Biodegradable and Non-Biodegradable Biomaterials and Their Effect on Cell Differentiation
Abstract
1. Introduction
2. Natural Biomaterials Directing Differentiation in Desired Directions
2.1. Collagen
Topology and Stiffness
2.2. Elastin
Topology and Stiffness
2.3. Fibrin
Topology and Stiffness
2.4. Gelatin
Topology and Stiffness
2.5. Silk Fibroin
Topology and Stiffness
2.6. Glycosaminoglycans
Topology and Stiffness
2.7. Alginate and Chitosan
Topology and Stiffness
2.8. Other Natural Biomaterials
2.8.1. Plant-Derived Biomorphous Carbon Materials
2.8.2. Matrigel
2.9. Decellularized Scaffolds as an Example of Natural Biomaterials with Differentiation-Supporting Properties
3. Synthetic Biomaterials or Dopants Supporting Differentiation of Stem Cells into Selected Cell TYPES
3.1. Synthetic Biomaterials
3.1.1. Polycaprolactone (PCL)
Topology
Stiffness
Biological Cues
3.1.2. Polylactic Acid, L-lactic Acid, and lactic-co-Glycolic Acid (PLA, PLLA, and PLGA)
Topology
Stiffness
Biological Cues
3.1.3. Polyethylene Glycol and (Ethylene Glycol) Diacrylate (PEG and PEGDA)
3.2. Other Polymeric Biomaterials
3.2.1. Polyurethane (PU)
3.2.2. Polyvinyl Alcohol (PVA)
3.2.3. Polyethylene Terephthalate (PET)
3.3. Ceramics
3.4. Metals
4. Natural and Synthetic Biomaterials and Dopants Attenuating or Impairing Stem Cell Differentiation
4.1. Two-Dimensional (2D) Surfaces Versus Three-Dimensional (3D) Biomaterial Scaffolds
4.2. Natural Versus Synthetic Biomaterials
4.3. Microenvironment-Related Factors Affecting Stem Cells Fate
4.4. Parameters for Designing Biomaterials
4.5. The Influence of Properties of Extracellular Matrices on Cell Fate
4.6. Assessment of Time-Dependent Responses of Stem Cells
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Arg | arginine |
| Asp | aspartic acid |
| Au | gold |
| bFGF | basic Fibroblast Growth Factor |
| BMP2 | Bone Morphogenic Protein-2 |
| BMSCs | Bone marrow Mesenchymal Stem Cell |
| CA | Chitosan Alginate |
| CNT | Carbon NanoTubes |
| CTGF | Connective Tissue Growth Factor |
| dECM | decellularized Extracellular Matrix |
| ECM | Extracellular Matrix |
| EGF | Epidermal Growth Factor |
| FGF | Fibroblast Growth Factor |
| GAIN | Graphene Augmented Inorganic Nanofiber |
| gelMA | gelatin Methacrylate |
| Gly | Glycine |
| GO | Graphene Oxide |
| hADSCs | human Adipose-Derived Stem Cells |
| HGF | Hepatocyte Growth Factor |
| hiPSCs | human induced Pluripotent Stem Cells |
| hMSCs | human Mesenchymal Stem Cells |
| HRP | HorseRadish Peroxidase |
| IGF1 | Insulin like Growth Factor-1 |
| IncRNAs | long noncoding RNAs |
| MAPK/ERK | Mitogen-Activated Protein Kinases/Extracellular signal-Regulated Kinases |
| MCWCNT | MultiWalled Carbon NanoTubes |
| Mg | Magnesium |
| MSCs | Mesenchymal Stem Cells |
| mTOR | mammalian Target Of Rapamycin |
| Nestin | neuroepithelial stem cell protein |
| PAMAM | PolyAMidoAMine |
| PCL | PolyCaproLactone |
| PDGF | Platelet-Derived Growth Factor |
| PEC | PolyElectrolyte Complex |
| PEG | PolyEthylene Glycol |
| PEGDA | Poly(Ethylene Glycol)DiAcrylate |
| PET | PolyEthylene Terephthalate |
| PHB | Poly3-HydroxyButyrate |
| PHBV | Poly(3-HydroxyButyrate-co-3-hydroxyValerate) |
| PLGA | PolyLactic-co-Glycolic Acid |
| PLLA | PolyL-Lactic Acid |
| Poly-P | Poly-Phospate |
| PTHF | PolyTetraHydroFuran |
| PVA | PolyVinyl Alcohol |
| RGD | aRginine-Glycine-aspartic aciD |
| RGD-MNP | Arg-Gly-Asp tripeptide-bearing magnetic nanoparticle |
| RGNPs | RGD conjugated Gold Nanoparticles |
| SMC | Smooth Muscle Cell |
| TGF-β3 | Transforming Growth Factor β3 |
| VEGF | Vascular Endothelial Growth Factor |
References
- Hubbell, J.A. Biomaterials in tissue engineering. Bio/technology 1995, 13, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Yoo, J.J.; Atala, A. Biomaterials and tissue engineering. Clin. Regen. Med. Urol. 2018, 13, 17–51. [Google Scholar]
- Erten, E.; Arslan, Y.E. The Great Harmony in Translational Medicine: Biomaterials and Stem Cells. Adv. Exp. Med. Biol. 2018, 1119, 21–39. [Google Scholar] [CrossRef] [PubMed]
- Basu, B. Biomaterials Science and Tissue Engineering: Principles and Methods; Cambridge University Press: Cambridge, UK, 2017. [Google Scholar]
- O’Brien, F.J. Biomaterials & scaffolds for tissue engineering. Mater. Today 2011, 14, 88–95. [Google Scholar] [CrossRef]
- Williams, D.F. On the nature of biomaterials. Biomaterials 2009, 30, 5897–5909. [Google Scholar] [CrossRef]
- Kitala, D.; Klama-Baryła, A.; Łabuś, W.; Ples, M.; Misiuga, M.; Kraut, M.; Szapski, M.; Bobiński, R.; Pielesz, A.; Łos, M.J.; et al. Amniotic cells share clusters of differentiation of fibroblasts and keratinocytes, influencing their ability to proliferate and aid in wound healing while impairing their angiogenesis capability. Eur. J. Pharmacol. 2019, 854, 167–178. [Google Scholar] [CrossRef]
- Kitala, D.; Klama-Baryła, A.; Misiuga, M.; Łabuś, W.; Kraut, M.; Szapski, M.; Lesiak, M.; Krakowian, D.; Sieroń, A.L.; Łos, M.J.; et al. Heterogeneous Mixture of Amniotic Cells is Likely a Better Source of Stem Cells than Adipose Tissue. Arch. Immunol. Ther. Exp. 2019, 67, 189–196. [Google Scholar] [CrossRef]
- Mazhari, S.; Gitiara, A.; Baghaei, K.; Hatami, B.; Rad, R.E.; Asadirad, A.; Joharchi, K.; Tokhanbigli, S.; Hashemi, S.M.; Łos, M.J.; et al. Therapeutic potential of bone marrow-derived mesenchymal stem cells and imatinib in a rat model of liver fibrosis. Eur. J. Pharmacol. 2020, 882, 173263. [Google Scholar] [CrossRef]
- Cieślar-Pobuda, A.; Knoflach, V.; Ringh, M.V.; Stark, J.; Likus, W.; Siemianowicz, K.; Ghavami, S.; Hudecki, A.; Green, J.L.; Łos, M.J. Transdifferentiation and reprogramming: Overview of the processes, their similarities and differences. Biochimica et biophysica acta. Mol. Cell Res. 2017, 1864, 1359–1369. [Google Scholar] [CrossRef]
- Kucharzewski, M.; Rojczyk, E.; Wilemska-Kucharzewska, K.; Wilk, R.; Hudecki, J.; Los, M.J. Novel trends in application of stem cells in skin wound healing. Eur. J. Pharmacol. 2019, 843, 307–315. [Google Scholar] [CrossRef]
- Hybiak, J.; Jankowska, K.; Machaj, F.; Rosik, J.; Broniarek, I.; Żyluk, A.; Hilderman, G.C.; Małecki, A.; Łos, M.J.; Urasińska, E. Reprogramming and transdifferentiation—Two key processes for regenerative medicine. Eur. J. Pharmacol. 2020, 882, 173202. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K.; Mujawar, M.A.; Kaushik, A. State-of-Art Functional Biomaterials for Tissue Engineering. Front. Mater. 2019, 6, 172. [Google Scholar] [CrossRef]
- Guilak, F.; Pferdehirt, L.; Ross, A.K.; Choi, Y.-R.; Collins, K.; Nims, R.J.; Katz, D.B.; Klimak, M.; Tabbaa, S.; Pham, C.T.N. Designer Stem Cells: Genome Engineering and the Next Generation of Cell-Based Therapies. J. Orthop. Res. 2019, 37, 1287–1293. [Google Scholar] [CrossRef]
- Pferdehirt, L.; Ross, A.K.; Brunger, J.M.; Guilak, F. A synthetic gene circuit for self-regulating delivery of biologic drugs in engineered tissues. Tissue Eng. Part A 2019, 25, 809–820. [Google Scholar] [CrossRef]
- Lin, T.; Pajarinen, J.; Nabeshima, A.; Lu, L.; Nathan, K.; Yao, Z.; Goodman, S.B. Establishment of NF-κB sensing and interleukin-4 secreting mesenchymal stromal cells as an “on-demand” drug delivery system to modulate inflammation. Cytotherapy 2017, 19, 1025–1034. [Google Scholar] [CrossRef] [PubMed]
- Staff, P.O. Correction: Paracrine effect of mesenchymal stem cells derived from human adipose tissue in bone regeneration. PLoS ONE 2015, 10, e0119262. [Google Scholar]
- Zhou, H.; Xiao, C.; Wang, Y.; Bi, X.; Ge, S.; Fan, X. In Vivo Efficacy of Bone Marrow Stromal Cells Coated with Beta-Tricalcium Phosphate for the Reconstruction of Orbital Defects in Canines. Investig. Ophthalmol. Vis. Sci. 2011, 52, 1735–1741. [Google Scholar] [CrossRef]
- Field, J.; McGee, M.; Stanley, R.; Ruthenbeck, G.; Papadimitrakis, T.; Zannettino, A.; Gronthos, S.; Itescu, S. The efficacy of allogeneic mesenchymal precursor cells for the repair of an ovine tibial segmental defect. Vet. Comp. Orthop. Traumatol. 2011, 24, 113–121. [Google Scholar] [CrossRef]
- Usas, A.; Ho, A.M.; Cooper, G.M.; Olshanski, A.; Peng, H.; Huard, J. Bone regeneration mediated by BMP4-expressing muscle-derived stem cells is affected by delivery system. Tissue Eng. Part A 2009, 15, 285–293. [Google Scholar] [CrossRef]
- Sijbesma, R.P.; Beijer, F.H.; Brunsveld, L.; Folmer, B.J.; Hirschberg, J.K.; Lange, R.F.; Lowe, J.K.; Meijer, E. Reversible polymers formed from self-complementary monomers using quadruple hydrogen bonding. Science 1997, 278, 1601–1604. [Google Scholar] [CrossRef]
- Liu, P.; Deng, Z.; Han, S.; Liu, T.; Wen, N.; Lu, W.; Geng, X.; Huang, S.; Jin, Y. Tissue-Engineered Skin Containing Mesenchymal Stem Cells Improves Burn Wounds. Artif. Organs 2008, 32, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Usas, A.; Proto, J.D.; Lu, A.; Cummins, J.H.; Proctor, A.; Chen, C.-W.; Huard, J. Role of donor and host cells in muscle-derived stem cell-mediated bone repair: Differentiation vs. paracrine effects. FASEB J. 2014, 28, 3792–3809. [Google Scholar] [CrossRef]
- Linero, I.; Chaparro, O. Paracrine effect of mesenchymal stem cells derived from human adipose tissue in bone regeneration. PLoS ONE 2014, 9, e107001. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Fan, H.; Zhang, Z.-Y.; Lou, A.-J.; Pei, G.-X.; Jiang, S.; Mu, T.-W.; Qin, J.-J.; Chen, S.-Y.; Jin, D. Osteogenesis and angiogenesis of tissue-engineered bone constructed by prevascularized β-tricalcium phosphate scaffold and mesenchymal stem cells. Biomaterials 2010, 31, 9452–9461. [Google Scholar] [CrossRef]
