Magnetic Fe/Fe3C@C Nanoadsorbents for Efficient Cr (VI) Removal
Abstract
1. Introduction
2. Results and Discussion
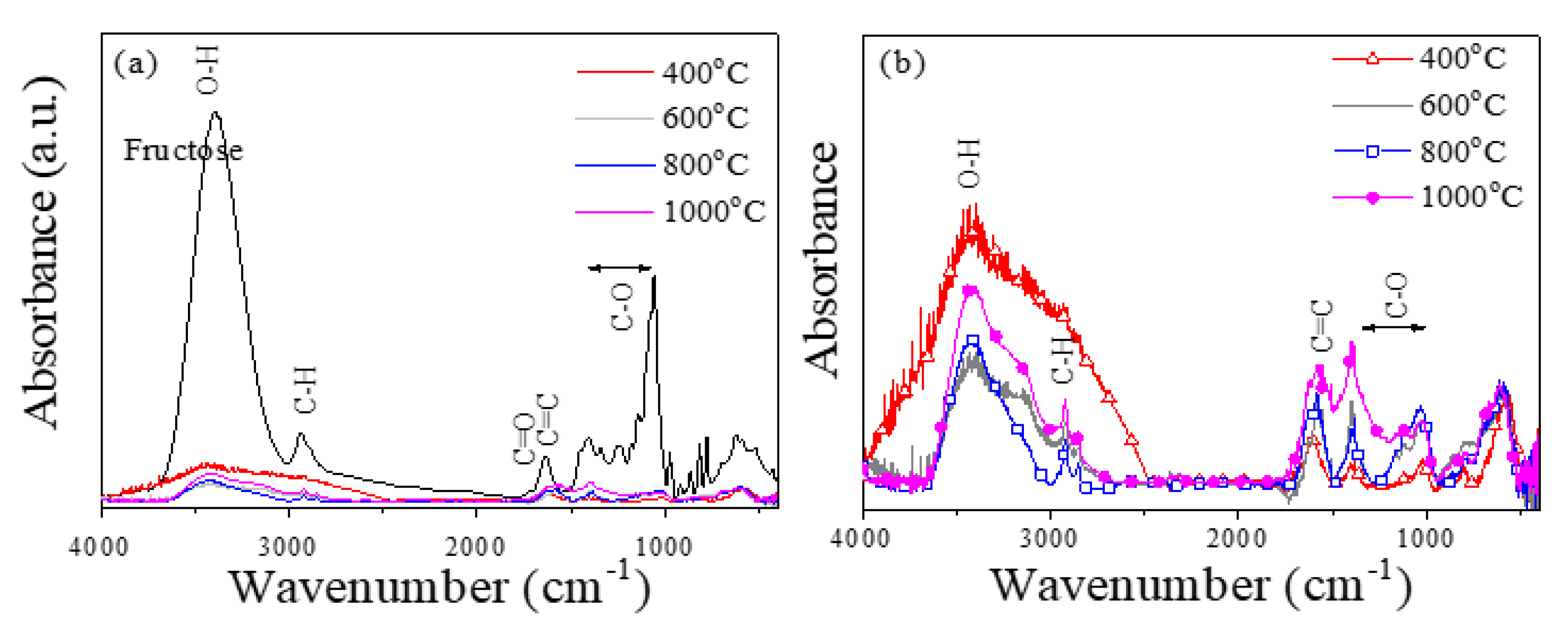
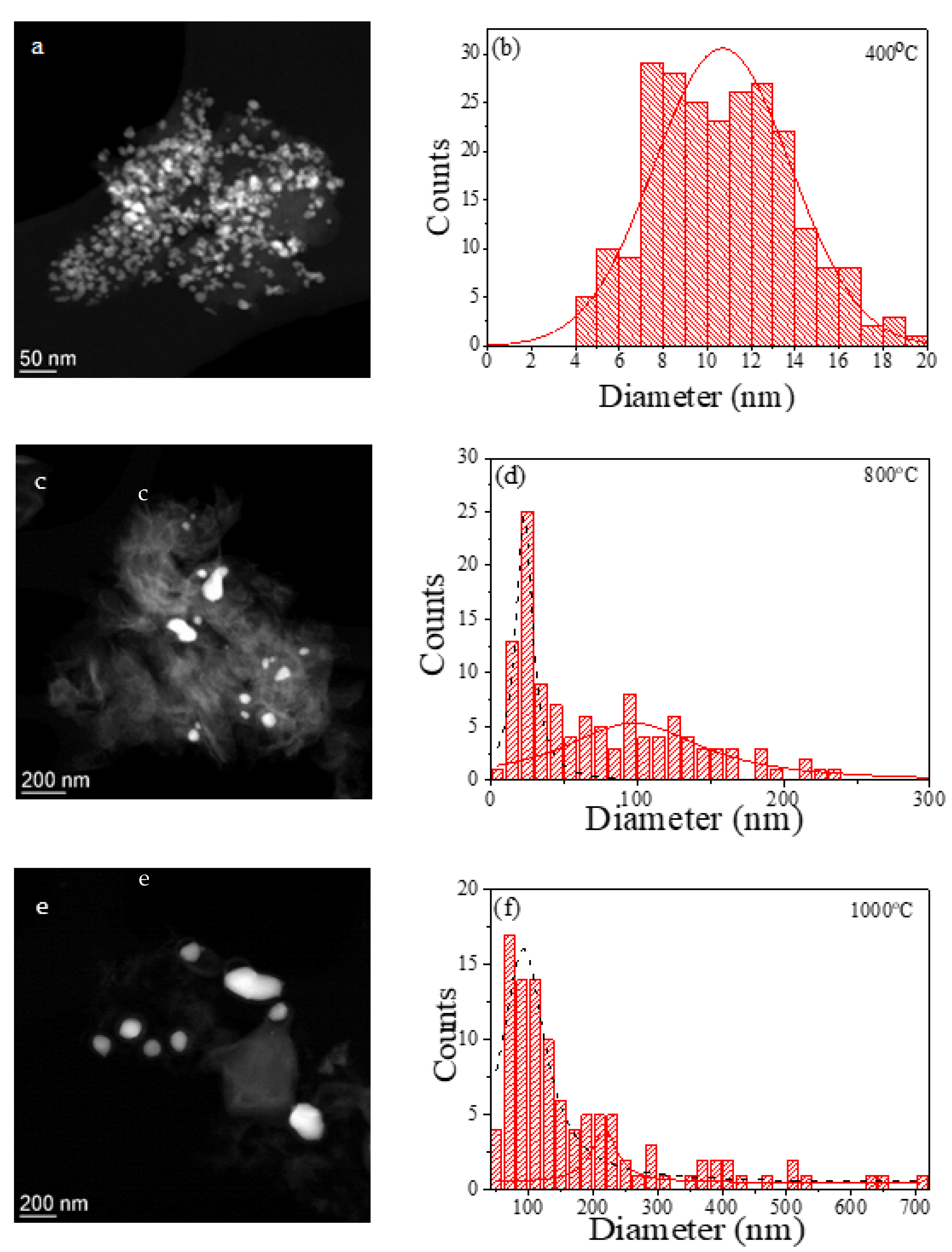
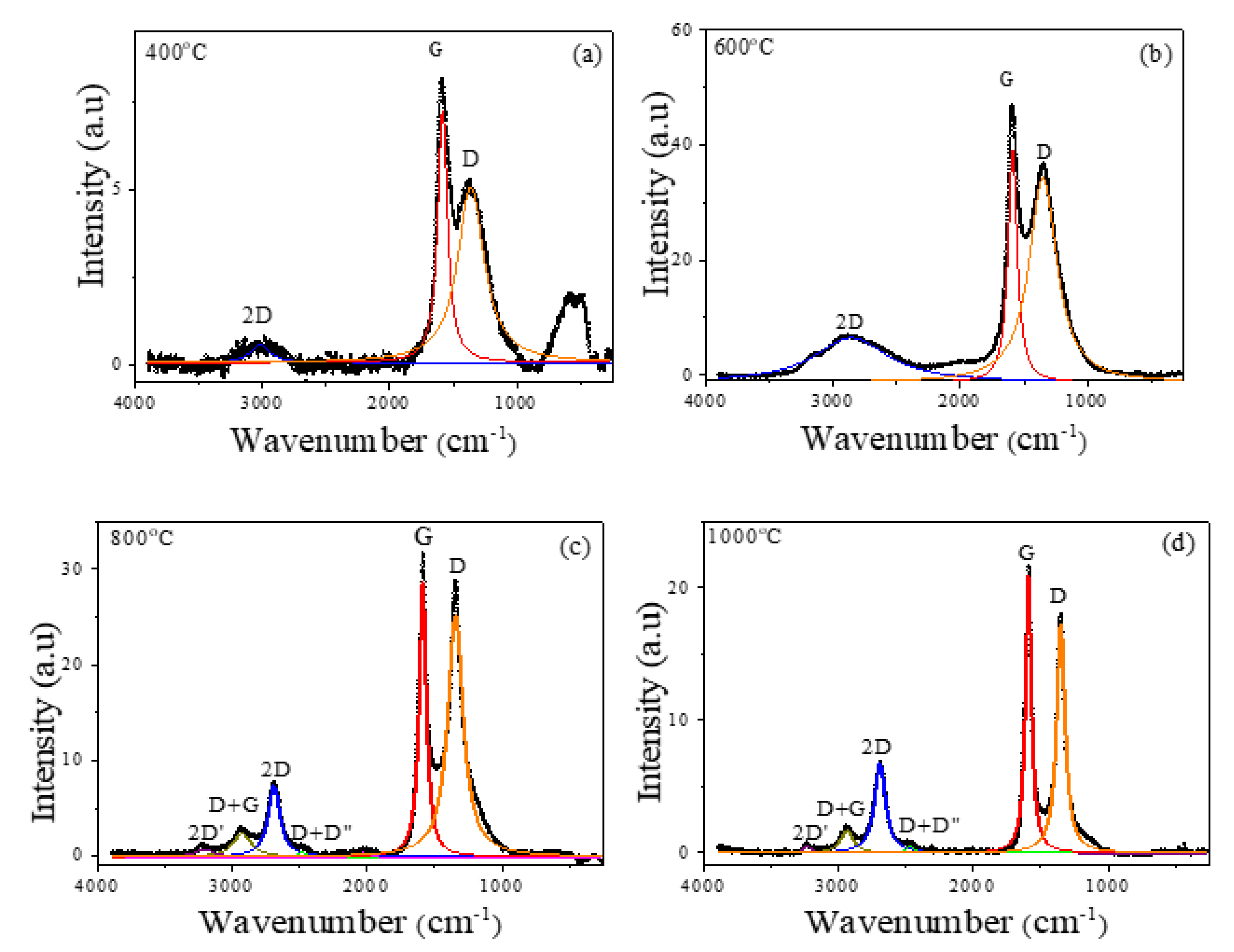
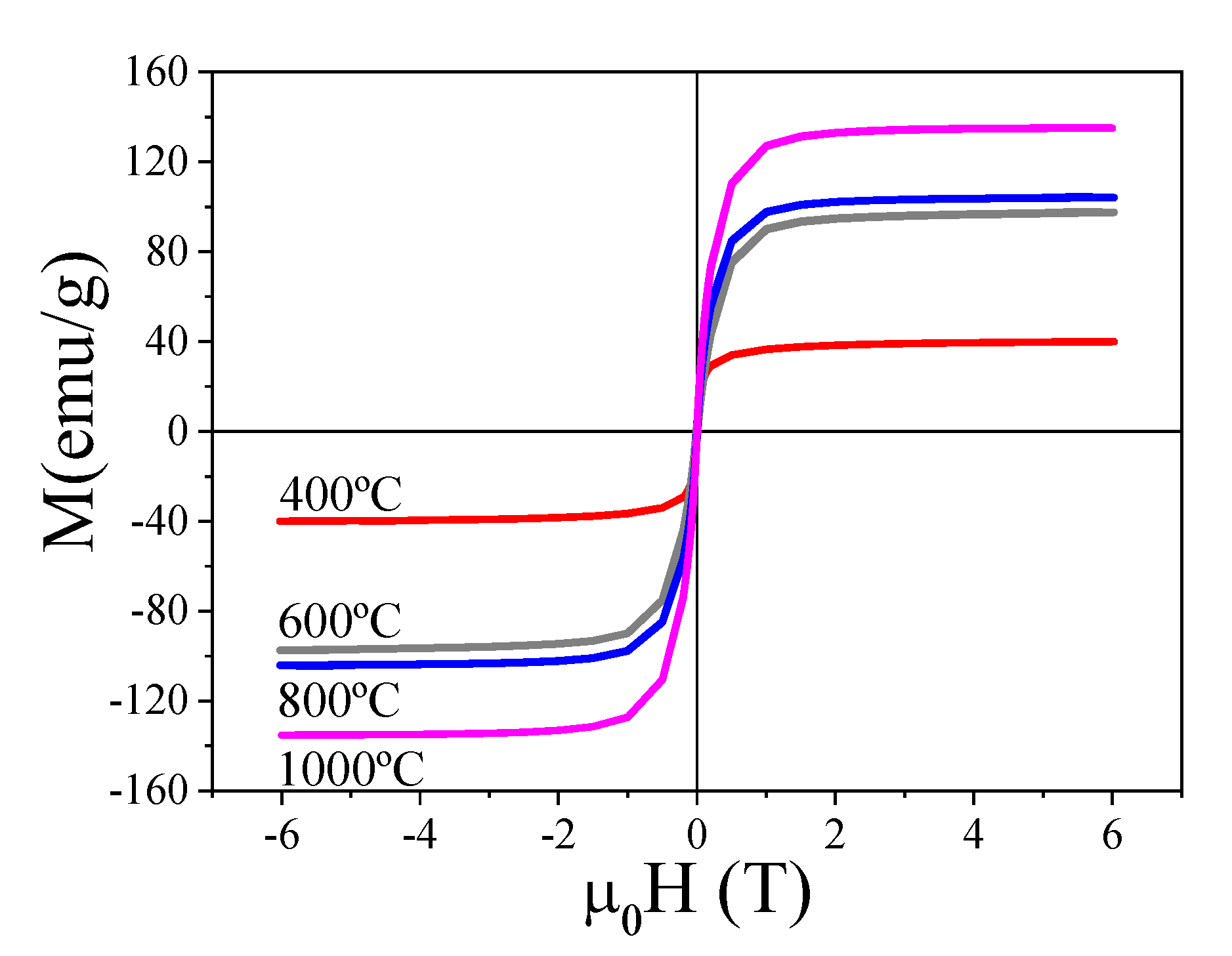
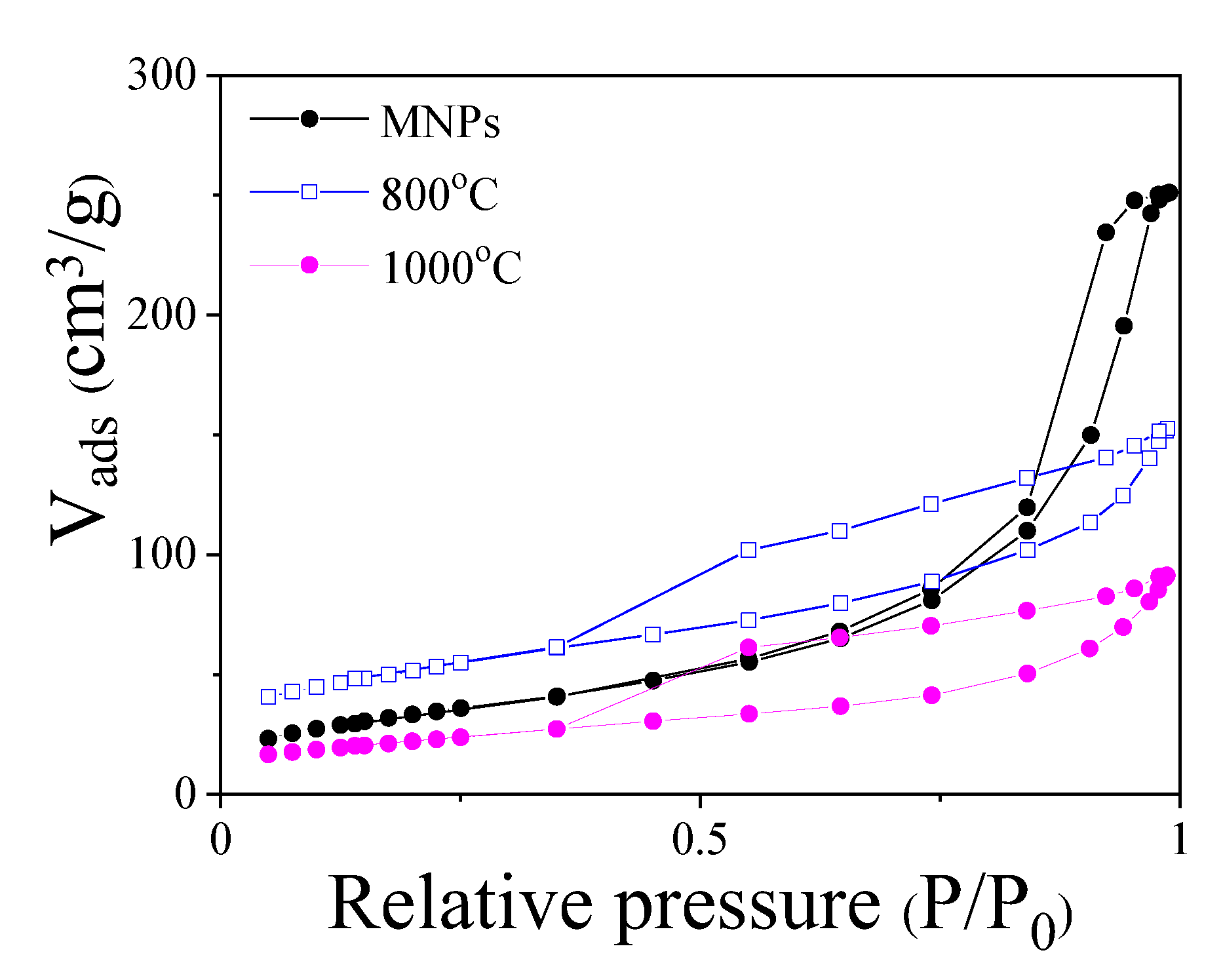
2.1. Structural and Magnetic Characterization
2.2. Cr (VI) Adsorption Tests
- Cr (VI) Adsorption Kinetics
- b.
- Cr (VI) Adsorption Isotherms
3. Materials and Methods
3.1. Synthesis of Fe–C Nanostructures
3.2. Structural and Magnetic Characterization
3.3. Cr (VI) Adsorption Tests
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Geissen, V.; Mol, H.; Klumpp, E.; Umlauf, G.; Nadal, M.; van der Ploeg, M.; van de Zee, S.E.A.T.M.; Ritsema, C.J. Emerging pollutants in the environment: A challenge for water resource management. Int. Soil Water Conserv. Res. 2015, 3, 57–65. [Google Scholar] [CrossRef]
- Zhitkovich, A. Chromium in Drinking Water: Sources, Metabolism, and Cancer Risks. Chem. Res. Toxicol. 2011, 24, 1617–1629. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wei, L.; Zhang, P.; Xiao, J.; Guo, Z.; Fu, Q. Bioaccumulation of dietary CrPic, Cr(III) and Cr(VI) in juvenile coral trout (Plectropomus leopardus). Ecotoxicol. Environ. Saf. 2022, 240, 113692. [Google Scholar] [CrossRef]
