A, B, C’s of Trk Receptors and Their Ligands in Ocular Repair
Abstract
1. Introduction
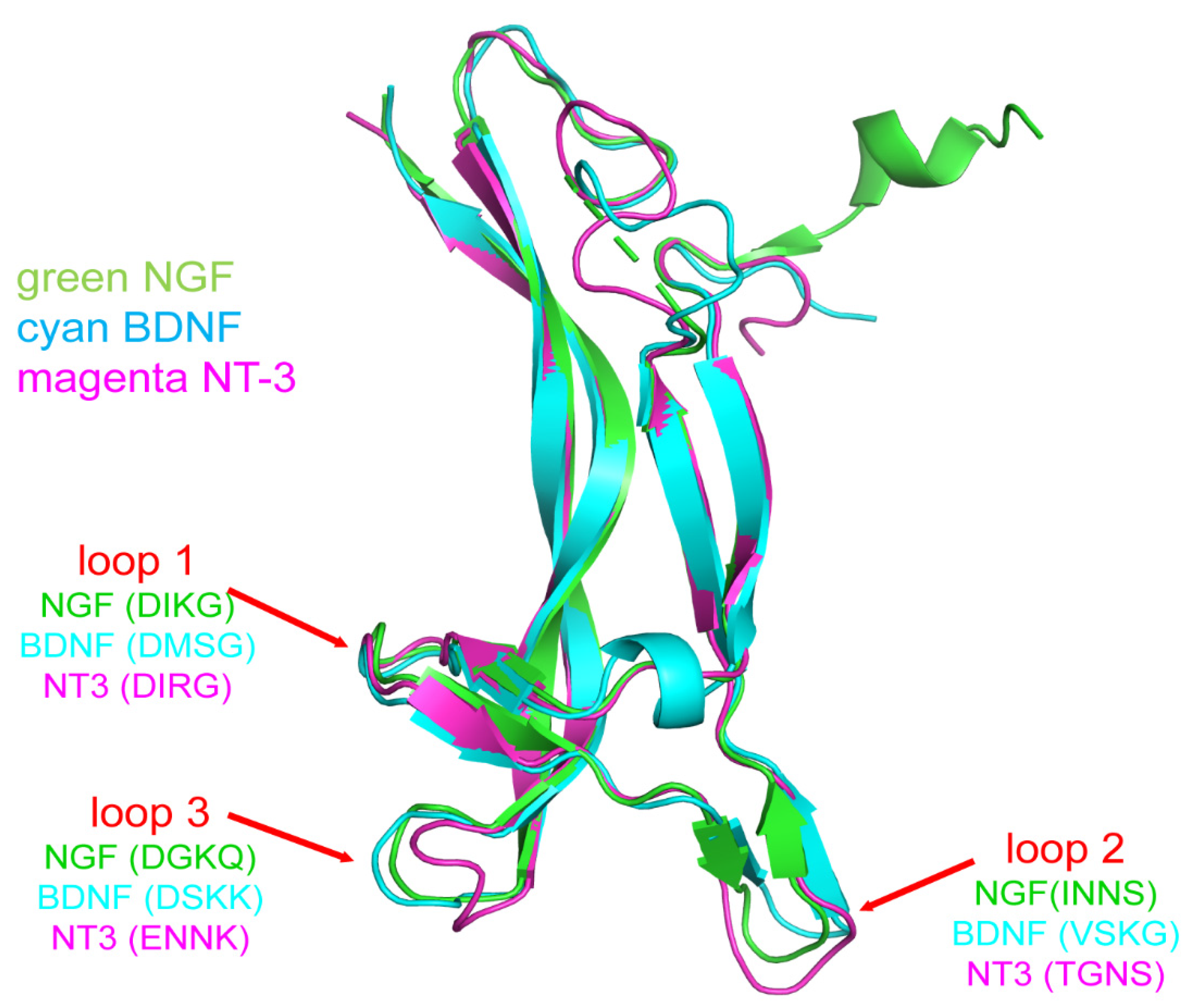
2. Molecular Structure
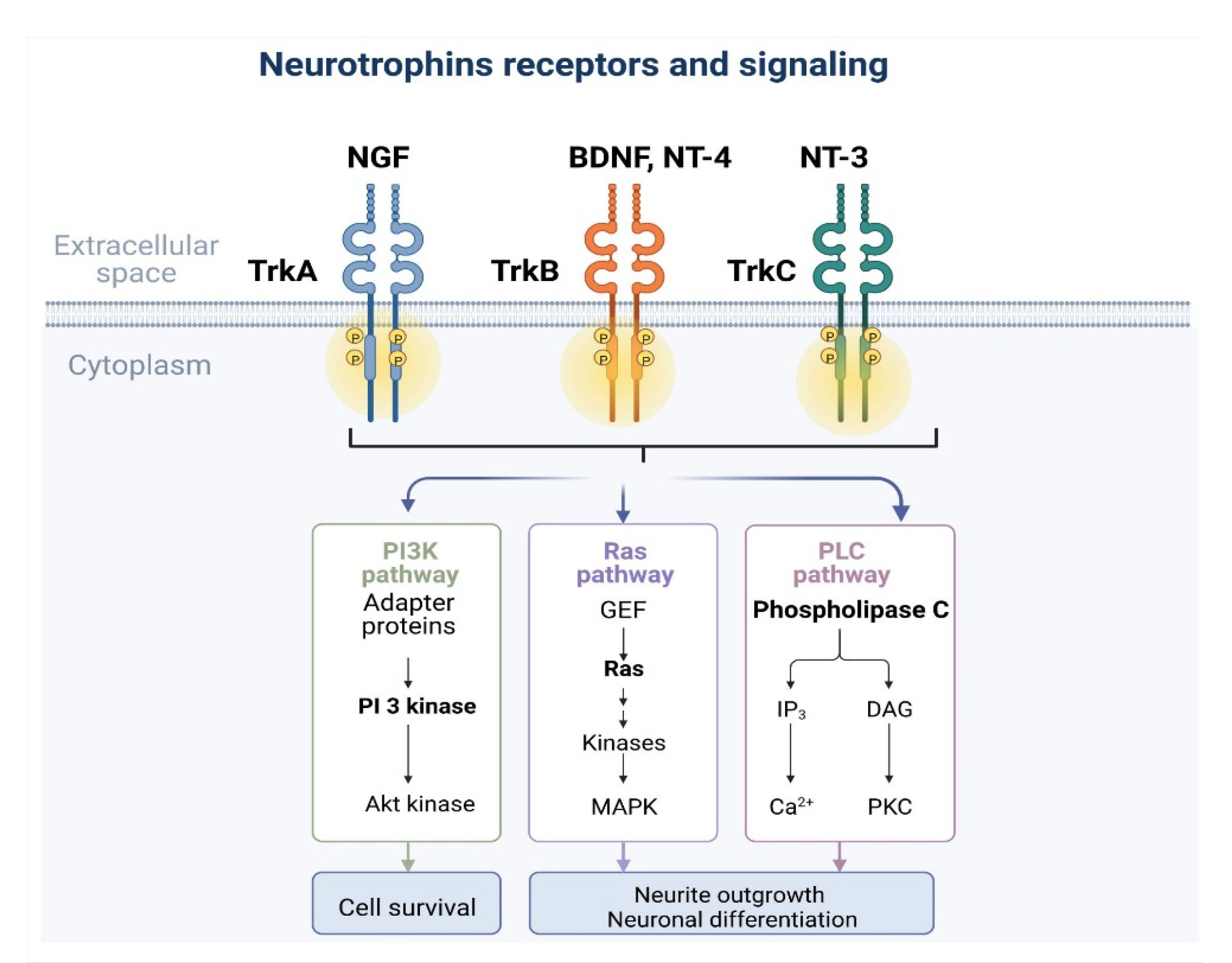
3. TrK and Signaling Pathways (TrkA–C)
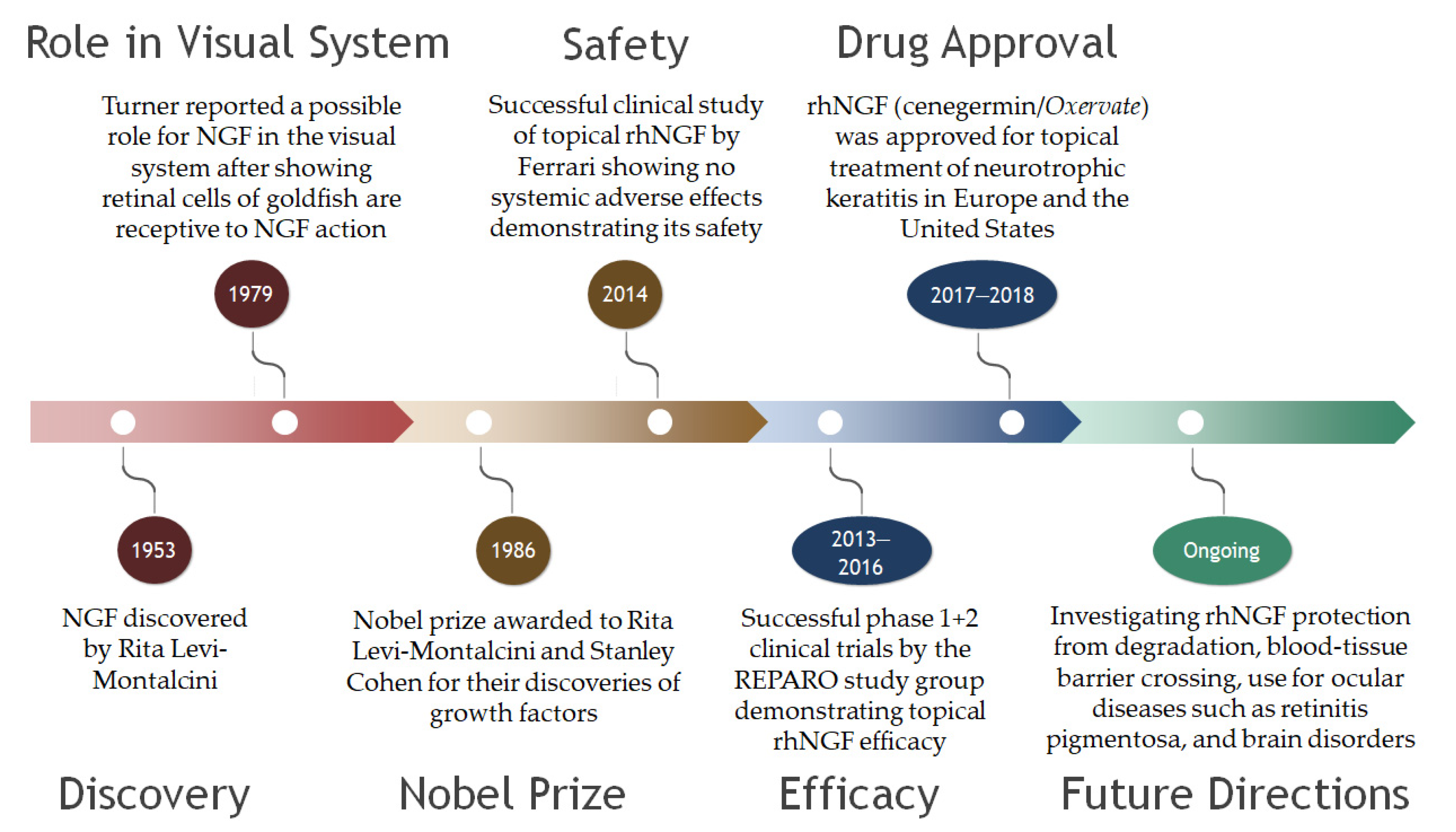
4. NGF
4.1. Wound Healing
4.2. Corneal Reinnervation
4.3. Dry Eye
5. Clinical Trials Featuring NGF
6. Brain-Derived Neurotrophic Factor, Neurotrophin-4, and Neurotrophin-3
7. Neurotrophin Analogs
8. Conclusions
9. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Benito-Gutiérrez, È.; Garcia-Fernàndez, J.; Comella, J.X. Origin and evolution of the Trk family of neurotrophic receptors. Mol. Cell. Neurosci. 2006, 31, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Huang, E.J.; Reichardt, L.F. Neurotrophins: Roles in neuronal development and function. Annu. Rev. Neurosci. 2001, 24, 677–736. [Google Scholar] [CrossRef] [PubMed]
- Ilag, L.L.; Lönnerberg, P.; Persson, H.; Ibáñez, C.F. Role of variable beta-hairpin loop in determining biological specificities in neurotrophin family. J. Biol. Chem. 1994, 269, 19941–19946. [Google Scholar] [CrossRef]
- van Kesteren, R.E.; Fainzilber, M.; Hauser, G.; van Minnen, J.; Vreugdenhil, E.; Smit, A.B.; Ibáñez, C.F.; Geraerts, W.P.; Bulloch, A.G. Early evolutionary origin of the neurotrophin receptor family. EMBO J. 1998, 17, 2534–2542. [Google Scholar] [CrossRef] [PubMed]
- Maliartchouk, S.; Saragovi, H.U. Optimal nerve growth factor trophic signals mediated by synergy of TrkA and p75 receptor-specific ligands. J. Neurosci. Off. J. Soc. Neurosci. 1997, 17, 6031–6037. [Google Scholar] [CrossRef]
- Carter, B.D.; Kaltschmidt, C.; Kaltschmidt, B.; Offenhäuser, N.; Böhm-Matthaei, R.; Baeuerle, P.A.; Barde, Y.A. Selective activation of NF-kappa B by nerve growth factor through the neurotrophin receptor p75. Science 1996, 272, 542–545. [Google Scholar] [CrossRef]
- Casaccia-Bonnefil, P.; Carter, B.D.; Dobrowsky, R.T.; Chao, M.V. Death of oligodendrocytes mediated by the interaction of nerve growth factor with its receptor p75. Nature 1996, 383, 716–719. [Google Scholar] [CrossRef]
- Frade, J.M.; Barde, Y.A. Nerve growth factor: Two receptors, multiple functions. Bioessays 1998, 20, 137–145. [Google Scholar] [CrossRef]
- Brennan, C.; Rivas-Plata, K.; Landis, S.C. The p75 neurotrophin receptor influences NT-3 responsiveness of sympathetic neurons in vivo. Nat. Neurosci. 1999, 2, 699–705. [Google Scholar] [CrossRef]
- Mischel, P.S.; Smith, S.G.; Vining, E.R.; Valletta, J.S.; Mobley, W.C.; Reichardt, L.F. The extracellular domain of p75NTR is necessary to inhibit neurotrophin-3 signaling through TrkA. J. Biol. Chem. 2001, 276, 11294–11301. [Google Scholar] [CrossRef]
- Hallböök, F. Evolution of the vertebrate neurotrophin and Trk receptor gene families. Curr. Opin. Neurobiol. 1999, 9, 616–621. [Google Scholar] [CrossRef]
- Korsching, S. The neurotrophic factor concept: A reexamination. J. Neurosci. Off. J. Soc. Neurosci. 1993, 13, 2739–2748. [Google Scholar] [CrossRef]
