Aging of Liver in Its Different Diseases
Abstract
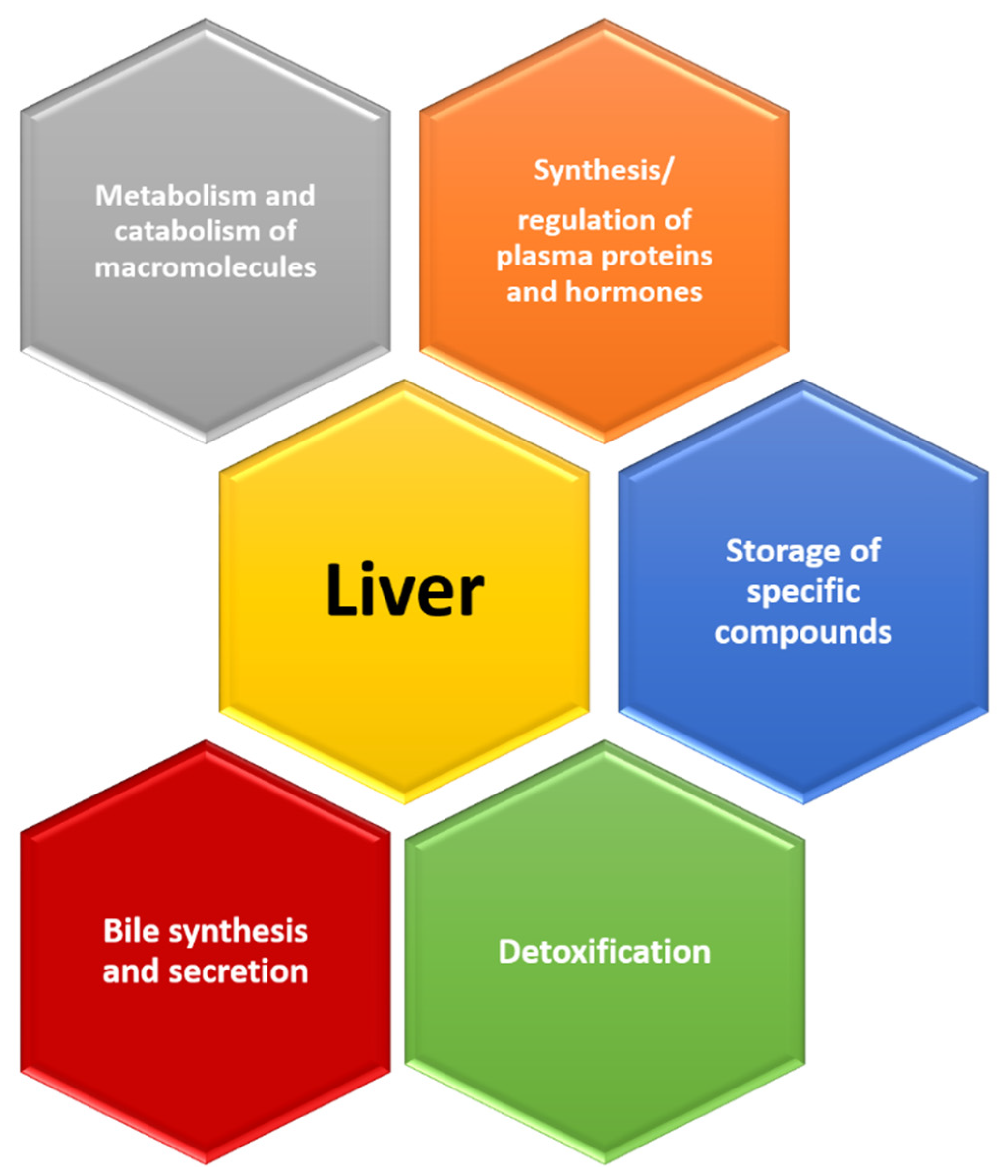
1. Introduction
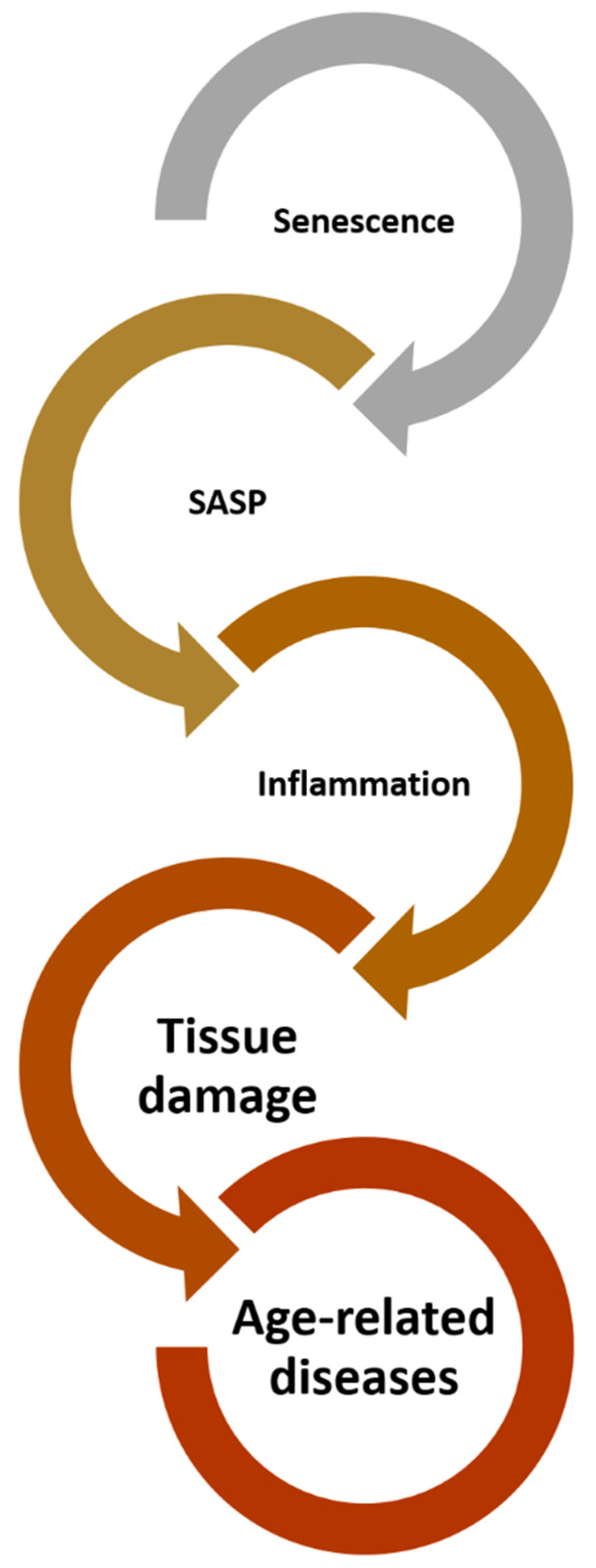
2. Cellular Senescence and Its Characteristics
3. Inflammaging
4. Signs That Reveal an Aging Liver
5. Liver Diseases and Aging
5.1. Non-Alcoholic Fatty Liver Disease (NAFLD), Non-Alcoholic Steatohepatitis (NASH), Alcoholic Steatohepatitis (ASH) and End-Stage Liver Disease
5.2. Viral Hepatitis
5.3. Hepatocellular Carcinoma (HCC)
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tchkonia, T.; Zhu, Y.; van Deursen, J.; Campisi, J.; Kirkland, J.L. Cellular Senescence and the Senescent Secretory Phenotype: Therapeutic Opportunities. J. Clin. Investig. 2013, 123, 966–972. [Google Scholar] [CrossRef] [PubMed]
- McHugh, D.; Gil, J. Senescence and Aging: Causes, Consequences, and Therapeutic Avenues. J. Cell Biol. 2018, 217, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Ogrodnik, M. Cellular Aging beyond Cellular Senescence: Markers of Senescence Prior to Cell Cycle Arrest in Vitro and in Vivo. Aging Cell 2021, 20, e13338. [Google Scholar] [CrossRef] [PubMed]
- Shmulevich, R.; Krizhanovsky, V. Cell Senescence, DNA Damage, and Metabolism. Antioxid. Redox Signal. 2021, 34, 324–334. [Google Scholar] [CrossRef] [PubMed]
- Kumari, R.; Jat, P. Mechanisms of Cellular Senescence: Cell Cycle Arrest and Senescence Associated Secretory Phenotype. Front. Cell Dev. Biol. 2021, 9, 645593. [Google Scholar] [CrossRef]
- d’Adda di Fagagna, F. Living on a Break: Cellular Senescence as a DNA-Damage Response. Nat. Rev. Cancer 2008, 8, 512–522. [Google Scholar] [CrossRef]
- Campisi, J. Aging, Cellular Senescence, and Cancer. Annu. Rev. Physiol. 2013, 75, 685–705. [Google Scholar] [CrossRef] [PubMed]
- Salama, R.; Sadaie, M.; Hoare, M.; Narita, M. Cellular Senescence and Its Effector Programs. Genes Dev. 2014, 28, 99–114. [Google Scholar] [CrossRef]
- Sikora, E.; Bielak-Zmijewska, A.; Mosieniak, G. A Common Signature of Cellular Senescence; Does It Exist? Ageing Res. Rev. 2021, 71, 101458. [Google Scholar] [CrossRef]
- Popper, H. Relations between Liver and Aging. Semin. Liver Dis. 1985, 5, 221–227. [Google Scholar] [CrossRef]
- Regev, A.; Schiff, E.R. Liver Disease in the Elderly. Gastroenterol. Clin. N. Am. 2001, 30, 547–563. [Google Scholar] [CrossRef]
- Hoare, M.; Das, T.; Alexander, G. Ageing, Telomeres, Senescence, and Liver Injury. J. Hepatol. 2010, 53, 950–961. [Google Scholar] [CrossRef] [PubMed]
- Morsiani, C.; Bacalini, M.G.; Santoro, A.; Garagnani, P.; Collura, S.; D’Errico, A.; de Eguileor, M.; Grazi, G.L.; Cescon, M.; Franceschi, C.; et al. The Peculiar Aging of Human Liver: A Geroscience Perspective within Transplant Context. Ageing Res. Rev. 2019, 51, 24–34. [Google Scholar] [CrossRef]
- Cho, E.; Cho, H.A.; Jun, C.H.; Kim, H.J.; Cho, S.B.; Choi, S.K. A Review of Hepatocellular Carcinoma in Elderly Patients Focused on Management and Outcomes. In Vivo 2019, 33, 1411–1420. [Google Scholar] [CrossRef]
- Saleh, T.; Tyutyunyk-Massey, L.; Gewirtz, D.A. Tumor Cell Escape from Therapy-Induced Senescence as a Model of Disease Recurrence after Dormancy. Cancer Res. 2019, 79, 1044–1046. [Google Scholar] [CrossRef] [PubMed]
- Milanovic, M.; Fan, D.N.Y.; Belenki, D.; Däbritz, J.H.M.; Zhao, Z.; Yu, Y.; Dörr, J.R.; Dimitrova, L.; Lenze, D.; Monteiro Barbosa, I.A.; et al. Senescence-associated reprogramming promotes cancer stemness. Nature 2018, 553, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Elmore, L.W.; Di, X.; Dumur, C.; Holt, S.E.; Gewirtz, D.A. Evasion of a single-step, chemotherapy-induced senescence in breast cancer cells: Implications for treatment response. Clin. Cancer Res. 2005, 11, 2637–2643. [Google Scholar] [CrossRef]
- Roberson, R.S.; Kussick, S.J.; Vallieres, E.; Chen, S.-Y.J.; Wu, D.Y. Escape from therapy-induced accelerated cellular senescence in p53-null lung cancer cells and in human lung cancers. Cancer Res. 2005, 65, 2795–2803. [Google Scholar] [CrossRef] [PubMed]
- Zampetidis, C.P.; Galanos, P.; Angelopoulou, A.; Zhu, Y.; Polyzou, A.; Karamitros, T.; Kotsinas, A.; Lagopati, N.; Mourkioti, I.; Mirzazadeh, R.; et al. A recurrent chromosomal inversion suffices for driving escape from oncogene-induced senescence via subTAD reorganization. Mol. Cell 2021, 81, 4907–4923.e8. [Google Scholar] [CrossRef]
- Kwon, S.M.; Hong, S.M.; Lee, Y.-K.; Min, S.; Yoon, G. Metabolic Features and Regulation in Cell Senescence. BMB Rep. 2019, 52, 5–12. [Google Scholar] [CrossRef]
- Young, A.R.J.; Narita, M.; Narita, M. Cell Senescence as Both a Dynamic and a Static Phenotype. Methods Mol. Biol. 2013, 965, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Birch, J.; Gil, J. Senescence and the SASP: Many Therapeutic Avenues. Genes Dev. 2020, 34, 1565–1576. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Espín, D.; Cañamero, M.; Maraver, A.; Gómez-López, G.; Contreras, J.; Murillo-Cuesta, S.; Rodríguez-Baeza, A.; Varela-Nieto, I.; Ruberte, J.; Collado, M.; et al. Programmed Cell Senescence during Mammalian Embryonic Development. Cell 2013, 155, 1104–1118. [Google Scholar] [CrossRef] [PubMed]