- Cui, L.; Liu, B.; Liu, G.; Zhang, W.; Cen, L.; Sun, J.; Yin, S.; Liu, W.; Cao, Y. Repair of cranial bone defects with adipose derived stem cells and coral scaffold in a canine model. Biomaterials 2007, 28, 5477–5486. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.P.; Leong, K.W. Scaffolding in tissue engineering: General approaches and tissue-specific considerations. Eur. Spine J. Off. Publ. Eur. Spine Soc. Eur. Spinal Deform. Soc. Eur. Sect. Cerv. Spine Res. Soc. 2008, 17 (Suppl. S4), 467–479. [Google Scholar] [CrossRef]
- Rahmati, M.; Pennisi, C.P.; Budd, E.; Mobasheri, A.; Mozafari, M. Biomaterials for Regenerative Medicine: Historical Perspectives and Current Trends. Adv. Exp. Med. Biol. 2018, 1119, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Sofi, H.S.; Ashraf, R.; Beigh, M.A.; Sheikh, F.A. Scaffolds Fabricated from Natural Polymers/Composites by Electrospinning for Bone Tissue Regeneration. Adv. Exp. Med. Biol. 2018, 1078, 49–78. [Google Scholar] [CrossRef] [PubMed]
- Dhandayuthapani, B.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. Polymeric Scaffolds in Tissue Engineering Application: A Review. Int. J. Polym. Sci. 2011, 2011, 290602. [Google Scholar] [CrossRef]
- Xing, H.; Lee, H.; Luo, L.; Kyriakides, T.R. Extracellular matrix-derived biomaterials in engineering cell function. Biotechnol. Adv. 2020, 42, 107421. [Google Scholar] [CrossRef]
- Burdick, J.A.; Vunjak-Novakovic, G. Engineered microenvironments for controlled stem cell differentiation. Tissue Eng. Part A 2009, 15, 205–219. [Google Scholar] [CrossRef] [PubMed]
- Takitoh, T.; Bessho, M.; Hirose, M.; Ohgushi, H.; Mori, H.; Hara, M. Gamma-cross-linked nonfibrillar collagen gel as a scaffold for osteogenic differentiation of mesenchymal stem cells. J. Biosci. Bioeng. 2015, 119, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zhu, Y.; Laslett, A.L.; Chan, H.F. Hepatic Differentiation of Stem Cells in 2D and 3D Biomaterial Systems. Bioengineering 2020, 7, 47. [Google Scholar] [CrossRef] [PubMed]
- Dawson, E.; Mapili, G.; Erickson, K.; Taqvi, S.; Roy, K. Biomaterials for stem cell differentiation. Adv. Drug Deliv. Rev. 2008, 60, 215–228. [Google Scholar] [CrossRef]
- Abdulghani, S.; Mitchell, G.R. Biomaterials for In Situ Tissue Regeneration: A Review. Biomolecules 2019, 9, 750. [Google Scholar] [CrossRef]
- Engler, A.J.; Sen, S.; Sweeney, H.L.; Discher, D.E. Matrix elasticity directs stem cell lineage specification. Cell 2006, 126, 677–689. [Google Scholar] [CrossRef]
- Singh, A.; Elisseeff, J. Biomaterials for stem cell differentiation. J. Mater. Chem. 2010, 20, 8832–8847. [Google Scholar] [CrossRef]
- Cross, V.L.; Zheng, Y.; Won Choi, N.; Verbridge, S.S.; Sutermaster, B.A.; Bonassar, L.J.; Fischbach, C.; Stroock, A.D. Dense type I collagen matrices that support cellular remodeling and microfabrication for studies of tumor angiogenesis and vasculogenesis in vitro. Biomaterials 2010, 31, 8596–8607. [Google Scholar] [CrossRef]
- Xu, Y.; Chen, C.; Hellwarth, P.B.; Bao, X. Biomaterials for stem cell engineering and biomanufacturing. Bioact. Mater. 2019, 4, 366–379. [Google Scholar] [CrossRef]
- Chattopadhyay, S.; Raines, R.T. Review collagen-based biomaterials for wound healing. Biopolymers 2014, 101, 821–833. [Google Scholar] [CrossRef]
- Liu, X.; Long, X.; Liu, W.; Zhao, Y.; Hayashi, T.; Yamato, M.; Mizuno, K.; Fujisaki, H.; Hattori, S.; Tashiro, S.I.; et al. Type I collagen induces mesenchymal cell differentiation into myofibroblasts through YAP-induced TGF-β1 activation. Biochimie 2018, 150, 110–130. [Google Scholar] [CrossRef] [PubMed]
- Even-Ram, S.; Artym, V.; Yamada, K.M. Matrix control of stem cell fate. Cell 2006, 126, 645–647. [Google Scholar] [CrossRef] [PubMed]
- Grier, W.G.; Moy, A.S.; Harley, B.A. Cyclic tensile strain enhances human mesenchymal stem cell Smad 2/3 activation and tenogenic differentiation in anisotropic collagen-glycosaminoglycan scaffolds. Eur. Cell Mater. 2017, 33, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Grier, W.K.; Tiffany, A.S.; Ramsey, M.D.; Harley, B.A.C. Incorporating β-cyclodextrin into collagen scaffolds to sequester growth factors and modulate mesenchymal stem cell activity. Acta Biomater. 2018, 76, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Ryan, E.J.; Ryan, A.J.; González-Vázquez, A.; Philippart, A.; Ciraldo, F.E.; Hobbs, C.; Nicolosi, V.; Boccaccini, A.R.; Kearney, C.J.; O’Brien, F.J. Collagen scaffolds functionalised with copper-eluting bioactive glass reduce infection and enhance osteogenesis and angiogenesis both in vitro and in vivo. Biomaterials 2019, 197, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.Y.; Agarwal, R.; Ko, K.R.; Ruthven, M.; Sarhan, H.T.; Frampton, J.P. Templated Assembly of Collagen Fibers Directs Cell Growth in 2D and 3D. Sci. Rep. 2017, 7, 9628. [Google Scholar] [CrossRef]
- Berger, A.J.; Linsmeier, K.M.; Kreeger, P.K.; Masters, K.S. Decoupling the effects of stiffness and fiber density on cellular behaviors via an interpenetrating network of gelatin-methacrylate and collagen. Biomaterials 2017, 141, 125–135. [Google Scholar] [CrossRef]
- Wang, M.; Yang, Y.; Han, L.; Han, S.; Liu, N.; Xu, F.; Li, F. Effect of three-dimensional ECM stiffness on cancer cell migration through regulating cell volume homeostasis. Biochem. Biophys. Res. Commun. 2020, 528, 459–465. [Google Scholar] [CrossRef]
- Branco da Cunha, C.; Klumpers, D.D.; Li, W.A.; Koshy, S.T.; Weaver, J.C.; Chaudhuri, O.; Granja, P.L.; Mooney, D.J. Influence of the stiffness of three-dimensional alginate/collagen-I interpenetrating networks on fibroblast biology. Biomaterials 2014, 35, 8927–8936. [Google Scholar] [CrossRef]
- Catoira, M.C.; Fusaro, L.; Di Francesco, D.; Ramella, M.; Boccafoschi, F. Overview of natural hydrogels for regenerative medicine applications. J. Mater. Sci. Mater. Med. 2019, 30, 115. [Google Scholar] [CrossRef]
- Daamen, W.F.; Veerkamp, J.H.; van Hest, J.C.M.; van Kuppevelt, T.H. Elastin as a biomaterial for tissue engineering. Biomaterials 2007, 28, 4378–4398. [Google Scholar] [CrossRef] [PubMed]
- Del Prado Audelo, M.L.; Mendoza-Muñoz, N.; Escutia-Guadarrama, L.; Giraldo-Gomez, D.; González-Torres, M.; Florán, B.; Cortés, H.; Leyva-Gomez, G. Recent Advances in Elastin-Based Biomaterial. J. Pharm. Pharm. Sci. 2020, 23, 314–332. [Google Scholar] [CrossRef] [PubMed]
- Skopinska-Wisniewska, J.; Kuderko, J.; Bajek, A.; Maj, M.; Sionkowska, A.; Ziegler-Borowska, M. Collagen/elastin hydrogels cross-linked by squaric acid. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 60, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Riopel, M.; Trinder, M.; Wang, R. Fibrin, a scaffold material for islet transplantation and pancreatic endocrine tissue engineering. Tissue Eng. Part B Rev. 2015, 21, 34–44. [Google Scholar] [CrossRef]
- Park, C.H.; Woo, K.M. Fibrin-Based Biomaterial Applications in Tissue Engineering and Regenerative Medicine. Adv. Exp. Med. Biol. 2018, 1064, 253–261. [Google Scholar] [CrossRef]
- Chrobak, M.O.; Hansen, K.J.; Gershlak, J.R.; Vratsanos, M.; Kanellias, M.; Gaudette, G.R.; Pins, G.D. Design of a Fibrin Microthread-Based Composite Layer for Use in a Cardiac Patch. ACS Biomater. Sci. Eng. 2017, 3, 1394–1403. [Google Scholar] [CrossRef]
- Echave, M.C.; Saenz del Burgo, L.; Pedraz, J.L.; Orive, G. Gelatin as Biomaterial for Tissue Engineering. Curr. Pharm. Des. 2017, 23, 3567–3584. [Google Scholar] [CrossRef] [PubMed]
- Bello, A.B.; Kim, D.; Park, H.; Lee, S.H. Engineering and Functionalization of Gelatin Biomaterials: From Cell Culture to Medical Applications. Tissue Eng. Part B Rev. 2020, 26, 164–180. [Google Scholar] [CrossRef]
- Pignatello, R. Biomaterials: Applications for Nanomedicine; BoD–Books on Demand: Norderstedt, Germany, 2011. [Google Scholar]
- Tajima, S.; Tabata, Y. Preparation and functional evaluation of cell aggregates incorporating gelatin microspheres with different degradabilities. J. Tissue Eng. Regen. Med. 2013, 7, 801–811. [Google Scholar] [CrossRef]
- García Cruz, D.M.; Sardinha, V.; Escobar Ivirico, J.L.; Mano, J.F.; Gómez Ribelles, J.L. Gelatin microparticles aggregates as three-dimensional scaffolding system in cartilage engineering. J. Mater. Sci. Mater. Med. 2013, 24, 503–513. [Google Scholar] [CrossRef]
- Kozlowska, J.; Stachowiak, N.; Sionkowska, A. Collagen/gelatin/hydroxyethyl cellulose composites containing microspheres based on collagen and gelatin: Design and evaluation. Polymers 2018, 10, 456. [Google Scholar] [CrossRef] [PubMed]
- Hiew, V.V.; Simat, S.F.B.; Teoh, P.L. The Advancement of Biomaterials in Regulating Stem Cell Fate. Stem Cell Rev. Rep. 2018, 14, 43–57. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Kim, H.J.; Vunjak-Novakovic, G.; Kaplan, D.L. Stem cell-based tissue engineering with silk biomaterials. Biomaterials 2006, 27, 6064–6082. [Google Scholar] [CrossRef] [PubMed]
- Kundu, B.; Rajkhowa, R.; Kundu, S.C.; Wang, X. Silk fibroin biomaterials for tissue regenerations. Adv. Drug Deliv. Rev. 2013, 65, 457–470. [Google Scholar] [CrossRef]