- Fariñas, I.; Wilkinson, G.A.; Backus, C.; Reichardt, L.F.; Patapoutian, A. Characterization of neurotrophin and Trk receptor functions in developing sensory ganglia: Direct NT-3 activation of TrkB neurons in vivo. Neuron 1998, 21, 325–334. [Google Scholar] [CrossRef]
- Brady, R.; Zaidi, S.I.A.; Mayer, C.; Katz, D.M. BDNF is a target-derived survival factor for arterial baroreceptor and chemoafferent primary sensory neurons. J. Neurosci. Off. J. Soc. Neurosci. 1999, 19, 2131–2142. [Google Scholar] [CrossRef]
- Wiesmann, C.; De Vos, A.M. Nerve growth factor: Structure and function. Cell. Mol. Life Sci. CMLS 2001, 58, 748–759. [Google Scholar] [CrossRef]
- Holden, P.H.; Asopa, V.; Robertson, A.G.S.; Clarke, A.R.; Tyler, S.; Bennett, G.S.; Brain, S.D.; Wilcock, G.K.; Allen, S.J.; Smith, S.K.F.; et al. Immunoglobulin-like domains define the nerve growth factor binding site of the TrkA receptor. Nat. Biotechnol. 1997, 15, 668–672. [Google Scholar] [CrossRef]
- Lewin, G.R.; Barde, Y.A. Physiology of the neurotrophins. Annu. Rev. Neurosci. 1996, 19, 289–317. [Google Scholar] [CrossRef]
- Huang, E.J.; Reichardt, L.F. Trk receptors: Roles in neuronal signal transduction. Annu. Rev. Biochem. 2003, 72, 609–642. [Google Scholar] [CrossRef]
- Huang, H.; Tang, X. Phenotypic determination and characterization of nestin-positive precursors derived from human fetal pancreas. Lab. Investig. 2003, 83, 539–547. [Google Scholar] [CrossRef][Green Version]
- Grewal, S.S.; York, R.D.; Stork, P.J.S. Extracellular-signal-regulated kinase signalling in neurons. Curr. Opin. Neurobiol. 1999, 9, 544–553. [Google Scholar] [CrossRef]
- Corbit, K.C.; Foster, D.A.; Rosner, M.R. Protein kinase Cdelta mediates neurogenic but not mitogenic activation of mitogen-activated protein kinase in neuronal cells. Mol. Cell. Biol. 1999, 19, 4209–4218. [Google Scholar] [CrossRef] [PubMed]
- Qi, H.; Chuang, E.Y.; Yoon, K.C.; de Paiva, C.S.; Shine, H.D.; Jones, D.B.; Pflugfelder, S.C.; Li, D.Q. Patterned expression of neurotrophic factors and receptors in human limbal and corneal regions. Mol. Vis. 2007, 13, 1934–1941. [Google Scholar] [PubMed]
- You, L.; Kruse, F.E.; Völcker, H.E. Neurotrophic Factors in the Human Cornea. Investig. Ophthalmol. Vis. Sci. 2000, 41, 692–702. [Google Scholar]
- Rios, J.D.; Ghinelli, E.; Gu, J.; Hodges, R.R.; Dartt, D.A. Role of neurotrophins and neurotrophin receptors in rat conjunctival goblet cell secretion and proliferation. Investig. Ophthalmol. Vis. Sci. 2007, 48, 1543–1551. [Google Scholar] [CrossRef]
- Ghinelli, E.; Johansson, J.; Ríos, J.D.; Chen, L.L.; Zoukhri, D.; Hodges, R.R.; Dartt, D.A. Presence and localization of neurotrophins and neurotrophin receptors in rat lacrimal gland. Investig. Ophthalmol. Vis. Sci. 2003, 44, 3352–3357. [Google Scholar] [CrossRef]
- Puri, S.; Kenyon, B.M.; Hamrah, P. Immunomodulatory Role of Neuropeptides in the Cornea. Biomedicines 2022, 10, 1985. [Google Scholar] [CrossRef]
- You, L.; Ebner, S.; Kruse, F.E. Glial cell-derived neurotrophic factor (GDNF)-induced migration and signal transduction in corneal epithelial cells. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2496–2504. [Google Scholar]
