Glucocorticoid Effects on Proteoglycans and Glycosaminoglycans
Abstract
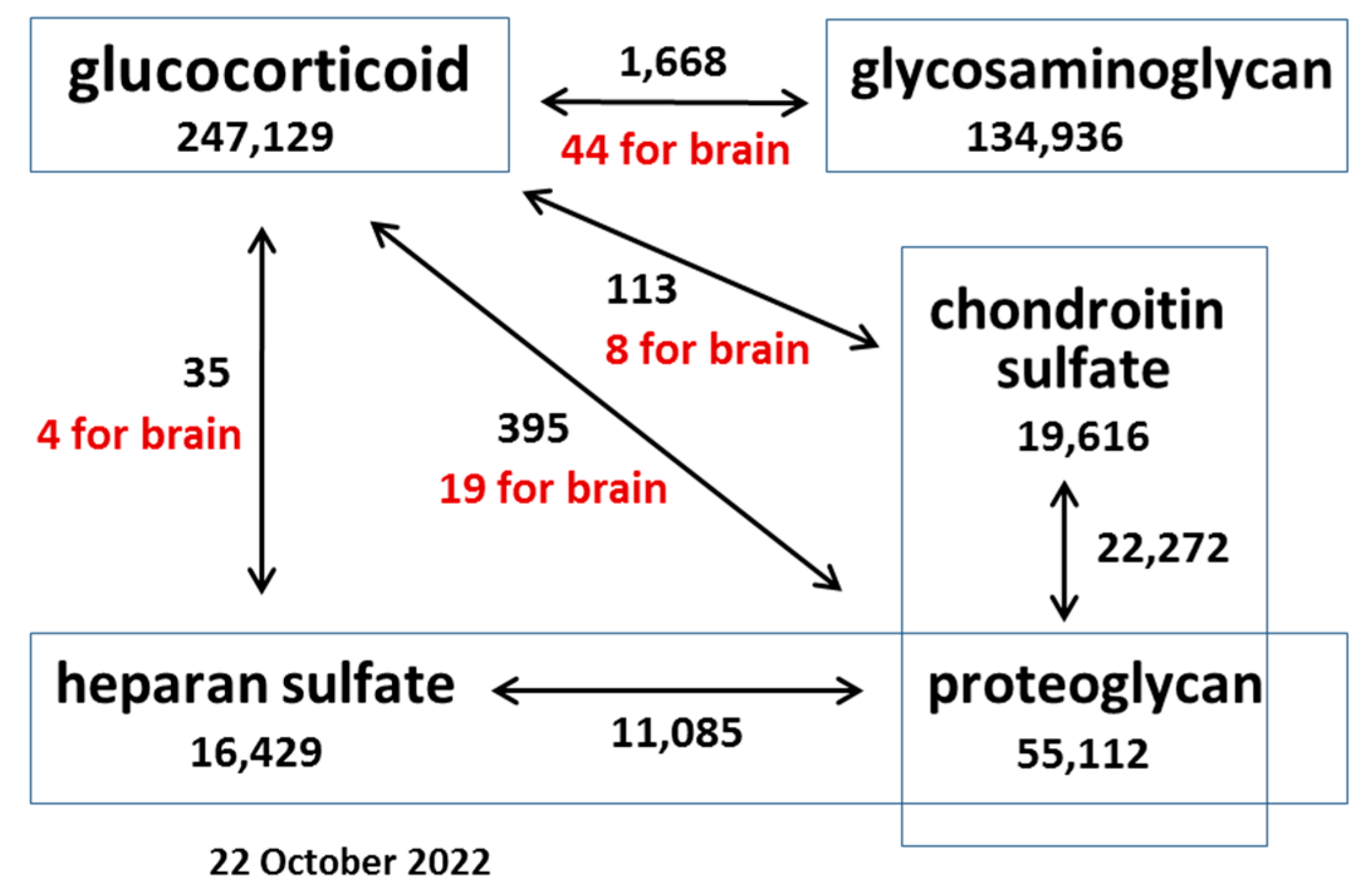
1. Introduction
2. Overview of the Literature Data
3. Effects of GCs on PG Expression and GAG Content in Normal Tissues and Cells
3.1. Cartilage and Chondrocytes
3.2. Lungs and Fibroblasts
3.3. Dermal Fibroblasts
3.4. Osteoblasts and Bone Marrow Stromal Cells
3.5. Other Tissues and Cells
4. Effects of GCs on PG Expression and GAG Content in Malignant Tissues and Cells
5. Effects of GCs on PG Expression and GAG Content in Normal and Malignant Brain Tissues and Cells
5.1. Brain ECM
5.2. Effects of GCs on Normal Brain PGs and GAGs
5.3. Effects of Glucocorticoids on PGs and GAGs in Gliomas
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Swarbrick, M.; Zhou, H.; Seibel, M. Mechanisms in endocrinology: Local and systemic effects of glucocorticoids on metabolism: New lessons from animal models. Eur. J. Endocrinol. 2021, 185, R113–R129. [Google Scholar] [CrossRef] [PubMed]
- Shimba, A.; Ikuta, K. Glucocorticoids Regulate Circadian Rhythm of Innate and Adaptive Immunity. Front. Immunol. 2020, 11, 2143. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Jimenez, D.; Kolb, J.P.; Cidlowski, J.A. Glucocorticoids as Regulators of Macrophage-Mediated Tissue Homeostasis. Front. Immunol. 2021, 12, 669891. [Google Scholar] [CrossRef]
- Mir, N.; Chin, S.A.; Riddell, M.C.; Beaudry, J.L. Genomic and Non-Genomic Actions of Glucocorticoids on Adipose Tissue Lipid Metabolism. Int. J. Mol. Sci. 2021, 22, 8503. [Google Scholar] [CrossRef] [PubMed]
- Taves, M.D.; Ashwell, J.D. Glucocorticoids in T cell development, differentiation and function. Nat. Rev. Immunol. 2021, 21, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Reichardt, S.D.; Amouret, A.; Muzzi, C.; Vettorazzi, S.; Tuckermann, J.P.; Lühder, F.; Reichardt, H.M. The Role of Glucocorticoids in Inflammatory Diseases. Cells 2021, 10, 2921. [Google Scholar] [CrossRef]
- Goodin, D.S. Glucocorticoid treatment of multiple sclerosis. Handb. Clin. Neurol. 2014, 122, 455–464. [Google Scholar] [CrossRef]
- Porta, S.; Danza, A.; Saavedra, M.A.; Carlomagno, A.; Goizueta, M.C.; Vivero, F.; Ruiz-Irastorza, G. Glucocorticoids in Systemic Lupus Erythematosus. Ten Questions and Some Issues. J. Clin. Med. 2020, 9, 2709. [Google Scholar] [CrossRef]
- Mejía-Vilet, J.M.; Ayoub, I. The Use of Glucocorticoids in Lupus Nephritis: New Pathways for an Old Drug. Front. Med. (Lausanne) 2021, 8, 622225. [Google Scholar] [CrossRef]
- Quattrocelli, M.; Zelikovich, A.S.; Salamone, I.M.; Fischer, J.A.; McNally, E.M. Mechanisms and Clinical Applications of Glucocorticoid Steroids in Muscular Dystrophy. J. Neuromuscul. Dis. 2021, 8, 39–52. [Google Scholar] [CrossRef]
- Garaffoni, C.; Adinolfi, A.; Bortoluzzi, A.; Filippou, G.; Giollo, A.; Sakellariou, G.; Sirotti, S.; Ughi, N.; Scirè, C.A.; Silvagni, E. Novel insights into the management of rheumatoid arthritis: One year in review 2022. Clin. Exp. Rheumatol. 2022, 40, 1247–1257. [Google Scholar] [CrossRef] [PubMed]
- Prasad, P.; Verma, S.; Ganguly, N.K.; Chaturvedi, V.; Mittal, S.A. Rheumatoid arthritis: Advances in treatment strategies. Mol. Cell. Biochem. 2022. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Burwick, N.; Sharma, S. Glucocorticoids in multiple myeloma: Past, present, and future. Ann. Hematol. 2019, 98, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Bhadri, V.A.; Trahair, T.N.; Lock, R.B. Glucocorticoid resistance in paediatric acute lymphoblastic leukaemia. J. Paediatr. Child. Health 2012, 48, 634–640. [Google Scholar] [CrossRef] [PubMed]
- Amratia, D.A.; Viola, H.; Ioachimescu, O.C. Glucocorticoid therapy in respiratory illness: Bench to bedside. J. Investig. Med. 2022. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Ferreto, L.E.D.; Bortoloti, D.S.; Fortes, P.C.N.; Follador, F.; Arruda, G.; Ximenez, J.P.; Wendt, G.W. Dexamethasone for treating SARS-CoV-2 infection: A systematic review and meta-analysis. Sao Paulo Med. J. 2021, 139, 657–661. [Google Scholar] [CrossRef]
- Landolf, K.M.; Lemieux, S.M.; Rose, C.; Johnston, J.P.; Adams, C.D.; Altshuler, J.; Berger, K.; Dixit, D.; Effendi, M.K.; Heavner, M.S.; et al. Corticosteroid use in ARDS and its application to evolving therapeutics for coronavirus disease 2019 (COVID-19): A systematic review. Pharmacotherapy 2022, 4, 71–90. [Google Scholar] [CrossRef]
- Kelly, W.J.; Gilbert, M.R. Glucocorticoids and immune checkpoint inhibitors in glioblastoma. J. Neurooncol. 2021, 151, 13–20. [Google Scholar] [CrossRef]
- Petrelli, F.; De Stefani, A.; Ghidini, A.; Bruschieri, L.; Riboldi, V.; Dottorini, L.; Iaculli, A.; Zaniboni, A.; Trevisan, F. Steroids use and survival in patients with glioblastoma multiforme: A pooled analysis. J. Neurol. 2021, 268, 440–447. [Google Scholar] [CrossRef]
