Differential Effect of Dopamine D4 Receptor Activation on Low-Frequency Oscillations in the Prefrontal Cortex and Hippocampus May Bias the Bidirectional Prefrontal–Hippocampal Coupling
Abstract
1. Introduction
2. Results
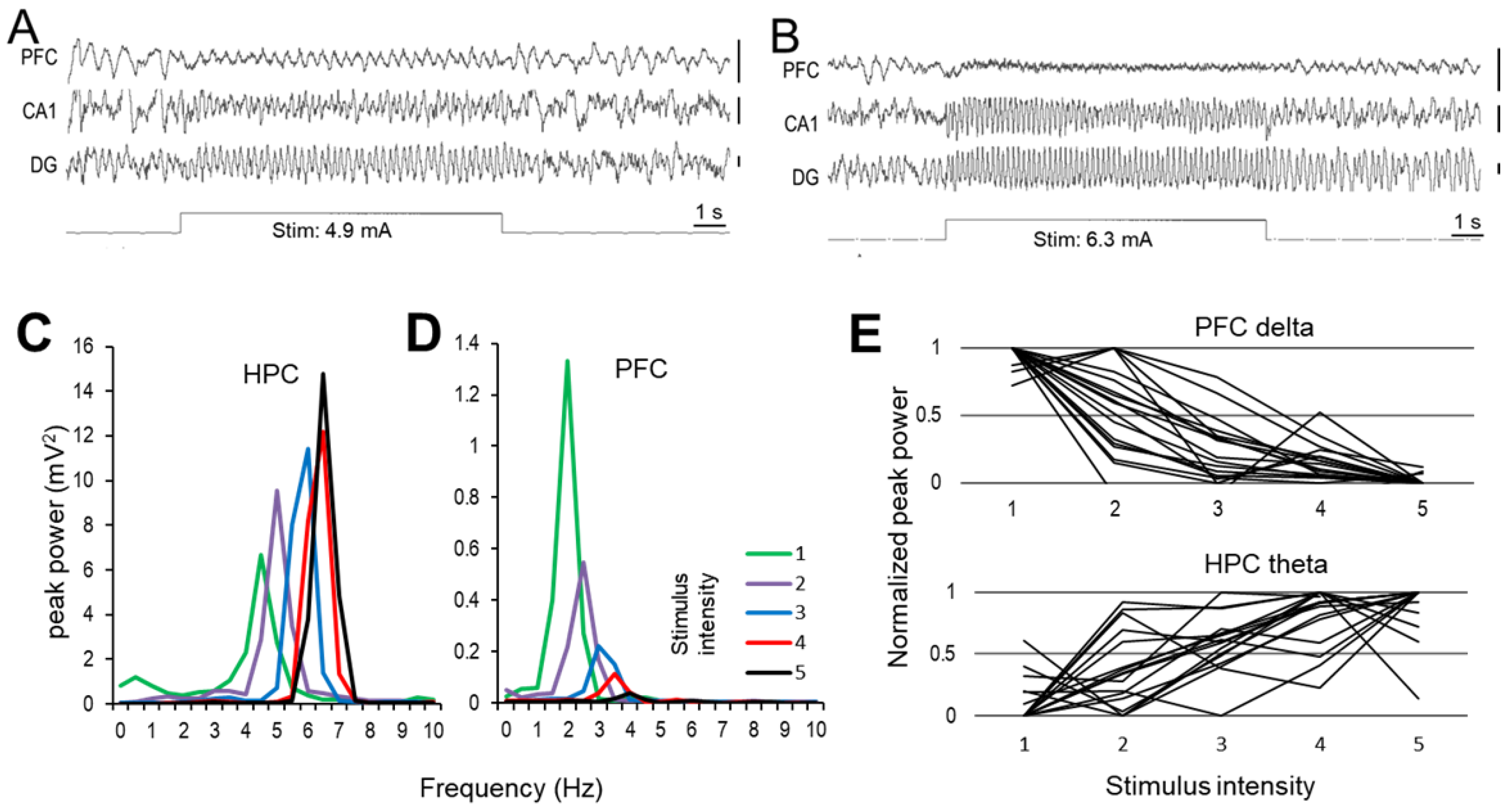
2.1. Effect of RPO Stimulation on Forebrain Oscillations
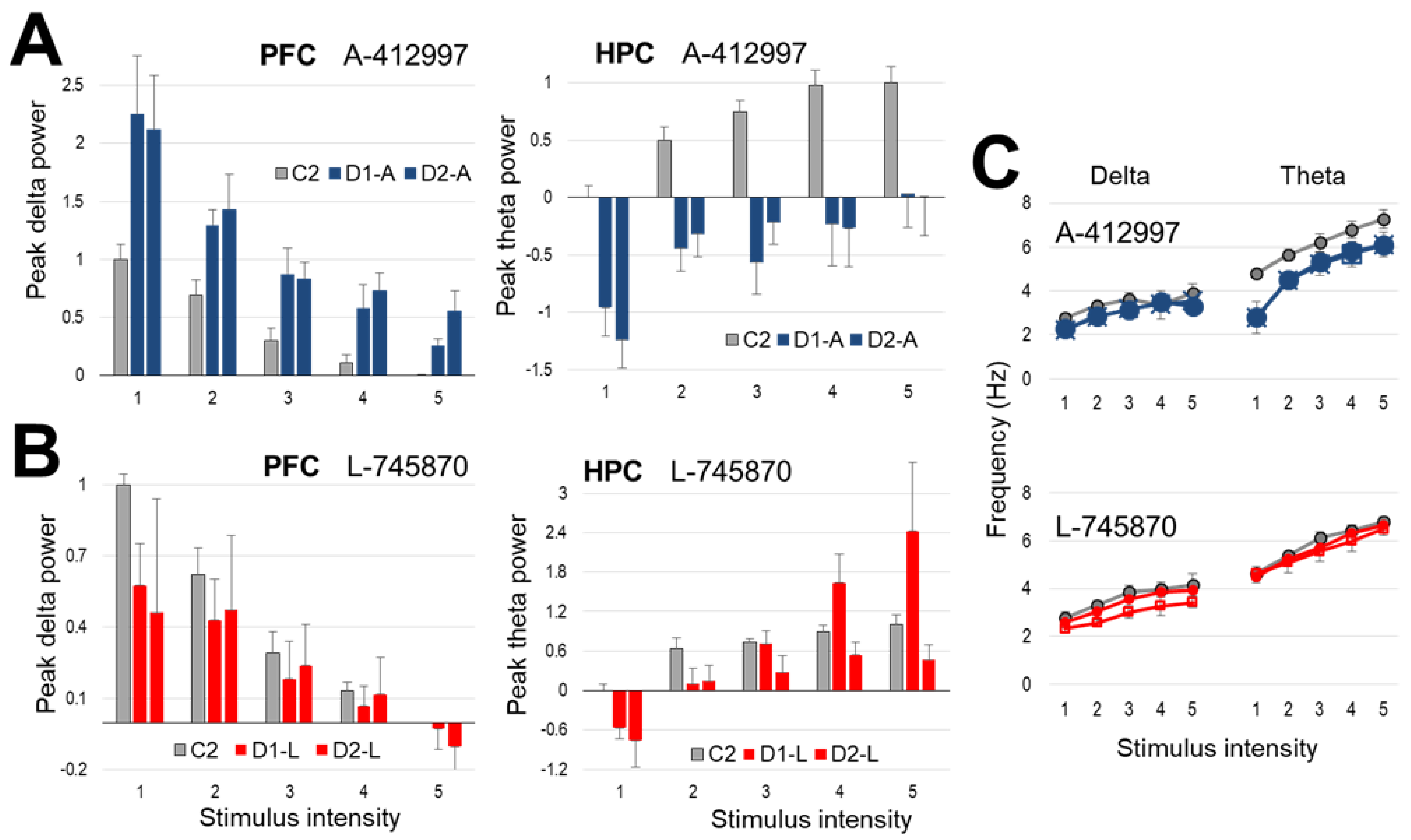
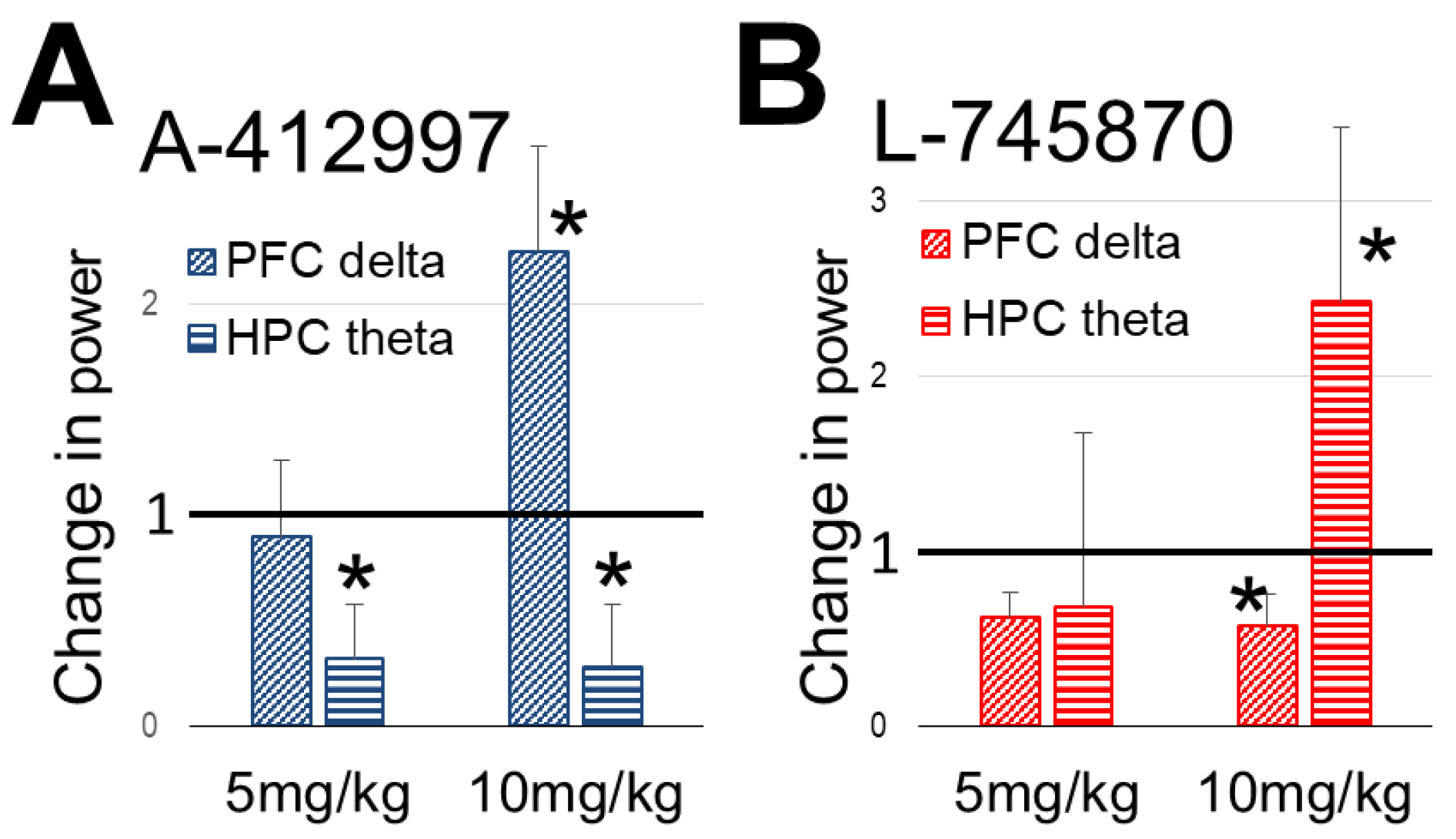
2.2. Effect of Dopamine D4-Receptor Activation on Forebrain Network Oscillations
2.3. Effect of Dopamine D4-Receptor Blockade on Forebrain Network Oscillations
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Drugs
4.3. Surgery
4.4. Brain Cryosectioning and Histology
4.5. RPO Stimulation and Data Recording
4.6. Data and Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Insel, T.; Cuthbert, B.; Garvey, M.; Heinssen, R.; Pine, D.S.; Quinn, K.; Sanislow, C.; Wang, P. Research domain criteria (RDoC): Toward a new classification framework for research on mental disorders. Am. J. Psychiatry 2010, 167, 748–751. [Google Scholar] [CrossRef] [PubMed]
- Stahl, S.M.; Buckley, P.F. Negative symptoms of schizophrenia: A problem that will not go away. Acta Psychiatr. Scand. 2007, 115, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, G.P.; Abdul-Monim, Z.; Neill, J.C.; Zhang, Z.J. Calcium binding protein markers of GABA deficits in schizophrenia--postmortem studies and animal models. Neurotox. Res. 2004, 6, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Fachim, H.A.; Srisawat, U.; Dalton, C.F.; Reynolds, G.P. Parvalbumin promoter hypermethylation in postmortem brain in schizophrenia. Epigenomics 2018, 10, 519–524. [Google Scholar] [CrossRef]
- Dienel, S.J.; Lewis, D.A. Alterations in cortical interneurons and cognitive function in schizophrenia. Neurobiol. Dis. 2019, 131, 104208. [Google Scholar] [CrossRef]
- Tan, H.Y.; Callicott, J.H.; Weinberger, D.R. Intermediate phenotypes in schizophrenia genetics redux: Is it a no brainer? Mol. Psychiatry 2008, 13, 233–238. [Google Scholar] [CrossRef]
- Sun, Y.; Farzan, F.; Barr, M.S.; Kirihara, K.; Fitzgerald, P.B.; Light, G.A.; Daskalakis, Z.J. gamma oscillations in schizophrenia: Mechanisms and clinical significance. Brain Res. 2011, 1413, 98–114. [Google Scholar] [CrossRef] [PubMed]
- Koshiyama, D.; Miyakoshi, M.; Tanaka-Koshiyama, K.; Sprock, J.; Light, G.A. High-power gamma-related delta phase alteration in schizophrenia patients at rest. Psychiatry Clin. Neurosci. 2022, 76, 179–186. [Google Scholar] [CrossRef]
