ENO1 Binds to ApoC3 and Impairs the Proliferation of T Cells via IL-8/STAT3 Pathway in OSCC
Abstract
1. Introduction
2. Results
2.1. ENO1 Was Overexpressed in Human OSCC Tissues
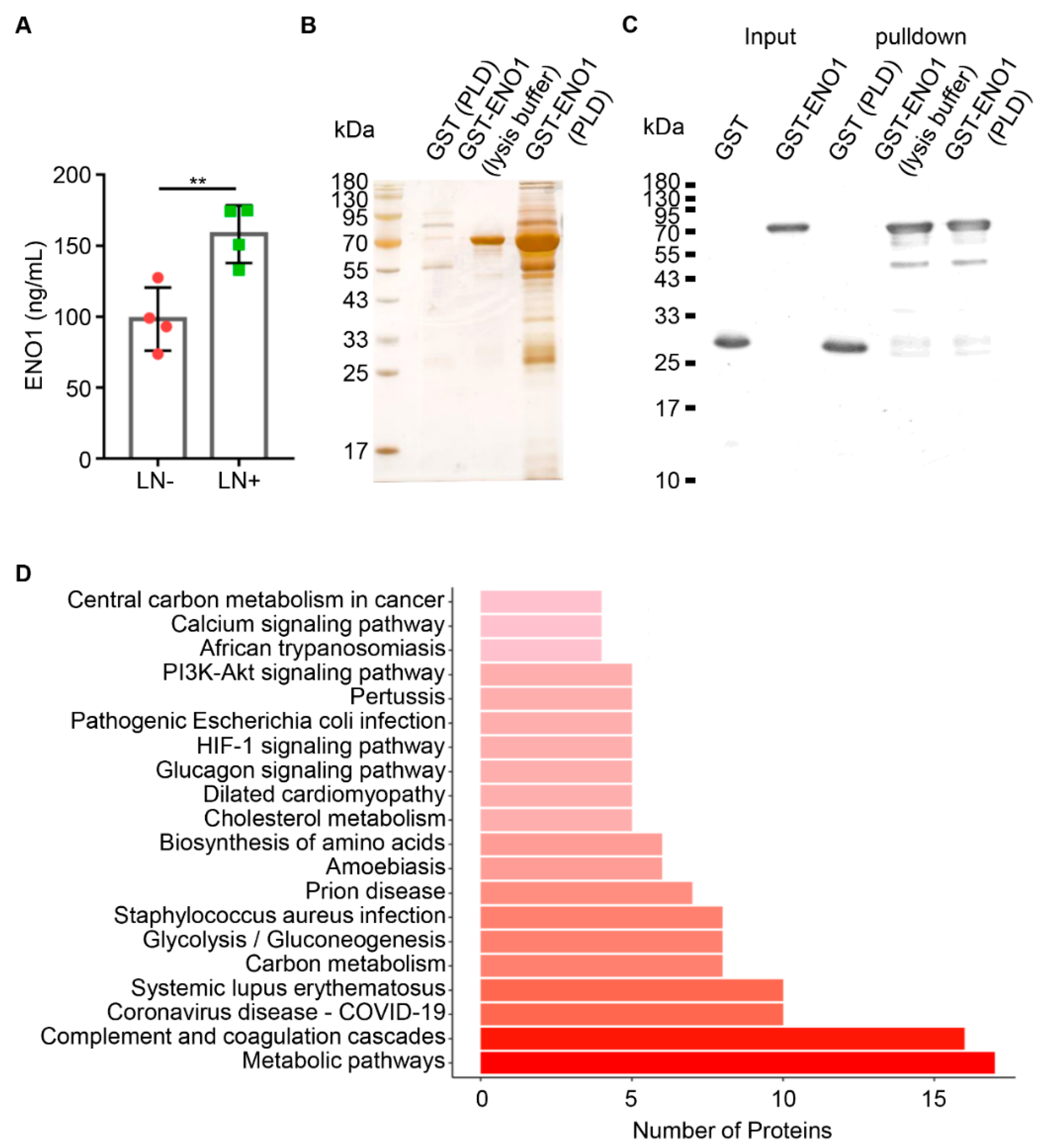
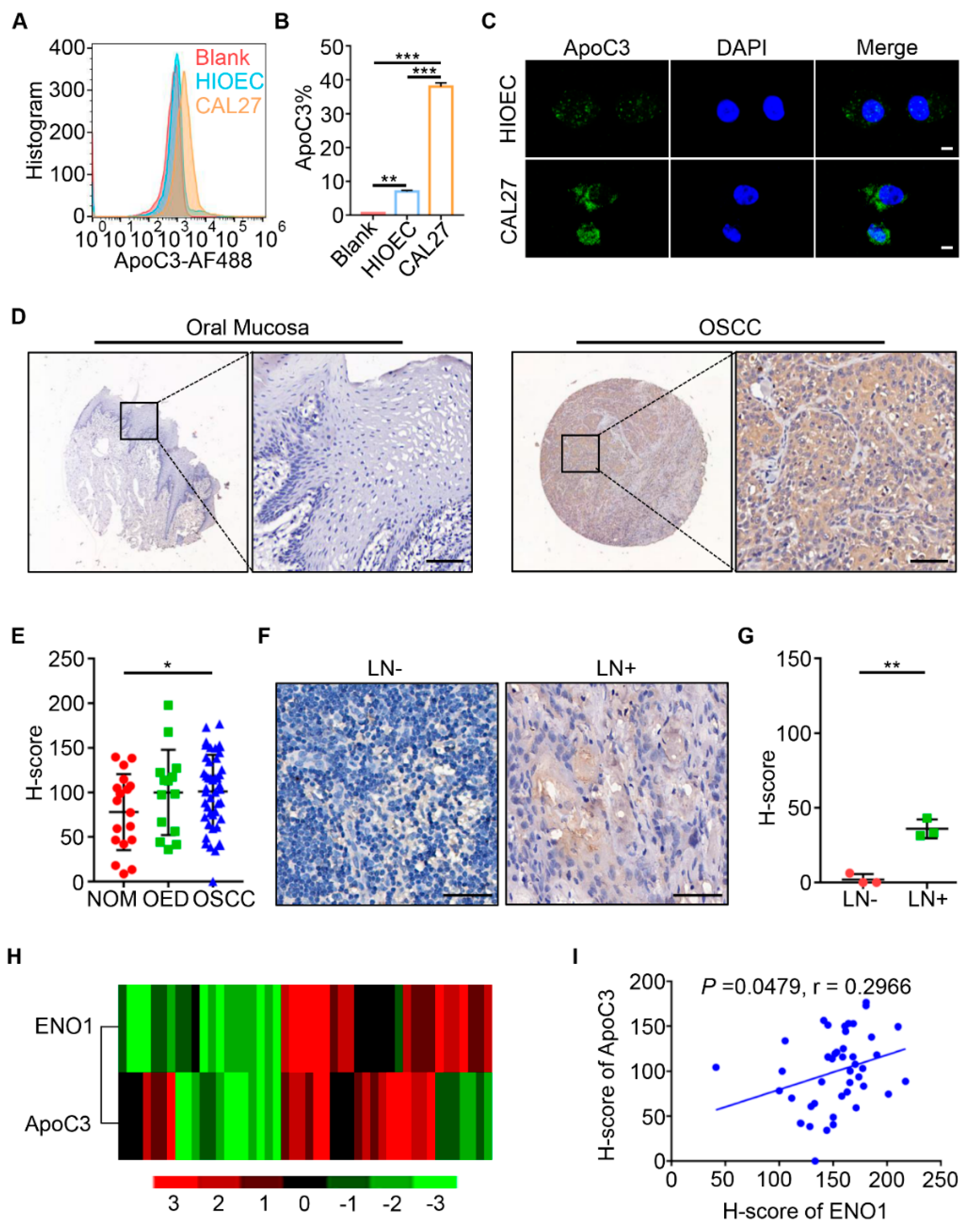
2.2. Clinicopathological Correlation of ENO1 Expression Level in OSCC
2.3. ENO1 Directly Interacted with ApoC3 in OSCC
2.4. ApoC3 was Overexpressed in Human OSCC
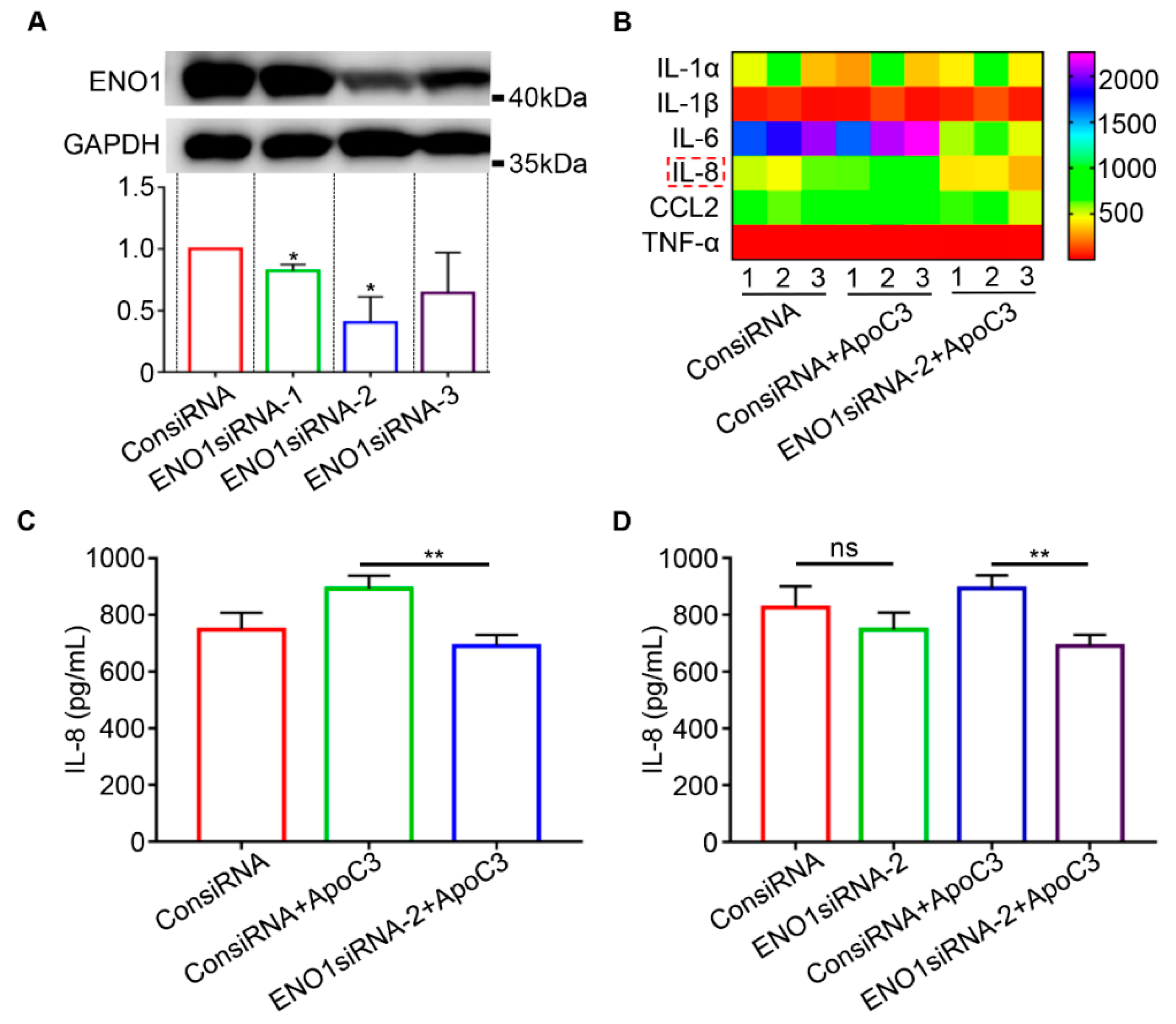
2.5. ENO1 Binding to ApoC3 Mediated the Production of Proinflammatory Cytokines
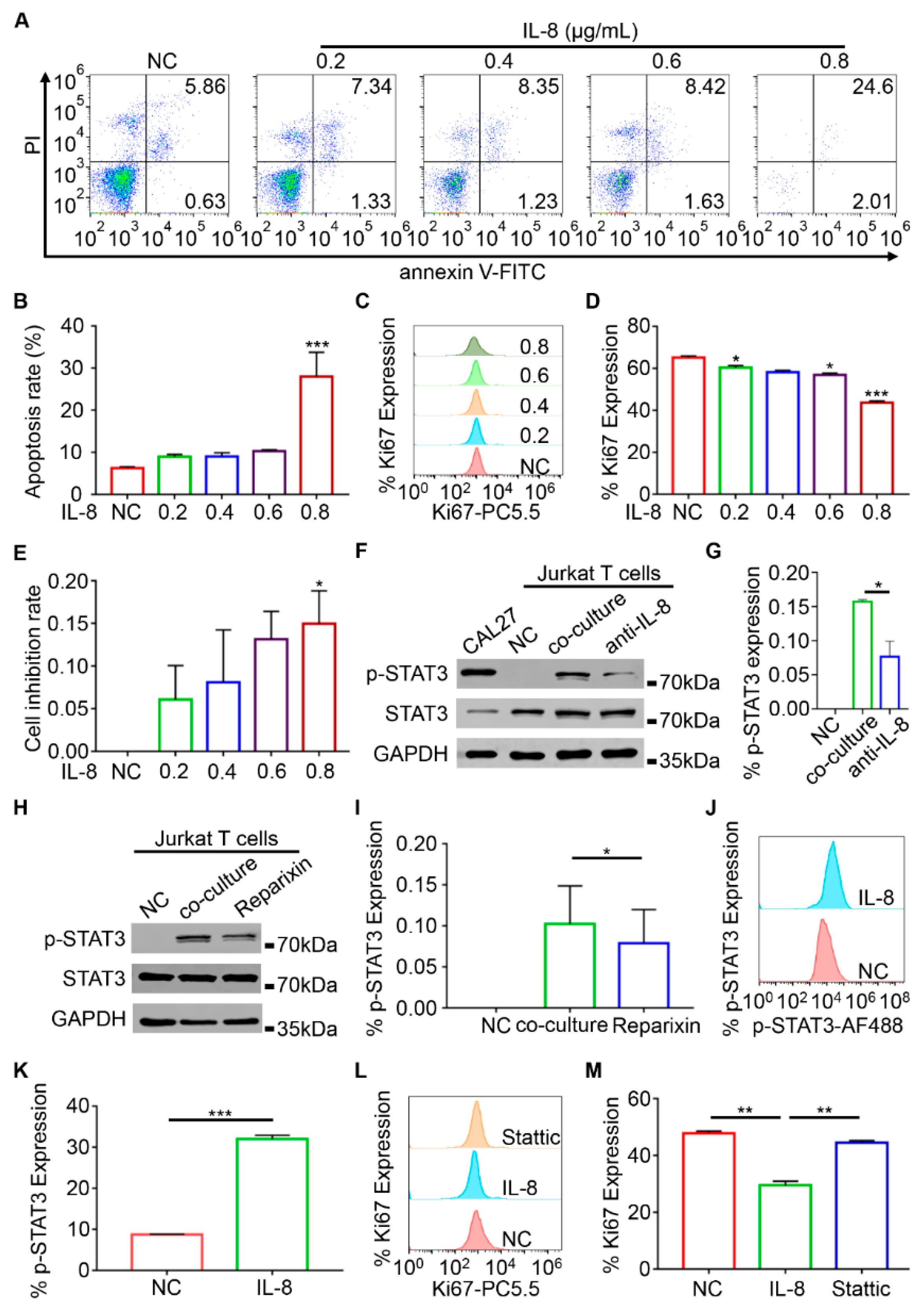
2.6. IL-8 Impaired the Proliferation of T Cells through the STAT3 Signaling Pathway
3. Discussion
4. Materials and Methods
4.1. Patient and Sample Collection
4.2. Human OSCC Tissue Microarrays
4.3. Immunohistochemical (IHC) Staining
4.4. Human Lymphatic Drainage
4.5. GST Pulldown Assay
4.6. Mass Spectrometry
4.7. KEGG Pathway Enrichment and PPI Network Analysis
4.8. Cell Culture
4.9. Immunofluorescence
4.10. Confocal Microscopy
4.11. Flow Cytometry
4.12. Co-Culture of Jurkat T and CAL27 Cells
4.13. SiRNA Knockdown of ENO1
4.14. Western Blot Analysis
4.15. Inflammatory Cytokine Assay
4.16. Enzyme-Linked Immunosorbent Assay (ELISA)
4.17. Cell Cytotoxicity Assay
4.18. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Givony, S. Oral squamous cell carcinoma (OSCC) an overview. Med. Moksl. 2020, 8, 67–74. [Google Scholar]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Cives, M.; Mannavola, F.; Lospalluti, L.; Sergi, M.C.; Cazzato, G.; Filoni, E.; Cavallo, F.; Giudice, G.; Stucci, L.S.; Porta, C. Non-melanoma skin cancers: Biological and clinical features. Int. J. Mol. Sci. 2020, 21, 5394. [Google Scholar] [CrossRef]
- Chamoli, A.; Gosavi, A.S.; Shirwadkar, U.P.; Wangdale, K.V.; Behera, S.K.; Kurrey, N.K.; Kalia, K.; Mandoli, A. Overview of oral cavity squamous cell carcinoma: Risk factors, mechanisms, and diagnostics. Oral Oncol. 2021, 121, 105451. [Google Scholar] [CrossRef] [PubMed]
- Halim, A.S.; Ramasenderan, N. High-risk cutaneous squamous cell carcinoma (CSCC): Challenges and emerging therapies. Asian J. Surg. 2022, in press. [Google Scholar]
- Wang, W.; Adeoye, J.; Thomson, P.; Choi, S.W. Multiple tumour recurrence in oral, head and neck cancer: Characterising the patient journey. J. Oral Pathol. Med. 2021, 50, 979–984. [Google Scholar] [CrossRef] [PubMed]
