AQP5-1364A/C Polymorphism Affects AQP5 Promoter Methylation
Abstract
1. Introduction
2. Results
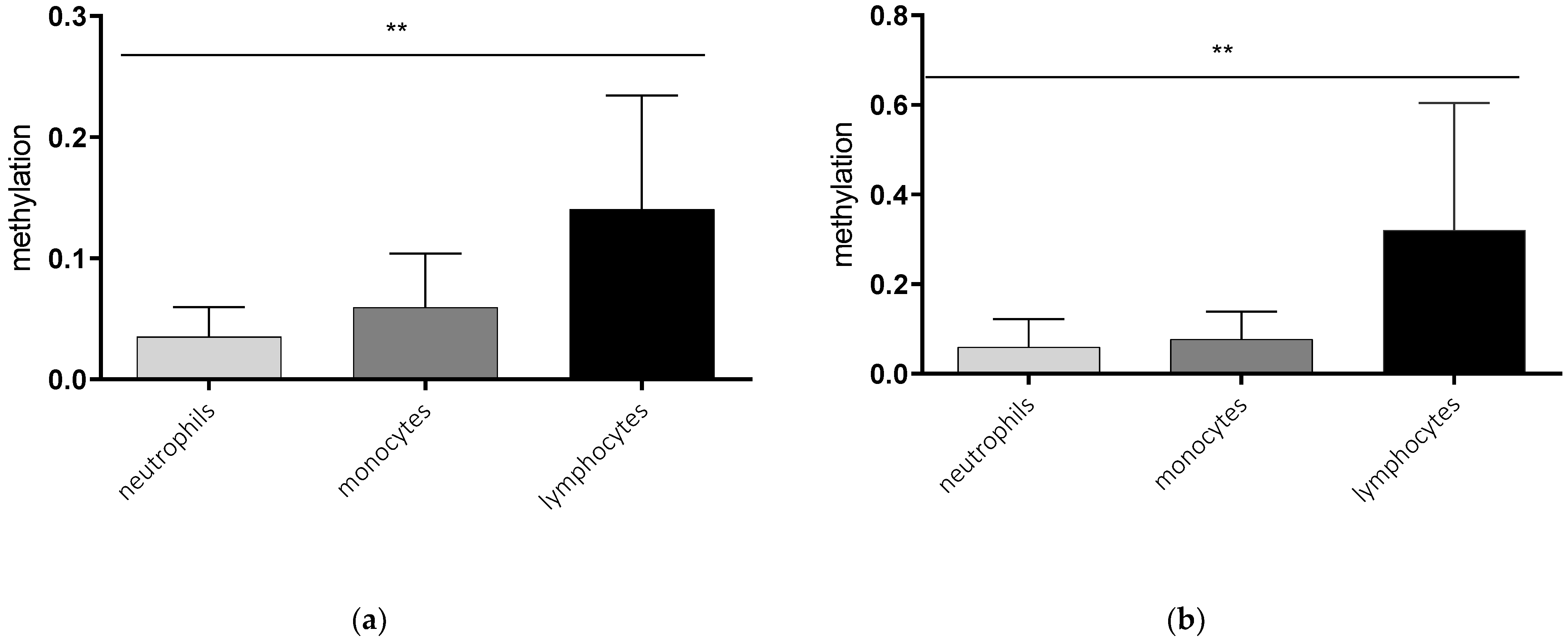
2.1. Cell Type-Specific Methylation of the AQP5 Promoter
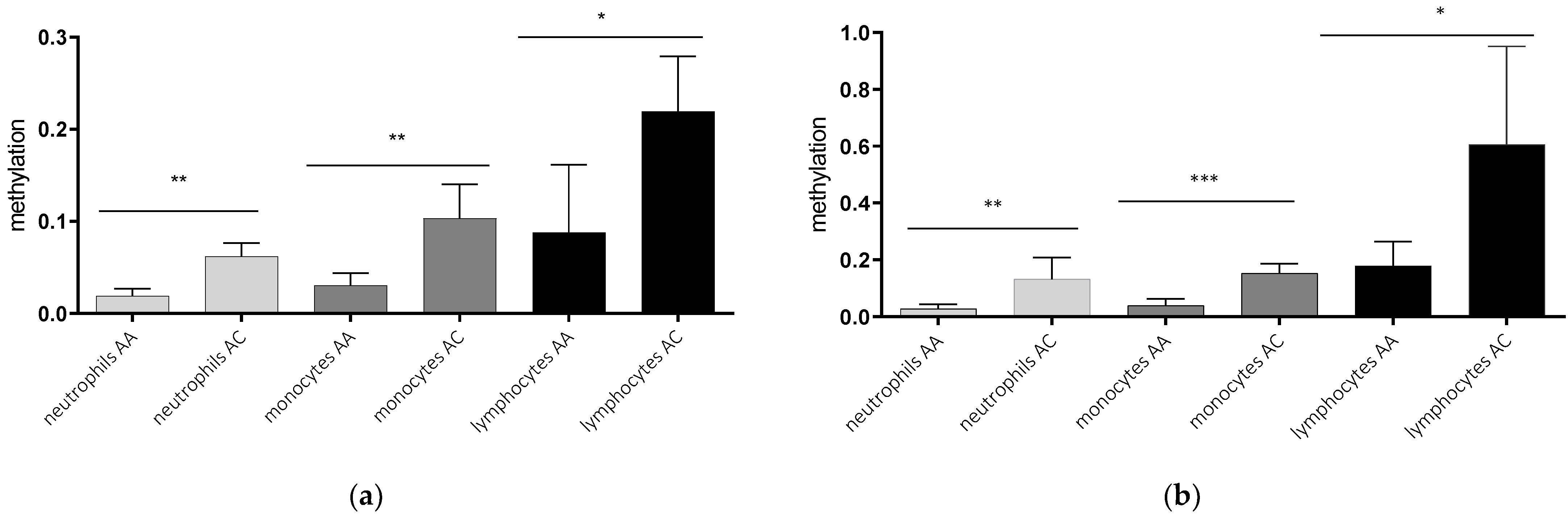
2.2. AQP5-1364A/C Genotype-Dependent AQP5 Promoter Methylation
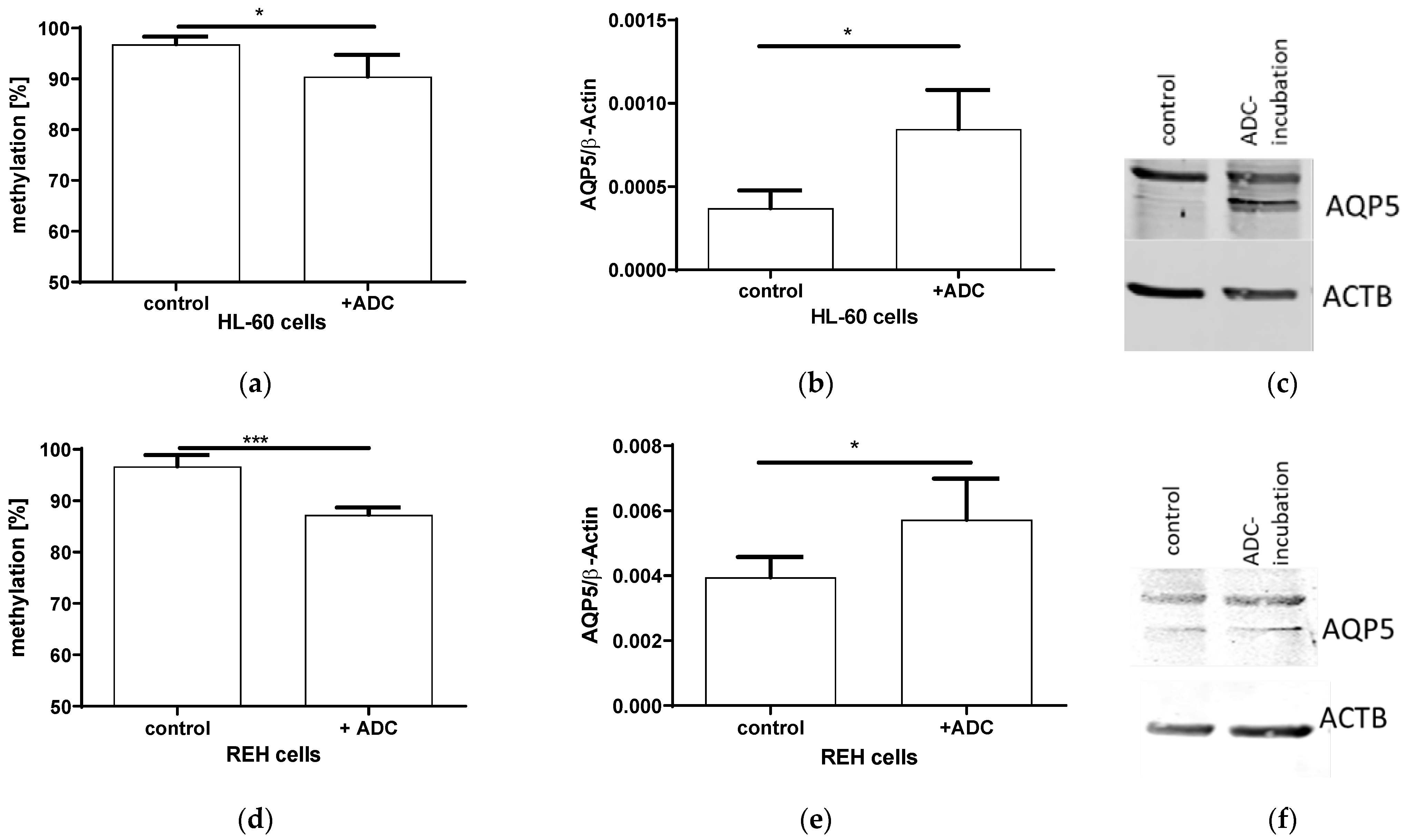
2.3. Expression and AQP5 Promoter Methylation after ADC Incubation
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Blood Sample Collection, Preparation and Storage
4.3. Genotyping of Patients and Healthy Volunteers
4.4. Overall DNA Methylation Analysis by Methylation-Specific Polymer Chain Reaction (PCR)
4.5. Methylation, mRNA and Protein Expression after 5-Aza-2′-Deoxycytidin (ADC) Incubation
4.6. Statistics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chousterman, B.G.; Swirski, F.K.; Weber, G.F. Cytokine storm and sepsis disease pathogenesis. Semin. Immunopathol. 2017, 39, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Gupta, E.; Kaushik, S.; Jyoti, A. Neutrophil Extracellular Traps: Formation and Involvement in Disease Progression. Iran. J. Allergy Asthma Immunol. 2018, 17, 208–220. [Google Scholar] [PubMed]
- Fleischmann, C.; Scherag, A.; Adhikari, N.K.; Hartog, C.S.; Tsaganos, T.; Schlattmann, P.; Angus, D.C.; Reinhart, K. Assessment of Global Incidence and Mortality of Hospital-treated Sepsis. Current Estimates and Limitations. Am. J. Respir. Crit. Care Med. 2016, 193, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Adamzik, M.; Frey, U.H.; Mohlenkamp, S.; Scherag, A.; Waydhas, C.; Marggraf, G.; Dammann, M.; Steinmann, J.; Siffert, W.; Peters, J. Aquaporin 5 gene promoter—1364A/C polymorphism associated with 30-day survival in severe sepsis. Anesthesiology 2011, 114, 912–917. [Google Scholar] [CrossRef] [PubMed]