- Dubinski, D.; Hattingen, E.; Senft, C.; Seifert, V.; Peters, K.G.; Reiss, Y.; Devraj, K.; Plate, K.H. Controversial roles for dexamethasone in glioblastoma—Opportunities for novel vascular targeting therapies. J. Cereb. Blood Flow Metab. 2019, 39, 1460–1468. [Google Scholar] [CrossRef]
- Herath, M.; Langdahl, B.; Ebeling, P.R.; Milat, F. Challenges in the diagnosis and management of glucocorticoid-induced osteoporosis in younger and older adults. Clin. Endocrinol. 2022, 96, 460–474. [Google Scholar] [CrossRef] [PubMed]
- Sarzani, R.; Spannella, F.; Giulietti, F.; Di Pentima, C.; Giordano, P.; Giacometti, A. Possible harm from glucocorticoid drugs misuse in the early phase of SARS-CoV-2 infection: A narrative review of the evidence. Intern. Emerg. Med. 2022, 17, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Li, J.X.; Cummins, C.L. Fresh insights into glucocorticoid-induced diabetes mellitus and new therapeutic directions. Nat. Rev. Endocrinol. 2022, 1–18. 2022, 1–18, Epub ahead of print. [Google Scholar] [CrossRef]
- Beaupere, C.; Liboz, A.; Fève, B.; Blondeau, B.; Guillemain, G. Molecular Mechanisms of Glucocorticoid-Induced Insulin Resistance. Int. J. Mol. Sci. 2021, 22, 623. [Google Scholar] [CrossRef]
- Hauser, B.; Raterman, H.; Ralston, S.H.; Lems, W.F. The Effect of Anti-rheumatic Drugs on the Skeleton. Calcif. Tissue Int. 2022. Epub ahead of print. [Google Scholar] [CrossRef]
- Laurent, M.R.; Goemaere, S.; Verroken, C.; Bergmann, P.; Body, J.J.; Bruyère, O.; Cavalier, E.; Rozenberg, S.; Lapauw, B.; Gielen, E. Prevention and Treatment of Glucocorticoid-Induced Osteoporosis in Adults: Consensus Recommendations from the Belgian Bone Club. Front. Endocrinol. 2022, 13, 908727. [Google Scholar] [CrossRef]
- Dhaibar, H.A.; Cruz-Topete, D. Predisposition of Women to Cardiovascular Diseases: A Side-Effect of Increased Glucocorticoid Signaling During the COVID-19 Pandemic? Front. Glob. Womens Health 2021, 2, 606833. [Google Scholar] [CrossRef]
- Karamanos, N.K.; Theocharis, A.D.; Piperigkou, Z.; Manou, D.; Passi, A.; Skandalis, S.S.; Vynios, D.H.; Orian-Rousseau, V.; Ricard-Blum, S.; Schmelzer, C.E.H.; et al. A guide to the composition and functions of the extracellular matrix. FEBS J. 2021, 288, 6850–6912. [Google Scholar] [CrossRef]
- Manou, D.; Caon, I.; Bouris, P.; Triantaphyllidou, I.E.; Giaroni, C.; Passi, A.; Karamanos, N.K.; Vigetti, D.; Theocharis, A.D. The Complex Interplay Between Extracellular Matrix and Cells in Tissues. Methods Mol. Biol. 2019, 1952, 1–20. [Google Scholar] [CrossRef]
- Karamanos, N.K.; Piperigkou, Z.; Theocharis, A.D.; Watanabe, H.; Franchi, M.; Baud, S.; Brézillon, S.; Götte, M.; Passi, A.; Vigetti, D.; et al. Proteoglycan Chemical Diversity Drives Multifunctional Cell Regulation and Therapeutics. Chem. Rev. 2018, 118, 9152–9232. [Google Scholar] [CrossRef]
- Theocharis, A.D.; Manou, D.; Karamanos, N.K. The extracellular matrix as a multitasking player in disease. FEBS J. 2019, 286, 2830–2869. [Google Scholar] [CrossRef] [PubMed]
- Theocharis, A.D.; Karamanos, N.K. Proteoglycans remodeling in cancer: Underlying molecular mechanisms. Matrix Biol. 2019, 75–76, 220–259. [Google Scholar] [CrossRef]
- Claassen, H.; Cellarius, C.; Scholz-Ahrens, K.E.; Schrezenmeir, J.; Glüer, C.C.; Schünke, M.; Kurz, B. Extracellular matrix changes in knee joint cartilage following bone-active drug treatment. Cell Tissue Res. 2006, 324, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Albano, M.B.; Skroch, G.P.; Ioshii, S.O.; Grahels, X.S.; de Alencar, P.G.; Matias, J.E. Computerized photocolorimetric analysis of the effects of intraarticular betamethasone on the proteoglycan concentration of leporine knee cartilage matrix: Influence of the number of intraarticular injections. Rev. Col. Bras. Cir. 2009, 36, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Barton, K.I.; Heard, B.J.; Sevick, J.L.; Martin, C.R.; Shekarforoush, S.M.M.; Chung, M.; Achari, Y.; Frank, C.B.; Shrive, N.G.; Hart, D.A. Posttraumatic Osteoarthritis Development and Progression in an Ovine Model of Partial Anterior Cruciate Ligament Transection and Effect of Repeated Intra-articular Methylprednisolone Acetate Injections on Early Disease. Am. J. Sport. Med. 2018, 46, 1596–1605. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.C.; Evans, C.H.; Grodzinsky, A.J. Effects of short-term glucocorticoid treatment on changes in cartilage matrix degradation and chondrocyte gene expression induced by mechanical injury and inflammatory cytokines. Arthritis Res. Ther. 2011, 13, R142. [Google Scholar] [CrossRef]
