Conductive Polymer PEDOT:PSS-Based Platform for Embryonic Stem-Cell Differentiation
Abstract
1. Introduction
2. Results
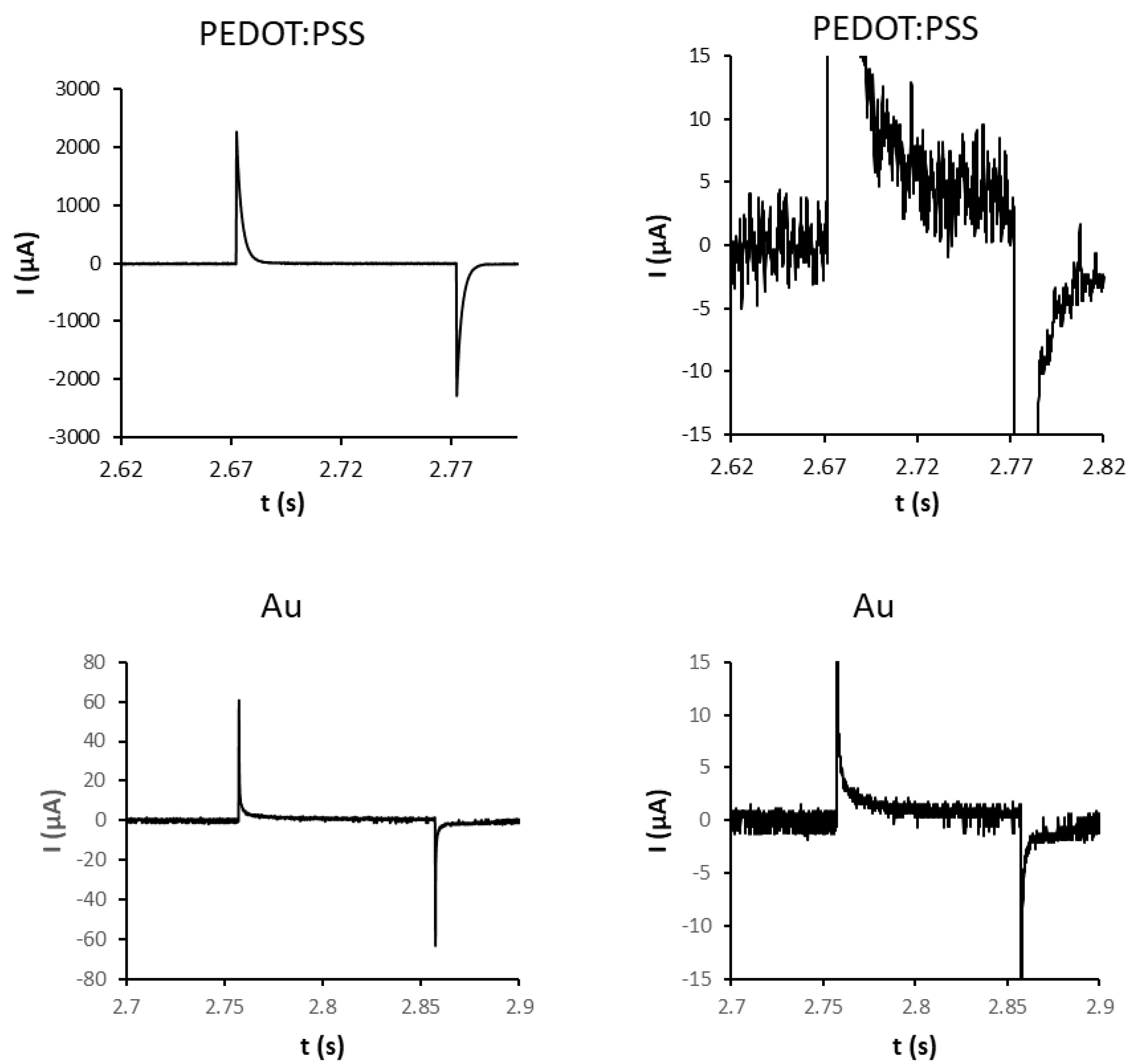
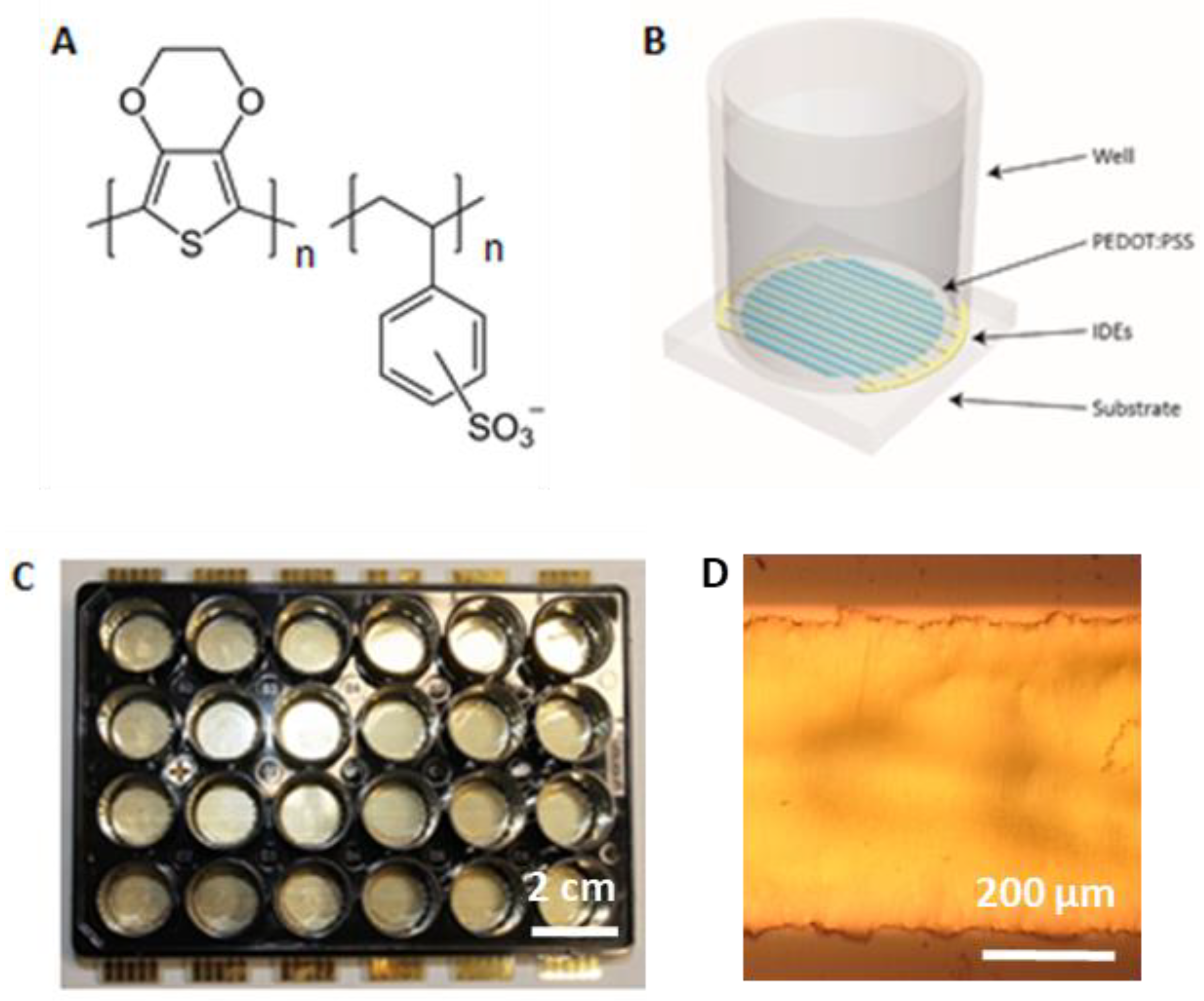
2.1. Platforms for Differentiation of Mouse Embryonic Stem Cells
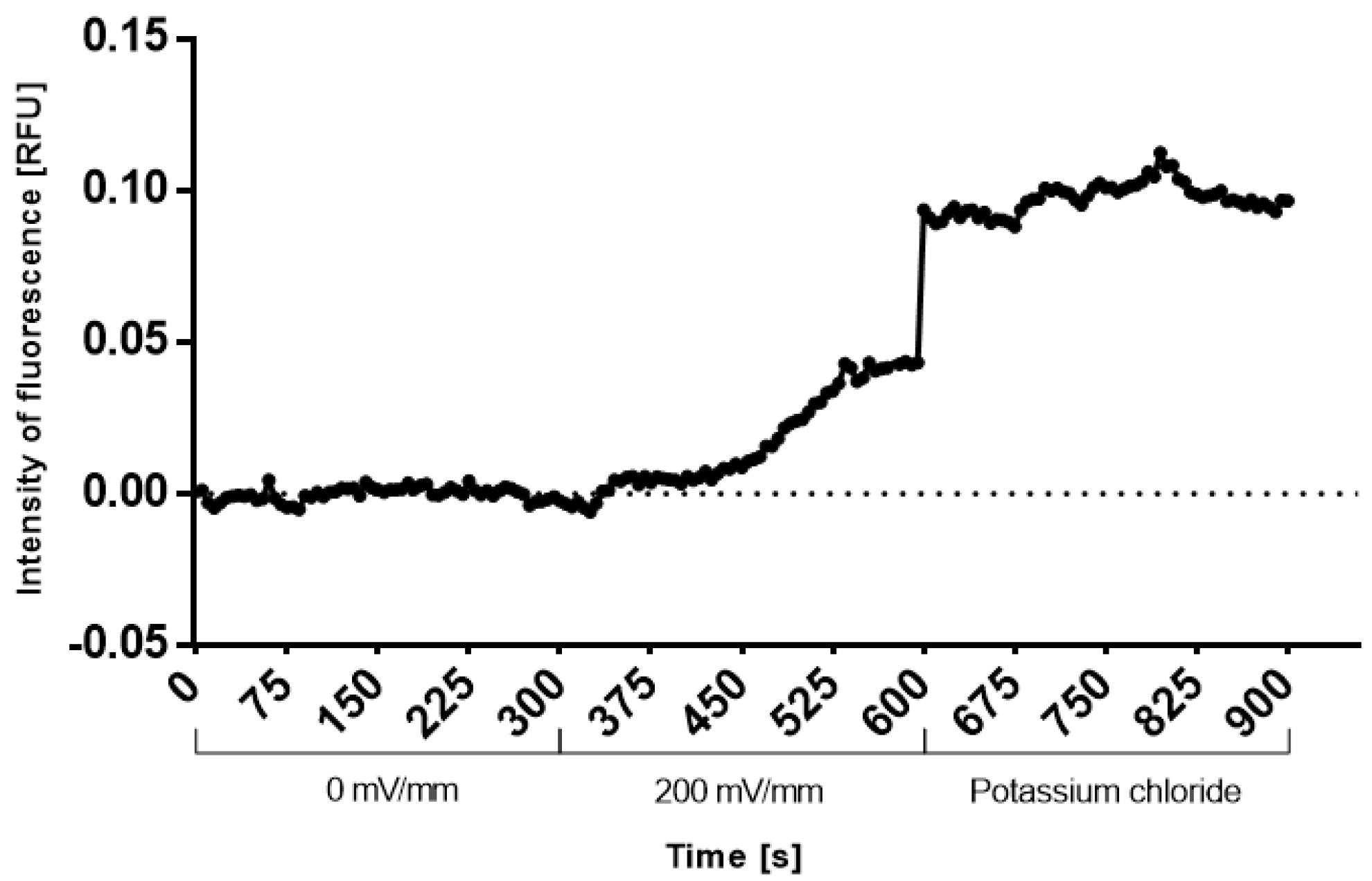
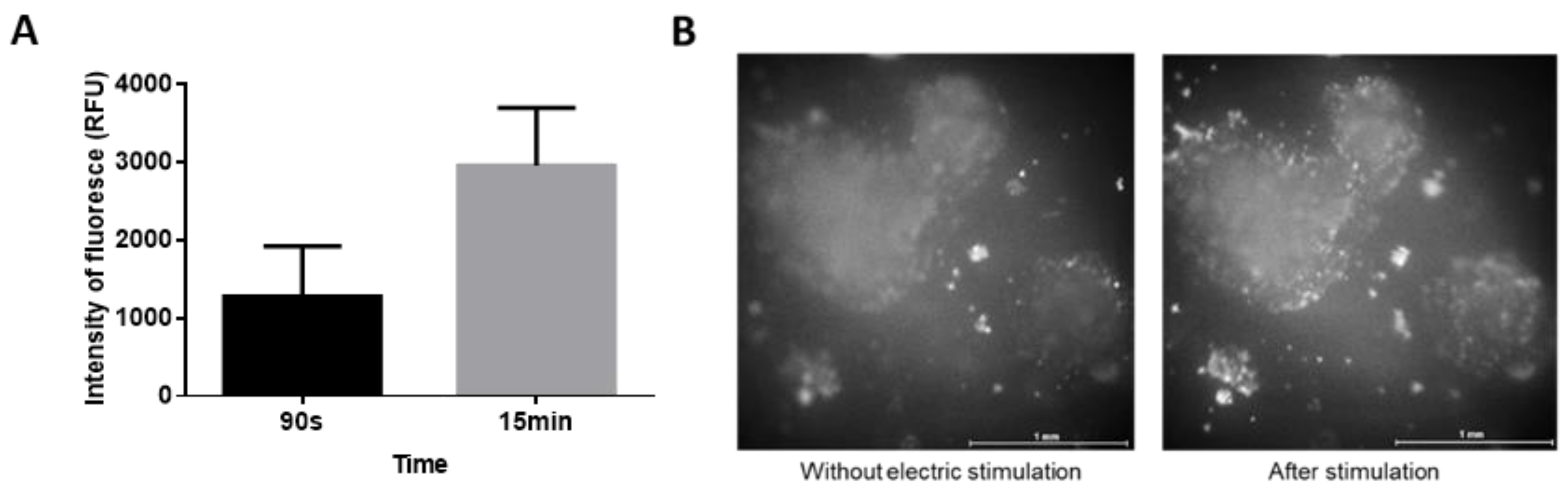
2.2. Early Response of Embryonic Stem Cells
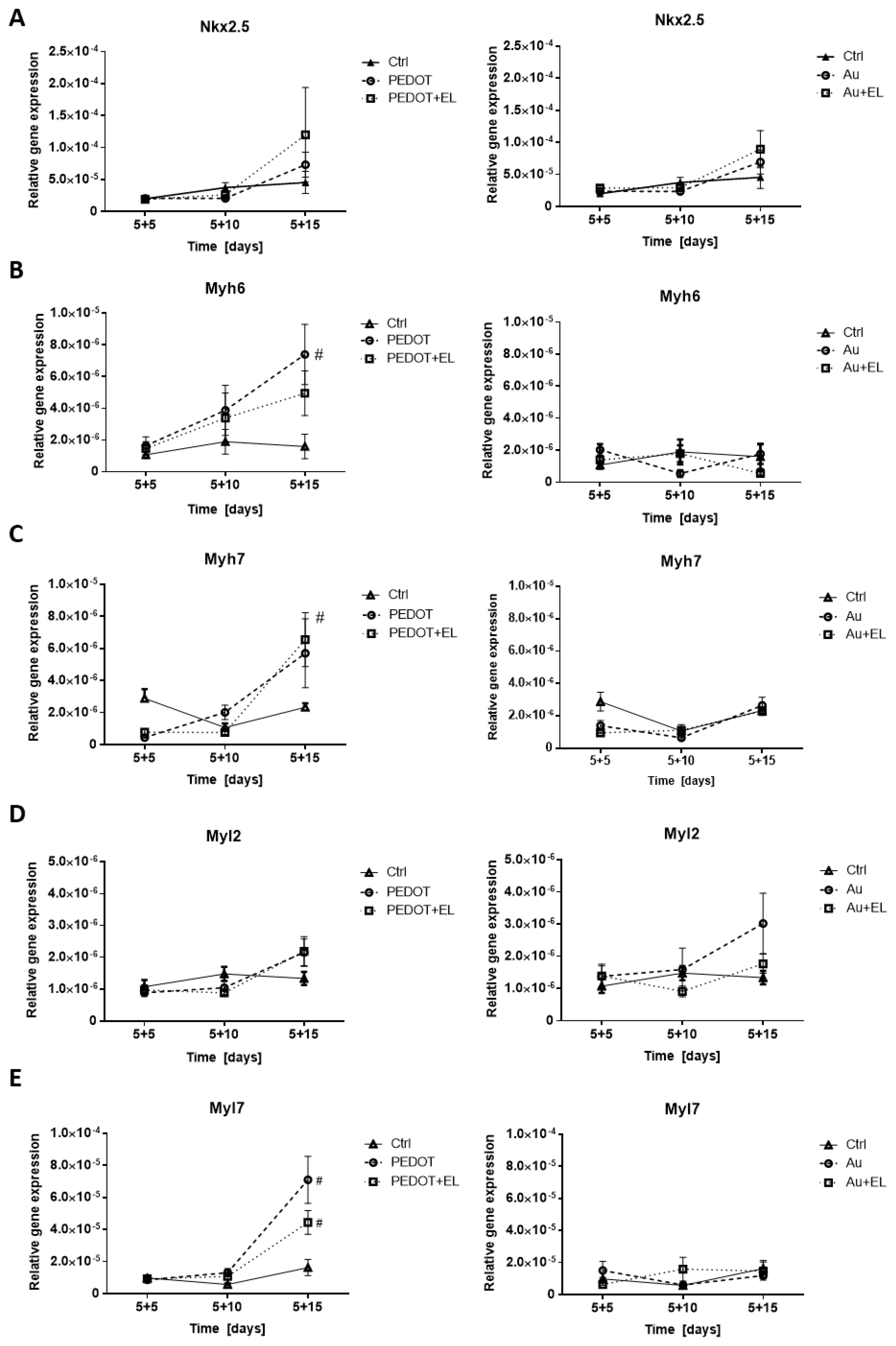
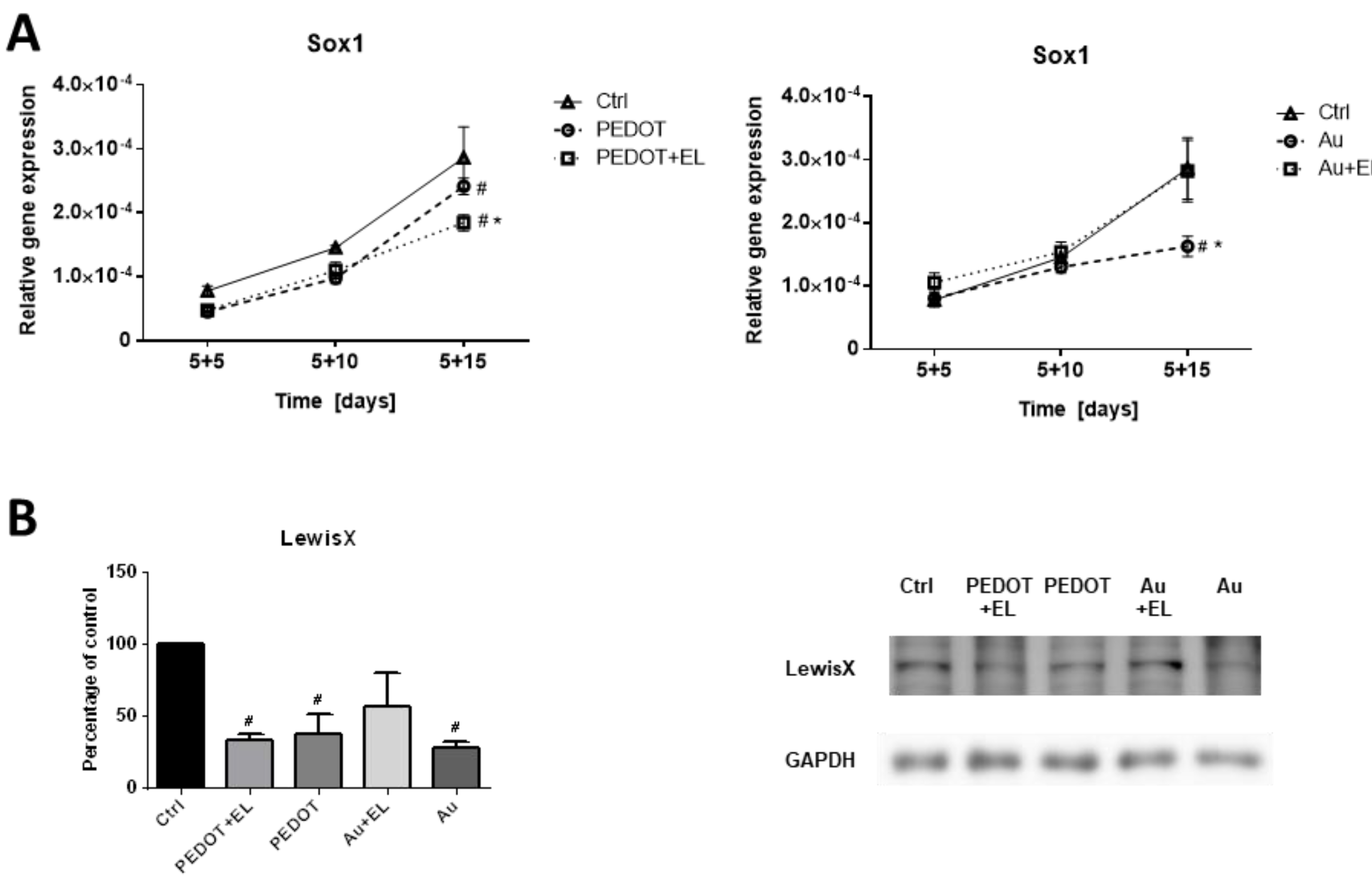
2.3. Impact of Electrostimulation to Gene and Protein Expression
3. Discussion
4. Material and Methods
4.1. Electrostimulation Platform
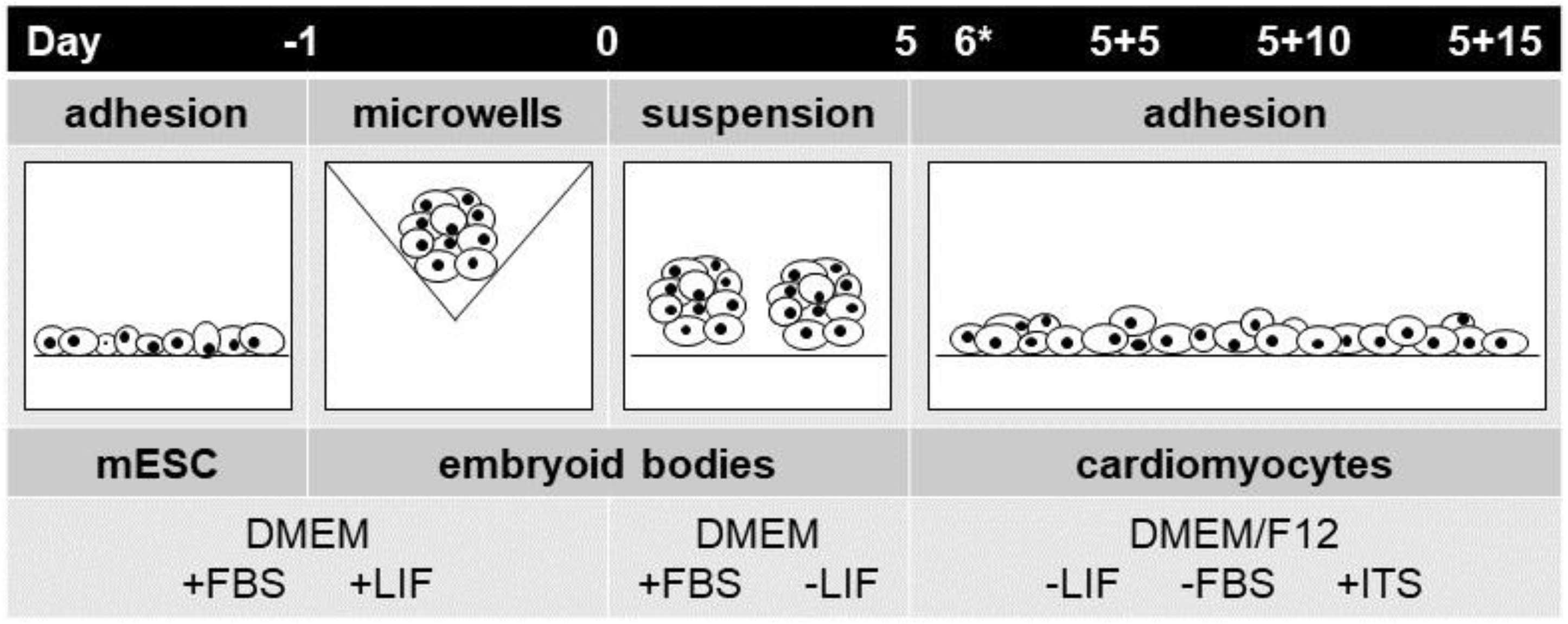
4.2. Mouse Embryonic Stem-Cell Differentiation
4.3. Stem-Cell Electrostimulation
4.4. Imaging of Intracellular Calcium
4.5. Depolarization Detection
4.6. Gene Expression Analysis
4.7. Protein Expression Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| embryoid bodies | (EBs) |
| stem cells | (SCs) |
| embryonic stem cells | (ESCs) |
| mouse embryonic stem cells | (mESCs) |
| poly(3:4-ethylenedioxythiophene):polystyrene sulfonate | (PEDOT:PSS) |
| direct current | (DC) |
References
- Sachinidis, A.; Fleischmann, B.K.; Kolossov, E.; Wartenberg, M.; Sauer, H.; Hescheler, J. Cardiac specific differentiation of mouse embryonic stem cells. Cardiovasc. Res. 2003, 58, 278–291. [Google Scholar] [CrossRef]
- Keller, G. Embryonic stem cell differentiation: Emergence of a new era in biology and medicine. Genes Dev. 2005, 19, 1129–1155. [Google Scholar] [CrossRef]
- Heng, B.C.; Cao, T.; Stanton, L.W.; Robson, P.; Olsen, B. Strategies for directing the differentiation of stem cells into the osteogenic lineage in vitro. J. Bone Miner. Res. 2004, 19, 1379–1394. [Google Scholar] [CrossRef]
- Amin, M.; Kushida, Y.; Wakao, S.; Kitada, M.; Tatsumi, K.; Dezawa, M. Cardiotrophic growth factor-driven induction of human muse cells into cardiomyocyte-like phenotype. Cell Transplant. 2018, 27, 285–298. [Google Scholar] [CrossRef]