- Padern, G.; Duflos, C.; Ferreira, R.; Assou, S.; Guilpain, P.; Maria, A.T.J.; Goulabchand, R.; Galea, P.; Jurtela, M.; Jorgensen, C.; et al. Identification of a Novel Serum Proteomic Signature for Primary Sjögren’s Syndrome. Front. Immunol. 2021, 12, 631539. [Google Scholar] [CrossRef]
- Li, Y.J.; Yang, C.S.; Lei, L.; Wu, K.F.; Yang, P.T.; Xiao, W.G. Serum nerve grow factor and brain-derived neurotrophic factor profiles in Sjögren’s syndrome concomitant with interstitial lung disease. Clin. Rheumatol. 2014, 33, 1161–1164. [Google Scholar] [CrossRef]
- Chaudhary, S.; Namavari, A.; Yco, L.; Chang, J.H.; Sonawane, S.; Khanolkar, V.; Sarkar, J.; Jain, S. Neurotrophins and nerve regeneration-associated genes are expressed in the cornea after lamellar flap surgery. Cornea 2012, 31, 1460–1467. [Google Scholar] [CrossRef]
- Afarid, M.; Torabi-Nami, M.; Nemati, A.; Khosravi, A.; Malekzadeh, M. Brain-derived neurotrophic factor in patients with advanced age-related macular degeneration. Int. J. Ophthalmol. 2015, 8, 991–995. [Google Scholar] [PubMed]
- Ghosh, T.; Maity, N.; Sur, V.P.; Konar, A.; Hazra, S. Mitigating fibrosis-An impediment to corneal re-innervation following lamellar flap surgery. Exp Eye Res. 2020, 194, 108009. [Google Scholar] [CrossRef] [PubMed]
- Lambiase, A.; Manni, L.; Bonini, S.; Rama, P.; Micera, A.; Aloe, L. Nerve growth factor promotes corneal healing: Structural, biochemical, and molecular analyses of rat and human corneas. Investig. Ophthalmol. Vis. Sci. 2000, 41, 1063–1069. [Google Scholar]
- Nguyen, D.H.; Beuerman, R.W.; Thompson, H.W.; DiLoreto, D.A. Growth factor and neurotrophic factor mRNA in human lacrimal gland. Cornea 1997, 16, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Vereertbrugghen, A.; Galletti, J.G. Corneal nerves and their role in dry eye pathophysiology. Exp Eye Res. 2022, 222, 109191. [Google Scholar] [CrossRef]
- Lambiase, A.; Micera, A.; Sacchetti, M.; Cortes, M.; Mantelli, F.; Bonini, S. Alterations of tear neuromediators in dry eye disease. Arch. Ophthalmol. 2011, 129, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Chang, E.J.; Young, S.I.; Kay, E.P.; Jong, Y.K.; Jong, E.L.; Hyung, K.L. The role of nerve growth factor in hyperosmolar stress induced apoptosis. J. Cell. Physiol. 2008, 216, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Kang, S.-S.; Kim, J.Y.; Tchah, H. Nerve Growth Factor Attenuates Apoptosis and Inflammation in the Diabetic Cornea. Investig. Ophthalmol. Vis. Sci. 2016, 57, 6767–6775. [Google Scholar] [CrossRef]
- Kang, S.S.; Ha, S.J.; Kim, E.S.; Shin, J.A.; Kim, J.Y.; Tchah, H. Effect of nerve growth factor on the in vitro induction of apoptosis of human conjunctival epithelial cells by hyperosmolar stress. Investig. Ophthalmol. Vis Sci 2014, 55, 535–541. [Google Scholar] [CrossRef][Green Version]
- Lambiase, A.; Bonini, S.; Manni, L.; Ghinelli, E.; Tirassa, P.; Rama, P.; Aloe, L. Intraocular production and release of nerve growth factor after iridectomy. Investig. Ophthalmol. Vis. Sci. 2002, 43, 2334–2340. [Google Scholar]
