Role of YAP as a Mechanosensing Molecule in Stem Cells and Stem Cell-Derived Hematopoietic Cells
Abstract
1. Introduction
1.1. The Hippo-YAP/TAZ Signaling Pathway
1.2. Stem Cells
2. Mechanosensing and Forces Regulating YAP/TAZ
2.1. ECM Stiffness Influences MSC Differentiation via YAP/TAZ
2.2. Fluid Shear Stress and Force Modulate YAP/TAZ Activity
3. YAP Regulates the Self-Renewal, Pluripotency, and Differentiation of PSCs, Dependent on ECM Stiffness and Cytoskeleton Tension
3.1. Role of YAP in Stem Cell Pluripotency, Self-Renewal, and Differentiation
3.2. ECM Stiffness and Cytoskeleton Tension Influence YAP Activity-Mediated Stem Cell Differentiation
3.3. Small Molecules Targeting YAP/TAZ Activity via the Actin Cytoskeleton
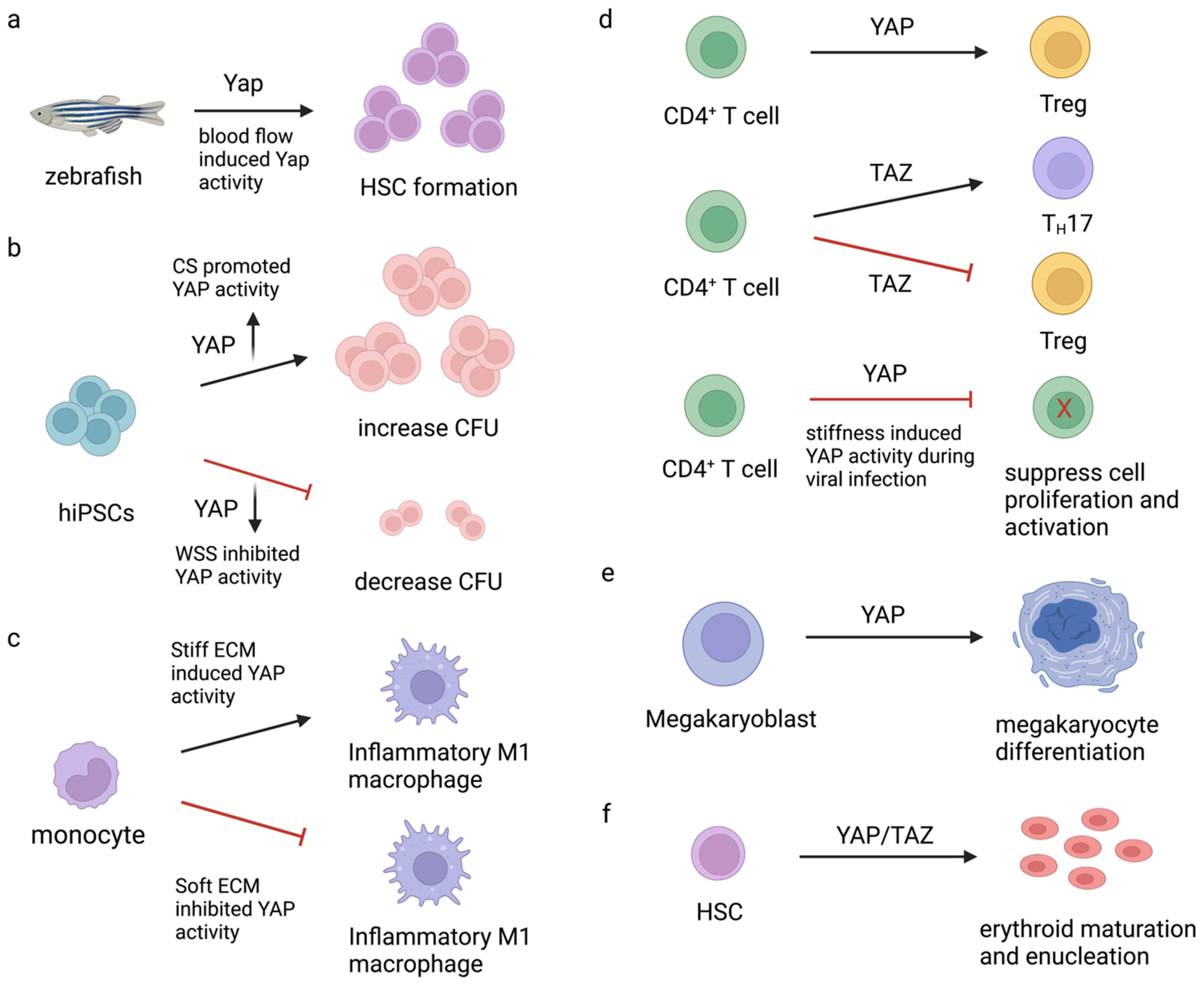
4. Role of YAP during HSC Formation and Blood Cell Production
4.1. Role of YAP during HSC Formation
4.2. Role of YAP in Myeloid and Lymphoid Lineage Development
4.2.1. Role of YAP in T-Cell Development and Activation
4.2.2. Role of YAP in Megakaryocyte Differentiation and Platelet Production
4.2.3. Role of YAP in Red Blood Cell Maturation and Enucleation
4.3. ECM Stiffness and Mechanical Force Affect YAP Activity during HSC Differentiation
4.4. Effect of Blood Flow on YAP in Endothelial Cell Maintenance, Diseases, and Anti-Inflammatory Effects
5. Concluding Remarks and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gattazzo, F.; Urciuolo, A.; Bonaldo, P. Extracellular matrix: A dynamic microenvironment for stem cell niche. Biochim. Biophys. Acta 2014, 1840, 2506–2519. [Google Scholar] [CrossRef] [PubMed]
- Kothapalli, C.; Mahajan, G.; Farrell, K. Substrate stiffness induced mechanotransduction regulates temporal evolution of human fetal neural progenitor cell phenotype, differentiation, and biomechanics. Biomater. Sci. 2020, 8, 5452–5464. [Google Scholar] [CrossRef] [PubMed]
- Selig, M.; Lauer, J.C.; Hart, M.L. Mechanotransduction and Stiffness-Sensing: Mechanisms and Opportunities to Control Multiple Molecular Aspects of Cell Phenotype as a Design Cornerstone of Cell-Instructive Biomaterials for Articular Cartilage Repair. Int. J. Mol. Sci. 2020, 21, 5399. [Google Scholar] [CrossRef] [PubMed]
- Pan, D. The unfolding of the Hippo signaling pathway. Dev. Biol. 2022, 487, 1–9. [Google Scholar] [CrossRef]
- Pan, D. The hippo signaling pathway in development and cancer. Dev. Cell 2010, 19, 491–505. [Google Scholar] [CrossRef]
- Huang, J.; Wu, S.; Barrera, J.; Matthews, K.; Pan, D. The Hippo signaling pathway coordinately regulates cell proliferation and apoptosis by inactivating Yorkie, the Drosophila Homolog of YAP. Cell 2005, 122, 421–434. [Google Scholar] [CrossRef]
- Tarkowski, A.K.; Wroblewska, J. Development of blastomeres of mouse eggs isolated at the 4- and 8-cell stage. J. Embryol. Exp. Morphol. 1967, 18, 155–180. [Google Scholar] [CrossRef]
- Kimber, S.J.; Surani, M.A.; Barton, S.C. Interactions of blastomeres suggest changes in cell surface adhesiveness during the formation of inner cell mass and trophectoderm in the preimplantation mouse embryo. J. Embryol. Exp. Morphol. 1982, 70, 133–152. [Google Scholar] [CrossRef]
- Lorthongpanich, C.; Doris, T.P.; Limviphuvadh, V.; Knowles, B.B.; Solter, D. Developmental fate and lineage commitment of singled mouse blastomeres. Development 2012, 139, 3722–3731. [Google Scholar] [CrossRef]
- Lorthongpanich, C.; Issaragrisil, S. Emerging Role of the Hippo Signaling Pathway in Position Sensing and Lineage Specification in Mammalian Preimplantation Embryos. Biol. Reprod. 2015, 92, 143. [Google Scholar] [CrossRef]
- Lorthongpanich, C.; Messerschmidt, D.M.; Chan, S.W.; Hong, W.; Knowles, B.B.; Solter, D. Temporal reduction of LATS kinases in the early preimplantation embryo prevents ICM lineage differentiation. Genes Dev. 2013, 27, 1441–1446. [Google Scholar] [CrossRef]
- Nishioka, N.; Inoue, K.; Adachi, K.; Kiyonari, H.; Ota, M.; Ralston, A.; Yabuta, N.; Hirahara, S.; Stephenson, R.O.; Ogonuki, N.; et al. The Hippo signaling pathway components Lats and Yap pattern Tead4 activity to distinguish mouse trophectoderm from inner cell mass. Dev. Cell 2009, 16, 398–410. [Google Scholar] [CrossRef]
- Dupont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S.; et al. Role of YAP/TAZ in mechanotransduction. Nature 2011, 474, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.C.; Wu, J.W.; Wang, C.W.; Jang, A.C. Hippo Signaling-Mediated Mechanotransduction in Cell Movement and Cancer Metastasis. Front. Mol. Biosci. 2019, 6, 157. [Google Scholar] [CrossRef]
- Xu, T.; Wang, W.; Zhang, S.; Stewart, R.A.; Yu, W. Identifying tumor suppressors in genetic mosaics: The Drosophila lats gene encodes a putative protein kinase. Development 1995, 121, 1053–1063. [Google Scholar] [CrossRef] [PubMed]
