Alarmins as a Possible Target of Future Therapies for Atrial Fibrillation
Abstract
1. Introduction
2. Methods
3. Results and Discussion
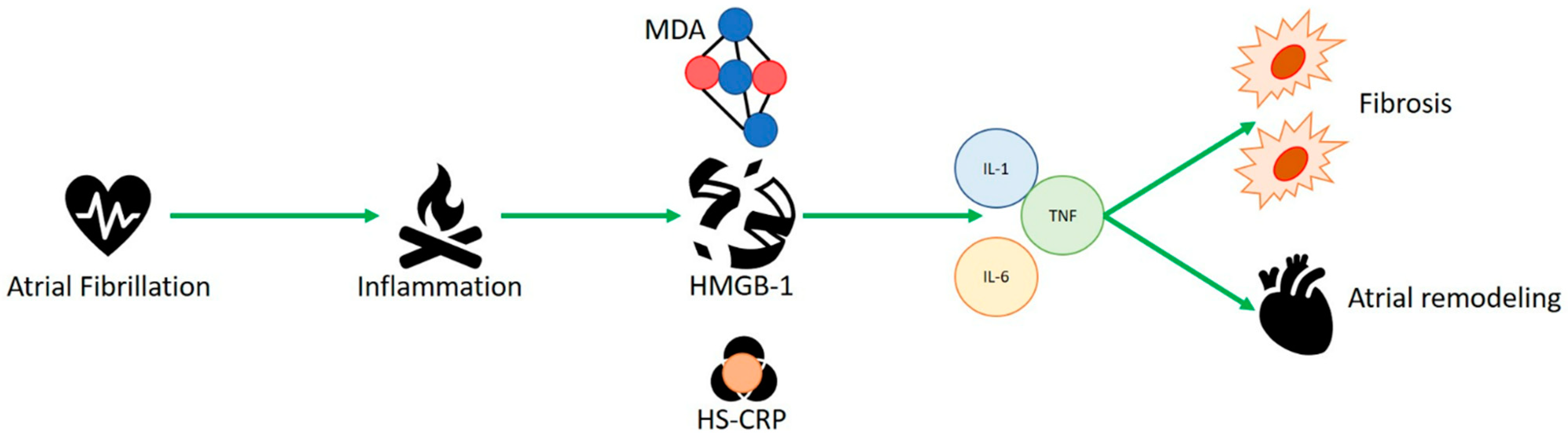
3.1. HMGB-1 and Human Studies
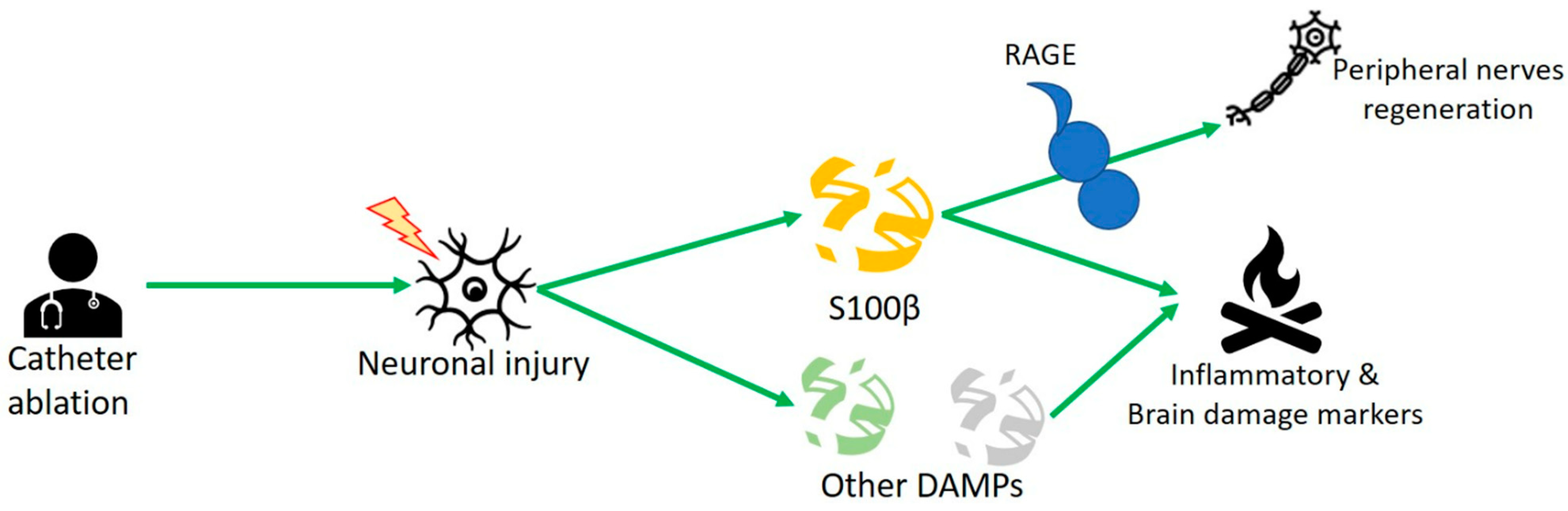
3.2. S100 Protein and Human Studies
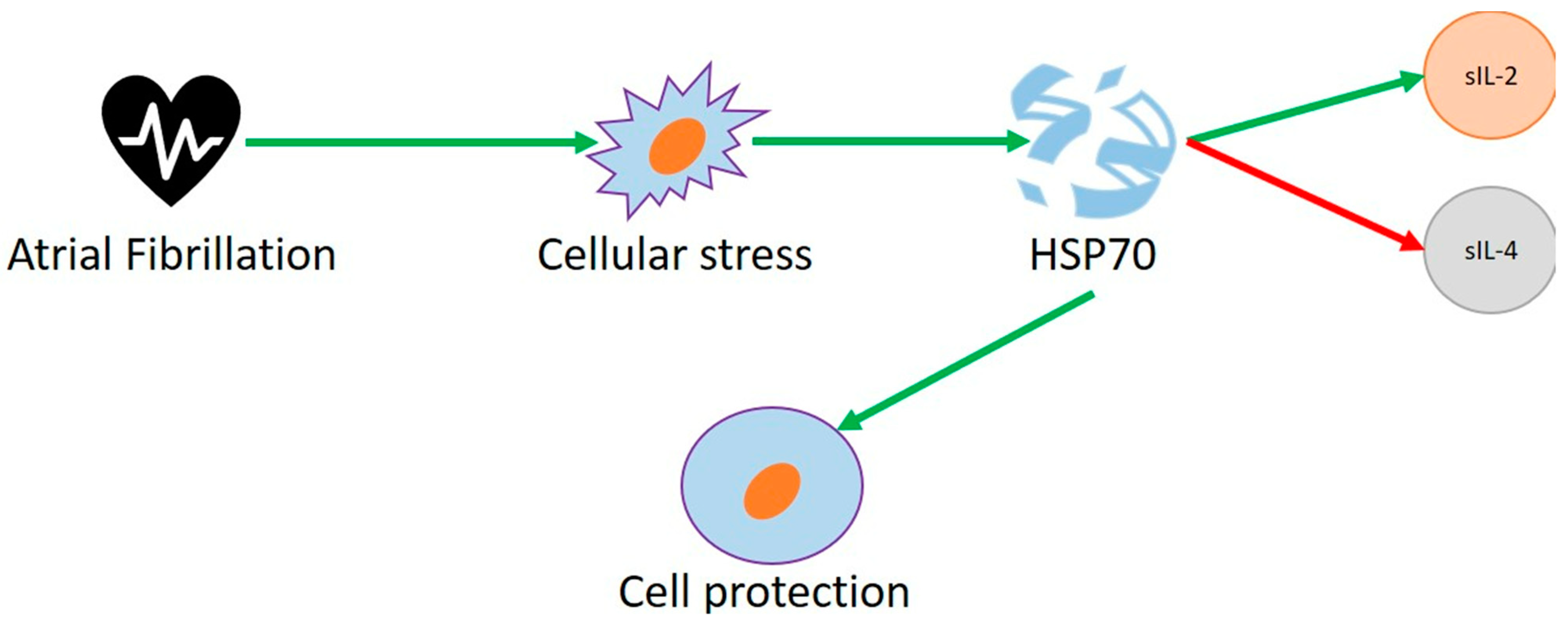
3.3. Heat Shock Proteins and Human Studies
3.4. Heat Shock Proteins and Animal Studies
3.5. Heat Shock Proteins and Cell Studies
4. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das, S.R.; et al. American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart Dis. Stroke Stat. 2019 Update A Rep. Am. Heart Assoc. Circ. 2019, 139, e56e528. [Google Scholar]
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomström-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.-A.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur. Heart J. 2021, 42, 373–498, Erratum in: Eur. Heart J. 2021, 42, 507. [Google Scholar] [CrossRef] [PubMed]
- Hart, R.G.; Pearce, L.; Aguilar, M.I. Meta-analysis: Antithrombotic Therapy to Prevent Stroke in Patients Who Have Nonvalvular Atrial Fibrillation. Ann. Intern. Med. 2007, 146, 857–867. [Google Scholar] [CrossRef]
- Colilla, S.; Crow, A.; Petkun, W.; Singer, D.E.; Simon, T.; Liu, X. Estimates of Current and Future Incidence and Prevalence of Atrial Fibrillation in the U.S. Adult Population. Am. J. Cardiol. 2013, 112, 1142–1147. [Google Scholar] [CrossRef]
- Krijthe, B.P.; Kunst, A.; Benjamin, E.; Lip, G.Y.; Franco, O.; Hofman, A.; Witteman, J.C.; Stricker, B.H.; Heeringa, J. Projections on the number of individuals with atrial fibrillation in the European Union, from 2000 to 2060. Eur. Heart J. 2013, 34, 2746–2751. [Google Scholar] [CrossRef] [PubMed]
- Lau, D.H.; Linz, D.; Schotten, U.; Mahajan, R.; Sanders, P.; Kalman, J.M. Pathophysiology of Paroxysmal and Persistent Atrial Fibrillation: Rotors, Foci and Fibrosis. Heart Lung Circ. 2017, 26, 887–893. [Google Scholar] [CrossRef] [PubMed]
- Lau, D.H.; Linz, D.; Sanders, P. New Findings in Atrial Fibrillation Mechanisms. Card Electrophysiol. Clin. 2019, 11, 563–571. [Google Scholar] [CrossRef]
- European Heart Rhythm Association (EHRA); European Cardiac Arrhythmia Scoiety (ECAS); American College of Cardiology (ACC); American Heart Association (AHA); Society of Thoracic Surgeons (STS); Calkins, H.; Brugada, J.; Packer, D.L.; Cappato, R.; Chen, S.A.; et al. HRS/EHRA/ECAS expert Consensus Statement on catheter and surgical ablation of atrial fibrillation: Recommendations for personnel, policy, procedures and follow-up. A report of the Heart Rhythm Society (HRS) Task Force on catheter and surgical ablation of atrial fibrillation. Heart Rhythm. 2007, 4, 816–861. [Google Scholar] [CrossRef]