- Tian, H.-C.; Liu, J.-Q.; Kang, X.-Y.; Wei, D.-X.; Zhang, C.; Du, J.-C.; Yang, B.; Chen, X.; Yang, C.-S. Biotic and abiotic molecule dopants determining the electrochemical performance, stability and fibroblast behavior of conducting polymer for tissue interface. RSC Adv. 2014, 4, 47461–47471. [Google Scholar] [CrossRef]
- Chen, C.; Bai, X.; Ding, Y.; Lee, I.-S. Electrical stimulation as a novel tool for regulating cell behavior in tissue engineering. Biomater. Res. 2019, 23, 25. [Google Scholar] [CrossRef] [PubMed]
- Broughton, K.M.; Wang, B.J.; Firouzi, F.; Khalafalla, F.; Dimmeler, S.; Fernandez-Aviles, F.; Sussman, M.A. Mechanisms of cardiac repair and regeneration. Circ. Res. 2018, 122, 1151–1163. [Google Scholar] [CrossRef]
- Du Pré, B.C.; Doevendans, P.A.; van Laake, L.W. Stem cells for cardiac repair: An introduction. J. Geriatr. Cardiol. JGC 2013, 10, 186. [Google Scholar]
- Parsa, H.; Ronaldson, K.; Vunjak-Novakovic, G. Bioengineering methods for myocardial regeneration. Adv. Drug Deliv. Rev. 2016, 96, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Thrivikraman, G.; Boda, S.K.; Basu, B. Unraveling the mechanistic effects of electric field stimulation towards directing stem cell fate and function: A tissue engineering perspective. Biomaterials 2018, 150, 60–86. [Google Scholar] [CrossRef]
- Ma, R.; Liang, J.; Huang, W.; Guo, L.; Cai, W.; Wang, L.; Paul, C.; Yang, H.-T.; Kim, H.W.; Wang, Y. Electrical Stimulation Enhances Cardiac Differentiation of Human Induced Pluripotent Stem Cells for Myocardial Infarction Therapy. Antioxid. Redox Signal. 2018, 28, 371–384. [Google Scholar] [CrossRef]
- Radisic, M.; Park, H.; Shing, H.; Consi, T.; Schoen, F.J.; Langer, R.; Freed, L.E.; Vunjak-Novakovic, G. Functional assembly of engineered myocardium by electrical stimulation of cardiac myocytes cultured on scaffolds. Proc. Natl. Acad. Sci. USA 2004, 101, 18129–18134. [Google Scholar] [CrossRef]
