Topical Omega-3 Fatty Acids Eyedrops in the Treatment of Dry Eye and Ocular Surface Disease: A Systematic Review
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Selection of Studies and Data Extraction
2.4. Outcome Measures
2.5. Data Analysis and Quality Assessment (Risk of Bias)
3. Results and Discussion
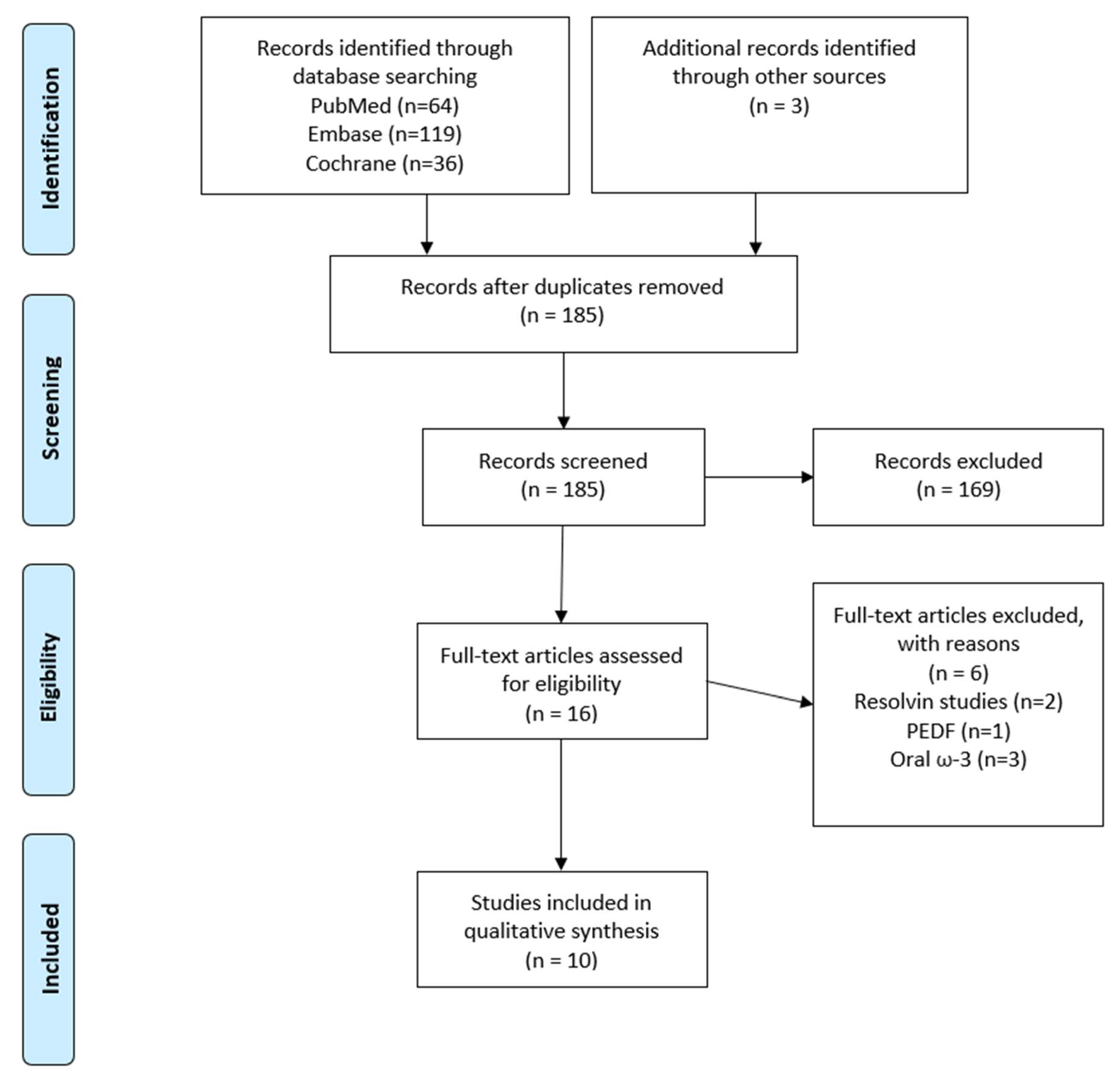
3.1. Systematic Search Strategy
3.2. Human Studies
3.2.1. Study Design
3.2.2. Improvements in Ocular Symptoms
3.2.3. Improvements in Tear Stability
3.2.4. Reduction of Meibomian Dysfunction (MGD)
3.2.5. Tear Production and Volume
3.2.6. Corneal Nerve Fiber Density
3.2.7. Biochemical Parameters
3.2.8. Adverse Events
3.2.9. Risk of Bias
3.3. Animal Studies
3.3.1. Overview of Animal Studies
| Author, Year | Condition | Fatty Acid; Regimen | Vehicle | Control | Other Tx | Outcomes Assessed and Their Results | Adverse Effects |
|---|---|---|---|---|---|---|---|
| Jacobi 2022 [40] | DED (old) | 0.2% DHA qid; 8 weeks | F6H8 (surfactant) | NA (single-arm) | Lid hygiene ^ | tCFS (NEI): CFB − 3.4 ± 2.1 (p < 0.0001) TBUT: CFB + 4.18 ± 2.77 (p < 0.0001) MGD score: CFB − 4.1 ± 3.3 (p < 0.0001) OSDI: clinically remarkable reduction by 17.5 ± 20 points (p < 0.0001) Slit-lamp: improved conjunctival injection (n = 14); improved lid redness (n = 2); improved PEE (n = 6); improved MGD (n = 2) NSD: Schirmer’s | None reported |
| Yilmaz 2021 [41] | CXL (young) | 1.2 mg EPA ester + 0.02 mg DHA ester qid; 4 weeks | VE, GLY, PAA, AAC, NaOH, Na3PO4 (Remogen Omega) | HA | Moxi-floxacin qid 1 week + FML qid 4 wks | tCFS (Oxford): omega-3: 0.68 ± 0.69; HA: 1.2 ± 0.72 p = 0.012 TBUT (postop): omega-3: 12.56 ± 2.8; hyaluronate: 9.72 ± 3.0 p < 0.001 Tear meniscus(postop): omega-3: 0.38 ± 0.08 hyaluronate: 0.34 ± 0.06 p = 0.047 NSD: Schirmer’s | None reported |
| Cagini 2020 [42] | CXL (young) | DHA-EPA 1 drop tid, 3 mo | VE, carbopol 980, GLY, Pemulen, NaOH, Na3PO4 (Resolvis Omega) | HA | Ofloxacin qid 1 week + Netidex qid 2 wks + HA qid 1 mo | OSDI: benefit over HA control NF density: NF density was 6 ± 0.82 in omega-3 group and 1 ± 0.51 in control group (sodium hyaluronate) p = 0.0001 at 3 month follow-up | None reported |
| Downie 2020 [43] | DED (old) | Flaxseed oil (ALA), bid, 3 mo | CMC 0.5%, GLY 1%, P80, ECO, osmoprotectants (levocarnitine, erythritol, trehalose) | ROA | NR ^ | tCFS: CFB − 1.5 ± 2.4 (p < 0.007 relative to control and p < 0.003 relative to baseline). Conj. staining: −0.85 ± 3.6 (p < 0.039 relative to control and p < 0.05 relative to baseline) NSD: TBUT, OSDI | Conjunctival hyperemia (n = 2); Eye irritation/itch (n = 3); Eyelid margin crusting (n = 1); Chalazion (n = 1); Foreign body sensation (n = 1) |