- Leemans, C.R.; Braakhuis, B.J.; Brakenhoff, R.H. The molecular biology of head and neck cancer. Nat. Rev. Cancer. 2011, 11, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Kim, J.-W.; Paeng, J.-Y. Clinical analysis of neck node metastasis in oral cavity cancer. J. Korean Assoc. Oral Maxillofac. Surg. 2018, 44, 282–288. [Google Scholar] [CrossRef]
- Reticker-Flynn, N.E.; Zhang, W.; Belk, J.A.; Basto, P.A.; Escalante, N.K.; Pilarowski, G.O.; Bejnood, A.; Martins, M.M.; Kenkel, J.A.; Linde, I.L. Lymph node colonization induces tumor-immune tolerance to promote distant metastasis. Cell 2022, 185, 1924–1942.e23. [Google Scholar] [CrossRef]
- Abdul-Aziz, M.A.; Amin, A.K.; El-Rouby, D.H.; Shaker, O.G. Lymphangiogenesis in oral squamous cell carcinoma: Correlation with VEGF-C expression and lymph node metastasis. Int. J. Dent. 2017, 2017, 7285656. [Google Scholar] [CrossRef]
- Włodzimierz, R.; Rutkowski, P.; Nowecki, Z.I.; Kulik, J.; Nasierowska-Guttmejer, A.; Siedlecki, J.A. Detection of melanoma cells in the lymphatic drainage after lymph node dissection in melanoma patients by using two-marker reverse transcriptase-polymerase chain reaction assay. Ann. Surg. Oncol. 2004, 11, 988–997. [Google Scholar] [CrossRef]
- Rutkowski, P.; Nowecki, Z.I.; Kulik, J.; Ruka, W.; Siedlecki, J.A. Molecular staging by multimarker reverse transcriptase-polymerase chain reaction assay of lymphatic drainage and blood from melanoma patients after lymph node dissection. Melanoma Res. 2008, 18, 246–252. [Google Scholar] [CrossRef] [PubMed]
- García-Silva, S.; Benito-Martín, A.; Sánchez-Redondo, S.; Hernández-Barranco, A.; Ximénez-Embún, P.; Nogués, L.; Mazariegos, M.S.; Brinkmann, K.; Amor López, A.; Meyer, L. Use of extracellular vesicles from lymphatic drainage as surrogate markers of melanoma progression and BRAF V600E mutation. J. Exp. Med. 2019, 216, 1061–1070. [Google Scholar] [CrossRef] [PubMed]
- Capello, M.; Ferri-Borgogno, S.; Cappello, P.; Novelli, F. α-enolase: A promising therapeutic and diagnostic tumor target. FEBS J. 2011, 278, 1064–1074. [Google Scholar] [CrossRef] [PubMed]
- Principe, M.; Ceruti, P.; Shih, N.-Y.; Chattaragada, M.S.; Rolla, S.; Conti, L.; Bestagno, M.; Zentilin, L.; Yang, S.-H.; Migliorini, P. Targeting of surface alpha-enolase inhibits the invasiveness of pancreatic cancer cells. Oncotarget 2015, 6, 11098. [Google Scholar] [CrossRef] [PubMed]
- Capello, M.; Ferri-Borgogno, S.; Riganti, C.; Chattaragada, M.S.; Principe, M.; Roux, C.; Zhou, W.; Petricoin, E.F.; Cappello, P.; Novelli, F. Targeting the Warburg effect in cancer cells through ENO1 knockdown rescues oxidative phosphorylation and induces growth arrest. Oncotarget 2016, 7, 5598. [Google Scholar] [CrossRef]