- Justice, R.W.; Zilian, O.; Woods, D.F.; Noll, M.; Bryant, P.J. The Drosophila tumor suppressor gene warts encodes a homolog of human myotonic dystrophy kinase and is required for the control of cell shape and proliferation. Genes Dev. 1995, 9, 534–546. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Huang, J.; Dong, J.; Pan, D. hippo encodes a Ste-20 family protein kinase that restricts cell proliferation and promotes apoptosis in conjunction with salvador and warts. Cell 2003, 114, 445–456. [Google Scholar] [CrossRef]
- Dong, J.; Feldmann, G.; Huang, J.; Wu, S.; Zhang, N.; Comerford, S.A.; Gayyed, M.F.; Anders, R.A.; Maitra, A.; Pan, D. Elucidation of a universal size-control mechanism in Drosophila and mammals. Cell 2007, 130, 1120–1133. [Google Scholar] [CrossRef]
- Zhao, B.; Wei, X.; Li, W.; Udan, R.S.; Yang, Q.; Kim, J.; Xie, J.; Ikenoue, T.; Yu, J.; Li, L.; et al. Inactivation of YAP oncoprotein by the Hippo pathway is involved in cell contact inhibition and tissue growth control. Genes Dev. 2007, 21, 2747–2761. [Google Scholar] [CrossRef]
- Pan, D.J. Hippo signaling in organ size control. Genes Dev. 2007, 21, 886–897. [Google Scholar] [CrossRef]
- Camargo, F.D.; Gokhale, S.; Johnnidis, J.B.; Fu, D.; Bell, G.W.; Jaenisch, R.; Brummelkamp, T.R. YAP1 increases organ size and expands undifferentiated progenitor cells. Curr. Biol. 2007, 17, 2054–2060. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Li, L.; Lei, Q.; Guan, K.L. The Hippo-YAP pathway in organ size control and tumorigenesis: An updated version. Genes Dev. 2010, 24, 862–874. [Google Scholar] [CrossRef] [PubMed]
- Yagi, R.; Chen, L.F.; Shigesada, K.; Murakami, Y.; Ito, Y.A. A WW domain-containing yes-associated protein (YAP) is a novel transcriptional co-activator. EMBO J. 1999, 18, 2551–2562. [Google Scholar] [CrossRef] [PubMed]
- Kanai, F.; Marignani, P.A.; Sarbassova, D.; Yagi, R.; Hall, R.A.; Donowitz, M.; Hisaminato, A.; Fujiwara, T.; Ito, Y.T.; Cantley, L.C.; et al. TAZ: A novel transcriptional co-activator regulated by interactions with 14-3-3 and PDZ domain proteins. EMBO J. 2000, 19, 6778–6791. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Taouk, G.M. A Potential Role of YAP/TAZ in the Interplay Between Metastasis and Metabolic Alterations. Front. Oncol. 2020, 10, 928. [Google Scholar] [CrossRef]
- Sun, X.; Ren, Z.; Cun, Y.; Zhao, C.; Huang, X.; Zhou, J.; Hu, R.; Su, X.; Ji, L.; Li, P.; et al. Hippo-YAP signaling controls lineage differentiation of mouse embryonic stem cells through modulating the formation of super-enhancers. Nucleic Acids Res. 2020, 48, 7182–7196. [Google Scholar] [CrossRef]
- Plouffe, S.W.; Lin, K.C.; Moore, J.L., 3rd; Tan, F.E.; Ma, S.; Ye, Z.; Qiu, Y.; Ren, B.; Guan, K.L. The Hippo pathway effector proteins YAP and TAZ have both distinct and overlapping functions in the cell. J. Biol. Chem. 2018, 293, 11230–11240. [Google Scholar] [CrossRef]
- Zhao, B.; Ye, X.; Yu, J.; Li, L.; Li, W.; Li, S.; Yu, J.; Lin, J.D.; Wang, C.Y.; Chinnaiyan, A.M.; et al. TEAD mediates YAP-dependent gene induction and growth control. Genes Dev. 2008, 22, 1962–1971. [Google Scholar] [CrossRef]
- Stein, C.; Bardet, A.F.; Roma, G.; Bergling, S.; Clay, I.; Ruchti, A.; Agarinis, C.; Schmelzle, T.; Bouwmeester, T.; Schübeler, D.; et al. YAP1 Exerts Its Transcriptional Control via TEAD-Mediated Activation of Enhancers. PLoS Genet. 2015, 11, e1005465. [Google Scholar] [CrossRef]
- Zanconato, F.; Forcato, M.; Battilana, G.; Azzolin, L.; Quaranta, E.; Bodega, B.; Rosato, A.; Bicciato, S.; Cordenonsi, M.; Piccolo, S. Genome-wide association between YAP/TAZ/TEAD and AP-1 at enhancers drives oncogenic growth. Nat. Cell Biol. 2015, 17, 1218–1227. [Google Scholar] [CrossRef]
- Zhang, J.M.; Smolen, G.A.; Haber, D.A. Negative regulation of YAP by LATS1 underscores evolutionary conservation of the Drosophila Hippo pathway. Cancer Res. 2008, 68, 2789–2794. [Google Scholar] [CrossRef]
- Varelas, X. The Hippo pathway effectors TAZ and YAP in development, homeostasis and disease. Development 2014, 141, 1614–1626. [Google Scholar] [CrossRef] [PubMed]
- Piccolo, S.; Dupont, S.; Cordenonsi, M. The biology of YAP/TAZ: Hippo signaling and beyond. Physiol. Rev. 2014, 94, 1287–1312. [Google Scholar] [CrossRef]
- Ma, S.; Meng, Z.; Chen, R.; Guan, K.L. The Hippo Pathway: Biology and Pathophysiology. Annu. Rev. Biochem. 2019, 88, 577–604. [Google Scholar] [CrossRef] [PubMed]
- Zakrzewski, W.; Dobrzyński, M.; Szymonowicz, M.; Rybak, Z. Stem cells: Past, present, and future. Stem Cell Res. Ther. 2019, 10, 68. [Google Scholar] [CrossRef]
- Singh, P.P.; Singh, S. Statins—The Holy Grail for cancer? Ann. Transl. Med. 2013, 1, 1. [Google Scholar]
- Dvash, T.; Ben-Yosef, D.; Eiges, R. Human embryonic stem cells as a powerful tool for studying human embryogenesis. Pediatr. Res. 2006, 60, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Okita, K.; Nakagawa, M.; Yamanaka, S. Induction of pluripotent stem cells from fibroblast cultures. Nat. Protoc. 2007, 2, 3081–3089. [Google Scholar] [CrossRef]