- Abubakr, N.; Salem, Z.; Ali, Z.; Assaly, M.E. Comparative evaluation of the early effects of the low-level laser therapy versus intra-articular steroids on temporomandibular joint acute osteoarthritis in rats: A histochemical, molecular and imaging evaluation. Dent. Med. Probl. 2018, 55, 359–366. [Google Scholar] [CrossRef]
- Malfait, A.M.; Tortorella, M.; Thompson, J.; Hills, R.; Meyer, D.M.; Jaffee, B.D.; Chinn, K.; Ghoreishi-Haack, N.; Markosyan, S.; Arner, E.C. Intra-articular injection of tumor necrosis factor-alpha in the rat: An acute and reversible in vivo model of cartilage proteoglycan degradation. Osteoarthr. Cartil. 2009, 17, 627–635. [Google Scholar] [CrossRef][Green Version]
- Suhovskih, A.V.; Molodykh, O.P.; Ushakov, V.S.; Politko, M.O.; Sokolov, D.K.; Koldysheva, E.V.; Grigorieva, E.V. Long-Term Exposure to Temozolomide Affects Locomotor Activity and Cartilage Structure of Elderly Experimental Rats. Biomedicines 2020, 8, 541. [Google Scholar] [CrossRef]
- Euppayo, T.; Siengdee, P.; Buddhachat, K.; Pradit, W.; Chomdej, S.; Ongchai, S.; Nganvongpanit, K. In vitro effects of triamcinolone acetonide and in combination with hyaluronan on canine normal and spontaneous osteoarthritis articular cartilage. In Vitro Cell Dev. Biol. Anim. 2016, 52, 723–735. [Google Scholar] [CrossRef]
- Byron, C.R.; Benson, B.M.; Stewart, A.A.; Pondenis, H.C. Effects of methylprednisolone acetate and glucosamine on proteoglycan production by equine chondrocytes in vitro. Am. J. Vet. Res. 2008, 69, 1123–1128. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Kang, X.; Qin, N.; Li, F.; Jin, X.; Ma, Z.; Qian, Z.; Wu, S. Melatonin protects chondrocytes from impairment induced by glucocorticoids via NAD+-dependent SIRT1. Steroids 2017, 126, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.W.; Zhang, T.; Wang, W.B. Gluococorticoid could influence extracellular matrix synthesis through Sox9 via p38 MAPK pathway. Rheumatol. Int. 2012, 32, 3669–3673. [Google Scholar] [CrossRef] [PubMed]
- Iwaniak, P.; Dobrowolski, P.; Tomaszewska, E.; Hułas-Stasiak, M.; Tomczyk, A.; Gawron, A. The influence of dexamethasone administered prenatally on cartilage of newborn spiny mouse (Acomys cahirinus) offspring. J. Dev. Orig. Health Dis. 2016, 7, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhao, Z.; Li, Y.; Zhang, X.; Li, B.; Chen, L.; Wang, H. Course-, dose-, and stage-dependent toxic effects of prenatal dexamethasone exposure on fetal articular cartilage development. Toxicol. Lett. 2018, 286, 1–9. [Google Scholar] [CrossRef]
- Randau, T.M.; Schildberg, F.A.; Alini, M.; Wimmer, M.D.; Haddouti, M.; Gravius, S.; Ito, K.; Stoddart, M.J. The effect of dexamethasone and triiodothyronine on terminal differentiation of primary bovine chondrocytes and chondrogenically differentiated mesenchymal stem cells. PLoS ONE 2013, 8, e72973. [Google Scholar] [CrossRef]
- Richardson, D.W.; Dodge, G.R. Dose-dependent effects of corticosteroids on the expression of matrix-related genes in normal and cytokine-treated articular chondrocytes. Inflamm. Res. 2003, 52, 39–49. [Google Scholar] [CrossRef]
- Black, R.M.; Flaman, L.L.; Lindblom, K.; Chubinskaya, S.; Grodzinsky, A.J.; Önnerfjord, P. Tissue catabolism and donor-specific dexamethasone response in a human osteochondral model of post-traumatic osteoarthritis. Arthritis Res. Ther. 2022, 24, 137. [Google Scholar] [CrossRef]
- Schwartze, J.T.; Becker, S.; Sakkas, E.; Wujak, Ł.A.; Niess, G.; Usemann, J.; Reichenberger, F.; Herold, S.; Vadász, I.; Mayer, K.; et al. Glucocorticoids recruit Tgfbr3 and Smad1 to shift transforming growth factor-β signaling from the Tgfbr1/Smad2/3 axis to the Acvrl1/Smad1 axis in lung fibroblasts. J. Biol. Chem. 2014, 289, 3262–3275. [Google Scholar] [CrossRef]
- McDougall, A.R.A.; Fosang, A.J.; Faggian, J.; Wallace, M.J.; Crossley, K.J.; Cole, T.J.; Hooper, S.B. Glucocorticoids influence versican and chondroitin sulphate proteoglycan levels in the fetal sheep lung. Respir. Res. 2018, 19, 155. [Google Scholar] [CrossRef]
- Todorova, L.; Gürcan, E.; Miller-Larsson, A.; Westergren-Thorsson, G. Lung fibroblast proteoglycan production induced by serum is inhibited by budesonide and formoterol. Am. J. Respir. Cell Mol. Biol. 2006, 34, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Kähäri, V.M.; Häkkinen, L.; Westermarck, J.; Larjava, H. Differential regulation of decorin and biglycan gene expression by dexamethasone and retinoic acid in cultured human skin fibroblasts. J. Invest. Dermatol. 1995, 104, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Watson, C.E.; Liu, C.; Williams, K.J.; Werth, V.P. Glucocorticoids induce a near-total suppression of hyaluronan synthase mRNA in dermal fibroblasts and in osteoblasts: A molecular mechanism contributing to organ atrophy. Biochem. J. 2000, 349, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Särnstrand, B.; Brattsand, R.; Malmström, A. Effect of glucocorticoids on glycosaminoglycan metabolism in cultured human skin fibroblasts. J. Invest. Dermatol. 1982, 79, 412–417. [Google Scholar] [CrossRef]
