Review on Magnetism in Catalysis: From Theory to PEMFC Applications of 3d Metal Pt-Based Alloys
Abstract
1. Introduction
2. Magnetism in Transition Metals: The 3d-Electrons Case
2.1. Types of Magnetic Behavior
2.1.1. Diamagnetism and Paramagnetism
2.1.2. Collective Magnetism
2.1.3. Strongly Correlated Electron Systems (SCES)
2.2. (Indirect) Exchange Interactions
2.2.1. Basic Quantum Concepts
- The operator for the kinetic energy of the electrons ();
- The operator for the kinetic energy of the nuclei ();
- The electron–nucleus Coulomb attraction term ();
- The electron–electron Coulomb repulsion (); and
- The nucleus–nucleus Coulomb repulsion ().
2.2.2. Exchange Effects
2.2.3. Coulomb and Exchange Integrals
- is a one-electron term (integral) that represents the average nuclear attraction and kinetic energy of an electron a described by (this integral is indicated as in Equation (15)).
- is a two-electron term (integral) that represents the classical Coulomb repulsion between and charge clouds, called Coulomb integral ().
- is another two-electron integral with a quantum mechanics interpretation and it is called exchange integral ().
- and are the averages of the nuclear attraction and kinetic energy for an electron a described by and;
- and are the Coulomb integrals between electrons with the same spin,
- is the Coulomb integral of an electron in with one in ;
- and are the exchange integrals among electrons with parallel spin (there is no exchange interaction between electrons with antiparallel spin);
- The summations, with upper limit and , are over occupied orbitals or and or , respectively; and
- The factor in the third and fourth terms removes the double counting in the free sum.
2.2.4. Exchange Mechanisms
2.3. Electronic Structure of Solids
2.3.1. Bloch Theorem
- The Bragg reflection is a feature of the wave propagation in periodic systems; hence, it must be also a characteristic of electron waves in crystalline structures [27,32,69]. The most relevant consequence of the Bragg reflection is that it leads to the creation of gaps in the distribution of energy states [27]. The energy spectrum of a crystal is transformed into a band structure featuring energy levels where the propagation of electrons becomes possible [32]. The Bragg reflection conditions can also be used to build the boundaries of the Brillouin zone [27].
- The Fermi energy (EF) is a concept of quantum mechanics used in solids. The Fermi energy defines the energy level for which all states having energy € smaller than EF are occupied at T = 0 K [15,26,32]. In other words, EF represents the highest occupied energy level [13,44]. There are no thermal energies at 0 K, so the occupation of one-electron states is determined only by placing one electron per state (in agreement with the Pauli exclusion principle). The position of EF in relation to the band energy level is important in determining the electronic and thermal properties of a solid, since it energetically separates the occupied from the non-occupied states [32,44]. Another useful concept related to Fermi energy is the Fermi surface. The Fermi surface is a surface, defined in the reciprocal space, that separates the occupied states from the empty ones at 0 K [32,71]. Its shape can provide information on the electrical properties of a solid [71]. The electronic bands placed below and above EF are called valence and conduction bands, respectively [32].
2.3.2. Electrons in Transition Metals
2.3.3. Heisenberg Model
2.3.4. Hubbard Model
- and are two nearest neighbor sites;
- is a vertex set that is normally assumed to form a translationally invariant lattice, whose characteristics are important to define the properties of the model [82];
- is the spin electron;
- is the transfer or hopping matrix element. It indicates that the dispersion energy band is now expressed in terms of hopping [32];
- indicates Bloch functions described by the spin index;
2.3.5. Additional Remarks
- is the kinetic energy of the electrons;
- is the Coulomb attraction between nucleus and electrons;
- is the energy factor of the electron–electron Coulomb repulsions;
- is the energy factor due to the spin–orbit coupling.
2.4. Magnetic Properties of 3d-Transition Metals and Their Alloys
- Copper (Cu) is a face-centered cubic (fcc) solid. It is the only 3d metal that exhibits a diamagnetic behavior (= −1.1 at 298 K) [15], since it has a completely filled 3d-band [72] (the sequential filling of the d-shell induces the related bands to become narrower and to energetically shift below the Fermi energy) [32].
- and are the atomic fractions of M and Pt in the same sample;
- and are the fractions of M and Pt sites; and
- and are fractions of Fe or Pt sites occupied by the correct atomic species.
| FePt | CoPt | NiPt | ||||
|---|---|---|---|---|---|---|
| Strukturbericht Designation | A1 | L10 | A1 | L10 | A1 | L10 |
| Chemical Ordering | D | O | D | O | D | O |
| Order–Disorder Critical Temperature (K) | ~1573 [99,108] | ~1106 [95]; ~1098 [99,144] | ~940 [128,144] | |||
| Heat of Formation (ΔHf) (eV/atom) | - | −0.73 a [145] | - | −0.140 [146] | - | −0.096 [128] |
| Magnetic Ordering | FM [108] | FM [108] | FM [147] | FM [147,148] | FM [94] | P [14,94] |
| Curie Temperature (TC) (K) | 585 [108] | 750 [108]; 670 [14] | - | 750 [14]; 710 [149]; 850 [133] | - | - |
| Magnetically | Soft | Hard | Soft | Hard | - | - |
| Maximum Energy Product (BH)max (MGOe) | - | ~13 [150] | - | ~9.7 [95] | - | - |
| Uniaxial MCA Energy Constant (Ku) (107 erg/cm3) | [110] | 7 [119]; 6.6–10 [133] | <4.9 [126] | 4.9 [133] | - | - |
| Saturation Magnetization (MS) (emu/cm3) | - | 1140 [133]; 1150 [151] | - | 800 [133] | - | - |
| Minimal Stable Grain Size (Dp) (nm) b,c | - | 3.3–2.8 [133] | - | 3.6 [133] | - | - |
3. Catalysis and Magnetism
3.1. Oxygen Reduction Reaction (ORR)
- The Griffith model, in which both oxygen atoms interact with a single atom of the catalytic surface (the less common type of adsorption);
- The Pauling model (or end-on configuration), in which only one of the two oxygen atoms is coordinated with one atom of the catalytic surface; and
- The bridge model, in which two bonds are formed involving both O atoms with two different atoms of the surface.
3.2. Applications of 3d Metals and 3d-Based Alloys
3.3. Bimetallic Pt-Based Alloys as ORR Catalysts
3.4. Catalytic Trends and Magnetism
- By engineering magnetic catalysts through the increment in their “internal” magnetic properties () (intrinsic fields);
- By the application of an external magnetic field () (extrinsic fields); and
- By combining the previous two options ().
3.5. Improvement of Magnetic Properties of Catalysts (, Intrinsic Magnetism)
3.6. Application of External Magnetic Field (, Extrinsic Magnetism)
- Changes in the mass transport;
- Modification of the heterogeneous electron transfer kinetics and electrochemical equilibria; and
- Influence on electrodeposit morphology.
3.7. Combination of Intrinsic and Extrinsic Magnetism in Catalysis ()
4. Basics of Fuel Cells
4.1. Renewable Energy Demand and Energy Storage Systems
4.2. Electrochemical Energy Storage Systems: Batteries and Fuel Cells
4.3. Fuel Cells
4.4. Thermodynamics of Fuel Cell
- Activation losses, due to kinetics of the electrochemical reaction at the electrodes. A part of the voltage is used to drive the electron transfer from one electrode to the other during the electrochemical reaction (major voltage loss).
- Fuel crossover and internal currents, caused by incomplete fuel utilization. The majority of the fuel reacts, but a small amount diffuses through the electrolyte unused (this loss increases in fuel cells operating at low temperatures).
- Ohmic losses, due to the electrical resistance of the material of the electrodes, the electrolyte solution and other components of the fuel cell.
- Mass transport or concentration losses, connected with the consumption of reactants at the electrode surface, that cause a change in their concentrations (or, more precisely, activities), thus to the voltage.
4.5. Kinetics of Fuel Cell
- is the current density, the current per unit area (A/cm2) (a more important parameter than the simple current, since the reaction takes place at the electrode/electrolyte interface);
- is known as exchange current density;
- is the activation overpotential;
- is the dimensionless charge transfer coefficient that corresponds to the quantity of the electrical energy used to modify the reaction rate at the anode and cathode (its value depends on the type of electrochemical reaction and electrode material, but it ranges from 0 to 1.0) [10];
- is the number of electrons involved in the electrochemical process;
- R is the universal gas constant;
- T is the absolute temperature; and
- F is the faraday constant.
4.6. Types of Fuel Cell
- Alkaline Fuel Cells (AFCs): They use an alkaline liquid (K2CO3 or KOH) as electrolyte. The first models operated at high temperatures (50–200 °C) [6], but AFCs can operate at lower temperature (20–80 °C) nowadays [3]. No high-profile research on these FCs is currently ongoing, due to their higher capital cost compared to the other fuel cell categories, but they were exploited in the 1960s for space programs with a great deal of success [6].
- Phosphoric Acid Fuel Cells (PAFCs): They work at high temperatures (~220 °C) and use an inorganic acid (100% concentrate phosphoric acid) as proton-conducting electrolyte [6]. These were the first examples of commercially available fuel cells, thanks to their reliability as a power source, durability and low maintenance [6]. PAFCs are exploited in power stations nowadays [3].
- Molten Carbonate Fuel Cells (MCFCs): They also operate at high temperatures (600–700 °C), use a molten mixture of alkaline metal carbonate (lithium and potassium or lithium and sodium carbonate) as an electrolyte ((CO3)2− is the mobile ion) [6] and, unlike AFCs and PACFs, exploit abundant metals as catalysts (nickel and nickel oxides). MCFCs display severe corrosion and stability issues at present, making them unappealing for the market [6].
- Solid Oxide Fuel Cells (SOFCs): They are solid-state devices composed of a solid and a gas phase. The anode contains ceramic zirconia cermet with nickel metal and the cathode contains a mixture of electronically conducting ions and ceramics (for example, strontium-doped lanthanum manganite) [3,10]. SOFCs are currently working at high temperatures (600–1000 °C) [3] and are still under development [10].
4.7. Proton Exchange Membrane Fuel Cells (PEMFCs)
4.7.1. PEMFCs Components
4.7.2. Applications of Magnetic Field in PEMFCs
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IEA. Energy Technology Perspectives 2020. Available online: https://www.iea.org/reports/energy-technology-perspectives-2020 (accessed on 29 August 2022).
- Suter, T.A.M.; Smith, K.; Hack, J.; Rasha, L.; Rana, Z.; Angel, G.M.A.; Shearing, P.R.; Miller, T.S.; Brett, D.J.L. Engineering Catalyst Layers for Next-Generation Polymer Electrolyte Fuel Cells: A Review of Design, Materials, and Methods. Adv. Energy Mater. 2021, 11, 2101025. [Google Scholar] [CrossRef]
- Fan, L.; Tu, Z.; Chan, S.H. Recent development of hydrogen and fuel cell technologies: A review. Energy Rep. 2021, 7, 8421–8446. [Google Scholar] [CrossRef]
- Wang, Y.; Ruiz Diaz, D.F.; Chen, K.S.; Wang, Z.; Adroher, X.C. Materials, technological status, and fundamentals of PEM fuel cells—A review. Mater. Today 2020, 32, 178–203. [Google Scholar] [CrossRef]
- Cullen, D.A.; Neyerlin, K.C.; Ahluwalia, R.K.; Mukundan, R.; More, K.L.; Borup, R.L.; Weber, A.Z.; Myers, D.J.; Kusoglu, A. New roads and challenges for fuel cells in heavy-duty transportation. Nat. Energy 2021, 6, 462–474. [Google Scholar] [CrossRef]
- Debe, M.K. Electrocatalyst approaches and challenges for automotive fuel cells. Nature 2012, 486, 43–51. [Google Scholar] [CrossRef]
- Wang, X.X.; Swihart, M.T.; Wu, G. Achievements, challenges and perspectives on cathode catalysts in proton exchange membrane fuel cells for transportation. Nat. Catal. 2019, 2, 578–589. [Google Scholar] [CrossRef]
- Tellez-Cruz, M.M.; Escorihuela, J.; Solorza-Feria, O.; Compañ, V. Proton Exchange Membrane Fuel Cells (PEMFCs): Advances and Challenges. Polymers 2021, 13, 3064. [Google Scholar] [CrossRef] [PubMed]
- Alaswad, A.; Omran, A.; Sodre, J.R.; Wilberforce, T.; Pignatelli, G.; Dassisti, M.; Baroutaji, A.; Olabi, A.G. Technical and Commercial Challenges of Proton-Exchange Membrane (PEM) Fuel Cells. Energies 2021, 14, 144. [Google Scholar] [CrossRef]
- Larminie, J.; Dicks, A.; McDonald, M.S. Fuel Cell Systems Explained; Wiley: Hoboken, NJ, USA, 2003. [Google Scholar]
- Cui, J.; Chen, Q.; Li, X.; Zhang, S. Recent advances in non-precious metal electrocatalysts for oxygen reduction in acidic media and PEMFCs: An activity, stability and mechanism study. Green Chem. 2021, 23, 6898–6925. [Google Scholar] [CrossRef]
- Mølmen, L.; Eiler, K.; Fast, L.; Leisner, P.; Pellicer, E. Recent advances in catalyst materials for proton exchange membrane fuel cells. APL Mater. 2021, 9, 040702. [Google Scholar] [CrossRef]
- Getzlaff, M. Fundamentals of Magnetism; Springer: Berlin/Heidelberg, Germany, 2008. [Google Scholar]
- Wijn, H.P.J. Magnetic Properties of Metals: D-Element, Alloys and Compounds; Springer: Berlin/Heidelberg, Germany, 1991. [Google Scholar]
- Blundell, S. Magnetism in Condensed Matter, 1st ed.; Oxford University Press: Oxford, UK, 2001. [Google Scholar]
- Heisenberg, W. Mehrkörperproblem und Resonanz in der Quantenmechanik. Z. Für Phys. 1926, 38, 411–426. [Google Scholar] [CrossRef]
- Dirac, P.A.M.; Fowler, R.H. On the theory of quantum mechanics. Proc. R. Soc. London. Ser. A Contain. Pap. A Math. Phys. Character 1926, 112, 661–677. [Google Scholar] [CrossRef]
- Szabo, A.; Ostlund, N.S. Modern Quantum Chemistry: Introduction to Advanced Electronic Structure Theory; Dover Publication: Mineola, NY, USA, 1996. [Google Scholar]
- Biz, C.; Fianchini, M.; Gracia, J. Strongly Correlated Electrons in Catalysis: Focus on Quantum Exchange. ACS Catal. 2021, 11, 14249–14261. [Google Scholar] [CrossRef]
- Wang, X.; Li, Z.; Qu, Y.; Yuan, T.; Wang, W.; Wu, Y.; Li, Y. Review of Metal Catalysts for Oxygen Reduction Reaction: From Nanoscale Engineering to Atomic Design. Chem 2019, 5, 1486–1511. [Google Scholar] [CrossRef]
- Li, D.; Liu, H.; Feng, L. A Review on Advanced FeNi-Based Catalysts for Water Splitting Reaction. Energy Fuels 2020, 34, 13491–13522. [Google Scholar] [CrossRef]
- Chernavskii, P.A.; Dalmon, J.A.; Perov, N.S.; Khodakov, A.Y. Magnetic Characterization of Fischer-Tropsch Catalysts. Oil Gas Sci. Technol. Rev. IFP 2009, 64, 25–48. [Google Scholar] [CrossRef]
- Zhu, J.; Hu, L.; Zhao, P.; Lee, L.Y.S.; Wong, K.-Y. Recent Advances in Electrocatalytic Hydrogen Evolution Using Nanoparticles. Chem. Rev. 2020, 120, 851–918. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.toyota-europe.com/download/cms/euen/Toyota%20Mirai%20FCV_Posters_LR_tcm-11-564265.pdf (accessed on 7 July 2022).
- Chiara, B. Electronic and Magnetic Factors in the Design of Optimum Catalysts for Hydrogen Fuel Cells; University of Jaume I: Castellón de la Plana, Spain, 2022; p. 292. [Google Scholar]
- Griffiths, D.J.; Schroeter, D.F. Introduction to Quantum Mechanics, 3rd ed.; Cambridge University Press: Cambridge, UK, 2018. [Google Scholar]
- Goodenough, J.B. Magnetism and the Chemical Bond; Wiley & Sons: Hoboken, NJ, USA, 1963. [Google Scholar]
- Bluhm, H.B.T.; Morgenstern, M.; von Plessen, G.; Stampfer, C. Electrons in Solids; Walter de Gruyter GmbH: Berlin/Boston, Germany, 2019. [Google Scholar]
- Thonhauser, T. Theory of Orbital Magnetization in Solids. Int. J. Mod. Phys. B 2011, 25, 1429–1458. [Google Scholar] [CrossRef]
- Aryasetiawan, F.; Karlsson, K. Modern theory of orbital magnetic moment in solids. J. Phys. Chem. Solids 2019, 128, 87–108. [Google Scholar] [CrossRef]
- McHenry, M.E.; Laughlin, D.E. Magnetic Moment and Magnetization. In Characterization of Materials; Kaufmann, E.N., Ed.; Carnegie Mellon University: Pittsburgh, PA, USA, 2012. [Google Scholar]
- Rössler, U. Solid State Theory: An Introduction; Physica-Verlag: Heidelberg, Germany, 2009. [Google Scholar]
- Machin, D.J.; Mabbs, F.E. Magnetism and Transition Metal Complexes; Dover Publications, Inc.: Mineola, NY, USA, 2008. [Google Scholar]
- Kotnala, R.K.; Shah, J. Chapter 4-Ferrite materials: Nano to spintronics regime. In Handbook of Magnetic Materials; Elsevier: Amsterdam, The Netherlands, 2015; pp. 291–379. [Google Scholar]
- Antonov, V.N.; Bekenov, L.V.; Yaresko, A.N. Electronic Structure of Strongly Correlated Systems. Adv. Condens. Matter Phys. 2011, 2011, 298928. [Google Scholar] [CrossRef]
- Edelstein, A.S. An overview of strongly correlated electron systems. J. Magn. Magn. Mater. 2003, 256, 430–448. [Google Scholar] [CrossRef]
- Dagotto, E. Complexity in Strongly Correlated Electronic Systems. Science 2005, 309, 257–262. [Google Scholar] [CrossRef]
- Dagotto, E.; Tokura, Y. 31 A brief introduction to strongly correlated electronic materials. In Multifunctional Oxide Heterostructures; Oxford University Press: Oxford, UK, 2012; pp. 3–37. [Google Scholar]
- Ngai, J.H.; Walker, F.J.; Ahn, C.H. Correlated Oxide Physics and Electronics. Annu. Rev. Mater. Res. 2014, 44, 1–17. [Google Scholar] [CrossRef]
- Zhou, Y.; Ramanathan, S. Correlated Electron Materials and Field Effect Transistors for Logic: A Review. Crit. Rev. Solid State Mater. Sci. 2013, 38, 286–317. [Google Scholar] [CrossRef]
- Pavarini, E.; Koch, E.; Anders, F.; Jarrell, M. Correlated electrons: From models to materials. Reihe Model. Simul. 2012, 2. Available online: https://www.cond-mat.de/events/correl12/manuscripts/correl12.pdf (accessed on 7 July 2022).