- Adamzik, M.; Schafer, S.; Frey, U.H.; Becker, A.; Kreuzer, M.; Winning, S.; Frede, S.; Steinmann, J.; Fandrey, J.; Zacharowski, K.; et al. The NFKB1 promoter polymorphism (−94ins/delATTG) alters nuclear translocation of NF-kappaB1 in monocytes after lipopolysaccharide stimulation and is associated with increased mortality in sepsis. Anesthesiology 2013, 118, 123–133. [Google Scholar] [CrossRef]
- Rump, K.; Adamzik, M. Function of aquaporins in sepsis: A systematic review. Cell Biosci. 2018, 8, 10. [Google Scholar] [CrossRef]
- Adamzik, M.; Frey, U.H.; Bitzer, K.; Jakob, H.; Baba, H.A.; Schmieder, R.E.; Schneider, M.P.; Heusch, G.; Peters, J.; Siffert, W. A novel-1364A/C aquaporin 5 gene promoter polymorphism influences the responses to salt loading of the renin-angiotensin-aldosterone system and of blood pressure in young healthy men. Basic Res. Cardiol. 2008, 103, 598–610. [Google Scholar] [CrossRef]
- Ahmed, T.; Ghafoor, S. Aquaporins; systemic, functional and therapeutic correlations in health and disease. J. Pak. Med. Assoc. 2021, 71, 1228–1233. [Google Scholar] [CrossRef]
- Bergmann, L.; Nowak, H.; Siffert, W.; Peters, J.; Adamzik, M.; Koos, B.; Rahmel, T. Major Adverse Kidney Events Are Associated with the Aquaporin 5-1364A/C Promoter Polymorphism in Sepsis: A Prospective Validation Study. Cells 2020, 9, 904. [Google Scholar] [CrossRef]
- Rump, K.; Unterberg, M.; Bergmann, L.; Bankfalvi, A.; Menon, A.; Schafer, S.; Scherag, A.; Bazzi, Z.; Siffert, W.; Peters, J.; et al. AQP5-1364A/C polymorphism and the AQP5 expression influence sepsis survival and immune cell migration: A prospective laboratory and patient study. J. Transl. Med. 2016, 14, 321. [Google Scholar] [CrossRef]
- Barros, S.P.; Offenbacher, S. Epigenetics: Connecting environment and genotype to phenotype and disease. J. Dent. Res. 2009, 88, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Yamamura, Y.; Aota, K.; Yamanoi, T.; Kani, K.; Takano, H.; Momota, Y.; Motegi, K.; Azuma, M. DNA demethylating agent decitabine increases AQP5 expression and restores salivary function. J. Dent. Res. 2012, 91, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Nomura, J.; Hisatsune, A.; Miyata, T.; Isohama, Y. The role of CpG methylation in cell type-specific expression of the aquaporin-5 gene. Biochem. Biophys. Res. Commun. 2007, 353, 1017–1022. [Google Scholar] [CrossRef] [PubMed]