- Kimoto, S.; Cheng, S.L.; Zhang, S.F.; Avioli, L.V. The effect of glucocorticoid on the synthesis of biglycan and decorin in human osteoblasts and bone marrow stromal cells. Endocrinology 1994, 135, 2423–2431. [Google Scholar] [CrossRef]
- Eklund, K.K.; Humphries, D.E.; Xia, Z.; Ghildyal, N.; Friend, D.S.; Gross, V.; Stevens, R.L. Glucocorticoids inhibit the cytokine-induced proliferation of mast cells, the high affinity IgE receptor-mediated expression of TNF-alpha, and the IL-10-induced expression of chymases. J. Immunol. 1997, 158, 4373–4380. [Google Scholar]
- Wen, Y.; Shi, H.; Wu, Z.; Xiao, H.; Wang, H.; Chen, L. GR/Sp3/HDAC1/UGDH signaling participated in the maternal dexamethasone-induced dysplasia of the rat fetal growth plate. FASEB J. 2020, 34, 12834–12846. [Google Scholar] [CrossRef]
- Florine, E.M.; Miller, R.E.; Porter, R.M.; Evans, C.H.; Kurz, B.; Grodzinsky, A.J. Effects of Dexamethasone on Mesenchymal Stromal Cell Chondrogenesis and Aggrecanase Activity: Comparison of Agarose and Self-Assembling Peptide Scaffolds. Cartilage 2013, 4, 63–74. [Google Scholar] [CrossRef]
- Bentley, S.A.; Kirby, S.L.; Anklesaria, P.; Greenberger, J.S. Bone marrow stromal proteoglycan heterogeneity: Phenotypic variability between cell lines and the effects of glucocorticoid. J. Cell Physiol. 1988, 136, 182–187. [Google Scholar] [CrossRef]
- Tsidulko, A.; Bezier, C.; De La Bourdonnaye, G.; Suhovskih, A.V.; Pankova, T.M.; Kazanskaya, G.M.; Aidagulova, S.; Grigorieva, E. Conventional Anti-glioblastoma Chemotherapy Affects Proteoglycan Composition of Brain Extracellular Matrix in Rat Experimental Model in vivo. Front. Pharmacol. 2018, 9, 1104. [Google Scholar] [CrossRef]
- Tsidulko, A.Y.; Shevelev, O.B.; Khotskina, A.S.; Kolpakova, M.A.; Suhovskih, A.V.; Kazanskaya, G.M.; Volkov, A.M.; Aidagulova, S.V.; Zavyalov, E.L.; Grigorieva, E.V. Chemotherapy-Induced Degradation of Glycosylated Components of the Brain Extracellular Matrix Promotes Glioblastoma Relapse Development in an Animal Model. Front. Oncol. 2021, 11, 713139. [Google Scholar] [CrossRef] [PubMed]
- Wei, F.; Song, J.; Zhang, C.; Lin, J.; Xue, R.; Shan, L.D.; Gong, S.; Zhang, G.X.; Qin, Z.H.; Xu, G.Y.; et al. Chronic stress impairs the aquaporin-4-mediated glymphatic transport through glucocorticoid signaling. Psychopharmacology 2019, 236, 1367–1384. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Bellamkonda, R.V. Dexamethasone-coated neural probes elicit attenuated inflammatory response and neuronal loss compared to uncoated neural probes. Brain Res. 2007, 1148, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.L.; Lee, Y.H.; Tsai, S.Y.; Hsu, C.Y.; Sun, Y.Y.; Yang, L.Y.; Tsai, S.H.; Yang, W.C. Methylprednisolone inhibits the expression of glial fibrillary acidic protein and chondroitin sulfate proteoglycans in reactivated astrocytes. Glia 2008, 56, 1390–1400. [Google Scholar] [CrossRef] [PubMed]
- Yanase, F.; Tosif, S.H.; Churilov, L.; Yee, K.; Bellomo, R.; Gunn, K.; Kim, C.; Krizhanovskii, C.; Hahn, R.G.; Riedel, B.; et al. A Randomized, Multicenter, Open-Label, Blinded End Point, Phase 2, Feasibility, Efficacy, and Safety Trial of Preoperative Microvascular Protection in Patients Undergoing Major Abdominal Surgery. Anesth. Analg. 2021, 133, 1036–1047. [Google Scholar] [CrossRef]
- Johnson, D.H.; Bradley, J.M.; Acott, T.S. The effect of dexamethasone on glycosaminoglycans of human trabecular meshwork in perfusion organ culture. Invest. Ophthalmol. Vis. Sci. 1990, 31, 2568–2571. [Google Scholar]
- Kuroda, M.; Sasamura, H.; Shimizu-Hirota, R.; Mifune, M.; Nakaya, H.; Kobayashi, E.; Hayashi, M.; Saruta, T. Glucocorticoid regulation of proteoglycan synthesis in mesangial cells. Kidney Int. 2002, 62, 780–789. [Google Scholar] [CrossRef]
- Wickert, L.; Abiaka, M.; Bolkenius, U.; Gressner, A.M. Corticosteroids stimulate selectively transforming growth factor (TGF)-beta receptor type III expression in transdifferentiating hepatic stellate cells. J. Hepatol. 2004, 40, 69–76. [Google Scholar] [CrossRef]
- Wong, M.W.; Tang, Y.Y.; Lee, S.K.; Fu, B.S. Glucocorticoids suppress proteoglycan production by human tenocytes. Acta Orthop. 2005, 76, 927–931. [Google Scholar] [CrossRef]
- Nakamura, T.; Ebihara, I.; Tomino, Y.; Koide, H. Glucocorticoid ameliorates altered gene expression of extracellular matrix components in kidneys of New Zealand black/white F1 mice. Clin. Sci. 1992, 83, 701–709. [Google Scholar] [CrossRef]