- Costa, M. Toxicity and Carcinogenicity of Cr(VI) in Animal Models and Humans. Crit. Rev. Toxicol. 1997, 27, 431–442. [Google Scholar] [CrossRef]
- World Health Organization. Chromium in Drinking-Water. In Background Document for Development of WHO Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Owlad, M.; Aroua, M.K.; Daud, W.A.W.; Baroutian, S. Removal of Hexavalent Chromium-Contaminated Water and Wastewater: A Review. Water Air Soil Pollut. 2009, 200, 59–77. [Google Scholar] [CrossRef]
- Xing, X.; Ren, X.; Alharbi, N.S.; Chen, C. Efficient adsorption and reduction of Cr(VI) from aqueous solution by Santa Barbara Amorphous-15 (SBA-15) supported Fe/Ni bimetallic nanoparticles. J. Colloid Interface Sci. 2022, 629, 744–754. [Google Scholar] [CrossRef]
- Abbo, H.S.; Gupta, K.C.; Khaligh, N.G.; Titinchi, S.J.J. Carbon Nanomaterials for Wastewater Treatment. ChemBioEng Rev. 2021, 8, 463–489. [Google Scholar] [CrossRef]
- Sinha, R.; Kumar, R.; Sharma, P.; Kant, N.; Shang, J.; Aminabhavi, T.M. Removal of hexavalent chromium via biochar-based adsorbents: State-of-the-art, challenges, and future perspectives. J. Environ. Manag. 2022, 317, 115356. [Google Scholar] [CrossRef]
- Babel, S. Low-cost adsorbents for heavy metals uptake from contaminated water: A review. J. Hazard. Mater. 2003, 97, 219–243. [Google Scholar] [CrossRef]
