Influence of Mn Ions’ Insertion in Pseudo-Tetragonal Phased CaBi4Ti4O15-Based Ceramics for Highly Efficient Energy Storage Devices and High-Temperature Piezoelectric Applications
Abstract
1. Introduction
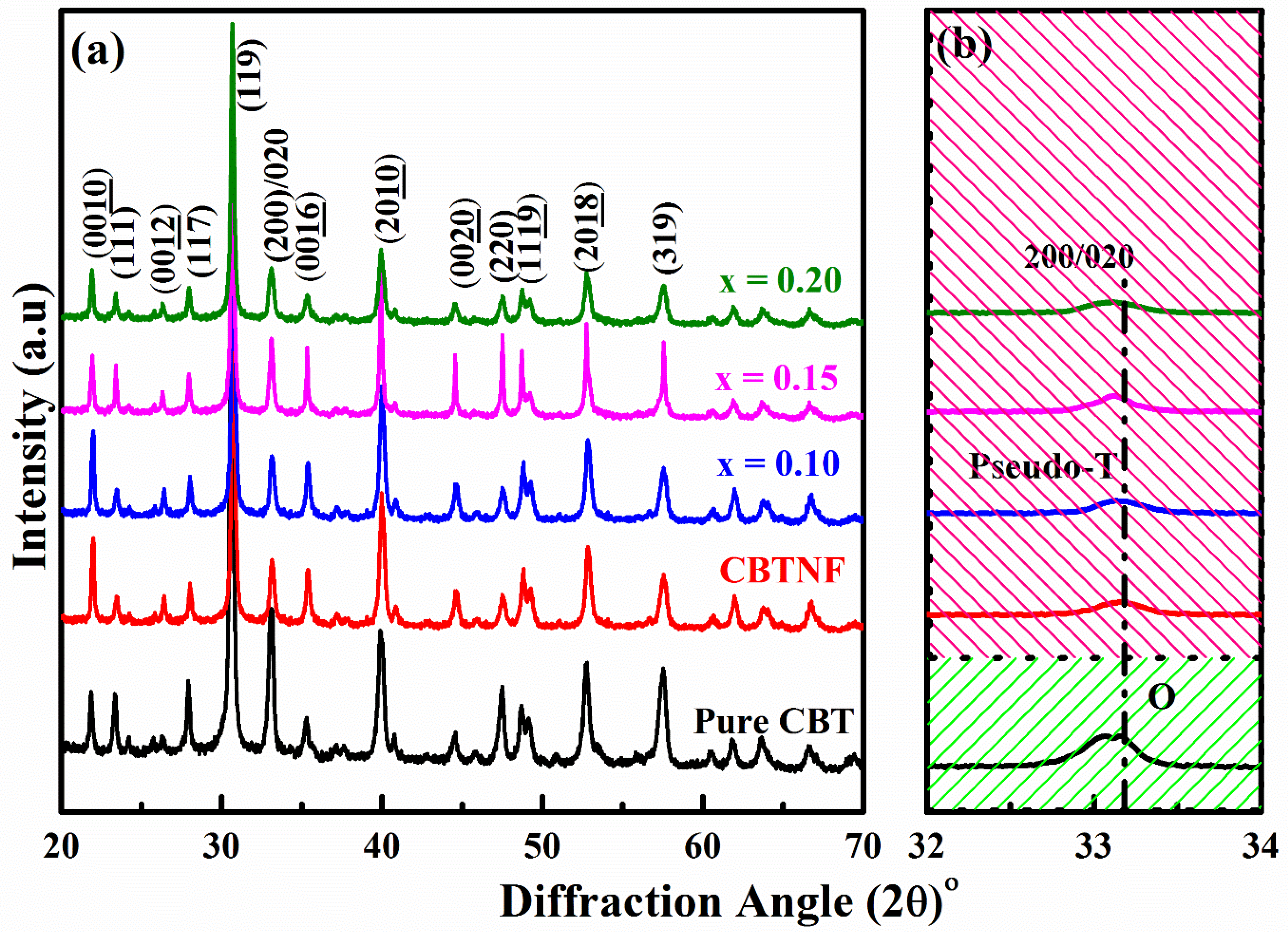
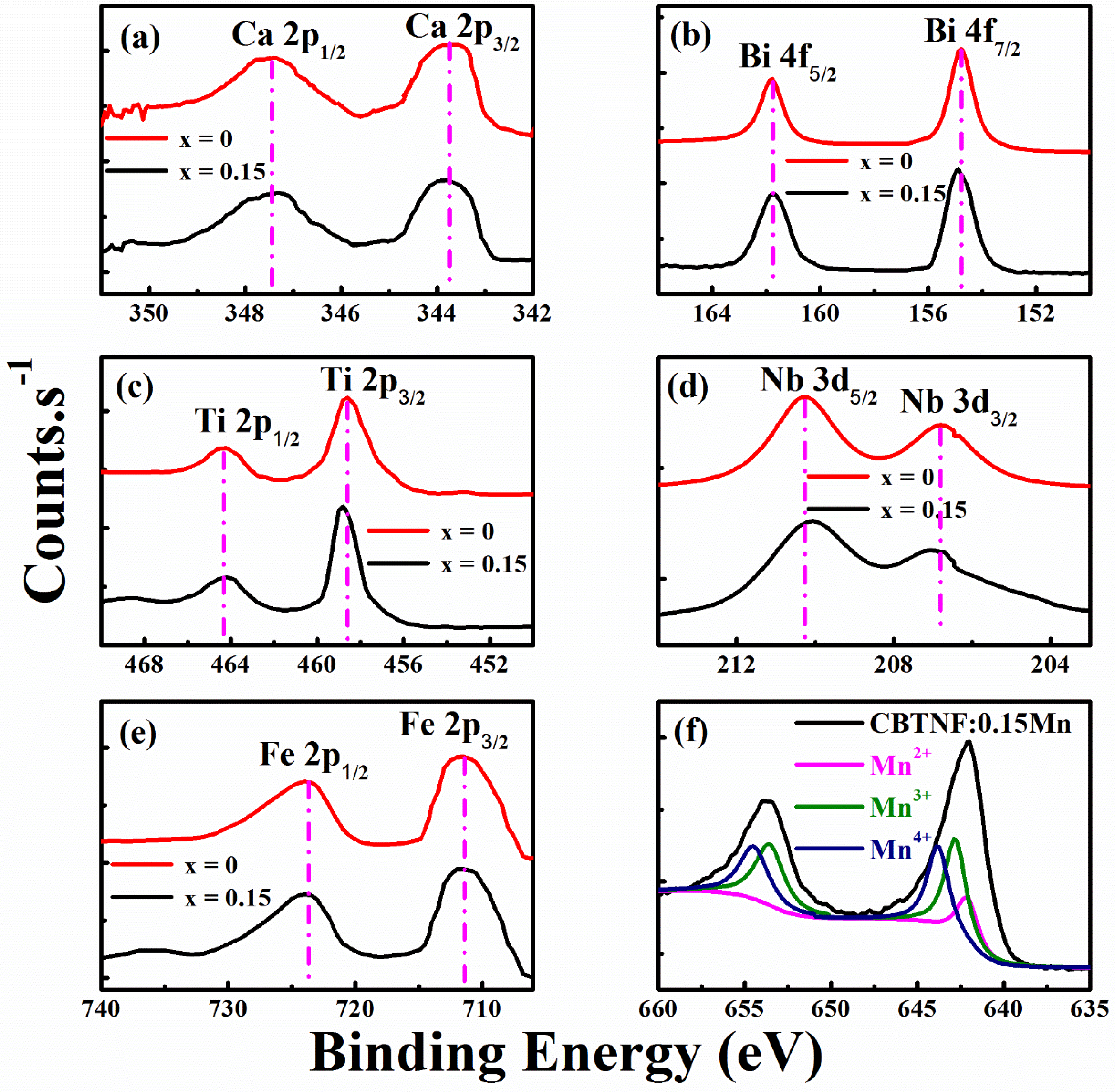
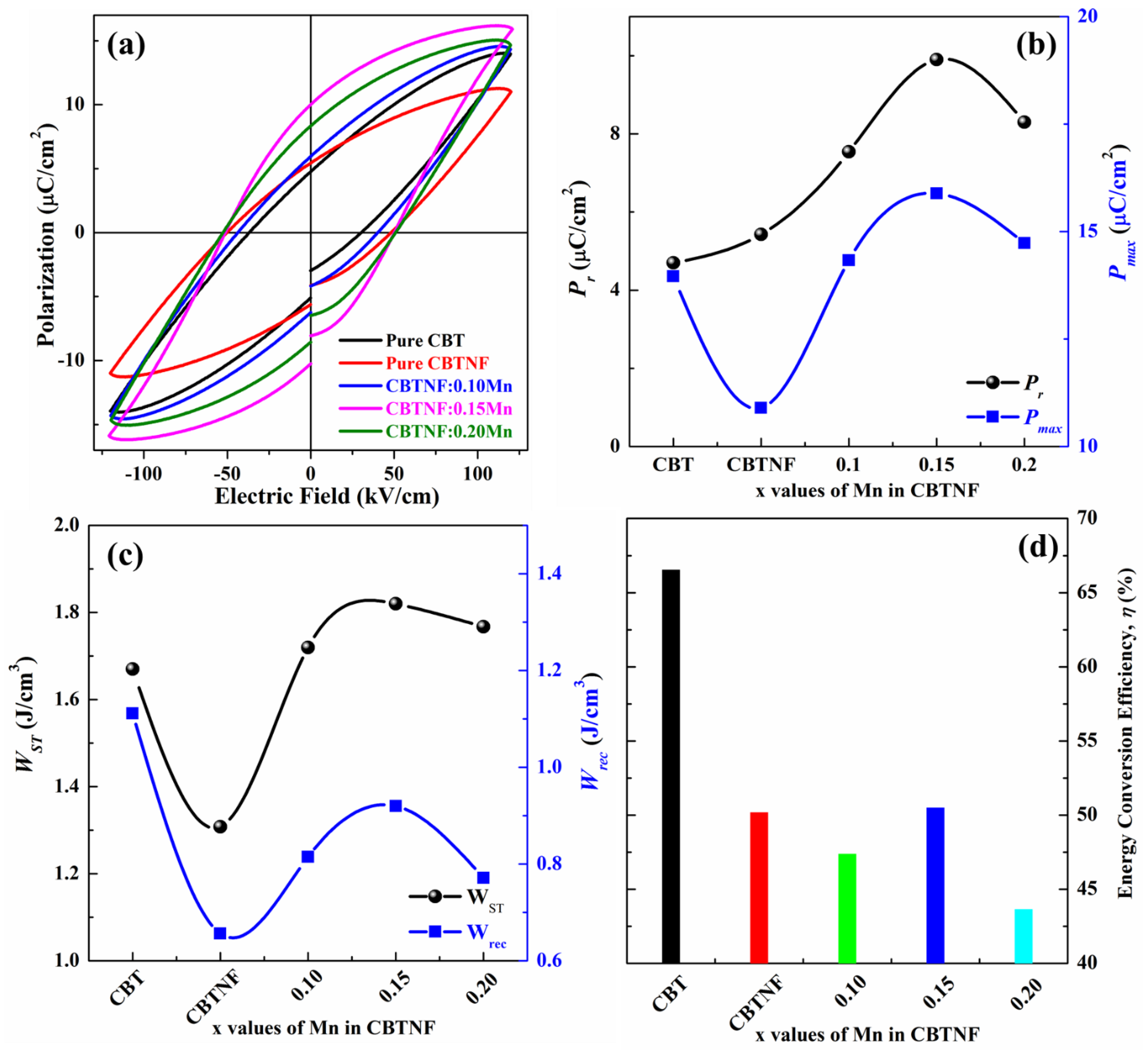
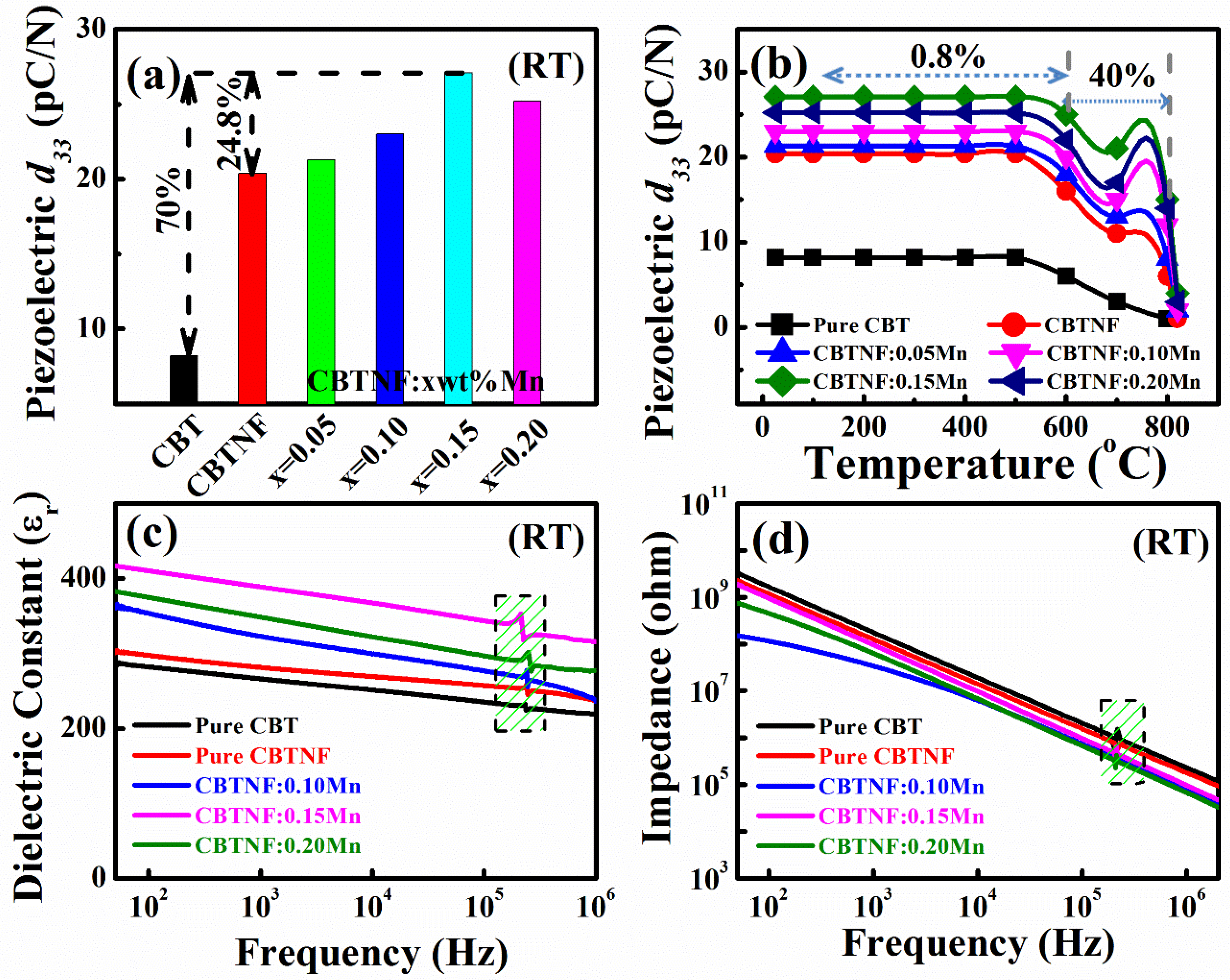
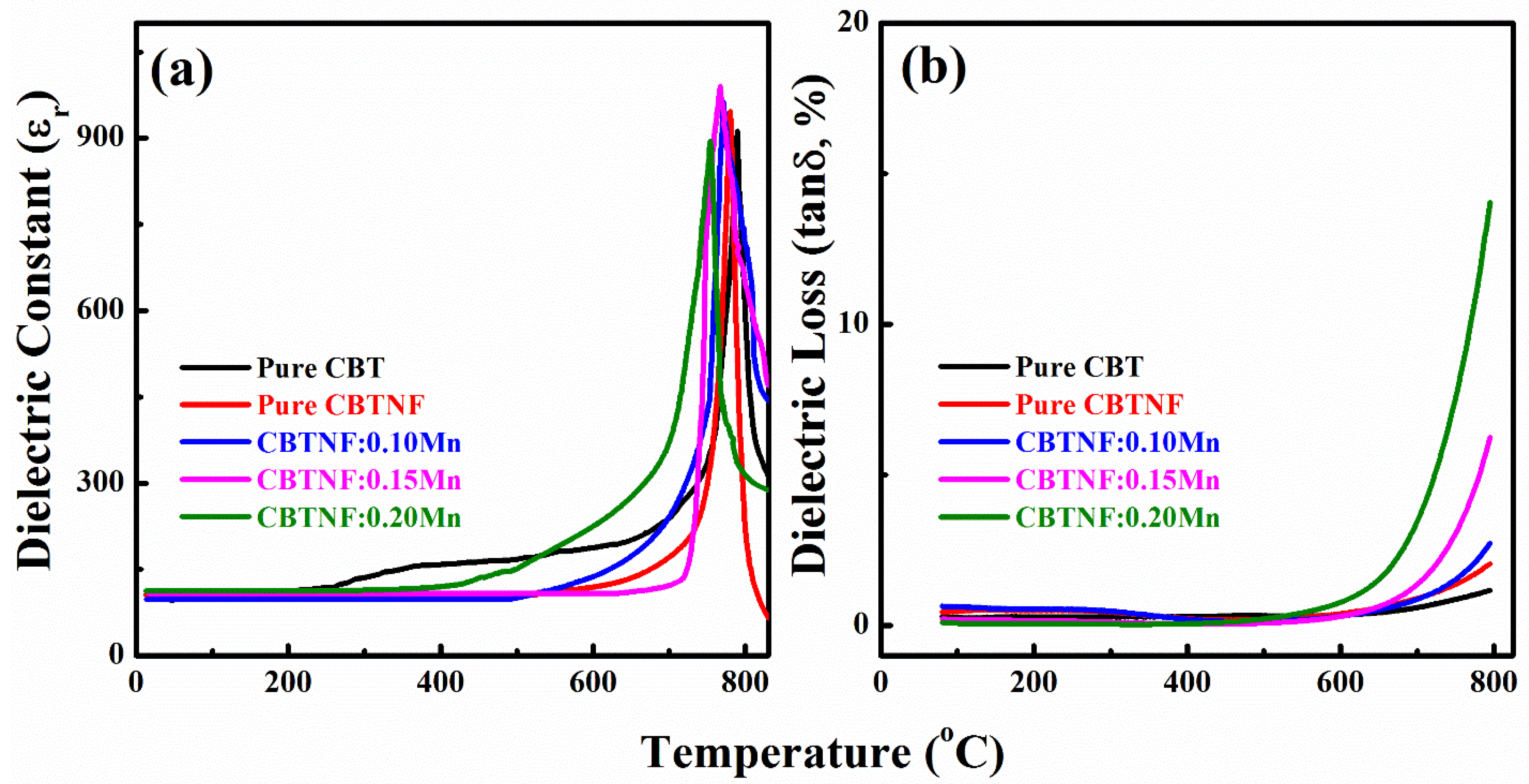
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sekhar, B.C.; Dhanalakshmi, B.; Rao, B.S.; Ramesh, S.; Prasad, K.V.; Rao, P.S.; Rao, B.P. Piezoelectricity and its applications. Multifunct. Ferroelectr. Mater. 2021, 71. [Google Scholar] [CrossRef]
- Kalimuldina, G.; Turdakyn, N.; Abay, I.; Medeubayev, A.; Nurpeissova, A.; Adair, D.; Bakenov, Z. A review of piezoelectric PVDF film by electrospinning and its applications. Sensors 2020, 20, 5214. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Cheng, J.; Dong, S. Review on high temperature piezoelectric ceramics and actuators based on BiScO3–PbTiO3 solid solutions. J. Adv. Dielectr. 2014, 4, 1430002. [Google Scholar] [CrossRef]
- Cutts, J.A.; Balint, T.S.; Chassefiere, E.; Kolawa, E.A. Technology perspectives in the future exploration of Venus. Geophys. Monogr.-Am. Geophys. Union 2007, 176, 207. [Google Scholar]
- Jordan, J.L.; Ponchak, G.E.; Neudeck, P.; Spry, D. First iteration communications circuit for a long-lived in-situ solar system explorer (LLISSE). In Proceedings of the 2020 IEEE Aerospace Conference, Big Sky, MT, USA, 7–14 March 2020; pp. 1–11. [Google Scholar]