- Koshiyama, D.; Miyakoshi, M.; Joshi, Y.B.; Molina, J.L.; Tanaka-Koshiyama, K.; Joyce, S.; Braff, D.L.; Swerdlow, N.R.; Light, G.A. Neural network dynamics underlying gamma synchronization deficits in schizophrenia. Prog. Neuropsychopharmacol. Biol. Psychiatry 2021, 107, 110224. [Google Scholar] [CrossRef] [PubMed]
- Rurup, L.; Mathes, B.; Schmiedt-Fehr, C.; Wienke, A.S.; Ozerdem, A.; Brand, A.; Basar-Eroglu, C. Altered gamma and theta oscillations during multistable perception in schizophrenia. Int. J. Psychophysiol. 2020, 155, 127–139. [Google Scholar] [CrossRef]
- Lanre-Amos, T.; Kocsis, B. Hippocampal oscillations in the rodent model of schizophrenia induced by amygdala GABA receptor blockade. Front. Psychiatry 2010, 1, 132. [Google Scholar] [CrossRef] [PubMed]
- Kocsis, B.; Lee, P.; Deth, R. Enhancement of gamma activity after selective activation of dopamine D4 receptors in freely moving rats and in a neurodevelopmental model of schizophrenia. Brain Struct. Funct. 2014, 219, 2173–2180. [Google Scholar] [CrossRef]
- Kocsis, B.; Brown, R.E.; McCarley, R.W.; Hajos, M. Impact of ketamine on neuronal network dynamics: Translational modeling of schizophrenia-relevant deficits. CNS Neurosci. Ther. 2013, 19, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Fejgin, K.; Nielsen, J.; Birknow, M.R.; Bastlund, J.F.; Nielsen, V.; Lauridsen, J.B.; Stefansson, H.; Steinberg, S.; Sorensen, H.B.; Mortensen, T.E.; et al. A mouse model that recapitulates cardinal features of the 15q13.3 microdeletion syndrome including schizophrenia- and epilepsy-related alterations. Biol. Psychiatry 2014, 76, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Meier, M.A.; Lemercier, C.E.; Kulisch, C.; Kiss, B.; Lendvai, B.; Adham, N.; Gerevich, Z. The novel antipsychotic cariprazine stabilizes gamma oscillations in rat hippocampal slices. Br. J. Pharmacol. 2020, 177, 1622–1634. [Google Scholar] [CrossRef]
- Perreault, M.L.; Fan, T.; Banasikowski, T.J.; Grace, A.A.; George, S.R. The atypical dopamine receptor agonist SKF 83959 enhances hippocampal and prefrontal cortical neuronal network activity in a rat model of cognitive dysfunction. Eur. J. Neurosci. 2017, 46, 2015–2025. [Google Scholar] [CrossRef]
- Hudson, M.R.; Hannan, A.J.; O’Brien, T.J.; Jones, N.C. High-Frequency Neuronal Oscillatory Abnormalities in the Phospholipase C-beta1 Knockout Mouse Model of Schizophrenia. Int. J. Neuropsychopharmacol. 2019, 22, 221–231. [Google Scholar] [CrossRef]
- Long, L.E.; Anderson, P.; Frank, E.; Shaw, A.; Liu, S.; Huang, X.F.; Pinault, D.; Karl, T.; O’Brien, T.J.; Shannon Weickert, C.; et al. Neuregulin 1 expression and electrophysiological abnormalities in the Neuregulin 1 transmembrane domain heterozygous mutant mouse. PLoS ONE 2015, 10, e0124114. [Google Scholar] [CrossRef]