- Uebersax, L.; Merkle, H.P.; Meinel, L. Insulin-like growth factor I releasing silk fibroin scaffolds induce chondrogenic differentiation of human mesenchymal stem cells. J. Control. Release 2008, 127, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Gregory, D.A.; Tomeh, M.A.; Zhao, X. Silk Fibroin as a Functional Biomaterial for Tissue Engineering. Int. J. Mol. Sci. 2021, 22, 1499. [Google Scholar] [CrossRef]
- Pérez, S.; Bonnardel, F.; Lisacek, F.; Imberty, A.; Ricard Blum, S.; Makshakova, O. GAG-DB, the New Interface of the Three-Dimensional Landscape of Glycosaminoglycans. Biomolecules 2020, 10, 1660. [Google Scholar] [CrossRef]
- Berdiaki, A.; Neagu, M.; Giatagana, E.M.; Kuskov, A.; Tsatsakis, A.M.; Tzanakakis, G.N.; Nikitovic, D. Glycosaminoglycans: Carriers and Targets for Tailored Anti-Cancer Therapy. Biomolecules 2021, 11, 395. [Google Scholar] [CrossRef]
- Scott, R.A.; Panitch, A. Glycosaminoglycans in biomedicine. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnology 2013, 5, 388–398. [Google Scholar] [CrossRef]
- Shan, J.; Chi, Q.; Wang, H.; Huang, Q.; Yang, L.; Yu, G.; Zou, X. Mechanosensing of cells in 3D gel matrices based on natural and synthetic materials. Cell Biol. Int. 2014, 38, 1233–1243. [Google Scholar] [CrossRef]
- Ruvinov, E.; Cohen, S. Alginate biomaterial for the treatment of myocardial infarction: Progress, translational strategies, and clinical outlook: From ocean algae to patient bedside. Adv. Drug Deliv. Rev. 2016, 96, 54–76. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.C.; Chang, Y.H. Differentiation of induced pluripotent stem cells toward neurons in hydrogel biomaterials. Colloids Surf. B Biointerfaces 2013, 102, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Ganapathy, K.; Andl, T.; Wang, Z.; Copland, J.A.; Chakrabarti, R.; Florczyk, S.J. 3D porous chitosan-alginate scaffold stiffness promotes differential responses in prostate cancer cell lines. Biomaterials 2019, 217, 119311. [Google Scholar] [CrossRef] [PubMed]
- Fakhri, E.; Eslami, H.; Maroufi, P.; Pakdel, F.; Taghizadeh, S.; Ganbarov, K.; Yousefi, M.; Tanomand, A.; Yousefi, B.; Mahmoudi, S.; et al. Chitosan biomaterials application in dentistry. Int. J. Biol. Macromol. 2020, 162, 956–974. [Google Scholar] [CrossRef] [PubMed]
- Le, M.N.; Xu, K.; Wang, Z.; Beverung, S.; Steward, R.L.; Florczyk, S.J. Evaluation of the effect of 3D porous Chitosan-alginate scaffold stiffness on breast cancer proliferation and migration. J. Biomed. Mater. Res. A 2021, 109, 1990–2000. [Google Scholar] [CrossRef]
- Aho, A.J.; Rekola, J.; Matinlinna, J.; Gunn, J.; Tirri, T.; Viitaniemi, P.; Vallittu, P. Natural composite of wood as replacement material for ostechondral bone defects. J. Biomed. Mater. Res. Part B Appl. Biomater. 2007, 83, 64–71. [Google Scholar] [CrossRef]
- Rekola, J.; Aho, A.J.; Gunn, J.; Matinlinna, J.; Hirvonen, J.; Viitaniemi, P.; Vallittu, P.K. The effect of heat treatment of wood on osteoconductivity. Acta Biomater. 2009, 5, 1596–1604. [Google Scholar] [CrossRef]
- Rekola, J.; Lassila, L.V.; Hirvonen, J.; Lahdenperä, M.; Grenman, R.; Aho, A.J.; Vallittu, P.K. Effects of heat treatment of wood on hydroxylapatite type mineral precipitation and biomechanical properties in vitro. J. Mater. Sci. Mater. Med. 2010, 21, 2345–2354. [Google Scholar] [CrossRef]
- Rekola, J.; Lassila, L.V.; Nganga, S.; Ylä-Soininmäki, A.; Fleming, G.J.; Grenman, R.; Aho, A.J.; Vallittu, P.K. Effect of heat treatment of wood on the morphology, surface roughness and penetration of simulated and human blood. Bio-Med. Mater. Eng. 2014, 24, 1595–1607. [Google Scholar] [CrossRef]
- Josan, C.; Kakar, S.; Raha, S. Matrigel® enhances 3T3-L1 cell differentiation. Adipocyte 2021, 10, 361–377. [Google Scholar] [CrossRef]
- Aisenbrey, E.A.; Murphy, W.L. Synthetic alternatives to Matrigel. Nat. Rev. Mater. 2020, 5, 539–551. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, A.; Kale, A.; Novozhilova, E.; Olivius, P. The Effects of Matrigel® on the Survival and Differentiation of a Human Neural Progenitor Dissociated Sphere Culture. Anat. Rec. 2020, 303, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Ji, R.; Zhang, N.; You, N.; Li, Q.; Liu, W.; Jiang, N.; Liu, J.; Zhang, H.; Wang, D.; Tao, K.; et al. The differentiation of MSCs into functional hepatocyte-like cells in a liver biomatrix scaffold and their transplantation into liver-fibrotic mice. Biomaterials 2012, 33, 8995–9008. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.A.; Sampaio, L.C.; Ferdous, Z.; Gobin, A.S.; Taite, L.J. Decellularized matrices in regenerative medicine. Acta Biomater. 2018, 74, 74–89. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Pérez, J.; Ahearne, M. The impact of decellularization methods on extracellular matrix derived hydrogels. Sci. Rep. 2019, 9, 14933. [Google Scholar] [CrossRef]
- Spang, M.T.; Christman, K.L. Extracellular matrix hydrogel therapies: In vivo applications and development. Acta Biomater. 2018, 68, 1–14. [Google Scholar] [CrossRef]
- Kargar-Abarghouei, E.; Vojdani, Z.; Hassanpour, A.; Alaee, S.; Talaei-Khozani, T. Characterization, recellularization, and transplantation of rat decellularized testis scaffold with bone marrow-derived mesenchymal stem cells. Stem Cell Res. Ther. 2018, 9, 324. [Google Scholar] [CrossRef]
- Liška, V.; Moulisová, V.; Pálek, R.; Rosendorf, J.; Červenková, L.; Bolek, L.; Třeška, V. Repopulation of decellularized pig scaffolds: Promising approach for liver tissue engineering. Rozhl. Chir. 2019, 98, 388–393. [Google Scholar]
- Yesmin, S.; Paget, M.B.; Murray, H.E.; Downing, R. Bio-scaffolds in organ-regeneration: Clinical potential and current challenges. Curr. Res. Transl. Med. 2017, 65, 103–113. [Google Scholar] [CrossRef]
- O’Neill, J.D.; Freytes, D.O.; Anandappa, A.J.; Oliver, J.A.; Vunjak-Novakovic, G.V. The regulation of growth and metabolism of kidney stem cells with regional specificity using extracellular matrix derived from kidney. Biomaterials 2013, 34, 9830–9841. [Google Scholar] [CrossRef]
- Bonandrini, B.; Figliuzzi, M.; Papadimou, E.; Morigi, M.; Perico, N.; Casiraghi, F.; Dipl, C.; Sangalli, F.; Conti, S.; Benigni, A.; et al. Recellularization of well-preserved acellular kidney scaffold using embryonic stem cells. Tissue Eng. Part A 2014, 20, 1486–1498. [Google Scholar] [CrossRef] [PubMed]
- Leclerc, C.J.; Cooper, T.T.; Bell, G.I.; Lajoie, G.A.; Flynn, L.E.; Hess, D.A. Decellularized adipose tissue scaffolds guide hematopoietic differentiation and stimulate vascular regeneration in a hindlimb ischemia model. Biomaterials 2021, 274, 120867. [Google Scholar] [CrossRef] [PubMed]
- Rasti, H.; Saghiri, N.; Baharara, J.; Mahdavi-Shahri, N.; Marjani, M.; Alavi, S.H.; Hoseini, B. Differentiation of blastema cells in decellularized bladder scaffold in vitro. Zahedan J. Res. Med. Sci. 2015, 17. [Google Scholar] [CrossRef]
- Navarro-Tableros, V.; Herrera Sanchez, M.B.; Figliolini, F.; Romagnoli, R.; Tetta, C.; Camussi, G. Recellularization of rat liver scaffolds by human liver stem cells. Tissue Eng. Part A 2015, 21, 1929–1939. [Google Scholar] [CrossRef]
- Rajabi, S.; Aghdami, N.; Varzideh, F.; Parchehbaf-Kashani, M.; Nobakht Lahrood, F. Decellularized muscle-derived hydrogels support in vitro cardiac microtissue fabrication. J. Biomed. Mater. Res. B Appl. Biomater. 2020, 108, 3302–3310. [Google Scholar] [CrossRef]
- Coronado, R.E.; Somaraki-Cormier, M.; Ong, J.L.; Halff, G.A. Hepatocyte-like cells derived from human amniotic epithelial, bone marrow, and adipose stromal cells display enhanced functionality when cultured on decellularized liver substrate. Stem Cell Res. 2019, 38, 101471. [Google Scholar] [CrossRef]
- Anasiz, Y.; Ozgul, R.K.; Uckan-Cetinkaya, D. A New Chapter for Mesenchymal Stem Cells: Decellularized Extracellular Matrices. Stem Cell Rev. Rep. 2017, 13, 587–597. [Google Scholar] [CrossRef]
- Rao Pattabhi, S.; Martinez, J.S.; Keller, T.C.S. 3rd. Decellularized ECM effects on human mesenchymal stem cell stemness and differentiation. Differentiation 2014, 88, 131–143. [Google Scholar] [CrossRef]
- French, K.M.; Boopathy, A.V.; DeQuach, J.A.; Chingozha, L.; Lu, H.; Christman, K.L.; Davis, M.E. A naturally derived cardiac extracellular matrix enhances cardiac progenitor cell behavior in vitro. Acta Biomater. 2012, 8, 4357–4364. [Google Scholar] [CrossRef]
- Viswanath, A.; Vanacker, J.; Germain, L.; Leprince, J.G.; Diogenes, A.; Shakesheff, K.M.; White, L.J.; des Rieux, A. Extracellular matrix-derived hydrogels for dental stem cell delivery. J. Biomed. Mater. Res. A 2017, 105, 319–328. [Google Scholar] [CrossRef]
- Hoch, A.I.; Mittal, V.; Mitra, D.; Vollmer, N.; Zikry, C.A.; Leach, J.K. Cell-secreted matrices perpetuate the bone-forming phenotype of differentiated mesenchymal stem cells. Biomaterials 2016, 74, 178–187. [Google Scholar] [CrossRef] [PubMed]
- García-Gareta, E.; Abduldaiem, Y.; Sawadkar, P.; Kyriakidis, C.; Lali, F.; Greco, K.V. Decellularised scaffolds: Just a framework? Current knowledge and future directions. J. Tissue Eng. 2020, 11, 2041731420942903. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, R.; Kumar, S.; Pandey, R.; Mahajan, A.; Nandana, D.; Katti, D.S.; Mehrotra, D. Polycaprolactone as biomaterial for bone scaffolds: Review of literature. J. Oral Biol. Craniofacial Res. 2020, 10, 381–388. [Google Scholar] [CrossRef]
- Jang, H.Y.; Shin, J.Y.; Oh, S.H.; Byun, J.-H.; Lee, J.H. PCL/HA Hybrid Microspheres for Effective Osteogenic Differentiation and Bone Regeneration. ACS Biomater. Sci. Eng. 2020, 6, 5172–5180. [Google Scholar] [CrossRef] [PubMed]
