Fusion Cell Markers in Circulating Tumor Cells from Patients with High-Grade Ovarian Serous Carcinoma
Abstract
:1. Introduction
2. Results
2.1. Clinical-Pathological Data
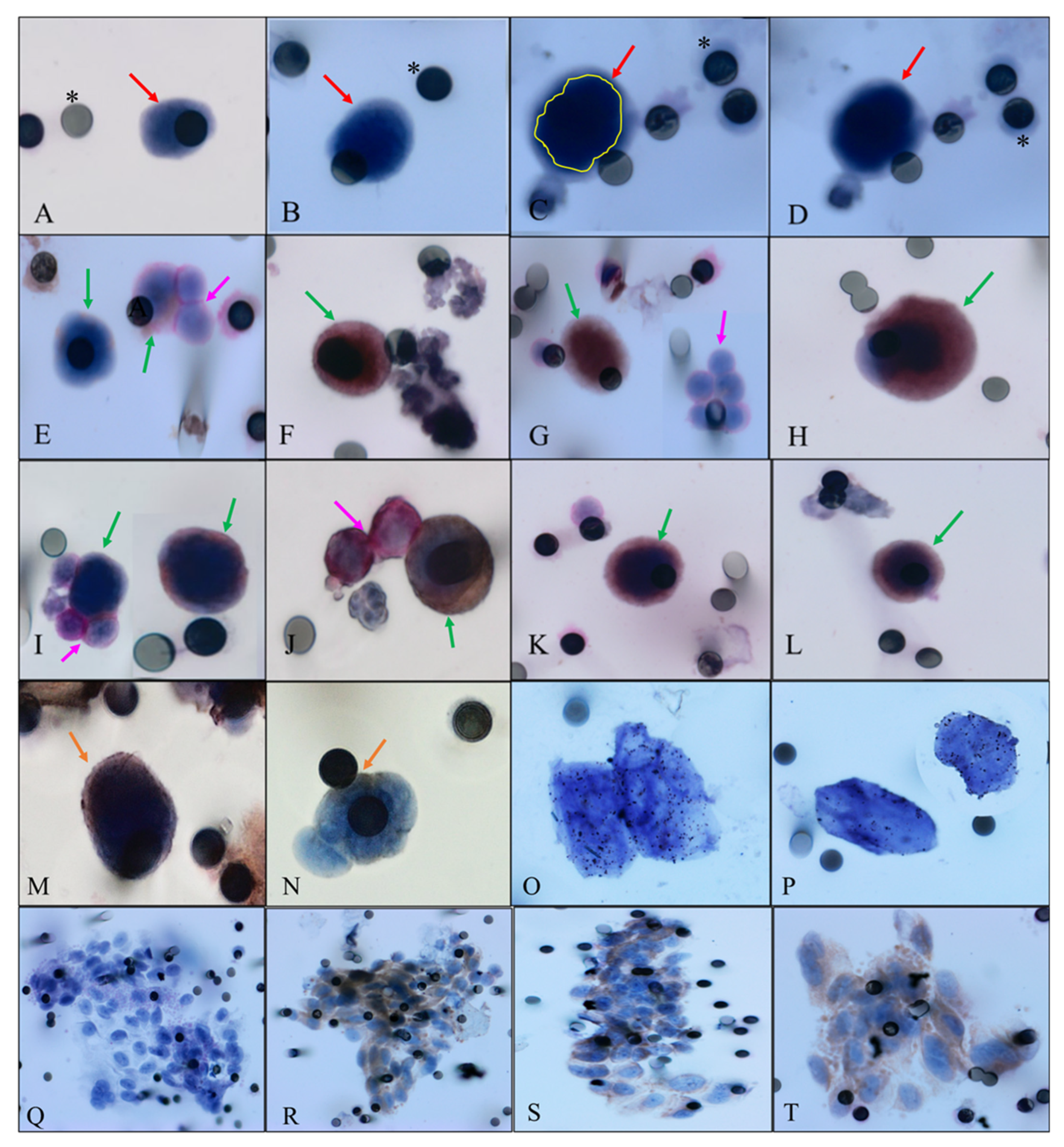
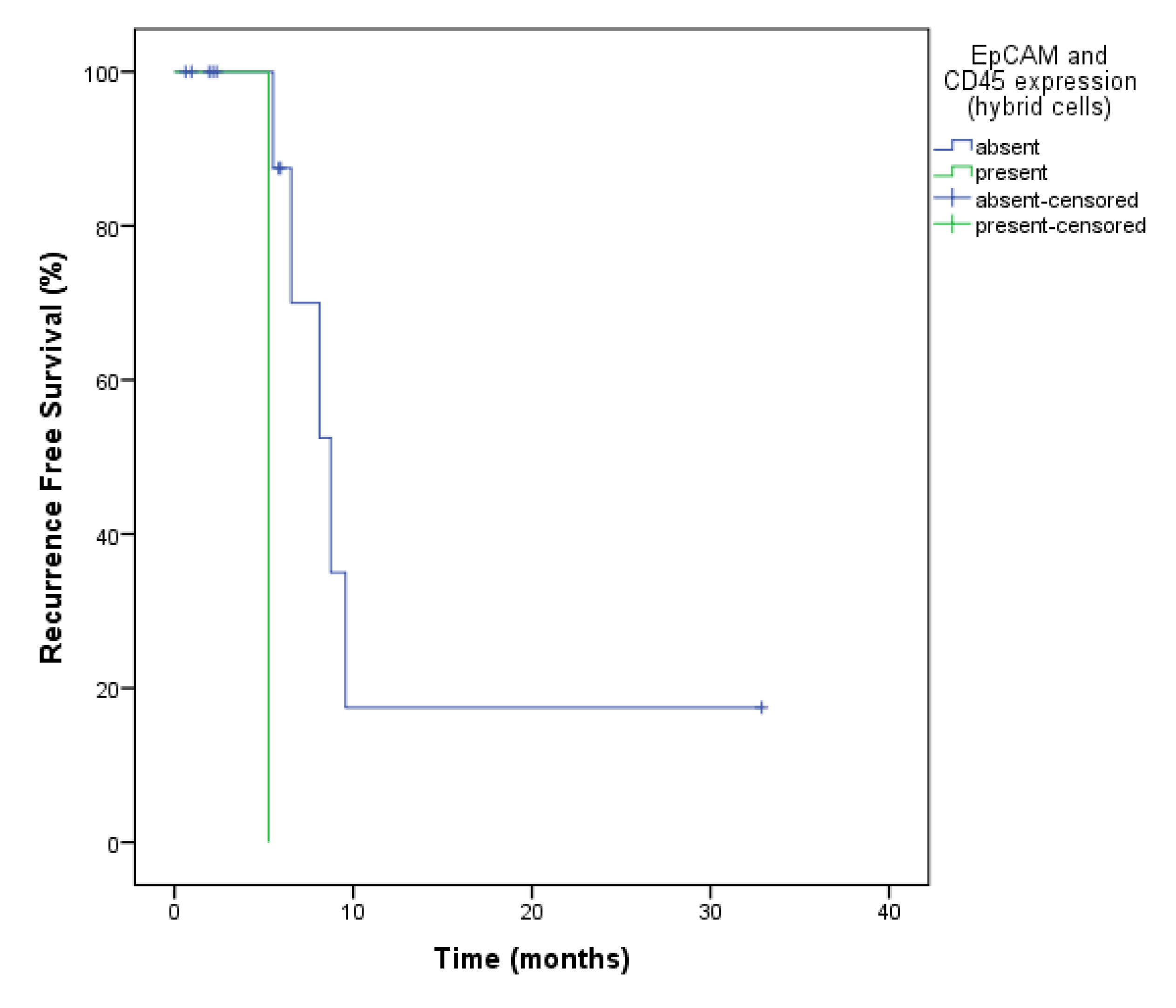
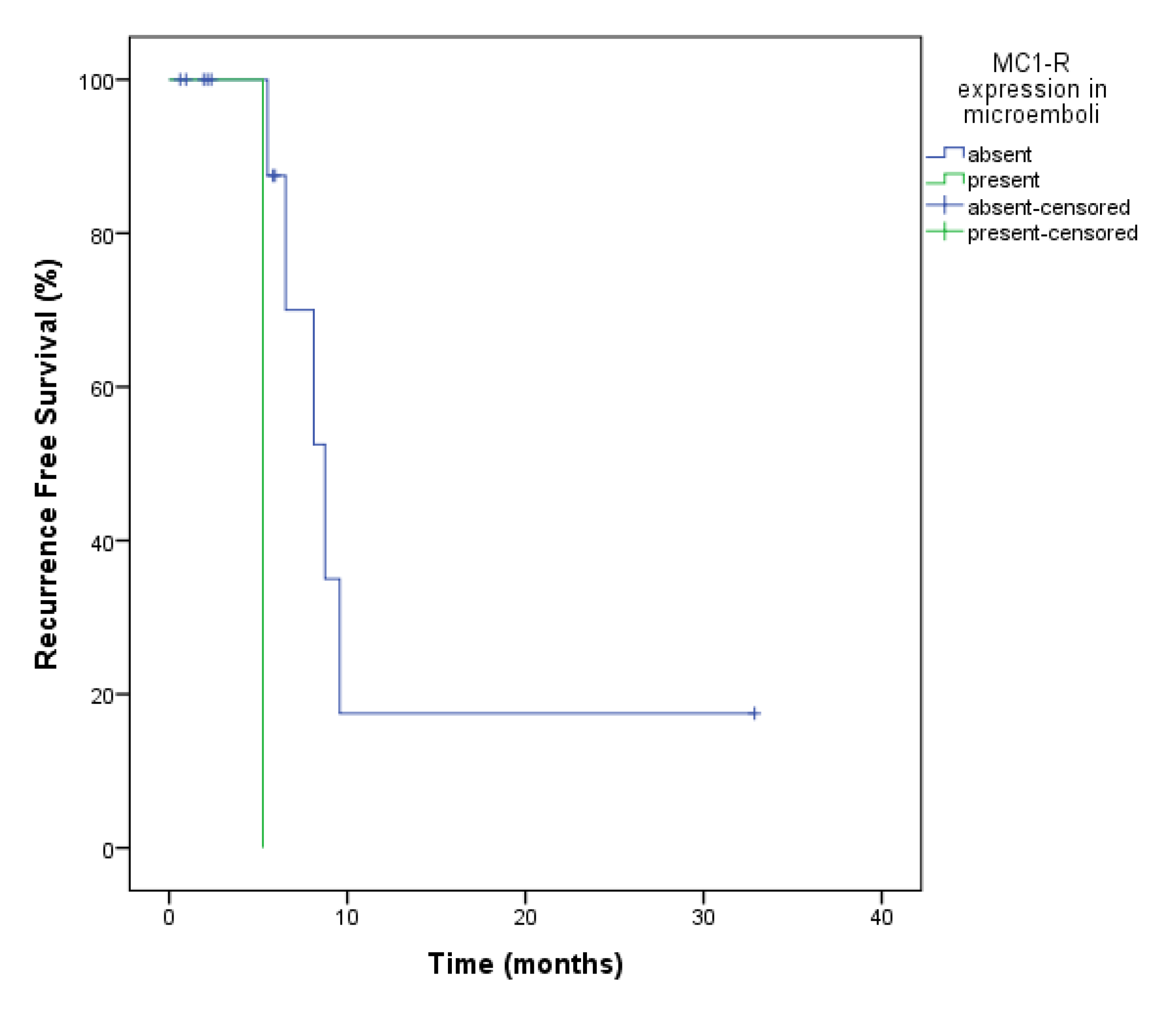
2.2. Analysis of Protein Expression
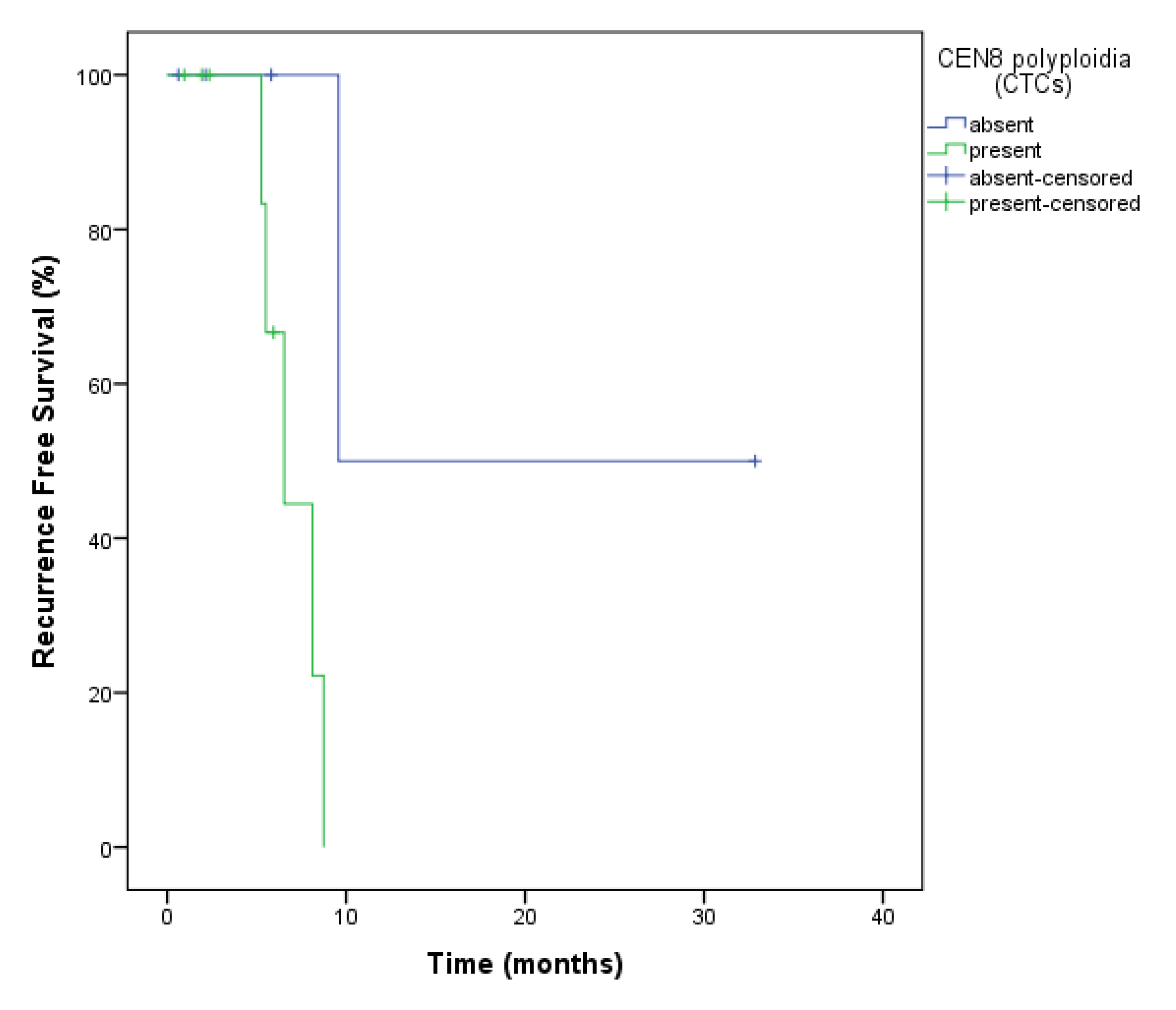
2.3. CEN8
2.4. CTCs and Recurrence-Free Survival (RFS)
2.5. CTCs and Overall Survival
2.6. CTCs Detected Using the CellSearch®
3. Discussion
4. Materials and Methods
4.1. Patient Population and Study Design
4.2. Ethics
4.3. Detection of CTCs and the Cell Fusion Phenotype
4.3.1. ISET Assay
4.3.2. Immunocytochemistry
4.3.3. Chromogenic In Situ Hybridization
4.3.4. The CellSearch® System (Menarini Biosystems)
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Msc, M.L.; Weiderpass, E.; Soerjomataram, I. The ever-increasing importance of cancer as a leading cause of premature death worldwide. Cancer 2021, 127, 3029–3030. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Instituto Nacional de Câncer-INCA. INCA. Published 2022. Available online: https://www.inca.gov.br/tipos-de-cancer/cancer-de-ovario (accessed on 20 September 2022).
- Maltoni, R.; Gallerani, G.; Fici, P.; Rocca, A.; Fabbri, F. CTCs in early breast cancer: A path worth taking. Cancer Lett. 2016, 376, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Pantel, K.; Alix-Panabières, C. Circulating tumour cells in cancer patients: Challenges and perspectives. Trends Mol. Med. 2010, 16, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Cristofanilli, M.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Matera, J.; Miller, M.C.; Reuben, J.M.; Doyle, G.V.; Allard, W.J.; Terstappen, L.W.M.M.; et al. Circulating Tumor Cells, Disease Progression, and Survival in Metastatic Breast Cancer. N. Engl. J. Med. 2004, 351, 781–791. [Google Scholar] [CrossRef] [Green Version]
- Cohen, S.J.A.; Punt, C.J.; Iannotti, N.; Saidman, B.H.; Sabbath, K.D.; Gabrail, N.Y.; Picus, J.; Morse, M.; Mitchell, E.; Miller, M.C.; et al. Relationship of Circulating Tumor Cells to Tumor Response, Progression-Free Survival, and Overall Survival in Patients With Metastatic Colorectal Cancer. J. Clin. Oncol. 2008, 26, 3213–3221. [Google Scholar] [CrossRef]
