Augmented CPT1A Expression Is Associated with Proliferation and Colony Formation during Barrett’s Tumorigenesis
Abstract
1. Introduction
2. Results
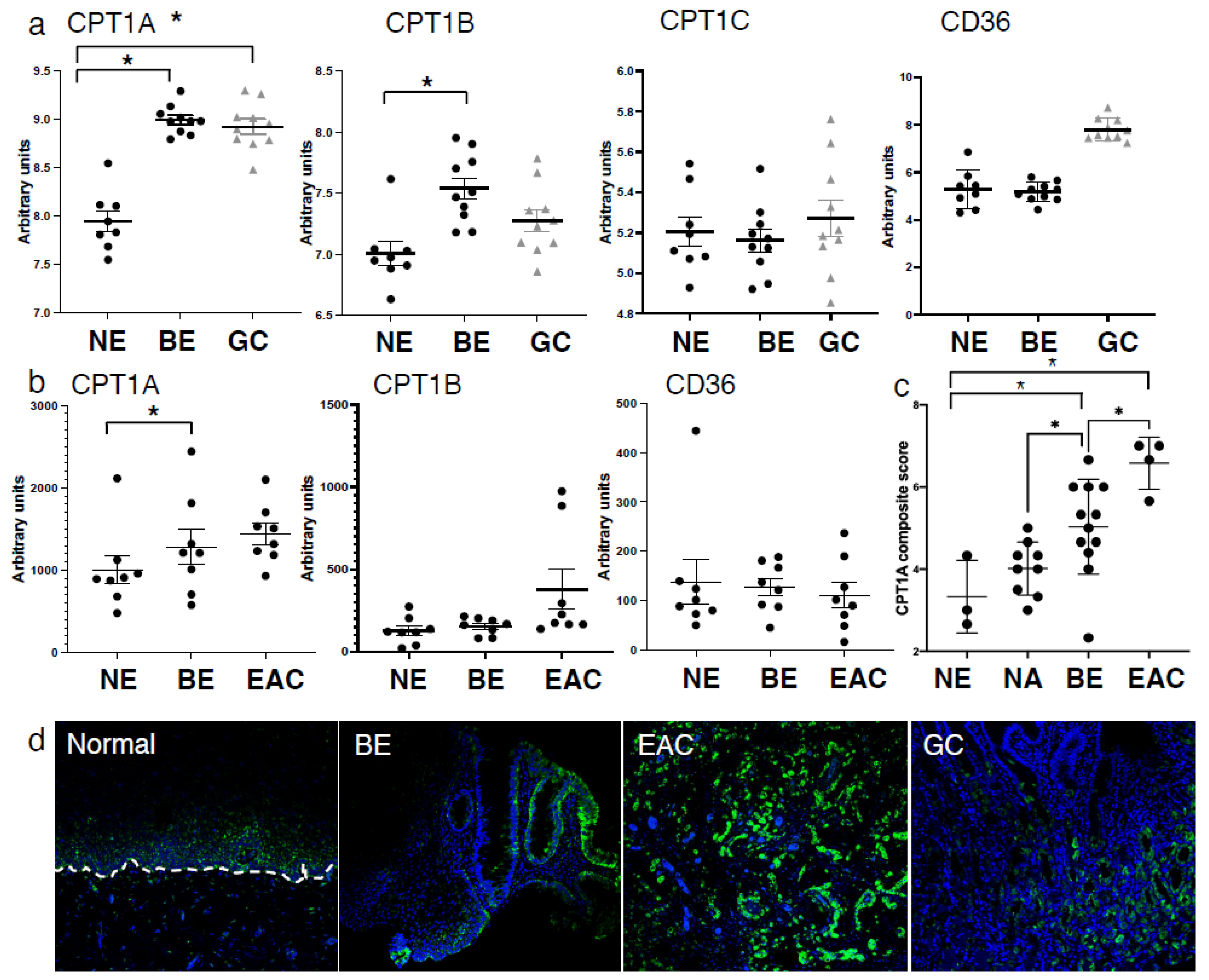
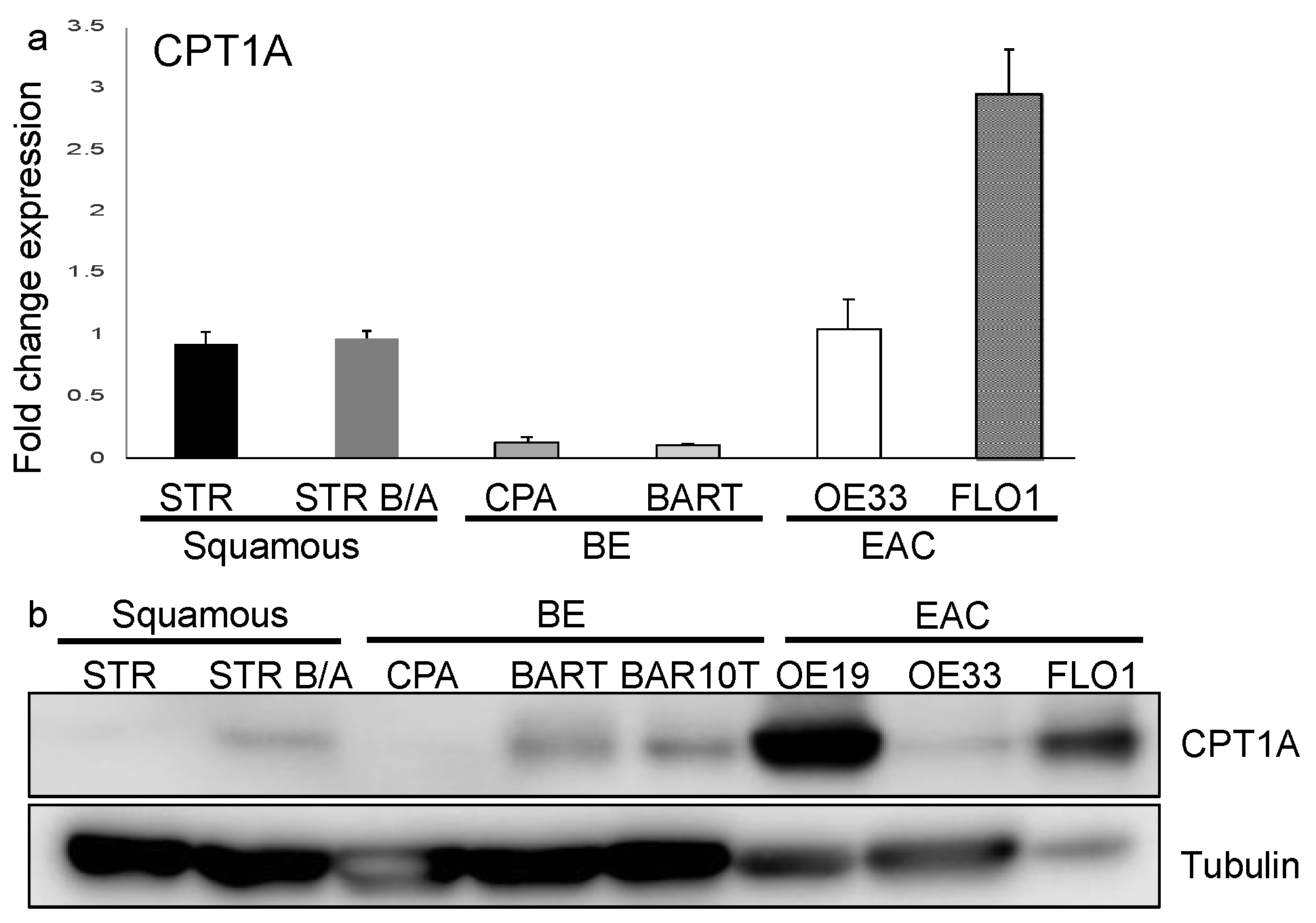
2.1. Expression of CPT1A Is the Predominant CPT1 Isoform Expressed in the Esophagus and Upregulated in Barrett’s Tumorigenesis
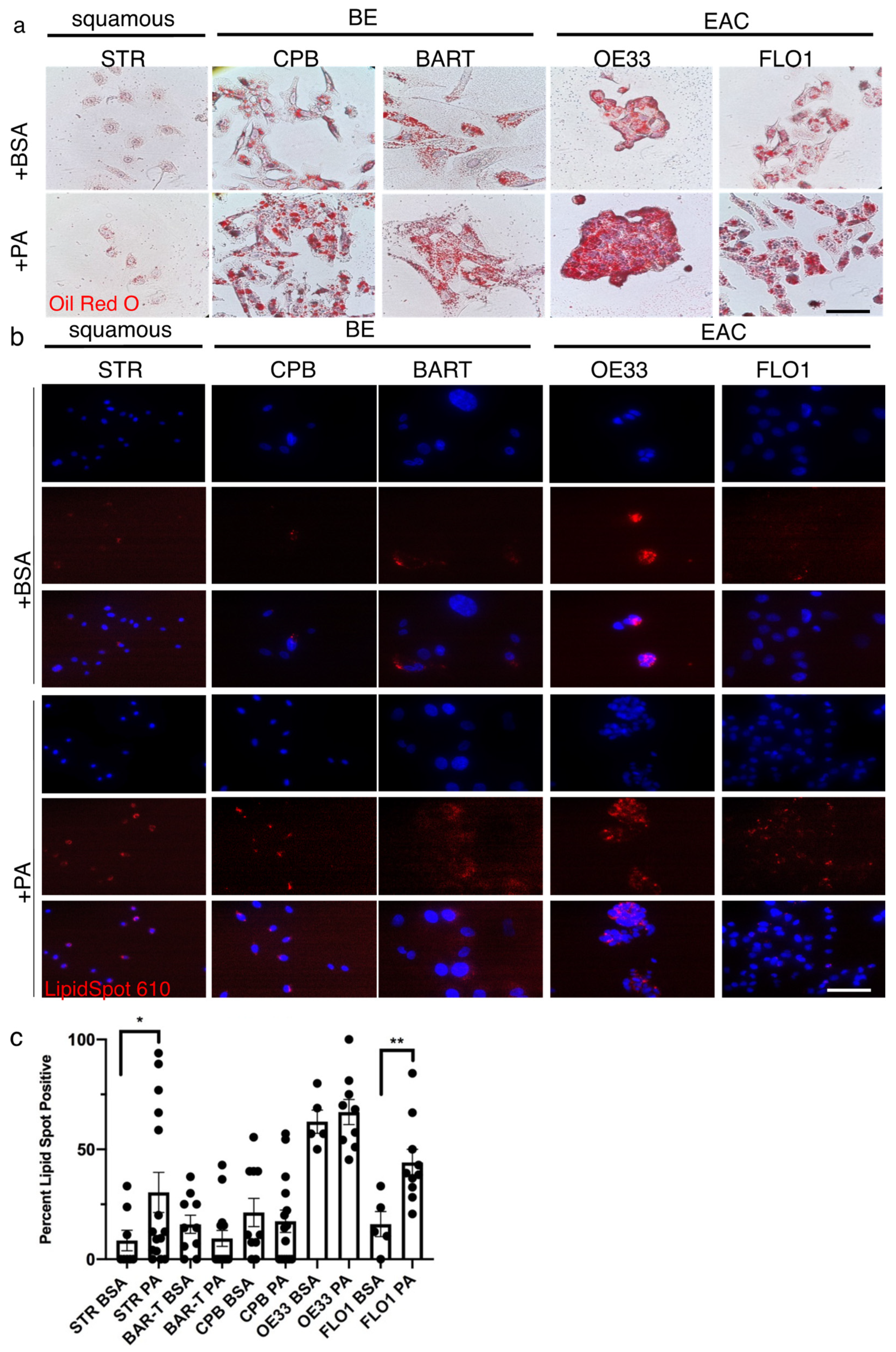
2.2. Esophageal Epithelial Cells Have the Capacity for Palmitic Acid Uptake and Incorporation into Mitochondria
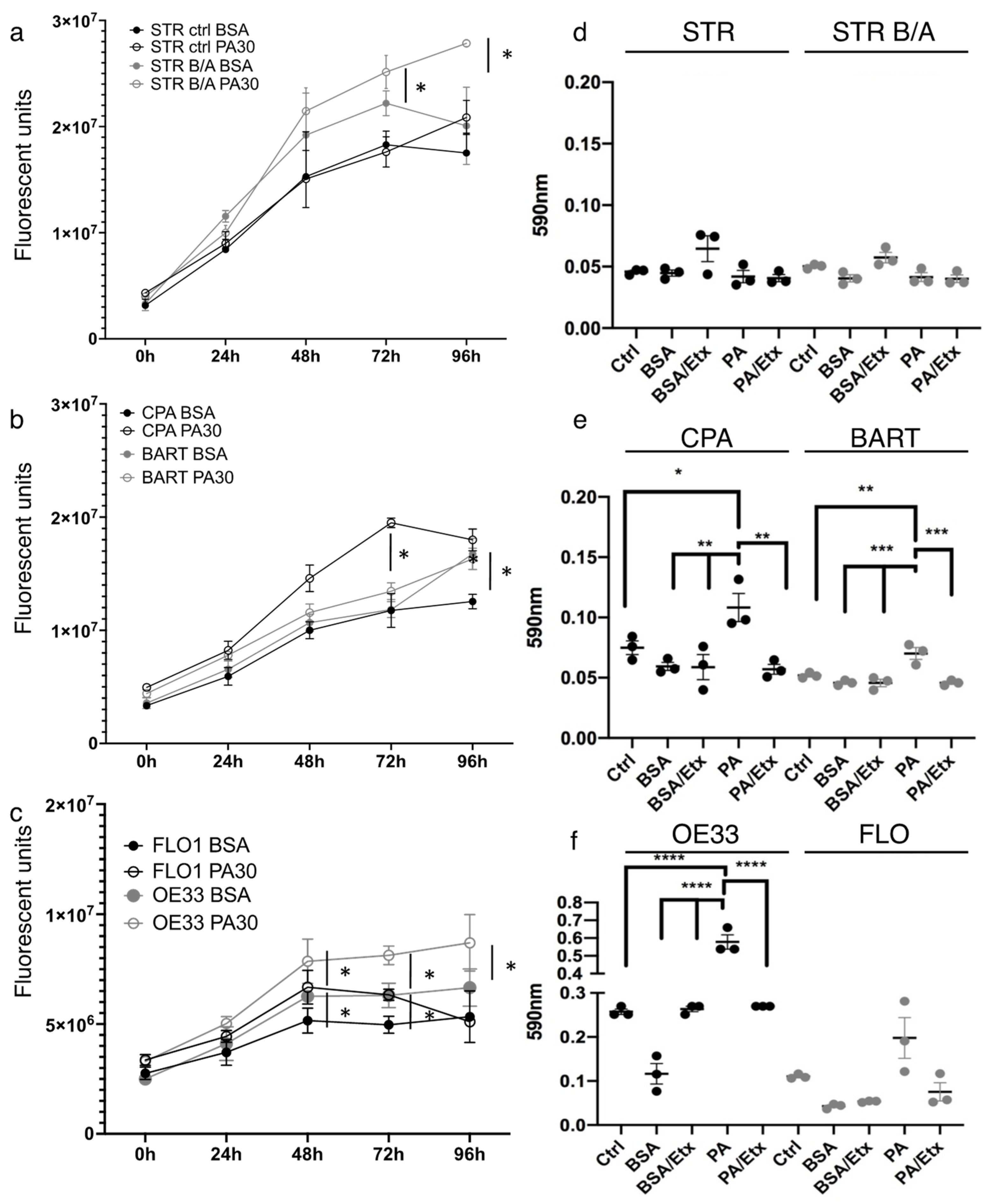
2.3. Palmitic Acid Promotes Esophageal Cell Proliferation and Colony Formation in BE and EAC Cell Lines
2.4. IL1β and IL8 Expression Is Induced by PA Stimulation in the Early Stages of BE Pathology
2.5. CPT1A Is Upregulated by a High Fat Diet in Mice and Correlates with an Increase in Ki67-Positive Cells in Inflamed Human BE and EAC
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Human Tissues
4.3. Animals
4.4. Histology
4.5. Conjugation of Palmitic Acid
4.6. Spheroid Cultures
4.7. Proliferation Assay
4.8. Colony Formation Assay
4.9. qRT-PCR
4.10. Western Blot
4.11. Immunofluorescence
4.12. Tyramide-Based Multiplex Immunofluorescence Staining
4.13. Lipid Detection Using Oil Red O Staining, BODIPY and LipoSpot 610
4.14. Biostatistical Analysis
4.15. Dataset Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- El-Serag, H.B.; Hashmi, A.; Garcia, J.; Richardson, P.; Alsarraj, A.; Fitzgerald, S.; Vela, M.; Shaib, Y.; Abraham, N.S.; Velez, M.; et al. Visceral abdominal obesity measured by CT scan is associated with an increased risk of Barrett’s oesophagus: A case-control study. Gut 2013, 63, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Hoyo, C.; Cook, M.B.; Kamangar, F.; Freedman, N.D.; Whiteman, D.; Bernstein, L.; Brown, L.M.; Risch, H.A.; Ye, W.; Sharp, L.; et al. Body mass index in relation to oesophageal and oesophagogastric junction adenocarcinomas: A pooled analysis from the International BEACON Consortium. Int. J. Epidemiol. 2012, 41, 1706–1718. [Google Scholar] [CrossRef]
- Malaty, H.M.; Fraley, J.K.; Abudayyeh, S.; Fairly, K.W.; Javed, U.S.; Aboul-Fotouh, H.; Mattek, N.; Gilger, M.A. Obesity and gastroesophageal reflux disease and gastroesophageal reflux symptoms in children. Clin. Exp. Gastroenterol. 2009, 2, 31–36. [Google Scholar] [CrossRef]
- Borodina, G.; Morozov, S. Children with Gastroesophageal Reflux Disease Consume More Calories and Fat Compared to Controls of Same Weight and Age. J. Pediatr. Gastroenterol. Nutr. 2020, 70, 808–814. [Google Scholar] [CrossRef]
- Delshad, S.D.; Almario, C.V.; Chey, W.D.; Spiegel, B.M. Prevalence of Gastroesophageal Reflux Disease and Proton Pump Inhibitor-Refractory Symptoms. Gastroenterology 2020, 158, 1250–1261. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, N.; Ransohoff, D.F. Gastroesophageal Reflux, Barrett Esophagus, and Esophageal Cancerr: Scientific Review. JAMA 2002, 287, 1972–1981. [Google Scholar] [CrossRef]