- Mahsa Khatami, S.; Parivar, K.; Naderi Sohi, A.; Soleimani, M.; Hanaee-Ahvaz, H. Acetylated hyaluronic acid effectively enhances chondrogenic differentiation of mesenchymal stem cells seeded on electrospun PCL scaffolds. Tissue Cell 2020, 65, 101363. [Google Scholar] [CrossRef]
- Pan, L.; Pei, X.; He, R.; Wan, Q.; Wang, J. Multiwall carbon nanotubes/polycaprolactone composites for bone tissue engineering application. Colloids Surf. B Biointerfaces 2012, 93, 226–234. [Google Scholar] [CrossRef]
- Alcala-Orozco, C.R.; Mutreja, I.; Cui, X.; Hooper, G.J.; Lim, K.S.; Woodfield, T.B. Hybrid biofabrication of 3D osteoconductive constructs comprising Mg-based nanocomposites and cell-laden bioinks for bone repair. Bone 2021, 154, 116198. [Google Scholar] [CrossRef]
- Halabian, R.; Moridi, K.; Korani, M.; Ghollasi, M. Composite Nanoscaffolds Modified with Bio-ceramic Nanoparticles (Zn2SiO4) Prompted Osteogenic Differentiation of Human Induced Pluripotent Stem Cells. Int. J. Mol. Cell. Med. 2019, 8, 24. [Google Scholar] [CrossRef]
- Herrero-Herrero, M.; Alberdi-Torres, S.; González-Fernández, M.L.; Vilariño-Feltrer, G.; Rodríguez-Hernández, J.C.; Vallés-Lluch, A.; Villar-Suárez, V. Influence of chemistry and fiber diameter of electrospun PLA, PCL and their blend membranes, intended as cell supports, on their biological behavior. Polym. Test. 2021, 103, 107364. [Google Scholar] [CrossRef]
- Xu, T.; Yao, Q.; Miszuk, J.M.; Sanyour, H.J.; Hong, Z.; Sun, H.; Fong, H. Tailoring weight ratio of PCL/PLA in electrospun three-dimensional nanofibrous scaffolds and the effect on osteogenic differentiation of stem cells. Colloids Surf. B Biointerfaces 2018, 171, 31–39. [Google Scholar] [CrossRef]
- Baudequin, T.; Gaut, L.; Mueller, M.; Huepkes, A.; Glasmacher, B.; Duprez, D.; Bedoui, F.; Legallais, C. The Osteogenic and Tenogenic Differentiation Potential of C3H10T1/2 (Mesenchymal Stem Cell Model) Cultured on PCL/PLA Electrospun Scaffolds in the Absence of Specific Differentiation Medium. Materials 2017, 10, 1387. [Google Scholar] [CrossRef] [PubMed]
- Ghobeira, R.; Philips, C.; Liefooghe, L.; Verdonck, M.; Asadian, M.; Cools, P.; Declercq, H.; De Vos, W.H.; De Geyter, N.; Morent, R. Synergetic effect of electrospun PCL fiber size, orientation and plasma-modified surface chemistry on stem cell behavior. Appl. Surf. Sci. 2019, 485, 204–221. [Google Scholar] [CrossRef]
- Su, N.; Gao, P.L.; Wang, K.; Wang, J.Y.; Zhong, Y.; Luo, Y. Fibrous scaffolds potentiate the paracrine function of mesenchymal stem cells: A new dimension in cell-material interaction. Biomaterials 2017, 141, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Kai, D.; Liu, S.; Huang, X.; Heng, S.; Zhao, J.; Yu Chan, B.Q.; Loh, X.J.; Zhu, Y.; Mao, C.; et al. Mechanically cartilage-mimicking poly(PCL-PTHF urethane)/collagen nanofibers induce chondrogenesis by blocking NF–kappa B signaling pathway. Biomaterials 2018, 178, 281–292. [Google Scholar] [CrossRef]
- Yao, Q.; Cosme, J.G.L.; Xu, T.; Miszuk, J.M.; Picciani, P.H.S.; Fong, H.; Sun, H. Three dimensional electrospun PCL/PLA blend nanofibrous scaffolds with significantly improved stem cells osteogenic differentiation and cranial bone formation. Biomaterials 2017, 115, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Olvera, D.; Sathy, B.N.; Carroll, S.F.; Kelly, D.J. Modulating microfibrillar alignment and growth factor stimulation to regulate mesenchymal stem cell differentiation. Acta Biomater. 2017, 64, 148–160. [Google Scholar] [CrossRef]
- Santoro, M.; Shah, S.R.; Walker, J.L.; Mikos, A.G. Poly(Lactic Acid) nanofibrous scaffolds for tissue engineering. Adv. Drug Deliv. Rev. 2016, 107, 206. [Google Scholar] [CrossRef]
- Jaidev, L.R.; Chatterjee, K. Surface functionalization of 3D printed polymer scaffolds to augment stem cell response. Mater. Des. 2019, 161, 44–54. [Google Scholar] [CrossRef]
- Teixeira, B.N.; Aprile, P.; Mendonça, R.H.; Kelly, D.J.; Thiré, R.M.d.S.M. Evaluation of bone marrow stem cell response to PLA scaffolds manufactured by 3D printing and coated with polydopamine and type I collagen. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 107, 37–49. [Google Scholar] [CrossRef]
- Heo, D.N.; Castro, N.J.; Lee, S.-J.; Noh, H.; Zhu, W.; Zhang, L.G. Enhanced bone tissue regeneration using a 3D printed microstructure incorporated with a hybrid nano hydrogel. Nanoscale 2017, 9, 5055–5062. [Google Scholar] [CrossRef]
- Ojaghi, M.; Soleimanifar, F.; Kazemi, A.; Ghollasi, M.; Soleimani, M.; Nasoohi, N.; Enderami, S.E. Electrospun poly-l-lactic acid/polyvinyl alcohol nanofibers improved insulin-producing cell differentiation potential of human adipose-derived mesenchymal stem cells. J. Cell. Biochem. 2019, 120, 9917–9926. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Cai, Y.; MacLachlan, A.; Chen, P. Novel nanoplasmonic-structure-based integrated microfluidic biosensors for label-free in situ immune functional analysis: A review of recent progress. IEEE Nanotechnol. Mag. 2020, 14, 46-C3. [Google Scholar] [CrossRef] [PubMed]
- Birhanu, G.; Javar, H.A.; Seyedjafari, E.; Zandi-Karimi, A.; Telgerd, M.D. An improved surface for enhanced stem cell proliferation and osteogenic differentiation using electrospun composite PLLA/P123 scaffold. Artif. Cells Nanomed. Biotechnol. 2017, 46, 1274–1281. [Google Scholar] [CrossRef] [PubMed]
- Didekhani, R.; Sohrabi, M.R.; Seyedjafari, E.; Soleimani, M.; Hanaee-Ahvaz, H. Electrospun composite PLLA/Oyster shell scaffold enhances proliferation and osteogenic differentiation of stem cells. Biologicals 2018, 54, 33–38. [Google Scholar] [CrossRef]
- Souza, D.C.d.; Abreu, H.d.L.V.d.; Oliveira, P.V.d.; Capelo, L.P.; Passos-Bueno, M.R.; Catalani, L.H. A fast degrading PLLA composite with a high content of functionalized octacalcium phosphate mineral phase induces stem cells differentiation. J. Mech. Behav. Biomed. Mater. 2019, 93, 93–104. [Google Scholar] [CrossRef]
- Mahdavi, F.S.; Salehi, A.; Seyedjafari, E.; Mohammadi-Sangcheshmeh, A.; Ardeshirylajimi, A. Bioactive glass ceramic nanoparticles-coated poly(l-lactic acid) scaffold improved osteogenic differentiation of adipose stem cells in equine. Tissue Cell 2017, 49, 565–572. [Google Scholar] [CrossRef]
- Fattahi, Z.; Hasanzadeh, M. Nanotechnology-assisted microfluidic systems for chemical sensing, biosensing, and bioanalysis. Trac-Trends Anal. Chem. 2022, 152. [Google Scholar] [CrossRef]
- Shafiee, A.; Kehtari, M.; Zarei, Z.; Soleimani, M.; Varshochian, R.; Ahmadi, A.; Atyabi, F.; Dinarvand, R. An in situ hydrogel-forming scaffold loaded by PLGA microspheres containing carbon nanotube as a suitable niche for neural differentiation. Mater. Sci. Eng. C 2021, 120, 111739. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, B.; Kasoju, N.; Ma, J.; Yang, A.; Cui, Z.; Wang, H.; Ye, H. Differential and Interactive Effects of Substrate Topography and Chemistry on Human Mesenchymal Stem Cell Gene Expression. Int. J. Mol. Sci. 2018, 19, 2344. [Google Scholar] [CrossRef]
- Izadpanahi, M.; Seyedjafari, E.; Arefian, E.; Hamta, A.; Hosseinzadeh, S.; Kehtari, M.; Soleimani, M. Nanotopographical cues of electrospun PLLA efficiently modulate non-coding RNA network to osteogenic differentiation of mesenchymal stem cells during BMP signaling pathway. Mater. Sci. Eng. C 2018, 93, 686–703. [Google Scholar] [CrossRef]
- Khatib, M.E.; Mauro, A.; Mattia, M.D.; Wyrwa, R.; Schweder, M.; Ancora, M.; Lazzaro, F.; Berardinelli, P.; Valbonetti, L.; Giacinto, O.D.; et al. Electrospun PLGA Fiber Diameter and Alignment of Tendon Biomimetic Fleece Potentiate Tenogenic Differentiation and Immunomodulatory Function of Amniotic Epithelial Stem Cells. Cells 2020, 9, 1207. [Google Scholar] [CrossRef]
- Sperling, L.E.; Reis, K.P.; Pozzobon, L.G.; Girardi, C.S.; Pranke, P. Influence of random and oriented electrospun fibrous poly(lactic-co-glycolic acid) scaffolds on neural differentiation of mouse embryonic stem cells. J. Biomed. Mater. Res. Part A 2017, 105, 1333–1345. [Google Scholar] [CrossRef]
- Qi, Z.; Guo, W.; Zheng, S.; Fu, C.; Ma, Y.; Pan, S.; Liu, Q.; Yang, X. Enhancement of neural stem cell survival, proliferation and differentiation by IGF-1 delivery in graphene oxide-incorporated PLGA electrospun nanofibrous mats. RSC Adv. 2019, 9, 8315–8325. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, S.; Karkhaneh, A.; Soleimani, M.; Ardeshirylajimi, A.; Zonouzi, H.S.; Hanaee-Ahvaz, H. Enhanced chondrogenic differentiation of stem cells using an optimized electrospun nanofibrous PLLA/PEG scaffolds loaded with glucosamine. J. Biomed. Mater. Res. Part A 2017, 105, 2461–2474. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yan, H.; Shen, Y.; Tang, H.; Yi, B.; Qin, C.; Zhang, Y. Shape Memory and Osteogenesis Capabilities of the Electrospun Poly(3-Hydroxybutyrate-co-3-Hydroxyvalerate) Modified Poly(l-Lactide) Fibrous Mats. Tissue Eng. Part A 2021, 27, 142–152. Available online: https://home.liebertpub.com/tea (accessed on 1 December 2022). [CrossRef]
- Levy-Mishali, M.; Zoldan, J.; Levenberg, S. Effect of Scaffold Stiffness on Myoblast Differentiation. Tissue Eng. Part A 2009, 15, 935–944. [Google Scholar] [CrossRef]