- Storer, M.; Mas, A.; Robert-Moreno, A.; Pecoraro, M.; Ortells, M.C.; Di Giacomo, V.; Yosef, R.; Pilpel, N.; Krizhanovsky, V.; Sharpe, J.; et al. Senescence Is a Developmental Mechanism That Contributes to Embryonic Growth and Patterning. Cell 2013, 155, 1119–1130. [Google Scholar] [CrossRef]
- Nassrally, M.S.; Lau, A.; Wise, K.; John, N.; Kotecha, S.; Lee, K.L.; Brooks, R.F. Cell Cycle Arrest in Replicative Senescence Is Not an Immediate Consequence of Telomere Dysfunction. Mech. Ageing Dev. 2019, 179, 11–22. [Google Scholar] [CrossRef]
- Gorgoulis, V.; Adams, P.D.; Alimonti, A.; Bennett, D.C.; Bischof, O.; Bishop, C.; Campisi, J.; Collado, M.; Evangelou, K.; Ferbeyre, G.; et al. Cellular Senescence: Defining a Path Forward. Cell 2019, 179, 813–827. [Google Scholar] [CrossRef]
- Neurohr, G.E.; Terry, R.L.; Lengefeld, J.; Bonney, M.; Brittingham, G.P.; Moretto, F.; Miettinen, T.P.; Vaites, L.P.; Soares, L.M.; Paulo, J.A.; et al. Excessive Cell Growth Causes Cytoplasm Dilution And Contributes to Senescence. Cell 2019, 176, 1083–1097.e18. [Google Scholar] [CrossRef]
- Ogrodnik, M.; Salmonowicz, H.; Gladyshev, V.N. Integrating Cellular Senescence with the Concept of Damage Accumulation in Aging: Relevance for Clearance of Senescent Cells. Aging Cell 2019, 18, e12841. [Google Scholar] [CrossRef]
- Yi, S.; Lin, K.; Jiang, T.; Shao, W.; Huang, C.; Jiang, B.; Li, Q.; Lin, D. NMR-Based Metabonomic Analysis of HUVEC Cells during Replicative Senescence. Aging 2020, 12, 3626–3646. [Google Scholar] [CrossRef]
- Zwerschke, W.; Mazurek, S.; Stöckl, P.; Hütter, E.; Eigenbrodt, E.; Jansen-Dürr, P. Metabolic Analysis of Senescent Human Fibroblasts Reveals a Role for AMP in Cellular Senescence. Biochem. J. 2003, 376 Pt 2, 403–411. [Google Scholar] [CrossRef]
- Lunt, S.Y.; Vander Heiden, M.G. Aerobic Glycolysis: Meeting the Metabolic Requirements of Cell Proliferation. Annu. Rev. Cell Dev. Biol. 2011, 27, 441–464. [Google Scholar] [CrossRef] [PubMed]
- Jolly, R.D.; Douglas, B.V.; Davey, P.M.; Roiri, J.E. Lipofuscin in Bovine Muscle and Brain: A Model for Studying Age Pigment. Gerontology 1995, 41 (Suppl. 2), 283–295. [Google Scholar] [CrossRef] [PubMed]
- Lim, C.-S.; Potts, M.; Helm, R.F. Nicotinamide Extends the Replicative Life Span of Primary Human Cells. Mech. Ageing Dev. 2006, 127, 511–514. [Google Scholar] [CrossRef] [PubMed]
- Matuoka, K.; Chen, K.Y.; Takenawa, T. Rapid Reversion of Aging Phenotypes by Nicotinamide through Possible Modulation of Histone Acetylation. Cell Mol. Life Sci. 2001, 58, 2108–2116. [Google Scholar] [CrossRef]
- Fumagalli, M.; Rossiello, F.; Clerici, M.; Barozzi, S.; Cittaro, D.; Kaplunov, J.M.; Bucci, G.; Dobreva, M.; Matti, V.; Beausejour, C.M.; et al. Telomeric DNA Damage Is Irreparable and Causes Persistent DNA-Damage-Response Activation. Nat. Cell Biol. 2012, 14, 355–365. [Google Scholar] [CrossRef]
- Mao, P.; Liu, J.; Zhang, Z.; Zhang, H.; Liu, H.; Gao, S.; Rong, Y.S.; Zhao, Y. Homologous Recombination-Dependent Repair of Telomeric DSBs in Proliferating Human Cells. Nat. Commun. 2016, 7, 12154. [Google Scholar] [CrossRef]
- Palm, W.; de Lange, T. How Shelterin Protects Mammalian Telomeres. Annu. Rev. Genet. 2008, 42, 301–334. [Google Scholar] [CrossRef]
- van Steensel, B.; Smogorzewska, A.; de Lange, T. TRF2 Protects Human Telomeres from End-to-End Fusions. Cell 1998, 92, 401–413. [Google Scholar] [CrossRef]
- Bernadotte, A.; Mikhelson, V.M.; Spivak, I.M. Markers of Cellular Senescence. Telomere Shortening as a Marker of Cellular Senescence. Aging 2016, 8, 3–11. [Google Scholar] [CrossRef]
- Rodier, F.; Coppé, J.-P.; Patil, C.K.; Hoeijmakers, W.A.M.; Muñoz, D.P.; Raza, S.R.; Freund, A.; Campeau, E.; Davalos, A.R.; Campisi, J. Persistent DNA Damage Signalling Triggers Senescence-Associated Inflammatory Cytokine Secretion. Nat. Cell Biol. 2009, 11, 973–979. [Google Scholar] [CrossRef]
- White, R.R.; Vijg, J. Do DNA Double-Strand Breaks Drive Aging? Mol. Cell. 2016, 63, 729–738. [Google Scholar] [CrossRef] [PubMed]
- Pustovalova, M.; Grekhova, A.; Astrelina, T.; Nikitina, V.; Dobrovolskaya, E.; Suchkova, Y.; Kobzeva, I.; Usupzhanova, D.; Vorobyeva, N.; Samoylov, A.; et al. Accumulation of Spontaneous ΓH2AX Foci in Long-Term Cultured Mesenchymal Stromal Cells. Aging 2016, 8, 3498–3506. [Google Scholar] [CrossRef] [PubMed]
- Gelot, C.; Magdalou, I.; Lopez, B.S. Replication Stress in Mammalian Cells and Its Consequences for Mitosis. Genes 2015, 6, 267–298. [Google Scholar] [CrossRef] [PubMed]
- Zorin, V.; Grekhova, A.; Pustovalova, M.; Zorina, A.; Smetanina, N.; Vorobyeva, N.; Kopnin, P.; Gilmutdinova, I.; Moskalev, A.; Osipov, A.N.; et al. Spontaneous ΓH2AX Foci in Human Dermal Fibroblasts in Relation to Proliferation Activity and Aging. Aging 2019, 11, 4536–4546. [Google Scholar] [CrossRef]
- Serrano, M.; Lin, A.W.; McCurrach, M.E.; Beach, D.; Lowe, S.W. Oncogenic Ras Provokes Premature Cell Senescence Associated with Accumulation of P53 and P16INK4a. Cell 1997, 88, 593–602. [Google Scholar] [CrossRef]