- De Bono, J.S.; Scher, H.I.; Montgomery, R.B.; Parker, C.; Miller, M.C.; Tissing, H.; Doyle, G.V.; Terstappen, L.W.W.M.; Pienta, K.J.; Raghavan, D. Circulating Tumor Cells Predict Survival Benefit from Treatment in Metastatic Castration-Resistant Prostate Cancer. Clin. Cancer Res. 2008, 14, 6302–6309. [Google Scholar] [CrossRef] [Green Version]
- Krebs, M.G.; Hou, J.-M.; Sloane, R.; Lancashire, L.; Priest, L.; Nonaka, D.; Ward, T.H.; Backen, A.; Clack, G.; Hughes, A.; et al. Analysis of Circulating Tumor Cells in Patients with Non-small Cell Lung Cancer Using Epithelial Marker-Dependent and -Independent Approaches. J. Thorac. Oncol. 2012, 7, 306–315. [Google Scholar] [CrossRef] [Green Version]
- Williams, E.D.; Gao, D.; Redfern, A.; Thompson, E.W. Controversies around epithelial–mesenchymal plasticity in cancer metastasis. Nat. Cancer 2019, 19, 716–732. [Google Scholar] [CrossRef] [Green Version]
- Szczerba, B.M.; Castro-Giner, F.; Vetter, M.; Krol, I.; Gkountela, S.; Landin, J.; Scheidmann, M.C.; Donato, C.; Scherrer, R.; Singer, J.; et al. Neutrophils escort circulating tumour cells to enable cell cycle progression. Nature 2019, 566, 553–557. [Google Scholar] [CrossRef]
- Yu, M. Metastasis Stemming from Circulating Tumor Cell Clusters. Trends Cell Biol. 2019, 29, 275–276. [Google Scholar] [CrossRef] [PubMed]
- Bobek, V.; Gurlich, R.; Eliasova, P.; Kolostova, K. Circulating tumor cells in pancreatic cancer patients: Enrichment and cultivation. World J. Gastroenterol. 2014, 20, 17163–17170. [Google Scholar] [CrossRef] [PubMed]
- Clawson, G. The Fate of Fusions. Cells 2018, 8, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Condeelis, J.; Pollard, J.W. Macrophages: Obligate Partners for Tumor Cell Migration, Invasion, and Metastasis. Cell 2006, 124, 263–266. [Google Scholar] [CrossRef] [Green Version]
- Shemer, G.; Podbilewicz, B. The story of cell fusion: Big lessons from little worms. BioEssays 2003, 25, 672–682. [Google Scholar] [CrossRef]