- Xiang, X.B.; Chen, H.; Wu, Y.L.; Wang, K.; Yue, X.; Cheng, X.Q. The Effect of Preoperative Methylprednisolone on Postoperative Delirium in Older Patients Undergoing Gastrointestinal Surgery: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Gerontol. Biol. Med. Sci. 2022, 77, 517–523. [Google Scholar] [CrossRef]
- Short, K.L.; Bird, A.D.; Seow, B.K.L.; Ng, J.; McDougall, A.R.A.; Wallace, M.J.; Hooper, S.B.; Cole, T.J. Glucocorticoid signalling drives reduced versican levels in the fetal mouse lung. J. Mol. Endocrinol. 2020, 64, 155–164. [Google Scholar] [CrossRef]
- Bird, A.D.; Choo, Y.L.; Hooper, S.B.; McDougall, A.R.; Cole, T.J. Mesenchymal glucocorticoid receptor regulates the development of multiple cell layers of the mouse lung. Am. J. Respir. Cell Mol. Biol. 2014, 50, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, H.; Ichikawa, F.; Andres, J.L.; Massagué, J.; Noda, M. Dexamethasone enhancement of betaglycan (TGF-beta type III receptor) gene expression in osteoblast-like cells. Exp. Cell Res. 1994, 211, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Iozzo, R.V.; Sanderson, R.D. Proteoglycans in cancer biology, tumour microenvironment and angiogenesis. J. Cell Mol. Med. 2011, 15, 1013–1031. [Google Scholar] [CrossRef] [PubMed]
- De Pasquale, V.; Pavone, L.M. Heparan Sulfate Proteoglycan Signaling in Tumor Microenvironment. Int. J. Mol. Sci. 2020, 21, 6588. [Google Scholar] [CrossRef]
- Lund, M.E.; Campbell, D.H.; Walsh, B.J. The Role of Glypican-1 in the Tumour Microenvironment. Adv. Exp. Med. Biol. 2020, 1245, 163–176. [Google Scholar] [CrossRef]
- Mellai, M.; Casalone, C.; Corona, C.; Crociara, P.; Favole, A.; Cassoni, P.; Schiffer, D.; Boldorini, R. Chondroitin Sulphate Proteoglycans in the Tumour Microenvironment. Adv. Exp. Med. Biol. 2020, 1272, 73–92. [Google Scholar] [CrossRef]
- Benad-Mehner, P.; Thiele, S.; Rachner, T.D.; Göbel, A.; Rauner, M.; Hofbauer, L.C. Targeting syndecan-1 in breast cancer inhibits osteoclast functions through up-regulation of osteoprotegerin. J. Bone Oncol. 2013, 3, 18–24. [Google Scholar] [CrossRef]
- Yevdokimova, N.; Freshney, R.I. Activation of paracrine growth factors by heparan sulphate induced by glucocorticoid in A549 lung carcinoma cells. Br. J. Cancer 1997, 76, 281–289. [Google Scholar] [CrossRef][Green Version]
- Xu, C.; Sun, M.; Zhang, X.; Xu, Z.; Miyamoto, H.; Zheng, Y. Activation of Glucocorticoid Receptor Inhibits the Stem-Like Properties of Bladder Cancer via Inactivating the β-Catenin Pathway. Front. Oncol. 2020, 10, 1332. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Fang, F.; Song, X.; Wang, Y.; Huang, G.; Su, J.; Hui, N.; Lu, J. The pro-adhesive and pro-survival effects of glucocorticoid in human ovarian cancer cells. J. Mol. Endocrinol. 2016, 57, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Mackie, A.E.; Freshney, R.I.; Akturk, F.; Hunt, G. Glucocorticoids and the cell surface of human glioma cells: Relationship to cytostasis. Br. J. Cancer Suppl. 1988, 9, 101–107. [Google Scholar] [PubMed]
- Cenciarini, M.; Valentino, M.; Belia, S.; Sforna, L.; Rosa, P.; Ronchetti, S.; D’Adamo, M.C.; Pessia, M. Dexamethasone in Glioblastoma Multiforme Therapy: Mechanisms and Controversies. Front. Mol. Neurosci. 2019, 12, 65. [Google Scholar] [CrossRef]
- Mistry, A.M.; Jonathan, S.V.; Monsour, M.A.; Mobley, B.C.; Clark, S.W.; Moots, P.L. Impact of postoperative dexamethasone on survival, steroid dependency, and infections in newly diagnosed glioblastoma patients. Neurooncol. Pract. 2021, 8, 589–600. [Google Scholar] [CrossRef]
- Madalena, K.M.; Lerch, J.K. The Effect of Glucocorticoid and Glucocorticoid Receptor Interactions on Brain, Spinal Cord, and Glial Cell Plasticity. Neural. Plast. 2017, 2017, 8640970. [Google Scholar] [CrossRef]
- Viho, E.M.G.; Buurstede, J.C.; Mahfouz, A.; Koorneef, L.L.; van Weert, L.T.C.M.; Houtman, R.; Hunt, H.J.; Kroon, J.; Meijer, O.C. Corticosteroid Action in the Brain: The Potential of Selective Receptor Modulation. Neuroendocrinology 2019, 109, 266–276. [Google Scholar] [CrossRef]
- Atucha, E.; Zalachoras, I.; van den Heuvel, J.K.; van Weert, L.T.; Melchers, D.; Mol, I.M.; Belanoff, J.K.; Houtman, R.; Hunt, H.; Roozendaal, B.; et al. A Mixed Glucocorticoid/Mineralocorticoid Selective Modulator with Dominant Antagonism in the Male Rat Brain. Endocrinology 2015, 156, 4105–4114. [Google Scholar] [CrossRef]
- Srinivasan, M.; Lahiri, D.K. Glucocorticoid-Induced Leucine Zipper in Central Nervous System Health and Disease. Mol. Neurobiol. 2017, 54, 8063–8070. [Google Scholar] [CrossRef]
- Aldaz, P.; Fernández-Celis, A.; López-Andrés, N.; Arozarena, I. Novel Insights into the Role of the Mineralocorticoid Receptor in Human Glioblastoma. Int. J. Mol. Sci. 2021, 22, 11656. [Google Scholar] [CrossRef]
- Kaup, B.; Schindler, I.; Knüpfer, H.; Schlenzka, A.; Preiss, R.; Knüpfer, M.M. Time-dependent inhibition of glioblastoma cell proliferation by dexamethasone. J. Neurooncol. 2001, 51, 105–110. [Google Scholar] [CrossRef]