- Lambiase, A.; Mantelli, F.; Bonini, S. Nerve growth factor eye drops to treat glaucoma. Drug News Perspect. 2010, 23, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Micera, A.; Lambiase, A.; Aloe, L.; Bonini, S.; Levi-Schaffer, F.; Bonini, S. Nerve growth factor involvement in the visual system: Implications in allergic and neurodegenerative diseases. Cytokine Growth Factor Rev. 2004, 15, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Kruse, F.E.; Tseng, S.C. Growth factors modulate clonal growth and differentiation of cultured rabbit limbal and corneal epithelium. Investig. Ophthalmol.Vis.Sci. 1993, 34, 1963–1976. [Google Scholar]
- Lambiase, A.; Bonini, S.; Micera, A.; Rama, P.; Bonini, S.; Aloe, L. Expression of nerve growth factor receptors on the ocular surface in healthy subjects and during manifestation of inflammatory diseases. Investig. Ophthalmol. Vis. Sci. 1998, 39, 1272–1275. [Google Scholar]
- Lambiase, A.; Centofanti, M.; Micera, A.; Manni, G.L.; Mattei, E.; De Gregorio, A.; De Feo, G.; Bucci, M.G.; Aloe, L. Nerve growth factor (NGF) reduces and NGF antibody exacerbates retinal damage induced in rabbit by experimental ocular hypertension. Graefe’s Arch. Clin. Exp. Ophthalmol. = Albrecht Von Graefes Arch. Fur Klin. Und Exp. Ophthalmol. 1997, 235, 780–785. [Google Scholar] [CrossRef]
- Di, G.; Qi, X.; Zhao, X.; Zhang, S.; Danielson, P.; Zhou, Q. Corneal Epithelium-Derived Neurotrophic Factors Promote Nerve Regeneration. Investig. Ophthalmol. Vis. Sci. 2017, 58, 4695–4702. [Google Scholar] [CrossRef]
- Lambiase, A.; Sacchetti, M.; Bonini, S. Nerve growth factor therapy for corneal disease. Curr. Opin. Ophthalmol. 2012, 23, 296–302. [Google Scholar] [CrossRef]
- Bonini, S.; Lambiase, A.; Rama, P.; Caprioglio, G.; Aloe, L. Topical treatment with nerve growth factor for neurotrophic keratitis. Ophthalmology 2000, 107, 1347–1351; discussion 1351–13512. [Google Scholar] [CrossRef]
- Joo, M.J.; Yuhan, K.R.; Hyon, J.Y.; Lai, H.; Hose, S.; Sinha, D.; O’Brien, T.P. The effect of nerve growth factor on corneal sensitivity after laser in situ keratomileusis. Arch. Ophthalmol. 2004, 122, 1338–1341. [Google Scholar] [CrossRef][Green Version]
- Wu, Y.; Chu, R.; Zhou, X.; Dai, J.; Qu, X. Determination of the nerve growth factor level in the central cornea after LASIK and Epi-LASIK treatment in a rabbit model system. Cornea 2009, 28, 1144–1148. [Google Scholar] [CrossRef]
- Lambiase, A.; Micera, A.; Pellegrini, G.; Merlo, D.; Rama, P.; De Luca, M.; Bonini, S.; Bonini, S. In vitro evidence of nerve growth factor effects on human conjunctival epithelial cell differentiation and mucin gene expression. Investig. Ophthalmol. Vis. Sci. 2009, 50, 4622–4630. [Google Scholar] [CrossRef] [PubMed]
- Fauchais, A.L.; Boumediene, A.; Lalloue, F.; Gondran, G.; Loustaud-Ratti, V.; Vidal, E.; Jauberteau, M.O. Brain-derived neurotrophic factor and nerve growth factor correlate with T-cell activation in primary Sjogren’s syndrome. Scand. J. Rheumatol. 2009, 38, 50–57. [Google Scholar] [CrossRef]