- Bhatt, H.V.; Fischer, G.W. Atrial Fibrillation: Pathophysiology and Therapeutic Options. J. Cardiothorac. Vasc. Anesthesia 2015, 29, 1333–1340. [Google Scholar] [CrossRef]
- Li, Q.; Lai, Y.; Gao, X.; Li, X.; Deng, C.-Y.; Guo, H.; Zhao, J.; Yang, H.; Xu, Y.; Wu, S.; et al. Involvement of plasminogen activator inhibitor-1 and its related molecules in atrial fibrosis in patients with atrial fibrillation. PeerJ 2021, 9, e11488. [Google Scholar] [CrossRef]
- Yagi, S.; Akaike, M.; Aihara, K.-I.; Ishikawa, K.; Iwase, T.; Ikeda, Y.; Soeki, T.; Yoshida, S.; Sumitomo-Ueda, Y.; Matsumoto, T.; et al. Endothelial Nitric Oxide Synthase–Independent Protective Action of Statin Against Angiotensin II–Induced Atrial Remodeling via Reduced Oxidant Injury. Hypertension 2010, 55, 918–923. [Google Scholar] [CrossRef] [PubMed]
- Harada, M.; Van Wagoner, D.R.; Nattel, S. Role of Inflammation in Atrial Fibrillation Pathophysiology and Management. Circ. J. 2015, 79, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Harada, M.; Nattel, S. Implications of Inflammation and Fibrosis in Atrial Fibrillation Pathophysiology. Card. Electrophysiol. Clin. 2021, 13, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Huxley, R.R.; Filion, K.B.; Konety, S.; Alonso, A. Meta-Analysis of Cohort and Case–Control Studies of Type 2 Diabetes Mellitus and Risk of Atrial Fibrillation. Am. J. Cardiol. 2011, 108, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Teshima, Y.; Fukui, A.; Kondo, H.; Nishio, S.; Nakagawa, M.; Saikawa, T.; Takahashi, N. Glucose fluctuations increase the incidence of atrial fibrillation in diabetic rats. Cardiovasc. Res. 2014, 104, 5–14. [Google Scholar] [CrossRef]
- Chen, G.; Chelu, M.G.; Dobrev, D.; Li, N. Cardiomyocyte Inflammasome Signaling in Cardiomyopathies and Atrial Fibrillation: Mechanisms and Potential Therapeutic Implications. Front. Physiol. 2018, 9, 1115. [Google Scholar] [CrossRef]
- Van Linthout, S.; Miteva, K.; Tscho¨pe, C. Crosstalk between fibroblasts and inflammatory cells. Cardiovasc Res. 2014, 102, 25. [Google Scholar] [CrossRef]
- Yang, D.; Han, Z.; Oppenheim, J.J. Alarmins and immunity. Immunol Rev. 2017, 280, 41–56. [Google Scholar] [CrossRef]
- Rider, P.; Voronov, E.; Dinarello, C.A.; Apte, R.N.; Cohen, I. Alarmins: Feel the Stress. J. Immunol. 2017, 198, 1395–1402. [Google Scholar] [CrossRef]
- Oppenheim, J.J.; Yang, D. Alarmins: Chemotactic activators of immune responses. Curr. Opin. Immunol. 2005, 17, 359–365. [Google Scholar] [CrossRef]
- Chen, G.Y.; Nuñez, G. Sterile inflammation: Sensing and reacting to damage. Nat. Rev. Immunol. 2010, 10, 826–837. [Google Scholar] [CrossRef] [PubMed]
- Roh, J.S.; Sohn, D.H. Damage-Associated Molecular Patterns in Inflammatory Diseases. Immune Netw. 2018, 18, e27. [Google Scholar] [CrossRef] [PubMed]