- Cao, H.; Kang, B.J.; Lee, C.-A.; Shung, K.K.; Hsiai, T.K. Electrical and mechanical strategies to enable cardiac repair and regeneration. IEEE Rev. Biomed. Eng. 2015, 8, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Gueye, M.N.; Carella, A.; Faure-Vincent, J.; Demadrille, R.; Simonato, J.-P. Progress in understanding structure and transport properties of PEDOT-based materials: A critical review. Prog. Mater. Sci. 2020, 108, 100616. [Google Scholar] [CrossRef]
- Rivnay, J.; Owens, R.M.; Malliaras, G.G. The rise of organic bioelectronics. Chem. Mater. 2013, 26, 679–685. [Google Scholar] [CrossRef]
- Stritesky, S.; Markova, A.; Vitecek, J.; Safarikova, E.; Hrabal, M.; Kubac, L.; Kubala, L.; Weiter, M.; Vala, M. Printing inks of electroactive polymer PEDOT:PSS: The study of biocompatibility, stability, and electrical properties. J. Biomed. Mater. Res. Part A 2018, 106, 1121–1128. [Google Scholar] [CrossRef]
- Dijk, G.; Rutz, A.L.; Malliaras, G.G. Stability of PEDOT:PSS-Coated Gold Electrodes in Cell Culture Conditions. Adv. Mater. Technol. 2020, 5, 1900662. [Google Scholar] [CrossRef]
- Moyen, E.; Hama, A.; Ismailova, E.; Assaud, L.; Malliaras, G.; Hanbücken, M.; Owens, R.M. Nanostructured conducting polymers for stiffness controlled cell adhesion. Nanotechnology 2016, 27, 074001. [Google Scholar] [CrossRef]
- Ritzau-Reid, K.I.; Spicer, C.D.; Gelmi, A.; Grigsby, C.L.; Ponder, J.F., Jr.; Bemmer, V.; Creamer, A.; Vilar, R.; Serio, A.; Stevens, M.M. An electroactive oligo-EDOT platform for neural tissue engineering. Adv. Funct. Mater. 2020, 30, 2003710. [Google Scholar] [CrossRef] [PubMed]
- Heo, D.N.; Acquah, N.; Kim, J.; Lee, S.-J.; Castro, N.J.; Zhang, L.G. Directly induced neural differentiation of human adipose-derived stem cells using three-dimensional culture system of conductive microwell with electrical stimulation. Tissue Eng. Part A 2018, 24, 537–545. [Google Scholar] [CrossRef]
- Aggas, J.R.; Abasi, S.; Phipps, J.F.; Podstawczyk, D.A.; Guiseppi-Elie, A. Microfabricated and 3-D printed electroconductive hydrogels of PEDOT:PSS and their application in bioelectronics. Biosens. Bioelectron. 2020, 168, 112568. [Google Scholar] [CrossRef] [PubMed]