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage potential of adult human mesenchymal stem cells. Science 1999, 284, 143–147. [Google Scholar] [CrossRef]
- Caplan, A.I.; Bruder, S.P. Mesenchymal stem cells: Building blocks for molecular medicine in the 21st century. Trends Mol. Med. 2001, 7, 259–264. [Google Scholar] [CrossRef]
- Engler, A.J.; Sen, S.; Sweeney, H.L.; Discher, D.E. Matrix elasticity directs stem cell lineage specification. Cell 2006, 126, 677–689. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Petersen, B.E.; Steindler, D.A.; Jorgensen, M.L.; Laywell, E.D. Mesenchymal stem cells spontaneously express neural proteins in culture and are neurogenic after transplantation. Stem Cells 2006, 24, 1054–1064. [Google Scholar] [CrossRef] [PubMed]
- Manochantr, S.; Marupanthorn, K.; Tantrawatpan, C.; Kheolamai, P. The expression of neurogenic markers after neuronal induction of chorion-derived mesenchymal stromal cells. Neurol. Res. 2015, 37, 545–552. [Google Scholar] [CrossRef]
- Gu, W.; Hong, X.; Le Bras, A.; Nowak, W.N.; Issa Bhaloo, S.; Deng, J.; Xie, Y.; Hu, Y.; Ruan, X.Z.; Xu, Q. Smooth muscle cells differentiated from mesenchymal stem cells are regulated by microRNAs and suitable for vascular tissue grafts. J. Biol. Chem. 2018, 293, 8089–8102. [Google Scholar] [CrossRef]
- Gong, Z.; Niklason, L.E. Small-diameter human vessel wall engineered from bone marrow-derived mesenchymal stem cells (hMSCs). FASEB J. 2008, 22, 1635–1648. [Google Scholar] [CrossRef] [PubMed]
- Bagno, L.; Hatzistergos, K.E.; Balkan, W.; Hare, J.M. Mesenchymal Stem Cell-Based Therapy for Cardiovascular Disease: Progress and Challenges. Mol. Ther. 2018, 26, 1610–1623. [Google Scholar] [CrossRef]
- Serwold, T.; Ehrlich, L.I.; Weissman, I.L. Reductive isolation from bone marrow and blood implicates common lymphoid progenitors as the major source of thymopoiesis. Blood 2009, 113, 807–815. [Google Scholar] [CrossRef]
- Karsunky, H.; Inlay, M.A.; Serwold, T.; Bhattacharya, D.; Weissman, I.L. Flk2+ common lymphoid progenitors possess equivalent differentiation potential for the B and T lineages. Blood 2008, 111, 5562–5570. [Google Scholar] [CrossRef]
- Kondo, M.; Weissman, I.L.; Akashi, K. Identification of clonogenic common lymphoid progenitors in mouse bone marrow. Cell 1997, 91, 661–672. [Google Scholar] [CrossRef]
- Akashi, K.; Traver, D.; Miyamoto, T.; Weissman, I.L. A clonogenic common myeloid progenitor that gives rise to all myeloid lineages. Nature 2000, 404, 193–197. [Google Scholar] [CrossRef]
- Nakorn, T.N.; Miyamoto, T.; Weissman, I.L. Characterization of mouse clonogenic megakaryocyte progenitors. Proc. Natl. Acad. Sci. USA 2003, 100, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Pronk, C.J.; Rossi, D.J.; Månsson, R.; Attema, J.L.; Norddahl, G.L.; Chan, C.K.; Sigvardsson, M.; Weissman, I.L.; Bryder, D. Elucidation of the phenotypic, functional, and molecular topography of a myeloerythroid progenitor cell hierarchy. Cell Stem Cell 2007, 1, 428–442. [Google Scholar] [CrossRef]
- Haas, S.; Trumpp, A.; Milsom, M.D. Causes and Consequences of Hematopoietic Stem Cell Heterogeneity. Cell Stem Cell 2018, 22, 627–638. [Google Scholar] [CrossRef] [PubMed]
- Mercier, F.E.; Scadden, D.T. Not All Created Equal: Lineage Hard-Wiring in the Production of Blood. Cell 2015, 163, 1568–1570. [Google Scholar] [CrossRef] [PubMed]
- Notta, F.; Zandi, S.; Takayama, N.; Dobson, S.; Gan, O.I.; Wilson, G.; Kaufmann, K.B.; McLeod, J.; Laurenti, E.; Dunant, C.F.; et al. Distinct routes of lineage development reshape the human blood hierarchy across ontogeny. Science 2016, 351, aab2116. [Google Scholar] [CrossRef] [PubMed]
- Paul, F.; Arkin, Y.; Giladi, A.; Jaitin, D.A.; Kenigsberg, E.; Keren-Shaul, H.; Winter, D.; Lara-Astiaso, D.; Gury, M.; Weiner, A.; et al. Transcriptional Heterogeneity and Lineage Commitment in Myeloid Progenitors. Cell 2015, 163, 1663–1677. [Google Scholar] [CrossRef]
- Velten, L.; Haas, S.F.; Raffel, S.; Blaszkiewicz, S.; Islam, S.; Hennig, B.P.; Hirche, C.; Lutz, C.; Buss, E.C.; Nowak, D.; et al. Human haematopoietic stem cell lineage commitment is a continuous process. Nat. Cell Biol. 2017, 19, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Macaulay, I.C.; Svensson, V.; Labalette, C.; Ferreira, L.; Hamey, F.; Voet, T.; Teichmann, S.A.; Cvejic, A. Single-Cell RNA-Sequencing Reveals a Continuous Spectrum of Differentiation in Hematopoietic Cells. Cell Rep. 2016, 14, 966–977. [Google Scholar] [CrossRef]
- Nestorowa, S.; Hamey, F.K.; Pijuan Sala, B.; Diamanti, E.; Shepherd, M.; Laurenti, E.; Wilson, N.K.; Kent, D.G.; Göttgens, B. A single-cell resolution map of mouse hematopoietic stem and progenitor cell differentiation. Blood 2016, 128, e20–e31. [Google Scholar] [CrossRef]