- Fu, Q.-F.; Liu, Y.; Fan, Y.; Hua, S.-N.; Qu, H.-Y.; Dong, S.-W.; Li, R.-L.; Zhao, M.-Y.; Zhen, Y.; Yu, X.-L. Alpha-enolase promotes cell glycolysis, growth, migration, and invasion in non-small cell lung cancer through FAK-mediated PI3K/AKT pathway. J. Hematol. Oncol. 2015, 8, 22. [Google Scholar] [CrossRef]
- Qian, X.; Xu, W.; Xu, J.; Shi, Q.; Li, J.; Weng, Y.; Jiang, Z.; Feng, L.; Wang, X.; Zhou, J. Enolase 1 stimulates glycolysis to promote chemoresistance in gastric cancer. Oncotarget 2017, 8, 47691. [Google Scholar] [CrossRef]
- Tu, S.-H.; Chang, C.-C.; Chen, C.-S.; Tam, K.-W.; Wang, Y.-J.; Lee, C.-H.; Lin, H.-W.; Cheng, T.-C.; Huang, C.-S.; Chu, J.-S. Increased expression of enolase α in human breast cancer confers tamoxifen resistance in human breast cancer cells. Breast Cancer Res. Treat. 2010, 121, 539–553. [Google Scholar] [CrossRef]
- Yin, H.; Wang, L.; Liu, H.-L. ENO1 overexpression in pancreatic cancer patients and its clinical and diagnostic significance. Gastroenterol. Res. Pract. 2018, 2018, 3842198. [Google Scholar] [CrossRef]
- Tsai, S.-T.; Chien, I.-H.; Shen, W.-H.; Kuo, Y.-Z.; Jin, Y.-T.; Wong, T.-Y.; Hsiao, J.-R.; Wang, H.-P.; Shih, N.-Y.; Wu, L.-W. ENO1, a potential prognostic head and neck cancer marker, promotes transformation partly via chemokine CCL20 induction. Eur. J. Cancer 2010, 46, 1712–1723. [Google Scholar] [CrossRef]
- Wu, W.; Tang, X.; Hu, W.; Lotan, R.; Hong, W.K.; Mao, L. Identification and validation of metastasis-associated proteins in head and neck cancer cell lines by two-dimensional electrophoresis and mass spectrometry. Clin. Exp. Metastasis 2002, 19, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Zhao, X.; Liu, W. NEDD4L inhibits glycolysis and proliferation of cancer cells in oral squamous cell carcinoma by inducing ENO1 ubiquitination and degradation. Cancer Biol. Ther. 2022, 23, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Kang, M.J.; Choi, J.Y.; Park, J.S.; Park, J.K.; Lee, E.Y.; Lee, E.B.; Pap, T.; Eugene, C.Y.; Song, Y.W. Apolipoprotein B binds to enolase-1 and aggravates inflammation in rheumatoid arthritis. Ann. Rheum. Dis. 2018, 77, 1480–1489. [Google Scholar] [CrossRef] [PubMed]
- Zewinger, S.; Reiser, J.; Jankowski, V.; Alansary, D.; Hahm, E.; Triem, S.; Klug, M.; Schunk, S.J.; Schmit, D.; Kramann, R. Apolipoprotein C3 induces inflammation and organ damage by alternative inflammasome activation. Nat. Immunol. 2020, 21, 30–41. [Google Scholar] [CrossRef]