- Rump, K.; Unterberg, M.; Dahlke, A.; Nowak, H.; Koos, B.; Bergmann, L.; Siffert, W.; Schafer, S.T.; Peters, J.; Adamzik, M.; et al. DNA methylation of a NF-kappaB binding site in the aquaporin 5 promoter impacts on mortality in sepsis. Sci. Rep. 2019, 9, 18511. [Google Scholar] [CrossRef] [PubMed]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Rump, K.; Siffert, W.; Peters, J.; Adamzik, M. The Transcription Factor NMP4 Binds to the AQP5 Promoter and Is a Novel Transcriptional Regulator of the AQP5 Gene. DNA Cell Biol. 2016, 35, 322–327. [Google Scholar] [CrossRef]
- Kerkel, K.; Spadola, A.; Yuan, E.; Kosek, J.; Jiang, L.; Hod, E.; Li, K.; Murty, V.V.; Schupf, N.; Vilain, E.; et al. Genomic surveys by methylation-sensitive SNP analysis identify sequence-dependent allele-specific DNA methylation. Nat. Genet. 2008, 40, 904–908. [Google Scholar] [CrossRef]
- Tycko, B. Allele-specific DNA methylation: Beyond imprinting. Hum. Mol. Genet. 2010, 19, R210–R220. [Google Scholar] [CrossRef]
- Martin-Trujillo, A.; Iglesias-Platas, I.; Coto, E.; Corral-Juan, M.; San Nicolás, H.; Corral, J.; Volpini, V.; Matilla-Dueñas, A.; Monk, D. Genotype of an individual single nucleotide polymorphism regulates DNA methylation at the TRPC3 alternative promoter. Epigenetics 2011, 6, 1236–1241. [Google Scholar] [CrossRef][Green Version]
- Onuma, H.; Tabara, Y.; Kawamura, R.; Ohashi, J.; Nishida, W.; Takata, Y.; Ochi, M.; Nishimiya, T.; Ohyagi, Y.; Kawamoto, R.; et al. Dual Effects of a RETN Single Nucleotide Polymorphism (SNP) at −420 on Plasma Resistin: Genotype and DNA Methylation. J. Clin. Endocrinol. Metab. 2017, 102, 884–892. [Google Scholar] [CrossRef]