- Mohammed, L.; Gomaa, H.G.; Ragab, D.; Zhu, J. Magnetic nanoparticles for environmental and biomedical applications: A review. Particuology 2017, 30, 1–14. [Google Scholar] [CrossRef]
- Sęk, J.P.; Kasprzak, A.; Bystrzejewski, M.; Poplawska, M.; Kaszuwara, W.; Stojek, Z.; Nowicka, A.M. Nanoconjugates of ferrocene and carbon-encapsulated iron nanoparticles as sensing platforms for voltammetric determination of ceruloplasmin in blood. Biosens. Bioelectron. 2018, 102, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, R.; Pang, X.; Zhao, X.; Zhang, Y.; Qin, G.; Zhang, X. Fe@C nanocapsules with substitutional sulfur heteroatoms in graphitic shells for improving microwave absorption at gigahertz frequencies. Carbon 2018, 126, 372–381. [Google Scholar] [CrossRef]
- El-Gendy, A.; Ibrahim, E.; Khavrus, V.; Krupskaya, Y.; Hampel, S.; Leonhardt, A.; Büchner, B.; Klingeler, R. The synthesis of carbon coated Fe, Co and Ni nanoparticles and an examination of their magnetic properties. Carbon 2009, 47, 2821–2828. [Google Scholar] [CrossRef]
- Ning, L.; Xiaojie, L.; Xiaohong, W.; Honghao, Y.; Fei, M.; Wei, S. Preparation and magnetic behavior of carbon-encapsulated iron nanoparticles by detonation method. Compos. Sci. Technol. 2009, 69, 2554–2558. [Google Scholar] [CrossRef]
