Role of Cannabidiol for Improvement of the Quality of Life in Cancer Patients: Potential and Challenges
Abstract
1. Introduction
2. Methodology for Search and Evaluation of Literature
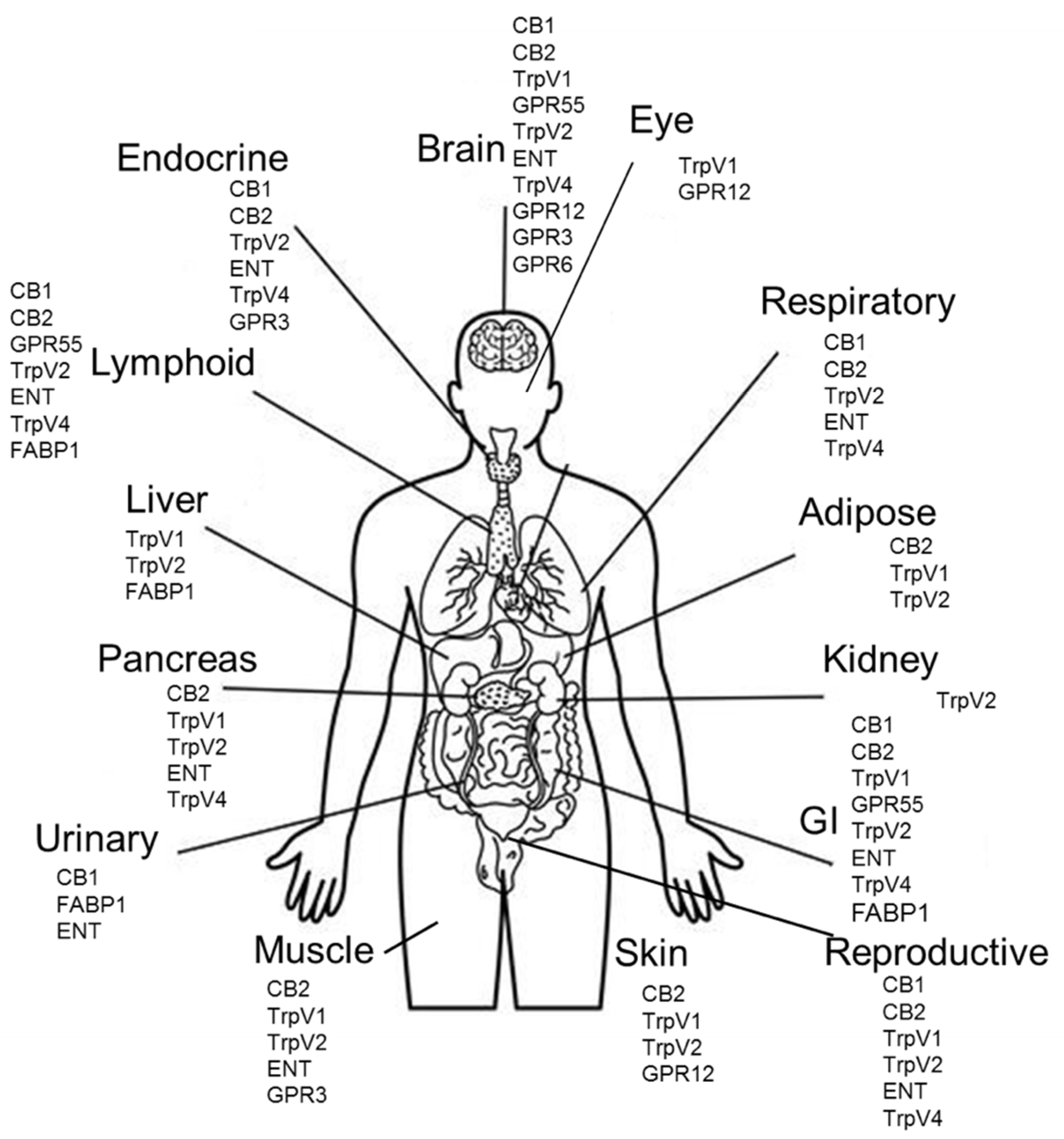
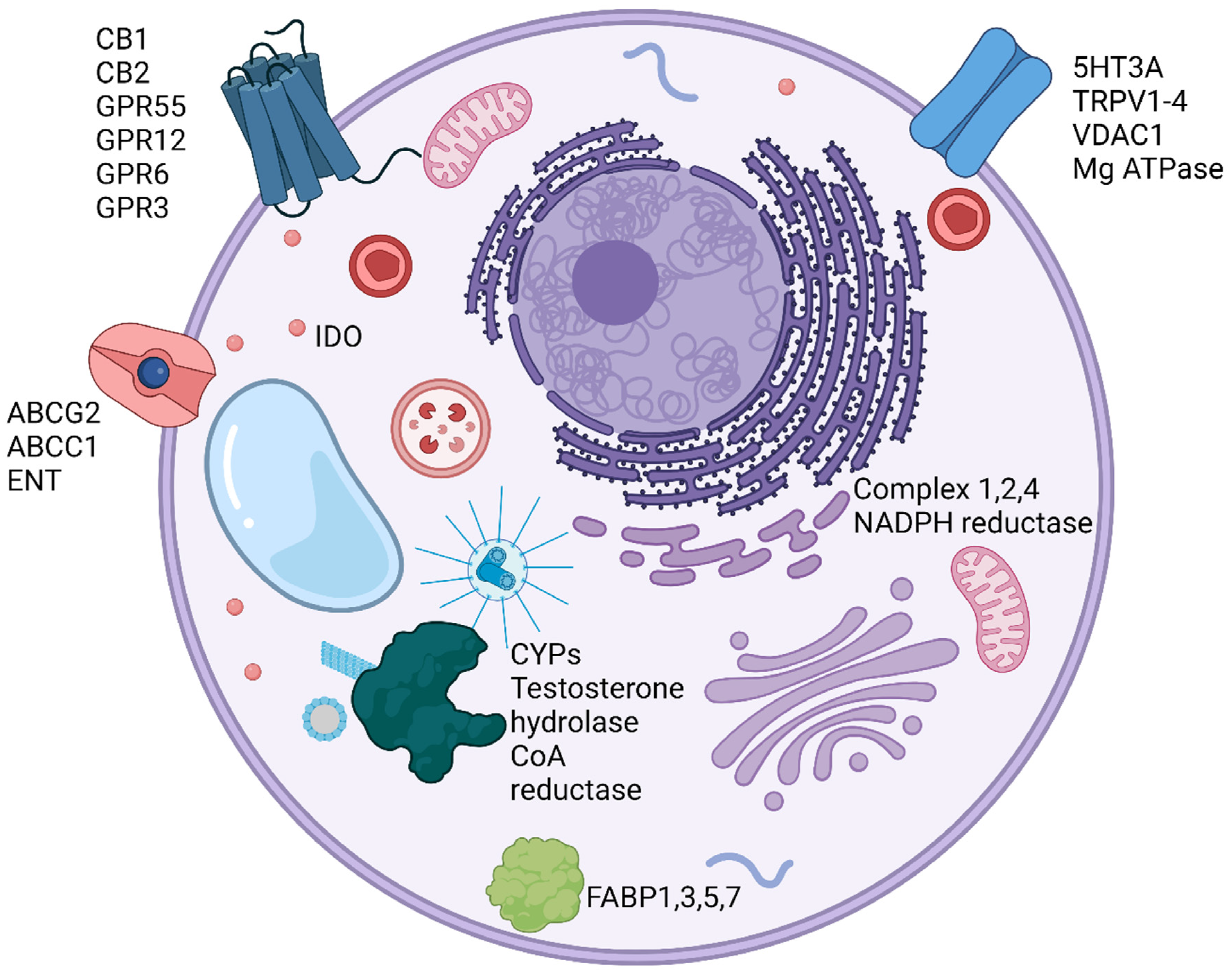
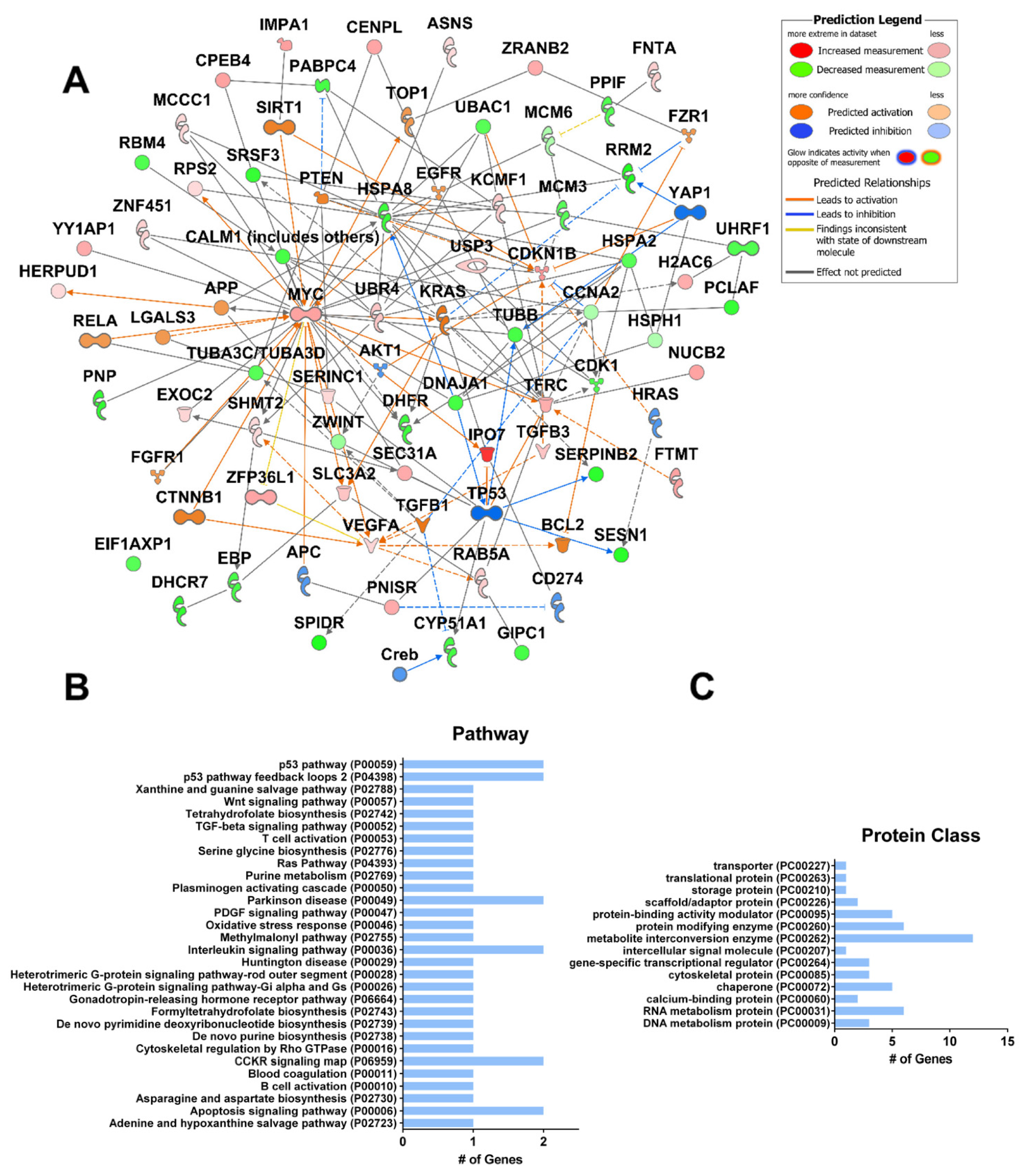
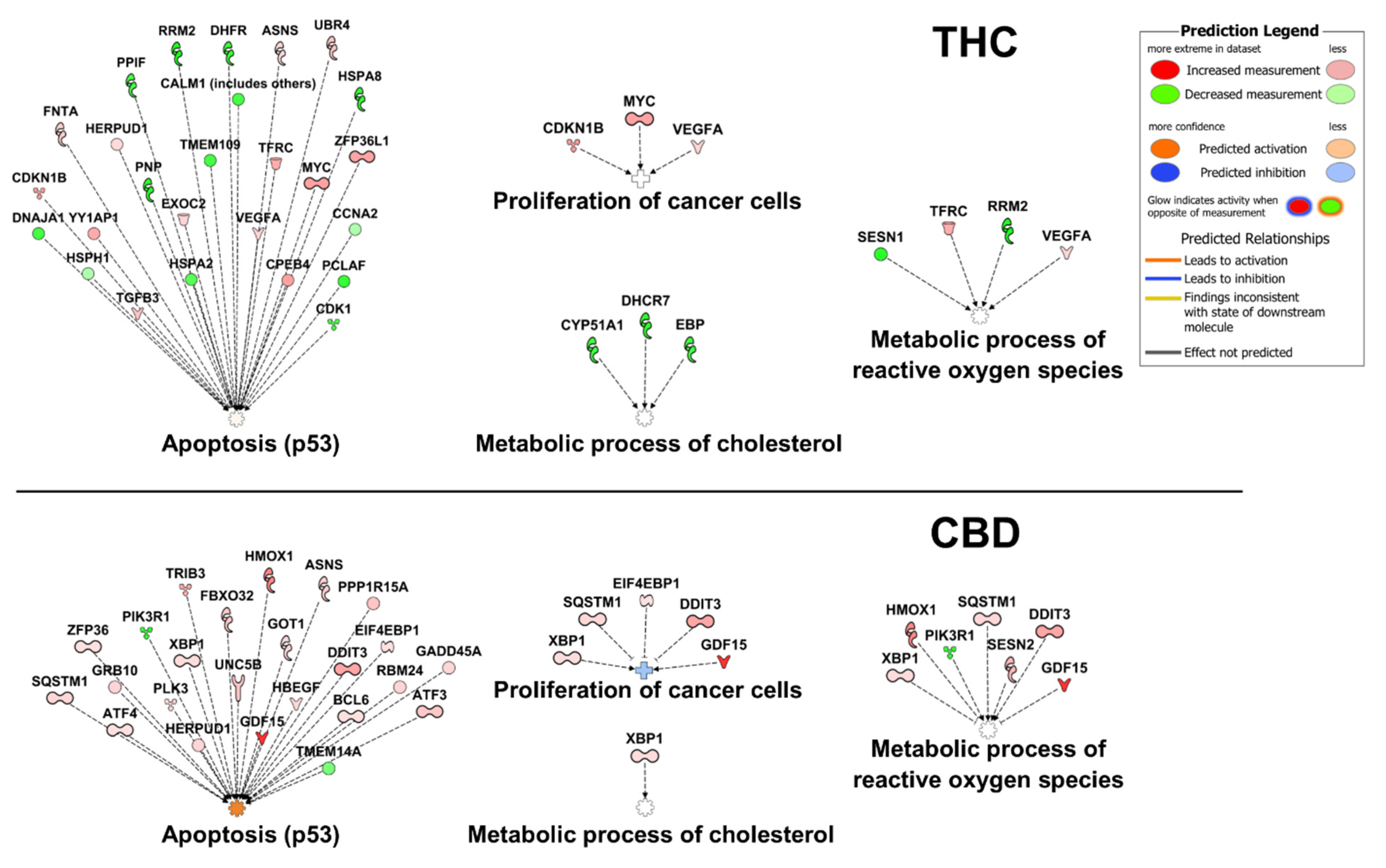
3. Biology and Molecular Targets of CBD
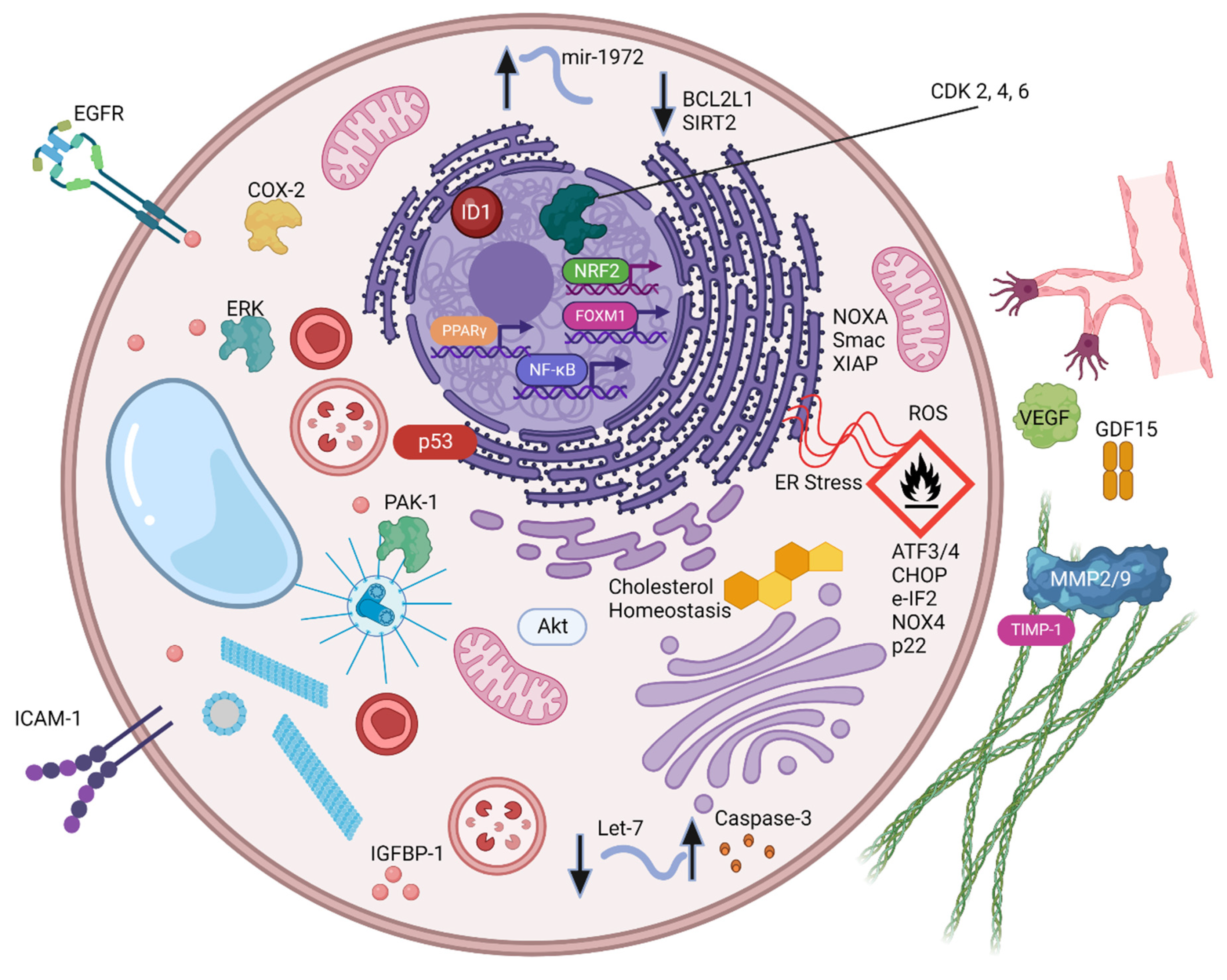
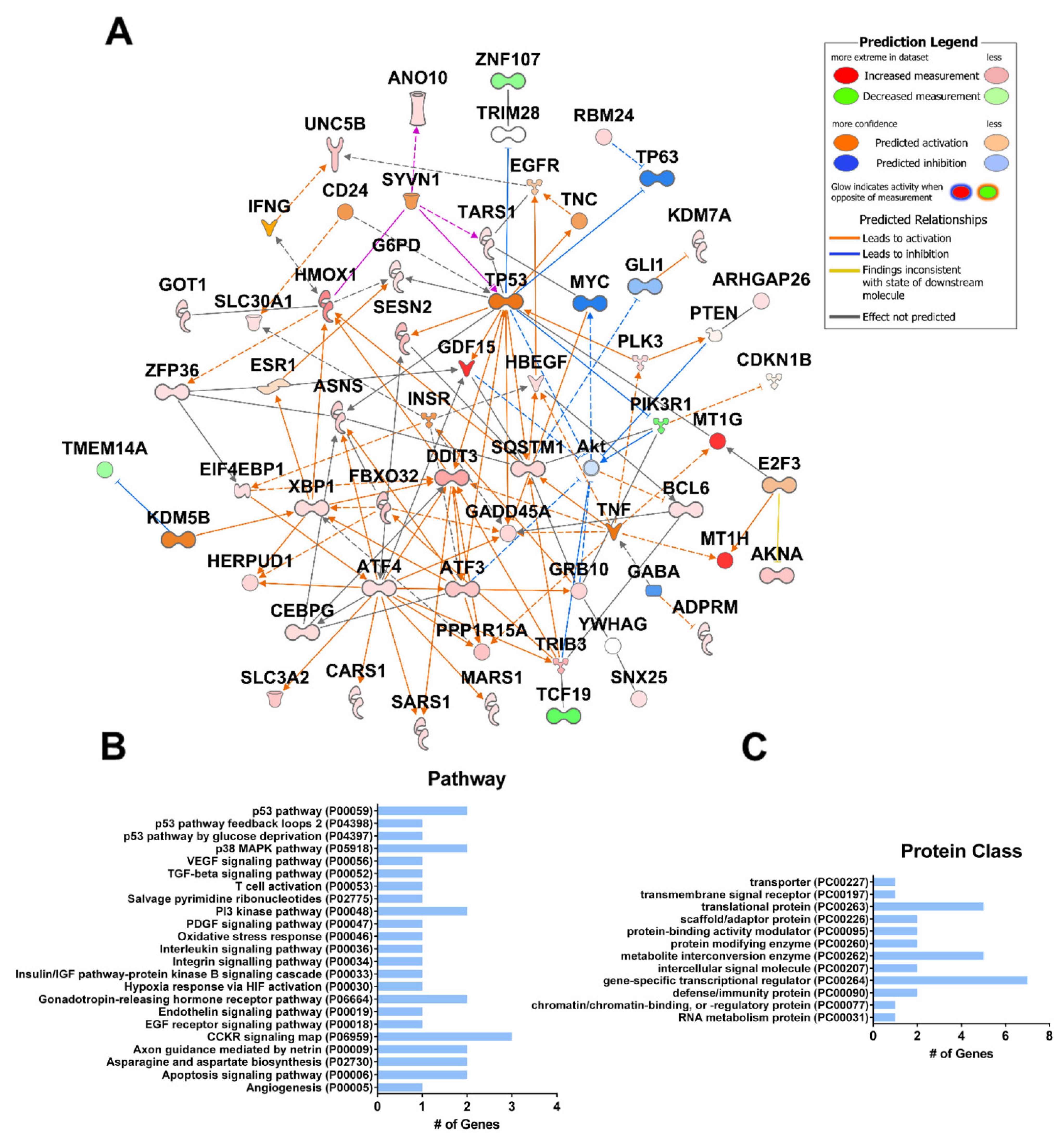
4. Pre-Clinical Studies: Role of CBD as a Component of Combination Therapies for Cancer
| Summary of Preclinical Studies Involving CBD Treatment of Cancer Cells and Mouse Tumors | |||
|---|---|---|---|
| Preclinical Systems | Pathways | Molecules and Mechanisms Involved | Ref. |
| Cell Culture Models | |||
| Colorectal cancer cells: HCT-116, SW480/620, Caco-2 | Cell cycle arrest | cyclin D3, CDK2, CDK4, and CDK6 | [12] |
| Apoptosis-ROS-ER stress pathway | p-eIF2 α, ATF3, ATF4, CHOP | [49] | |
| EMT | WNT/β-catenin | [68] | |
| Leukemia cells | ROS | NADPH oxidases Nox4, p22-phox | [50] |
| Glioblastoma cells | ROS | SoX2, ID1, STAT3 | [46] |
| ROS/apoptosis | Cholesterol | [55] | |
| Hepatocellular carcinoma cells: HCC | Pyroptosis | AKT pathway: inducing IGFBP-1 and inhibiting the AKT/GSK3β LDH release and cleavage of caspase-3, PARP, and GSDME | [65] |
| Lung cells: A549, H460, and primary lung tumor cells | Apoptosis | COX-2, PPAR-γ, and prostaglandin | [41] |
| Breast (ER+ and ER−): T-47D | EGF signaling | EGFR, AKT, NF-κB, MMP2, and MMP9 | [58] |
| Gastric cells | Increased ROS cell cycle arrest, apoptosis | Cleaved caspases-3 and 9, decreased Bcl-2 levels and mitochondrial dysfunction | [13,61] |
| Cervical/Pancreatic cells | Apoptosis | p53, caspase 3, BAX, and inhibiting KRAS-activated PAK-1 signaling | [62,63] |
| Neuroblastoma cells | Apoptosis | hsa-let-7a, has-mir-1972: caspase-3(UR) and BCL2L1 and SIRT2 (DR) | [51,64] |
| Animal Models | |||
| Colorectal cancer HCT-116 subcutaneous model | Anti-tumor | ROS, NOXA activation | [49] |
| Mouse model of glioblastoma | Anti-tumor | ROS | [46] |
| Colon: CT26 | Apoptosis | VEGF | [59] |
| Lung: A549 xenograft mouse model | Anti-tumor | ICAM-1 and TIMP-1, p42/44 MAPK | [60] |
| Trial (N)-Design | Intervention | Results | Ref. |
|---|---|---|---|
| NCT00530764 (N = 360) A Study of Sativex® for Pain Relief in Patients with Advanced Malignancy (SPRAY). Duration: 9 weeks Design: Double blind, randomized, placebo-controlled, parallel group, dose-range exploration study. Results Posted: 17 June 2011 | Experimental: Sativex Each 100 μL oromucosal spray actuation delivered: (2.7 mg THC + 2.5 mg CBD) Low Dose: Range of 1 to 4 sprays per day. Maximum daily dose: 10.8 mg THC and 10 mg CBD. Medium Dose: Range of 6 to 10 sprays per day. Maximum daily dose: 27 mg THC and 25 mg CBD. High Dose: Range of 11 to 16 sprays per day. Maximum daily dose: 43.2 mg THC and 40 mg CBD. Placebo: 1–16 sprays per day | -Number of patients with at least 30% improvement in Numerical Rating Scale (NRS) average pain score from baseline was not significant with any dose. -Change in cumulative average pain response was significant in low and med doses. -Change in sleep disruption NRS was significant in low dose. -No change in Montgomery Asberg Depression Rating Scale (MADRS). | [78] |
| NCT00675948 (N = 43) Study to Compare the Safety and Tolerability of Sativex® in Patients with Cancer-Related Pain. Duration: 2 weeks Design: multicenter, open-label, self-titrated dose Results Posted: 13 September 2012 | Sativex® or GW-2000-02 oromucosal spray (self-administered, self-titrated). Sativex: THC 27 mg/mL + CBD 25 mg/mL Maximum tolerated/allowable dose was 130 mg THC and 120 mg CBD in each 24 h period. GW-2000-02: THC 27 mg/mL. Maximum tolerated/allowable dose was 130 mg THC in each 24 h period. | -Change from baseline in mean Brief Pain Inventory-Short Form scores for “pain severity” and “worst pain” decreased (improvement) at each visit in the THC/CBD spray group. -The EORTC Quality of Life Questionnaire (C30 scores) decreased (improvement) from baseline for insomnia, pain, and fatigue. -No new safety concerns associated with the extended use of THC/CBD spray. | [79] |
| NCT01337089 (N = 660) Long-Term Safety of Sativex as Adjunctive Therapy in Patients with Uncontrolled Persistent Chronic Cancer-Related Pain. Duration: 6 months Design: multicenter, non-comparative, open label extension Results Posted: 23 April 2018 | Nabiximols (self-administered, self-titrated) oromucosal spray. Maximum of 10 sprays per day for 6 months. Nabiximols contained: THC 27 mg/mL CBD 25 mg/mL Each 100 μL actuation delivered: 2.7 mg THC 2.5 mg CBD | -No change from baseline in mean NRS average pain score. -No change from baseline in mean sleep disruption NRS. -Overall patient satisfaction was high. # of patients extremely satisfied: 56 (9.1%); very satisfied: 230 (37.2%); slightly satisfied: 185 (29.9%); neutral: 82 (13.3%). | [80] |