- Landskron, G.; De la Fuente, M.; Thuwajit, P.; Thuwajit, C.; Hermoso, M.A. Chronic inflammation and cytokines in the tumor microenvironment. J. Immunol. Res. 2014, 2014, 149185. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zhou, B.P. Inflammation: A driving force speeds cancer metastasis. Cell Cycle 2009, 8, 3267–3273. [Google Scholar] [CrossRef]
- Ha, H.; Debnath, B.; Neamati, N. Role of the CXCL8-CXCR1/2 axis in cancer and inflammatory diseases. Theranostics 2017, 7, 1543. [Google Scholar] [CrossRef]
- Schalper, K.A.; Carleton, M.; Zhou, M.; Chen, T.; Feng, Y.; Huang, S.-P.; Walsh, A.M.; Baxi, V.; Pandya, D.; Baradet, T. Elevated serum interleukin-8 is associated with enhanced intratumor neutrophils and reduced clinical benefit of immune-checkpoint inhibitors. Nat. Med. 2020, 26, 688–692. [Google Scholar] [CrossRef]
- Zou, S.; Tong, Q.; Liu, B.; Huang, W.; Tian, Y.; Fu, X. Targeting STAT3 in cancer immunotherapy. Mol. Cancer 2020, 19, 145. [Google Scholar] [CrossRef]
- Wang, H.-Q.; Man, Q.-W.; Huo, F.-Y.; Gao, X.; Lin, H.; Li, S.-R.; Wang, J.; Su, F.-C.; Cai, L.; Shi, Y. STAT3 pathway in cancers: Past, present, and future. MedComm 2022, 3, e124. [Google Scholar] [CrossRef]
- Valeta-Magara, A.; Gadi, A.; Volta, V.; Walters, B.; Arju, R.; Giashuddin, S.; Zhong, H.; Schneider, R.J. Inflammatory breast cancer promotes development of M2 tumor-associated macrophages and cancer mesenchymal cells through a complex chemokine network. Cancer Res. 2019, 79, 3360–3371. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Gao, F.-X.; Wang, C.; Qin, M.; Han, F.; Xu, T.; Hu, Z.; Long, Y.; He, X.-M.; Deng, X. IL-6 and IL-8 secreted by tumour cells impair the function of NK cells via the STAT3 pathway in oesophageal squamous cell carcinoma. J. Exp. Clin. Cancer Res. 2019, 38, 321. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhang, R.; Wang, C.; Gong, W.; Xue, M.; Liu, L.; Zhang, Y. Central neck lymph node metastasis in oral squamous cell carcinoma at the floor of mouth. BMC Cancer 2021, 21, 225. [Google Scholar] [CrossRef]
- Biswas, N.K.; Das, C.; Das, S.; Maitra, A.; Nair, S.; Gupta, T.; D’Cruz, A.K.; Sarin, R.; Majumder, P.P. Lymph node metastasis in oral cancer is strongly associated with chromosomal instability and DNA repair defects. Int. J. Cancer 2019, 145, 2568–2579. [Google Scholar] [CrossRef] [PubMed]