- Platt, J.L.; Cascalho, M. Cell Fusion in Malignancy: A Cause or Consequence? A Provocateur or Cure? Cells 2019, 8, 587. [Google Scholar] [CrossRef] [Green Version]
- Halaban, R.; Nordlund, J.; Francke, U.; Moellmann, G.; Eisenstadt, J.M. Supermelanotic hybrids derived from mouse melanomas and normal mouse cells. Somat. Cell Mol. Genet. 1980, 6, 29–44. [Google Scholar] [CrossRef]
- Lindström, A.; Midtbö, K.; Arnesson, L.-G.; Garvin, S.; Shabo, I. Fusion between M2-macrophages and cancer cells results in a subpopulation of radioresistant cells with enhanced DNA-repair capacity. Oncotarget 2017, 8, 51370–51386. [Google Scholar] [CrossRef] [Green Version]
- Hernandez, L.D.; Hoffman, L.R.; Wolfsberg, T.G.; White, J.M. VIRUS-CELL AND CELL-CELL FUSION. Annu. Rev. Cell Dev. Biol. 1996, 12, 627–661. [Google Scholar] [CrossRef]
- Lu, X.; Kang, Y. Cell Fusion as a Hidden Force in Tumor Progression. Cancer Res. 2009, 69, 8536–8539. [Google Scholar] [CrossRef]
- Jiang, E.; Yan, T.; Xu, Z.; Shang, Z. Tumor Microenvironment and Cell Fusion. BioMed Res. Int. 2019, 2019, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bastida-Ruiz, D.; Van Hoesen, K.; Cohen, M. The Dark Side of Cell Fusion. Int. J. Mol. Sci. 2016, 17, 638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brzoska, T.; Luger, T.A.; Maaser, C.; Abels, C.; Böhm, M. α-Melanocyte-Stimulating Hormone and Related Tripeptides: Biochemistry, Antiinflammatory and Protective Effects in Vitro and in Vivo, and Future Perspectives for the Treatment of Immune-Mediated Inflammatory Diseases. Endocr. Rev. 2008, 29, 581–602. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moscowitz, A.E.; Asif, H.; Lindenmaier, L.B.; Calzadilla, A.; Zhang, C.; Mirsaeidi, M. The Importance of Melanocortin Receptors and Their Agonists in Pulmonary Disease. Front. Med. 2019, 6, 145. [Google Scholar] [CrossRef] [Green Version]
- Chakraborty, A.K.; Funasaka, Y.; Slominski, A.; Bolognia, J.; Sodi, S.; Ichihashi, M.; Pawelek, J.M. UV Light and MSH Receptors. Ann. N. Y. Acad. Sci. 2006, 885, 100–116. [Google Scholar] [CrossRef] [PubMed]