- Nomura, J.; Horie, I.; Seto, M.; Nagai, K.; Hisatsune, A.; Miyata, T.; Isohama, Y. All-trans retinoic acid increases expression of aquaporin-5 and plasma membrane water permeability via transactivation of Sp1 in mouse lung epithelial cells. Biochem. Biophys. Res. Commun. 2006, 351, 1048–1053. [Google Scholar] [CrossRef] [PubMed]
- Moon, C.; Rousseau, R.; Soria, J.C.; Hoque, M.O.; Lee, J.; Jang, S.J.; Trink, B.; Sidransky, D.; Mao, L. Aquaporin expression in human lymphocytes and dendritic cells. Am. J. Hematol. 2004, 75, 128–133. [Google Scholar] [CrossRef]
- Rump, K.; Brendt, P.; Frey, U.H.; Schafer, S.T.; Siffert, W.; Peters, J.; Adamzik, M. Aquaporin 1 and 5 expression evoked by the beta2 adrenoreceptor agonist terbutaline and lipopolysaccharide in mice and in the human monocytic cell line THP-1 is differentially regulated. Shock 2013, 40, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, N.R.; Chau, E.; Garibaldi, B.T.; Mock, J.R.; Sussan, T.; Rao, K.; Rao, K.; Menon, A.G.; D’Alessio, F.R.; Damarla, M.; et al. Aquaporin 5 regulates cigarette smoke induced emphysema by modulating barrier and immune properties of the epithelium. Tissue Barriers 2013, 1, e25248. [Google Scholar] [CrossRef] [PubMed]
- Agre, P. Membrane water transport and aquaporins: Looking back. Biol. Cell 2005, 97, 355–356. [Google Scholar] [CrossRef]
- Papadopoulos, M.C.; Saadoun, S.; Verkman, A.S. Aquaporins and cell migration. Pflugers Arch. 2008, 456, 693–700. [Google Scholar] [CrossRef]
- Huang, D.; Ding, Q.; Chen, S.; Lü, S.; Zhang, Y.; Long, M. E-selectin negatively regulates polymorphonuclear neutrophil transmigration through altered endothelial junction integrity. FASEB J. 2021, 35, e21521. [Google Scholar] [CrossRef]
- Salvermoser, M.; Begandt, D.; Alon, R.; Walzog, B. Nuclear Deformation During Neutrophil Migration at Sites of Inflammation. Front. Immunol. 2018, 9, 2680. [Google Scholar] [CrossRef]