- Iandolo, D.; Sheard, J.; Levy, G.K.; Pitsalidis, C.; Tan, E.; Dennis, A.; Kim, J.-S.; Markaki, A.E.; Widera, D.; Owens, R.M. Biomimetic and electroactive 3D scaffolds for human neural crest-derived stem cell expansion and osteogenic differentiation. MRS Commun. 2020, 10, 179–187. [Google Scholar] [CrossRef]
- Tomaskovic-Crook, E.; Zhang, P.; Ahtiainen, A.; Kaisvuo, H.; Lee, C.-Y.; Beirne, S.; Aqrawe, Z.; Svirskis, D.; Hyttinen, J.; Wallace, G.G.; et al. Human neural tissues from neural stem cells using conductive biogel and printed polymer microelectrode arrays for 3D electrical stimulation. Adv. Healthc. Mater. 2019, 8, 1900425. [Google Scholar] [CrossRef]
- Tsai, N.-C.; She, J.-W.; Wu, J.-G.; Chen, P.; Hsiao, Y.-S.; Yu, J. Poly (3,4-ethylenedioxythiophene) polymer composite bioelectrodes with designed chemical and topographical cues to manipulate the behavior of pc12 neuronal cells. Adv. Mater. Interfaces 2019, 6, 1801576. [Google Scholar] [CrossRef]
- Pires, F.; Ferreira, Q.; Rodrigues, C.A.; Morgado, J.; Ferreira, F.C. Neural stem cell differentiation by electrical stimulation using a cross-linked PEDOT substrate: Expanding the use of biocompatible conjugated conductive polymers for neural tissue engineering. Biochim. Biophys. Acta Gen. Subj. 2015, 1850, 1158–1168. [Google Scholar] [CrossRef] [PubMed]
- Decataldo, F.; Druet, V.; Pappa, A.-M.; Tan, E.; Savva, A.; Pitsalidis, C.; Inal, S.; Kim, J.-S.; Fraboni, B.; Owens, R.M.; et al. BMP-2 functionalized PEDOT:PSS-based OECTs for stem cell osteogenic differentiation monitoring. Flex. Print. 2019, 4, 044006. [Google Scholar] [CrossRef]
- Roshanbinfar, K.; Vogt, L.; Greber, B.; Diecke, S.; Boccaccini, A.R.; Scheibel, T.; Engel, F.B. Electroconductive biohybrid hydrogel for enhanced maturation and beating properties of engineered cardiac tissues. Adv. Funct. Mater. 2018, 28, 1803951. [Google Scholar] [CrossRef]
- Yoshida, S.; Sumomozawa, K.; Nagamine, K.; Nishizawa, M. Hydrogel Microchambers Integrated with Organic Electrodes for Efficient Electrical Stimulation of Human iPSC-Derived Cardiomyocytes. Macromol. Biosci. 2019, 19, 1900060. [Google Scholar] [CrossRef]