- Kambhampati, S.; Tieu, A.H.; Luber, B.; Wang, H.; Meltzer, S.J. Risk Factors for Progression of Barrett’s Esophagus to High Grade Dysplasia and Esophageal Adenocarcinoma. Sci. Rep. 2020, 10, 489. [Google Scholar] [CrossRef]
- Alexandre, L.; Long, E.; Beales, I.L. Pathophysiological mechanisms linking obesity and esophageal adenocarcinoma. World J. Gastrointest. Pathophysiol. 2014, 5, 534–549. [Google Scholar] [CrossRef]
- Schlottmann, F.; Dreifuss, N.H.; Patti, M.G. Obesity and esophageal cancer: GERD, Barrett’s esophagus, and molecular carcinogenic pathways. Expert Rev. Gastroenterol. Hepatol. 2020, 14, 425–433. [Google Scholar] [CrossRef]
- De Jonge, P.J.F.; Van Blankenstein, M.; Grady, W.M.; Kuipers, E.J. Barrett’s oesophagus: Epidemiology, cancer risk and implications for management. Gut 2013, 63, 191–202. [Google Scholar] [CrossRef]
- Valezi, A.C.; Herbella, F.A.; Schlottmann, F.; Patti, M.G. Gastroesophageal Reflux Disease in Obese Patients. J. Laparoendosc. Adv. Surg. Tech. 2018, 28, 949–952. [Google Scholar] [CrossRef]
- Molendijk, J.; Nguyen, T.-M.; Brown, I.; Mohamed, A.; Lim, Y.; Barclay, J.; Hodson, M.P.; Hennessy, T.P.; Krause, L.; Morrison, M.; et al. Chronic High-Fat Diet Induces Early Barrett’s Esophagus in Mice through Lipidome Remodeling. Biomolecules 2020, 10, 776. [Google Scholar] [CrossRef]
- Münch, N.S.; Fang, H.-Y.; Ingermann, J.; Maurer, H.C.; Anand, A.; Kellner, V.; Sahm, V.; Wiethaler, M.; Baumeister, T.; Wein, F.; et al. High-Fat Diet Accelerates Carcinogenesis in a Mouse Model of Barrett’s Esophagus via Interleukin 8 and Alterations to the Gut Microbiome. Gastroenterology 2019, 157, 492–506.e2. [Google Scholar] [CrossRef]
- Zhang, H.Y.; Zhang, Q.; Zhang, X.; Yu, C.; Huo, X.; Cheng, E.; Wang, D.H.; Spechler, S.J.; Souza, R.F. Cancer-related inflammation and Barrett’s carcinogenesis: Interleukin-6 and STAT3 mediate apoptotic resistance in transformed Barrett’s cells. Am. J. Physiol. Liver Physiol. 2011, 300, G454–G460. [Google Scholar] [CrossRef]
- Poehlmann, A.; Kuester, D.; Malfertheiner, P.; Guenther, T.; Roessner, A. Inflammation and Barrett’s carcinogenesis. Pathol. Res. Pract. 2012, 208, 269–280. [Google Scholar] [CrossRef]
- Souza, R.F.; Huo, X.; Mittal, V.; Schuler, C.M.; Carmack, S.W.; Zhang, H.Y.; Zhang, X.; Yu, C.; Hormi–Carver, K.; Genta, R.M.; et al. Gastroesophageal Reflux Might Cause Esophagitis Through a Cytokine-Mediated Mechanism Rather than Caustic Acid Injury. Gastroenterology 2009, 137, 1776–1784. [Google Scholar] [CrossRef]
- Huo, X.; Dunbar, K.B.; Zhang, X.; Zhang, Q.; Spechler, S.J.; Souza, R.F. In Barrett’s epithelial cells, weakly acidic bile salt solutions cause oxidative DNA damage with response and repair mediated by p38. Am. J. Physiol. Liver Physiol. 2020, 318, G464–G478. [Google Scholar] [CrossRef]
- Han, D.; Zhang, C. The Oxidative Damage and Inflammation Mechanisms in GERD-Induced Barrett’s Esophagus. Front. Cell Dev. Biol. 2022, 10, 885537. [Google Scholar] [CrossRef]