- Shah, J.P.; Gil, Z. Current concepts in management of oral cancer—Surgery. Oral Oncol. 2009, 45, 394–401. [Google Scholar] [CrossRef] [PubMed]
- de Bree, R.; Takes, R.P.; Shah, J.P.; Hamoir, M.; Kowalski, L.P.; Robbins, K.T.; Rodrigo, J.P.; Sanabria, A.; Medina, J.E.; Rinaldo, A. Elective neck dissection in oral squamous cell carcinoma: Past, present and future. Oral Oncol. 2019, 90, 87–93. [Google Scholar] [CrossRef]
- Didiasova, M.; Schaefer, L.; Wygrecka, M. When place matters: Shuttling of enolase-1 across cellular compartments. Front. Cell Dev. Biol. 2019, 7, 61. [Google Scholar] [CrossRef]
- Li, H.-J.; Ke, F.-Y.; Lin, C.-C.; Lu, M.-Y.; Kuo, Y.-H.; Wang, Y.-P.; Liang, K.-H.; Lin, S.-C.; Chang, Y.-H.; Chen, H.-Y. ENO1 Promotes Lung Cancer Metastasis via HGFR and WNT Signaling–Driven Epithelial-to-Mesenchymal Transition. Cancer Res. 2021, 81, 4094–4109. [Google Scholar] [CrossRef]
- Chang, K.-P.; Kao, H.-K.; Yen, T.-C.; Chang, Y.-L.; Liang, Y.; Liu, S.-C.; Lee, L.-Y.; Chang, Y.-L.; Kang, C.-J.; Chen, I.-H. Overexpression of macrophage inflammatory protein-3α in oral cavity squamous cell carcinoma is associated with nodal metastasis. Oral Oncol. 2011, 47, 108–113. [Google Scholar] [CrossRef]
- Xu, X.; Chen, B.; Zhu, S.; Zhang, J.; He, X.; Cao, G.; Chen, B. Hyperglycemia promotes Snail-induced epithelial—Mesenchymal transition of gastric cancer via activating ENO1 expression. Cancer Cell Int. 2019, 19, 344. [Google Scholar] [CrossRef]
- van Dijk, K.W.; Rensen, P.C.; Voshol, P.J.; Havekes, L.M. The role and mode of action of apolipoproteins CIII and AV: Synergistic actors in triglyceride metabolism? Curr. Opin. Lipidol. 2004, 15, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Gong, T.; Zhou, R. ApoC3: An ‘alarmin’triggering sterile inflammation. Nat. Immunol. 2020, 21, 9–11. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.-W.; Karin, M. A cytokine-mediated link between innate immunity, inflammation, and cancer. J. Clin. Investig. 2007, 117, 1175–1183. [Google Scholar] [CrossRef] [PubMed]
- Celià-Terrassa, T.; Kang, Y. Metastatic niche functions and therapeutic opportunities. Nat. Cell Biol. 2018, 20, 868–877. [Google Scholar] [CrossRef] [PubMed]
- Zhong, D.-N.; Ning, Q.-Y.; Wu, J.-Z.; Zang, N.; Wu, J.-L.; Hu, D.-F.; Luo, S.-Y.; Huang, A.-C.; Li, L.-L.; Li, G.-J. Comparative proteomic profiles indicating genetic factors may involve in hepatocellular carcinoma familial aggregation. Cancer Sci. 2012, 103, 1833–1838. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-L.; Lin, T.-S.; Tsai, C.-H.; Wu, C.-C.; Chung, T.; Chien, K.-Y.; Wu, M.; Chang, Y.-S.; Yu, J.-S.; Chen, Y.-T. Identification of potential bladder cancer markers in urine by abundant-protein depletion coupled with quantitative proteomics. J. Proteom. 2013, 85, 28–43. [Google Scholar] [CrossRef]