- Sun, M.; Chi, G.; Li, P.; Lv, S.; Xu, J.; Xu, Z.; Xia, Y.; Tan, Y.; Xu, J.; Li, L.; et al. Effects of Matrix Stiffness on the Morphology, Adhesion, Proliferation and Osteogenic Differentiation of Mesenchymal Stem Cells. Int. J. Med. Sci. 2018, 15, 257. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Qiao, R.; Dao, J.; Su, J.; Jiang, C.; Wang, X.; Gao, M.; Zhong, J. Soybean Lecithin-Mediated Nanoporous PLGA Microspheres with Highly Entrapped and Controlled Released BMP-2 as a Stem Cell Platform. Small 2018, 14, 1800063. [Google Scholar] [CrossRef]
- Zhao, T.; Qi, Y.; Xiao, S.; Ran, J.; Wang, J.; Ghamor-Amegavi, E.P.; Zhou, X.; Li, H.; He, T.; Gou, Z.; et al. Integration of mesenchymal stem cell sheet and bFGF-loaded fibrin gel in knitted PLGA scaffolds favorable for tendon repair. J. Mater. Chem. B 2019, 7, 2201–2211. [Google Scholar] [CrossRef] [PubMed]
- Noh, M.; Kim, S.-H.; Kim, J.; Lee, J.-R.; Jeong, G.-J.; Yoon, J.-K.; Kang, S.; Bhang, S.H.; Yoon, H.H.; Lee, J.-C.; et al. Graphene oxide reinforced hydrogels for osteogenic differentiation of human adipose-derived stem cells. RSC Adv. 2017, 7, 20779–20788. [Google Scholar] [CrossRef]
- Amiri, B.; Ghollasi, M.; Shahrousvand, M.; Kamali, M.; Salimi, A. Osteoblast differentiation of mesenchymal stem cells on modified PES-PEG electrospun fibrous composites loaded with Zn2SiO4 bioceramic nanoparticles. Differentiation 2016, 92, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Nam, S.; Stowers, R.; Lou, J.; Xia, Y.; Chaudhuri, O. Varying PEG density to control stress relaxation in alginate-PEG hydrogels for 3D cell culture studies. Biomaterials 2019, 200, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Bi, X.; Maturavongsadit, P.; Tan, Y.; Watts, M.; Bi, E.; Kegley, Z.; Morton, S.; Lu, L.; Wang, Q.; Liang, A. Polyamidoamine dendrimer-PEG hydrogel and its mechanical property on differentiation of mesenchymal stem cells. Bio-Med. Mater. Eng. 2019, 30, 111–123. [Google Scholar] [CrossRef] [PubMed]
- Nachlas, A.L.Y.; Li, S.; Jha, R.; Singh, M.; Xu, C.; Davis, M.E. Human iPSC-derived mesenchymal stem cells encapsulated in PEGDA hydrogels mature into valve interstitial-like cells. Acta Biomater. 2018, 71, 235–246. [Google Scholar] [CrossRef]
- Shahrousvand, M.; Sadeghi, G.M.M.; Shahrousvand, E.; Ghollasi, M.; Salimi, A. Superficial physicochemical properties of polyurethane biomaterials as osteogenic regulators in human mesenchymal stem cells fates. Colloids Surf. B Biointerfaces 2017, 156, 292–304. [Google Scholar] [CrossRef]
- Shahrousvand, M.; Ghollasi, M.; Zarchi, A.A.K.; Salimi, A. Osteogenic differentiation of hMSCs on semi-interpenetrating polymer networks of polyurethane/poly(2-hydroxyethyl methacrylate)/cellulose nanowhisker scaffolds. Int. J. Biol. Macromol. 2019, 138, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Hazeri, Y.; Irani, S.; Zandi, M.; Pezeshki-Modaress, M. Polyvinyl alcohol/sulfated alginate nanofibers induced the neuronal differentiation of human bone marrow stem cells. Int. J. Biol. Macromol. 2020, 147, 946–953. [Google Scholar] [CrossRef]
- Mokhames, Z.; Rezaie, Z.; Ardeshirylajimi, A.; Basiri, A.; Taheri, M.; Omrani, M.D. Efficient smooth muscle cell differentiation of iPS cells on curcumin-incorporated chitosan/collagen/polyvinyl-alcohol nanofibers. Vitr. Cell. Dev. Biol. Anim. 2020, 56, 313–321. [Google Scholar] [CrossRef]
- Hou, Y.; Xie, W.; Achazi, K.; Cuellar-Camacho, J.L.; Melzig, M.F.; Chen, W.; Haag, R. Injectable degradable PVA microgels prepared by microfluidic technology for controlled osteogenic differentiation of mesenchymal stem cells. Acta Biomater. 2018, 77, 28–37. [Google Scholar] [CrossRef]
- Kajtez, J.; Nilsson, F.; Fiorenzano, A.; Parmar, M.; Emnéus, J. 3D biomaterial models of human brain disease. Neurochem. Int. 2021, 147, 105043. [Google Scholar] [CrossRef]
- Kazantseva, J.; Hussainova, I.; Ivanov, R.; Neuman, T.; Gasik, M. Hybrid graphene–ceramic nanofibre network for spontaneous neural differentiation of stem cells. Interface Focus 2018, 8, 20170037. [Google Scholar] [CrossRef] [PubMed]
- Hudecki, A.; Łyko-Morawska, D.; Kasprzycka, A.; Kazek-Kęsik, A.; Likus, W.; Hybiak, J.; Jankowska, K.; Kolano-Burian, A.; Włodarczyk, P.; Wolany, W.; et al. Comparison of Physicochemical, Mechanical, and (Micro-)Biological Properties of Sintered Scaffolds Based on Natural- and Synthetic Hydroxyapatite Supplemented with Selected Dopants. Int. J. Mol. Sci. 2022, 23, 4692. [Google Scholar] [CrossRef] [PubMed]
- Khaw, J.S.; Xue, R.; Cassidy, N.J.; Cartmell, S.H. Electrical stimulation of titanium to promote stem cell orientation, elongation and osteogenesis. Acta Biomater. 2021, 139, 204–217. [Google Scholar] [CrossRef]
- Hudecki, A.; Łyko-Morawska, D.; Likus, W.; Skonieczna, M.; Markowski, J.; Wilk, R.; Kolano-Burian, A.; Maziarz, W.; Adamska, J.; Łos, M.J. Composite Nanofibers Containing Multiwall Carbon Nanotubes as Biodegradable Membranes in Reconstructive Medicine. Nanomaterials 2019, 9, 63. [Google Scholar] [CrossRef] [PubMed]
- Cha, C.; Liechty, W.B.; Khademhosseini, A.; Peppas, N.A. Designing biomaterials to direct stem cell fate. ACS Nano 2012, 6, 9353–9358. [Google Scholar] [CrossRef]
- Watt, F.M.; Hogan, B.L. Out of Eden: Stem cells and their niches. Science 2000, 287, 1427–1430. [Google Scholar] [CrossRef]
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic stem cell lines derived from human blastocysts. Science 1998, 282, 1145–1147. [Google Scholar] [CrossRef]
- Willerth, S.M.; Sakiyama-Elbert, S.E. Combining stem cells and biomaterial scaffolds for constructing tissues and cell delivery. StemJournal 2019, 1, 1–25. [Google Scholar] [CrossRef]
- Higuchi, A.; Ling, Q.-D.; Hsu, S.-T.; Umezawa, A. Biomimetic cell culture proteins as extracellular matrices for stem cell differentiation. Chem. Rev. 2012, 112, 4507–4540. [Google Scholar] [CrossRef]
- Saha, K.; Pollock, J.F.; Schaffer, D.V.; Healy, K.E. Designing synthetic materials to control stem cell phenotype. Curr. Opin. Chem. Biol. 2007, 11, 381–387. [Google Scholar] [CrossRef]
- Ruoslahti, E. RGD and other recognition sequences for integrins. Annu. Rev. Cell Dev. Biol. 1996, 12, 697–715. [Google Scholar] [CrossRef]
- Weber, L.M.; Hayda, K.N.; Haskins, K.; Anseth, K.S. The effects of cell–matrix interactions on encapsulated β-cell function within hydrogels functionalized with matrix-derived adhesive peptides. Biomaterials 2007, 28, 3004–3011. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; McNamara, L.E.; Gadegaard, N.; Alakpa, E.V.; Burgess, K.V.; Meek, R.D.; Dalby, M.J. Nanotopographical induction of osteogenesis through adhesion, bone morphogenic protein cosignaling, and regulation of microRNAs. ACS Nano 2014, 8, 9941–9953. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; van Rijt, S. 2D biointerfaces to study stem cell–ligand interactions. Acta Biomater. 2021, 131, 80–96. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.N.; Sahoo, J.K.; McNamara, L.E.; Burgess, K.V.; Yang, J.; Alakpa, E.V.; Anderson, H.J.; Hay, J.; Turner, L.-A.; Yarwood, S.J. Dynamic surfaces for the study of mesenchymal stem cell growth through adhesion regulation. ACS Nano 2016, 10, 6667–6679. [Google Scholar] [CrossRef]
- Ito, Y. Growth factor engineering for biomaterials. ACS Biomater. Sci. Eng. 2019, 5, 5597–5609. [Google Scholar] [CrossRef]
- West-Livingston, L.N.; Park, J.; Lee, S.J.; Atala, A.; Yoo, J.J. The role of the microenvironment in controlling the fate of bioprinted stem cells. Chem. Rev. 2020, 120, 11056–11092. [Google Scholar] [CrossRef]
- Cheng, H.; Jiang, W.; Phillips, F.M.; Haydon, R.C.; Peng, Y.; Zhou, L.; Luu, H.H.; An, N.; Breyer, B.; Vanichakarn, P. Osteogenic activity of the fourteen types of human bone morphogenetic proteins (BMPs). JBJS 2003, 85, 1544–1552. [Google Scholar] [CrossRef]
- Urist, M.R.; Strates, B.S. Bone morphogenetic protein. J. Dent. Res. 1971, 50, 1392–1406. [Google Scholar] [CrossRef]
- Reddi, A.H.; Cunningham, N.S. Initiation and promotion of bone differentiation by bone morphogenetic proteins. J. Bone Miner. Res. 1993, 8, S499–S502. [Google Scholar] [CrossRef]
- Luu, H.H.; Song, W.X.; Luo, X.; Manning, D.; Luo, J.; Deng, Z.L.; Sharff, K.A.; Montag, A.G.; Haydon, R.C.; He, T.C. Distinct roles of bone morphogenetic proteins in osteogenic differentiation of mesenchymal stem cells. J. Orthop. Res. 2007, 25, 665–677. [Google Scholar] [CrossRef] [PubMed]
- Lou, J.; Xu, F.; Merkel, K.; Manske, P. Gene therapy: Adenovirus-mediated human bone morphogenetic protein-2 gene transfer induces mesenchymal progenitor cell proliferation and differentiation in vitro and bone formation in vivo. J. Orthop. Res. 1999, 17, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.C.N.; Chung, H.Y.; Tai, C.L.; Chen, P.K.T.; Lin, T.M.; Jeng, L.B. Repair of large cranial defects by hBMP-2 expressing bone marrow stromal cells: Comparison between alginate and collagen type I systems. J. Biomed. Mater. Res. Part A 2010, 94, 433–441. [Google Scholar]
- Fan, V.H.; Au, A.; Tamama, K.; Littrell, R.; Richardson, L.B.; Wright, J.W.; Wells, A.; Griffith, L.G. Tethered epidermal growth factor provides a survival advantage to mesenchymal stem cells. Stem Cells 2007, 25, 1241–1251. [Google Scholar] [CrossRef] [PubMed]
- Krampera, M.; Pasini, A.; Rigo, A.; Scupoli, M.T.; Tecchio, C.; Malpeli, G.; Scarpa, A.; Dazzi, F.; Pizzolo, G.; Vinante, F. HB-EGF/HER-1 signaling in bone marrow mesenchymal stem cells: Inducing cell expansion and reversibly preventing multilineage differentiation. Blood 2005, 106, 59–66. [Google Scholar] [CrossRef]