- Peng, D.; Zaika, A.; Que, J.; El-Rifai, W. The antioxidant response in Barrett’s tumorigenesis: A double-edged sword. Redox Biol. 2021, 41, 101894. [Google Scholar] [CrossRef]
- Koundouros, N.; Poulogiannis, G. Reprogramming of fatty acid metabolism in cancer. Br. J. Cancer 2019, 122, 4–22. [Google Scholar] [CrossRef]
- Carracedo, A.; Cantley, L.C.; Pandolfi, P.P. Cancer metabolism: Fatty acid oxidation in the limelight. Nat. Rev. Cancer 2013, 13, 227–232. [Google Scholar] [CrossRef]
- Rufer, A.C.; Thoma, R.; Hennig, M. Structural insight into function and regulation of carnitine palmitoyltransferase. Cell Mol. Life Sci. 2009, 66, 2489–2501. [Google Scholar] [CrossRef]
- Schlaepfer, I.R.; Joshi, M. CPT1A-mediated Fat Oxidation, Mechanisms, and Therapeutic Potential. Endocrinology 2020, 161, bqz046. [Google Scholar] [CrossRef]
- Qu, Q.; Zeng, F.; Liu, X.; Wang, Q.J.; Deng, F. Fatty acid oxidation and carnitine palmitoyltransferase I: Emerging therapeutic targets in cancer. Cell Death Dis. 2016, 7, e2226. [Google Scholar] [CrossRef]
- Gatza, M.L.; Silva, G.O.; Parker, J.S.; Fan, C.; Perou, C.M. An integrated genomics approach identifies drivers of proliferation in luminal-subtype human breast cancer. Nat. Genet. 2014, 46, 1051–1059. [Google Scholar] [CrossRef]
- Pucci, S.; Zonetti, M.J.; Fisco, T.; Polidoro, C.; Bocchinfuso, G.; Palleschi, A.; Novelli, G.; Spagnoli, L.G.; Mazzarelli, P. Carnitine palmitoyl transferase-1A (CPT1A): A new tumor specific target in human breast cancer. Oncotarget 2016, 7, 19982–19996. [Google Scholar] [CrossRef]
- Wang, Y.-N.; Zeng, Z.-L.; Lu, J.; Wang, Y.; Liu, Z.-X.; He, M.-M.; Zhao, Q.; Wang, Z.-X.; Li, T.; Lu, Y.-X.; et al. CPT1A-mediated fatty acid oxidation promotes colorectal cancer cell metastasis by inhibiting anoikis. Oncogene 2018, 37, 6025–6040. [Google Scholar] [CrossRef]
- Xiong, Y.; Liu, Z.; Zhao, X.; Ruan, S.; Zhang, X.; Wang, S.; Huang, T. CPT1A regulates breast cancer-associated lymphangiogenesis via VEGF signaling. Biomed. Pharmacother. 2018, 106, 1–7. [Google Scholar] [CrossRef]
- Mihaylova, M.M.; Cheng, C.-W.; Cao, A.Q.; Tripathi, S.; Mana, M.D.; Bauer-Rowe, K.E.; Abu-Remaileh, M.; Clavain, L.; Erdemir, A.; Lewis, C.A.; et al. Fasting Activates Fatty Acid Oxidation to Enhance Intestinal Stem Cell Function during Homeostasis and Aging. Cell Stem Cell 2018, 22, 769–778.e4. [Google Scholar] [CrossRef]
- Barisas, D.A.; Stappenbeck, T.S. Intestinal Stem Cells Live off the Fat of the Land. Cell Stem Cell 2018, 22, 611–612. [Google Scholar] [CrossRef]
- Arber, N.; Lightdale, C.; Rotterdam, H.; Han, K.H.; Sgambato, A.; Yap, E.; Ahsan, H.; Finegold, J.; Stevens, P.D.; Green, P.H.; et al. Increased expression of the cyclin D1 gene in Barrett’s esophagus. Cancer Epidemiol. Biomark. Prev. 1996, 5, 457–459. [Google Scholar]
- Bani-Hani, K.; Martin, I.G.; Hardie, L.J.; Mapstone, N.; Briggs, J.A.; Forman, D.; Wild, C.P. Prospective Study of Cyclin D1 Overexpression in Barrett’s Esophagus: Association with Increased Risk of Adenocarcinoma. JNCI J. Natl. Cancer Inst. 2000, 92, 1316–1321. [Google Scholar] [CrossRef]