- Yang, C.; Lv, P.; Qian, J.; Han, Y.; Ouyang, J.; Lin, X.; Huang, S.; Cheng, Z. Fatigue-free and bending-endurable flexible Mn-doped Na0.5Bi0. 5TiO3-BaTiO3-BiFeO3 film capacitor with an ultrahigh energy storage performance. Adv. Energy Mater. 2019, 9, 1803949. [Google Scholar] [CrossRef]
- Lv, P.; Yang, C.; Qian, J.; Wu, H.; Huang, S.; Cheng, X.; Cheng, Z. Flexible lead-free perovskite oxide multilayer film capacitor based on (Na0.8K0.2)0.5 Bi0.5TiO3/Ba0.5Sr0.5(Ti0.97Mn0.03)O3 for high-performance dielectric energy storage. Adv. Energy Mater. 2020, 10, 1904229. [Google Scholar] [CrossRef]
- Kurchania, R.; Subohi, O. Aurivillius Phase Materials: Exploring Lead-Free Ferroelectrics; IOP Publishing: Bristol, UK, 2021. [Google Scholar]
- Qaiser, M.A.; Hussain, A.; Zhang, J.; Wang, Y.; Zhang, S.; Chen, L.; Yuan, G. 0–3 type Bi3TaTiO9:40wt%BiFeO3 composite with improved high-temperature piezoelectric properties. J. Alloys Compd. 2018, 740, 1–6. [Google Scholar] [CrossRef]
- Jabeen, N.; Nawaz, S.; Qaiser, M.A.; Rana, M.A.; Hassan, F.; Abbas, Z.; Ahmed, F.; Hussain, A. Stable piezoelectric response of 0–3 type CaBi2Nb2O9:xwt%BiFeO3 composites for high-temperature piezoelectric applications. J. Asian Ceram. Soc. 2021, 9, 312–322. [Google Scholar] [CrossRef]
- Hussain, A.; Qaiser, M.A.; Zhang, J.; Zhang, S.T.; Wang, Y.; Yang, Y.; Liu, Z.; Yuan, G. High-temperature piezoelectric properties of 0–3 type CaBi4Ti4O15:xwt%BiFeO3 composites. J. Am. Ceram. Soc. 2017, 100, 3522–3529. [Google Scholar] [CrossRef]