- Marín-Vicente, C.; Mendes, M.; de Los Ríos, V.; Fernández-Aceñero, M.J.; Casal, J.I. Identification and Validation of Stage-Associated Serum Biomarkers in Colorectal Cancer Using MS-Based Procedures. Proteom. Clin. Appl. 2020, 14, 1900052. [Google Scholar] [CrossRef]
- Lopez-Bujanda, Z.A.; Haffner, M.C.; Chaimowitz, M.G.; Chowdhury, N.; Venturini, N.J.; Patel, R.A.; Obradovic, A.; Hansen, C.S.; Jacków, J.; Maynard, J.P. Castration-mediated IL-8 promotes myeloid infiltration and prostate cancer progression. Nat. Cancer 2021, 2, 803–818. [Google Scholar] [CrossRef]
- Sanmamed, M.; Perez-Gracia, J.; Schalper, K.; Fusco, J.; Gonzalez, A.; Rodriguez-Ruiz, M.; Oñate, C.; Perez, G.; Alfaro, C.; Martín-Algarra, S. Changes in serum interleukin-8 (IL-8) levels reflect and predict response to anti-PD-1 treatment in melanoma and non-small-cell lung cancer patients. Ann. Oncol. 2017, 28, 1988–1995. [Google Scholar] [CrossRef]
- Zhai, J.; Shen, J.; Xie, G.; Wu, J.; He, M.; Gao, L.; Zhang, Y.; Yao, X.; Shen, L. Cancer-associated fibroblasts-derived IL-8 mediates resistance to cisplatin in human gastric cancer. Cancer Lett. 2019, 454, 37–43. [Google Scholar] [CrossRef]
- Visciano, C.; Liotti, F.; Prevete, N.; Cali, G.; Franco, R.; Collina, F.; De Paulis, A.; Marone, G.; Santoro, M.; Melillo, R. Mast cells induce epithelial-to-mesenchymal transition and stem cell features in human thyroid cancer cells through an IL-8–Akt–Slug pathway. Oncogene 2015, 34, 5175–5186. [Google Scholar] [CrossRef] [PubMed]
- Lalla, R.; Goralnick, S.; Tanzer, M.; Kreutzer, D. Fibrin induces IL-8 expression from human oral squamous cell carcinoma cells. Oral Oncol. 2001, 37, 234–242. [Google Scholar] [CrossRef]
- Watanabe, H.; Iwase, M.; Ohashi, M.; Nagumo, M. Role of interleukin-8 secreted from human oral squamous cell carcinoma cell lines. Oral Oncol. 2002, 38, 670–679. [Google Scholar] [CrossRef]
- Alfaro, C.; Sanmamed, M.F.; Rodríguez-Ruiz, M.E.; Teijeira, Á.; Oñate, C.; González, Á.; Ponz, M.; Schalper, K.A.; Pérez-Gracia, J.L.; Melero, I. Interleukin-8 in cancer pathogenesis, treatment and follow-up. Cancer Treat. Rev. 2017, 60, 24–31. [Google Scholar] [CrossRef]