| NCT01361607 (N = 399) Sativex® for Relieving Persistent Pain in Patients with Advanced Cancer (SPRAY III). Duration: 9 weeks Design: multi-center, double-blind, randomized, placebo-controlled. Results Posted: 23 April 2018 | Experimental: Nabiximols (self-administered, self-titrated) oromucosal spray. Minimum 3 and maximum of 10 sprays per day for 5 weeks. Nabiximols contained: THC 27 mg/mL CBD 25 mg/mL Each 100 μL actuation delivered: 2.7 mg THC 2.5 mg CBD Placebo (GA-0034): Placebo (self-administered) 100 μL oromucosal spray. Maximum of 10 sprays per day for 5 weeks. Placebo contained ethanol: propylene glycol (50:50) | -Percent improvement from baseline in mean NRS average pain score: Ex. 7.2% (0.0 to 30.0) Placebo 9.5% (−2.9 to 25.7) -Change from baseline in mean sleep disruption NRS: Ex. −0.9 Placebo −1.1 -Change from baseline in daily total opioid use (morphine equivalent): Ex. −6.5 Placebo 2.3 -Change from baseline in daily breakthrough opioid dose (morphine equivalent): Ex. −4.4 Placebo 0.5 | [81] |
| NCT01262651 (N = 397) Sativex® for Relieving Persistent Pain in Participants with Advanced Cancer Duration: 9 weeks Design: multi-center, double-blind, randomized, placebo-controlled Results Posted: 23 April 2018 | Nabiximols (self-administered, self-titrated) oromucosal spray. Minimum 3 and maximum of 10 sprays per day for 5 weeks. Nabiximols contained: THC 27 mg/mL + CBD 25 mg/mL Each 100 μL actuation delivered: 2.7 mg THC + 2.5 mg CBD Placebo (GA-0034): Placebo (self-administered) 100 μL oromucosal spray. Maximum of 10 sprays per day for 5 weeks. Placebo contained ethanol: propylene glycol (50:50). | -Median percent improvements from baseline in average pain NRS score: Intention-to-treat population: Nabiximols: 10.7% (p = 0.0854), Placebo: 4.5% -Per-protocol population: Nabiximols: 15.5% (p = 0.0378), Placebo: 6.3% “Nabiximols was statistically superior to placebo on two of three quality-of-life instruments at Week 3 and on all three at Week 5. The safety profile of Nabiximols was consistent with earlier studies.” | [78,82] |
| NCT00674609 (N = 177) A Study of Sativex® for Pain Relief in Patients with Advanced Malignancy (SPRAY) Duration: 2 weeks Design: multicenter, double blind, randomized, placebo-controlled, parallel group study to evaluate the efficacy of Sativex® and GW-2000-02 Results Posted: 13 August 2012 | Sativex® (self-administered, self-titrated) oromucosal spray (100 µL/actuation). THC 27 mg/mL CBD 25 mg/mL Maximum tolerated/allowable dose was 130 mg THC and 120 mg CBD in each 24 h period. GW-1000-02: (THC Alone) (self-administered, self-titrated) THC 27 mg/mL. Maximum tolerated/allowable dose was 130 mg THC in each 24 h period. Placebo contained colorants and excipients. | -Change from baseline in mean pain NRS score was statistically significant for Sativex, but not GW-1000-02. -Patients with a reduction of more than 30% from baseline pain NRS score. -No change from baseline in median dose of opioid background medication or mean number of doses of breakthrough medication across treatment groups. -No significant group differences were found in the NRS sleep quality or nausea scores or the pain control assessment. | [83] |
| NCT01424566 (N = 406) A Two-Part Study of Sativex® Oromucosal Spray for Relieving Uncontrolled Persistent Pain in Patients with Advanced Cancer Duration: 11 weeks Design: multi-center; placebo-controlled; aimed to determine the efficacy, safety, and tolerability of Nabiximols. Results Posted: 23 April 2018 | Nabiximols (self-administered, self-titrated) oromucosal spray. Minimum 3 and maximum of 10 sprays per day for 2 or 7 weeks. Nabiximols contained: THC 27 mg/mL CBD 25 mg/mL With excipients Each 100 μL actuation delivered: 2.7 mg THC + 2.5 mg CBD Placebo was self-administered by participants as a 100 μL oromucosal spray up to a maximum of 10 sprays per day for 5 weeks. Placebo oromucosal spray contained ethanol:propylene glycol (50:50) with excipients. | -Overall percent improvement or mean change in pain NRS score was not statistically significant. -Statistically significant treatment effect for Sativex was identified in US patients <65 years (p = 0.040). “Treatment effect in favor of Sativex was observed on quality-of-life questionnaires, despite the fact that similar effects were not observed on NRS score. The safety profile of Sativex was consistent with earlier studies, and no evidence of abuse or misuse was identified.” | [81] |
| EudraCT trial no. 2014-005553-39 (N = 23): Effects of Sativex on Blood Leukocytes in Patients with Lymphoma/Chronic Lymphocytic Leukemia Duration: Single dose Design: Therapeutic exploratory (Phase II), open label, not placebo-controlled Results Posted: 17 January 2022 | Asymptomatic patients with CLL or marginal zone lymphomas received a single dose of THC/CBD, starting from 2.7 mg THC and 2.5 mg CBD (one actuation of Sativex) to 18.9 mg THC and 17.5 mg CBD. | -A significant reduction in leukemic B cells (median, 11%) occurred in the blood within two hours (p = 0.014), and remained for 6 h without induction of apoptosis or proliferation. All effects were gone by 24 h. Normal non-leukemic B cells and T cells were also reduced. | [84] |