- Gandal, M.J.; Edgar, J.C.; Klook, K.; Siegel, S.J. Gamma synchrony: Towards a translational biomarker for the treatment-resistant symptoms of schizophrenia. Neuropharmacology 2012, 62, 1504–1518. [Google Scholar] [CrossRef]
- Buzsaki, G. Theta oscillations in the hippocampus. Neuron 2002, 33, 325–340. [Google Scholar] [CrossRef]
- Fujisawa, S.; Buzsaki, G. A 4 Hz oscillation adaptively synchronizes prefrontal, VTA, and hippocampal activities. Neuron 2011, 72, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.; Svensson, F.P.; Mazeh, A.; Kocsis, B. Prefrontal-hippocampal coupling by theta rhythm and by 2-5 Hz oscillation in the delta band: The role of the nucleus reuniens of the thalamus. Brain Struct. Funct. 2017, 222, 2819–2830. [Google Scholar] [CrossRef]
- Zhong, P.; Yan, Z. Distinct Physiological Effects of Dopamine D4 Receptors on Prefrontal Cortical Pyramidal Neurons and Fast-Spiking Interneurons. Cereb. Cortex 2016, 26, 180–191. [Google Scholar] [CrossRef] [PubMed]
- Andersson, R.; Johnston, A.; Fisahn, A. Dopamine D4 receptor activation increases hippocampal gamma oscillations by enhancing synchronization of fast-spiking interneurons. PLoS ONE 2012, 7, e40906. [Google Scholar] [CrossRef]
- de Almeida, J.; Mengod, G. D2 and D4 dopamine receptor mRNA distribution in pyramidal neurons and GABAergic subpopulations in monkey prefrontal cortex: Implications for schizophrenia treatment. Neuroscience 2010, 170, 1133–1139. [Google Scholar] [CrossRef]
- Mrzljak, L.; Bergson, C.; Pappy, M.; Huff, R.; Levenson, R.; Goldman-Rakic, P.S. Localization of dopamine D4 receptors in GABAergic neurons of the primate brain. Nature 1996, 381, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Wedzony, K.; Chocyk, A.; Mackowiak, M.; Fijal, K.; Czyrak, A. Cortical localization of dopamine D4 receptors in the rat brain--immunocytochemical study. J. Physiol. Pharmacol. 2000, 51, 205–221. [Google Scholar] [PubMed]
- Wang, Y.; Jin, Y.K.; Guo, T.C.; Li, Z.R.; Feng, B.Y.; Han, J.H.; Vreugdenhil, M.; Lu, C.B. Activation of Dopamine 4 Receptor Subtype Enhances Gamma Oscillations in Hippocampal Slices of Aged Mice. Front. Aging Neurosci. 2022, 14, 838803. [Google Scholar] [CrossRef]
- Furth, K.E.; McCoy, A.J.; Dodge, C.; Walters, J.R.; Buonanno, A.; Delaville, C. Neuronal correlates of ketamine and walking induced gamma oscillations in the medial prefrontal cortex and mediodorsal thalamus. PLoS ONE 2017, 12, e0186732. [Google Scholar] [CrossRef]
- Deth, R.C. Molecular Origins of Attention: The Dopamine-Folate Connection; Kluwer Academic Publishers: Amsterdam, The Netherlands, 2003. [Google Scholar]