- Lin, A.W.; Barradas, M.; Stone, J.C.; van Aelst, L.; Serrano, M.; Lowe, S.W. Premature Senescence Involving P53 and P16 Is Activated in Response to Constitutive MEK/MAPK Mitogenic Signaling. Genes Dev. 1998, 12, 3008–3019. [Google Scholar] [CrossRef] [PubMed]
- Bartkova, J.; Rezaei, N.; Liontos, M.; Karakaidos, P.; Kletsas, D.; Issaeva, N.; Vassiliou, L.-V.F.; Kolettas, E.; Niforou, K.; Zoumpourlis, V.C.; et al. Oncogene-Induced Senescence Is Part of the Tumorigenesis Barrier Imposed by DNA Damage Checkpoints. Nature 2006, 444, 633–637. [Google Scholar] [CrossRef] [PubMed]
- Di Micco, R.; Fumagalli, M.; Cicalese, A.; Piccinin, S.; Gasparini, P.; Luise, C.; Schurra, C.; Garre’, M.; Nuciforo, P.G.; Bensimon, A.; et al. Oncogene-Induced Senescence Is a DNA Damage Response Triggered by DNA Hyper-Replication. Nature 2006, 444, 638–642. [Google Scholar] [CrossRef] [PubMed]
- Mallette, F.A.; Gaumont-Leclerc, M.-F.; Ferbeyre, G. The DNA Damage Signaling Pathway Is a Critical Mediator of Oncogene-Induced Senescence. Genes Dev. 2007, 21, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Cesare, A.J.; Karlseder, J. A Three-State Model of Telomere Control over Human Proliferative Boundaries. Curr. Opin. Cell Biol. 2012, 24, 731–738. [Google Scholar] [CrossRef]
- Campisi, J. Senescent Cells, Tumor Suppression, and Organismal Aging: Good Citizens, Bad Neighbors. Cell 2005, 120, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Kuilman, T.; Peeper, D.S. Senescence-Messaging Secretome: SMS-Ing Cellular Stress. Nat. Rev. Cancer 2009, 9, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Passos, J.F.; Nelson, G.; Wang, C.; Richter, T.; Simillion, C.; Proctor, C.J.; Miwa, S.; Olijslagers, S.; Hallinan, J.; Wipat, A.; et al. Feedback between P21 and Reactive Oxygen Production Is Necessary for Cell Senescence. Mol. Syst. Biol. 2010, 6, 347. [Google Scholar] [CrossRef]
- Coppé, J.-P.; Desprez, P.-Y.; Krtolica, A.; Campisi, J. The Senescence-Associated Secretory Phenotype: The Dark Side of Tumor Suppression. Annu. Rev. Pathol. 2010, 5, 99–118. [Google Scholar] [CrossRef] [PubMed]
- Lujambio, A.; Akkari, L.; Simon, J.; Grace, D.; Tschaharganeh, D.F.; Bolden, J.E.; Zhao, Z.; Thapar, V.; Joyce, J.A.; Krizhanovsky, V.; et al. Non-Cell-Autonomous Tumor Suppression by P53. Cell 2013, 153, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Kuilman, T.; Michaloglou, C.; Vredeveld, L.C.W.; Douma, S.; van Doorn, R.; Desmet, C.J.; Aarden, L.A.; Mooi, W.J.; Peeper, D.S. Oncogene-Induced Senescence Relayed by an Interleukin-Dependent Inflammatory Network. Cell 2008, 133, 1019–1031. [Google Scholar] [CrossRef] [PubMed]
- Acosta, J.C.; O’Loghlen, A.; Banito, A.; Guijarro, M.V.; Augert, A.; Raguz, S.; Fumagalli, M.; Da Costa, M.; Brown, C.; Popov, N.; et al. Chemokine Signaling via the CXCR2 Receptor Reinforces Senescence. Cell 2008, 133, 1006–1018. [Google Scholar] [CrossRef]
- Jing, H.; Kase, J.; Dörr, J.R.; Milanovic, M.; Lenze, D.; Grau, M.; Beuster, G.; Ji, S.; Reimann, M.; Lenz, P.; et al. Opposing Roles of NF-ΚB in Anti-Cancer Treatment Outcome Unveiled by Cross-Species Investigations. Genes Dev. 2011, 25, 2137–2146. [Google Scholar] [CrossRef]
- Orjalo, A.V.; Bhaumik, D.; Gengler, B.K.; Scott, G.K.; Campisi, J. Cell Surface-Bound IL-1alpha Is an Upstream Regulator of the Senescence-Associated IL-6/IL-8 Cytokine Network. Proc. Natl. Acad. Sci. USA 2009, 106, 17031–17036. [Google Scholar] [CrossRef]
- Hoare, M.; Narita, M. Transmitting Senescence to the Cell Neighbourhood. Nat. Cell Biol. 2013, 15, 887–889. [Google Scholar] [CrossRef]
- Nelson, G.; Wordsworth, J.; Wang, C.; Jurk, D.; Lawless, C.; Martin-Ruiz, C.; von Zglinicki, T. A Senescent Cell Bystander Effect: Senescence-Induced Senescence. Aging Cell 2012, 11, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Nelson, G.; Kucheryavenko, O.; Wordsworth, J.; von Zglinicki, T. The Senescent Bystander Effect Is Caused by ROS-Activated NF-ΚB Signalling. Mech. Ageing Dev. 2018, 170, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Acosta, J.C.; Banito, A.; Wuestefeld, T.; Georgilis, A.; Janich, P.; Morton, J.P.; Athineos, D.; Kang, T.-W.; Lasitschka, F.; Andrulis, M.; et al. A Complex Secretory Program Orchestrated by the Inflammasome Controls Paracrine Senescence. Nat. Cell Biol. 2013, 15, 978–990. [Google Scholar] [CrossRef] [PubMed]
- Bavik, C.; Coleman, I.; Dean, J.P.; Knudsen, B.; Plymate, S.; Nelson, P.S. The Gene Expression Program of Prostate Fibroblast Senescence Modulates Neoplastic Epithelial Cell Proliferation through Paracrine Mechanisms. Cancer Res. 2006, 66, 794–802. [Google Scholar] [CrossRef]
- Krtolica, A.; Parrinello, S.; Lockett, S.; Desprez, P.Y.; Campisi, J. Senescent Fibroblasts Promote Epithelial Cell Growth and Tumorigenesis: A Link between Cancer and Aging. Proc. Natl. Acad. Sci. USA 2001, 98, 12072–12077. [Google Scholar] [CrossRef]
- Krizhanovsky, V.; Yon, M.; Dickins, R.A.; Hearn, S.; Simon, J.; Miething, C.; Yee, H.; Zender, L.; Lowe, S.W. Senescence of Activated Stellate Cells Limits Liver Fibrosis. Cell 2008, 134, 657–667. [Google Scholar] [CrossRef]