5. Clinical Studies of CBD in Cancer
6. CBD Use in Rehabilitation of Cancer Patients
7. Pain
8. Sleep
9. Anxiety/Depression
10. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Moreno, T.; Montanes, F.; Tallon, S.J.; Fenton, T.; King, J.W. Extraction of cannabinoids from hemp (Cannabis sativa L.) using high pressure solvents: An overview of different processing options. J. Supercrit. Fluids 2020, 161, 104850. [Google Scholar] [CrossRef]
- Maccarrone, M.; Maldonado, R.; Casas, M.; Henze, T.; Centonze, D. Cannabinoids therapeutic use: What is our current understanding following the introduction of THC, THC: CBD oromucosal spray and others? Expert Rev. Clin. Pharmacol. 2017, 10, 443–455. [Google Scholar] [CrossRef]
- Nahler, G. Cannabidiol and Other Phytocannabinoids as Cancer Therapeutics. Pharm. Med. 2022, 36, 99–129. [Google Scholar] [CrossRef]
- Mlost, J.; Bryk, M.; Starowicz, K. Cannabidiol for Pain Treatment: Focus on Pharmacology and Mechanism of Action. Int. J. Mol. Sci. 2020, 21, 8870. [Google Scholar] [CrossRef]
- Shannon, S.; Lewis, N.; Lee, H.; Hughes, S. Cannabidiol in Anxiety and Sleep: A Large Case Series. Perm. J. 2019, 23, 18–041. [Google Scholar] [CrossRef]
- Spanagel, R.; Bilbao, A. Approved cannabinoids for medical purposes—Comparative systematic review and meta-analysis for sleep and appetite. Neuropharmacology 2021, 196, 108680. [Google Scholar] [CrossRef]
- Gaston, T.E.; Martin, R.C.; Szaflarski, J.P. Cannabidiol (CBD) and cognition in epilepsy. Epilepsy Behav. 2021, 124, 108316. [Google Scholar] [CrossRef]
- Lazarini-Lopes, W.; Silva, R.A.D.V.-D.; da Silva-Júnior, R.M.; Leite, J.P.; Garcia-Cairasco, N. The anticonvulsant effects of cannabidiol in experimental models of epileptic seizures: From behavior and mechanisms to clinical insights. Neurosci. Biobehav. Rev. 2020, 111, 166–182. [Google Scholar] [CrossRef]
- Good, P.; Greer, R.M.; Huggett, G.E.; Hardy, J.R. An Open-Label Pilot Study Testing the Feasibility of Assessing Total Symptom Burden in Trials of Cannabinoid Medications in Palliative Care. J. Palliat. Med. 2020, 23, 650–655. [Google Scholar] [CrossRef]
- Hardy, J.; Haywood, A.; Gogna, G.; Martin, J.; Yates, P.; Greer, R.; Good, P. Oral medicinal cannabinoids to relieve symptom burden in the palliative care of patients with advanced cancer: A double-blind, placebo-controlled, randomised clinical trial of efficacy and safety of 1:1 delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD). Trials 2020, 21, 110. [Google Scholar] [CrossRef]
- Todorova, J.; Lazarov, L.I.; Petrova, M.; Tzintzarov, A.; Ugrinova, I. The antitumor activity of cannabidiol on lung cancer cell lines A549 and H1299: The role of apoptosis. Biotechnol. Biotechnol. Equip. 2021, 35, 873–879. [Google Scholar] [CrossRef]
- Lee, H.-S.; Tamia, G.; Song, H.-J.; Amarakoon, D.; Wei, C.-I.; Lee, S.-H. Cannabidiol exerts anti-proliferative activity via a cannabinoid receptor 2-dependent mechanism in human colorectal cancer cells. Int. Immunopharmacol. 2022, 108, 108865. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Qin, Y.; Pan, Z.; Li, M.; Liu, X.; Chen, X.; Qu, G.; Zhou, L.; Xu, M.; Zheng, Q.; et al. Cannabidiol Induces Cell Cycle Arrest and Cell Apoptosis in Human Gastric Cancer SGC-7901 Cells. Biomolecules 2019, 9, 302. [Google Scholar] [CrossRef] [PubMed]
- Hamad, H.; Olsen, B.B. Cannabidiol Induces Cell Death in Human Lung Cancer Cells and Cancer Stem Cells. Pharmaceuticals 2021, 14, 1169. [Google Scholar] [CrossRef] [PubMed]
- Go, Y.Y.; Kim, S.R.; Kim, D.Y.; Chae, S.-W.; Song, J.-J. Cannabidiol enhances cytotoxicity of anti-cancer drugs in human head and neck squamous cell carcinoma. Sci. Rep. 2020, 10, 20622. [Google Scholar] [CrossRef]
- Jiang, R.; Yamaori, S.; Takeda, S.; Yamamoto, I.; Watanabe, K. Identification of cytochrome P450 enzymes responsible for metabolism of cannabidiol by human liver microsomes. Life Sci. 2011, 89, 165–170. [Google Scholar] [CrossRef]
- Lucas, C.J.; Galettis, P.; Schneider, J. The pharmacokinetics and the pharmacodynamics of cannabinoids. Br. J. Clin. Pharmacol. 2018, 84, 2477–2482. [Google Scholar] [CrossRef]
- Twelves, C.; Sabel, M.; Checketts, D.; Miller, S.; Tayo, B.; Jove, M.; Brazil, L.; Short, S.C. A phase 1b randomised, placebo-controlled trial of nabiximols cannabinoid oromucosal spray with temozolomide in patients with recurrent glioblastoma. Br. J. Cancer 2021, 124, 1379–1387. [Google Scholar] [CrossRef]
- Schloss, J.; Lacey, J.; Sinclair, J.; Steel, A.; Sughrue, M.; Sibbritt, D.; Teo, C. A Phase 2 Randomised Clinical Trial Assessing the Tolerability of Two Different Ratios of Medicinal Cannabis in Patients With High Grade Gliomas. Front. Oncol. 2021, 11, 649555. [Google Scholar] [CrossRef]
- Taylor, L.; Gidal, B.; Blakey, G.; Tayo, B.; Morrison, G. A Phase I, Randomized, Double-Blind, Placebo-Controlled, Single Ascending Dose, Multiple Dose, and Food Effect Trial of the Safety, Tolerability and Pharmacokinetics of Highly Purified Cannabidiol in Healthy Subjects. CNS Drugs 2018, 32, 1053–1067. [Google Scholar] [CrossRef]