- Pina, C.; Fugazza, C.; Tipping, A.J.; Brown, J.; Soneji, S.; Teles, J.; Peterson, C.; Enver, T. Inferring rules of lineage commitment in haematopoiesis. Nat. Cell Biol. 2012, 14, 287–294. [Google Scholar] [CrossRef]
- Wattanapanitch, M.; Damkham, N.; Potirat, P.; Trakarnsanga, K.; Janan, M.; U-Pratya, Y.; Kheolamai, P.; Klincumhom, N.; Issaragrisil, S. One-step genetic correction of hemoglobin E/beta-thalassemia patient-derived iPSCs by the CRISPR/Cas9 system. Stem Cell Res. Ther. 2018, 9, 46. [Google Scholar] [CrossRef] [PubMed]
- Laowtammathron, C.; Chingsuwanrote, P.; Choavaratana, R.; Phornwilardsiri, S.; Sitthirit, K.; Kaewjunun, C.; Makemaharn, O.; Terbto, P.; Waeteekul, S.; Lorthongpanich, C.; et al. High-efficiency derivation of human embryonic stem cell lines using a culture system with minimized trophoblast cell proliferation. Stem Cell Res. Ther. 2018, 9, 138. [Google Scholar] [CrossRef] [PubMed]
- Ebert, A.D.; Liang, P.; Wu, J.C. Induced pluripotent stem cells as a disease modeling and drug screening platform. J. Cardiovasc. Pharmacol. 2012, 60, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Han, Y.; Cao, X. Induced pluripotent stem cell (iPSCs) and their application in immunotherapy. Cell Mol. Immunol. 2014, 11, 17–24. [Google Scholar] [CrossRef]
- Doss, M.X.; Sachinidis, A. Current Challenges of iPSC-Based Disease Modeling and Therapeutic Implications. Cells 2019, 8, 403. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, S.; Wang, T. How the mechanical microenvironment of stem cell growth affects their differentiation: A review. Stem Cell Res. Ther. 2022, 13, 415. [Google Scholar] [CrossRef]
- Cobbaut, M.; Karagil, S.; Bruno, L.; Diaz de la Loza, M.D.C.; Mackenzie, F.E.; Stolinski, M.; Elbediwy, A. Dysfunctional Mechanotransduction through the YAP/TAZ/Hippo Pathway as a Feature of Chronic Disease. Cells 2020, 9, 151. [Google Scholar] [CrossRef]
- Hwang, J.H.; Byun, M.R.; Kim, A.R.; Kim, K.M.; Cho, H.J.; Lee, Y.H.; Kim, J.; Jeong, M.G.; Hwang, E.S.; Hong, J.H. Extracellular Matrix Stiffness Regulates Osteogenic Differentiation through MAPK Activation. PLoS ONE 2015, 10, e0135519. [Google Scholar] [CrossRef]
- Hsiao, C.; Lampe, M.; Nillasithanukroh, S.; Han, W.; Lian, X.; Palecek, S.P. Human pluripotent stem cell culture density modulates YAP signaling. Biotechnol. J. 2016, 11, 662–675. [Google Scholar] [CrossRef]
- Calvo, F.; Ege, N.; Grande-Garcia, A.; Hooper, S.; Jenkins, R.P.; Chaudhry, S.I.; Harrington, K.; Williamson, P.; Moeendarbary, E.; Charras, G.; et al. Mechanotransduction and YAP-dependent matrix remodelling is required for the generation and maintenance of cancer-associated fibroblasts. Nat. Cell Biol. 2013, 15, 637–646. [Google Scholar] [CrossRef]
- Halder, G.; Dupont, S.; Piccolo, S. Transduction of mechanical and cytoskeletal cues by YAP and TAZ. Nat. Rev. Mol. Cell Biol. 2012, 13, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Panciera, T.; Azzolin, L.; Cordenonsi, M.; Piccolo, S. Mechanobiology of YAP and TAZ in physiology and disease. Nat. Rev. Mol. Cell Biol. 2017, 18, 758–770. [Google Scholar] [CrossRef] [PubMed]
- Lorthongpanich, C.; Thumanu, K.; Tangkiettrakul, K.; Jiamvoraphong, N.; Laowtammathron, C.; Damkham, N.; U-Pratya, Y.; Issaragrisil, S. YAP as a key regulator of adipo-osteogenic differentiation in human MSCs. Stem Cell Res. Ther. 2019, 10, 402. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Zhou, Y.; Lee, M.S.; Zhang, Y.; Li, W.J. A newly identified mechanism involved in regulation of human mesenchymal stem cells by fibrous substrate stiffness. Acta Biomater. 2016, 42, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Tibbitt, M.W.; Basta, L.; Anseth, K.S. Mechanical memory and dosing influence stem cell fate. Nat. Mater. 2014, 13, 645–652. [Google Scholar] [CrossRef]
- Olivares-Navarrete, R.; Lee, E.M.; Smith, K.; Hyzy, S.L.; Doroudi, M.; Williams, J.K.; Gall, K.; Boyan, B.D.; Schwartz, Z. Substrate Stiffness Controls Osteoblastic and Chondrocytic Differentiation of Mesenchymal Stem Cells without Exogenous Stimuli. PLoS ONE 2017, 12, e0170312. [Google Scholar] [CrossRef]
- Karystinou, A.; Roelofs, A.J.; Neve, A.; Cantatore, F.P.; Wackerhage, H.; De Bari, C. Yes-associated protein (YAP) is a negative regulator of chondrogenesis in mesenchymal stem cells. Arthritis Res. Ther. 2015, 17, 147. [Google Scholar] [CrossRef]
- Deng, Y.; Wu, A.; Li, P.; Li, G.; Qin, L.; Song, H.; Mak, K.K. Yap1 Regulates Multiple Steps of Chondrocyte Differentiation during Skeletal Development and Bone Repair. Cell Rep. 2016, 14, 2224–2237. [Google Scholar] [CrossRef]
- Yang, B.; Sun, H.; Song, F.; Yu, M.; Wu, Y.; Wang, J. YAP1 negatively regulates chondrocyte differentiation partly by activating the beta-catenin signaling pathway. Int. J. Biochem. Cell Biol. 2017, 87, 104–113. [Google Scholar] [CrossRef]
- Li, Y.; Yang, S.; Qin, L.; Yang, S. TAZ is required for chondrogenesis and skeletal development. Cell Discov. 2021, 7, 26. [Google Scholar] [CrossRef]