- Nicholson, C.; Hrabětová, S. Brain Extracellular Space: The Final Frontier of Neuroscience. Biophys. J. 2017, 113, 2133–2142. [Google Scholar] [CrossRef] [PubMed]
- Melrose, J.; Hayes, A.J.; Bix, G. The CNS/PNS Extracellular Matrix Provides Instructive Guidance Cues to Neural Cells and Neuroregulatory Proteins in Neural Development and Repair. Int. J. Mol. Sci. 2021, 22, 5583. [Google Scholar] [CrossRef] [PubMed]
- Perus, L.J.M.; Walsh, L.A. Microenvironmental Heterogeneity in Brain Malignancies. Front. Immunol. 2019, 10, 2294. [Google Scholar] [CrossRef] [PubMed]
- Reinhard, J.; Brösicke, N.; Theocharidis, U.; Faissner, A. The extracellular matrix niche microenvironment of neural and cancer stem cells in the brain. Int. J. Biochem. Cell Biol. 2016, 81, 174–183. [Google Scholar] [CrossRef]
- Brown, Y.; Hua, S.; Tanwar, P.S. Extracellular matrix-mediated regulation of cancer stem cells and chemoresistance. Int. J. Biochem. Cell Biol. 2019, 109, 90–104. [Google Scholar] [CrossRef]
- So, J.S.; Kim, H.; Han, K.S. Mechanisms of Invasion in Glioblastoma: Extracellular Matrix, Ca2+ Signaling, and Glutamate. Front. Cell Neurosc. 2021, 15, 663092. [Google Scholar] [CrossRef]
- Mohiuddin, E.; Wakimoto, H. Extracellular matrix in glioblastoma: Opportunities for emerging therapeutic approaches. Am. J. Cancer Res. 2021, 11, 3742–3754. [Google Scholar]
- Belousov, A.; Titov, S.; Shved, N.; Garbuz, M.; Malykin, G.; Gulaia, V.; Kagansky, A.; Kumeiko, V. The Extracellular Matrix and Biocompatible Materials in Glioblastoma Treatment. Front. Bioeng. Biotechnol. 2019, 7, 341. [Google Scholar] [CrossRef]
- Hatoum, A.; Mohammed, R.; Zakieh, O. The unique invasiveness of glioblastoma and possible drug targets on extracellular matrix. Cancer Manag. Res. 2019, 11, 1843–1855. [Google Scholar] [CrossRef]
- Butler, M.; Prasad, S.; Srivastava, S.K. Targeting Glioblastoma Tumor Microenvironment. Adv. Exp. Med. Biol. 2020, 1296, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wade, A.; Robinson, A.E.; Engler, J.R.; Petritsch, C.; James, C.D.; Phillips, J.J. Proteoglycans and their roles in brain cancer. FEBS J. 2013, 280, 2399–2417. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, N.B.; Domowicz, M.S. Proteoglycans in brain development and pathogenesis. FEBS Lett. 2018, 592, 3791–3805. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Wang, S. Proteoglycans as Therapeutic Targets in Brain Cancer. Front. Oncol. 2020, 10, 1358. [Google Scholar] [CrossRef]
- Xiong, A.; Spyrou, A.; Forsberg-Nilsson, K. Involvement of Heparan Sulfate and Heparanase in Neural Development and Pathogenesis of Brain Tumors. Adv. Exp. Med. Biol. 2020, 1221, 365–403. [Google Scholar] [CrossRef]
- Silver, D.J.; Siebzehnrubl, F.A.; Schildts, M.J.; Yachnis, A.T.; Smith, G.M.; Smith, A.A.; Scheffler, B.; Reynolds, B.A.; Silver, J.; Steindler, D.A. Chondroitin sulfate proteoglycans potently inhibit invasion and serve as a central organizer of the brain tumor microenvironment. J. Neurosci. 2013, 33, 15603–15617. [Google Scholar] [CrossRef]
- Xiao, W.; Wang, S.; Zhang, R.; Sohrabi, A.; Yu, Q.; Liu, S.; Ehsanipour, A.; Liang, J.; Bierman, R.D.; Nathanson, D.A.; et al. Bioengineered scaffolds for 3D culture demonstrate extracellular matrix-mediated mechanisms of chemotherapy resistance in glioblastoma. Matrix Biol. 2020, 85–86, 128–146. [Google Scholar] [CrossRef]
- Grigorieva, E.V. Radiation Effects on Brain Extracellular Matrix. Front. Oncol. 2020, 10, 576701. [Google Scholar] [CrossRef]
- Politko, M.O.; Prokaeva, A.I.; Pashkovskaya, O.A.; Kuper, K.E.; Zheravin, A.A.; Kliver, E.E.; Tsidulko, A.Y.; Aidagulova, S.V.; Grigorieva, E.V. Single X-ray irradiation modulates proteoglycan expression in brain tissue: Investigation using mouse model. Mol. Biol. Rep. 2020, 47, 5657–5663. [Google Scholar] [CrossRef]
- Cho, J.A.; Kim, B.J.; Hwang, Y.J.; Woo, S.W.; Noh, T.S.; Suh, M.W. Effect and Biocompatibility of a Cross-Linked Hyaluronic Acid and Polylactide-co-glycolide Microcapsule Vehicle in Intratympanic Drug Delivery for Treating Acute Acoustic Trauma. Int. J. Mol. Sci. 2021, 22, 5720. [Google Scholar] [CrossRef]
- D’Amico, R.; Siracusa, R.; Fusco, R.; Cordaro, M.; Genovese, T.; Peritore, A.F.; Gugliandolo, E.; Crupi, R.; Impellizzeri, D.; Cuzzocrea, S.; et al. Protective effects of Colomast®, A New Formulation of Adelmidrol and Sodium Hyaluronate, in A Mouse Model of Acute Restraint Stress. Int. J. Mol. Sci. 2020, 21, 8136. [Google Scholar] [CrossRef] [PubMed]