- Malrieu, J.P.; Caballol, R.; Calzado, C.J.; de Graaf, C.; Guihéry, N. Magnetic Interactions in Molecules and Highly Correlated Materials: Physical Content, Analytical Derivation, and Rigorous Extraction of Magnetic Hamiltonians. Chem. Rev. 2014, 114, 429–492. [Google Scholar] [CrossRef] [PubMed]
- Kent, P.R.C.; Kotliar, G. Toward a predictive theory of correlated materials. Science 2018, 361, 348–354. [Google Scholar] [CrossRef]
- Sakurai, J.J.; Napolitano, J. Modern Quantum Mechanics; Cambridge University Press: Cambridge, UK, 2021. [Google Scholar]
- Goodenough, J.B. Localized vs Collective Descriptions of Magnetic Electrons. J. Appl. Phys. 1968, 39, 403–411. [Google Scholar] [CrossRef]
- Hazewinkel, M. Encyclopaedia of Mathematics; Springer: Berlin/Heidelberg, Germany, 1990. [Google Scholar]
- Slater, J.C. The Electronic Structure of Metals. Rev. Mod. Phys. 1934, 6, 209–280. [Google Scholar] [CrossRef]
- Wigner, E.; Seitz, F. On the Constitution of Metallic Sodium. Phys. Rev. 1933, 43, 804–810. [Google Scholar] [CrossRef]
- Wigner, E.; Seitz, F. On the Constitution of Metallic Sodium. II. Phys. Rev. 1934, 46, 509–524. [Google Scholar] [CrossRef]
- Bader, R.F.W.; Streitwieser, A.; Neuhaus, A.; Laidig, K.E.; Speers, P. Electron Delocalization and the Fermi Hole. J. Am. Chem. Soc. 1996, 118, 4959–4965. [Google Scholar] [CrossRef]
- Giner, E.; Tenti, L.; Angeli, C.; Malrieu, J.-P. The “Fermi hole” and the correlation introduced by the symmetrization or the anti-symmetrization of the wave function. J. Chem. Phys. 2016, 145, 124114. [Google Scholar] [CrossRef] [PubMed]
- Holthausen, M.C.; Koch, W. A Chemist’s Guide to Density Functional Theory; Wiley: Hoboken, NJ, USA, 2002. [Google Scholar]
- Gong, C.; Zhang, X. Two-dimensional magnetic crystals and emergent heterostructure devices. Science 2019, 363, eaav4450. [Google Scholar] [CrossRef]
- Gracia, J. What Are the Electrons Really Doing in Molecules? A Space-Time Picture. Eur. J. Phys. Educ. 2020, 11, 1–19. [Google Scholar]
- Harlander, R. Feynman diagrams. Synthese 2021, 199, 15087–15111. [Google Scholar] [CrossRef]
- Gracia, J. Spin dependent interactions catalyse the oxygen electrochemistry. Phys. Chem. Chem. Phys. 2017, 19, 20451–20456. [Google Scholar] [CrossRef] [PubMed]
- Ruderman, M.A.; Kittel, C. Indirect Exchange Coupling of Nuclear Magnetic Moments by Conduction Electrons. Phys. Rev. 1954, 96, 99–102. [Google Scholar] [CrossRef]
- Kasuya, T. A Theory of Metallic Ferro- and Antiferromagnetism on Zener’s Model. Prog. Theor. Phys. 1956, 16, 45–57. [Google Scholar] [CrossRef]
- Yosida, K. Magnetic Properties of Cu-Mn Alloys. Phys. Rev. 1957, 106, 893–898. [Google Scholar] [CrossRef]
- Ishikawa, Y.; Miura, N. Physics and Engineering Applications of Magnetism; Springer: Berlin/Heidelberg, Germany, 1991. [Google Scholar]
- Bruno, P. Interlayer exchange coupling: A unified physical picture. J. Magn. Magn. Mater. 1993, 121, 248–252. [Google Scholar] [CrossRef]
- Slonczewski, J.C. Mechanism of interlayer exchange in magnetic multilayers. J. Magn. Magn. Mater. 1993, 126, 374–379. [Google Scholar] [CrossRef]
- Dzyaloshinsky, I. A thermodynamic theory of “weak” ferromagnetism of antiferromagnetics. J. Phys. Chem. Solids 1958, 4, 241–255. [Google Scholar] [CrossRef]
- Moriya, T. New Mechanism of Anisotropic Superexchange Interaction. Phys. Rev. Lett. 1960, 4, 228–230. [Google Scholar] [CrossRef]
- Goodenough, J.B. Electronic and ionic transport properties and other physical aspects of perovskites. Rep. Prog. Phys. 2004, 67, 1915–1993. [Google Scholar] [CrossRef]
- Anderson, P.W. Theory of Magnetic Exchange Interactions:Exchange in Insulators and Semiconductors. In Solid State Physics; Seitz, F., Turnbull, D., Eds.; Academic Press: Cambridge, MA, USA, 1963; pp. 99–214. [Google Scholar]
- Mullin, J.W. Crystallization; Butterworth-Heinemann: Oxford, UK, 2001. [Google Scholar]
- Kittel, C. Introduction to Solid State Physics; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005. [Google Scholar]
- Cracknell, A.P.; Bradley, C. The Mathematical Theory of Symmetry in Solids: Representation Theory for Point Groups and Space Groups; Oxford University Press, Clarendon Press: Oxford, UK, 2009. [Google Scholar]
- Dronskowski, R. Computational Chemistry of Solid State Materials; Wiley: Hoboken, NJ, USA, 2007. [Google Scholar]
- Dugdale, S.B. Life on the edge: A beginner’s guide to the Fermi surface. Phys. Scr. 2016, 91, 053009. [Google Scholar] [CrossRef]
- Mott, N.F. Electrons in transition metals. Adv. Phys. 1964, 13, 325–422. [Google Scholar] [CrossRef]
- Khomskii, D.I. (Ed.) Localized and itinerant electrons in solids. In Transition Metal Compounds; Cambridge University Press: Cambridge, UK, 2014; pp. 1–24. [Google Scholar] [CrossRef]
- Buschow, K.H.J.; de Boer, F.R. Itinerant-Electron Magnetism. In Physics of Magnetism and Magnetic Materials; Buschow, K.H.J., de Boer, F.R., Eds.; Springer: Boston, MA, USA, 2003; pp. 63–73. [Google Scholar]
- Mostafanejad, M. Basics of the spin Hamiltonian formalism. Int. J. Quantum Chem. 2014, 114, 1495–1512. [Google Scholar] [CrossRef]
- Hotta, T. Orbital ordering phenomena in d- and f-electron systems. Rep. Prog. Phys. 2006, 69, 2061–2155. [Google Scholar] [CrossRef]
- Landrum, G.A.; Dronskowski, R. The Orbital Origins of Magnetism: From Atoms to Molecules to Ferromagnetic Alloys. Angew. Chem. Int. Ed. 2000, 39, 1560–1585. [Google Scholar] [CrossRef]
- Tasaki, H. From Nagaoka’s Ferromagnetism to Flat-Band Ferromagnetism and Beyond: An Introduction to Ferromagnetism in the Hubbard Model. Prog. Theor. Phys. 1998, 99, 489–548. [Google Scholar] [CrossRef]
- Arai, T. Exchange Interaction and Heisenberg’s Spin Hamiltonian. Phys. Rev. 1962, 126, 471–488. [Google Scholar] [CrossRef]
- Slater, J.C. Ferromagnetism and the Band Theory. Rev. Mod. Phys. 1953, 25, 199–210. [Google Scholar] [CrossRef]
- Galperin, F.M. The Electronic Structure and Magnetic Moment of Fe, Co, and Ni Atoms. Phys. Status Solidi B 1973, 57, 715–718. [Google Scholar] [CrossRef]
- Pavarini, E.; Koch, E.; Coleman, P. Manybody Physics: From Kondo to Hubbard. The Hubbard Model and Its Properties; Forschungszentrum Jülich and the German Research School for Simulation Sciences: Jülich, Germany, 2015. [Google Scholar]
- Li, X.; Yu, H.; Lou, F.; Feng, J.; Whangbo, M.-H.; Xiang, H. Spin Hamiltonians in Magnets: Theories and Computations. Molecules 2021, 26, 803. [Google Scholar] [CrossRef]
- Eisenstein, J. Superconducting Elements. Rev. Mod. Phys. 1954, 26, 277–291. [Google Scholar] [CrossRef]
- Coey, J.M.D. (Ed.) Magnetic materials. In Magnetism and Magnetic Materials; Cambridge University Press: Cambridge, UK, 2010; pp. 374–438. [Google Scholar]
- Albert, H.J.; Rubin, L.R. Magnetic Properties of the Platinum Metals and Their Alloys. In Platinum Group Metals and Compounds; American Chemical Society: Washington, DC, USA, 1971; pp. 1–16. [Google Scholar]
- Coles, B.R. The Magnetic Properties of Platinum Metals and Alloys. A survay of recent research. Platin. Met. Rev. 1964, 8, 9–11. [Google Scholar]
- Hou, D.; Qiu, Z.; Iguchi, R.; Sato, K.; Vehstedt, E.K.; Uchida, K.; Bauer, G.E.W.; Saitoh, E. Observation of temperature-gradient-induced magnetization. Nat. Commun. 2016, 7, 12265. [Google Scholar] [CrossRef][Green Version]
- Hoshino, T.; Zeller, R.; Dederichs, P.H.; Weinert, M. Magnetic Energy Anomalies of 3d Systems. Europhys. Lett. (EPL) 1993, 24, 495–500. [Google Scholar] [CrossRef]
- Philipsen, P.H.T.; Baerends, E.J. Cohesive energy of 3d transition metals: Density functional theory atomic and bulk calculations. Phys. Rev. B 1996, 54, 5326–5333. [Google Scholar] [CrossRef] [PubMed]
- Aldén, M.; Skriver, H.L.; Mirbt, S.; Johansson, B. Calculated surface-energy anomaly in the 3d metals. Phys. Rev. Lett. 1992, 69, 2296–2298. [Google Scholar] [CrossRef]
- Dai, J.; Ni, C.; Kan, L.; Fang, D. Characterization, Stability, and Magnetic Properties of Bimetallic Pt3Co Nanoparticles. Synth. React. Inorg. Met.-Org. Nano-Met. Chem. 2011, 41, 1188–1192. [Google Scholar] [CrossRef]
- Liu, Y.; Jiang, Y.; Zhang, X.; Wang, Y.; Zhang, Y.; Liu, H.; Zhai, H.; Liu, Y.; Yang, J.; Yan, Y. Structural and magnetic properties of the ordered FePt3, FePt and Fe3Pt nanoparticles. J. Solid State Chem. 2014, 209, 69–73. [Google Scholar] [CrossRef]
- Gillespie, D.J.; Schindler, A.I. The Magnetic Properties of Concentrated Pt-Ni Alloys in the Ordered and Disordered States. AIP Conf. Proc. 1972, 5, 461–465. [Google Scholar] [CrossRef]
- Darling, A.S. A.M.I.Mech.E. at Research Laboratories, Johnson Matthey & Co Limited. Cobalt-Platinum Alloys. A Critical Review of their Constitution and Properties. Platin. Met. Rev. 1963, 7, 96–104. [Google Scholar]
- Kim, H.Y.; Joo, S.H. Recent advances in nanostructured intermetallic electrocatalysts for renewable energy conversion reactions. J. Mater. Chem. A 2020, 8, 8195–8217. [Google Scholar] [CrossRef]
- Cheng, H.; Gui, R.; Yu, H.; Wang, C.; Liu, S.; Liu, H.; Zhou, T.; Zhang, N.; Zheng, X.; Chu, W.; et al. Subsize Pt-based intermetallic compound enables long-term cyclic mass activity for fuel-cell oxygen reduction. Proc. Natl. Acad. Sci. USA 2021, 118, e2104026118. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Pu, M.; Chen, Q.; Zhu, H. Pt-Based Intermetallic Nanocrystals in Cathode Catalysts for Proton Exchange Membrane Fuel Cells: From Precise Synthesis to Oxygen Reduction Reaction Strategy. Catalysts 2021, 11, 1050. [Google Scholar] [CrossRef]
- Laughlin, D.E.; Srinivasan, K.; Tanase, M.; Wang, L. Crystallographic aspects of L10 magnetic materials. Scr. Mater. 2005, 53, 383–388. [Google Scholar] [CrossRef]
- Pearson, W.B. A Handbook of Lattice Spacings and Structures of Metals and Alloys; Pergamon Press: Oxford, UK, 1967. [Google Scholar]
- Mehl, M.J.; Hicks, D.; Toher, C.; Levy, O.; Hanson, R.M.; Hart, G.; Curtarolo, S. The AFLOW Library of Crystallographic Prototypes: Part 1. Comput. Mater. Sci. 2017, 136, S1–S828. [Google Scholar] [CrossRef]
- Hicks, D.; Mehl, M.J.; Gossett, E.; Toher, C.; Levy, O.; Hanson, R.M.; Hart, G.; Curtarolo, S. The AFLOW Library of Crystallographic Prototypes: Part 2. Comput. Mater. Sci. 2019, 161, S1–S1011. [Google Scholar] [CrossRef]
- Hicks, D.; Mehl, M.J.; Esters, M.; Oses, C.; Levy, O.; Hart, G.L.W.; Toher, C.; Curtarolo, S. The AFLOW Library of Crystallographic Prototypes: Part 3. Comput. Mater. Sci. 2021, 199, 110450. [Google Scholar] [CrossRef]
- Nguyen, H.L.; Howard, L.E.M.; Stinton, G.W.; Giblin, S.R.; Tanner, B.K.; Terry, I.; Hughes, A.K.; Ross, I.M.; Serres, A.; Evans, J.S.O. Synthesis of Size-Controlled fcc and fct FePt Nanoparticles. Chem. Mater. 2006, 18, 6414–6424. [Google Scholar] [CrossRef]
- Rellinghaus, B.; Stappert, S.; Acet, M.; Wassermann, E.F. Magnetic properties of FePt nanoparticles. J. Magn. Magn. Mater. 2003, 266, 142–154. [Google Scholar] [CrossRef]
- Rong, C.-B.; Li, Y.; Liu, J.P. Curie temperatures of annealed FePt nanoparticle systems. J. Appl. Phys. 2007, 101, 09K505. [Google Scholar] [CrossRef]
- Seo, O.; Lee, J.Y.; Kim, J.M.; Kim, J.-W.; Kang, H.C.; Chung, J.; Noh, D.Y. Chemical ordering in PtNi nanocrystals. J. Alloys Compd. 2016, 666, 232–236. [Google Scholar] [CrossRef]
- Lyubina, J.; Rellinghaus, B.; Gutfleisch, O.; Albrecht, M. Chapter Five—Structure and Magnetic Properties of L10-Ordered Fe–Pt Alloys and Nanoparticles. In Handbook of Magnetic Materials; Buschow, K.H.J., Ed.; Elsevier: Amsterdam, The Netherlands, 2011; pp. 291–407. [Google Scholar]
- Howard, L.E.M.; Nguyen, H.L.; Giblin, S.R.; Tanner, B.K.; Terry, I.; Hughes, A.K.; Evans, J.S.O. A Synthetic Route to Size-Controlled fcc and fct FePt Nanoparticles. J. Am. Chem. Soc. 2005, 127, 10140–10141. [Google Scholar] [CrossRef]
- Hu, J.; Chen, J.; Ju, G. L10 Fept for Magnetic Recording Media Application. In Developments in Data Storage; Wiley: Hoboken, NJ, USA, 2011; pp. 223–255. [Google Scholar]
- Liang, J.; Ma, F.; Hwang, S.; Wang, X.; Sokolowski, J.; Li, Q.; Wu, G.; Su, D. Atomic Arrangement Engineering of Metallic Nanocrystals for Energy-Conversion Electrocatalysis. Joule 2019, 3, 956–991. [Google Scholar] [CrossRef]
- Alloyeau, D.; Ricolleau, C.; Mottet, C.; Oikawa, T.; Langlois, C.; Le Bouar, Y.; Braidy, N.; Loiseau, A. Size and shape effects on the order–disorder phase transition in CoPt nanoparticles. Nat. Mater. 2009, 8, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Brück, E.; Xiao, Q.F.; Thang, P.D.; Toonen, M.J.; de Boer, F.R.; Buschow, K.H.J. Influence of phase transformation on the permanent-magnetic properties of Fe–Pt based alloys. Phys. B Condens. Matter 2001, 300, 215–229. [Google Scholar] [CrossRef]
- Cowley, J.M. Short- and Long-Range Order Parameters in Disordered Solid Solutions. Phys. Rev. 1960, 120, 1648–1657. [Google Scholar] [CrossRef]
- Warren, B.E. X-ray Diffraction; Dover Publication: Mineola, NY, USA, 1990. [Google Scholar]
- Malheiro, A.R.; Perez, J.; Villullas, H.M. Well-Alloyed PtFe/C Nanocatalysts of Controlled Composition and Same Particle Size: Oxygen Reduction and Methanol Tolerance. J. Electrochem. Soc. 2009, 156, B51. [Google Scholar] [CrossRef]
- Xiong, L.; Manthiram, A. Effect of Atomic Ordering on the Catalytic Activity of Carbon Supported PtM (M=Fe, Co, Ni, and Cu) Alloys for Oxygen Reduction in PEMFCs. J. Electrochem. Soc. 2005, 152, A697. [Google Scholar] [CrossRef]
- Chen, L.; Bock, C.; Mercier, P.H.J.; MacDougall, B.R. Ordered alloy formation for Pt3Fe/C, PtFe/C and Pt5.75Fe5.75Cuy/CO2-reduction electro-catalysts. Electrochim. Acta 2012, 77, 212–224. [Google Scholar] [CrossRef]
- Loukrakpam, R.; Luo, J.; He, T.; Chen, Y.; Xu, Z.; Njoki, P.N.; Wanjala, B.N.; Fang, B.; Mott, D.; Yin, J.; et al. Nanoengineered PtCo and PtNi Catalysts for Oxygen Reduction Reaction: An Assessment of the Structural and Electrocatalytic Properties. J. Phys. Chem. C 2011, 115, 1682–1694. [Google Scholar] [CrossRef]
- Watanabe, M.; Tsurumi, K.; Mizukami, T.; Nakamura, T.; Stonehart, P. Activity and Stability of Ordered and Disordered Co-Pt Alloys for Phosphoric Acid Fuel Cells. J. Electrochem. Soc. 1994, 141, 2659–2668. [Google Scholar] [CrossRef]
- Oezaslan, M.; Hasché, F.; Strasser, P. Oxygen Electroreduction on PtCo3, PtCo and Pt3Co Alloy Nanoparticles for Alkaline and Acidic PEM Fuel Cells. J. Electrochem. Soc. 2012, 159, B394–B405. [Google Scholar] [CrossRef]
- Travitsky, N.; Ripenbein, T.; Golodnitsky, D.; Rosenberg, Y.; Burshtein, L.; Peled, E. Pt-, PtNi- and PtCo-supported catalysts for oxygen reduction in PEM fuel cells. J. Power Sources 2006, 161, 782–789. [Google Scholar] [CrossRef]
- Carpenter, M.K.; Moylan, T.E.; Kukreja, R.S.; Atwan, M.H.; Tessema, M.M. Solvothermal Synthesis of Platinum Alloy Nanoparticles for Oxygen Reduction Electrocatalysis. J. Am. Chem. Soc. 2012, 134, 8535–8542. [Google Scholar] [CrossRef]
- Ding, Y.; Majetich, S.A. Saturation of Nuclei Concentration in the Phase Transformation of FePt Nanoparticles. IEEE Trans. Magn. 2007, 43, 3100–3102. [Google Scholar] [CrossRef]
- Rellinghaus, B.; Mohn, E.; Schultz, L.; Gemming, T.; Acet, M.; Kowalik, A.; Kock, B.F. On the L10 Ordering Kinetics in Fe-Pt Nanoparticles. IEEE Trans. Magn. 2006, 42, 3048–3050. [Google Scholar] [CrossRef]
- Xiao, Q.F.; Brück, E.; Zhang, Z.D.; de Boer, F.R.; Buschow, K.H.J. Phase transformation and magnetic properties of bulk CoPt alloy. J. Alloys Compd. 2004, 364, 64–71. [Google Scholar] [CrossRef]
- Sun, X.; Jia, Z.Y.; Huang, Y.H.; Harrell, J.W.; Nikles, D.E.; Sun, K.; Wang, L.M. Synthesis and magnetic properties of CoPt nanoparticles. J. Appl. Phys. 2004, 95, 6747–6749. [Google Scholar] [CrossRef]
- Amador, C.; Lambrecht, W.R.L.; van Schilfgaarde, M.; Segall, B. Internal strain effects on the phase diagram of Ni-Pt alloys. Phys. Rev. B 1993, 47, 15276–15279. [Google Scholar] [CrossRef]
- Imaizumi, M.; Soufen, C.A.; Pintão, C.A.F.; Varanda, L.C.; Jafelicci, M. Structural phase transition study of FePt alloys using ab initio calculation. Mater. Sci. Eng. A 2009, 521–522, 167–168. [Google Scholar] [CrossRef]
- Müller, M.; Erhart, P.; Albe, K. Thermodynamics of L10 ordering in FePt nanoparticles studied by Monte Carlo simulations based on an analytic bond-order potential. Phys. Rev. B 2007, 76, 155412. [Google Scholar] [CrossRef]
- Hizi, A.; Garbouj, H.; Mottet, C.; Said, M. Chemical ordering and surface segregation in Ni1-cPtc system: A theoretical study from the alloys to the nanoalloys. Results Phys. 2019, 14, 102493. [Google Scholar] [CrossRef]
- Kolhatkar, A.G.; Jamison, A.C.; Litvinov, D.; Willson, R.C.; Lee, T.R. Tuning the Magnetic Properties of Nanoparticles. Int. J. Mol. Sci. 2013, 14, 15977–16009. [Google Scholar] [CrossRef]
- Weller, D.; Moser, A.; Folks, L.; Best, M.E.; Wen, L.; Toney, M.F.; Schwickert, M.; Thiele, J.U.; Doerner, M.F. High Ku materials approach to 100 Gbits/in2. IEEE Trans. Magn. 2000, 36, 10–15. [Google Scholar] [CrossRef]
- Johnson, M.T.; Bloemen, P.J.H.; Broeder, F.J.A.d.; Vries, J.J.d. Magnetic anisotropy in metallic multilayers. Rep. Prog. Phys. 1996, 59, 1409–1458. [Google Scholar] [CrossRef]
- Sato, K.; Hirotsu, Y. Magnetoanisotropy, long-range order parameter and thermal stability of isolated L10 FePt nanoparticles with mutual fixed orientation. J. Magn. Magn. Mater. 2004, 272–276, 1497–1499. [Google Scholar] [CrossRef]