| Downie 2018 [44] | CLD-DED (young) | 0.025% EPA + 0.0025% DHA qid, 3 mo | VE, GLY, PAA, AAC, NaOH, Na3PO4 (Remogen Omega) | Olive oil | AT | IL-17A (tear): −76.2 ± 10.8% relative to baseline (p < 0.05) and placebo (p < 0.05) NSD: corneal staining, conj. staining, TBUT, Schirmer’s, MGD score, CLDEQ-8 score, slit-lamp examination |
| Author, Year | Q1 | Q2 | Q3 | Q4 | Q5 | Q6 | Q7 | Q8 | Q9 | Q10 | Q11 | Q12 | Q13 | Q14 |
Quality
Rating |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Jacobi 2022 * [40] | Y | Y | Y | Y | Y | Y | Y | N | Y | Y | N | NA | Good | ||
| Yilmaz 2021 [41] | Y | Y | NR | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Good |
| Cagini 2020 [42] | Y | Y | NR | NR | NR | Y | Y | Y | Y | Y | Y | Y | Y | Y | Fair |
| Downie 2020 [43] | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Good |
| Downie 2018 [44] | Y | Y | NR | N | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Good |
3.3.2. Effects on Tear Function Parameters
3.3.3. Effects on Cytokine Elaboration and Immune Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Appendix A. Criteria for Quality (Risk of Bias) Assessment among Included Studies
- Was the study described as randomized, a randomized trial, a randomized clinical trial, or an RCT?
- Was the method of randomization adequate (i.e., use of randomly generated assignment)?
- Was the treatment allocation concealed (so that assignments could not be predicted)?
- Were study participants and providers blinded to treatment group assignment?
- Were the people assessing the outcomes blinded to the participants’ group assignments?
- Were the groups similar at baseline on important characteristics that could affect outcomes (e.g., demographics, risk factors, co-morbid conditions)?
- Was the overall dropout rate from the study at endpoint 20% or lower of the number allocated to treatment?
- Was the differential dropout rate (between treatment groups) at endpoint 15 percentage points or lower?
- Was there high adherence to the intervention protocols for each treatment group?
- Were other interventions avoided or similar in the groups (e.g., similar background treatments)?
- Were outcomes assessed using valid and reliable measures, implemented consistently across all study participants?
- Did the authors report that the sample size was sufficiently large to be able to detect a difference in the main outcome between groups with at least 80% power?
- Were outcomes reported or subgroups analyzed prespecified (i.e., identified before analyses were conducted)?
- Were all randomized participants analyzed in the group to which they were originally assigned, i.e., did they use an intention-to-treat analysis?