- Ehrhard, P.B.; Erb, P.; Graumann, U.; Schmutz, B.; Otten, U. Expression of functional trk tyrosine kinase receptors after T cell activation. J. Immunol. 1994, 152, 2705–2709. [Google Scholar] [PubMed]
- Coassin, M.; Lambiase, A.; Costa, N.; De Gregorio, A.; Sgrulletta, R.; Sacchetti, M.; Aloe, L.; Bonini, S. Efficacy of topical nerve growth factor treatment in dogs affected by dry eye. Graefe’s Arch. Clin. Exp. Ophthalmol. = Albrecht Von Graefes Arch. Fur Klin. Und Exp. Ophthalmol. 2005, 243, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Leao, F.; Sendra, V.G.; Kenyon, B.; Harris, D.L.; Hamrah, P. Dry Eye Disease Results in an Imbalance of Neuropeptides and Neurotrophins in the Cornea and Trigeminal Ganglion. Investig. Ophthalmol. Vis. Sci. 2021, 62, 1319. [Google Scholar]
- Ferrari, M.P.; Mantelli, F.; Sacchetti, M.; Antonangeli, M.I.; Cattani, F.; D’Anniballe, G.; Sinigaglia, F.; Ruffini, P.A.; Lambiase, A. Safety and pharmacokinetics of escalating doses of human recombinant nerve growth factor eye drops in a double-masked, randomized clinical trial. BioDrugs 2014, 28, 275–283. [Google Scholar] [CrossRef]
- Bonini, S.; Lambiase, A.; Rama, P.; Filatori, I.; Allegretti, M.; Chao, W.; Mantelli, F. Phase I Trial of Recombinant Human Nerve Growth Factor for Neurotrophic Keratitis. Ophthalmology 2018, 125, 1468–1471. [Google Scholar] [CrossRef]
- Bonini, S.; Lambiase, A.; Rama, P.; Sinigaglia, F.; Allegretti, M.; Chao, W.; Mantelli, F. Phase II Randomized, Double-Masked, Vehicle-Controlled Trial of Recombinant Human Nerve Growth Factor for Neurotrophic Keratitis. Ophthalmology 2018, 125, 1332–1343. [Google Scholar] [CrossRef]
- Pflugfelder, S.C.; Massaro-Giordano, M.; Perez, V.L.; Hamrah, P.; Deng, S.X.; Espandar, L.; Foster, C.S.; Affeldt, J.; Seedor, J.A.; Afshari, N.A.; et al. Topical Recombinant Human Nerve Growth Factor (Cenegermin) for Neurotrophic Keratopathy: A Multicenter Randomized Vehicle-Controlled Pivotal Trial. Ophthalmology 2020, 127, 14–26. [Google Scholar] [CrossRef]
- Mastropasqua, L.; Lanzini, M.; Dua, H.S.; Uffizi, A.D.; Di Nicola, M.; Calienno, R.; Bondi, J.; Said, D.G.; Nubile, M. In Vivo Evaluation of Corneal Nerves and Epithelial Healing After Treatment with Recombinant Nerve Growth Factor for Neurotrophic Keratopathy. Am. J. Ophthalmol. 2020, 217, 278–286. [Google Scholar] [CrossRef]
- Pieragostino, D.; Lanzini, M.; Cicalini, I.; Cufaro, M.C.; Damiani, V.; Mastropasqua, L.; De Laurenzi, V.; Nubile, M.; Lanuti, P.; Bologna, G.; et al. Tear proteomics reveals the molecular basis of the efficacy of human recombinant nerve growth factor treatment for Neurotrophic Keratopathy. Sci. Rep. 2022, 12, 1229. [Google Scholar] [CrossRef] [PubMed]
- Saricay, L.Y.; Bayraktutar, B.N.; Lilley, J.; Mah, F.S.; Massaro-Giordano, M.; Hamrah, P. Efficacy of Recombinant Human Nerve Growth Factor in Stage 1 Neurotrophic Keratopathy. Ophthalmology, 2022; in press. [Google Scholar] [CrossRef] [PubMed]