- Laberge, G.S.; Duvall, E.; Grasmick, Z.; Haedicke, K.; Pawelek, J. A Melanoma Lymph Node Metastasis with a Donor-Patient Hybrid Genome following Bone Marrow Transplantation: A Second Case of Leucocyte-Tumor Cell Hybridization in Cancer Metastasis. PLoS ONE 2017, 12, e0168581. [Google Scholar] [CrossRef] [PubMed]
- Losick, V.P.; Fox, D.T.; Spradling, A.C. Polyploidization and Cell Fusion Contribute to Wound Healing in the Adult Drosophila Epithelium. Curr. Biol. 2013, 23, 2224–2232. [Google Scholar] [CrossRef] [Green Version]
- Tamori, Y.; Deng, W.-M. Tissue Repair through Cell Competition and Compensatory Cellular Hypertrophy in Postmitotic Epithelia. Dev. Cell 2013, 25, 350–363. [Google Scholar] [CrossRef] [Green Version]
- Liu, J. The dualistic origin of human tumors. Semin. Cancer Biol. 2018, 53, 1–16. [Google Scholar] [CrossRef]
- Rheinländer, A.; Schraven, B.; Bommhardt, U. CD45 in human physiology and clinical medicine. Immunol. Lett. 2018, 196, 22–32. [Google Scholar] [CrossRef]
- Keller, L.; Werner, S.; Pantel, K. Biology and clinical relevance of EpCAM. Cell Stress 2019, 3, 165–180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, L.; Yang, Y.; Yang, F.; Liu, S.; Zhu, Z.; Lei, Z.; Guo, J. Functions of EpCAM in physiological processes and diseases (Review). Int. J. Mol. Med. 2018, 42, 1771–1785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carneiro, J. Histologia Básica, 12th ed.; Guanabara Koogan: Rio de Janeiro, Brazil, 2013. [Google Scholar]
- Broncy, L.; Ben Njima, B.; Méjean, A.; Béroud, C.; Ben Romdhane, K.; Ilie, M.; Hofman, V.; Muret, J.; Hofman, P.; Bouhamed, H.C.; et al. Single-cell genetic analysis validates cytopathological identification of circulating cancer cells in patients with clear cell renal cell carcinoma. Oncotarget 2018, 9, 20058–20074. [Google Scholar] [CrossRef] [Green Version]
- Castillo, L.N.G.; Mejean, A.; Vielh, P.; Anract, J.; Decina, A.; Nalpas, B.; Benali-Furet, N.; Desitter, I.; Paterlini-Bréchot, P. Predictive Value of Circulating Tumor Cells Detected by ISET® in Patients with Non-Metastatic Prostate Cancer Undergoing Radical Prostatectomy. Life 2022, 12, 165. [Google Scholar] [CrossRef] [PubMed]