- Dall’Stella, P.B.; Docema, M.F.L.; Maldaun, M.V.C.; Feher, O.; Lancellotti, C.L.P. Case Report: Clinical Outcome and Image Response of Two Patients With Secondary High-Grade Glioma Treated With Chemoradiation, PCV, and Cannabidiol. Front. Oncol. 2019, 8, 643. [Google Scholar] [CrossRef] [PubMed]
- Liew, K.L.; Capuano, E.; Yung, B. Lung cancer patient who had declined conventional cancer treatment: Could the self-administration of ‘CBD oil’ be contributing to the observed tumour regression? BMJ Case Rep. 2021, 14, e244195. [Google Scholar] [CrossRef] [PubMed]
- Zou, S.; Kumar, U. Cannabinoid Receptors and the Endocannabinoid System: Signaling and Function in the Central Nervous System. Int. J. Mol. Sci. 2018, 19, 833. [Google Scholar] [CrossRef] [PubMed]
- Pagano, C.; Navarra, G.; Coppola, L.; Bifulco, M.; Laezza, C. Molecular Mechanism of Cannabinoids in Cancer Progression. Int. J. Mol. Sci. 2021, 22, 3680. [Google Scholar] [CrossRef]
- Proto, M.C.; Fiore, D.; Piscopo, C.; Franceschelli, S.; Bizzarro, V.; Laezza, C.; Lauro, G.; Feoli, A.; Tosco, A.; Bifulco, G.; et al. Inhibition of Wnt/β-Catenin pathway and Histone acetyltransferase activity by Rimonabant: A therapeutic target for colon cancer. Sci. Rep. 2017, 7, 11678. [Google Scholar] [CrossRef]
- Scarlett, K.A.; White, E.S.Z.; Coke, C.J.; Carter, J.R.; Bryant, L.K.; Hinton, C.V. Agonist-induced CXCR4 and CB2 Heterodimerization Inhibits Gα13/RhoA-mediated Migration. Mol. Cancer Res. 2018, 16, 728–739. [Google Scholar] [CrossRef]
- Rajesh, M.; Mukhopadhyay, P.; Bátkai, S.; Haskó, G.; Liaudet, L.; Huffman, J.W.; Csiszar, A.; Ungvari, Z.; Mackie, K.; Chatterjee, S.; et al. CB2-receptor stimulation attenuates TNF-α-induced human endothelial cell activation, transendothelial migration of monocytes, and monocyte-endothelial adhesion. Am. J. Physiol. Circ. Physiol. 2007, 293, H2210–H2218. [Google Scholar] [CrossRef]
- Morales, P.; Reggio, P.H. An Update on Non-CB1, Non-CB2 Cannabinoid Related G-Protein-Coupled Receptors. Cannabis Cannabinoid Res. 2017, 2, 265–273. [Google Scholar] [CrossRef]
- Holland, M.L.; Allen, J.D.; Arnold, J.C. Interaction of plant cannabinoids with the multidrug transporter ABCC1 (MRP1). Eur. J. Pharmacol. 2008, 591, 128–131. [Google Scholar] [CrossRef]
- Holland, M.L.; Lau, D.T.T.; Allen, J.D.; Arnold, J.C. The multidrug transporter ABCG2 (BCRP) is inhibited by plant-derived cannabinoids. J. Cereb. Blood Flow Metab. 2007, 152, 815–824. [Google Scholar] [CrossRef]
- Liou, G.I.; Auchampach, J.A.; Hillard, C.J.; Zhu, G.; Yousufzai, B.; Mian, S.; Khan, S.; Khalifa, Y. Mediation of Cannabidiol Anti-inflammation in the Retina by Equilibrative Nucleoside Transporter and A2AAdenosine Receptor. Investig. Opthalmology Vis. Sci. 2008, 49, 5526–5531. [Google Scholar] [CrossRef] [PubMed]
- Elmes, M.W.; Kaczocha, M.; Berger, W.T.; Leung, K.; Ralph, B.P.; Wang, L.; Sweeney, J.M.; Miyauchi, J.T.; Tsirka, S.E.; Ojima, I.; et al. Fatty Acid-binding Proteins (FABPs) Are Intracellular Carriers for Δ9-Tetrahydrocannabinol (THC) and Cannabidiol (CBD). J. Biol. Chem. 2015, 290, 8711–8721. [Google Scholar] [CrossRef] [PubMed]
- Muller, C.; Morales, P.; Reggio, P.H. Cannabinoid Ligands Targeting TRP Channels. Front. Mol. Neurosci. 2019, 11, 487. [Google Scholar] [CrossRef] [PubMed]
- Bakas, T.; van Nieuwenhuijzen, P.; Devenish, S.; McGregor, I.; Arnold, J.; Chebib, M. The direct actions of cannabidiol and 2-arachidonoyl glycerol at GABA A receptors. Pharmacol. Res. 2017, 119, 358–370. [Google Scholar] [CrossRef]
- Pretzsch, C.; Freyberg, J.; Voinescu, B.; Lythgoe, D.; Horder, J.; Mendez, M.A.; Wichers, R.; Ajram, L.; Ivin, G.; Heasman, M.; et al. Effects of cannabidiol on brain excitation and inhibition systems; a randomised placebo-controlled single dose trial during magnetic resonance spectroscopy in adults with and without autism spectrum disorder. Neuropsychopharmacology 2019, 44, 1398–1405. [Google Scholar] [CrossRef]
- Uhlén, M.; Fagerberg, L.; Hallström, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, Å.; Kampf, C.; Sjöstedt, E.; Asplund, A.; et al. Proteomics. Tissue-Based Map of the Human Proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef]
- Singh, N.; Hroudová, J.; Fišar, Z. Cannabinoid-Induced Changes in the Activity of Electron Transport Chain Complexes of Brain Mitochondria. J. Mol. Neurosci. 2015, 56, 926–931. [Google Scholar] [CrossRef]
- Cosentino, M.; Legnaro, M.; Luini, A.; Ferrari, M.; Sodergren, M.; Pacchetti, B.; Marino, F. Effect of Cannabidiol on Cyclooxygenase Type 1 and 2 Expression and Function in Human Neutrophils. Cannabis Cannabinoid Res. 2022. [Google Scholar] [CrossRef]
- Massi, P.; Valenti, M.; Vaccani, A.; Gasperi, V.; Perletti, G.; Marras, E.; Fezza, F.; Maccarrone, M.; Parolaro, D. 5-Lipoxygenase and anandamide hydrolase (FAAH) mediate the antitumor activity of cannabidiol, a non-psychoactive cannabinoid. J. Neurochem. 2008, 104, 1091–1100. [Google Scholar] [CrossRef]