- Zwingelberg, S.B.; Bachmann, B.O.; Cursiefen, C. Real Life Data on Efficacy and Safety of Topical NGF Eye Drops (Cenegermin). Klin. Mon. Augenheilkd. 2020, 237, 1455–1461. [Google Scholar] [CrossRef] [PubMed]
- Pedrotti, E.; Bonacci, E.; Chierego, C.; De Gregorio, A.; Cozzini, T.; Brighenti, T.; Caldarella, G.; Pastore, G.; Fasolo, A.; Marchini, G. Eight months follow-up of corneal nerves and sensitivity after treatment with cenegermin for neurotrophic keratopathy. Orphanet. J. Rare Dis. 2022, 17, 63. [Google Scholar] [CrossRef] [PubMed]
- Di Zazzo, A.; Varacalli, G.; Mori, T.; Coassin, M. Long-term restoration of corneal sensitivity in neurotrophic keratopathy after rhNGF treatment. Eur. J. Ophthalmol. 2022, 32, NP15–NP18. [Google Scholar] [CrossRef] [PubMed]
- Bruscolini, A.; Marenco, M.; Albanese, G.M.; Lambiase, A.; Sacchetti, M. Long-term clinical efficacy of topical treatment with recombinant human nerve growth factor in neurotrophic keratopathy: A novel cure for a rare degenerative corneal disease? Orphanet. J. Rare Dis. 2022, 17, 57. [Google Scholar] [CrossRef]
- Papadopoulos, K.; Besgen, V.; Sekundo, W. Successful Treatment of a Pediatric Neurotrophic Keratopathy With Cenegermin. Cornea 2021, 40, 516–518. [Google Scholar] [CrossRef] [PubMed]
- Leto, M.G.; Toro, M.E.; Indemini, P.E.; Fruttero, C.; Denina, M.; Dalmazzo, C.; Sannia, A.; Vaiano, A.S. Pediatric Use of Recombinant Human Nerve Growth Factor 20 mug/mL Eye Drops (Cenegermin) for Bilateral Neurotrophic Keratopathy in Congenital Corneal Anesthesia. Cornea 2021, 40, 228–231. [Google Scholar] [CrossRef]
- Fausto, R.; Ceccuzzi, R.; Micheletti, E.; Clerici, R.; Riva, I.; Katsanos, A.; Oddone, F.; Quaranta, L. A case report of pediatric neurotrophic keratopathy in pontine tegmental cap dysplasia treated with cenegermin eye drops. Medicine 2020, 99, e20816. [Google Scholar] [CrossRef] [PubMed]
- Hatcher, J.B.; Soifer, M.; Morales, N.G.; Farooq, A.V.; Perez, V.L.; Shieh, C. Aftermarket effects of cenegermin for neurotrophic keratopathy in pediatric patients. Ocul. Surf. 2021, 21, 52–57. [Google Scholar] [CrossRef]
- Elhusseiny, A.M.; Traish, A.S.; Saeed, H.N.; Mantagos, I.S. Topical cenegermin 0.002% for pediatric neurotrophic keratopathy. Eur. J. Ophthalmol. 2022, 32. [Google Scholar] [CrossRef] [PubMed]
- Sacchetti, M.; Lambiase, A.; Schmidl, D.; Schmetterer, L.; Ferrari, M.; Mantelli, F.; Allegretti, M.; Garhoefer, G. Effect of recombinant human nerve growth factor eye drops in patients with dry eye: A phase IIa, open label, multiple-dose study. Br. J. Ophthalmol. 2020, 104, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Beykin, G.; Stell, L.; Halim, M.S.; Nunez, M.; Popova, L.; Nguyen, B.T.; Groth, S.L.; Dennis, A.; Li, Z.; Atkins, M.; et al. Phase 1b Randomized Controlled Study of Short Course Topical Recombinant Human Nerve Growth Factor (rhNGF) for Neuroenhancement in Glaucoma: Safety, Tolerability, and Efficacy Measure Outcomes. Am. J. Ophthalmol. 2022, 234, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Falsini, B.; Iarossi, G.; Chiaretti, A.; Ruggiero, A.; Manni, L.; Galli-Resta, L.; Corbo, G.; Abed, E. NGF eye-drops topical administration in patients with retinitis pigmentosa, a pilot study. J. Transl. Med. 2016, 14, 8. [Google Scholar] [CrossRef] [PubMed]