- Asfaha, S.; Dubeykovskiy, A.N.; Tomita, H.; Yang, X.; Stokes, S.; Shibata, W.; Friedman, R.A.; Ariyama, H.; Dubeykovskaya, Z.A.; Muthupalani, S. Mice that express human interleukin-8 have increased mobilization of immature myeloid cells, which exacerbates inflammation and accelerates colon carcinogenesis. Gastroenterology 2013, 144, 155–166. [Google Scholar] [CrossRef]
- Bakouny, Z.; Choueiri, T.K. IL-8 and cancer prognosis on immunotherapy. Nat. Med. 2020, 26, 650–651. [Google Scholar] [CrossRef]
- Zheng, T.; Ma, G.; Tang, M.; Li, Z.; Xu, R. IL-8 secreted from M2 macrophages promoted prostate tumorigenesis via STAT3/MALAT1 pathway. Int. J. Mol. Sci. 2018, 20, 98. [Google Scholar] [CrossRef]
- Brierley, J.D.; Gospodarowicz, M.K.; Wittekind, C. TNM Classification of Malignant Tumours; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Zhang, L.-Z.; Man, Q.-W.; Liu, J.-Y.; Zhong, W.-Q.; Zheng, Y.-Y.; Zhao, Y.-F.; Liu, B. Overexpression of Fra-1, c-Jun and c-Fos in odontogenic keratocysts: Potential correlation with proliferative and anti-apoptotic activity. Histopathology 2018, 73, 933–942. [Google Scholar] [CrossRef]
- Li, R.-F.; Man, Q.-W.; Liu, J.-Y.; Zheng, Y.-Y.; Gao, X.; Liu, H.-M. Overexpression of T-type calcium channel Cav3. 1 in oral squamous cell carcinoma: Association with proliferation and anti-apoptotic activity. J. Mol. Histol. 2021, 52, 511–520. [Google Scholar] [CrossRef]
- Bu, D.; Luo, H.; Huo, P.; Wang, Z.; Zhang, S.; He, Z.; Wu, Y.; Zhao, L.; Liu, J.; Guo, J. KOBAS-i: Intelligent prioritization and exploratory visualization of biological functions for gene enrichment analysis. Nucleic Acids Res. 2021, 49, W317–W325. [Google Scholar] [CrossRef]
- Kanehisa, M.; Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M. Toward understanding the origin and evolution of cellular organisms. Protein Sci. 2019, 28, 1947–1951. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Furumichi, M.; Sato, Y.; Ishiguro-Watanabe, M.; Tanabe, M. KEGG: Integrating viruses and cellular organisms. Nucleic Acids Res. 2021, 49, D545–D551. [Google Scholar] [CrossRef] [PubMed]
- Szklarczyk, D.; Gable, A.L.; Nastou, K.C.; Lyon, D.; Kirsch, R.; Pyysalo, S.; Doncheva, N.T.; Legeay, M.; Fang, T.; Bork, P. The STRING database in 2021: Customizable protein–protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res. 2021, 49, D605–D612. [Google Scholar] [CrossRef] [PubMed]
- Szklarczyk, D.; Gable, A.L.; Lyon, D.; Junge, A.; Wyder, S.; Huerta-Cepas, J.; Simonovic, M.; Doncheva, N.T.; Morris, J.H.; Bork, P. STRING v11: Protein—Protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019, 47, D607–D613. [Google Scholar] [CrossRef]