- Klemmer, T.J.; Shukla, N.; Liu, C.; Wu, X.W.; Svedberg, E.B.; Mryasov, O.; Chantrell, R.W.; Weller, D.; Tanase, M.; Laughlin, D.E. Structural studies of L10 FePt nanoparticles. Appl. Phys. Lett. 2002, 81, 2220–2222. [Google Scholar] [CrossRef]
- Kabir, A.; Hu, J.; Turkowski, V.; Wu, R.; Camley, R.; Rahman, T.S. Effect of structure on the magnetic anisotropy of L10 FePt nanoparticles. Phys. Rev. B 2015, 92, 054424. [Google Scholar] [CrossRef]
- Sakuma, A. First Principle Calculation of the Magnetocrystalline Anisotropy Energy of FePt and CoPt Ordered Alloys. J. Phys. Soc. Jpn. 1994, 63, 3053–3058. [Google Scholar] [CrossRef]
- Ke, L. Intersublattice magnetocrystalline anisotropy using a realistic tight-binding method based on maximally localized Wannier functions. Phys. Rev. B 2019, 99, 054418. [Google Scholar] [CrossRef]
- Alsaad, A.; Ahmad, A.A.; Shukri, A.A.; Bani-Younes, O.A. Determination of Magneto-crystalline Anisotropy Energy (MAE) Of ordered L10 CoPt and FePt nanoparticles. IOP Conf. Ser. Mater. Sci. Eng. 2018, 305, 012017. [Google Scholar] [CrossRef]
- Šipr, O.; Minár, J.; Mankovsky, S.; Ebert, H. Influence of composition, many-body effects, spin-orbit coupling, and disorder on magnetism of Co-Pt solid-state systems. Phys. Rev. B 2008, 78, 144403. [Google Scholar] [CrossRef]
- Coey, J.M.D. Permanent magnets: Plugging the gap. Scr. Mater. 2012, 67, 524–529. [Google Scholar] [CrossRef]
- Paudyal, D.; Saha-Dasgupta, T.; Mookerjee, A. Magnetic properties of X–Pt (X = Fe,Co,Ni) alloy systems. J. Phys. Condens. Matter 2004, 16, 2317–2334. [Google Scholar] [CrossRef]
- Okamoto, H.; Schlesinger, M.E.; Mueller, E.M. (Eds.) ASM Handbook: Alloy Phase Diagrams; ASM International: Materials Park, OH, USA, 1992; Volume 3. [Google Scholar]
- Fredriksson, P.; Sundman, B. A thermodynamic assessment of the Fe-Pt system. Calphad 2001, 25, 535–548. [Google Scholar] [CrossRef]
- Hultgren, R.; Desai, P.D.; Hawkins, D.T.; Gleiser, M.; Kelley, K.K. Selected Values of the Thermodynamic Properties of Binary Alloys; ASM: Metal Park, OH, USA, 1973. [Google Scholar]
- Leroux, C.; Cadeville, M.C.; Pierron-Bohnes, V.; Inden, G.; Hinz, F. Comparative investigation of structural and transport properties of L10 NiPt and CoPt phases; the role of magnetism. J. Phys. F Met. Phys. 1988, 18, 2033–2051. [Google Scholar] [CrossRef]
- Woolley, J.C.; Phillips, J.H.; Clark, J.A. Ordering in CoPt-CrPt and CoPt-MnPt alloys. J. Less Common Met. 1964, 6, 461–471. [Google Scholar] [CrossRef]
- Kootte, A.; Haas, C.; Groot, R.A.d. The electronic structure of ordered binary Co-Pt compounds. J. Phys. Condens. Matter 1991, 3, 1133–1152. [Google Scholar] [CrossRef]
- Weller, D.; Moser, A. Thermal effect limits in ultrahigh-density magnetic recording. IEEE Trans. Magn. 1999, 35, 4423–4439. [Google Scholar] [CrossRef]
- Vlasova, N.I.; Kandaurova, G.S.; Shchegoleva, N.N. Effect of the polytwinned microstructure parameters on magnetic domain structure and hysteresis properties of the CoPt-type alloys. J. Magn. Magn. Mater. 2000, 222, 138–158. [Google Scholar] [CrossRef]
- Pierre, J.-L. Chemistry of dioxygen and its activated species. In Analysis of Free Radicals in Biological Systems; Favier, A.E., Cadet, J., Kalyanaraman, B., Fontecave, M., Eds.; Birkhäuser: Basel, Switzerland, 1995. [Google Scholar]
- Chen, G.; Sun, Y.; Chen, R.R.; Biz, C.; Fisher, A.C.; Sherburne, M.P.; Ager Iii, J.W.; Gracia, J.; Xu, Z.J. A discussion on the possible involvement of singlet oxygen in oxygen electrocatalysis. J. Phys. Energy 2021, 3, 031004. [Google Scholar] [CrossRef]
- Ebsworth, E.A.V.; Connor, J.A.; Turner, J.J. The Chemistry of Oxygen; Pergamon Press: Oxford, UK, 1973; Volume 16. [Google Scholar]
- Borden, W.T.; Hoffmann, R.; Stuyver, T.; Chen, B. Dioxygen: What Makes This Triplet Diradical Kinetically Persistent? J. Am. Chem. Soc. 2017, 139, 9010–9018. [Google Scholar] [CrossRef]
- Fridovich, I. Oxygen: How Do We Stand It? Med. Princ. Pract. 2013, 22, 131–137. [Google Scholar] [CrossRef]
- Green, M.J.; Hill, H.A.O. Chemistry of Dioxygen in Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1984; Volume 105, pp. 3–22. [Google Scholar]
- Poli, R.; Harvey, J.N. Spin forbidden chemical reactions of transition metal compounds. New ideas and new computational challenges. Chem. Soc. Rev. 2003, 32, 1–8. [Google Scholar] [CrossRef]
- Minaev, B. Spin effects in reductive activation of O2 by oxydase enzymes. RIKEN Rev. 2002, 44, 147–149. [Google Scholar]
- Li, J.; Ma, J.; Ma, Z.; Zhao, E.; Du, K.; Guo, J.; Ling, T. Spin Effect on Oxygen Electrocatalysis. Adv. Energy Sustain. Res. 2021, 2, 2100034. [Google Scholar] [CrossRef]
- Sun, Y.; Sun, S.; Yang, H.; Xi, S.; Gracia, J.; Xu, Z.J. Spin-Related Electron Transfer and Orbital Interactions in Oxygen Electrocatalysis. Adv. Mater. 2020, 32, 2003297. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Xu, Z.J. Oxygen evolution in spin-sensitive pathways. Curr. Opin. Electrochem. 2021, 30, 100804. [Google Scholar] [CrossRef]
- Li, X.; Cheng, Z.; Wang, X. Understanding the Mechanism of the Oxygen Evolution Reaction with Consideration of Spin. Electrochem. Energy Rev. 2021, 4, 136–145. [Google Scholar] [CrossRef]
- Minaev, B.F. Role of exchange interaction in spin catalysis mechanisms. Theor. Exp. Chem. 1996, 32, 1–12. [Google Scholar] [CrossRef]
- Morozan, A.; Jousselme, B.; Palacin, S. Low-platinum and platinum-free catalysts for the oxygen reduction reaction at fuel cell cathodes. Energy Environ. Sci. 2011, 4, 1238–1254. [Google Scholar] [CrossRef]
- Ge, X.; Sumboja, A.; Wuu, D.; An, T.; Li, B.; Goh, F.W.T.; Hor, T.S.A.; Zong, Y.; Liu, Z. Oxygen Reduction in Alkaline Media: From Mechanisms to Recent Advances of Catalysts. ACS Catal. 2015, 5, 4643–4667. [Google Scholar] [CrossRef]
- Gómez-Marín, A.; Feliu, J.; Edson, T. Reaction Mechanism for Oxygen Reduction on Platinum: Existence of a Fast Initial Chemical Step and a Soluble Species Different from H2O2. ACS Catal. 2018, 8, 7931–7943. [Google Scholar] [CrossRef]
- Sepa, D.B.; Vojnovic, M.V.; Damjanovic, A. Reaction intermediates as a controlling factor in the kinetics and mechanism of oxygen reduction at platinum electrodes. Electrochim. Acta 1981, 26, 781–793. [Google Scholar] [CrossRef]
- Gómez-Marín, A.M.; Rizo, R.; Feliu, J.M. Some reflections on the understanding of the oxygen reduction reaction at Pt(111). Beilstein J. Nanotechnol. 2013, 4, 956–967. [Google Scholar] [CrossRef]
- Dong, J.-C.; Zhang, X.-G.; Briega-Martos, V.; Jin, X.; Yang, J.; Chen, S.; Yang, Z.-L.; Wu, D.-Y.; Feliu, J.M.; Williams, C.T.; et al. In situ Raman spectroscopic evidence for oxygen reduction reaction intermediates at platinum single-crystal surfaces. Nat. Energy 2019, 4, 60–67. [Google Scholar] [CrossRef]
- Wei, C.; Rao, R.R.; Peng, J.; Huang, B.; Stephens, I.E.L.; Risch, M.; Xu, Z.J.; Shao-Horn, Y. Recommended Practices and Benchmark Activity for Hydrogen and Oxygen Electrocatalysis in Water Splitting and Fuel Cells. Adv. Mater. 2019, 31, 1806296. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Han, H.; Jin, S.; Choi, S.M.; Kim, H.J.; Seo, M.H.; Kim, W.B. A Review on Recent Progress in the Aspect of Stability of Oxygen Reduction Electrocatalysts for Proton-Exchange Membrane Fuel Cell: Quantum Mechanics and Experimental Approaches. Energy Technol. 2019, 7, 1900312. [Google Scholar] [CrossRef]
- Keith, J.A.; Jerkiewicz, G.; Jacob, T. Theoretical Investigations of the Oxygen Reduction Reaction on Pt(111). ChemPhysChem 2010, 11, 2779–2794. [Google Scholar] [CrossRef] [PubMed]
- Jinnouchi, R.; Kodama, K.; Hatanaka, T.; Morimoto, Y. First principles based mean field model for oxygen reduction reaction. Phys. Chem. Chem. Phys. 2011, 13, 21070–21083. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, V.; Hansen, H.A.; Rossmeisl, J.; Nørskov, J.K. Universality in Oxygen Reduction Electrocatalysis on Metal Surfaces. ACS Catal. 2012, 2, 1654–1660. [Google Scholar] [CrossRef]
- Wang, S.; Zhu, E.; Huang, Y.; Heinz, H. Direct correlation of oxygen adsorption on platinum-electrolyte interfaces with the activity in the oxygen reduction reaction. Sci. Adv. 2021, 7, eabb1435. [Google Scholar] [CrossRef]
- Tripković, V.; Skúlason, E.; Siahrostami, S.; Nørskov, J.K.; Rossmeisl, J. The oxygen reduction reaction mechanism on Pt(111) from density functional theory calculations. Electrochim. Acta 2010, 55, 7975–7981. [Google Scholar] [CrossRef]
- Van Santen, R.A.; Neurock, M. Molecular Heterogeneus Catalysis. A Conceptual an Computational Approach; Wiley-VCH: Hoboken, NJ, USA, 2006. [Google Scholar]
- Nørskov, J.K.; Rossmeisl, J.; Logadottir, A.; Lindqvist, L.; Kitchin, J.R.; Bligaard, T.; Jónsson, H. Origin of the Overpotential for Oxygen Reduction at a Fuel-Cell Cathode. J. Phys. Chem. B 2004, 108, 17886–17892. [Google Scholar] [CrossRef]
- Adžić, R.R.; Wang, J.X. Configuration and Site of O2 Adsorption on the Pt(111) Electrode Surface. J. Phys. Chem. B 1998, 102, 8988–8993. [Google Scholar] [CrossRef]
- Christensen, P.A.; Hamnett, A.; Linares-Moya, D. Oxygen reduction and fuel oxidation in alkaline solution. Phys. Chem. Chem. Phys. 2011, 13, 5206–5214. [Google Scholar] [CrossRef] [PubMed]
- Zinola, C.F.; Arvia, A.J.; Estiu, G.L.; Castro, E.A. A Quantum Chemical Approach to the Influence of Platinum Surface Structure on the Oxygen Electroreduction Reaction. J. Phys. Chem. 1994, 98, 7566–7576. [Google Scholar] [CrossRef]
- Shao, M.-H.; Liu, P.; Adzic, R.R. Superoxide Anion is the Intermediate in the Oxygen Reduction Reaction on Platinum Electrodes. J. Am. Chem. Soc. 2006, 128, 7408–7409. [Google Scholar] [CrossRef] [PubMed]
- Lang, P.; Yuan, N.; Jiang, Q.; Zhang, Y.; Tang, J. Recent Advances and Prospects of Metal-Based Catalysts for Oxygen Reduction Reaction. Energy Technol. 2020, 8, 1900984. [Google Scholar] [CrossRef]
- Shao, M.; Liu, P.; Zhang, J.; Adzic, R. Origin of Enhanced Activity in Palladium Alloy Electrocatalysts for Oxygen Reduction Reaction. J. Phys. Chem. B 2007, 111, 6772–6775. [Google Scholar] [CrossRef]
- Park, Y.; Lee, B.; Kim, C.; Kim, J.; Nam, S.; Oh, Y.; Park, B. Modification of Gold Catalysis with Aluminum Phosphate for Oxygen-Reduction Reaction. J. Phys. Chem. C 2010, 114, 3688–3692. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, K.S.; Mishler, J.; Cho, S.C.; Adroher, X.C. A review of polymer electrolyte membrane fuel cells: Technology, applications, and needs on fundamental research. Appl. Energy 2011, 88, 981–1007. [Google Scholar] [CrossRef]
- Xia, W.; Mahmood, A.; Liang, Z.; Zou, R.; Guo, S. Earth-Abundant Nanomaterials for Oxygen Reduction. Angew. Chem. Int. Ed. 2016, 55, 2650–2676. [Google Scholar] [CrossRef]
- Serrà, A.; Grau, S.; Gimbert-Suriñach, C.; Sort, J.; Nogués, J.; Vallés, E. Magnetically-actuated mesoporous nanowires for enhanced heterogeneous catalysis. Appl. Catal. B Environ. 2017, 217, 81–91. [Google Scholar] [CrossRef]
- Du, S. Recent Advances in Electrode Design Based on One-Dimensional Nanostructure Arrays for Proton Exchange Membrane Fuel Cell Applications. Engineering 2021, 7, 33–49. [Google Scholar] [CrossRef]
- He, J.; Gao, T.; Jiang, T.; Mu, B.; Suo, Y.; Zhang, Z.; Su, J. Nonprecious Nanoalloys Embedded in N-Enriched Mesoporous Carbons Derived from a Dual-MOF as Highly Active Catalyst towards Oxygen Reduction Reaction. ChemistrySelect 2018, 3, 7913–7920. [Google Scholar] [CrossRef]
- Othman, R.; Dicks, A.L.; Zhu, Z. Non precious metal catalysts for the PEM fuel cell cathode. Int. J. Hydrogen Energy 2012, 37, 357–372. [Google Scholar] [CrossRef]
- Zhang, J.; Yuan, Y.; Gao, L.; Zeng, G.; Li, M.; Huang, H. Stabilizing Pt-Based Electrocatalysts for Oxygen Reduction Reaction: Fundamental Understanding and Design Strategies. Adv. Mater. 2021, 33, 2006494. [Google Scholar] [CrossRef]
- Malacrida, P.; Escudero-Escribano, M.; Verdaguer-Casadevall, A.; Stephens, I.E.L.; Chorkendorff, I. Enhanced activity and stability of Pt–La and Pt–Ce alloys for oxygen electroreduction: The elucidation of the active surface phase. J. Mater. Chem. A 2014, 2, 4234–4243. [Google Scholar] [CrossRef]
- Vej-Hansen, U.G.; Escudero-Escribano, M.; Velázquez-Palenzuela, A.; Malacrida, P.; Rossmeisl, J.; Stephens, I.E.L.; Chorkendorff, I.; Schiøtz, J. New Platinum Alloy Catalysts for Oxygen Electroreduction Based on Alkaline Earth Metals. Electrocatalysis 2017, 8, 594–604. [Google Scholar] [CrossRef]
- Kim, C.; Dionigi, F.; Beermann, V.; Wang, X.; Möller, T.; Strasser, P. Alloy Nanocatalysts for the Electrochemical Oxygen Reduction (ORR) and the Direct Electrochemical Carbon Dioxide Reduction Reaction (CO2RR). Adv. Mater. 2019, 31, 1805617. [Google Scholar] [CrossRef] [PubMed]
- Oezaslan, M.; Hasché, F.; Strasser, P. PtCu3, PtCu and Pt3Cu Alloy Nanoparticle Electrocatalysts for Oxygen Reduction Reaction in Alkaline and Acidic Media. J. Electrochem. Soc. 2012, 159, B444–B454. [Google Scholar] [CrossRef]
- Jong Yoo, S.; Kim, S.-K.; Jeon, T.-Y.; Jun Hwang, S.; Lee, J.-G.; Lee, S.-C.; Lee, K.-S.; Cho, Y.-H.; Sung, Y.-E.; Lim, T.-H. Enhanced stability and activity of Pt–Y alloy catalysts for electrocatalytic oxygen reduction. Chem. Commun. 2011, 47, 11414–11416. [Google Scholar] [CrossRef] [PubMed]
- Greeley, J.; Stephens, I.E.L.; Bondarenko, A.S.; Johansson, T.P.; Hansen, H.A.; Jaramillo, T.F.; Rossmeisl, J.; Chorkendorff, I.; Nørskov, J.K. Alloys of platinum and early transition metals as oxygen reduction electrocatalysts. Nat. Chem. 2009, 1, 552–556. [Google Scholar] [CrossRef]
- Brown, R.; Vorokhta, M.; Khalakhan, I.; Dopita, M.; Vonderach, T.; Skála, T.; Lindahl, N.; Matolínová, I.; Grönbeck, H.; Neyman, K.M.; et al. Unraveling the Surface Chemistry and Structure in Highly Active Sputtered Pt3Y Catalyst Films for the Oxygen Reduction Reaction. ACS Appl. Mater. Interfaces 2020, 12, 4454–4462. [Google Scholar] [CrossRef]
- Wang, C.; Chi, M.; Li, D.; van der Vliet, D.; Wang, G.; Lin, Q.; Mitchell, J.F.; More, K.L.; Markovic, N.M.; Stamenkovic, V.R. Synthesis of Homogeneous Pt-Bimetallic Nanoparticles as Highly Efficient Electrocatalysts. ACS Catal. 2011, 1, 1355–1359. [Google Scholar] [CrossRef]
- Stamenkovic, V.R.; Mun, B.S.; Arenz, M.; Mayrhofer, K.J.J.; Lucas, C.A.; Wang, G.; Ross, P.N.; Markovic, N.M. Trends in electrocatalysis on extended and nanoscale Pt-bimetallic alloy surfaces. Nat. Mater. 2007, 6, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Gasteiger, H.A.; Kocha, S.S.; Sompalli, B.; Wagner, F.T. Activity benchmarks and requirements for Pt, Pt-alloy, and non-Pt oxygen reduction catalysts for PEMFCs. Appl. Catal. B Environ. 2005, 56, 9–35. [Google Scholar] [CrossRef]
- Stamenkovic, V.; Mun, B.S.; Mayrhofer, K.J.J.; Ross, P.N.; Markovic, N.M.; Rossmeisl, J.; Greeley, J.; Nørskov, J.K. Changing the Activity of Electrocatalysts for Oxygen Reduction by Tuning the Surface Electronic Structure. Angew. Chem. Int. Ed. 2006, 45, 2897–2901. [Google Scholar] [CrossRef] [PubMed]
- Kitchin, J.R.; Nørskov, J.K.; Barteau, M.A.; Chen, J.G. Modification of the surface electronic and chemical properties of Pt(111) by subsurface 3d transition metals. J. Chem. Phys. 2004, 120, 10240–10246. [Google Scholar] [CrossRef]
- Biz, C.; Fianchini, M.; Polo, V.; Gracia, J. Magnetism and Heterogeneous Catalysis: In Depth on the Quantum Spin-Exchange Interactions in Pt3M (M = V, Cr, Mn, Fe, Co, Ni, and Y)(111) Alloys. ACS Appl. Mater. Interfaces 2020, 12, 50484–50494. [Google Scholar] [CrossRef]
- Kim, S.-H.; Kang, Y.; Ham, H.C. First-Principles Study of Pt-Based Bifunctional Oxygen Evolution & Reduction Electrocatalyst: Interplay of Strain and Ligand Effects. Energies 2021, 14, 7814. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Goodman, D.W. Surface science studies of the electronic and chemical properties of bimetallic systems. J. Phys. Chem. 1991, 95, 4196–4206. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Goodman, D.W. The Nature of the Metal-Metal Bond in Bimetallic Surfaces. Science 1992, 257, 897–903. [Google Scholar] [CrossRef]
- Yu, W.; Porosoff, M.D.; Chen, J.G. Review of Pt-Based Bimetallic Catalysis: From Model Surfaces to Supported Catalysts. Chem. Rev. 2012, 112, 5780–5817. [Google Scholar] [CrossRef]
- Bligaard, T.; Nørskov, J.K. Ligand effects in heterogeneous catalysis and electrochemistry. Electrochim. Acta 2007, 52, 5512–5516. [Google Scholar] [CrossRef]
- Mavrikakis, M.; Hammer, B.; Nørskov, J.K. Effect of Strain on the Reactivity of Metal Surfaces. Phys. Rev. Lett. 1998, 81, 2819–2822. [Google Scholar] [CrossRef]
- Liu, P.; Nørskov, J.K. Ligand and ensemble effects in adsorption on alloy surfaces. Phys. Chem. Chem. Phys. 2001, 3, 3814–3818. [Google Scholar] [CrossRef]
- Burch, R. Importance of electronic ligand effects in metal alloy catalysts. Acc. Chem. Res. 1982, 15, 24–31. [Google Scholar] [CrossRef]
- Li, H.; Shin, K.; Henkelman, G. Effects of ensembles, ligand, and strain on adsorbate binding to alloy surfaces. J. Chem. Phys. 2018, 149, 174705. [Google Scholar] [CrossRef]
- Colón-Mercado, H.R.; Popov, B.N. Stability of platinum based alloy cathode catalysts in PEM fuel cells. J. Power Sources 2006, 155, 253–263. [Google Scholar] [CrossRef]
- Peng, L.; Ringe, E.; Van Duyne, R.P.; Marks, L.D. Segregation in bimetallic nanoparticles. Phys. Chem. Chem. Phys. 2015, 17, 27940–27951. [Google Scholar] [CrossRef]
- Menning, C.A.; Chen, J.G. General trend for adsorbate-induced segregation of subsurface metal atoms in bimetallic surfaces. J. Chem. Phys. 2009, 130, 174709. [Google Scholar] [CrossRef]