- Jabeen, N.; Hussain, A.; Nawaz, S.; Qaiser, M.A.; Zafar, R.; Ishtiaq, S.; Ahmed, F.; Usman, M.; Shultan, T.; Muntaha, S.T. Thermally Stable Piezoelectric Performance of MnO2 Inserted Pseudo-tetragonal Phase Existent CaBi2Nb2O9-based Ceramics. Mater. Technol. 2022, 1–9. [Google Scholar] [CrossRef]
- Xi, J.; Xing, J.; Yuan, J.; Nie, R.; Chen, H.; Zhang, W.; Chen, Q.; Zhu, J. Preparation and characterization of Zn-modified CaBi4Ti4O15 piezoelectric ceramics with lower sintering temperature. J. Mater. Sci. Mater. Electron. 2020, 31, 8805–8814. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, P.; Zhang, Y.; Du, J.; Bai, W.; Li, L.; Wen, F.; Zheng, P.; Wu, W.; Zhang, Y. Improved electrical properties in Nb/Fe co-modified CaBi4Ti4O15 high-temperature piezoceramics. Mater. Res. Express 2020, 6, 126334. [Google Scholar] [CrossRef]
- Fan, M.; Wu, G.; Yu, F.; Li, L.; Li, Y.; Cheng, X.; Zhao, X. Spontaneous Growth of CaBi4Ti4O15 Piezoelectric Crystal Using Mixed Flux Agents. Crystals 2020, 10, 698. [Google Scholar] [CrossRef]
- Shen, Z.Y.; Sun, H.; Tang, Y.; Li, Y.; Zhang, S. Enhanced piezoelectric properties of Nb and Mn co-doped CaBi4Ti4O15 high temperature piezoceramics. Mater. Res. Bull. 2015, 63, 129–133. [Google Scholar] [CrossRef]
- Hajra, S.; Sahu, M.; Oh, D.; Kim, H.J. Lead-free and flexible piezoelectric nanogenerator based on CaBi4Ti4O15 Aurivillius oxides/PDMS composites for efficient biomechanical energy harvesting. Ceram. Int. 2021, 47, 15695–15702. [Google Scholar] [CrossRef]
- Wu, D.; Zhou, H.; Li, L.; Chen, Y. Gd/Mn Co-Doped CaBi4Ti4O15 Aurivillius-Phase Ceramics: Structures, Electrical Conduction and Dielectric Relaxation Behaviors. Materials 2022, 15, 5810. [Google Scholar] [CrossRef] [PubMed]