- Bih, C.I.; Chen, T.; Nunn, A.V.W.; Bazelot, M.; Dallas, M.; Whalley, B.J. Molecular Targets of Cannabidiol in Neurological Disorders. Neurotherapeutics 2015, 12, 699–730. [Google Scholar] [CrossRef]
- Ramer, R.; Heinemann, K.; Merkord, J.; Rohde, H.; Salamon, A.; Linnebacher, M.; Hinz, B. COX-2 and PPAR-γ Confer Cannabidiol-Induced Apoptosis of Human Lung Cancer Cells. Mol. Cancer Ther. 2013, 12, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Jastrząb, A.; Gęgotek, A.; Skrzydlewska, E. Cannabidiol Regulates the Expression of Keratinocyte Proteins Involved in the Inflammation Process through Transcriptional Regulation. Cells 2019, 8, 827. [Google Scholar] [CrossRef] [PubMed]
- Khaksar, S.; Bigdeli, M.R. Intra-cerebral cannabidiol infusion-induced neuroprotection is partly associated with the TNF-α/TNFR1/NF-κB pathway in transient focal cerebral ischaemia. Brain Inj. 2017, 31, 1932–1943. [Google Scholar] [CrossRef]
- Ivanov, V.N.; Grabham, P.W.; Wu, C.-C.; Hei, T.K. Inhibition of autophagic flux differently modulates cannabidiol-induced death in 2D and 3D glioblastoma cell cultures. Sci. Rep. 2020, 10, 2687. [Google Scholar] [CrossRef] [PubMed]
- Juknat, A.; Pietr, M.; Kozela, E.; Rimmerman, N.; Levy, R.; Gao, F.; Coppola, G.; Geschwind, D.; Vogel, Z. Microarray and Pathway Analysis Reveal Distinct Mechanisms Underlying Cannabinoid-Mediated Modulation of LPS-Induced Activation of BV-2 Microglial Cells. PLoS ONE 2013, 8, e61462. [Google Scholar] [CrossRef]
- Singer, E.; Judkins, J.; Salomonis, N.; Matlaf, L.; Soteropoulos, P.; McAllister, S.; Soroceanu, L. Reactive oxygen species-mediated therapeutic response and resistance in glioblastoma. Cell Death Dis. 2015, 6, e1601. [Google Scholar] [CrossRef]
- Soroceanu, L.; Singer, E.; Dighe, P.; Sidorov, M.; Limbad, C.; Rodriquez-Brotons, A.; Rix, P.; Woo, R.W.L.; Dickinson, L.; Desprez, P.-Y.; et al. Cannabidiol inhibits RAD51 and sensitizes glioblastoma to temozolomide in multiple orthotopic tumor models. Neuro-Oncol. Adv. 2022, 4, vdac019. [Google Scholar] [CrossRef]
- De la Harpe, A.; Beukes, N.; Frost, C.L. CBD activation of TRPV1 induces oxidative signaling and subsequent ER stress in breast cancer cell lines. Biotechnol. Appl. Biochem. 2021, 69, 420–430. [Google Scholar] [CrossRef]
- Jeong, S.; Yun, H.K.; Jeong, Y.A.; Jo, M.J.; Kang, S.H.; Kim, J.L.; Kim, D.Y.; Park, S.H.; Kim, B.R.; Na, Y.J.; et al. Cannabidiol-induced apoptosis is mediated by activation of Noxa in human colorectal cancer cells. Cancer Lett. 2019, 447, 12–23. [Google Scholar] [CrossRef]
- McKallip, R.J.; Jia, W.; Schlomer, J.; Warren, J.W.; Nagarkatti, P.S.; Nagarkatti, M. Cannabidiol-Induced Apoptosis in Human Leukemia Cells: A Novel Role of Cannabidiol in the Regulation of p22phox and Nox4 Expression. Mol. Pharmacol. 2006, 70, 897–908. [Google Scholar] [CrossRef]
- Guard, S.E.; Chapnick, D.A.; Poss, Z.C.; Ebmeier, C.C.; Jacobsen, J.; Nemkov, T.; Ball, K.A.; Webb, K.J.; Simpson, H.L.; Coleman, S.; et al. Multi-Omic Analysis Reveals Disruption of Cholesterol Homeostasis by Cannabidiol in Human Cell Lines. Mol. Cell. Proteom. 2022, 21, 100262. [Google Scholar] [CrossRef] [PubMed]
- Howell, M.C.; Green, R.; Khalil, R.; Foran, E.; Quarni, W.; Nair, R.; Stevens, S.; Grinchuk, A.; Hanna, A.; Mohapatra, S.; et al. Lung cancer cells survive epidermal growth factor receptor tyrosine kinase inhibitor exposure through upregulation of cholesterol synthesis. FASEB BioAdv. 2019, 2, 90–105. [Google Scholar] [CrossRef] [PubMed]
- Shangguan, F.; Zhou, H.; Ma, N.; Wu, S.; Huang, H.; Jin, G.; Wu, S.; Hong, W.; Zhuang, W.; Xia, H.; et al. A Novel Mechanism of Cannabidiol in Suppressing Hepatocellular Carcinoma by Inducing GSDME Dependent Pyroptosis. Front. Cell Dev. Biol. 2021, 9, 697832. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Xu, T.; Wang, Y.; Zhou, Y.; Yu, D.; Wang, Z.; He, L.; Chen, Z.; Zhang, Y.; Davidson, D.; et al. Cannabidiol inhibits human glioma by induction of lethal mitophagy through activating TRPV4. Autophagy 2021, 17, 3592–3606. [Google Scholar] [CrossRef]
- Desprez, P.-Y.; Murase, R.; Limbad, C.; Woo, R.W.; Adrados, I.; Weitenthaler, K.; Soroceanu, L.; Salomonis, N.; McAllister, S.D. Cannabidiol Treatment Results in a Common Gene Expression Response Across Aggressive Cancer Cells from Various Origins. Cannabis Cannabinoid Res. 2021, 6, 148–155. [Google Scholar] [CrossRef]
- Buchtova, T.; Skrott, Z.; Chroma, K.; Rehulka, J.; Dzubak, P.; Hajduch, M.; Lukac, D.; Arampatzis, S.; Bartek, J.; Mistrik, M. Cannabidiol-induced activation of the metallothionein pathway impedes anticancer effects of disulfiram and its metabolite CuET. Mol. Oncol. 2021, 16, 1541–1554. [Google Scholar] [CrossRef]
- Dobovišek, L.; Novak, M.; Krstanović, F.; Borštnar, S.; Turnšek, T.L.; Debeljak, N. Effect of combining CBD with standard breast cancer therapeutics. Adv. Cancer Biol. Metastasis 2022, 4, 100038. [Google Scholar] [CrossRef]