- Karam, B.S.; Chavez-Moreno, A.; Koh, W.; Akar, J.G.; Akar, F.G. Oxidative stress and inflammation as central mediators of atrial fibrillation in obesity and diabetes. Cardiovasc. Diabetol. 2017, 16, 120. [Google Scholar] [CrossRef] [PubMed]
- Packer, M. Characterization, Pathogenesis, and Clinical Implications of Inflammation-Related Atrial Myopathy as an Important Cause of Atrial Fibrillation. J. Am. Heart Assoc. 2020, 9, e015343. [Google Scholar] [CrossRef]
- Marchi, N.; Rasmussen, P.; Kapural, M.; Fazio, V.; Kight, K.; Mayberg, M.R.; Kanner, A.; Ayumar, B.; Albensi, B.; Cavaglia, M.; et al. Peripheral markers of brain damage and blood-brain barrier dysfunction. Restor. Neurol. Neurosci. 2003, 21, 109–121. [Google Scholar]
- Scherschel, K.; Hedenus, K.; Jungen, C.; Münkler, P.; Willems, S.; Anwar, O.; Klatt, N.; Eickholt, C.; Meyer, C. Impact of the ablation technique on release of the neuronal injury marker S100B during pulmonary vein isolation. EP Eur. 2020, 22, 1502–1508. [Google Scholar] [CrossRef]
- Kato, T.; Sekiguchi, A.; Sagara, K.; Tanabe, H.; Takamura, M.; Kaneko, S.; Aizawa, T.; Fu, L.-T.; Yamashita, T. Endothelial–mesenchymal transition in human atrial fibrillation. J. Cardiol. 2016, 69, 706–711. [Google Scholar] [CrossRef]
- Sramko, M.; Peichl, P.; Wichterle, D.; Tintera, J.; Maxian, R.; Weichet, J.; Knesplova, L.; Franekova, J.; Pasnisinova, S.; Kautzner, J. A Novel Biomarker-Based Approach for the Detection of Asymptomatic Brain Injury During Catheter Ablation of Atrial Fibrillation. J. Cardiovasc. Electrophysiol. 2013, 25, 349–354. [Google Scholar] [CrossRef]
- Galenko, O.; Jacobs, V.; Knight, S.; Bride, D.; Cutler, M.J.; Muhlestein, J.B.; Carlquist, J.L.; Anderson, J.L.; Knowlton, K.U.; Bunch, T.J. Circulating Levels of Biomarkers of Cerebral Injury in Patients with Atrial Fibrillation. Am. J. Cardiol. 2019, 124, 1697–1700. [Google Scholar] [CrossRef]
- Dumitriu, I.E.; Baruah, P.; Manfredi, A.; E Bianchi, M.; Rovere-Querini, P. HMGB1: An immmune odyssey. Discov. Med. 2005, 5, 388–392. [Google Scholar]
- Echahidi, N.; Pibarot, P.; O’Hara, G.; Mathieu, P. Mechanisms, Prevention, and Treatment of Atrial Fibrillation After Cardiac Surgery. J. Am. Coll. Cardiol. 2008, 51, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Gurses, K.M.; Kocyigit, D.; Yalcin, M.U.; Canpinar, H.; Evranos, B.; Canpolat, U.; Yorgun, H.; Sahiner, L.; Guc, D.; Aytemir, K. Platelet Toll-like receptor and its ligand HMGB-1 expression is increased in the left atrium of atrial fibrillation patients. Cytokine 2018, 103, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Migdady, I.; Russman, A.; Buletko, A.B. Atrial Fibrillation and Ischemic Stroke: A Clinical Review. Semin. Neurol. 2021, 41, 348–364. [Google Scholar] [CrossRef]