- Shi, Z.-Z.; Liang, J.-W.; Zhan, T.; Wang, B.-S.; Lin, D.-C.; Liu, S.-G.; Hao, J.-J.; Yang, H.; Zhang, Y.; Zhan, Q.-M.; et al. Genomic alterations with impact on survival in esophageal squamous cell carcinoma identified by array comparative genomic hybridization. Genes Chromosom. Cancer 2011, 50, 518–526. [Google Scholar] [CrossRef]
- Roudebush, C.; Catala-Valentin, A.; Andl, T.; Le Bras, G.F.; Andl, C.D. Activin A-mediated epithelial de-differentiation contributes to injury repair in an in vitro gastrointestinal reflux model. Cytokine 2019, 123, 154782. [Google Scholar] [CrossRef]
- Jaiswal, K.R.; Morales, C.P.; Feagins, L.A.; Gandia, K.G.; Zhang, X.; Zhang, H.-Y.; Hormi-Carver, K.; Shen, Y.; Elder, F.; Ramirez, R.D.; et al. Characterization of telomerase-immortalized, non-neoplastic, human Barrett’s cell line (BAR-T). Dis. Esophagus 2007, 20, 256–264. [Google Scholar] [CrossRef]
- Hegardt, F.G.; Serra, D.; Asins, G. Influence of etomoxir on the expression of several genes in liver, testis and heart. Gen. Pharmacol. Vasc. Syst. 1995, 26, 897–904. [Google Scholar] [CrossRef]
- Bristow, M. Etomoxir: A new approach to treatment of chronic heart failure. Lancet 2000, 356, 1621–1622. [Google Scholar] [CrossRef]
- Björntorp, P.; Bergman, H.; Varnauskas, E. Plasma free fatty acid turnover rate in obesity. Acta Med. Scand. 2009, 185, 351–356. [Google Scholar] [CrossRef]
- Jensen, M.D. Adipose Tissue Metabolism—An Aspect We Should not Neglect? Horm. Metab. Res. 2007, 39, 722–725. [Google Scholar] [CrossRef]
- Boden, G.; Lebed, B.; Schatz, M.; Homko, C.; Lemieux, S. Effects of Acute Changes of Plasma Free Fatty Acids on Intramyocellular Fat Content and Insulin Resistance in Healthy Subjects. Diabetes 2001, 50, 1612–1617. [Google Scholar] [CrossRef]
- Bi, X.; Yeo, P.L.Q.; Loo, Y.T.; Henry, C.J. Associations between circulating fatty acid levels and metabolic risk factors. J. Nutr. Intermed. Metab. 2019, 15, 65–69. [Google Scholar] [CrossRef]
- Carta, G.; Murru, E.; Banni, S.; Manca, C. Palmitic Acid: Physiological Role, Metabolism and Nutritional Implications. Front. Physiol. 2017, 8, 902. [Google Scholar] [CrossRef]
- Gong, J.; Zhao, H.; Liu, T.; Li, L.; Cheng, E.; Zhi, S.; Kong, L.; Yao, H.-W.; Li, J. Cigarette Smoke Reduces Fatty Acid Catabolism, Leading to Apoptosis in Lung Endothelial Cells: Implication for Pathogenesis of COPD. Front. Pharmacol. 2019, 10, 941. [Google Scholar] [CrossRef] [PubMed]
- Febbraio, M.A. Role of interleukins in obesity: Implications for metabolic disease. Trends Endocrinol. Metab. 2014, 25, 312–319. [Google Scholar] [CrossRef] [PubMed]
- O’Riordan, J.M.; Abdel-Latif, M.; Ravi, N.; McNamara, D.; Byrne, P.J.; McDonald, G.S.A.; Keeling, P.W.N.; Kelleher, D.; Reynolds, J.V. Proinflammatory Cytokine and Nuclear Factor Kappa-B Expression along the Inflammation-Metaplasia-Dysplasia-Adenocarcinoma Sequence in the Esophagus. Am. J. Gastroenterol. 2005, 100, 1257–1264. [Google Scholar] [CrossRef] [PubMed]