- Nucifora, F.C., Jr.; Mihaljevic, M.; Lee, B.J.; Sawa, A. Clozapine as a Model for Antipsychotic Development. Neurotherapeutics 2017, 14, 750–761. [Google Scholar] [CrossRef]
- Wenthur, C.J.; Lindsley, C.W. Classics in chemical neuroscience: Clozapine. ACS Chem. Neurosci. 2013, 4, 1018–1025. [Google Scholar] [CrossRef]
- Lindsley, C.W.; Hopkins, C.R. Return of D4 Dopamine Receptor Antagonists in Drug Discovery. J. Med. Chem. 2017, 60, 7233–7243. [Google Scholar] [CrossRef] [PubMed]
- Hwang, R.; Tiwari, A.K.; Zai, C.C.; Felsky, D.; Remington, E.; Wallace, T.; Tong, R.P.; Souza, R.P.; Oh, G.; Potkin, S.G.; et al. Dopamine D4 and D5 receptor gene variant effects on clozapine response in schizophrenia: Replication and exploration. Prog. Neuropsychopharmacol. Biol. Psychiatry 2012, 37, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Vertes, R.P.; Kocsis, B. Brainstem-diencephalo-septohippocampal systems controlling the theta rhythm of the hippocampus. Neuroscience 1997, 81, 893–926. [Google Scholar]
- Kirk, I.J. Frequency modulation of hippocampal theta by the supramammillary nucleus, and other hypothalamo-hippocampal interactions: Mechanisms and functional implications. Neurosci. Biobehav. Rev. 1998, 22, 291–302. [Google Scholar] [CrossRef]
- Bland, B.H.; Oddie, S.D. Anatomical, electrophysiological and pharmacological studies of ascending brainstem hippocampal synchronizing pathways. Neurosci. Biobehav. Rev. 1998, 22, 259–273. [Google Scholar] [CrossRef]
- Totah, N.K.; Neves, R.M.; Panzeri, S.; Logothetis, N.K.; Eschenko, O. The Locus Coeruleus Is a Complex and Differentiated Neuromodulatory System. Neuron 2018, 99, 1055–1068 e6. [Google Scholar] [CrossRef]
- Szkudlarek, H.J.; Orlowska, P.; Lewandowski, M.H. Light-induced responses of slow oscillatory neurons of the rat olivary pretectal nucleus. PLoS ONE 2012, 7, e33083. [Google Scholar] [CrossRef]
- Kramis, R.; Vanderwolf, C.H.; Bland, B.H. Two types of hippocampal rhythmical slow activity in both the rabbit and the rat: Relations to behavior and effects of atropine, diethyl ether, urethane, and pentobarbital. Exp. Neurol. 1975, 49 Pt 1, 58–85. [Google Scholar] [CrossRef]
- Kocsis, B.; Gyimesi-Pelczer, K. Patterns of relationship between activity of sympathetic nerves in rabbits and rats. Auton. Neurosci. 2004, 114, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Mofleh, R.; Kocsis, B. Respiratory coupling between prefrontal cortex and hippocampus of rats anaesthetized with urethane in theta and non-theta states. Eur. J. Neurosci. 2021, 54, 5507–5517. [Google Scholar] [CrossRef] [PubMed]