- Xiao, P.; Guo, Y.; Tian, M.; Zheng, Q.; Jiang, N.; Wu, X.; Xia, Z.; Lin, D. Improved ferroelectric/piezoelectric properties and bright green/UC red emission in (Li, Ho)-doped CaBi4Ti4O15 multifunctional ceramics with excellent temperature stability and superior water-resistance performance. Dalton Trans. 2015, 44, 17366–17380. [Google Scholar] [CrossRef] [PubMed]
- Jabeen, N.; Xia, Q.; Savilov, S.V.; Aldoshin, S.M.; Yu, Y.; Xia, H. Enhanced pseudocapacitive performance of α-MnO2 by cation preinsertion. ACS Appl. Mater. Interfaces 2016, 8, 33732–33740. [Google Scholar] [CrossRef]
- Jabeen, N.; Hussain, A.; Xia, Q.; Sun, S.; Zhu, J.; Xia, H. High-performance 2.6 V aqueous asymmetric supercapacitors based on in situ formed Na0.5MnO2 nanosheet assembled nanowall arrays. Adv. Mater. 2017, 29, 1700804. [Google Scholar] [CrossRef] [PubMed]
- Risch, M.; Stoerzinger, K.A.; Han, B.; Regier, T.Z.; Peak, D.; Sayed, S.Y.; Wei, C.; Xu, Z.; Shao-Horn, Y. Redox processes of manganese oxide in catalyzing oxygen evolution and reduction: An in situ soft X-ray absorption spectroscopy study. J. Phys. Chem. C 2017, 121, 17682–17692. [Google Scholar] [CrossRef]
- Hussain, A.; Nawaz, S.; Jabeen, N.; Zafar, R.; Qaiser, M.A.; Abbas, Z.; Ahmed, F.; Khan, M.U.; Waseen, M.; Aslam, S. Enhanced ferroelectric and piezoelectric response by MnO2 added Bi0.5(K0.2Na0.8)0.5TiO3 ceramics. J. Solid State Chem. 2022, 306, 122716. [Google Scholar] [CrossRef]