- Fan, G.; Jiang, Y.; Xin, J.; Zhang, Z.; Fu, X.; Xie, P.; Cheng, C.; Liu, Y.; Qu, Y.; Sun, K.; et al. Facile Synthesis of Fe@Fe3C/C Nanocomposites Derived from Bulrush for Excellent Electromagnetic Wave-Absorbing Properties. ACS Sustain. Chem. Eng. 2019, 7, 18765–18774. [Google Scholar] [CrossRef]
- Cervera, L.; Peréz-Landazábal, J.; Garaio, E.; Monteserín, M.; Larumbe, S.; Martín, F.; Gómez-Polo, C. Fe-C nanoparticles obtained from thermal decomposition employing sugars as reducing agents. J. Alloys Compd. 2021, 863, 158065. [Google Scholar] [CrossRef]
- Bai, L.; Su, X.; Feng, J.; Ma, S. Preparation of sugarcane bagasse biochar/nano-iron oxide composite and mechanism of its Cr (VI) adsorption in water. J. Clean. Prod. 2021, 320, 128723. [Google Scholar] [CrossRef]
- Qi, X.; Xu, J.; Zhong, W.; Du, Y. A facile route to synthesize core/shell structured carbon/magnetic nanoparticles hybrid and their magnetic properties. Mater. Res. Bull. 2015, 67, 162–169. [Google Scholar] [CrossRef]
- Yue, Z.; Bender, S.E.; Wang, J.; Economy, J. Removal of chromium Cr(VI) by low-cost chemically activated carbon materials from water. J. Hazard. Mater. 2009, 166, 74–78. [Google Scholar] [CrossRef]
