in Non-Small Cell Lung Cancer: Periostin Silencing Inhibits the Migration and Invasion of Lung Cancer Cells via Regulation of MMP-2 Expression. International Journal of Molecular Sciences. 2022; 23(3):1240. https://doi.org/10.3390/ijms23031240
Chicago/Turabian StyleRatajczak-Wielgomas, Katarzyna, Alicja Kmiecik, and Piotr Dziegiel. 2022. "Role of Periostin Expression in Non-Small Cell Lung Cancer: Periostin Silencing Inhibits the Migration and Invasion of Lung Cancer Cells via Regulation of MMP-2 Expression" International Journal of Molecular Sciences 23, no. 3: 1240. https://doi.org/10.3390/ijms23031240
APA StyleRatajczak-Wielgomas, K., Kmiecik, A., & Dziegiel, P. (2022). Role of Periostin Expression in Non-Small Cell Lung Cancer: Periostin Silencing Inhibits the Migration and Invasion of Lung Cancer Cells via Regulation of MMP-2 Expression. International Journal of Molecular Sciences, 23(3), 1240. https://doi.org/10.3390/ijms23031240