- Zhou, J.Y.; Szasz, T.P.; Stewart-Hutchinson, P.J.; Sivapalan, J.; Todd, E.M.; Deady, L.E.; Cooper, J.A.; Onken, M.D.; Morley, S.C. L-Plastin promotes podosome longevity and supports macrophage motility. Mol. Immunol. 2016, 78, 79–88. [Google Scholar] [CrossRef]
- Kogan, A.N.; von Andrian, U.H. Lymphocyte trafficking. In Microcirculation; Elsevier: Amsterdam, The Netherlands, 2008; pp. 449–482. [Google Scholar]
- Liu, Y.; Gao, H.; Wang, X.; Zeng, Y. Methylation of Inflammatory Cells in Lung Diseases. Adv. Exp. Med. Biol. 2020, 1255, 63–72. [Google Scholar] [CrossRef]
- Gorica, E.; Mohammed, S.A.; Ambrosini, S.; Calderone, V.; Costantino, S.; Paneni, F. Epi-Drugs in Heart Failure. Front. Cardiovasc. Med. 2022, 9, 923014. [Google Scholar] [CrossRef] [PubMed]
- Guenin, S.; Mouallif, M.; Deplus, R.; Lampe, X.; Krusy, N.; Calonne, E.; Delbecque, K.; Kridelka, F.; Fuks, F.; Ennaji, M.M.; et al. Aberrant promoter methylation and expression of UTF1 during cervical carcinogenesis. PLoS ONE 2012, 7, e42704. [Google Scholar] [CrossRef] [PubMed]
- Leclerc, D.; Cao, Y.; Deng, L.; Mikael, L.G.; Wu, Q.; Rozen, R. Differential gene expression and methylation in the retinoid/PPARA pathway and of tumor suppressors may modify intestinal tumorigenesis induced by low folate in mice. Mol. Nutr. Food Res. 2013, 57, 686–697. [Google Scholar] [CrossRef] [PubMed]
- Yao, D.M.; Zhou, J.D.; Zhang, Y.Y.; Yang, L.; Wen, X.M.; Yang, J.; Guo, H.; Chen, Q.; Lin, J.; Qian, J. GPX3 promoter is methylated in chronic myeloid leukemia. Int. J. Clin. Exp. Pathol. 2015, 8, 6450–6457. [Google Scholar] [PubMed]
- Levy, M.M.; Fink, M.P.; Marshall, J.C.; Abraham, E.; Angus, D.; Cook, D.; Cohen, J.; Opal, S.M.; Vincent, J.L.; Ramsay, G. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Intensive Care Med. 2003, 29, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.C.; Balk, R.A.; Cerra, F.B.; Dellinger, R.P.; Fein, A.M.; Knaus, W.A.; Schein, R.M.; Sibbald, W.J. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest 1992, 101, 1644–1655. [Google Scholar] [CrossRef]