- Jadhav, N.V.; Prasad, A.I.; Kumar, A.; Mishra, R.; Dhara, S.; Babu, K.; Prajapat, C.; Misra, N.; Ningthoujam, R.; Pandey, B.; et al. Synthesis of oleic acid functionalized Fe3O4 magnetic nanoparticles and studying their interaction with tumor cells for potential hyperthermia applications. Colloids Surf. B Biointerfaces 2013, 108, 158–168. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, L.; Liu, R.; Li, D.; Ge, X.; Ge, G. The Role of the OH Group in Citric Acid in the Coordination with Fe3O4 Nanoparticles. Langmuir 2019, 35, 8325–8332. [Google Scholar] [CrossRef] [PubMed]
- Bera, M.; Chandravati; Gupta, P.; Maji, P.K. Facile One-Pot Synthesis of Graphene Oxide by Sonication Assisted Mechanochemical Approach and Its Surface Chemistry. J. Nanosci. Nanotechnol. 2018, 18, 902–912. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Kumar, A.; Verma, N.; Anupama, A.; Philip, R.; Sahoo, B. Modulating non-linear optical absorption through controlled graphitization of carbon nanostructures containing Fe3C-graphite core-shell nanoparticles. Carbon 2019, 153, 545–556. [Google Scholar] [CrossRef]
- Howe, J.Y.; Rawn, C.J.; Jones, L.E.; Ow, H. Improved crystallographic data for graphite. Powder Diffr. 2003, 18, 150–154. [Google Scholar] [CrossRef]
- The Iron Carbon Phase Diagram. Available online: https://www.tf.uni-kiel.de/matwis/amat/iss/kap_6/illustr/s6_1_2.html (accessed on 6 September 2021).