- Ma, Y.; Balbuena, P.B. Surface segregation in bimetallic Pt3M (M = Fe, Co, Ni) alloys with adsorbed oxygen. Surf. Sci. 2009, 603, 349–353. [Google Scholar] [CrossRef]
- Wei, G.-F.; Liu, Z.-P. Towards active and stable oxygen reduction cathodes: A density functional theory survey on Pt2M skin alloys. Energy Environ. Sci. 2011, 4, 1268–1272. [Google Scholar] [CrossRef]
- Balandin, A.A. Modern State of the Multiplet Theory of Heterogeneous Catalysis. In Advances in Catalysis; Eley, D.D., Pines, H., Weisz, P.B., Eds.; Academic Press: Cambridge, MA, USA, 1969; pp. 1–210. [Google Scholar]
- Menning, C.A.; Chen, J.G. Thermodynamics and kinetics of oxygen-induced segregation of 3d metals in Pt–3d–Pt(111) and Pt–3d–Pt(100) bimetallic structures. J. Chem. Phys. 2008, 128, 164703. [Google Scholar] [CrossRef]
- Cui, C.; Gan, L.; Heggen, M.; Rudi, S.; Strasser, P. Compositional segregation in shaped Pt alloy nanoparticles and their structural behaviour during electrocatalysis. Nat. Mater. 2013, 12, 765–771. [Google Scholar] [CrossRef]
- Ruban, A.V.; Skriver, H.L.; Nørskov, J.K. Surface segregation energies in transition-metal alloys. Phys. Rev. B 1999, 59, 15990–16000. [Google Scholar] [CrossRef]
- Ramírez-Caballero, G.E.; Ma, Y.; Callejas-Tovar, R.; Balbuena, P.B. Surface segregation and stability of core–shell alloy catalysts for oxygen reduction in acid medium. Phys. Chem. Chem. Phys. 2010, 12, 2209–2218. [Google Scholar] [CrossRef]
- Farsi, L.; Deskins, N.A. First principles analysis of surface dependent segregation in bimetallic alloys. Phys. Chem. Chem. Phys. 2019, 21, 23626–23637. [Google Scholar] [CrossRef]
- Wang, Y.-H.; Le, J.-B.; Li, W.-Q.; Wei, J.; Radjenovic, P.M.; Zhang, H.; Zhou, X.-S.; Cheng, J.; Tian, Z.-Q.; Li, J.-F. In situ Spectroscopic Insight into the Origin of the Enhanced Performance of Bimetallic Nanocatalysts towards the Oxygen Reduction Reaction (ORR). Angew. Chem. Int. Ed. 2019, 58, 16062–16066. [Google Scholar] [CrossRef]
- Toda, T.; Igarashi, H.; Uchida, H.; Watanabe, M. Enhancement of the Electroreduction of Oxygen on Pt Alloys with Fe, Ni, and Co. J. Electrochem. Soc. 1999, 146, 3750–3756. [Google Scholar] [CrossRef]
- Jung, C.; Lee, C.; Bang, K.; Lim, J.; Lee, H.; Ryu, H.J.; Cho, E.; Lee, H.M. Synthesis of Chemically Ordered Pt3Fe/C Intermetallic Electrocatalysts for Oxygen Reduction Reaction with Enhanced Activity and Durability via a Removable Carbon Coating. ACS Appl. Mater. Interfaces 2017, 9, 31806–31815. [Google Scholar] [CrossRef]
- Yang, D.; Gu, J.; Liu, X.; He, H.; Wang, M.; Wang, P.; Zhu, Y.; Fan, Q.; Huang, R. Monodispersed Pt3Ni Nanoparticles as a Highly Efficient Electrocatalyst for PEMFCs. Catalysts 2019, 9, 588. [Google Scholar] [CrossRef]
- Wang, C.; van der Vliet, D.; Chang, K.-C.; You, H.; Strmcnik, D.; Schlueter, J.A.; Markovic, N.M.; Stamenkovic, V.R. Monodisperse Pt3Co Nanoparticles as a Catalyst for the Oxygen Reduction Reaction: Size-Dependent Activity. J. Phys. Chem. C 2009, 113, 19365–19368. [Google Scholar] [CrossRef]
- Koh, S.; Toney, M.F.; Strasser, P. Activity–stability relationships of ordered and disordered alloy phases of Pt3Co electrocatalysts for the oxygen reduction reaction (ORR). Electrochim. Acta 2007, 52, 2765–2774. [Google Scholar] [CrossRef]
- Xiong, Y.; Xiao, L.; Yang, Y.; DiSalvo, F.J.; Abruña, H.D. High-Loading Intermetallic Pt3Co/C Core–Shell Nanoparticles as Enhanced Activity Electrocatalysts toward the Oxygen Reduction Reaction (ORR). Chem. Mater. 2018, 30, 1532–1539. [Google Scholar] [CrossRef]
- Cheng, Z.; Liao, S.; Zhou, W.; Luo, G.; Huang, H. Straightforward synthesis of chemically ordered Pt3Co/C nanoparticles by a solid phase method for oxygen-reduction reaction. Ionics 2021, 27, 2553–2560. [Google Scholar] [CrossRef]
- Haile, A.S.; Yohannes, W.; Mekonnen, Y.S. Oxygen reduction reaction on Pt-skin Pt3V(111) fuel cell cathode: A density functional theory study. RSC Adv. 2020, 10, 27346–27356. [Google Scholar] [CrossRef] [PubMed]
- Biz, C.; Fianchini, M.; Gracia, J. Catalysis Meets Spintronics; Spin Potentials Associated with Open-Shell Orbital Configurations Enhance the Activity of Pt3Co Nanostructures for Oxygen Reduction: A Density Functional Theory Study. ACS Appl. Nano Mater. 2020, 3, 506–515. [Google Scholar] [CrossRef]
- Zosiak, L.; Goyhenex, C.; Kozubski, R.; Tréglia, G. Electronic structure of CoPt based systems: From bulk to nanoalloys. J. Phys. Condens. Matter 2015, 27, 455503. [Google Scholar] [CrossRef]
- Sharma, S.; Zeng, C.; Peterson, A.A. Face-centered tetragonal (FCT) Fe and Co alloys of Pt as catalysts for the oxygen reduction reaction (ORR): A DFT study. J. Chem. Phys. 2018, 150, 041704. [Google Scholar] [CrossRef]
- Sui, S.; Wang, X.; Zhou, X.; Su, Y.; Riffat, S.; Liu, C.-J. A comprehensive review of Pt electrocatalysts for the oxygen reduction reaction: Nanostructure, activity, mechanism and carbon support in PEM fuel cells. J. Mater. Chem. A 2017, 5, 1808–1825. [Google Scholar] [CrossRef]
- Antolini, E. Alloy vs. intermetallic compounds: Effect of the ordering on the electrocatalytic activity for oxygen reduction and the stability of low temperature fuel cell catalysts. Appl. Catal. B Environ. 2017, 217, 201–213. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, X.; Guan, J. Applications of Magnetic Nanomaterials in Heterogeneous Catalysis. ACS Appl. Nano Mater. 2019, 2, 4681–4697. [Google Scholar] [CrossRef]
- Li, J.; Xi, Z.; Pan, Y.-T.; Spendelow, J.S.; Duchesne, P.N.; Su, D.; Li, Q.; Yu, C.; Yin, Z.; Shen, B.; et al. Fe Stabilization by Intermetallic L10-FePt and Pt Catalysis Enhancement in L10-FePt/Pt Nanoparticles for Efficient Oxygen Reduction Reaction in Fuel Cells. J. Am. Chem. Soc. 2018, 140, 2926–2932. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sharma, S.; Liu, X.; Pan, Y.T.; Spendelow, J.S.; Chi, M.; Jia, Y.; Zhang, P.; Cullen, D.A.; Xi, Z.; et al. Hard-Magnet L10-CoPt Nanoparticles Advance Fuel Cell Catalysis. Joule 2019, 3, 124–135. [Google Scholar] [CrossRef]
- Chung, D.Y.; Jun, S.W.; Yoon, G.; Kwon, S.G.; Shin, D.Y.; Seo, P.; Yoo, J.M.; Shin, H.; Chung, Y.-H.; Kim, H.; et al. Highly Durable and Active PtFe Nanocatalyst for Electrochemical Oxygen Reduction Reaction. J. Am. Chem. Soc. 2015, 137, 15478–15485. [Google Scholar] [CrossRef] [PubMed]
- Antolini, E. Iron-containing platinum-based catalysts as cathode and anode materials for low-temperature acidic fuel cells: A review. RSC Adv. 2016, 6, 3307–3325. [Google Scholar] [CrossRef]
- Li, Q.; Wu, L.; Wu, G.; Su, D.; Lv, H.; Zhang, S.; Zhu, W.; Casimir, A.; Zhu, H.; Mendoza-Garcia, A.; et al. New Approach to Fully Ordered fct-FePt Nanoparticles for Much Enhanced Electrocatalysis in Acid. Nano Lett. 2015, 15, 2468–2473. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, X.; Jiang, G.; Zhu, H.; Guo, S.; Su, D.; Lu, G.; Sun, S. Tuning Nanoparticle Structure and Surface Strain for Catalysis Optimization. J. Am. Chem. Soc. 2014, 136, 7734–7739. [Google Scholar] [CrossRef]
- Liu, H.; Dou, M.; Wang, F.; Liu, J.; Ji, J.; Li, Z. Ordered intermetallic PtFe@Pt core–shell nanoparticles supported on carbon nanotubes with superior activity and durability as oxygen reduction reaction electrocatalysts. RSC Adv. 2015, 5, 66471–66475. [Google Scholar] [CrossRef]
- Ying, J.; Li, J.; Jiang, G.; Cano, Z.P.; Ma, Z.; Zhong, C.; Su, D.; Chen, Z. Metal-organic frameworks derived platinum-cobalt bimetallic nanoparticles in nitrogen-doped hollow porous carbon capsules as a highly active and durable catalyst for oxygen reduction reaction. Appl. Catal. B Environ. 2018, 225, 496–503. [Google Scholar] [CrossRef]
- Du, X.X.; He, Y.; Wang, X.X.; Wang, J.N. Fine-grained and fully ordered intermetallic PtFe catalysts with largely enhanced catalytic activity and durability. Energy Environ. Sci. 2016, 9, 2623–2632. [Google Scholar] [CrossRef]
- Gong, Y.; Yeboah, Y.D.; Lvov, S.N.; Balashov, V.; Wang, Z. Fe-Modified, Pt-Based Cathodic Electrocatalysts for Oxygen Reduction Reaction with Enhanced Methanol Tolerance. J. Electrochem. Soc. 2007, 154, B560. [Google Scholar] [CrossRef]
- Cui, Z.; Chen, H.; Zhou, W.; Zhao, M.; DiSalvo, F.J. Structurally Ordered Pt3Cr as Oxygen Reduction Electrocatalyst: Ordering Control and Origin of Enhanced Stability. Chem. Mater. 2015, 27, 7538–7545. [Google Scholar] [CrossRef]
- Zou, L.; Li, J.; Yuan, T.; Zhou, Y.; Li, X.; Yang, H. Structural transformation of carbon-supported Pt3Cr nanoparticles from a disordered to an ordered phase as a durable oxygen reduction electrocatalyst. Nanoscale 2014, 6, 10686–10692. [Google Scholar] [CrossRef] [PubMed]
- Gallego, S.; Ocal, C.; Muñoz, M.C.; Soria, F. Surface-layered ordered alloy (Pt/Pt3Mn) on Pt(111). Phys. Rev. B 1997, 56, 12139–12142. [Google Scholar] [CrossRef]
- Wang, J.X.; Inada, H.; Wu, L.; Zhu, Y.; Choi, Y.; Liu, P.; Zhou, W.-P.; Adzic, R.R. Oxygen Reduction on Well-Defined Core−Shell Nanocatalysts: Particle Size, Facet, and Pt Shell Thickness Effects. J. Am. Chem. Soc. 2009, 131, 17298–17302. [Google Scholar] [CrossRef] [PubMed]
- Bing, Y.; Liu, H.; Zhang, L.; Ghosh, D.; Zhang, J. Nanostructured Pt-alloy electrocatalysts for PEM fuel cell oxygen reduction reaction. Chem. Soc. Rev. 2010, 39, 2184–2202. [Google Scholar] [CrossRef] [PubMed]
- Stephens, I.E.L.; Bondarenko, A.S.; Grønbjerg, U.; Rossmeisl, J.; Chorkendorff, I. Understanding the electrocatalysis of oxygen reduction on platinum and its alloys. Energy Environ. Sci. 2012, 5, 6744–6762. [Google Scholar] [CrossRef]
- Selwood, P.W. Magnetism and Catalysis. Chem. Rev. 1946, 38, 41–82. [Google Scholar] [CrossRef]
- Richardson, J.T. Magnetism and catalysis. J. Appl. Phys. 1978, 49, 1781–1786. [Google Scholar] [CrossRef]
- Dalmon, J.A. Magnetic Measurements and Catalysis. In Catalyst Characterization: Physical Techniques for Solid Materials; Imelik, B., Vedrine, J.C., Eds.; Springer: Boston, MA, USA, 1994; pp. 585–609. [Google Scholar]
- Sabatier, P. La Catalyse en Chimie Organique; CH. Béranger Éditeur: Paris, France, 1920. [Google Scholar]
- Rossi, L.M.; Costa, N.J.S.; Silva, F.P.; Gonçalves, R.V. Magnetic nanocatalysts: Supported metal nanoparticles for catalytic applications. Nanotechnol. Rev. 2013, 2, 597–614. [Google Scholar] [CrossRef]
- Ma, Z.; Mohapatra, J.; Wei, K.; Liu, J.P.; Sun, S. Magnetic Nanoparticles: Synthesis, Anisotropy, and Applications. Chem. Rev. 2021. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Xu, J.; Li, W.; Sun, S.; Gao, S.; Hou, Y. Magnetic Nanostructures: Rational Design and Fabrication Strategies toward Diverse Applications. Chem. Rev. 2022, 122, 5411–5475. [Google Scholar] [CrossRef] [PubMed]
- Thomas, N.; Dionysiou, D.D.; Pillai, S.C. Heterogeneous Fenton catalysts: A review of recent advances. J. Hazard. Mater. 2021, 404, 124082. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Shah, T.; Ullah, R.; Zhou, P.; Guo, M.; Ovais, M.; Tan, Z.; Rui, Y. Review on Recent Progress in Magnetic Nanoparticles: Synthesis, Characterization, and Diverse Applications. Front. Chem. 2021, 9, 629054. [Google Scholar] [CrossRef] [PubMed]
- Hirohata, A.; Yamada, K.; Nakatani, Y.; Prejbeanu, I.-L.; Diény, B.; Pirro, P.; Hillebrands, B. Review on spintronics: Principles and device applications. J. Magn. Magn. Mater. 2020, 509, 166711. [Google Scholar] [CrossRef]
- Koksharov, Y.A.; Gubin, S.P.; Taranov, I.V.; Khomutov, G.B.; Gulyaev, Y.V. Magnetic Nanoparticles in Medicine: Progress, Problems, and Advances. J. Commun. Technol. Electron. 2022, 67, 101–116. [Google Scholar] [CrossRef]
- Shinde, P.V.; Rout, C.S. Magnetic gas sensing: Working principles and recent developments. Nanoscale Adv. 2021, 3, 1551–1568. [Google Scholar] [CrossRef]
- Carlin, R.L. Magnetochemistry; Springer: Berlin/Heidelberg, Germany, 1986. [Google Scholar]
- Selwood, P.W. Magnetochemistry; Interscience Publishers: New York, NY, USA, 1956. [Google Scholar]
- Dowden, D.A. 56. Heterogeneous catalysis. Part I. Theoretical basis. J. Chem. Soc. 1950, 242–265. [Google Scholar] [CrossRef]
- Huang, W.; Li, W.-X. Surface and interface design for heterogeneous catalysis. Phys. Chem. Chem. Phys. 2019, 21, 523–536. [Google Scholar] [CrossRef]
- Fianchini, M. Synthesis meets theory: Past, present and future of rational chemistry. Phys. Sci. Rev. 2017, 2, 20170134. [Google Scholar] [CrossRef]
- Hammes-Schiffer, S. Catalysts by Design: The Power of Theory. Acc. Chem. Res. 2017, 50, 561–566. [Google Scholar] [CrossRef]
- Seh, Z.W.; Kibsgaard, J.; Dickens, C.F.; Chorkendorff, I.; Nørskov, J.K.; Jaramillo, T.F. Combining theory and experiment in electrocatalysis: Insights into materials design. Science 2017, 355, eaad4998. [Google Scholar] [CrossRef] [PubMed]
- Cramer, C.J.; Truhlar, D.G. Density functional theory for transition metals and transition metal chemistry. Phys. Chem. Chem. Phys. 2009, 11, 10757–10816. [Google Scholar] [CrossRef]
- Shishkin, M.; Sato, H. DFT+U in Dudarev’s formulation with corrected interactions between the electrons with opposite spins: The form of Hamiltonian, calculation of forces, and bandgap adjustments. J. Chem. Phys. 2019, 151, 024102. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, F.; Aryasetiawan, F. Recent Progress in First-Principles Methods for Computing the Electronic Structure of Correlated Materials. Computation 2018, 6, 26. [Google Scholar] [CrossRef]
- Georges, A.; Medici, L.d.; Mravlje, J. Strong Correlations from Hund’s Coupling. Annu. Rev. Condens. Matter Phys. 2013, 4, 137–178. [Google Scholar] [CrossRef]
- Gracia, J.; Sharpe, R.; Munarriz, J. Principles determining the activity of magnetic oxides for electron transfer reactions. J. Catal. 2018, 361, 331–338. [Google Scholar] [CrossRef]
- Munarriz, J.; Polo, V.; Gracia, J. On the Role of Ferromagnetic Interactions in Highly Active Mo-Based Catalysts for Ammonia Synthesis. ChemPhysChem 2018, 19, 2843–2847. [Google Scholar] [CrossRef]
- Lim, T.; Niemantsverdriet, J.W.; Gracia, J. Layered Antiferromagnetic Ordering in the Most Active Perovskite Catalysts for the Oxygen Evolution Reaction. ChemCatChem 2016, 8, 2968–2974. [Google Scholar] [CrossRef]
- Liu, Q.; Xu, L.; Wang, R.; Gong, J.; Tang, Y.; Liu, X. Kinetic Atomic-Scale Reproducibility of the Oxygen Reduction Reaction Process and a Newly Suggested Strong Correlation Descriptor: A Case Study of BaCo0.75Fe0.25O3−δ. J. Phys. Chem. Lett. 2022, 13, 4227–4234. [Google Scholar] [CrossRef]
- Zhou, G.; Wang, P.; Hu, B.; Shen, X.; Liu, C.; Tao, W.; Huang, P.; Liu, L. Spin-related symmetry breaking induced by half-disordered hybridization in BixEr2-xRu2O7 pyrochlores for acidic oxygen evolution. Nat. Commun. 2022, 13, 4106. [Google Scholar] [CrossRef]
- Yamada, I.; Toda, F.; Kawaguchi, S.; Yagi, S. Multiple Factors on Catalytic Activity for Oxygen Evolution Reaction in Magnetoplumbite Fe–Co Oxide BaFe12–xCoxO19. ACS Appl. Energy Mater. 2022, 5, 5995–6002. [Google Scholar] [CrossRef]
- Lin, C.-C.; Liu, T.-R.; Lin, S.-R.; Boopathi, K.M.; Chiang, C.-H.; Tzeng, W.-Y.; Chien, W.-H.C.; Hsu, H.-S.; Luo, C.-W.; Tsai, H.-Y.; et al. Spin-Polarized Photocatalytic CO2 Reduction of Mn-Doped Perovskite Nanoplates. J. Am. Chem. Soc. 2022, 144, 15718–15726. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Tian, H.; Ma, L.; Wang, Y.; Liu, X.; Gao, X. Low-temperature water electrolysis: Fundamentals, progress, and new strategies. Mater. Adv. 2022, 3, 5598–5644. [Google Scholar] [CrossRef]
- Minaev, B.F.; Ågren, H. Spin-catalysis phenomena. Int. J. Quantum Chem. 1996, 57, 519–532. [Google Scholar] [CrossRef]
- Usharani, D.; Janardanan, D.; Li, C.; Shaik, S. A Theory for Bioinorganic Chemical Reactivity of Oxometal Complexes and Analogous Oxidants: The Exchange and Orbital-Selection Rules. Acc. Chem. Res. 2013, 46, 471–482. [Google Scholar] [CrossRef]
- Shaik, S.; Chen, H.; Janardanan, D. Exchange-enhanced reactivity in bond activation by metal–oxo enzymes and synthetic reagents. Nat. Chem. 2011, 3, 19–27. [Google Scholar] [CrossRef]
- Voorhoeve, R.J.H. Experimental Relationships between Catalysis and Magnetism. AIP Conf. Proc. 1974, 18, 19–32. [Google Scholar] [CrossRef]
- Cohn, G.; Hedvall, J.A. The influence of the transition from ferromagnetism to paramagnetism on catalytic activity. J. Phys. Chem. 1942, 46, 841–847. [Google Scholar] [CrossRef]
- Hedvall, J.A. Changes in Crystal Structure and Their Influence on the Reactivity and Catalytic Effect of Solids. Chem. Rev. 1934, 15, 139–168. [Google Scholar] [CrossRef]
- Lielmezs, J.; Morgan, J.P. Magneto-catalytic effect in ethylene hydrogenation reaction. Chem. Eng. Sci. 1967, 22, 781–791. [Google Scholar] [CrossRef]
- Zeiger, H.J.; Wasserman, B.; Dresselhaus, M.S.; Dresselhaus, G. Rate of ethylene hydrogenation on Ni1−xCux catalysts-effect of magnetic ordering. Surf. Sci. 1983, 124, 583–590. [Google Scholar] [CrossRef]
- Modestov, A.D.; Kazarinov, V.E. Electrochemical reduction of 2,6-diphenylpyrylium cations by spin-polarized electrons. The Hedvall effect. Chem. Phys. Lett. 1993, 206, 401–404. [Google Scholar] [CrossRef]
- Yamaguchi, S. Measurement of Magnetic Gradient on the Surface of Catalyst. Oyo Buturi 1959, 28, 726–727. [Google Scholar] [CrossRef]
- Mehta, R.S.; Dresselhaus, M.S.; Dresselhaus, G.; Zeiger, H.J. Magnetic Phase Dependence of the Nickel-CO Reaction. Phys. Rev. Lett. 1979, 43, 970–973. [Google Scholar] [CrossRef]
- Cardwell, A.B. Photoelectric and Thermionic Properties of Nickel. Phys. Rev. 1949, 76, 125–127. [Google Scholar] [CrossRef]
