Human Genetic Diseases Linked to the Absence of NEMO: An Obligatory Somatic Mosaic Disorder in Male
Abstract
:1. Introduction
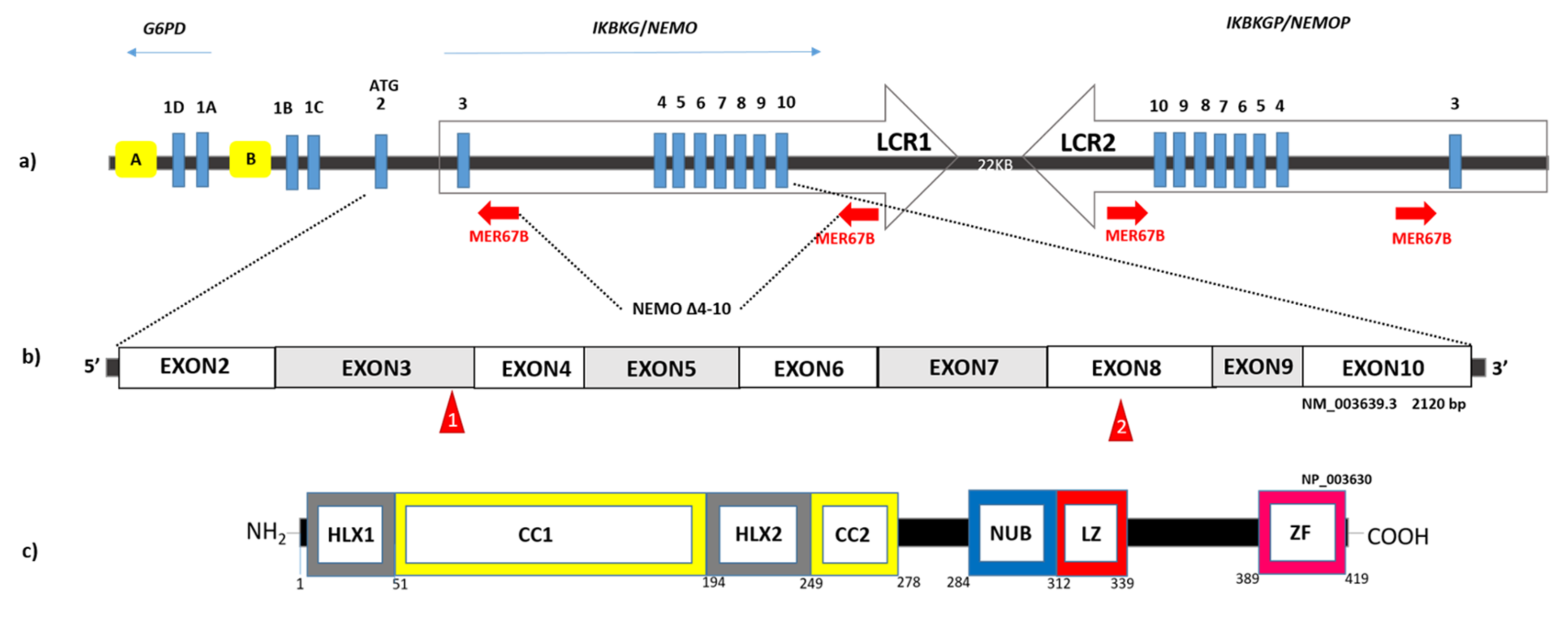
2. What Distinguishes an EDA-ID Male from an IP Male?
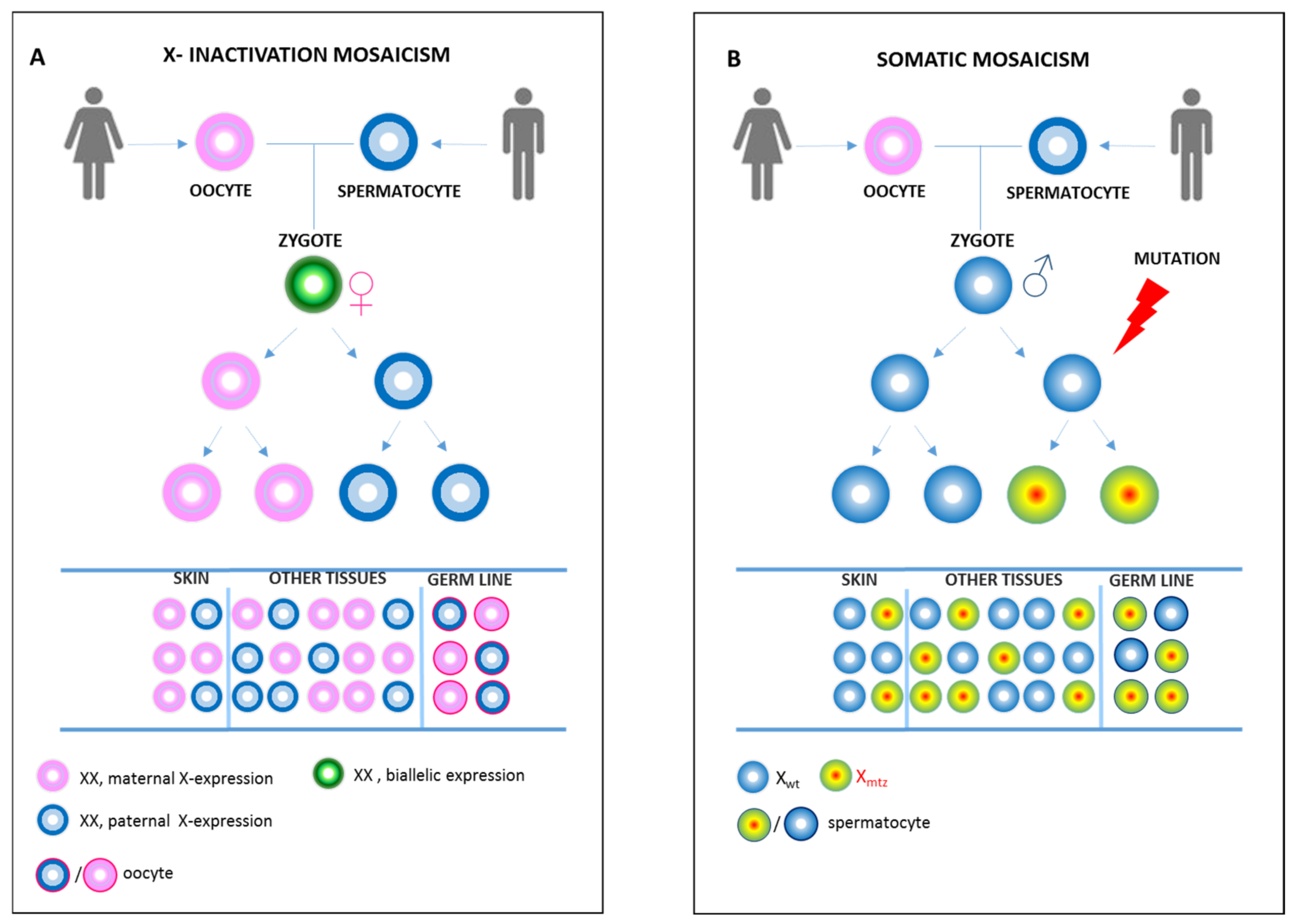
3. Phenotype Associated to a Mosaic NEMO Deficiency: Differences and Similarities between Mosaic IP Males and IP Females
4. Pitfall in Demonstrating Mosaicism in IP Males
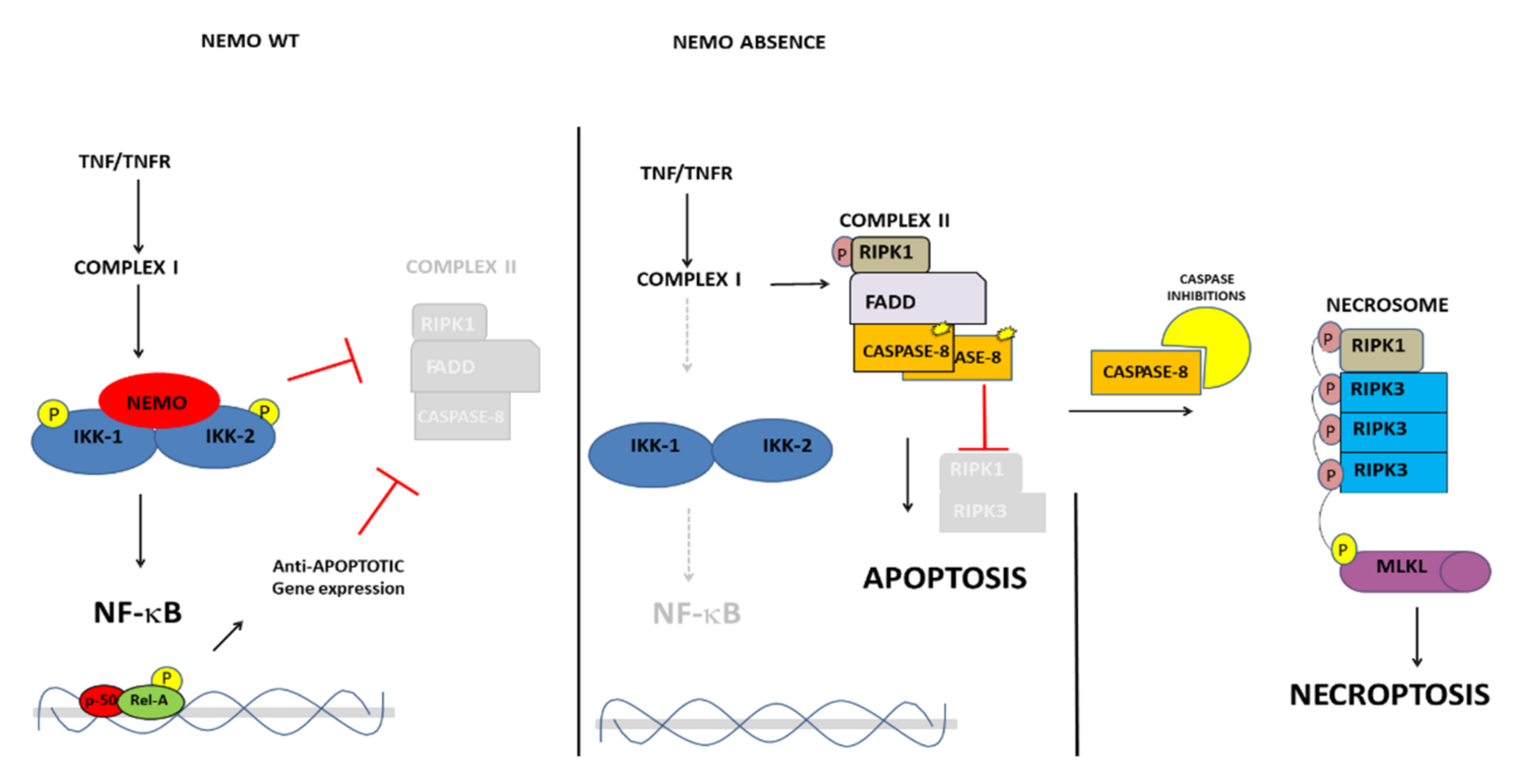
5. NEMO Deficiency and Cell Death: Implications for the IP-Phenotype
6. Final Remarks on the Genetics of IP
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Forsberg, L.A.; Gisselsson, D.; Dumanski, J.P. Mosaicism in health and disease—Clones picking up speed. Nat. Rev. Genet. 2017, 18, 128–142. [Google Scholar] [CrossRef] [PubMed]
- Aduna-Hidalgo, R.; Bo, T.; Kwint, M.P.; van de Vorst, M.; Pinelli, M.; Veltman, J.A.; Hoischen, A.; Vissers, L.E.; Gilissen, C. Post-zygotic Point Mutations Are an Underrecognized Source of De Novo Genomic Variation. Am. J. Hum. Genet. 2015, 97, 67–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lyon, M.F. Gene action in the X-chromosome of the mouse (Mus musculus L.). Nature 1961, 190, 372–373. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, I.M.; Laven, J.S.; Stevens, M.; Jonkers, I.; Galjaard, R.J.; Gribnau, J.; van Doorninck, J.H. X chromosome inactivation is initiated in human preimplantation embryos. Am. J. Hum. Genet. 2009, 84, 771–779. [Google Scholar] [CrossRef] [Green Version]
- Courtois, G.; Gilmore, T.D. Mutations in the NF-kappaB signaling pathway: Implications for human disease. Oncogene 2006, 25, 6831–6843. [Google Scholar] [CrossRef] [Green Version]
- Courtois, G.; Pescatore, A.; Gautheron, J.; Fusco, F.; Ursini, M.V.; Senegas, A. NF-kB-Related Genetic Diseases, 1st ed.; Springer: Cham, Switzerland, 2006; ISBN 978-3-319-25848-5. [Google Scholar]
- Zonana, J.; Elder, M.E.; Schneider, L.C.; Orlow, S.J.; Moss, C.; Golabi, M.; Shapira, S.K.; Farndon, P.A.; Wara, D.W.; Emmal, S.A.; et al. A novel X-linked disorder of immune deficiency and hypohidrotic ectodermal dysplasia is allelic to incontinentia pigmenti and due to mutations in IKK-gamma (NEMO). Am. J. Hum. Genet. 2000, 67, 1555–1562. [Google Scholar] [CrossRef] [Green Version]