- Menzies, K.L.; Jones, L. The impact of contact angle on the biocompatibility of biomaterials. Optom. Vis. Sci. 2010, 87, 387–399. [Google Scholar] [CrossRef] [PubMed]
- Safarikova, E.; Sindlerova, L.S.; Stritesky, S.; Kubala, L.; Vala, M.; Weiter, M.; Vitecek, J. Evaluation and improvement of organic semiconductors’ biocompatibility towards fibroblasts and cardiomyocytes. Sens. Actuators B Chem. 2018, 260, 418–425. [Google Scholar] [CrossRef]
- Radaszkiewicz, K.A.; Sykorova, D.; Bino, L.; Kudova, J.; Bebarova, M.; Prochazkova, J.; Kotasova, H.; Kubala, L.; Pachernik, J. The acceleration of cardiomyogenesis in embryonic stem cells in vitro by serum depletion does not increase the number of developed cardiomyocytes. PLoS ONE 2017, 12, e0173140. [Google Scholar] [CrossRef] [PubMed]
- Lanza, R.; Gearhart, J.; Hogan, B.; Melton, D.; Pedersen, R.; Thomas, E.D.; Thomson, J.A.; West, M. Essentials of Stem Cell Biology; Elsevier: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Hernandez, D.; Millard, R.; Sivakumaran, P.; Wong, R.C.; Crombie, D.E.; Hewitt, A.W.; Liang, H.; Hung, S.S.; Pebay, A.; Shepherd, R.K.; et al. Electrical stimulation promotes cardiac differentiation of human induced pluripotent stem cells. Stem Cells Int. 2015, 2016, 1718041. [Google Scholar] [CrossRef] [PubMed]
- Sauer, H.; Bekhite, M.M.; Hescheler, J.; Wartenberg, M. Redox control of angiogenic factors and CD31-positive vessel-like structures in mouse embryonic stem cells after direct current electrical field stimulation. Exp. Cell Res. 2005, 304, 380–390. [Google Scholar] [CrossRef]
- Yamada, M.; Tanemura, K.; Okada, S.; Iwanami, A.; Nakamura, M.; Mizuno, H.; Ozawa, M.; Ohyama-Goto, R.; Kitamura, N.; Kawano, M.; et al. Electrical stimulation modulates fate determination of differentiating embryonic stem cells. Stem Cells 2007, 25, 562–570. [Google Scholar] [CrossRef]
- Serena, E.; Figallo, E.; Tandon, N.; Cannizzaro, C.; Gerecht, S.; Elvassore, N.; Vunjak-Novakovic, G. Electrical stimulation of human embryonic stem cells: Cardiac differentiation and the generation of reactive oxygen species. Exp. Cell Res. 2009, 315, 3611–3619. [Google Scholar] [CrossRef] [PubMed]