- Agrawal, S.; Unterberg, M.; Koschmieder, S.; zur Stadt, U.; Brunnberg, U.; Verbeek, W.; Büchner, T.; Berdel, W.E.; Serve, H.; Müller-Tidow, C. DNA methylation of tumor suppressor genes in clinical remission predicts the relapse risk in acute myeloid leukemia. Cancer Res. 2007, 67, 1370–1377. [Google Scholar] [CrossRef]
- Thangavel, J.; Malik, A.B.; Elias, H.K.; Rajasingh, S.; Simpson, A.D.; Sundivakkam, P.K.; Vogel, S.M.; Xuan, Y.T.; Dawn, B.; Rajasingh, J. Combinatorial therapy with acetylation and methylation modifiers attenuates lung vascular hyperpermeability in endotoxemia-induced mouse inflammatory lung injury. Am. J. Pathol. 2014, 184, 2237–2249. [Google Scholar] [CrossRef]
| Septic Patients | AA Genotype (n = 7) | AC Genotype (n = 3) | p-Value |
| male/female | 5/2 | 2/1 | 0.8803 |
| age years (±SD) | 59.1 (±21) | 65.3 (±9.7) | 0.6459 |
| Hb (g/dL (±SD)) day 1 | 10.9 (±1.5) | 10.1 (±2.3) | 0.5228 |
| leucocytes (×1000 µL (±SD)) | 14.06 (±6.7) | 4.0 (±3.1) | 0.0414 |
| thrombocytes (×1000 µL (±SD)) | 209.6 (±136.3) | 145.5 (±96.9) | 0.4544 |
| CRP (mg/dL (±SD)) | 13.2 (±12.6) | 12.17 (±6.4) | 0.8988 |
| PCT (ng/L (±SD)) | 3.1 (±4.7) | 1.4 (±1.9) | 0.5719 |
| temperature (°C (±SD)) | 37.7 (±1.5) | 37.0 (±1.5) | 0.5719 |
| noradrenaline (mg/h (±SD)) | 0.95 (±0.8) | 0.6 (±0.7) | 0.5318 |
| 30-day survival n (%) | 43% | 33% | 0.7782 |
| survival on ITS n (%) | 43% | 33% | 0.7782 |
| Gram-positive pathogen (%) | 28% | 67% | 0.2598 |
| Gram-negative pathogen detection n (%) | 14% | 0% | 0.4902 |
| no pathogen | 57% | 33% | 0.4902 |
| SOFA score day 1 | 8.4 (1.59) | 6.3 (2.49) | 0.1398 |
| Healthy Controls | AA Genotype (n = 6) | AC Genotype (n = 4) | p-Value |
| male/female | (2/4) | (2/2) | 0.999 |
| age years ((±SD) | 29.83 (±8.9) | 28.75 (±5.6) | 0.8365 |
| qSOFA | 0 | 0 |
| Septic Patients | Healthy Controls | p Value | |