- Döffinger, R.; Smahi, A.; Bessia, C.; Geissmann, F.; Feinberg, J.; Durandy, A.; Bodemer, C.; Kenwrick, S.; Dupuis-Girod, S.; Blanche, S.; et al. X-linked anhidrotic ectodermal dysplasia with immunodeficiency is caused by impaired NF-kappaB signaling. Nat. Genet. 2001, 27, 277–285. [Google Scholar] [CrossRef]
- Smahi, A.; Courtois, G.; Vabres, P.; Yamaoka, S.; Heuertz, S.; Munnich, A.; Israël, A.; Heiss, N.S.; Klauck, S.M.; Kioschis, P.; et al. Genomic rearrangement in NEMO impairs NF-kappaB activation and is a cause of incontinentia pigmenti. The International Incontinentia Pigmenti (IP) Consortium. Nature 2000, 405, 466–472. [Google Scholar] [CrossRef]
- Hadj-Rabia, S.; Froidevaux, D.; Bodak, N.; Hamel-Teillac, D.; Smahi, A.; Touil, Y.; Fraitag, S.; de Prost, Y.; Bodemer, C. Clinical study of 40 cases of incontinentia pigmenti. Arch. Dermatol. 2003, 139, 1163–1170. [Google Scholar] [CrossRef] [Green Version]
- Fusco, F.; Bardaro, T.; Fimiani, G.; Mercadante, V.; Miano, M.G.; Falco, G.; Israël, A.; Courtois, G.; D’Urso, M.; Ursini, M.V. Molecular analysis of the genetic defect in a large cohort of IP patients and identification of novel NEMO mutations interfering with NF-kappaB activation. Hum. Mol. Genet. 2004, 13, 1763–1773. [Google Scholar] [CrossRef]
- Fusco, F.; Paciolla, M.; Conte, M.I.; Pescatore, A.; Esposito, E.; Mirabelli, P.; Lioi, M.B.; Ursini, M.V. Incontinentia pigmenti: Report on data from 2000 to 2013. Orphanet J. Rare Dis. 2014, 9, 93. [Google Scholar] [CrossRef] [Green Version]
- Bastard, P.; Rosen, L.B.; Zhang, Q.; Michailidis, E.; Hoffmann, H.H.; Zhang, Y.; Dorgham, K.; Philippot, Q.; Rosain, J.; Béziat, V.; et al. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science 2020, 370, 4585. [Google Scholar] [CrossRef]
- Puel, A.; Picard, C.; Ku, C.L.; Smahi, A.; Casanova, J.L. Inherited disorders of NF-kappaB-mediated immunity in man. Curr. Opin. Immunol. 2004, 16, 34–41. [Google Scholar] [CrossRef]
- Fusco, F.; Pescatore, A.; Conte, M.I.; Mirabelli, P.; Paciolla, M.; Esposito, E.; Lioi, M.B.; Ursini, M.V. EDA-ID and IP, two faces of the same coin: How the same IKBKG/NEMO mutation affecting the NF-κB pathway can cause immunodeficiency and/or inflammation. Int. Rev. Immunol. 2015, 34, 445–459. [Google Scholar] [CrossRef]