- Elbaz, M.; Nasser, M.W.; Ravi, J.; Wani, N.A.; Ahirwar, D.K.; Zhao, H.; Oghumu, S.; Satoskar, A.R.; Shilo, K.; Carson, W.E.; et al. Modulation of the tumor microenvironment and inhibition of EGF/EGFR pathway: Novel anti-tumor mechanisms of Cannabidiol in breast cancer. Mol. Oncol. 2015, 9, 906–919. [Google Scholar] [CrossRef]
- Honarmand, M.; Namazi, F.; Mohammadi, A.; Nazifi, S. Can cannabidiol inhibit angiogenesis in colon cancer? Comp. Clin. Pathol. 2019, 28, 165–172. [Google Scholar] [CrossRef]
- Ramer, R.; Bublitz, K.; Freimuth, N.; Merkord, J.; Rohde, H.; Haustein, M.; Borchert, P.; Schmuhl, E.; Linnebacher, M.; Hinz, B. Cannabidiol inhibits lung cancer cell invasion and metastasis via intercellular adhesion molecule-1. FASEB J. 2011, 26, 1535–1548. [Google Scholar] [CrossRef]
- Jeong, S.; Jo, M.J.; Yun, H.K.; Kim, D.Y.; Kim, B.R.; Kim, J.L.; Park, S.H.; Na, Y.J.; Jeong, Y.A.; Kim, B.G.; et al. Cannabidiol promotes apoptosis via regulation of XIAP/Smac in gastric cancer. Cell Death Dis. 2019, 10, 846. [Google Scholar] [CrossRef] [PubMed]
- Lukhele, S.T.; Motadi, L.R. Cannabidiol rather than Cannabis sativa extracts inhibit cell growth and induce apoptosis in cervical cancer cells. BMC Complement. Altern. Med. 2016, 16, 335. [Google Scholar] [CrossRef]
- Yang, Y.; Huynh, N.; Dumesny, C.; Wang, K.; He, H.; Nikfarjam, M. Cannabinoids Inhibited Pancreatic Cancer via P-21 Activated Kinase 1 Mediated Pathway. Int. J. Mol. Sci. 2020, 21, 8035. [Google Scholar] [CrossRef] [PubMed]
- Alharris, E.; Singh, N.P.; Nagarkatti, P.S.; Nagarkatti, M. Role of miRNA in the regulation of cannabidiol-mediated apoptosis in neuroblastoma cells. Oncotarget 2019, 10, 45–59. [Google Scholar] [CrossRef] [PubMed]
- Marzęda, P.; Wróblewska-Łuczka, P.; Drozd, M.; Florek-Łuszczki, M.; Załuska-Ogryzek, K.; Łuszczki, J.J. Cannabidiol Interacts Antagonistically with Cisplatin and Additively with Mitoxantrone in Various Melanoma Cell Lines—An Isobolographic Analysis. Int. J. Mol. Sci. 2022, 23, 6752. [Google Scholar] [CrossRef]
- Morelli, M.B.; Offidani, M.; Alesiani, F.; Discepoli, G.; Liberati, S.; Olivieri, A.; Santoni, M.; Santoni, G.; Leoni, P.; Nabissi, M. The effects of cannabidiol and its synergism with bortezomib in multiple myeloma cell lines. A role for transient receptor potential vanilloid type-2. Int. J. Cancer 2013, 134, 2534–2546. [Google Scholar] [CrossRef]
- Ferro, R.; Adamska, A.; Lattanzio, R.; Mavrommati, I.; Edling, C.E.; Arifin, S.A.; Fyffe, C.A.; Sala, G.; Sacchetto, L.; Chiorino, G.; et al. GPR55 signalling promotes proliferation of pancreatic cancer cells and tumour growth in mice, and its inhibition increases effects of gemcitabine. Oncogene 2018, 37, 6368–6382. [Google Scholar] [CrossRef]
- Feng, P.; Zhu, L.; Jie, J.; Yang, P.; Sheng, N.; Chen, X.; Chen, X. Cannabidiol inhibits invasion and metastasis in colorectal cancer cells by reversing epithelial-mesenchymal transition through the Wnt/beta-catenin signaling pathway. J. Cancer Res. Clin. Oncol. 2022. [Google Scholar] [CrossRef]
- Ivanov, V.N.; Wu, J.; Wang, T.J.C.; Hei, T.K. Inhibition of ATM kinase upregulates levels of cell death induced by cannabidiol and γ-irradiation in human glioblastoma cells. Oncotarget 2019, 10, 825–846. [Google Scholar] [CrossRef]
- Nguyen, L.C.; Yang, D.; Nicolaescu, V.; Best, T.J.; Gula, H.; Saxena, D.; Gabbard, J.D.; Chen, S.-N.; Ohtsuki, T.; Friesen, J.B.; et al. Cannabidiol inhibits SARS-CoV-2 replication through induction of the host ER stress and innate immune responses. Sci. Adv. 2022, 8, eabi6110. [Google Scholar] [CrossRef]
- Preet, A.; Ganju, R.K.; Groopman, J.E. Δ9-Tetrahydrocannabinol inhibits epithelial growth factor-induced lung cancer cell migration in vitro as well as its growth and metastasis in vivo. Oncogene 2008, 27, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Anis, O.; Vinayaka, A.; Shalev, N.; Namdar, D.; Nadarajan, S.; Anil, S.; Cohen, O.; Belausov, E.; Ramon, J.; Gati, E.M.; et al. Cannabis-Derived Compounds Cannabichromene and Δ9-Tetrahydrocannabinol Interact and Exhibit Cytotoxic Activity against Urothelial Cell Carcinoma Correlated with Inhibition of Cell Migration and Cytoskeleton Organization. Molecules 2021, 26, 465. [Google Scholar] [CrossRef]
- Caffarel, M.M.; Moreno-Bueno, G.; Cerutti, C.; Palacios, J.; Guzman, M.; Mechta-Grigoriou, F.; Sanchez, C. JunD is involved in the antiproliferative effect of Δ9-tetrahydrocannabinol on human breast cancer cells. Oncogene 2008, 27, 5033–5044. [Google Scholar] [CrossRef] [PubMed]
- Milián, L.; Monleón-Guinot, I.; Sancho-Tello, M.; Galbis, J.M.; Cremades, A.; Almenar-Ordaz, M.; Peñaroja-Martinez, J.; Farras, R.; de Llano, J.J.M.; Carda, C.; et al. In Vitro Effect of Δ9-Tetrahydrocannabinol and Cannabidiol on Cancer-Associated Fibroblasts Isolated from Lung Cancer. Int. J. Mol. Sci. 2022, 23, 6766. [Google Scholar] [CrossRef] [PubMed]
- McKallip, R.J.; Nagarkatti, M.; Nagarkatti, P.S. Δ-9-Tetrahydrocannabinol Enhances Breast Cancer Growth and Metastasis by Suppression of the Antitumor Immune Response. J. Immunol. 2005, 174, 3281–3289. [Google Scholar] [CrossRef]