- Kumar, R.; Sahoo, B. One-step pyrolytic synthesis and growth mechanism of core–shell type Fe/Fe3C-graphite nanoparticles-embedded carbon globules. Nano-Struct. Nano-Objects 2018, 16, 77–85. [Google Scholar] [CrossRef]
- Puech, P.; Plewa, J.-M.; Mallet-Ladeira, P.; Monthioux, M. Spatial confinement model applied to phonons in disordered graphene-based carbons. Carbon 2016, 105, 275–281. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Robertson, J. Interpretation of Raman spectra of disordered and amorphous carbon. Phys. Rev. B 2000, 61, 14095–14107. [Google Scholar] [CrossRef]
- De Gisi, S.; Lofrano, G.; Grassi, M.; Notarnicola, M. Characteristics and adsorption capacities of low-cost sorbents for wastewater treatment: A review. Sustain. Mater. Technol. 2016, 9, 10–40. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Sitko, R.; Turek, E.; Zawisza, B.; Malicka, E.; Talik, E.; Heimann, J.; Gagor, A.; Feist, B.; Wrzalik, R. Adsorption of divalent metal ions from aqueous solutions using graphene oxide. Dalton Trans. 2013, 42, 5682–5689. [Google Scholar] [CrossRef]
- Kazak, O. Fabrication of in situ magnetic activated carbon by co-pyrolysis of sucrose with waste red mud for removal of Cr(VI) from waters. Environ. Technol. Innov. 2021, 24, 101856. [Google Scholar] [CrossRef]
- Ma, B.; Zhu, J.; Sun, B.; Chen, C.; Sun, D. Influence of pyrolysis temperature on characteristics and Cr(VI) adsorption performance of carbonaceous nanofibers derived from bacterial cellulose. Chemosphere 2022, 291, 132976. [Google Scholar] [CrossRef]
- Sahoo, T.R.; Prelot, B. Chapter 7—Adsorption processes for the removal of contaminants from wastewater: The perspective role of nanomaterials and nanotechnology. In Micro and Nano Technologies, Nanomaterials for the Detection and Removal of Wastewater Pollutant; Bonelli, B., Freyria, F.S., Rossetti, I., Sethi, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 161–222. [Google Scholar] [CrossRef]
- Li, B.; Yin, W.; Xu, M.; Tan, X.; Li, P.; Gu, J.; Chiang, P.; Wu, J. Facile modification of activated carbon with highly dispersed nano-sized α-Fe2O3 for enhanced removal of hexavalent chromium from aqueous solutions. Chemosphere 2019, 224, 220–227. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, J.; Zhang, C.; Wang, Y.; Li, Y. Adsorptive removal of Cr(VI) by Fe-modified activated carbon prepared from Trapa natans husk. Chem. Eng. J. 2010, 162, 677–684. [Google Scholar] [CrossRef]
- Wen, R.; Tu, B.; Guo, X.; Hao, X.; Wu, X.; Tao, H. An ion release controlled Cr(VI) treatment agent: Nano zero-valent iron/carbon/alginate composite gel. Int. J. Biol. Macromol. 2020, 146, 692–704. [Google Scholar] [CrossRef]