- Wakisaka, O.; Takahashi, N.; Shinohara, T.; Ooie, T.; Nakagawa, M.; Yonemochi, H.; Hara, M.; Shimada, T.; Saikawa, T.; Yoshimatsu, H. Hyperthermia treatment prevents angiotensin II-mediated atrial fibrosis and fibrillation via induction of heat-shock protein 72. J. Mol. Cell Cardiol. 2007, 43, 616–626. [Google Scholar] [CrossRef]
- Demidov, O.N.; Tyrenko, V.V.; Svistov, A.; Komarova, Y.Y.; Karpishenko, A.I.; Margulis, B.A.; Leonidovich Shevchenko, Y. Heat shock proteins in cardiosurgery patients. Eur. J. Cardio-Thoracic Surg. 1999, 16, 444–449. [Google Scholar] [CrossRef]
- Afzal, A.R.; Mandal, K.; Nyamweya, S.; Foteinos, G.; Poloniecki, J.; Camm, A.J.; Jahangiri, M.; Xu, Q. Association of Met439Thr Substitution in Heat Shock Protein 70 Gene with Postoperative Atrial Fibrillation and Serum HSP70 Protein Levels. Cardiology 2007, 110, 45–52. [Google Scholar] [CrossRef]
- Rammos, K.S.; Koullias, G.J.; Hassan, M.O.; Argyrakis, N.P.; Voucharas, C.G.; Scarupa, S.J.; Cowte, T.G. Low preoperative HSP70 atrial myocardial levels correlate significantly with high incidence of postoperative atrial fibrillation after cardiac surgery. Cardiovasc. Surg. 2002, 10, 228–232. [Google Scholar] [CrossRef]
- Hu, L.; Wang, Z.; Carmone, C.; Keijer, J.; Zhang, D. Role of Oxidative DNA Damage and Repair in Atrial Fibrillation and Ischemic Heart Disease. Int. J. Mol. Sci. 2021, 22, 3838. [Google Scholar] [CrossRef]
- Vitadello, M.; Ausma, J.; Borgers, M.; Gambino, A.; Casarotto, D.C.; Gorza, L. Increased Myocardial GRP94 Amounts During Sustained Atrial Fibrillation. Circulation 2001, 103, 2201–2206. [Google Scholar] [CrossRef]
- Li, P.; Kurata, Y.; Maharani, N.; Mahati, E.; Higaki, K.; Hasegawa, A.; Shirayoshi, Y.; Yoshida, A.; Kondo, T.; Kurozawa, Y.; et al. E3 ligase CHIP and Hsc70 regulate Kv1.5 protein expression and function in mammalian cells. J. Mol. Cell Cardiol. 2015, 86, 138–146. [Google Scholar] [CrossRef]
- Min, T.J.; Jo, W.-M.; Shin, S.Y.; Lim, H.E. The protective effect of heat shock protein 70 (Hsp70) in atrial fibrillation in various cardiomyopathy conditions. Heart Vessel. 2014, 30, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Kornej, J.; Reinhardt, C.; Kosiuk, J.; Arya, A.; Hindricks, G.; Adams, V.; Husser, D.; Bollmann, A. Response of circulating heat shock protein 70 and anti-heat shock protein 70 antibodies to catheter ablation of atrial fibrillation. J. Transl. Med. 2013, 11, 49. [Google Scholar] [CrossRef] [PubMed]
- Milner, C.; Campbell, R. Polymorphic analysis of the three MHC-linked HSP70 genes. Immunogenetics 1992, 36, 357–362. [Google Scholar] [CrossRef]