- Xue, W.; Zender, L.; Miething, C.; Dickins, R.A.; Hernando, E.; Krizhanovsky, V.; Cordon-Cardo, C.; Lowe, S.W. Senescence and Tumour Clearance Is Triggered by P53 Restoration in Murine Liver Carcinomas. Nature 2007, 445, 656–660. [Google Scholar] [CrossRef]
- Iannello, A.; Thompson, T.W.; Ardolino, M.; Lowe, S.W.; Raulet, D.H. P53-Dependent Chemokine Production by Senescent Tumor Cells Supports NKG2D-Dependent Tumor Elimination by Natural Killer Cells. J. Exp. Med. 2013, 210, 2057–2069. [Google Scholar] [CrossRef]
- Kohli, J.; Wang, B.; Brandenburg, S.M.; Basisty, N.; Evangelou, K.; Varela-Eirin, M.; Campisi, J.; Schilling, B.; Gorgoulis, V.; Demaria, M. Algorithmic Assessment of Cellular Senescence in Experimental and Clinical Specimens. Nat. Protoc. 2021, 16, 2471–2498. [Google Scholar] [CrossRef]
- Chung, H.Y.; Cesari, M.; Anton, S.; Marzetti, E.; Giovannini, S.; Seo, A.Y.; Carter, C.; Yu, B.P.; Leeuwenburgh, C. Molecular Inflammation: Underpinnings of Aging and Age-Related Diseases. Ageing Res. Rev. 2009, 8, 18–30. [Google Scholar] [CrossRef]
- Yu, B.P.; Chung, H.Y. Adaptive Mechanisms to Oxidative Stress during Aging. Mech. Ageing Dev. 2006, 127, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Campisi, J. Chronic Inflammation (Inflammaging) and Its Potential Contribution to Age-Associated Diseases. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69 (Suppl. 1), S4–S9. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.J.; Wijshake, T.; Tchkonia, T.; LeBrasseur, N.K.; Childs, B.G.; van de Sluis, B.; Kirkland, J.L.; van Deursen, J.M. Clearance of P16Ink4a-Positive Senescent Cells Delays Ageing-Associated Disorders. Nature 2011, 479, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.J.; Childs, B.G.; Durik, M.; Wijers, M.E.; Sieben, C.J.; Zhong, J.; Saltness, R.A.; Jeganathan, K.B.; Verzosa, G.C.; Pezeshki, A.; et al. Naturally Occurring P16(Ink4a)-Positive Cells Shorten Healthy Lifespan. Nature 2016, 530, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Capri, M.; Monti, D.; Giunta, S.; Olivieri, F.; Sevini, F.; Panourgia, M.P.; Invidia, L.; Celani, L.; Scurti, M.; et al. Inflammaging and Anti-Inflammaging: A Systemic Perspective on Aging and Longevity Emerged from Studies in Humans. Mech. Ageing Dev. 2007, 128, 92–105. [Google Scholar] [CrossRef]
- Vasto, S.; Candore, G.; Balistreri, C.R.; Caruso, M.; Colonna-Romano, G.; Grimaldi, M.P.; Listi, F.; Nuzzo, D.; Lio, D.; Caruso, C. Inflammatory Networks in Ageing, Age-Related Diseases and Longevity. Mech. Ageing Dev. 2007, 128, 83–91. [Google Scholar] [CrossRef]
- Fagiolo, U.; Cossarizza, A.; Scala, E.; Fanales-Belasio, E.; Ortolani, C.; Cozzi, E.; Monti, D.; Franceschi, C.; Paganelli, R. Increased Cytokine Production in Mononuclear Cells of Healthy Elderly People. Eur. J. Immunol. 1993, 23, 2375–2378. [Google Scholar] [CrossRef]
- Naugler, W.E.; Karin, M. The Wolf in Sheep’s Clothing: The Role of Interleukin-6 in Immunity, Inflammation and Cancer. Trends Mol. Med. 2008, 14, 109–119. [Google Scholar] [CrossRef]
- Sparmann, A.; Bar-Sagi, D. Ras-Induced Interleukin-8 Expression Plays a Critical Role in Tumor Growth and Angiogenesis. Cancer Cell 2004, 6, 447–458. [Google Scholar] [CrossRef]
- Hubackova, S.; Krejcikova, K.; Bartek, J.; Hodny, Z. IL1- and TGFβ-Nox4 Signaling, Oxidative Stress and DNA Damage Response Are Shared Features of Replicative, Oncogene-Induced, and Drug-Induced Paracrine “Bystander Senescence”. Aging 2012, 4, 932–951. [Google Scholar] [CrossRef]
- Kim, E.-C.; Kim, J.-R. Senotherapeutics: Emerging Strategy for Healthy Aging and Age-Related Disease. BMB Rep. 2019, 52, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Tchkonia, T.; Jiang, J.; Kirkland, J.L.; Sun, Y. Targeting Senescent Cells for a Healthier Aging: Challenges and Opportunities. Adv. Sci. 2020, 7, 2002611. [Google Scholar] [CrossRef] [PubMed]
- Moiseeva, O.; Deschênes-Simard, X.; St-Germain, E.; Igelmann, S.; Huot, G.; Cadar, A.E.; Bourdeau, V.; Pollak, M.N.; Ferbeyre, G. Metformin Inhibits the Senescence-Associated Secretory Phenotype by Interfering with IKK/NF-ΚB Activation. Aging Cell 2013, 12, 489–498. [Google Scholar] [CrossRef]
- Childs, B.G.; Baker, D.J.; Wijshake, T.; Conover, C.A.; Campisi, J.; van Deursen, J.M. Senescent Intimal Foam Cells Are Deleterious at All Stages of Atherosclerosis. Science 2016, 354, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Bussian, T.J.; Aziz, A.; Meyer, C.F.; Swenson, B.L.; van Deursen, J.M.; Baker, D.J. Clearance of Senescent Glial Cells Prevents Tau-Dependent Pathology and Cognitive Decline. Nature 2018, 562, 578–582. [Google Scholar] [CrossRef]
- Blagosklonny, M.V. Anti-Aging: Senolytics or Gerostatics (Unconventional View). Oncotarget 2021, 12, 1821–1835. [Google Scholar] [CrossRef]
- Zhu, Y.; Tchkonia, T.; Pirtskhalava, T.; Gower, A.C.; Ding, H.; Giorgadze, N.; Palmer, A.K.; Ikeno, Y.; Hubbard, G.B.; Lenburg, M.; et al. The Achilles’ Heel of Senescent Cells: From Transcriptome to Senolytic Drugs. Aging Cell 2015, 14, 644–658. [Google Scholar] [CrossRef]