- Silver, N.R.G.; Ward-Flanagan, R.; Dickson, C.T. Long-term stability of physiological signals within fluctuations of brain state under urethane anesthesia. PLoS ONE 2021, 16, e0258939. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Topchiy, I.; Kocsis, B. The effect of atropine administered in the medial septum or hippocampus on high- and low-frequency theta rhythms in the hippocampus of urethane anesthetized rats. Synapse 2007, 61, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Kiss, T.; Hoffmann, W.E.; Hajos, M. Delta oscillation and short-term plasticity in the rat medial prefrontal cortex: Modelling NMDA hypofunction of schizophrenia. Int. J. Neuropsychopharmacol. 2011, 14, 29–42. [Google Scholar] [CrossRef]
- Ly, S.; Pishdari, B.; Lok, L.L.; Hajos, M.; Kocsis, B. Activation of 5-HT6 receptors modulates sleep-wake activity and hippocampal theta oscillation. ACS Chem. Neurosci. 2013, 4, 191–199. [Google Scholar] [CrossRef]
- Sorman, E.; Wang, D.; Hajos, M.; Kocsis, B. Control of hippocampal theta rhythm by serotonin: Role of 5-HT2c receptors. Neuropharmacology 2011, 61, 489–494. [Google Scholar] [CrossRef]
- Furth, K.E.; Mastwal, S.; Wang, K.H.; Buonanno, A.; Vullhorst, D. Dopamine, cognitive function, and gamma oscillations: Role of D4 receptors. Front. Cell Neurosci. 2013, 7, 102. [Google Scholar] [CrossRef]
- Hirano, Y.; Oribe, N.; Kanba, S.; Onitsuka, T.; Nestor, P.G.; Spencer, K.M. Spontaneous Gamma Activity in Schizophrenia. JAMA Psychiatry 2015, 72, 813–821. [Google Scholar] [CrossRef]
- Spencer, K.M.; Salisbury, D.F.; Shenton, M.E.; McCarley, R.W. Gamma-band auditory steady-state responses are impaired in first episode psychosis. Biol. Psychiatry 2008, 64, 369–375. [Google Scholar] [CrossRef]
- Kittelberger, K.; Hur, E.E.; Sazegar, S.; Keshavan, V.; Kocsis, B. Comparison of the effects of acute and chronic administration of ketamine on hippocampal oscillations: Relevance for the NMDA receptor hypofunction model of schizophrenia. Brain Struct. Funct. 2012, 217, 395–409. [Google Scholar] [CrossRef]
- Pittman-Polletta, B.; Hu, K.; Kocsis, B. Subunit-specific NMDAR antagonism dissociates schizophrenia subtype-relevant oscillopathies associated with frontal hypofunction and hippocampal hyperfunction. Sci. Rep. 2018, 8, 11588. [Google Scholar] [CrossRef]
- Lakatos, P.; Shah, A.S.; Knuth, K.H.; Ulbert, I.; Karmos, G.; Schroeder, C.E. An oscillatory hierarchy controlling neuronal excitability and stimulus processing in the auditory cortex. J. Neurophysiol. 2005, 94, 1904–1911. [Google Scholar] [CrossRef] [PubMed]