- Gollob, M.H. Atrial fibrillation as an autoimmune disease? Heart Rhythm. 2013, 10, 442–443. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, S.; Yoo, S.; Chakrabarti, S.; Zhang, T.; Ke, T.; Oberti, C.; Yong, S.L.; Fang, F.; Li, L.; et al. Mutation in Nuclear Pore Component NUP155 Leads to Atrial Fibrillation and Early Sudden Cardiac Death. Cell 2008, 135, 1017–1027. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, B.C.; Sapra, G.; Patterson, N.L.; Cemerlang, N.; Kiriazis, H.; Ueyama, T.; Febbraio, M.A.; McMullen, J.R. Long-Term Overexpression of Hsp70 Does Not Protect against Cardiac Dysfunction and Adverse Remodeling in a MURC Transgenic Mouse Model with Chronic Heart Failure and Atrial Fibrillation. PLoS ONE 2015, 10, e0145173. [Google Scholar] [CrossRef]
- Sapra, G.; Tham, Y.K.; Cemerlang, N.; Matsumoto, A.; Kiriazis, H.; Bernardo, B.C.; Henstridge, D.C.; Ooi, J.Y.Y.; Pretorius, L.; Boey, E.J.H.; et al. The small-molecule BGP-15 protects against heart failure and atrial fibrillation in mice. Nat. Commun. 2014, 5, 5705. [Google Scholar] [CrossRef]
- Wiersma, M.; Meijering, R.A.M.; Qi, X.; Zhang, D.; Liu, T.; Hoogstra-Berends, F.; Sibon, O.C.M.; Henning, R.H.; Nattel, S.; Brundel, B.J.J.M. Endoplasmic Reticulum Stress Is Associated With Autophagy and Cardiomyocyte Remodeling in Experimental and Human Atrial Fibrillation. J. Am. Heart Assoc. 2017, 6, e006458. [Google Scholar] [CrossRef]
- Westphal, S.; RIPHeart-Study Collaborators; Stoppe, C.; Gruenewald, M.; Bein, B.; Renner, J.; Cremer, J.; Coburn, M.; Schaelte, G.; Boening, A.; et al. Genome-wide association study of myocardial infarction, atrial fibrillation, acute stroke, acute kidney injury and delirium after cardiac surgery—A sub-analysis of the RIPHeart-Study. BMC Cardiovasc. Disord. 2019, 19, 26. [Google Scholar] [CrossRef]
- Atwood, J.E.; Albers, G.W. Anticoagulation and atrial fibrillation. Herz 1993, 18, 27–38. [Google Scholar]
- Bravo-San Pedro, J.M.; Kroemer, G.; Galluzzi, L. Autophagy and Mitophagy in Cardiovascular Disease. Circ. Res. 2017, 120, 1812–1824. [Google Scholar] [CrossRef] [PubMed]
- Van Marion, D.M.S.; Lanters, E.A.H.; Ramos, K.S.; Li, J.; Wiersma, M.; Bulte, L.B.-T.; Muskens, A.J.Q.M.; Boersma, E.; De Groot, N.M.S.; Brundel, B.J.J.M. Evaluating Serum Heat Shock Protein Levels as Novel Biomarkers for Atrial Fibrillation. Cells 2020, 9, 2105. [Google Scholar] [CrossRef] [PubMed]
- Brundel, B.J.; Henning, R.H.; Ke, L.; van Gelder, I.C.; Crijns, H.J.; Kampinga, H.H. Heat shock protein upregulation protects against pacing-induced myolysis in HL-1 atrial myocytes and in human atrial fibrillation. J. Mol. Cell Cardiol. 2006, 41, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Casciaro, M.; Gangemi, S.; Imbalzano, E.; Quartuccio, S.; Di Salvo, E.; Crea, T. Association between HMGB1 and asthma: A literature review. Clin. Mol. Allergy 2017, 15, 12. [Google Scholar] [CrossRef]
- Taverna, S.; Tonacci, A.; Ferraro, M.; Cammarata, G.; Cuttitta, G.; Bucchieri, S.; Pace, E.; Gangemi, S. High Mobility Group Box 1: Biological Functions and Relevance in Oxidative Stress Related Chronic Diseases. Cells 2022, 11, 849. [Google Scholar] [CrossRef]