- Was the study question or objective clearly stated?
- Were eligibility/selection criteria for the study population prespecified and clearly described?
- Were the participants in the study representative of those who would be eligible for the test/service/intervention in the general or clinical population of interest?
- Were all eligible participants that met the prespecified entry criteria enrolled?
- Was the sample size sufficiently large to provide confidence in the findings?
- Was the test/service/intervention clearly described and delivered consistently across the study population?
- Were the outcome measures prespecified, clearly defined, valid, reliable, and assessed consistently across all study participants?
- Were the people assessing the outcomes blinded to the participants’ exposures/interventions?
- Was the loss to follow-up after baseline 20% or less? Were those lost to follow-up accounted for in the analysis?
- Did the statistical methods examine changes in outcome measures from before to after the intervention? Were statistical tests done that provided p values for the pre-to-post changes?
- Were outcome measures of interest taken multiple times before the intervention and multiple times after the intervention (i.e., did they use an interrupted time-series design)?
- If the intervention was conducted at a group level (e.g., a whole hospital, a community, etc.) did the statistical analysis take into account the use of individual-level data to determine effects at the group level?
References
- Stapleton, F.; Alves, M.; Bunya, V.Y.; Jalbert, I.; Lekhanont, K.; Malet, F.; Na, K.-S.; Schaumberg, D.; Uchino, M.; Vehof, J.; et al. TFOS DEWS II Epidemiology Report. Ocul. Surf. 2017, 15, 334–365. [Google Scholar] [CrossRef] [PubMed]
- Asbell, P.A.; Maguire, M.G.; Peskin, E.; Bunya, V.Y.; Kuklinski, E.J. Dry Eye Assessment and Management (DREAM©) Study: Study design and baseline characteristics. Contemp. Clin. Trials 2018, 71, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Asche, C.V.; Fairchild, C.J. The economic burden of dry eye disease in the United States: A decision tree analysis. Cornea 2011, 30, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, W.; Chauhan, S.K.; Dana, R. Dry eye disease: An immune-mediated ocular surface disorder. Arch. Ophthalmol. 2012, 130, 90–100. [Google Scholar] [CrossRef]
- Lubis, R.R.; Gultom, M.T.H. The Correlation between Daily Lens Wear Duration and Dry Eye Syndrome. Open Access Maced. J. Med. Sci. 2018, 6, 829–834. [Google Scholar] [CrossRef]
- Uchino, M.; Schaumberg, D.A.; Dogru, M.; Uchino, Y.; Fukagawa, K.; Shimmura, S.; Satoh, T.; Takebayashi, T.; Tsubota, K. Prevalence of dry eye disease among Japanese visual display terminal users. Ophthalmology 2008, 115, 1982–1988. [Google Scholar] [CrossRef] [PubMed]
- Ishrat, S.; Nema, N.; Chandravanshi, S.C.L. Incidence and pattern of dry eye after cataract surgery. Saudi J. Ophthalmol. 2019, 33, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Pflugfelder, S.C.; Stern, M.E. Biological functions of tear film. Exp. Eye Res. 2020, 197, 108115. [Google Scholar] [CrossRef]
- Guo, O.L.; Akpek, E. The negative effects of dry eye disease on quality of life and visual function. Turk. J. Med. Sci. 2020, 50, 1611–1615. [Google Scholar] [CrossRef]
- Ambaw, Y.A.; Chao, C.; Ji, S.; Raida, M.; Torta, F.; Wenk, M.R.; Tong, L. Tear eicosanoids in healthy people and ocular surface disease. Sci. Rep. 2018, 8, 11296. [Google Scholar] [CrossRef]
- Johnson, M.E.; Murphy, P.J. Changes in the tear film and ocular surface from dry eye syndrome. Prog. Retin. Eye Res. 2004, 23, 449–474. [Google Scholar] [CrossRef] [PubMed]
- Dartt, D.A.; Willcox, M.D. Complexity of the tear film: Importance in homeostasis and dysfunction during disease. Exp. Eye Res. 2013, 117, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Baudouin, C.; Messmer, E.M.; Aragona, P.; Geerling, G.; Akova, Y.A.; Benítez-del-Castillo, J.; Boboridis, K.G.; Merayo-Lloves, J.; Rolando, M.; Labetoulle, M. Revisiting the vicious circle of dry eye disease: A focus on the pathophysiology of meibomian gland dysfunction. Br. J. Ophthalmol. 2016, 100, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Messmer, E.M. The pathophysiology, diagnosis, and treatment of dry eye disease. Dtsch. Arztebl. Int. 2015, 112, 71–81; quiz 82. [Google Scholar] [CrossRef] [PubMed]
- Bron, A.J.; de Paiva, C.S.; Chauhan, S.K.; Bonini, S.; Gabison, E.E.; Jain, S.; Knop, E.; Markoulli, M.; Ogawa, Y.; Perez, V.; et al. TFOS DEWS II pathophysiology report. Ocul. Surf. 2017, 15, 438–510. [Google Scholar] [CrossRef] [PubMed]
- Findlay, Q.; Reid, K. Dry eye disease: When to treat and when to refer. Aust. Prescr. 2018, 41, 160–163. [Google Scholar] [CrossRef] [PubMed]