- Searles, S.C.; Santosa, E.K.; Bui, J.D. Cell-cell fusion as a mechanism of DNA exchange in cancer. Oncotarget 2018, 9, 6156–6173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lengauer, C.; Kinzler, K.W.; Vogelstein, B. Genetic instabilities in human cancers. Nature 1998, 396, 643–649. [Google Scholar] [CrossRef]
- Ye, Z.; Ding, Y.; Chen, Z.; Li, Z.; Ma, S.; Xu, Z.; Cheng, L.; Wang, X.; Zhang, X.; Ding, N.; et al. Detecting and phenotyping of aneuploid circulating tumor cells in patients with various malignancies. Cancer Biol. Ther. 2019, 20, 546–551. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Shi, H.; Jiang, T.; Liu, Z.; Lin, P.P.; Chen, N. Circulating tumor cells with karyotyping as a novel biomarker for diagnosis and treatment of nasopharyngeal carcinoma. BMC Cancer 2018, 18, 1133. [Google Scholar] [CrossRef]
- Yu, S.; Fiedler, S.; Stegner, A.; Graf, W. Genomic profile of copy number variants on the short arm of human chromosome 8. Eur. J. Hum. Genet. 2010, 18, 1114–1120. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, X.; Ge, S.; Gao, J.; Gong, J.; Lu, M.; Zhang, Q.; Cao, Y.; Wang, D.D.; Lin, P.P.; et al. Clinical significance of phenotyping and karyotyping of circulating tumor cells in patients with advanced gastric cancer. Oncotarget 2014, 5, 6594–6602. [Google Scholar] [CrossRef]
- Clawson, G.A.; Kimchi, E.; Patrick, S.D.; Xin, P.; Harouaka, R.; Zheng, S.; Berg, A.; Schell, T.; Staveley-O’Carroll, K.F.; Neves, R.I.; et al. Circulating Tumor Cells in Melanoma Patients. PLoS ONE 2012, 7, e41052. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Itakura, E.; Huang, R.-R.; Wen, D.-R.; Cochran, A.J. “Stealth” Melanoma Cells in Histology-negative Sentinel Lymph Nodes. Am. J. Surg. Pathol. 2011, 35, 1657–1665. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pawelek, J.M.; Chakraborty, A.K. Fusion of tumour cells with bone marrow-derived cells: A unifying explanation for metastasis. Nat. Cancer 2008, 8, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhao, L.; Zhou, P.; Ma, H.; Huang, F.; Jin, M.; Dai, X.; Zheng, X.; Huang, S.; Zhang, T. Circulating tumor microemboli (CTM) and vimentin+ circulating tumor cells (CTCs) detected by a size-based platform predict worse prognosis in advanced colorectal cancer patients during chemotherapy. Cancer Cell Int. 2017, 17, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chu, H.