- Kenwrick, S.; Woffendin, H.; Jakins, T.; Shuttleworth, S.G.; Mayer, E.; Greenhalgh, L.; Whittaker, J.; Rugolotto, S.; Bardaro, T.; Esposito, T.; et al. International IP Consortium. Survival of male patients with incontinentia pigmenti carrying a lethal mutation can be explained by somatic mosaicism or Klinefelter syndrome. Am. J. Hum. Genet. 2001, 69, 1210–1217. [Google Scholar] [CrossRef] [Green Version]
- Kunze, J.; Frenzel, U.H.; Hüttig, E.; Grosse, F.R.; Wiedemann, H.R. Klinefelter’s syndrome and Incontinentia pigmenti Bloch-Sulzberger. Hum. Genet. 1977, 35, 237–240. [Google Scholar] [CrossRef]
- Ormerod, A.D.; White, M.I.; McKay, E.; Johnston, A.W. Incontinentia pigmenti in a boy with Klinefelter’s syndrome. J. Med. Genet. 1987, 24, 439–444. [Google Scholar] [CrossRef]
- Prendiville, J.S.; Gorski, J.L.; Stein, C.K.; Esterly, N.B. Incontinentia pigmenti in a male infant with Klinefelter syndrome. Am. Acad. Dermatol. 1989, 20, 937–940. [Google Scholar] [CrossRef]
- García-Dorado, J.; de Unamuno, P.; Fernández-López, E.; Salazar Veloz, J.; Armijo, M. Incontinentia pigmenti: XXY male with a family history. Clin. Genet. 1990, 38, 128–138. [Google Scholar] [CrossRef]
- Buinauskaite, E.; Buinauskiene, J.; Kucinskiene, V.; Strazdiene, D.; Valiukeviciene, S. Incontinentia pigmenti in a male infant with Klinefelter syndrome: A case report and review of the literature. Pediatr. Dermatol. 2010, 27, 492–495. [Google Scholar] [CrossRef]
- Williams, A.; Chandrashekar, L.; Srivastava, V.M.; Thomas, M.; Horo, S.; George, R. Incontinentia pigmenti, an x-linked dominant disorder, in a 2-year-old boy with Klinefelter syndrome. Indian J. Pathol. Microbiol. 2017, 60, 424–426. [Google Scholar] [CrossRef]
- Moro, R.; Fabiano, A.; Calzavara-Pinton, P.; Cardinale, J.; Palumbo, G.; Giliani, S.; Lanzi, G.; Antonelli, F.; De Simone, M.; Martelli, P.; et al. Incontinentia Pigmenti Associated with Aplasia Cutis Congenita in a Newborn Male with Klinefelter Syndrome: Is the Severity of Neurological Involvement Linked to Skin Manifestations? Dermatol. Ther. 2020, 10, 213–220. [Google Scholar] [CrossRef] [Green Version]
- Pacheco, T.R.; Levy, M.; Collyer, J.C.; de Parra, N.P.; Parra, C.A.; Garay, M.; Aprea, G.; Moreno, S.; Mancini, A.J.; Paller, A.S. Incontinentia pigmenti in male patients. J. Am. Acad. Dermatol. 2006, 55, 251–255. [Google Scholar] [CrossRef]