- Tamama, K.; Fan, V.H.; Griffith, L.G.; Blair, H.C.; Wells, A. Epidermal growth factor as a candidate for ex vivo expansion of bone marrow–derived mesenchymal stem cells. Stem Cells 2006, 24, 686–695. [Google Scholar] [CrossRef]
- Eswarakumar, V.; Lax, I.; Schlessinger, J. Cellular signaling by fibroblast growth factor receptors. Cytokine Growth Factor Rev. 2005, 16, 139–149. [Google Scholar] [CrossRef]
- Stewart, A.A.; Byron, C.R.; Pondenis, H.; Stewart, M.C. Effect of fibroblast growth factor-2 on equine mesenchymal stem cell monolayer expansion and chondrogenesis. Am. J. Vet. Res. 2007, 68, 941–945. [Google Scholar] [CrossRef]
- Tsutsumi, S.; Shimazu, A.; Miyazaki, K.; Pan, H.; Koike, C.; Yoshida, E.; Takagishi, K.; Kato, Y. Retention of multilineage differentiation potential of mesenchymal cells during proliferation in response to FGF. Biochem. Biophys. Res. Commun. 2001, 288, 413–419. [Google Scholar] [CrossRef]
- Forte, G.; Minieri, M.; Cossa, P.; Antenucci, D.; Sala, M.; Gnocchi, V.; Fiaccavento, R.; Carotenuto, F.; De Vito, P.; Baldini, P.M. Hepatocyte growth factor effects on mesenchymal stem cells: Proliferation, migration, and differentiation. Stem Cells 2006, 24, 23–33. [Google Scholar] [CrossRef]
- Laron, Z. Insulin-like growth factor 1 (IGF-1): A growth hormone. Mol. Pathol. 2001, 54, 311. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hu, Q.; Mansoor, A.; Lee, J.; Wang, Z.; Lee, T.; From, A.H.; Zhang, J. Bioenergetic and functional consequences of stem cell-based VEGF delivery in pressure-overloaded swine hearts. Am. J. Physiol.-Heart Circ. Physiol. 2006, 290, H1393–H1405. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Duffy, A.M.; Bouchier-Hayes, D.J.; Harmey, J.H. Vascular endothelial growth factor (VEGF) and its role in non-endothelial cells: Autocrine signalling by VEGF. In Madame Curie Bioscience Database [Internet]; Landes Bioscience: Austin, TX, USA, 2013. [Google Scholar]
- Youssef, A.; Aboalola, D.; Han, V.K. The roles of insulin-like growth factors in mesenchymal stem cell niche. Stem Cells Int. 2017, 2017, 9453108. [Google Scholar] [CrossRef] [PubMed]
- Pons, J.; Huang, Y.; Arakawa-Hoyt, J.; Washko, D.; Takagawa, J.; Ye, J.; Grossman, W.; Su, H. VEGF improves survival of mesenchymal stem cells in infarcted hearts. Biochem. Biophys. Res. Commun. 2008, 376, 419–422. [Google Scholar] [CrossRef] [PubMed]
- Hellstrom, M.; Kaln, M.; Lindahl, P.; Abramsson, A.; Betsholtz, C. Role of PDGF-B and PDGFR-beta in recruitment of vascular smooth muscle cells and pericytes during embryonic blood vessel formation in the mouse. Development 1999, 126, 3047–3055. [Google Scholar] [CrossRef]
- Hu, K.; Olsen, B.R. Osteoblast-derived VEGF regulates osteoblast differentiation and bone formation during bone repair. J. Clin. Investig. 2016, 126, 509–526. [Google Scholar] [CrossRef]
- Klemm, D.J.; Leitner, J.W.; Watson, P.; Nesterova, A.; Reusch, J.E.-B.; Goalstone, M.L.; Draznin, B. Insulin-induced adipocyte differentiation: Activation of CREB rescues adipogenesis from the arrest caused by inhibition of prenylation. J. Biol. Chem. 2001, 276, 28430–28435. [Google Scholar] [CrossRef]
- Krausgrill, B.; Vantler, M.; Burst, V.; Raths, M.; Halbach, M.; Frank, K.; Schynkowski, S.; Schenk, K.; Hescheler, J.; Rosenkranz, S. Influence of cell treatment with PDGF-BB and reperfusion on cardiac persistence of mononuclear and mesenchymal bone marrow cells after transplantation into acute myocardial infarction in rats. Cell Transplant. 2009, 18, 847–853. [Google Scholar] [CrossRef]
- Andrae, J.; Gallini, R.; Betsholtz, C. Role of platelet-derived growth factors in physiology and medicine. Genes Dev. 2008, 22, 1276–1312. [Google Scholar] [CrossRef]
- Chase, L.G.; Lakshmipathy, U.; Solchaga, L.A.; Rao, M.S.; Vemuri, M.C. A novel serum-free medium for the expansion of human mesenchymal stem cells. Stem Cell Res. Ther. 2010, 1, 8. [Google Scholar] [CrossRef]
- Clark, D.A.; Coker, R. Transforming growth factor-beta (TGF-beta). Int. J. Biochem. Cell Biol. 1998, 30, 293–298. [Google Scholar] [CrossRef]
- Moustakas, A.; Pardali, K.; Gaal, A.; Heldin, C.-H. Mechanisms of TGF-β signaling in regulation of cell growth and differentiation. Immunol. Lett. 2002, 82, 85–91. [Google Scholar] [CrossRef]
- Longobardi, L.; ORear, L.; Aakula, S.; Johnstone, B.; Shimer, K.; Chytil, A.; Horton, W.A.; Moses, H.L.; Spagnoli, A. Effect of IGF-I in the chondrogenesis of bone marrow mesenchymal stem cells in the presence or absence of TGF-beta signaling. J. Bone Miner. Res. 2006, 21, 626–636. [Google Scholar] [CrossRef] [PubMed]
- McKinnon, R.D.; Piras, G.; Ida Jr, J.A.; Dubois-Dalcq, M. A role for TGF-beta in oligodendrocyte differentiation. J. Cell Biol. 1993, 121, 1397–1407. [Google Scholar] [CrossRef] [PubMed]
- Weiss, S.; Hennig, T.; Bock, R.; Steck, E.; Richter, W. Impact of growth factors and PTHrP on early and late chondrogenic differentiation of human mesenchymal stem cells. J. Cell. Physiol. 2010, 223, 84–93. [Google Scholar] [CrossRef]
- Komiya, Y.; Habas, R. Wnt signal transduction pathways. Organogenesis 2008, 4, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Baksh, D.; Boland, G.M.; Tuan, R.S. Cross-talk between Wnt signaling pathways in human mesenchymal stem cells leads to functional antagonism during osteogenic differentiation. J. Cell. Biochem. 2007, 101, 1109–1124. [Google Scholar] [CrossRef]
- Visweswaran, M.; Pohl, S.; Arfuso, F.; Newsholme, P.; Dilley, R.; Pervaiz, S.; Dharmarajan, A. Multi-lineage differentiation of mesenchymal stem cells–To Wnt, or not Wnt. Int. J. Biochem. Cell Biol. 2015, 68, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Seims, K.B.; Hunt, N.K.; Chow, L.W. Strategies to Control or Mimic Growth Factor Activity for Bone, Cartilage, and Osteochondral Tissue Engineering. Bioconjugate Chem. 2021, 32, 861–878. [Google Scholar] [CrossRef]
- Sun, X.; Wang, J.; Wang, Y.; Huang, C.; Yang, C.; Chen, M.; Chen, L.; Zhang, Q. Scaffold with orientated microtubule structure containing polylysine-heparin sodium nanoparticles for the controlled release of TGF-β1 in cartilage tissue engineering. ACS Appl. Bio Mater. 2018, 1, 2030–2040. [Google Scholar] [CrossRef]
- Guan, Z.-Y.; Wu, C.-Y.; Chen, H.-Y. Stepwise and programmable cell differentiation pathways of controlled functional biointerfaces. ACS Biomater. Sci. Eng. 2017, 3, 1815–1821. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, A.; Ling, Q.-D.; Chang, Y.; Hsu, S.-T.; Umezawa, A. Physical cues of biomaterials guide stem cell differentiation fate. Chem. Rev. 2013, 113, 3297–3328. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Villa-Diaz, L.G.; Sun, Y.; Weng, S.; Kim, J.K.; Lam, R.H.; Han, L.; Fan, R.; Krebsbach, P.H.; Fu, J. Nanotopography influences adhesion, spreading, and self-renewal of human embryonic stem cells. ACS Nano 2012, 6, 4094–4103. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, F.; Na, S.; Li, D.; Poh, Y.-C.; Tanaka, T.S.; Wang, F.; Wang, N. Material properties of the cell dictate stress-induced spreading and differentiation in embryonic stem cells. Nat. Mater. 2010, 9, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Wang, X.; Zeng, L.; Wu, Z.L.; Guo, H.; Hourdet, D. Stimuli-Responsive Toughening of Hydrogels. Chem. Mater. 2021, 33, 7633–7656. [Google Scholar] [CrossRef]
- Zhang, J.; Wehrle, E.; Vetsch, J.R.; Paul, G.R.; Rubert, M.; Müller, R. Alginate dependent changes of physical properties in 3D bioprinted cell-laden porous scaffolds affect cell viability and cell morphology. Biomed. Mater. 2019, 14, 065009. [Google Scholar] [CrossRef]
- Di Giuseppe, M.; Law, N.; Webb, B.; Macrae, R.A.; Liew, L.J.; Sercombe, T.B.; Dilley, R.J.; Doyle, B.J. Mechanical behaviour of alginate-gelatin hydrogels for 3D bioprinting. J. Mech. Behav. Biomed. Mater. 2018, 79, 150–157. [Google Scholar] [CrossRef]
- Floren, M.; Bonani, W.; Dharmarajan, A.; Motta, A.; Migliaresi, C.; Tan, W. Human mesenchymal stem cells cultured on silk hydrogels with variable stiffness and growth factor differentiate into mature smooth muscle cell phenotype. Acta Biomater. 2016, 31, 156–166. [Google Scholar] [CrossRef]
- Sanz-Fraile, H.; Amoros, S.; Mendizabal, I.; Galvez-Monton, C.; Prat-Vidal, C.; Bayes-Genis, A.; Navajas, D.; Farre, R.; Otero, J. Silk-reinforced collagen hydrogels with raised multiscale stiffness for mesenchymal cells 3D culture. Tissue Eng. Part A 2020, 26, 358–370. [Google Scholar] [CrossRef]
- Buitrago, J.O.; Patel, K.D.; El-Fiqi, A.; Lee, J.-H.; Kundu, B.; Lee, H.-H.; Kim, H.-W. Silk fibroin/collagen protein hybrid cell-encapsulating hydrogels with tunable gelation and improved physical and biological properties. Acta Biomater. 2018, 69, 218–233. [Google Scholar] [CrossRef]
- Lu, X.; Ding, Z.; Xu, F.; Lu, Q.; Kaplan, D.L. Subtle regulation of scaffold stiffness for the optimized control of cell behavior. ACS Appl. Bio Mater. 2019, 2, 3108–3119. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Liu, S.; Yao, D.; Ding, Z.; Fan, Z.; Lu, Q.; Kaplan, D.L. Fabrication of silk scaffolds with nanomicroscaled structures and tunable stiffness. Biomacromolecules 2017, 18, 2073–2079. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ding, Z.; Lu, G.; Wang, J.; Wang, L.; Lu, Q. Amorphous silk fibroin nanofiber hydrogels with enhanced mechanical properties. Macromol. Biosci. 2019, 19, 1900326. [Google Scholar] [CrossRef]