- Busani, S.; Tosi, M.; Mighali, P.; Vandelli, P.; D’Amico, R.; Marietta, M.; Forfori, F.; Donati, A.; Cinnella, G.; De Monte, A.; et al. Multi-centre, three arm, randomized controlled trial on the use of methylprednisolone and unfractionated heparin in critically ill ventilated patients with pneumonia from SARS-CoV-2 infection: A structured summary of a study protocol for a randomised controlled trial. Trials 2020, 21, 724. [Google Scholar] [CrossRef] [PubMed]
- Mamelle, E.; Kechai, N.E.; Granger, B.; Sterkers, O.; Bochot, A.; Agnely, F.; Ferrary, E.; Nguyen, Y. Effect of a liposomal hyaluronic acid gel loaded with dexamethasone in a guinea pig model after manual or motorized cochlear implantation. Eur. Arch. Otorhinolaryngol. 2017, 274, 729–736. [Google Scholar] [CrossRef] [PubMed]
| GC | Dose, Regimen | Tissue/Cell/Animal | Detection | Changes in PGs and GAGs | Ref. |
|---|---|---|---|---|---|
| Cartilage and Chondrocytes | |||||
| DXM | 2.5 mg/kg for five cycles (5 consecutive days with breaks of 16 days) | Wistar rats | 6 months | Total GAGs (↗); sulfated GAGs in TMZ-treated animals (↘) | [39] |
| 125 μg/kg from gestation day 20 until parturition | Pregnant spiny mice | Offspring | Total PGs in articular and growth cartilages in the offspring (↘) | [44] | |
| 0.2., 0.8, and 1.2 mg/kg once a day from day 12 to day 17 of gestation | Pregnant C57BL/6 mice | Offspring | Gene and protein expression of aggrecan in offspring (↘) | [45] | |
| 1 mg/kg | Rats | Prevented TNFα-induced degradation of aggrecan | [38] | ||
| 100 nM and higher | Bovine and human cartilage explants | 6 days | Prevented TNFα-induced GAG and PG loss | [36] | |
| 100 nM | Human osteochondral explants | 21 days | Prevented a loss of sulfated GAGs from injury- and cytokine-treated explants | [48] | |
| 10−6, 10−7, and 10−8 M | Chondrocytes isolated from mouse knee joints | 24 h | Total PG content (↘) | [42] | |
| 1 µM | Primary bovine articular chondrocytes Chondrogenically differentiated mesenchymal stem cells | Day 21, day 14, and days 21–35 | GAG production (↗) | [46] | |
| 50–150 μM | Human chondrocytes from knee joints | 1–7 days | Aggrecan (↘) | [43] | |
| DXM triamcinolone prednisolone | 10−10 to 10−4 M | Primary equine articular chondrocytes from foals, as well as 7-year-old and adult horses | 24 h | Biglycan (=) Decorin (=) | [47] |
| Prednisolone | 0.5–1 mg/kg/day for 6 months and 13 months | Gottingen miniature pigs | 6 months 13 months | No change; GAG content (↘) | [33] |
| Methylprednisolone acetate | 80 mg/mL, 4 injections | Suffolk cross sheep | 20 weeks | PG content (↘) | [35] |
| 0.05 and 0.5 mg/mL | Articular cartilage from1–10-year-old horses | Total PGs (↘) | [41] | ||
| Betamethasone | Californian rabbits | 1–5 weeks 6–8 weeks | No change; total PG content (↘) | [34] | |
| Triamcinolone acetonide | 1.25, 2.5, and 5 mg/mL | Canine cartilage explant | PG content (↘) | [40] | |
| 0.11 mg/mL | Primary canine normal chondrocytes Osteoarthritic chondrocytes | Aggrecan (↘) Aggrecan (↗) | [40] | ||
| Cortisone | Rats | 1 and 4 weeks | Restored osteoarthritis-induced decrease in total GAG content | [37] | |
| Lung and Fibroblasts | |||||
| DXM | 20 nM | C57Bl/6J mice | 24 h | Betaglycan at mRNA and protein levels (↗) | [49] |
| DXM, methylprednisolone, budesonide, and fluticasone | Mouse fibroblasts | Betaglycan (↗) | [49] | ||
| Cortisol | 1.5–4 mg/day | Fetal sheep lung | 9 days | Chondrotin-6-sulfate (↘); Versican (=); chondroitin-4-sulfate (=) | [50] |
| Betamethasone | 11.4 mg | Fetal sheep lung | 24 h and 36 h | Versican (↘); chondrotin-6-sulfate (↘); chondroitin-4-sulfate (↘) | [50] |
| Budesonide | 10−8 M | Human lung fibroblasts | 24 h | Serum-induced total PG increase (↘); Decorin (↘); Versican (↘) | [51] |
| Dermal Fibroblasts | |||||
| DXM | 1µM | Human skin fibroblasts | Decorin at mRNA and protein levels (↗); Biglycan (=); the length of CS/DS chains attached to Decorin and Biglycan core proteins (=); prevented TGFβ-induced downregulation of Decorin and upregulation of Biglycan | [52] | |
| 1 µM | Human skin fibroblasts MG-63 osteoblast-like osteosarcoma cell line | 1–2 h | Hyaluronan synthase 2 (HAS2) (↘) | [53] | |
| Fluocinolone acetonide, budesonide, and hydrocortisone | 10−11–10−5 M | Human skin fibroblasts | Accumulation of hyaluronic acid (HA) (↘); DS content (↘) in the culture medium on the cell surface and in the cells; decrease in HS content (↘) in culture medium but not on cell surface; considerably less effect | [54] | |
| Osteoblasts and Bone Marrow Stromal Cells | |||||
| DXM | 10−10–10−7 M | Human osteoblasts (HOBs) and adult human bone marrow stromal cells (BMSC) | 7 days | Decorin at mRNA and protein levels (↗); Biglycan at mRNA and protein levels (↘) in both the conditioned medium and the cell layer | [55] |
| 10−10–10−7 M | Osteoblast-like and preosteoblast-like cell lines (murine MC3T3-E1 and rat RCT1) | 9–48 h | Betaglycan (↗) | [56] | |