- Eftimiadi, G.; Soligo, M.; Manni, L.; Di Giuda, D.; Calcagni, M.L.; Chiaretti, A. Topical delivery of nerve growth factor for treatment of ocular and brain disorders. Neural Regen. Res. 2021, 16, 1740–1750. [Google Scholar] [PubMed]
- Gao, X.; He, G.H.; Zhang, X.T.; Chen, S. Protective effect of human umbilical cord mesenchymal stem cell-derived exosomes on rat retinal neurons in hyperglycemia through the brain-derived neurotrophic factor/TrkB pathway. Int. J. Ophthalmol. 2021, 14, 1683–1689. [Google Scholar] [CrossRef]
- Cha, Y.W.; Kim, S.T. Serum and aqueous humor levels of brain-derived neurotrophic factor in patients with primary open-angle glaucoma and normal-tension glaucoma. Int. Ophthalmol. 2021, 41, 3869–3875. [Google Scholar] [CrossRef]
- Proenca, C.C.; Song, M.; Lee, F.S. Differential effects of BDNF and neurotrophin 4 (NT4) on endocytic sorting of TrkB receptors. J. Neurochem. 2016, 138, 397–406. [Google Scholar] [CrossRef]
- Jeanneteau, F.; Chao, M.V. Are BDNF and glucocorticoid activities calibrated? Neuroscience 2013, 239, 173–195. [Google Scholar] [CrossRef]
- Hallak, J.A.; Tibrewal, S.; Mohindra, N.; Gao, X.; Jain, S. Single Nucleotide Polymorphisms in the BDNF, VDR, and DNASE 1 Genes in Dry Eye Disease Patients: A Case-Control Study. Investig. Ophthalmol. Vis. Sci. 2015, 56, 5990–5996. [Google Scholar] [CrossRef]
- Stepp, M.A.; Pal-Ghosh, S.; Tadvalkar, G.; Williams, A.R.; Pflugfelder, S.C.; de Paiva, C.S. Reduced Corneal Innervation in the CD25 Null Model of Sjogren Syndrome. Int. J. Mol. Sci. 2018, 19, 3821. [Google Scholar] [CrossRef]
- Kempfle, J.S.; Duro, M.V.; Zhang, A.; Amador, C.D.; Kuang, R.; Lu, R.; Kashemirov, B.A.; Edge, A.S.; McKenna, C.E.; Jung, D.H. A Novel Small Molecule Neurotrophin-3 Analogue Promotes Inner Ear Neurite Outgrowth and Synaptogenesis In vitro. Front. Cell Neurosci. 2021, 15, 666706. [Google Scholar] [CrossRef] [PubMed]
- Brahimi, F.; Galan, A.; Siegel, S.; Szobota, S.; Sarunic, M.V.; Foster, A.C.; Saragovi, H.U. Therapeutic Neuroprotection by an Engineered Neurotrophin that Selectively Activates Tropomyosin Receptor Kinase (Trk) Family Neurotrophin Receptors but Not the p75 Neurotrophin Receptor. Mol. Pharmacol. 2021, 100, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Maliartchouk, S.; Feng, Y.; Ivanisevic, L.; Debeir, T.; Cuello, A.C.; Burgess, K.; Saragovi, H.U. A designed peptidomimetic agonistic ligand of TrkA nerve growth factor receptors. Mol. Pharmacol. 2000, 57, 385–391. [Google Scholar] [PubMed]
- Yu, Z.; Joy, S.; Mi, T.; Yazdanpanah, G.; Burgess, K.; de Paiva, C.S. New, potent, small molecule agonists of tyrosine kinase receptors attenuate dry eye disease. Front. Med. 2022, 9, 937142. [Google Scholar] [CrossRef] [PubMed]