- Zhong, W.; Tian, K.; Zheng, X.; Li, L.; Zhang, W.; Wang, S.; Qin, J. Mesenchymal stem cell and chondrocyte fates in a multishear microdevice are regulated by Yes-associated protein. Stem Cells Dev. 2013, 22, 2083–2093. [Google Scholar] [CrossRef] [PubMed]
- Klincumhom, N.; Lorthongpanich, C.; Thumanu, K.; Septham, P.; Phomyu, W.; Issaragrisil, S.; Pavasant, P. Intermittent compressive force regulates human periodontal ligament cell behavior via yes-associated protein. Heliyon 2022, 8, e10845. [Google Scholar] [CrossRef] [PubMed]
- Nieto-Miguel, T.; Galindo, S.; Reinoso, R.; Corell, A.; Martino, M.; Pérez-Simón, J.A.; Calonge, M. In vitro simulation of corneal epithelium microenvironment induces a corneal epithelial-like cell phenotype from human adipose tissue mesenchymal stem cells. Curr. Eye Res. 2013, 38, 933–944. [Google Scholar] [CrossRef] [PubMed]
- Chavez-Munoz, C.; Nguyen, K.T.; Xu, W.; Hong, S.J.; Mustoe, T.A.; Galiano, R.D. Transdifferentiation of adipose-derived stem cells into keratinocyte-like cells: Engineering a stratified epidermis. PLoS ONE 2013, 8, e80587. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.C.; Yeh, Y.T.; Nguyen, P.; Limqueco, E.; Lopez, J.; Thorossian, S.; Guan, K.L.; Li, Y.J.; Chien, S. Flow-dependent YAP/TAZ activities regulate endothelial phenotypes and atherosclerosis. Proc. Natl. Acad. Sci. USA 2016, 113, 11525–11530. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, H.; Yamamoto, K.; Agarwala, S.; Terai, K.; Fukui, H.; Fukuhara, S.; Ando, K.; Miyazaki, T.; Yokota, Y.; Schmelzer, E.; et al. Flow-Dependent Endothelial YAP Regulation Contributes to Vessel Maintenance. Dev. Cell 2017, 40, 523–536. [Google Scholar] [CrossRef] [PubMed]
- Lundin, V.; Sugden, W.W.; Theodore, L.N.; Sousa, P.M.; Han, A.; Chou, S.; Wrighton, P.J.; Cox, A.G.; Ingber, D.E.; Goessling, W.; et al. YAP Regulates Hematopoietic Stem Cell Formation in Response to the Biomechanical Forces of Blood Flow. Dev. Cell 2020, 52, 446–460. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Luo, J.Y.; Li, B.; Tian, X.Y.; Chen, L.J.; Huang, Y.; Liu, J.; Deng, D.; Lau, C.W.; Wan, S.; et al. Integrin-YAP/TAZ-JNK cascade mediates atheroprotective effect of unidirectional shear flow. Nature 2016, 540, 579–582. [Google Scholar] [CrossRef] [PubMed]
- Gumbiner, B.M. and N.G. Kim, The Hippo-YAP signaling pathway and contact inhibition of growth. J. Cell Sci. 2014, 127 Pt 4, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Heng, B.C.; Zhang, X.; Aubel, D.; Bai, Y.; Li, X.; Wei, Y.; Fussenegger, M.; Deng, X. Role of YAP/TAZ in Cell Lineage Fate Determination and Related Signaling Pathways. Front. Cell Dev. Biol. 2020, 8, 735. [Google Scholar] [CrossRef] [PubMed]
- Lian, I.; Kim, J.; Okazawa, H.; Zhao, J.; Zhao, B.; Yu, J.; Chinnaiyan, A.; Israel, M.A.; Goldstein, L.S.; Abujarour, R.; et al. The role of YAP transcription coactivator in regulating stem cell self-renewal and differentiation. Genes Dev. 2010, 24, 1106–1118. [Google Scholar] [CrossRef]
- Tamm, C.; Bower, N.; Anneren, C. Regulation of mouse embryonic stem cell self-renewal by a Yes-YAP-TEAD2 signaling pathway downstream of LIF. J. Cell Sci. 2011, 124 Pt 7, 1136–1144. [Google Scholar] [CrossRef] [PubMed]
- Pijuan-Galito, S.; Tamm, C.; Anneren, C. Serum Inter-alpha-inhibitor activates the Yes tyrosine kinase and YAP/TEAD transcriptional complex in mouse embryonic stem cells. J. Biol. Chem. 2014, 289, 33492–33502. [Google Scholar] [CrossRef] [PubMed]
- Lorthongpanich, C.; Jiamvoraphong, N.; Klaihmon, P.; Lueangamornnara, U.; U-Pratya, Y.; Laowtammathron, C.; Issaragrisil, S. Effect of YAP/TAZ on megakaryocyte differentiation and platelet production. Biosci. Rep. 2020, 40, BSR20201780. [Google Scholar] [CrossRef] [PubMed]
- Varelas, X.; Sakuma, R.; Samavarchi-Tehrani, P.; Peerani, R.; Rao, B.M.; Dembowy, J.; Yaffe, M.B.; Zandstra, P.W.; Wrana, J.L. TAZ controls Smad nucleocytoplasmic shuttling and regulates human embryonic stem-cell self-renewal. Nat. Cell Biol. 2008, 10, 837–848. [Google Scholar] [CrossRef]
- Musah, S.; Wrighton, P.J.; Zaltsman, Y.; Zhong, X.; Zorn, S.; Parlato, M.B.; Hsiao, C.; Palecek, S.P.; Chang, Q.; Murphy, W.L.; et al. Substratum-induced differentiation of human pluripotent stem cells reveals the coactivator YAP is a potent regulator of neuronal specification. Proc. Natl. Acad. Sci. USA 2014, 111, 13805–13810. [Google Scholar] [CrossRef]
- Ohgushi, M.; Minaguchi, M.; Sasai, Y. Rho-Signaling-Directed YAP/TAZ Activity Underlies the Long-Term Survival and Expansion of Human Embryonic Stem Cells. Cell Stem Cell 2015, 17, 448–461. [Google Scholar] [CrossRef]
- Zhao, Y.; Fei, X.; Guo, J.; Zou, G.; Pan, W.; Zhang, J.; Huang, Y.; Liu, T.; Cheng, W. Induction of reprogramming of human amniotic epithelial cells into iPS cells by overexpression of Yap, Oct4, and Sox2 through the activation of the Hippo-Yap pathway. Exp. Ther. Med. 2017, 14, 199–206. [Google Scholar] [CrossRef]