- Fusco, F.; Fimiani, G.; Tadini, G.; Michele, D.; Ursini, M.V. Clinical diagnosis of incontinentia pigmenti in a cohort of male patients. J. Am. Acad. Dermatol. 2007, 56, 264–267. [Google Scholar] [CrossRef]
- Fusco, F.; Conte, M.I.; Diociaiuti, A.; Bigoni, S.; Branda, M.F.; Ferlini, A.; El Hachem, M.; Ursini, M.V. Unusual Father-to-Daughter Transmission of Incontinentia Pigmenti Due to Mosaicism in IP Males. Pediatrics 2017, 140, e20162950. [Google Scholar] [CrossRef] [Green Version]
- Rashidghamat, E.; Hsu, C.K.; Nanda, A.; Liu, L.; Al-Ajmi, H.; McGrath, J.A. Incontinentia pigmenti in a father and daughter. Br. J. Dermatol. 2016, 175, 1059–1060. [Google Scholar] [CrossRef] [Green Version]
- Minić, S.; Obradović, M.; Kovacević, I.; Trpinac, D. Ocular anomalies in incontinentia pigmenti: Literature review and meta-analysis. Srp. Arh. Celok. Lek. 2010, 138, 408–413. [Google Scholar] [CrossRef]
- Minić, S.; Trpinac, D.; Gabriel, H.; Gencik, M.; Obradović, M. Dental and oral anomalies in incontinentia pigmenti: A systematic review. Clin. Oral Investig. 2013, 17, 1–8. [Google Scholar] [CrossRef]
- Minić, S.; Trpinac, D.; Obradović, M. Systematic review of central nervous system anomalies in incontinentia pigmenti. Orphanet J. Rare Dis. 2013, 8, 25. [Google Scholar] [CrossRef] [Green Version]
- Meuwissen, M.E.; Mancini, G.M. Neurological findings in incontinentia pigmenti; a review. Eur. J. Med. Genet. 2012, 55, 323–331. [Google Scholar] [CrossRef]
- Ohnishi, H.; Kishimoto, Y.; Taguchi, T.; Kawamoto, N.; Nakama, M.; Kawai, T.; Nakayama, M.; Ohara, O.; Orii, K.; Fukao, T. Immunodeficiency in Two Female Patients with Incontinentia Pigmenti with Heterozygous NEMO Mutation Diagnosed by LPS Unresponsiveness. J. Clin. Immunol. 2017, 37, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Bal, E.; Laplantine, E.; Hamel, Y.; Dubosclard, V.; Boisson, B.; Pescatore, A.; Picard, C.; Hadj-Rabia, S.; Royer, G.; Steffann, J.; et al. Lack of interaction between NEMO and SHARPIN impairs linear ubiquitination and NF-κB activation and leads to incontinentia pigmenti. J. Allergy Clin. Immunol. 2017, 140, 1671–1682. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alabdullatif, Z.; Coulombe, J.; Steffann, J.; Bodemer, C.; Hadj-Rabia, S. Postzygotic mosaicism and incontinentia pigmenti in male patients: Molecular diagnosis yield. Br. J. Dermatol. 2018, 178, e261–e262. [Google Scholar] [CrossRef] [PubMed]