- Tandon, N.; Marsano, A.; Maidhof, R.; Numata, K.; Montouri-Sorrentino, C.; Cannizzaro, C.; Voldman, J.; Vunjak-Novakovic, G. Surface-patterned electrode bioreactor for electrical stimulation. Lab Chip 2010, 10, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.-C.; Ting, S.; Lee, Y.-K.; Ng, K.-M.; Zhang, J.; Chen, Z.; Siu, C.-W.; Oh, S.K.W.; Tse, H.-F. Electrical stimulation promotes maturation of cardiomyocytes derived from human embryonic stem cells. J. Cardiovasc. Transl. Res. 2013, 6, 989–999. [Google Scholar] [CrossRef]
- Nuccitelli, R. Endogenous ionic currents and DC electric fields in multicellular animal tissues. Bioelectromagnetics 1992, 13, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Proctor, C.M.; Rivnay, J.; Malliaras, G.G. Understanding volumetric capacitance in conducting polymers. J. Polym. Sci. B Polym. Phys. 2016, 54, 1433–1436. [Google Scholar] [CrossRef]
- Winther-Jensen, B.; Winther-Jensen, O.; Forsyth, M.; MacFarlane, D.R. High rates of oxygen reduction over a vapor phase-polymerized PEDOT electrode. Science 2008, 321, 671–674. [Google Scholar] [CrossRef]
- Cottis, P.P.; Evans, D.; Fabretto, M.; Pering, S.; Murphy, P.; Hojati-Talemi, P. Metal-free oxygen reduction electrodes based on thin PEDOT films with high electrocatalytic activity. RSC Adv. 2014, 4, 9819–9824. [Google Scholar] [CrossRef]
- Kudova, J.; Prochazkova, J.; Vasicek, O.; Perecko, T.; Sedlackova, M.; Pesl, M.; Pachernik, J.; Kubala, L. HIF-1alpha deficiency attenuates the cardiomyogenesis of mouse embryonic stem cells. PLoS ONE 2016, 11, e0158358. [Google Scholar] [CrossRef] [PubMed]
- Vecera, J.; Kudova, J.; Kucera, J.; Kubala, L.; Pachernik, J. Neural Differentiation Is Inhibited through HIF1α/β-Catenin Signaling in Embryoid Bodies. Stem Cells Int. 2017, 2017, 8715798. [Google Scholar] [CrossRef]