- Karamipour, A.; Parsi, P.K.; Zahedi, P.; Moosavian, S.M.A. Using Fe3O4-coated nanofibers based on cellulose acetate/chitosan for adsorption of Cr(VI), Ni(II) and phenol from aqueous solutions. Int. J. Biol. Macromol. 2020, 154, 1132–1139. [Google Scholar] [CrossRef]
- Nethaji, S.; Sivasamy, A.; Mandal, A. Preparation and characterization of corn cob activated carbon coated with nano-sized magnetite particles for the removal of Cr(VI). Bioresour. Technol. 2013, 134, 94–100. [Google Scholar] [CrossRef]
- Xie, Y.; Lin, J.; Liang, J.; Li, M.; Fu, Y.; Wang, H.; Tu, S.; Li, J. Hypercrosslinked mesoporous poly (ionic liquid) s with high density of ion pairs: Efficient adsorbents for Cr(VI) removal via ion-exchange. Chem. Eng. J. 2019, 378, 122107. [Google Scholar] [CrossRef]
- Dong, L.; Deng, R.; Xiao, H.; Chen, F.; Zhou, Y.; Li, J.; Chen, S.; Yan, B. Hierarchical polydopamine coated cellulose nanocrystal microstructures as efficient nanoadsorbents for removal of Cr(VI) ions. Cellulose 2019, 26, 6401–6414. [Google Scholar] [CrossRef]
- Xiang, L.; Lin, J.; Yang, Q.; Lin, S.; Chen, S.; Yan, B. Facile preparation of hierarchical porous polydopamine microspheres for rapid removal of chromate from the wastewater. J. Leather Sci. Eng. 2020, 2, 22. [Google Scholar] [CrossRef]
- Li, L.; Chi, L.; Zhang, H.; Wu, S.; Wang, H.; Luo, Z.; Li, Y.; Li, Y. Fabrication of Ti-PDA nanoparticles with enhanced absorption and photocatalytic activities for hexavalent chromium Cr(VI) removal. Appl. Surf. Sci. 2022, 580, 152168. [Google Scholar] [CrossRef]
- Tristão, J.C.; De Mendonça, F.G.; Lago, R.M.; Ardisson, J.D. Controlled formation of reactive Fe particles dispersed in a carbon matrix active for the oxidation of aqueous contaminants with H2O2. Environ. Sci. Pollut. Res. 2015, 22, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Carvajal, J. Recent advances in magnetic structure determination by neutron powder diffraction. Phys. B Condens. Matter 1993, 192, 55–69. [Google Scholar] [CrossRef]

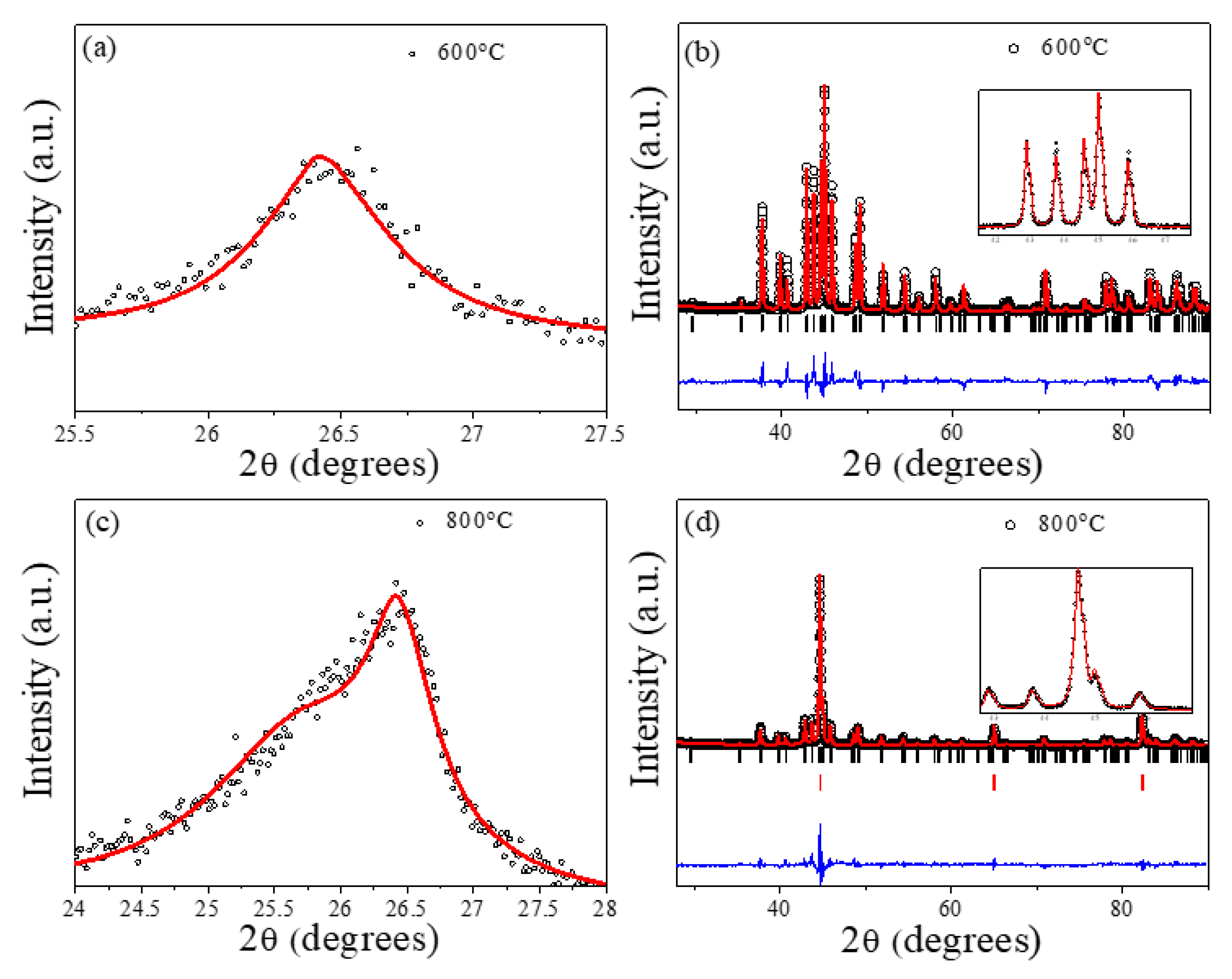


| Tann | Graphitic Phase | Cell Parameter (Å) | Fe-Based Phases | Cell Parameter (Å) | Crystallite Size (Å) |
|---|---|---|---|---|---|
| 400 °C | Graphite1 (100%) | a = b = 2.454 (1) c = 6.696 (6) | Fe3O4 (100%) | a = b = c = 8.3619 (6) | 79.14 (4) |
| 600 °C | Graphite1 (100%) | a = b = 2.439 (8) c = 6.742 (2) | Fe3C (100%) | a = 5.0920 b = 6.7441 (1) c = 4.5269 | 777.45 (8) |
| 800 °C | Graphite1 (31.2%) | a = b = 2.446 (2) c = 6.741 (1) | Fe3C (61.1%) | a = 5.0926 (1) b = 6.745 (2) c = 4.5281 (1) | 603.32 (5) |
| Graphite2 (68.8%) | a = b = 2.557 (2) c = 6.932 (5) | α-Fe (38.9%) | a = b = c = 2.8682 (1) | 689.76 (0) | |
| 1000 °C | Graphite1 (70.7) | a = b = 2.5681 (6) c = 6.7478 (6) | Fe3C (57.8%) | a = 5.0934 (1) b = 6.7456 (2) c = 4.5290 (1) | 542.91 (2) |