-Y.; Yang, C.-Y.; Yeh, P.-H.; Hsu, C.-J.; Chang, L.-W.; Chan, W.-J.; Lin, C.-P.; Lyu, Y.-Y.; Wu, W.-C.; Lee, C.-W.; et al. Highly Correlated Recurrence Prognosis in Patients with Metastatic Colorectal Cancer by Synergistic Consideration of Circulating Tumor Cells/Microemboli and Tumor Markers CEA/CA19-9. Cells 2021, 10, 1149. [Google Scholar] [CrossRef]
- de Oliveira, T.B.; Braun, A.C.; Nicolau, U.R.; Abdallah, E.A.; Alves, V.D.S.; de Jesus, V.H.F.; Calsavara, V.F.; Kowaslki, L.P.; Chinen, L.T.D. Prognostic impact and potential predictive role of baseline circulating tumor cells in locally advanced head and neck squamous cell carcinoma. Oral Oncol. 2021, 121, 105480. [Google Scholar] [CrossRef]
- Gast, C.E.; Silk, A.D.; Zarour, L.; Riegler, L.; Burkhart, J.G.; Gustafson, K.T.; Parappilly, M.S.; Roh-Johnson, M.; Goodman, J.R.; Olson, B.; et al. Cell fusion potentiates tumor heterogeneity and reveals circulating hybrid cells that correlate with stage and survival. Sci. Adv. 2018, 4, eaat7828. [Google Scholar] [CrossRef] [Green Version]
- Chakraborty, A.K.; Sodi, S.; Rachkovsky, M.; Kolesnikova, N.; Platt, J.T.; Bolognia, J.L.; Pawelek, J.M. A spontaneous murine melanoma lung metastasis comprised of host x tumor hybrids. Cancer Res. 2000, 60, 2512–2519. Available online: http://www.ncbi.nlm.nih.gov/pubmed/10811133 (accessed on 22 August 2022).
- Pawelek, J.M. Tumour cell hybridization and metastasis revisited. Melanoma Res. 2000, 10, 507–514. [Google Scholar] [CrossRef]
- Kemény, L.V.; Kurgyis, Z.; Buknicz, T.; Groma, G.; Jakab, Á.; Zänker, K.; Dittmar, T.; Németh, I.B. Melanoma Cells Can Adopt the Phenotype of Stromal Fibroblasts and Macrophages by Spontaneous Cell Fusion in Vitro. Int. J. Mol. Sci. 2016, 17, 826. [Google Scholar] [CrossRef] [Green Version]
- Adams, D.L.; Martin, S.S.; Alpaugh, R.K.; Charpentier, M.; Tsai, S.; Bergan, R.C.; Ogden, I.M.; Catalona, W.; Chumsri, S.; Tang, C.-M.; et al. Circulating giant macrophages as a potential biomarker of solid tumors. Proc. Natl. Acad. Sci. USA 2014, 111, 3514–3519. [Google Scholar] [CrossRef] [PubMed]