- Jain, P.; Li, R.; Lama, T.; Saragovi, H.U.; Cumberlidge, G.; Meerovitch, K. An NGF mimetic, MIM-D3, stimulates conjunctival cell glycoconjugate secretion and demonstrates therapeutic efficacy in a rat model of dry eye. Exp. Eye Res. 2011, 93, 503–512. [Google Scholar] [CrossRef]
- Meerovitch, K.; Torkildsen, G.; Lonsdale, J.; Goldfarb, H.; Lama, T.; Cumberlidge, G.; Ousler, G.W., 3rd. Safety and efficacy of MIM-D3 ophthalmic solutions in a randomized, placebo-controlled Phase 2 clinical trial in patients with dry eye. Clin. Ophthalmol. 2013, 7, 1275–1285. [Google Scholar] [CrossRef]
| Akt | Protein kinase B |
| Erk | Extracellular signal regulated kinase |
| MAPK | Mitogen-activated protein kinase |
| NGF | Nerve growth factor |
| Raf | Rapidly accelerated fibrosarcoma |
| Ras | Rat sarcoma virus |
| rhNGF | Recombinant human nerve growth factor |
| Trk | Tropomyosin receptor kinase |
| NCT#/Reference | Phase | Sample (N, Sex, Age | Design | Results | Adverse Events (AE) |
|---|---|---|---|---|---|
| NCT01744704 [56] | I | 74 healthy volunteers (24 F and 50 M, 40 ± 11 years) | Single, single ascending, or multiple ascending doses of rhNGF | No increase in serum NGF and no antidrug antibodies | Only mild, transient ocular AE (warm/pressure feeling, blurry vision, mild pain) |
| NCT01756456 [57] | I | 18 Stage 2–3 NK patients (9 F and 9 M, range 24–86 years) | 10 or 20 μg/mL rhNGF or vehicle, 6 drops/day for 8 weeks, then 48-week follow-up | Detectable serum NGF in 2 patients, no antidrug antibodies | Only mild ocular AE in 4 patients (29%) |
| NCT01756456 [58] | II | 156 Stage 2–3 NK patients (95 F and 61 M, ~60 ± 14 years) | 10 or 20 μg/mL rhNGF or vehicle, 6 drops/day for 8 weeks, then 48-week follow-up | 55%, 58%, and 20% followed by 75%, 74%, and 43% corneal healing rates at 4 and 8 weeks, respectively | Only mild treatment-related AE in 25 patients (16%) |
| NCT02227147 [59] | II | 48 Stage 2–3 NK patients (29 F and 19 M, 65 ± 14 years) | 20 μg/mL rhNGF or vehicle, 6 drops daily for 8 weeks, then 24-week follow-up | 70% vs. 29% corneal healing rate at 8 weeks, respectively | Mostly mild treatment-related AE in 18 patients (38%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gupta, A.; Galletti, J.G.; Yu, Z.; Burgess, K.; de Paiva, C.S. A, B, C’s of Trk Receptors and Their Ligands in Ocular Repair. Int. J. Mol. Sci. 2022, 23, 14069. https://doi.org/10.3390/ijms232214069
Gupta A, Galletti JG, Yu Z, Burgess K, de Paiva CS. A, B, C’s of Trk Receptors and Their Ligands in Ocular Repair. International Journal of Molecular Sciences. 2022; 23(22):14069. https://doi.org/10.3390/ijms232214069
Chicago/Turabian StyleGupta, Akash, Jeremias G. Galletti, Zhiyuan Yu, Kevin Burgess, and Cintia S. de Paiva. 2022. "A, B, C’s of Trk Receptors and Their Ligands in Ocular Repair" International Journal of Molecular Sciences 23, no. 22: 14069. https://doi.org/10.3390/ijms232214069
APA StyleGupta, A., Galletti, J. G., Yu, Z., Burgess, K., & de Paiva, C. S. (2022). A, B, C’s of Trk Receptors and Their Ligands in Ocular Repair. International Journal of Molecular Sciences, 23(22), 14069. https://doi.org/10.3390/ijms232214069