- Whorton, J.; Sureban, S.M.; May, R.; Qu, D.; Lightfoot, S.A.; Madhoun, M.; Johnson, M.; Tierney, W.M.; Maple, J.T.; Vega, K.J.; et al. DCLK1 Is Detectable in Plasma of Patients with Barrett’s Esophagus and Esophageal Adenocarcinoma. Am. J. Dig. Dis. 2014, 60, 509–513. [Google Scholar] [CrossRef]
- Christman, E.M.; Chandrakesan, P.; Weygant, N.; Maple, J.T.; Tierney, W.M.; Vega, K.J.; Houchen, C.W. Elevated doublecortin-like kinase 1 serum levels revert to baseline after therapy in early stage esophageal adenocarcinoma. Biomark. Res. 2019, 7, 5. [Google Scholar] [CrossRef]
- Mohammed, A.; Janakiram, N.B.; Madka, V.; Brewer, M.; Ritchie, R.L.; Lightfoot, S.; Kumar, G.; Sadeghi, M.; Patlolla, J.M.R.; Yamada, H.Y.; et al. Targeting pancreatitis blocks tumor-initiating stem cells and pancreatic cancer progression. Oncotarget 2015, 6, 15524–15539. [Google Scholar] [CrossRef] [PubMed]
- Quante, M.; Bhagat, G.; Abrams, J.A.; Marache, F.; Good, P.; Lee, M.D.; Lee, Y.; Friedman, R.; Asfaha, S.; Dubeykovskaya, Z.; et al. Bile Acid and Inflammation Activate Gastric Cardia Stem Cells in a Mouse Model of Barrett-Like Metaplasia. Cancer Cell 2012, 21, 36–51. [Google Scholar] [CrossRef]
- Vazquez, A.; Kamphorst, J.J.; Markert, E.K.; Schug, Z.T.; Tardito, S.; Gottlieb, E. Cancer metabolism at a glance. J. Cell Sci. 2016, 129, 3367–3373. [Google Scholar] [CrossRef] [PubMed]
- Thrift, A.P.; Shaheen, N.J.; Gammon, M.D.; Bernstein, L.; Reid, B.J.; Onstad, L.; Risch, H.A.; Liu, G.; Bird, N.C.; Wu, A.H.; et al. Obesity and Risk of Esophageal Adenocarcinoma and Barrett’s Esophagus: A Mendelian Randomization Study. JNCI J. Natl. Cancer Inst. 2014, 106, dju252. [Google Scholar] [CrossRef]
- Long, E.; Beales, I.L. The role of obesity in oesophageal cancer development. Ther. Adv. Gastroenterol. 2014, 7, 247–268. [Google Scholar] [CrossRef] [PubMed]
- Corley, D.A.; Kubo, A.; Levin, T.R.; Block, G.; Habel, L.; Zhao, W.; Leighton, P.; Quesenberry, C.; Rumore, G.J.; Buffler, P.A. Abdominal Obesity and Body Mass Index as Risk Factors for Barrett’s Esophagus. Gastroenterology 2007, 133, 34–41. [Google Scholar] [CrossRef]
- Progatzky, F.; Sangha, N.J.; Yoshida, N.; McBrien, M.; Cheung, J.; Shia, A.; Scott, J.; Marchesi, J.; Lamb, J.R.; Bugeon, L.; et al. Dietary cholesterol directly induces acute inflammasome-dependent intestinal inflammation. Nat. Commun. 2014, 5, 5864. [Google Scholar] [CrossRef] [PubMed]
- Reddy, B.S.; Mangat, S.; Sheinfil, A.; Weisburger, J.H.; Wynder, E.L. Effect of type and amount of dietary fat and 1,2-dimethylhydrazine on biliary bile acids, fecal bile acids, and neutral sterols in rats. Cancer Res. 1977, 37, 2132–2137. [Google Scholar]