- Suresh, S.N.; Chakravorty, A.; Giridharan, M.; Garimella, L.; Manjithaya, R. Pharmacological Tools to Modulate Autophagy in Neurodegenerative Diseases. J. Mol. Biol. 2020, 432, 2822–2842. [Google Scholar] [CrossRef]
- Lin, C.H.; Su, J.J.M.; Lee, S.Y.; Lin, Y.M. Stiffness modification of photopolymerizable gelatin-methacrylate hydrogels influences endothelial differentiation of human mesenchymal stem cells. J. Tissue Eng. Regen. Med. 2018, 12, 2099–2111. [Google Scholar] [CrossRef]
- Kim, C.; Young, J.L.; Holle, A.W.; Jeong, K.; Major, L.G.; Jeong, J.H.; Aman, Z.M.; Han, D.-W.; Hwang, Y.; Spatz, J.P. Stem cell mechanosensation on gelatin methacryloyl (GelMA) stiffness gradient hydrogels. Ann. Biomed. Eng. 2020, 48, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.-N.; Dobre, O.; Richards, D.; Ballestrem, C.; Curran, J.M.; Hunt, J.A.; Richardson, S.M.; Swift, J.; Wong, L.S. Photoresponsive hydrogels with photoswitchable mechanical properties allow time-resolved analysis of cellular responses to matrix stiffening. ACS Appl. Mater. Interfaces 2018, 10, 7765–7776. [Google Scholar] [CrossRef]
- Wong, S.H.D.; Wong, W.K.R.; Lai, C.H.N.; Oh, J.; Li, Z.; Chen, X.; Yuan, W.; Bian, L. Soft polymeric matrix as a macroscopic cage for magnetically modulating reversible nanoscale ligand presentation. Nano Lett. 2020, 20, 3207–3216. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Jung, H.J.; Wong, D.S.H.; Kim, S.K.; Lin, S.; Chan, K.F.; Zhang, L.; Li, G.; Dravid, V.P.; Bian, L. Remote control of heterodimeric magnetic nanoswitch regulates the adhesion and differentiation of stem cells. J. Am. Chem. Soc. 2018, 140, 5909–5913. [Google Scholar] [CrossRef]
- Dobson, J. Remote control of cellular behaviour with magnetic nanoparticles. Nat. Nanotechnol. 2008, 3, 139–143. [Google Scholar] [CrossRef]
- Rössler, K.; Donat, M.; Lanzenberger, R.; Novak, K.; Geissler, A.; Gartus, A.; Tahamtan, A.; Milakara, D.; Czech, T.; Barth, M. Evaluation of preoperative high magnetic field motor functional MRI (3 Tesla) in glioma patients by navigated electrocortical stimulation and postoperative outcome. J. Neurol. Neurosurg. Psychiatry 2005, 76, 1152–1157. [Google Scholar] [CrossRef]
- Colombo, M.; Carregal-Romero, S.; Casula, M.F.; Gutiérrez, L.; Morales, M.P.; Böhm, I.B.; Heverhagen, J.T.; Prosperi, D.; Parak, W.J. Biological applications of magnetic nanoparticles. Chem. Soc. Rev. 2012, 41, 4306–4334. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Wong, D.S.H.; Yan, X.; Jung, H.J.; Kim, S.; Lin, S.; Wei, K.; Li, G.; Dravid, V.P.; Bian, L. Remote control of multimodal nanoscale ligand oscillations regulates stem cell adhesion and differentiation. ACS Nano 2017, 11, 9636–9649. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, T.; Khademhosseini, A.; Langer, R. Chasing the Paradigm: Clinical Translation of 25 Years of Tissue Engineering. Tissue Eng. Part A 2019, 25, 679–687. [Google Scholar] [CrossRef] [PubMed]
- Trounson, A.; McDonald, C. Stem cell therapies in clinical trials: Progress and challenges. Cell Stem Cell 2015, 17, 11–22. [Google Scholar] [CrossRef]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef]
- Pagliuca, F.W.; Millman, J.R.; Gürtler, M.; Segel, M.; Van Dervort, A.; Ryu, J.H.; Peterson, Q.P.; Greiner, D.; Melton, D.A. Generation of functional human pancreatic β cells in vitro. Cell 2014, 159, 428–439. [Google Scholar] [CrossRef]
- Xu, J.; Du, Y.; Deng, H. Direct lineage reprogramming: Strategies, mechanisms, and applications. Cell Stem Cell 2015, 16, 119–134. [Google Scholar] [CrossRef]
- Srivastava, D.; DeWitt, N. In vivo cellular reprogramming: The next generation. Cell 2016, 166, 1386–1396. [Google Scholar] [CrossRef]
- DeWitt, N.D.; Trounson, A. Direct conversion in the heart: A simple twist of fate. EMBO J. 2012, 31, 2244–2246. [Google Scholar] [CrossRef] [PubMed]
- Gornalusse, G.G.; Hirata, R.K.; Funk, S.E.; Riolobos, L.; Lopes, V.S.; Manske, G.; Prunkard, D.; Colunga, A.G.; Hanafi, L.-A.; Clegg, D.O. HLA-E-expressing pluripotent stem cells escape allogeneic responses and lysis by NK cells. Nat. Biotechnol. 2017, 35, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Kretlow, J.D.; Mikos, A.G. From material to tissue: Biomaterial development, scaffold fabrication, and tissue engineering. AIChE J. 2008, 54, 3048–3067. [Google Scholar] [CrossRef] [PubMed]
- Badylak, S.F. Xenogeneic extracellular matrix as a scaffold for tissue reconstruction. Transpl. Immunol. 2004, 12, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Dahms, S.; Piechota, H.; Dahiya, R.; Lue, T.; Tanagho, E. Composition and biomechanical properties of the bladder acellular matrix graft: Comparative analysis in rat, pig and human. Br. J. Urol. 1998, 82, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Parmaksiz, M.; Dogan, A.; Odabas, S.; Elçin, A.E.; Elçin, Y.M. Clinical applications of decellularized extracellular matrices for tissue engineering and regenerative medicine. Biomed. Mater. 2016, 11, 022003. [Google Scholar] [CrossRef]
- Gilpin, A.; Yang, Y. Decellularization strategies for regenerative medicine: From processing techniques to applications. BioMed Res. Int. 2017, 2017, 9831534. [Google Scholar] [CrossRef]
- Ott, H.C.; Matthiesen, T.S.; Goh, S.-K.; Black, L.D.; Kren, S.M.; Netoff, T.I.; Taylor, D.A. Perfusion-decellularized matrix: Using nature’s platform to engineer a bioartificial heart. Nat. Med. 2008, 14, 213–221. [Google Scholar] [CrossRef]
- Chen, K.-L.; Eberli, D.; Yoo, J.J.; Atala, A. Bioengineered corporal tissue for structural and functional restoration of the penis. Proc. Natl. Acad. Sci. USA 2010, 107, 3346–3350. [Google Scholar] [CrossRef]
- Petersen, T.H.; Calle, E.A.; Zhao, L.; Lee, E.J.; Gui, L.; Raredon, M.B.; Gavrilov, K.; Yi, T.; Zhuang, Z.W.; Breuer, C. Tissue-engineered lungs for in vivo implantation. Science 2010, 329, 538–541. [Google Scholar] [CrossRef]
- Zhang, Y.S.; Khademhosseini, A. Advances in engineering hydrogels. Science 2017, 356, eaaf3627. [Google Scholar] [CrossRef] [PubMed]
- Yue, K.; Trujillo-de Santiago, G.; Alvarez, M.M.; Tamayol, A.; Annabi, N.; Khademhosseini, A. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials 2015, 73, 254–271. [Google Scholar] [CrossRef] [PubMed]
- Klotz, B.J.; Lim, K.S.; Chang, Y.X.; Soliman, B.G.; Pennings, I.; Melchels, F.P.; Woodfield, T.B.; Rosenberg, A.; Malda, J.; Gawlitta, D. Engineering of a complex bone tissue model with endothelialised channels and capillary-like networks. Eur. Cells Mater. 2018, 35, 335–349. [Google Scholar] [CrossRef] [PubMed]
- Edri, R.; Gal, I.; Noor, N.; Harel, T.; Fleischer, S.; Adadi, N.; Green, O.; Shabat, D.; Heller, L.; Shapira, A. Personalized hydrogels for engineering diverse fully autologous tissue implants. Adv. Mater. 2019, 31, 1803895. [Google Scholar] [CrossRef]
- Webber, M.J.; Khan, O.F.; Sydlik, S.A.; Tang, B.C.; Langer, R. A perspective on the clinical translation of scaffolds for tissue engineering. Ann. Biomed. Eng. 2015, 43, 641–656. [Google Scholar] [CrossRef] [PubMed]
- Burke, J.F.; Yannas, I.V.; Quinby Jr, W.C.; Bondoc, C.C.; Jung, W.K. Successful use of a physiologically acceptable artificial skin in the treatment of extensive burn injury. Ann. Surg. 1981, 194, 413. [Google Scholar] [CrossRef]
- di Summa, P.G.; Kingham, P.J.; Campisi, C.C.; Raffoul, W.; Kalbermatten, D.F. Collagen (NeuraGen®) nerve conduits and stem cells for peripheral nerve gap repair. Neurosci. Lett. 2014, 572, 26–31. [Google Scholar] [CrossRef]
- Guo, Y.; Yuan, T.; Xiao, Z.; Tang, P.; Xiao, Y.; Fan, Y.; Zhang, X. Hydrogels of collagen/chondroitin sulfate/hyaluronan interpenetrating polymer network for cartilage tissue engineering. J. Mater. Sci. Mater. Med. 2012, 23, 2267–2279. [Google Scholar] [CrossRef]
- Hillel, A.T.; Unterman, S.; Nahas, Z.; Reid, B.; Coburn, J.M.; Axelman, J.; Chae, J.J.; Guo, Q.; Trow, R.; Thomas, A. Photoactivated composite biomaterial for soft tissue restoration in rodents and in humans. Sci. Transl. Med. 2011, 3, ra67–ra93. [Google Scholar] [CrossRef]
- Sharma, B.; Fermanian, S.; Gibson, M.; Unterman, S.; Herzka, D.A.; Cascio, B.; Coburn, J.; Hui, A.Y.; Marcus, N.; Gold, G.E. Human cartilage repair with a photoreactive adhesive-hydrogel composite. Sci. Transl. Med. 2013, 5, ra166–ra167. [Google Scholar] [CrossRef]
- Foster, L.J.R.; Karsten, E. A chitosan based, laser activated thin film surgical adhesive, ‘SurgiLux’: Preparation and demonstration. JoVE (J. Vis. Exp.) 2012, 68, e3527. [Google Scholar] [CrossRef] [PubMed]
- Daly, K.A.; Liu, S.; Agrawal, V.; Brown, B.N.; Huber, A.; Johnson, S.A.; Reing, J.; Sicari, B.; Wolf, M.; Zhang, X. The host response to endotoxin-contaminated dermal matrix. Tissue Eng. Part A 2012, 18, 1293–1303. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.D.; Wright, N.M. Polyethylene glycol hydrogel spinal sealant (DuraSeal Spinal Sealant) as an adjunct to sutured dural repair in the spine: Results of a prospective, multicenter, randomized controlled study. LWW 2011, 36, 1906–1912. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.E.; Park, S.; Park, M.; Kim, M.H.; Park, C.G.; Lee, S.H.; Choi, S.Y.; Kim, B.H.; Park, H.J.; Park, J.-H. Surgical suture assembled with polymeric drug-delivery sheet for sustained, local pain relief. Acta Biomater. 2013, 9, 8318–8327. [Google Scholar] [CrossRef]