- Aragona, P.; Giannaccare, G.; Mencucci, R.; Rubino, P.; Cantera, E.; Rolando, M. Modern approach to the treatment of dry eye, a complex multifactorial disease: A P.I.C.A.S.S.O. board review. Br. J. Ophthalmol. 2021, 105, 446. [Google Scholar] [CrossRef]
- Bibb, L.A.; Winter, R.P.; Leicht, S.S. Cyclosporine-induced Erythromelalgia. Cureus 2018, 10, e3506. [Google Scholar] [CrossRef]
- Nasri, H.; Baradaran, A.; Shirzad, H.; Rafieian-Kopaei, M. New concepts in nutraceuticals as alternative for pharmaceuticals. Int. J. Prev. Med. 2014, 5, 1487–1499. [Google Scholar]
- Grad, R. Cod and the Consumptive: A Brief History of Cod-Liver Oil in the Treatment of Pulmonary Tuberculosis. Pharm. Hist. 2004, 46, 106–120. [Google Scholar]
- Griffing, G.T. Mother was right about cod liver oil. Medscape J. Med. 2008, 10, 8. [Google Scholar] [PubMed]
- Sheppe Austin, E.F.; Edelmann Mariola, J. Roles of Eicosanoids in Regulating Inflammation and Neutrophil Migration as an Innate Host Response to Bacterial Infections. Infect. Immun. 2021, 89, e00095-21. [Google Scholar] [CrossRef]
- Alvarez, Y.; Valera, I.; Municio, C.; Hugo, E.; Padrón, F.; Blanco, L.; Rodríguez, M.; Fernández, N.; Crespo, M.S. Eicosanoids in the Innate Immune Response: TLR and Non-TLR Routes. Mediat. Inflamm. 2010, 2010, 201929. [Google Scholar] [CrossRef] [PubMed]
- Rees, D.; Miles, E.A.; Banerjee, T.; Wells, S.J.; Roynette, C.E.; Wahle, K.W.; Calder, P.C. Dose-related effects of eicosapentaenoic acid on innate immune function in healthy humans: A comparison of young and older men. Am. J. Clin. Nutr. 2006, 83, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Wall, R.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Fatty acids from fish: The anti-inflammatory potential of long-chain omega-3 fatty acids. Nutr. Rev. 2010, 68, 280–289. [Google Scholar] [CrossRef]
- Serhan, C.N.; Chiang, N.; Dalli, J. The resolution code of acute inflammation: Novel pro-resolving lipid mediators in resolution. Semin. Immunol. 2015, 27, 200–215. [Google Scholar] [CrossRef]
- Downie, L.E.; Ng, S.M.; Lindsley, K.B.; Akpek, E.K. Omega-3 and omega-6 polyunsaturated fatty acids for dry eye disease. Cochrane Database Syst. Rev. 2019, 12, Cd011016. [Google Scholar] [CrossRef]
- Barabino, S.; Horwath-Winter, J.; Messmer, E.M.; Rolando, M.; Aragona, P.; Kinoshita, S. The role of systemic and topical fatty acids for dry eye treatment. Prog. Retin. Eye Res. 2017, 61, 23–34. [Google Scholar] [CrossRef]
- Asbell, P.A.; Maguire, M.G.; Pistilli, M.; Ying, G.S.; Szczotka-Flynn, L.B.; Hardten, D.R.; Lin, M.C.; Shtein, R.M. n-3 Fatty Acid Supplementation for the Treatment of Dry Eye Disease. N. Engl. J. Med. 2018, 378, 1681–1690. [Google Scholar] [CrossRef]
- Hussain, M.; Shtein, R.M.; Pistilli, M.; Maguire, M.G.; Oydanich, M.; Asbell, P.A. The Dry Eye Assessment and Management (DREAM) extension study—A randomized clinical trial of withdrawal of supplementation with omega-3 fatty acid in patients with dry eye disease. Ocul. Surf. 2020, 18, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Cholkar, K.; Agrahari, V.; Mitra, A.K. Ocular drug delivery systems: An overview. World J. Pharmacol. 2013, 2, 47–64. [Google Scholar] [CrossRef] [PubMed]
- Cortina, M.S.; Bazan, H.E. Docosahexaenoic acid, protectins and dry eye. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Walter, S.D.; Gronert, K.; McClellan, A.L.; Levitt, R.C.; Sarantopoulos, K.D.; Galor, A. ω-3 Tear Film Lipids Correlate With Clinical Measures of Dry Eye. Investig. Ophthalmol. Vis. Sci. 2016, 57, 2472–2478. [Google Scholar] [CrossRef] [PubMed]
- Agrahari, V.; Mandal, A.; Agrahari, V.; Trinh, H.M.; Joseph, M.; Ray, A.; Hadji, H.; Mitra, R.; Pal, D.; Mitra, A.K. A comprehensive insight on ocular pharmacokinetics. Drug Deliv. Transl. Res. 2016, 6, 735–754. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef]
- Cortina, M.S.; He, J.; Li, N.; Bazan, N.G.; Bazan, H.E. Neuroprotectin D1 synthesis and corneal nerve regeneration after experimental surgery and treatment with PEDF plus DHA. Investig. Ophthalmol. Vis. Sci. 2010, 51, 804–810. [Google Scholar] [CrossRef]
- de Paiva, C.S.; Schwartz, C.E.; Gjörstrup, P.; Pflugfelder, S.C. Resolvin E1 (RX-10001) reduces corneal epithelial barrier disruption and protects against goblet cell loss in a murine model of dry eye. Cornea 2012, 31, 1299–1303. [Google Scholar] [CrossRef]
- Li, N.; He, J.; Schwartz, C.E.; Gjorstrup, P.; Bazan, H.E. Resolvin E1 improves tear production and decreases inflammation in a dry eye mouse model. J. Ocul. Pharmacol. Ther. 2010, 26, 431–439. [Google Scholar] [CrossRef]
- National Heart, Lung, and Blood Institute. Study Quality Assessment Tools. 2014. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 16 September 2022).