- Lorthongpanich, C.; Jiamvoraphong, N.; Supakun, P.; Damkham, N.; Terbto, P.; Waeteekul, S.; U-Pratya, Y.; Laowtammathron, C.; Issaragrisil, S. Generation of a WWTR1 mutation induced pluripotent stem cell line, MUSIi012-A-1, using CRISPR/Cas9. Stem Cell Res. 2019, 41, 101634. [Google Scholar] [CrossRef]
- Lorthongpanich, C.; Laowtammathron, C.; Jiamvoraphong, N.; Srisook, P.; Chingsuwanrote, P.; Klaihmon, P.; Damkham, N.; Terbto, P.; Waeteekul, S.; U-Pratya, Y.; et al. YAP-depleted iPSC MUSIi012-A-2 maintained all normal stem cell characteristics. Stem Cell Res. 2020, 43, 101723. [Google Scholar] [CrossRef]
- Qin, H.; Hejna, M.; Liu, Y.; Percharde, M.; Wossidlo, M.; Blouin, L.; Durruthy-Durruthy, J.; Wong, P.; Qi, Z.; Yu, J.; et al. YAP Induces Human Naive Pluripotency. Cell Rep. 2016, 14, 2301–2312. [Google Scholar] [CrossRef] [PubMed]
- Kumari, D. States of pluripotency: Naïve and primed pluripotent stem cells. In Pluripotent Stem Cells—From the Bench to the Clinic; Books on Demand: Norderstedt, Germany, 2016; pp. 31–45. [Google Scholar]
- Gafni, O.; Weinberger, L.; Mansour, A.A.; Manor, Y.S.; Chomsky, E.; Ben-Yosef, D.; Kalma, Y.; Viukov, S.; Maza, I.; Zviran, A.; et al. Derivation of novel human ground state naive pluripotent stem cells. Nature 2013, 504, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Musah, S.; Morin, S.A.; Wrighton, P.J.; Zwick, D.B.; Jin, S.; Kiessling, L.L. Glycosaminoglycan-binding hydrogels enable mechanical control of human pluripotent stem cell self-renewal. ACS Nano 2012, 6, 10168–10177. [Google Scholar] [CrossRef] [PubMed]
- Abagnale, G.; Sechi, A.; Steger, M.; Zhou, Q.; Kuo, C.C.; Aydin, G.; Schalla, C.; Müller-Newen, G.; Zenke, M.; Costa, I.G.; et al. Surface Topography Guides Morphology and Spatial Patterning of Induced Pluripotent Stem Cell Colonies. Stem Cell Rep. 2017, 9, 654–666. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Guan, K.L. Hippo Signaling in Embryogenesis and Development. Trends Biochem. Sci. 2021, 46, 51–63. [Google Scholar] [CrossRef]
- Kumari, S.; Vermeulen, S.; van der Veer, B.; Carlier, A.; de Boer, J.; Subramanyam, D. Shaping Cell Fate: Influence of Topographical Substratum Properties on Embryonic Stem Cells. Tissue Eng. Part B Rev. 2018, 24, 255–266. [Google Scholar] [CrossRef]
- Li, Y.; Wang, J.; Zhong, W. Regulation and mechanism of YAP/TAZ in the mechanical microenvironment of stem cells (Review). Mol. Med. Rep. 2021, 24, 506. [Google Scholar] [CrossRef]
- Lee, S.; Stanton, A.E.; Tong, X.; Yang, F. Hydrogels with enhanced protein conjugation efficiency reveal stiffness-induced YAP localization in stem cells depends on biochemical cues. Biomaterials 2019, 202, 26–34. [Google Scholar] [CrossRef]
- Sun, Y.; Yong, K.M.; Villa-Diaz, L.G.; Zhang, X.; Chen, W.; Philson, R.; Weng, S.; Xu, H.; Krebsbach, P.H.; Fu, J. Hippo/YAP-mediated rigidity-dependent motor neuron differentiation of human pluripotent stem cells. Nat. Mater. 2014, 13, 599–604. [Google Scholar] [CrossRef]
- Yamashita, A.; Yoshitomi, H.; Kihara, S.; Toguchida, J.; Tsumaki, N. Culture substrate-associated YAP inactivation underlies chondrogenic differentiation of human induced pluripotent stem cells. Stem Cells Transl. Med. 2021, 10, 115–127. [Google Scholar] [CrossRef]
- Smith, Q.; Chan, X.Y.; Carmo, A.M.; Trempel, M.; Saunders, M.; Gerecht, S. Compliant substratum guides endothelial commitment from human pluripotent stem cells. Sci. Adv. 2017, 3, e1602883. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Wang, K.; Li, Y.; Yang, Y. Nanotopography promoted neuronal differentiation of human induced pluripotent stem cells. Colloids Surf. B Biointerfaces 2016, 148, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.; Cho, S.; Discher, D.E. Mechanosensing of matrix by stem cells: From matrix heterogeneity, contractility, and the nucleus in pore-migration to cardiogenesis and muscle stem cells in vivo. Semin. Cell Dev. Biol. 2017, 71, 84–98. [Google Scholar] [CrossRef] [PubMed]
- Mo, J.S.; Yu, F.X.; Gong, R.; Brown, J.H.; Guan, K.L. Regulation of the Hippo-YAP pathway by protease-activated receptors (PARs). Genes Dev. 2012, 26, 2138–2143. [Google Scholar] [CrossRef] [PubMed]
- Wada, K.; Itoga, K.; Okano, T.; Yonemura, S.; Sasaki, H. Hippo pathway regulation by cell morphology and stress fibers. Development 2011, 138, 3907–3914. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, F.; Xie, Z.; Chen, L.; Sinkemani, A.; Yu, H.; Wu, X. AMOT130 linking F-actin to YAP is involved in intervertebral disc degeneration. Cell Prolif. 2018, 51, e12492. [Google Scholar] [CrossRef] [PubMed]