- Deng, D.; Novoselov, K.S.; Fu, Q.; Zheng, N.; Tian, Z.; Bao, X. Catalysis with two-dimensional materials and their heterostructures. Nat. Nanotechnol. 2016, 11, 218–230. [Google Scholar] [CrossRef]
- Tan, C.; Cao, X.; Wu, X.-J.; He, Q.; Yang, J.; Zhang, X.; Chen, J.; Zhao, W.; Han, S.; Nam, G.-H.; et al. Recent Advances in Ultrathin Two-Dimensional Nanomaterials. Chem. Rev. 2017, 117, 6225–6331. [Google Scholar] [CrossRef] [PubMed]
- Lin, L. Synthesis, characterization and application of monatomic catalysts. IOP Conf. Ser. Mater. Sci. Eng. 2018, 392, 032026. [Google Scholar] [CrossRef]
- Itoh, T.; Uebayashi, M.; Tohji, K.; Jeyadevan, B. Inhibition of the Dissolution of Fe from Fe-Pt Nano Particles by a Structural Phase Transitions. Electrochemistry 2010, 78, 157–160. [Google Scholar] [CrossRef]
- Lebedeva, M.V.; Pierron-Bohnes, V.; Goyhenex, C.; Papaefthimiou, V.; Zafeiratos, S.; Nazmutdinov, R.R.; Da Costa, V.; Acosta, M.; Zosiak, L.; Kozubski, R.; et al. Effect of the chemical order on the electrocatalytic activity of model PtCo electrodes in the oxygen reduction reaction. Electrochim. Acta 2013, 108, 605–616. [Google Scholar] [CrossRef]
- Kim, J.; Lee, Y.; Sun, S. Structurally Ordered FePt Nanoparticles and Their Enhanced Catalysis for Oxygen Reduction Reaction. J. Am. Chem. Soc. 2010, 132, 4996–4997. [Google Scholar] [CrossRef]
- Li, X.; An, L.; Wang, X.; Li, F.; Zou, R.; Xia, D. Supported sub-5nm Pt–Fe intermetallic compounds for electrocatalytic application. J. Mater. Chem. 2012, 22, 6047–6052. [Google Scholar] [CrossRef]
- Zeng, J.; Liao, S.; Lee, J.Y.; Liang, Z. Oxygen reduction reaction operated on magnetically-modified PtFe/C electrocatalyst. Int. J. Hydrogen Energy 2010, 35, 942–948. [Google Scholar] [CrossRef]
- Leslie-Pelecky, D.L.; Rieke, R.D. Magnetic Properties of Nanostructured Materials. Chem. Mater. 1996, 8, 1770–1783. [Google Scholar] [CrossRef]
- Poole, C., Jr. Encyclopedic Dictionary of Condensed Matter Physics, 1st ed.; Academic Press: Cambridge, MA, USA, 2004. [Google Scholar]
- Nguyen, H.L.; Howard, L.E.M.; Giblin, S.R.; Tanner, B.K.; Terry, I.; Hughes, A.K.; Ross, I.M.; Serres, A.; Bürckstümmer, H.; Evans, J.S.O. Synthesis of monodispersed fcc and fct FePt/FePd nanoparticles by microwave irradiation. J. Mater. Chem. 2005, 15, 5136–5143. [Google Scholar] [CrossRef]
- Medwal, R.; Sehdev, N.; Annapoorni, S. Temperature-dependent magnetic and structural ordering of self-assembled magnetic array of FePt nanoparticles. J. Nanoparticle Res. 2013, 15, 1423. [Google Scholar] [CrossRef]
- Sun, S.; Murray, C.B.; Weller, D.; Folks, L.; Moser, A. Monodisperse FePt Nanoparticles and Ferromagnetic FePt Nanocrystal Superlattices. Science 2000, 287, 1989–1992. [Google Scholar] [CrossRef] [PubMed]
- Rong, C.B.; Li, D.; Nandwana, V.; Poudyal, N.; Ding, Y.; Wang, Z.L.; Zeng, H.; Liu, J.P. Size-Dependent Chemical and Magnetic Ordering in L10-FePt Nanoparticles. Adv. Mater. 2006, 18, 2984–2988. [Google Scholar] [CrossRef]
- Srivastava, S.; Gajbhiye, N.S. Exchange coupled L10-FePt/fcc-FePt nanomagnets: Synthesis, characterization and magnetic properties. J. Magn. Magn. Mater. 2016, 401, 969–976. [Google Scholar] [CrossRef]
- Yano, K.; Nandwana, V.; Poudyal, N.; Rong, C.-B.; Liu, J.P. Rapid thermal annealing of FePt nanoparticles. J. Appl. Phys. 2008, 104, 013918. [Google Scholar] [CrossRef]
- Chinnasamy, C.N.; Jeyadevan, B.; Shinoda, K.; Tohji, K. Polyol-process-derived CoPt nanoparticles: Structural and magnetic properties. J. Appl. Phys. 2003, 93, 7583–7585. [Google Scholar] [CrossRef]
- Park, J.-I.; Cheon, J. Synthesis of “Solid Solution” and “Core-Shell” Type Cobalt−Platinum Magnetic Nanoparticles via Transmetalation Reactions. J. Am. Chem. Soc. 2001, 123, 5743–5746. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Tung, L.D.; Stokes, K.L.; He, J.; Caruntu, D.; Zhou, W.L.; O’Connor, C.J. Synthesis and magnetic properties of CoPt–poly(methylmethacrylate) nanostructured composite material. J. Appl. Phys. 2002, 91, 8816–8818. [Google Scholar] [CrossRef]
- Bigot, J.-Y.; Kesserwan, H.; Halté, V.; Ersen, O.; Moldovan, M.S.; Kim, T.H.; Jang, J.-T.; Cheon, J. Magnetic Properties of Annealed Core–Shell CoPt Nanoparticles. Nano Lett. 2012, 12, 1189–1197. [Google Scholar] [CrossRef] [PubMed]
- Ge, J.; Chen, R.R.; Ren, X.; Liu, J.; Ong, S.J.H.; Xu, Z.J. Ferromagnetic–Antiferromagnetic Coupling Core–Shell Nanoparticles with Spin Conservation for Water Oxidation. Adv. Mater. 2021, 33, 2101091. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Ren, X.; Sun, S.; Liu, Z.; Xi, S.; Xu, Z.J. Engineering High-Spin State Cobalt Cations in Spinel Zinc Cobalt Oxide for Spin Channel Propagation and Active Site Enhancement in Water Oxidation. Angew. Chem. Int. Ed. 2021, 60, 14536–14544. [Google Scholar] [CrossRef]
- Jarju, J.J.; Díez, A.M.; Frey, L.; Sousa, V.; Carbó-Argibay, E.; Gonçalves, L.P.L.; Medina, D.D.; Lebedev, O.I.; Kolen’ko, Y.V.; Salonen, L.M. Synthetic strategy for metallophthalocyanine covalent organic frameworks for electrochemical water oxidation. Mater. Today Chem. 2022, 26, 101032. [Google Scholar] [CrossRef]
- Delprat, S.; Galbiati, M.; Tatay, S.; Quinard, B.; Barraud, C.; Petroff, F.; Seneor, P.; Mattana, R. Molecular spintronics: The role of spin-dependent hybridization. J. Phys. D Appl. Phys. 2018, 51, 473001. [Google Scholar] [CrossRef]
- Wolf, S.A.; Awschalom, D.D.; Buhrman, R.A.; Daughton, J.M.; von Molnár, S.; Roukes, M.L.; Chtchelkanova, A.Y.; Treger, D.M. Spintronics: A Spin-Based Electronics Vision for the Future. Science 2001, 294, 1488–1495. [Google Scholar] [CrossRef]
- Ivanov, K.L.; Wagenpfahl, A.; Deibel, C.; Matysik, J. Spin-chemistry concepts for spintronics scientists. Beilstein J. Nanotechnol. 2017, 8, 1427–1445. [Google Scholar] [CrossRef]
- Fert, A.; Campbell, I.A. Two-Current Conduction in Nickel. Phys. Rev. Lett. 1968, 21, 1190–1192. [Google Scholar] [CrossRef]
- Fert, A.; Campbell, I.A. Electrical resistivity of ferromagnetic nickel and iron based alloys. J. Phys. F Met. Phys. 1976, 6, 849–871. [Google Scholar] [CrossRef]
- Chappert, C.; Fert, A.; Van Dau, F.N. The emergence of spin electronics in data storage. Nat. Mater. 2007, 6, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Morris, H.; Selwood, P.W. Magnetic Measurements on Some Catalytically Active Substances. J. Am. Chem. Soc. 1943, 65, 2245–2252. [Google Scholar] [CrossRef]
- Shovkovy, I.A. Magnetic Catalysis: A Review. In Strongly Interacting Matter in Magnetic Fields; Kharzeev, D., Landsteiner, K., Schmitt, A., Yee, H.-U., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 13–49. [Google Scholar]
- Chen, H.-L.; Fukushima, K.; Huang, X.-G.; Mameda, K. Surface magnetic catalysis. Phys. Rev. D 2017, 96, 054032. [Google Scholar] [CrossRef]
- Atkins, P.W. The Effects of Magnetic Fields on Chemical Reactions. In Chemically Induced Magnetic Polarization; Springer: Dordrecht, The Netherlands, 1977. [Google Scholar]
- Rodgers, C.T. Magnetic field effects in chemical systems. Pure Appl. Chem. 2009, 81, 19–43. [Google Scholar] [CrossRef]
- Steiner, U.E.; Ulrich, T. Magnetic field effects in chemical kinetics and related phenomena. Chem. Rev. 1989, 89, 51–147. [Google Scholar] [CrossRef]
- Ilisca, E. Ortho-para conversion of hydrogen molecules physisorbed on surfaces. Prog. Surf. Sci. 1992, 41, 217–335. [Google Scholar] [CrossRef]
- Ilisca, E.; Houssais, L.; Ghiglieno, F. Symmetry Breakings in the interactions of Molecular Hydrogen with Solids. EPJ Web Conf. 2022, 263, 01013. [Google Scholar] [CrossRef]
- Taylor, H.S.; Diamond, H. The Para-Hydrogen Conversion at Paramagnetic Surfaces. J. Am. Chem. Soc. 1933, 55, 2613–2614. [Google Scholar] [CrossRef]
- Misono, M.; Selwood, P.W. Extrinsic field acceleration of the magnetic parahydrogen conversion. J. Am. Chem. Soc. 1969, 91, 1300–1303. [Google Scholar] [CrossRef]
- Selwood, P.W. The Effect of a Magnetic Field on the Catalyzed Nondissociative Para hydrogen Conversion Rate. In Advances in Catalysis; Eley, D.D., Pines, H., Weisz, P.B., Eds.; Academic Press: Cambridge, MA, USA, 1979; pp. 23–57. [Google Scholar]
- Selwood, P.W. The effect of a weak magnetic field on the rare earth catalyzed parahydrogen conversion rate. J. Catal. 1971, 22, 123–129. [Google Scholar] [CrossRef]
- Justi, E.; Vieth, G. Über den Einfluß äußerer Magnetfelder auf die Aktivität ferromagnetischer Katalysatoren. Z. Für Nat. A 1953, 8, 538–546. [Google Scholar] [CrossRef]
- Zhang, X.; Karman, T.; Groenenboom, G.C.; van der Avoird, A. Para-ortho hydrogen conversion: Solving a 90-year old mystery. Nat. Sci. 2021, 1, e10002. [Google Scholar] [CrossRef]
- Ilisca, E.; Gallais, E. Orientation Effect of the Surface Magnetization of Ferromagnetic Catalysts on Ortho-Para Hydrogen Conversion. Phys. Rev. B 1972, 6, 2858–2859. [Google Scholar] [CrossRef]
- Ilisca, E.; Debauche, M.; Motchane, J.L. Quantum formulation of a magneto-catalytic reaction. Phys. Rev. B 1980, 22, 687–701. [Google Scholar] [CrossRef]
- Petzinger, K.G.; Scalapino, D.J. Para- to Ortho-Hydrogen Conversion on Magnetic Surfaces. Phys. Rev. B 1973, 8, 266–279. [Google Scholar] [CrossRef]
- Selwood, P.W. Effect of Low Magnetic Fields on the Catalysed Parahydrogen Conversion Rate over Certain Rare Earths. Nature 1970, 228, 278. [Google Scholar] [CrossRef]
- Luo, S.; Elouarzaki, K.; Xu, Z.J. Electrochemistry in Magnetic Fields. Angew. Chem. Int. Ed. 2022, 61, e202203564. [Google Scholar] [CrossRef]
- Gatard, V.; Deseure, J.; Chatenet, M. Use of magnetic fields in electrochemistry: A selected review. Curr. Opin. Electrochem. 2020, 23, 96–105. [Google Scholar] [CrossRef]
- Coey, J.M.D. Magnetoelectrochemistry. Europhys. News 2003, 34, 246–248. [Google Scholar] [CrossRef]
- Alemany, A.; Chopart, J.-P. An Outline of Magnetoelectrochemistry. In Magnetohydrodynamics: Historical Evolution and Trends; Molokov, S., Moreau, R., Moffatt, K., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 391–407. [Google Scholar]
- Monzon, L.M.A.; Coey, J.M.D. Magnetic fields in electrochemistry: The Lorentz force. A mini-review. Electrochem. Commun. 2014, 42, 38–41. [Google Scholar] [CrossRef]
- Monzon, L.M.A.; Coey, J.M.D. Magnetic fields in electrochemistry: The Kelvin force. A mini-review. Electrochem. Commun. 2014, 42, 42–45. [Google Scholar] [CrossRef]
- Coey, J.M.D.; Rhen, F.M.F.; Dunne, P.; McMurry, S. The magnetic concentration gradient force—Is it real? J. Solid State Electrochem. 2007, 11, 711–717. [Google Scholar] [CrossRef]
- Fahidy, T.Z. Hydrodynamic models in magnetoelectrolysis. Electrochim. Acta 1973, 18, 607–614. [Google Scholar] [CrossRef]
- Waskaas, M.; Kharkats, Y.I. Magnetoconvection Phenomena: A Mechanism for Influence of Magnetic Fields on Electrochemical Processes. J. Phys. Chem. B 1999, 103, 4876–4883. [Google Scholar] [CrossRef]
- Pullins, M.D.; Grant, K.M.; White, H.S. Microscale Confinement of Paramagnetic Molecules in Magnetic Field Gradients Surrounding Ferromagnetic Microelectrodes. J. Phys. Chem. B 2001, 105, 8989–8994. [Google Scholar] [CrossRef]
- Hinds, G.; Coey, J.M.D.; Lyons, M.E.G. Influence of magnetic forces on electrochemical mass transport. Electrochem. Commun. 2001, 3, 215–218. [Google Scholar] [CrossRef]
- Tacken, R.A.; Janssen, L.J.J. Applications of magnetoelectrolysis. J. Appl. Electrochem. 1995, 25, 1–5. [Google Scholar] [CrossRef]
- Kołodziejczyk, K.; Miękoś, E.; Zieliński, M.; Jaksender, M.; Szczukocki, D.; Czarny, K.; Krawczyk, B. Influence of constant magnetic field on electrodeposition of metals, alloys, conductive polymers, and organic reactions. J. Solid State Electrochem. 2018, 22, 1629–1647. [Google Scholar] [CrossRef]
- Zhang, L.; Wu, D.; Yan, X. Applications of magnetic field for electrochemical energy storage. Appl. Phys. Rev. 2022, 9, 031307. [Google Scholar] [CrossRef]
- Huan, Y.; Zhijia, Z.; Zhenyang, Y.; Mengmeng, Z.; Yifang, Z.; Yong, J. Research Progress of Spin-Dependent Effects in Catalysis and Energy Storage. Mater. Lab 2022, 1, 220016. [Google Scholar] [CrossRef]
- Yan, J.; Wang, Y.; Zhang, Y.; Xia, S.; Yu, J.; Ding, B. Direct Magnetic Reinforcement of Electrocatalytic ORR/OER with Electromagnetic Induction of Magnetic Catalysts. Adv. Mater. 2021, 33, 2007525. [Google Scholar] [CrossRef] [PubMed]
- Westsson, E.; Picken, S.; Koper, G. The Effect of Magnetic Field on Catalytic Properties in Core-Shell Type Particles. Front. Chem. 2020, 8, 163. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Zhang, T.; Liu, Y.; Zhang, W.; Yin, Z.; Ji, Z.; Wei, J. Magnetic Field-Enhanced 4-Electron Pathway for Well-Aligned Co3O4/Electrospun Carbon Nanofibers in the Oxygen Reduction Reaction. ChemSusChem 2018, 11, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Tufa, L.T.; Jeong, K.-J.; Tran, V.T.; Lee, J. Magnetic-Field-Induced Electrochemical Performance of a Porous Magnetoplasmonic Ag@Fe3O4 Nanoassembly. ACS Appl. Mater. Interfaces 2020, 12, 6598–6606. [Google Scholar] [CrossRef]
- Monzon, L.M.A.; Rode, K.; Venkatesan, M.; Coey, J.M.D. Electrosynthesis of Iron, Cobalt, and Zinc Microcrystals and Magnetic Enhancement of the Oxygen Reduction Reaction. Chem. Mater. 2012, 24, 3878–3885. [Google Scholar] [CrossRef]
- Wu, T.; Ren, X.; Sun, Y.; Sun, S.; Xian, G.; Scherer, G.G.; Fisher, A.C.; Mandler, D.; Ager, J.W.; Grimaud, A.; et al. Spin pinning effect to reconstructed oxyhydroxide layer on ferromagnetic oxides for enhanced water oxidation. Nat. Commun. 2021, 12, 3634. [Google Scholar] [CrossRef]
- Ren, X.; Wu, T.; Sun, Y.; Li, Y.; Xian, G.; Liu, X.; Shen, C.; Gracia, J.; Gao, H.-J.; Yang, H.; et al. Spin-polarized oxygen evolution reaction under magnetic field. Nat. Commun. 2021, 12, 2608. [Google Scholar] [CrossRef]
- Hunt, C.; Zhang, Z.; Ocean, K.; Jansonius, R.P.; Abbas, M.; Dvorak, D.J.; Kurimoto, A.; Lees, E.W.; Ghosh, S.; Turkiewicz, A.; et al. Quantification of the Effect of an External Magnetic Field on Water Oxidation with Cobalt Oxide Anodes. J. Am. Chem. Soc. 2022, 144, 733–739. [Google Scholar] [CrossRef]
- Kim, D.; Efe, I.; Torlakcik, H.; Terzopoulou, A.; Veciana, A.; Siringil, E.; Mushtaq, F.; Franco, C.; von Arx, D.; Sevim, S.; et al. Magnetoelectric Effect in Hydrogen Harvesting: Magnetic Field as a Trigger of Catalytic Reactions. Adv. Mater. 2022, 34, 2110612. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Chen, M.; Guo, M.; Hong, A.; Yu, T.; Luo, X.; Yuan, C.; Lei, W.; Wang, S. Magnetic Enhancement for Hydrogen Evolution Reaction on Ferromagnetic MoS2 Catalyst. Nano Lett. 2020, 20, 2923–2930. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Yang, Q.; Manna, K.; Mu, Q.; Fu, C.; Sun, Y.; Felser, C. Magnetocatalysis: The Interplay between the Magnetic Field and Electrocatalysis. CCS Chem. 2021, 3, 2259–2267. [Google Scholar] [CrossRef]
- Zieliñski, M. Positive and Negative Aspects of Electrode Reactions of Hydrogen Evolution and the Influence of a Constant Magnetic Field. J. Adv. Chem. Eng. 2014, 4, 2. [Google Scholar] [CrossRef]
- Yu, L.; Wang, J.; Xu, Z.J. A Perspective on the Behavior of Lithium Anodes under a Magnetic Field. Small Struct. 2021, 2, 2000043. [Google Scholar] [CrossRef]
- Shen, K.; Xu, X.; Tang, Y. Recent progress of magnetic field application in lithium-based batteries. Nano Energy 2022, 92, 106703. [Google Scholar] [CrossRef]
- Chaure, N.B.; Coey, J.M.D. Enhanced Oxygen Reduction at Composite Electrodes Producing a Large Magnetic Gradient. J. Electrochem. Soc. 2009, 156, F39. [Google Scholar] [CrossRef]
- Chaure, N.B.; Rhen, F.M.F.; Hilton, J.; Coey, J.M.D. Design and application of a magnetic field gradient electrode. Electrochem. Commun. 2007, 9, 155–158. [Google Scholar] [CrossRef]
- Lu, F.; Wang, J.; Li, J.; Du, Y.; Kong, X.-P.; Liu, S.; Yi, D.; Takahashi, Y.K.; Hono, K.; Wang, X.; et al. Regulation of oxygen reduction reaction by the magnetic effect of L10-PtFe alloy. Appl. Catal. B Environ. 2020, 278, 119332. [Google Scholar] [CrossRef]
- Wang, L.; Yang, H.; Yang, J.; Yang, Y.; Wang, R.; Li, S.; Wang, H.; Ji, S. The effect of the internal magnetism of ferromagnetic catalysts on their catalytic activity toward oxygen reduction reaction under an external magnetic field. Ionics 2016, 22, 2195–2202. [Google Scholar] [CrossRef]
- Okada, T.; Wakayama, N.I.; Wang, L.; Shingu, H.; Okano, J.-I.; Ozawa, T. The effect of magnetic field on the oxygen reduction reaction and its application in polymer electrolyte fuel cells. Electrochim. Acta 2003, 48, 531–539. [Google Scholar] [CrossRef]
- Zakrzewska, B.; Adamczyk, L.; Marcinek, M.; Miecznikowski, K. The Effect of an External Magnetic Field on the Electrocatalytic Activity of Heat-Treated Cyanometallate Complexes towards the Oxygen Reduction Reaction in an Alkaline Medium. Materials 2022, 15, 1418. [Google Scholar] [CrossRef] [PubMed]
- Kiciński, W.; Sęk, J.P.; Matysiak-Brynda, E.; Miecznikowski, K.; Donten, M.; Budner, B.; Nowicka, A.M. Enhancement of PGM-free oxygen reduction electrocatalyst performance for conventional and enzymatic fuel cells: The influence of an external magnetic field. Appl. Catal. B Environ. 2019, 258, 117955. [Google Scholar] [CrossRef]
- Shi, J.; Xu, H.; Zhao, H.; Lu, L.; Wu, X. Preparation of Nd2Fe14B/C magnetic powder and its application in proton exchange membrane fuel cells. J. Power Sources 2014, 252, 189–199. [Google Scholar] [CrossRef]
- IEA. Net Zero by 2050. Available online: https://www.iea.org/reports/net-zero-by-2050 (accessed on 29 August 2022).