- Qu, C.; Wang, X.-W.; Huang, C.; Qiu, F.; Xiang, X.-Y.; Lu, Z.-Q. High mobility group box 1 gene polymorphism is associated with the risk of postoperative atrial fibrillation after coronary artery bypass surgery. J. Cardiothorac. Surg. 2015, 10, 88. [Google Scholar] [CrossRef][Green Version]
- Xu, Q.; Bo, L.; Hu, J.; Geng, J.; Chen, Y.; Li, X.; Chen, F.; Song, J. High mobility group box 1 was associated with thrombosis in patients with atrial fibrillation. Medicine 2018, 97, e0132. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, K.; Zhao, L.; Guo, J.; Hu, X.; Chen, Z. Increased serum HMGB1 is related to oxidative stress in patients with atrial fibrillation. J. Int. Med. Res. 2013, 41, 1796–1802. [Google Scholar] [CrossRef]
- Donato, R.; Cannon, B.R.; Sorci, G.; Riuzzi, F.; Hsu, K.; Weber, D.J.; Geczy, C.L. Functions of S100 proteins. Curr. Mol. Med. 2013, 13, 24–57. [Google Scholar] [CrossRef]
- Gonzalez, L.L.; Garrie, K.; Turner, M.D. Role of S100 proteins in health and disease. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2020, 1867, 118677. [Google Scholar] [CrossRef]
- Kozlyuk, N.; Monteith, A.J.; Garcia, V.; Damo, S.M.; Skaar, E.P.; Chazin, W.J. S100 Proteins in the Innate Immune Response to Pathogens. Adv. Struct. Saf. Stud. 2019, 1929, 275–290. [Google Scholar] [CrossRef]
- Scherschel, K.; Hedenus, K.; Jungen, C.; Lemoine, M.D.; Rübsamen, N.; Veldkamp, M.W.; Klatt, N.; Lindner, D.; Westermann, D.; Casini, S.; et al. Cardiac glial cells release neurotrophic S100B upon catheter-based treatment of atrial fibrillation. Sci. Transl. Med. 2019, 11, eaav7770. [Google Scholar] [CrossRef] [PubMed]
- Tsan, M.-F.; Gao, B. Heat shock proteins and immune system. J. Leukoc. Biol. 2009, 85, 905–910. [Google Scholar] [CrossRef] [PubMed]
- Mandal, K.; Torsney, E.; Poloniecki, J.; Camm, A.J.; Xu, Q.; Jahangiri, M. Association of High Intracellular, But Not Serum, Heat Shock Protein 70 With Postoperative Atrial Fibrillation. Ann. Thorac. Surg. 2005, 79, 865–871. [Google Scholar] [CrossRef]
- Pizon, M.T.; Gburek, T.; Sztefko, K. Kinetics of plasma heat shock protein HSP-70 release in coronary artery surgery: On-pump versus off-pump. Minerva Chir. 2006, 61, 483–491. [Google Scholar]
- Rigopoulos, A.G.; Kalogeropoulos, A.S.; Tsoporis, J.N.; Sakadakis, E.A.; Triantafyllis, A.S.; Noutsias, M.; Gupta, S.; Parker, T.G.; Rizos, I. Heat Shock Protein 70 Is Associated With Cardioversion Outcome and Recurrence of Symptomatic Recent Onset Atrial Fibrillation in Hypertensive Patients. J. Cardiovasc. Pharmacol. 2020, 77, 360–369. [Google Scholar] [CrossRef]
- Oc, M.; Ucar, H.I.; Pinar, A.; Akbulut, B.; Oc, B.; Akyon, Y.; Kanbak, M.; Dogan, R. Heat Shock Protein70: A New Marker for Subsequent Atrial Fibrillation Development? Artif. Organs 2008, 32, 846–850. [Google Scholar] [CrossRef]