- Luo, C.-K.; Chou, P.-H.; Ng, S.-K.; Lin, W.-Y.; Wei, T.-T. Cannabinoids orchestrate cross-talk between cancer cells and endothelial cells in colorectal cancer. Cancer Gene Ther. 2021, 29, 597–611. [Google Scholar] [CrossRef]
- Miller, H.; Bonawitz, S.C.; Ostrovsky, O. The effects of delta-9-tetrahydrocannabinol (THC) on inflammation: A review. Cell. Immunol. 2020, 352, 104111. [Google Scholar] [CrossRef]
- Portenoy, R.K.; Ganae-Motan, E.D.; Allende, S.; Yanagihara, R.; Shaiova, L.; Weinstein, S.; McQuade, R.; Wright, S.; Fallon, M.T. Nabiximols for Opioid-Treated Cancer Patients With Poorly-Controlled Chronic Pain: A Randomized, Placebo-Controlled, Graded-Dose Trial. J. Pain 2012, 13, 438–449. [Google Scholar] [CrossRef]
- Johnson, J.R.; Lossignol, D.; Burnell-Nugent, M.; Fallon, M.T. An Open-Label Extension Study to Investigate the Long-Term Safety and Tolerability of THC/CBD Oromucosal Spray and Oromucosal THC Spray in Patients with Terminal Cancer-Related Pain Refractory to Strong Opioid Analgesics. J. Pain Symptom Manag. 2012, 46, 207–218. [Google Scholar] [CrossRef]
- Pharmaceuticals, J. Long Term Safety of Sativex Oromucosal Spray (Sativex®; Nabiximols) as Adjunctive Therapy in Patients with Uncontrolled Persistent Chronic Cancer Related Pain NCT01337089; National Library of Medicine: Bethesda, MD, USA, 2018. [Google Scholar]
- Fallon, M.T.; Lux, E.A.; McQuade, R.; Rossetti, S.; Sanchez, R.; Sun, W.; Wright, S.; Lichtman, A.H.; Kornyeyeva, E. Sativex oromucosal spray as adjunctive therapy in advanced cancer patients with chronic pain unalleviated by optimized opioid therapy: Two double-blind, randomized, placebo-controlled phase 3 studies. Br. J. Pain 2017, 11, 119–133. [Google Scholar] [CrossRef]
- Lichtman, A.H.; Lux, E.A.; McQuade, R.; Rossetti, S.; Sanchez, R.; Sun, W.; Wright, S.; Kornyeyeva, E.; Fallon, M.T. Results of a Double-Blind, Randomized, Placebo-Controlled Study of Nabiximols Oromucosal Spray as an Adjunctive Therapy in Advanced Cancer Patients with Chronic Uncontrolled Pain. J. Pain Symptom Manag. 2018, 55, 179–188.e1. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.R.; Burnell-Nugent, M.; Lossignol, D.; Ganae-Motan, E.D.; Potts, R.; Fallon, M.T. Multicenter, Double-Blind, Randomized, Placebo-Controlled, Parallel-Group Study of the Efficacy, Safety, and Tolerability of THC:CBD Extract and THC Extract in Patients with Intractable Cancer-Related Pain. J. Pain Symptom Manag. 2010, 39, 167–179. [Google Scholar] [CrossRef]
- Melén, C.M.; Merrien, M.; Wasik, A.M.; Panagiotidis, G.; Beck, O.; Sonnevi, K.; Junlén, H.-R.; Christensson, B.; Sander, B.; Wahlin, B.E. Clinical effects of a single dose of cannabinoids to patients with chronic lymphocytic leukemia. Leuk. Lymphoma 2022, 63, 1387–1397. [Google Scholar] [CrossRef] [PubMed]
- Tayo, B.; Taylor, L.; Sahebkar, F.; Morrison, G. A Phase I, Open-Label, Parallel-Group, Single-Dose Trial of the Pharmacokinetics, Safety, and Tolerability of Cannabidiol in Subjects with Mild to Severe Renal Impairment. Clin. Pharmacokinet. 2019, 59, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Millar, S.A.; Stone, N.L.; Yates, A.S.; O’Sullivan, S.E. A Systematic Review on the Pharmacokinetics of Cannabidiol in Humans. Front. Pharmacol. 2018, 9, 1365. [Google Scholar] [CrossRef] [PubMed]
- Hagmann, C.; Cramer, A.; Kestenbaum, A.; Durazo, C.; Downey, A.; Russell, M.; Geluz, J.; Ma, J.D.; Roeland, E.J. Evidence-based Palliative Care Approaches to Non-pain Physical Symptom Management in Cancer Patients. Semin. Oncol. Nurs. 2018, 34, 227–240. [Google Scholar] [CrossRef]
- Sleight, A.G.; Gerber, L.H.; Marshall, T.F.; Livinski, A.; Alfano, C.M.; Harrington, S.; Flores, A.M.; Virani, A.; Hu, X.; Mitchell, S.A.; et al. Systematic Review of Functional Outcomes in Cancer Rehabilitation. Arch. Phys. Med. Rehabil. 2022, 103, 1807–1826. [Google Scholar] [CrossRef]
- Corli, O.; Roberto, A.; Corsi, N.; Galli, F.; Pizzuto, M. Opioid switching and variability in response in pain cancer patients. Support. Care Cancer 2018, 27, 2321–2327. [Google Scholar] [CrossRef]
- Vitzthum, L.K.; Riviere, P.; Sheridan, P.; Nalawade, V.; Deka, R.; Furnish, T.; Mell, L.K.; Rose, B.; Wallace, M.; Murphy, J.D. Predicting Persistent Opioid Use, Abuse, and Toxicity Among Cancer Survivors. JNCI J. Natl. Cancer Inst. 2019, 112, 720–727. [Google Scholar] [CrossRef]
- Bhamra, S.K.; Desai, A.; Imani-Berendjestanki, P.; Horgan, M. The emerging role of cannabidiol (CBD) products; a survey exploring the public’s use and perceptions of CBD. Phytother. Res. 2021, 35, 5734–5740. [Google Scholar] [CrossRef]
- Wheeler, M.; Merten, J.W.; Gordon, B.T.; Hamadi, H. CBD (Cannabidiol) Product Attitudes, Knowledge, and Use among Young Adults. Subst. Use Misuse 2020, 55, 1138–1145. [Google Scholar] [CrossRef] [PubMed]
- U.S. National Library of Medicine. ClinicalTrials.gov [Internet]. Available online: https://clinicaltrials.gov/ (accessed on 31 August 2022).
- Thomas, P.A.; Carter, G.T.; Bombardier, C.H. A scoping review on the effect of cannabis on pain intensity in people with spinal cord injury. J. Spinal Cord Med. 2022, 45, 656–667. [Google Scholar] [CrossRef] [PubMed]