- Yang, C.; Han, Y.; Qian, J.; Lv, P.; Lin, X.; Huang, S.; Cheng, Z. Flexible, temperature-resistant, and fatigue-free ferroelectric memory based on Bi(Fe0.93Mn0.05Ti0.02)O3 thin film. ACS Appl. Mater. Interfaces 2019, 11, 12647–12655. [Google Scholar] [CrossRef] [PubMed]
- Hui, X.; Peng, D.; Zou, H.; Li, J.; Cao, Q.; Li, Y.; Wang, X.; Yao, X. A new multifunctional Aurivillius oxide Na0.5Er0.5Bi4Ti4O15: Up-conversion luminescent, dielectric, and piezoelectric properties. Ceram. Int. 2014, 40, 12477–12483. [Google Scholar] [CrossRef]
- Chen, J.N.; Kang, C.; Hou, R.M.; Zhao, X.; Wang, C.M. Dielectric, ferroelectric, and piezoelectric properties of Gd-modified CaBi2Nb2O9 high Curie temperature ceramics. J. Mater. Res. 2021, 36, 1086–1096. [Google Scholar] [CrossRef]
- Yang, L.I.U.; Yang, Y.U.; Yin, C.-y.; Zheng, L.; Zheng, P.; Bai, W.-f.; Li, L.-l.; Wen, F.; Zhang, Y. Achieving remarkable piezoelectric activity in Sb–Mn co-modified CaBi4Ti4O15 piezoelectric ceramics. Trans. Nonferrous Met. Soc. China 2021, 31, 2442–2453. [Google Scholar]
- Wendari, T.P.; Ramadhani, R.; Putri, Y.E. La3+ substitution induced structural transformation in CaBi4Ti4O15 Aurivillius phases: Synthesis, morphology, dielectric and optical properties. Ceram. Int. 2021, 47, 23549–23557. [Google Scholar]
- Suresh, M.B.; Ramana, E.V.; Babu, S.N.; Suryanarayana, S.V. Comparison of electrical and dielectric properties of BLSF materials in Bi-Fe-Ti-O and Bi-Mn-Ti-O systems. Ferroelectrics 2006, 332, 57–63. [Google Scholar] [CrossRef]
- Naumkin, A.V.; Kraut-Vass, A.; Gaarenstroom, S.W.; Powell, C.J. NIST X-ray Photoelectron Spectroscopy Database, NIST Standard Reference Database 20; Version 4.1; US Department of Commerce: Washington, DC, USA, 2012. [Google Scholar]