- Jacobi, C.; Angstmann-Mehr, S.; Lange, A.; Kaercher, T. A Water-Free Omega-3 Fatty Acid Eye Drop Formulation for the Treatment of Evaporative Dry Eye Disease: A Prospective, Multicenter Noninterventional Study. J. Ocul. Pharmacol. Ther. 2022, 38, 348–353. [Google Scholar] [CrossRef]
- Ondes Yilmaz, F.; Kepez Yildiz, B.; Tunc, U.; Kandemir Besek, N.; Yildirim, Y.; Demirok, A. Comparison of topical omega-3 fatty acids with topical sodium hyaluronate after corneal crosslinking: Short term results. Ocul. Immunol. Inflamm. 2021, 30, 959–965. [Google Scholar] [CrossRef]
- Cagini, C.; Messina, M.; Torroni, G.; Riccitelli, F.; Mariniello, M.; Dua, H.S. Efficacy of topical microemulsion of fatty acids of the ω-3 series on the sub-epithelial corneal nerves regeneration after epithelium-off corneal collagen cross-linking for keratoconus. Int. Ophthalmol. 2020, 40, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Downie, L.E.; Hom, M.M.; Berdy, G.J.; El-Harazi, S.; Verachtert, A.; Tan, J.; Liu, H.; Carlisle-Wilcox, C.; Simmons, P.; Vehige, J. An artificial tear containing flaxseed oil for treating dry eye disease: A randomized controlled trial. Ocul. Surf. 2020, 18, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Downie, L.E.; Gad, A.; Wong, C.Y.; Gray, J.H.V.; Zeng, W.; Jackson, D.C.; Vingrys, A.J. Modulating Contact Lens Discomfort With Anti-Inflammatory Approaches: A Randomized Controlled Trial. Investig. Ophthalmol. Vis. Sci. 2018, 59, 3755–3766. [Google Scholar] [CrossRef] [PubMed]
- Damani, M.R.; Iwabe, S.; Aguirre, G.D.; Carlisle, K.; Pistilli, M.; Bunya, V.Y. Safety and Efficacy of Topical 1% docosahexaenoic acid (DHA) and 1% alpha-linoleic acid (ALA) in a Canine Model. Investig. Ophthalmol. Vis. Sci. 2014, 55, 3688. [Google Scholar]
- Lidich, N.; Garti-Levy, S.; Aserin, A.; Garti, N. Potentiality of microemulsion systems in treatment of ophthalmic disorders: Keratoconus and dry eye syndrome—In vivo study. Colloids Surf. B Biointerfaces 2019, 173, 226–232. [Google Scholar] [CrossRef]
- Neves, M.L.; Yamasaki, L.; Sanches Ode, C.; do Amaral, M.S.; Stevanin, H.; Giuffrida, R.; Candido, E.R.; Góes, J.E.; Zulim, L.F.; Schweigert, A.; et al. Use of linseed oil to treat experimentally induced keratoconjunctivitis sicca in rabbits. J. Ophthalmic Inflamm. Infect. 2013, 3, 4. [Google Scholar] [CrossRef]
- Rashid, S.; Jin, Y.; Ecoiffier, T.; Barabino, S.; Schaumberg, D.A.; Dana, M.R. Topical omega-3 and omega-6 fatty acids for treatment of dry eye. Arch. Ophthalmol. 2008, 126, 219–225. [Google Scholar] [CrossRef]
- Li, Z.; Choi, J.H.; Oh, H.J.; Park, S.H.; Lee, J.B.; Yoon, K.C. Effects of eye drops containing a mixture of omega-3 essential fatty acids and hyaluronic acid on the ocular surface in desiccating stress-induced murine dry eye. Curr. Eye Res. 2014, 39, 871–878. [Google Scholar] [CrossRef]
- Parissi, M.; Randjelovic, S.; Poletti, E.; Guimarães, P.; Ruggeri, A.; Fragkiskou, S.; Wihlmark, T.B.; Utheim, T.P.; Lagali, N. Corneal Nerve Regeneration After Collagen Cross-Linking Treatment of Keratoconus: A 5-Year Longitudinal Study. JAMA Ophthalmol. 2016, 134, 70–78. [Google Scholar] [CrossRef]
- Wolffsohn, J.S.; Arita, R.; Chalmers, R.; Djalilian, A.; Dogru, M.; Dumbleton, K.; Gupta, P.K.; Karpecki, P.; Lazreg, S.; Pult, H.; et al. TFOS DEWS II Diagnostic Methodology report. Ocul. Surf. 2017, 15, 539–574. [Google Scholar] [CrossRef]
- Sun, M.; Moreno, I.Y.; Dang, M.; Coulson-Thomas, V.J. Meibomian Gland Dysfunction: What Have Animal Models Taught Us? Int. J. Mol. Sci. 2020, 21, 8822. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, M.; Sattar, M.; Saba, A.; Saba, A.; Sonum, S.; Batool, A. Evaluation of tear film instability pre and post corneal collagen cross-linking procedure in keratoconus patients. Adv. Ophthalmol. Vis. Syst. 2021, 11, 23–26. [Google Scholar] [CrossRef]