| 10−10–10−7 M | Immature, non-transformed mouse bone-marrow-derived mast cells (mBMMCs) | 9–48 h | Serglycin (↗) | [56] | |
| 0.2 mg/kg once a day from gestational day 9 to 20 | Rats | Total PG content (↘) in the growth plate | [57] | ||
| 100 nM | Young bovine bone marrow stromal cells (BMSCs) seeded on agarose hydrogels | PG biosynthesis (↗) | [58] | ||
| 100 nM | Young bovine bone marrow stromal cells (BMSCs) seeded on peptide scaffolds | PG accumulation (↘) | [58] | ||
| Hydrocortisone | 1 μM | D2XRII, Bl6, and 14F1 bone marrow stromal cell lines | HSPG synthesis (↘) in all three cell lines; altered pattern of CS/DSPGs in the culture media by DS1 and DS3 (↗) DS2 (↘) | [59] | |
| Brain | |||||
| DXM | 1 mg/kg 2.5 and 5 mg/kg | Wistar rats | 24 h | PG expression (=); overall transcriptional activity of PGs (↗); syndecan-1 (↗); glypican-1 (↗); brevican (↗); CSPG4/NG2 (↗); Lumican (↗); Decorin (↘); CS content in hippocampus (↗); CS content in cortex (↘); HS content (↘) in both | [60] |
| 1 mg/kg | SCID mice | Glypican-1 (↗); syndecan-1 (↗); Versican (↗); CS content (↘) in the subcortex; Biglycan in cortex (↗) | [61] | ||
| 5 mg/kg once daily for 6 consecutive weeks | Adult mice | Agrin (↘) | [62] | ||
| 100 μg DEX in 20 μL nitrocellulose Local delivery into rat brain | Rats | 7 days | CS content (↘) | [63] | |
| 0.01–0.5 μM 50–200 μM | Rat organotypic hippocampal cultures | Glypican-1 (↗); Versican (↗); syndecan-1 (↘); Biglycan (↘) | [60] | ||
| Methylprednisolone | 30 mg/kg body weight intravenously immediately after SCI surgery | Female Long–Evans rats (acute spinal cord injury model) | 24 h | Neurocan (↘) | [64] |
| Pretreatment 10 and 50 μM | AMPA + cyclothiazide-induced reactivated primary cultured astrocytes (Sprague–Dawley rats) | Neurocan (↘) Phosphacan (↘) | [64] | ||
| Other Tissue and Cells | |||||
| DXM | 16 mg | Patients undergoing colorectal, pancreas, or liver surgery | Postoperative day 1 | Plasma syndecan-1 level (=); heparan sulfate content (↘) | [65] |
| 0.55 μM | Human eyes in ex vivo perfusion organ culture | 7 days and 14–21 days after treatment | No change In total GAG (CS, DS, HS, and HA) content in human trabecular meshwork (↗) | [66] | |
| Rat mesangial cells (RMCs) and human mesangial cells (HMCs) | Total CS/DSPGs synthesis and secretion (↘); Decorin (↘) Biglycan (↗) | [67] | |||
| DXM, hydrocortisone Aldosterone | 200 or 400 ng/mL 200 ng/mL 500 ng/mL | Hepatic stellate cells (HSCs) | Betaglycan mRNA level (↗) | [68] | |
| DXM triamcinolone | 1 μM 1 μM | Primary cultures of human tenocytes from explants of healthy human patellar tendon | Total PG content (↘) | [69] | |
| Methylprednisolone | New Zealand Black/White F1 mice | Onset of murine lupus nephritis (5 months of age) | Expression, distribution, and intensity of HSPG staining in renal cortex (↘) | [70] | |
| 2 mg/kg single preoperative dose | Patients undergoing gastrointestinal surgery | 1 and 3 days after surgery | Syndecan-1 (↘); HS content (↘) in endothelial glycocalyx | [71] | |
| GC | Dose | Tissue/Cells/Animals | Detection | Changes in PGs and GAGs | Ref. |
|---|---|---|---|---|---|
| DXM | Up to 10−6 M | MCF-7 breast cancer cells | 72 h | mRNA and membrane-bound form of syndecan-1 (↗); concentration of the soluble form of syndecan-1 (=) | [79] |
| 0.25 µM | Subconfluent A549 cells | 72 h | Heparan sulfate (↗); hyaluronic acid (↗) | [80] | |
| 100 nmol/L | Bladder cancer cells | 48 h | CD44 expression (↘) | [81] | |
| 10−7 mol/L | HO-8910 and SKOV-3 human ovarian cancer cells | 6–48 h | CD44 protein level (=); hyaluronic acid secretion (=) | [82] | |
| DXM methylprednisolone | 25 μM | Primary cultures of human gliomas (mainly anaplastic astrocytomas) | Total GAG content (↗); both in the cell medium and pronase digest of the cells’ total GAG content: in the culture medium (↘); in the protease cell digest (↗) | [83] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Strokotova, A.V.; Grigorieva, E.V. Glucocorticoid Effects on Proteoglycans and Glycosaminoglycans. Int. J. Mol. Sci. 2022, 23, 15678. https://doi.org/10.3390/ijms232415678
Strokotova AV, Grigorieva EV. Glucocorticoid Effects on Proteoglycans and Glycosaminoglycans. International Journal of Molecular Sciences. 2022; 23(24):15678. https://doi.org/10.3390/ijms232415678
Chicago/Turabian StyleStrokotova, Anastasia V., and Elvira V. Grigorieva. 2022. "Glucocorticoid Effects on Proteoglycans and Glycosaminoglycans" International Journal of Molecular Sciences 23, no. 24: 15678. https://doi.org/10.3390/ijms232415678
APA StyleStrokotova, A. V., & Grigorieva, E. V. (2022). Glucocorticoid Effects on Proteoglycans and Glycosaminoglycans. International Journal of Molecular Sciences, 23(24), 15678. https://doi.org/10.3390/ijms232415678