- Lagoumtzi, S.M.; Chondrogianni, N. Senolytics and Senomorphics: Natural and Synthetic Therapeutics in the Treatment of Aging and Chronic Diseases. Free Radic. Biol. Med. 2021, 171, 169–190. [Google Scholar] [CrossRef]
- Schmucker, D.L. Hepatocyte Fine Structure during Maturation and Senescence. J. Electron. Microsc. Tech. 1990, 14, 106–125. [Google Scholar] [CrossRef]
- Kudryavtsev, B.N.; Kudryavtseva, M.V.; Sakuta, G.A.; Stein, G.I. Human Hepatocyte Polyploidization Kinetics in the Course of Life Cycle. Virchows Arch. B Cell Pathol. Incl. Mol. Pathol. 1993, 64, 387–393. [Google Scholar] [CrossRef]
- Salmonowicz, H.; Passos, J.F. Detecting Senescence: A New Method for an Old Pigment. Aging Cell 2017, 16, 432–434. [Google Scholar] [CrossRef] [PubMed]
- Skoczyńska, A.; Budzisz, E.; Trznadel-Grodzka, E.; Rotsztejn, H. Melanin and Lipofuscin as Hallmarks of Skin Aging. Postep. Dermatol. Alergol. 2017, 34, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Rivas, S.; Mucyn, T.; Van Den Burg, H.A.; Vervoort, J.; Jones, J.D.G. An ~400 KDa Membrane-Associated Complex That Contains One Molecule of the Resistance Protein Cf-4. Plant J. 2017, 90, 1214. [Google Scholar] [CrossRef] [PubMed]
- Jung, T.; Bader, N.; Grune, T. Lipofuscin: Formation, distribution, and metabolic consequences. Ann. N. Y. Acad. Sci. 2007, 1119, 97–111. [Google Scholar] [CrossRef]
- Georgakopoulou, E.A.; Tsimaratou, K.; Evangelou, K.; Fernandez Marcos, P.J.; Zoumpourlis, V.; Trougakos, I.P.; Kletsas, D.; Bartek, J.; Serrano, M.; Gorgoulis, V.G. Specific lipofuscin staining as a novel biomarker to detect replicative and stress-induced senescence. A method applicable in cryo-preserved and archival tissues. Aging 2013, 5, 37–50. [Google Scholar] [CrossRef]
- Moreno-García, A.; Kun, A.; Calero, O.; Medina, M.; Calero, M. An Overview of the Role of Lipofuscin in Age-Related Neurodegeneration. Front. Neurosci. 2018, 12, 464. [Google Scholar] [CrossRef]
- Verma, S.; Tachtatzis, P.; Penrhyn-Lowe, S.; Scarpini, C.; Jurk, D.; Von Zglinicki, T.; Coleman, N.; Alexander, G.J.M. Sustained Telomere Length in Hepatocytes and Cholangiocytes with Increasing Age in Normal Liver. Hepatology 2012, 56, 1510–1520. [Google Scholar] [CrossRef]
- Kitada, T.; Seki, S.; Kawakita, N.; Kuroki, T.; Monna, T. Telomere Shortening in Chronic Liver Diseases. Biochem. Biophys. Res. Commun. 1995, 211, 33–39. [Google Scholar] [CrossRef]
- Urabe, Y.; Nouso, K.; Higashi, T.; Nakatsukasa, H.; Hino, N.; Ashida, K.; Kinugasa, N.; Yoshida, K.; Uematsu, S.; Tsuji, T. Telomere Length in Human Liver Diseases. Liver 1996, 16, 293–297. [Google Scholar] [CrossRef]
- Sekoguchi, S.; Nakajima, T.; Moriguchi, M.; Jo, M.; Nishikawa, T.; Katagishi, T.; Kimura, H.; Minami, M.; Itoh, Y.; Kagawa, K.; et al. Role of Cell-Cycle Turnover and Oxidative Stress in Telomere Shortening and Cellular Senescence in Patients with Chronic Hepatitis C. J. Gastroenterol. Hepatol. 2007, 22, 182–190. [Google Scholar] [CrossRef]
- Kojima, H.; Yokosuka, O.; Imazeki, F.; Saisho, H.; Omata, M. Telomerase Activity and Telomere Length in Hepatocellular Carcinoma and Chronic Liver Disease. Gastroenterology 1997, 112, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Wiemann, S.U.; Satyanarayana, A.; Buer, J.; Kamino, K.; Manns, M.P.; Rudolph, K.L. Contrasting Effects of Telomere Shortening on Organ Homeostasis, Tumor Suppression, and Survival during Chronic Liver Damage. Oncogene 2005, 24, 1501–1509. [Google Scholar] [CrossRef]
- Plentz, R.R.; Caselitz, M.; Bleck, J.S.; Gebel, M.; Flemming, P.; Kubicka, S.; Manns, M.P.; Rudolph, K.L. Hepatocellular Telomere Shortening Correlates with Chromosomal Instability and the Development of Human Hepatoma. Hepatology 2004, 40, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Capri, M.; Olivieri, F.; Lanzarini, C.; Remondini, D.; Borelli, V.; Lazzarini, R.; Graciotti, L.; Albertini, M.C.; Bellavista, E.; Santoro, A.; et al. Identification of MiR-31-5p, MiR-141-3p, MiR-200c-3p, and GLT1 as Human Liver Aging Markers Sensitive to Donor-Recipient Age-Mismatch in Transplants. Aging Cell 2017, 16, 262–272. [Google Scholar] [CrossRef] [PubMed]
- Shenvi, S.V.; Smith, E.; Hagen, T.M. Identification of Age-Specific Nrf2 Binding to a Novel Antioxidant Response Element Locus in the Gclc Promoter: A Compensatory Means for the Loss of Glutathione Synthetic Capacity in the Aging Rat Liver? Aging Cell 2012, 11, 297–304. [Google Scholar] [CrossRef]
- Bacalini, M.G.; Franceschi, C.; Gentilini, D.; Ravaioli, F.; Zhou, X.; Remondini, D.; Pirazzini, C.; Giuliani, C.; Marasco, E.; Gensous, N.; et al. Molecular Aging of Human Liver: An Epigenetic/Transcriptomic Signature. J. Gerontol. A Biol. Sci. Med. Sci. 2019, 74, 1–8. [Google Scholar] [CrossRef]
- Calder, P.C.; Bosco, N.; Bourdet-Sicard, R.; Capuron, L.; Delzenne, N.; Doré, J.; Franceschi, C.; Lehtinen, M.J.; Recker, T.; Salvioli, S.; et al. Health Relevance of the Modification of Low Grade Inflammation in Ageing (Inflammageing) and the Role of Nutrition. Ageing Res. Rev. 2017, 40, 95–119. [Google Scholar] [CrossRef]