|---|---|---|---|
| AA-genotype | 7 | 6 | 0.6392 |
| AC-genotype | 3 | 4 | |
| male | 3 | 4 | 0.9999 |
| female | 7 | 6 | |
| Alter (Jahre (±SD)) | 67.3 (±13) | 29.5 (±9.4) | <0.0001 |
| Oligonucleotide Name | Sequence | Annealing Temperature |
|---|---|---|
| AQP5_M_SE | CGTTTTCGTCGTATTTATTTTTTTC | 55° |
| AQP5_M_AS | CTCCTTCTTCATAATAACCGCGA | |
| AQP5_U_SE | TGTTTTTGTTGTATTTATTTTTTTT | 55° |
| AQP5_U_AS | CACCTCCTTCTTCATAATAACCACAA |
| Oligonucleotide Name | Sequence | |
|---|---|---|
| AQP5 forward | 5′-TCGGTTCAGCCCCGCTCACT-3′ | 60° |
| AQP5 reverse | 5′-GCCACACGCTCACTCAGGCT-3′ | |
| Actin forward | 5′-CTGGAACGGTGAAGGTGACA-3′ | 60° |
| Actin reverse | 5′-AAGGGACTTCCTGTAACAATGCA-3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rump, K.; Spellenberg, T.; von Busch, A.; Wolf, A.; Ziehe, D.; Thon, P.; Rahmel, T.; Adamzik, M.; Koos, B.; Unterberg, M. AQP5-1364A/C Polymorphism Affects AQP5 Promoter Methylation. Int. J. Mol. Sci. 2022, 23, 11813. https://doi.org/10.3390/ijms231911813
Rump K, Spellenberg T, von Busch A, Wolf A, Ziehe D, Thon P, Rahmel T, Adamzik M, Koos B, Unterberg M. AQP5-1364A/C Polymorphism Affects AQP5 Promoter Methylation. International Journal of Molecular Sciences. 2022; 23(19):11813. https://doi.org/10.3390/ijms231911813
Chicago/Turabian StyleRump, Katharina, Theresa Spellenberg, Alexander von Busch, Alexander Wolf, Dominik Ziehe, Patrick Thon, Tim Rahmel, Michael Adamzik, Björn Koos, and Matthias Unterberg. 2022. "AQP5-1364A/C Polymorphism Affects AQP5 Promoter Methylation" International Journal of Molecular Sciences 23, no. 19: 11813. https://doi.org/10.3390/ijms231911813
APA StyleRump, K., Spellenberg, T., von Busch, A., Wolf, A., Ziehe, D., Thon, P., Rahmel, T., Adamzik, M., Koos, B., & Unterberg, M. (2022). AQP5-1364A/C Polymorphism Affects AQP5 Promoter Methylation. International Journal of Molecular Sciences, 23(19), 11813. https://doi.org/10.3390/ijms231911813