- Margari, L.; Lamanna, A.L.; Buttiglione, M.; Craig, F.; Petruzzelli, M.G.; Terenzio, V. Long-term follow-up of neurological manifestations in a boy with incontinentia pigmenti. Eur. J. Pediatr. 2013, 172, 1259–1262. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.L.; Lai, S.H.; Huang, J.L.; Lee, N.C.; Lee, W.I. Serial cytokine expressions in infants with incontinentia pigmenti. Immunobiology 2013, 218, 772–779. [Google Scholar] [CrossRef] [Green Version]
- Gregersen, P.A.; Sommerlund, M.; Ramsing, M.; Gjørup, H.; Rasmussen, A.A.; Aggerholm, A. Diagnostic and molecular genetic challenges in male incontinentia pigmenti: A case report. Acta Derm. Venereol. 2013, 93, 741–742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, K.D.; Padhiar, B.B.; Karia, U.K.; Shah, B.J. Case reports of incontinentia pigmenti in males. Indian J. Dermatol. 2013, 58, 328. [Google Scholar] [CrossRef]
- Mullan, E.; Barbarian, M.; Trakadis, Y.; Moroz, B. Incontinentia pigmenti in an XY boy: Case report and review of the literature. J. Cutan. Med. Surg. 2014, 18, 119–122. [Google Scholar] [CrossRef]
- Yang, Y.; Guo, Y.; Ping, Y.; Zhou, X.G.; Li, Y. Neonatal incontinentia pigmenti: Six cases and a literature review. Exp. Ther. Med. 2014, 8, 1797–1806. [Google Scholar] [CrossRef] [Green Version]
- Gonzalez, E.M.; DeKlotz, C.C.; Eichenfield, L.F. A 6-day-old male infant with linear band of skin-colored papules. Incontinentia pigmenti. JAMA Pediatrics 2014, 168, 859–860. [Google Scholar] [CrossRef]
- Hull, S.; Arno, G.; Thomson, P.; Mutch, S.; Webster, A.R.; Rai, H.; Hill, V.; Moore, A.T. Somatic mosaicism of a novel IKBKG mutation in a male patient with incontinentia pigmenti. Am. J. Med. Genet. A 2015, 167, 1601–1604. [Google Scholar] [CrossRef]
- Swamy, D.K.; Arunagirinathan, A.; Krishnakumar, R.; Sangili, S. Incontinentia pigmenti: A rare genodermatosis in a male child. J. Clin. Diagn. Res. 2015, 9, 6–8. [Google Scholar] [CrossRef]
- Adashek, J.; Mu, W.; Francis, J.; Cohen, B.; Pruette, C.; Grossberg, A.; Bodurtha, J. Incontinentia Pigmenti with Persistent Hypercalcemia: Case Report. Pediatr. Dermatol. 2016, 33, e315–e317. [Google Scholar] [CrossRef]
- Mangalesh, S.; Chen, X.; Tran-Viet, D.; Viehland, C.; Freedman, S.F.; Toth, C.A. Assessment of the retinal structure in children with incontinentia pigmenti. Retina 2017, 37, 1568–1574. [Google Scholar] [CrossRef]