- Dehghani-Sanij, A.R.; Tharumalingam, E.; Dusseault, M.B.; Fraser, R. Study of energy storage systems and environmental challenges of batteries. Renew. Sustain. Energy Rev. 2019, 104, 192–208. [Google Scholar] [CrossRef]
- Available online: https://ec.europa.eu/docsroom/documents/37824 (accessed on 8 March 2021).
- Fathabadi, H. Fuel cell hybrid electric vehicle (FCHEV): Novel fuel cell/SC hybrid power generation system. Energy Convers. Manag. 2018, 156, 192–201. [Google Scholar] [CrossRef]
- IEA. Hydrogen. Available online: https://www.iea.org/reports/hydrogen (accessed on 29 August 2022).
- Yonoff, R.E.; Ochoa, G.V.; Cardenas-Escorcia, Y.; Silva-Ortega, J.I.; Meriño-Stand, L. Research trends in proton exchange membrane fuel cells during 2008–2018: A bibliometric analysis. Heliyon 2019, 5, e01724. [Google Scholar] [CrossRef]
- Kulikovsky, A.A. Analytical Modeling of Fuel Cells; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- O’Hayre, R.; Cha, S.-W.; Colella, W.G.; Prinz, F.B. Fuel Cell Fundamentals, 3rd ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016. [Google Scholar]
- Sazali, N.; Wan Salleh, W.N.; Jamaludin, A.S.; Mhd Razali, M.N. New Perspectives on Fuel Cell Technology: A Brief Review. Membranes 2020, 10, 99. [Google Scholar] [CrossRef]
- Parekh, A. Recent developments of proton exchange membranes for PEMFC: A review. Front. Energy Res. 2022, 10, 01. [Google Scholar] [CrossRef]
- Tawalbeh, M.; Alarab, S.; Al-Othman, A.; Javed, R.M. The Operating Parameters, Structural Composition, and Fuel Sustainability Aspects of PEM Fuel Cells: A Mini Review. Fuels 2022, 3, 449–474. [Google Scholar] [CrossRef]
- Shukla, A.K.; Suresh, P.; Berchmans, S.; Rajendran, A. Biological Fuel Cells and Their Applications. Cur. Sci. 2004, 87, 455–468. [Google Scholar]
- Matsushima, H.; Iida, T.; Fukunaka, Y.; Bund, A. PEMFC Performance in a Magnetic Field. Fuel Cells 2008, 8, 33–36. [Google Scholar] [CrossRef]
- Hermann, A.; Chaudhuri, T.; Spagnol, P. Bipolar plates for PEM fuel cells: A review. Int. J. Hydrogen Energy 2005, 30, 1297–1302. [Google Scholar] [CrossRef]
- Pourrahmani, H.; Moghimi, M.; Siavashi, M. Thermal management in PEMFCs: The respective effects of porous media in the gas flow channel. Int. J. Hydrogen Energy 2019, 44, 3121–3137. [Google Scholar] [CrossRef]
- Saadat, N.; Dhakal, H.N.; Tjong, J.; Jaffer, S.; Yang, W.; Sain, M. Recent advances and future perspectives of carbon materials for fuel cell. Renew. Sustain. Energy Rev. 2021, 138, 110535. [Google Scholar] [CrossRef]
- Ogungbemi, E.; Wilberforce, T.; Ijaodola, O.; Thompson, J.; Olabi, A.G. Review of operating condition, design parameters and material properties for proton exchange membrane fuel cells. Int. J. Energy Res. 2021, 45, 1227–1245. [Google Scholar] [CrossRef]
- Wang, H.; Wang, R.; Sui, S.; Sun, T.; Yan, Y.; Du, S. Cathode Design for Proton Exchange Membrane Fuel Cells in Automotive Applications. Automot. Innov. 2021, 4, 144–164. [Google Scholar] [CrossRef]
- Santangelo, P.E.; Cannio, M.; Romagnoli, M. Review of catalyst-deposition techniques for PEMFC electrodes. TECNICA ITALIANA-Ital. J. Eng. Sci. 2019, 63, 65–72. [Google Scholar] [CrossRef]
- Deng, X.; Zhang, J.; Fan, Z.; Tan, W.; Yang, G.; Wang, W.; Zhou, W.; Shao, Z. Understanding and Engineering of Multiphase Transport Processes in Membrane Electrode Assembly of Proton-Exchange Membrane Fuel Cells with a Focus on the Cathode Catalyst Layer: A Review. Energy Fuels 2020, 34, 9175–9188. [Google Scholar] [CrossRef]
- Jiao, K.; Xuan, J.; Du, Q.; Bao, Z.; Xie, B.; Wang, B.; Zhao, Y.; Fan, L.; Wang, H.; Hou, Z.; et al. Designing the next generation of proton-exchange membrane fuel cells. Nature 2021, 595, 361–369. [Google Scholar] [CrossRef]
- Majlan, E.H.; Rohendi, D.; Daud, W.R.W.; Husaini, T.; Haque, M.A. Electrode for proton exchange membrane fuel cells: A review. Renew. Sustain. Energy Rev. 2018, 89, 117–134. [Google Scholar] [CrossRef]
- Sun, Y.; Polani, S.; Luo, F.; Ott, S.; Strasser, P.; Dionigi, F. Advancements in cathode catalyst and cathode layer design for proton exchange membrane fuel cells. Nat. Commun. 2021, 12, 5984. [Google Scholar] [CrossRef] [PubMed]
- Xie, M.; Chu, T.; Wang, T.; Wan, K.; Yang, D.; Li, B.; Ming, P.; Zhang, C. Preparation, Performance and Challenges of Catalyst Layer for Proton Exchange Membrane Fuel Cell. Membranes 2021, 11, 879. [Google Scholar] [CrossRef] [PubMed]
- Bevilacqua, N.; Asset, T.; Schmid, M.A.; Markötter, H.; Manke, I.; Atanassov, P.; Zeis, R. Impact of catalyst layer morphology on the operation of high temperature PEM fuel cells. J. Power Sources Adv. 2021, 7, 100042. [Google Scholar] [CrossRef]
- Guo, Y.; Pan, F.; Chen, W.; Ding, Z.; Yang, D.; Li, B.; Ming, P.; Zhang, C. The Controllable Design of Catalyst Inks to Enhance PEMFC Performance: A Review. Electrochem. Energy Rev. 2021, 4, 67–100. [Google Scholar] [CrossRef]
- Kobayashi, A.; Fujii, T.; Takeda, K.; Tamoto, K.; Kakinuma, K.; Uchida, M. Effect of Pt Loading Percentage on Carbon Blacks with Large Interior Nanopore Volume on the Performance and Durability of Polymer Electrolyte Fuel Cells. ACS Appl. Energy Mater. 2022, 5, 316–329. [Google Scholar] [CrossRef]
- Gummalla, M.; Ball, S.C.; David, A.C.; Rasouli, S.; Yu, K.; Ferreira, P.J.; Myers, D.J.; Yang, Z. Effect of Particle Size and Operating Conditions on Pt3Co, PEMFC Cathode Catalyst Durability. Catalysts 2015, 5, 926–948. [Google Scholar] [CrossRef]
- Marappan, M.; Palaniswamy, K.; Velumani, T.; Chul, K.B.; Velayutham, R.; Shivakumar, P.; Sundaram, S. Performance Studies of Proton Exchange Membrane Fuel Cells with Different Flow Field Designs—Review. Chem. Rec. 2021, 21, 663–714. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Mao, Y.; Zhang, W.; Tang, Y.; Wang, P. Reviews on the effects of contaminations and research methodologies for PEMFC. Int. J. Hydrogen Energy 2020, 45, 23174–23200. [Google Scholar] [CrossRef]
- Prince Abraham, B.; Kalidasa, M.K. Influence of Catalyst Layer and Gas Diffusion Layer Porosity in Proton Exchange Membrane Fuel Cell Performance. Electrochim. Acta 2021, 389, 138793. [Google Scholar] [CrossRef]
- Sanchez, D.G.; Ruiu, T.; Friedrich, K.A.; Sanchez-Monreal, J.; Vera, M. Analysis of the Influence of Temperature and Gas Humidity on the Performance Stability of Polymer Electrolyte Membrane Fuel Cells. J. Electrochem. Soc. 2015, 163, F150–F159. [Google Scholar] [CrossRef]
- Liu, X.; Li, Y.; Xue, J.; Zhu, W.; Zhang, J.; Yin, Y.; Qin, Y.; Jiao, K.; Du, Q.; Cheng, B.; et al. Magnetic field alignment of stable proton-conducting channels in an electrolyte membrane. Nat. Commun. 2019, 10, 842. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Rehim, A.A. The influence of electromagnetic field on the performance and operation of a PEM fuel cell stack sub-jected to a relatively low electromagnetic field. Energy Convers. Manag. 2019, 198, 111906. [Google Scholar] [CrossRef]
- Lang, X.; Wu, Z.; Li, R.; Wu, M. Study on the improvement of performance of proton exchange membrane Fuel Cell by Magnetic effect. J. Phys. Conf. Ser. 2022, 2263, 012003. [Google Scholar] [CrossRef]
- Ruksawong, K.; Songprakorp, R.; Monyakul, V.; David, N.A.; Sui, P.C.; Djilali, N. Investigation of PEMFC under Static Magnetic Field: Temperature, Relative Humidity and Performance. J. Electrochem. Soc. 2017, 164, F1. [Google Scholar] [CrossRef]
- Sun, X.; Xu, H.; Lu, L.; Xing, W.; Zhao, H. Preparing a catalyst layer in magnetic field to improve the performance of proton exchange membrane fuel cells. J. Appl. Electrochem. 2014, 44, 1179–1184. [Google Scholar] [CrossRef]
- Tang, J.; Li, H.; Zhang, Y.; Huang, F. Performance Study on Magnetic Field Inducing Perfluorosulfonate Ionomer Membranes for PEMFC. J. Electrochem. Soc. 2016, 163, F1605. [Google Scholar] [CrossRef]
- Liu, L.; Li, X.; Liu, Z.; Zhang, S.; Qian, L.; Chen, Z.; Li, J.; Fang, P.; He, C. High-performance fuel cells using Nafion composite membranes with alignment of sulfonated graphene oxides induced by a strong magnetic field. J. Membr. Sci. 2022, 653, 120516. [Google Scholar] [CrossRef]
- Hyun, J.; Doo, G.; Yuk, S.; Lee, D.-H.; Lee, D.W.; Choi, S.; Kwen, J.; Kang, H.; Tenne, R.; Lee, S.G.; et al. Magnetic Field-Induced Through-Plane Alignment of the Proton Highway in a Proton Exchange Membrane. ACS Appl. Energy Mater. 2020, 3, 4619–4628. [Google Scholar] [CrossRef]
- Baker, A.M.; Wang, L.; Advani, S.G.; Prasad, A.K. Nafion membranes reinforced with magnetically controlled Fe3O4–MWCNTs for PEMFCs. J. Mater. Chem. 2012, 22, 14008–14012. [Google Scholar] [CrossRef]
- Cai, J.; Wang, L.; Wu, P. Oxygen enrichment from air by using the interception effect of gradient magnetic field on oxygen molecules. Phys. Lett. A 2007, 362, 105–108. [Google Scholar] [CrossRef]
- Kwon, K.; Kang, H.; Kang, S.; Kim, D. Evaluation of reciprocating electromagnetic air pumping for portable PEMFC. J. Micromech. Microeng. 2013, 23, 065007. [Google Scholar] [CrossRef]