- Tomlinson, A.; Bron, A.J.; Korb, D.R.; Amano, S.; Paugh, J.R.; Pearce, E.I.; Yee, R.; Yokoi, N.; Arita, R.; Dogru, M. The international workshop on meibomian gland dysfunction: Report of the diagnosis subcommittee. Investig. Ophthalmol. Vis. Sci. 2011, 52, 2006–2049. [Google Scholar] [CrossRef]
- Shaheen, B.S.; Bakir, M.; Jain, S. Corneal nerves in health and disease. Surv. Ophthalmol. 2014, 59, 263–285. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Moreno, A.; Baudouin, C.; Melik Parsadaniantz, S.; Réaux-Le Goazigo, A. Morphological and Functional Changes of Corneal Nerves and Their Contribution to Peripheral and Central Sensory Abnormalities. Front. Cell. Neurosci. 2020, 14, 610342. [Google Scholar] [CrossRef]
- Jackson, D.C.; Zeng, W.; Wong, C.Y.; Mifsud, E.J.; Williamson, N.A.; Ang, C.S.; Vingrys, A.J.; Downie, L.E. Tear Interferon-Gamma as a Biomarker for Evaporative Dry Eye Disease. Investig. Ophthalmol. Vis. Sci. 2016, 57, 4824–4830. [Google Scholar] [CrossRef]
- Hampel, U.; Krüger, M.; Kunnen, C.; Garreis, F.; Willcox, M.; Paulsen, F. In vitro effects of docosahexaenoic and eicosapentaenoic acid on human meibomian gland epithelial cells. Exp. Eye Res. 2015, 140, 139–148. [Google Scholar] [CrossRef]
- Amerio, P.; Frezzolini, A.; Feliciani, C.; Verdolini, R.; Teofoli, P.; De Pità, O.; Puddu, P. Eotaxins and CCR3 receptor in inflammatory and allergic skin diseases: Therapeutical implications. Curr. Drug Targets Inflamm. Allergy 2003, 2, 81–94. [Google Scholar] [CrossRef]
- Crawford, A.; Angelosanto, J.M.; Nadwodny, K.L.; Blackburn, S.D.; Wherry, E.J. A role for the chemokine RANTES in regulating CD8 T cell responses during chronic viral infection. PLoS Pathog. 2011, 7, e1002098. [Google Scholar] [CrossRef]
- Guerrero-Moreno, A.; Liang, H.; Moreau, N.; Luzu, J.; Rabut, G.; Melik Parsadaniantz, S.; Labbé, A.; Baudouin, C.; Réaux-Le Goazigo, A. Corneal Nerve Abnormalities in Painful Dry Eye Disease Patients. Biomedicines 2021, 9, 1424. [Google Scholar] [CrossRef]
- Labetoulle, M.; Baudouin, C.; Calonge, M.; Merayo-Lloves, J.; Boboridis, K.G.; Akova, Y.A.; Aragona, P.; Geerling, G.; Messmer, E.M.; Benítez-del-Castillo, J. Role of corneal nerves in ocular surface homeostasis and disease. Acta Ophthalmol. 2019, 97, 137–145. [Google Scholar] [CrossRef]
- Bazan, N.G. Cellular and molecular events mediated by docosahexaenoic acid-derived neuroprotectin D1 signaling in photoreceptor cell survival and brain protection. Prostaglandins Leukot. Essent. Fat. Acids 2009, 81, 205–211. [Google Scholar] [CrossRef]
- Zhao, Y.; Calon, F.; Julien, C.; Winkler, J.W.; Petasis, N.A.; Lukiw, W.J.; Bazan, N.G. Docosahexaenoic Acid-Derived Neuroprotectin D1 Induces Neuronal Survival via Secretase- and PPARγ-Mediated Mechanisms in Alzheimer’s Disease Models. PLoS ONE 2011, 6, e15816. [Google Scholar] [CrossRef]
- Tsai, W.-C.; Kalyanaraman, C.; Yamaguchi, A.; Holinstat, M.; Jacobson, M.P.; Holman, T.R. In Vitro Biosynthetic Pathway Investigations of Neuroprotectin D1 (NPD1) and Protectin DX (PDX) by Human 12-Lipoxygenase, 15-Lipoxygenase-1, and 15-Lipoxygenase-2. Biochemistry 2021, 60, 1741–1754. [Google Scholar] [CrossRef]
- Li, N.; Kenchegowda, S.; Cortina, M.S.; Bazan, H.E.P. Pigment Epithelium Derived Factor (PEDF) Increases After Corneal Epithelial Injury and Stimulates the Expression of 15-Lox1. Investig. Ophthalmol. Vis. Sci. 2009, 50, 6304. [Google Scholar]
- Pham, T.L.; Bazan, H.E.P. Docosanoid signaling modulates corneal nerve regeneration: Effect on tear secretion, wound healing, and neuropathic pain. J. Lipid Res. 2021, 62, 100033. [Google Scholar] [CrossRef]