- Nie, P.; Li, Y.; Suo, H.; Jiang, N.; Yu, D.; Fang, B. Dasatinib Promotes Chondrogenic Differentiation of Human Mesenchymal Stem Cells via the Src/Hippo-YAP Signaling Pathway. ACS Biomater. Sci. Eng. 2019, 5, 5255–5265. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.J.; Nakagawa, K.; Yang, Z.; Ikeda, M.; Withanage, K.; Ishigami-Yuasa, M.; Okuno, Y.; Hata, S.; Nishina, H.; Hata, Y. A cell-based assay to screen stimulators of the Hippo pathway reveals the inhibitory effect of dobutamine on the YAP-dependent gene transcription. J. Biochem. 2011, 150, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Driscoll, T.P.; Cosgrove, B.D.; Heo, S.J.; Shurden, Z.E.; Mauck, R.L. Cytoskeletal to Nuclear Strain Transfer Regulates YAP Signaling in Mesenchymal Stem Cells. Biophys. J. 2015, 108, 2783–2793. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.; Yang, J.; DeRan, M.; Wu, C.; Su, A.I.; Bonamy, G.M.; Liu, J.; Peters, E.C.; Wu, X. Identification of serum-derived sphingosine-1-phosphate as a small molecule regulator of YAP. Chem. Biol. 2012, 19, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Damkham, N.; Lorthongpanich, C.; Klaihmon, P.; Lueangamornnara, U.; Kheolamai, P.; Trakarnsanga, K.; Issaragrisil, S. YAP and TAZ play a crucial role in human erythrocyte maturation and enucleation. Stem Cell Res. Ther. 2022, 13, 467. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Kang, W.; Du, L.; Ge, S. Rho-kinase inhibitor Y-27632 facilitates the proliferation, migration and pluripotency of human periodontal ligament stem cells. J. Cell Mol. Med. 2017, 21, 3100–3112. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, G.B.; Martinez-Agosto, J.A. Kicking it up a Notch for the best in show: Scalloped leads Yorkie into the haematopoietic arena. Fly 2014, 8, 206–217. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, G.B.; Martinez-Agosto, J.A. Yorkie and Scalloped signaling regulates Notch-dependent lineage specification during Drosophila hematopoiesis. Curr. Biol. 2014, 24, 2665–2672. [Google Scholar] [CrossRef] [PubMed]
- Milton, C.C.; Grusche, F.A.; Degoutin, J.L.; Yu, E.; Dai, Q.; Lai, E.C.; Harvey, K.F. The Hippo pathway regulates hematopoiesis in Drosophila melanogaster. Curr. Biol. 2014, 24, 2673–2680. [Google Scholar] [CrossRef] [PubMed]
- Goode, D.K.; Obier, N.; Vijayabaskar, M.S.; Lie-A-Ling, M.; Lilly, A.J.; Hannah, R.; Lichtinger, M.; Batta, K.; Florkowska, M.; Patel, R.; et al. Dynamic Gene Regulatory Networks Drive Hematopoietic Specification and Differentiation. Dev. Cell 2016, 36, 572–587. [Google Scholar] [CrossRef] [PubMed]
- Althoff, M.J.; Nayak, R.C.; Hegde, S.; Wellendorf, A.M.; Bohan, B.; Filippi, M.D.; Xin, M.; Lu, Q.R.; Geiger, H.; Zheng, Y.; et al. Yap1-Scribble polarization is required for hematopoietic stem cell division and fate. Blood 2020, 136, 1824–1836. [Google Scholar] [CrossRef] [PubMed]
- Jansson, L.; Larsson, J. Normal Hematopoietic Stem Cell Function in Mice with Enforced Expression of the Hippo Signaling Effector YAP1. PLoS ONE 2012, 7, e32013. [Google Scholar] [CrossRef]
- Donato, E.; Biagioni, F.; Bisso, A.; Caganova, M.; Amati, B.; Campaner, S. YAP and TAZ are dispensable for physiological and malignant haematopoiesis. Leukemia 2018, 32, 2037–2040. [Google Scholar] [CrossRef]
- Lee, D.H.; Kim, T.S.; Lee, D.; Lim, D.S. Mammalian sterile 20 kinase 1 and 2 are important regulators of hematopoietic stem cells in stress condition. Sci. Rep. 2018, 8, 942. [Google Scholar] [CrossRef]
- Ni, X.H.; Tao, J.; Barbi, J.; Chen, Q.; Park, B.V.; Li, Z.; Zhang, N.; Lebid, A.; Ramaswamy, A.; Wei, P.; et al. YAP Is Essential for Treg-Mediated Suppression of Antitumor Immunity. Cancer Discov. 2018, 8, 1026–1043. [Google Scholar] [CrossRef] [PubMed]
- Geng, J.; Yu, S.; Zhao, H.; Sun, X.; Li, X.; Wang, P.; Xiong, X.; Hong, L.; Xie, C.; Gao, J.; et al. The transcriptional coactivator TAZ regulates reciprocal differentiation of T(H)17 cells and T-reg cells. Nat. Immunol. 2017, 18, 800–812. [Google Scholar] [CrossRef] [PubMed]
- Meng, K.P.; Majedi, F.S.; Thauland, T.J.; Butte, M.J. Mechanosensing through YAP controls T cell activation and metabolism. J. Exp. Med. 2020, 217, e20200053. [Google Scholar] [CrossRef] [PubMed]
- Lorthongpanich, C.; Jiamvoraphong, N.; Supraditaporn, K.; Klaihmon, P.; U-Pratya, Y.; Issaragrisil, S. The Hippo pathway regulates human megakaryocytic differentiation. Thromb. Haemost. 2017, 117, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Hao, S.; Matsui, Y.; Lai, Z.C.; Paulson, R.F. Yap1 promotes proliferation of transiently amplifying stress erythroid progenitors during erythroid regeneration. Exp. Hematol. 2019, 80, 42–54.e4. [Google Scholar] [CrossRef]
- Rausch, V.; Bostrom, J.R.; Park, J.; Bravo, I.R.; Feng, Y.; Hay, D.C.; Link, B.A.; Hansen, C.G. The Hippo Pathway Regulates Caveolae Expression and Mediates Flow Response via Caveolae. Curr. Biol. 2019, 29, 242–255.e6. [Google Scholar] [CrossRef]
- Rausch, V.; Hansen, C.G. The Hippo Pathway, YAP/TAZ, and the Plasma Membrane. Trends Cell Biol. 2020, 30, 32–48. [Google Scholar] [CrossRef]