- Belluscio, M.A.; Mizuseki, K.; Schmidt, R.; Kempter, R.; Buzsaki, G. Cross-frequency phase-phase coupling between theta and gamma oscillations in the hippocampus. J. Neurosci. 2012, 32, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, C.E.; Lakatos, P. The gamma oscillation: Master or slave? Brain Topogr. 2009, 22, 24–26. [Google Scholar] [CrossRef]
- Sirota, A.; Montgomery, S.; Fujisawa, S.; Isomura, Y.; Zugaro, M.; Buzsaki, G. Entrainment of neocortical neurons and gamma oscillations by the hippocampal theta rhythm. Neuron 2008, 60, 683–697. [Google Scholar] [CrossRef] [PubMed]
- Vertes, R.P.; Hoover, W.B.; Szigeti-Buck, K.; Leranth, C. Nucleus reuniens of the midline thalamus: Link between the medial prefrontal cortex and the hippocampus. Brain Res. Bull. 2007, 71, 601–609. [Google Scholar] [CrossRef]
- Hunt, M.J.; Kopell, N.J.; Traub, R.D.; Whittington, M.A. Aberrant Network Activity in Schizophrenia. Trends Neurosci. 2017, 40, 371–382. [Google Scholar] [CrossRef]
- Anderson, K.L.; Rajagovindan, R.; Ghacibeh, G.A.; Meador, K.J.; Ding, M. Theta oscillations mediate interaction between prefrontal cortex and medial temporal lobe in human memory. Cereb. Cortex 2010, 20, 1604–1612. [Google Scholar] [CrossRef]
- Pinault, D. N-methyl d-aspartate receptor antagonists ketamine and MK-801 induce wake-related aberrant gamma oscillations in the rat neocortex. Biol. Psychiatry 2008, 63, 730–735. [Google Scholar] [CrossRef]
- Ahnaou, A.; Huysmans, H.; Van de Casteele, T.; Drinkenburg, W. Cortical high gamma network oscillations and connectivity: A translational index for antipsychotics to normalize aberrant neurophysiological activity. Transl. Psychiatry 2017, 7, 1285. [Google Scholar] [CrossRef]
- Kocsis, B. Differential role of NR2A and NR2B subunits in N-methyl-D-aspartate receptor antagonist-induced aberrant cortical gamma oscillations. Biol. Psychiatry 2012, 71, 987–995. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.; Kim, Y.; Moghaddam, B. Disruption of prefrontal cortex large scale neuronal activity by different classes of psychotomimetic drugs. J. Neurosci. 2012, 32, 3022–3031. [Google Scholar] [CrossRef]
- Bernaerts, P.; Tirelli, E. Facilitatory effect of the dopamine D4 receptor agonist PD168,077 on memory consolidation of an inhibitory avoidance learned response in C57BL/6J mice. Behav. Brain Res. 2003, 142, 41–52. [Google Scholar] [CrossRef]