- Liu, G.; Wang, D.; Wu, C.; Wu, J.; Chen, Q. A realization of excellent piezoelectricity and good thermal stability in CaBi2Nb2O9: Pseudo phase boundary. J. Am. Ceram. Soc. 2019, 102, 1794–1804. [Google Scholar] [CrossRef]
- Nawaz, R.; Kait, C.F.; Chia, H.Y.; Isa, M.H.; Huei, L.W. Glycerol-mediated facile synthesis of colored titania nanoparticles for visible light photodegradation of phenolic compounds. Nanomaterials 2019, 9, 1586. [Google Scholar] [CrossRef] [PubMed]
- Shvab, R.; Hryha, E.; Nyborg, L. Surface chemistry of the titanium powder studied by XPS using internal standard reference. Powder Metall. 2017, 60, 42–48. [Google Scholar] [CrossRef]
- Zhuk, N.A.; Lutoev, V.P.; Makeev, B.A.; Nekipelov, S.V.; Koroleva, A.V.; Fedorova, A.V.; Yermolina, V.D.; Beznosikov, S.; Karlova, L.O. Magnetic susceptibility, EPR, NEXAFS and XPS spectra of Fe-doped CaBi2Nb2O9. J. Mater. Res. Technol. 2020, 9, 4173–4182. [Google Scholar] [CrossRef]
- Yamashita, T.; Hayes, P. Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials. Appl. Surf. Sci. 2008, 254, 2441–2449. [Google Scholar] [CrossRef]
- Zhao, Z.; Dai, Y.; Li, X.; Zhao, Z.; Zhang, X. The evolution mechanism of defect dipoles and high strain in MnO2-doped KNN lead-free ceramics. Appl. Phys. Lett. 2016, 108, 172906. [Google Scholar] [CrossRef]
- Li, G.; Zheng, L.; Yin, Q.; Jiang, B.; Cao, W. Microstructure and ferroelectric properties of MnO2-doped bismuth-layer (Ca, Sr)Bi4Ti4O15 ceramics. J. Appl. Phys. 2005, 98, 064108. [Google Scholar] [CrossRef]