- Zheng, X.; Huang, F.; Zhao, A.; Lei, S.; Zhang, Y.; Xie, G.; Chen, T.; Qu, C.; Rajani, C.; Dong, B.; et al. Bile acid is a significant host factor shaping the gut microbiome of diet-induced obese mice. BMC Biol. 2017, 15, 120. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Niu, C.; Li, Y.; Gao, B.; Zheng, J.; Guo, X.; Ma, W. Fatty acid synthase expression and esophageal cancer. Mol. Biol. Rep. 2012, 39, 9733–9739. [Google Scholar] [CrossRef] [PubMed]
- Orita, H.; Coulter, J.; Tully, E.; Abe, M.; Montgomery, E.; Alvarez, H.; Sato, K.; Hino, O.; Kajiyama, Y.; Tsurumaru, M.; et al. High levels of fatty acid synthase expression in esophageal cancers represent a potential target for therapy. Cancer Biol. Ther. 2010, 10, 549–554. [Google Scholar] [CrossRef]
- Menendez, J.; Lupu, R. Fatty acid synthase and the lipogenic phenotype in cancer pathogenesis. Nat. Cancer 2007, 7, 763–777. [Google Scholar] [CrossRef] [PubMed]
- Pandey, P.R.; Xing, F.; Sharma, S.; Watabe, M.; Pai, S.K.; Iiizumi-Gairani, M.; Fukuda, K.; Hirota, S.; Mo, Y.-Y.; Watabe, K. Elevated lipogenesis in epithelial stem-like cell confers survival advantage in ductal carcinoma in situ of breast cancer. Oncogene 2012, 32, 5111–5122. [Google Scholar] [CrossRef]
- Tanosaki, S.; Tohyama, S.; Fujita, J.; Someya, S.; Hishiki, T.; Matsuura, T.; Nakanishi, H.; Ohto-Nakanishi, T.; Akiyama, T.; Morita, Y.; et al. Fatty Acid Synthesis Is Indispensable for Survival of Human Pluripotent Stem Cells. iScience 2020, 23, 101535. [Google Scholar] [CrossRef] [PubMed]
- Jariwala, N.; A Mehta, G.; Bhatt, V.; Hussein, S.; Parker, K.A.; Yunus, N.; Parker, J.S.; Guo, J.Y.; Gatza, M.L. CPT1A and fatty acid β-oxidation are essential for tumor cell growth and survival in hormone receptor-positive breast cancer. NAR Cancer 2021, 3. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Dong, X.; Xiao, L.; Tan, Z.; Luo, X.; Yang, L.; Li, W.; Shi, F.; Li, Y.; Zhao, L.; et al. CPT1A-mediated fatty acid oxidation promotes cell proliferation via nucleoside metabolism in nasopharyngeal carcinoma. Cell Death Dis. 2022, 13, 331. [Google Scholar] [CrossRef]
- Jammula, S.; Katz-Summercorn, A.C.; Li, X.; Linossi, C.; Smyth, E.; Killcoyne, S.; Biasci, D.; Subash, V.V.; Abbas, S.; Blasko, A.; et al. Identification of Subtypes of Barrett’s Esophagus and Esophageal Adenocarcinoma Based on DNA Methylation Profiles and Integration of Transcriptome and Genome Data. Gastroenterology 2020, 158, 1682–1697. [Google Scholar] [CrossRef] [PubMed]
- Fabisiak, A.; Bartoszek, A.; Talar, M.; Binienda, A.; Dziedziczak, K.; Krajewska, J.; Mosińska, P.; Niewinna, K.; Tarasiuk, A.; Mokrowiecka, A.; et al. Expression of FFAR3 and FFAR4 Is Increased in Gastroesophageal Reflux Disease. J. Clin. Med. 2020, 9, 4111. [Google Scholar] [CrossRef]
- Zhou, B.-R.; Zhang, J.-A.; Zhang, Q.; Permatasari, F.; Xu, Y.; Wu, D.; Yin, Z.-Q.; Luo, D. Palmitic Acid Induces Production of Proinflammatory Cytokines Interleukin-6, Interleukin-1β, and Tumor Necrosis Factor-α via a NF-κB-Dependent Mechanism in HaCaT Keratinocytes. Mediat. Inflamm. 2013, 2013, 530429. [Google Scholar] [CrossRef] [PubMed]