- Man, Q.-W.; Zhang, L.-Z.; Zhao, Y.; Liu, J.-Y.; Zheng, Y.-Y.; Zhao, Y.-F.; Liu, B. Lymphocyte-derived microparticles stimulate osteoclastogenesis by inducing RANKL in fibroblasts of odontogenic keratocysts. Oncol. Rep. 2018, 40, 3335–3345. [Google Scholar] [CrossRef]
- Li, R.-F.; Zhang, W.; Man, Q.-W.; Zhao, Y.-F.; Zhao, Y. Tunneling nanotubes mediate intercellular communication between endothelial progenitor cells and osteoclast precursors. J. Mol. Histol. 2019, 50, 483–491. [Google Scholar] [CrossRef]

| Accession | Gene | Identified Proteins | Coverage | Peptides |
|---|---|---|---|---|
| P02042 | HBD | Hemoglobin subunit delta | 70.07 | 37 |
| A0A0C4DH69 | IGKV1-9 | Immunoglobulin kappa variable 1-9 | 48.72 | 5 |
| A0A0C4DH38 | IGHV5-51 | Immunoglobulin heavy variable 5-51 | 45.30 | 6 |
| P02656 | APOC3 | Apolipoprotein C-III | 44.44 | 3 |
| P01611 | IGKV1D-12 | Immunoglobulin kappa variable 1D-12 | 43.59 | 7 |
| P09382 | LGALS1 | Galectin-1 | 42.21 | 4 |
| A0A087WSY6 | IGKV3D-15 | Immunoglobulin kappa variable 3D-15 | 40.00 | 8 |
| P00915 | CA1 | Carbonic anhydrase 1 | 35.62 | 7 |
| P61626 | LYZ | Lysozyme C | 35.13 | 3 |
| P06312 | IGKV4-1 | Immunoglobulin kappa variable 4-1 | 34.70 | 7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Man, Q.; Zhong, N.; Wang, H.; Zhang, C.; Li, S.; Bu, L.; Liu, B. ENO1 Binds to ApoC3 and Impairs the Proliferation of T Cells via IL-8/STAT3 Pathway in OSCC. Int. J. Mol. Sci. 2022, 23, 12777. https://doi.org/10.3390/ijms232112777
Wang J, Man Q, Zhong N, Wang H, Zhang C, Li S, Bu L, Liu B. ENO1 Binds to ApoC3 and Impairs the Proliferation of T Cells via IL-8/STAT3 Pathway in OSCC. International Journal of Molecular Sciences. 2022; 23(21):12777. https://doi.org/10.3390/ijms232112777
Chicago/Turabian StyleWang, Jing, Qiwen Man, Niannian Zhong, Hanqi Wang, Chenxi Zhang, Suran Li, Linlin Bu, and Bing Liu. 2022. "ENO1 Binds to ApoC3 and Impairs the Proliferation of T Cells via IL-8/STAT3 Pathway in OSCC" International Journal of Molecular Sciences 23, no. 21: 12777. https://doi.org/10.3390/ijms232112777
APA StyleWang, J., Man, Q., Zhong, N., Wang, H., Zhang, C., Li, S., Bu, L., & Liu, B. (2022). ENO1 Binds to ApoC3 and Impairs the Proliferation of T Cells via IL-8/STAT3 Pathway in OSCC. International Journal of Molecular Sciences, 23(21), 12777. https://doi.org/10.3390/ijms232112777