- Matsuzaki, Y.; Rokunohe, A.; Minakawa, S.; Nomura, K.; Nakano, H.; Ito, E.; Sawamura, D. Incontinentia pigmenti in a male (XY) infant with long-term follow up over 8 years. J. Dermatol. 2018, 45, 100–103. [Google Scholar] [CrossRef]
- Moosajee, M.; Ali, M.A.; Wong, S.C. Retinal Angiography Findings in Male Infant With Incontinentia Pigmenti and Sickle Cell Trait. JAMA Ophthalmol. 2018, 136, e183140. [Google Scholar] [CrossRef]
- Wang, R.; Lara-Corrales, I.; Kannu, P.; Pope, E. Unraveling incontinentia pigmenti: A comparison of phenotype and genotype variants. J. Am. Acad. Dermatol. 2019, 81, 1142–1149. [Google Scholar] [CrossRef]
- Thorsness, S.; Eyler, J.; Mudaliar, K.; Speiser, J.; Kim, W. Asymptomatic Rash in a Male Infant with Incontinentia Pigmenti. J. Pediatr. 2019, 215, 278. [Google Scholar] [CrossRef] [Green Version]
- Sen, A.; Shenoy, P.; Mitra, A.; Jain, T. Multimodal retinal imaging of a 6-year-old male child with incontinentia pigmenti. Indian J. Ophthalmol. 2019, 67, 942–943. [Google Scholar] [CrossRef]
- Haque, M.N.; Ohtsubo, M.; Nishina, S.; Nakao, S.; Yoshida, K.; Hosono, K.; Kurata, K.; Ohishi, K.; Fukami, M.; Sato, M.; et al. Analysis of IKBKG/NEMO gene in five Japanese cases of incontinentia pigmenti with retinopathy: Fine genomic assay of a rare male case with mosaicism. J. Hum. Genet. 2021, 66, 205–214. [Google Scholar] [CrossRef]
- Michel, S.; Reynaud, C.; Daruich, A.; Hadj-Rabia, S.; Bremond-Gignac, D.; Bodemer, C.; Robert, M.P. Early management of sight threatening retinopathy in incontinentia pigmenti. Orphanet. J. Rare Dis. 2020, 15, 223. [Google Scholar] [CrossRef] [PubMed]
- Nakao, S.; Nishina, S.; Tanaka, S.; Yoshida, T.; Yokoi, T.; Azuma, N. Early laser photocoagulation for extensive retinal avascularity in infants with incontinentia pigmenti. Jpn. J. Ophthalmol. 2020, 64, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Kawai, M.; Kato, T.; Tsutsumi, M.; Shinkai, Y.; Inagaki, H.; Kurahashi, H. Molecular analysis of low-level mosaicism of the IKBKG mutation using the X Chromosome Inactivation pattern in Incontinentia Pigmenti. Mol. Genet. Genomic Med. 2020, 8, e1531. [Google Scholar] [CrossRef] [PubMed]