| Graphite2 (29.3) | a = b = 2.3812 (9) c = 6.869 (3) | α-Fe (42.2%) | a = b = c = 2.8682 (1) | 931.48 (5) |
| Tann | ωD (cm−1) | ωG (cm−1) | ω2D (cm−1) | FWHMD (cm−1) | FWHMG (cm−1) | ID/IG | I2D/ID |
|---|---|---|---|---|---|---|---|
| 400 °C | 1362 ± 1 | 1585 ± 1 | 3018 ± 10 | 253 ± 5 | 92 ± 2 | 0.69 | 0.10 |
| 600 °C | 1351.3 ± 0.3 | 1589.6 ± 0.1 | 2859 ± 3 | 262 ± 1 | 86.3 ± 0.4 | 0.88 | 0.22 |
| 800 °C | 1344.6 ± 0.3 | 1587.6 ± 0.1 | 2692 ± 1 | 139 ± 1 | 63.9 ± 0.4 | 0.88 | 0.29 |
| 1000 °C | 1350.4 ± 0.1 | 1590.1 ± 0.1 | 2693.7 ± 0.3 | 75.8 ± 0.3 | 53.7 ± 0.2 | 0.83 | 0.39 |
| Sample | Pseudo-First-Order Model | Pseudo-Second-Order Model | ||||
|---|---|---|---|---|---|---|
| qe (mg/g) | k1 (min−1) | R2 | qe (mg/g) | k2 (g/mg min) | R2 | |
| MNPs + fructose (Tann = 800 °C) | 0.519 | 0.61 ± 0.01 | 0.994 | 0.611 | 0.72 ± 0.06 | 0.887 |
| MNPs | 0.746 | 0.047 ± 0.005 | 0.909 | 0.937 | 0.042 ± 0.004 | 0.928 |
| Sample | Langmuir Model | Freundlich Model | |||||
|---|---|---|---|---|---|---|---|
| qmax (mg/g) | KL (L/mg) | RL | R2 | KF (L/g) | n | R2 | |
| MNPs + fructose (Tann = 800 °C) | 41.5 | 0.182 | 0.499 | 0.907 | 11.1 | 3.18 | 0.955 |
| MNPs | 11.2 | 0.709 | 0.940 | 0.869 | 4.85 | 5.99 | 0.724 |
| Sample | Isotherm Values | Kinetic Constants | References |
|---|---|---|---|
| MNPs + fructose (Tann = 800 °C) | qmax = 41.5 mg/g | k1 = 0.61 min−1 | This work |
| KF = 11.1 L/g | k2 = 0.72 g/mg·min | ||
| Poly(ionic liquid) | qmax = 236.8 mg/g | k2 = 0.0211 g/mg·min | [41] |
| Hierarchical polydopamine coated cellulose nanocrystal microstructures | qmax = 205 mg/g | k1 = 0.0028 min−1 | [42] |
| KF = 43.27 L/g | k2 = 3.96 × 10−4 g/mg·min | ||
| Hierarchical porous polydopamine microspheres | qmax = 307.7 mg/g | k1 = 2.508 min−1 | [43] |
| KF = 94.99 L/g | k2 = 3.39 g/mg·min | ||
| Ti-PDA nanoparticles | qmax = 625 mg/g | k1 = 0.01 min−1 | [44] |
| KF = 0.28111 L/g | k2 = 2.45 × 10−3 g/mg·min |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cervera-Gabalda, L.; Gómez-Polo, C. Magnetic Fe/Fe3C@C Nanoadsorbents for Efficient Cr (VI) Removal. Int. J. Mol. Sci. 2022, 23, 15135. https://doi.org/10.3390/ijms232315135
Cervera-Gabalda L, Gómez-Polo C. Magnetic Fe/Fe3C@C Nanoadsorbents for Efficient Cr (VI) Removal. International Journal of Molecular Sciences. 2022; 23(23):15135. https://doi.org/10.3390/ijms232315135
Chicago/Turabian StyleCervera-Gabalda, Laura, and Cristina Gómez-Polo. 2022. "Magnetic Fe/Fe3C@C Nanoadsorbents for Efficient Cr (VI) Removal" International Journal of Molecular Sciences 23, no. 23: 15135. https://doi.org/10.3390/ijms232315135
APA StyleCervera-Gabalda, L., & Gómez-Polo, C. (2022). Magnetic Fe/Fe3C@C Nanoadsorbents for Efficient Cr (VI) Removal. International Journal of Molecular Sciences, 23(23), 15135. https://doi.org/10.3390/ijms232315135