- Meli, V.S.; Atcha, H.; Veerasubramanian, P.K.; Nagalla, R.R.; Luu, T.U.; Chen, E.Y.; Guerrero-Juarez, C.F.; Yamaga, K.; Pandori, W.; Hsieh, J.Y.; et al. YAP-mediated mechanotransduction tunes the macrophage inflammatory response. Sci. Adv. 2020, 6, eabb8471. [Google Scholar] [CrossRef]
- Klaihmon, P.; Lorthongpanich, C.; Kheolamai, P.; Luanpitpong, S.; Issaragrisil, S. Distinctive Roles of YAP and TAZ in Human Endothelial Progenitor Cells Growth and Functions. Biomedicines 2022, 10, 147. [Google Scholar] [CrossRef]




| Agents | Effectiveness | Cell Types | Treatment Results | References |
|---|---|---|---|---|
| Dasatinib | Chemotherapeutic agent | Human MSC-derived chondrocyte | YAP and TAZ inhibitors increased YAP phosphorylation at serine 127 | [118] |
| Dobutamine hydrochloride (DH) | YAP inhibition | Human osteoblastoma U2OS cells | Induced YAP phosphorylation at serine 127 | [119] |
| Lysophosphatidic acid (LPA) | YAP/TAZ nuclear activation | iPSC-induced neuron | Increased YAP nuclear localization; inhibited neuron induction | [111] |
| MSC derived from bovine bone marrow | Increased stress fiber; decreased contractility | [120] | ||
| Statin | Anti-atherosclerotic drug; YAP/TAZ inhibition | Human endothelial cells | Inhibited cell proliferation and anti-inflammatory effect | [86] |
| Sphingosine-1-phosphate (S1P) | YAP activation | HEK293A | Induced YAP nuclear localization | [121] |
| Verteporfin (VP) | YAP inhibition; blocked YAP-TEAD interaction | Human HSC-derived erythroid cells | Inhibited cell growth and differentiation | [122] |
| Agents | Effectiveness | Cell Types | Treatment Results | References |
|---|---|---|---|---|
| Blebbistatin | Myosin inhibition | Mouse embryonic fibroblast NIH3T3 | Reduced stress fibers F-actin and nuclear YAP | [116] |
| Endothelial cells | Block shear stress-induced YAP translocation | [87] | ||
| Cytochalasin D | Actin inhibition; blocked actin polymerization, resulting in reduction of YAP and TEAD nuclear activity | Mouse embryonic fibroblast NIH3T3 | Reduced stress fiber and nuclear YAP | [116] |
| HEK293A | Blocked YAP dephosphorylation | [115] | ||
| MSC derived from bovine bone marrow | Loss of stress fiber; decreased contractility | [120] | ||
| C3 exoenzyme | Rho-specific inhibition | HEK293A | Suppressed YAP/TAZ dephosphorylation | [115] |
| Human ESCs | Massive cell death | [98] | ||
| Human endothelial cells | Strongly regulated YAP/TAZ activity | [86] | ||
| CT04 | Rho-GTPase inhibition | Human iPSC-derived hemogenic endothelial cells | Decreased Runx1/Cmyb expression | [88] |
| CN02, CN03 and CN04 | Rho-GTPase activation | Human iPSC-derived hemogenic endothelial cells | Induced YAP signaling and promoted HSC formation | [88] |
| Latrunculin A | Actin inhibition; prevented the conversion of globular G-actin into filamentous F-actin and disrupted actin polymerization | MSCs | YAP/TAZ nuclear exclusion | [13] |
| Mouse embryonic fibroblast NIH3T3 | Reduced stress fiber and nuclear YAP | [116] | ||
| Human PSCs | Decreased YAP/TEAD reporter activity | [70] | ||
| Latrunculin B | F-actin inhibition | HEK293A | Blocked YAP dephosphorylation and prevented YAP nuclear localization | [115] |
| Nucleus pulposus cells of intervertebral disc | Reduced cell proliferation | [117] | ||
| Y-27632 | ROCK inhibition | iPSC-induced neurons | Promoted YAP/TAZ cytoplasmic retention; increased the percentage of neuroepithelial progenitors | [111] |
| Human periodontal ligament stem cells | Promoted cell proliferation, chemotaxis, and wound healing | [123] | ||
| MSC derived from bovine bone marrow | Loss of stress fiber; decreased contractility | [120] | ||
| V14-Rho | RhoA activation | Human endothelial cells | Strongly regulated YAP/TAZ activity | [86] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Damkham, N.; Issaragrisil, S.; Lorthongpanich, C. Role of YAP as a Mechanosensing Molecule in Stem Cells and Stem Cell-Derived Hematopoietic Cells. Int. J. Mol. Sci. 2022, 23, 14634. https://doi.org/10.3390/ijms232314634
Damkham N, Issaragrisil S, Lorthongpanich C. Role of YAP as a Mechanosensing Molecule in Stem Cells and Stem Cell-Derived Hematopoietic Cells. International Journal of Molecular Sciences. 2022; 23(23):14634. https://doi.org/10.3390/ijms232314634
Chicago/Turabian StyleDamkham, Nattaya, Surapol Issaragrisil, and Chanchao Lorthongpanich. 2022. "Role of YAP as a Mechanosensing Molecule in Stem Cells and Stem Cell-Derived Hematopoietic Cells" International Journal of Molecular Sciences 23, no. 23: 14634. https://doi.org/10.3390/ijms232314634
APA StyleDamkham, N., Issaragrisil, S., & Lorthongpanich, C. (2022). Role of YAP as a Mechanosensing Molecule in Stem Cells and Stem Cell-Derived Hematopoietic Cells. International Journal of Molecular Sciences, 23(23), 14634. https://doi.org/10.3390/ijms232314634