- Schmidt-Arras, D.; Rose-John, S. IL-6 Pathway in the Liver: From Physiopathology to Therapy. J. Hepatol. 2016, 64, 1403–1415. [Google Scholar] [CrossRef]
- Grizzi, F.; Di Caro, G.; Laghi, L.; Hermonat, P.; Mazzola, P.; Nguyen, D.D.; Radhi, S.; Figueroa, J.A.; Cobos, E.; Annoni, G.; et al. Mast Cells and the Liver Aging Process. Immun. Ageing 2013, 10, 9. [Google Scholar] [CrossRef]
- Lebeaupin, C.; Proics, E.; de Bieville, C.H.D.; Rousseau, D.; Bonnafous, S.; Patouraux, S.; Adam, G.; Lavallard, V.J.; Rovere, C.; Thuc, O.L.; et al. ER Stress Induces NLRP3 Inflammasome Activation and Hepatocyte Death. Cell Death Dis. 2015, 6, e1879. [Google Scholar] [CrossRef]
- Saito, Y.; Morine, Y.; Shimada, M. Mechanism of Impairment on Liver Regeneration in Elderly Patients: Role of Hepatic Stellate Cell Function. Hepatol. Res. 2017, 47, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Benkdane, M.; Teixeira-Clerc, F.; Bonnafous, S.; Louvet, A.; Lafdil, F.; Pecker, F.; Tran, A.; Gual, P.; Mallat, A.; et al. M2 Kupffer Cells Promote M1 Kupffer Cell Apoptosis: A Protective Mechanism against Alcoholic and Nonalcoholic Fatty Liver Disease. Hepatology 2014, 59, 130–142. [Google Scholar] [CrossRef] [PubMed]
- Sheedfar, F.; Di Biase, S.; Koonen, D.; Vinciguerra, M. Liver Diseases and Aging: Friends or Foes? Aging Cell 2013, 12, 950–954. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.; Tas, E.; Yakar, S.; Muzumdar, R. Hepatic Lipid Metabolism and Non-Alcoholic Fatty Liver Disease in Aging. Mol. Cell Endocrinol. 2017, 455, 115–130. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Charlton, M.; Cusi, K.; Rinella, M.; Harrison, S.A.; Brunt, E.M.; Sanyal, A.J. The Diagnosis and Management of Nonalcoholic Fatty Liver Disease: Practice Guidance from the American Association for the Study of Liver Diseases. Hepatology 2018, 67, 328–357. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global Epidemiology of Nonalcoholic Fatty Liver Disease-Meta-Analytic Assessment of Prevalence, Incidence, and Outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef]
- Cariou, B.; Byrne, C.D.; Loomba, R.; Sanyal, A.J. Nonalcoholic Fatty Liver Disease as a Metabolic Disease in Humans: A Literature Review. Diabetes Obes. Metab. 2021, 23, 1069–1083. [Google Scholar] [CrossRef]
- Ratziu, V.; Giral, P.; Charlotte, F.; Bruckert, E.; Thibault, V.; Theodorou, I.; Khalil, L.; Turpin, G.; Opolon, P.; Poynard, T. Liver Fibrosis in Overweight Patients. Gastroenterology 2000, 118, 1117–1123. [Google Scholar] [CrossRef]
- Angulo, P.; Keach, J.C.; Batts, K.P.; Lindor, K.D. Independent Predictors of Liver Fibrosis in Patients with Nonalcoholic Steatohepatitis. Hepatology 1999, 30, 1356–1362. [Google Scholar] [CrossRef] [PubMed]
- Branković, M.; Jovanović, I.; Dukić, M.; Radonjić, T.; Oprić, S.; Klašnja, S.; Zdravković, M. Lipotoxicity as the Leading Cause of Non-Alcoholic Steatohepatitis. Int. J. Mol. Sci. 2022, 23, 5146. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R. Alcoholic Steatohepatitis (ASH) and Alcoholic Hepatitis (AH): Cascade of Events, Clinical Aspects, and Pharmacotherapy Options. Expert Opin. Pharmacother. 2018, 19, 779–793. [Google Scholar] [CrossRef] [PubMed]
- Engin, A. Non-Alcoholic Fatty Liver Disease. Adv. Exp. Med. Biol. 2017, 960, 443–467. [Google Scholar] [CrossRef] [PubMed]
- Marra, F.; Svegliati-Baroni, G. Lipotoxicity and the Gut-Liver Axis in NASH Pathogenesis. J. Hepatol. 2018, 68, 280–295. [Google Scholar] [CrossRef]
- Forrest, E.H.; Evans, C.D.J.; Stewart, S.; Phillips, M.; Oo, Y.H.; McAvoy, N.C.; Fisher, N.C.; Singhal, S.; Brind, A.; Haydon, G.; et al. Analysis of Factors Predictive of Mortality in Alcoholic Hepatitis and Derivation and Validation of the Glasgow Alcoholic Hepatitis Score. Gut 2005, 54, 1174–1179. [Google Scholar] [CrossRef] [PubMed]
- Durand, F.; Levitsky, J.; Cauchy, F.; Gilgenkrantz, H.; Soubrane, O.; Francoz, C. Age and Liver Transplantation. J. Hepatol. 2019, 70, 745–758. [Google Scholar] [CrossRef]
- Lué, A.; Solanas, E.; Baptista, P.; Lorente, S.; Araiz, J.J.; Garcia-Gil, A.; Serrano, M.T. How Important Is Donor Age in Liver Transplantation? World J. Gastroenterol. 2016, 22, 4966–4976. [Google Scholar] [CrossRef]
- Capri, M.; Franceschi, C.; Cescon, M. Biological Age of Transplanted Livers. Aging 2018, 10, 156–157. [Google Scholar] [CrossRef] [PubMed]
- Sanz, N.; Díez-Fernández, C.; Alvarez, A.M.; Fernández-Simón, L.; Cascales, M. Age-Related Changes on Parameters of Experimentally-Induced Liver Injury and Regeneration. Toxicol. Appl. Pharmacol. 1999, 154, 40–49. [Google Scholar] [CrossRef]
- Fry, M.; Silber, J.; Loeb, L.A.; Martin, G.M. Delayed and Reduced Cell Replication and Diminishing Levels of DNA Polymerase-Alpha in Regenerating Liver of Aging Mice. J. Cell Physiol. 1984, 118, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Gagliano, N.; Grizzi, F.; Annoni, G. Mechanisms of Aging and Liver Functions. Dig. Dis. 2007, 25, 118–123. [Google Scholar] [CrossRef]