- Browman, K.E.; Curzon, P.; Pan, J.B.; Molesky, A.L.; Komater, V.A.; Decker, M.W.; Brioni, J.D.; Moreland, R.B.; Fox, G.B. A-412997, a selective dopamine D4 agonist, improves cognitive performance in rats. Pharmacol. Biochem. Behav. 2005, 82, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Woolley, M.L.; Waters, K.A.; Reavill, C.; Bull, S.; Lacroix, L.P.; Martyn, A.J.; Hutcheson, D.M.; Valerio, E.; Bate, S.; Jones, D.N.; et al. Selective dopamine D4 receptor agonist (A-412997) improves cognitive performance and stimulates motor activity without influencing reward-related behaviour in rat. Behav. Pharmacol. 2008, 19, 765–776. [Google Scholar] [CrossRef] [PubMed]
- Vertes, R.P.; Hoover, W.B.; Do Valle, A.C.; Sherman, A.; Rodriguez, J.J. Efferent projections of reuniens and rhomboid nuclei of the thalamus in the rat. J. Comp. Neurol. 2006, 499, 768–796. [Google Scholar] [CrossRef] [PubMed]
- Vertes, R.P. Analysis of projections from the medial prefrontal cortex to the thalamus in the rat, with emphasis on nucleus reuniens. J. Comp. Neurol. 2002, 442, 163–187. [Google Scholar] [CrossRef]
- Cassel, J.C.; Ferraris, M.; Quilichini, P.; Cholvin, T.; Boch, L.; Stephan, A.; Pereira de Vasconcelos, A. The reuniens and rhomboid nuclei of the thalamus: A crossroads for cognition-relevant information processing? Neurosci. Biobehav. Rev. 2021, 126, 338–360. [Google Scholar] [CrossRef]
- Kafetzopoulos, V.; Kokras, N.; Sousa, N.; Antoniou, K.; Sotiropoulos, I.; Dalla, C. Nucleus Reuniens Lesion and Antidepressant Treatment Prevent Hippocampal Neurostructural Alterations Induced by Chronic Mild Stress in Male Rats. Neuroscience 2021, 454, 85–93. [Google Scholar] [CrossRef]
- Kafetzopoulos, V.; Kokras, N.; Sotiropoulos, I.; Oliveira, J.F.; Leite-Almeida, H.; Vasalou, A.; Sardinha, V.M.; Papadopoulou-Daifoti, Z.; Almeida, O.F.X.; Antoniou, K.; et al. The nucleus reuniens: A key node in the neurocircuitry of stress and depression. Mol. Psychiatry 2018, 23, 579–586. [Google Scholar] [CrossRef]
- Linley, S.B.; Athanason, A.C.; Rojas, A.K.P.; Vertes, R.P. Role of the reuniens and rhomboid thalamic nuclei in anxiety-like avoidance behavior in the rat. Hippocampus 2021, 31, 756–769. [Google Scholar] [CrossRef] [PubMed]
- Viena, T.D.; Linley, S.B.; Vertes, R.P. Inactivation of nucleus reuniens impairs spatial working memory and behavioral flexibility in the rat. Hippocampus 2018, 28, 297–311. [Google Scholar] [CrossRef] [PubMed]
- Rahman, F.; Nanu, R.; Schneider, N.A.; Katz, D.; Lisman, J.; Pi, H.J. Optogenetic perturbation of projections from thalamic nucleus reuniens to hippocampus disrupts spatial working memory retrieval more than encoding. Neurobiol. Learn. Mem. 2021, 179, 107396. [Google Scholar] [CrossRef]
- Duan, A.R.; Varela, C.; Zhang, Y.; Shen, Y.; Xiong, L.; Wilson, M.A.; Lisman, J. Delta frequency optogenetic stimulation of the thalamic nucleus reuniens is sufficient to produce working memory deficits: Relevance to schizophrenia. Biol. Psychiatry 2015, 77, 1098–1107. [Google Scholar] [CrossRef] [PubMed]