- Chambelland, A.; Aubert, H.; Bourrat, E.; Morice-Picard, F.; Puzenat, E.; Lacour, J.P.; Chiaverini, C.; Société Française de Dermatologie Pédiatrique Research Group. Incontinentia pigmenti in boys: Causes and consequences. Ann. Dermatol. Venereol. 2020, 147, 188–193. [Google Scholar] [CrossRef]
- Fusco, F.; Valente, V.; Fergola, D.; Pescatore, A.; Lioi, M.B.; Ursini, M.V. The Incontinentia Pigmenti Genetic Biobank: Study design and cohort profile to facilitate research into a rare disease worldwide. Eur. J. Hum. Genet. 2019, 27, 1509–1518. [Google Scholar] [CrossRef]
- Fusco, F.; Pescatore, A.; Steffann, J.; Royer, G.; Bonnefont, J.P.; Ursini, M.V. Clinical Utility Gene Card for: Incontinentia pigmenti. Eur. J. Hum. Genet. 2019, 21, 227. [Google Scholar] [CrossRef] [Green Version]
- Scheuerle, A.E.; Ursini, M.V. Incontinentia Pigmenti, 1st ed.; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Stephens, K., Amemiya, A., Eds.; GeneReviews®: Seattle, DC, USA, 1999; pp. 1993–2020. [Google Scholar]
- Maubach, G.; Schmädicke, A.C.; Naumann, M. NEMO Links Nuclear Factor-κB to Human Diseases. Trends Mol. Med. 2017, 23, 1138–1155. [Google Scholar] [CrossRef]
- Pescatore, A.; Esposito, E.; Draber, P.; Walczak, H.; Ursini, M.V. NEMO regulates a cell death switch in TNF signaling by inhibiting recruitment of RIPK3 to the cell death-inducing complex II. Cell Death Dis. 2016, 7, e2346. [Google Scholar] [CrossRef]
- Rudolph, D.; Yeh, W.C.; Wakeham, A.; Rudolph, B.; Nallainathan, D.; Potter, J.; Elia, A.J.; Mak, T.W. Severe liver degeneration and lack of NF-kappaB activation in NEMO/IKKgamma-deficient mice. Genes Dev. 2000, 14, 854–862. [Google Scholar] [CrossRef]
- Makris, C.; Godfrey, V.L.; Krähn-Senftleben, G.; Takahashi, T.; Roberts, J.L.; Schwarz, T.; Feng, L.; Johnson, R.S.; Karin, M. Female mice heterozygous for IKK gamma/NEMO deficiencies develop a dermatopathy similar to the human X-linked disorder incontinentia pigmenti. Mol. Cell 2000, 5, 969–979. [Google Scholar] [CrossRef]
- Schmidt-Supprian, M.; Bloch, W.; Courtois, G.; Addicks, K.; Israël, A.; Rajewsky, K.; Pasparakis, M. NEMO/IKK gamma-deficient mice model incontinentia pigmenti. Mol. Cell 2000, 5, 981–992. [Google Scholar] [CrossRef]
- Vlantis, K.; Wullaert, A.; Polykratis, A.; Kondylis, V.; Dannappel, M.; Schwarzer, R.; Welz, P.; Corona, T.; Walczak, H.; Weih, F.; et al. NEMO Prevents RIP Kinase 1-Mediated Epithelial Cell Death and Chronic Intestinal Inflammation by NF-κB-Dependent and-Independent Functions. Immunity 2016, 15, 553–567. [Google Scholar] [CrossRef] [Green Version]