- Ogawa, M.; Ishihara, T.; Isobe, Y.; Kato, T.; Kuba, K.; Imai, Y.; Uchino, Y.; Tsubota, K.; Arita, M. Eosinophils promote corneal wound healing via the 12/15-lipoxygenase pathway. FASEB J. 2020, 34, 12492–12501. [Google Scholar] [CrossRef]
- Singh, N.K.; Rao, G.N. Emerging role of 12/15-Lipoxygenase (ALOX15) in human pathologies. Prog. Lipid Res. 2019, 73, 28–45. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, P.; Craig, J.P.; Rupenthal, I.D. Formulation Considerations for the Management of Dry Eye Disease. Pharmaceutics 2021, 13, 207. [Google Scholar] [CrossRef] [PubMed]
- Mofidfar, M.; Abdi, B.; Ahadian, S.; Mostafavi, E.; Desai, T.A.; Abbasi, F.; Sun, Y.; Manche, E.E.; Ta, C.N.; Flowers, C.W. Drug delivery to the anterior segment of the eye: A review of current and future treatment strategies. Int. J. Pharm. 2021, 607, 120924. [Google Scholar] [CrossRef] [PubMed]
- Messerli, F.H.; Bangalore, S.; Schmieder, R.E. Wilder’s principle: Pre-treatment value determines post-treatment response. Eur. Heart J. 2015, 36, 576–579. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Tourmouzis, K.; Perry, H.; Honkanen, R.A.; Rigas, B. Animal models of dry eye disease: Useful, varied and evolving (Review). Exp. Ther. Med. 2021, 22, 1394. [Google Scholar] [CrossRef] [PubMed]
- Tong, L.; Lim, L.; Tan, D.; Heng, W.J.; Lim, J.; Chan, C.; Arundhati, A.; Tan, A. Assessment and Management of Dry Eye Disease and Meibomian Gland Dysfunction: Providing a Singapore Framework. Asia-Pac. J. Ophthalmol. (Phila) 2021, 10, 530–541. [Google Scholar] [CrossRef] [PubMed]
| Author, Year | Sample Size | Study Type, Masking | Condition | Gender | Age Range | Significant Inclusion Criteria | Significant Exclusion Criteria |
|---|---|---|---|---|---|---|---|
| Jacobi 2022 [40] | T: 33 | PO, open-label | DED (old) | T: 69.7% | T: 54.8 ± 17.86 |
History of DED at least 6 mos, tCFS (NEI) < 11, MGD > 3, TBUT < 8, OSDI > 25, Schirmer’s I > 5 mm
Pts must have applied lid hygiene for 14 days prior to and continue during study |
|
| Yilmaz 2021 [41] |
T: 25
C: 25 | RCT, double-masked | CXL (young) | T: 24% C: 36% | T: 24.24 ± 2.46 C: 24.64 ± 2.22 |
|
|
| Cagini 2020 [42] |
T: 20
C: 20 | RCT, NR | CXL (young) | T + C: 45% | T + C: 28 (22–37) |
| NR |
| Downie 2020 [43] |
T: 120 C: 120 | RCT, double-masked | DED (old) | T: 70% C: 77.9% | T: 54.3 ± 17.3 C: 52.8 ± 16.7 | Aged > 18, with DED, OSDI > 18 and <65 at baseline |
|
| Downie 2018 [44] |
T1: 14
T2: 14 T3: 14 C: 14 # | RCT, single-masked | CLD (young) | T1: 86% T2: 59% T3: 83% C: 75% | T1: 25.9 ± 1.9 T2: 29.4 ± 1.3 T3: 23.3 ± 0.6 C: 24.6 ± 1.3 |
|
|
|
Author,
Year | Animal, Model | Description of Study |
|---|---|---|
|
Damani
2014 [45] |
Dogs,
healthy | This study investigates the outcomes of a topical w-3 formulation of DHA and ALA applied to the eye of healthy dogs, with their fellow eye used as control. Slit-lamp examination, ocular surface fluorescein staining, and tear cytokine levels (both protein and mRNA) were measured from Schirmer’s test strips. |
|
Lidich
2018 [46] |
Rabbit,
induced DED | This study describes the preparation of various microemulsions containing riboflavin 5-phosphate (RFP) and various other surfactants, to be mixed with triglyceride DHA (TG-DHA), to be used in ex vivo and in vivo experiments on a rabbit model of induced DED. Ex vivo experimentation sought to determine the effect of RFP on biomechanical strength of the cornea. In vivo experimentation aimed to determine the effect of TG-DHA on tear breakup time and Schirmer’s test. |
|
Neves
2013 [47] |
Rabbit,
induced DED | This study investigates efficacy of linseed oil given orally, topical, and oral–topical combined, on ocular surface staining with fluorescein and rose bengal, and on Schirmer’s test. Histopathologic analysis was also performed to evaluate conjunctival goblet cell density. |