- Forbes, A.; Williams, R. Changing Epidemiology and Clinical Aspects of Hepatitis A. Br. Med. Bull. 1990, 46, 303–318. [Google Scholar] [CrossRef] [PubMed]
- Junaidi, O.; Di Bisceglie, A.M. Aging Liver and Hepatitis. Clin. Geriatr. Med. 2007, 23, 889–903. [Google Scholar] [CrossRef] [PubMed]
- Forbes, A.; Williams, R. Increasing Age—An Important Adverse Prognostic Factor in Hepatitis A Virus Infection. J. R. Coll. Physicians 1988, 22, 237–239. [Google Scholar]
- Advisory Committee on Immunization Practices (ACIP); Fiore, A.E.; Wasley, A.; Bell, B.P. Prevention of Hepatitis A through Active or Passive Immunization: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm. Rep. 2006, 55, 1–23. [Google Scholar]
- Trépo, C.; Chan, H.L.Y.; Lok, A. Hepatitis B Virus Infection. Lancet 2014, 384, 2053–2063. [Google Scholar] [CrossRef]
- Sugauchi, F.; Mizokami, M.; Orito, E.; Ohno, T.; Kato, H.; Maki, M.; Suzuki, H.; Ojika, K.; Ueda, R. Hepatitis B Virus Infection among Residents of a Nursing Home for the Elderly: Seroepidemiological Study and Molecular Evolutionary Analysis. J. Med. Virol. 2000, 62, 456–462. [Google Scholar] [CrossRef]
- Karakousis, N.D.; Papatheodoridi, A.; Chatzigeorgiou, A.; Papatheodoridis, G. Cellular Senescence and Hepatitis B-Related Hepatocellular Carcinoma: An Intriguing Link. Liver Int. 2020, 40, 2917–2927. [Google Scholar] [CrossRef]
- Dekker, S.E.; Green, E.W.; Ahn, J. Treatment and Prevention of Acute Hepatitis B Virus. Clin. Liver Dis. 2021, 25, 711–724. [Google Scholar] [CrossRef]
- Schillie, S.; Vellozzi, C.; Reingold, A.; Harris, A.; Haber, P.; Ward, J.W.; Nelson, N.P. Prevention of Hepatitis B Virus Infection in the United States: Recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm. Rep. 2018, 67, 1–31. [Google Scholar] [CrossRef]
- Pardee, M. Diagnosis and Management of Hepatitis B and C. Nurs. Clin. N. Am. 2019, 54, 277–284. [Google Scholar] [CrossRef]
- Manns, M.P.; Buti, M.; Gane, E.; Pawlotsky, J.-M.; Razavi, H.; Terrault, N.; Younossi, Z. Hepatitis C Virus Infection. Nat. Rev. Dis. Prim. 2017, 3, 17006. [Google Scholar] [CrossRef] [PubMed]
- Ansaldi, F.; Orsi, A.; Sticchi, L.; Bruzzone, B.; Icardi, G. Hepatitis C Virus in the New Era: Perspectives in Epidemiology, Prevention, Diagnostics and Predictors of Response to Therapy. World J. Gastroenterol. 2014, 20, 9633–9652. [Google Scholar] [CrossRef]
- Yen, H.-H.; Su, P.-Y.; Liu, I.-L.; Zeng, Y.-Y.; Huang, S.-P.; Hsu, Y.-C.; Yang, C.-W.; Chen, Y.-Y. Direct-Acting Antiviral Treatment for Hepatitis C Virus in Geriatric Patients: A Real-World Retrospective Comparison between Early and Late Elderly Patients. PeerJ 2021, 9, e10944. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-F.; Yu, M.-L. Treating Hepatitis C in the Elderly: Pharmacotherapeutic Considerations and Developments. Expert Opin. Pharmacother. 2017, 18, 1867–1874. [Google Scholar] [CrossRef]
- Strader, D.B. Understudied Populations with Hepatitis C. Hepatology 2002, 36 (Suppl. 1), S226–S236. [Google Scholar] [CrossRef] [PubMed]
- Imai, Y.; Kasahara, A.; Tanaka, H.; Okanoue, T.; Hiramatsu, N.; Tsubouchi, H.; Yoshioka, K.; Kawata, S.; Tanaka, E.; Hino, K.; et al. Interferon Therapy for Aged Patients with Chronic Hepatitis C: Improved Survival in Patients Exhibiting a Biochemical Response. J. Gastroenterol. 2004, 39, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Thabut, D.; Le Calvez, S.; Thibault, V.; Massard, J.; Munteanu, M.; Di Martino, V.; Ratziu, V.; Poynard, T. Hepatitis C in 6,865 Patients 65 Yr or Older: A Severe and Neglected Curable Disease? Am. J. Gastroenterol. 2006, 101, 1260–1267. [Google Scholar] [CrossRef]
- Nudo, C.G.; Wong, P.; Hilzenrat, N.; Deschênes, M. Elderly Patients Are at Greater Risk of Cytopenia during Antiviral Therapy for Hepatitis C. Can. J. Gastroenterol. 2006, 20, 589–592. [Google Scholar] [CrossRef] [PubMed]
- El-Serag, H.B.; Rudolph, K.L. Hepatocellular Carcinoma: Epidemiology and Molecular Carcinogenesis. Gastroenterology 2007, 132, 2557–2576. [Google Scholar] [CrossRef]
- Nishikawa, H.; Kimura, T.; Kita, R.; Osaki, Y. Treatment for Hepatocellular Carcinoma in Elderly Patients: A Literature Review. J. Cancer 2013, 4, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Tsujita, E.; Utsunomiya, T.; Ohta, M.; Tagawa, T.; Matsuyama, A.; Okazaki, J.; Yamamoto, M.; Tsutsui, S.-I.; Ishida, T. Outcome of Repeat Hepatectomy in Patients with Hepatocellular Carcinoma Aged 75 Years and Older. Surgery 2010, 147, 696–703. [Google Scholar] [CrossRef]
- Hung, A.K.; Guy, J. Hepatocellular Carcinoma in the Elderly: Meta-Analysis and Systematic Literature Review. World J. Gastroenterol. 2015, 21, 12197–12210. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, A.; Koike, K.; Nishino, H. Clinical Features and Prognosis of Elderly Patients with Hepatocellular Carcinoma Not Indicated for Surgical Resection. Geriatr. Gerontol. Int. 2017, 17, 189–201. [Google Scholar] [CrossRef]