- Delanghe, T.; Dondelinger, Y.; Bertrand, M.J.M. RIPK1 Kinase-Dependent Death: A Symphony of Phosphorylation Events. Trends Cell Biol. 2020, 30, 189–200. [Google Scholar] [CrossRef]
- Nenci, A.; Huth, M.; Funteh, A.; Schmidt-Supprian, M.; Bloch, W.; Metzger, D.; Chambon, P.; Rajewsky, K.; Krieg, T.; Haase, I.; et al. Skin lesion development in a mouse model of incontinentia pigmenti is triggered by NEMO deficiency in epidermal keratinocytes and requires TNF signaling. Hum. Mol. Genet. 2006, 15, 531–542. [Google Scholar] [CrossRef]
- Nelson, D.L. NEMO, NF-kappaB signaling and incontinentia pigmenti. Curr. Opin. Genet. Dev. 2006, 16, 282–288. [Google Scholar] [CrossRef]
| * IP Patients | ** Ocular Disease | Dental Anomalies | *** Neurological Changes | |
|---|---|---|---|---|
| Total | 91 * | 26 | 13 | 24 |
| Number of IP patients undergoing molecular investigation | 77 | 23 | 11 | 21 |
| Positive Genetic test | 32 | 13 | 6 | 12 |
| Negative Genetic test | 45 | 10 | 5 | 9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pescatore, A.; Spinosa, E.; Casale, C.; Lioi, M.B.; Ursini, M.V.; Fusco, F. Human Genetic Diseases Linked to the Absence of NEMO: An Obligatory Somatic Mosaic Disorder in Male. Int. J. Mol. Sci. 2022, 23, 1179. https://doi.org/10.3390/ijms23031179
Pescatore A, Spinosa E, Casale C, Lioi MB, Ursini MV, Fusco F. Human Genetic Diseases Linked to the Absence of NEMO: An Obligatory Somatic Mosaic Disorder in Male. International Journal of Molecular Sciences. 2022; 23(3):1179. https://doi.org/10.3390/ijms23031179
Chicago/Turabian StylePescatore, Alessandra, Ezia Spinosa, Carmela Casale, Maria Brigida Lioi, Matilde Valeria Ursini, and Francesca Fusco. 2022. "Human Genetic Diseases Linked to the Absence of NEMO: An Obligatory Somatic Mosaic Disorder in Male" International Journal of Molecular Sciences 23, no. 3: 1179. https://doi.org/10.3390/ijms23031179
APA StylePescatore, A., Spinosa, E., Casale, C., Lioi, M. B., Ursini, M. V., & Fusco, F. (2022). Human Genetic Diseases Linked to the Absence of NEMO: An Obligatory Somatic Mosaic Disorder in Male. International Journal of Molecular Sciences, 23(3), 1179. https://doi.org/10.3390/ijms23031179