- Kong, J.; Sai, H.; Crissey, M.A.S.; Jhala, N.; Falk, G.W.; Ginsberg, G.G.; Abrams, J.A.; Nakagawa, H.; Wang, K.; Rustgi, A.K.; et al. Immature myeloid progenitors promote disease progression in a mouse model of Barrett’s-like metaplasia. Oncotarget 2015, 6, 32980–33005. [Google Scholar] [CrossRef] [PubMed]
- Usui, G.; Shinozaki, T.; Jinno, T.; Fujibayashi, K.; Morikawa, T.; Gunji, T.; Matsuhashi, N. Association between visceral abdominal obesity and long-segment Barrett’s esophagus in a Japanese population. J. Gastroenterol. 2019, 55, 189–197. [Google Scholar] [CrossRef]
- Westphalen, C.B.; Quante, M.; Wang, T.C. Functional implication of Dclk1 and Dclk1-expressing cells in cancer. Small GTPases 2016, 8, 164–171. [Google Scholar] [CrossRef]
- Middelhoff, M.; Westphalen, C.B.; Hayakawa, Y.; Yan, K.S.; Gershon, M.D.; Wang, T.C.; Quante, M. Dclk1-expressing tuft cells: Critical modulators of the intestinal niche? Am. J. Physiol. Liver Physiol. 2017, 313, G285–G299. [Google Scholar] [CrossRef] [PubMed]
- Saqui-Salces, M.; Keeley, T.M.; Grosse, A.S.; Qiao, X.T.; El-Zaatari, M.; Gumucio, D.L.; Samuelson, L.C.; Merchant, J.L. Gastric tuft cells express DCLK1 and are expanded in hyperplasia. Histochem. Cell Biol. 2011, 136, 191–204. [Google Scholar] [CrossRef]
- Gerbe, F.; Sidot, E.; Smyth, D.J.; Ohmoto, M.; Matsumoto, I.; Dardalhon, V.; Cesses, P.; Garnier, L.; Pouzolles, M.; Brulin, B.; et al. Intestinal epithelial tuft cells initiate type 2 mucosal immunity to helminth parasites. Nature 2016, 529, 226–230. [Google Scholar] [CrossRef]
- Han, S.; Wei, R.; Zhang, X.; Jiang, N.; Fan, M.; Huang, J.H.; Xie, B.; Zhang, L.; Miao, W.; Butler, A.C.-P.; et al. CPT1A/2-Mediated FAO Enhancement—A Metabolic Target in Radioresistant Breast Cancer. Front. Oncol. 2019, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.; Xiao, L.; Tang, M.; Bai, F.; Li, J.; Li, L.; Shi, F.; Li, N.; Li, Y.; Du, Q.; et al. Targeting CPT1A-mediated fatty acid oxidation sensitizes nasopharyngeal carcinoma to radiation therapy. Theranostics 2018, 8, 2329–2347. [Google Scholar] [CrossRef] [PubMed]
- Kamphorst, J.J.; Cross, J.R.; Fan, J.; de Stanchina, E.; Mathew, R.; White, E.P.; Thompson, C.B.; Rabinowitz, J.D. Hypoxic and Ras-transformed cells support growth by scavenging unsaturated fatty acids from lysophospholipids. Proc. Natl. Acad. Sci. USA 2013, 110, 8882–8887. [Google Scholar] [CrossRef] [PubMed]
- Mana, M.D.; Hussey, A.M.; Tzouanas, C.N.; Imada, S.; Millan, Y.B.; Bahceci, D.; Saiz, D.R.; Webb, A.T.; Lewis, C.A.; Carmeliet, P.; et al. High-fat diet-activated fatty acid oxidation mediates intestinal stemness and tumorigenicity. Cell Rep. 2021, 35, 109212. [Google Scholar] [CrossRef]
- Barrera, G.; Toaldo, C.; Pizzimenti, S.; Cerbone, A.; Pettazzoni, P.; Dianzani, M.U.; Ferretti, C. The Role of PPAR Ligands in Controlling Growth-Related Gene Expression and their Interaction with Lipoperoxidation Products. PPAR Res. 2008, 2008, 524671. [Google Scholar] [CrossRef]