- Yasui, K.; Hashimoto, E.; Tokushige, K.; Koike, K.; Shima, T.; Kanbara, Y.; Saibara, T.; Uto, H.; Takami, S.; Kawanaka, M.; et al. Clinical and Pathological Progression of Non-Alcoholic Steatohepatitis to Hepatocellular Carcinoma. Hepatol. Res. 2012, 42, 767–773. [Google Scholar] [CrossRef] [PubMed]
- Honda, T.; Miyaaki, H.; Ichikawa, T.; Taura, N.; Miuma, S.; Shibata, H.; Isomoto, H.; Takeshima, F.; Nakao, K. Clinical Characteristics of Hepatocellular Carcinoma in Elderly Patients. Oncol. Lett. 2011, 2, 851–854. [Google Scholar] [CrossRef]
- Shirabe, K.; Kajiyama, K.; Harimoto, N.; Gion, T.; Tsujita, E.; Abe, T.; Wakiyama, S.; Nagaie, T.; Maehara, Y. Early Outcome Following Hepatic Resection in Patients Older than 80 Years of Age. World J. Surg. 2009, 33, 1927–1932. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, H.; Arimoto, A.; Wakasa, T.; Kita, R.; Kimura, T.; Osaki, Y. Surgical Resection for Hepatocellular Carcinoma: Clinical Outcomes and Safety in Elderly Patients. Eur. J. Gastroenterol. Hepatol. 2013, 25, 912–919. [Google Scholar] [CrossRef] [PubMed]
- Ueno, M.; Hayami, S.; Tani, M.; Kawai, M.; Hirono, S.; Yamaue, H. Recent Trends in Hepatectomy for Elderly Patients with Hepatocellular Carcinoma. Surg. Today 2014, 44, 1651–1659. [Google Scholar] [CrossRef]
- Poulou, L.S.; Botsa, E.; Thanou, I.; Ziakas, P.D.; Thanos, L. Percutaneous Microwave Ablation vs Radiofrequency Ablation in the Treatment of Hepatocellular Carcinoma. World J. Hepatol. 2015, 7, 1054–1063. [Google Scholar] [CrossRef] [PubMed]
- Oishi, K.; Itamoto, T.; Kobayashi, T.; Oshita, A.; Amano, H.; Ohdan, H.; Tashiro, H.; Asahara, T. Hepatectomy for Hepatocellular Carcinoma in Elderly Patients Aged 75 Years or More. J. Gastrointest. Surg. 2009, 13, 695–701. [Google Scholar] [CrossRef]
- Hiraoka, A.; Michitaka, K.; Horiike, N.; Hidaka, S.; Uehara, T.; Ichikawa, S.; Hasebe, A.; Miyamoto, Y.; Ninomiya, T.; Sogabe, I.; et al. Radiofrequency Ablation Therapy for Hepatocellular Carcinoma in Elderly Patients. J. Gastroenterol. Hepatol. 2010, 25, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, H.; Osaki, Y.; Iguchi, E.; Takeda, H.; Ohara, Y.; Sakamoto, A.; Hatamaru, K.; Henmi, S.; Saito, S.; Nasu, A.; et al. Percutaneous Radiofrequency Ablation for Hepatocellular Carcinoma: Clinical Outcome and Safety in Elderly Patients. J. Gastrointestin. Liver Dis. 2012, 21, 397–405. [Google Scholar] [PubMed]
- Cohen, M.J.; Bloom, A.I.; Barak, O.; Klimov, A.; Nesher, T.; Shouval, D.; Levi, I.; Shibolet, O. Trans-Arterial Chemo-Embolization Is Safe and Effective for Very Elderly Patients with Hepatocellular Carcinoma. World J. Gastroenterol. 2013, 19, 2521–2528. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, H.; Kita, R.; Kimura, T.; Ohara, Y.; Takeda, H.; Sakamoto, A.; Saito, S.; Nishijima, N.; Nasu, A.; Komekado, H.; et al. Transcatheter Arterial Chemoembolization for Intermediate-Stage Hepatocellular Carcinoma: Clinical Outcome and Safety in Elderly Patients. J. Cancer 2014, 5, 590–597. [Google Scholar] [CrossRef]


| Virus | Genetic Material | Family | Transmission | Complication |
|---|---|---|---|---|
| Hepatitis A (HAV) | RNA | Picornaviridae | Feco-oral | Acute liver failure |
| Hepatitis B (HBV) | DNA | Hepadnaviridae | Parenteral or sexual | Liver cirrhosis, HCC |
| Hepatitis C (HCV) | RNA | Flaviviridae | Parenteral or sexual | Liver cirrhosis, HCC |
| Hepatocellular Carcinoma (HCC) Facts | Treatment |
|---|---|
| Elderly patients are mostly negative for hepatotropic viruses. | Surgical |
| Elderly patients are more likely to have HCV than other hepatotropic viruses. | Radiofrequency ablation (RFA) |
| Elderly patients are more likely to be female. | Transarterial chemoembolization (TACE) |
| NASH-related-HCC occurs more often in elderly than in young people. | |
| Liver fibrosis is less pronounced in elderly patients. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radonjić, T.; Dukić, M.; Jovanović, I.; Zdravković, M.; Mandić, O.; Popadić, V.; Popović, M.; Nikolić, N.; Klašnja, S.; Divac, A.; et al. Aging of Liver in Its Different Diseases. Int. J. Mol. Sci. 2022, 23, 13085. https://doi.org/10.3390/ijms232113085
Radonjić T, Dukić M, Jovanović I, Zdravković M, Mandić O, Popadić V, Popović M, Nikolić N, Klašnja S, Divac A, et al. Aging of Liver in Its Different Diseases. International Journal of Molecular Sciences. 2022; 23(21):13085. https://doi.org/10.3390/ijms232113085
Chicago/Turabian StyleRadonjić, Tijana, Marija Dukić, Igor Jovanović, Marija Zdravković, Olga Mandić, Višeslav Popadić, Maja Popović, Novica Nikolić, Slobodan Klašnja, Anica Divac, and et al. 2022. "Aging of Liver in Its Different Diseases" International Journal of Molecular Sciences 23, no. 21: 13085. https://doi.org/10.3390/ijms232113085
APA StyleRadonjić, T., Dukić, M., Jovanović, I., Zdravković, M., Mandić, O., Popadić, V., Popović, M., Nikolić, N., Klašnja, S., Divac, A., Todorović, Z., & Branković, M. (2022). Aging of Liver in Its Different Diseases. International Journal of Molecular Sciences, 23(21), 13085. https://doi.org/10.3390/ijms232113085