- Ma, H.; Hsu, Y.; Hsu, P. A Novel Hybrid Actuator Driven Magnetically in the Bi-Cell PEM Fuel Cell Stack. Metals 2017, 7, 453. [Google Scholar] [CrossRef]
- Liu, Z.; Sun, Y.; Mao, L.; Zhang, H.; Jackson, L.; Wu, Q.; Lu, S. Efficient fault diagnosis of proton exchange membrane fuel cell using external magnetic field measurement. Energy Convers. Manag. 2022, 266, 115809. [Google Scholar] [CrossRef]
- Sun, Y.; Mao, L.; He, K.; Liu, Z.; Lu, S. Imaging PEMFC performance heterogeneity by sensing external magnetic field. Cell Rep. Phys. Sci. 2022, 3, 101083. [Google Scholar] [CrossRef]
- Ifrek, L.; Rosini, S.; Cauffet, G.; Chadebec, O.; Rouveyre, L.; Bultel, Y. Fault detection for polymer electrolyte membrane fuel cell stack by external magnetic field. Electrochim. Acta 2019, 313, 141–150. [Google Scholar] [CrossRef]
- Hamaz, T.; Cadet, C.; Druart, F.; Cauffet, G. Diagnosis of PEM fuel cell stack based on magnetic fields measurements. IFAC Proc. Vol. 2014, 47, 11482–11487. [Google Scholar] [CrossRef]
- Le Ny, M.; Chadebec, O.; Cauffet, G.; Rosini, S.; Bultel, Y. PEMFC stack diagnosis based on external magnetic field measurements. J. Appl. Electrochem. 2015, 45, 667–677. [Google Scholar] [CrossRef]
- Akimoto, Y.; Izawa, Y.; Suzuki, S.-N.; Okajima, K. Experimental investigation of stable the proton exchange membrane fuel cell control using magnetic sensor probes. Fuel Cells 2022, 22, 2–11. [Google Scholar] [CrossRef]
- Sun, Y.; Mao, L.; Wang, H.; Liu, Z.; Lu, S. Simulation study on magnetic field distribution of PEMFC. Int. J. Hydrogen Energy 2022, 47, 33439–33452. [Google Scholar] [CrossRef]
- Plait, A.; Dubas, F. A 2D Multi-Layer Model to Study the External Magnetic Field Generated by a Polymer Exchange Membrane Fuel Cell. Mathematics 2022, 10, 3883. [Google Scholar] [CrossRef]
- Feng, S.; Chen, Q.; Han, X. Effect of Magnetic Field on Corrosion Behaviors of Gold-Coated Titanium as Cathode Plates for Proton Exchange Membrane Fuel Cells. J. Electrochem. Soc. 2022, 169, 091503. [Google Scholar] [CrossRef]
- Ručinskien, A.; Bikulčius, G.; Gudavičiūt, L.; Juzeliūnas, E. Magnetic field effect on stainless steel corrosion in FeCl3 solution. Electrochem. Commun. 2002, 4, 86–91. [Google Scholar] [CrossRef]
- Chiba, A.; Kawazu, K.; Nakano, O.; Tamura, T.; Yoshihara, S.; Sato, E. The effects of magnetic fields on the corrosion of aluminum foil in sodium chloride solutions. Corros. Sci. 1994, 36, 539–543. [Google Scholar] [CrossRef]
- Mitre, C.I.N.; Tosin, G.; Colnago, L.A. In-operando analysis of the corrosion patterns and rates under magnetic fields using metallic film. npj Mater. Degrad. 2022, 6, 24. [Google Scholar] [CrossRef]
- Nozières, J.P.; Taylor, D.W.; Bala, H.; Malik, M.; Szymura, S.; Stokłosa, H. Corrosion behaviour of hot-worked Nd-Fe-B and Nd-Fe-Cu-B permanent magnets. J. Alloys Compd. 1992, 186, 201–208. [Google Scholar] [CrossRef]
- Zheng, J.W.; Xia, Q.P.; Qiao, L.; Jiang, L.Q.; Li, J.; Mao, L.Y. Study on the Electrochemical Corrosion Behavior of Nd-Fe-B-Sintered Magnets in Different Remanence States. Corrosion 2008, 64, 800–804. [Google Scholar] [CrossRef]
- Costa, I.; Oliveira, M.C.L.; de Melo, H.G.; Faria, R.N. The effect of the magnetic field on the corrosion behavior of Nd–Fe–B permanent magnets. J. Magn. Magn. Mater. 2004, 278, 348–358. [Google Scholar] [CrossRef]
- Tu, C.-S.; Hsu, F.-T. Magnetic Fuel Cell-Based Battery. US20120268071A1, 25 October 2012. [Google Scholar]
- Marsacq, D.; Nayoze, C.; Roux, C.; Franco, A. Fuel Cell Comprising a Magnetic Cathode with Static Pumping. US7569289B2, 4 August 2009. [Google Scholar]
- Lersch, J.; Mattejat, A.; Strasser, K. Fuel Cell Module Comprising a Magnetic Shielding. US20040033411A1, 19 February 2004. [Google Scholar]
- Nitta, S.; Taki, M.; Kawahara, T.; Miura, M. Fuel-Cells System Having Means for Oxagen Enrichment of the Oxidant Gas. EP0878859A1, 18 November 1998. [Google Scholar]
- Yan, G.; Hao, T.; Zemin, Q.; Cong, Y.; Kai, L. Preparation Method of Fuel Cell Membrane Electrode Catalyst Layer. CN111740119B, 6 July 2021. [Google Scholar]

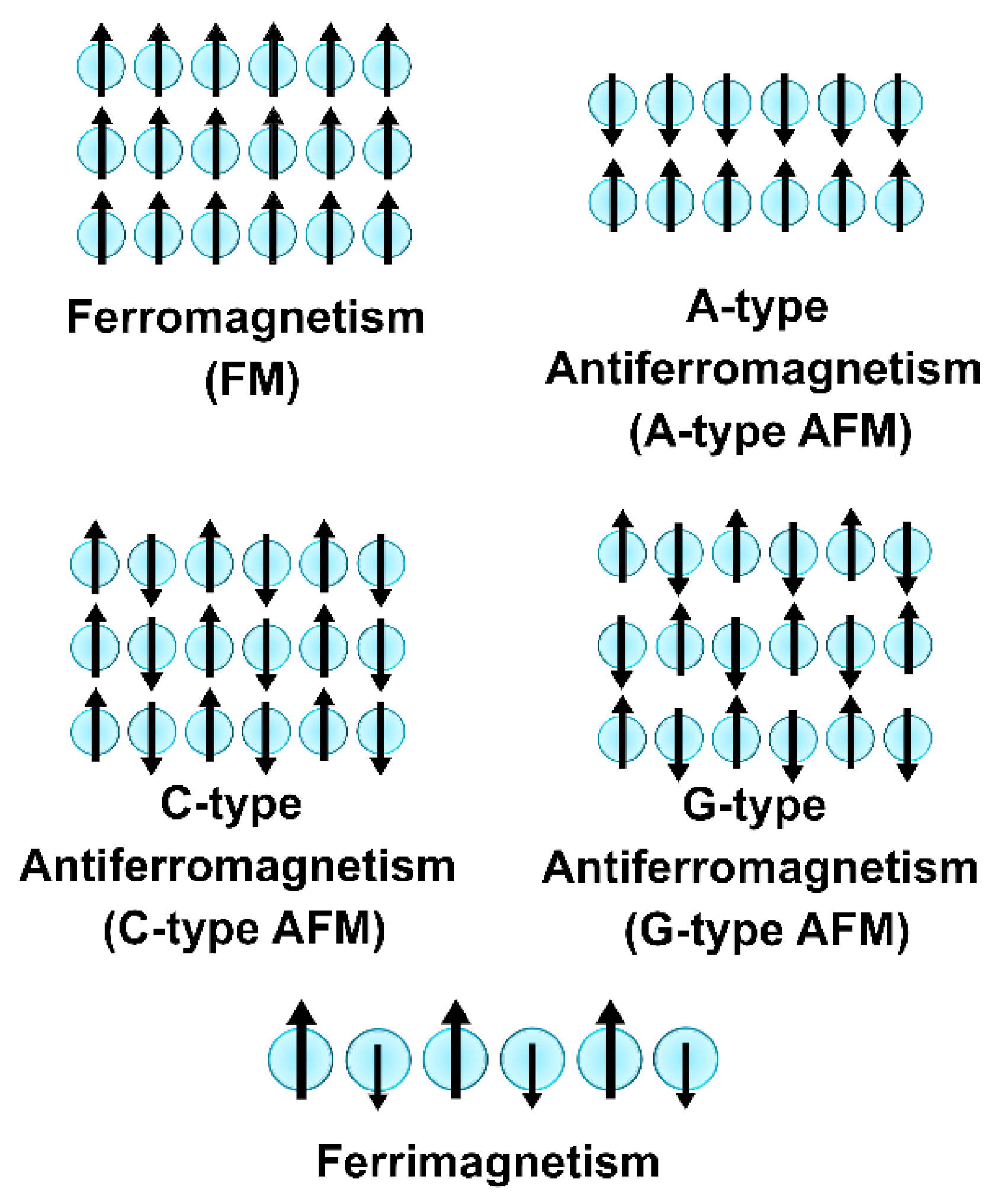
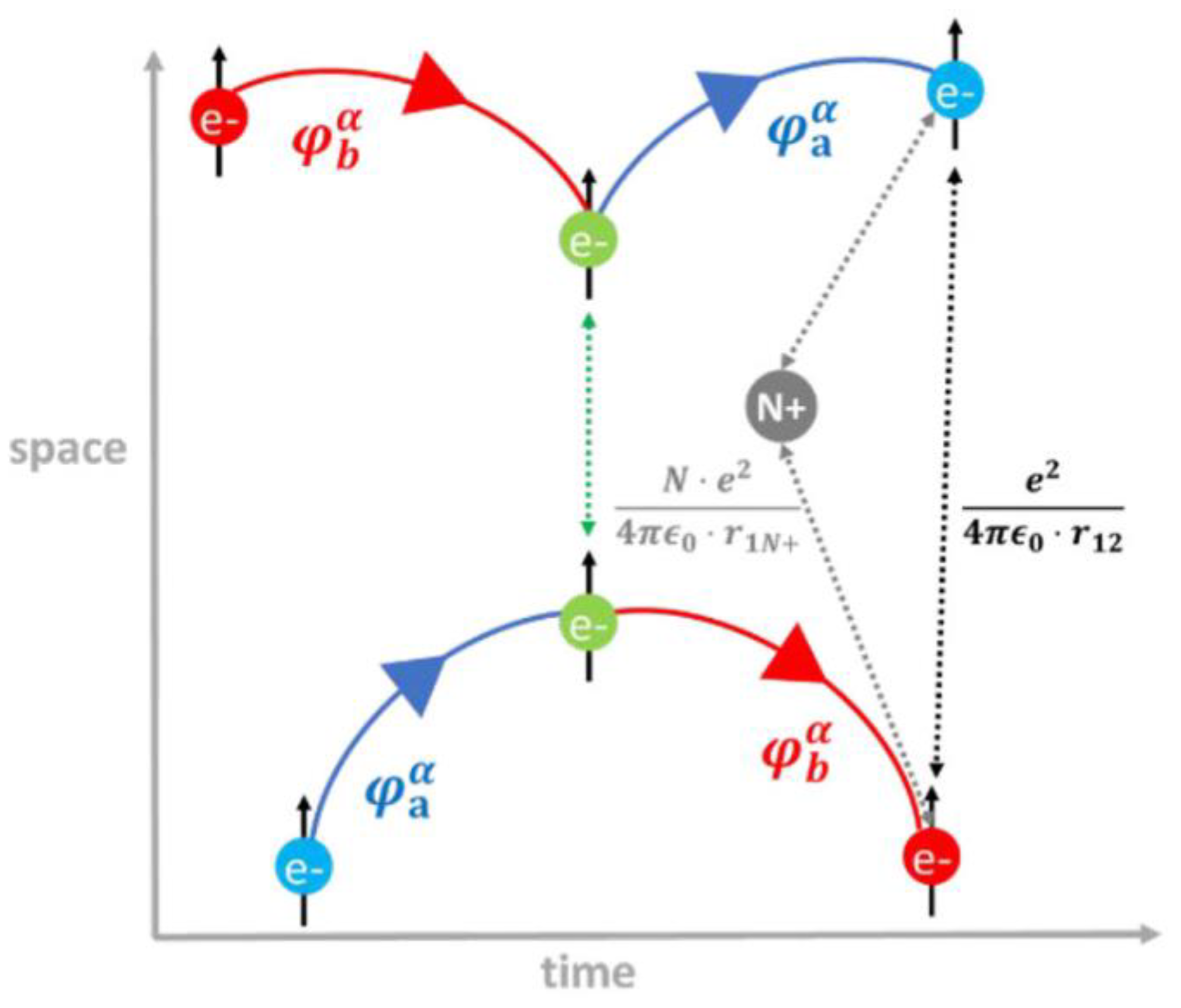

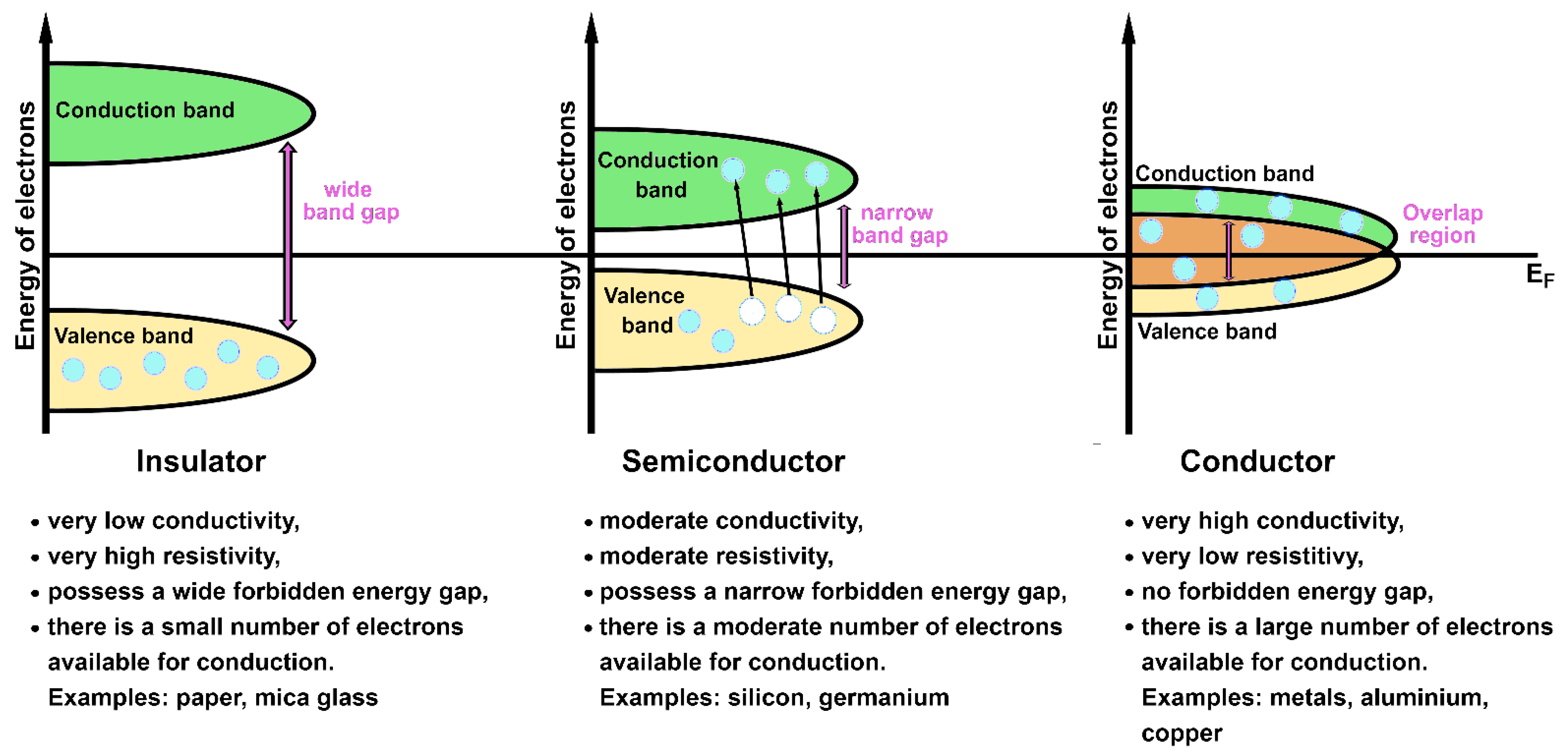
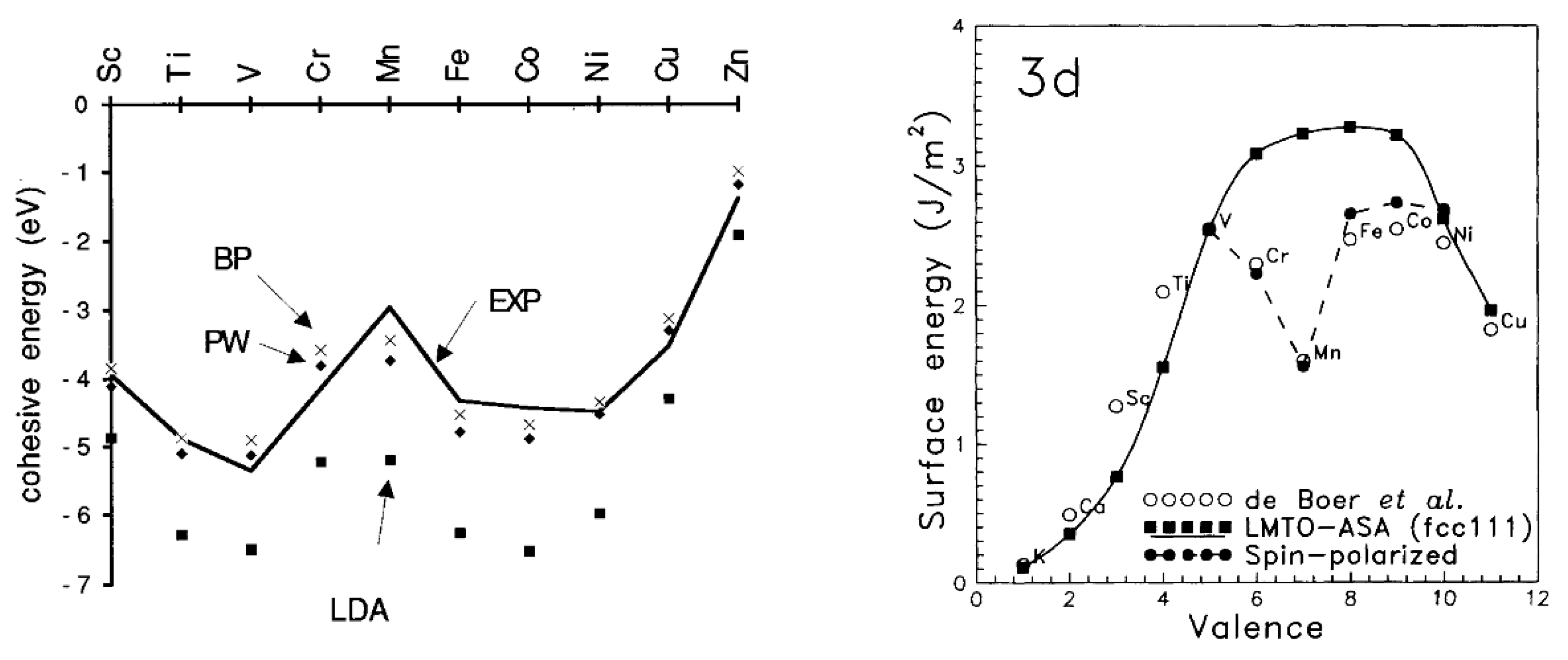

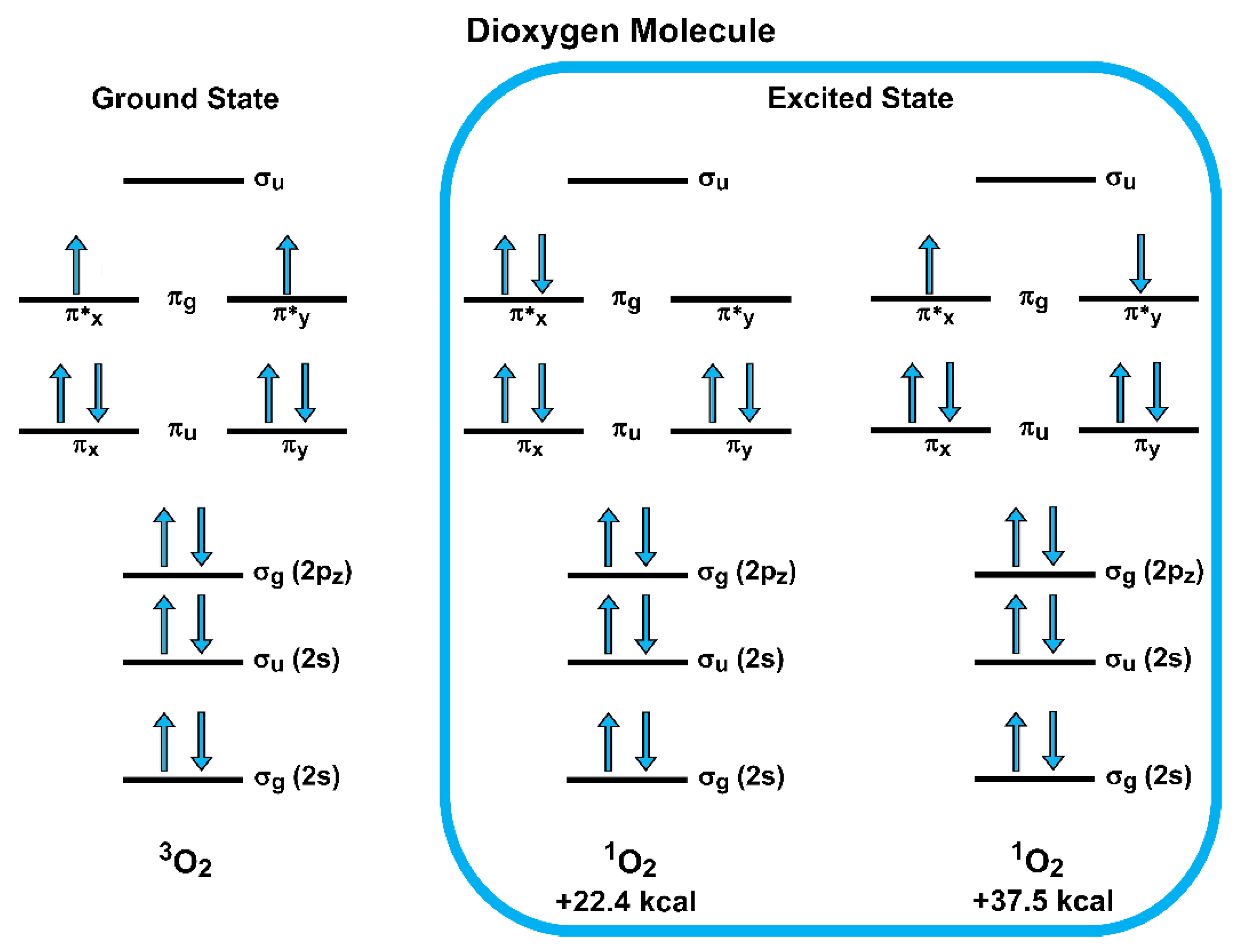







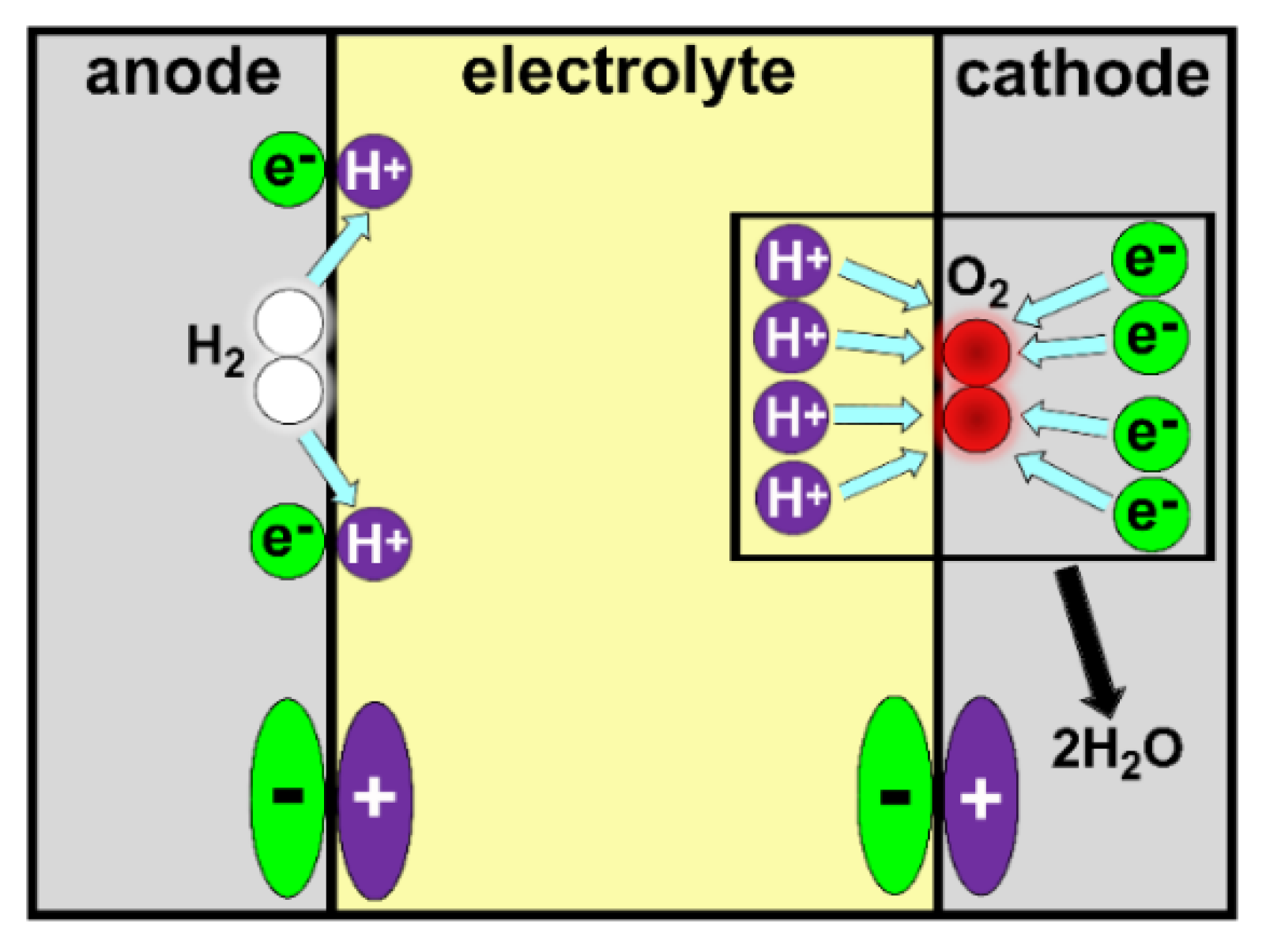
| System | NPs Size (nm) | Cell Parameters (nm) | c/a Ratio | Reference | |
|---|---|---|---|---|---|
| a | c | ||||
| FePt | |||||
| fcc FePt | - | 0.3884 | 0.3884 | 1 | Malheiro [116] |
| fct FePt | 6.5 ± 0.3 | 0.3848 | 0.3724 | 0.96 | Xiong [117] |
| fct FePt | ~6.1 | 0.27069 | 0.3709 | 1.37 | Chen [118] |
| fct FePt | ~6.1 | 0.27248 | 0.37312 | 1.37 | Chen [118] |
| CoPt | |||||
| fcc CoPt | 2.5 ± 0.2 | 0.3844 | 0.3844 | 1 | Loukrakpam [119] |
| fcc CoPt | ~6.2 | 0.3803 | 0.3803 | 1 | Watanabe [120] |
| fcc CoPt | 2.5 ± 0.1 | 0.3797 | 0.3797 | 1 | Oezaslan [121] |
| fcc CoPt | 2.7 | 0.38732 | 0.38732 | 1 | Travitsky [122] |
| fct CoPt | 12.4 ± 1.4 | 0.3814 | 0.3704 | 0.97 | Oezaslan [121] |
| fct CoPt | ~6.2 | 0.2692 | 0.3662 | 1.36 | Watanabe [120] |
| fct CoPt | ~6 | 0.3780 | 0.3705 | 0.98 | Xiong [117] |
| NiPt | |||||
| fcc NiPt | 4.7 | 0.3821 | 0.3821 | 1 | Xiong [117] |
| fcc NiPt | 4.8 ± 0.5 | 0.3817 | 0.3817 | 1 | Loukrakpam [119] |
| fcc NiPt | 2.2 | 0.38486 | 0.38486 | 1 | Travitsky [122] |
| fcc NiPt | 3.2 | 0.38204 | 0.38204 | 1 | Travitsky [122] |
| fcc NiPt | 6.1 | 0.37368 | 0.37368 | 1 | Carpenter [123] |
| Entry | NPs Size (nm) | SA (mA·cm−2) | Tafel Slope (mV·dec−1) | Reference | ||
|---|---|---|---|---|---|---|
| Pt | ||||||
| Commercial Pt/C | 2–3 | 0.22 | 0.12 | 75 | 0.883 | Li [243] |
| Commercial Pt/C | - | 0.28 | 0.13 | - | - | Li [246] |
| Commercial Pt/C | 2.5–3.5 | 0.264 a | - | - | 0.531 | Zhang [247] |
| Commercial Pt/C | 3.2 | - | - | - | 0.905 | Liu [248] |
| Commercial Pt/C | - | 0.177 | 0.102 | - | 0.864 | Ying [249] |
| Pt/C | ~2.7 | 0.07 | - | 68.9 | 0.818 | Du [250] |
| Pt/C | - | 0.22 | 0.14 | - | - | Chung [244] |
| Pt/C | <7.1 ± 1.6 b | 0.20 | 0.11 | - | - | Loukrakpam [119] |
| Pt/C | - | 1.70 c | - | - | - | Gong [251] |
| Pt black | - | 0.221 | 0.042 | - | 0.868 | Ying [249] |
| FePt | ||||||
| fcc FePt/C | 8.5 ± 0.5 | 0.89 a | - | - | 0.533 | Zhang [247] |
| fcc FePt/C | <10 | - | - | - | 0.890 | Li [246] |
| fcc FePt/C | 2.6 c | 3.95 | - | - | - | Gong [251] |
| fcc FePt/CNT | 2–3 | - | - | - | 0.894 | Liu [248] |
| fct FePt/C | ~6.1 | 0.578 | 0.272 | - | - | Chen [118] |
| fct FePt/C | 8.5 ± 0.5 | 2.1 a | - | - | 0.562 | Zhang [247] |
| fct FePt/C | 8.8 ± 0.5 | 3.16 | 0.69 | - | 0.958 | Li [246] |
| fct FePt/C | 8.0 ± 0.5 | - | 0.7 | - | 0.945 | Li [242] |
| fct FePt/C | 6.5 | 2.3 | 1.6 | - | - | Chung [244] |
| fct FePt/C | ~6.1 | 0.589 | 0.230 | - | - | Chen [118] |
| fct FePt | ~3.6 | 0.37 | - | 65.8 | 0.893 | Du [250] |
| fct FePt/CNT | 3–13 | 0.26 | 0.308 | - | 0.921 | Liu [248] |
| CoPt | ||||||
| fcc CoPt | 8.9 ± 0.8 | 0.70 | 0.15 | 86 | - | Li [243] |
| fcc CoPt/C | 2.5 ± 0.2 b | 0.57 | 0.25 | - | - | Loukrakpam [119] |
| fcc CoPt/Co@NHPCC | - | 0.876 | 0.566 | - | 0.883 | Ying [249] |
| fct PtCo | 3.8 ± 1.1 | - | 0.25 ± 0.07 | 88 | - | Oezaslan [121] |
| fct CoPt | 8.9 ± 0.8 | 8.26 | 2.26 | 66 | 0.967 | Li [243] |
| NiPt | ||||||
| fcc NiPt/C | 4.8 ± 0.5 b | 0.69 | 0.17 | - | - | Loukrakpam [119] |
| fcc NiPt/C | 6.1 | 2.977 | 0.68 | - | - | Carpenter [123] |
| MPt | Preparation Method | MxPt1−x | Structural Parameters | Magnetic Properties | Reference |
|---|---|---|---|---|---|
| FePt | |||||
| fcc | DC sputtering (pprep = 1 mbar) | Fe62Pt38 | NPsize = 4.6, Polycrystalline | Hc = 1.48·103, TB = 53–100 | Rellinghaus [105] |
| fcc | Microwave heating method | - | NPsize = 2.7 | SP | Nguyen [311] |
| fcc | Synthetic chemical method | - | NPsize = 2.25 | SP, TB = 14 | Nguyen [104] |
| fcc | Co-reduction chemical method | - | NPsize~3 | SP | Medwal [312] |
| fcc | Modified polyol process | Fe52Pt48 | - | SP, TB = 20–30 | Sun [313] |
| fcc | Chemical solution method | Fe52Pt48 | NPsize = 4 | - | Rong [314] |
| fct Partially Ordered | Microwave heating method + annealing under Ar + 5%H2 flowing atmosphere at ~637 K for ~6 min | Fe48Pt52 (fct > fcc) | NPsize~24 c/a = 0.9675 | Hc = 10.6·103 | Nguyen [311] |
| fct Partially Ordered | DC sputtering (pprep = 1 mbar) + gas-phase sintering at 1073 K | Fe62Pt38 | NPsize~7.5 | TB = 309 | Rellinghaus [105] |
| fct Partially Ordered | DC sputtering (pprep = 1.5 mbar) + gas-phase sintering at 1273 K | Fe51Pt49 | NPsize~7.2 | TB = 309 | Rellinghaus [105] |
| fct Partially Ordered | DC sputtering (pprep = 1 mbar) + gas-phase sintering at 1273 K | Fe62Pt38 | NPsize~7.7 | TB = 530, Hc = 1.2·103 | Rellinghaus [105] |
| fct Partially Ordered | Synthetic chemical method + annealing under Ar + 5%H2 flowing atmosphere at 662 K for 18 h | Fe56Pt44 %fcc > %fct | NPsize = 6.09 | Hc = 1.3·103 | Nguyen [104] |
| fct Partially Ordered | Co-reduction chemical method + annealing at 873 K | 38% fcc +62% fct | NPsize~5 c/a = 0.9848 | Hc soft-phase = 890 + Hc hard-phase = 11,930 | Medwal [312] |