- Capano, A.; Weaver, R.; Burkman, E. Evaluation of the effects of CBD hemp extract on opioid use and quality of life indicators in chronic pain patients: A prospective cohort study. Postgrad. Med. 2019, 132, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Arout, C.A.; Haney, M.; Herrmann, E.S.; Bedi, G.; Cooper, Z.D. A placebo-controlled investigation of the analgesic effects, abuse liability, safety and tolerability of a range of oral cannabidiol doses in healthy humans. Br. J. Clin. Pharmacol. 2021, 88, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Vela, J.; Dreyer, L.; Petersen, K.K.; Arendt-Nielsen, L.; Duch, K.S.; Kristensen, S. Cannabidiol treatment in hand osteoarthritis and psoriatic arthritis: A randomized, double-blind, placebo-controlled trial. Pain 2021, 163, 1206–1214. [Google Scholar] [CrossRef]
- Leehey, M.A.; Liu, Y.; Hart, F.; Epstein, C.; Cook, M.; Sillau, S.; Klawitter, J.; Newman, H.; Sempio, C.; Forman, L.; et al. Safety and Tolerability of Cannabidiol in Parkinson Disease: An Open Label, Dose-Escalation Study. Cannabis Cannabinoid Res. 2020, 5, 326–336. [Google Scholar] [CrossRef]
- Bonn-Miller, M.O.; Sisley, S.; Riggs, P.; Yazar-Klosinski, B.; Wang, J.B.; Loflin, M.J.E.; Shechet, B.; Hennigan, C.; Matthews, R.; Emerson, A.; et al. The short-term impact of 3 smoked cannabis preparations versus placebo on PTSD symptoms: A randomized cross-over clinical trial. PLoS ONE 2021, 16, e0246990. [Google Scholar] [CrossRef]
- Wade, D.T.; Makela, P.; Robson, P.; House, H.; Bateman, C. Do cannabis-based medicinal extracts have general or specific effects on symptoms in multiple sclerosis? A double-blind, randomized, placebo-controlled study on 160 patients. Mult. Scler. J. 2004, 10, 434–441. [Google Scholar] [CrossRef]
- Bickel, E.A.; Schellekens, M.P.J.; Smink, J.G.; Mul, V.E.M.; Ranchor, A.V.; Fleer, J.; Schroevers, M.J. Looking at individual symptoms: The dynamic network structure of depressive symptoms in cancer survivors and their preferences for psychological care. J. Cancer Surviv. 2022, 1–10. [Google Scholar] [CrossRef]
- Linden, W.; Vodermaier, A.; MacKenzie, R.; Greig, D. Anxiety and depression after cancer diagnosis: Prevalence rates by cancer type, gender, and age. J. Affect. Disord. 2012, 141, 343–351. [Google Scholar] [CrossRef]
- De Faria, S.M.; Fabrício, D.D.M.; Tumas, V.; Castro, P.C.; Ponti, M.; Hallak, J.E.; Zuardi, A.W.; Crippa, J.A.S.; Chagas, M.H.N. Effects of acute cannabidiol administration on anxiety and tremors induced by a Simulated Public Speaking Test in patients with Parkinson’s disease. J. Psychopharmacol. 2020, 34, 189–196. [Google Scholar] [CrossRef]
- Berger, M.; Li, E.; Rice, S.; Davey, C.G.; Ratheesh, A.; Adams, S.; Jackson, H.; Hetrick, S.; Parker, A.; Spelman, T.; et al. Cannabidiol for Treatment-Resistant Anxiety Disorders in Young People. J. Clin. Psychiatry 2022, 83, 42111. [Google Scholar] [CrossRef] [PubMed]
- Heider, C.G.; Itenberg, S.A.; Rao, J.; Ma, H.; Wu, X. Mechanisms of Cannabidiol (CBD) in Cancer Treatment: A Review. Biology 2022, 11, 817. [Google Scholar] [CrossRef] [PubMed]
- Valenti, C.; Billi, M.; Pancrazi, G.L.; Calabria, E.; Armogida, N.G.; Tortora, G.; Pagano, S.; Barnaba, P.; Marinucci, L. Biological effects of cannabidiol on human cancer cells: Systematic review of the literature. Pharmacol. Res. 2022, 181. [Google Scholar] [CrossRef] [PubMed]
- Gorzo, A.; Havași, A.; Spînu, Ș.; Oprea, A.; Burz, C.; Sur, D. Practical Considerations for the Use of Cannabis in Cancer Pain Management—What a Medical Oncologist Should Know. J. Clin. Med. 2022, 11, 5036. [Google Scholar] [CrossRef] [PubMed]
- Vinette, B.; Côté, J.; El-Akhras, A.; Mrad, H.; Chicoine, G.; Bilodeau, K. Routes of administration, reasons for use, and approved indications of medical cannabis in oncology: A scoping review. BMC Cancer 2022, 22, 319. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Green, R.; Khalil, R.; Mohapatra, S.S.; Mohapatra, S. Role of Cannabidiol for Improvement of the Quality of Life in Cancer Patients: Potential and Challenges. Int. J. Mol. Sci. 2022, 23, 12956. https://doi.org/10.3390/ijms232112956
Green R, Khalil R, Mohapatra SS, Mohapatra S. Role of Cannabidiol for Improvement of the Quality of Life in Cancer Patients: Potential and Challenges. International Journal of Molecular Sciences. 2022; 23(21):12956. https://doi.org/10.3390/ijms232112956
Chicago/Turabian StyleGreen, Ryan, Roukiah Khalil, Shyam S. Mohapatra, and Subhra Mohapatra. 2022. "Role of Cannabidiol for Improvement of the Quality of Life in Cancer Patients: Potential and Challenges" International Journal of Molecular Sciences 23, no. 21: 12956. https://doi.org/10.3390/ijms232112956
APA StyleGreen, R., Khalil, R., Mohapatra, S. S., & Mohapatra, S. (2022). Role of Cannabidiol for Improvement of the Quality of Life in Cancer Patients: Potential and Challenges. International Journal of Molecular Sciences, 23(21), 12956. https://doi.org/10.3390/ijms232112956