- Jabeen, N.; Rehman, A.U.; Hassan, N.U.; Qaiser, M.A.; Zaidi, A.; Khan, M.U.; Khan, I.A.; Nouman, M. Boosting of Magnetic, Ferroelectric, Energy Storage Efficiency, and Piezoelectric Properties of Zn Intercalated SrBi4Ti4O15-Based Ceramics. Materials 2022, 15, 5057. [Google Scholar] [CrossRef]
- Zhang, H.; Wei, T.; Zhang, Q.; Ma, W.; Fan, P.; Salamon, D.; Zhang, S.-T.; Nan, B.; Tan, H.; Ye, Z.G. A review on the development of lead-free ferroelectric energy-storage ceramics and multilayer capacitors. J. Mater. Chem. C 2020, 8, 16648–16667. [Google Scholar] [CrossRef]
- Hussain, A.; Zheng, P.; Xu, X.; Chen, X.; Li, T.; Wang, Y.; Yuan, G.; Yin, J.; Liu, Z. Structural and electrical properties of multiferroic (1− x)BiFeO3−xBi0.5K0.5TiO3 ceramics. J. Alloys Compd. 2016, 678, 228–233. [Google Scholar] [CrossRef]
- Zhang, J.; Pan, Z.; Guo, F.F.; Liu, W.C.; Ning, H.; Chen, Y.B.; Lu, M.-H.; Yang, B.; Chen, J.; Zhang, S.H.; et al. Semiconductor/relaxor 0–3 type composites without thermal depolarization in Bi0.5Na0.5TiO3-based lead-free piezoceramics. Nat. Commun. 2015, 6, 6615. [Google Scholar] [CrossRef]
- Qaiser, M.A.; Hussain, A.; Xu, Y.; Wang, Y.; Wang, Y.; Yang, Y.; Yuan, G. CuO added Pb0.92Sr0.06Ba0.02(Mg1/3Nb2/3)0.25(Ti0.53Zr0.47)0.75O3 ceramics sintered with Ag electrodes at 900°C for multilayer piezoelectric actuator. Chin. Phys. B 2017, 26, 037702. [Google Scholar] [CrossRef]