| fct Partially Ordered | Co-reduction chemical method + annealing at 973 K | 10% fcc +90% fct | NPsize~5 c/a = 0.9801 | Hc soft-phase = 3250 + Hc hard-phase = 12,310 | Medwal [312] |
| fct Partially Ordered | Co-reduction chemical method + annealing at 1023 K | 5% fcc + 95% fct | NPsize > 5 c/a = 0.9692 | Hc soft-phase = 6970 + Hc hard-phase = 13,940 | Medwal [312] |
| fct Partially Ordered | Chemical synthesis + annealing at 973 K for 2 h under atmosphere of 4%H2 | Fe52Pt48 24% fcc +76% fct | NPsize~20 c/a = 0.9626 S = 0.64 | Hc = 7212, (BH)max~6.31 Ms = 34.90 | Srivastava [315] |
| fct | Chemical synthesis + annealing at 973 K for 4 h under 4% H2 atmosphere | Fe52Pt48 11%fcc + 89%fct | NPsize~20 c/a = 0.9646 S = 0.88 | Hc = 8617, (BH)max~10.92 Ms = 30.80 | Srivastava [315] |
| fct | Chemical synthesis + annealing at 973 K for 6 h under atmosphere of 4% H2 | Fe52Pt48 9% fcc + 91% fct | NPsize~20 c/a = 0.9626 S = 0.95 | Hc = 9040, (BH)max~7.60 Ms = 32.45, Ku~6.02·107 | Srivastava [315] |
| fct | Chemical solution route + 1 h annealing under forming gas (Ar + 7% H2) at 973 K | Fe55Pt45 | NPsize = 10–20 | Hc = 18·103 | Rong [106] |
| fct | Chemical solution route + 1 h annealing under forming gas (Ar + 7% H2) at 973 K | Fe66Pt34 | NPsize = 10–20 | Hc = 7.6·103, (BH)max~17 | Rong [106] |
| fct | Chemical solution route + rapid thermal annealing at 923 K for 10 s | Close to Fe50Pt50 | NPsize = 8 S = 0.97 | (BH)max~12.7 | Yano [316] |
| fct | Modified polyol process + annealing at 833 K for 30 min in static N2 atmosphere (p = 1 atm) | Fe52Pt48 | NPsize = 4 | FM | Sun [313] |
| fct | Chemical solution method + annealing at 973 K for 4 h under forming gas (93%Ar + 7%H2) | Fe52Pt48 | NPsize = 8.2 S~0.93 | FM | Rong [314] |
| CoPt | |||||
| fcc | Polyol process | Close to Co50Pt50 | NPsize = 4 ± 1 | Hc = 380, Ms = 8 | Chinnasamy [317] |
| fcc | Redox transmetallation reaction | Co46Pt54 | NPsize = 1.9 ± 0.3 | SP, TB = 15 | Park [318] |
| fcc | Soft chemical processing route + annealing at 673 K for 3 h under Ar atmosphere | Co46Pt54 | NPsize = 4–7 elongated shape c/a = 0.9732 | Hc = 260 | Fang [319] |
| fcc | Redox transmetallation reaction | - | NPsize~5 with Pt-shell~1.5 nm | Hc = 0, TB = 66 Ms = 27(//) and 27(⊥) | Bigot [320] |
| fct | Chemical process + annealing at 923 K for 1 h under Ar/5% H2 flowing atmosphere | Co50Pt50 | NPsize = 7.6 rod-like shape | Hc = 12·103, Ku = 1.7·107 | Sun [127] |
| fct | Polyol process + annealing at 823 K for 1 h under H2/N2 atmosphere | Close to Co50Pt50 | NPsize = 4 ± 1 | Hc = 1.34·103 | Chinnasamy [317] |
| fct | Polyol process + annealing at 873 K or 1 h under H2/N2 atmosphere | Close to Co50Pt50 | NPsize = 4 ± 1 | Hc = 3.67·103 | Chinnasamy [317] |
| fct | Polyol process + annealing at 973 K for 1 h under H2/N2 atmosphere | Close to Co50Pt50 | NPsize = 4 ± 1 | Hc = 7.57·103 | Chinnasamy [317] |
| fct | Pellet of NPs obtain from fcc core–shell with a Co-core and a Pt-shell after annealing at 723 K for 1 h in primary vacuum | - | NPsize = 8 | Hc = 50(//) and 50(⊥), TB = 347 Ms = 181(//) and 151(⊥) Ku ≈ 1.8·105 | Bigot [320] |
| System | Preparation Method | Size | Composition | SA | Tafel Slope | Magnetic Properties | Reference |
|---|---|---|---|---|---|---|---|
| FePt | |||||||
| fcc FePt/C | Chemical synthesis | 8.5 ± 0.5 | As-prepared: Fe51Pt49; Core–shell: Fe26Pt74 with a Pt-shell of ~3 atomic layers (~0.6 nm) | 0.89 a | - | - | Zhang [247] |
| fcc FePt/C | Chemical method | <10 | Fe52Pt48 | - | - | SP | Li [246] |
| fcc FePt/C | Bönnemann colloidal method | 2.6 | ~Fe50Pt50 | 3.95 b | - | - | Gong [251] |
| fcc FePt/CNT | Chemical reduction method | 2–3 | - | - | - | - | Liu [248] |
| fct FePt/C | Impregnation method + annealing at ~873 K for 3 h under an 8% H2/Ar gas mixture | ~6.1 | Core–shell with ~0.6 nm of Pt coating (2–4 atomic layers) | 0.578 | - | - | Chen [118] |
| fct FePt/C | Chemical synthesis + annealing at ~923 K for 1 h under 95% Ar + 5% H2 atmosphere | 8.5 ± 0.5 | As-prepared: Fe51Pt49; Core–shell: Fe26Pt74 with a Pt-shell of ~3 atomic layers (~0.6 nm) | 2.1 a | - | - | Zhang [247] |
| fct FePt/C | Chemical method + annealing at ~973 K for 6 h under Ar + 5% H2 | 8.8 ± 0.5 | As-prepared: Fe52Pt48; Core–shell: Fe50Pt50 with a Pt-shell of ~0.6 nm (~2–4 atomic layers) | 3.16 | - | Hc = 33·103 for as-prepared NPs | Li [246] |
| fct FePt/C | Modified chemical method + annealing at ~973 K for 6 h under 95% Ar + 5% H2 atmosphere | 8.0 ± 0.5 | Core–shell: Fe42Pt58 with a Pt-shell of 0.53 nm (~2 atomic layers); degree of ordering >80% | - | - | FM; Hc = 33.8·103 | Li [242] |
| fct FePt/C | Chemical synthesis + annealing at ~973 K | 6.5 | Core–shell with ~0.43 nm of N-doped carbon shell (~2 atomic layers) | 2.3 | - | - | Chung [244] |
| fct FePt/C | Impregnation method + annealing at ~1073 K for 3 h under an 8% H2/Ar gas mixture | ~6.1 | Core–shell with ~0.6 nm of Pt coating (2–4 atomic layers) | 0.589 | - | - | Chen [118] |
| fct FePt/C | Liquid-phase reduction method + annealing at ~1173 K in a tube furnace under vacuum | ~3.6 | - | 0.37 | 65.8 | - | Du [250] |
| fct FePt/CNT | Chemical reduction method + annealing at ~923 K under H2-free inert atmosphere | 3–13 | Core–shell with ~3 atomic layers of Pt coating | 0.26 | - | - | Liu [248] |
| CoPt | |||||||
| fcc CoPt/C | Chemical method | 8.9 ± 0.8 | As-prepared: Co49Pt51 | 0.70 | 86 | SP | Li [243] |
| fcc CoPt/C | Chemical synthesis | 2.5 ± 0.2 | Pt52Co48 | 0.57 c | - | - | Loukrakpam [119] |
| fcc CoPt/ Co@NHPCC | Chemical synthesis | - | - | 0.876 | - | - | Ying [249] |
| fct PtCo/C | Liquid precursor impregnation–freeze-drying method + annealing at ~1037 K for 7 h under 4 Vol% H2/96 Vol% Ar atmosphere | 3.8 ± 1.1 | As-prepared: Pt59Co41 After stability treatment: Pt77Co23 85% fct + 15% fcc | - | 88 | - | Oezaslan [121] |
| fct CoPt/C | Chemical method + annealing at ~923 K for 6 h under 95% Ar + 5% H2 atmosphere | 8.9 ± 0.8 | As-prepared: Co49Pt51; Core–shell with a Pt-shell of 3 atomic layers 88% fct + 12% fcc | 8.26 | 66 | FM; HC = 7.1·103 for as-prepared NPs | Li [243] |
| NiPt | |||||||
| fcc NiPt/C | Chemical synthesis | 4.8 ± 0.5 | Pt56Ni44 | 0.69 c | - | - | Loukrakpam [119] |
| fcc NiPt/C | Solvothermal reaction | 6.1 | Ni47Pt53 | 2.977 | - | - | Carpenter [123] |
| Catalyst | Three-Electrode and External Magnetic Field Parameters | Experimental Observations | Ref. |
|---|---|---|---|
| Direct application of external magnetic fields | |||
| Pt/ FM CoPt nanowire/alumina membrane |
|
| Chaure [377] |
| Pt/alumina membrane | |||
| Indirect application of external magnetic fields (magnetization of catalyst) | |||
| L10-PtFe nanopillar (FM) |
|
| Lu [379] |
| Pt/Ag/CoPt nanowire |
|
| Chaure [378] |
| Battery | Characteristics | Advantages | Limitations | Examples |
|---|---|---|---|---|
| Primary batteries |
|
|
| Zinc–carbon, alkaline, lithium primary cells |
| Secondary batteries |
|
|
| Lead acid, lithium ion, nickel–cadmium |
| Battery systems for grid-scale energy |
|
|
| Flow, sodium–sulfur |
| Fuel cells |
|
|
| Proton exchange membrane, direct methanol, solid oxide |
| Electrochem. capacitors (or supercapacitors) |
|
|
| Carbon-based, metal oxide, polymeric |
| Strength | Weakness | Opportunities | Threats |
|---|---|---|---|
|
|
|
|
| Catalyst (Cathode) | PEMFC and MF Parameters | Experimental Observations | Reference |
|---|---|---|---|
| Single Fuel Cell experiments under an applied magnetic field | |||
| Pt |
|
| Matsushima [397] |
| Pt |
|
| Lang [419] |
| Not reported |
|
| Ruksawong [420] |
| Single Fuel Cell experiments with magnetized catalyst | |||
| Nd-Fe-B + 20% Pt/Vulcan XC72 |
|
| Okada [381] |
| Pt-Co/ MWCNTs |
|
| Sun [421] |
| Nd2Fe14B/C+ 50% Pt/C |
|
| Shi [384] |
| Fuel cell stack experiments | |||
| Pt |
|
| Abdel-Rehim [418] |
| Effects of Engineering Magnetic Catalysts with FM Metals | Effects of Applying an External Magnetic Field | Effects of Combining Engineered Magnetic Catalysts with FM Metals and Applied External Magnetic Field |
|---|---|---|
|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Biz, C.; Gracia, J.; Fianchini, M. Review on Magnetism in Catalysis: From Theory to PEMFC Applications of 3d Metal Pt-Based Alloys. Int. J. Mol. Sci. 2022, 23, 14768. https://doi.org/10.3390/ijms232314768
Biz C, Gracia J, Fianchini M. Review on Magnetism in Catalysis: From Theory to PEMFC Applications of 3d Metal Pt-Based Alloys. International Journal of Molecular Sciences. 2022; 23(23):14768. https://doi.org/10.3390/ijms232314768
Chicago/Turabian StyleBiz, Chiara, José Gracia, and Mauro Fianchini. 2022. "Review on Magnetism in Catalysis: From Theory to PEMFC Applications of 3d Metal Pt-Based Alloys" International Journal of Molecular Sciences 23, no. 23: 14768. https://doi.org/10.3390/ijms232314768
APA StyleBiz, C., Gracia, J., & Fianchini, M. (2022). Review on Magnetism in Catalysis: From Theory to PEMFC Applications of 3d Metal Pt-Based Alloys. International Journal of Molecular Sciences, 23(23), 14768. https://doi.org/10.3390/ijms232314768