- Takahashi, M.; Chesley, A.; Freyssenet, D.; Hood, D.A. Contractile activity-induced adaptations in the mitochondrial protein import system. Am. J. Physiol. Physiol. 1998, 274, C1380–C1387. [Google Scholar] [CrossRef]
- Kirmanoglou, K.; Hannekum, A. Expression of mortalin in patients with chronic atrial fibrillation. Basic Res. Cardiol. 2004, 99, 404–408. [Google Scholar] [CrossRef]
- Allende, M.; Molina, E.; Guruceaga, E.; Tamayo, I.; González-Porras, J.R.; Gonzalez-López, T.J.; Toledo, E.; Rabal, O.; Ugarte, A.; Roldán, V.; et al. Hsp70 protects from stroke in atrial fibrillation patients by preventing thrombosis without increased bleeding risk. Cardiovasc. Res. 2016, 110, 309–318. [Google Scholar] [CrossRef]
- Theodorakis, G. Coronary artery disease and atrial fibrillation. Hell. J. Cardiol. 2017, 58, 213–214. [Google Scholar] [CrossRef] [PubMed]
- Asanin, M.; Vasiljevic, Z.; Matic, M.; Vujisic-Tesic, B.; Arandjelovic, A.; Marinkovic, J.; Ostojic, M. Outcome of Patients in Relation to Duration of New-Onset Atrial Fibrillation following Acute Myocardial Infarction. Cardiology 2006, 107, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Sakabe, M.; Shiroshita-Takeshita, A.; Maguy, A.; Brundel, B.J.; Fujiki, A.; Inoue, H.; Nattel, S. Effects of a heat shock protein inducer on the atrial fibrillation substrate caused by acute atrial ischaemia. Cardiovasc. Res. 2008, 78, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Abdellatif, M.; Sedej, S.; Carmona-Gutierrez, D.; Madeo, F.; Kroemer, G. Autophagy in Cardiovascular Aging. Circ. Res. 2018, 123, 803–824. [Google Scholar] [CrossRef]
- Ren, J.; Zhang, Y. Targeting Autophagy in Aging and Aging-Related Cardiovascular Diseases. Trends Pharmacol. Sci. 2018, 39, 1064–1076. [Google Scholar] [CrossRef]
- Sciarretta, S.; Maejima, Y.; Zablocki, D.; Sadoshima, J. The Role of Autophagy in the Heart. Annu. Rev. Physiol. 2018, 80, 31–38. [Google Scholar] [CrossRef]
- Han, M.; Zhao, M.; Cheng, C.; Huang, Y.; Han, S.; Li, W.; Tu, X.; Luo, X.; Yu, X.; Liu, Y.; et al. Lamin A mutation impairs interaction with nucleoporin NUP155 and disrupts nucleocytoplasmic transport in atrial fibrillation. Hum. Mutat. 2018, 40, 310–325. [Google Scholar] [CrossRef]
- López-Sendón, J.; Merino, J.L. Poor Anticoagulation Control in Atrial Fibrillation: How Much Longer? Rev. Esp. Cardiol. 2015, 68, 740–742. [Google Scholar] [CrossRef]
- Steffel, J.; Verhamme, P.; Potpara, T.S.; Albaladejo, P.; Antz, M.; Desteghe, L.; Haeusler, K.G.; Oldgren, J.; Reinecke, H.; Roldan-Schilling, V.; et al. The 2018 European Heart Rhythm Association Practical Guide on the use of non-vitamin K antagonist oral anticoagulants in patients with atrial fibrillation. Eur. Heart J. 2018, 39, 1330–1393. [Google Scholar] [CrossRef]