- Xu, X.; Jiang, J.; Ahmad, H.; Yuan, G.; Yin, J.; Liu, Z. Improved ferroelectricity of (1−x)Na0.5Bi0.5TiO3–xBaTiO3 ceramics rapidly sintered at low temperature. Ceram. Int. 2014, 40, 11819–11824. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hussain, A.; Jabeen, N.; Hassan, N.U.; Rahman, A.U.; Khan, M.U.; Naz, A.; Yousef, E.S. Influence of Mn Ions’ Insertion in Pseudo-Tetragonal Phased CaBi4Ti4O15-Based Ceramics for Highly Efficient Energy Storage Devices and High-Temperature Piezoelectric Applications. Int. J. Mol. Sci. 2022, 23, 12723. https://doi.org/10.3390/ijms232112723
Hussain A, Jabeen N, Hassan NU, Rahman AU, Khan MU, Naz A, Yousef ES. Influence of Mn Ions’ Insertion in Pseudo-Tetragonal Phased CaBi4Ti4O15-Based Ceramics for Highly Efficient Energy Storage Devices and High-Temperature Piezoelectric Applications. International Journal of Molecular Sciences. 2022; 23(21):12723. https://doi.org/10.3390/ijms232112723
Chicago/Turabian StyleHussain, Ahmad, Nawishta Jabeen, Najam Ul Hassan, Altaf Ur Rahman, Muhammad Usman Khan, Adeela Naz, and El Sayed Yousef. 2022. "Influence of Mn Ions’ Insertion in Pseudo-Tetragonal Phased CaBi4Ti4O15-Based Ceramics for Highly Efficient Energy Storage Devices and High-Temperature Piezoelectric Applications" International Journal of Molecular Sciences 23, no. 21: 12723. https://doi.org/10.3390/ijms232112723
APA StyleHussain, A., Jabeen, N., Hassan, N. U., Rahman, A. U., Khan, M. U., Naz, A., & Yousef, E. S. (2022). Influence of Mn Ions’ Insertion in Pseudo-Tetragonal Phased CaBi4Ti4O15-Based Ceramics for Highly Efficient Energy Storage Devices and High-Temperature Piezoelectric Applications. International Journal of Molecular Sciences, 23(21), 12723. https://doi.org/10.3390/ijms232112723