- Prante, O.; Tietze, R.; Hocke, C.; Lober, S.; Hubner, H.; Kuwert, T.; Gmeiner, P. Synthesis, radiofluorination, and in vitro evaluation of pyrazolo[1,5-a]pyridine-based dopamine D4 receptor ligands: Discovery of an inverse agonist radioligand for PET. J. Med. Chem. 2008, 51, 1800–1810. [Google Scholar] [CrossRef]
- Bristow, L.J.; Kramer, M.S.; Kulagowski, J.; Patel, S.; Ragan, C.I.; Seabrook, G.R. Schizophrenia and L-745,870, a novel dopamine D4 receptor antagonist. Trends Pharmacol. Sci. 1997, 18, 186–188. [Google Scholar] [CrossRef]
- Van Tol, H.H.; Wu, C.M.; Guan, H.C.; Ohara, K.; Bunzow, J.R.; Civelli, O.; Kennedy, J.; Seeman, P.; Niznik, H.B.; Jovanovic, V. Multiple dopamine D4 receptor variants in the human population. Nature 1992, 358, 149–152. [Google Scholar]
- Demiralp, T.; Herrmann, C.S.; Erdal, M.E.; Ergenoglu, T.; Keskin, Y.H.; Ergen, M.; Beydagi, H. DRD4 and DAT1 polymorphisms modulate human gamma band responses. Cereb. Cortex 2007, 17, 1007–1019. [Google Scholar] [CrossRef]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates; Academic Press: Sydney, Australia, 1986. [Google Scholar]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thörn, C.W.; Kafetzopoulos, V.; Kocsis, B. Differential Effect of Dopamine D4 Receptor Activation on Low-Frequency Oscillations in the Prefrontal Cortex and Hippocampus May Bias the Bidirectional Prefrontal–Hippocampal Coupling. Int. J. Mol. Sci. 2022, 23, 11705. https://doi.org/10.3390/ijms231911705
Thörn CW, Kafetzopoulos V, Kocsis B. Differential Effect of Dopamine D4 Receptor Activation on Low-Frequency Oscillations in the Prefrontal Cortex and Hippocampus May Bias the Bidirectional Prefrontal–Hippocampal Coupling. International Journal of Molecular Sciences. 2022; 23(19):11705. https://doi.org/10.3390/ijms231911705
Chicago/Turabian StyleThörn, Carolina Wilnerzon, Vasilios Kafetzopoulos, and Bernat Kocsis. 2022. "Differential Effect of Dopamine D4 Receptor Activation on Low-Frequency Oscillations in the Prefrontal Cortex and Hippocampus May Bias the Bidirectional Prefrontal–Hippocampal Coupling" International Journal of Molecular Sciences 23, no. 19: 11705. https://doi.org/10.3390/ijms231911705
APA StyleThörn, C. W., Kafetzopoulos, V., & Kocsis, B. (2022). Differential Effect of Dopamine D4 Receptor Activation on Low-Frequency Oscillations in the Prefrontal Cortex and Hippocampus May Bias the Bidirectional Prefrontal–Hippocampal Coupling. International Journal of Molecular Sciences, 23(19), 11705. https://doi.org/10.3390/ijms231911705