- Gironda, D.J.; Adams, D.L.; He, J.; Xu, T.; Gao, H.; Qiao, Y.; Komaki, R.; Reuben, J.M.; Liao, Z.; Blum-Murphy, M.; et al. Cancer associated macrophage-like cells and prognosis of esophageal cancer after chemoradiation therapy. J. Transl. Med. 2020, 18, 413. [Google Scholar] [CrossRef] [PubMed]
- Racila, E.; Euhus, D.; Weiss, A.J.; Rao, C.; McConnell, J.; Terstappen, L.W.M.M.; Uhr, J.W. Detection and characterization of carcinoma cells in the blood. Proc. Natl. Acad. Sci. USA 1998, 95, 4589–4594. [Google Scholar] [CrossRef] [Green Version]
- Sutton, T.L.; Walker, B.S.; Wong, M.H. Circulating Hybrid Cells Join the Fray of Circulating Cellular Biomarkers. Cell. Mol. Gastroenterol. Hepatol. 2019, 8, 595–607. [Google Scholar] [CrossRef] [Green Version]
- Pawelek, J.M.; Chakraborty, A.K. Chapter 10 The Cancer Cell–Leukocyte Fusion Theory of Metastasis. Adv. Cancer Res. 2008, 101, 397–444. [Google Scholar] [CrossRef] [PubMed]
- Shabo, I.; Svanvik, J.; Lindström, A.; Lechertier, T.; Trabulo, S.; Hulit, J.; Sparey, T.; Pawelek, J. Roles of cell fusion, hybridization and polyploid cell formation in cancer metastasis. World J. Clin. Oncol. 2020, 11, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Kaigorodova, E.V.; Kozik, A.V.; Zavaruev, I.S.; Grishchenko, M.Y. Hybrid/Atypical Forms of Circulating Tumor Cells: Current State of the Art. Biochemistry 2022, 87, 380–390. [Google Scholar] [CrossRef]

| Variables | No. | (%) |
|---|---|---|
| Median age at baseline, years | 45 years (29–55) | |
| FIGO stage | ||
| IIIc | 8 | 57.1 |
| IV | 3 | 21.4 |
| No information | 3 | 21.4 |
| Treatment for platinum-sensitive recurrence | ||
| Carboplatin and Paclitaxel | 14 | 100 |
| Treatment cycles | ||
| 6 cycles | 9 | 64.3 |
| 8 cycles | 2 | 14.3 |
| No information | 3 | 21.4 |
| Recurrence site before the 1st collection | ||
| Pelvic | 4 | 28.6 |
| Pelvic and mediastinum | 2 | 14.3 |
| Mediastinum | 1 | 7.1 |
| Pelvic/retroperitoneum | 1 | 7.1 |
| Retroperitoneum | 1 | 7.1 |
| No information | 5 | 35.7 |
| Surgery | ||
| Optimal surgery | 8 | 57.1 |
| Suboptimal surgery | 3 | 21.4 |