| References | Year | Target |
|---|---|---|
| Sramko et al. [25] | 2014 | Humans |
| Kato et al. [26] | 2016 | Humans |
| Scherschel et al. [27] | 2019 | Humans |
| Galenko et al. [28] | 2019 | Humans |
| Scherschel et al. [29] | 2020 | Humans |
| References | Year | Target |
|---|---|---|
| Yongbo Wu [30] | 2013 | Humans |
| Can Qu [31] | 2015 | Humans |
| Qiwen Xu et al. [32] | 2018 | Humans |
| Kadri Murat Gurses [33] | 2018 | Humans |
| References | Year | Target |
|---|---|---|
| Maurizio Vitadello et al. [34] | 2001 | Animals |
| Kyriakos St. Rammos et al. [35] | 2002 | Humans |
| Kiriakos Kirmanoglou et al. [36] | 2004 | Humans |
| Kaushik Mandal et al. [37] | 2005 | Humans |
| Bianca J.J.M. Brundel et al. [38] | 2006 | Cells |
| Bianca J.J.M. Brundel et al. [38] | 2006 | Humans |
| Masao Sakabe et al. [39] | 2007 | Animals |
| Osamu Wakisaka et al. [40] | 2007 | Cells |
| Xianqin Zhang et al. [41] | 2008 | Animals |
| Mehmet Oc et al. [42] | 2008 | Humans |
| Ali R. Afzal et al. [43] | 2008 | Humans |
| Jelena Kornej et al. [44] | 2013 | Humans |
| Geeta Sapra et al. [45] | 2014 | Animals |
| Too Jae Min et al. [46] | 2014 | Animals |
| Peili Li et al. [47] | 2015 | Cells |
| Can Qu et al. [31] | 2015 | Humans |
| Bianca C. Bernardo et al. [48] | 2015 | Animals |
| Mikel Allende et al. [49] | 2016 | Humans |
| Meng Han et al. [50] | 2016 | Cells |
| Marit Wiersma et al. [51] | 2017 | Cells |
| Sabine Westphal et al. [52] | 2019 | Humans |
| Denise M. S. van Marion et al. [53] | 2020 | Humans |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imbalzano, E.; Murdaca, G.; Orlando, L.; Gigliotti-De Fazio, M.; Terranova, D.; Tonacci, A.; Gangemi, S. Alarmins as a Possible Target of Future Therapies for Atrial Fibrillation. Int. J. Mol. Sci. 2022, 23, 15946. https://doi.org/10.3390/ijms232415946
Imbalzano E, Murdaca G, Orlando L, Gigliotti-De Fazio M, Terranova D, Tonacci A, Gangemi S. Alarmins as a Possible Target of Future Therapies for Atrial Fibrillation. International Journal of Molecular Sciences. 2022; 23(24):15946. https://doi.org/10.3390/ijms232415946
Chicago/Turabian StyleImbalzano, Egidio, Giuseppe Murdaca, Luana Orlando, Marianna Gigliotti-De Fazio, Dario Terranova, Alessandro Tonacci, and Sebastiano Gangemi. 2022. "Alarmins as a Possible Target of Future Therapies for Atrial Fibrillation" International Journal of Molecular Sciences 23, no. 24: 15946. https://doi.org/10.3390/ijms232415946
APA StyleImbalzano, E., Murdaca, G., Orlando, L., Gigliotti-De Fazio, M., Terranova, D., Tonacci, A., & Gangemi, S. (2022). Alarmins as a Possible Target of Future Therapies for Atrial Fibrillation. International Journal of Molecular Sciences, 23(24), 15946. https://doi.org/10.3390/ijms232415946