- Humpolicek, P.; Radaszkiewicz, K.A.; Kasparkova, V.; Stejskal, J.; Trchova, M.; Kucekova, Z.; Vicarova, H.; Pachernik, J.; Lehocky, M.; Minarik, A. Stem cell differentiation on conducting polyaniline. RSC Adv. 2015, 5, 68796–68805. [Google Scholar] [CrossRef]
- Radaszkiewicz, K.A.; Beckerova, D.; Woloszczukova, L.; Radaszkiewicz, T.W.; Lesakova, P.; Blanarova, O.V.; Kubala, L.; Humpolicek, P.; Pachernik, J. 12-O-Tetradecanoylphorbol-13-acetate increases cardiomyogenesis through PKC/ERK signaling. Sci. Rep. 2020, 10, 15922. [Google Scholar] [CrossRef] [PubMed]
- Tandon, N.; Cannizzaro, C.; Chao, P.-H.G.; Maidhof, R.; Marsano, A.; Au, H.T.H.; Radisic, M.; Vunjak-Novakovic, G. Electrical stimulation systems for cardiac tissue engineering. Nat. Protoc. 2009, 4, 155–173. [Google Scholar] [CrossRef]
- Sauer, H.; Rahimi, G.; Hescheler, J.; Wartenberg, M. Effects of electrical fields on cardiomyocyte differentiation of embryonic stem cells. J. Cell. Biochem. 1999, 75, 710–723. [Google Scholar] [CrossRef]
- Coelho, N.M.; González-Garcia, C.; Planell, J.; Salmerón-Sánchez, M.; Altankov, G. Different assembly of type IV collagen on hydrophilic and hydrophobic substrata alters endothelial cells interaction. Eur. Cell. Mater. 2010, 19, 262–272. [Google Scholar] [CrossRef]
- Nagy, A.; Rossant, J.; Nagy, R.; Abramow-Newerly, W.; Roder, J.C. Derivation of completely cell culture-derived mice from early-passage embryonic stem cells. Proc. Natl. Acad. Sci. USA 1993, 90, 8424–8428. [Google Scholar] [CrossRef]
- Dahlmann, J.; Kensah, G.; Kempf, H.; Skvorc, D.; Gawol, A.; Elliott, D.A.; Dräger, G.; Zweigerdt, R.; Martin, U.; Gruh, I. The use of agarose microwells for scalable embryoid body formation and cardiac differentiation of human and murine pluripotent stem cells. Biomaterials 2013, 34, 2463–2471. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W.; Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; et al. NIH image to imageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Adams, D.S.; Levin, M. Measuring resting membrane potential using the fluorescent voltage reporters DiBAC4 (3) and CC2-DMPE. Cold Spring Harb. Protoc. 2012, 2012, pdb-prot067702. [Google Scholar] [CrossRef] [PubMed]