| No information | 3 | 21.4 |
| Mutation | ||
| BRCA1/BRCA2 | 8 | 57.1 |
| No mutation | 3 | 21.4 |
| No information | 3 | 21.4 |
| CA125 pre olaparib treatment median | 268 (7–2183.72) | |
| Relapse post-olaparib treatment | 6 | 42.9 |
| CEN 8 | 9/11 | 81.8 |
| CTC/mL median (baseline) | 0.58 CTCs/mL (0.33–9.91) | |
| CTC/mL median (1° follow-up) | 0.60 CTCs/mL (0.20–7.20) | |
| CTC/mL median (2° follow-up) | 1 CTCs/mL (1.00–8.58) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruano, A.P.C.; Gadelha Guimarães, A.P.; Braun, A.C.; Flores, B.C.T.C.P.; Tariki, M.S.; Abdallah, E.A.; Torres, J.A.; Nunes, D.N.; Tirapelli, B.; de Lima, V.C.C.; et al. Fusion Cell Markers in Circulating Tumor Cells from Patients with High-Grade Ovarian Serous Carcinoma. Int. J. Mol. Sci. 2022, 23, 14687. https://doi.org/10.3390/ijms232314687
Ruano APC, Gadelha Guimarães AP, Braun AC, Flores BCTCP, Tariki MS, Abdallah EA, Torres JA, Nunes DN, Tirapelli B, de Lima VCC, et al. Fusion Cell Markers in Circulating Tumor Cells from Patients with High-Grade Ovarian Serous Carcinoma. International Journal of Molecular Sciences. 2022; 23(23):14687. https://doi.org/10.3390/ijms232314687
Chicago/Turabian StyleRuano, Anna Paula Carreta, Andrea Paiva Gadelha Guimarães, Alexcia C. Braun, Bianca C. T. C. P. Flores, Milena Shizue Tariki, Emne A. Abdallah, Jacqueline Aparecida Torres, Diana Noronha Nunes, Bruna Tirapelli, Vladmir C. Cordeiro de Lima, and et al. 2022. "Fusion Cell Markers in Circulating Tumor Cells from Patients with High-Grade Ovarian Serous Carcinoma" International Journal of Molecular Sciences 23, no. 23: 14687. https://doi.org/10.3390/ijms232314687
APA StyleRuano, A. P. C., Gadelha Guimarães, A. P., Braun, A. C., Flores, B. C. T. C. P., Tariki, M. S., Abdallah, E. A., Torres, J. A., Nunes, D. N., Tirapelli, B., de Lima, V. C. C., Fanelli, M. F., Colombo, P.-E., da Costa, A. A. B. A., Alix-Panabières, C., & Chinen, L. T. D. (2022). Fusion Cell Markers in Circulating Tumor Cells from Patients with High-Grade Ovarian Serous Carcinoma. International Journal of Molecular Sciences, 23(23), 14687. https://doi.org/10.3390/ijms232314687