- Beyaz, S.; Mana, M.D.; Roper, J.; Kedrin, D.; Saadatpour, A.; Hong, S.-J.; Bauer-Rowe, K.E.; Xifaras, M.E.; Akkad, A.; Arias, E.; et al. High-fat diet enhances stemness and tumorigenicity of intestinal progenitors. Nature 2016, 531, 53–58. [Google Scholar] [CrossRef]
- Li, S.; Lu, C.-W.; Diem, E.C.; Li, W.; Guderian, M.; Lindenberg, M.; Kruse, F.; Buettner, M.; Floess, S.; Winny, M.R.; et al. Acetyl-CoA-Carboxylase 1-mediated de novo fatty acid synthesis sustains Lgr5+ intestinal stem cell function. Nat. Commun. 2022, 13, 3998. [Google Scholar] [CrossRef]
- Bernal, C.; Araya, C.; Palma, V.; Bronfman, M. PPARβ/δ and PPARγ maintain undifferentiated phenotypes of mouse adult neural precursor cells from the subventricular zone. Front. Cell. Neurosci. 2015, 9, 78. [Google Scholar] [CrossRef]
- Liu, K.; Lin, B.; Zhao, M.; Yang, X.; Chen, M.; Gao, A.; Liu, F.; Que, J.; Lan, X. The multiple roles for Sox2 in stem cell maintenance and tumorigenesis. Cell. Signal. 2013, 25, 1264–1271. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Jiang, M.; Lu, Y.; Chen, H.; Sun, J.; Wu, S.; Ku, W.-Y.; Nakagawa, H.; Kita, Y.; Natsugoe, S.; et al. Sox2 Cooperates with Inflammation-Mediated Stat3 Activation in the Malignant Transformation of Foregut Basal Progenitor Cells. Cell Stem Cell 2013, 12, 304–315. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Zhou, B.; Yang, Q.; Pan, Y.; Yang, W.; Freedland, S.J.; Ding, L.-W.; Freeman, M.R.; Breunig, J.J.; Bhowmick, N.A.; et al. A Transcriptional Regulatory Loop of Master Regulator Transcription Factors, PPARG, and Fatty Acid Synthesis Promotes Esophageal Adenocarcinoma. Cancer Res. 2021, 81, 1216–1229. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bernard, J.N.; Chinnaiyan, V.; Andl, T.; Le Bras, G.F.; Qureshi, M.N.; Altomare, D.A.; Andl, C.D. Augmented CPT1A Expression Is Associated with Proliferation and Colony Formation during Barrett’s Tumorigenesis. Int. J. Mol. Sci. 2022, 23, 11745. https://doi.org/10.3390/ijms231911745
Bernard JN, Chinnaiyan V, Andl T, Le Bras GF, Qureshi MN, Altomare DA, Andl CD. Augmented CPT1A Expression Is Associated with Proliferation and Colony Formation during Barrett’s Tumorigenesis. International Journal of Molecular Sciences. 2022; 23(19):11745. https://doi.org/10.3390/ijms231911745
Chicago/Turabian StyleBernard, Joshua N., Vikram Chinnaiyan, Thomas Andl, Gregoire F. Le Bras, M. Nasar Qureshi, Deborah A. Altomare, and Claudia D. Andl. 2022. "Augmented CPT1A Expression Is Associated with Proliferation and Colony Formation during Barrett’s Tumorigenesis" International Journal of Molecular Sciences 23, no. 19: 11745. https://doi.org/10.3390/ijms231911745
APA StyleBernard, J. N., Chinnaiyan, V., Andl, T., Le Bras, G. F., Qureshi, M. N., Altomare, D. A., & Andl, C. D. (2022). Augmented CPT1A Expression Is Associated with Proliferation and Colony Formation during Barrett’s Tumorigenesis. International Journal of Molecular Sciences, 23(19), 11745. https://doi.org/10.3390/ijms231911745