- Lan, J.-Y.; Williams, C.; Levin, M.; Black, L.D. Depolarization of cellular resting membrane potential promotes neonatal cardiomyocyte proliferation in vitro. Cell. Mol. Bioeng. 2014, 7, 432–445. [Google Scholar] [CrossRef] [PubMed][Green Version]
| Gene of Interest | Forward Primer 5′→3′ | Reverse Primer 5′→3′ | UPL Probe No. |
|---|---|---|---|
| Rpl13a | CATGAGGTCGGGTGGAAGTA | GCCTGTTTCCGTAACCTCAA | #25 |
| Nkx2.5 | GACGTAGCCTGGTGTCTCG | GTGTGGAATCCGTCGAAAGT | #53 |
| Myh6 | CGCATCAAGGAGCTCACC | CCTGCAGCCGCATTAAGT | #6 |
| Myh7 | CGCATCAAGGAGCTCACC | CTGCAGCCGCAGTAGGTT | #6 |
| Myl2 | CCCAGATCCAGGAGTTCAAG | CTGCAGCCGCAGTAGGTT | #95 |
| Myl7 | CCCATCAACTTCACCGTCTT | AACATGCGGAAGGCACTC | #7 |
| Sox1 | GTGACATCTGCCCCCATC | GAGGCCAGTCTGGTGTCAG | #60 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Šafaříková, E.; Ehlich, J.; Stříteský, S.; Vala, M.; Weiter, M.; Pacherník, J.; Kubala, L.; Víteček, J. Conductive Polymer PEDOT:PSS-Based Platform for Embryonic Stem-Cell Differentiation. Int. J. Mol. Sci. 2022, 23, 1107. https://doi.org/10.3390/ijms23031107
Šafaříková E, Ehlich J, Stříteský S, Vala M, Weiter M, Pacherník J, Kubala L, Víteček J. Conductive Polymer PEDOT:PSS-Based Platform for Embryonic Stem-Cell Differentiation. International Journal of Molecular Sciences. 2022; 23(3):1107. https://doi.org/10.3390/ijms23031107
Chicago/Turabian StyleŠafaříková, Eva, Jiří Ehlich, Stanislav Stříteský, Martin Vala, Martin Weiter, Jiří Pacherník, Lukáš Kubala, and Jan Víteček. 2022. "Conductive Polymer PEDOT:PSS-Based Platform for Embryonic Stem-Cell Differentiation" International Journal of Molecular Sciences 23, no. 3: 1107. https://doi.org/10.3390/ijms23031107
APA StyleŠafaříková, E., Ehlich, J., Stříteský, S., Vala, M., Weiter, M., Pacherník, J., Kubala, L., & Víteček, J. (2022). Conductive Polymer PEDOT:PSS-Based Platform for Embryonic Stem-Cell Differentiation. International Journal of Molecular Sciences, 23(3), 1107. https://doi.org/10.3390/ijms23031107