|
Rashid
2008 [48] |
Mouse,
induced DED | This study evaluates effectiveness of various topical w-3 formulations of ALA and LA. Corneal fluorescein staining was used to assess the integrity of the ocular surface pre- and post-treatment. Immunohistochemistry with microscopy was used to evaluate CD11b+ cells in various regions of the cornea. Corneal and conjunctival tissues were separately homogenized and the mRNA levels of various inflammatory cytokines were quantified with RT-PCR and qPCR. |
| Li 2014 [49] |
Mouse,
induced DED | This study evaluates the efficacy of various mixtures of w-3 with hyaluronic acid applied topically. Corneal fluorescein staining was used to assess the integrity of the ocular surface pre- and post-treatment. Conjunctival tissues were homogenized and immunobead assays were used to quantify concentrations of inflammatory cytokines IL-1b, IL-17, and IP-10, as well as lipid peroxidation markers hexanoyl-lys and 4-hydroxynonenal. |
|
Author,
Year | Animal, Model | Method of DE Induction | Fatty Acid and Regimen | Control | Vehicle | Outcomes Assessed and Their Results |
|---|---|---|---|---|---|---|
|
Damani
2014 [45] |
Dogs,
healthy | N/A | 1% DHA + 1% EPA tid, 1 month | Vehicle | None specified | NSD: Schirmer’s, cytokines (IFN-y, TNFa, IL-1a, IL-1b, IL-2, IL-6, IL-8, and IL-10 (analysis from tears and conjunctival biopsy) |
|
Lidich
2018 [46] |
Rabbit,
induced DED | BZA | 1.2, 0.85, 0.45 wt% DHA @ ^ | NS | T80, Cremophor EL | TBUT: at 14 days: ω-3 > 10 s; control = 5–7 s (no statistical analysis done) Schirmer’s: at 14 days: ω-3 = 28 mm; control = NA (no statistical analysis) |
|
Neves
2013 [47] |
Rabbit,
induced DED | Lacrimal gland Removal + topical atropine | linseed oil (57% ω-3, 16% ω-6) bid @ | Placebo | NR | NSD: conjunctival goblet cell density |
|
Rashid
2008 [48] |
Mouse,
induced DED | s/c scopolamine + wind + low humidity | 0.2% ALA, 0.1% ALA + 0.1% LA, 0.2% LA 1 x/day @ | Vehicle | T80, Glucam e-20, VE, packing solution | Staining: decreased (compared to control) at days 5, 10 Cells: CD11b+ cells in the center of cornea significantly decreased (p = 0.03) in ALA-only group compared to vehicle control Cytokines: ALA-only group showed decreased corneal and conjunctival expression of IL-1A and TNF-a at day 10, compared to vehicle control |
| Li 2014 [49] |
Mouse,
induced DED | s/c scopolamine + wind + low humidity | ω-3 qid @ | HA | NR | Staining: 0.2% ω-3 + HA = 4.35 ± 1.40; significantly higher compared to 0.2% ω-3 and 0.02% ω-3 + HA. Cytokines: 0.2% ω-3 + HA: Conjunctival IL-1b, IL-17 significantly lower compared to HA only, 0.02% ω-3 only, 0.2% ω-3 only, and 0.02% ω-3 + HA. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paik, B.; Tong, L. Topical Omega-3 Fatty Acids Eyedrops in the Treatment of Dry Eye and Ocular Surface Disease: A Systematic Review. Int. J. Mol. Sci. 2022, 23, 13156. https://doi.org/10.3390/ijms232113156
Paik B, Tong L. Topical Omega-3 Fatty Acids Eyedrops in the Treatment of Dry Eye and Ocular Surface Disease: A Systematic Review. International Journal of Molecular Sciences. 2022; 23(21):13156. https://doi.org/10.3390/ijms232113156
Chicago/Turabian StylePaik, Benjamin, and Louis Tong. 2022. "Topical Omega-3 Fatty Acids Eyedrops in the Treatment of Dry Eye and Ocular Surface Disease: A Systematic Review" International Journal of Molecular Sciences 23, no. 21: 13156. https://doi.org/10.3390/ijms232113156
APA StylePaik, B., & Tong, L. (2022). Topical Omega-3 Fatty Acids Eyedrops in the Treatment of Dry Eye and Ocular Surface Disease: A Systematic Review. International Journal of Molecular Sciences, 23(21), 13156. https://doi.org/10.3390/ijms232113156