- Utzschneider, S.; Paulus, A.; Schröder, C.; Jansson, V. Possibilities and limits of modern polyethylenes. With respect to the application profile. Der Orthop. 2014, 43, 515–521. [Google Scholar] [CrossRef]
- Odian, G. Principles of Polymerization; John Wiley & Sons: Hoboken, NJ, USA, 2004. [Google Scholar]

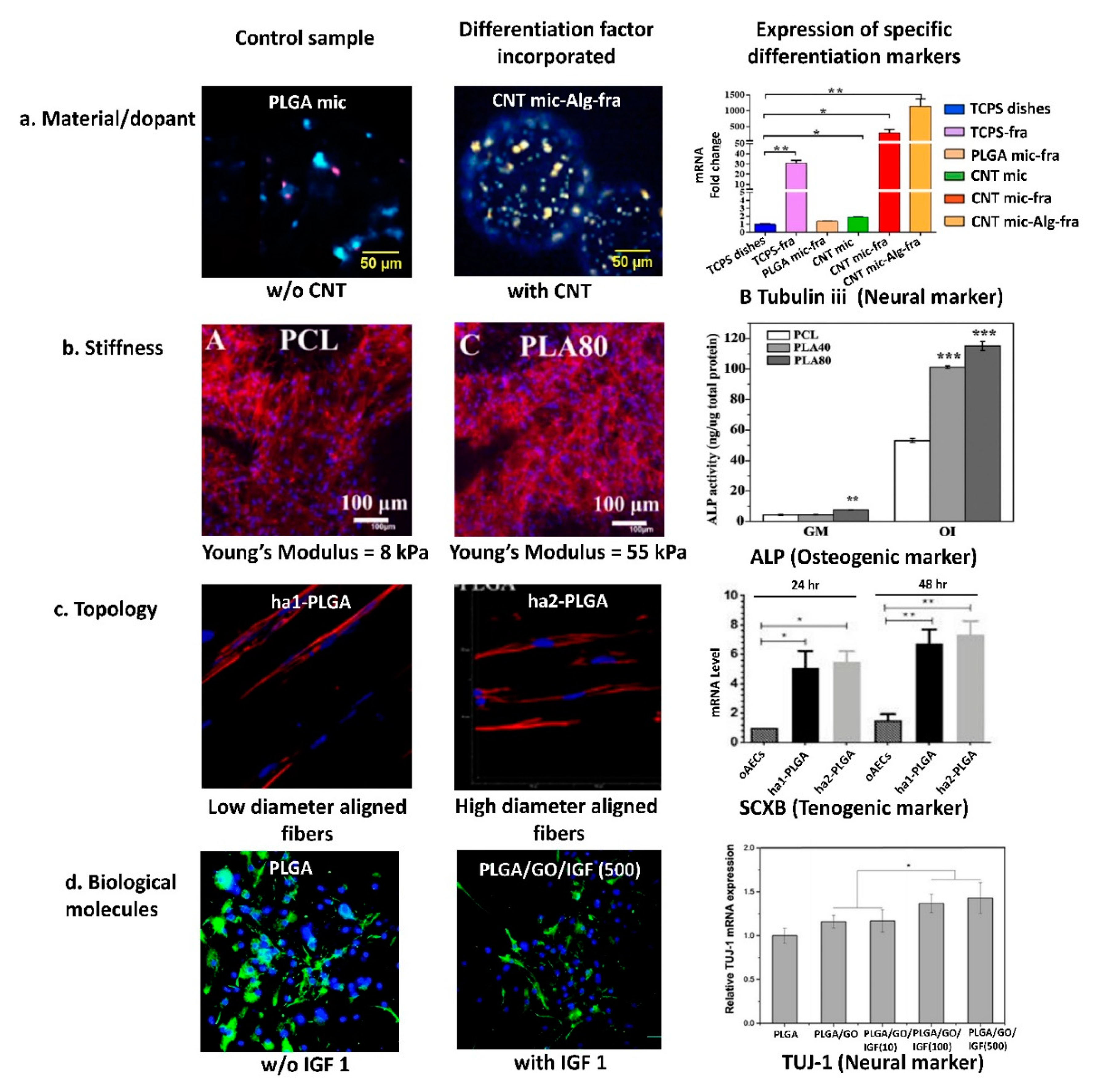
| Differentiation Factor | Type of Scaffold | Differentiation | Ref. | |
|---|---|---|---|---|
| Material/dopant | HA | PCL/HA microsphere prepared by spray precipitation | Osteogenic | [106] |
| Acetylated HA | PLC/HA electrospun nanofibrous scaffold | Chondrogenic | [107] | |
| CNT | Cylindrical scaffold of PCL/CNT prepared by solution evaporation technique | Osteogenic | [108] | |
| Magnesium | PCL/Mg nano composite film | Osteogenic | [109] | |
| Bioceramic (Zn2SiO4) | Polyaniline-gelatin-PCL composite electrospun nanofiber with ceramic nanoparticle (Zn2SiO4) coating | Osteogenic | [110] | |
| Topology/stiffness | Relative increase | PCL/PLA electrospun mesh | Osteogenic | [117] |
| PCL/PLA random meshes of 1.8 µm diameter | PCL/PLA electrospun mesh | Chondrogenic | [111] | |
| Young’s Modulus of 55 kPa | PCL/PLA electrospun mesh | Osteogenic | [112] | |
| PCL aligned fiber of 1000 nm diameter Coaxial PCL/PLA blend of 2000 nm diameter | Electrospun scaffold of coaxial PCL/PLA fibers | Osteogenic Tenogenic | [113] | |
| Soft scaffold of PTHF/PCL/collagen type 1 (Modulus of 4.3 MPa) | PTHF/PCL/collagen type 1 electrospun nanofiber | Chondrogenic | [116] | |
| Biological molecules/topology | TGF-β3/random fibers TGF-β3/aligned fibers TGF-β/CTGF/aligned fibers CTGF/aligned fibers | PCL electrospun sheets ca. 300 µm thick | Endochondral Chondrogenic Fibrochondrogenic Ligamentous | [118] |
| Differentiation Factor | Type of Scaffold | Differentiation | Ref. | |
|---|---|---|---|---|
| Material/dopant | Polyethyleneimine, citric acid and hydroxyapatite | 3D printed PLA with the functionalized surface | Osteogenic | [120] |
| PDL/collagen type 1 | 3D printed PLA with PDL and collagen type 1 coating | Osteogenic | [121] | |
| Gelatin/RGNPs | 3D printed PLA embedded in Gelatin/RGNPs | Osteogenic | [122] | |
| PVA addition | Electrospun nano fibrous scaffold | Pancreatic cells | [123] | |
| PHB (stiffness) curcumin | Electrospun nano fibrous scaffold | Osteogenic | [124] | |
| Pluronic | Electrospun nano fibrous scaffold | Osteogenic | [125] | |
| Oyster shell | Electrospun nano fibrous scaffold | Osteogenic | [126] | |
| Octa calcium phosphate | Electrospun nano fibrous scaffold | Osteogenic | [127] | |
| Bioactive glass ceramic nanoparticle | Electrospun nano fibrous scaffold coated with ceramic nanoparticle | Osteogenic | [128] | |
| Poly-P solution | Electrospun nano fibrous scaffold | Myogenic | [129] | |
| CNT | PLGA-CNT microspheres with alginate hydrogel | Neurogenic | [130] | |
| Stiffness | Relatively high stiffness | Electrospun nano fibrous scaffold | Chondrogenic | [136] |
| Relatively high Young’s modulus value | Electrospun nano fibrous scaffold | Osteogenic | [137] | |
| Young’s modulus > 200 kPa | 3D porous composite bulk scaffold prepared by salt leaching | Myogenic | [138] | |
| Young’s modulus 62–68 MPa | Injectable micro-ribbon shaped fibronectin coated PLGA | Osteogenic | [139] | |
| Topology | Aligned fibers | Electrospun fibrous scaffold | Osteogenic | [132] |
| Lower fiber diameter (1.27 μm) | Electrospun nano fibrous scaffold | Tenogenic | [133] | |
| Aligned fibres | Electrospun nano fibrous scaffold | Neurogenic | [134] | |
| Biological molecules | BMP2 | Soybean lectin mediated PLGA microspheres | Osteogenic | [141] |
| GO/IGF1 | Electrospun nano fibrous scaffold | Neuronal | [133] | |
| Fibrin/bFGF | Knitted gel scaffold | Tenogenic | [141] | |
| Differentiation Factor | Type of Scaffold | Differentiation | Ref. | |
|---|---|---|---|---|
| Material/dopant | PEGDA/RGD | Bulk hydrogel | Heart valve interstitial cells | [146] |
| GO | Bulk hydrogel | Osteogenic | [142] | |
| Zn2SiO4 | Electrospun nano fibrous scaffold with Zn2SiO4 coating | Osteogenic | [143] | |
| Stiffness | Relatively increased relaxation time and creep | Bulk hydrogel | Osteogenic | [144] |
| Hard gel (5.7 kPa) Soft gel (77 Pa) | Bulk hydrogel | Osteogenic Adipogenic | [145] | |
| Differentiation Factor | Type of Scaffold | Differentiation | Ref. | |
|---|---|---|---|---|
| Material/dopant | Low stiffness (Young’s modulus = 26 MPa) and high roughness | Bulk scaffold | Osteogenic | [147] |
| Cellulose nanowhisker | Porous bulk scaffold | Osteogenic | [148] | |
| PVA/sulphated alginate | Electrospun nanofibrous scaffold | Neurogenic | [149] | |
| Chitosan/collagen/PVA nanofiber with Curcumin | Electrospun nanofibrous scaffold | Myogenic | [150] | |
| PET/SR-HA | Bulk material | Osteogenic | [152] | |
| Biological Molecules | BMP-2 | Microgel system | Osteogenic | [151] |
| Differentiation Factor | Type of Scaffold | Differentiation | Ref. | |
|---|---|---|---|---|
| Material/dopant | Ceramic/GO | Nanofibrous scaffold | Neurogenic | [153] |
| Titanium | Bulk material | Osteogenic | [155] | |
| Growth Factor | Observed Effects | Ref. |
|---|---|---|
| BMP subfamily | Enhanced proliferation, and differentiation to osteogenic and chondrogenic phenotype | [170,171,172,173,174,175] |
| EGF | Enhanced proliferation, viability, and migration | [176,177,178] |
| FGF | Enhanced proliferation and differentiation to chondrogenic phenotype | [179,180,181] |
| HGF | Enhanced proliferation and viability | [182] |
| IGF-1 | Enhance proliferation and viability, influencing apoptosis, differentiation to neural phenotype | [183,184,185,186,187,188,189] |
| Insulin | Differentiation to adipogenic phenotype | [190] |
| PDGF | Enhanced proliferation and viability, differentiation to the pulmonary, neural, dermal, skeletal, gastrointestinal, vessel, and hematopoietic cell types | [191,192,193] |
| TGF-β family | Enhanced proliferation, differentiation to oligodendric and chondrogenic phenotype | [194,195,196,197,198] |
| VEGF | Enhanced proliferation and viability, differentiation to hematopoietic, osteogenic, and chondrogenic phenotypes | [184,185,187,189] |
| Wnt family | Enhanced proliferation and differentiation | [199,200,201] |
| Materials | Concentration | Preparation Method | Range of Elastic Modulus | Cells | Ref. |
|---|---|---|---|---|---|
| Alginate hydrogelsGelatin | 0.8, 1.3, 1.8, and 2.3 wt% 4.1 wt% | CaCl2 cross-linking | 1.4−14,2 kPa | a hMSCs | [209] |
| Alginate hydrogelsGelatin | 1, 3, 5, 7, and 9 wt% 2, 4, 6, 8, and 10 wt% | CaCl2 cross-linking | 29.8−48 kPa | b MSCs | [210] |
| Silk fibroin | 1.5−4 wt% | high-pressure CO2 | 6−64 kPa | MSCs | [211] |
| Silk fibroin Collagen | various ratio starting from silk fibroin (70 mg/mL) collagen (7.8 mg/mL) | Gelatin | 9.93−31.16 kPa | c BMSCs | [212] |
| Silk fibroin Collagen | 0.175−2.45 wt% 0.5−3.5 mg/mL | sonication and gelation | 0.05−20.4 kPa | hMSCs | [213] |
| Silk fibroin | 2 wt% | freeze-drying | 3−58.4 kPa | BMSCs differentiation in endothelial cells | [214] |
| Silk fibroin nanofiber | 5 wt% | concentrating silk fibroin nanofiber solution and salt leaching | 2−18 kPa | BMSCs myogenic differentiation | [215] |
| Silk fibroin Silk fibroin nanofiber | various ratio starting from 6% (silk fibroin) and 2% (silk fibroin nanofiber) | d HRP cross-linking | 9−60 kPa | BMSCs differentiation in different lineages | [215] |
| Silk fibroin nanofiber | 1, 2, and 4 wt% | HRP-cross-linking | 0.6−160 kPa | BMSCs | [216] |
| Gelatin methacrylamide | 5, 10, and 15 wt% | photo-crosslinking | 1.7−16.4 kPa | dental stem cells | [217] |
| Gelatin methacrylamide | 7.5, 10, and 15 wt% | photo-crosslinking | 25.59−41.78 kPa | BMSCs differentiation into endothelial cells | [218] |
| Gelatin methacrylamide | 10 wt% | photo-crosslinking | 3.5−13.1 kPa | human adipose- derived stem cells | [219] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geevarghese, R.; Sajjadi, S.S.; Hudecki, A.; Sajjadi, S.; Jalal, N.R.; Madrakian, T.; Ahmadi, M.; Włodarczyk-Biegun, M.K.; Ghavami, S.; Likus, W.; et al. Biodegradable and Non-Biodegradable Biomaterials and Their Effect on Cell Differentiation. Int. J. Mol. Sci. 2022, 23, 16185. https://doi.org/10.3390/ijms232416185
Geevarghese R, Sajjadi SS, Hudecki A, Sajjadi S, Jalal NR, Madrakian T, Ahmadi M, Włodarczyk-Biegun MK, Ghavami S, Likus W, et al. Biodegradable and Non-Biodegradable Biomaterials and Their Effect on Cell Differentiation. International Journal of Molecular Sciences. 2022; 23(24):16185. https://doi.org/10.3390/ijms232416185
Chicago/Turabian StyleGeevarghese, Rency, Seyedeh Sara Sajjadi, Andrzej Hudecki, Samad Sajjadi, Nahid Rezvani Jalal, Tayyebeh Madrakian, Mazaher Ahmadi, Małgorzata K. Włodarczyk-Biegun, Saeid Ghavami, Wirginia Likus, and et al. 2022. "Biodegradable and Non-Biodegradable Biomaterials and Their Effect on Cell Differentiation" International Journal of Molecular Sciences 23, no. 24: 16185. https://doi.org/10.3390/ijms232416185
APA StyleGeevarghese, R., Sajjadi, S. S., Hudecki, A., Sajjadi, S., Jalal, N. R., Madrakian, T., Ahmadi, M., Włodarczyk-Biegun, M. K., Ghavami, S., Likus, W., Siemianowicz, K., & Łos, M. J. (2022). Biodegradable and Non-Biodegradable Biomaterials and Their Effect on Cell Differentiation. International Journal of Molecular Sciences, 23(24), 16185. https://doi.org/10.3390/ijms232416185









